Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
A review of C-based cathode composites for advanced K–Se batteries
Narasimharao Kitchamsetti *a,
Sungwook Mhin*b and
HyukSu Han
*a,
Sungwook Mhin*b and
HyukSu Han *c
*c
aDepartment of Microsystems, University of South-Eastern Norway, Campus Vestfold, Raveien 215, 3184 Borre, Norway. E-mail: kinar9318@usn.no
bDepartment of Energy and Materials Engineering, Dongguk University, Seoul, 04620, Republic of Korea. E-mail: swmhin@dongguk.edu
cDivision of Materials Science and Engineering, Hanyang University, Seoul, 04763, Republic of Korea. E-mail: hyuksuhan@hanyang.ac.kr
First published on 23rd April 2026
Abstract
Potassium–selenium batteries (KSeBs) are increasingly recognized as attractive energy-storage candidates thanks to their high theoretical energy density and relatively low production cost. Yet, their practical deployment remains limited. Issues such as the migration of soluble polyselenides, sluggish redox kinetics, and substantial cathode volume changes during charge–discharge cycles lead to poor coulombic efficiency (CE) and short service life. To overcome these obstacles, carbon (C)-based materials have received significant attention. Owing to their excellent electrical conductivity, tunable porosity, and strong chemical stability, they have been widely explored as cathode hosts for KSeBs and have shown considerable promise in mitigating the aforementioned drawbacks. Given the rapid expansion of research in this field, this review provides a comprehensive overview of recent progress in C-based cathodes for KSeBs. We first outline the fundamental characteristics of KSeBs, the major technical challenges, and common optimization approaches, such as confining Se within C scaffolds, introducing chemisorptive host structures, and employing catalytic strategies to accelerate redox reactions. We then examine how different fabrication methods and micro/nanostructural designs influence electrochemical behavior. Lastly, we offer perspectives on future directions for the intelligent design of C-based cathodes in KSeBs and broader applications in other alkali–metal chalcogen battery systems.
Broader contextDriven by the growing demand for sustainable, high energy density electrochemical storage systems, the exploration of alternative alkali–metal batteries beyond lithium has become a critical research focus. Potassium-based batteries are particularly attractive due to the natural abundance, low cost, and favorable redox potential of potassium, while also offering fast ionic transport in conventional electrolytes. Within this family, potassium–selenium batteries (KSeBs) have emerged as promising candidates because selenium delivers high theoretical energy density through multi-electron redox reactions and exhibits higher intrinsic electrical conductivity than sulfur, enabling improved reaction kinetics. However, the practical development of KSeBs is severely constrained by intrinsic challenges, including the dissolution and shuttling of potassium polyselenides, sluggish conversion kinetics, pronounced cathode volume expansion, and unstable electrode–electrolyte interfaces, all of which result in low coulombic efficiency and rapid capacity decay. Carbon-based materials have therefore attracted extensive attention as advanced cathode hosts, owing to their excellent electrical conductivity, tunable pore structures, lightweight nature, and outstanding chemical stability. Rationally designed carbon frameworks can physically confine selenium species, buffer volumetric changes, suppress polyselenide migration, and provide efficient electron/ion transport pathways. Furthermore, heteroatom doping, defect engineering, and catalytic functionalization of carbon hosts enable strong chemical adsorption and accelerated redox kinetics. In this context, a comprehensive and critical assessment of recent advances in carbon-based cathodes is highly timely for guiding future KSeB development. |
1. Introduction
Lithium-ion batteries (LIBs) are currently the dominant power source for portable electronics, electric vehicles, and smart grid technologies due to their long cycle life and low self-discharge rates.1–4 Despite these advantages, the limited availability of lithium, accounting for only about 0.0017 wt% of the Earth's crust, combined with its uneven geographic distribution and rising cost, poses significant constraints on the continued expansion of LIB technology.5–8 Given these limitations, potassium-ion batteries (PIBs) have drawn growing interest. PIBs share similar electrochemical mechanisms with LIBs, yet benefit from the natural abundance and low cost of potassium (K), which constitutes roughly 2.1–2.6 wt% of the crust.9–12 Moreover, the relatively high radius of K+ ions often results in notable volume changes in electrodes and sluggish ion diffusion, prompting ongoing efforts to develop more advanced K-based energy-storage systems.13–16 The electrochemical behavior of PIBs is strongly influenced by the properties of their cathode materials, especially in terms of capacity and redox potential.17–19 Achieving cathodes that combine high capacity with elevated operating voltages is essential for advancing PIB technology, yet this goal remains difficult to realize. Among the various options, selenium (Se)-based cathodes have emerged as attractive candidates owing to their exceptionally greater theoretical volumetric capacity (3253 mAh g−1) and relatively high electrical conductivity (1 × 10−3 S m−1).20 Compared with lithium–selenium (Li–Se) and sodium–selenium (Na–Se) systems, where extensive polyselenide dissolution and slow reaction kinetics severely hinder performance, KSeBs benefit from the intrinsically lower solubility of K polyselenides (KPSes).21,22 This reduced solubility effectively mitigates the shuttle effect, enabling improved coulombic efficiency (CE) and extended cycling stability. As a result, KSeBs show strong potential as competitive next-generation energy-storage technologies.23,24The KSeBs operate through a reversible conversion reaction between Se and K ions.25 Despite this straightforward mechanism, its real-world performance is hindered by several intrinsic issues of the Se cathode, such as polyselenide shuttling, poor electrical conductivity, sluggish redox kinetics, and pronounced volume fluctuations during cycling.26,27 To tackle these limitations, various strategies, such as constructing Se–C composites and designing Se/S hybrid molecular structures, have been proposed.28,29 Among these approaches, C-based composites stand out as particularly promising cathode hosts for KSeBs. Their tunable porous architectures and excellent electrical conductivity allow them to effectively confine Se physically while providing chemical adsorption sites for polyselenides.30,31 To date, extensive research has centered on integrating C-based composites into the cathodes of KSeBs. For instance, Yao and colleagues designed a Se@NOPC-CNT cathode that delivered a capacity of 335 mAh g−1 even after 700 cycles at 0.8 A g−1. This durable behavior was attributed to combined contributions of N/O heteroatom doping, a 3D conductive C network that facilitates rapid electron and ion transport, and abundant nanopores that effectively confine polyselenides.32 In another study, Lim's team prepared a Se@h-NMCNF composite cathode whose hierarchical porous C nanofiber (CNF) framework greatly enhanced Se utilization, resulting in improved rate capability and long-term cycling stability.33 Several review articles have discussed the progress of C-based composites in KSeBs. For example, Huang et al. provided a broad overview of recent developments in K-chalcogen systems (S, Se, and Te), including various cathode composites.24 Du and co-workers also offered a systematic summary of biomass-derived C used in (Li, Na, K)–Se battery technologies.34 Although several review articles have discussed Se-based batteries,35 K-chalcogen systems,36 or C materials for energy storage,37 most of these works focus on broader battery chemistries or other alkali–metal systems such as Li–Se and Na–Se batteries.38 A systematic review that specifically examines C-based cathode engineering strategies for KSeBs, including Se confinement, chemisorptive host design, and electrocatalytic regulation, remains relatively limited.
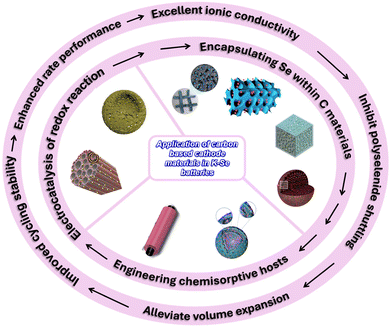
In this report, we offer an in-depth investigation of recent progress in employing C-based composites as cathodes for high-performance KSeBs, covering key strategies such as C encapsulation of Se, the design of chemisorptive host structures, and catalytic enhancement of redox reactions, as illustrated in Fig. 1. We also discuss the working mechanism of KSeBs, identify the major challenges, and outline current optimization approaches, while emphasizing the existing gaps in mechanistic understanding, particularly for systems involving small-molecule Se. Furthermore, we offer future perspectives for rational development of C-based cathodes aimed at improving KSeB performance. It is anticipated that this review will stimulate broader interest in this research direction and help accelerate the practical implementation of KSeB technology.
2. Mechanistic insights, performance barriers, and enhancement strategies in KSeBs
Because of their inherent benefits, Se-based cathodes have attracted widespread interest. However, in Li–Se and Na–Se battery systems, severe polyselenide dissolution and shuttling, along with slow reaction kinetics, often result in limited cycling durability.39 In comparison, KSeBs show much lower solubility of Se and KPSes in carbonate electrolytes, which effectively mitigates a shuttle effect and leads to higher energy density, better CE, and enhanced long-term stability.40 As a result, rechargeable KSeBs are emerging as promising candidates for next-generation energy-storage devices. This section provides an overview of their key characteristics, major limitations, and possible strategies for performance optimization.2.1. Mechanistic insights and performance barriers of KSeBs
A typical rechargeable KSeB is composed of a Se-based cathode, a K metal anode, an electrolyte, and a separator.41 Its electrochemical operation relies on the oxidation of K and the corresponding reduction of Se. The specific reaction pathway is closely linked to the structural form of the Se species. Ring-shaped Se8 generally converts to K2Se through a two-step process involving polyselenide intermediates, whereas polymeric Sen can either react directly to form K2Se or proceed through intermediate polyselenide states. In contrast, small-molecule Se follows a solid–solid conversion route to yield K2Se, a mechanism that is advantageous for suppressing the shuttle effect.41 Although notable progress has been made in uncovering the reaction mechanisms of KSeBs, their full electrochemical behavior, particularly in systems employing small-molecule Se, has not yet been fully clarified. Despite these uncertainties, Se remains highly appealing for K–Se chemistry because of its outstanding properties, such as high electronic conductivity (1 × 10−3 S m−1), a large theoretical capacity of 675 mAh g−1, and an impressive volumetric capacity of 3253 mAh cm−3.42 Importantly, Se-based cathodes exhibit exceptional stability in carbonate electrolytes. Their direct conversion pathway eliminates the construction of soluble polyselenides, reducing parasitic reactions and greatly improving the interfacial compatibility between the electrode and electrolyte.26The electrochemical conversion pathway of Se in KSeBs is strongly influenced by the structural form of Se, the physicochemical properties of the host matrix, and the electrolyte environment. Bulk Se8 typically follows a stepwise conversion process involving soluble polyselenide intermediates (K2Sex, 2 ≤ x ≤ 8), which can lead to shuttle phenomena and capacity decay.43 In contrast, when Se is confined as polymeric chains or highly dispersed small-molecule species within porous C frameworks, the reaction pathway can shift toward shorter solid–solid conversion routes with reduced formation of soluble intermediates.44 Moreover, the polarity and catalytic activity of the host matrix play a crucial role in stabilizing Se intermediates and accelerating redox kinetics.45 Polar or heteroatom-doped C hosts can enhance chemisorption of polyselenides and regulate their conversion dynamics, while the electrolyte composition further modulates intermediate solubility and interfacial charge-transfer processes.46 Therefore, the interplay between the Se structural form, host polarity, and electrolyte chemistry collectively determines the dominant reaction pathway and the stability of reaction intermediates in KSeBs.
However, KSeBs continue to encounter several major obstacles. First, the shuttle effect of soluble polyselenides remains severe; these species form through reactions between the Se cathode and the K metal anode and lead to continuous loss of the active material and rapid capacity fading.47 Second, the Se electrode undergoes substantial structural stress because K+ insertion induces nearly a 400% volume expansion, arising from the density difference between elemental Se and the resulting K2Sex phases, ultimately deteriorating electrode integrity.47 Third, the intrinsically large atomic radius of Se slows down redox kinetics, thereby limiting CE and long-term cycling durability.27,40
2.2. Enhancement strategies of KSeBs
In response to the challenges outlined above, various cathode-optimization strategies have been introduced for KSeBs. These approaches include confining Se within conductive C matrices, designing hosts capable of strong chemisorption toward intermediate species, and incorporating electrocatalytic components to accelerate redox reactions (Fig. 2).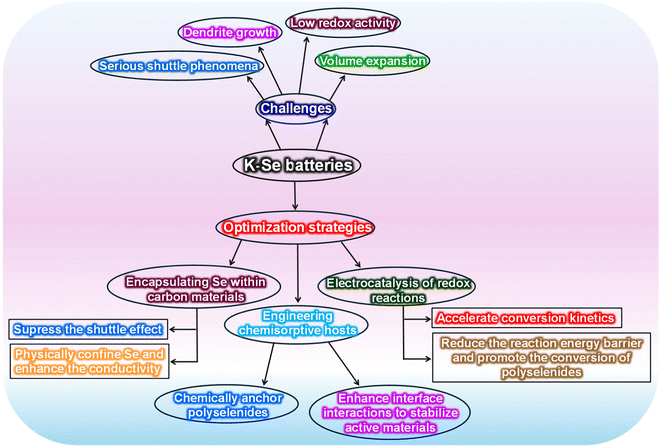 | ||
| Fig. 2 Schematic illustration of the major challenges in KSeBs and the corresponding cathode-engineering strategies developed to address them. | ||
Research on KSeBs has largely centered on strategies aimed at suppressing polyselenide shuttling, accelerating reaction kinetics, and enhancing electrode durability. Most current efforts focus on cathode engineering, with C-based encapsulation of Se receiving considerable attention due to its structural benefits.31 Porous C frameworks can buffer the substantial volume change of Se and provide efficient ion pathways through their hierarchical pores,42 while hollow C structures confine active species within a closed space, thereby reducing polyselenide dissolution.48 Additionally, heteroatom doping strengthens chemical interactions with polyselenides, improves electronic conductivity, and promotes redox conversion.32 Biomass-derived carbons further offer natural porosity and inherent heteroatoms, enabling strong electrochemical behavior at lower cost.49 Recent studies also highlight that restricting Se to smaller molecular units (Se2–4) reduces phase-transition-induced stress and enhances reaction kinetics, providing an alternative to the sluggish conversion of the conventional Se8 structure.50 Moreover, polar hosts and single-atom catalysts contribute to stronger adsorption and more efficient transformation of polyselenides.51 Beyond these approaches, electrocatalytic strategies can further decrease the conversion energy barrier and accelerate redox kinetics.52
3. Application of C-based cathodes for KSeBs
The cathode remains one of the primary bottlenecks for KSeBs, making the stabilization of Se-based electrodes crucial for achieving enhanced electrochemical behavior. Despite Se's higher electronic conductivity compared to S, elemental Se alone is unsuitable as a cathode material and must be integrated with conductive hosts to support efficient electron transport.53 During cycling, the reversible conversion of Se forms soluble KPSes that readily diffuse into the electrolyte, causing rapid capacity decay and eventual cell failure.24 Addressing this issue requires the design of spatially confining host structures that can suppress the polyselenide shuttle, with cathodes that provide both high conductivity for charge transport and strong confinement of active Se species. C-based composites have emerged as highly promising components for KSeBs due to their excellent conductivity, tunable porosity, large specific surface area (SSA), and strong chemical stability. These intrinsic features position C composites as effective platforms for addressing the challenges discussed above. This section outlines design principles for C-based cathodes in KSeBs, focusing on three main strategies: Se encapsulation, construction of chemisorptive host structures, and electrocatalytic enhancement of redox reactions. Performance comparisons of various C-based cathode architectures are summarized in Table 1.| Composites | Se content (wt %)/loading (mg cm−2) | Preparation approach | Design strategy | Electrolytea | Voltage window (V) | Electrochemical performance (DCb, CDc, CRRd, CNe) | Ref. |
|---|---|---|---|---|---|---|---|
| 1C = 675 mAh g−1.a EC: ethylene carbonate; DEC: diethyl carbonate; PC: propylene carbonate; DME: 1,2-dimethoxyethane; and DOL: 1,3-dioxolane.b DC: discharge capacity (mAh g−1).c CD: current density.d CRR: capacity retention rate.e CN: cycle number. | |||||||
| Se–O-PCS | 51/∼1.0 | Template and melt-diffusion | Encapsulating Se within C composites | 1 M KFSI in EC/DEC | 0.5–3.0 | 514, 0.2C, ∼80%, 45 | 42 |
| Se@HCR | 55/∼0.8 | Solvothermal and melt-diffusion | 0.8 M KPF6 in EC/DEC | 581.4, 0.1C, 86%, 200 | 54 | ||
| NSHPC-700/Se | 57–59/1.8–2.1 | Spray pyrolysis and melt-diffusion | 1 M KFSI in EC/DEC | 461, 0.2C, ∼95%, 120 | 55 | ||
| Se@NO-nanocage/CNT | 49/— | Solution method and melt-diffusion | 0.7 M KPF6 in EC/DEC | 470, 1.5C, ∼82%, 200 | 48 | ||
| Se@N-HCNS | 45.6/— | Melt-diffusion | 610, 0.3C, ∼89%, 100 | 56 | |||
| HPC/Se | —/1–1.2 | 1 M KPF6 in EC/DEC | 527, 0.1C, ∼85%, 100 | 57 | |||
| Se@NCHS | 60/∼1.2 | 3 M KFSI in EC/DEC | ∼320, 0.5C, ∼62%, 500 | 58 | |||
| Se@NOPC-CNT | 60/∼1.5 | 0.7 M KPF6 in EC/DEC | ∼420, 1.2C, ∼80%, 700 | 32 | |||
| MSTC@Se | 1 M KFSI in EC/DEC | ∼300, 3C, ∼70%, 2000 | 25 | ||||
| Se2–3/Se4–7@MMCFs | 49.4/1.2–1.5 | Electrospinning and melt-diffusion | 0.7 M KPF6 in EC/DEC | 443, 1.5C, 90%, 2000 | 59 | ||
| 3D-N-CPC/Se | 54/1.0–1.5 | Freeze-drying and pyrolysis | 0.85 M KPF6 in EC/DEC | ∼590, 2C, ∼39%, 800 | 28 | ||
| FNDPC@Se | 40/— | Melt-diffusion | 0.8 M KPF6 in EC/DEC | ∼150, 3C, ∼72%, 500 | 49 | ||
| Se@NPCFs | 62/∼1.5 | Electrospinning and melt-diffusion | 0.7 M KPF6 in EC/DEC | ∼580, 0.75C, ∼63%, 1670 | 50 | ||
| Se/MDPC | 53/1.0–1.8 | Melt-diffusion | 1 M KPF6 in EC/PC | 0.7–2.3 | ∼280, 0.2C, ∼50%, 100 | 60 | |
| Se@NPCS | 60/— | Encapsulating Se within C composites | 1 M KPF6 in EC/DEC | 0.5–2.5 | 604, 0.5C, ∼52%, 300 | 61 | |
| Se50/SO-HPC3 | 50/∼1.5 | ∼620, 0.5C, ∼88%, 1700 | 62 | ||||
| Se-HPC | 42/0.5–1.0 | Freeze-drying and melt-diffusion | 1 M KPF6 in EC/PC | ∼580, 0.2C, ∼80%, 100 | 63 | ||
| Se/HHPC | 47/∼1.5 | Melt-diffusion | 0.1 M KTFSI in DOL/DME | 589, 0.2C, 39%, 200 | 64 | ||
| PC/Se/GO | 40/— | 0.8 M KPF6 in EC/DEC | 426.3, 0.5C, 74%, 150 | 65 | |||
| C-PAN-Se | 40/0.3–0.5 | Mixed sintering | 1 M KPF6 in EC/PC | 0.7–2.3 | 652, 0.2C, ∼61%, 100 | 39 | |
| Se@AHCS | 51.7/∼1.0 | Melt-diffusion | 0.8 M KPF6 in EC/DEC | 0.5–2.8 | 547.8, 0.3C, 59%, 300 | 22 | |
| Se@PWC-NS | 9.75/∼3.4 | Two-step carbonization and melt-diffusion | 1 M KTFSI in EC/DEC | 0.001–3.0 | 642.7, 0.2C, 90%, 200 | 29 | |
| CFS@N–C | 55/0.8–1.0 | Self-assembly | 4 M KFSI in DME | — | 392, 0.15C, ∼97%, 500 | 41 | |
| Se@h-NMCNF | 60/∼1.9 | Electrospinning and melt-diffusion | 3 M KFSI in EC/DEC | — | 384, 0.5C, 54.9%, 1000 | 33 | |
| C-DWHCSs/Se | 62/1.6–2.0 | Template and melt-diffusion | Engineering chemisorptive hosts | 1 M KPF6 in EC/DEC | 0.5–3.0 | 612.5, 0.2C, 91%, 100 | 51 |
| Se@MoSe2-HPC | 35/— | Freeze-dried carbonization and melt-diffusion | 1 M KPF6 in EC/PC | 572.4, 1C, 56%, 500 | 66 | ||
| Se/CoNiSe2-NR | 6.05/1.0–1.5 (typical); 1.7–3.8 (3D printing) | Melt-diffusion and 3D printing | Electrocatalysis of redox reactions | 1 M KPF6 in EC/DEC | 0.5–3.0 | ∼450, 0.1C, ∼87%, 150 | 21 |
| Se@P–N–C@Mo2C | 58/∼1.0 | Electrospinning and melt-diffusion | 0.7 M KPF6 in EC/DEC | ∼300, 1C, ∼78%, 220 | 52 | ||
| Se-W2N/C | 45/∼1.0 | Melt-diffusion | 1 M KTFSI in EC/DEC | 540.7, 0.15C, 74%, 100 | 67 | ||
It should be noted that direct comparisons of electrochemical performances across different KSeB studies must be interpreted with caution because experimental conditions are often not standardized. Key parameters such as Se loading, electrolyte-to-Se (E/S) ratio, current density definition (based on total electrode mass or active material mass), and cell configuration can vary significantly between reports. These differences may strongly influence the reported specific capacities, rate capabilities, and cycling stability. Therefore, while the summarized data provide a useful overview of the recent progress in cathode design, the performance metrics should be considered within the context of the specific testing conditions reported in each study. Establishing more consistent reporting standards, particularly with respect to practical parameters such as high Se loading and lean electrolyte conditions, will be important for enabling more reliable cross-study comparisons and accelerating the development of practically relevant KSeB systems.
Beyond qualitative descriptions, several measurable physicochemical parameters have been shown to correlate strongly with the electrochemical performance of C-based cathodes in KSeBs.48 First, pore size distribution plays a critical role in balancing Se confinement and ion transport. Micropores (<2 nm) effectively confine Se and suppress polyselenide dissolution, while mesopores (2–10 nm) facilitate electrolyte penetration and K+ diffusion, leading to improved rate capability.21 Second, the binding energy between Se species and the host matrix, which can be tuned through heteroatom doping (e.g., N, S, or P), determines the stability of intermediate polyselenides. Moderate binding energies are generally favorable because they stabilize intermediates without significantly impeding their redox conversion.52 Third, the intrinsic electronic conductivity of the C framework directly influences charge-transfer kinetics and high-rate performance. Finally, catalytically active sites introduced by heteroatom dopants or metal–N–C structures can reduce the activation energy of polyselenide conversion reactions, thereby improving reaction reversibility and overall conversion efficiency.66 These structure–property–performance correlations highlight the importance of simultaneously optimizing the pore architecture, surface chemistry, and electronic transport in designing advanced C-based cathodes.
3.1. Incorporating Se into C composites
Incorporating Se into C frameworks helps overcome its inherent limitations by taking advantage of C's superior conductivity, which promotes fast electron transfer and enhances overall reaction kinetics. The porous architecture of C enables high Se loading and ensures efficient electrolyte penetration, while its large SSA offers abundant electroactive sites. Moreover, C matrices can restrict polyselenide migration and accommodate the large volume changes of Se during cycling, helping to preserve electrode stability. Presently, a variety of C materials, such as porous C,56,57 hollow C,63 doped C,58 and biomass-derived C47 have been used in Se-based cathodes.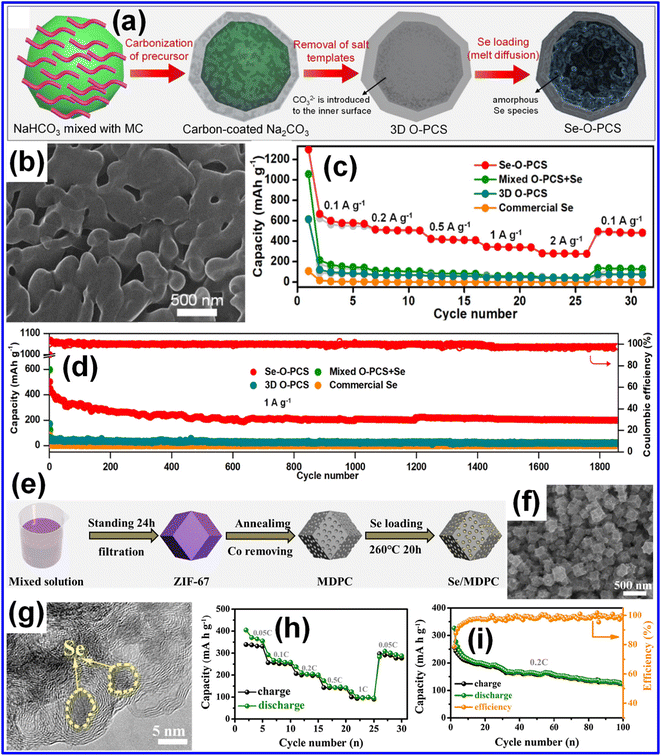 | ||
| Fig. 3 (a) Pictorial representation of the fabrication of the Se–O-PCS composite. (b) SEM micrograph of the resulting Se–O-PCS structure. (c) Comparison of rate capabilities of different electrode materials. (d) Long-term cycling behavior at 1.0 A g−1 for the examined electrodes. Reproduced from ref. 42, Copyright 2021, Elsevier B.V. (e) Illustration of the fabrication route for the Se/MDPC composite. (f) FE-SEM micrograph of the Se/MDPC composite. (g) TEM micrograph showing the internal morphology, (h) rate capability, and (i) cycling stability of the Se/MDPC composite. Reproduced from ref. 60, Copyright 2019, Elsevier B.V. | ||
MOFs have emerged as excellent templates for producing porous C materials.5,71,72 Owing to their metal–ligand coordination structures, these frameworks yield highly porous C networks after pyrolysis and subsequent removal of metal species. Such derived carbons can effectively confine Se73 while preserving the structural features and large SSAs characteristic of their parent MOFs.24 As shown in Fig. 3e, Huang and co-workers utilized ZIF-67 to generate N-doped porous C (MDPC) through controlled carbonization followed by acid etching.60 A Se/MDPC composite containing 53 wt% Se was then obtained using a melt-diffusion strategy. The SEM micrograph in Fig. 3f confirms that the MDPC framework preserves its polyhedral morphology, while the TEM micrograph (Fig. 3g) reveals uniform infiltration of Se throughout the hierarchical pore system. Consequently, the Se/MDPC half-cell achieved discharge capacities of 405, 267, 211, 150, and 101 mAh g−1 at 0.05, 0.1, 0.2, 0.5, and 1C, respectively, with the CE approaching 90% (Fig. 3h). In addition, the cathode demonstrated strong cycling durability, retaining ∼130 mAh g−1 and >95% CE after 100 cycles (Fig. 3i). These outstanding electrochemical behaviors stem from the hierarchical porosity of MDPC, which buffers the large ∼400% volume expansion of Se, while N doping improves conductivity and the confined structure suppresses polyselenide migration. Using a comparable strategy, Huang and co-workers produced a Se host (N-HCNS) by combining ZIF-8 derived microporous C with N-doped porous C nanosheets (NSs) by the melt-diffusion technique.56 This hybrid structure mitigated polyselenide shuttling, improved Se usage, and buffered the volume variation during cycling. In parallel, Sun and co-workers synthesized interconnected microporous C nanorods (Se@HCR) employing Zn-MOF-74, which exhibited enhanced rate performance owing to their optimized porous network.54 Together, these works underscore the importance of MOF-derived C frameworks for overcoming the principal limitations of Se cathodes, especially regarding polyselenide confinement and volume-expansion management.
Besides MOF-derived carbons, other engineered C materials featuring controlled porosity or heteroatom incorporation have also yielded strong KSeB performance. Huang et al. developed a N-doped porous C sponge,61 while Lim et al.33 produced N-doped CNFs (h-NMCNFs) via electrospinning, both showing improved electrochemical properties due to their optimized porous configurations. Furthermore, Kim and co-workers synthesized N,S co-doped HPC microspheres (NSHPC) through spray pyrolysis followed by chemical activation at 650–800 °C (Fig. 4a).55 As illustrated in Fig. 4b and c, NSHPC-700 and its Se-loaded counterpart maintained their hierarchical pore networks. By fine-tuning micropore volume and the ratio between micro- and mesopores, the NSHPC-700/Se cathode achieved outstanding rate performance. As illustrated in Fig. 4d, the NSHPC-700/Se cathode delivered stable reversible capacities of 492, 461, 356, 256, and 137 mAh g−1 at 0.1, 0.2, 0.5, 1, and 2C, respectively. When the current density was returned to 0.1C, the capacity recovered to 482 mAh g−1, indicating minimal degradation. Fig. 4e further shows that the electrode exhibits excellent cycling stability at 0.2C, delivering a reversible capacity of 436 mAh g−1 over 120 cycles. The CE quickly reached 98.5% and consistently remained above 99% in subsequent cycles. This superior electrochemical behavior results from the large micropore volume that confines Se and the optimized micro/mesopore ratio that promotes efficient mass transport. The Se-filled micropores restrict polyselenide formation/dissolution, while the mesoporous channels enhance ion mobility and buffer the volume changes occurring during cycling. In a related study, Zhao's team introduced a S/O-co-doped HPC (SO-HPC) as a robust host matrix for Se storage.62 Following the melt-diffusion incorporation of Se, the resulting Se50/SO-HPC3 composite retained its initial porous morphology (Fig. 4f), confirming the uniform permeation of Se into the hierarchical pore network. Electrochemical evaluation revealed an impressive reversible capacity of 450 mAh g−1 at 0.5C, which was sustained for 1700 cycles with nearly 100% CE (Fig. 4g). The outstanding behavior is attributed to the cooperative physicochemical adsorption provided by S and O dopants, which strengthens Se immobilization, curtails anion dissolution, and mitigates parasitic reactions.
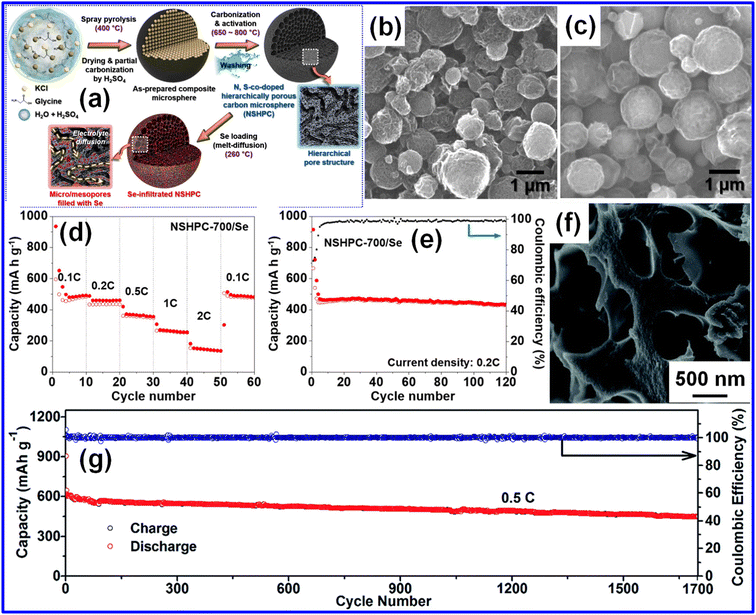 | ||
| Fig. 4 (a) Schematic illustration of the preparation of Se-loaded N, S co-doped hierarchical porous C. SEM micrographs of (b) NSHPC-700 and (c) NSHPC-700/Se. (d) Rate capability and (e) cycling behavior of NSHPC-700/Se. Reproduced from ref. 55, Copyright 2020, American Chemical Society. (f) SEM micrograph and (g) cycling behavior of Se50/SO-HPC3 at 0.5C. Reproduced from ref. 62, Copyright 2019, Royal Society of Chemistry. | ||
To fabricate hollow C architectures, numerous studies have focused on tailoring MOF-derived nanostructures. Huang's team, for instance, developed Se-HPC using MOF-based porous C polyhedra produced through freeze-drying followed by a melt-diffusion process.63 As a versatile class of engineered materials, MOFs have found extensive application in gas adsorption and separation. As illustrated in Fig. 5a, Zhou and co-workers constructed an NO-nanocage/CNT composite by synthesizing ZIF-8 via a solution method employing Zn(NO3)2 and 2-methylimidazole as precursors.48 TEM and SEM images (Fig. 5b) confirm its polyhedral morphology. The resulting Se@NO-nanocage/CNT exhibited remarkable rate performance, carrying 304 mAh g−1 at 5 A g−1 and maintaining 80% of its capacity at 2 A g−1 (Fig. 5c). Furthermore, its abundant micro/mesoporosity, N/O doping, and high conductivity endowed it with excellent K-storage capabilities. Impressively, after 3500 cycles, the material retained a capacity of 274 mAh g−1 (Fig. 5d). To enhance electronic conductivity and thereby improve rate capability, Jia's group designed a CFS@N–C composite utilizing hollow C spheres through a self-assembly strategy (Fig. 5e).41 As shown in the SEM image (Fig. 5f), the material features uniformly distributed hollow C spheres. The CFS@N–C electrode delivered stable cycling over 500 cycles at 100 mA g−1 while maintaining a high reversible capacity (Fig. 5g). This strong performance stems from its self-assembled architecture, which boosts K-ion storage, reinforces structural integrity, and increases the electrode–electrolyte interfacial area. In related work, hollow C spheres have been directly employed as hosts. Ding's group synthesized Se@AHCS using a conventional procedure, where the O-containing functional groups and defect-rich graphitic domains within the C network offered strong chemisorption spots for Se intermediates, thus inhibiting polyselenide diffusion.22
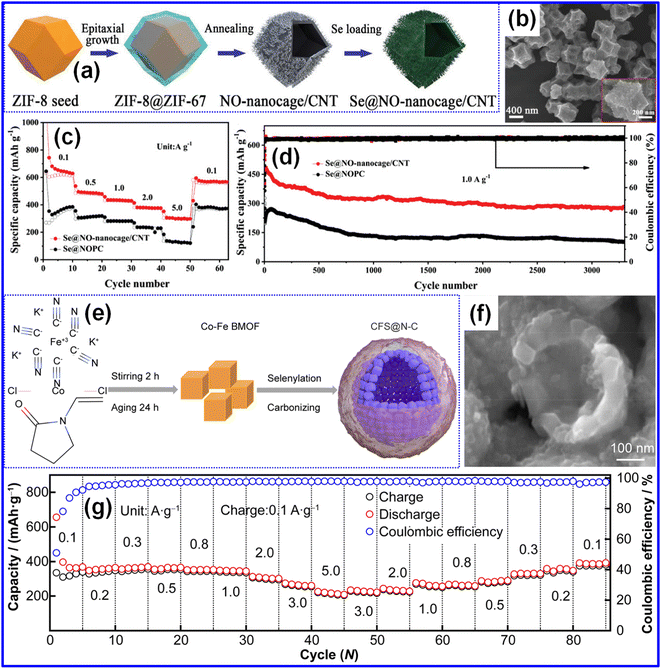 | ||
| Fig. 5 (a) Pictorial representation of the formation process of the Se@NO-nanocage/CNT composite. (b) SEM micrograph and (c) rate capability of the Se@NO-nanocage/CNT cathode. (d) Cycling behavior and CE at 1 A g−1 over 3500 cycles. Reproduced from ref. 48, Copyright 2020, Wiley-VCH. (e) Diagram illustrating the preparation route of the CFS@N–C composite. (f) SEM micrograph and (g) rate capability of the CFS@N–C electrode. Reproduced from ref. 41, Copyright 2021, Springer Nature. | ||
Beyond single-atom doping, dual-doped C composites can introduce synergistic interactions that further enhance cathode functionality.32,64 In particular, N and O co-doping is known to improve electronic carrier density, surface polarity, and chemical binding strength.59,64 Yao and co-workers reported the fabrication of porous C NSs intertwined with CNTs and simultaneously doped with N and O (referred to as NOPC-CNT), which were subsequently infused with Se to produce a Se@NOPC-CNT cathode.32 The synthesis pathway for the thin-film cathode is illustrated in Fig. 6a. Electrochemical tests revealed that the Se@NOPC-CNT electrode maintained excellent stability over 700 cycles at 0.8 A g−1, delivering a reversible capacity of 335 mAh g−1 and maintaining nearly 100% CE (Fig. 6b). Furthermore, when the current density was returned to 0.1 A g−1 after high-rate cycling at 5 A g−1, the electrode recovered a capacity of 546 mAh g−1 with negligible degradation (Fig. 6c), highlighting its fast reaction kinetics and robust structural durability. These superior properties stem from the flexible and conductive nature of the NOPC-CNT host, which accommodates volume fluctuations and offers strong adsorption sites for Se species. To improve electronic conductivity and facilitate faster reaction kinetics, both essential for achieving high capacity and long-term cycling performance, Du's group synthesized an N/O co-doped hollow porous C material (MSTC) by employing carrageenan and SiO2 as precursors through a molten-salt approach. The Se was subsequently incorporated using a melt-diffusion process, yielding an efficient cathode material for KSeBs.25 The resulting 3D, interconnected, freestanding C film provided a robust conductive network and superior mechanical integrity, which translated into excellent electrochemical behavior. As illustrated in Fig. 6d, the MSTC@Se electrode delivered 507 mAh g−1 at 0.1 A g−1 and retained 201 mAh g−1 even after 2000 cycles at 2 A g−1 (Fig. 6e). These enhancements stem from the optimized structure that supports high Se loading (∼60 wt%) due to strong chemical affinity from the abundant N and O functionalities. Additionally, the N-strengthened O-sites within the C framework contributed to improved conductivity and effectively mitigated the polyselenide shuttle. Li and colleagues further developed a flexible KSeB by employing N/O doped porous CNFs (MMCFs) as the Se host.59 The engineered C framework provides effective physical confinement, limiting polyselenide generation and thereby enhancing cycling stability.
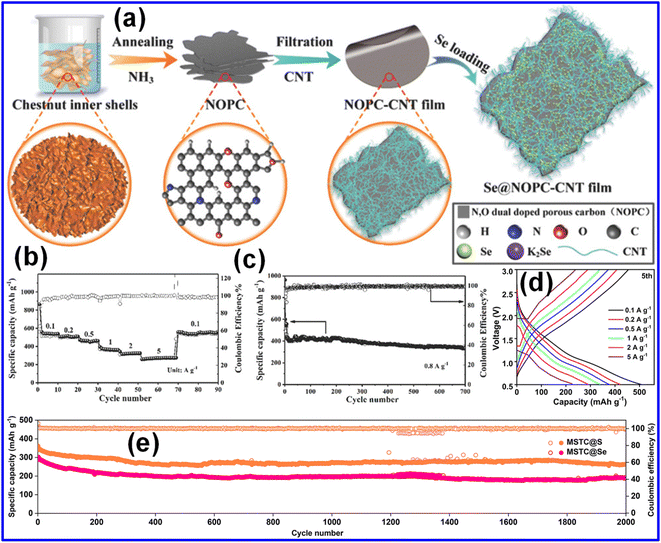 | ||
| Fig. 6 (a) Illustration of the synthesis route for the Se@NOPC-CNT composite. (b) Rate capability measured across current densities ranging from 0.1 to 5 A g−1. (c) Cycling behavior of Se@NOPC-CNTs at 0.8 A g−1. Reproduced from ref. 32, Copyright 2018, Wiley-VCH. (d) GCD profiles of the MSTC@Se electrode under various current densities. (e) Cycling behavior of MSTC@S and MSTC@Se at 2 A g−1. Reproduced from ref. 25, Copyright 2025, Royal Society of Chemistry. | ||
Biomass-derived porous carbons are commonly utilized as Se hosts in cathode configurations. Ma et al. recently developed a 3D N-doped cross-linked C (3D-N-CPC) from eggplant biomass via freeze-drying followed by pyrolysis (Fig. 7a).28 The resulting material exhibited a highly interconnected hierarchical microporous network. Following Se incorporation, the 3D-N-CPC framework remained structurally intact, with Se uniformly distributed throughout the continuous pore channels (Fig. 7b and c). In another study, Qiu and colleagues produced a foam-type N-doped C (FNDPC) using natural cotton as the precursor (Fig. 7d and e),49 demonstrating a promising approach for developing low-cost Se host materials. In addition, efforts have been made to further enhance cathode properties by integrating biomass-derived C with graphene or similar C frameworks to serve as a dual-host system for Se. Cai and co-workers introduced a composite cathode, PC/Se/GO, synthesized by amalgamating walnut shell-derived biochar with GO (Fig. 7f).65 The resulting composite displayed an irregular cubic morphology (Fig. 7g). This architecture significantly improved the electrochemical performance, delivering a reversible capacity of 426.3 mAh g−1 in the second cycle (Fig. 7h). The enhanced behavior is primarily attributed to the uniform dispersion of Se within the PC/Se matrix.
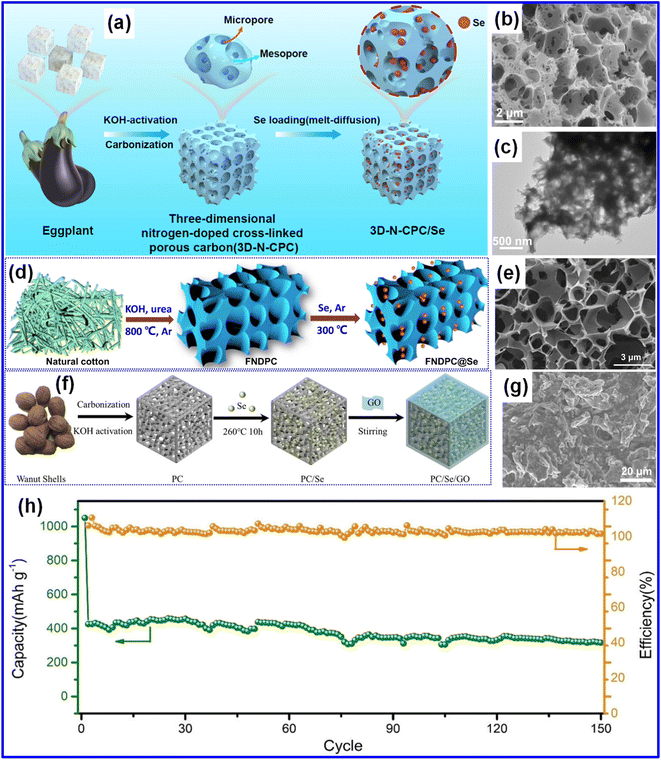 | ||
| Fig. 7 (a) Schematic representation of the synthesis of the 3D-N-CPC/Se composite. (b and c) SEM and TEM micrographs of 3D-N-CPC/Se. Reproduced from ref. 28, Copyright 2024, American Chemical Society. (d) Illustration of the fabrication process for FNDPC@Se. (e) TEM micrograph of FNDPC. Reproduced from ref. 49, Copyright 2020, Elsevier B.V. (f) Schematic of the preparation approach for PC/Se/GO. (g) SEM micrograph of PC/Se/GO. (h) Cycling behavior of PC/Se/GO measured at 0.5C. Reproduced from ref. 65, Copyright 2020, Wiley-VCH. | ||
Xu and co-workers developed a flexible KSeB using small-molecule Se confined within thin N-doped CNF films (Se@NPCFs) as a cathode composite (Fig. 8a).77 The resulting fibers exhibited a distinctive pod-shaped morphology rich in hierarchical pores. Benefiting from this architecture, the Se@NPCFs delivered outstanding cycling durability, maintaining 367 mAh g−1 at 0.5 A g−1 for 1670 cycles with nearly 100% CE (Fig. 8b). These remarkable electrochemical characteristics stem from the 1D N-doped C “pea-pod” architecture, which facilitates electrolyte infiltration and K+ transport, accommodates volume changes, and lowers internal resistance. To further improve the cycling stability of Se-based cathodes, Wang's group infused small-molecule Se into the intrinsic nanopores of N/S doped walnut-shell-derived C through sequential carbonization and melt-diffusion processes (Fig. 8c).29 As shown in Fig. 8d, the Se@PWC-NS electrode preserved a capacity of 211.7 mAh g−1 after 3500 cycles. This stability is attributed to the N/S-co-doped biomass C host, which effectively mitigates Se volume expansion and enhances structural robustness. The C-derived composites have also been widely utilized as supports to augment the characteristics of small-molecule Se cathodes.39
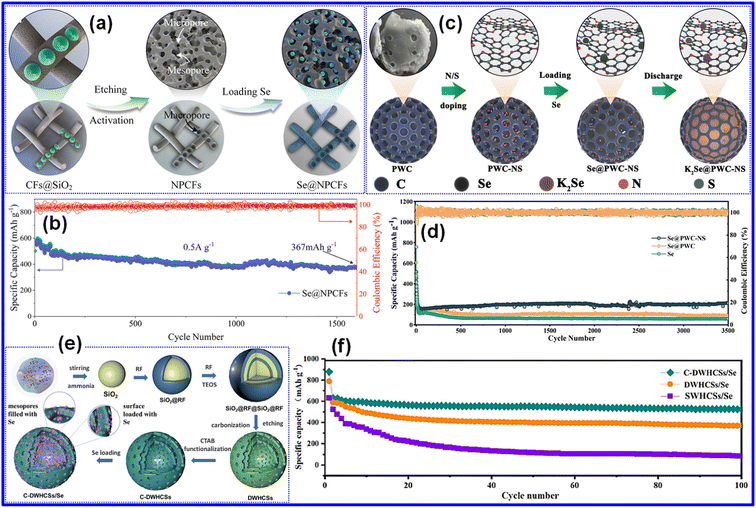 | ||
| Fig. 8 (a) Illustration depicting the preparation route of the Se@NPCF composite cathode. (b) Cycling behavior of Se@NPCFs at 0.5 A g−1. Reproduced from ref. 77, Copyright 2020, Wiley-VCH. (c) Pictorial representation of the fabrication strategy for the Se@PWC-NS material. (d) Cycling behavior comparison between Se@PWC-NS and pristine Se composites at 10C. Reproduced from ref. 29, Copyright 2022, Elsevier B.V. (e) Diagram of the fabrication procedure for C-DWHCSs/Se. (f) Cycling results of different composites evaluated at 0.1C. Reproduced from ref. 51, Copyright 2022, Elsevier B.V. | ||
Although various C host engineering strategies have been reported, their relative importance strongly depends on the operating regime of KSeBs.56 Under high Se loading conditions, structural confinement provided by hierarchical porous C frameworks becomes critical to maintain effective electron/ion transport pathways and to accommodate the large volume changes associated with Se conversion reactions.55 In lean electrolyte systems, where polyselenide dissolution and shuttle effects become more pronounced, polar or heteroatom-doped C hosts play a dominant role by enhancing chemisorption interactions with Se intermediates and stabilizing them at the cathode interface.58 In contrast, under high-rate operation, the reaction kinetics of polyselenide conversion become the primary limitation; therefore, highly conductive C frameworks and electrocatalytically active sites are particularly important for reducing activation barriers and accelerating redox reactions.60 These observations suggest that an effective cathode design should simultaneously integrate structural confinement, chemical adsorption, and catalytic regulation, with the relative contribution of each mechanism depending on the targeted operating conditions. However, despite the promising electrochemical performance achieved through such structural and chemical engineering, many reported C host architectures rely on complex synthetic strategies, including hard/soft templating, MOF derivation, or multi-step heteroatom doping processes.60,61,64 While these approaches enable precise regulation of pore size distribution, surface functionality, and catalytic activity, their scalability and cost-effectiveness remain important considerations for practical applications. For example, templating methods often require additional template removal steps and chemical treatments,75 whereas MOF-derived carbons typically involve expensive precursors and high-temperature pyrolysis processes, which may limit their feasibility for large-scale electrode manufacturing.77 In contrast, more scalable approaches such as the use of biomass-derived carbons, direct pyrolysis of polymer precursors, or template-free porous C synthesis offer attractive alternatives due to their lower cost, simplified processing, and compatibility with industrial production.60,75 Therefore, future research should aim to balance structural sophistication with synthetic simplicity in order to develop cathode host materials that combine high electrochemical performance with practical manufacturability.
3.2. Engineering chemisorptive hosts
An alternative effective approach for enhancing the durability of Se-based cathodes involves engineering chemisorptive host materials.26 By tailoring the C matrix or introducing specific surface functional groups, these hosts create strong chemical binding sites for Se and polyselenides. Such interactions help suppress polyselenide dissolution and the associated shuttle effect, strengthen the attachment of Se to the C scaffold, and minimize loss of active Se moieties during repeated cycling.23,47Among the various implementations of chemisorptive host design, Wu and colleagues developed dual-wall hollow C spheres (DWHCSs) modified with CTAB as an effective Se host (C-DWHCSs), which were subsequently used to assemble the C-DWHCSs/Se cathode (Fig. 8e).51 Owing to their double-layered hollow architecture and high SSA, the C-DWHCSs demonstrated strong affinity toward polyselenides, enabling a higher Se loading in the cathode. As a result, the C-DWHCSs/Se electrode delivered a capacity of 411 mAh g−1 at 0.1C after 100 cycles (Fig. 8f). Notable cycling stability is attributed to the large SSA and hollow dual-wall structure, which provide abundant storage sites for Se and support higher Se mass loading. Additionally, the internal cavity acts as a buffer to accommodate volume fluctuations during cycling. In addition, Yang's group incorporated the polar compound MoSe2 into a chitosan-derived hierarchical porous C framework (MoSe2-HPC) using a straightforward freeze-drying and carbonization route, producing an effective host for Se with excellent cycling stability and rate capability.66 Collectively, these findings highlight the strong promise of chemisorptive host materials in enhancing the electrochemical characteristics of Se-based cathodes.
3.3. Electrocatalysis of redox processes
Beyond the aforementioned approaches, recent work has focused on incorporating catalysts to promote the adsorption and conversion of polyselenides.21 Evidence indicates that the cycling durability and rate behavior of KSeBs can be greatly enhanced by designing C-based architectures featuring porous structures in combination with suitable electrocatalysts. Collectively, these studies offer valuable theoretical guidance and experimental support for the development of high-performance KSeBs.21,67Cho and co-workers fabricated a 1D N-doped C framework (P–N–C@Mo2C) through electrospinning followed by carbonization and KOH activation, and subsequently infused Se into this structure to obtain Se@P–N–C@Mo2C (Fig. 9a).52 The P–N–C@Mo2C scaffold featured a porous 1D morphology, which was retained after Se loading, as evidenced in Fig. 9b and c. This porous network facilitated electrolyte access and promoted efficient electron and ion transport, shortening diffusion pathways and accommodating the volume variation during cycling. The N-incorporation enhanced the electronic conductivity of the C matrix, while the ultrafine Mo2C nanoparticles (NPs) acted as catalysts capable of anchoring and accelerating the redox conversion of KPSes via electrophilic coupling with Mo and Se, thereby increasing active material employment.
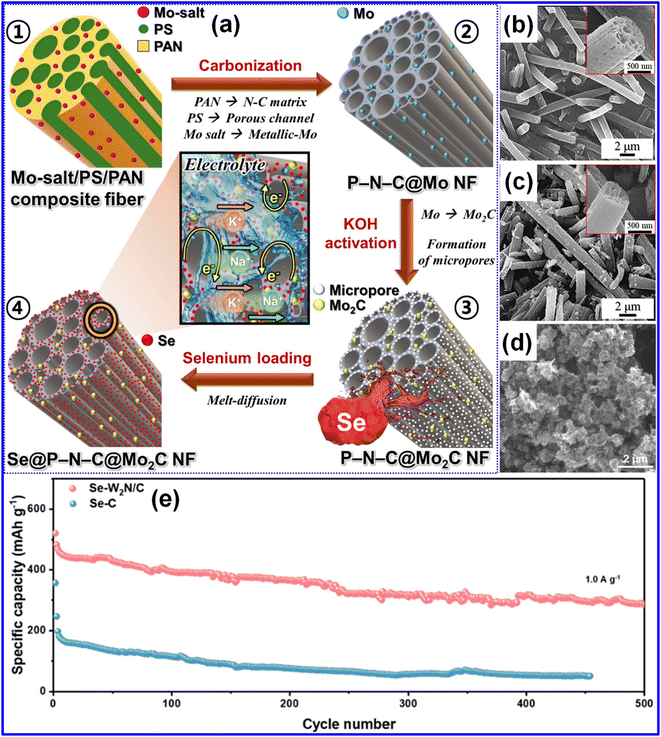 | ||
| Fig. 9 (a) Pictorial representation of the fabrication route for the Se@P–N–C@Mo2C composite. FE-SEM micrograph of (b) P–N–C@Mo2C and (c) Se@P–N–C@Mo2C NF framework. Reproduced from ref. 52, Copyright 2025, Elsevier B.V. (d) SEM imaging of the Se-W2N/C composite. (e) Cycling behaviors of Se-W2N/C and Se–C electrodes at 1 A g−1. Reproduced from ref. 67, Copyright 2023, American Chemical Society. | ||
Chemical adsorption and electrocatalysis are often integrated within a single host material. Zhou and co-workers prepared a Se-W2N/C cathode using a melt-diffusion technique to uniformly distribute Se within a W2N/C porous C matrix.67 This configuration leverages both the catalytic activity of W2N and the adsorption capacity of the C framework. Owing to the microporous characteristics of the C host (Fig. 9d), sufficient space was available to accommodate Se species and buffer their volume variation during cycling. As illustrated in Fig. 9e, the Se-W2N/C electrode delivered a capacity of 286 mAh g−1 after 500 cycles at 1 A g−1. Its amended electrochemical characteristics stem from the strong chemical affinity and high conductivity of W2N, which help suppress polyselenide dissolution and shuttle effects. Additionally, W2N provides bidirectional catalytic activity, accelerating both oxidation and reduction reactions of Se.
3.4. Electrolyte chemistry and interfacial reaction thermodynamics
In KSeBs, electrolyte chemistry and interfacial reactions play a crucial role in determining reaction reversibility, shuttle suppression, and long-term cycling stability. During battery operation, the reduction of electrolyte components and dissolved polyselenide intermediates leads to the formation of a SEI on both the cathode surface and the K metal anode.78 The composition and stability of this SEI layer strongly depend on the electrolyte formulation, including the solvent system, salt concentration, and anion chemistry. Electrolytes with appropriate solvation structures can regulate the dissolution behavior of polyselenide intermediates and influence their transport within the electrolyte.79 In many cases, the formation of inorganic-rich SEI components such as K fluoride (KF),80 K carbonate (K2CO3),81 or K selenide (K2Se)35 has been shown to enhance interfacial stability and suppress the shuttle effect by limiting the diffusion of soluble intermediates.From a thermodynamic perspective, the reduction potentials of electrolyte solvents and salt anions determine the preferential pathways for SEI formation and the resulting interfacial composition,82 while kinetic factors such as interfacial charge-transfer resistance and the catalytic activity of the host material influence the rate of polyselenide conversion and the evolution of the SEI layer during repeated cycling.83 Although cathode design plays a central role in determining the electrochemical performance of KSeBs, the overall cell stability is also strongly influenced by the behavior of the anode. Dissolved polyselenide intermediates generated during Se conversion reactions can migrate through the electrolyte and react with the K metal anode, leading to parasitic side reactions, unstable SEI formation, and gradual loss of the active material, which ultimately contribute to the shuttle effect and capacity fading during long-term cycling.26 Therefore, effective suppression of polyselenide migration requires not only optimized cathode hosts but also stable anode interfaces. Recent studies have explored several strategies to address this challenge, including the construction of artificial SEI layers on K metal,80 electrolyte engineering to regulate polyselenide solubility,84 and the development of alternative anode materials such as C-based or alloy-type hosts that exhibit improved interfacial stability.36,85 Considering these coupled cathode–anode interactions is therefore essential for achieving durable and high-performance KSeB systems.
4. Conclusions and outlook
Encapsulating Se within C-based matrices has emerged as a promising method for enhancing the electrochemical behavior of KSeBs. The C composites, characterized by their high conductivity, tunable porosity, and large SSAs, facilitate rapid electron transport and favorable reaction kinetics, while simultaneously restricting polyselenide dissolution, suppressing shuttle effects, and buffering Se's volume fluctuations during cycling.24 Various forms of C hosts have been explored, including porous C, hollow C, heteroatom-doped structures, biomass-derived composites, and Se/C composites constructed from small molecular precursors.47 Each offers distinct functional benefits: porous carbons accommodate Se expansion and enable efficient ion diffusion;57 hollow carbons confine polyselenides within their closed cavities;63 heteroatom doping introduces active sites that enhance both adsorption strength and electrical conductivity;64 biomass-derived carbons provide a sustainable and low-cost option;28 and small-molecule-derived Se/C systems improve reaction kinetics through size-dependent effects.50 Despite its advantages, this approach still faces several challenges. The melt-diffusion technique frequently results in nonuniform Se dispersion within the C host, ultimately limiting the effective utilization of the active material.60 The C obtained from MOFs or heteroatom-doping usually requires intricate and expensive preparation steps, restricting its suitability for large-scale fabrication.54 Biomass-derived carbons often possess inherently low electrochemical activity and thus demand further modification.28 In addition, molecular Se is prone to instability during storage and may gradually aggregate.77 At high Se loadings, the ability of C matrices to physically confine Se species is also substantially diminished.47Developing chemisorptive hosts is essential for establishing strong chemical interactions with Se and polyselenides by tailoring the chemical composition or surface functionality of C materials. Such strategies can overcome the limitations of purely physical confinement and enable more effective anchoring of polyselenides. Enhanced Se–host binding contributes to improved cycling stability and higher utilization of the active material in batteries.51 Nevertheless, several challenges remain. Precisely controlling both the density and strength of chemisorption sites is difficult; excessive heteroatom doping or incorporation of polar components may compromise the intrinsic conductivity of C; and under high Se loading, these chemical sites may become saturated, failing to prevent polyselenide shuttling.66 Furthermore, the synthesis of these additional components often involves complex and costly procedures, limiting scalability. The dynamic evolution of adsorption processes under real operating conditions, along with long-term stability and failure mechanisms, also lacks sufficient in situ investigation, leaving the long-term persistence of chemisorption efficacy uncertain.76
Incorporating electrocatalysts into KSeBs to strengthen polyselenide adsorption and conversion has recently emerged as a promising research direction. Constructing C-based composites with porous structures and catalytic sites has been shown to markedly enhance cycling stability and rate performance.76 This strategy effectively reduces the energy barriers associated with Se conversion and accelerates redox kinetics, leading to improved overall cell behavior.21 Nevertheless, electrocatalytic approaches still present notable challenges. Catalyst NPs tend to aggregate or detach during extended cycling due to insufficient interfacial bonding, resulting in diminished catalytic activity. Additionally, many synthesis routes involve high-temperature reduction or elaborate processes, increasing cost and limiting scalability. Critically, the catalytic mechanisms are not yet fully elucidated, and the absence of in situ characterization of intermediate polyselenides impedes rational catalyst optimization.67 Thus, developing low-cost, robust catalysts and advancing mechanism-oriented studies are vital for future practical deployment.
In conclusion, this review systematically discusses the recent progress in C-based cathodes for KSeBs, focusing on approaches such as Se encapsulation, the construction of chemisorptive C hosts, and electrocatalytic enhancement of redox reactions. These approaches collectively improve electronic conductivity, mitigate volume expansion, and suppress polyselenide shuttling through distinct mechanisms. C materials exhibit several intrinsic advantages for boosting the electrochemical performance of KSeBs: their tunable porosity and high SSA facilitate effective Se confinement and accommodate volume changes; their conductive frameworks promote rapid redox kinetics; and heteroatom doping introduces anchoring sites that impede polyselenide diffusion. Despite significant advancements, key challenges remain unresolved. Therefore, this review also outlines several recommendations for further optimizing C-based cathodes in KSeBs, as summarized in Fig. 10.
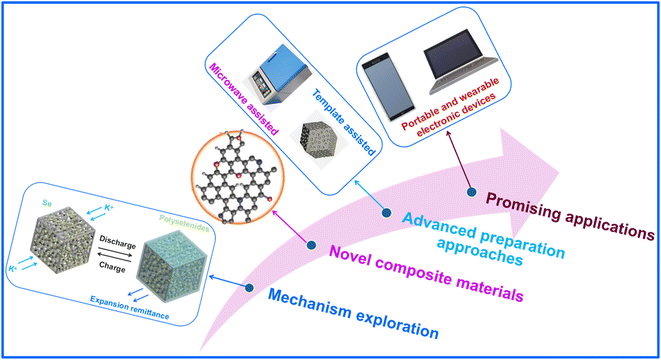
1. Mechanism investigation: in KSeB cathodes, C-based composites function primarily as Se hosts, conductive scaffolds, and polyselenide-trapping media, thereby enhancing performance through the combined effects of physical confinement, chemical adsorption, and conductivity improvement. However, the existing literature has yet to establish clear quantitative correlations among the pore characteristics of C materials and their Se uptake, the varying adsorption strengths of polyselenides at different heteroatom spots, and mechanistic relationship among Se phase-transition routes (especially the transient species involved in the Se8 → K2Se conversion) and the structural integrity of C matrices. To address these gaps, advanced in situ characterization methods, such as in situ XRD and Raman spectroscopy, should be employed to monitor the evolution of Se species and polyselenides. Coupling these techniques with theoretical calculations (e.g., DFT-based adsorption energy simulations)23,86 will enable deeper insight into dynamic interactions among C frameworks and Se, thus guiding the rational design of superior cathode composites.
2. Development of novel composite cathodes: current C-based cathode composites for KSeBs mainly involve Se/porous C, Se/heteroatom-doped C, Se/hollow C, and Se/biomass-derived C materials. Even though these systems display considerable structural diversity and have markedly improved electrochemical performance, there remains a strong need to design new C composites with even higher activity. Hybridizing C with graphene, MOFs and MXenes, or incorporating carbides and nitrides can yield composites with enhanced electrochemical behavior. In addition, the integration of artificial intelligence (AI) to accelerate material screening and optimization represents an emerging research frontier.23 Such advanced composites not only provide fast ion and electron transport pathways but also suppress polyselenide migration through strong chemical interactions, thereby improving overall battery stability. Moreover, refined control over morphological features offers further opportunities for performance enhancement, highlighting the great promise of these materials for future endeavors.
3. Advanced preparation approaches: the melt-diffusion technique has been extensively used for fabricating Se/C materials owing to its easiness, low cost, and suitability for batch scale production. By finely tuning the pore structure of the C host and optimizing diffusion temperature, both the homogeneity and loading efficiency of Se can be greatly improved. Recent studies have introduced optimized variations of this method. Microwave-assisted melting, for example, shortens processing time and mitigates Se volatilization, whereas template-directed melting enhances the confinement of Se through predefined pore architectures. In addition, the emergence of new C-based composite systems is expected to broaden the scope of synthesis and performance enhancement. Beyond melt-diffusion, alternative methods such as electrospinning are being actively explored. A comprehensive assessment of the strengths and limitations of each preparation route, followed by the selection of suitable strategies, will be essential for advancing the application of C-based cathode composites in KSeBs.
4. Operando characterization: a deeper mechanistic understanding of Se redox chemistry requires advanced operando characterization techniques capable of capturing the dynamic evolution of intermediate species during battery operation. In particular, the formation, transformation, and dissolution behavior of soluble polyselenides remain insufficiently understood under realistic electrochemical conditions. Operando spectroscopic techniques such as Raman spectroscopy, X-ray absorption spectroscopy (XAS), and UV-Vis spectroscopy can provide valuable insights into the chemical states and reaction pathways of Se species during cycling. Meanwhile, operando microscopy and synchrotron-based techniques, including transmission X-ray microscopy and X-ray diffraction, can help visualize structural evolution and phase transformations in the cathode. Combining these experimental approaches with electrochemical analysis would enable a more comprehensive understanding of polyselenide conversion mechanisms, interfacial reactions, and shuttle behavior. Future efforts integrating multimodal operando characterization with theoretical modeling will be essential for clarifying the complex reaction network of Se cathodes and guiding the rational design of high-performance Se-based batteries.
5. Promising applications: with the rapid development of flexible energy-storage systems, KSeBs are emerging as promising candidates for portable and wearable electronics owing to their high energy density and the cost advantages of K resources. By integrating self-supporting, flexible C-based cathodes, the inherent flexibility and conductivity of C composites can be fully exploited to deliver durable power to lightweight, foldable, and wearable devices. Additionally, the ionic conductivity of KSeBs remains relatively stable at low temperatures, enabling their use in wearable devices designed for extreme environments. As fabrication technologies for C-based cathodes continue to improve, scalable production of flexible KSeBs is anticipated, which will help drive the commercialization of advanced wearable systems. Furthermore, coupling KSeBs with energy-harvesting systems, for instance biomechanical or environmental energy capture, may significantly extend the operating life of self-powered sensors and other sustainable electronic platforms. Such integration offers enhanced functionality, improved reliability, and greater energy efficiency, paving the way for next-generation wearable electronics.
In conclusion, this work delivers a comprehensive summary of the recent progress in the development of C-based composites for KSeBs. Despite notable advancements, several key issues highlighted above still warrant deeper investigation and optimization. We anticipate that continued research efforts will gradually address these challenges, ultimately enabling the rational design of next-generation Se/C composites. Overall, the insights presented here are expected to support the broader applications of C-based composites in KSeBs and other advanced energy-storage technologies.
Conflicts of interest
The authors declare no conflict of Interest.Data availability
Data sharing is not applicable to this article as no new data were created or analyzed in this study.Acknowledgements
The authors acknowledge the financial support from the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (RS-2024-00449682). The authors acknowledge financial support from the National Research Foundation of Korea (NRF), funded by the Ministry of Science and ICT (RS-2024-00433118).References
- N. Kitchamsetti, D. Narsimulu, S. Payyavula, C. S. Chakra and A. L. F. de Barros, J. Energy Storage, 2025, 109, 115144 CrossRef CAS.
- J. S. Lee, K. W. Baek, N. Kitchamsetti, H. W. Kim and J. S. Cho, J. Mater. Sci. Technol., 2025, 223, 76–90 CrossRef CAS.
- N. Kitchamsetti, C. K. Mannem, D. Narsimulu, C. S. Chakra and A. L. F. de Barros, Chem. Eng. J., 2025, 505, 159354 CrossRef CAS.
- N. Kitchamsetti, A. L. F. de Barros and S. Mhin, Small Sci., 2025, 5, 2500331 CrossRef CAS PubMed.
- N. Kitchamsetti and J. S. Cho, J. Energy Storage, 2024, 84, 110947 CrossRef CAS.
- N. Kitchamsetti and J. S. Cho, J. Energy Storage, 2024, 80, 110293 CrossRef CAS.
- N. Kitchamsetti, S. Payyavula and J. S. Cho, J. Energy Storage, 2024, 98, 113143 CrossRef CAS.
- N. Kitchamsetti, C. K. Mannem, C. S. Chakra and A. L. F. de Barros, J. Energy Storage, 2025, 106, 114836 CrossRef CAS.
- N. Kitchamsetti and C. K. Mannem, J. Alloys Compd., 2025, 1027, 180664 CrossRef CAS.
- N. Kitchamsetti, A. L. F. de Barros, H. Han and S. Mhin, Adv. Sustainable Syst., 2025, 9, e00581 CrossRef CAS.
- N. Kitchamsetti, A. L. F. de Barros, H. Han and S. Mhin, J. Alloys Compd., 2025, 1044, 184389 CrossRef CAS.
- J. S. Lee, N. Kitchamsetti and J. S. Cho, Chem. Eng. J., 2024, 487, 150465 CrossRef CAS.
- N. Kitchamsetti, K. Kim, H. Han and S. Mhin, Nanomaterials, 2025, 15, 1554 CrossRef CAS PubMed.
- N. Kitchamsetti and S. Mhin, Polymers, 2025, 17, 2801 CrossRef CAS PubMed.
- N. Kitchamsetti and S. Mhin, J. Alloys Compd., 2025, 1046, 184742 CrossRef CAS.
- N. Kitchamsetti, A. L. F. de Barros and S. Mhin, Batteries Supercaps, 2025, 8, e202500482 CrossRef CAS.
- N. Kitchamsetti, J. Energy Storage, 2023, 73, 108958 CrossRef.
- N. Kitchamsetti, H. Han and S. Mhin, Batteries, 2025, 11, 206 CrossRef CAS.
- H. C. Kim, H. Kim, S. O. Moon, C. Jo and H. S. Park, J. Energy Chem., 2025, 105, 764–796 CrossRef CAS.
- W. Zhang, Y. Liu and Z. Guo, Sci. Adv., 2019, 5, eaav7412 CrossRef CAS PubMed.
- Y. Ding, J. Cai, Y. Sun, Z. Shi, Y. Yi, B. Liu and J. Sun, ACS Nano, 2022, 16, 3373–3382 CrossRef CAS PubMed.
- J. Ding, Y. Wang, Z. Huang, W. Song, C. Zhong, J. Ding and W. Hu, ACS Appl. Mater. Interfaces, 2022, 14, 6828–6840 CrossRef CAS PubMed.
- J. Feng, S. Yu, C. Shi, X. Tang, X. Zhao, S. Chen and J. Song, Adv. Funct. Mater., 2025, 35, 2422013 CrossRef CAS.
- X. Huang, J. Sun, L. Wang, X. Tong, S. X. Dou and Z. M. Wang, Small, 2021, 17, 2004369 CrossRef CAS PubMed.
- Y. Du, H. Fan, Y. Zhu, X. Zhang, D. Wei, C. Jin, Y. Cui and M. Lv, Green Chem., 2025, 27, 2309–2318 RSC.
- J. Park, H. Kim, A. Celeste, H. Park, S. Kansara, R. Zumpano, V. Piacentini, S. Brutti, A. Matic, M. Agostini and J. Y. Hwang, ACS Energy Lett., 2025, 10, 2512–2531 CrossRef CAS PubMed.
- X. Zhou, X. Rui and Y. Yu, Acc. Mater. Res., 2023, 4, 467–471 CrossRef CAS.
- Q. Ma, S. Chen, Y. Li, Z. Yan, Y. Liu and Z. Fang, ACS Appl. Nano Mater., 2024, 7, 16599–16608 CrossRef CAS.
- H. Wang, P. Wang, J. Cao, C. Liang and K. Yu, Electrochim. Acta, 2022, 432, 141158 CrossRef CAS.
- Y. M. Gao, Y. Liu, K. J. Feng, J. Q. Ma, Y. J. Miao, B. R. Xu, K. M. Pan, O. Akiyoshi, G. X. Wang, K. K. Zhang and Q. B. Zhang, Rare Met., 2024, 43, 1–19 CrossRef.
- H. Liu, Q. Wang, X. Sun, H. Gou, C. Zhang and G. Wang, Carbon, 2023, 203, 141–151 CrossRef CAS.
- Y. Yao, M. Chen, R. Xu, S. Zeng, H. Yang, S. Ye, F. Liu, X. Wu and Y. Yu, Adv. Mater., 2018, 30, 1805234 CrossRef PubMed.
- J. B. Lim, H. J. Kim, J. H. Na, J. K. Kim, S. Y. Jeong and S. K. Park, Rare Met., 2025, 44, 3839–3851 CrossRef CAS.
- Y. Du, S. Ma, J. Dai, J. Lin, X. Zhou, T. Chen and X. Gu, Batteries, 2022, 8, 123 CrossRef CAS.
- J. Park, H. Kim, A. Celeste, H. Park, S. Kansara, R. Zumpano, V. Piacentini, S. Brutti, A. Matic, M. Agostini and J. Y. Hwang, ACS Energy Lett., 2025, 10, 2512–2531 CrossRef CAS PubMed.
- N. Kitchamsetti, K. Kim, H. Han and S. Mhin, J. Energy Storage, 2026, 144, 119817 CrossRef CAS.
- Y. Du, S. Ma, J. Dai, J. Lin, X. Zhou, T. Chen and X. Gu, Batteries, 2022, 8, 123 CrossRef CAS.
- J. Feng, S. Yu, C. Shi, X. Tang, X. X. Zhao, S. Q. Chen and J. Song, Adv. Funct. Mater., 2025, 35, 2422013 CrossRef CAS.
- Y. Liu, Z. Tai, Q. Zhang, H. Wang, W. K. Pang, H. K. Liu, K. Konstantinov and Z. Guo, Nano Energy, 2017, 35, 36–43 CrossRef CAS.
- Q. Liu, W. Deng, Y. Pan and C. F. Sun, Chem. Sci., 2020, 11, 6045–6052 RSC.
- X. X. Jia, X. Z. Yu and B. A. Lu, Rare Met., 2021, 40, 2455–2463 CrossRef CAS.
- L. Zhou, Y. Cui, D. Kong, W. Feng, X. Gao, Y. Yan, H. Ren, H. Hu, Q. Xue, Z. Yan and W. Xing, J. Energy Chem., 2021, 61, 319–326 CrossRef CAS.
- Z. Wang, C. Qiu, Z. Chen, Z. Wu, H. X. Yu, L. Yan, L. Zhang, T. F. Yi and J. Shu, Adv. Mater., 2026, e22085 CrossRef CAS PubMed.
- H. Hao, T. Hutter, B. L. Boyce, J. Watt, P. Liu and D. Mitlin, Chem. Rev., 2022, 122, 8053–8125 CrossRef CAS PubMed.
- Y. Wei, M. Huang, Y. Wu, X. Tang, K. Yuan and Y. Chen, Adv. Funct. Mater., 2024, 34, 2404787 CrossRef CAS.
- S. Zhi, Q. Dai, H. Wang, D. Wu, L. Zhao, C. Hu and L. Dai, ACS Nano, 2025, 19, 29860–29897 CrossRef CAS PubMed.
- S. Lee, J. Lee, J. Kim, M. Agostini, S. Xiong, A. Matic and J. Y. Hwang, Energies, 2020, 13, 2791 CrossRef CAS.
- X. Zhou, L. Wang, Y. Yao, Y. Jiang, R. Xu, H. Wang, X. Wu and Y. Yu, Adv. Funct. Mater., 2020, 30, 2003871 CrossRef CAS.
- R. Qiu, R. Fei, T. Zhang, X. Liu, J. Jin, H. Fan, R. Wang, B. He, Y. Gong and H. Wang, Electrochim. Acta, 2020, 356, 136832 CrossRef CAS.
- R. Xu, Y. Yao, H. Wang, Y. Yuan, J. Wang, H. Yang, Y. Jiang, P. Shi, X. Wu, Z. Peng, Z. S. Wu, J. Lu and Y. Yu, Adv. Mater., 2020, 32, 2003879 CrossRef PubMed.
- X. Wu, X. Chen, H. Wu, B. Xie, D. Wang, R. Wang, X. Zhang, Y. Piao, G. Diao and M. Chen, Carbon, 2022, 187, 354–364 CrossRef CAS.
- S. W. Cho, H. H. Choi, T. G. Senthamaraikannan, D. H. Lim, G. D. Park, C. Cho, S. M. Jeong, R. Saroha and J. S. Cho, Chem. Eng. J., 2025, 512, 162456 CrossRef CAS.
- X. W. Luo, M. R. Zhou, M. J. Ran, D. X. Xun, J. L. He, Z. Y. Hu, H. S. H. Mohamed, J. Liu, L. H. Chen, Y. Li and B. L. Su, Chem. Eng. J., 2024, 488, 150885 CrossRef CAS.
- Y. Sun, J. Liao, J. Sun, L. Duan, Y. Du, J. Bao and X. Zhou, ACS Appl. Energy Mater., 2022, 5, 13023–13030 CrossRef CAS.
- J. K. Kim and Y. C. Kang, ACS Nano, 2020, 14, 13203–13216 CrossRef CAS PubMed.
- H. Huang, X. Luo, Y. Yao, X. Zhou, Y. Jiang, C. Guo, J. Liu, X. Wu and Y. Yu, InfoMat, 2021, 3, 421–431 CrossRef CAS.
- L. Cheng, C. Ma, W. Lu, X. Wang, H. Yue, D. Zhang and Z. Xing, Chem. Eng. J., 2022, 433, 133527 CrossRef CAS.
- H. J. Kim, J. H. Na and S. K. Park, Batteries, 2025, 11, 101 CrossRef CAS.
- D. Li, L. Wang, X. Cheng, Y. Yao, Y. Jiang, P. Shi, Y. Wu, X. Wu, C. Ma and Y. Yu, J. Energy Chem., 2021, 62, 581–589 CrossRef CAS.
- X. Huang, Q. Xu, W. Gao, T. Yang, R. Zhan, J. Deng, B. Guo, M. Tao, H. Liu and M. Xu, J. Colloid Interface Sci., 2019, 539, 326–331 CrossRef CAS PubMed.
- X. Huang, J. Deng, Y. Qi, D. Liu, Y. Wu, W. Gao, W. Zhong, F. Zhang, S. Bao and M. Xu, Inorg. Chem. Front., 2020, 7, 1182–1189 RSC.
- X. Zhao, L. Yin, Z. Yang, G. Chen, H. Yue, D. Zhang, Z. Sun and F. Li, J. Mater. Chem. A, 2019, 7, 21774–21782 RSC.
- X. Huang, W. Wang, J. Deng, W. Gao, D. Liu, Q. Ma and M. Xu, Inorg. Chem. Front., 2019, 6, 2118–2125 RSC.
- X. Zhao, D. Wang, H. Qi, Z. Li and H. Yue, J. Alloys Compd., 2023, 930, 167395 CrossRef CAS.
- R. Cai, X. Chen, P. Liu, T. Chen, W. Liu, X. Fan, B. Ouyang and K. Liu, ChemElectroChem, 2020, 7, 4477–4483 CrossRef CAS.
- W. Yang, S. Jamil and M. Xu, ChemistrySelect, 2023, 8, e202300470 CrossRef CAS.
- L. Zhou, B. Li, H. Hu, H. Liu, J. Nan, W. Wu, H. Xu, T. Cai, P. Liu, X. Li, Q. g. Xue, Z. Yan, Y. Cui and W. Xing, Energy Fuels, 2023, 37, 18103–18110 CrossRef CAS.
- F. Gao, X. A. Yue, X. Y. Xu, P. Xu, F. Zhang, H. S. Fan, Z. L. Wang, Y. T. Wu, X. Liu and Y. Zhang, Rare Met., 2023, 42, 2670–2678 CrossRef CAS.
- J. Li, C. Qian, Y. Hu, J. Huang, G. Chen, L. Cao, F. Wang, K. Kajiyoshi, Y. Zhao, Y. Liu, Z. Li, H. Yang and Z. Xu, Small, 2023, 19, 2302100 CrossRef CAS PubMed.
- P. Phogat, S. Rawat, S. Dey and M. Wan, J. Alloys Compd., 2025, 1020, 179544 CrossRef CAS.
- N. Kitchamsetti and D. Kim, J. Alloys Compd., 2023, 959, 170483 CrossRef CAS.
- N. Kitchamsetti and D. Kim, Electrochim. Acta, 2023, 441, 141824 CrossRef CAS.
- T. Li, J. Zheng, J. Wu, Z. Li, A. Xu, S. Wu and Y. Yan, Small, 2025, 21, 2409766 CrossRef CAS PubMed.
- W. N. Deng, Y. H. Li, D. F. Xu, W. Zhou, K. X. Xiang and H. Chen, Rare Met., 2022, 41, 3432–3445 CrossRef CAS.
- M. Khan, S. Yan, G. Li, J. Liu, M. R. Ali and Y. Wang, Arabian J. Chem., 2024, 18, 106077 CrossRef.
- X. L. Huang, Z. Guo, S. X. Dou and Z. M. Wang, Adv. Funct. Mater., 2021, 31, 2102326 CrossRef CAS.
- R. Xu, Y. Yao, H. Wang, Y. Yuan, J. Wang, H. Yang, Y. Jiang, P. Shi, X. Wu, Z. Peng, Z. S. Wu, J. Lu and Y. Yu, Adv. Mater., 2020, 32, 2003879 CrossRef PubMed.
- J. Zhang, M. Wu, Z. Zhang, N. Li, C. Tian and W. Xue, Angew. Chem., Int. Ed., 2025, 64, e11223 CrossRef CAS PubMed.
- Q. Liu, C. Zhang, F. Nie, W. Chen, W. Hao, H. Jin, D. Li, F. Yu and Y. Chen, Chem. Eng. J., 2025, 519, 165236 CrossRef CAS.
- L. Zhang, J. Xu, L. Li, H. Fei, Q. Wu, H. Yu, L. Yan, J. Shu and L. Zhang, Adv. Energy Mater., 2025, 15, 2500434 CrossRef CAS.
- Y. P. Han, P. He and Y. Xu, Joule, 2025, 9, 102053 CrossRef CAS.
- D. A. Skiba, A. M. Melemed and B. M. Gallant, J. Phys. Chem. C, 2026, 130, 3–17 CrossRef CAS.
- Q. Yang, X. Liu, J. Zhu, Y. Wen, Y. Zhang, G. Chen, K. Li, Y. Li and Y. Zhao, Chem. Eng. J., 2025, 522, 167105 CrossRef CAS.
- J. Li, J. Cong, H. Ji, T. Shi, L. Yuan and Y. Huang, J. Energy Chem., 2024, 95, 611–617 CrossRef CAS.
- Q. Yang, Q. Fan, J. Peng, S. Chou, H. Liu and J. Wang, Microstructures, 2023, 3, 2023013 CAS.
- B. Antil, S. Olhan and R. L. V. Wal, Minerals, 2025, 15, 262 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |