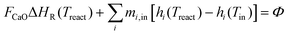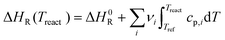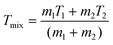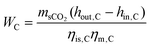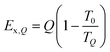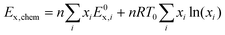Open Access Article
Open Access ArticleHybrid thermochemical Carnot battery with calcium looping, methane dry reforming, and supercritical carbon dioxide cycle: thermodynamic and techno-economic optimization
Jun Shena,
Yongqing Zhanga,
Zihao Sunb,
Dabiao Wangb,
Sheng Lia,
Wei Hanc,
Kezhen Zhangc,
Xiao Yangc,
Chenglong Yangc and
Qianghui Xu *a
*a
aSchool of Mechanical Engineering, Beijing Institute of Technology, Beijing, 100081, P. R. China. E-mail: xuqh12@bit.edu.cn
bSchool of Mechanical Engineering and Automation, Fuzhou University, Fuzhou 350108, China
cXi'an Thermal Power Research Institute Co., Ltd, Xi'an, 710032, China
First published on 12th March 2026
Abstract
This study proposes a polygeneration hybrid thermochemical Carnot battery that integrates calcium-looping thermochemical storage with methane dry reforming in an electrically heated calciner, coupled to a supercritical CO2 Brayton cycle for power discharge. Operating the calciner in a methane atmosphere lowers the calcination temperature by ∼200 °C, mitigates sorbent deactivation, and enables in situ CO2 utilization for synthesis-gas coproduction. A steady-state system model combined with multi-objective optimization is developed to examine efficiency–cost trade-offs. The Pareto-optimal compromise achieves a levelized cost of electricity (LCOE) of 0.94 ¥ per kWh, with a simplified recuperated cycle delivering 22.6 MW net output at a round-trip efficiency of 20.5%. Accounting for synthesis gas coproduct and ∼90% CO2 capture increases the overall energy and exergy efficiencies to 88.0% and 85.0%, respectively. Sensitivity analyses identify favorable operating windows of 30–50% CaO conversion and 5–10 °C minimum temperature approach in recuperators. Economies of scale are evident: as capacity expands from 1 to 100 MW, the LCOE declines from 2.37 to 0.83 ¥ per kWh. These findings demonstrate that incorporating methane dry reforming into calcium looping enables long-duration storage with inherent CO2 capture and valuable syngas coproduction, offering attractive thermo-economic trade-offs for grid-scale applications.
Broader contextThe global transition to carbon neutrality is critically dependent on solving two interconnected challenges: the large-scale integration of intermittent renewable energy through affordable long-duration storage, and the urgent need for decarbonizing industrial processes and chemical production. This study introduces a polygeneration Carnot battery that innovatively combines calcium-looping thermochemical energy storage with methane dry reforming and a supercritical CO2 power cycle. By operating the calciner in a methane atmosphere, the system simultaneously achieves a significant reduction in the calcination temperature, mitigates sorbent deactivation, and enables in situ CO2 utilization for syngas coproduction. The research demonstrates that this integrated approach not only provides a pathway for long-duration electricity storage with inherent CO2 capture, but also transforms the storage system into a multi-product energy hub. The favorable thermo-economic trade-offs and clear economies of scale revealed by the analysis highlight the potential of this technology to enhance grid flexibility, promote a circular carbon economy, and offer a viable strategy for the synergistic decarbonization of the power and chemical sectors. |
1. Introduction
Achieving net-zero emissions by 2050 will require a rapid scale-up of solar and wind generation, as projected by the International Energy Agency and the International Renewable Energy Agency.1,2 Yet integrating these variable resources poses significant operational challenges: the temporal mismatch between generation and demand complicates unit commitment and dispatch,3 driven by pronounced diurnal and seasonal variability.4 Therefore, the advancement of carbon capture, utilization, and storage (CCUS) and power-to-X (PtX) technologies is critical for addressing grid stability and achieving low-carbon emissions.5Carnot battery systems, considered a promising route for deep decarbonization, alleviate operational constraints of power-to-heat-to-power schemes by shifting electricity into a thermal reservoir; however, when implemented with sensible or latent heat storage they still suffer from relatively low volumetric energy density—typically around or below 100 kWh m−3 for practical materials—along with cumulative thermal losses during long holding times and limited functional flexibility in matching discharge profiles to grid needs.6–8 Thermochemical energy storage based on reversible reactions offers a compelling alternative with superior energy density approaching 500 kWh m−3, theoretically indefinite storage duration, and temperature-independent energy transport capabilities.9–11 Among these systems, calcium looping technology utilizing the reversible calcination–carbonation reaction of limestone stands out due to its ultra-high theoretical energy density of 889 kWh m−3, natural abundance of limestone precursors, and intrinsic carbon capture potential during the carbonation phase.12–14
Since its proposal, CaCO3/CaO cycling (CaL) systems have attracted significant research attention. Edwards et al.15 pioneered an integrated system combining concentrating solar power, CaCO3/CaO storage, and an open Brayton cycle, achieving 40–50% electrical efficiency with significantly reduced storage volume versus molten salt systems. Chacartegui et al.12 integrated CaL with a closed CO2 Brayton cycle, attaining over 45% efficiency through optimization. Ortiz et al.16 demonstrated direct integration of CaL discharge with closed CO2 cycles delivers optimal energy efficiency at 45–46%. Zhang et al.17 enhanced a 50 MW Concentrated Solar Power (CSP) plant by integrating CaL thermochemical storage with PV-driven compressed CO2 storage, doubling both energy efficiency from 14.6% to 29.4% and exergy efficiency from 15.7% to 31.6%, while achieving a levelized cost of electricity between $0.010 and $0.159 per kWh. Nevertheless, most CaL thermochemical storage and carbon capture integrations target CSP plants,14 which incur high solar field investment costs and exhibit low-efficiency radiation heat transfer between compound parabolic concentrators and calcination reactors.18 Therefore, this study directly employs electrical heating to meet the calcination reaction's energy demands and utilizes either coal-fired power plant steam Rankine cycles or Brayton cycles for energy discharge, establishing a novel CaL Carnot battery system for high-efficiency clean power generation.
Our previous study proposed a CaL Carnot battery integrated with a coal-fired power plant steam Rankine cycle.19 By supplying high-temperature heat to partially replace the boiler reheater duty, the system reduced coal consumption by approximately 7.07% relative to the reference plant, achieved a round-trip efficiency of 36.93%, and realized a specific energy consumption for CO2 capture of 3.66 MJ kg−1. Nonetheless, two limitations remain: (i) the inherently high calcination temperature accelerates sorbent deactivation, undermining long-term capacity retention; and (ii) although lowering the reheater load curbs coal use and emissions, the configuration cannot deliver zero-emission operation while it remains coupled to a coal-fired unit—necessitating either full decoupling from fossil heat or complete capture of residual stack CO2.
Simultaneously, Carnot battery systems based on calcium looping face progressive activity decay of calcium-based sorbents. Sun et al.20 demonstrated that multi-cycle operation promotes CaO sintering, leading to pore coarsening and reduced sorbent conversion. Benitez-Guerrero et al.21 revealed through microstructural analysis that rapid formation of dense CaCO3 product layers under high-temperature, high-pressure CO2 causes pore blockage and diffusion-limited reaction kinetics. To address sorbent deactivation, researchers have proposed several mitigation strategies, including: (i) recarbonation treatments to restore reactive surface area and slow sintering;22,23 (ii) the use of additives or supports to stabilize the microstructure and sustain multi-cycle capacity;24,25 and (iii) conducting calcination under a reducing atmosphere to suppress sintering and improve cyclic performance.26,27 Notably, Shao et al.28 reported that coupling calcium looping with dry reforming of methane enables calcination at 700 °C in a CH4 atmosphere—approximately 200 °C lower than conventional operation—thereby markedly suppressing high-temperature sorbent deactivation. However, the consequences of this deactivation mitigation for system-level performance have yet to be systematically evaluated.
Building upon the aforementioned issues and prior work,19 this study proposes an integrated hybrid thermochemical Carnot battery system coupling CaL thermochemical energy storage, dry reforming of methane, and a supercritical CO2 Brayton cycle (CaL-DRM-sCO2). The proposed system offers multifunctional benefits: accommodating surplus renewable electricity, enabling long-duration energy storage, capturing carbon, and co-producing synthesis gas (syngas) feedstock. Thermodynamic and economic models of the system were developed using Python. Key energy and economic performance metrics were defined, and a multi-objective optimization framework was established to identify Pareto-optimal trade-off solutions balancing efficiency and cost. The optimization results were evaluated through energy analysis, exergy analysis, and economic assessment to quantify system performance. Furthermore, the influence of key parameters on system behavior was investigated.
2. Models and methods
2.1 Integrated CaL-DRM-sCO2 system
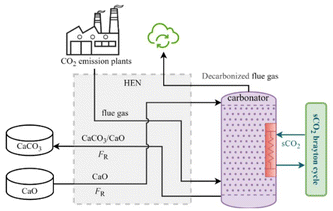
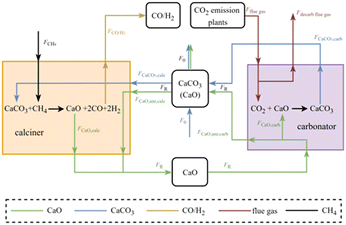
The charging subsystem of the proposed CaL-DRM-sCO2 system is depicted in Fig. 1. The calciner is electrically heated using excess renewable electricity from the grid (a coal gasification module similar to those implemented in integrated gasification combined cycle (IGCC) systems). A stream of partially carbonated solids (FR − F0), comprising CaCO3 and unreacted CaO (collectively termed ‘CaCO3 + CaO’), exits the CaCO3 storage tank and combines with a fresh limestone makeup stream (F0) before entering the Heat Exchange Network (HEN). Simultaneously with limestone introduction, an equivalent molar flow (F0) of circulating solids is purged from the CaCO3 storage tank. This purge compensates for adsorbent deactivation caused by high-temperature sintering and the decline in CO2 capture capacity resulting from repeated carbonation/calcination cycles. Within HEN, the combined solid feed and the CH4 stream (is assumed to be supplied via a dedicated gas pipeline), acting as cold streams, undergo thorough preheating prior to entering the calciner. Following calcination, the resulting CaO solids (FR) and the syngas mixture, primarily CO and H2, exit the calciner as hot streams. These hot streams then pass through HEN, transferring heat to preheat the incoming reactants while simultaneously cooling themselves. Subsequently, the cooled CaO solids and the cooled syngas mixture are separately conveyed to their respective storage tanks.The proposed CaL-DRM-sCO2 system distinguishes itself from the Calcium Looping integrated with a Coal-Fired Power Plant19 (CaL-CPP) primarily through methane injection into the calciner. This enables calcination decomposition to proceed within a methane atmosphere, which achieves three synergistic effects: approximately 200 °C reduction in calcination temperature,28 effective suppression of adsorbent thermal deactivation, and in situ reaction between released high-temperature CO2 and CH4. This integrated approach circumvents separate transportation and storage requirements for CO2. The produced syngas, primarily comprising carbon monoxide and hydrogen, offers versatile downstream applications, notably serving as valuable feedstock for processes such as methanol synthesis and Fischer–Tropsch synthesis.27 This syngas is generated via the governing reaction between carbon dioxide and methane, expressed as follows:
| CO2 + CH4 → 2CO + 2H2 ΔH = +247 kJ mol−1. | (1) |
Collectively, the integrated reaction scheme within the calciner is governed by the following equation:
| CaCO3 + CH4 → CaO + 2CO + 2H2 ΔH = +425 kJ mol−1. | (2) |
This study idealizes methane conversion as unity in order to decouple and highlight the thermochemical energy storage behavior of the CaCO3/CaO system; however, this assumption may underestimate additional heat transfer losses or efficiency penalties arising from incomplete reforming. Fig. 2 illustrates the discharging subsystem of the CaL-DRM-sCO2 hybrid thermochemical Carnot battery system. CaO solid particles are discharged from the storage tank, and CO2-rich flue gas from plants (including but not limited to coal-fired power plants, cement plants, and steel mills) enter the carbonator reactor together after being preheated in the HEN. Within the carbonator, CaO reacts with CO2 at approximately 650 °C through the carbonation reaction shown in:
| CaO + CO2 → CaCO3 ΔH = −178 kJ mol−1. | (3) |
This process converts the chemical energy of CaO solids into high-temperature heat while achieving approximately 90% carbon capture efficiency. The resulting partially carbonated solids, designated as CaCO3 + CaO, and the decarbonized flue gas are subsequently cooled in HEN. During cooling, they transfer heat to the incoming CaO particles and CO2-rich flue gas streams destined for the carbonator, thereby preheating these reactants. Critically, a key design feature involves direct heat transfer through the carbonator walls for thermal energy release during discharge. This integrated approach significantly simplifies HEN configuration on the carbonator side and enhances heat transfer efficiency compared to systems recovering heat from reaction products, yielding superior performance as demonstrated in ref. 29. Crucially, the substantial heat generated by the exothermic carbonation reaction is harnessed to drive a sCO2 Brayton cycle for power generation. This cycle employs sCO2 as the working fluid, leveraging its advantageous properties—notably high density akin to liquids and low viscosity like gases—which enable exceptional compressibility, efficient heat transfer, and excellent flow properties. For clarity, Fig. 2 presents a simplified schematic of the sCO2 Brayton cycle configuration; detailed cycle design optimization is reserved for subsequent sections.
2.2 Simulation models
This study employs a steady-state process modeling framework to simulate mass and energy balances across all system components. A customized in-house thermodynamic program was developed to resolve the coupled heat transfer and chemical reaction processes, ensuring numerical stability and convergence under varying operational conditions. Thermophysical properties of gaseous and liquid streams (e.g., sCO2, flue gas) were sourced from the NIST REFPROP database, while thermodynamic data for solid reactants (e.g., CaO, CaCO3) were obtained from the Aspen Plus® V11 property library. This dual-source approach guarantees consistency with widely accepted industrial standards.Fig. 3 illustrates the mass balances of material streams within the CaL system. CaO enters the carbonator from the storage tank at a molar flow rate FR. Within the carbonator, a portion of the incoming CaO undergoes carbonation, with the reacted molar flow designated as FCaO,carb, and this process produces the same molar flow of CaCO3 (FCaCO3,carb). Simultaneously, the unconverted CaO flow rate is designated FCaO,unr,carb. Collectively, these molar flows thus satisfy the relationship:
| FCaO,carb = FCaCO3,carb = XFR, | (4) |
| FCaO,unr,carb = (1 − X)FR. | (5) |
Here, X represents the carbonation conversion of CaO. Critically, it should be emphasized that CaO and CaCO3 physically coexist as indivisible composite particles – rather than discrete phases. The mass balance model's representation of CaO and CaCO3 as separate fluid streams constitutes a deliberate idealization for computational tractability and conceptual clarity. The molar flow rate of CO2-rich flue gas entering the carbonator is denoted Fflue-gas, the value of which is determined by:
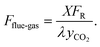 | (6) |
Here, λ denotes the CO2 capture rate of the flue gas, and yCO2 represents the mole fraction of CO2 in the inlet flue gas. The molar flow rate of CO2 participating in the carbonation reaction is calculated as:
| FCO2-capture = XFR. | (7) |
The molar flow rate of decarbonized flue gas exiting the carbonator is denoted Fdecarb-flue-gas, the value of which is calculated as:
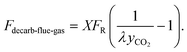 | (8) |
The total molar flow rate of solid particles exiting the carbonator is FR. Fresh CaCO3 is supplied to the CaCO3 storage vessel at a molar flow rate F0, and mixed particles (CaCO3 + CaO) are simultaneously removed at molar flow rate F0. The makeup ratio, r, is defined as the ratio of the fresh makeup flow rate F0 to the total recirculating solid flow rate FR, expressed as:
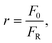 | (9) |
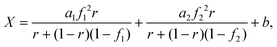 | (10) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 charge/discharge duration ratio and complete decomposition of CaCO3, the total molar flow rate of the mixed-particle stream exiting the CaCO3 storage tank is denoted as FR. Within this stream, the molar flow rate of the reactive CaCO3 stream undergoing calcination in the calciner is denoted as FCaCO3,calc, which generates an equimolar flow of CaO (FCaO,calc) through thermal decomposition. These values are calculated as follows:14
1 charge/discharge duration ratio and complete decomposition of CaCO3, the total molar flow rate of the mixed-particle stream exiting the CaCO3 storage tank is denoted as FR. Within this stream, the molar flow rate of the reactive CaCO3 stream undergoing calcination in the calciner is denoted as FCaCO3,calc, which generates an equimolar flow of CaO (FCaO,calc) through thermal decomposition. These values are calculated as follows:14| FCaCO3,calc = FCaO,calc = X(FR − F0) + F0, | (11) |
| FCaO,unr,calc = (FR − F0)(1 − X). | (12) |
During cyclic operation, two types of solid particles enter the calciner: partially carbonated particles with a CaO core and a CaCO3 outer layer, and freshly supplied CaCO3 particles. In the calciner, CaCO3 is assumed to undergo complete decomposition; the CaCO3 layer of the partially carbonated particles is fully converted to CaO, and the fresh CaCO3 particles are also entirely transformed into CaO. As a result, only CaO particles exit the calciner, with a total molar flow rate denoted as FR.
It should be noted that the solid particles exiting the calciner consist exclusively of CaO, with a total molar flow rate of FR. CH4 at a molar flow rate of FCH4 enters the calciner where it reacts to produce syngas (CO/H2), with the combined molar flow rate denoted as FCO/H2. The inlet and outlet gaseous molar flow rates for the calciner are, respectively:
| FCH4 = X(FR − F0) + F0, | (13) |
| FCO/H2 = 4X(FR − F0) + 4F0. | (14) |
Carbon deposition via methane cracking and the Boudouard reaction is a known operational risk in dry reforming systems. In practical operation, these effects can be effectively mitigated by maintaining an appropriate CH4/CO2 feed ratio, operating at sufficiently high temperatures, and, where necessary, introducing limited amounts of steam. Such measures are commonly adopted to suppress carbon formation without fundamentally altering the system configuration. In the present study, these mitigation strategies are assumed to be in place, and carbon deposition is therefore not explicitly modeled at the system level.
The design parameters for the calcium looping section of the CaL-DRM-sCO2 system are specified in Table 1. Parameters for the carbonator side follow ref. 19 and are not listed separately herein.
| Parameters | Components/streams | Values |
|---|---|---|
| Calciner temperature | Calciner | 700 °C (ref. 28) |
| Standard enthalpy of reaction | Calciner | 425 kJ mol−1 |
| Thermal loss | Calciner | 4% |
| Pressure loss | Calciner | 15 kPa (ref. 31) |
| CH4 | 1%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14 14 |
|
| CO/H2 syngas | 2%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 14 14 |
|
| Minimum temperature difference | HENs | 15 °C (ref. 11) |
| Specific electrical work of solid conveying | CaO, CaCO3 | 10 kJ (kg 100 m)−1 (ref. 32) |
| Storage vessel-calciner distance | CaO, CaCO3 | 100 m (ref. 32) |
| Isentropic efficiency | Fan | 80%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 29 29 |
| Mechanical efficiency | Fan | 97%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 32 32 |
| Heat transfer coefficient | Gas | 600 W (m2 K)−1 (ref. 14) |
| Solid particle | 200 W (m2 K)−1 (ref. 14) | |
| Heat discharge | Carbonator | 40 MW |
| Annual operating time | Calciner | 2922 h |
| Carbonator | 2922 h |
The hybrid thermochemical Carnot battery system employs an intercooled recompression sCO2 Brayton cycle as the power block during discharge. Building upon this baseline configuration, a reheating process is implemented between the two turbines to enhance cycle efficiency and reduce turbine operating pressures and temperatures. The finalized cycle layout and key design parameters are presented in Table 2 and Fig. 4. An optional waste heat heater can be strategically positioned within this configuration between the high-temperature recuperator (HTR) and the low-temperature recuperator (LTR). This heater utilizes industrial waste heat to elevate the temperature of the sCO2 working fluid exiting the LTR, bringing it closer to the inlet temperature requirement of the HTR, thereby optimizing thermal matching across the recuperative heat exchange process.
| Parameters | Components/streams | Values |
|---|---|---|
| Heat source temperature | Carbonator | 650 °C (ref. 33) |
| Minimum temperature | sCO2 | 32 °C |
| Waste heat exhaust temperature | Waste heat exhaust gas | 200 °C |
| Isentropic efficiency | Compressor | 88%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 29 29 |
| Turbine | 92%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 29 29 |
|
| Mechanical efficiency | Compressor/turbine | 98%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 29 29 |
| Minimum pressure | sCO2 | 7.5 MPa (ref. 29) |
| Minimum pinch point temperature | Heater/recuperator | 10 °C |
| Heat exchanger thermal loss | 1%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 34 34 |
All reactors in the system are treated as zero-dimensional open reacting systems for the present analysis, while detailed reactor configurations and spatially resolved designs are currently under active investigation. In the ongoing engineering studies, the reactors are primarily being developed based on fixed-bed or moving-bed concepts, with their specific hydrodynamics, heat transfer characteristics, and scalability addressed in dedicated follow-up work. Consequently, chemical reaction kinetics are excluded from their thermodynamic modeling. The system energy and exergy balance equations are summarized in Tables 3 and 4, with detailed methodologies available in ref. 11, 14 and 19.
Investment cost constitutes a critical factor in assessing the techno-economic feasibility of the system, primarily comprising two components: the Total Plant Cost (TPC) and the Total Annualized Cost (TAC). The TPC is defined as follows:31
| TPC = iTASC[(1 + iLC + iE&PC)(1 + iP&C)∑ICi], | (15) |
Reactors are configured as circulating fluidized-bed reactors (CFBRs). This design promotes intensive gas–solid contact under fluidization regimes, thereby enhancing reaction rates. The cost function for the calciner is defined as follows:33
ICcalc = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 831 831![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 151Q0.65calc, 151Q0.65calc,
| (16) |
ICcarb = 133![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 671Q0.5carb, 671Q0.5carb,
| (17) |
ICHEX = 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 409A0.67HEXp0.28HEX, 409A0.67HEXp0.28HEX,
| (18) |
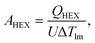 | (19) |
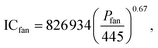 | (20) |
Within the sCO2 Brayton cycle architecture, the compressor cost correlation adopts:36
ICcomp = 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 917 917![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 503W0.3992comp, 503W0.3992comp,
| (21) |
ICturb = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 323 323![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 850W0.5561turbfT,turb, 850W0.5561turbfT,turb,
| (22) |
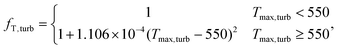 | (23) |
| ICrecup = 359(UArecap)0.7544fT,recup, | (24) |
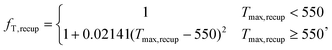 | (25) |
Regarding the main heater in the CO2 Brayton cycle, its cost model follows Carlson et al.,37 which is calculated via eqn (26):
| ICabs = UAabscabs, | (26) |
| UAabs (W K−1) | cabs (¥ per (W per K)) |
|---|---|
| 5 × 103 | 13.78 |
| 3 × 104 | 9.43 |
| 1 × 105 | 7.98 |
| 3 × 105 | 7.25 |
| 1 × 106 | 7.25 |
The cost function for the cooler in the CO2 Brayton cycle is given by:31
| ICcooler = 258.8Qcooler, | (27) |
ICEG = 789![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 525P0.5463EG, 525P0.5463EG,
| (28) |
The net annual operating cost (TAC) is defined as follows:31
 | (29) |
2.3 Technical assessments
This study adopts multiple performance metrics to evaluate the proposed integrated system. Among these, round-trip efficiency serves as the fundamental metric for assessing hybrid thermochemical Carnot battery performance, defined as:
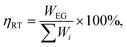 | (30) |
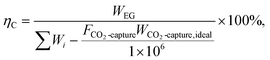 | (31) |
WCO2-capture,ideal = RTflue-gas(∑yout,i![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(yout,i) − ∑yin,i ln(yout,i) − ∑yin,i![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(yin,i)), ln(yin,i)),
| (32) |
For the CaL-DRM-sCO2 system, the energy outputs comprise electrical power generation (WEG) and the chemical energy of syngas (Echem,syngas). The energy inputs include the total electrical energy input (∑Wi), chemical energy of consumed methane (Echem,CH4), and industrial waste heat (Qwaste-heat). Thus, the energy efficiency of the CaL-DRM-sCO2 system is derived as:
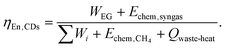 | (33) |
Similarly, the exergy outputs include the exergy of net electrical output (Ex,WEG) and chemical exergy of syngas (Exchem,syngas), while exergy inputs comprise the exergy of electrical input (Ex,Wi), chemical exergy of methane feed (Ex,chem,CH4), and exergy of industrial waste heat (Ex,Qwaste-heat). Thus, the exergy efficiency of the CaL-DRM-sCO2 system is defined as:
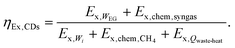 | (34) |
The specific energy consumption per unit CO2 captured (SEC) serves as a key metric for evaluating the system's carbon capture capability. With units of MJ kg−1, it is defined as:
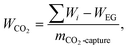 | (35) |
Additionally, this study employs the levelized cost methodology to evaluate electricity costs. This approach integrates the system's TPC, TAC, and discount rate over its operational lifetime. The levelized cost of electricity (LCOE), expressed in ¥ per kWh, is defined as:
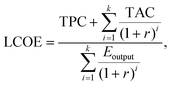 | (36) |
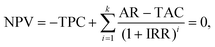 | (37) |
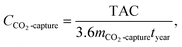 | (38) |
2.4 Optimization method
This study conducts multi-objective optimization of the CaL-DRM-sCO2 system using the NSGA-II algorithm, where the defined objective functions (Table 6) comprise exergy efficiency maximization for energy performance and LCOE minimization for economic performance. These conflicting objectives necessitate a balanced compromise solution. Consequently, the configuration closest to the exergy efficiency midpoint within the Pareto frontier is selected as the optimal trade-off solution to reconcile thermodynamic and economic trade-offs.| Optimization objectives | Objective functions |
|---|---|
| Energy performance | Max. ηEx,CDs |
| Economic performance | Min. LCOE |
The decision variables and their parametric boundaries for the multi-objective optimization model are detailed in Table 7, with the CaO conversion (X) – a performance-dominant parameter in the energy storage subsystem – selected as the primary decision variable for storage optimization. For the discharging subsystem, critical decision variables include the sCO2 Brayton cycle's maximum pressure (pH), intermediate turbine pressure (pMH), intermediate compressor pressure (pML), and compression flow split ratio (β) – the latter defined as the mass flow ratio of sCO2 entering the main compressor (C1) to the total working fluid mass flow according to eqn (39).
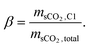 | (39) |
| Decision variables | Lower bounds | Upper bounds |
|---|---|---|
| X | 10% | 70% |
| pH | 20 MPa | 30 MPa |
| pMH | 10 MPa | 18 MPa |
| pML | 8 MPa | 15 MPa |
| β | 0 | 1 |
The constrained parameters in this multi-objective optimization include the main compressor outlet temperature (TC1,out), waste heat recuperator thermal load (QWH), cold stream outlet temperatures of the LTR and HTR (TLTR,cold,out/THTR,cold,out), and HEN hot utility for the carbonator side (Qhot-utility), as tabulated in Table 8. The constraint principles fundamentally derive from thermodynamic imperatives: the main compressor outlet temperature (defining the cycle minimum temperature) must exceed the sCO2 threshold at ≥32 °C, while the waste heat recuperator thermal load is constrained to non-negative values ensuring unidirectional heat transfer. Furthermore, recuperator temperature differences are governed by the second law's irreversibility requirement, mandating cold-stream outlet temperatures to remain below hot-stream inlets minus the 10 °C pinch point difference. Additionally, the absence of auxiliary heating for the carbonator subsystem necessitates zero hot utility consumption in HEN.
| Parameters | Constraint conditions |
|---|---|
| TC1,out | ≥32 °C |
| QWH | ≥0 |
| TLTR,cold,out | ≤TLTR,hot,in – 10 °C |
| THTR,cold,out | ≤THTR,hot,in – 10 °C |
| Qhot utility | =0 |
Furthermore, the population size and parameter configuration for the NSGA-II algorithm are specified in Table 9.
| Parameters | Values |
|---|---|
| Population size | 60 |
| Generations | 80 |
| Crossover probability | 0.95 |
| Mutation probability | 0.9 |
3. Validation
The credibility of the proposed hybrid thermochemical Carnot battery system is established through component-level validation. The operational reliability of the core reversible Brayton cycle (compressor, turbine, heat exchangers) was confirmed by our prior experimental work11,19,38 and benchmarking against literature data. The designs for the heat exchanger network and reactor are adopted from ref. 14.Furthermore, the system's cost models are grounded in industrial data: component costs for the supercritical CO2 cycle are derived from 4–24 supplier quotations,36 while costs for the heat network and reactor are sourced from ref. 14. By integrating these proven performance data and credible cost models, the overall system analysis presented here is highly reliable.
4. Results and discussion
4.1 System optimization results
As illustrated in Fig. 6, the Pareto frontier obtained through multi-objective optimization reveals a pronounced trade-off relationship between LCOE and exergy efficiency. When LCOE decreases from approximately 1.106 ¥ per kWh to 0.916 ¥ per kWh, the system's exergy efficiency increases from 84.70% to 85.36%, indicating that pursuing higher energy efficiency incurs higher economic costs, and vice versa. The distribution of non-dominated solutions demonstrates a nonlinear correlation between these objectives: within the exergy efficiency range of 84.70–85.50%, LCOE increases marginally by only 0.006 ¥ per kWh per 0.1% efficiency gain, suggesting a favorable compromise zone between economy and performance. Conversely, beyond 85.0% exergy efficiency, each 0.1% improvement requires a steep LCOE increment of 0.048 ¥ per kWh, causing drastic economic deterioration. Consequently, the non-dominated solution closest to the exergy efficiency midpoint within the Pareto set is selected as the optimal compromise, with corresponding decision variables and objective values detailed in Table 10. Notably, the compression β of 1 indicates the reversion from the recompression Brayton cycle configuration to a simple recuperated cycle topology, eliminating the need for working fluid splitting and inter-stage compression.| Parameters | Result | |
|---|---|---|
| Objective function values | Exergy efficiency | 85.03% |
| LCOE | 0.94 ¥ per kWh | |
| Decision variables | X | 48.74% |
| pH | 29.97 MPa | |
| pMH | 16.25 MPa | |
| pML | 8.00 MPa | |
| β | 1 |
Fig. 7 shows the energy and exergy analyses of the baseline case, which is the optimal trade-off solution obtained through multi-objective optimization. Producing 22.57 MW of electrical power requires a significant combined input of surplus and auxiliary electrical energy totaling 110.25 MW. This high demand mainly comes from the calciner, where simultaneous calcination and CH4 dry reforming reactions greatly increase its energy needs. As a result, the system has a relatively low electrical round-trip efficiency of 20.47%. However, relying only on round-trip efficiency to assess system performance has limitations. During the charging phase, the system produces valuable syngas as a co-product, while the discharging phase captures carbon with a capacity of 8.41 MJ kg−1. Including these polygeneration benefits results in much higher overall system efficiencies: 88.04% on an energy basis and 85.03% on an exergy basis.
 | ||
| Fig. 7 Energy and exergy analyses of the baseline CaL-DRM-sCO2 system: (a) energy flow diagram; (b) exergy flow diagram; (c) energy loss diagram; (d) exergy destruction diagram. | ||
Fig. 7(a) depicts the energy flow diagram for the baseline case. Total system input energy reaches 331.67 MW, comprising surplus electricity (107.87 MW), auxiliary power consumption (2.38 MW), CH4 chemical energy (210.87 MW), and industrial waste heat (10.55 MW). Total output energy is 292.00 MW, consisting of generated electricity (22.57 MW) and syngas chemical energy (269.43 MW). Within the calciner, a portion of the input surplus electricity is converted and stored as chemical energy within CaO solid particles (42.23 MW). During subsequent energy discharge, the stored chemical energy in CaO is released as 39.60 MW of thermal energy within the carbonator. This thermal energy heats the sCO2 working fluid, ultimately driving the sCO2 Brayton cycle to generate 22.57 MW electrical output. Fig. 7(c) details the distribution of energy losses. Irreversible thermal and mechanical losses within the sCO2 Brayton cycle dominate, accounting for 69.53% of the total energy dissipation. These are followed by heat losses from the HEN (5.13 MW) and thermal losses from the two reactors (4.59 MW). Auxiliary power consumption includes energy for solids transport (1.30 MW) and fan operation (1.08 MW).
Fig. 7(b) depicts the exergy flow diagram for the baseline case. Total exergy input reaches 310.94 MW, comprising exergy of surplus electricity (107.87 MW), parasitic exergy consumption (2.38 MW combined), chemical exergy of methane (196.68 MW), and exergy of industrial waste heat (4.01 MW). Total exergy output is 264.38 MW, including generated electricity exergy (22.57 MW) and chemical exergy of syngas (241.81 MW). Within this flow, 31.00 MW of electrical exergy is converted and stored as chemical exergy in CaO. During discharge, this stored chemical exergy transforms into 27.02 MW of thermal exergy via carbonation, ultimately driving the Brayton cycle to produce 22.57 MW of electrical output. Fig. 7(d) details the distribution of exergy destruction. The calciner dominates exergy destruction at 65.51%, primarily due to significant energy-grade degradation during electrical-to-thermal conversion. The Brayton cycle contributes substantially less to exergy loss (18.17%) than energy loss, attributable to the low-grade exergy of the discharged low-pressure sCO2 from the compressor.
Economic analysis of the baseline case reveals a TPC of 295.34 million CNY (M¥), with the cost structure detailed in Fig. 8(a). Equipment expenditure dominates at 176.56 M¥, constituting 59.78% of TPC. As labor, engineering, piping/integration, and miscellaneous costs scale proportionally with equipment investment, the latter represents the primary cost driver. Fig. 8(b) details the equipment cost breakdown, identifying five major components: the calciner (38.38 M¥; 21.74% of total equipment costs), carbonator (26.73 M¥), recuperator (28.31 M¥), turbine (21.43 M¥), and compressor (20.95 M¥). Collectively, these components represent 76.91% of equipment expenditure, with the calciner constituting the largest individual share. Notably, the sCO2 Brayton cycle's recuperator incurs higher relative costs than the entire HEN in the calcium looping system, attributable to stringent material and sealing requirements under high-pressure operating conditions.
 | ||
| Fig. 8 Economic analysis of the baseline CaL-DRM-sCO2 system: (a) TPC breakdown; (b) equipment cost breakdown. | ||
Fig. 9 shows the cost and revenue breakdown within the TAC framework. CH4 consumption dominates operational expenses at 71.87%, driven by high calciner demand that requires a continuous input of 13.64 t h−1 during charging. Green electricity accounts for a significant 23.96% due to the calciner's high power needs, while CaO, maintenance, and labor costs remain small. Conversely, syngas sales provide the main revenue stream (159.45 M¥), supported by carbon capture credits (21.92 M¥). The net economic assessment results in a TAC of 40.48 M¥ after balancing costs and revenue.
4.2 Sensitivity analysis
The Pareto-optimal trade-off solution derived from multi-objective optimization was selected as the baseline scenario for the system, accounting for the uncertainties in system design parameters and the potential for optimizing both energetic and economic performance. A sensitivity analysis is conducted to evaluate the impact of fluctuations in key parameters on the performance of this baseline system. This analysis focuses on four critical parameters: the CaO conversion rate, the minimum temperature (ΔTmin) in heat exchangers, the system scale, and the annual operating hours. The objective is to investigate their sensitivity to system performance metrics, particularly energy efficiency and economic indicators, thereby identifying stable operational boundaries and potential for further optimization. | ||
| Fig. 10 Comparison of the CaCO3 makeup ratio under different operational scenarios as a function of CaO conversion rate. | ||
Fig. 11 illustrates the influence of X on the system's energy performance. As X increases from 10% to 50%, the solid circulation rate of the calcium loop decreases from 2460 mol s−1 to 474 mol s−1. Since no fresh CaCO3 makeup is required in this range, both the energy consumption for solid transport and the excess power demand of the calciner are reduced, leading to improved system efficiency. At X = 10%, the total power input is 120.55 MW, with a round-trip efficiency, energy efficiency, and exergy efficiency of 18.72%, 86.38%, and 83.25%, respectively. When X reaches 50%, the total power input decreases to 110.19 MW, while the round-trip, energy, and exergy efficiencies increase to 20.48%, 88.05%, and 85.04%, respectively. Beyond X = 50%, sorbent deactivation occurs, necessitating the addition of fresh CaCO3. The required makeup amount increases with higher X, which elevates the calcination reaction's heat demand and raises the calciner's excess power consumption. The solid circulation rate declines, saving transport energy; however, this benefit is offset by the calciner's higher power demand. For instance, when X increases from 50% to 70%, the excess power for calcination rises from 107.84 MW to 114.71 MW. Consequently, the overall system efficiency slightly decreases beyond the 50% conversion threshold. At X = 70%, the round-trip, energy, and exergy efficiencies decline to 19.33%, 87.95%, and 84.81%, respectively.
Similar to the variations in system efficiency, the relationship between the specific energy consumption for carbon capture and X is shown in Fig. 11(c). As X increases from 10% to 50%, energy consumption declines from 9.05 MJ kg−1 to 8.41 MJ kg−1 due to reduced total system power input. Beyond this range, further increases in X cause higher power demand because of sorbent deactivation, raising energy consumption to 9.07 MJ kg−1 at X = 70%. Based on this energetic analysis, the system performs optimally at around a 50% conversion rate.
Fig. 12 illustrates the influence of X on the economic performance of the CaL-DRM-sCO2 system. The most cost-sensitive components to variations in X are the calciner, the calciner-side HEN, and the carbonator-side HEN. As X increases from 10% to 50%, the heat load on the calciner-side and carbonator-side HENs gradually decrease, reducing their combined cost from 44.38 to 17.68 M¥. At the same time, the cost of the calciner drops from 39.72 to 38.37 M¥, leading to a downward trend in the total capital cost of the system. When X increases from 50% to 60%, the rising power demand causes the calciner cost to rise by 0.72 M¥. However, this increase is offset by a decrease of 1.44 M¥ in the HEN costs. As a result, the total equipment cost continues to decline as X increases.
 | ||
| Fig. 12 Effects of CaO conversion (X) on the economic performance of the CaL-DRM-sCO2 system: (a) equipment costs by component; (b) total investment cost; (c) LCOE and IRR. | ||
As the TPC is proportional to the total capital cost, it decreases with increasing X, as shown in Fig. 12(b). The TPC declines from 341.98 M¥ to 293.82 M¥ as X rises from 10% to 60%. The overall trend of the TAC with increasing X is characterized by an initial gradual decrease due to reduced power demand, followed by a slow increase caused by higher limestone expenditure. Finally, when X exceeds 50%, the requirement for fresh limestone makeup drives a rapid escalation in TAC, which reaches 113.52 M¥ at X = 60%. It is evident that further increases in X beyond this point would lead to a sharp rise in the TAC, significantly impairing the system's economic viability.
Fig. 12(c) shows that the trend of LCOE closely follows that of TAC, indicating that TAC is the dominant factor influencing LCOE at a given X. In contrast, IRR exhibits a trend opposite to LCOE. As X increases from 10% to 35%, LCOE decreases from 1.02 ¥ per kWh to 0.92 ¥ per kWh, while IRR increases from 10.14% to 13.32%. When X further increases to 50%, LCOE rises slightly to 0.94 ¥ per kWh due to the elevated TAC, and IRR declines to 13.07%. Beyond this point, further increase in X leads to a severe deterioration in economic performance, making the project investment hardly profitable.
Based on a thorough analysis of X's impact on energy and economic performance, the ideal range for X is identified as 30%–50%. In this range, lower X values support economic growth, while higher values enhance energy efficiency.
Fig. 14 illustrates the impact of the ΔTmin in the heat exchangers on the economic performance of the CaL-DRM-sCO2 system. As the ΔTmin increases from 5 °C to 30 °C, major equipment costs, including the turbine, compressor, and recuperator—which contribute significantly to the total capital cost—all exhibit a decreasing trend, as shown in Fig. 14(a). At a ΔTmin of 5 °C, the costs of these components are 22.94 M¥, 21.14 M¥, and 36.73 M¥, respectively. When the ΔTmin increased to 30 °C, the costs decrease to 20.26 M¥, 16.41 M¥, and 15.40 M¥, respectively. This reduction can be attributed to two primary factors: the increase in the minimum temperature lowers the temperature of the sCO2 working fluid after the heating section, thereby reducing the maximum cycle temperature. As a result, the power output of both the turbine and the compressor decreases, leading to lower equipment costs. Additionally, the required heat transfer area of the heat exchangers decreases with a larger temperature approach, which also contributes to the reduction in cost.
Meanwhile, the decrease in individual equipment costs results in a lower TPC, which then causes a slight drop in the TAC, as shown in Fig. 14(b). As the ΔTmin increases from 5 °C to 30 °C, the TAC slightly declines from 40.90 M¥ to 39.76 M¥. However, the unit operating cost for power generation rises with a larger temperature difference. This is due to the strong negative relationship between system power output and ΔTmin, as previously explained. Specifically, when ΔTmin increases from 5 °C to 30 °C, the unit operating cost for power generation goes up from 1.761 M¥ per MW to 1.974 M¥ per MW.
Fig. 14(c) illustrates the influence of the ΔTmin on the carbon capture cost. The heat release from the carbonator remains constant, resulting in an unchanged system carbon capture rate and annual CO2 capture capacity. Consequently, as the ΔTmin increases from 5 °C to 30 °C, the carbon capture cost follows the same trend as the TAC, decreasing slightly from 373.11 ¥ per t to 362.68 ¥ per t.
Fig. 14(d) shows the influence of the heat exchanger ΔTmin on the LCOE and IRR. The LCOE initially decreases and then increases with a rising ΔTmin, while the IRR exhibits the opposite trend. As the ΔTmin increases from 5 °C to 10 °C, the LCOE decreases from approximately 0.943 ¥ per kWh to 0.939 ¥ per kWh. This reduction occurs because the decline in TAC outweighs the decrease in net power output. Correspondingly, the IRR increases from 12.35% to 12.73%. However, when the ΔTmin further increases to 30 °C, the LCOE gradually rises to 0.99 ¥ per kWh and the IRR declines to 11.59%, indicating deteriorated economic performance. Thus, the optimal economic performance is achieved within a minimum temperature range of 5–10 °C.
In summary, both the system's energy and economic performance are favorable when the ΔTmin is relatively small. However, since very small values can cause significant technical challenges and uncertainties in practical engineering, a ΔTmin of 10 °C is recommended as the best balance.
The TAC also exhibits a pattern of diminishing growth with system scale, albeit more gradually compared to the TPC, as shown in Fig. 15(b). Specifically, when the system scale increases from 1 MW to 50 MW, the TAC rises from 1.49 M¥ to 50.07 M¥—an increase of 48.58 M¥. As the scale further expands to 100 MW, the TAC reaches 97.43 M¥, representing an incremental rise of 47.36 M¥. The unit operating cost for power generation also follows a step-like decreasing trend as the system scale enlarges. In the system scale range of 1–10 MW, the unit operating cost decreases rapidly from 2.64 M¥ per MW to 1.96 M¥ per MW, a reduction of 0.68 M¥ per MW. Beyond 10 MW, however, the rate of decrease slows significantly. By the time the scale reaches 100 MW, the cumulative reduction amounts to a mere 0.23 M¥ per MW.
As shown in Fig. 15(c), the LCOE follows the variation of the unit capital and operating costs, demonstrating clear economies of scale. When system capacity increases from 1 MW to 10 MW, the LCOE drops sharply from 2.37 to 1.23 ¥ per kWh; with further expansion, the decline becomes gradual, reaching 0.83 ¥ per kWh at 100 MW. In contrast, the IRR exhibits a mirror-image trend relative to LCOE. For capacities up to 3 MW, IRR is negative, indicating economic infeasibility. As capacity expands, IRR rises and exceeds the 6% benchmark discount rate at 11 MW (6.01%), marking the onset of positive returns; continued scale-up to 100 MW elevates IRR to 18.67%. Balancing these effects, a moderate installation size of approximately 40–70 MW is reasonable and can achieve superior techno-economic performance.
 | ||
| Fig. 16 Effects of annual operating hours on the CaL-DRM-sCO2 system: (a) TAC and annual electricity revenue; (b) LCOE and IRR; (c) annual CO2 captured and capture cost. | ||
As shown in Fig. 16(c), annual CO2 capture scales linearly with annual operating hours. When utilization increases from 3000 h to 8000 h, the captured CO2 rises by 93.8 kt (i.e., about 18.76 t h−1 for each additional operating hour). In parallel, the CO2 capture cost declines with higher utilization: it drops from 449.20 ¥ per t-CO2 at 3000 h to 346.53 ¥ per t-CO2 at 8000 h. Taken together, annual operating hours are strongly and positively correlated with both economic and environmental performance. Nevertheless, continuous year-round operation is constrained by maintenance requirements and staffing. Within these practical limits, maximizing feasible annual operating hours enhances economic performance.
As shown in Fig. 17, extending the operational lifetime enhances techno-economic performance by amortizing upfront capital over a longer period. With a 20-year lifetime, the LCOE is 1.00 ¥ per kWh and the IRR is 11.64%; when the lifetime is extended to 60 years, the LCOE decreases to 0.89 ¥ per kWh and the IRR increases to 13.08%, strengthening investment returns. Nevertheless, lifetime extension must account for whole-life dynamics: equipment aging may raise repair frequency and operation and maintenance expenditures, and rapid technology turnover can induce performance degradation and obsolescence risks. Hence, lifetime should not be pushed arbitrarily long; it should be selected by balancing initial capital expenditures, escalation of operation and maintenance costs, overhaul/replacement strategies, and technology currency to maximize life-cycle economic value.
5. Conclusions
This work proposes a CaL-DRM-sCO2 system. The CH4 atmosphere lowers the calcination temperature by about 200 °C, mitigates sorbent deactivation, and reacts in situ with the released carbon dioxide to co-produce synthesis gas, while the carbonation stage enables approximately 90% CO2 capture. Multi-objective optimization reveals clear efficiency–cost trade-offs:The Pareto-optimal compromise yields an exergy efficiency of 85.03% at a levelized cost of electricity of 0.94 ¥ per kWh for a 40 MW plant, with the power block simplified to a recuperated cycle configuration. Under these conditions, the net electric output reaches 22.57 MW, corresponding to a round-trip electrical efficiency of 20.47%. When credit is given to the synthesis-gas co-product and ∼90% carbon dioxide capture, the overall system efficiency rises markedly to 88.04%, with a specific capture energy of 8.41 MJ per kg-CO2.
An operating window with 30–50% CaO conversion, combined with a 10 °C minimum temperature in the recuperators, yields favorable thermo-economic performance.
As plant capacity expands from 1 to 100 MW, the levelized cost of electricity declines sharply from 2.37 to 0.83 ¥ per kWh. A capacity range of 40–70 MW appears most practical when considering both investment scale and associated risks. Moreover, higher annual utilization further enhances economics, reducing the LCOE to 0.81 ¥ per kWh and boosting the internal rate of return to 18.97%.
The present analysis relies on steady-state models employing zero-dimensional reactor representations and cost correlations. Future work should extend beyond these simplifications to incorporate detailed reaction kinetics and multi-scale coupling, dynamic operation and dispatch optimization, long-term sorbent cycling and regeneration strategies, pilot-scale validation, and comprehensive assessment of methane supply-chain leakage on life-cycle performance.
Conflicts of interest
There are no conflicts to declare.Data availability
This paper is a methodological study and does not utilize any specific datasets. The code is available in the GitHub repository: gitee.com/sun-zihao/nbCaLtrees-zh.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5eb00225g.
Acknowledgements
This work is supported by National Key Research and Development Program of China (no. 2024YFE0212800).References
- I. R. E. Agency, Renewable power generation costs in 2022, Abu Dhabi, United Arab Emirates, 2023 Search PubMed.
- I. E. Agency, Net Zero by 2050: A Roadmap for the Global Energy Sector, Paris, France, 2021 Search PubMed.
- A. Toktarova, L. Gruber, M. Hlusiak, D. Bogdanov and C. Breyer, Int. J. Electr. Power Energy Syst., 2019, 111, 160–181 CrossRef.
- Y. Wang, R. Wang, K. Tanaka, P. Ciais, J. Penuelas, Y. Balkanski, J. Sardans, D. Hauglustaine, W. Liu, X. Xing, J. Li, S. Xu, Y. Xiong, R. Yang, J. Cao, J.-M. Chen, L. Wang, X. Tang and R. Zhang, Nature, 2023, 619, 761–767 CrossRef CAS PubMed.
- H. Jiang, L. Yao, J. Qin, Y. Bai, M. Brandt, X. Lian, S. J. Davis, N. Lu, W. Zhao, T. Liu and C. Zhou, Nat. Commun., 2025, 16, 4523 CrossRef CAS PubMed.
- I. Sarbu and C. Sebarchievici, Sustainability, 2018, 10(1), 191 CrossRef.
- E. Borri, A. Tafone, A. Romagnoli and G. Comodi, Energy Convers. Manage., 2017, 143, 275–285 CrossRef.
- G. Feng, Y. Li, X. Cheng, B. Hou, D. Qu, R. Xu and L. Zhang, Therm. Power Gener., 2024, 53, 1–9 Search PubMed.
- A. H. Abedin and M. A. Rosen, Energy, 2012, 41, 83–92 CrossRef CAS.
- P. Pardo, A. Deydier, Z. Anxionnaz-Minvielle, S. Rougé, M. Cabassud and P. Cognet, Renewable Sustainable Energy Rev., 2014, 32, 591–610 CrossRef CAS.
- H. Liu, Y. Zhang, Q. Xu, W. Han and J. Shen, Appl. Energy, 2025, 377, 124535 CrossRef CAS.
- R. Chacartegui, A. Alovisio, C. Ortiz, J. M. Valverde, V. Verda and J. A. Becerra, Appl. Energy, 2016, 173, 589–605 CrossRef CAS.
- C. Ortiz, J. M. Valverde, R. Chacartegui, L. A. Perez-Maqueda and P. Giménez, Renewable Sustainable Energy Rev., 2019, 113, 109252 CrossRef CAS.
- Q. Xu, l. Wang, Z. Li and L. Shi, Energy Convers. Manage., 2023, 276, 116592 CrossRef CAS.
- S. E. B. Edwards and V. Materić, Sol. Energy, 2012, 86, 2494–2503 CrossRef CAS.
- C. Ortiz, R. Chacartegui, J. M. Valverde, A. Alovisio and J. A. Becerra, Energy Convers. Manage., 2017, 149, 815–829 CrossRef CAS.
- C. Zhang, J. Sun, Y. Qiao and J. Wei, Energy, 2024, 312, 133687 CrossRef CAS.
- L. Teng, Y. Xuan, Y. Da, X. Liu and Y. Ding, Energy Storage Mater., 2020, 25, 836–845 CrossRef.
- D. Wang, Z. Sun, Q. Xu, R. Tian, W. Han and J. Shen, Energy Convers. Manage., 2024, 318, 118888 CrossRef CAS.
- P. Sun, J. R. Grace, C. J. Lim and E. J. Anthony, AIChE J., 2007, 53, 2432–2442 CrossRef CAS.
- M. Benitez-Guerrero, J. M. Valverde, P. E. Sanchez-Jimenez, A. Perejon and L. A. Perez-Maqueda, Sol. Energy, 2017, 153, 188–199 CrossRef CAS.
- M. E. Diego, B. Arias, G. Grasa, J. C. Abanades, L. Díaz, M. Lorenzo and A. Sánchez-Biezma, Energy Procedia, 2014, 63, 2060–2069 CrossRef CAS.
- Z. He, Y. Li, W. Zhang, X. Ma, L. Duan and H. Song, Energy Convers. Manage., 2017, 148, 1468–1477 CrossRef CAS.
- R. Anwar, A. Al Kape, M. Lusi, E. Voutsas, A. Cammarata, A. Rafferty and M. V. Sofianos, J. Energy Storage, 2025, 114, 115784 CrossRef CAS.
- H. Guo, X. Kou, Y. Zhao, S. Wang, Q. Sun and X. Ma, Chem. Eng. J., 2018, 334, 237–246 CrossRef CAS.
- I. Wang, S. Huang, S. Wang, X. Bie, H. Zhou and Z. Li, Sep. Purif. Technol., 2024, 333, 125975 CrossRef CAS.
- L. Teng, Y. Xuan, Y. Da, C. Sun, X. Liu and Y. Ding, Chem. Eng. J., 2022, 440, 135955 CrossRef CAS.
- B. Shao, Y. Zhu, J. Hu, Y. Zong, Z. Xie, S. Li, W. Du, M. Wang, H. Liu and F. Qian, Chem. Eng. J., 2024, 483, 149098 CrossRef CAS.
- U. Tesio, E. Guelpa and V. Verda, Energy Convers. Manage.: X, 2020, 6, 100038 CAS.
- L. Zhen-shan, C. Ning-sheng and E. Croiset, AIChE J., 2008, 54, 1912–1925 CrossRef.
- S. Michalski, D. P. Hanak and V. Manovic, J. Cleaner Prod., 2019, 219, 540–551 CrossRef CAS.
- U. Tesio, E. Guelpa, C. Ortiz, R. Chacartegui and V. Verda, Energy Convers. Manage.: X, 2019, 4, 100025 CAS.
- S. Khosravi, S. Hossainpour, H. Farajollahi and N. Abolzadeh, Energy, 2022, 240, 122466 CrossRef CAS.
- Summary of the Sandia Supercritical CO2 Development Program. [electronic resource], Washington, D.C.: United States. National Nuclear Security Administration; Oak Ridge, Tenn.: distributed by the Office of Scientific and Technical Information, U.S. Dept. of Energy, 2011.
- U. Tesio, E. Guelpa and V. Verda, Energy Convers. Manage.: X, 2020, 6, 100039 CAS.
- N. T. Weiland, B. W. Lance and S. R. Pidaparti, “sCO2 Power Cycle Component Cost Correlations From DOE Data Spanning Multiple Scales and Applications.” proceedings of the ASME Turbo Expo 2019: Turbomachinery Technical Conference and Exposition, Volume 9: Oil and Gas Applications; Supercritical CO2 Power Cycles; Wind Energy, Phoenix, Arizona, USA, June 17–21, 2019, V009T38A008, ASME. https://doi.org/10.1115/GT2019-90493.
- M. D. Carlson, B. M. Middleton and C. K. Ho, V001T05A009 T1 - Techno-Economic Comparison of Solar-Driven SCO2 Brayton Cycles Using Component Cost Models Baselined With Vendor Data and Estimates VL - DO - 10.1115/ES2017-3590 ER, 2017.
- Y. Zhang, H. Liu, J. Lu, Q. Xu and C. Xu, J. Energy Storage, 2025, 130, 117487 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |