Open Access Article
Open Access ArticleGassing behavior and safety issues of Li-rich cathodes
Haitang Zhangabc,
Jian Wangabcd,
Lingqiao Wuabc,
Shiqi Liuabc,
Yuan Liangabc,
Shu Zhaoabc and
Haijun Yu *abc
*abc
aInstitute of Advanced Battery Materials and Devices, College of Materials Science and Engineering, Beijing University of Technology, Beijing 100124, China. E-mail: hj-yu@bjut.edu.cn
bInstitute of Gongda-Guochuang Advanced Battery Materials and Devices, Beijing, 100176, P. R. China
cState Key Laboratory of Materials Low-Carbon Recycling, Institute of Matter Science, Beijing University of Technology, Beijing 100124, China
dPhylion Battery Co., Ltd, Suzhou 320500, China
First published on 6th March 2026
Abstract
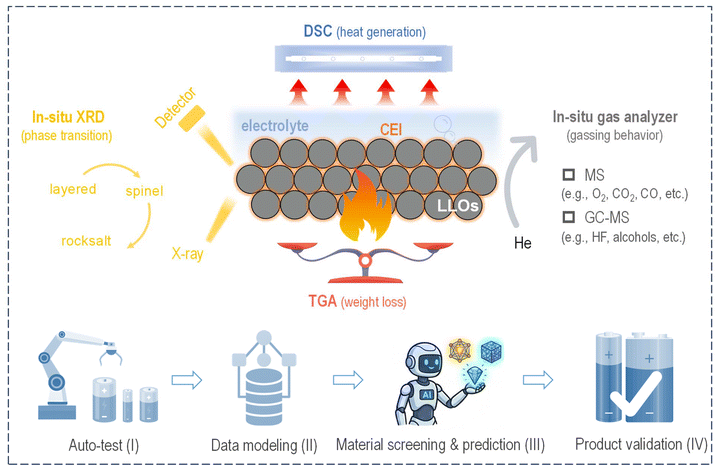
Over the past few decades, lithium-rich layered oxides (LLOs) have attracted significant research attention on account of their prominent advantage of high energy density. However, gas evolution is a crucial issue, exerting a detrimental impact on the cycle performance and intrinsic safety of a battery. This perspective systematically shows that gas evolution is largely attributable to the release of high-reactivity oxygen species under high-voltage or high-temperature conditions. We suggest that future research into the gas-related thermal runaway (TR) safety issue should focus on two scientific aspects. (i) The thermal stability of a delithiated LLO cathode: what is the intrinsic correlation between the microstructural characteristics and thermal stability of delithiated LLOs, and what are the key governing factors that dictate their thermal stability? (ii) The thermal stability of the cathode–electrolyte interface: what are the detailed transformation pathways of reactive oxygen species and the specific interfacial reaction mechanisms occurring at the cathode–electrolyte interface (for delithiated LLOs) under the heating process? On this basis, the development of a multimodal combined characterization platform is innovatively proposed, including in situ XRD, an in situ gas analyzer, DSC, TGA, and other complementary technologies. Based on collected comprehensive information (e.g., phase transitions, gassing behavior, heat generation, and weight loss), the intrinsic structure–activity relationships between microstructural characteristics, gassing behavior, and TR risks are expected to be unveiled. Assisted by artificial intelligence (AI), the R&D cycle for low-gas-emission, high-safety LLO materials and matched electrolytes could be substantially shortened.
Broader contextIn view of the environmental goal of “farbon peaking and carbon neutrality” and the thriving development of “the low-altitude economy, electric vehicles and energy storage”, the industrial market is imposing higher standards on the cycling performance and safety risks of lithium-ion batteries, especially “on-flammable and non-explosive” high-energy-density battery systems. Li-rich cathode materials exhibit a prominent advantage of ultrahigh specific capacity derived from the “formed π-bond ring” in the LiMn6 unit, and are regarded as promising candidates for next-generation high-energy-density cathodes. Meanwhile, some ensuing problems hinder further practical applications, among which serious gas evolution is one of the most noticeable problems. From the perspective of materials research, this perspective focuses on research progress, sorts out the gas generation mechanism, and, in particular, discusses key scientific questions regarding the gas-related safety issue in LLO-based battery systems. The safety issue largely originates from the thermal stability of a de-lithiated LLO cathode and the corresponding cathode–electrolyte interface. Thus, the development of a multimodal combined characterization platform (including in situ XRD, an in situ gas analyzer, DSC and TGA) is suggested to decouple the internal connections of microstructural characteristics, thermal stability, and thermal runaway risks, providing valuable guidance for achieving low-gas-emission, high-safety LLO-based battery systems. |
1. Introduction
With the vigorous development of the low-altitude economy, electric vehicles, and consumer electronics over the past few decades, there is an urgent demand for high-energy-density lithium-ion batteries (LIBs) featuring reliable intrinsic safety and long cycle life to meet market requirements.1 Obviously, research into cathode materials plays a crucial role in advancing the development of the entire battery system. Among various cathode candidates, lithium-rich layered oxides (LLOs) have garnered widespread attention from both academic and industrial communities, owing to their advantage of high specific energy—stemming largely from an extra oxygen anionic redox (OAR) reaction. However, the ensuing severe gas generation issues (e.g., O2 and CO2)—particularly under various extremely harsh working conditions (e.g., overcharging and high temperature)—can trigger rapid performance degradation and pose potential safety hazards.Once the battery is charged to a high voltage, the surface of highly delithiated LLOs would present high reactivity, and irreversible oxygen species would escape from the cathode lattice. Meanwhile, the released oxygen species act as a strong oxidizing agent, initiating the decomposition of other surrounding battery components—including the electrolyte, binder, and conductive additives—accompanied by the generation of various gaseous species, such as CO2 and CO. Moreover, gases originating from the cathode side shuttle through the porous separator to the anode side and interfere with the dynamic equilibrium of the formation of a solid electrolyte interphase (SEI) film, affecting the quality of the formed SEI film. More importantly, the gases released during long-term cycling will gradually accumulate inside the sealed battery casing, as the generated gas cannot be effectively dissipated. This gas accumulation leads to a sharp rise in internal pressure and stress, a continuous increase in overall battery impedance, and even obvious battery swelling or bulging. The swollen battery not only suffers from poor electrode contact and reduced ion transport efficiency but also poses a risk of damage to packaging due to excessive internal stress, which in turn accelerates the rate of capacity fade and performance degradation, significantly shortening the cycle life of the battery.2,3
In addition, if the battery suffers from internal defects (e.g., micro-shorts) or external abuse (e.g., overcharging, crushing, or high temperature), its internal temperature will inevitably rise rapidly. At elevated temperature, the surface of highly delithiated LLOs exhibits extremely high reactivity, far exceeding that observed under normal operating conditions. The irreversible release of oxygen species could occur when the delithiated LLO cathodes are heated to approximately 200 °C. The released oxygen species can ignite the combustion of electrolyte, binder, separator, anode, and other flammable battery components. Such violent oxidation reactions are bound to trigger an uncontrollable thermal runaway (TR) process. The temperature could surge instantly to about 1000 °C within seconds, posing significant safety hazards for practical applications.4–6
Therefore, it is crucial to obtain an in-depth understanding of the gas evolution mechanism and to develop effective strategies to suppress gas evolution, especially under extreme TR conditions. Although several key reviews have extensively covered gassing behavior7,8 or safety issues4,9 within various battery systems, there remains a notable absence of a dedicated review or perspective article focusing specifically on the Li-rich cathode system. Such a perspective article is essential to elucidate the distinct mechanisms of electrochemical and TR-induced gas evolution, thereby providing critical guidance for rational material design. Here, we systematically outline current research progress into gas generation for LLO-based batteries, further assess available approaches to inhibit gas release, refine key scientific questions, and provide critical insights into their practical applicability. Most importantly, we tentatively present a personal perspective on multimodal characterizations for heat stability analysis of delithiated LLOs (with electrolyte), including in situ X-ray diffraction (XRD), an in situ gas analyzer (dual detector of MS and GC-MS), differential scanning calorimetry (DSC), thermogravimetric analysis (TGA), and other complementary characterizations. When integrated with artificial intelligence (AI) algorithms, these multimodal characterization techniques are expected to unlock the intrinsic correlations between the phase transitions, heat release, gas evolution, and weight loss of LLOs. This synergistic approach can not only provide robust support for mechanistic research on gas generation and thermal stability but also offer valuable guidance for material screening, structural/component optimization, and performance prediction of a low-gas-emission, high-safety LLO cathode or matching electrolyte.
2. In-depth analysis of the gassing mechanism during the cycling process
2.1 O2 evolution derived from an oxygen anionic redox reaction
When LLO cathodes are charged to a high voltage (>4.5 V vs. Li+/Li) or a highly delithiated state, a unique OAR reaction could be activated, corresponding to an observed characteristic voltage platform around 4.5 V in the charging curve (Fig. 1a, top panel). Apart from metal cationic redox (MCR), lattice oxygen (O2−) in LLOs would participate in the charge compensation process, and extra capacity could be obtained, which is the core advantage of their ultrahigh specific energy. In 2016, Bruce's group utilized 18O to label a portion of the lattice oxygen in an Li1.2[Ni0.13Co0.13Mn0.54]O2 cathode and carried out differential electrochemical mass spectrometry (DEMS). Based on the collected gas evolution profiles of O2 and CO2 (Fig. 1a, middle and bottom panels), substantial amounts of 16/18O2 and 16/18CO2 were clearly detected upon charging to a high voltage, demonstrating that the vast majority of the detected O2 gas originates directly from the release of lattice oxygen rather than electrolyte oxidation. In 2016, Ceder's group further argued that the intrinsic origin of the OAR reaction in LLOs lies in the presence of Li–O–Li local configurations within the octahedral coordinated framework.10 Such configurations give rise to unhybridized O 2p states, whose energy levels are higher than those of hybridized O 2p states (t1ub, a1gb, egb). Consequently, these unhybridized states are more susceptible to oxidation to peroxo, superoxo, and molecular oxygen species. Among them, molecular oxygen can be easily analyzed using high-resolution mass spectrometry. Thus, numerous groups have adopted a variety of strategies to stabilize anionic redox reactions and suppress the irreversible release of O2, including structural engineering, surface modification, and electrolyte optimization.11–16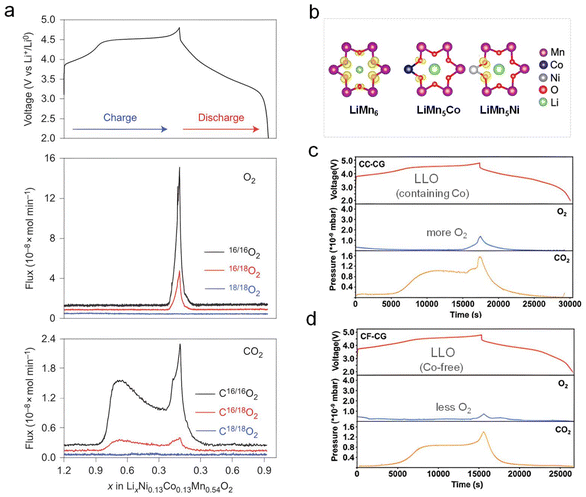 | ||
| Fig. 1 O2 evolution derived from O-related redox chemistry. (a) The DEMS test results of the Li1.2Ni0.13Co0.13Mn0.54O2 cathode during the first cycle. The observation of 18O-labelled O2 and CO2 proves that O is extracted from the lattice when charged above the 4.5 V plateau. Reproduced with permission.17 Copyright 2016, Springer Nature. (b) The arrangements of LiMn6, LiMn5Co, and LiMn6Ni functional units in the TM layer. (c and d) The influence of Co on the O2 evolution behavior of LLOs. Co would disrupt the symmetry of the π-bonded ring in the LiMn6 structure. Consequently, the synthesized Co-free LLOs exhibit reduced release of O2 during the initial cycle, indicating more reversible oxygen redox behavior. Reproduced with permission.18 Copyright 2025, National Academy of Sciences. | ||
Compared to traditional layered cathode materials (e.g., LiCoO2 or LiNixCoyMn1−x−yO2), LLO cathodes feature unique LiMn6 hexatomic-ring functional units in transition metal (TM) layers. Each Li atom in an LiMn6 unit is coordinated by six Mn atoms, and each Mn atom is shared by three LiMn6 units. In addition, each oxygen atom in an LiMn6 unit uses two O 2p orbitals to form σ-bonds with Mn atoms, whereas the remaining O 2p orbital along the Li–O axis exhibits only weak overlaps with Li 2s orbitals, rather than the formation of σ-type hybridization. These O 2p orbitals interact with Mn 3d(t2g) orbitals to form π-type bonding (t2g) and antibonding (t2g*) molecular orbitals. The π-type hybridization of six O 2p and six Mn 3d(t2g) orbitals within the LiMn6 unit further constructs an intact [Mn–O] π-bond ring. In particular, recent pioneering studies conducted in our group found that this π-bond ring behaves as a collective redox center and plays a decisive role in governing OAR reversibility in LLOs. The capacity, voltage, and structural stability of LLOs are therefore determined by the intrinsic properties of the [Mn–O] π-bond ring, rather than by any specific bonding environment. Importantly, partial substitution of Mn by Co or Ni disrupts the symmetry and integrity of the π-bond ring, leading to reduced oxygen stability. In contrast, the intact π-bond ring preserved in Co-free LiMn6 frameworks enables stable redox reactions to proceed as a collective unit, thereby exhibiting enhanced electrochemical stability (Fig. 1b). As depicted in Fig. 1c and d, the designed/synthesized Co-free LLOs (CF-CG-LLOs) cathode materials exhibit reduced O2/CO2 evolution compared with their Co-containing counterparts (CC-CG-LLOs), indicating a more reversible OAR reaction. Undoubtedly, pouch cells assembled with CF-CG-LLOs exhibit superior electrochemical performance compared to those with CC-CG-LLOs, featuring outstanding capacity retention and remarkably low voltage fade. This important research provides a new perspective for the design of low-cost, high-energy-density LLO cathode materials.18 In addition, surface coatings (e.g., metal oxides, fast-ion conductors, or spinel heterostructures) could effectively stabilize lattice oxygen and suppress parasitic reactions with the electrolyte. Meanwhile, bulk doping with heteroatoms (e.g., Mg or Ti) enhances M–O bond covalency or modulates the local coordination environment of oxygen, thermodynamically inhibiting irreversible oxygen evolution.19 These strategies collectively provide important insights for the design of high-stability LLOs.
2.2 Gas evolution derived from the cathode–electrolyte interface
When charged to a high voltage, the surface of an LLO always exhibits exceptionally high oxidation ability owing to the presence of high-valence TMs and oxidized oxygen. Apart from the bulk material being a source of gas evolution, more gas evolution is likely to originate from the interface between the LLO cathode and the electrolyte.As we all know, synthesized LLOs are highly sensitive and residual lithium species (e.g., LiOH, Li2O, Li2O2 or LiHCO3)—which originate from the excess lithium source used during the synthetic process—are prone to react with CO2, O2 and H2O in ambient air during storage, thus forming Li2CO3.20 As shown in Fig. 2a, these Li2CO3 impurities would be electrochemically oxidized to generate CO2 gas and oxygen species. Afterwards, the oxygen species would continue to react with a carbonate-based electrolyte solvent to produce CO2/CO gas and organic impurities.21 Notably, no signals of molecular oxygen (O2) have been detected using the DEMS method until now—a rather confounding observation. Furthermore, Gasteiger's group argued that Li2CO3 decomposition is a chemical pathway, where HF species react with Li2CO3 to release CO2 gas.22 Based on the aforementioned findings, McCloskey's group23 employed a rapid acid-based surface treatment strategy to eliminate residual Li2CO3, thereby achieving marked reductions in gas generation and substantial improvements in electrochemical performance (Fig. 2b). Furthermore, Li2CO3 is commonly used as a cathode pre-lithiation agent. If effective measures can be taken to fully activate the surface Li2CO3 impurities and replenish Li loss, then we could turn this originally detrimental substance into a valuable component.24
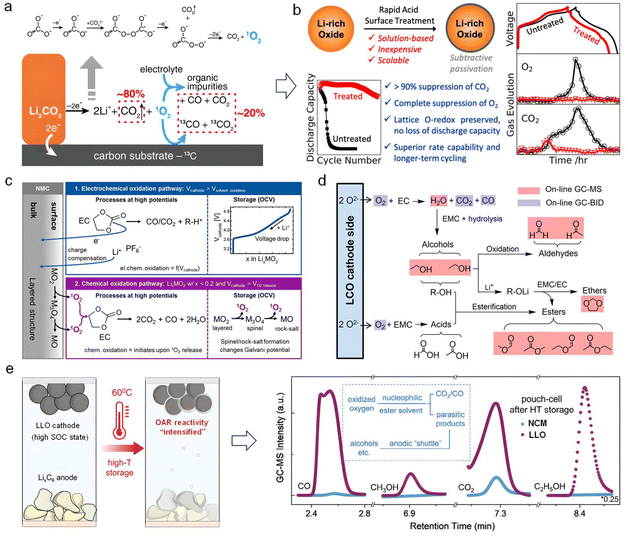 | ||
| Fig. 2 The gassing behavior on the cathode–electrolyte interface during cycling. (a) The influence of surface Li2CO3 on gassing behavior. The (electro-)chemical decomposition of surface Li2CO3 impurities occurs throughout the charge process, accompanied by the generation of CO2. Reproduced with permission.21 Copyright 2022, Springer Nature. (b) Depressed release of O2 and CO2 via rapid acid treatment to eliminate surface Li2CO3. Reproduced with permission.23 Copyright 2020, American Chemical Society. (c) The gassing behavior derived from the (electro-)chemical oxidation decomposition of carbonate-based electrolyte. Reproduced with permission.25 Copyright 2021, The Electrochemical Society. (d) The chain reactions induced by released O2 based on online GC-BID/MS test results. Reproduced with permission.26 Copyright 2024, Wiley. (e) Gas production comparison between NCM and LLO pouch cells (at the same holding voltage of 4.43 V) after high-temperature storage. Reproduced with permission.27 Copyright 2024, Wiley. | ||
Another critical source of gas generation is the decomposition of a carbonate-based electrolyte, especially for cyclic electrolyte solvents (e.g., EC solvent). Similar to Li2CO3, the decomposition of electrolyte solvents involves two pathways: electrochemical oxidation and chemical oxidation, as shown in Fig. 2c. High-valence TM ions on the surface of LLOs tend to catalyze the electrochemical oxidation decomposition of electrolyte solvents, thereby generating CO2/CO gases and proton species (R–H+). In addition, the generated 1O2 can attack the EC solvent. Gasteiger's group proposed the corresponding chemical reaction equation:25
| C3H4O3(EC) + 31O2 → 2CO2 + CO + 2H2O | (1) |
Recently, Qiao's group developed an original online electrochemistry gas chromatography–barrier discharge ionization/mass spectrometry (GC-BID/MS) technology beyond the traditional DEMS method. Not limited to O2, CO2, and CO gases, a range of other volatile species (including alcohols, ethers, carboxylic esters, and aldehydes) could also be detected in an LCO-Li half-cell system. Given the wide range of detected gaseous products, the chain reactions triggered by released O2 are being systematically perfected, as listed in Fig. 2d. Obviously, not all gaseous by-products are harmful to a battery system, such as ether and carboxylic ester by-products. Among them, ethers are friendly for a lithium metal anode, and carboxylic esters are more suitable for low-temperature working conditions. If we follow the principle of “seeking benefits and avoiding harm” and take measures to promote the generation of beneficial species and suppress the generation of harmful species, the overall performance will be significantly enhanced.
The gas evolution behavior at the cathode–electrolyte interface could drastically deteriorate under extreme operating conditions. For fully charged pouch-type cells, a cell assembled with LLO cathodes presents much more serious gassing issues than an NCM811-based cell after prolonged high-temperature (HT) storage (Fig. 2e). This phenomenon could be explained by the intensified OAR reactions induced by HT storage, followed by the escape of oxidized oxygen species (On−). Such a process would disrupt the coordination environment of TMs and exacerbate electrolyte decomposition, producing a large amount of gas.27 In addition, gas accumulation during electrochemical processes could increase interfacial impedance, generate particle cracking, and create an unstable cathode electrolyte interface (CEI) film, which will inevitably lower the onset temperature of the TR reaction and/or exacerbate the TR reaction.
3. Gassing behavior and safety issues during the thermal runaway process
3.1 O2 evolution during the TR process
In recent years, concerns about battery safety have grown increasingly prominent within both academic and industrial communities, driven by the escalating demand for high-performance energy storage devices in electric vehicles and grid-scale applications. In response to this critical challenge, numerous researchers have directed their efforts toward comprehensively investigating crucial trigger factors, underlying reaction mechanisms, and corresponding modification strategies for safer LIBs. Nevertheless, the operational safety of LIB products still cannot be fully ensured, and this is particularly pronounced for high-energy-density battery systems (e.g., batteries assembled with NCM and LLO cathodes), which face heightened TR risks under extreme operating conditions. We propose that concerns about battery safety stem largely from the poor thermal stability of battery components, especially delithiated cathodes, which take primary responsibility for the TR risks of full cells.19,28Ouyang's group has reported a series of far-reaching and outstanding works on the battery TR safety issue. As described in Fig. 3a, they proposed that NCM cathodes would undergo severe O2 evolution, phase transition (from a layered structure to spinel and further rock salt phases), and TM dissolution during the TR process, when the temperature of the battery increases to approximately 200 °C.29 The high-reactivity O2 would immediately react with surrounding combustible battery components, accompanied by the generation of a large amount of heat and the occurrence of uncontrollable TR events. Thus, the release of O2 from delithiated cathodes has been considered a key trigger for the TR process. Compared with traditional layered cathode materials, the TR reaction mechanism for highly delithiated LLO cathodes probably tends to be more complex, attributable to their high charge voltage, excessive OAR reaction, and the unique LiMn6 units. Thus, it is essential to clarify the intrinsic connection between structural evolution, O2 release, and heat generation during the TR process.
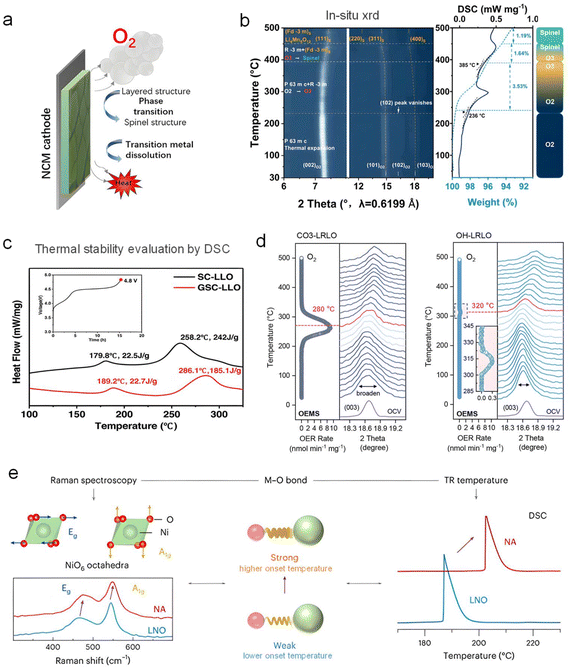 | ||
| Fig. 3 O2 evolution from delithiated LLO cathodes during the gradual heating process. (a) Schematic illustration for the thermal stability of delithiated layered cathodes, accompanied by phase transition (from a layered structure to a spinel structure), O2 evolution, heat release, and transition metal dissolution. Reproduced with permission.29 Copyright 2022 Elsevier. (b) The in situ XRD and TG-DSC test results for a highly delithiated O2-type LLO cathode (Li0.390Ni0.133Co0.133Mn0.564O2, 30–500 °C, 5 °C min−1). Reproduced with permission.30 Copyright 2025, Wiley. (c) Thermal stability evaluation by DSC. Less heat release always represents superior thermal stability. Reproduced with permission.31 Copyright 2022, Wiley. (d) Thermal stability evaluation by in situ XRD and MS. Hydroxide-precursor-derived (OH−) LLO cathodes show superior thermal stability to carbonate-precursor-derived (CO32−). Reproduced with permission.32 Copyright 2025, Wiley. (e) Schematics illustrating the correlation between M–O bond strength and thermal stability. Reproduced with permission.28 Copyright 2025, Springer Nature. | ||
As shown in Fig. 3b, Li's group employed in situ XRD and TG-DSC technologies to investigate the thermal decomposition mechanisms and differences in inherent stability between O3 and O2 types of LLOs. During the linear heating process, highly delithiated O2-LLOs would undergo a phase transition from the metastable O2-type layered structure to an intermediate O3-type layered phase, followed by further conversion to a spinel phase. In contrast, delithiated O3-LLOs would directly transform from their original O3-type layered phase to the spinel phase.30 As shown in Fig. 3c, our group has consistently utilized DSC as a powerful characterization tool to fairly evaluate the thermal stability of synthesized LLO materials (charged to 4.8 V Li+/Li). Clearly, GSC-LLO, which exhibits a higher onset temperature and reduced heat release of the exothermic peak, indicates superior thermal stability compared to SC-LLO.31 In addition, Qiao's group used in situ XRD coupled with mass spectrometry (MS) to compare the thermal stability of LLO cathodes derived from carbonate (CO32−) and hydroxide (OH−) precursors (Fig. 3d). Evidently, the hydroxide-precursor-derived LLOs exhibit a higher O2 release temperature, indicating superior thermal stability. Furthermore, as presented in Fig. 3e, Manthiram's group employed Raman spectroscopy to investigate the thermal stability of various high-nickel cathodes, and found that thermal stability is determined by the metal–oxygen (TM–O) bond covalency. Specifically, a stronger TM–O bond corresponds to a higher onset temperature of the exothermic peak and superior thermal stability. Meanwhile, thermal stability is also influenced by factors such as cation mixing and particle size.
Clearly, the thermal stability of delithiated LLOs directly governs the safety performance of full cells. Thus, it is crucial to elucidate the inter-relationships among crystal structure, O2 release, heat generation, and weight loss, as well as to identify the factors governing LLO thermal stability, providing a clear direction for structural design and regulation for safer LLO cathode materials, such as heteroatom doping and surface coating.
3.2 Gassing behavior derived from the cathode–electrolyte interface during the TR process
Overall, a delithiated cathode sample with an electrolyte exhibits significantly more heat release compared to a cathode-only sample without electrolyte, suggesting that heat generation on the cathode side is derived primarily from interfacial reactions between the cathode and electrolyte. Thus, elucidating the detailed reaction mechanisms at the electrode–electrolyte interface is crucial to mitigating heat generation. As depicted in Fig. 4a, Ouyang's group proposed a transformation process for O2 gas on a layered oxide cathode surface during the heating process. (i) Initially, the oxygen species may be chemically adsorbed as oxidized oxygen species (O22−/O2−/O−) located at lattice positions. (ii) As the temperature continues to rise, a portion of these oxygen species will escape from the lattice and convert into physically adsorbed molecular oxygen, which is localized primarily on the particle surface. (iii) Afterwards, further evolved O2 gas would desorb from the cathode surface (Fig. 4a, top panel). They proposed that reactions between these reactive oxygen species and the electrolyte have been identified as key factors responsible for initiating TR (Fig. 4a, middle panel). Given the strong anion-coordination capability of tris(pentafluorophenyl)borane (TPFPB), these oxidized oxygen species are expected to be trapped by TPFPB, thereby protecting the electrolyte from direct attack (Fig. 4a, down panel).33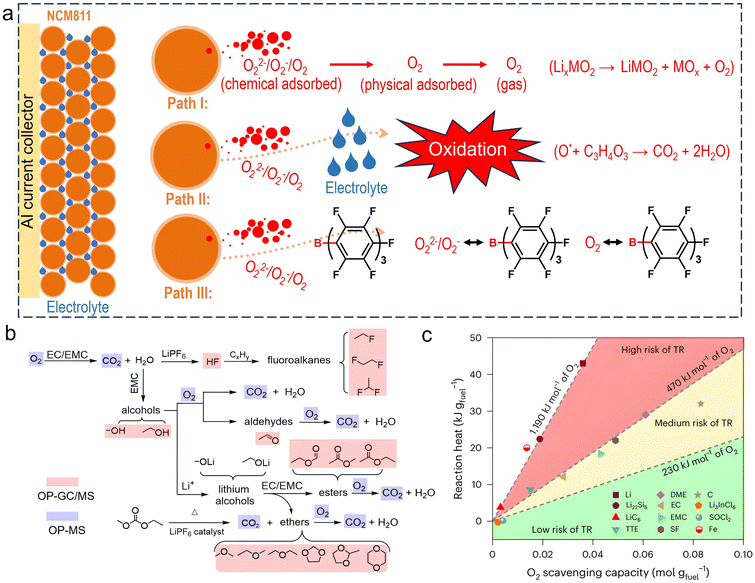 | ||
| Fig. 4 Gassing behavior on the cathode–electrolyte interfacial surface during the heating process. (a) Proposed transformation pathway of oxidized oxygen species to O2 gas evolution and modification mechanism of TPFPB additive for scavenging highly reactive oxidized oxygen species. Reproduced with permission.33 Copyright 2021, Elsevier. (b) O2-release-induced chain reactions at the electrode–electrolyte interface, based on OP-MS and OP-GCMS test results during the heating process. Reproduced with permission.34 Copyright 2024, Wiley. (c) The reaction heat and O2 scavenging capacity of various battery components. Reproduced with permission.35 Copyright 2025, Springer Nature. | ||
Based on the results obtained from online pyrolysis mass spectrometry/gas chromatography–mass spectrometry (OP-MS/GC-MS) for delithiated NCM cathodes in the presence of carbonate-based electrolytes (Fig. 4b), various gaseous species generated at high temperature were monitored in real time during heating, and the chain reactions initiated by oxygen species were further elucidated. (i) Clearly, oxygen species will attack the EC/EMC solvents, generating H2O and CO2 gases. (ii) The resulting H2O then induces the hydrolysis of the LiPF6 salt, accompanied by the production of corrosive HF gas. (iii) Subsequently, the generated HF reacts with the Li+-intercalated carbon anode, generating substantial heat. Meanwhile, a series of organic vapors were detected, and most of them are flammable, explosive, and even poisonous, such as fluoroalkane, alcohols, aldehydes, carboxylic esters, ethers, and ketones. When a TR event occurs, the secondary products triggered by oxygen species may be even more detrimental, and they deserve greater focus in subsequent research.34 Furthermore, sacrificing oxygen species or interrupting the oxygen-mediated chain reaction represents key strategies for battery safety protection.
Surprisingly, Wang's group found that batteries with an all-solid-state electrolyte or non-flammable liquid electrolytes may face greater security risks compared to those with traditional carbonate- and ether-based electrolytes. As for the former, oxygen species released from delithiated cathodes can pass directly to the lithiated carbon anode without any O-loss, thereby triggering the highly energetic lithium oxidation reaction (LOR). As for the latter, the oxygen species released from delithiated cathodes would react rapidly with traditional carbonate- or ether-based electrolytes, and the oxygen species may be partially consumed or completely depleted in transit, alleviating or avoiding LOR. As shown in Fig. 4c, they summarized the reaction heat (KJ gfuel−1) and O2 scavenging capacity of various battery materials. They proposed that safe electrolytes should have high O2-scavenging capacity while generating low reaction heat, thereby providing new guidance for the development of safer battery systems.35
3.3 Gas-related crosstalk reactions during the TR process
Prior to battery explosion, the generated gas would gradually accumulate inside the battery, and gas-related crosstalk reactions between the delithiated cathode and the lithiated anode would proceed extremely vigorously, accompanied by the generation of a substantial amount of heat. Thus, it is imperative to establish an in-depth understanding of the gas-related crosstalk reaction mechanism during the TR process.As can be seen in Fig. 5a, the O2 and CO2 gas from the cathode side would shuttle to the anode side, and react with lithiated carbon to produce solid Li2O and Li2CO3, respectively.36 In addition, the reductive gases (e.g., H2 and CH4) from the anode side would also pass through the separator to the cathode surface, and attack the delithiated cathode to produce H2O and CO2, leading to degradation of the layered cathode crystal (Fig. 5b).37 The two crosstalk reactions above are both strongly exothermic and may be the key triggers for TR. Thus, Wang's group innovatively introduced a gas blocker (e.g., Cu foil or PTFE film) between the NCM cathode and Li metal anode, preventing gas-related crosstalk reactions, which effectively delays the onset of fire/explosion (Fig. 5c).35
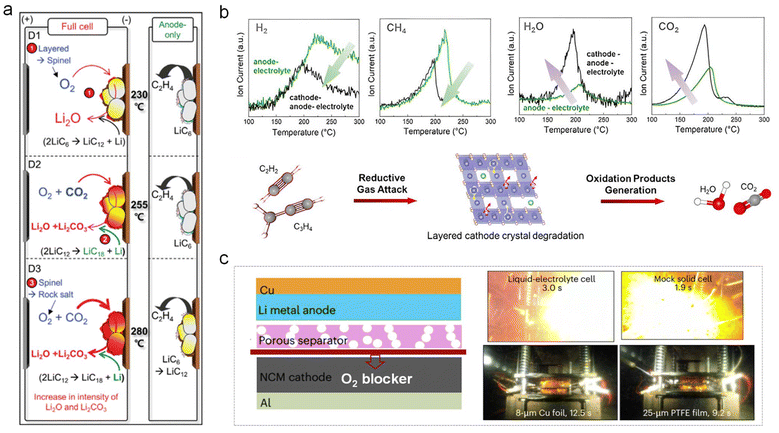 | ||
| Fig. 5 Gas crosstalk phenomenon between the delithiated cathode and the lithiated anode during the heating process. (a) The O2 released from the delithiated layered cathode would react with the LiCx anode or reductive gas. Reproduced with permission.36 Copyright 2024, Springer Nature. (b) The released reductive gas would attack the delithiated layered cathode, accompanied by the generation of oxidation products. Reproduced with permission.37 Copyright 2023, Elsevier. (c) The addition of an O2 blocker between the cathode and anode to effectively delay the onset of fire/explosion. Reproduced with permission.35 Copyright 2025, Springer Nature. | ||
4. Proposed multimodal combined characterization platform for safer material screening and prediction
On the one hand, substantial research has been conducted on the TR mechanisms of NCM cathode systems, while related studies on Li-rich cathode materials are comparatively scarce, mainly stemming from the more intricate TR reaction mechanisms associated with the latter compared to conventional layered cathodes. On the other hand, with the advances of LLO-type pre-lithiation agents, high-voltage solid-state electrolytes, and non-flammable electrolyte systems over the past decades, research into the triggers and mechanisms of the TR process has faced increasingly formidable challenges. Importantly, the root cause of TR safety hazards lies primarily in the thermal instability of battery components, particularly unstable delithiated LLO cathodes and the vulnerable cathode–electrolyte interface under high-temperature conditions—with the entire reaction accompanied by substantial evolution of gas and the generation of intense heat. Thus, we originally proposed a multimodal combined characterization techniques platform to unveil the intrinsic connections between microstructural characteristics, oxygen species, and battery safety for the screening and prediction of safer battery materials (electrodes and electrolyte) (Fig. 6, top section). Focusing on delithiated LLO cathodes with an electrolyte, this platform integrates in situ XRD, an in situ gas analyzer, DSC, and TGA, enabling the real-time collection of comprehensive data throughout the heating process. Specifically, in situ XRD characterization is employed to monitor the phase transition process, particularly the structural transformation from the layered configuration to spinel and further rocksalt phases. DSC is utilized to track the heat generation behavior of the sample. In situ gas analysis techniques are applied to monitor gassing behavior, including in situ MS for the detection of various permanent gases and in situ GC-MS for the identification/quantification of volatile organic compounds (VOCs).26 TGA is used to quantify the weight loss of the samples throughout the heating process. Thus, comprehensive information—encompassing phase transitions, gassing behavior, heat generation, and weight loss—can be obtained, while the intrinsic correlations between microstructural characteristics, thermal stability, and TR risks can be further elucidated.With the rapid development of artificial intelligence (AI), the capabilities for data acquisition and processing have also been greatly improved. Looking ahead, the integration of AI into battery safety research represents a promising avenue, and the establishment of a specialized AI laboratory has emerged as a strategic direction for pioneering next-generation battery systems with intrinsic safety. The AI laboratory consists of four technical modules (Fig. 6, bottom section). Module I: developing AI-driven robotic auto-test systems enables the continuous collection of multi-dimensional test data, including gas evolution, structural evolution, and heat generation. Module II: based on collected massive datasets, AI models (machine learning, deep learning, neural networks, and even large models) have been constructed to decode the complex correlations between material composition, microstructural characteristics (e.g., LiMn6 ring), and gassing behavior, and the further intrinsic mechanisms of gas-evolution-induced TR risks. Module III: pre-trained AI models are applied to conduct high-throughput virtual screening of candidate materials, such as LLO materials, high-voltage electrolytes, and interface modifiers, thereby rapidly optimizing material composition and structural design, overcoming the limitations of the traditional trial-and-error R&D mode, accelerating the R&D cycle of low-gas-emission, high-safety battery materials, and providing robust technical support for the commercialization of next-generation high-energy-density battery systems. Module IV: the product validation module serves as the critical final checkpoint for translating lab-scale material innovations into practical, high-safety battery products. It conducts comprehensive performance evaluations on the AI-screened optimal material systems and battery products under simulated real-world working conditions, including electrochemical testing under extreme conditions and TR trigger safety tests. This module verifies the effectiveness of the suppression of gas evolution and enhancement in thermal stability in actual battery configurations, providing targeted optimization suggestions to bridge the gap between theoretical predictions and industrial applications.
5. Conclusion and perspectives
In summary, we reviewed the current research progress of LLO-related gassing behavior during both cycling and the TR process. Evidently, gas evolution during the cycling process stems primarily from two interrelated mechanisms: an unstable OAR reaction induced by the unique LiMn6 functional units inherent in the LLO crystal structure, and the severe parasitic reactions triggered by irreversible loss of oxygen from the cathode lattice. Until now, the mainstream research focus has consistently centered on stabilizing the OAR reaction, either by optimizing a cationic doping strategy to regulate the local electronic structure of LLOs, constructing robust surface coating layers to isolate the cathode from direct contact with the electrolyte, or modifying the electrolyte composition to suppress interfacial side reactions. While these strategies have yielded notable advances in mitigating gassing issues, they still face certain practical limitations. For instance, cationic doping often involves a trade-off between structural stability and reversible capacity. More importantly, these singular strategies are often insufficient to completely eliminate the generation of reactive oxygen species from the lattice, meaning that the gassing issue—primarily oxygen release and subsequent electrolyte decomposition—remains an unresolved challenge for the practical application of LLO-based high-energy-density battery systems, especially under high-voltage operation and elevated temperature. We propose that future research should prioritize the organic combination and synergistic effects of multiple modification strategies.Furthermore, gas evolution behavior during the TR process is far more severe than that during electrochemical cycling, while the underlying gas generation mechanism is correspondingly more intricate. We propose that future research should prioritize two scientific aspects. (i) The thermal stability of delithiated LLOs: it is critical to figure out the intrinsic correlation between microstructural characteristics and thermal stability, as well as to identify the key factors governing thermal stability. On this basis, another core challenge is developing effective targeted modification strategies to elevate the onset temperature of oxygen release and reduce the amount of irreversible O-loss. (ii) The thermal stability of the cathode–electrolyte interface: it is imperative to elucidate the detailed transformation pathways of oxygen species and the specific reaction mechanisms at the cathode–electrolyte interface during the heating process, particularly the cascade reactions between oxygen species/high-valence TMs and the electrolyte. Another critical question to address is how to implement efficient countermeasures to rapidly terminate/interrupt such detrimental chain reactions, or to scavenge oxygen species in a timely way through low-exothermic pathways. In addition, with the speedy development of new technologies, gas-related safety issues are becoming more diverse and complex, such as LLO-type pre-lithiation agents (e.g., Li5FeO4),38 solid electrolytes (e.g., polymer electrolytes),39 and non-flammable electrolytes.40 Previously common modification strategies may no longer be blindly applicable. Focusing on the aforementioned key scientific questions, we innovatively proposed the development of an integrated multimodal combined characterization platform for safer material screening and prediction, including in situ XRD, an in situ gas analyzer, DSC, TGA, and other characterization techniques. The structure–activity relationships between phase transition, gas evolution, heat generation, and weight loss could be uncovered, leveraging the comprehensive data collected during the heating process. In addition, assisted by AI, it is expected that auto-test, data modelling, material screening and prediction, and production validation will be realized, accelerating the R&D cycle of low-gas-emission, high-safety battery materials (LLO cathodes and electrolytes).
Author contributions
Conceptualization: H. J. Y., H. T. Z. and J. W.; investigation: H. T. Z., J. W.; formal analysis: H. T. Z., J. W., S. Q. L., Y. L., S. Z.; writing – original draft: H. T. Z.; writing – review and editing: H. T. Z, L. Q. W. and H. J. Y.; supervision: H. J. Y.Conflicts of interest
The authors declare no conflict of interest.Data availability
No new experimental data were generated in this study and all analyzed datasets are publicly available.Acknowledgements
This work was financially supported by the National Key R&D Program of China (No. 2022YFB2404400), the National Natural Science Foundation of China (No. 92572203 and 92263206), the National Natural Science Foundation of China (No. U23A20577, 52372168, 52472181 and U24A2065, U24A20549, 52302209, 22375010), “The Youth Beijing Scholars Program” (No. PXM2021_014204_000023), and the Beijing Natural Science Foundation (No. L233004 and 2252003).References
- C. Yang, A. Singh, X. Pu, A. Mallarapu, K. Smith, M. Keyser, M. R. Haberman, H. Khani, P. Misztal, R. Spray, O. A. Ezekoye and D. P. Finegan, Nature, 2025, 645, 603–613 Search PubMed.
- W. Wang, W. Li, F. Yu, Q. Pu, J. Miao, S. Han, Y. Wu, L. Chen, Y. Shen and Y. Chen, Nat. Chem., 2026, 18, 561–572 Search PubMed.
- L. Pang, H. Li, X. Feng, Z. Zhao, C. Ouyang and Z. Peng, ACS Energy Lett., 2024, 9, 3587–3594 CrossRef.
- Y. Wang, X. Feng, W. Huang, X. He, L. Wang and M. Ouyang, Adv. Energy Mater., 2023, 13, 2203841 Search PubMed.
- Z. Jiang, C. Liu, L. Huang, S. Zhu, X. Zhang, R. Wu, T. Gong, Y. Wu, L. Guo, P. Han, J. Ma, G. Xu and G. Cui, Energy Environ. Sci., 2025, 18, 8232–8243 RSC.
- G. Xu, L. Huang, C. Lu, X. Zhou and G. Cui, Energy Storage Mater., 2020, 31, 72–86 Search PubMed.
- S. Kim, H.-S. Kim, B. Kim, Y.-J. Kim, J.-W. Jung and W.-H. Ryu, Adv. Energy Mater., 2023, 13, 2301983 Search PubMed.
- P. Liu, L. Yang, B. Xiao, H. Wang, L. Li, S. Ye, Y. Li, X. Ren, X. Ouyang, J. Hu, F. Pan, Q. Zhang and J. Liu, Adv. Funct. Mater., 2022, 32, 2208586 CrossRef.
- Kausthubharam, B. S. Vishnugopi, A. S. J. Alujjage, V. Premnath, W. S. Tang, J. A. Jeevarajan and P. P. Mukherjee, Chem. Rev., 2026, 126, 404–447 CrossRef PubMed.
- D.-H. Seo, J. Lee, A. Urban, R. Malik, S. Kang and G. Ceder, Nat. Chem., 2016, 8, 692–697 Search PubMed.
- B. Wang, Z. Zhuo, H. Li, S. Liu, S. Zhao, X. Zhang, J. Liu, D. Xiao, W. Yang and H. Yu, Adv. Mater., 2023, 35, 2207904 CrossRef.
- S. Liu, Y. Wang, D. Xiao, H. Li, T. Wu, B. Wang, G. Hu, L. Wu, Y. Wang, G. Wang, N. Zhang and H. Yu, EES Batteries, 2025, 1, 185–194 RSC.
- Y. Wu, Z. Ran, Y. Li, Y. Peng, L. Gao, D. Ren, Z. Ma, C. Xu, X. Feng, L. Wang, L. Lu, J. Chen, X. He and M. Ouyang, Joule, 2025, 102274 Search PubMed.
- B. Qiu, Y. Zhou, H. Liang, M. Zhang, K. Gu, T. Zeng, Z. Zhou, W. Wen, P. Miao, L. He, Y. Xiao, S. Burke, Z. Liu and Y. S. Meng, Nature, 2025, 640, 941–946 CrossRef.
- X.-Y. Huang, C.-Z. Zhao, W.-J. Kong, N. Yao, Z.-Y. Shuang, P. Xu, S. Sun, Y. Lu, W.-Z. Huang, J.-L. Li, L. Shen, X. Chen, J.-Q. Huang, L. A. Archer and Q. Zhang, Nature, 2025, 646, 343–350 Search PubMed.
- S. Liu, X. Zhang and H. Yu, Sci. China Mater., 2025, 69, 585–586 Search PubMed.
- K. Luo, M. R. Roberts, R. Hao, N. Guerrini, D. M. Pickup, Y.-S. Liu, K. Edström, J. Guo, A. V. Chadwick, L. C. Duda and P. G. Bruce, Nat. Chem., 2016, 8, 684–691 Search PubMed.
- H. Yang, L. Wang, Y. Li, Z. Zhuo, T. Wu, J. Liu, L. Xu, H. Du, S. Liu, L. Wu, S. Zhao, M. Tang, W. Yang and H. Yu, Proc. Natl. Acad. Sci. U. S. A., 2024, 121, e2412460121 CrossRef.
- X. Zhang, W. Zuo, S. Liu, C. Zhao, Q. Li, Y. Gao, X. Liu, D. Xiao, I. Hwang, Y. Ren, C.-J. Sun, Z. Chen, B. Wang, Y. Feng, W. Yang, G.-L. Xu, K. Amine and H. Yu, Adv. Mater., 2024, 36, 2310659 CrossRef.
- H. Zhang, J. Chen, Y. Hong, X. Wu, X. Huang, P. Dai, H. Luo, B. Zhang, Y. Qiao and S.-G. Sun, Nano Lett., 2022, 22, 9972–9981 Search PubMed.
- D. Cao, C. Tan and Y. Chen, Nat. Commun., 2022, 13, 4908 CrossRef PubMed.
- M. Song, D. Lee, J. Kim, Y. Heo, C. Nam, D. Ko and J. Lim, J. Power Sources, 2023, 560, 232699 CrossRef.
- S. Ramakrishnan, B. Park, J. Wu, W. Yang and B. D. McCloskey, J. Am. Chem. Soc., 2020, 142, 8522–8531 Search PubMed.
- Y. Zhu, Y. Chen, J. Chen, J. Yin, Z. Sun, G. Zeng, X. Wu, L. Chen, X. Yu, H. Luo, Y. Yan, H. Zhang, B. Zhang, X. Kuai, Y. Tang, J. Xu, W. Yin, Y. Qiu, Q. Zhang, Y. Qiao and S.-G. Sun, Adv. Mater., 2024, 36, 2312159 CrossRef PubMed.
- R. Jung, M. Metzger, F. Maglia, C. Stinner and H. A. Gasteiger, J. Electrochem. Soc., 2017, 164, A1361–A1377 CrossRef.
- H. Zhang, X. Wu, Z. Li, Y. Zou, J. Wang, X. Yu, J. Chen, J. Xue, B. Zhang, J.-H. Tian, Y.-h. Hong, Y. Qiao and S.-G. Sun, Adv. Energy Mater., 2024, 14, 2400397 CrossRef.
- B. Zhang, K. Zhang, X. Wu, Q. Zheng, H. Luo, H. Zhang, Y. Chen, S. Zhou, Y. Zhu, J. Yin, Y. Zou, H.-G. Liao, W. Jiao, N. Liu, Y. Qin, B.-W. Zhang, C. Shen, Y. Qiao and S.-G. Sun, Adv. Energy Mater., 2025, 15, 2404391 Search PubMed.
- Z. Cui, C. Liu, F. Wang and A. Manthiram, Nat. Energy, 2025, 10, 490–501 CrossRef.
- X. Liu, D. Ren, H. Hsu, X. Feng, G.-L. Xu, M. Zhuang, H. Gao, L. Lu, X. Han, Z. Chu, J. Li, X. He, K. Amine and M. Ouyang, Joule, 2018, 2, 2047–2064 Search PubMed.
- M. Ren, S. Jiao, Y. Zhao, L. Gan, Z. Wang, F. Wu, J. Wang, X. Yu and H. Li, Adv. Funct. Mater., 2025, e30009 Search PubMed.
- T. Wu, X. Zhang, Y. Wang, N. Zhang, H. Li, Y. Guan, D. Xiao, S. Liu and H. Yu, Adv. Funct. Mater., 2023, 33, 2210154 Search PubMed.
- Y. Huang, C. Li, K. Zhang, Y. Tang, W. Tu, Y. Tian, J. Wang, Y. Yan, Y. Chen, Y. Zou, L. Li, B. Zhang, J. Bao, C. Ding, Y. Wang, T. Qiu, X. Sun, Y. Qiao and S.-G. Sun, Angew. Chem., Int. Ed., 2025, 64, e18277 CrossRef.
- Y. Li, X. Liu, L. Wang, X. Feng, D. Ren, Y. Wu, G. Xu, L. Lu, J. Hou, W. Zhang, Y. Wang, W. Xu, Y. Ren, Z. Wang, J. Huang, X. Meng, X. Han, H. Wang, X. He, Z. Chen, K. Amine and M. Ouyang, Nano Energy, 2021, 85, 105878 CrossRef.
- H. Zhang, J. Xue, Y. Qin, J. Chen, J. Wang, X. Yu, B. Zhang, Y. Zou, Y.-h. Hong, Z. Li, Y. Qiao and S.-G. Sun, Small, 2024, 20, 2406110 CrossRef.
- C.-Y. Wang, K. Qin, S. Ge, N. Gupta, T. Sasaki and K. Aotani, Nat. Energy, 2025, 10, 1382–1390 CrossRef.
- S. Jo, S. Seo, S. K. Kang, I. Na, S. Kunze, M. Song, H. San, S. P. Woo, S. Kim, W. B. Kim and J. Lim, Adv. Mater., 2024, 36, 2402024 CrossRef.
- Y. Wang, X. Feng, Y. Peng, F. Zhang, D. Ren, X. Liu, L. Lu, Y. Nitta, L. Wang and M. Ouyang, Joule, 2022, 6, 2810–2820 CrossRef.
- Y. Zhu, R. Xu, Y. Zheng, Y. Chen, J. Yin, J. Xue, B. Zhang, L. Li, G. Zeng, H. Luo, X. Wu, K. Zhang, Z. Wu, S. Yang, S. Li, Y. Sun, D. Zhang, Y. Qiao and S.-G. Sun, Angew. Chem., Int. Ed., 2025, 64, e202502126 CrossRef PubMed.
- P. Ding, L. Wu, Z. Lin, C. Lou, M. Tang, X. Guo, H. Guo, Y. Wang and H. Yu, J. Am. Chem. Soc., 2023, 145, 1548–1556 CrossRef.
- J. Xie and Y.-C. Lu, Adv. Mater., 2024, 37, 2312451 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |