Open Access Article
Open Access ArticleTiO2-coated rutile oxide catalysts for acidic oxygen evolution: a design principle
Georgios K.
Stavroglou
 ,
Adrian M.
Frandsen
,
Adrian M.
Frandsen
 ,
Henrik H.
Kristoffersen
,
Henrik H.
Kristoffersen
 ,
Katrine L.
Svane
,
Katrine L.
Svane
 and
Jan
Rossmeisl
and
Jan
Rossmeisl
 *
*
Center for High Entropy Alloy Catalysis (CHEAC), Department of Chemistry, University of Copenhagen, Universitetsparken 5, 2100 Copenhagen, Denmark. E-mail: jan.rossmeisl@chem.ku.dk
First published on 23rd February 2026
Abstract
Green hydrogen production through water electrolysis under acidic conditions is limited by the efficiency of the anodic oxygen evolution reaction (OER). In this study, we outline a design principle for developing active and potentially stable rutile oxide electrocatalysts for acidic OER. Using density functional theory, we find that the adsorption energies for Ti, Sn, and Zr active sites correlate with the position of the Fermi level relative to the oxygen 2s states as the surrounding oxide composition is varied. This trend is not observed for platinum-group elements. We attribute this difference to the inability of Ti, Sn, and Zr active sites to access oxidation states above the 4+ of the rutile structure, according to changes in geometry and charge. Ti emerges as an adsorption site of special interest, since varying the composition of the surrounding rutile oxide allows titanium sites to reach theoretical overpotentials as low as 0.14 V. We then demonstrate tunable activity for TiO2-coated conductive rutile substrates. Based on literature evidence that TiO2 coatings improve stability under acidic OER conditions without substantially compromising activity, we suggest that a TiO2 overlayer can serve as both an active site and corrosion protection. We propose a composition-based model that successfully finds Ir and Ru free compositions (WxPt1−xO2, W0.46Pd0.54O2) that can activate the applied TiO2 overlayer.
Broader contextProton-exchange membrane water electrolysers offer significant advantages over alkaline water electrolysers in the production of green hydrogen, including higher current densities, a high-purity hydrogen gas and a compact device design. As catalysts for the anodic oxygen evolution reaction (OER), oxides based on Ir offer the best compromise between activity and stability, however, the scarcity of Ir limits the industrial scalability of this technology. Earth-abundant alternatives are usually either active but unstable under acidic conditions, or stable but not sufficiently active. This work outlines a design principle for the fabrication of active and stable TiO2-coated rutile oxide catalysts for the acidic OER. Our calculations demonstrate that the activity of a Ti site correlates with the position of the Fermi level of the surrounding oxide. By choosing the right surrounding oxide composition, Ti sites can be activated to achieve low OER overpotential. The same principle is found to apply for TiO2 overlayer(s) which can then act as both active site and corrosion protection. The Fermi level energy of the host oxide can be predicted based on the composition, making it possible to construct Ir- and Ru-free catalysts with predicted high OER activity. |
Introduction
Practical strategies for storing electricity from renewable energy sources, e.g. in the form of chemical fuels, are crucial to reduce dependence on fossil fuels. Molecular hydrogen is a suitable fuel in this context, since it can be efficiently generated through water electrolysis (WE).1 Alkaline WE is a well-established technology relying on transition metal based catalysts,2,3 however proton exchange membrane water electrolysis (PEMWE), which operates under acidic conditions, offers several advantages over the alkaline setup, including higher current densities, purer product gas and more compact devices.4 The oxygen evolution reaction (OER), occurring at the anode, is recognised as the kinetic bottleneck in both alkaline and acidic water electrolysis.5 The limitation arises because the OER consists of four proton-coupled electron transfer steps, e.g. for the conventionally assumed adsorbate evolution mechanism (AEM, see Section S1 in the SI for illustration):| * + H2O(l) → *OH + H+ + e− | (1) |
| ΔG1 = ΔG*OH − eURHE |
| *OH → *O + H+ + e− | (2) |
| ΔG2 = ΔG*O − ΔG*OH − eURHE |
| *O + H2O(l) → *OOH + H+ + e− | (3) |
| ΔG3 = ΔG*OOH − ΔG*O − eURHE |
| *OOH → * + O2(g) + H+ + e− | (4) |
| ΔG4 = ΔGO2 − ΔG*OOH − eURHE |
Within this limitation, oxides based on Ir and Ru are the most efficient catalysts for acidic OER.8–14 IrO2 is less active than RuO2, but IrO2 is substantially more stable.8,10 Both IrO2 and RuO2 are metallic conductors,15 facilitating electron transport in the anode catalyst and offering low electronic resistance. However, the low abundance of Ir and Ru precludes industrial upscaling of this technology.16 Materials based on earth-abundant elements are usually either stable but inert or active but unstable under acidic OER conditions. Research on catalysts based on earth-abundant elements has therefore often followed one of two approaches: activating stable materials or stabilising active materials.17,18
Stable materials that are based on low-cost elements have been doped to enhance their OER activity. For example, TiO2 is stable in acid,19 but it is a poor OER catalyst because it is a semiconductor.20 Both experimental and theoretical studies have demonstrated that the catalytic activity can be enhanced by doping.21–24 The dopant introduces new states in the band gap, which determine the Fermi level. In particular, introducing one or two subsurface layers of SrRuO3 into inert SrTiO3 enhanced the experimentally measured OER activity under alkaline conditions, which was attributed to new electronic states introduced by the dopant.25 In a later computational study, this concept was extended by introducing a single subsurface layer of different transition metals into SrTiO3, SrZrO3, TiO2, and ZrO2, and a correlation between the Fermi level and catalytic activity was established.26 Since the catalytic activity is influenced by the energy position of the dopant states in the band gap, it is desirable to be able to tune these states, which is difficult to achieve with a single dopant. Co-doping with both n-type (electron-donor) and p-type (electron-acceptor) dopants has therefore been investigated, demonstrating that the OER activity of TiO2 and SrTiO3 can be tuned.20,21 As an extreme case of the co-doping strategy, Ti atoms incorporated in a conducting high-entropy oxide (HEO), i.e. an oxide containing five different metals, were theoretically predicted to be highly active.27
Protecting active but unstable catalysts by coating or enriching them with a stable oxide such as TiO2 is an alternative strategy for improving catalyst performance. Dimensionally stable anodes (DSAs), used in the commercial production of chlorine, employ this strategy.28 DSAs consist of a mixture of active precious-metal oxides such as RuO2 and IrO2, together with TiO2, which offers stability. Such mixtures have therefore been extensively studied, and it has been shown that the formation of an ultrathin TiO2 coating on oxide nanoparticles consisting of Ru, Ti, and Nb resulted in improved stability during chlorine evolution.29 Thin TiO2 coatings have also been formed on active catalysts such as MnO2 and Co3O4, resulting in improved stability without significant loss of activity.30,31 Such coatings can be readily made by atomic layer deposition (ALD),30,32 thermal diffusion,29 sputter deposition,31 and electrodeposition.33
The catalytic activity for OER on oxides can be estimated computationally by calculating the free energies of the individual steps along the reaction pathway (eqn (1)–(4)). Correlations between catalytic activity and the adsorption energies of reaction intermediates are often represented by volcano plots.34,35 In particular, ΔG2 has been proposed as a universal descriptor of OER activity.7 It is attractive to relate these adsorption energies to electronic properties of the material to provide an understanding of the adsorption process and reduce the effort required to predict the catalytic activity of new materials.36 A range of electronic descriptors have been widely studied in the literature, inspired by the success of the d-band model from Hammer and Nørskov. They showed that the binding energy on metal surfaces is linearly dependent on the energy difference between the center of the metal d-band and the Fermi level of the material.37,38 This approach has been less successful in describing the adsorption energies on oxides. In contrast, the positions of the O2p band, t2g band, the occupancy of the eg orbital, and the vacuum level relative to the Fermi level have all been identified as alternative descriptors for different systems.22,26,39–43
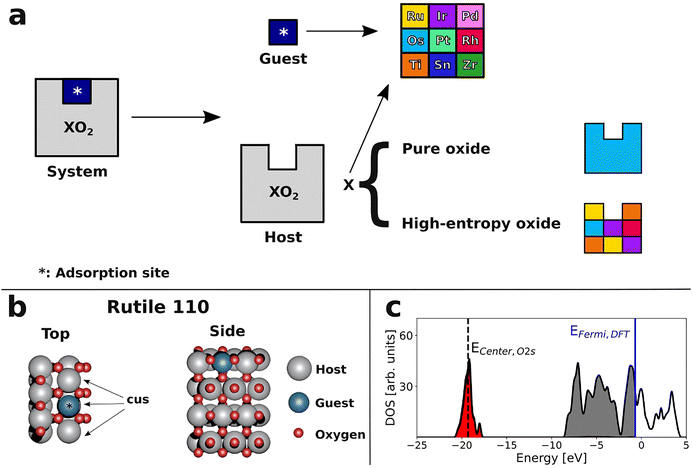
Here, guided by the fundamental ideas from these previous efforts, we outline a new design principle for developing active and potentially stable electrocatalysts for the OER under acidic conditions. We first study Ru, Rh, Ir, Os, Pd, Pt, Ti, Sn, and Zr “guest” atoms substituted at surface sites in rutile oxide slabs (“host”) with a range of element compositions. Using density functional theory (DFT), we show how the adsorption energies of *OH and *O intermediates on these guest sites change as the host composition, and therefore its Fermi level, is varied. For Ti, Sn, and Zr guests, the adsorption energies show a pronounced variation that correlates with the host Fermi level, whereas Ru, Rh, Pd, Pt, Os, and Ir guests show only low variations, which are independent of the Fermi level. By studying changes in the geometry and the charge at the adsorption site, we show that this difference is related to the inability of the Ti, Sn, and Zr guest elements to access oxidation states higher than the 4+ oxidation state of the rutile structure. Ti is particularly interesting, as it spans binding energies corresponding to OER overpotentials as low as 0.14 V. Motivated by this, we further extend the concept from single Ti sites to TiO2 overlayers applied as coatings on conductive rutile oxides (substrates). The overpotentials on TiO2 overlayers coating substrates of various compositions showed trends similar to those for single Ti sites. Finally, we show that a simple linear model can successfully predict the Fermi level of the TiO2-coated system from the composition of the underlying substrate oxide. Thus, substrate oxides with a Fermi level that optimally activates the TiO2 overlayer for the OER can be designed from combinations of metal oxides that are not necessarily active or stable by themselves, making it possible to avoid Ru and Ir entirely. As an example, our model accurately predicts WxPt1−xO2 and W0.46Pd0.54O2 substrate compositions that, when coated with TiO2, exhibit calculated OER overpotentials as low as 0.39 V.
Methodology
DFT (cf. Section S2 in the SI for computational details) is used to calculate the adsorption energies of the reaction intermediates *OH, *O + Hb, *O, *OOH, *O2 + Hb, and O2(g) + Hb, where Hb is a hydrogen atom adsorbed on a bridging oxygen atom adjacent to the adsorption site (*). These calculations are carried out for a range of adsorption site elements (guests) and surrounding oxide compositions (hosts), as illustrated in Fig. 1a. More specifically, Ru, Rh, Ir, Os, Pd, Pt, Ti, Sn, and Zr have been considered as guests, while both pure oxides of these elements (except for ZrO2, which does not have a rutile polymorph),44,45 and high entropy oxides have been employed as hosts. More details and structural illustrations of this dataset are provided in Sections S2 and S4 of the SI.We focus on the (110) surfaces of rutile structures because RuO2, IrO2, RhO2, OsO2, PdO2, PtO2, SnO2, and TiO2 are all known to form a rutile polymorph.15,46,47 Rutile is the preferred crystal structure for TiO2,48 RuO2, IrO2, OsO2,49 and SnO2,50 under standard conditions. PtO2 prefers a distorted rutile structure,51 rutile RhO2 is possibly only a high-pressure phase,52 while PdO2 appears to have been synthesised by only one group and is unstable under standard conditions.47 The (110) surface is the rutile facet that preserves the highest coordination number for surface atoms, and for TiO2 it is calculated to have the lowest surface energy.53 Furthermore, corrosion measurements on RuO2 single crystals54 and IrO2 crystalline films55 demonstrate low dissolution rates from the (110) facets, which indicate increased stability compared to other facets.
In our analysis, we included previously published adsorption energies for *O and *OH on the rutile (110) surface of near-equimolar HEOs consisting of Ru, Ti, Ir, Os and Rh (details about the HEOs dataset are provided in Sections S3 and S4 in the SI).27 Similar HEOs in the IrOsPdPtRu, IrOsPtRhRu, AuOsPdPtRu, and AuIrOsPdPtReRhRu composition spaces have been prepared as nanoparticles using microwave solvothermal synthesis, and their OER activities were studied under acidic conditions.56
The surface model (Fig. 1b) consists of three coordinatively unsaturated sites (cus). One of these is the adsorption site and the other two are either bare or occupied with a pre-adsorbed oxygen, depending on the O* affinity of the adsorbing metal element (details are provided in Section S2 in the SI).
The adsorption free energies of the catalytic intermediates (ΔGintermediate) are calculated as:
| ΔGintermediate = ΔEDFT + ΔZPE − TΔS | (5) |
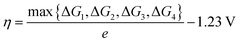 | (6) |
As a descriptor for the adsorption free energies, we investigate the Fermi level, which was previously observed to correlate with the adsorption energies of OER intermediates for doped SrTiO3 and TiO2.26 In that study, the vacuum level was used as a reference to compare DFT-calculated Fermi levels (EFermi,DFT) across different oxides; here, we adopt the approach in ref. 58, where EFermi,DFT values are aligned using a sharp low-energy peak in the density of states (DOS) as a reference. In our case the oxygen 2s band centroid (Ecenter,O2s) is used as the reference point, because it is common to systems studied, lies at relatively low energies (approximately −18 eV), and generally exhibits a sharp, non-interacting peak as illustrated in Fig. 1c for RuO2 (see Section S5 in the SI for the DOS of all pure oxides studied). In the case of RuO2, the EFermi,DFT relative to the oxygen 2s band center is 18.6 eV (EFermi,RuO2) and we use this value to normalise the Fermi level energies of all systems in this study
| EFermi = [EFermi,DFT − Ecenter,O2s] −EFermi,RuO2 | (7) |
In addition to the host–guest dataset described above, we studied TiO2 overlayers applied as coatings on conductive rutile oxides (substrates). We modelled pure RuO2, RhO2, IrO2, OsO2, PdO2, and PtO2 substrates, as well as substrates containing 89%, 78%, and 56% of each oxide, with the remainder being a random mixture of the other five oxides. To study the effect of increasing the coating thickness, the substrate oxides were investigated with both one and two TiO2 overlayers. We also studied TiO2-coated rutiles of WxPt1−xO2 (W = tungsten, x = 0.04, 0.08, 0.12, 0.17, 0.21, and WO2) and W0.46Pd0.54O2, as Ir and Ru free OER candidates. For each WxPt1−xO2 composition, we construct two distinct rutile slabs covered with one TiO2 overlayer and calculate adsorption energies at all six Ti cus sites. The lattice constants of all TiO2-coated doped structures were set to the composition weighted averages of the lattice parameters of the constituent pure oxides. More details on the TiO2-coated dataset are provided in Sections S2 and S4 of the SI.
GGA functionals such as RPBE are known to over-delocalise electrons and to underestimate band gaps. To assess how this affects our EFermi descriptor and the band gaps of the systems in our study, we benchmarked the EFermi (as defined in eqn (7)) and the band gaps using the HSE06 hybrid functional.59 We perform non self-consistent calculations, which have shown good agreement with self-consistent hybrid calculations60 and are widely used,61–63 while avoiding the computationally too demanding self-consistent HSE06 calculations for large surface slabs.
The HSE06 EFermi values calculated for the TiO2-coated systems are on average 0.08 eV lower than the RPBE EFermi values, although there are significant outliers (Fig. S9 in the SI). We also compared the band gaps between HSE06 and RPBE for the bare surface slabs of the pure oxides (Table S4). HSE06 band gaps remain in the metallic, near-metallic regime for RuO2, IrO2, OsO2, RhO2, and WO2, in agreement with resistivity measurements.15 PdO2 is also calculated to be metallic, and ref. 64 shows that Pd doping in RuO2 improves OER activity, suggesting that Pd incorporation is unlikely to severely diminish electronic conductivity. TiO2 and SnO2 show much larger HSE06 band gaps than RPBE, consistent with the known GGA underestimation on semiconductors. In Fig. S8, the RPBE band gaps of the bare surface slabs for the mixed oxides studied in this work indicate metallic or near-metallic behaviour (band gap range around 0–0.15 eV).
We also studied the D3 dispersion correction65 in the RPBE calculations for the TiO2-coated systems. D3 introduces an approximately constant shift of 0.25 eV in ΔG2 but the EFermi − ΔG2 trend remains essentially unchanged (Fig. S16 in the SI).
Results and discussion
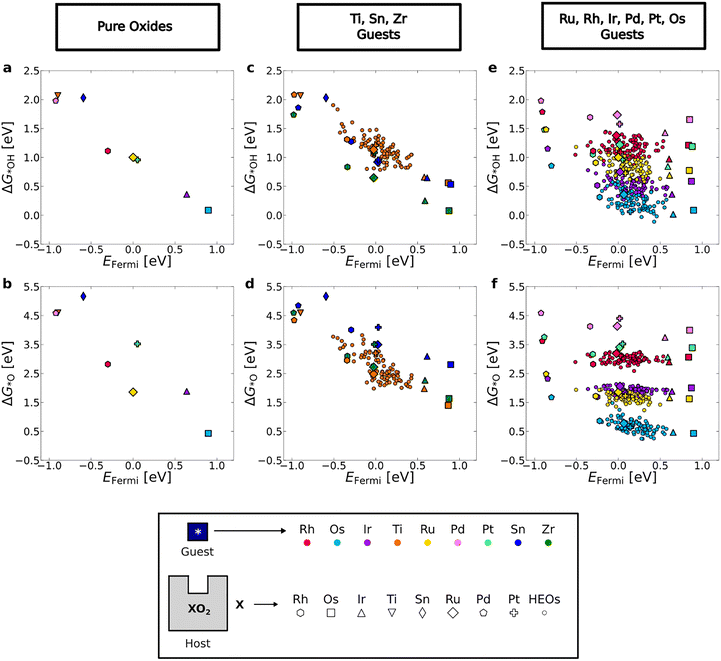
The adsorption free energies of *OH (ΔG*OH) and *O (ΔG*O) intermediates on the pure oxides as a function of the EFermi of the bare surface slabs are shown in Fig. 2a and b, demonstrating clear linear correlations. Looking at the individual guest elements in the host–guest systems, two significantly different trends in the adsorption free energies are observed (Fig. 2c–f). For Ti, Sn, and Zr guests, a notable variation in the adsorption free energies that correlates with the variation in EFermi is observed (Fig. 2c and d), indicating that the adsorption energy is primarily determined by the host composition. In contrast to this, the adsorption energies for the other guest elements (Ru, Rh, Ir, Os, Pd, Pt) span a much smaller energy range and are not correlated with EFermi (Fig. 2e and f), indicating that, in these systems, the adsorption energy is primarily determined by the guest element. We note that the data corresponding to SnO2 and TiO2 hosts are excluded from Fig. 2e and f, because their semiconducting electronic structure leads to a significantly different behaviour. Analysis of the trends in ΔG*O and EFermi, as the guest element is varied for each pure-host composition (including data where TiO2 and SnO2 are hosts) can be found in Section S6 in the SI.In ref. 27, a model based on the local atomic environments could predict ΔG*OH and ΔG*O adsorption energies. The model worked very well for Ru, Rh, Ir, and Os sites, but less well for Ti sites. We now know that *OH and *O adsorption on Ti sites depend strongly on EFermi, which was completely missing from the developed model.
To further understand the interactions between the adsorbates and the surface, we derived the scaling relation ΔG*O = aΔG*OH + b, where a is the slope and b is the intercept, for each of the guest elements under study (see Section S7 in the SI). Ti, Sn, and Zr exhibit strong correlations (R2 = 0.85–0.99) with slopes approaching 2 (a = 1.5–1.9). A relationship between OH and O adsorption energies with a slope of around 2 has been observed on metals,66 which have multiple electrons at the Fermi level, and on oxides, except when the computational cell only contains one dopant,67i.e. when there is only one electron at the Fermi level.
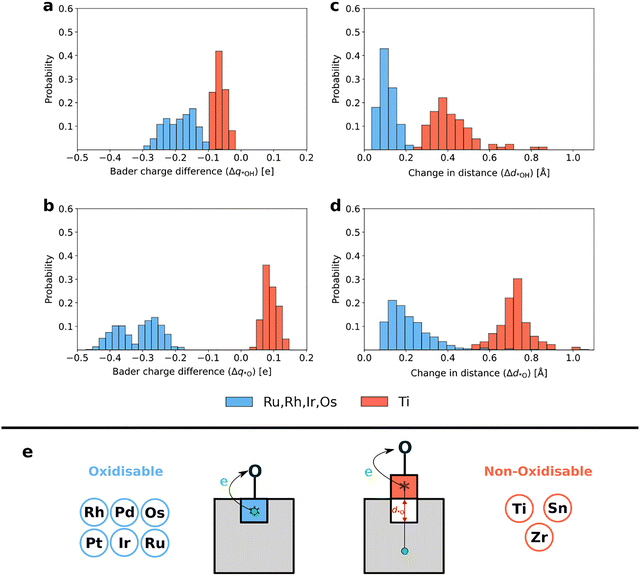
To investigate the origin of the two different trends in the adsorption energies, we conducted Bader charge analysis68 on the structures before and after the adsorption of *OH and *O. The Bader charge difference of the adsorption site atom upon adsorption of A (Δq*A, A = OH or O) is calculated as the difference between the Bader charge of that atom in the bare surface (q*) and the charge when the adsorbate is added (q*A):
| Δq*A = q*A − q* | (8) |
To further elucidate the distinct trends in adsorption behaviour, we calculated the change in the distance between the adsorption site atom and the oxygen atom beneath it upon adsorption of A (Δd*A, A = OH or O):
| Δd*A = d*A − d* | (9) |
To evaluate the impact of the different adsorption behaviour on the OER activity, the theoretical overpotential (η) for OER has been calculated using eqn (6). We have considered both the adsorbate evolution mechanism (AEM) given in eqn (1)–(4), along with alternative pathways which have previously been studied for rutile oxides, where the proton of the *OH and *OOH intermediates is transferred to a neighbouring bridging oxygen atom to form *O + Hb and *O2 + Hb (eqn (S1)–(S4)).27,69–72 Additionally, we considered the pathway in which molecular oxygen desorbs from the surface already in the third step, represented as O2(g) + Hb. Further details of the reaction pathways and illustrations of the intermediates can be found in Section S1 in the SI.
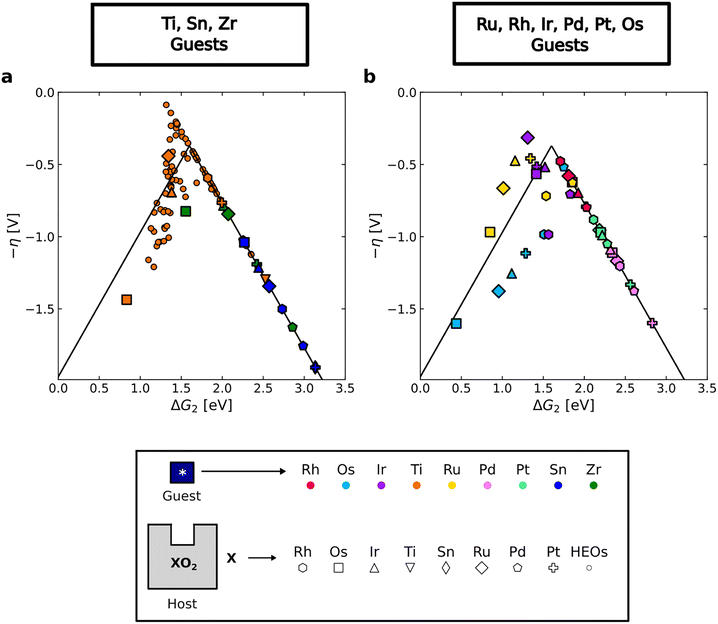
Volcano plots of the calculated theoretical overpotential based on the minimum energy pathway as a function of ΔG2 are shown in Fig. 4. Ti, Zr, and Sn guests (Fig. 4a) exhibit a significant variation in the overpotential compared to the Ru, Rh, Ir, Os, Pd and Pt sites (Fig. 4b) in accordance with the corresponding trends in the adsorption energies. The Ti ensemble is particularly remarkable, because it includes the high-activity region near the peak of the volcano plot. Notably, many Ti sites demonstrate activities beyond the conventional volcano (as low as η = 0.14 V), due to the absence of the known scaling relation ΔG*OOH ≈ ΔG*OH + 3.2 eV (see Fig. S14b). In particular, O2(g) + Hb and *O2 +Hb intermediates are found to be thermodynamically more stable than *OOH. Meanwhile, high catalytic activity is only marginally achieved at Zr sites, and not at all at Sn sites, due to the weak binding of the *O intermediate across the entire EFermi range. Among the platinum group elements, the Os guest data covers the widest activity range. This could be attributed to OsO2 having the highest EFermi among the studied oxides (Fig. 2), resulting in electrons donated by Os adsorption sites having similar energies to the electrons at the EFermi.
Inspired by the tunable activity observed for Ti guest sites across different host compositions (Fig. 4a), we now extend this single-site concept to a more advantageous catalyst design by considering TiO2 coatings on conductive rutile oxide substrates. Previous studies have shown that a TiO2 coating can be applied on conductive oxides, often with a minor decrease in the activity and improvement of the stability under acidic OER conditions.30–32 The feasibility of producing TiO2 coatings of one or two monolayers is supported by ref. 29, where an ultrathin protective TiO2 layer was formed by annealing RuO2 nanoparticles on a Nb-doped TiO2 support. The thickness of the TiO2 layer was ∼4.4 Å, compared with ∼4.0 Å for a single TiO2 monolayer. Moreover, the atomic layer deposition (ALD) technique has demonstrated exceptional control for fabricating TiO2 coatings on IrO2, RuO2, and F-doped SnO2 films.32 In ref. 32, the average growth rate of TiO2 was estimated via profilometric and scanning electron microscopy measurements to be 0.50–0.65 Å per ALD cycle. It is also possible that thicker TiO2 coatings can be used for OER catalysis without strongly compromising activity, since ref. 30 reports that TiO2 coatings up to 5.5 nm on Co3O4 increase durability without significantly affecting the activity relative to bare Co3O4.
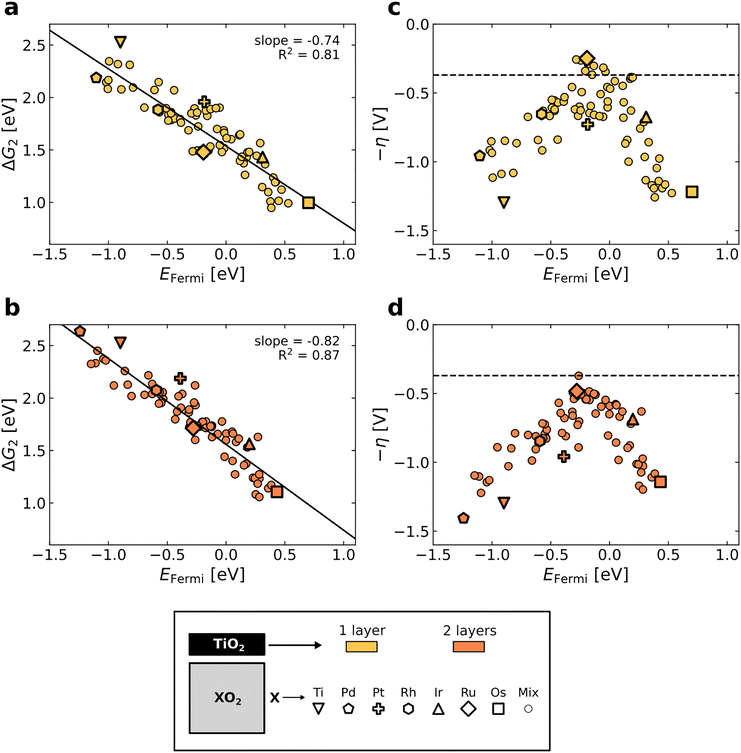
To investigate whether TiO2 coatings exhibit tunable activity, as observed for Ti guest sites, we calculated the adsorption energies and overpotentials on TiO2-coated rutile oxides composed of the conductive elements studied above, namely Ru, Rh, Ir, Os, Pt, and Pd (see Methodology section). The ΔG2 of these systems is plotted against the EFermi in Fig. 5a and b. A strong correlation is demonstrated (R2 > 0.8), with the linear fit showing a consistent slope of around −0.8 for one TiO2 and two TiO2 overlayers, close to the slope obtained for the single Ti sites (−0.92). This demonstrates the robustness of the EFermi as a descriptor for the adsorption energies on both single Ti sites and TiO2 overlayers. More details and illustrations of the slab structures in this dataset are provided in Sections S2 and S4 of the SI.
The theoretical overpotential based on the minimum energy pathway is directly compared to the EFermi for these systems in Fig. 5c and d. From these plots, it is evident that TiO2-coated oxides can be highly active if they have a suitable EFermi. Notably, a system with a EFermi close to 0 eV (compared to the EFermi of RuO2), can yield low overpotentials beyond the conventional volcano (Fig. 5c). The shape of these distributions suggests a volcano-type activity trend. Adding a second TiO2 overlayer increases the overpotential (Fig. 5d), consistent with experimental observations that thicker TiO2 coatings on Co3O4 progressively decrease OER performance.30 Scaling relations of the adsorption energies and conventional volcano plots for the TiO2-coated oxides data can be found in the Section S11 of the SI.
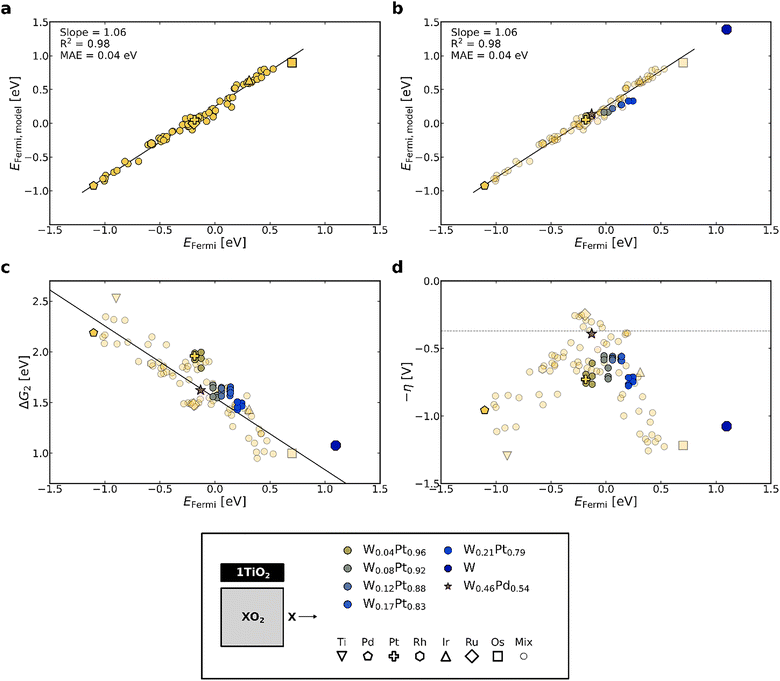
To further expand the use of the EFermi as a descriptor, we propose a model (EFermi,model) which predicts the EFermi of TiO2-coated oxides based on the composition of the substrate oxide (excluding the TiO2 overlayer). Specifically, this model is a weighted average of the EFermi of the pure oxides,
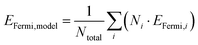 | (10) |
In Fig. 6a, EFermi,model values predicted by eqn (10) are plotted against the EFermi (calculated from eqn (7)) for the TiO2-coated oxides with one TiO2 overlayer. A strong correlation is observed (R2 = 0.98 and MAE = 0.04 eV), showing that the composition of the substrate oxide is a straightforward way to engineer the EFermi of TiO2-coated oxides. We note that while the model is clearly successful for mixtures primarily consisting of platinum-group elements, it may be too simplistic for some combinations of elements, for example, when mixing results in changes in oxidation states.
 | ||
| Fig. 6 (a) Correlation between the composition-based model (eqn (10)) and the real EFermi data obtained from DFT (eqn (7)) for oxides covered with one TiO2 overlayer. The black line corresponds to the best linear fit. (b) The WxPt1−xO2 and W0.46Pd0.54O2 substrate compositions have been predicted based on the model (eqn (10)). (c) Correlation between EFermi and ΔG2 for the WxPt1−xO2 and W0.46Pd0.54O2 substrate oxides. The semi-transparent data points and the linear fit (black line) correspond to the data shown in Fig. 5a. (d) Negative theoretical overpotentials (−η) as a function of the EFermi for the WxPt1−xO2 and W0.46Pd0.54O2 substrate oxides. The semi-transparent points correspond to the data shown in Fig. 5c. The dashed horizontal line indicates the value of the overpotential at the apex of the activity volcano defined by the conventional scaling relation ΔG*OOH ≈ ΔG*OH + 3.2 eV. | ||
Based on the observation that the activity of TiO2-coated rutile oxides can be tuned through the substrate oxide EFermi (Fig. 5), and that the EFermi of the TiO2-coated systems can be predicted from the substrate oxide composition using the linear model in eqn (10) (Fig. 6a), we now apply this to predict Ir- and Ru-free OER candidates. We also exclude Rh and Os, because they are equally or even more scarce and expensive than Ru and Ir.16 Instead, we select TiO2-coated PtO2 as a representative system, whose activity is not yet optimal, and dope it with tungsten (W) to tune EFermi.
In Fig. 6b, we show that the model (eqn (10)) successfully predicts the EFermi of the TiO2-coated WxPt1−xO2 data. The two distinct rutile slabs for each composition are found to have similar EFermi, indicating that EFermi is largely controlled by composition rather than the specific arrangement of metal atoms in the slab. In Fig. 6c, we see that the adsorption energies vary almost linearly with the EFermi, and the trend is shifted smoothly from the TiO2-coated PtO2 towards the TiO2-coated WO2 direction, as the amount of W increases. The observed scatter arises from an inequivalent local coordination environment associated with the distinct atomic configurations at the three different Ti-cus sites sampled on each composition. This indicates that EFermi is not a perfect descriptor, since all six adsorption sites (three Ti sites for each of the two atomic configurations) experience similar EFermi. In Fig. 6d, it is evident that these systems follow the expected volcano trend. The largest improvement in overpotential is observed for W0.12Pt0.88O2 and W0.17Pt0.83O2 (as low as 0.55 V), compared with the PtO2 substrate composition (0.73 V), indicating that higher activity can be achieved while reducing precious-metal content. Increasing the W content further to 21 at% reduces the activity, indicating that the WxPt1−xO2 substrate composition is optimal at x = 12–17 at%.
We applied the same design principle to predict active Pd-based catalysts. In Fig. 6, we show TiO2-coated W0.46Pd0.54O2 (purple star), which has a significantly lower overpotential (η = 0.39 V) than the TiO2-coated PdO2 (η = 0.96 V). Together with the TiO2-coated WxPt1−xO2 results, this demonstrates that tuning the substrate EFermi provides a robust route to designing Ru and Ir free catalysts.
Conclusions
In summary, our work presents a theoretical design principle for active and potentially stable OER electrocatalysts under acidic conditions. We find that elements which cannot be oxidised beyond the 4+ oxidation state in the rutile structure (Ti, Sn, and Zr) exhibit adsorption energies and activities that are correlated with the EFermi of the surrounding oxide, whereas this behaviour is not observed for Pt, Pd, Ir, Os, Ru, and Rh sites. Among these, Ti is found to be particularly special, as it spans a wide range of adsorption energies, many of which correspond to very low overpotentials (η = 0.14 V).We then show that these observations on single Ti sites can be transferred to a more advantageous catalyst design by studying TiO2 coatings on conductive rutile oxide substrates. For these systems, a volcano-type relationship is observed between activity and EFermi, demonstrating that a TiO2-coated oxide with a suitable EFermi can be highly active and potentially stable under acidic OER conditions, consistent with experimental reports that TiO2 coatings can preserve activity while improving stability in acid.19,29–32 Furthermore, we show that a simple linear model can accurately predict the EFermi of TiO2-coated oxides based on the composition of the substrate oxide. Using this model, we identify Ir and Ru free substrate compositions, WxPt1−xO2 and W0.46Pd0.54O2, with EFermi values that can activate the TiO2 overlayer, illustrating a practical route to designing Ir and Ru free acidic OER catalysts.
Author contributions
J. R. supervised the whole project. G. K. S. performed the DFT calculations and data analysis supervised by K. L. S., H. H. K., and J. R. G. K. S., H. H. K., and K. L. S. wrote the manuscript. All authors discussed the results, provided insights and suggestions, and helped improve the manuscript.Conflicts of interest
The authors have submitted a patent application (European application under number EP 25387103.2) covering this work.Data availability
The data and analysis that support the findings of this study are openly available at https://www.erda.dk/archives/f73eb5ea1d7a1897bfa54659f714b3cd/published-archive.html.Supplementary information (SI): reaction pathways for the OER; DFT calculations setup; RuRhIrOsTi high entropy oxide dataset; description of the model structures; Fermi level alignment using O 2s band centroid; trends in ΔG*O and EFermi; scaling relations between adsorption energies; Bader charge differences in pure oxide hosts; change in distance between adsorption site and oxygen atom beneath in pure oxide hosts; conventional volcano plots for TiO2-coated metal oxides; and D3-corrections. See DOI: https://doi.org/10.1039/d5ee07605f.
Acknowledgements
We acknowledge financial support from the Danish National Research Foundation, Center for High Entropy Alloy Catalysis (CHEAC) DNRF149, the European Union under ERC Synergy grant DEMI, GA no. 101118768, and V-Sustain: The VILLUM Centre for the Science of Sustainable Fuels and Chemicals (#9455) from VILLUM FONDEN.Notes and references
- M. K. Singla, P. Nijhawan and A. S. Oberoi, Environ. Sci. Pollut. Res., 2021, 28, 15607–15626 CrossRef CAS PubMed.
- A. Grimaud, K. J. May, C. E. Carlton, Y. L. Lee, M. Risch, W. T. Hong, J. Zhou and Y. Shao-Horn, Nat. Commun., 2013, 4, 2439 Search PubMed.
- M. N. Lakhan, A. Hanan, A. Hussain, I. Ali Soomro, Y. Wang, M. Ahmed, U. Aftab, H. Sun and H. Arandiyan, Chem. Commun., 2024, 60, 5104–5135 RSC.
- S. A. Grigoriev, V. N. Fateev, D. G. Bessarabov and P. Millet, Int. J. Hydrogen Energy, 2020, 45, 26036–26058 CrossRef CAS.
- E. Fabbri and T. J. Schmidt, ACS Catal., 2018, 8, 9765–9774 CrossRef CAS.
- J. Rossmeisl, A. Logadottir and J. K. Nørskov, Chem. Phys., 2005, 319, 178–184 CrossRef CAS.
- I. C. Man, H. Y. Su, F. Calle-Vallejo, H. A. Hansen, J. I. Martínez, N. G. Inoglu, J. Kitchin, T. F. Jaramillo, J. K. Nørskov and J. Rossmeisl, ChemCatChem, 2011, 3, 1159–1165 CrossRef CAS.
- N. Danilovic, R. Subbaraman, K. C. Chang, S. H. Chang, Y. J. Kang, J. Snyder, A. P. Paulikas, D. Strmcnik, Y. T. Kim, D. Myers, V. R. Stamenkovic and N. M. Markovic, J. Phys. Chem. Lett., 2014, 5, 2474–2478 CrossRef CAS PubMed.
- M. Escudero-Escribano, A. F. Pedersen, E. A. Paoli, R. Frydendal, D. Friebel, P. Malacrida, J. Rossmeisl, I. E. L. Stephens and I. Chorkendorff, J. Phys. Chem. B, 2018, 122, 947–955 CrossRef CAS PubMed.
- Y. Lee, J. Suntivich, K. J. May, E. E. Perry and Y. Shao-Horn, J. Phys. Chem. Lett., 2012, 3, 399–404 Search PubMed.
- D. Lebedev, M. Povia, K. Waltar, P. M. Abdala, I. E. Castelli, E. Fabbri, M. V. Blanco, A. Fedorov, C. Copéret, N. Marzari and T. J. Schmidt, Chem. Mater., 2017, 29, 5182–5191 Search PubMed.
- D. F. Abbott, D. Lebedev, K. Waltar, M. Povia, M. Nachtegaal, E. Fabbri, C. Copéret and T. J. Schmidt, Chem. Mater., 2016, 28, 6591–6604 Search PubMed.
- K. A. Stoerzinger, L. Qiao, M. D. Biegalski and Y. Shao-Horn, J. Phys. Chem. Lett., 2014, 5, 1636–1641 Search PubMed.
- P. E. Pearce, C. Yang, A. Iadecola, J. Rodriguez-Carvajal, G. Rousse, R. Dedryvère, A. M. Abakumov, D. Giaume, M. Deschamps, J.-M. Tarascon and A. Grimaud, Chem. Mater., 2019, 31, 5845–5855 Search PubMed.
- D. B. Rogers, R. D. Shannon, A. W. Sleight and J. L. Gillson, Inorg. Chem., 1969, 8, 841–849 CrossRef CAS.
- P. C. K. Vesborg and T. F. Jaramillo, RSC Adv., 2012, 2, 7933–7947 RSC.
- R. Wan, T. Yuan, L. Wang, B. Li, M. Liu and B. Zhao, Nat. Catal., 2024, 7, 1288–1304 CrossRef CAS.
- W. T. Hong, M. Risch, K. A. Stoerzinger, A. Grimaud, J. Suntivich and Y. Shao-Horn, Energy Environ. Sci., 2015, 8, 1404–1427 Search PubMed.
- M. Pourbaix, Atlas of Electrochemical Equilibria in Aqueous Solutions, National Association of Corrosion Engineers, TX, 2 edn, 1974 Search PubMed.
- T. Kutlusoy, S. Divanis, R. Pittkowski, R. Marina, A. M. Frandsen, K. Minhova-Macounova, R. Nebel, D. Zhao, S. F. L. Mertens, H. Hoster, P. Krtil and J. Rossmeisl, Chem. Sci., 2022, 13, 13879–13892 RSC.
- R. K. B. Karlsson, A. Cornell and L. G. M. Pettersson, Electrochim. Acta, 2015, 180, 514–527 Search PubMed.
- M. García-Mota, A. Vojvodic, F. Abild-Pedersen and J. K. Nørskov, J. Phys. Chem. C, 2012, 117, 460–465 CrossRef.
- N. Roy, Y. Sohn, K. T. Leung and D. Pradhan, J. Phys. Chem. C, 2014, 118, 29499–29506 CrossRef CAS.
- D. M. Jang, I. H. Kwak, E. L. Kwon, C. S. Jung, H. S. Im, K. Park and J. Park, J. Phys. Chem. C, 2015, 119, 1921–1927 Search PubMed.
- A. R. Akbashev, L. Zhang, J. T. Mefford, J. Park, B. Butz, H. Luftman, W. C. Chueh and A. Vojvodic, Energy Environ. Sci., 2018, 11, 1762–1769 RSC.
- L. Zhang, A. S. Raman and A. Vojvodic, Chem. Mater., 2020, 32, 5569–5578 Search PubMed.
- K. L. Svane and J. Rossmeisl, Angew. Chem., 2022, 61, e202201146 CrossRef CAS PubMed.
- S. Trasatti, Electrochim. Acta, 2000, 45, 2377–2385 CrossRef CAS.
- H. W. Lim, D. K. Cho, J. H. Park, S. G. Ji, Y. J. Ahn, J. Y. Kim and C. W. Lee, ACS Catal., 2021, 11, 12423–12432 CrossRef CAS.
- T. Tran-Phu, H. Chen, R. Daiyan, M. Chatti, B. Liu, R. Amal, Y. Liu, D. R. Macfarlane, A. N. Simonov and A. Tricoli, ACS Appl. Mater. Interfaces, 2022, 14, 33130–33140 CrossRef CAS PubMed.
- R. Frydendal, E. A. Paoli, I. Chorkendorff, J. Rossmeisl and I. E. L. Stephens, Adv. Energy Mater., 2015, 5, 1500991 Search PubMed.
- C. E. Finke, S. T. Omelchenko, J. T. Jasper, M. F. Lichterman, C. G. Read, N. S. Lewis and M. R. Hoffmann, Energy Environ. Sci., 2019, 12, 358–365 RSC.
- B. Endrődi, E. Kecsenovity, K. Rajeshwar and C. Janáky, ACS Appl. Energy Mater., 2018, 1, 851–858 Search PubMed.
- J. Rossmeisl, Z. W. Qu, H. Zhu, G. J. Kroes and J. K. Nørskov, J. Electroanal. Chem., 2007, 607, 83–89 CrossRef CAS.
- J. K. Nørskov, J. Rossmeisl, A. Logadottir, L. Lindqvist, J. R. Kitchin, T. Bligaard and H. Jónsson, J. Phys. Chem. B, 2004, 108, 17886–17892 CrossRef PubMed.
- E. Fabbri, A. Habereder, K. Waltar, R. Kötz and T. J. Schmidt, Catal. Sci. Technol., 2014, 4, 3800–3821 Search PubMed.
- B. Hammer and J. K. Nørskov, Surf. Sci., 1995, 343, 211–220 CrossRef CAS.
- B. Hammer and J. K. Norskov, Nature, 1995, 376, 238–240 CrossRef CAS.
- S. A. Akhade and J. R. Kitchin, J. Chem. Phys., 2012, 137, 084703 CrossRef PubMed.
- J. Suntivich, K. J. May, H. A. Gasteiger, J. B. Goodenough and Y. Shao-Horn, Science, 2011, 334, 1383–1385 CrossRef CAS PubMed.
- Y.-L. Lee, J. Kleis, J. Rossmeisl, Y. Shao-Horn and D. Morgan, Energy Environ. Sci., 2011, 4, 3966–3970 RSC.
- Z. Xu and J. R. Kitchin, J. Chem. Phys., 2015, 142, 104703 CrossRef PubMed.
- L. Giordano, K. Akkiraju, R. Jacobs, D. Vivona, D. Morgan and Y. Shao-Horn, Acc. Chem. Res., 2022, 55, 298–308 CrossRef CAS PubMed.
- G. Jomard, T. Petit, A. Pasturel, L. Magaud, G. Kresse and J. Hafner, Phys. Rev. B: Condens. Matter Mater. Phys., 1999, 59, 4044–4052 CrossRef CAS.
- Y. Han and J. Zhu, Top. Catal., 2013, 56, 1525–1541 Search PubMed.
- S. F. Matar, G. Demazeau, M. H. Möller and R. Pöttgen, Chem. Phys. Lett., 2011, 508, 215–218 CrossRef CAS.
- I. Shaplygin, G. Aparnikov and V. Lazarev, Russ. J. Inorg. Chem., 1978, 23, 488 Search PubMed.
- A. A. Levchenko, G. Li, J. Boerio-Goates, B. F. Woodfield and A. Navrotsky, Chem. Mater., 2006, 18, 6324–6332 CrossRef CAS.
- H. S. C. O'Neill and J. Nell, Geochim. Cosmochim. Acta, 1997, 61, 5279–5293 CrossRef.
- L. Gracia, A. Beltrán and J. Andrés, J. Phys. Chem. B, 2007, 111, 6479–6485 CrossRef CAS PubMed.
- R. K. Nomiyama, M. J. Piotrowski and J. L. F. Da Silva, Phys. Rev. B: Condens. Matter Mater. Phys., 2011, 84, 100101(R) CrossRef.
- R. D. Shannon, Solid State Commun., 1968, 6, 139–143 CrossRef CAS.
- H. Perron, C. Domain, J. Roques, R. Drot, E. Simoni and H. Catalette, Theor. Chem. Acc., 2007, 117, 565–574 Search PubMed.
- C. Roy, R. R. Rao, K. A. Stoerzinger, J. Hwang, J. Rossmeisl, I. Chorkendorff, Y. Shao-Horn and I. E. L. Stephens, ACS Energy Lett., 2018, 3, 2045–2051 Search PubMed.
- T. Weber, V. Vonk, D. Escalera-López, G. Abbondanza, A. Larsson, V. Koller, M. J. S. Abb, Z. Hegedüs, T. Bäcker, U. Lienert, G. S. Harlow, A. Stierle, S. Cherevko, E. Lundgren and H. Over, ACS Catal., 2021, 11, 12651–12660 Search PubMed.
- V. A. Mints, K. L. Svane, J. Rossmeisl and M. Arenz, ACS Catal., 2024, 14, 6936–6944 Search PubMed.
- M. Busch, N. B. Halck, U. I. Kramm, S. Siahrostami, P. Krtil and J. Rossmeisl, Nano Energy, 2016, 29, 126–135 Search PubMed.
- H. H. Kristoffersen and H. Metiu, J. Phys. Chem. C, 2015, 119, 10500–10506 CrossRef CAS.
- J. Heyd, G. E. Scuseria and M. Ernzerhof, J. Chem. Phys., 2003, 118, 8207–8215 CrossRef CAS.
- J. Paier, M. Marsman, K. Hummer, G. Kresse, I. C. Gerber and J. G. Angyan, J. Chem. Phys., 2006, 124, 154709 CrossRef CAS PubMed.
- H. Moustafa, J. J. Mortensen, J. Rossmeisl and K. W. Jacobsen, Appl. Surf. Sci., 2024, 649, 159168 CrossRef CAS.
- I. E. Castelli, F. Hüser, M. Pandey, H. Li, K. S. Thygesen, B. Seger, A. Jain, K. A. Persson, G. Ceder and K. W. Jacobsen, Adv. Energy Mater., 2014, 5, 1400915 CrossRef.
- I. E. Castelli, I.-C. Man, S.-G. Soriga, V. Parvulescu, N. B. Halck and J. Rossmeisl, J. Phys. Chem. C, 2017, 121, 18608–18614 CrossRef CAS.
- Y. Liu, X. Hu, C. Liu, S. Zhu, K. Jiang, F. Liu and S. Zheng, Green Energy Environ., 2024, 9, 937–948 CrossRef CAS.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, J. Chem. Phys., 2010, 132, 154104 CrossRef PubMed.
- F. Abild-Pedersen, J. Greeley, F. Studt, J. Rossmeisl, T. R. Munter, P. G. Moses, E. Skúlason, T. Bligaard and J. K. Nørskov, Phys. Rev. Lett., 2007, 99, 016105 CrossRef CAS PubMed.
- S. Divanis, T. Kutlusoy, I. M. Ingmer Boye, I. C. Man and J. Rossmeisl, Chem. Sci., 2020, 11, 2943–2950 RSC.
- G. Henkelman, A. Arnaldsson and H. Jónsson, Comput. Mater. Sci., 2006, 36, 354–360 CrossRef.
- N. B. Halck, V. Petrykin, P. Krtil and J. Rossmeisl, Phys. Chem. Chem. Phys., 2014, 16, 13682–13688 RSC.
- A. Grimaud, O. Diaz-Morales, B. Han, W. T. Hong, Y. L. Lee, L. Giordano, K. A. Stoerzinger, M. T. M. Koper and Y. Shao-Horn, Nat. Chem., 2017, 9, 457–465 CrossRef CAS PubMed.
- E. Inico, G. Di Liberto and L. Giordano, ChemCatChem, 2024, 16, e202400813 Search PubMed.
- R. R. Rao, M. J. Kolb, N. B. Halck, A. F. Pedersen, A. Mehta, H. You, K. A. Stoerzinger, Z. Feng, H. A. Hansen, H. Zhou, L. Giordano, J. Rossmeisl, T. Vegge, I. Chorkendorff, I. E. L. Stephens and Y. Shao-Horn, Energy Environ. Sci., 2017, 10, 2626–2637 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |