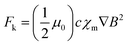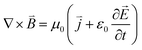Spontaneous magnetic field enhancement in nano-frameworks enables efficient ORR
Zhijie
Qi
a,
Zhenjie
Lu
a,
Pengcheng
Yao
*b,
Wuxin
Ba
 a,
Lianjin
Wei
a,
Duansheng
Liu
a,
Jun
Jiang
a,
Shujun
Liu
a,
Pawel J.
Kulesza
a,
Lianjin
Wei
a,
Duansheng
Liu
a,
Jun
Jiang
a,
Shujun
Liu
a,
Pawel J.
Kulesza
 c,
Jingwen
Sun
c,
Jingwen
Sun
 a,
Pan
Xiong
a,
Pan
Xiong
 a,
Xin
Wang
a,
Junwu
Zhu
a,
Xin
Wang
a,
Junwu
Zhu
 *a and
Yongsheng
Fu
*a and
Yongsheng
Fu
 *a
*a
aKey Laboratory for Soft Chemistry and Functional Materials, School of Chemistry and Chemical Engineering, Nanjing University of Science and Technology, Nanjing 210094, China. E-mail: zhujw@njust.edu.cn; fuyongsheng@njust.edu.cn
bDepartment of Chemical and Biomolecular Engineering and Ralph O'Connor Sustainable Energy Institute, Johns Hopkins University, Baltimore, Maryland 21218, USA. E-mail: pyao4@jh.edu
cFaculty of Chemistry, University of Warsaw, Pasteura 1, PL 02-093 Warsaw, Poland
First published on 13th February 2026
Abstract
The oxygen reduction reaction (ORR) is a pivotal process in sustainable energy conversion and storage devices, such as fuel cells and metal–air batteries. However, its efficiency is severely restricted by sluggish oxygen (O2) mass transfer. Herein, we report the construction of CoPt3-based nano-framework catalysts (CoPt3@NC-NFs) with a unique architecture composed solely of edges and corners. The framework geometry concentrates current density during electrochemical reactions, thereby amplifying the surface magnetic field strength and enhancing the Kelvin force acting on paramagnetic O2 molecules. This effect actively drives O2 toward high-field regions on the catalyst surface, enabling directional and accelerated mass transport. The increased Kelvin force promotes the movement of O2 to high magnetic field regions on the catalysts’ surface, thereby enhancing mass transport of O2. Consequently, the CoPt3@NC-NFs exhibit outstanding performance in both the ORR (Eonset = 1.02 V, E1/2 = 0.89 V, j = −5.8 mA cm−2 in 0.1 M KOH) and Zn–air batteries (with a power density of 184.8 mW cm−2 at 305 mA cm−2). This work establishes a clear correlation between catalyst microstructure and surface magnetic field, revealing a spontaneous magnetic enhancement mechanism that introduces a new physical perspective for enhancing electrocatalytic activity.
Broader contextThe oxygen reduction reaction (ORR) plays a central role in energy-conversion technologies such as fuel cells and metal–air batteries. However, the overall efficiency of ORR systems is critically constrained by the rate of O2 mass transfer, which governs the supply of reactants to catalytic active sites. Conventional strategies mainly rely on structural engineering to shorten diffusion pathways or increase accessible surface area, yet they still suffer from inherently slow and nondirectional diffusion. This limitation becomes particularly prominent in complex micro/nanostructured catalysts, where a mismatch often persists between O2 transport and active site distribution. This work introduces a fundamentally different concept: leveraging nano-framework architectures to spontaneously enhance the surface magnetic field of the catalyst during ORR. The unique edge- and corner-dominated framework concentrates current, strengthens the intrinsic magnetic field, and actively directs paramagnetic O2 toward catalytic sites through a Kelvin-force-driven mechanism. By integrating structural design with magnetic-field-regulated mass transport, this study provides a generalizable approach for overcoming ORR mass-transfer limitations and offers new design principles for advanced electrocatalysts in next-generation energy-conversion technologies. |
Introduction
The oxygen reduction reaction (ORR) is a crucial cathode reaction in energy conversion devices such as fuel cells and metal–air batteries, with its efficiency directly determining the overall device performance.1,2 However, the ORR process is fundamentally constrained by the inherent limitations of the four-electron transfer multi-step reaction mechanism and slow mass transport, resulting in low reaction efficiency that severely compromises the overall device performance.3–5 In recent years, strategies based on electronic structure engineering,6–8 including heteroatoms doping,9,10 defect engineering,11,12 and nano-confinement effects,13,14 have been extensively explored to modulate the d-band center and charge distribution of catalytic active sites. These approaches have significantly enhanced ORR catalytic activity and reaction kinetics. However, these strategies remain primarily confined to enhancing catalytic activity, with limited progress made in addressing the critical bottleneck of O2 mass transfer kinetics. This leads to underutilization of active sites and ultimately impairs the full catalytic potential.In electrochemical catalytic systems, the mass transport at the catalyst–electrolyte interface is jointly governed by three fundamental mechanisms: concentration diffusion, electric field migration, and convection. Current research predominantly focuses on enhancing O2 transport in the ORR by promoting concentration diffusion through micro/nano-structural engineering—such as constructing hollow and porous structures with larger specific surface areas.15–18 However, as the reaction proceeds, local concentration gradients diminish, leading to a gradual decline in diffusion-driven transport efficiency. Moreover, the inherently stochastic nature of diffusion fails to achieve directional enrichment of O2 at the catalytic active centers. This spatial mismatch between mass transport and reaction sites results in a substantial portion of active sites remaining idle, constituting a critical barrier to further improving ORR performance.
It is worth noting that ground-state O2 molecules exhibit intrinsic paramagnetism (↑O·O↑), which provides a unique physical basis for magnetic-field-mediated regulation of mass transport.19–22 In an inhomogeneous magnetic field, paramagnetic species are subject to a magnetic body force, commonly referred to as the Kelvin force. This force originates from the interaction between the magnetic susceptibility of the species and the spatial gradient of the magnetic field, and acts to drive paramagnetic molecules toward regions of higher magnetic field intensity.23,24 The magnitude of this force is given by:
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) m−3, χm is the molar susceptibility, and ∇B2 is the gradient of the magnetic induction.
m−3, χm is the molar susceptibility, and ∇B2 is the gradient of the magnetic induction.
Importantly, this relationship highlights that the Kelvin force scales with both the magnetic field intensity and, more critically, its spatial gradient. In practical electrocatalytic systems, although the Kelvin force acting on individual O2 molecules is relatively weak compared to thermal motion, it can function as a biasing force at the micro/nanoscale. When combined with concentration-driven diffusion and electrochemically induced convection, the Kelvin force can preferentially guide paramagnetic O2 molecules toward regions of enhanced magnetic field near the catalyst surface, thereby facilitating local O2 enrichment and surface adsorption. Existing studies primarily rely on externally applied magnetic fields to establish magnetic field gradients, thereby accelerating the transport of O2 in the electrolyte via magnetophoresis.25,26 However, this approach fails to achieve directional enrichment of O2 molecules at the active sites on the micro/nanoscale.27,28 Therefore, designing catalysts with intrinsic magnetic fields through precise structural engineering to actively and directionally guide O2 molecules toward catalytic active sites remains a largely unexplored strategy.
It is well known that an uneven magnetic field will generate around the material when current flows through it during electrochemical processes. These intrinsic magnetic fields exert a Kelvin force on paramagnetic O2 molecules, thereby providing a driving force for their active and directional mass transport. According to Maxwell–Ampère's law:
This relation reveals that the local magnetic field intensity (B) is linearly proportional to the surface current density (J). When the geometric design of the catalyst induces current focusing, a significant enhancement of the surface magnetic field can be achieved. According to the Kelvin force equation, the driving force for the directional transport of O2 molecules is directly proportional to the magnetic field gradient. Therefore, designing catalyst structures to concentrate surface current density and thereby enhance their intrinsic magnetic fields provides an effective strategy to actively guide O2 molecules toward catalytic active sites, maximizing their utilization.
Herein, we developed a simple yet effective strategy for spontaneous surface magnetic field enhancement by constructing a CoPt3-based nano-catalyst (CoPt3@NC-NFs) with a unique nano-framework structure composed exclusively of edges and corners. This design enables the active and directional enrichment of O2 molecules at catalytic active sites, thereby significantly enhancing the ORR catalytic performance. Finite element simulations reveal that during electrocatalysis, the current is spatially confined along the boundaries of the nano-framework, resulting in locally intensified current densities along specific paths and consequently inducing spontaneous surface magnetic field enhancement. This enhanced magnetic field further enhances the Kelvin force acting on paramagnetic O2 molecules, facilitating their rapid and directional mass transport toward the catalyst surface and ultimately boosting the overall catalytic activity. In situ Raman spectroscopy and scanning electrochemical microscopy (SECM) confirmed that the magnetic field enhancement significantly accelerates the directional enrichment of O2 molecules at the catalyst surface, resulting in a substantial enhancement of ORR performance. This study underscores the unique capability of nano-framework structures to induce spontaneous magnetic field enhancement during electrocatalysis, and through the integration of simulation and experimental results, elucidates the underlying mechanism by which magnetic field enhancement drives the active and directional accumulation of O2 at catalytic active sites.
Results and discussion
DFT calculation and finite element simulation of CoPt3-based nano-framework materials in ORR
Density functional theory (DFT) calculations were performed on three Pt-based catalysts, Pt (111), CoPt (111) and CoPt3 (111), to explore their potential reactivity in ORR. Fig. 1a shows the Gibbs free energy diagrams of the 4e− pathway processes for the three Pt-based catalysts constructed at U = 0 V and U = 1.23 V. It can be observed that the energy barriers of all reaction steps for these catalysts exhibit a decreasing trend, indicating that all three catalysts undergo thermodynamically favorable processes during the ORR (U = 0 V). At U = 1.23 V, the three catalysts exhibit different potential-determining step (PDS). The PDS for CoPt3 is the formation of *OOH (O2 + e− → *OOH) with a Gibbs free energy (ΔG) value of 0.51 eV. For Pt, O*+ e− → *OH is the PDS in ORR process (ΔG = 0.79 eV). For CoPt, it is desorption of *OH (*OH + e− → OH−) (ΔG = 1.07 eV). Among these catalysts, CoPt3 exhibits the lowest reaction energy barrier for the PDS, indicating its superior potential catalytic activity in the ORR. Fig. 1b shows the optimized ORR pathway on CoPt3.The project density of states (PDOS) in Fig. 1c shows that CoPt3 has a lower d-band center (−2.29 eV), which is adverse to the bonding between the catalysts and oxygen, further weakening the adsorption of O2 on the catalysts as well as blocking the transfer of electrons and protons to O2. This also explains the high energy barrier for *OOH formation during the reaction process (Fig. S1). Therefore, promoting the adsorption of O2 on CoPt3 will effectively enhance the reactivity of the catalysts in the ORR. Moreover, the TDOS of O2 exhibited asymmetry between the upper and lower electronic states, indicative of the magnetic properties of O2 (Fig. S2). During the electrocatalytic process, the surface magnetic field of the CoPt3 catalysts exerts a Kelvin force on paramagnetic species, driving their migration toward regions of higher magnetic field intensity. Considering the paramagnetic nature of O2 molecules, the stronger Kelvin force facilitates their active transport toward the catalysts’ surface, enhancing their adsorption at active sites and thereby improving ORR catalytic performance (Fig. 1d). Therefore, constructing a stronger magnetic field gradient at the catalysts’ surface offers an effective means to modulate the influence of the Kelvin force on paramagnetic reactants during ORR, ultimately promoting catalytic activity.
In order to verify the hypothesis that nano-framework structure can spontaneously produce stronger surface magnetic fields, finite element simulation method is used to study the surface magnetic fields generated by different structures in the electrochemical process. Based on the structural characteristics of CoPt3 catalysts, three models were constructed: a cubic model, a hollow model, and a framework model, each with dimensions of 500 × 500 × 500 nm. The hollow model had a shell thickness of 20 nm, while the framework model had an edge thickness of 100 nm (Fig. 1e). The simulations were performed under a series of applied currents, allowing the current-dependent evolution of the magnetic field to be examined (Fig. S3–S5). The results demonstrate that, across the investigated current range, the CoPt3 framework model consistently generates stronger magnetic fields than both the cubic and hollow models. For example, at an applied current of 10 nA, magnetic flux lines are more densely concentrated around the CoPt3 framework structure. In this region, the maximum magnetic field intensity is approximately 1.25 times that of the cubic model and 1.34 times that of the hollow model, while the minimum magnetic field intensity is 2.4 and 2.27 times higher, respectively (Fig. 1f–h). Notably, as the applied current increases, the enhancement of magnetic field strength becomes more pronounced for the framework-structured catalyst compared to the other morphologies, indicating a stronger average magnetic field distribution under higher current conditions. These illustrate that the CoPt3 framework model exhibits a stronger average magnetic field distribution. Observations of the magnetic field lines revealed additional distributions within the hollow core of the CoPt3 framework model. Analysis of the XY plane further demonstrated a higher density of magnetic field lines in the CoPt3 framework material model, which directly corresponds to an increase in the magnetic field B value in the Kelvin force (Fig. S6). Moreover, the simulated magnetic field distribution reveals that the field intensity increases progressively toward the catalyst surface, which favors the directional migration of paramagnetic species to the active interface. These results support our initial hypothesis that the CoPt3 nano-framework structure can effectively enhance the local magnetic field intensity at the surface of catalysts.
Synthesis and characterization of catalysts
To prepare CoPt3-based catalysts with the aforementioned nano-framework structure, we employed a self-template method previously reported by our research group, using cubic ZIF-67 as a precursor to synthesize the special ZIF-L nano-framework structure through the Marangoni effect.29 Subsequently, the ZIF-L NFs was annealed in an N2 atmosphere to obtain Co nano-particle-loaded nitrogen-doped carbon with framework structure (Co@NC-NFs). Finally, after exchanging Pt2+ on the Co@NC-NFs and recombining them under a nitrogen atmosphere, we successfully prepared N-doped carbon loaded with CoPt3 nanoparticles with a nano-framework structure (CoPt3@NC-NF) as shown in Fig. 2a. In addition, to more effectively exclude the influence of large surface area and porosity, hollow H-ZIF-67 obtained via tannic acid etching and pristine ZIF-67 were subjected to the aforementioned two-step pyrolysis, yielding CoPt3@NC with a cubic structure and CoPt3@NC-H with a hollow structure, respectively (Fig. S7–S9). Field emission scanning electron microscopy (FE-SEM) images demonstrate the transformation of the cubic structure of ZIF-67 to the ZIF-L nano framework structure (Fig. S10 and S11). The average size of ZIF-L is approximately around 500 nm. FE-SEM confirms that CoPt3@NC-NFs maintains a well-defined nano-frame structure (Fig. 2b).X-ray diffraction (XRD) analysis characterized the crystal structure of the prepared material, revealing a successful transformation of crystal structure after the two-step pyrolysis (Fig. 2c). The obtained CoPt3@NC-NFs exhibited three diffraction peaks, which matched well with the typical FCC structure of CoPt3 (PDF#29-0499). High-resolution transmission electron microscopy (HR-TEM) reveals uniform distribution of metal particles with diameters ranging from 5 to 20 nm (Fig. S12a). Further HR-TEM observations (Fig. S12b) displayed distinct lattice fringes, with the interplanar distance corresponding to the CoPt3 (111) plane measured at 0.226 nm and that of the (200) plane at 0.197 nm. Subsequent EDS-mapping illustrates the homogeneous distribution of elements such as Co, Pt, N, and C in the material (Fig. S13). In order to obtain detailed structural information of CoPt3 nanoparticles, aberration-corrected high-angle annular dark field scanning transmission electron microscopy (AC-HAADF-STEM) was employed. CoPt3 nanoparticles placed along the [101] direction and the [001] direction are shown in Fig. 2d and f, and their fast Fourier transform (FFT) results lead to the identification of the neighboring CoPt3 structure (Fig. 2e).30 As shown in Fig. 2g, the nanoparticles in this area along the [001] axis are composed of periodic activation of pure Co–Pt. The line intensity distribution along the white dashed rectangular area in Fig. 2h further confirms the formation of an ordered metallic interphase, because the lattice spacing = 0.38 nm corresponds to the (100) plane of CoPt3. In addition, the uniform distribution of Pt and Co elements can also be seen in the element mapping of the single particles (Fig. 2i–k). The above proves the successful preparation of the framework material of CoPt3 particles.
Through Raman spectroscopy analysis (Fig. S14), CoPt3@NC-NFs show a lower ID/IG ratio (0.95), indicating a higher level of graphitization in the material, which will enhance its conductivity. The pore characteristics of the CoPt3@NC-NFs and CoPt3@NC were determined through N2 adsorption–desorption isotherm measurements. Both materials exhibited typical type-IV adsorption curves, indicating a predominance of mesoporous structures (Fig. S15–S17). Specifically, the average pore size in CoPt3@NC-NFs was larger than that in CoPt3@NC, with the pore size distribution of CoPt3@NC concentrated around 4 nm, while in CoPt3@NC-NFs the distribution was greater than 6 nm, facilitating the transport of reactants and products. Additionally, CoPt3@NC-NFs had a larger BET specific surface area (180.0 m2 g−1) than CoPt3@NC (120.3 m2 g−1), providing an increased surface area available for reaction. The X-ray photoelectron spectroscopy (XPS) analysis of the synthesized CoPt3@NC-NFs and CoPt3@NC catalysts confirmed the presence of Pt, Co, N, and C elements (Fig. S18–S23), with a Pt/Co atomic ratio close to 3 (Table S1). As shown in Fig. S23, the high-resolution Pt 4f and Co 3d spectra of CoPt3@NC-NFs were compared to CoPt3@NC, revealing a slight negative shift in binding energy for the elements. This shift may be attributed to differences in N doping levels in the carbon materials derived from different precursors. By finely resolving the XPS peaks of Pt, we identified that 71.1 eV and 74.4 eV correspond to the Pt0, while 72.2 eV and 75.5 eV correspond to the Pt2+.31 The analysis of Co 2p yields 778.3 eV and 793.2 eV corresponding to Co0, 782.5 eV and 797.4 eV corresponding to Co2+, and 780.5 eV and 795.7 eV corresponding to Co3+.32 In order to further investigate the influence of the special structure on the magnetic properties of the materials, we compared the hysteresis loops of CoPt3@NC and CoPt3@NC-NFs. The results indicate that CoPt3@NC has a narrow and elongated hysteresis loop, while CoPt3@NC-NFs exhibit significant hysteresis characteristics (Fig. S24). And CoPt3@NC-NFs demonstrated superior ferromagnetic behavior with a coercivity of 300 Oe, far exceeding CoPt3@NC (14 Oe), along with optimal saturation magnetization (Ms). The stronger magnetic properties of CoPt3@NC-NFs would enhance the interaction between the catalyst and paramagnetic O2.
Electrochemical ORR tests
To investigate the impact of the special framework with enhanced magnetic field on the ORR performance, we evaluated the prepared samples and commercial Pt/C for ORR performance using a rotating disk electrode (RDE) in a O2-saturated 0.1 M KOH electrolyte. The cyclic voltammetry (CV) diagram of the catalysts shows that CoPt3@NC-NFs has the optimal ORR peak potential of 0.92 V (vs. RHE, all the following potentials are relative to RHE), suggesting that CoPt3@NC-NFs has a stronger ORR activity (Fig. S25). Fig. 3a presents the linear sweep voltammetry (LSV) curves of CoPt3@NC-NFs, CoPt3@NC, CoPt3@NC-H, and Pt/C catalysts. At 1600 rpm, The CoPt3@NC-NFs exhibit the best ORR activity with the highest Eonset of 1.02 V and E1/2 of 0.89 V, accompanied by the optimal j of 5.8 mA cm−2. Moreover, CoPt3@NC-H exhibits better performance than CoPt3@NC and is comparable to Pt/C. These findings indicate that the magnetic-field enhancement induced by the unique framework structure plays a crucial role in significantly boosting ORR activity. Considering the electronic selectivity of the ORR reaction, we evaluated the LSV curves of the catalysts at different rotation speeds, revealing an increasing limiting current density with the expansion of the rotation speed (Fig. S26). The Koutecky–Levich plot of CoPt3@NC-NFs exhibits an approximately linear relationship between ω−1/2 and j−1, as shown in Fig. S27. At potentials of 0.3, 0.4, 0.5, and 0.6 V, the average electron transfer number n of CoPt3@NC-NFs is approximately 3.96, indicating a four-electron transfer mechanism. Furthermore, the production of hydrogen peroxide during the ORR process was further studied through RRDE measurements (Fig. 3b). As shown in Fig. 3c, within the potential range of 0.2–0.8 V, the average electron transfer number of CoPt3@NC-NFs is the highest at approximately 3.9, while that of CoPt3@NC is 3.8. Meanwhile, its peroxide yield is approximately 4.8%, much lower than CoPt3@NC (10%). These findings clearly demonstrate that CoPt3@NC-NFs exhibit the best 4e− pathway catalytic selectivity. Further electrochemical durability tests on the CoPt3@NC-NFs demonstrated that there was almost no significant current decay after 12 h, indicating that the material possesses outstanding catalytic activity (Fig. S28).Based on the excellent ORR activity of CoPt3@NC-NFs catalyst, we assembled ZABs with CoPt3@NC-NFs as the cathode catalyst and zinc (Zn) foil as the anode. The ZABs were subjected to electrochemical activity tests at room temperature. The open circuit voltage (OCV) of CoPt3@NC-NFs-based ZABs were very stable at 1.50 V, higher than Pt/C (1.41 V) and close to the theoretical value (Fig. S29). The charge–discharge curves of ZAB are shown in Fig. S30. Compared to Pt/C, NFs-based ZAB exhibited higher discharge current and smaller charge–discharge voltage difference, indicating its excellent rechargeability. As shown in Fig. 3d, CoPt3@NC-NFs-based ZABs achieved a power density of 184.8 mW cm−2 at 305 mA cm−2, which was much higher than Pt/C-based ZABs (150.6 mW cm−2 at 248.2 mA cm−2). Moreover, in contrast to three-electrode measurements conducted at high rotation rates-where mass transport is predominantly governed by forced convection—the performance enhancement becomes more pronounced at higher current densities. This behavior can be attributed to the generation of stronger current-induced magnetic fields, which more effectively promote ORR under such conditions. Gradual discharge curves (Fig. S31) showed that the discharge plateau of CoPt3@NC-NFs-based ZABs were higher than that of Pt/C at different current densities. Even at a high current density of 30 mA cm−2, the discharge voltage of CoPt3@NC-NFs-based ZABs remained above 1.1 V. When the current density was restored to 1 mA cm−2, the discharge voltage hardly changed compared to the initial current density. Fig. S32 shows the constant current discharge curve obtained at a current density of 10 mA cm−2, where the discharge voltage of CoPt3@NC-NFs-based ZABs maintained at around 1.2 V until the Zn foil was completely corroded and penetrated by a concentrated alkaline solution with a concentration of 6 M. Moreover, the calculated specific capacity reached an impressive 815.2 mA h g−1, while the corresponding Pt/C-based ZABs was only 678.6 mA h g−1. Furthermore, the charge–discharge cycling performance of CoPt3@NC-NFs-based ZABs was evaluated (Fig. 3e). After 400 continuous cycles, the voltage gap showed no significant increase. As shown in the Fig. 3e, the initial round-trip efficiency was 56.0%, which eventually increased to 56.2%. In contrast, Pt/C exhibited an initial round-trip efficiency of only 54.2%, which further decreased to 51.3% before becoming deactivated after 300 cycles. These results clearly demonstrate the superior charging performance and cycling stability of CoPt3@NC-NFs-based ZABs. The above results indicate that the catalyst possesses significant potential for practical applications.
Mechanism analysis of ORR activity promoted by magnetic field enhancement caused by framework structure
To further elucidate the reaction mechanisms of CoPt3@NC-NFs and CoPt3@NC, we employed in situ Raman spectroscopy to monitor the evolution of active sites and ORR intermediates throughout the entire ORR process (Fig. 3f and g). Experiments were conducted using custom-designed cells. Both CoPt3@NC-NFs and CoPt3@NC initially exhibited vibration peaks at 1068 cm−1, attributed to the *O2− adsorption on metal sites.33,34 As the potential was further reduced, subsequent C sites likely participated in small amounts in the ORR reaction, evidenced by the appearance of peaks at 1153 and 1532 cm−1, corresponding to *O2− and *OOH adsorption on C sites.35 This observation indicates that the formation of *OOH is the PDS for both materials, consistent with DFT calculation results. Additionally, it is noteworthy that the *O2− peak on metal sites for CoPt3@NC-NFs appeared at 0.9 V, while for CoPt3@NC it appeared at 0.7 V. Similarly, for CoPt3@NC-NFs, the *O2− and *OOH peaks on C sites first appeared at 0.6 V, whereas for CoPt3@NC, the *O2− and *OOH peaks started to appear at 0.4 V. These results indicate that CoPt3@NC-NFs exhibit accelerated O2 adsorption and promote the formation of *OOH intermediates. This enhancement is attributed to the enhanced magnetic field induced by the framework structure, which facilitates rapid and directional transport of O2 toward the catalysts’ surface.To further validate the role of magnetic field enhancement within the nano-framework structure in promoting the active and directional transport of O2 to the catalysts’ surface, finite element simulations were conducted to investigate the influence of the magnetic field on O2 distribution. As shown in Fig. S33–S35, O2 molecules were positioned around the cubic, framework and hollow models. Under a certain current, it was observed that in the framework model, where the surface magnetic field is stronger, the diffusion rate of O2 molecules toward the material surface increased significantly compared to the cubic model (Fig. S36–S38). Within the same time, there are more O2 deposition on the surface of the framework model than that of the cubic and hollow model (Fig. S39–S41). For example, after 1 hour, the O2 deposition on the framework surface reaches 9.3 × 10−7 mg cm−2, whereas the corresponding value for the cubic model is significantly lower at 8.4 × 10−7 mg cm−2 and hollow model is 8.8 × 10 −7 mg cm−2.(Fig. 4a–c). In addition, it can be seen that the high deposition area on the frame surface occupies a larger area. This result confirms that the framework structure facilitates the easier directional diffusion of O2 to the materials’ surface. To further clarify this point, we examined O2 adsorption and deposition on catalyst surfaces under conditions without current flow (without magnetic field effects). The results show that the hollow structure exhibits the strongest O2 adsorption, while the framework structure is intermediate and the cubic structure is the weakest (Fig. S42–S44). This confirms that in the non-magnetic case, the hollow structure is intrinsically more favorable for oxygen uptake. By contrast, under operating conditions with current flow, the framework structure displays markedly accelerated O2 adsorption, which can be attributed to the unique magnetic fields generated within its architecture. In this context, the Kelvin force serves as an additional driving bias, effectively enhancing O2 transport and promoting preferential accumulation at the framework catalyst surface.
Scanning electrochemical microscopy (SECM) was employed to visualize the localized catalytic processes of the catalysts at the micro/nanoscale. Fig. S45 schematically illustrates the working principle of the ultramicroelectrode (UME) and the catalyst electrode in an electrolyte containing a redox mediator. Initially, in a solution of 5 mM K3[Fe(CN)6] with 0.1 M KCl as the supporting electrolyte, the electrochemical reaction proceeds according to equation: [Fe(CN)6]3− + e− →[Fe(CN)6]4−. The UME records the steady-state Pt tip current (iT) generated by the substrate–tip redox processes occurring at a defined working distance. At the open-circuit voltage (OCV) of the substrate, the presence of oxidizing species in the solution facilitates the spontaneous oxidation of [Fe(CN)6]4− to [Fe(CN)6]3− in the electrolyte, which subsequently diffuses to the tip electrode due to the concentration gradient. At the tip, electrons are transferred to [Fe(CN)6]3−, reducing it back to [Fe(CN)6]4−. This [Fe(CN)6]4−/[Fe(CN)6]3− redox cycling significantly enhances the tip current, making it suitable for various SECM generation–collection and feedback modes. The tip voltammogram (Fig. S46) exhibits a characteristic sigmoidal (S-shaped) profile, confirming that the Pt tip is appropriate for electrochemical mapping. The CV recorded at the tip also reflects the electrochemical reactivity of the electrode surface. Benefiting from its large specific surface area provided by the framework structure, CoPt3@NC-NFs exhibits superior reactivity. As shown in Fig. S47, in both CoPt3@NC and CoPt3@NC-NFs systems, the Pt tip current gradually increases as it approaches the substrate, indicating that both materials operate in a positive feedback mode. Fig. 4d and e present two-dimensional (2D) SECM images of the CoPt3@NC-NFs and CoPt3@NC working electrodes. Under the typical constant-height mode, the tip was positioned 10 µm above the catalyst electrode and scanned over the x–y plane. The recorded current varied with the lateral position of the tip relative to the substrate electrode surface. Driven by a piezoelectric-controlled 3D positioning system, the tip was moved across the substrate electrode surface to record the tip current, thereby providing the microscopic distribution of the electrode material and enabling imaging of both insulating and conductive regions. The results show that the current distribution of CoPt3@NC-NFs is more uniform, and the current magnitude in the reactive region is higher, indicating a more homogeneous distribution and greater surface reactivity on the substrate electrode.
Furthermore, to further elucidate the role of framework-induced magnetic field enhancement in promoting ORR kinetics, SECM measurements were conducted under constant-height conditions in 0.1 M KOH electrolyte, employing the tip generation/substrate collection (TG/SC) mode to investigate the local O2 transport and electrocatalytic activity of CoPt3@NC-NFs and CoPt3@NC during the ORR process (Fig. 4f). In this mode, a potential of 1.3 V (vs. RHE) is applied to the Pt tip electrode to generate a large flux of O2 in its vicinity, thereby enabling oxygen reduction reactions on the catalyst-loaded substrate at the micro/nanoscale. Subsequently, an appropriate ORR potential is applied to the substrate electrode to probe the catalytic response within the localized reaction zone. Here, the potential for rapid ORR is selected to scan the substrate voltage (0.6 V vs. RHE). The potential at which the *OOH intermediate appears in the CoPt3@NC-NFs measured by the in situ Raman spectroscopy is selected to scan the substrate voltage (0.6 V vs. RHE), and the response current of the catalyst in the micro-region is obtained in real time. As shown in Fig. 4g and h, in the local area, the current value of the CoPt3@NC-NFs substrate is uniformly distributed above 20 nA, while the current value of CoPt3@NC is only around 10 nA. This indicates that CoPt3@NC-NFs can quickly capture O2 generated by the probe electrode and promote the rapid conversion of O2. This is consistent with the above simulation results, confirming that the surface magnetic field enhancement induced by the unique nano-framework structure effectively promotes the rapid and active directional transport of O2 molecules toward the catalysts’ surface. This, in turn, facilitates accelerated O2 adsorption and *OOH formation, thereby boosting ORR activity.
Conclusions
In summary, we successfully prepared a nano-framework-structured CoPt3 nanoparticle catalyst (CoPt3@NC-NFs) by utilizing the unique ZIF-L framework as the precursor. Our studies reveal that this distinctive framework structure generates a significantly stronger surface magnetic field during electrochemical processes compared to cubic structures, thereby enhancing the Kelvin force's effect on paramagnetic substances. This enhancement enables the active and directional enrichment of O2 molecules toward the catalysts’ surface, thereby increasing O2 adsorption capacity and promoting the formation of *OOH (the PDS). The catalyst exhibits excellent catalytic activity for the ORR, with an onset potential of 1.02 V, a half-wave potential of 0.89 V, and a limiting current density of −5.8 mA cm−2 in 0.1 M KOH, outperforming conventional cubic structure catalysts (CoPt3@NC) and Pt/C. In assembled alkaline ZABs using CoPt3@NC-NFs as the cathode, the OCV reaches 1.50 V, with a peak power density of 184.8 mW cm−2 and good durability. These findings provide important strategies for designing efficient ORR catalysts, revealing the enhancement mechanism of magnetic fields in the ORR, and offering constructive insights for further exploring the magnetic effects of materials in the ORR process.Author contributions
Zhijie Qi: investigation; data curation; formal analysis; writing – original draft. Zhenjie Lu: investigation. Pengcheng Yao: formal analysis; writing – review & editing. Wuxin Ba: investigation. Lianjin Wei: validation. Duansheng Liu: investigation. Jun Jiang: data curation. Shujun Liu: investigation. Pawel J. Kulesza: formal analysis. Jingwen Sun: validation. Pan Xiong: validation. Xin Wang: resources; supervision. Junwu Zhu: supervision; project administration; funding acquisition; writing – review & editing. Yongsheng Fu: conceptualization; methodology; project administration; supervision; funding acquisition; writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
All data needed to support the conclusions of this manuscript are included in the main text or the supplementary information (SI). The supplementary information contains additional experimental details, characterization data (SEM, TEM, XRD, XPS, etc.), and supporting electrochemical, COMSOL and DFT results. See DOI: https://doi.org/10.1039/d5ee06929g.Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant No. 52125202, 52372083, 52173255), the financial support from the National Key R&D Program of China (Grant No. 2024YFB3815301), the Opening Project of the Jiangsu Key Laboratory for Chemistry of Low-Dimensional Materials (No. JSKC24025), Special Funds for the Trans-formation of Scientific and Technological Achievements in Jiangsu Province (Grant No. BA2023003), the Collaborative Innovation Center for Advanced Micro/nanomaterials and Equipment (Co-constructed by Jiangsu Province and Ministry of Education).Notes and references
- M. Luo, Z. Zhao, Y. Zhang, Y. Sun, Y. Xing and F. Lv, et al., PdMo bimetallene for oxygen reduction catalysis, Nature, 2019, 574(7776), 81–85 CrossRef CAS PubMed.
- W. Zhou, B. Li, X. Liu, J. Jiang, S. Bo and C. Yang, et al., In situ tuning of platinum 5d valence states for four-electron oxygen reduction, Nat. Commun., 2024, 15(1), 6650 CrossRef CAS PubMed.
- W. Li, B. Liu, D. Liu, P. Guo, J. Liu and R. Wang, et al., Alloying Co Species into Ordered and Interconnected Macroporous Carbon Polyhedra for Efficient Oxygen Reduction Reaction in Rechargeable Zinc–Air Batteries, Adv. Mater., 2022, 34(17), 2109605 CrossRef CAS PubMed.
- G. W. Sievers, A. W. Jensen, J. Quinson, A. Zana, F. Bizzotto and M. Oezaslan, et al., Self-supported Pt–CoO networks combining high specific activity with high surface area for oxygen reduction, Nat. Mater., 2021, 20(2), 208–213 CrossRef CAS PubMed.
- W. Zhou, H. Su, W. Cheng, Y. Li, J. Jiang and M. Liu, et al., Regulating the scaling relationship for high catalytic kinetics and selectivity of the oxygen reduction reaction, Nat. Commun., 2022, 13(1), 6414 CrossRef CAS.
- M. Liu, Z. Zhao, X. Duan and Y. Huang, Nanoscale Structure Design for High-Performance Pt-Based ORR Catalysts, Adv. Mater., 2019, 31(6), 1802234 CrossRef PubMed.
- Y.-J. Wang, W. Long, L. Wang, R. Yuan, A. Ignaszak and B. Fang, et al., Unlocking the door to highly active ORR catalysts for PEMFC applications: polyhedron-engineered Pt-based nanocrystals, Energy Environ. Sci., 2018, 11(2), 258–275 RSC.
- S. Wang, E. Zhu, Y. Huang and H. Heinz, Direct correlation of oxygen adsorption on platinum-electrolyte interfaces with the activity in the oxygen reduction reaction, Sci. Adv., 2021, 7(24), eabb1435 CrossRef CAS PubMed.
- Z. Qi, Y. Zhou, R. Guan, Y. Fu and J.-B. Baek, Tuning the Coordination Environment of Carbon-Based Single-Atom Catalysts via Doping with Multiple Heteroatoms and Their Applications in Electrocatalysis, Adv. Mater., 2023, 35(38), 2210575 CrossRef CAS PubMed.
- Y. Hu, T. Shen, X. Zhao, J. Zhang, Y. Lu and J. Shen, et al., Combining structurally ordered intermetallics with N-doped carbon confinement for efficient and anti-poisoning electrocatalysis, Appl. Catal., B, 2020, 279, 119370 CrossRef CAS.
- M. Park, C. Liang, T. H. Lee, D. A. Agyeman, J. Yang and V. W.-H. Lau, et al., Regulating the Catalytic Dynamics Through a Crystal Structure Modulation of Bimetallic Catalyst, Adv. Energy Mater., 2020, 10(8), 1903225 CrossRef CAS.
- M. Xie, Z. Lyu, R. Chen, M. Shen, Z. Cao and Y. Xia, Pt–Co@Pt Octahedral Nanocrystals: Enhancing Their Activity and Durability toward Oxygen Reduction with an Intermetallic Core and an Ultrathin Shell, J. Am. Chem. Soc., 2021, 143(22), 8509–8518 CrossRef CAS PubMed.
- J. Yang, D. Kang, Y. Jeon, J. H. Lee, H. Y. Jeong and H. S. Shin, et al., Sphere-to-Multipod Transmorphic Change of Nanoconfined Pt Electrocatalyst during Oxygen Reduction Reaction, Small, 2019, 15(2), 1802228 CrossRef PubMed.
- J. Wordsworth, T. M. Benedetti, S. V. Somerville, W. Schuhmann, R. D. Tilley and J. J. Gooding, The Influence of Nanoconfinement on Electrocatalysis, Angew. Chem., Int. Ed., 2022, 61(28), e202200755 CrossRef CAS PubMed.
- Z. Li, Y. Yao, Y. Niu, W. Zhang, B. Chen and X. Zeng, et al., Multi-heteroatom-doped hollow carbon tubes as robust electrocatalysts for the oxygen reduction reaction, oxygen and hydrogen evolution reaction, Chem. Eng. J., 2021, 418, 129321 CrossRef CAS.
- K. Shi, K. Wang, T. Li, J. Li, J. Ren and X. Li, et al., Apically guiding electron/mass transfer reaction induced by Ag/FeNx Mott-Schottky effect within a hollow star reactor toward high performance zinc-air batteries, J. Energy Chem., 2024, 95, 106–116 CrossRef CAS.
- M. Guo, M. Xu, Y. Qu, C. Hu, P. Yan and T. T. Isimjan, et al., Electronic/mass transport increased hollow porous Cu3P/MoP nanospheres with strong electronic interaction for promoting oxygen reduction in Zn-air batteries, Appl. Catal., B, 2021, 297, 120415 CrossRef CAS.
- Y. Zhou, Y. Liang, Z. Zhao, X. Wang, R. Guan and C. Li, et al., Hollow structures derived from metal-glycerates toward efficient electrochemical energy storage and conversion, Nano Energy, 2025, 144, 111348 CrossRef CAS.
- Y. Sun, S. Sun, H. Yang, S. Xi, J. Gracia and Z. J. Xu, Spin-Related Electron Transfer and Orbital Interactions in Oxygen Electrocatalysis, Adv. Mater., 2020, 32(39), 2003297 CrossRef CAS PubMed.
- Y. Yu, J. Shi, M. Li, L. Li, J. Hu and S. Li, et al., Local Magnetic Effect-Induced Electron Configuration Regulation: Spin Flipping of Iron Centers for Molecular Catalysis, ACS Catal., 2024, 14(9), 7191–7200 Search PubMed.
- P. Vensaus, Y. Liang, J.-P. Ansermet, J. Fransson and M. Lingenfelder, Spin-Polarized Electron Transport Promotes the Oxygen Reduction Reaction, ACS Nano, 2025, 19(44), 38709–38715 CrossRef PubMed.
- Y. Yang, G. Han, M. Xie, G. V. D. O. Silva, G.-X. Miao and Y. Huang, et al., Magnetic Field Enhanced Oxygen Reduction Reaction via Oxygen Diffusion Speedup, Small Methods, 2024, 8(10), 2301594 CrossRef CAS PubMed.
- S. Luo, K. Elouarzaki and Z. J. Xu, Electrochemistry in Magnetic Fields, Angew. Chem., Int. Ed., 2022, 61(27), e202203564 CrossRef CAS PubMed.
- J.-B. Chen, J. Ying, Y. Tian, Y.-X. Xiao and X.-Y. Yang, Electrocatalysis under Magnetic Fields, Adv. Funct. Mater., 2024, 2415660 Search PubMed.
- L. B. Wang, N. I. Wakayama and T. Okada, Numerical simulation of a new water management for PEM fuel cell using magnet particles deposited in the cathode side catalyst layer, Electrochem. Commun., 2002, 4(7), 584–588 CrossRef CAS.
- L. T. Tufa, K.-J. Jeong, V. T. Tran and J. Lee, Magnetic-Field-Induced Electrochemical Performance of a Porous Magnetoplasmonic Ag@Fe3O4 Nanoassembly, ACS Appl. Mater. Interfaces, 2020, 12(5), 6598–6606 CrossRef CAS.
- S.-L. Tian, L.-N. Song, L.-M. Chang, W.-Q. Liu, H.-F. Wang and J.-J. Xu, A magnetic/force coupling assisted lithium-oxygen battery based on magnetostriction and piezoelectric catalysis of CoFe2O4/BiFeO3 cathode, Nano Energy, 2024, 126, 109677 CrossRef CAS.
- P. Vensaus, Y. Liang, J.-P. Ansermet, G. Soler-Illia and M. Lingenfelder, Enhancement of electrocatalysis through magnetic field effects on mass transport, Nat. Commun., 2024, 15(1), 2867 CrossRef CAS PubMed.
- W. Bai, J. Chen, X. Wang, J. Zhu and Y. Fu, Transformation of ZIF-67 Nanocubes to ZIF-L Nanoframes, J. Am. Chem. Soc., 2024, 146(1), 79–83 CrossRef CAS PubMed.
- Z. Wang, S. Chen, W. Wu, R. Chen, Y. Zhu and H. Jiang, et al., Tailored Lattice Compressive Strain of Pt-Skins by the L12-Pt3M Intermetallic Core for Highly Efficient Oxygen Reduction, Adv. Mater., 2023, 35(36), 2301310 CrossRef CAS PubMed.
- J. Zhang, P. Ming, B. Li and C. Zhang, Ultralow platinum catalysts for high performance fuel cells: in situ encapsulation of platinum atoms and CoPt3 in 3D hollow nanoshells, J. Mater. Chem. A, 2023, 11(38), 20488–20496 RSC.
- Z. Lu, J. Xie, J. Hu, K. Wang and Y. Cao, In Situ Replacement Synthesis of Co@NCNT Encapsulated CoPt3@Co2P Heterojunction Boosting Methanol Oxidation and Hydrogen Evolution, Small, 2021, 17(52), 2104656 CrossRef CAS PubMed.
- J. Wei, D. Xia, Y. Wei, X. Zhu, J. Li and L. Gan, Probing the Oxygen Reduction Reaction Intermediates and Dynamic Active Site Structures of Molecular and Pyrolyzed Fe–N–C Electrocatalysts by In Situ Raman Spectroscopy, ACS Catal., 2022, 12(13), 7811–7820 CrossRef CAS.
- S. Ji, Y. Mou, H. Liu, X. Lu, Y. Zhang and C. Guo, et al., Manipulating the Electronic Properties of an Fe Single Atom Catalyst via Secondary Coordination Sphere Engineering to Provide Enhanced Oxygen Electrocatalytic Activity in Zinc-Air Batteries, Adv. Mater., 2024, 36(44), 2410121 CrossRef CAS PubMed.
- L. Zhao, Y. Dai, Y. Zhang, B. Liu, P. Guo and Z. Zhang, et al., Atomically Dispersed p-Block Aluminum-Based Catalysts for Oxygen Reduction Reaction, Angew. Chem., Int. Ed., 2024, 63(20), e202402657 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |