Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Recent advances and scientometric insights into alkali metal–CO2 batteries: materials, performance, and future directions
Camila Stockey Erhardt *a,
Moyra Freitas Vieira
*a,
Moyra Freitas Vieira a,
Darlan Pontina,
Chrisminder Dainb,
Felipe Dalla Vecchiaa and
Victor Hugo Jacks Mendes dos Santos
a,
Darlan Pontina,
Chrisminder Dainb,
Felipe Dalla Vecchiaa and
Victor Hugo Jacks Mendes dos Santos *a
*a
aPontifical Catholic University of Rio Grande do Sul, PUCRS, Institute of Petroleum and Natural Resources, Avenida Ipiranga, 6681, – TECNOPUC, Building 96J, 90619-900, Porto Alegre, Brazil. E-mail: camila.erhardt@pucrs.br; victor.santos@pucrs.br
bPETRONAS Research Sdn. Bhd., Jalan Ayer Hitam, 43000 Bandar Baru Bangi, Selangor, Malaysia
First published on 30th March 2026
Abstract
Alkali metal–CO2 batteries are emerging as key sustainable energy storage technologies due to their high energy density and CO2 capture capabilities. This review provides a comprehensive overview of lithium (Li), sodium (Na), and potassium (K)–CO2 batteries, emphasizing materials development, cell configurations, and electrochemical performance. Particular attention is given to cathode materials, transition metal catalysts, anode degradation, and the influence of electrolytes and separators on battery stability. Beyond technical discussions, a scientometric meta-analysis was conducted following the PRISMA 2020 methodology, analysing 206 articles from Web of Science and Scopus (2015–2025). VOSviewer was used to identify research hotspots, keyword clusters, and global collaboration networks. The results show that Li–CO2 batteries dominate the field, while Na–CO2 and K–CO2 systems have gained attention due to their cost and conductivity advantages. However, rate-limited kinetics, anode instability, and electrolyte degradation remain significant barriers to commercialization. Integrating advanced catalysts, solid-state or hybrid electrolytes, and functional separators presents promising pathways toward efficient and durable systems. This review concludes by proposing future research directions to accelerate the development of high-performance, scalable metal–CO2 batteries for energy and environmental applications.
Broader contextThe urgent global pursuit of carbon neutrality and sustainable energy storage has positioned alkali metal–CO2 batteries as a crucial technology. These systems offer the unique dual functionality of high-density energy storage and effective carbon dioxide utilization, aligning with United Nations Sustainable Development Goals for climate action and clean energy. This work provides a critical synthesis of the recent advancements in lithium, sodium, and potassium–CO2 batteries, integrating a rigorous scientometric analysis of over 200 studies to map the evolving research landscape. By identifying key technological bottlenecks, such as sluggish reaction kinetics, electrolyte degradation, and anode instability, this study highlights the strategic importance of advanced materials like MXenes, doped carbons, and hybrid electrolytes in overcoming current performance limits. This review serves as a roadmap for the scientific community, pinpointing emerging trends and research gaps that must be addressed to transition from fundamental lab-scale experiments to scalable, commercially viable energy storage solutions. Ultimately, this work contributes to the development of next-generation batteries that not only power our future but also actively participate in global carbon mitigation efforts. |
Introduction
The growing global demand for sustainable energy storage systems, coupled with the urgent need to mitigate climate change through the reduction of atmospheric CO2 levels, has intensified interest in metal–CO2 batteries.1,2 In alignment with the United Nations Sustainable Development Goals (SDGs), metal–CO2 battery technologies directly support several key global objectives. Specifically, SDG 7 (Affordable and Clean Energy) advocates for the expansion of reliable and modern energy systems; SDG 12 (Responsible Consumption and Production) promotes the use of sustainable materials and the development of circular technologies; and SDG 13 (Climate Action) calls for urgent efforts to combat climate change and its impact.3 By enabling both energy storage and CO2 utilization, metal–CO2 batteries offer a unique technological pathway. Their dual functionality makes them particularly attractive as next-generation technologies capable of addressing two of the most pressing challenges of our time: clean energy and carbon neutrality.4 These systems are being developed with an emphasis on material abundance, lower environmental impact, and decarbonization potential, positioning them as innovative tools for a more sustainable and resilient energy future.Among the various approaches under investigation, metal–CO2 batteries have gained attention due to their high theoretical energy densities, tunable redox chemistries, environmental relevance, and the potential to significantly reduce the weight and size of future battery systems. These batteries utilize metals as anodes and CO2 as a cathodic reactant, resulting in the electrochemical conversion of CO2 into solid products. While lithium (Li) has been the most extensively studied, alternative metals such as sodium (Na) and potassium (K) have also gained increasing attention due to their potential.5–12 Each metal offers distinct advantages in terms of abundance, cost, safety, redox potential, and reaction reversibility, opening new avenues for tuning the battery's performance and sustainability profile. At the same time, each metal–CO2 system presents its own specific challenges and corresponding technological development pathways that must be addressed to enable practical implementation.
Li–CO2 batteries, in particular, have drawn significant attention due to their high theoretical energy density (∼1876 Wh kg−1) and ability to capture and convert CO2 into lithium carbonate (Li2CO3) and carbon.13,14 These properties position Li–CO2 batteries as attractive alternatives to conventional Li-ion systems, especially for applications demanding higher energy capacities such as electric vehicles, aerospace, and grid-scale storage.15,16 In parallel, Na–CO2 and K–CO2 batteries are increasingly being investigated as cost-effective and safer alternatives, owing to natural abundance and reduced reactivity of sodium and potassium compared to those of lithium. While their energy densities are lower (∼1.10 Wh kg−1 for Na and ∼1.0 Wh kg−1 for K systems), their enhanced ion transport properties and potential for high-rate operation make them attractive candidates for specific applications and further exploration in metal–CO2 battery research.17,18
Given the expanding interest and diversification of materials and architectures, a systematic review is both timely and essential to summarize the rapidly growing body of knowledge on metal–CO2 batteries, identify dominant configurations, and highlight key performance trends. In parallel, a scientometric meta-analysis can provide quantitative insights into the evolution of the field, emphasizing influential contributors, emerging themes, and existing research gaps. Accordingly, this review has three overarching goals: (i) to provide a comprehensive technical assessment of the current state of alkali metal–CO2 batteries by systematically evaluating cell architectures, electrode and electrolyte materials, and reaction mechanisms, with special emphasis on critical limitations such as poor reversibility and metal anode degradation; (ii) to conduct a rigorous scientometric analysis of the literature published between 2015 and 2025 following PRISMA 2020 guidelines, employing tools such as VOSviewer to map thematic clusters, leading institutions, and global research collaborations; and (iii) to propose a forward-looking perspective to guide future developments in efficient, stable, and scalable metal–CO2 batteries that contribute simultaneously to sustainable energy storage and carbon mitigation. The present review focuses exclusively on alkali metal–CO2 batteries, leaving out other systems such as Zn–CO2, and restricts its scope to peer-reviewed scientific articles. Patents were not included at this stage, given the overall early maturity level of the technology.
The paper is organised as follows: section 2 examines fundamental operating principles, section 3 discusses methodology and scientometric results, section 4 analyses cell components and materials, section 5 discusses current challenges and future opportunities, and section 6 presents concluding remarks. The final aim is to shed light on potential development priorities and to enable the advancement of the technology's TRL by addressing, in a multifactorial manner, the various aspects required to develop and improve scalable and economically viable battery technologies, while also understanding the application context of future products. Through this structured analysis, we seek to accelerate progress toward sustainable energy storage solutions that simultaneously address energy and environmental challenges.
Fundamentals of alkali metal–CO2 batteries
Basic operating principles
Alkali metal–CO2 batteries operate based on the redox reactions between alkali metal anodes (Li, Na, and K) and CO2 at the cathode. These systems offer high theoretical energy densities and the potential for CO2 capture, making them effective systems to store energy while reducing greenhouse gas emissions. The general operational mechanism involves the oxidation of the alkali metal at the anode, releasing electrons to the external circuit, and the reduction of CO2 at the cathode, leading to the formation of metal–carbon products such as metal carbonates, reduced solid carbon or oxalates, depending on the alkali metal and electrolyte used.19,20The electrolyte facilitates the movement of ions between the anode and cathode. Despite growing research efforts, the mechanisms of the CO2 Reduction Reaction (CO2RR) and CO2 Electroreduction Reaction (CO2ER) remain debated due to the system's inherent complexity. For instance, reported discharge products vary widely, from Li2CO3 and Li2C2O4 to carbon and carbon monoxide, raising questions about their origin. It remains unclear whether these products stem from the direct CO2RR or electrolyte decomposition.21
The energy release is determined by the enthalpy of the CO2 reduction reaction, and the overall battery performance is dependent on various factors such as the choice of electrolyte, electrode materials, and reaction kinetics.
Alkali metals and their specificities
Each alkali metal provides unique electrochemical properties and reaction pathways that affect battery performance (Table 1):• Lithium is the most widely studied alkali metal in CO2 batteries due to its high theoretical capacity (∼3.860 mAh g−1) and corresponding energy density (∼1.876 Wh kg−1).22 These exceptional values make Li–CO2 systems highly attractive for high-energy applications. Consequently, research on Li–CO2 batteries is the most mature, including the development and testing of diverse battery systems (e.g., solid-state and flexible designs), various electrolytes (e.g., non-aqueous liquid, gel, and solid), and numerous advanced catalysts and cathode materials.
• Sodium is more abundant and cost-effective than lithium, making it an attractive alternative and highly promising for large-scale, stationary energy storage where material costs are a major factor.23 While its theoretical specific capacity (∼1.160 mAh g−1) and energy density (∼1.10 Wh kg−1) are lower than those of Li–CO2, Na–CO2 systems offer potential for sustainable and economical battery production.
• Potassium batteries are a more recent addition to the field. Based on their redox potential, K–CO2 systems exhibit a favourable theoretical specific capacity of approximately 687 mA h g−1 and an energy density of around 1.0 Wh kg−1. Despite the lower theoretical metrics compared to those of Li and Na, K offers unique kinetic advantages. Specifically, potassium offers superior ionic conductivity due to its low Stokes radius, resulting in rapid charge transport and enhanced rate capabilities. K–CO2 systems also benefit from lower charge–discharge overpotentials in some reports.
| Metal | Standard potential (V vs. SHE) | Theoretical capacity (mAh g−1) | Energy density (Wh g−1) | Electrochemical performance and specific descriptors |
|---|---|---|---|---|
| Lithium (Li) | −3.04 V | 3.860 | ∼1.876 | High theoretical voltage (2.80 V vs. Li/Li+); suffers from severe charge overpotentials (ηch > 1.0 V) due to the insulating nature of Li2CO3 and C products. |
| Sodium (Na) | −2.71 V | 1.160 | ∼1.13 | Theoretical cell voltage: 2.35 V; typically shows lower discharge overpotentials than Li but faces stability issues with superoxide intermediates (NaO2) in certain electrolytes. |
| Potassium (K) | −2.93 V | 687 | ∼1.0 | Exhibits higher exchange current density (i0) compared to Li/Na systems; demonstrates a narrower voltage gap (typically ΔV < 1.2 V at low current densities) due to more reversible K2CO3 formation. |
Additionally, since K+ has a weaker Lewis acidity than Li+ and Na+, it possesses a faster rate of migration between the electrolyte and electrode, leading to a better rate performance of the battery. The selection of alkali metal significantly impacts the battery's configuration, discharge products, and overall electrochemical behaviour.26
Main challenges
Currently, there are several challenges impeding the practical application of high-performance metal–CO2 batteries, such as:• Anode instability: Alkali metal anodes are prone to dendrite formation, which can lead to short-circuiting and reduced cycle life. The metal also undergoes volume expansion during cycling, which may cause mechanical stress and deterioration of the electrode.27
• Sluggish CO2RR/CO2ER kinetics and low CO2 conversion: The kinetic limitations of the CO2RR and CO2ER are a major challenge. These sluggish reactions occur at the cathode during discharge and charge, respectively, and involve the deposition and decomposition of insulating discharge products (e.g., metal carbonates or oxalates).22,28 This slow decomposition often leads to accumulation of residual discharge products, which passivate the cathode surface and diminish battery capacity over time. Consequently, the overall energy efficiency and cyclability of the battery are significantly compromised. Enhancing the kinetics of the CO2RR and CO2ER requires the development of high-surface-area, porous cathode structures and catalytic materials capable of promoting product reversibility and facilitating charge transfer.29,30
• Electrolyte degradation: The electrolyte plays a critical role in mediating ion transport and maintaining interfacial stability. However, in metal–CO2 systems, electrolytes are exposed to highly reactive alkali metals and intermediate species such as superoxide and carbonate radicals. These species can trigger electrolyte decomposition, especially under high voltage conditions and during high recharge overpotentials, leading to capacity fade, gas evolution, and poor cycling stability. Moreover, the electrochemical stability window of the electrolyte must be wide enough to support both CO2 reduction and product oxidation.27
• CO2 diffusion issues: The diffusion of CO2 in non-aqueous electrolytes is generally slow, limiting the rate at which CO2 can participate in the reduction reaction and reducing the overall charge/discharge rates. Furthermore, gas bubble formation during cycling can block active sites, further impeding mass transport.31
• Side reactions: Several side reactions, including the formation of unwanted by-products, can occur during cycling. Such reactions not only consume active materials but also contaminate the electrode surfaces, interfere with redox pathways, and alter the selectivity of the desired products, often taking place through irreversible reaction pathways, therefore, compromising the coulombic efficiency and lifespan of the battery.28,32
Despite these challenges, ongoing research aims to optimize the performance of alkali metal–CO2 batteries by improving electrolyte formulations, stabilizing anode materials, and enhancing the CO2 reduction process. These improvements could lead to more efficient and cost-effective systems for energy storage and CO2 sequestration.
Scientometric methodology
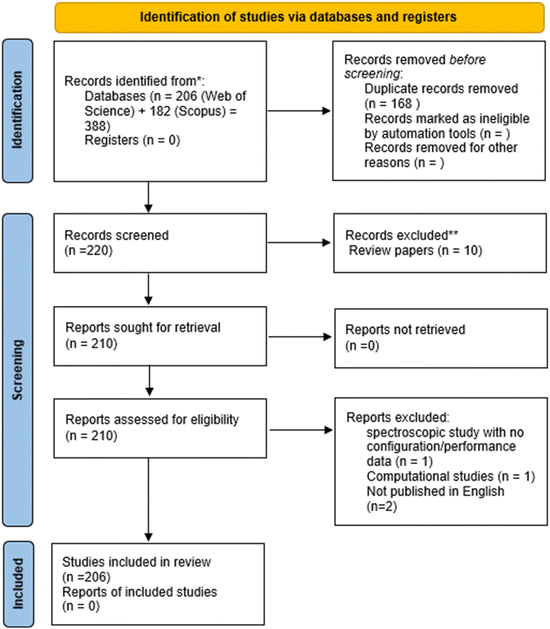
The scientometric analysis in this review is based on publications retrieved from Web of Science and Scopus. The search strategy was structured across three primary axes to ensure thematic depth: (i) battery chemistry, (ii) components, and (iii) performance metrics. The systematic retrieval employed the following parameters: (“Li–CO2 batter*” OR “Na–CO2 batter*” OR “K–CO2 batter*” OR “alkali metal–CO2 batter*”) AND (“cathode” OR “anode” OR “electrolyte” OR “stability” OR “catalyst”) AND (“cycling performance” OR “energy density” OR “power density” OR “coulombic efficiency” OR “electrochemical performance” OR “cycle life” OR “capacity retention” OR “overpotential” OR “discharge capacity”).This integrated approach was deliberately designed to prioritize studies that provide robust and comparable electrochemical data. To ensure a systematic and transparent selection, the PRISMA guidelines were employed. Fig. 1 presents the PRISMA flowchart documenting the literature screening and selection process. The initial search yielded 388 records, which were subsequently filtered through duplicate removal, title/abstract screening, and full-text evaluation.
In total, 206 studies met the predefined inclusion criteria. While this rigorous filtering may exclude purely fundamental or mechanistic studies that do not explicitly report the specified performance metrics in their metadata, it ensures a cohesive and representative dataset for analysing the current state-of-the-art in alkali metal–CO2 battery technology.
This standardised protocol ensures the reproducibility of the results and maintains a high level of methodological consistency throughout the evaluation of developmental milestones within the 2015–2025 period.
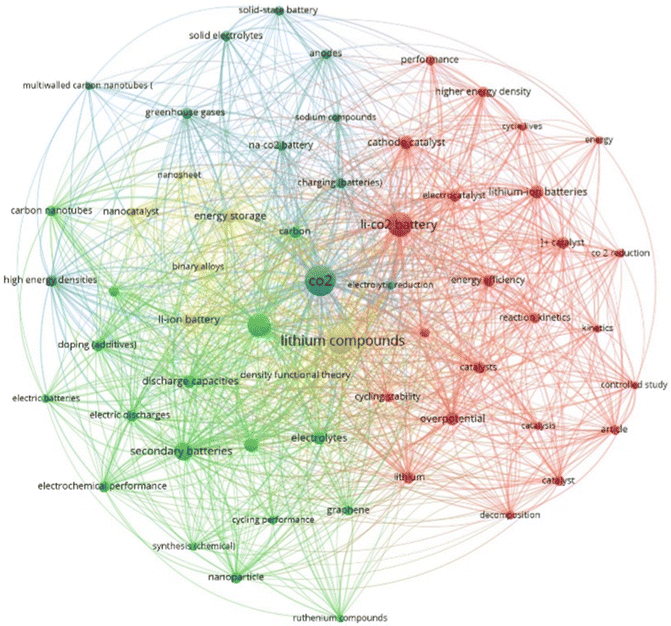
Using VOSviewer (1.6.20), the final dataset was processed to extract bibliometric data on keyword co-occurrence, co-authorship networks, and international collaborations. A total of 1581 keywords were identified in the research. By applying a minimum threshold of 10 occurrences, a keyword co-occurrence map was generated, as shown in Fig. 2. In this map, the size of each circle represents the frequency of the corresponding keyword in the analysed articles—the larger the circle, the higher its incidence. Notably, the central and most prominent keyword is “CO2”, both in terms of position and size, indicating its dominant presence across the dataset.
Surrounding it, three other significant and centrally located nodes are observed: “lithium compounds”, “cathodes”, and “Li–CO2 batteries”. Additionally, the thickness of the lines connecting the nodes represents the strength of their co-occurrence—how frequently they appear together in the same articles. The colours indicate clusters of keywords that are thematically related. The analysis revealed four distinct clusters, with a total of 1327 links between the keywords. The first cluster is centred around “Li–CO2 batteries”, which is primarily connected to “catalyst”, and includes a total of 94 keyword occurrences.
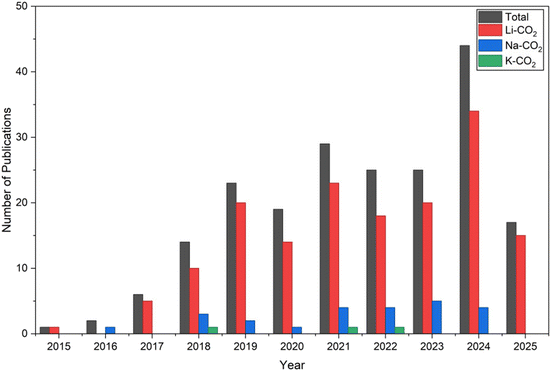
The second cluster is dominated by the keyword “cathodes”, with 81 occurrences, and shows strong association with “carbon” and “carbon nanotubes”. The third cluster is the largest, featuring “CO2” as the most frequently occurring keyword across the dataset, appearing in 145 articles. Finally, the fourth cluster revolves around “lithium compounds”, which appears 111 times and comprises 7 related keywords. Based on this map, it is evident that the vast majority of articles focus on Li–CO2 batteries. Na–CO2 batteries appear within the largest cluster—centred on “CO2”—while K–CO2 batteries are not represented in the map at all. Among the articles in the database used for this analysis, only three focused on K–CO2 batteries, which explains their absence from the co-occurrence map.11,33,34 Fig. 3 illustrates the annual number of scientific publications related to CO2 batteries, categorized into total publications and those specifically focused on Li–CO2, Na–CO2, and K–CO2 systems.
From the data, it is evident that Li–CO2 batteries dominate the research landscape, with a consistently increasing number of studies over the years. This strong upward trend reflects the scientific community's growing interest in lithium-based CO2 batteries, likely due to their high theoretical energy density and potential applications in next-generation energy storage systems.
In contrast, Na–CO2 batteries appear much less frequently, with only a modest number of publications each year. Despite this, their presence in the literature suggests a niche but emerging field, motivated by the abundance and low cost of sodium. K–CO2 batteries are nearly absent, showing minimal representation in the database. This indicates that potassium-based systems remain largely unexplored or are still in their very early stages of investigation.
When comparing with the total number of articles published each year, it is clear that Li–CO2 research contributes a substantial portion of the CO2 battery literature. The overall publication count also reveals the growing attention the field of CO2 electrochemistry has received over the past decade.
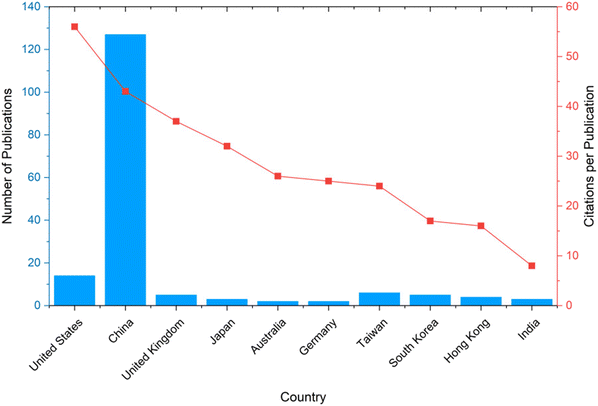
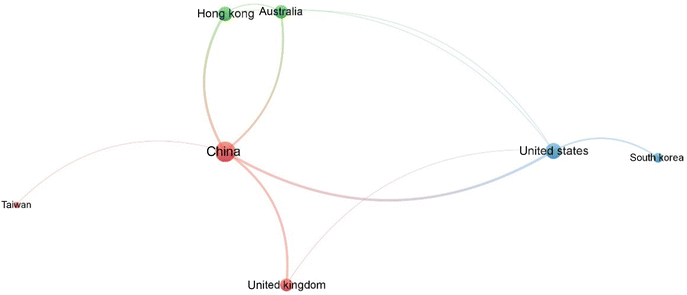
Fig. 4 presents the average number of citations per publication for the ten countries with the highest number of scientific articles on CO2 batteries. At the top of the list is the United States, with an impressive 57.07 citations per publication, indicating that although it has fewer publications than China (14 vs. 127), its research tends to be highly influential. Following closely is China, the most prolific in terms of publication volume, with 127 papers and an average of 44.50 citations per article, showing a strong balance between quantity and impact.
The United Kingdom ranks third, with 38.6 citations per article from just 5 publications, suggesting a highly selective yet impactful contribution. Japan, Australia, and Germany similarly appear in the list with high impact scores despite having only 2 publications each, emphasizing the significance of quality over volume. In contrast, Taiwan and South Korea show moderate citation averages, despite having a slightly higher number of publications compared to those from Japan, Australia, and Germany. Hong Kong also appears with 4 publications and an average of 17.75 citations, while India completes the list with 9 citations per publication from 3 studies. This analysis highlights that a high number of publications does not always equate to higher impact per article. Countries like the UK, Japan, and Australia demonstrate that even with a smaller volume of work, it is possible to achieve strong recognition and influence in the scientific community. The data also indicate growing interest from the Asian market in this research area, particularly in Japan, China, Taiwan, South Korea, Hong Kong, and India.
The most cited article in the CO2 battery research field is titled “The first introduction of graphene to rechargeable Li–CO2 batteries: a highly efficient catalyst for Li2CO3 decomposition”.35 With 343 citations, this work is highly influential because it was the first to explore the use of graphene as a catalyst in Li–CO2 batteries, addressing a critical limitation: the poor decomposition of lithium carbonate during charging. By improving the reversibility of the system, the study opened new paths for developing rechargeable Li–CO2 batteries.
Its pioneering approach, strong experimental validation, and relevance across materials science and energy storage fields have contributed to its widespread recognition within the field.
At the international level (Fig. 5), China has emerged as the most prolific and interconnected country in this domain, maintaining strong collaborative ties with countries such as the United States, South Korea, Japan, and the United Kingdom. The United States positions itself as the second most connected country. This reflects the collaborative nature of energy storage research, where partnerships between leading scientific nations are essential for rapid technological advancements. Interestingly, Taiwan and Hong Kong appear as regional hubs closely connected to China, which likely reflects the shared linguistic and cultural ties, as well as similar research priorities. While nations like France, Greece, and Poland appear less central in the network, they still contribute meaningfully, particularly in niche areas like solid-state electrolytes and advanced characterisation techniques.
Together, these scientometric insights reveal a dynamic and rapidly evolving research landscape. While lithium remains the dominant chemistry explored in metal–CO2 batteries, the increasing presence of sodium- and potassium-based systems, innovative materials, and transnational collaborations point toward a maturing field with expanding horizons. This comprehensive mapping of the literature provides a valuable guide for researchers and funding agencies seeking to identify hotspots, research gaps, and strategic opportunities for advancing dual-purpose energy storage and CO2 conversion technologies.
Cell architectures and materials
Understanding the structural and compositional aspects of alkali metal–CO2 batteries is crucial to optimising their performance, stability, and reversibility. The overall architecture of a battery (Fig. 6) – including the cathode, anode, electrolyte, and separator – governs its energy density, reaction kinetics, and long-term cycling behaviour.In recent years, significant advances have been made in the development of multifunctional materials and innovative cell designs tailored to overcome the intrinsic limitations of metal–CO2 systems, such as insulating discharge products, dendrite formation, and electrolyte degradation. This section provides a comprehensive overview of the main components that define the electrochemical configuration of Li, Na, and K–CO2 batteries, highlighting recent trends, material choices, fabrication strategies, and their impact on device performance.
Cathodes of alkali metal–CO2 batteries
In metal–CO2 batteries, the reduction of CO2 at the cathode during discharge is a crucial process, playing an important role in the battery's overall operation.32 The choice of cathode material and the type of electrolyte system (aprotic or aqueous) used directly influence the selectivity and efficiency of this reaction, which may result in the formation of different discharge products such as CO, HCOOH, and other hydrocarbons. Significant research efforts focus on optimising the cathode through various modifications, including advanced synthesis techniques, doping strategies, defect engineering, and surface treatments of the precursor materials.36–38These approaches aim to enhance catalytic activity, improve CO2 adsorption, and ensure greater stability and efficiency of the cathode, thus advancing the performance and practical viability of metal–CO2 batteries. Based on the bibliometric analysis, a diverse range of materials are commonly employed for cathode fabrication due to their favourable properties. Among these, carbon-based materials are widely utilised, including carbon cloth,39 porous carbon,19 carbon nanotube (CNT)-based structures,7,40,41 graphene,42–44 and functionalised variants such as doped graphene45 and carbon nanotubes.46 These materials are favoured for their high electrical conductivity, large surface area, and ability to improve the electrochemical reactions involved in CO2 reduction and product decomposition.
Carbon cloth, which typically ranges from 0.08 to 0.15 mm in thickness, is a preferred material for its high electrical conductivity, which is crucial for efficient electron transfer during electrochemical reactions. Its three-dimensional porous structure offers an extensive surface area,47 promoting high CO2 adsorption and effectively accommodating solid discharge products, such as carbonates. These features not only improve the efficiency of electrochemical reactions but also significantly enhance the energy density of the battery, making carbon cloth a key component in the development of high-performance cathodes.
Porous carbon is also widely used as a cathode material due to its high surface area, three-dimensional structure, and gas adsorption capacity, particularly for CO2. Its unique properties significantly enhance energy density by providing multiple active sites for electrochemical reactions and facilitating the efficient diffusion of CO2 and electrolytes. Various types of porous carbon, including activated carbon, carbon aerogels,48 and carbon nanotubes,47,49,50 can be functionalised or combined with catalysts to further improve performance. For example, Deng et al. developed a novel metal-free cathode based on nitrogen (N), phosphorus (P), and fluorine (F) co-doped carbon nanotube networks (NPF-CNTs).51 These materials were synthesised using an ammonium hexafluorophosphate (NH4PF6)-assisted method, resulting in a three-dimensional interconnected structure that enhances charge transfer and increases the accessibility of active sites. Heteroatom doping plays a crucial role in improving catalytic activity, facilitating the efficient and reversible decomposition of Li2CO3, and enhancing the performance of CO2 reduction and evolution reactions. Additionally, the porous structure of the nanotubes promotes efficient reagent diffusion and contributes to the structural stability of the cathode throughout the cycling process. These findings indicate that NPF-CNTs represent a promising alternative for the development of high-performance and long-lasting Li–CO2 batteries. The porous structure of carbon is particularly important in preventing cathode blockage caused by solid reaction products (e.g., carbonates), thereby ensuring prolonged cycling stability. Additionally, porous carbon is lightweight, chemically stable, and exhibits high electrical conductivity—key characteristics for advanced battery applications. Research on pore size engineering and distribution remains an active field, aiming to optimize the interaction between porous carbon and electrochemical reactants to maximize battery efficiency and durability. In addition to carbon-based materials, MXenes,49,50,52 which are two-dimensional transition metal carbides and nitrides, and molybdenum nitride (Mo2N)53,54 also stand out as promising cathodes due to their excellent conductivity, high ion storage capacity, and tunable surface chemistry, significantly improving CO2 reaction kinetics.
Composites comprising carbonaceous materials and transition metal-based compounds can serve as effective catalysts for the CO2 reduction reaction (CO2RR). The catalysts employed in the cathodes of metal–CO2 batteries are fundamental in enhancing the kinetics of electrochemical reactions, facilitating the efficient conversion of CO2 into desired products, such as carbonates,17 while simultaneously helping to reduce the recharge overpotential and supporting the development of rechargeable battery systems through reversible chemical pathways.
These catalysts are typically based on metallic materials, including metal oxides (e.g., MnO2,55 Co3O4,56 and Fe3O4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 57), noble metals (e.g., Pt,58 Au,59 and Ru60), and bimetallic compounds or alloys, known for their high catalytic activity and selectivity. A representative example is a cathode composed of Ru/RuO2 supported on high-porosity carbon (HPC) or Ketjenblack combined with ruthenium (KB/Ru), which integrates the high electrical conductivity of carbon with the catalytic functionality of transition metals. Such systems are engineered to accelerate CO2 conversion and promote the decomposition of metal carbonates (M2CO3, where M = Li, Na, or K), thereby improving cycle efficiency and overall reversibility.
57), noble metals (e.g., Pt,58 Au,59 and Ru60), and bimetallic compounds or alloys, known for their high catalytic activity and selectivity. A representative example is a cathode composed of Ru/RuO2 supported on high-porosity carbon (HPC) or Ketjenblack combined with ruthenium (KB/Ru), which integrates the high electrical conductivity of carbon with the catalytic functionality of transition metals. Such systems are engineered to accelerate CO2 conversion and promote the decomposition of metal carbonates (M2CO3, where M = Li, Na, or K), thereby improving cycle efficiency and overall reversibility.
However, a significant challenge in cathode design is that aggressive catalysts can exacerbate side reactions, such as electrolyte decomposition and electrode oxidation. Thus, tailoring the electronic structure of these catalysts offers a pathway to stabilize more efficiently the discharge products. Recent studies have demonstrated the potential of producing Li2C2O4 as the discharge product, which theoretically offers faster kinetics and a lower charging voltage (<3.3 V) due to a two-electron process (2Li+ + 2CO2 + 2e− ↔ Li2C2O4) compared to the more complex four-electron oxidation of the C/Li2CO3 system.61
The selection between these pathways is governed by orbital interactions at the catalyst interface. Specifically, the overlap between the metal d-orbitals and the CO2π* orbitals determines the adsorption geometry; a strong C–C coupling is required to favour oxalate formation. Furthermore, the dynamic surface reconstruction of transition metal cathodes during cycling plays a dual role: while it may create active sites that lower redox overpotentials, it can also accelerate the disproportionation of Li2C2O4 into the more thermodynamically stable Li2CO3 and C. Therefore, modulating the surface electronic states remains a significant challenge to achieving high energy efficiency and stability.62
Shifting from traditional strategies focused solely on the d-band centre, engineering d–p hybridization, specifically between Mn 3d and C 2p orbitals, promotes stronger coupling. This optimises both CO2 adsorption and Li2CO3 decomposition kinetics. Nevertheless, the systematic modulation of these hybridization states remains a scientific challenge yet to be fully explored.63 Similarly, distinct catalytic sites within the same material can exhibit varying catalytic activities for the CORR and COER.
Specifically, Mn(II) sites exhibit preferential adsorption of CO2, whereas Mn(III) sites demonstrate a preference for Li2CO3 adsorption.64 Moreover, recent studies have shown that the use of electrocatalytic materials, such as manganese-based transition metal oxides (AMn2O4, where A = Ni, Zn, Co), significantly enhances reaction kinetics and the cycling stability of metal–CO2 batteries. These nanostructured materials provide a high surface area and abundant active sites, facilitating the efficient decomposition of discharge products, such as metal carbonates (M2CO3, where M = Li, Na, or K), while ensuring effective diffusion of reactants within the cathode.
The integration of metallic catalysts with porous supports, such as carbon cloth or activated carbon, enhances CO2 adsorption, reduces reaction overpotentials, and improves the overall reversibility of the system. The strategic selection and design of catalysts are therefore critical to achieving enhanced efficiency, stability, and cycling performance in metal–CO2 battery systems.32,58
Despite these common choices, there is a wide range of variations in how the cathodes are prepared. This includes differences in fabrication techniques, such as slurry coating, in situ growth, and direct assembly on conductive substrates, as well as in the incorporation of catalysts like ruthenium (Ru),99,107,134 cobalt (Co),102,148 or nickel (Ni),129,149 which enhance the kinetics of the redox reactions. Additionally, many studies explore the functionalization of the active materials to improve CO2 adsorption, reaction selectivity, and long-term cycling stability.
In the studies on K–CO2 batteries, the active materials used in the cathode included carbon nanotubes (CNTs33 and MWCNTs11), and graphite.34 On Na–CO2 batteries, the majority employ CNTs or MWCNTs as the active material in the cathode, using carbon paper and nickel foam as substrates. Additionally, a significant number of works make use of ruthenium (Ru) as a catalyst.34,135,140
Overall, the choice of cathode is a critical factor in the performance of Li–CO2 and Na–CO2 batteries, directly influencing energy density, cycling stability, and CO2 conversion efficiency. Continuous optimization of these materials, including the incorporation of metallic catalysts and nanoporous structures, is essential to advancing the commercial viability of these technologies.
Table 2 summarizes the annual distribution of published articles related to metal–CO2 batteries that employed carbon nanotubes (CNTs) or ruthenium-based (Ru) materials as cathode components between 2015 and 2025. The use of CNTs began modestly in 2016, with only one article, but experienced a sharp rise in subsequent years, peaking in 2023 with 13 publications. The highest total number of CNT-related studies occurred in 2023 and 2024, highlighting a continued focus on nanocarbon-based cathodes. Ru-based materials, known for their high catalytic efficiency in CO2 reduction and evolution reactions, appeared consistently from 2016 onward. A notable surge occurred in 2022, with 11 articles referencing the use of Ru, possibly driven by advancements in hybrid systems combining Ru with porous carbons or conductive frameworks. Studies on Ru-based catalysts are also directly associated with the early development of rechargeable metal–CO2 batteries—particularly Li–CO2 systems—and with the emergence of strategies aimed at enabling long-term cyclability and improving the stability of this new class of batteries.
| Year | Articles (no) | CNTa (no) | Ref. | Rua (no) | Ref. |
|---|---|---|---|---|---|
| a Indicates that different forms or variants of the material are included. | |||||
| 2015 | 1 | 0 | — | 0 | — |
| 2016 | 2 | 1 | 65 | 1 | 66 |
| 2017 | 6 | 3 | 67–69 | 2 | 70 and 71 |
| 2018 | 13 | 6 | 72–77 | 2 | 75 and 78 |
| 2019 | 22 | 8 | 60 and 79–85 | 7 | 60, 81, 83 and 86–89 |
| 2020 | 19 | 4 | 90–93 | 5 | 94–98 |
| 2021 | 28 | 11 | 7, 11, 12, 59 and 99–104 | 8 | 12, 99 and 104–108 |
| 2022 | 23 | 3 | 33, 109 and 110 | 11 | 48, 109 and 111–119 |
| 2023 | 28 | 13 | 43 and 120–130 | 7 | 29, 122, 124, 126, 127, 131 and 132 |
| 2024 | 43 | 10 | 40, 46 and 133–139 | 6 | 134, 135 and 140–143 |
| 2025 | 17 | 6 | 41, 51, 58 and 144–146 | 1 | 147 |
The overlap in references suggests that some studies explored synergistic effects between CNTs and Ru-based catalysts.
This combination is of particular interest because CNTs can serve as conductive scaffolds that support dispersed Ru nanoparticles, thereby enhancing catalytic efficiency while minimizing noble metal usage. Overall, the data illustrate an evolving research landscape in which CNTs maintain a strong and growing presence, while Ru-based catalysts continue to be explored—though with some fluctuation—as high-performance yet resource-intensive components in metal–CO2 battery systems.
Anodes of alkali metal–CO2 batteries
In alkali–CO2 batteries, the anode is typically made of a metal foil, such as lithium, sodium, or potassium. The choice of anode material is critical, as it directly influences the battery's electrochemical performance, including its operating voltage and practical capacity.150 Each anode material has unique physical and chemical properties that set an upper theoretical limit on these characteristics. For instance, lithium is the most widely used anode material due to its low standard hydrogen potential (−3.04 V) and exceptionally high theoretical capacity of 3860 mAh g−1, making it ideal for high-performance batteries.69 However, the selection of the anode material not only dictates the cell's performance but also affects the charge and discharge processes. The chemical interactions between the anode and other electrochemically active components, such as CO2, vary significantly depending on the anode material. Even in systems with similar electrochemistry, such as lithium and sodium, distinct differences in electrochemical behaviour have been observed.150 This variability underscores the importance of carefully selecting the anode material to optimise battery performance for specific applications. One of the major challenges in advancing alkali–CO2 battery technology is the degradation of metal anodes, which significantly impacts both performance and long-term stability.11,69,150Alkali metals are highly reactive, making them susceptible to corrosion, pulverization, and dendrite formation during cycling. These issues lead to the formation of insulating oxide layers and the accumulation of inactive metal, which hinder ion transport and disrupt electrochemical processes. Additionally, the metal anode reacts with the electrolyte or dissolved gases, forming a solid electrolyte interphase (SEI) layer.
While the SEI layer is essential for stabilising the anode, its formation increases battery polarization and limits cycle life.11 Dendrite growth, in particular, poses a serious risk, as it can pierce the separator and cause short circuits, further degrading the cell's performance.151 These degradation mechanisms are exacerbated in the presence of CO2, which reacts with the metal anode to form by-products that accelerate deterioration. To address these challenges, strategies such as protective coatings, alternative anode materials, and electrolyte engineering have been developed.11,84,105,152–154 These approaches aim to enhance anode stability, minimize side reactions, and mitigate degradation, ultimately improving cycling performance and extending the battery lifespan. Such advancements are critical for the development of more durable alkali–CO2 battery systems. To mitigate anode degradation, various protective strategies have been employed to shield the anode from direct contact with the electrolyte and CO2. Protective layers, composed of carbon-based materials, metal alloys, or polymers, have proven effective in preventing corrosion and the formation of harmful by-products.
For example, Qi and co-workers demonstrated that depositing a Li3N layer on a lithium metal anode using atomic layer deposition (referred to as Li@Li3N) significantly enhanced the service life of Li–CO2 batteries.155 The Li3N layer not only protected the lithium anode from CO2-induced corrosion but also improved lithium-ion transport due to the material's high ionic conductivity. In symmetric cell tests, the Li@Li3N anode exhibited stable plating/stripping behaviour for over 900 hours at 1 mA cm−1, outperforming pure lithium, which lasted only about 500 hours (Fig. 7a). Moreover, the discharge–charge profiles for the Li@Li3N anode remained consistent, indicating that the protective layer did not compromise electrochemical performance.
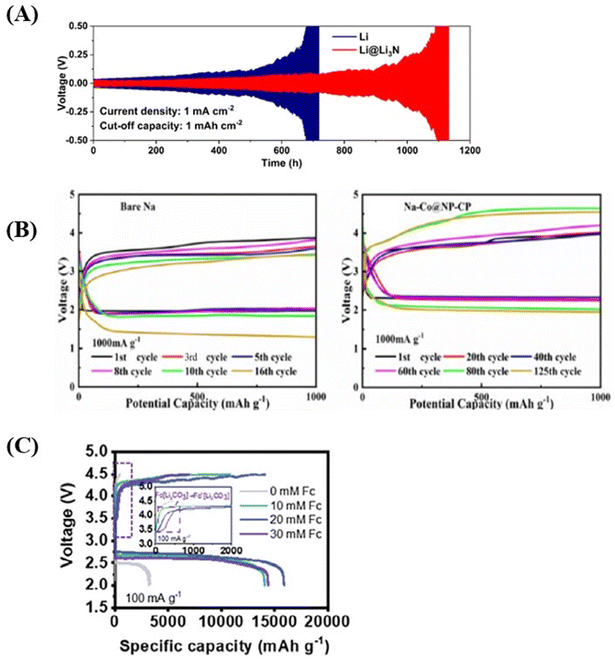 | ||
| Fig. 7 (A) Comparison between symmetric cells using pure Li and interface-protected Li (Li@Li3N) at a current density of 1 mA cm−2. Reproduced with permission.150 Copyright 2024, Energy Storage Materials. (B) Voltage profiles of the Na–CO2 batteries using a bare Na anode and a Na–Co@NP-CP anode under a limited capacity of 1000 mAh g−1 at a current density of 1000 mA g−1. Reproduced with permission.140 Copyright 2024, RSC Advances. (C) Comparison of discharge–charge curves of electrolytes with different FC concentrations at 100 mA g−1 in 2.0–4.5 V. Reproduced with permission.11 Copyright 2024, Advanced Functional Materials. | ||
Similarly, Liu and co-workers developed a composite anode consisting of nitrogen and phosphorus co-doped carbon paper modified with cobalt tetroxide (Co3O4) nanoparticles.140
This Na–Co@NP-CP anode demonstrated exceptional cycling stability in Na–CO2 batteries, maintaining over 100 cycles at a high current density of 1000 mA g−1 (Fig. 7b). Post-cycling analysis using scanning electron microscopy (SEM) revealed that the anode retained a smooth surface and intact crystal structure, highlighting its ability to suppress dendrite growth and maintain electrochemical stability.
In another innovative approach, Wang and co-workers incorporated ferrocene (Fc), an organic transition metal compound, into the electrolyte of Li–CO2 batteries.156 This modification stabilised discharge intermediates and promoted a two-electron reaction pathway during the CO2 reduction reaction (CO2 RR), leading to more stable battery operation. The addition of 20 mM Fc resulted in a first-cycle discharge capacity of 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 mAh g−1, nearly five times higher than the 3192 mAh g−1 achieved without Fc (Fig. 7c). This improvement suggests that ferrocene reduces polarization during discharge, enhancing overall battery performance and stability.
900 mAh g−1, nearly five times higher than the 3192 mAh g−1 achieved without Fc (Fig. 7c). This improvement suggests that ferrocene reduces polarization during discharge, enhancing overall battery performance and stability.
For K–CO2 batteries, the instability of potassium metal anodes also remains a critical challenge in K–CO2 batteries, primarily due to dendrite growth and parasitic reactions. In this context, Li and co-workers have demonstrated a breakthrough by in situ construction of a KF-rich artificial SEI layer using a concentrated 2.7 M KFSI/TEGDME electrolyte.33 This SEI layer, composed of KF and organic species (e.g., carboxylic esters), homogenises K+ flux and suppresses dendrite formation, enabling 450 cycles at 300 mA g−1 with a curtailed capacity of 500 mAh g−1, a low overpotential gap of 0.81 V at 50 mA g−1 and 100% coulombic efficiency in symmetric cells, contrasting with untreated K anodes (<60% efficiency). This work shows the potential of concentrated electrolytes for interfacial stabilization, though challenges like cost and viscosity must be addressed for scalability.
These advancements highlight the importance of tailored strategies for optimizing alkali–CO2 batteries. By addressing anode degradation and improving reaction kinetics, these approaches offer promising solutions for developing more durable and efficient energy storage systems.
Electrolytes of alkali metal–CO2 batteries
The electrolyte is a critical component in alkali metal–CO2 batteries, facilitating the conduction of alkali ions between the anode and cathode during charge and discharge cycles. Typically, these electrolytes are composed of non-aqueous solvents paired with alkali–metal salts that remain stable in CO2-rich environments. Solvents such as dimethyl sulfoxide (DMSO) and tetraethylene glycol dimethyl ether (TEGDME) are favoured for their high thermal stability, while carbonates like ethylene carbonate (EC) and diethyl carbonate (DEC) are valued for their excellent ionic conductivity.84 However, the reactivity of CO2 with these solvents can lead to their degradation, resulting in the formation of unwanted by-products that compromise battery performance, particularly in systems that operate under high recharge overpotentials. To overcome these challenges, researchers are exploring alternative electrolyte systems, including solid and hybrid electrolytes, which offer enhanced safety and stability. Additionally, the incorporation of stabilizing additives has shown promise in improving ionic conductivity and extending the battery lifespan. Developing more efficient and stable electrolytes is essential for advancing the performance and commercial viability of alkali metal–CO2 batteries, paving the way for their broader adoption in energy storage applications.33,84Electrolyte additives play a crucial role in enhancing the performance and stability of metal–CO2 batteries and can create a favourable environment for redox reactions involving CO2, improving reaction kinetics and efficiency. Additionally, certain additives help form a stable passivation layer on the electrode surface, reducing degradation and minimizing unwanted side reactions that could compromise battery longevity. Additives also modify the chemical environment of the electrolyte, improving its ionic conductivity and facilitating better ion transport within the system. This enhanced ion mobility leads to more efficient redox reactions at the cathode, as ions move more freely between electrodes. Furthermore, some additives contribute to electrolyte stabilization by preventing decomposition or undesired side reactions, while others actively participate in redox processes, further boosting battery performance.33,61,84
For example, Lu and co-workers have investigated the use of brominated organic additives, specifically 3-benzyloxy bromopropane (B3BPE), 4-bromo-3-chloroethyl acetate (E4BCT), and 3-phenoxypropyl bromide (BPB), to enhance the performance and lifespan of Li–CO2 batteries.84 Among these, B3BPE, with its longer chain length and stronger polarity, demonstrated the most significant improvement in battery performance (Fig. 8a). The additive facilitated the decomposition of Li2CO3, a key discharge product, and maintained a stable charging voltage of ∼3.90 V over 120 cycles. The study also revealed that additives with oxygen-containing functional groups, such as ethers and esters, can modulate the interaction strength with Li2CO3, further enhancing battery stability. These findings provide valuable insights into the design and selection of electrolyte additives, paving the way for more durable and efficient Li–CO2 batteries. In a related study, Dan and co-workers have applied halogenated organic additives, such as N-phenylpyrrolidine (PPD) and 1-(3-bromophenyl) pyrrole (Br-PPD) to improve the performance of Li–CO2 batteries.6 PPD reduced the charging potential to 3.65 V, the lowest reported for a pure Super P cathode, while Br-PPD enhanced the stability of the lithium anode, enabling long-term cycling of 800 hours (Fig. 8b). The introduction of bromine functional groups in Br-PPD not only improved the CO2 evolution reaction (CO2ER) but also protected the lithium anode from side reactions, forming a stable SEI.
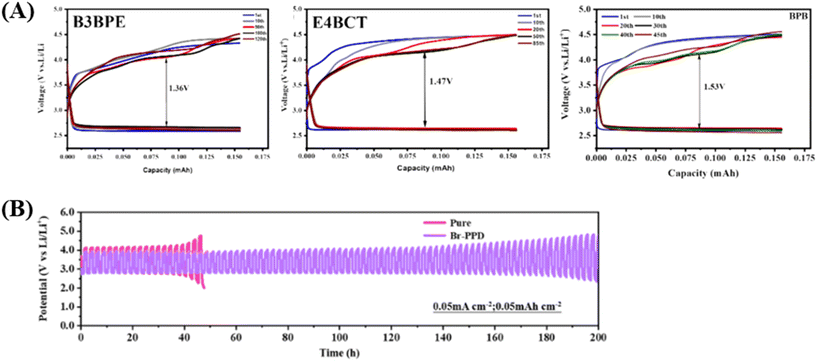 | ||
| Fig. 8 (A) Cycling performance of Li–CO2 cells with (a) B3BPE, (b) E4BCT and (c) BPB electrolytes at a current density of 0.1 mA cm2 and a cut-off capacity of 0.2 mAh cm2. Reproduced with permission.166 Copyright 2024, Chemical Engineering Journal. (B) Time–voltage curves of Li–CO2 cells at 0.05 mA cm−2. Reproduced with permission.6 Copyright 2023, ACS Applied Material Interface. | ||
While traditional aprotic electrolytes offer high ionic conductivity, their high cost and flammability pose significant challenges, particularly for cost-effective metal-based CO2 batteries. To address these limitations, solid-state electrolytes have emerged as a promising alternative. Solid electrolytes are essential for enhancing the safety, stability, and long-term performance of metal–CO2 batteries. Unlike liquid electrolytes, they eliminate issues such as electrolyte volatilization and leakage while offering benefits like high film-forming stability, flexibility, low cost, and superior mechanical properties. However, polymer electrolytes often face challenges such as low ionic conductivity and parasitic reactions with highly reactive alkali metals like sodium, which limit their effectiveness.33,46,122,157–160
To overcome these limitations, Lu and co-workers explored the impact of magnesium ion doping on the ionic conductivity of Na3Zr2Si2PO12 (NZSP).127 Their study revealed that Mg2+ doping significantly enhances ionic conductivity, achieving 1.16 mS cm−1 at room temperature while reducing activation energy. A composite electrolyte, consisting of the polymer PVDF-HFP and Na3.2Zr1.9Mg0.1Si2PO12 (NZM1SP) powder, was tested in Na–CO2 batteries using Ru-carbon nanotubes (CNTs) as the cathode catalyst. The battery assembled with PVDF-HFP-NZM1SP demonstrated a high discharge capacity of 7720 mAh g−1 at 200 mA g−1. At the same current density with a cut-off capacity of 500 mAh g−1, it maintained a discharge voltage above 2 V after 120 cycles, demonstrating its potential for long-term stability.
Another innovative approach involves the use of super-concentrated aqueous electrolytes, known as “water-in-salt” (WiSE) electrolytes. These electrolytes offer several advantages over traditional systems, including cost-effectiveness and non-flammability. WiSEs use water as the solvent, with the solute salt having a significantly larger mass and volume than the solvent. This unique structure endows WiSEs with excellent physical and chemical properties, such as enhanced stability and improved ionic conductivity.161,162 To further optimise WiSEs, strategies such as maintaining high salt concentrations and introducing a second salt with low viscosity and high ionic conductivity are commonly employed.163 While WiSEs have shown promise in lithium- and sodium-ion batteries, their direct use in alkali metal–CO2 batteries is hindered by the highly reactive nature of alkali metals, which can react violently with unsolvated water molecules, compromising safety and stability. Therefore, integrating WiSEs into alkali metal–CO2 battery systems through a hybrid electrolyte design is crucial to achieving both high performance and long-term stability.7
In this context, Yang and co-workers investigated the use of a hybrid electrolyte in a Na–CO2 battery.164 Their design combined a high-concentration water-based electrolyte with a non-aqueous electrolyte, separated by a NASICON solid electrolyte. The water-based electrolyte, composed of NaFSI salt (known for its high solubility in water) and NaClO4 salt (valued for its low viscosity and high solubility), was used on the cathode side to ensure high electrolyte concentration and enhanced ionic conductivity. On the anode side, a non-aqueous electrolyte was employed to protect the highly reactive Na anode from water. The NASICON solid electrolyte, which selectively allows the transport of Na+ ions, serves as a separator, preventing anode corrosion while maintaining stability in the anolyte. The hybrid Na–CO2 battery delivered exceptional performance, achieving a discharge specific capacity of 148.1 mAh cm−2, a power density of 6.56 mW cm−2, and remarkable cycling stability, completing over 1200 cycles at 30 °C. Additionally, the battery demonstrated impressive low-temperature performance, operating stably for more than 105 days at −20 °C, thanks to the high reversibility of the discharge product HCOOH and the robust properties of the electrolyte (Fig. 9a). Ionic liquids (ILs) have emerged as promising electrolytes for metal–CO2 batteries due to their unique physicochemical properties. Unlike traditional organic electrolytes, ILs exhibit negligible volatility, a wide electrochemical window, and high thermal stability, making them particularly suitable for high-temperature applications. In a recent study, Zhang and co-workers employed an imidazolium-based ionic liquid, 1-butyl-3-methyl-imidazolium bis(trifluoromethanesulfonyl)imide (BMImTFSI), as the electrolyte solvent in Li–CO2 batteries.165
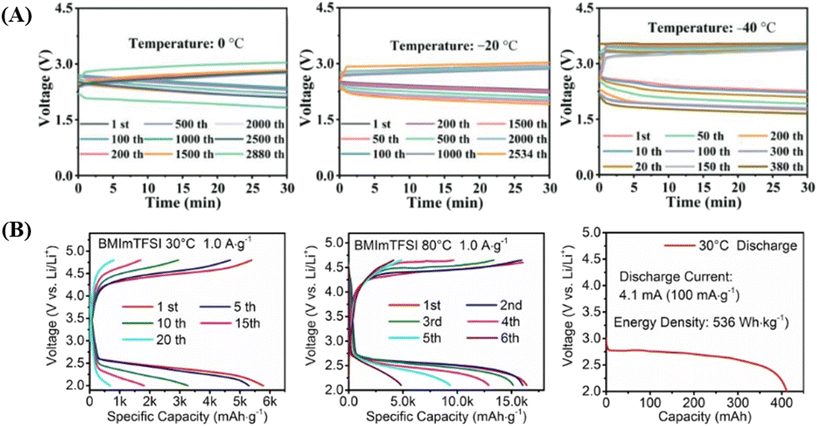 | ||
| Fig. 9 (A) Discharge/charge curves of a hybrid Na–CO2 battery at different temperatures: 0 °C, −20 °C, and −40 °C. Reproduced with permission.164 Copyright 2024, Advanced Energy Materials. (B) Full discharge–recharge profiles of Li–CO2 batteries at temperatures of 30 °C and at 80 °C; full discharge capacity of a Li–CO2 pouch cell fabricated with an FGDY cathode catalyst and a BMImTFSI based electrolyte. Reproduced with permission.165 Copyright 2025, Angewandte. | ||
The IL-based electrolyte mitigated issues related to electrolyte volatilization and leakage, thereby enhancing battery safety and stability at elevated temperatures. The Li–CO2 battery using BMImTFSI exhibited remarkable cycling performance, sustaining 240 cycles at 80 °C with a current density of 1.0 A g−1 and a cutoff capacity of 1000 mAh g−1. Additionally, the system demonstrated a high discharge capacity of 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 399 mAh g−1 and a reversible charge capacity of 16
399 mAh g−1 and a reversible charge capacity of 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 038 mAh g−1 at 80 °C, significantly outperforming conventional organic electrolytes (Fig. 9b). These results highlight the potential of IL-based electrolytes in advancing the performance and durability of Li–CO2 batteries.
038 mAh g−1 at 80 °C, significantly outperforming conventional organic electrolytes (Fig. 9b). These results highlight the potential of IL-based electrolytes in advancing the performance and durability of Li–CO2 batteries.
Based on the database used in this article, the most commonly used electrolytes in Li–CO2 and Na–CO2 batteries play a critical role in the stability, efficiency, and rechargeability of these systems. In Li–CO2 batteries, the most common electrolytes include lithium salts dissolved in organic solvents, such as 1 M LiTFSI in TEGDME and 1 M LiCF3SO3 in TEGDME. TEGDME (tetraethylene glycol dimethyl ether) is widely used due to its high chemical stability, low volatility, and high dielectric constant, which facilitate the dissolution of large concentrations of lithium salts. These properties are essential for ensuring efficient ionic conduction and the formation of a stable electrode/electrolyte interface, critical factors for improving battery cyclability and energy efficiency.
For Na–CO2 batteries, the most commonly used electrolytes include 1 M NaClO4 in TEGDME, which shares many of the benefits of TEGDME as a solvent, including excellent chemical stability and the ability to form stable passivation layers on the surface of sodium metal. Additionally, formulations such as 1.0 M NaCF3SO3 in DIGLYME have also been reported, where DIGLYME (bis(2-methoxyethyl) ether) is chosen for its high oxidative stability, low viscosity, and good compatibility with sodium metal, promoting better ionic diffusion and transport efficiency. In more advanced systems, combinations such as 2 M NaClO4 in MeCN as a catholyte and 1 M NaOTf in TEGDME as an anolyte are also used to explore new CO2 storage mechanisms and improve cycling stability, addressing challenges such as dendritic growth and long-term stability.
Overall, the choice of electrolyte in Li–CO2 and Na–CO2 batteries is a critical factor in determining the electrochemical performance, safety, and commercial viability of these devices, directly influencing energy efficiency, durability, and reliability in practical applications.
Separators of alkali metal–CO2 batteries
In metal–CO2 batteries, separators are essential components that prevent physical contact between electrodes while enabling ion transport and, in some cases, facilitating CO2 diffusion to the cathode.The choice of separator is influenced by the electrolyte type (liquid, gel, or solid), the need for dendrite suppression, and the specific challenges posed by CO2 utilization. The most commonly used separators in metal–CO2 batteries are primarily porous polymer-based membranes, such as polyethylene (PE) and polypropylene (PP). These materials are cost-effective, mechanically robust, and chemically stable. They are often manufactured in multilayer structures to enhance thermal stability and resistance to high temperatures.167 Their high porosity ensures efficient ion transport, but they struggle to suppress dendrite growth. Additionally, their poor wettability with certain electrolytes can hinder performance. Glass fiber (GF) separators, another liquid electrolyte option, offer superior porosity and electrolyte retention but suffer from brittleness and low mechanical strength, limiting their durability in long-term cycling.
Modification of conventional separators presents a promising solution to critical challenges in alkali metal–CO2 batteries such as the shuttle effect of redox mediators (RMs), which causes parasitic reactions and accelerates anode degradation, severely limiting battery cycle life. For instance, Lian and co-workers engineered a modified glass fiber (GF) separator by coating it with a zinc-based metal–organic framework (Zn-MOF, ZIF-L) nanoplate layer. This innovation effectively blocked the migration of the cathode catalyst, ruthenium acetylacetonate – Ru(acac)3, to the lithium anode.110 The Zn-MOF-modified separator (M-GF) not only physically restricted catalyst shuttling through its uniform micropores (∼3.4 Å) but also facilitated homogeneous Li+ flux, suppressing dendritic growth and electrode pulverization. Experimental results demonstrated remarkable improvements: the cycle life of Li–CO2 batteries increased from 36 cycles (GF separator) to 142 cycles (M-GF separator), while coulombic efficiency reached 100% (Fig. 10).
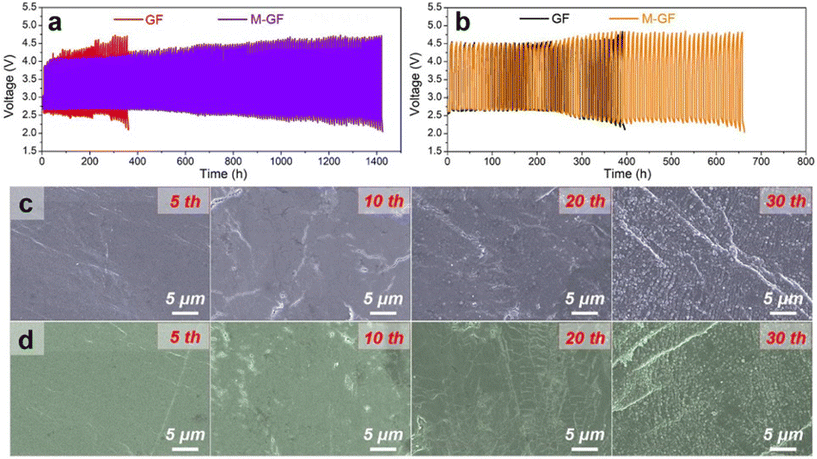 | ||
| Fig. 10 Cycling stability of Li–CO2 batteries based on GF and M-GF separators at a current density of 200 mA g−1 with a cut-off capacity of 1000 mAh g−1 with (a) and (b) without Ru(acac)3 addition. SEM images of Li anodes after 5, 10, 20 and 30 cycles when batteries are based on (c) GF and (d) M-GF separators. Reproduced with permission.110 Copyright 2022, Chemical Engineering Journal. | ||
Beyond conventional polymeric separators, Lu and co-workers have developed a multi-functional Janus separator for high-stability Li–CO2 batteries, addressing critical challenges such as cathodic passivation, lithium dendrite formation, and CO2 utilization.127 The separator, constructed by depositing Prussian blue (PB) on a nylon-6 membrane (PA) and coating it with a PB-reduced graphene oxide (rGO) composite, offers dual functionality: PB inhibits lithium dendrite growth on the anode side, while the PB-rGO composite acts as a cathode extension, accommodating discharge products and enhancing conductivity. The Li–CO2 battery with the Janus separator achieved an ultra-high discharge capacity of 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 mAh g−1 and an exceptionally long cycle life of 3144 hours, with a round-trip energy efficiency of around 83%. The separator also demonstrated superior mechanical strength, thermal stability, and the ability to block air pollutants, preventing anode corrosion, compared to conventional separators.
100 mAh g−1 and an exceptionally long cycle life of 3144 hours, with a round-trip energy efficiency of around 83%. The separator also demonstrated superior mechanical strength, thermal stability, and the ability to block air pollutants, preventing anode corrosion, compared to conventional separators.
Challenges and future opportunities
Li–CO2 batteries
One of the major challenges in the development of Li–CO2 batteries lies in the rate-limited kinetics of both the CO2 reduction reaction (CO2RR) and CO2 evolution reaction (CO2ER), primarily due to the formation of Li2CO3 – an insulating discharge product whose reversible decomposition is difficult. This leads to high overpotentials, rapid capacity fading, catalyst passivation, and electrolyte degradation.149 Moreover, the high cost and limited utilization efficiency of noble metal catalysts like Ru restrict their practical application.127The persistence of Li2CO3 on the cathode surface after repeated cycling also blocks active catalytic sites, impairs ion transport, and gradually limits the battery's capacity.127 Various strategies have been proposed to mitigate this issue, such as the introduction of metallic catalysts (e.g., Ru,60 MnO2,123 MoS2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 144) and the use of conductive interfaces based on graphene71 and MXenes.168 Nonetheless, achieving fast, complete, and reversible decomposition of Li2CO3 remains one of the key technical barriers to commercializing Li–CO2 batteries.
144) and the use of conductive interfaces based on graphene71 and MXenes.168 Nonetheless, achieving fast, complete, and reversible decomposition of Li2CO3 remains one of the key technical barriers to commercializing Li–CO2 batteries.
To overcome this limitation, there has been growing interest in the use of operando techniques, such as in situ Raman spectroscopy, electrochemical impedance spectroscopy under CO2 flow, and solid-state nuclear magnetic resonance, which allow the observation of reactions in real time and under operating conditions. In addition, density functional theory (DFT) calculations have been employed to predict the stability of intermediate species and to map the free energy profiles of the reactions. Integrating these experimental and computational tools is essential to uncover the fundamental mechanisms and to guide the development of Li–CO2 batteries with improved efficiency, stability, and reversibility.169–172 Future work can focus on scaling these findings to macroscopic systems and exploring hybrid gas environments, multifunctional catalysts, and novel electrolyte formulations to maximize battery life, capacity, and safety.110
Electrolyte stability is a critical factor for the performance and durability of Li–CO2 batteries. However, many of the organic solvents commonly used in these systems, such as tetraethylene glycol dimethyl ether (TEGDME), exhibit high volatility and limited oxidative stability, particularly under the high charging voltages required to decompose Li2CO3. Under such conditions, partial decomposition of the electrolyte often occurs, leading to the formation of unstable by-products that can react with the lithium metal, form undesired passivation layers, and reduce coulombic efficiency. To mitigate such degradation, recent studies have focused on protecting the lithium anode through artificial solid electrolyte interphases (SEI), polymer coatings, or solid-state protective layers, which can suppress parasitic reactions and improve long-term cycling stability.
Additionally, dissolved CO2 may chemically interact with the solvents or lithium salts, triggering parasitic reactions that accelerate cell aging. These degradation processes compromise not only electrochemical performance but also system safety, especially in sealed configurations.
To address these issues, various approaches have been investigated, including the development of electrolytes with high electrochemical stability, such as ionic liquids,112 stabilizing additives,6 and solid-state electrolytes.122 Furthermore, engineering the electrode–electrolyte interface through protective coatings and electrolyte composition optimization has shown promise in minimizing chemical degradation.
Looking ahead, the integration of Li–CO2 batteries into practical systems will require a holistic approach that combines material innovation with cell engineering. Tailoring battery configurations for niche applications—such as extraterrestrial energy storage, wearable electronics, or grid-scale CO2 capture—will demand advances not only in electrochemistry but also in thermal management, gas regulation, and safety protocols.
Na–CO2 batteries
Sodium–CO2 batteries face several critical challenges, including high overpotentials, low reaction reversibility due to the formation of stable discharge products such as Na2CO3, and cathode passivation caused by the accumulation of amorphous carbon. These factors hinder the decomposition of reaction products and limit the rechargeability and energy efficiency of the system. To overcome these issues, researchers have focused on developing catalysts with oxygen vacancies, structural defects, and heteroatom doping, which can facilitate the breakdown of Na2CO3 and improve overall battery performance.173–175Another major limitation is the instability of the sodium metal anode. It is highly reactive with traditional electrolytes and prone to dendrite growth, leading to short circuits and reduced cycle life. Several strategies have been proposed to mitigate these effects, including the use of artificial SEI layers, sodiophilic substrates, and NASICON-type solid electrolytes to physically isolate the anode and cathode environments.127,175,176
Although these strategies have enhanced performance, persistent challenges such as limited ionic conductivity and suboptimal energy density still hinder long-term stability. To address these issues, recent advancements in structural modulation and dual-carbon confinement (e.g., CNTs/MoS2@NC) have been explored to provide mechanical buffering against volume expansion and to create synergistic ‘electron highways’ that facilitate fast sodiation kinetics. These strategies, combined with interface stabilization through electrolyte optimization and film-forming additives, are essential for suppressing structural degradation and ensuring long-term cyclability.177–179
From the cathode perspective, high-surface-area and conductive materials such as MWCNTs, graphene, co-doped porous carbons, and transition metal-based catalysts (e.g., Co9S8, MoS2/SnS2, and Fe–Cu) have demonstrated strong potential to reduce overpotentials and extend cycle life. Techniques like dual heteroatom doping (e.g., N/S) and p-band tuning have also been successfully applied to optimize CO2 adsorption and facilitate product desorption.174,180,181
In parallel, emerging cell architectures—such as anode-free configurations (where Na is generated in situ during charging) and flexible solid-state designs with freestanding electrodes—are expanding the applicability of Na–CO2 batteries from stationary storage to portable and aerospace applications.72,182
K–CO2 batteries
Despite being in an early stage of development, K–CO2 batteries offer distinct theoretical advantages that justify their potential as a cost-effective alternative to lithium and sodium systems. Thermodynamically, the K–CO2 system operates via the discharge reaction 4K + 3CO2 + 4e− → 2K2CO3 + C, which presents a standard Gibbs free energy change (ΔfG°) of −1069.12 kJ mol−1. This results in a theoretical discharge potential of 2.48 V, which is notably higher than the corresponding value of 2.35 V for Na–CO2 batteries and approaches the levels of Li–based systems. Furthermore, K+ ions exhibit superior transport kinetics in organic solvents; for instance, in propylene carbonate (PC), K+ has the smallest Stokes radius (3.6 Å) compared to Li+ (4.8 Å) and Na+ (4.6 Å), leading to the highest ionic mobility and conductivity among them.183However, multiple technical challenges still persist. Chief among them is the instability of the potassium metal anode, which leads to dendritic growth, severe parasitic reactions with electrolytes, and poor cycling performance. To address this, recent studies have explored the use of artificial SEI layers and potassium-based alloys such as KSn, which help mitigate dendrite formation and enhance anode stability.11,33
Another significant challenge is the complex reaction mechanism at the cathode. While the electrochemical pathways for Li and K–CO2 batteries are generally consistent when carbonate is the final product, their decomposition energy barriers differ significantly, leading to distinct thermodynamic charge potentials. Notably, compared to the various reaction pathways observed in Li–CO2 systems, options for K–CO2 batteries are more limited. Although intermediates like potassium superoxide (KO2) could theoretically lower overpotentials, K2CO3 remains the dominant and most problematic product.184 To address these kinetic constraints, innovative cathode materials, including bamboo-like nitrogen-doped carbon nanotubes (B-NCNTs) and carboxyl-functionalized MWCNTs, have been developed. These materials have shown the ability to weaken C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds in K2CO3 through strong electrostatic interactions, enhancing its decomposition kinetics and thereby improving cycling stability.11,33
O bonds in K2CO3 through strong electrostatic interactions, enhancing its decomposition kinetics and thereby improving cycling stability.11,33
Mechanistic insights from in situ environmental TEM revealed the nucleation and decomposition pathways of K2CO3 at the nanoscale, providing valuable information about redox processes in these systems. These findings underscore the importance of rational electrode design, interface engineering, and advanced diagnostics for the advancement of K–CO2 technologies.34
Looking ahead, future research must focus on developing robust bifunctional catalysts, optimizing electrolyte formulations to stabilize both electrodes, and designing safe, scalable cell configurations. By tackling these scientific and engineering challenges, K–CO2 batteries could emerge as a cost-effective and sustainable energy storage solution with broad applications in both terrestrial and extraterrestrial environments.
Conclusions
This review provides a comprehensive analysis of the current state of alkali metal–CO2 batteries, encompassing lithium-, sodium-, and potassium-based systems, through both technical discussion and scientometric evaluation. The study highlights the substantial progress made in the development of advanced materials for cathodes, anodes, electrolytes, and separators, as well as emerging cell configurations and protective strategies. Among the key technical challenges identified are the rate-limited kinetics of the CO2 reduction and evolution reactions, instability of alkali metal anodes, and electrolyte degradation under CO2-rich environments. These limitations result in poor reversibility, high overpotentials, and short cycle life, all of which hinder the commercial viability of these systems.Scientometric analysis confirms that Li–CO2 batteries remain the most studied, with a sharp increase in publications over the past decade, while Na–CO2 and K–CO2 batteries are gaining momentum due to their cost-effectiveness and abundant raw materials. The integration of catalytic nanostructures, heteroatom-doped carbon frameworks, solid-state and hybrid electrolytes, and redox mediators has shown significant potential in enhancing energy efficiency, cycling stability, and safety.
Future efforts must focus on bridging fundamental understanding with practical design, emphasizing the development of multifunctional catalysts, in situ characterization techniques, and scalable, safe architectures. Moreover, tailoring battery systems for specific applications, ranging from wearable electronics to grid-scale CO2 capture and extraterrestrial energy storage, will be critical to unlocking the full potential of metal–CO2 technologies. In addition, testing metal–CO2 systems at higher TRL levels—such as in pouch-type batteries—is part of a learn-by-doing approach to understand scalability factors and the gap between theoretical and practically achievable charge potentials. A decay in performance and potential is expected as the system transitions from the coin cell level to the full battery or system level, but only through this process will it be possible to assess the economic viability of the technology and identify suitable application contexts.
Despite significant advancements, several critical research gaps continue to hinder the practical development of metal–CO2 batteries. One of the most pressing challenges is the rate limited kinetics of the CO2RR/CO2ER, which results in high overpotentials and poor energy efficiency. Additionally, the degradation of electrolytes, particularly under CO2-rich and high-voltage conditions, leads to reduced cycle life and instability. The limited diffusion of CO2 in non-aqueous electrolytes and the formation of insulating discharge products further restrict mass transport and electrochemical performance. Mechanistic uncertainties surrounding the origin and reversibility of discharge products demonstrate the need for advanced in situ characterization techniques.
This review highlights the need for targeted research in underexplored systems such as K–CO2 batteries. Despite their potential advantages, such as faster ion transport and low material cost, potassium-based systems remain largely overlooked. Practical recommendations include prioritizing the development of bifunctional catalysts to improve K2CO3 decomposition, designing artificial SEI layers for enhanced anode stability, and exploring compatible high-concentration or hybrid electrolytes.
Overall, this work provides strategic insights and defines clear directions for advancing efficient, durable, and sustainable alkali metal–CO2 batteries as next-generation solutions for energy storage and environmental mitigation. These technologies are closely aligned with United Nations Sustainable Development Goals (SDGs), particularly SDG 7 (Affordable and Clean Energy), SDG 12 (Responsible Consumption and Production), and SDG 13 (Climate Action). By simultaneously enabling high-efficiency energy storage and electrochemical CO2 utilization, metal–CO2 batteries represent a transformative approach to addressing climate change and resource sustainability, while adding value to CO2 as a useful chemical feedstock. Continued innovation in this field not only drives battery science forward but also plays a critical role in supporting global efforts toward decarbonization, circular technologies, and climate resilience.
Author contributions
C. S. E.: conceptualization, investigation, methodology, data curation, and writing – original draft, review & editing. M. F. V.: investigation, validation, and writing – original draft, review & editing. D. P.: conceptualization and visualization. C. D.: project administration. F. D.: project administration and supervision. V. H. J. M. S.: writing – review & editing, supervision, and funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
The authors confirm that the data supporting the findings of this study are available within the article.Acknowledgements
The authors thank the National Agency of Petroleum, Natural Gas and Biofuels (ANP), Petronas Petróleo Brasil LTDA (PPBL), and Petronas Research Sdn Bhd (PRSB) for the strategic partnership, the investment in the project, and the technical–scientific support for its execution, in alignment with the respective development plan and the R&D Investment Clause. The authors also thank the Pontifical Catholic University of Rio Grande do Sul (PUCRS) for the support in the preparation and implementation of the executive infrastructure projects.References
- M. Höök and X. Tang, Energy Policy, 2013, 52, 797–809 CrossRef.
- R. S. Haszeldine, Science, 2009, 325, 1647–1652 CrossRef CAS PubMed.
- N. United, Sustainable Development Goals. Transforming our world: the 2030 agenda for sustainable development, United Nations, New York, 2015 Search PubMed.
- S. Mukhopadhyay, S. Khan, A. Banerjee and D. Pramanik, Inorg. Chim. Acta, 2025, 577, 122441 CrossRef CAS.
- Z. Zhang, W. L. Bai, K. X. Wang and J. S. Chen, Energy Environ. Sci., 2020, 13, 4717–4737 RSC.
- B. Dan, L. Li, S. Li, L. Liu, Z. Wang, D. Wang and X. Liu, ACS Appl. Mater. Interfaces, 2023, 15, 49116–49122 CrossRef CAS PubMed.
- N. Feng, B. Wang, Z. Yu, Y. Gu, L. Xu, J. Ma, Y. Wang and Y. Xia, ACS Appl. Mater. Interfaces, 2021, 13, 7396–7404 CrossRef CAS PubMed.
- C. Xu, P. Hong, Y. Dong, Y. Li, Y. Shen, J. Biskupek, H. Zhao, U. Kaiser, G. Shao and Y. Lei, Adv. Mater., 2024, 36, 2409533 CrossRef CAS PubMed.
- Z. Zheng, C. Wu, Q. Gu, K. Konstantinov and J. Wang, Energy Environ. Mater., 2021, 4, 158–177 CrossRef CAS.
- M. Tang, J. Y. Du, J. L. Ma, X. D. Wang, X. Zhang, Q. Y. Shen, F. P. Wang and Y. Wang, J. Alloys Compd., 2022, 911, 165054 CrossRef CAS.
- Y. Lu, Y. Cai, Q. Zhang, Y. Ni, K. Zhang and J. Chen, Angew. Chem., Int. Ed., 2021, 60, 9540–9545 CrossRef CAS PubMed.
- S. Thoka, C. M. Tsai, Z. Tong, A. Jena, F. M. Wang, C. C. Hsu, H. Chang, S. F. Hu and R. S. Liu, ACS Appl. Mater. Interfaces, 2021, 13, 480–490 CrossRef CAS.
- Y. Gao, Z. Pan, J. Sun, Z. Liu and J. Wang, Nano-Micro Lett., 2022, 14, 94 CrossRef CAS PubMed.
- A. D. Pathak, P. R. Adhikari and W. Choi, Front. Energy Res., 2023, 11, 1150737 CrossRef.
- H. Ajibade, C. O. Ujah, K. C. Nnakwo and D. V. V. Kallon, Discover Appl. Sci., 2024, 6, 374 CrossRef CAS.
- Y. Ding, Z. P. Cano, A. Yu, J. Lu and Z. Chen, Electrochem. Energy Rev., 2019, 2, 1–18 CrossRef CAS.
- X. Mu, P. He and H. Zhou, Acc. Mater. Res., 2024, 5, 467–478 CrossRef CAS.
- C. Yang, K. Guo, D. Yuan, J. Cheng and B. Wang, J. Am. Chem. Soc., 2020, 142, 6983–6990 CrossRef CAS PubMed.
- L. Guo, B. Li, V. Thirumal and J. Song, Chem. Commun., 2019, 55, 7946–7949 RSC.
- J. Wang, B. Marchetti, X. D. Zhou and S. Wei, ACS Energy Lett., 2023, 8, 1818–1838 CrossRef CAS.
- C. Xu, P. Hong, Y. Dong, M. Robert, G. Shao and Y. Lei, Adv. Energy Mater., 2026, 16, 2406146 CrossRef CAS.
- B. Liu, Y. Sun, L. Liu, J. Chen, B. Yang, S. Xu and X. Yan, Energy Environ. Sci., 2019, 12, 887–922 RSC.
- B. L. Ellis and L. F. Nazar, Curr. Opin. Solid State Mater. Sci., 2012, 16, 168–177 CrossRef CAS.
- D. Gong, C. Wei, Z. Liang and Y. Tang, Small Sci., 2021, 1, 2100014 CrossRef CAS PubMed.
- Sarkar, V. R. Dharmaraj, C. H. Yi, K. Iputera, S. Y. Huang, R. J. Chung, S. F. Hu and R. S. Liu, Chem. Rev., 2023, 123, 9497–9564 CrossRef CAS PubMed.
- W. Zhang, J. Yin, W. Wang, Z. Bayhan and H. N. Alshareef, Nano Energy, 2021, 83, 105792 CrossRef CAS.
- C. Xu, Y. Dong, Y. Shen, H. Zhao, L. Li, G. Shao and Y. Lei, Small, 2023, 19, 2206445 CrossRef CAS PubMed.
- J. Zou, G. Liang, F. Zhang, S. Zhang, K. Davey and Z. Guo, Adv. Mater., 2023, 35, 2210671 CrossRef CAS PubMed.
- X. Sun, X. Mu, W. Zheng, L. Wang, S. Yang, C. Sheng, H. Pan, W. Li, C. H. Li, P. He and H. Zhou, Nat. Commun., 2023, 14, 536 CrossRef CAS PubMed.
- X. Xiao, Z. Zhang and P. Tan, Proc. Natl. Acad. Sci. U. S. A., 2023, 120, e2217454120 CrossRef CAS PubMed.
- Z. Zhang, X. G. Wang, X. Zhang, Z. Xie, Y. N. Chen, L. Ma, Z. Peng and Z. Zhou, Adv. Sci., 2018, 5, 1700567 CrossRef PubMed.
- W. Liu, C. Cai, Z. Zhang, Y. Li, Y. Hao, Y. Wang, R. Xu, D. Luo and Z. Chen, Nano Energy, 2024, 129, 109998 CrossRef CAS.
- X. L. Li, G. C. Qi, J. X. Zhang, J. L. Cheng and B. Wang, Adv. Funct. Mater., 2022, 32, 2105029 CrossRef CAS.
- L. Q. Zhang, Y. S. Tang, Q. N. Liu, T. T. Yang, C. C. Du, P. Jia, Z. F. Wang, Y. F. Tang, Y. F. Li, T. D. Shen and J. Y. Huang, Nano Energy, 2018, 53, 544–549 CrossRef CAS.
- Z. Zhang, Q. Zhang, Y. Chen, J. Bao, X. Zhou, Z. Xie, J. Wei and Z. Zhou, Angew. Chem., 2015, 127, 6650–6653 CrossRef.
- Y. Han, X. Zhou, R. Fang, C. Lu, K. Wang, Y. Gan, X. He, J. Zhang, H. Huang, W. Zhang, X. Xia and Y. Xia, Carbon Neutralization, 2023, 2, 169–185 CrossRef CAS.
- D. Weber, Đ. Tripković, K. Kretschmer, M. Bianchini and T. Brezesinski, Eur. J. Inorg. Chem., 2020, 3117–3130 CrossRef CAS.
- S. Q. Li, J. N. Yang, K. X. Wang and J. S. Chen, Acc. Mater. Res., 2024, 5, 1496–1506 CrossRef CAS.
- D. Na, H. Jeong, J. Baek, H. Yu, S. M. Lee, C. R. Lee, H. K. Seo, J. K. Kim and I. Seo, Electrochim. Acta, 2022, 419, 140408 CrossRef CAS.
- N. Wang, Y. M. Guo, F. H. Ning, X. Y. Liu, F. J. Li, J. J. Zhang, S. G. Lu and J. Yi, Energy Fuels, 2024, 38, 10324–10332 CrossRef.
- R. L. Song, J. R. Han, S. W. Tian, D. Wang and D. Liu, J. Power Sources, 2025, 626, 235763 CrossRef CAS.
- Q. C. Zhu, X. B. Xin, S. H. Zhou, Z. Y. Wang, D. Y. Mao and A. J. Fu, Electrochim. Acta, 2025, 514, 145683 CrossRef CAS.
- C. M. Tseng, C. C. Huang, J. Y. Pai and Y. Y. Li, ACS Sustainable Chem. Eng., 2023, 11, 8120–8130 CrossRef CAS.
- Y. Y. Xu, X. J. Li, Y. J. Li, Y. Wang, L. Song, J. C. Ding, X. L. Fan, J. P. He, T. Wang and Z. S. Wu, Energy Storage Mater., 2024, 68, 103354 CrossRef.
- W. Yu, L. Liu, Y. Yang, N. Li, Y. Chen, X. Yin, J. Niu, J. Wang and S. Ding, Chem. Eng. J., 2023, 465, 142787 CrossRef CAS.
- J. Liu, Z. Li, C. Chen and C. Sun, ACS Appl. Energy Mater., 2024, 7, 10730–10737 CrossRef CAS.
- S. Zhang, S. Xiao, D. Li, J. Liao, F. Ji, H. Liu and L. Ci, Energy Storage Mater., 2022, 48, 172–190 CrossRef.
- L. M. Liu, Y. Y. Qin, K. Wang, H. Mao, H. Wu, W. Yu, D. Y. Zhang, H. Y. Zhao, H. R. Wang, J. H. Wang, C. H. Xiao, Y. Q. Su and S. J. Ding, Adv. Energy Mater., 2022, 12, 2103681 CrossRef CAS.
- Y. H. Wang, J. W. Zhou, C. Lin, B. Chen, Z. Q. Guan, A. M. Ebrahim, G. N. Qian, C. L. Ye, L. Chen, Y. Y. Ge, Q. B. Yun, X. X. Wang, X. C. Zhou, G. Wang, K. D. Li, P. Y. Lu, Y. B. Ma, Y. C. Xiong, T. S. Wang, L. Zheng, S. Q. Chu, Y. Chen, B. Wang, C. S. Lee, Y. J. Liu, Q. F. Zhang and Z. X. Fan, Adv. Funct. Mater., 2022, 32, 2202737 CrossRef CAS.
- H. Liu, H. Lai, B. Cao, X. Tian, D. Zhang, R. A. Soomro and Y. Wu, Carbon, 2024, 230, 119676 CrossRef CAS.
- Y. G. Deng, W. W. Zhang, C. Ma, C. J. Zhang, C. Li, Y. X. Hong, Y. C. Sun, J. Q. Niu, S. Guo and S. S. Yao, ACS Appl. Nano Mater., 2025, 8, 1499–1507 CrossRef CAS.
- Z. Hu, Y. Xie, D. Yu, Q. Liu, L. Zhou, K. Zhang, P. Li, F. Hu, L. Li, S. Chou and S. Peng, ACS Nano, 2021, 15, 8407–8417 CrossRef CAS PubMed.
- R. Zheng, M. Yang, X. Zhu, Q. Fang, X. Wang, P. Lei, J. Zhou, B. Wang and J. Cheng, Adv. Funct. Mater., 2025, 35, 2412999 CrossRef CAS.
- J. Zhang, G. Qi, J. Cheng, P. Ratajczak, Z. Wang, F. Beguin and B. Wang, ACS Sustainable Chem. Eng., 2023, 11, 16185–16193 CrossRef CAS.
- Y. Wang, X. Wang, B. Ge, J. Guo, C. Fernandez and Q. Peng, ACS Appl. Energy Mater., 2021, 4, 9961–996855 CrossRef CAS.
- Z. Lian, Y. Lu, C. Wang, X. Zhu, S. Ma, Z. Li, Q. Liu and S. Zang, Adv. Sci., 2021, 8, 2102550 CrossRef CAS PubMed.
- H. Ren, H. Li, P. Barry, Z. Wang, A. R. Campos, E. S. Takeuchi, A. C. Marschilok, S. Yan, K. J. Takeuchi and E. Reichmanis, Am. Chem. Soc., 2024, 36, 9299–9319 CAS.
- J. Z. Wang, X. Chen, X. M. Cui, M. Zhou, J. N. Wang, W. B. Liu, H. Ma, J. V. Anguita, S. R. P. Silva, K. Yang and W. Yan, Sustainable Energy Fuels, 2025, 9, 1084–1094 RSC.
- Y. L. Kong, H. Gong, L. Song, C. Jiang, T. Wang and J. P. He, Eur. J. Inorg. Chem., 2021, 590–596 CrossRef CAS.
- S. Y. Bie, M. L. Du, W. X. He, H. G. Zhang, Z. T. Yu, J. G. Liu, M. Liu, W. W. Yan, L. Zhou and Z. G. Zou, ACS Appl. Mater. Interfaces, 2019, 11, 5146–5151 CrossRef CAS PubMed.
- Y. He, Z. Chen and Y. Zhang, iScience, 2024, 27, 110491 CrossRef CAS PubMed.
- L. Liu, S. Shen, J. Li, N. Zhao, X. Yin, H. Zhao, W. Yu, Y. Su, B. Y. Xia and S. Ding, Angew. Chem., Int. Ed., 2024, 64, e202415728 CrossRef PubMed.
- J. Li, L. Liu, D. Guan, H. Liang, Z. Lu, H. Wang, Y. Zhang, J. Tang, S. Chen, G. Feng, L. Zeng, R. V. Kumar, J. Xu, S. Ding, Y. L. He and K. Xi, Adv. Funct. Mater., 2026, 36, e14223 CrossRef CAS.
- L. Liu, S. Shen, N. Zhao, H. Zhao, K. Wang, X. Cui, B. Wen, J. Wang, C. Xiao, X. Hu, Y. Su and S. Ding, Adv. Mater., 2024, 36, 2403229 CrossRef CAS PubMed.
- X. F. Hu, J. C. Sun, Z. F. Li, Q. Zhao, C. C. Chen and J. Chen, Angew. Chem., Int. Ed., 2016, 55, 6482–648665 CrossRef CAS PubMed.
- P. Tan, Z. H. Wei, W. Shyy, T. S. Zhao and X. B. Zhu, Energy Environ. Sci., 2016, 9, 1783–1793 RSC.
- Y. Hou, J. Wang, L. Liu, Y. Liu, S. Chou, D. Shi, H. Liu, Y. Wu, W. Zhang and J. Chen, Adv. Funct. Mater., 2017, 27, 1700564 CrossRef.
- X. Hu, Z. Li and J. Chen, Angew. Chem., Int. Ed., 2017, 56, 5785–5789 CrossRef CAS PubMed.
- C. Li, Z. Y. Guo, B. C. Yang, Y. Liu, Y. G. Wang and Y. Y. Xia, Angew. Chem., Int. Ed., 2017, 56, 9126–9130 CrossRef CAS PubMed.
- S. Yang, Y. Qiao, P. He, Y. Liu, Z. Cheng, J.-J. Zhu and H. Zhou, Energy Environ. Sci., 2017, 10, 972–978 RSC.
- L. J. Wang, W. R. Dai, L. P. Ma, L. L. Gong, Z. Y. Lyu, Y. Zhou, J. Liu, M. Lin, M. Lai, Z. Q. Peng and W. Chen, ACS Omega, 2017, 2, 9280–9286 CrossRef CAS PubMed.
- X. C. Wang, X. J. Zhang, Y. Lu, Z. H. Yan, Z. L. Tao, D. Z. Jia and J. Chen, ChemElectroChem, 2018, 5, 3628–3632 CrossRef CAS.
- C. Y. Wang, Q. M. Zhang, X. Zhang, X. G. Wang, Z. J. Xie and Z. Zhou, Small, 2018, 14, 1800641 CrossRef PubMed.
- X. Zhang, C. Wang, H. Li, X.-G. Wang, Y.-N. Chen, Z. Xie and Z. Zhou, J. Mater. Chem. A, 2018, 6, 2792–2796 RSC.
- S. M. Xu, C. J. Chen, Y. D. Kuang, J. W. Song, W. T. Gan, B. Y. Liu, E. M. Hitz, J. W. Connell, Y. Lin and L. B. Hu, Energy Environ. Sci., 2018, 11, 3231–3237 RSC.
- M. Mushtaq, X. W. Guo, J. P. Bi, Z. X. Wang and H. J. Yu, Rare Met., 2018, 37, 520–526 CrossRef CAS.
- J. Sun, Y. Lu, H. Yang, M. Han, L. Shao and J. Chen, Research, 2018, 2018, 6914626 Search PubMed.
- S. M. Xu, Z. C. Ren, X. Liu, X. Liang, K. X. Wang and J. S. Chen, Energy Storage Mater., 2018, 15, 291–298 CrossRef.
- R. Wang, X. J. Zhang, Y. C. Cai, Q. S. Nian, Z. L. Tao and J. Chen, Nano Res., 2019, 12, 2543–2548 CrossRef CAS.
- D. L. Lei, S. Y. Ma, Y. C. Lu, Q. C. Liu and Z.J. Li, High-Performance Li-CO2 Batteries with -MnO2/CNT Cathodes, J. Electron. Mater., 2019, 48, 4653–4659 CrossRef CAS.
- P. F. Zhang, Y. Q. Lu, Y. J. Wu, Z. W. Yin, J. T. Li, Y. Zhou, Y. H. Hong, Y. Y. Li, L. Huang and S. G. Sun, Chem. Eng. J., 2019, 363, 224–233 CrossRef CAS.
- X. Li, J. Zhou, J. Zhang, M. Li, X. Bi, T. Liu, T. He, J. Cheng, F. Zhang, Y. Li, X. Mu, J. Lu and B. Wang, Adv. Mater., 2019, 31, 1903852 CrossRef PubMed.
- X. Li, H. Wang, Z. X. Chen, H. S. Xu, W. Yu, C. B. Liu, X. W. Wang, K. Zhang, K. Y. Xie and K. P. Loh, Adv. Mater., 2019, 31, 1905879 CrossRef CAS PubMed.
- Y. Lu, Y. Cai, Q. Zhang, L. Liu, Z. Niu and J. Chen, Chem. Sci., 2019, 10, 4306–4312 RSC.
- R. Pipes, J. R. He, A. Bhargav and A. Manthiram, ACS Appl. Energy Mater., 2019, 2, 8685–8694 CrossRef CAS.
- Z. Y. Guo, J. L. Li, H. C. Qi, X. M. Sun, H. D. Li, A. G. Tamirat, J. Liu, Y. G. Wang and L. Wang, Small, 2019, 15, 1803246 CrossRef PubMed.
- Y. C. Jin, F. Y. Chen, J. L. Wang and R. L. Johnston, Chem. Eng. J., 2019, 375, 121978 CrossRef CAS.
- Y. Qiao, S. M. Xu, Y. Liu, J. Q. Dai, H. Xie, Y. G. Yao, X. W. Mu, C. J. Chen, D. J. Kline, E. M. Hitz, B. Y. Liu, J. W. Song, P. He, M. R. Zachariah and L. B. Hu, Energy Environ. Sci., 2019, 12, 1100–1107 RSC.
- Z. Zhang, C. Yang, S. S. Wu, A. N. Wang, L. L. Zhao, D. D. Zhai, B. Ren, K. Z. Cao and Z. Zhou, Adv. Energy Mater., 2019, 9, 1802805 CrossRef.
- X. Xiao, W. Yu, W. Shang, P. Tan, Y. Dai, C. Cheng and M. Ni, Energy Fuels, 2020, 34, 16870–16878 CrossRef CAS.
- A. Jena, H. C. Hsieh, S. Thoka, S. F. Hu, H. Chang and R. S. Liu, ChemSusChem, 2020, 13, 2719–2725 CrossRef CAS PubMed.
- S. Thoka, C. J. Chen, A. Jena, F. M. Wang, X. C. Wang, H. Chang, S. F. Hu and R. S. Liu, ACS Appl. Mater. Interfaces, 2020, 12, 17353–17363 CrossRef CAS PubMed.
- Y. Xiao, F. Du, C. Hu, Y. Ding, Z. L. Wang, A. Roy and L. Dai, ACS Energy Lett., 2020, 5, 916–921 CrossRef CAS.
- G. Yue, X. Luo, Z. Hu, W. Xu, J. Li, J. Liu and R. Cao, Chem. Commun., 2020, 56, 11693–11696 RSC.
- C. Z. Wang, Y. Shang, Y. C. Lu, L. B. Qu, H. C. Yao, Z. J. Li and Q. C. Liu, J. Power Sources, 2020, 475, 228703 CrossRef CAS.
- Y. Xing, K. Wang, N. Li, D. Su, W.-T. Wong, B. Huang and S. Guo, Matter, 2020, 2, 1494–1508 CrossRef.
- Y. Qiao, J. W. Wu, J. Zhao, Q. L. Li, P. J. Zhang, C. S. Hao, X. L. Liu, S. T. Yang and Y. Liu, Energy Storage Mater., 2020, 27, 133–139 CrossRef.
- Y. C. Jin, F. Y. Chen and J. L. Wang, ACS Sustainable Chem. Eng., 2020, 8, 2783–2792 CrossRef CAS.
- K. V. Savunthari, C. H. Chen, Y. R. Chen, Z. Tong, K. Iputera, F. M. Wang, C. C. Hsu, D. H. Wei, S. F. Hu and R. S. Liu, ACS Appl. Mater. Interfaces, 2021, 13, 44266–44273 CrossRef CAS PubMed.
- C. F. Xu, J. Zhan, H. W. Wang, Y. Kang and F. Liang, J. Mater. Chem. A, 2021, 9, 22114–22128 RSC.
- M. H. Chen, Y. Liu, X. Q. Liang, F. Wang, Y. Li and Q. G. Chen, Energy Technol., 2021, 9, 2100547 CrossRef CAS.
- X. Xiao, Z. J. Zhang, W. T. Yu, W. X. Shang, Y. Y. Ma, X. B. Zhu and P. Tan, ACS Appl. Energy Mater., 2021, 4, 11858–11866 CrossRef CAS.
- H.-S. Kim, J.-Y. Lee, J.-K. Yoo and W.-H. Ryu, ACS Mater. Lett., 2021, 3, 815–825 CrossRef CAS.
- Y. M. Du, Y. J. Liu, S. X. Yang, C. Li, Z. Cheng, F. L. Qiu, P. He and H. S. Zhou, J. Mater. Chem. A, 2021, 9, 9581–9585 RSC.
- S. S. Zhang, X. Q. Liu, Y. Y. Feng, D. Wang, H. X. Yin, Z. Z. Chi, L. Li, J. Liu, S. X. Li, J. F. Huang, Z. Y. Guo and L. Wang, J. Power Sources, 2021, 506, 230226 CrossRef CAS.
- Z. Zhang, W. L. Bai, Z. P. Cai, J. H. Cheng, H. Y. Kuang, B. X. Dong, Y. B. Wang, K. X. Wang and J. S. Chen, Angew. Chem., Int. Ed., 2021, 60, 16404–16408 CrossRef CAS PubMed.
- D. Wang, J. G. Yang, P. He and H. S. Zhou, Energy Environ. Sci., 2021, 14, 4107–4114 RSC.
- K. Iputera, J. Y. Huang, S. C. Haw, J. M. Chen, S. F. Hu and R. S. Liu, J. Mater. Chem. A, 2022, 10, 3460–3468 RSC.
- Y. H. Wang, J. W. Zhou, C. Lin, B. Chen, Z. Q. Guan, A. M. Ebrahim, G. N. Qian, C. L. Ye, L. Chen, Y. Y. Ge, Q. B. Yun, X. X. Wang, X. C. Zhou, G. Wang, K. D. Li, P. Y. Lu, Y. B. Ma, Y. C. Xiong, T. S. Wang, L. Zheng, S. Q. Chu, Y. Chen, B. Wang, C. S. Lee, Y. J. Liu, Q. F. Zhang and Z. X. Fan, Adv. Funct. Mater., 2022, 32, 2202737 CrossRef CAS.
- Z. Lian, Y. Lu, S. Ma, L. Wang, Z. Li and Q. Liu, Chem. Eng. J., 2022, 450, 138400 CrossRef CAS.
- S. Wang, K. Xu, H. C. Song, T. Zhu, Z. Q. Yu, X. P. Song, D. K. Li, L. W. Yu, J. Xu and K. J. Chen, Adv. Energy Mater., 2022, 12, 2201866 CrossRef CAS.
- G. R. Qiu, Y. P. Shi and B. L. Huang, Nano Res., 2022, 15, 5153–5160 CrossRef CAS.
- L. Fan, H. Shen, D. Ji, Y. Xing, L. Tao, Q. Sun and S. Guo, Adv. Mater., 2022, 34, 2204134 CrossRef CAS PubMed.
- P. Jia, M. Yu, X. Zhang, T. Yang, D. Zhu, T. Shen, L. Zhang, Y. Tang and J. Huang, Nano Res., 2022, 15, 542–550 CrossRef CAS.
- S. Wang, H. C. Song, T. Zhu, J. M. Chen, Z. Q. Yu, P. F. Wang, L. W. Yu, J. Xu, H. S. Zhou and K. J. Chen, Nano Energy, 2022, 100, 107521 CrossRef CAS.
- J. Cheng, Y. Q. Bai, Y. B. Lian, Y. Ma, Z. Yin, L. Wei, H. Sun, Y. H. Su, Y. T. Gu, P. Kuang, J. Zhong, Y. Peng, H. B. Wang and Z. Deng, ACS Appl. Mater. Interfaces, 2022, 14, 18561–18569 CrossRef CAS PubMed.
- Z. Cheng, Z. Wu, Y. Tang, X. Fan, J. Zhang, Y. Chen, S. Xiang and Z. Zhang, Nanoscale, 2022, 14, 15073–15078 RSC.
- H. Hong, J. He, Y. Wang, X. Guo, X. Zhao, X. Wang, C. Zhi, H. Li and C. Han, J. Mater. Chem. A, 2022, 10, 18396–18407 RSC.
- Z. Lian, Y. Pei, S. Ma, Y. Lu and Q. Liu, ChemistrySelect, 2022, 7, e202104549 CrossRef CAS.
- W. Y. Huang, J. M. Qiu, Y. C. Ji, W. G. Zhao, Z. H. Dong, K. Yang, M. Yang, Q. D. Chen, M. J. Zhang, C. Lin, K. Xu, L. Y. Yang and F. Pan, ACS Nano, 2023, 17, 5570–5578 CrossRef CAS PubMed.
- Z. Y. Chen, M. M. Yuan, Z. L. Tang, H. L. Zhu and G. Zeng, Phys. Chem. Chem. Phys., 2023, 25, 7662–7668 RSC.
- D. Na, R. K. Kampara, D. H. Y. Yu, B. Yoon, S. W. Martin and I. Seo, Mater. Today Energy, 2023, 38, 101418 CrossRef CAS.
- X. Chen, J. Chen, Y. Liu, Y. Liu, Y. Gao, S. Fan, X. He, X. Liu, C. Shen, Y. Jiang, L. Li, Y. Qiao and S. Chou, ACS Appl. Mater. Interfaces, 2023, 15, 28106–28115 CrossRef CAS PubMed.
- Z. Cheng, Y. Fang, Y. Yang, H. Zhang, Z. Fan, J. Zhang, S. Xiang, B. Chen and Z. Zhang, Angew. Chem., 2023, 135, e202311480 CrossRef.
- H. K. Wu, Z. Wang, X. Y. Zeng, Z. T. Fei, Y. Gu, K. Ding, P. Dong, Y. J. Zhang and Q. Meng, Ceram. Int., 2023, 49, 4153–4159 CrossRef CAS.
- K. M. Naik, A. K. Chourasia, M. Shavez and C. S. Sharma, ChemSusChem, 2023, 16, e202300734 CrossRef CAS PubMed.
- L. Lu, C. Sun, J. Hao, Z. Wang, S. F. Mayer, M. T. Fernández-Díaz, J. A. Alonso and B. Zou, Energy Environ. Mater., 2023, 6, e12364 CrossRef CAS.
- K. Chourasia, M. Shavez, K. M. Naik, C. Bongu and C. S. Sharma, ACS Appl. Energy Mater., 2023, 6, 378–386 CrossRef.
- M. Chen, H. Meng, F. Wang, Q. Liu, Y. Liu, X. Liu, Q. Chen and Z. Chen, Chem. Eng. J., 2023, 455, 140564 CrossRef CAS.
- Y. Y. Fan, Y. Liu, Z. Chen, F. J. Lou, X. D. Lou and Y. Qiao, Inorg. Chem. Front., 2023, 10, 880–887 RSC.
- C. Guo, F. Zhang, X. Han, L. Zhang, Q. Hou, L. Gong, J. Wang, Z. Xia, J. Hao and K. Xie, Adv. Mater., 2023, 35, 2302325 CrossRef CAS PubMed.
- S. Li, J.-H. Wang, L.-Z. Dong, Y. Zhang, X.-M. Yao, Y. Chen, S.-L. Li and Y.-Q. Lan, Chin. Chem. Lett., 2023, 34, 107633 CrossRef CAS.
- W. Li, M. Zhang, X. Sun, C. Sheng, X. Mu, L. Wang, P. He and H. Zhou, Nat. Commun., 2024, 15, 803 CrossRef CAS PubMed.
- D. Na, D. Yu, H. Kim, B. Yoon, D. D. Lee and I. Seo, Nanomaterials, 2024, 14, 1894 CrossRef CAS PubMed.
- K. Iputera, C. H. Yi, J. Y. Huang, M. Nakayama, B. H. Liu, C. H. Wang, Y. W. Yang and R. S. Liu, J. Energy Storage, 2024, 100, 113467 CrossRef CAS.
- D.-H. Guan, X.-X. Wang, C.-L. Miao, J.-X. Li, J.-Y. Li, X.-Y. Yuan, X.-Y. Ma and J.-J. Xu, ACS Nano, 2024, 18, 34299–34311 CrossRef CAS PubMed.
- O. V. Korchagin and O. Tripachev, J. Electroanal. Chem., 2024, 973, 118662 CrossRef CAS.
- X. Ji, Y. Liu, Z. X. Zhang, J. B. Cui, Y. Y. Fan and Y. Qiao, Chem. – Eur. J., 2024, 30, e202303319 CrossRef CAS PubMed.
- J. W. Wu, J. Chen, X. Y. Chen, Y. Liu, Z. Hu, F. J. Lou, S. L. Chou and Y. Qiao, Chem. Sci., 2024, 15, 9591–9598 RSC.
- Z. Liu, Q. Zhang, L. Li and J. Guo, RSC Adv., 2024, 14, 5588–5593 RSC.
- Z. Wang, Y. Cai, Y. Ni, Y. Lu, L. Lin, H. Sun, H. Li, Z. Yan, Q. Zhao and J. Chen, Chin. Chem. Lett., 2023, 34, 107405 CrossRef CAS.
- Y. R. Ma, H. Q. Qu, W. N. Wang, Z. Y. Guo, Y. Q. Yu, F. Liu, B. Yu, M. E. Tian, Z. J. Li, B. Li and L. Wang, Chin. Chem. Lett., 2024, 35, 108352 CrossRef CAS.
- X. Y. Sun, D. Wang, Z. Wen, W. Li, H. S. Zhou and P. He, Chem. Commun., 2024, 60, 8772–8775 RSC.
- Y. G. Deng, W. W. Zhang, C. Ma, C. Li, Y. X. Hong, Y. C. Sun, J. Q. Niu, S. Guo and S. S. Yao, J. Energy Storage, 2025, 113, 115640 CrossRef CAS.
- T. Hu, W. Lian, K. Hu, Q. Li, X. Cui, T. Yao and L. Shen, Nano-Micro Lett., 2025, 17, 5 CrossRef CAS PubMed.
- D. Yu, D. Na, H. Kim, D. I. Son, D. D. Lee and I. Seo, J. Alloys Compd., 2025, 1010, 177722 CrossRef CAS.
- Z. Lin, Q. Yao, S. Yang, H. Song, Z. Yu, Z. Li, S. Chen, M. Wang, Z. Wang, G. Zhang, L. Zhang, Z. Yu, X. Song, K. Zhou, W. Li, L. Yu, J. Xu and K. Chen, Adv. Funct. Mater., 2025, 35, 2424110 CrossRef CAS.
- K. M. Naik, A. K. Chourasia and C. S. Sharma, J. Power Sources, 2024, 608, 234623 CrossRef CAS.
- Y. Liu, X. Wu, H. Qu, G. Lu, Y. Chen, B. Lu, Y. Song, G. Zhou and H.-M. Cheng, Adv. Mater., 2025, 37, 2411652 CrossRef CAS PubMed.
- C. J. Fetrow, C. Carugati, X. D. Zhou and S. Wei, Energy Storage Mater., 2022, 45, 911–933 CrossRef.
- Hagopian, M. L. Doublet and J. S. Filhol, Energy Environ. Sci., 2020, 13, 5186–5197 RSC.
- Z. Wang, Y. Mao, L. Sheng and C. Sun, ACS Appl. Mater. Interfaces, 2024, 16, 12706–12716 CrossRef CAS PubMed.
- J. Xu, Y. Xie, J. Zheng, C. Liu, Y. Lai and Z. Zhang, J. Electroanal. Chem., 2021, 903, 115853 CrossRef CAS.
- Y. Mao, X. Chen, H. Cheng, Y. Lu, J. Xie, T. Zhang, J. Tu, X. Xu, T. Zhu and X. Zhao, Energy Environ. Mater., 2022, 5, 572–581 CrossRef CAS.
- G. Qi, J. Zhang, J. Cheng, L. Chen, Y. Su and B. Wang, Small, 2024, 20, 2309064 CrossRef CAS PubMed.
- Z. Wang, L. Deng, X. R. Yang, J. X. Lin, D. Q. Cao, J. K. Liu, Z. Tong, J. Zhang, G. Y. Bai, Y. X. Luo, Z. W. Yin, Y. Zhou and J. Li, Adv. Funct. Mater., 2024, 34, 2404137 CrossRef CAS.
- M. K. Aslam, H. Wang, S. Chen, Q. Li and J. Duan, Mater. Today Energy, 2023, 31, 101196 CrossRef CAS.
- Z. Tong, S. B. Wang, M. H. Fang, Y. T. Lin, K. T. Tsai, S. Y. Tsai, L. C. Yin, S. F. Hu and R. S. Liu, Nano Energy, 2021, 85, 105972 CrossRef CAS.
- Bharti, D. Deb, G. Achutharao and A. J. Bhattacharyya, J. Phys. Chem. C, 2024, 128, 11543–11551 CrossRef CAS.
- W. Lyu, X. Yu, Y. Lv, A. M. Rao, J. Zhou and B. Lu, Adv. Mater., 2024, 36, 2305795 CrossRef PubMed.
- J. Han, A. Mariani, S. Passerini and A. Varzi, Energy Environ. Sci., 2023, 16, 1480–1501 RSC.
- S. Chen, M. Zhang, P. Zou, B. Sun and S. Tao, Energy Environ. Sci., 2022, 15, 1805–1839 RSC.
- E. Im, J. H. Ryu, K. Baek, G. D. Moon and S. J. Kang, Energy Storage Mater., 2021, 37, 424–432 CrossRef.
- X. Yang, D. Zhang, L. Zhao, C. Peng, K. Ren, C. Xu, P. Liu, Y. Zhou, Y. Lei, B. Yang, D. Xue and F. Liang, Adv. Energy Mater., 2024, 14, 2304365 CrossRef CAS.
- J. Zhang, D. Ding, Q. Fang, J. Cheng, H. Xiao and B. Wang, Angew. Chem., Int. Ed., 2025, 64, e202420892 CrossRef CAS PubMed.
- S. Li, Y. Wang, B. Dan, Z. Wang, X. Liu and D. Wang, Chem. Eng. J., 2024, 498, 155801 CrossRef CAS.
- S. S. Zhang, J. Power Sources, 2007, 164, 351–364 CrossRef CAS.
- X. Tian, H. Liu, B. Cao, P. Zhang, Y. Z. Li, Y. C. Ou, H. N. Cui, M. Y. Xu and B. Xu, ACS Nano, 2024, 18, 35738–35748 CrossRef CAS PubMed.
- R. Mao, Y. Liu, P. Shu, B. Lu, B. Chen, Y. Chen, Y. Song, Y. Jia, Z. Zheng, Q. Peng and G. Zhou, EcoMat, 2024, 6, e12449 CrossRef CAS.
- M. Wu, J. Y. Kim, H. Park, D. Y. Kim, K. M. Cho, E. Lim, O. B. Chae, S. Choi, Y. Kang, J. Kim and H.-T. Jung, ACS Appl. Mater. Interfaces, 2020, 12, 32633–32641 CrossRef CAS PubMed.
- S. M. Chen, K. Yang, H. Y. Zhu, J. Wang, Y. Gong, H. X. Li, M. M. Wang, W. G. Zhao, Y. C. Ji, F. Pan, S. R. P. Silva, Y. L. Zhao and L. Y. Yang, Nano Energy, 2023, 117, 108872 CrossRef CAS.
- Q. Deng, Y. Yang, C. Mao, T. Wang, Z. Fang, W. Yan, K. Yin and Y. Zhang, Adv. Energy Mater., 2022, 12, 2103667 CrossRef CAS.
- Z. Zheng, X. Zheng, J. Jiang, Q. Zhang, P. Li, C. Li, Q. Gu, L. Wei, K. Konstantinov, W. Yang, Y. Chen and J. Wang, ACS Appl. Mater. Interfaces, 2024, 16, 17657–17665 CrossRef CAS PubMed.
- Y. Wang, Y. Cheng, B. Chen, J. Zhou, H. Xie, Y. Fan, J. Sha, E. Liu, F. He, C. He, W. Hu and N. Zhao, Energy Storage Mater., 2024, 71, 103655 CrossRef.
- C. F. Xu, K. W. Zhang, D. Zhang, S. L. Chang, F. Liang, P. F. Yan, Y. C. Yao, T. Qu, J. Zhan, W. H. Ma, B. Yang, Y. N. Dai and X. L. Sun, Nano Energy, 2020, 68, 104318 CrossRef CAS.
- E. Im, J. Mun, S. Pourasad, K. Baek, J. H. Ha, Y. E. Durmus, H. Tempel, R.-A. Eichel, G. Lee, G. D. Moon and S. J. Kang, Chem. Eng. J., 2023, 476, 146740 CrossRef CAS.
- L. Habib, G. Suo, J. Li, C. Lin, X. Luo, G. Yang, Z. K. Kalkozova and K. Naseem, Energy Storage Mater., 2026, 84, 104863 CrossRef.
- L. Habib, G. Suo, N. Habib, M. Aqdas, C. Lin, J. Li and S. Javed, Energy Storage Mater., 2025, 139, 118821 CrossRef CAS.
- J. Li, G. Suo, C. Lin, J. Li, X. Luo, G. Yang, L. Habib, Z. K. Kalkozova and K. Naseem, Chem. Eng. J., 2026, 529, 173031 CrossRef CAS.
- K. Pichaimuthu, A. Jena, H. Chang, C. Su, S.-F. Hu and R.-S. Liu, ACS Appl. Mater. Interfaces, 2022, 14, 5834–5842 CrossRef CAS PubMed.
- C. Xu, H. Wang, J. Zhan, Y. Kang and F. Liang, J. Power Sources, 2018, 520, 230909 CrossRef.
- J. Kim, S. Lee, S. Kim, C. Y. Park, J. Kim, K. B. Lee, J. Oh and J. Lee, ACS Energy Lett., 2023, 8, 5079–5087 CrossRef CAS.
- W. Zhang, C. Hu, Z. Guo and L. Dai, Angew. Chem., 2020, 132, 3498–3502 CrossRef.
- J. Lin, W. Song, C. Xiao, J. Ding, Z. Huang, C. Zhong, J. Ding and W. Hu, Carbon Energy, 2023, 5, e313 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |