Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Dynamic reconstruction defines true active states in the hydrogen evolution reaction
Xingyu
Ding
 ab and
Xianbiao
Fu
ab and
Xianbiao
Fu
 *ac
*ac
aDepartment of Materials Science and Engineering, National University of Singapore, Singapore 117576, Singapore. E-mail: xbfu@nus.edu.sg
bCollege of Smart Materials and Future Energy, State Key Laboratory of Coatings for Advanced Equipment, Fudan University, Shanghai 200438, China
cCentre for Hydrogen Innovations, National University of Singapore, Singapore 117580, Singapore. E-mail: xbfu@nus.edu.sg
First published on 4th February 2026
Abstract
The hydrogen evolution reaction (HER) plays a pivotal role in sustainable hydrogen production and the transition to a carbon-neutral energy future. Traditionally, HER catalyst design has focused on optimizing as-synthesized structures such as composition, morphology, and electronic states, under the assumption that these features remain static during operation. However, accumulating evidence reveals that HER catalysts undergo profound reconstruction, including phase transformation, compositional change, and atomic rearrangement, which fundamentally redefine the true active states. Neglecting this dynamic evolution risks misidentifying catalytic sites, misinterpreting mechanisms, and misguiding design strategies. In this Perspective, we advocate a reconstruction-centered framework for the HER. We outline key reconstruction modes, argue that reconstruction is thermodynamically driven and shaped by intrinsic and extrinsic factors, and emphasize that catalysts should be designed as precursors engineered to evolve in situ into their most active and durable forms. Finally, we advocate for stability assessments that capture steady-state reconstructed phases instead of transient initial states. Adopting this dynamic viewpoint establishes a coherent foundation for mechanistic understanding and rational catalyst design, paving the way toward predictive control of catalytic activity and long-term durability.
Broader contextThe hydrogen evolution reaction (HER) is a cornerstone of sustainable hydrogen production and the global transition toward a carbon-neutral energy system. Conventional catalyst design has long focused on static structural features such as composition, morphology, and electronic states, assuming that these properties remain unchanged during operation. However, a growing body of evidence reveals that HER catalysts undergo extensive dynamic reconstruction under electrochemical conditions, giving rise to the true active states that govern catalytic performance. Recognizing reconstruction as an intrinsic and thermodynamically driven process prevents the misidentification of active sites, misinterpretation of mechanisms, and misdirection of design strategies, thereby establishing a more coherent and predictive foundation for understanding the hydrogen evolution reaction.Understanding and controlling reconstruction is therefore essential not only for mechanistic insight but also for ensuring the long-term reliability of catalysts in industrial-scale electrolyzers operating at high current densities. Incorporating reconstruction into catalyst design carries profound implications across multiple scales. At the mechanistic level, it challenges the static view of the HER and calls for frameworks that describe dynamically evolving active states under realistic conditions. At the materials design level, catalysts should be conceived as precursors engineered to evolve into their most active and durable forms, integrating reconstruction principles into synthesis and activation strategies. At the technological level, coupling operando diagnostics with controlled synthesis and electrochemical protocols enables the deliberate tuning of reconstruction pathways, promoting activity, stability, and scalability. Looking ahead, emerging concepts such as programmable reconstruction, using time-dependent potentials guided by artificial intelligence (AI)-driven feedback and physics-informed modeling, promise to transform reconstruction from an uncontrolled response into a design variable. Embracing this dynamic perspective provides a unifying framework linking fundamental mechanisms to practical hydrogen technologies, paving the way toward intelligent catalyst systems for the next generation of sustainable energy conversion. |
1. Introduction
The hydrogen evolution reaction (HER), as the cathodic half of water electrolysis, lies at the heart of renewable hydrogen production.1 For decades, researchers have pursued better catalysts by tuning the composition, morphology, crystallinity, and electronic states of as-synthesized catalysts.2–5 These studies implicitly assumed that such static properties persist during operation, and activity–structure relationships have therefore been built on the premise of structural immutability. But are we truly studying the catalysts we design? When the electrochemical environment persistently reshapes catalyst surfaces, can mechanistic models based on static structures remain valid? If catalytic activity and durability ultimately emerge from the reconstructed states, does optimizing pre-catalysts alone still hold meaning? These questions expose the growing inadequacy of the traditional static paradigm.Operando/in situ and post-reaction characterizations consistently reveal extensive structural evolution in HER catalysts, ranging from phase transitions to compositional change, and atomic rearrangement.6–9 The active state that drives hydrogen production is often fundamentally different from the material first introduced into the electrolyte. This recognition compels a fundamental shift in perspective. Sole reliance on pre-operando characterizations risks misidentifying the true active sites; mechanistic models grounded in static snapshots may mislead; and optimizing pre-catalyst structures alone may have limited impact when their evolution is dominated by reconstruction. We therefore call for HER research to explicitly acknowledge reconstruction and to embrace a new paradigm centered on the interplay between dynamic structures and catalytic performance.
In this perspective, we argue that reconstruction is an intrinsic thermodynamically driven phenomenon, not an accidental complication. We classify principal reconstruction modes and analyze their determining factors, then propose a reconstruction-driven framework as a new paradigm for HER catalyst research, linking pre-catalyst structure to reconstruction pathways, and reconstructed active structure to HER performance. This integrated understanding enables rational design of pre-catalysts that evolve into optimal active phases under operating conditions. We also emphasize that only long-term testing captures the steady-state reconstructed structures governing catalytic durability. Through this dynamic perspective, reconstruction transforms from a mechanistic complication into a powerful design principle, unveiling new strategies for advanced HER catalyst development.
2. Can we continue to treat reconstruction as peripheral rather than central in HER catalysis?
For decades, mechanistic studies of the HER have rested on a fundamental assumption: synthesized catalysts maintain their structures during the reaction process. This view persisted largely because conventional techniques struggled to resolve the subtle, transient surface changes where the reaction actually proceeds. Within this static paradigm, catalytic activity is directly attributed to pre-catalyst structures.10 As illustrated in Fig. 1a, HER processes, from reactant adsorption and intermediate formation to hydrogen evolution, are viewed as occurring on structurally invariant catalyst surfaces. Consequently, researchers have developed various descriptors for HER catalyst design, including hydrogen adsorption free energy (ΔGH), d-band center position,11–16 work function (Φ), hydrogen binding energy (HBE),17,18 and hydroxyl binding energy (OHBE),19,20 all of which presume structural stability during operation. However, this understanding is increasingly challenged by experimental evidence. Operando/in situ and post-reaction characterizations reveal that HER catalysts rarely maintain their pristine states under electrochemical conditions.6–9 As shown in Fig. 1b, HER is not merely an HER process but involves continuous catalyst surface reconstruction. This means the true active species under operating conditions often differs from the initial catalyst structure.These reconstruction phenomena can be broadly categorized into three types, each representing different levels of structural evolution. First, phase transformation (Fig. 1c) is defined as a reconstruction process involving changes in crystallographic structure under HER conditions. For example, MoS2 exemplifies the semiconducting 2H to the metallic 1T conversion under cathodic bias,21 while transition metal chalcogenides often evolve from chalcogen-rich to chalcogen-poor phases, as in the cubic NiSe2 to hexagonal NiSe transformation.22 More broadly, oxides, sulfides, phosphides, carbides, and borides typically reduce to metallic or hydride states that function as the true active phases.23–35 Earlier studies generally attributed HER activity to metallic species generated under cathodic potentials, exemplified by the transformation of Ni sulfides into Ni metal.23,24 More recent work has revealed that cathodic bias can also induce the formation of metal hydroxides, with systems such as CoF2 and CoP converting into Co(OH)2 as the operative HER phase.28–32 Second, Compositional change, as illustrated in Fig. 1d, is defined as a reconstruction process involving modification of elemental species and/or ratios while preserving the parent crystal structure. This manifests as a gradual evolution of chemical composition, typically driven by selective dissolution or ion exchange. In multi-metallic systems, electrochemically less stable elements preferentially dissolve, creating thermodynamically more stable surface compositions. A representative example is the Mo element undergoing dynamic dissolution and redeposition on the catalyst surface during HER.36–39 For example, Du et al. reported that NiMo alloys undergo an in situ transformation during the HER process, where Mo dissolves as MoO42−, re-adsorbs and polymerizes into Mo2O72−, and ultimately redeposits to enhance catalytic activity.36 Similarly, Mo-doped NiP exhibits Mo dissolution and subsequent redeposition, forming new MoO3 species on the Mo–NiP surface under HER conditions.39 Third, atomic rearrangement, shown in Fig. 1e, is defined as a reconstruction process involving spatial redistribution of atoms without inducing compositional change. This occurs at highly localized scales, typically resulting from surface or subsurface atom migration under electric fields. Such rearrangement leads to crystalline facet reorganization, which in turn generates surface defects, step edges, elemental segregation, and in extreme cases, surface amorphization. A more extreme case is single-atom catalysts, which are inherently unstable under HER conditions and tend to migrate and aggregate into clusters with lower surface energy. For example, Pt and Ru single atoms have been shown to undergo such atom migration, evolving into cluster states during HER.40,41 These three reconstruction types are ubiquitous under HER conditions and often occur synergistically. For instance, Ding et al. revealed that NiX (X = S or Se) catalysts first undergo a phase transformation into Ni3X2 under cathodic polarization. Subsequently, continuous leaching of surface anions (S or Se) and their replacement by oxygen leads to the formation of NiO, ultimately yielding a Ni3X2/NiO heterostructure catalyst.42,43 Notably, the three types of reconstruction discussed above may occur either in the bulk or at the surface. In practice, surface phase transformation and surface compositional change often represent the earliest manifestations of reconstruction under electrochemical conditions, yet they are typically manifested only as amorphous or atomic-scale layers at the surface and therefore remain difficult to detect or unambiguously characterize. By contrast, phase transformation or compositional change occurring in the bulk is generally more readily detectable by conventional structural or spectroscopic techniques, such as X-ray diffraction (XRD) and X-ray absorption spectroscopy (XAS). Even after such bulk reconstruction has taken place, the resulting observations remain inherently bulk-averaged and do not necessarily reflect the true surface state. This underscores the importance of incorporating surface-sensitive characterization techniques, including X-ray photoelectron spectroscopy (XPS), Raman spectroscopy, and time-of-flight secondary ion mass spectrometry (TOF-SIMS), to elucidate dynamic surface reconstruction under electrochemical conditions.
Nevertheless, despite being the equally essential half-reaction to the oxygen evolution reaction (OER) in water splitting, and although reconstruction has been reported in many HER systems, systematic, predictive, and closed-loop control of reconstruction is still an open challenge and opportunity. We call for increased efforts to systematically unveil HER catalyst reconstruction: in the short term, identifying and cataloging reconstruction types and manifestations across different systems; in the long term, distilling universally applicable reconstruction mechanisms from extensive studies, establishing reliable dynamic structure–performance relationships, and ultimately developing new paradigms for HER catalyst design that harness reconstruction processes.
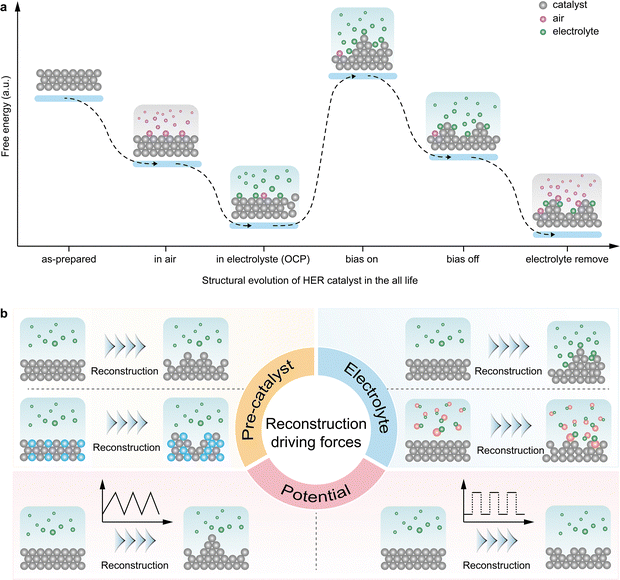
3. HER catalyst reconstruction: accident of operation or inevitable thermodynamic pathway?
The ubiquity of reconstruction in HER catalysts raises a fundamental question: Is reconstruction merely an operational perturbation or a thermodynamically inevitable evolution? From an energetic perspective, catalyst surface reconstruction represents not random events but the system's spontaneous pursuit of lower free energy states under specific conditions. As illustrated in Fig. 2a, we propose a lifecycle model encompassing catalyst reconstruction from synthesis through HER operation to post-reaction states. Freshly as-prepared catalysts exposed to ambient atmosphere inevitably undergo mild reconstruction through oxidation or species adsorption from air, reducing surface energy. Upon immersion in electrolyte at open circuit potential (OCP), catalyst surfaces interact with the solution environment through species adsorption or chemical corrosion, primarily from H+ or OH− interactions in HER, driving further reconstruction and surface energy minimization. For example, Ledendecker et al. show that non-noble materials such as MoS2, Ni5P4, WC, and Co2P undergo significant metal dissolution at open circuit, demonstrating their intrinsic instability under OCP.44 When cathodic bias initiates HER, the system's electrochemical potential shifts dramatically. Surface atoms become activated and reorganize through intense electrolyte interactions, forming higher energy excited states that subsequently relax into metastable structures sustainable under reaction conditions. Critically, the true catalytically active sites exist within these metastable configurations. Upon bias removal, the system's free energy changes again, destabilizing these metastable states and driving structural relaxation toward lower surface energy configurations compatible with the new environment. Finally, when catalysts are removed from the electrolyte, rinsed, and dried, these structures undergo further reconstruction through air exposure, continuing their evolution toward thermodynamic minima.This lifecycle model reveals that reconstruction pervades every stage from catalyst synthesis through HER operation, naturally raising critical questions: What factors govern surface reconstruction behavior and thus active site formation? Can we identify universal principles to deliberately control reconstruction for optimal electrocatalytic performance? Understanding this model proves essential for mastering surface reconstruction and advancing HER catalysis. Three interconnected factors emerge as reconstruction drivers: pre-catalyst structure, electrolyte environment, and applied potential. Pre-catalyst structure manifests through intrinsic atomic properties that dictate responses to electrochemical stimuli (Fig. 2b). Even identical elements exhibit divergent reconstruction behaviors depending on initial atomic arrangements; crystalline facets, defect densities, and coordination environments all influence transformation pathways. For example, while cubic CoSe2 remains stable under HER operating conditions, P-doped CoSe2 can be induced to generate metallic Co0 as the catalytically active species.45 Electrolyte environment creates distinct reconstruction trajectories even for identical precursors under the same potential (Fig. 2b). This arises from surface-specific adsorption during HER, which modulates atomic properties and triggers divergent restructuring. Leveraging this principle, surface reconstruction can be indirectly controlled by tuning the pH, introducing additives with tailored adsorption affinities, or employing electrochemically inert species.46–49 For example, Sun et al. examined the pH-dependent reconstruction of Co0.5W0.5Sx. Under neutral or alkaline conditions, the bulk converted to CoO and the surface to CoO/Co(OH)2, whereas in acid, only the surface was reconstructed.46 Other studies report that adding extra MoO42− to Mo-based HER catalysts, such as MoSe2 or Mo2Co3O8, facilitates surface reconstruction into active Mo2O72− species.47,48 Beyond deliberate manipulation of bulk electrolyte composition, the electrolyte environment itself exhibits pronounced dynamic evolution during the HER, particularly within the localized interfacial region, which may in turn influence catalyst restructuring pathways.50,51 The sustained consumption of protons during HER inevitably establishes local pH gradients.52,53 Such dynamically evolving interfacial pH directly modulates surface protonation states, adsorption energetics, and phase stability, thereby providing thermodynamic driving forces for catalyst restructuring. Consistent with this perspective, Bao et al. demonstrated that the locally acidic interfacial environment generated under HER conditions can induce the transformation of WO2 into HxWOy, establishing a direct link between pH dynamics and specific restructuring pathways.54 The electrolyte conductivity during HER is likewise not static. HER-induced concentration polarization, particularly the nucleation, growth, and accumulation of hydrogen bubbles, intermittently disrupts ionic conduction pathways, elevates effective ohmic resistance, and redistributes local current density and overpotential, thereby inducing divergent catalyst restructuring trajectories.1,55 Concurrently, sustained H2 evolution exerts physical impingement that drives morphological restructuring or even mechanical disintegration and detachment of catalysts.56 Moreover, the sequential processes of H2 generation, dissolution, supersaturation, bubble formation, and detachment give rise to variations in the interfacial microenvironment, including local pH fluctuations, mass transport limitations, and active site blockage. This cascade of microenvironmental evolution collectively influences catalyst restructuring pathways.55 Although studies directly correlating electrolyte dynamics with catalyst restructuring pathways remain limited, particularly regarding the effects of local conductivity variations and bubble evolution on dynamic catalyst restructuring, these analyses collectively underscore that electrolyte evolution and catalyst restructuring constitute intrinsically coupled and inseparable processes during prolonged HER operation. Applied potential serves as the primary energetic driver, directly programming reconstruction pathways (Fig. 2b). It controls energy input across reaction stages, determining initial excited states and sustainable metastable configurations, ultimately dictating the reconstruction direction. Notably, Gisbert-González et al. revealed that applied bias regulates surface hydrogen coverage, leading to systematic evolution of the catalytic transition state. This finding suggests that electrochemical potential provides a powerful lever to control transition states and, in turn, direct catalyst reconstruction pathways.57 For example, under HER conditions at overpotentials below 500 mV, NiS2 transforms into the active Ni3S2 phase. Feng et al. demonstrated that higher overpotentials produce a Ni/Ni3S2 composite, dramatically boosting HER activity.58
Current efforts have revealed diverse reconstruction behaviors, yet predictive and programmable control over reconstruction remains insufficiently developed. We advocate for more systematic investigations on how initial structures influence reconstruction pathways, how electrolyte environment directs transformation, and how potential protocols control evolution.
4. If reconstruction redefines the active state, can we trust short-term stability tests?
In the preceding sections, we discussed whether HER catalysts reconstruct and the driving forces behind this process, yet the extent of reconstruction has received little attention. As illustrated in Fig. 3a, a central unresolved question is whether reconstruction under HER conditions is confined to the surface, penetrates into the subsurface, or ultimately transforms the entire structure. The depth of reconstruction dictates the spatial distribution and stability of active sites, making it a critical determinant of both mechanism and design. However, systematic studies of reconstruction extent in HER catalysts remain scarce, leaving an important gap.This gap is compounded by the dominance of short-term stability tests, typically lasting only a few to several tens of hours (Fig. 3b). While convenient, such protocols overlook the ongoing structural evolution of catalysts. Early activity often reflects transient states rather than the steady reconstructed configurations that define durability. In fact, the most active or stable phases may only emerge after extended reconstruction, far beyond conventional testing windows. Catalysts that initially appear robust may later deactivate, while others with modest initial activity may evolve into highly durable systems (Fig. 3b).
As shown in Fig. 3b, we argue that HER stability assessment requires a paradigm shift. Long-term testing is indispensable to capture the kinetics of reconstruction and the durability of the reconstructed states. By combining extended operando characterization with electrochemical measurements, researchers can identify when reconstruction reaches completion and evaluate whether the resulting phases sustain activity or drift toward deactivation. Before assessing reconstruction depth, it is essential to establish whether the catalyst has reached a stable reconstructed state. Operando or in situ spectroscopic techniques, such as Raman spectroscopy, XAS, and XRD, enable real-time tracking of structural and chemical-state evolution during operation. The convergence of these spectroscopic signatures under prolonged operating conditions provides a key criterion for identifying reconstruction completion. Complementarily, operando TOF-SIMS can monitor the dynamic evolution of surface and near-surface species, offering additional insight into whether reconstruction has reached a steady state. In parallel, the stabilization of electrochemical parameters, including current density evaluated by linear sweep voltammetry (LSV), cyclic voltammetry (CV), or chronopotentiometry (CP), the Tafel slope derived from polarization analysis, and impedance features probed by electrochemical impedance spectroscopy (EIS), serves as an electrochemical indicator of reconstruction completion.5 Once a steady reconstructed state is established, ex situ characterization can be employed to resolve the spatial depth of reconstruction. Surface-sensitive techniques, such as angle-resolved XPS and depth-profiling methods (e.g., Ar+ sputtering XPS and TOF-SIMS), reveal compositional gradients from the surface to subsurface regions, while cross-sectional transmission electron microscopy (TEM) combined with electron energy loss spectroscopy (EELS) mapping enables direct visualization of reconstructed domains. For crystalline phase evolution, grazing-incidence XRD probes near-surface structural changes, whereas conventional XRD reflects bulk phase behavior, allowing surface and bulk reconstruction to be distinguished.
Such an approach establishes a more rigorous definition of stability, grounded in the intrinsic properties of reconstructed catalysts rather than in transient behavior. Recognizing durability as inseparable from reconstruction, we call for long-term stability testing to be established as a new standard for HER catalysis, enabling the rational development of catalysts capable of delivering sustained performance under realistic operating conditions.
5. From static illusions to dynamic realities: establishing a reconstruction-driven paradigm for HER catalysts
The pervasive and thermodynamically driven nature of reconstruction fundamentally challenges traditional HER research frameworks: so-called “static” catalysts are merely transition states, while true catalytic identity is progressively established through reconstruction under electrochemical conditions. Ignoring this dynamic evolution leads to misidentification of active sites, as true catalytic phases often emerge only during operation; mechanistic model deviations, since kinetic features (Tafel slopes, reaction orders) likely reflect reconstructed rather than pristine states; and design inefficiencies, because optimizing pre-catalysts alone yields limited benefits when reconstruction dominates final identity. The key imperative going forward is to harness structural evolution as a tool for performance enhancement rather than treating it as an uncontrolled variable. As shown in Fig. 4, we propose a research paradigm that systematically correlates pre-catalyst structures, operando-evolved active structures, and HER performance. This approach comprises three integrated steps: (i) elucidating structure–reconstruction relationships by mapping how the pre-catalyst transforms under electrochemical conditions into their active forms, and (ii) establishing structure–performance correlations between reconstructed active phases and HER performance. Building on these two research steps, (iii) pre-catalyst can be rationally designed via composition and structure regulation (e.g., doping, facet engineering, defect engineering, and heterojunction construction), in combination with operational parameters such as electrolyte composition (pH, ionic species) and applied potential to steer reconstruction toward optimal catalytic configurations. This pre-catalyst structure–active structure–HER performance framework, detailed in the following section, provides a comprehensive blueprint for advancing HER catalyst development.We begin with step (i), where the key objective is to resolve the pre-catalyst structure and the active structure formed under defined operational parameters such as electrolyte composition (pH, ionic species) and applied potential, in order to establish their structural relationship. Elucidating these structures necessitates an integrated characterization strategy that combines bulk and surface probes, with priority given to surface-sensitive techniques, since catalytic function is ultimately defined at the surface. A rigorous understanding of the pre-catalyst requires comprehensive ex situ characterization. Surface-sensitive probes such as temperature-programmed desorption (TPD), XPS, SIMS, ion scattering spectroscopy (ISS), low-energy electron diffraction (LEED), and Auger electron spectroscopy (AES) resolve surface composition, while TEM, atomic force microscopy (AFM), and scanning tunneling microscopy (STM) uncover morphological structure. Complementarily, bulk-sensitive techniques, including XRD and XAS, reveal long-range crystallinity and local coordination geometric structure, with XPS and XAS jointly elucidating electronic structure. For the active structure, operando or in situ techniques are essential for tracking its dynamic evolution under HER conditions. In situ TOF-SIMS and differential electrochemical mass spectrometry (DEMS) capture compositional changes; scanning electrochemical microscopy (SECM), electrochemical transmission electron microscopy (EC-TEM), electrochemical scanning tunneling microscopy (EC-STM), and electrochemical atomic force microscopy (EC-AFM) monitor surface restructuring of morphological structure in real time; in situ XRD probes global phase transitions; and in situ XAS simultaneously provides information on local geometric and electronic structure under HER conditions. Integrating these experimental insights with theoretical modeling enables the mapping of explicit structure–reconstruction relationships, bridging pre-catalyst attributes, operational environments, and reconstructed configurations. The step (ii) of this paradigm focuses on the structure–performance correlation of reconstructed active phases. While delineating the transformation pathways of pre-catalysts is essential, the ultimate objective is to quantitatively link these reconstructed states to HER performance metrics, including overpotential, Tafel slope, exchange current density, and long-term stability. Establishing such structure–activity relationships requires, beyond identifying the operando active structure in step (i), direct probing of reaction intermediates, including H2O*, H*, and OH* adsorption. Operando techniques that can capture these intermediates, such as Raman spectroscopy and infrared spectroscopy (IR), are therefore indispensable. In addition, electrochemical probe techniques such as CV, electrochemical stripping analysis (ESA), underpotential deposition (UPD), electrosorption, and EIS can provide complementary insights into surface states and adsorbed intermediates under HER conditions, by revealing changes in active site availability, adsorption behavior, and surface reconstruction.59–64 When combined with theoretical calculations, these approaches reveal the HER pathways on reconstructed active surfaces and enable the rational derivation of structure–performance correlations and mechanistic insights. Together, steps (i) and (ii) establish a unified “pre-catalyst structure–reconstructed active structure–HER performance” framework, turning reconstruction from an uncontrolled variable into a design tool. Building on this foundation, step (iii) elevates the paradigm to a prescriptive strategy: a reverse-engineering strategy identifies optimal reconstructed states, then directs the design of pre-catalyst structures (composition, geometric and electronic structure, etc.), electrolytes (pH, ionic species, etc.), and applied potentials to achieve it. By treating operando reconstruction as a feature rather than a limitation, this paradigm offers a pathway to simultaneously maximize activity and durability.
6. Conclusion and outlook
In summary, this perspective calls for increased attention to the dynamic reconstruction of HER catalysts. Future studies should broaden the range of material systems, expand the catalog of reconstruction types, and deepen understanding of how pre-catalyst structure, electrolyte environment, and applied potential govern reconstruction, enabling deliberate and directional control. Stability evaluations should be extended to ensure catalysts reach their fully reconstructed state, reflecting true durability. Ultimately, the research paradigm for HER catalysis must shift from static catalysts toward dynamic, reconstruction-driven catalysts, fostering a more accurate understanding of intrinsic activity and long-term stability.While the reconstruction-driven paradigm offers a prescriptive framework for HER catalyst design, its experimental validation is often constrained by limited access to specialized operando spectroscopies. Techniques such as in situ Raman, XPS, XAS, and IR typically require substantial capital investment, complex infrastructure, and sustained access to vacuum or synchrotron facilities, which are resources not universally available. This perspective therefore emphasizes complementary, broadly accessible operando electrochemical probes, including CV, ESA, UPD, electrosorption, and EIS, that can resolve surface site evolution, adsorption dynamics, and interfacial kinetic signatures without reliance on large-scale facilities. These techniques require only standard instrumentation available in most electrochemistry laboratories, namely a three-electrode cell and a commercial potentiostat. Operando CV tracks potential-resolved redox peak evolution to fingerprint phase conversion or coordination changes during reconstruction. ESA quantifies reconstruction-induced variations in active site availability by integrating stripping charges from oxidative removal of adsorbed species. UPD, implemented in dilute electrolytes (<5 mM trace metal ions) with slow-scan voltammetry, resolves facet- and site-selective adsorption energetics, constraining phase persistence and interfacial stability. Electrosorption under inert electrolytes and discrete potential holds probes potential-dependent adsorption of non-complexing spectator ions, capturing interfacial state transitions accompanying reconstruction. Among various electrochemical characterization techniques, EIS, as a non-invasive, frequency-resolved, and readily implementable operando technique, has been extensively employed to investigate the dynamic reconstruction of OER catalysts.65–67 This methodology can be equally applicable to HER catalysts. By analyzing the frequency-dependent impedance response, EIS enables discrete extraction of charge transfer kinetics (Rct) in the high-frequency region, interfacial capacitance characteristics (Cdl) in the mid-frequency region, and mass transport limitations coupled with bubble coverage effects in the low-frequency region (Zw).68 The temporal or potential-dependent evolution of these parameters serves as indirect yet highly sensitive fingerprint signatures of catalyst surface reconstruction. Mechanistically, a progressive decrease in Rct typically signifies the formation of more active phases, while significant Cdl variations reflect the evolution of electrochemically active surface area (ECSA), intimately associated with surface roughening, layer exfoliation, or exposure of new active sites.69 In the low-frequency regime, the Zw response exhibits particular sensitivity to bubble dynamics and local mass transfer heterogeneity, enabling discrimination between intrinsically kinetic-limited and diffusion or hydrodynamically dominated regimes.70 Recent HER studies have unambiguously demonstrated the intrinsic correlation between these EIS features and catalytic kinetics.69–74 For instance, operando EIS tracking of H-TaS2 revealed progressive reduction in Rct and increased effective Cdl, evidencing self-optimizing morphological reconstruction through layer thinning and basal plane activation that promotes HER kinetics.69
In addition, while fundamental understanding of HER catalyst reconstruction has advanced considerably, translating these insights into industrial electrolyzers operating at high current densities (>500 mA cm−2) remains challenging.75–77 In practical electrolyzers, catalysts experience hydrodynamic and electrochemical environments fundamentally different from laboratory three-electrode cells. Local flow fields governed by electrolyzer architecture, including serpentine and parallel channels as well as flow by and flow through configurations and channel geometry, directly influence mass transport, local pH gradients, and interfacial electric fields.78 High-current-density operation brings elevated local temperatures, intensified electric fields, and enhanced mass transport, while vigorous bubble nucleation, growth, and detachment impose periodic mechanical stresses and generate transient concentration gradients.79–81 As discussed, applied potential, electrolyte environment, and pre-catalyst structure govern reconstruction pathways; flow field design and bubble dynamics indirectly yet profoundly influence these driving forces by modulating local potential distribution and electrolyte environment (e.g., interfacial pH and ionic concentration). However, direct investigations of the effects of flow field design, bubble dynamics, and electrolyzer architecture on catalyst reconstruction remain scarce. Existing reconstruction studies are largely performed in simplified three-electrode systems, while studies on flow fields and bubble behavior mainly address mass transport and reaction kinetics. Bridging this gap by correlating electrolyzer environment, reconstruction behavior, and catalytic performance will open new opportunities for reconstruction-guided industrial catalyst design. First, engineering flow fields and channel geometry to homogenize local environments (potential, pH, ionic concentration) can minimize reconstruction heterogeneity across electrodes. Second, the design of reconstruction ready catalyst precursors that take advantage of high current density conditions, such as elevated temperatures, intensified fields, and enhanced mass transport, enables their rapid evolution into reconstructed phases that combine high activity with mechanical robustness.82–84 Third, coupling reconstruction understanding with accelerated stress tests mimicking industrial transients (load cycling, start-up/shut-down) enables more predictive durability assessments.85,86 Incorporating hydrodynamic, thermal, and mechanical factors into the reconstruction framework, future research can bridge fundamental understanding and rational design, enabling HER catalysts for sustained high-rate hydrogen production in real-world electrolyzers.
Looking ahead, the next Frontier in HER catalysis lies in actively controlling rather than passively observing catalyst reconstruction. We envision a new paradigm, termed Programmable Reconstruction, that treats catalyst evolution as an actively tunable process instead of an inevitable structural response. Wu et al. showed that square-wave potentials rapidly reconstruct oxide precursors into active metal/oxide heterointerfaces with superior efficiency and scalability over constant potential, validating a blueprint for Programmable Reconstruction.87 The emerging concept of programmable electrochemical potentials offers a route to realize Programmable Reconstruction by modulating atomic migration and interfacial phase transformation pathways, encoding reconstruction kinetics as a controllable design parameter.88 This is achieved through potential waveform engineering in the time and frequency domains, in line with the pulsed electrolysis framework articulated by Casebolt and colleagues. Detailly, time-domain potential pulses defined by amplitude, frequency, and duty cycle periodically amplify interfacial electric fields, accelerating atomic mobility and inducing defects at programmed densities. Frequency-domain excitation using characteristic frequencies and harmonics can be synchronized with intrinsic time scales of surface atom rearrangement or metastable phase formation, selectively activating reconstruction branches. When coupled with artificial intelligence (AI)-driven operando diagnostics, such as adaptive spectroscopic feedback and automated potential modulation, real-time mapping and control of dynamic structure–function relationships will become feasible.88 Sheng et al. demonstrate essential components of a closed-loop system by establishing an autonomous electrochemical platform where real-time signal streams inform AI-enabled state inference and actuate adaptive potential control, indicating the practical potential of closed-loop regulation for catalyst reconstruction.89 Furthermore, physics-informed inverse modeling powered by machine learning (ML) can reconstruct free-energy landscapes from operando data, revealing hidden metastable intermediates and predicting optimal excitation patterns for targeted reconstruction. To operationalize this goal, Bayesian optimization-guided, ML-driven waveform search provides a concrete, executable approach to efficiently explore pulsed-potential spaces and iteratively refine adaptive waveforms that align with inverse-inferred energetic and kinetic targets, enabling practical AI-assisted training of HER catalyst reconstruction pathways.90 Based on this analysis, we propose an editable closed-loop workflow for HER catalyst reconstruction. (1) Acquire operando signals to track evolving interfaces. (2) Use AI state inference to identify stages and update descriptors. (3) Apply ML-based inverse modelling to map free energies and predict target phases and excitation motifs. (4) Perform automated waveform actuation in time/frequency domains. (5) Refine waveforms via adaptive AI feedback to steer catalysts toward desired reconstructed active phases under operating conditions.88 By integrating these tools, reconstruction can evolve from an intrinsic response into a programmable feature, a data-driven, feedback-controlled paradigm for catalyst evolution. Ultimately, embracing programmable reconstruction will enable the rational “training” of catalysts to self-organize into thermodynamically favorable yet kinetically optimized active states, paving the way toward intelligent catalyst systems for scalable and durable hydrogen technologies.
Author contributions
X. Fu conceptualized the work, supervised the project, and revised the manuscript. X. Ding wrote the original draft and prepared the figures. All authors discussed the content and approved the final versionConflicts of interest
The authors declare no competing interests.Data availability
The authors declare that the data supporting the finding of this study are available in this paper.Acknowledgements
The authors acknowledge support from the startup grant provided by the National University of Singapore. X. Ding is grateful for funding sponsored by Shanghai Sailing Program under Grant No. 24YF2701900.Notes and references
- M. Chatenet, B. G. Pollet, D. R. Dekel, F. Dionigi, J. Deseure, P. Millet, R. D. Braatz, M. Z. Bazant, M. Eikerling, I. Staffell, P. Balcombe, Y. Shao-Horn and H. Schäfer, Chem. Soc. Rev., 2022, 51, 4583–4762 RSC.
- Y. Liu, Y. Guo, Y. Liu, Z. Wei, K. Wang and Z. Shi, Energy Fuels, 2023, 37, 2608–2630 CrossRef CAS.
- Y. Luo, Y. Zhang, J. Zhu, X. Tian, G. Liu, Z. Feng, L. Pan, X. Liu, N. Han and R. Tan, Small Methods, 2024, 8, 2400158 CrossRef CAS PubMed.
- J. Zhang, C. Ma, S. Jia, Y. Gu, D. Sun, Y. Tang and H. Sun, Adv. Energy Mater., 2023, 13, 2302436 CrossRef CAS.
- M. A. Qadeer, X. Zhang, M. A. Farid, M. Tanveer, Y. Yan, S. Du, Z.-F. Huang, M. Tahir and J.-J. Zou, J. Power Sources, 2024, 613, 234856 CrossRef CAS.
- Y. Zeng, M. Zhao, Z. Huang, W. Zhu, J. Zheng, Q. Jiang, Z. Wang and H. Liang, Adv. Energy Mater., 2022, 12, 2201713 CrossRef CAS.
- W. Zhang, Y. Yang, Y. Tang and Q. Gao, J. Energy Chem., 2022, 70, 414–436 CrossRef CAS.
- R. Zhang, B. Qian, K. Xu, A. Said, K. Chen, C. Yang, S. Komarneni and D. Xue, Rev. Mater. Res., 2025, 1, 100045 Search PubMed.
- Z. Li, X. Ding, D. Liu, J. Zhou, Y. Gao, Y. Liu, L. Jiang, R. Wu and H. Pan, Mater. Sci. Eng., R, 2025, 166, 101061 CrossRef.
- Y. Zheng, Y. Jiao, A. Vasileff and S.-Z. Qiao, Angew. Chem., Int. Ed., 2018, 57, 7568–7579 CrossRef CAS PubMed.
- N. Du, C. Wang, X. Wang, Y. Lin, J. Jiang and Y. Xiong, Adv. Mater., 2016, 28, 2077–2084 CrossRef CAS.
- C. Wei, Y. Sun, G. G. Scherer, A. C. Fisher, M. Sherburne, J. W. Ager and Z. J. Xu, J. Am. Chem. Soc., 2020, 142, 7765–7775 CrossRef CAS PubMed.
- Z. Chen, Y. Song, J. Cai, X. Zheng, D. Han, Y. Wu, Y. Zang, S. Niu, Y. Liu, J. Zhu, X. Liu and G. Wang, Angew. Chem., Int. Ed., 2018, 57, 5076–5080 CrossRef CAS PubMed.
- C. Miao, Y. Zang, H. Wang, X. Zhuang, N. Han, Y. Yin, Y. Ma, M. Chen, Y. Dai, S. Yip, J. C. Ho and Z.-X. Yang, Adv. Mater. Interfaces, 2022, 9, 2200739 CrossRef CAS.
- Y. Wang, X. Li, M. Zhang, Y. Zhou, D. Rao, C. Zhong, J. Zhang, X. Han, W. Hu, Y. Zhang, K. Zaghib, Y. Wang and Y. Deng, Adv. Mater., 2020, 32, 2000231 CrossRef CAS PubMed.
- I. S. Kwon, I. H. Kwak, G. M. Zewdie, S. J. Lee, J. Y. Kim, S. J. Yoo, J.-G. Kim, J. Park and H. S. Kang, Adv. Mater., 2022, 34, 2205524 CrossRef CAS PubMed.
- W. Sheng, M. Myint, J. G. Chen and Y. Yan, Energy Environ. Sci., 2013, 6, 1509–1512 RSC.
- W. Sheng, Z. Zhuang, M. Gao, J. Zheng, J. G. Chen and Y. Yan, Nat. Commun., 2015, 6, 5848 CrossRef CAS PubMed.
- I. T. McCrum and M. T. M. Koper, Nat. Energy, 2020, 5, 891–899 CrossRef CAS.
- J. Zhang, L. Zhang, J. Liu, C. Zhong, Y. Tu, P. Li, L. Du, S. Chen and Z. Cui, Nat. Commun., 2022, 13, 5497 CrossRef CAS PubMed.
- Y. Zhao, H. Li, R. Yang, S. Xie, T. Liu, P. Li, Y. Liu, H. Li, F. Yang and T. Zhai, Energy Environ. Sci., 2023, 16, 3951–3959 RSC.
- L. Zhai, T. W. Benedict Lo, Z.-L. Xu, J. Potter, J. Mo, X. Guo, C. C. Tang, S. C. Edman Tsang and S. P. Lau, ACS Energy Lett., 2020, 5, 2483–2491 CrossRef CAS.
- Q. Ma, C. Hu, K. Liu, S.-F. Hung, D. Ou, H. M. Chen, G. Fu and N. Zheng, Nano Energy, 2017, 41, 148–153 CrossRef CAS.
- C. Hu, Q. Ma, S.-F. Hung, Z.-N. Chen, D. Ou, B. Ren, H. M. Chen, G. Fu and N. Zheng, Chem, 2017, 3, 122–133 CAS.
- R. Wu, H. Liu, J. Xu, M.-R. Qu, Y.-Y. Qin, X.-S. Zheng, J.-F. Zhu, H. Li, X.-Z. Su and S.-H. Yu, Adv. Energy Mater., 2025, 15, 2405846 CrossRef CAS.
- F. Le, W. Jia, W. Shu, Z. Lu, Y. Lv, T. Wang, X. Wu, X. Yang, F. Ma and D. Jia, Small, 2024, 20, 2404894 CrossRef CAS PubMed.
- S. Jeong, Y. Zhang, J. Kang, S. Lee, J. M. Baik and H. Park, ACS Energy Lett., 2025, 10, 3591–3599 CrossRef CAS.
- P. Ji, R. Yu, P. Wang, X. Pan, H. Jin, D. Zheng, D. Chen, J. Zhu, Z. Pu, J. Wu and S. Mu, Adv. Sci., 2022, 9, 2103567 CrossRef CAS PubMed.
- J. Yao, Y. Zhang, F. Gao, Q. Jin, L. Zhang, L. Xu, M. Zhang, H. Gao and P. Yu, Energy Environ. Mater., 2025, 8, e70013 CrossRef CAS.
- X.-H. Zhang, M.-T. Zhang, H.-G. Du, H.-H. Huang, X.-F. Zhang, X. Wen, L.-D. Wang, W.-Z. Deng, Y.-M. He, J. Bai, L.-W. Ding and C.-T. He, Angew. Chem., Int. Ed., 2025, 64, e202507040 CrossRef CAS PubMed.
- Y. Xu, Y. Zhao, M. Sun, W. Xie, Y. Wu, G. Cheng, Y. Zhong, S. Han and L. Yu, Chem. Eng. J., 2024, 490, 151697 CrossRef CAS.
- L. Su, X. Cui, T. He, L. Zeng, H. Tian, Y. Song, K. Qi and B. Y. Xia, Chem. Sci., 2019, 10, 2019–2024 RSC.
- L. Wang, Y. Hao, L. Deng, F. Hu, S. Zhao, L. Li and S. Peng, Nat. Commun., 2022, 13, 5785 CrossRef CAS PubMed.
- H. Wang, X. Shao, Y. Wei, X. Ai, J. Yu, N. Xiao, R. Gan and Y. Qu, Appl. Catal., B, 2025, 367, 125110 CrossRef CAS.
- C. Chen, C. Du, Z. Wang, X. Chen, Y. Wang, N. Ju, Y. Fu, Z. Liu, M. Jia, S. Luo, G. Xu, J. Xu and H.-B. Sun, Energy Fuels, 2025, 39, 12154–12164 CrossRef CAS.
- W. Du, Y. Shi, W. Zhou, Y. Yu and B. Zhang, Angew. Chem., Int. Ed., 2021, 60, 7051–7055 CrossRef CAS PubMed.
- J. A. Bau, H. Haspel, S. Ould-Chikh, A. Aguilar-Tapia, J.-L. Hazemann, H. Idriss and K. Takanabe, J. Mater. Chem. A, 2019, 7, 15031–15035 RSC.
- J. A. Bau, S. M. Kozlov, L. M. Azofra, S. Ould-Chikh, A.-H. Emwas, H. Idriss, L. Cavallo and K. Takanabe, ACS Catal., 2020, 10, 12858–12866 CrossRef CAS.
- J. Nie, J. Shi, L. Li, M.-Y. Xie, Z.-Y. Ouyang, M.-H. Xian, G.-F. Huang, H. Wan, W. Hu and W.-Q. Huang, Adv. Energy Mater., 2025, 15, 2404246 CrossRef CAS.
- J. Wang, H.-Y. Tan, T.-R. Kuo, S.-C. Lin, C.-S. Hsu, Y. Zhu, Y.-C. Chu, T. L. Chen, J.-F. Lee and H. M. Chen, Small, 2021, 17, 2005713 CrossRef CAS PubMed.
- Y. Zhu, K. Fan, C.-S. Hsu, G. Chen, C. Chen, T. Liu, Z. Lin, S. She, L. Li, H. Zhou, Y. Zhu, H. M. Chen and H. Huang, Adv. Mater., 2023, 35, 2301133 CrossRef CAS PubMed.
- X. Ding, D. Liu, P. Zhao, X. Chen, H. Wang, F. E. Oropeza, G. Gorni, M. Barawi, M. García-Tecedor, V. A. de la Peña O’Shea, J. P. Hofmann, J. Li, J. Kim, S. Cho, R. Wu and K. H. L. Zhang, Nat. Commun., 2024, 15, 5336 CrossRef CAS PubMed.
- X. Ding, D. Liu, A. wang, P. Zhang, F. E. Oropeza, P. Zhao, G. Gorni, M. Barawi, V. de la Peña O'Shea, R. Wu and K. H. L. Zhang, Adv. Funct. Mater., 2025, e21566 CrossRef.
- M. Ledendecker, J. S. Mondschein, O. Kasian, S. Geiger, D. Göhl, M. Schalenbach, A. Zeradjanin, S. Cherevko, R. E. Schaak and K. Mayrhofer, Angew. Chem., Int. Ed., 2017, 56, 9767–9771 CrossRef CAS PubMed.
- Y. Zhu, H.-C. Chen, C.-S. Hsu, T.-S. Lin, C.-J. Chang, S.-C. Chang, L.-D. Tsai and H. M. Chen, ACS Energy Lett., 2019, 4, 987–994 CrossRef CAS.
- K. Fan, H. Zou, N. V. R. A. Dharanipragada, L. Fan, A. K. Inge, L. Duan, B. Zhang and L. Sun, J. Mater. Chem. A, 2021, 9, 11359–11369 RSC.
- L. Zhou, C. Yang, W. Zhu, R. Li, X. Pang, Y. Zhen, C. Wang, L. Gao, F. Fu, Z. Gao and Y. Liang, Adv. Energy Mater., 2022, 12, 2202367 CrossRef CAS.
- A. Zhu, L. Qiao, K. Liu, G. Gan, C. Luan, D. Lin, Y. Zhou, S. Bu, T. Zhang, K. Liu, T. Song, H. Liu, H. Li, G. Hong and W. Zhang, Nat. Commun., 2025, 16, 1880 CrossRef CAS PubMed.
- P. Fang, M. Zhu, J. Liu, Z. Zhu, J. Hu and X. Xu, Adv. Energy Mater., 2023, 13, 2301222 CrossRef CAS.
- C. Chen, H. Jin, P. Wang, X. Sun, M. Jaroniec, Y. Zheng and S.-Z. Qiao, Chem. Soc. Rev., 2024, 53, 2022–2055 RSC.
- X. Wang, Q. Ruan and Z. Sun, Energy Fuels, 2023, 37, 17667–17680 CrossRef CAS.
- X. Zheng, X. Shi, H. Ning, R. Yang, B. Lu, Q. Luo, S. Mao, L. Xi and Y. Wang, Nat. Commun., 2023, 14, 4209 CrossRef CAS PubMed.
- H. Tan, B. Tang, Y. Lu, Q. Ji, L. Lv, H. Duan, N. Li, Y. Wang, S. Feng, Z. Li, C. Wang, F. Hu, Z. Sun and W. Yan, Nat. Commun., 2022, 13, 2024 Search PubMed.
- D. Bao, L. Huang, Y. Gao, K. Davey, Y. Zheng and S.-Z. Qiao, J. Am. Chem. Soc., 2024, 146, 34711–34719 CrossRef CAS PubMed.
- A. Angulo, P. van der Linde, H. Gardeniers, M. Modestino and D. Fernández Rivas, Joule, 2020, 4, 555–579 Search PubMed.
- J. Feng, X. Wang and H. Pan, Adv. Mater., 2024, 36, 2411688 Search PubMed.
- J. M. Gisbert-González, C. G. Rodellar, J. Druce, E. Ortega, B. R. Cuenya and S. Z. Oener, J. Am. Chem. Soc., 2025, 147, 5472–5485 CrossRef PubMed.
- C. Feng, J. Shao, H. Wu, A. Hassan, H. Yang, J. Yu, Q. Hu and C. He, Chin. J. Catal., 2025, 72, 230–242 CrossRef CAS.
- N. Elgrishi, K. J. Rountree, B. D. McCarthy, E. S. Rountree, T. T. Eisenhart and J. L. Dempsey, J. Chem. Educ., 2018, 95, 197–206 CrossRef CAS.
- C. Ariño, C. E. Banks, A. Bobrowski, R. D. Crapnell, A. Economou, A. Królicka, C. Pérez-Ràfols, D. Soulis and J. Wang, Nat. Rev. Methods Primers, 2022, 2, 62 CrossRef.
- D. Hochfilzer, A. Tiwari, E. L. Clark, A. S. Bjørnlund, T. Maagaard, S. Horch, B. Seger, I. Chorkendorff and J. Kibsgaard, Langmuir, 2022, 38, 1514–1521 CrossRef CAS PubMed.
- N. Mayet, K. Servat, K. B. Kokoh and T. W. Napporn, Surfaces, 2019, 2, 257–276 Search PubMed.
- M. Rocca, T. Rahman and L. Vattuone, Springer handbook of surface science, Springer Nature, 2021 Search PubMed.
- J. Tymoczko, W. Schuhmann and A. S. Bandarenka, ChemElectroChem, 2014, 1, 213–219 CrossRef.
- Z. Xiao, Y.-C. Huang, C.-L. Dong, C. Xie, Z. Liu, S. Du, W. Chen, D. Yan, L. Tao, Z. Shu, G. Zhang, H. Duan, Y. Wang, Y. Zou, R. Chen and S. Wang, J. Am. Chem. Soc., 2020, 142, 12087–12095 CrossRef CAS PubMed.
- H.-Y. Wang, S.-F. Hung, H.-Y. Chen, T.-S. Chan, H. M. Chen and B. Liu, J. Am. Chem. Soc., 2016, 138, 36–39 CrossRef CAS PubMed.
- X. Liu, J. Meng, J. Zhu, M. Huang, B. Wen, R. Guo and L. Mai, Adv. Mater., 2021, 33, 2007344 CrossRef CAS PubMed.
- H. S. Magar, R. Y. A. Hassan and A. Mulchandani, Sensors, 2021, 21, 6578 CrossRef CAS PubMed.
- Y. Liu, J. Wu, K. P. Hackenberg, J. Zhang, Y. M. Wang, Y. Yang, K. Keyshar, J. Gu, T. Ogitsu, R. Vajtai, J. Lou, P. M. Ajayan, B. C. Wood and B. I. Yakobson, Nat. Energy, 2017, 2, 17127 Search PubMed.
- K. Dastafkan, S. Wang, S. Song, Q. Meyer, Q. Zhang, Y. Shen and C. Zhao, EES Catal., 2023, 1, 998–1008 RSC.
- J. Jiang, G. Xu, B. Gong, J. Zhu, W. Wang, T. Zhao, Y. Feng, Q. Wu, S. Liu and L. Zhang, Adv. Funct. Mater., 2025, 35, 2412685 CrossRef CAS.
- H. Wang, Y. Jiao, G. Zhang, W. Ma, W. Fan, X. Liu, Y. Zhao, H. Xie, W. Ma and X. Zong, Adv. Funct. Mater., 2025, 35, 2418617 CrossRef CAS.
- Z. Zhao, Y. Chen, Y. Liu, S. Qin, Z. Li, Z. Zhang and X. Meng, Adv. Funct. Mater., 2025, e28280 CrossRef.
- X. Chen, M. Bi, Q. Yan, D. Fan, B. Huang, B. Sun, C. Qin, C. Chen, D. Sun, Q. He and M. Zhao, Adv. Mater., 2025, 37, e08893 CrossRef CAS PubMed.
- J. Kwon, S. Choi, C. Park, H. Han and T. Song, Mater. Chem. Front., 2024, 8, 41–81 RSC.
- L. Zhang, Z. Shi, Y. Lin, F. Chong and Y. Qi, Front. Chem., 2022, 10, 866415 Search PubMed.
- X. Gao, P. Wang, X. Sun, M. Jaroniec, Y. Zheng and S. Z. Qiao, Angew. Chem., 2025, 137, e202417987 CrossRef.
- R. Lin, Y. Lu, J. Xu, J. Huo and X. Cai, Appl. Energy, 2022, 326, 120011 CrossRef CAS.
- A. Bashkatov, S. Park, Ç. Demirkır, J. A. Wood, M. T. M. Koper, D. Lohse and D. Krug, J. Am. Chem. Soc., 2024, 146, 10177–10186 CrossRef CAS PubMed.
- Q.-N. Wang, Y. Qiao, W. Qin, H. Zheng, Z. Wang, B. Chang, X. Qu, W. Zhang, T. Liu and C. Li, Ind. Eng. Chem. Res., 2025, 64, 5087–5098 Search PubMed.
- B. T. Sangtam and H. Park, Micromachines, 2023, 14, 2234 CrossRef PubMed.
- Y. Lin, D. Huang, Q. Wen, R. Yang, B. Chen, Y. Shen, Y. Liu, J. Fang, H. Li and T. Zhai, Proc. Natl. Acad. Sci. U. S. A., 2024, 121, e2407350121 Search PubMed.
- L. Xia, B. F. Gomes, W. Jiang, D. Escalera-López, Y. Wang, Y. Hu, A. Y. Faid, K. Wang, T. Chen, K. Zhao, X. Zhang, Y. Zhou, R. Ram, B. Polesso, A. Guha, J. Su, C. M. S. Lobo, M. Haumann, R. Spatschek, S. Sunde, L. Gan, M. Huang, X. Zhou, C. Roth, W. Lehnert, S. Cherevko, L. Gan, F. P. García de Arquer and M. Shviro, Nat. Mater., 2025, 24, 753–761 CrossRef CAS PubMed.
- X. Liu, J. Meng, K. Ni, R. Guo, F. Xia, J. Xie, X. Li, B. Wen, P. Wu, M. Li, J. Wu, X. Wu, L. Mai and D. Zhao, Cell Rep. Phys. Sci., 2020, 1, 100241 Search PubMed.
- E. Kuhnert, V. Hacker and M. Bodner, Int. J. Energy Res., 2023, 2023, 3183108 CrossRef.
- P. Aßmann, A. S. Gago, P. Gazdzicki, K. A. Friedrich and M. Wark, Curr. Opin. Electrochem., 2020, 21, 225–233 CrossRef.
- H.-C. Wu, Y.-W. Huang, Y.-C. Lin, C.-H. Yang and T.-C. Liu, ACS Appl. Mater. Interfaces, 2025, 17, 56991–57001 CrossRef CAS PubMed.
- A. Samreen, M. Azim and F. Chen, AI Mater., 2025, 1, 1–2 Search PubMed.
- H. Sheng, J. Sun, O. Rodríguez, B. B. Hoar, W. Zhang, D. Xiang, T. Tang, A. Hazra, D. S. Min, A. G. Doyle, M. S. Sigman, C. Costentin, Q. Gu, J. Rodríguez-López and C. Liu, Nat. Commun., 2024, 15, 2781 Search PubMed.
- C. S. Movassaghi, K. A. Perrotta, M. E. Curry, A. N. Nashner, K. K. Nguyen, M. E. Wesely, M. Alcañiz Fillol, C. Liu, A. S. Meyer and A. M. Andrews, Digital Discovery, 2025, 4, 1812–1832 RSC.
| This journal is © The Royal Society of Chemistry 2026 |