Open Access Article
Open Access ArticleResearch progress on molybdenum- and tungsten-based materials for sodium-ion batteries: fundamental mechanisms and optimization strategies
Bingrong
Shen
a,
Chenhui
Yan
a,
Hui
Peng
 *a,
Guofu
Ma
*a,
Guofu
Ma
 a and
Yuxi
Xu
a and
Yuxi
Xu
 *b
*b
aKey Laboratory of Eco-functional Polymer Materials of the Ministry of Education, Key Laboratory of Polymer Materials of Gansu Province, College of Chemistry and Chemical Engineering, College of Engineering, Northwest Normal University, Lanzhou 730070, China. E-mail: penghui@nwnu.edu.cn
bSchool of Engineering, Westlake University, Hangzhou 310024, China. E-mail: xuyuxi@westlake.edu.cn
First published on 6th February 2026
Abstract
In sodium-ion battery (SIB) anode systems, molybdenum (Mo)- and tungsten (W)-based materials have shown great potential in the field of energy storage due to their high theoretical capacity, adjustable layered structure, and multi-electron characteristics. However, their practical applications are limited by challenges such as structural collapse caused by volume expansion, instability of the solid electrolyte interface (SEI) due to interface side reactions, and poor conductivity. To date, many reports have shown that through strategies such as structural design, compounding and hybridization, defect and interface engineering, their conductivity can be significantly improved, mechanical stress alleviated and ion transport paths optimized, achieving a breakthrough in cycling stability. In this review, we first analyze the challenges faced by Mo- and W-based materials in SIB anodes. Then, we systematically focus on the sodium storage mechanisms and performance optimization strategies for Mo- and W-based materials, as well as research progress on Mo- and W-based oxides/chalcogenides, carbon composites and polymetallic molybdenum/tungstate. Finally, we discuss the bottlenecks they face and provide an outlook for the future development of Mo- and W-based materials in the SIB field.
Broader contextWith growing demand for sustainable energy storage, sodium-ion batteries (SIBs) are emerging as a promising alternative to lithium-ion batteries (LIBs) due to their abundant sodium resources, low cost, and environmental friendliness. However, the slow diffusion kinetics of Na+ in traditional anode materials remains a major challenge limiting the development of high-performance SIBs. This is not only due to limitations of the anode material, but also closely related to its sodium storage capacity, structural stability, and interfacial dynamics. Molybdenum (Mo)- and tungsten (W)-based materials, with their unique layered structures and rich valence states, offer a high theoretical capacity, structural stability, and excellent rate performance in SIBs, demonstrating great application potential. Nevertheless, issues such as interfacial stability, volume effects, and conductivity still hinder their performance improvement. Therefore, improving the overall performance of SIBs requires some efficient and significant optimization strategies. Against this backdrop, we highlight the recent progress made in sodium storage mechanisms and optimization strategies for SIB anode materials. In addition, we also conducted an in-depth analysis of research progress on various Mo- and W-based materials. Therefore, continued progress in these directions is expected to accelerate the practical application of SIBs in next-generation energy storage applications. |
1. Introduction
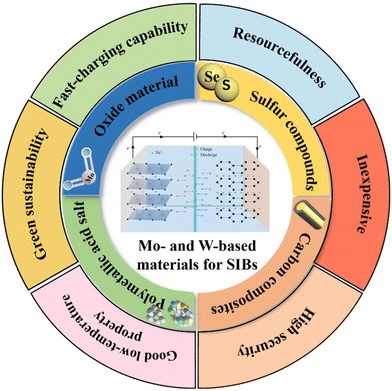
At a critical time in the global energy transition, efficient and low-cost energy storage technology has become a core element in supporting the large-scale deployment of renewable energy.1,2 Although lithium-ion batteries (LIBs) dominate the current market, they face severe challenges due to the uneven distribution of lithium resources and sharp price fluctuations, which seriously restrict their application in the electrochemical energy storage area.3,4 Sodium-ion batteries (SIBs) are widely regarded as an important representative of the next generation of energy storage systems due to the abundance of sodium resources in Earth's crust, low raw material costs, high intrinsic safety, outstanding fast charging capabilities, green sustainability, and a similar working mechanism to that of LIBs (Fig. 1).5–7 However, the large radius and molar mass of sodium ions (Na+) lead to sluggish diffusion kinetics in traditional anode materials, causing significant volume strain and capacity decay during charging and discharging, which has become the main bottleneck in the development of high-performance SIBs.8–10As the core component of SIBs, the performance of anode materials directly affects the energy density, cycle life and rate capability of batteries.11–13 The ideal anode needs to have a high specific capacity, excellent ionic/electronic conductivity and structural stability to achieve efficient and stable operation of the battery.14–16 Although traditional hard carbon materials have a good cycling performance, their limited theoretical capacity makes it difficult to meet the needs of high energy density, and new materials are urgently needed to break through the performance bottleneck.17–19 Among the many candidate materials, molybdenum (Mo)- and tungsten (W)-based materials stand out with their significant advantages and they have become the most promising candidate materials for anodes.20–22 From the perspective of structural characteristics, Mo- and W-based materials mostly present a layered crystal structure with natural ion transport channels between layers, and their interlayer spacing can be flexibly controlled by means of intercalation and exfoliation.23–25 Compared with other materials, this adjustable interlayer spacing not only provides a more suitable space for Na+ insertion/de-insertion, effectively reducing the insertion energy barrier, but also buffers volume changes during charging and discharging, maintaining structural stability.26–29 At the level of the Na+ storage mechanism, the rich valence changes of Mo/W elements enable multiple redox reactions such as intercalation, conversion and alloying to occur, providing a high theoretical capacity, far exceeding that of traditional hard carbon anode materials (250–350 mAh g−1).30–33 In addition, Mo- and W-based materials also have good chemical stability and thermal stability, and can maintain their structural integrity under extreme conditions such as high temperature and high voltage, greatly improving the safety and environmental adaptability of the battery.34–37 Meanwhile, its unique electronic structure endows the material with excellent intrinsic electronic conductivity, and the ion transport advantages brought by the layered structure can effectively improve the rate performance of the battery.38–41 In 2025, Liu et al. thoroughly investigated the effect of W doping on the electrochemical performance of a manganese phosphate material with a NASICON structure (sodium superionic conductor type structure), analyzing the mechanism of W doping and proposing an optimization framework to improve its cycling stability and rate performance.42 On the other hand, Khammuang et al. integrated the electrochemical mechanisms of Mo2C, Mo2CO2, and MXenes through density functional theory (DFT) calculations, highlighting Na+ adsorption, diffusion, and maximum capacity under different strains, and the Mo-based MXenes exhibited lower energy diffusion barriers and excellent dendrite suppression capabilities compared to existing materials.43 These studies provide a theoretical basis for the performance optimization of Mo- and W-based materials and point the way for further improving the performance of SIBs. In recent years, research has focused on further unlocking the performance potential of Mo/W based materials through strategies such as nanostructure design and atomic-level regulation. By enhancing the exposure of active sites in materials and optimizing interfacial charge transfer efficiency, SIBs are being pushed toward the practical goal of high energy density and long cycle life.44–47
Mo- and W-based materials have shown multi-dimensional performance advantages and application potential in the field of SIB anodes, but there are still many technical difficulties to be overcome before large-scale commercial applications.48–51 This review introduces research on Mo- and W-based materials in SIB anodes through the aspects of material classification, sodium storage mechanisms, performance optimization strategies, practical application challenges and future development directions, and provides a comprehensive overview and analysis of recent research strategies for high-performance SIB anode materials.52–55 With continued in depth research and development, Mo- and W-based anode materials will become the core breakthrough to overcome the performance bottleneck of SIBs, help the global energy storage industry move toward a green, efficient, and sustainable direction, and inject new impetus into the energy transformation goals.56–59
2. Challenges of Mo- and W-based materials in SIB anodes
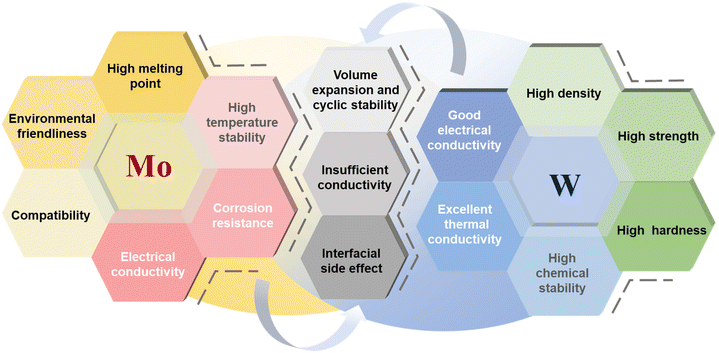
Mo-based materials have the advantages of high temperature stability, strong corrosion resistance, relatively excellent conductivity, good compatibility with carbon materials, and environmental protection and non-toxicity.60,61 W-based materials have the advantages of high density, high strength, high hardness, high chemical stability, excellent thermal conductivity and good electrical conductivity.62,63 However, both of them face multiple challenges when applied in SIB anodes: when storing sodium, multiple reactions such as intercalating–conversion–alloying will cause volume expansion, resulting in pulverization and shedding of electrode materials, structural instability, and shortened cycle life; although the intrinsic conductivity is better than some materials, the interlayer electron conduction resistance is large, and the conductivity is still lower than that of carbon-based materials; the abundant active sites on the surface react with the electrolyte to form an unstable SEI membrane, which continuously consumes sodium ions and electrolyte, exacerbating capacity decay and increased internal resistance.64–66 The above features and challenges are described in Fig. 2.Although both Mo-based and W-based materials show potential as anode materials for SIBs, significant differences exist between them that affect their performance. W has a higher atomic mass than Mo, leading to differences in specific capacity and mass energy density. At the atomic level, W-based materials generally exhibit higher theoretical capacity and energy density than Mo-based materials, but considering the actual material density, their volumetric efficiency is lower because the higher atomic mass of tungsten reduces the actual density of the material. Furthermore, due to stronger atomic bonds, W-based materials typically possess higher structural stability than Mo-based materials, which can mitigate volume expansion during cycling. However, this advantage often comes with higher synthesis complexity and higher material costs, making W-based materials less cost-effective than Mo-based materials for large-scale applications. When selecting the appropriate material for a specific application, the trade-offs between cost, capacity, and scalability are crucial, especially in balancing battery energy density and overall cost to enable the practical large-scale application of SIBs.67,68
2.1 Volume expansion and poor cycling stability
During the Na+ insertion/de-insertion process, Mo- and W-based materials undergo significant volume changes. Taking MoS2 as an example, because Na+ has a relatively large radius, when it is intercalated into the layered structure of MoS2, it will expand the interlayer distance and cause changes to the lattice parameters.69 This volume expansion will cause serious damage to the crystal structure of the material. Upon charge and discharge cycling, the layered structure is gradually stretched and distorted, and even interlayer peeling occurs.70 From a microscopic perspective, the bond lengths and bond angles between atoms will also change, causing the crystal structure of the material to gradually degenerate, which in turn affects the electrochemical properties of the material.71 After multiple cycles, the layered structure of MoS2 may transform into a disordered nanoparticle stacking structure, which will not only reduce the specific capacity of the material but also affect the diffusion path and kinetic process of Na+.Volume expansion will cause stress concentration inside the material. Since the expansion degree of different parts of the material may be different, stress gradients will form inside and between particles.72 This stress concentration will accelerate the pulverization process of the material. When the stress exceeds the tolerance limit of materials, cracks will appear in the material. As the number of cycles increases, the cracks continue to expand, eventually causing the material to break into fine particles.73 Taking WS2 nanosheets as an example, during the sodification process, the stress distribution at the edges and inside of the nanosheets is uneven, and the edges are more susceptible to stress and curl and break. After the material is pulverized, the electrical contact between the active material and the current collector deteriorates, and electron transmission is hindered, resulting in increased internal resistance of the battery and accelerated capacity decay. In addition, the pulverized particles may also fall off from the electrode surface, further reducing the active material loading of the electrode, seriously affecting the cycling stability and rate performance of the battery.74
Structural damage and pulverization will seriously weaken the cycling stability of the material. As the number of cycles increases, the structure of the material gradually deteriorates, resulting in the obstruction of Na+ intercalation/deintercalation, reduced specific capacity, increased battery polarization, reduced energy efficiency, and even safety hazards.75 The key to improving the cycling stability of Mo- and W-based materials lies in the synergy of “structural confinement” and “interface optimization”, building physical support through nano-scaling and composite formation, combining doping and defect engineering to enhance chemical stability, and regulating the electrolyte to stabilize the interface reaction, balancing volume expansion and long life requirements, providing key technical support for the commercialization of SIBs.76
2.2 Insufficient conductivity
In the actual application of the SIB anodes, the insufficient conductivity of Mo- and W-based materials has a significant negative impact on battery performance. Firstly, the low conductivity will greatly increase the ohmic resistance inside the electrode, leading to severe overpotential during the charge and discharge process, which will reduce the energy efficiency of the battery, limit the power density, and make it difficult to meet the needs of high-power fast charging and discharging.77 Secondly, the hysteresis of the electron conduction rate directly restricts the intercalation/deintercalation kinetics of Na+. The active materials cannot fully participate in the electrochemical reaction, resulting in a decrease in the battery's specific capacity. Especially under high rate conditions, the capacity decay is more obvious.78 Furthermore, uneven conductivity can cause imbalanced current distribution inside the electrode, resulting in local overheating and stress concentration, accelerating the structural destruction and pulverization of active materials, and significantly shortening the cycle life of the battery.79The poor conductivity of Mo- and W-based materials is mainly restricted by the characteristics of electronic structure and crystal structure. From the perspective of electronic structure, Mo- and W-based materials are mostly semiconductors or insulators, all of which have relatively wide band gaps. The energy consumption of the electron transition is high, resulting in fewer free electrons and a weak electron conduction ability.80 Taking MoS2 as an example, the chemical bonds within its layers are mainly covalent bonds, while the interlayers are bound by weaker van der Waals forces. This structure greatly hinders the conduction of electrons between layers.81 In terms of crystal structure, there are often problems with lattice defects, which scatter electrons and increase the resistance to electron transmission.82,83 At the same time, Mo- and W-based materials will undergo phase changes (i.e. conversion reactions or alloying reactions) during the Na+ intercalation/deintercalation process. The stress and structural changes generated during the phase change will further destroy the conductive network of the material, exacerbating the problem of insufficient conductivity.84
To address the aforementioned issues, researchers have conducted a lot of research on material structure design, composite modification, etc. In terms of structural optimization, nanofabrication can significantly shorten the electron transmission path, and the introduction of porous structures can improve the transmission efficiency of electrons and ions. Heterogeneous structure design utilizes interface effects to regulate the distribution of electron clouds, thereby improving the overall conductivity of the material.85–87 In terms of the composite strategy, constructing a continuous conductive network by combining it with highly conductive carbon materials can not only effectively improve the conductivity of the electrode, but also buffer the volume change during charging and discharging. These methods provide theoretical guidance for the design of high-performance Mo- and W-based anode materials in SIBs.88,89
2.3 Interfacial side reactions
In the anode system of SIBs, Mo- and W-based materials have become research hotspots due to their high theoretical capacity and unique layered structure, but interfacial side reactions seriously hinder improving their performance.90,91 The interface side reaction will first lead to irreversible capacity loss, consume limited Na+ and electrolyte resources, greatly reduce the initial coulombic efficiency of the battery, and continuously consume active materials during the cycling process, accelerating capacity decay.92,93 Meanwhile, the side reaction products will form an unstable SEI layer on the electrode surface. The repeated rupture and regeneration of the membrane will not only increase the internal resistance of the battery, but also cause the electron transfer efficiency to decrease, thus deteriorating the rate performance of the battery.94,95In addition to the issue of interfacial side reactions, the instability of the SEI layer remains a significant challenge for Mo- and W-based anodes. The chemical composition of the SEI on Mo- and W-based materials is still not fully understood. Studies have shown that, in carbonate-based electrolytes, the SEI consists of both inorganic components such as Na2CO3 and organic fragments derived from solvent decomposition, such as alkyl carbonates and ester compounds. In ether-based electrolytes, the SEI may contain more organic components, with the presence of compounds like Na2O and ether-derived organic salts.96 The differences in the SEI composition between these two types of electrolytes highlight a major issue for Mo- and W-based materials, namely electrolyte compatibility. This issue is less pronounced in more widely used anode materials like hard carbon or titanium-based oxides, where the SEI layer tends to be more stable. Additionally, high-concentration electrolytes can further complicate SEI formation due to their different interactions with the surface of Mo- and W-based materials. In these systems, the SEI tends to be thicker and less stable, leading to increased internal resistance and reduced overall performance. These differences underscore the need for further research into the stability and composition of the SEI layer on Mo- and W-based anodes, as well as strategies for improving electrolyte compatibility and interface stability.97
The root cause lies in the thermodynamic and kinetic incompatibility between Mo- and W-based materials and the electrolyte. In terms of thermodynamics, there are a large number of unsaturated coordinated atoms and active sites on the surface of Mo- and W-based materials, which have high reactivity with organic solvents and electrolyte salts in the electrolyte and are prone to reduction and decomposition.98,99 For example, after the carbonate solvent in the electrolyte gains electrons on the electrode surface, it decomposes to produce gases such as CO2 and CO as well as organic fragments. These fragments combine with Na+ to form SEI membrane components.100 Kinetically, during the Na+ intercalation/deintercalation process, the material structure changes significantly, exposing new active surfaces, resulting in continuous electrolyte infiltration and side reactions.101 In addition, surface defects and grain boundary regions of the material will further reduce the activation energy of the reaction.
The problem of interface byproducts needs to be solved through aspects such as material structure optimization and interface modification. In terms of material structure optimization, nano-scaling can be used to reduce the material size, shorten ion diffusion paths, reduce local concentration gradients, and reduce side reactions caused by uneven ion migration.102,103 In terms of interface modification, atomic layer deposition (ALD) technology is used to coat the material surface with inert oxide layers such as Al2O3 and TiO2, or conductive polymers are coated by chemical deposition. This can not only inhibit electrolyte penetration, but also optimize electron conduction and reduce interfacial charge accumulation. Element doping can also be used to regulate the surface electron cloud density and weaken the reaction activity of active sites with the electrolyte.104,105
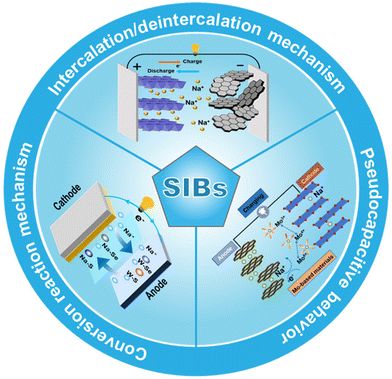
3. Sodium storage mechanisms in Mo- and W-based materials
In research on SIBs, Mo- and W-based materials have attracted much attention due to their diverse crystal structures and rich redox chemistry.106 Their sodium storage mechanism is not dominated by a single mode, but is a complex and synergistic system composed of intercalation/deintercalation, conversion reactions, and pseudocapacitive behavior.107 Among them, the intercalation/deintercalation mechanism lays the foundation for reversible cycling, relying on the reversible diffusion of Na+ in the material interlayer or tunnel; the conversion reaction mechanism contributes a high specific capacity through violent phase transitions, but is also accompanied by structural reconstruction and challenges; and pseudocapacitive behavior, as a surface-controlled rapid process, significantly improves the rate performance of the material (Fig. 3).108–110 A deep understanding of the interaction and balance of these three mechanisms, and the ability to artificially control them through nanostructure design, composite formation and defect engineering of materials, is key to unlocking the high-performance sodium storage capacity of Mo- and W-based materials.3.1 Intercalation/deintercalation mechanism
In the SIB system, Mo- and W-based materials exhibit unique sodium storage properties, and their intercalation/deintercalation mechanism is the key to understanding their energy storage performance. (1) Some Mo-based oxides have layered or tunnel-like structures. For example, in layered MoO3, there are certain gaps between the layers, which provide channels for the insertion of Na+. When the battery is charged, electrons in the external circuit flow into the material and combine with incoming Na+. Na+ is gradually intercalated in the MoO3 interlayer, resulting in an increase in the interlayer spacing. This process is accompanied by a change in the oxidation state of the Mo ions, from a high valence state to a low valence state, to maintain the electrical neutrality of the material.111 During the discharge process, Na+ is deintercalated from the interlayer and returns to the electrolyte, while electrons flow out through the external circuit, and the oxidation state of Mo ions returns to a higher valence state, completing a complete intercalation/deintercalation cycle. Mo sulfide also has a special structure for sodium storage.112 (2) Taking Mo-based sulfides as an example, MoS2 has a sandwich-like structure consisting of a Mo atomic layer sandwiched between two S atomic layers.113 When storing sodium, Na+ tends to embed into the interlayer van der Waals gap of MoS2. As Na+ is intercalated, the crystal structure of MoS2 expands to a certain extent, and the electron cloud distribution of Mo atoms and S atoms changes, causing slight changes in the Mo–S bond length and bond angle.114 This structural change affects the intercalation/deintercalation dynamics of Na+ to a certain extent. In the subsequent extraction and insertion process, the structure gradually recovers. After multiple cycles, the repeated structural changes may cause partial structural damage to the material, affecting the cycling stability of the battery.115 (3) For some Mo-based composites, the sodium storage mechanism is more complicated. When Mo-based materials are composited with carbon materials, the carbon materials can improve the conductivity of the materials. In addition, their porous structures or functional groups can provide additional adsorption sites for Na+.116 During the charge and discharge process, in addition to the Na+ intercalation/deintercalation reaction of the Mo-based material, the carbon material may also participate in sodium storage, storing a certain amount of Na+ through surface adsorption or the formation of weak chemical bonds. This synergistic effect helps to improve the overall sodium storage capacity and rate performance of the material.117 Moreover, W-based materials (such as WOx) undergo similar redox reactions and accompanying crystal phase transitions to those of Mo-based materials when storing sodium. When Na+ is embedded, the original crystal structure may transform from one crystal phase to another crystal phase that is more conducive to the accommodation of Na+. This process is also accompanied by a change in the oxidation state of W ions to balance the charge. During the deintercalation process, the crystal phase undergoes a reverse transformation. At the same time, some defects or amorphous areas on the surface of the material may also play an important role in the rapid adsorption and desorption of Na+, affecting the overall sodium storage kinetics.118The sodium storage mechanism of Mo- and W-based materials involves many factors, such as changes to the material structure, changes to the ion oxidation state, and synergistic effects with other composite components. Understanding these mechanisms is of great significance for further optimizing the sodium storage performance of materials.
3.2 Conversion reaction mechanism
The conversion reaction mechanism of Mo- and W-based sulfide compounds in the SIB system is the core path for sodium storage, which is characterized by “chemical bond breakage and recombination–product phase change–reversible redox”. At the beginning of discharging, Na+ is intercalated in the layered structure, triggering the dissociation of Mo/W–X (X = S/Se) bonds, and generating metallic Mo/W nanoparticles and Na2X (such as Na2S, Na2Se) through electrochemical reactions. This process is accompanied by the influx of electrons and the collapse of the crystal structure to form a nanocomposite phase. The reverse reaction theoretically restores the original material during the charging process, but in reality, it is affected by product agglomeration, side reactions and kinetic hysteresis, resulting in capacity decay.119–121 Structural differences in the materials will affect the conversion process. Compared with MoS2, the high oxidation state of W in WS2 results in a higher reaction potential and stronger W–S bond energy, leading to slower initial intercalating kinetics. MoSe2 and WSe2 have a large Se atomic radius, wide interlayer spacing and ion diffusion channels, but the difference in Se electronegativity changes the electron cloud distribution, resulting in different Mo/W–Se bond dissociation energies.122 It is worth noting that although W-based materials follow a similar transformation path, the high oxidation state of W makes the reaction potential higher, the interlayer van der Waals force weaker, and the ion diffusion energy barrier lower; this increases the risk of structural collapse during cycling.123,124To overcome the problems in the conversion reaction faced by Mo- and W-based materials when applied to SIBs, various strategies have been adopted.125 On one hand, by carefully designing the microstructure of the material, such as constructing nanostructures and porous structures, the influence of volume changes can be mitigated, the structural stability of the material can be increased, and the transmission and diffusion of Na+ can be facilitated. On the other hand, by adopting the strategy of forming composites with other materials, such as composites with highly conductive carbon materials, the overall conductivity and rate performance of the Mo- and W-based materials can be improved, which will indicate the direction for designing high specific energy and long-life anode materials for SIBs.126,127
3.3 Pseudocapacitive behavior
The pseudocapacitive behavior of Mo- and W-based materials and the sodium storage mechanism originate from the rapid and reversible redox reaction and ion adsorption process on the surface.128 This mechanism uses the atomic layer (edge sites or defect areas) on the surface of the material as the active center. Na+ diffuses to the surface of the material through the electrolyte, and rapid electron transfer occurs with the surface active site, triggering a change in the valence state of the Mo/W ions. At the same time, it is accompanied by surface adsorption or short-range embedding of Na+, forming a pseudocapacitive response.129,130 This process is different from traditional diffusion-controlled intercalation/deintercalation, in that its kinetics only depends on the surface ion adsorption–electron transfer rate.131 Specifically, during the Na+ intercalation/deintercalation process, Mo-based oxides and sulfides can achieve multi-electron transfer through the valence change of the Mo element (such as Mo6+ → Mo4+ → Mo0), and the rapid adsorption and desorption reaction of surface ions produces pseudocapacitive behavior.132 For example, MoO3 nanosheets construct a high specific surface area structure, allowing Na+ to undergo rapid redox reactions on the surface, significantly improving the rate performance, while the layered structure of MoS2 not only enables Na+ intercalation, but also accelerates ion storage dynamics through pseudocapacitive reactions at the edge active sites.133 W-based materials also exhibit excellent pseudocapacitive properties. The high valence state and stable lattice structure of the W element help maintain the reversibility of the pseudocapacitive reaction. After nano-scaling or composite modification, the active sites can be further exposed.134,135 However, the structure and defect engineering of materials have a significant impact on pseudocapacitive behavior. Nanosizing can significantly increase the specific surface area and expose more edge active sites, while the introduction of defects such as oxygen vacancies and sulfur vacancies can optimize the electronic structure and reduce the reaction energy barrier.136,137However, the pseudocapacitive behavior of Mo- and W-based materials still faces challenges in practical applications. On one hand, the surface structure is easily destroyed by repeated intercalation/deintercalation of ions during long-term cycling, resulting in pseudocapacitive capacity decay. On the other hand, insufficient intrinsic conductivity limits the surface reaction rate.138 Current research optimizes the electron transmission pathways and surface activity of materials through defect engineering, nanostructure design and carbon-based composite strategies, synergistically improving the pseudocapacitance contribution and cycling stability, which will provide important theoretical support for the development of high-performance sodium-ion battery electrode materials.139Table 1 summarizes the analysis of sodium storage mechanisms for some representative molybdenum and tungsten materials. Generally, a thorough understanding of the three sodium storage mechanisms (intercalation/deintercalation, conversion reactions and pseudocapacitive behavior) is key to unlocking the high-performance sodium storage capabilities of W- and Mo-based materials.
| Representative substances | Sodium storage mechanisms | Sodium storage principle | Main causes of failure | Ref. |
|---|---|---|---|---|
| Mo-based sulfides (MoS2) | Intercalation/deintercalation | Reversible diffusion and intercalation of Na+ between the layers; the Mo valence state reversibly changes in conjunction with electron/Na+ coupling to maintain electrical neutrality | Interlayer spacing changes/unstable SEI layer | 118 |
| Mo-based materials/carbon composites (MoS2/C) | Intercalation/deintercalation | Carbon enhances electronic conductivity, and the porous structure/functional groups provide additional Na+ adsorption sites | Volume change | 115 |
| W/Mo-based selenides (WSe2/MoSe2) | Conversion reaction | This is a “bond breaking–product phase transition–reversible redox” reaction pathway; Se influences the bond energy and reaction kinetics | Structural collapse | 119 |
| Mo-based sulfides (MoS2) | Conversion reaction | The reaction involves a transformation from interlayer intercalation to Mo–S bond breaking; the charging reversibility is limited by kinetics | Phase transition or volume change | 120 |
| Mo-based sulfides (MoS2) | Pseudocapacitive behavior | Na+ undergoes rapid and reversible redox reactions at surface defects; its kinetics depend on the surface reaction rate, not bulk diffusion | Structural collapse | 135 |
4. Performance optimization strategy for Mo- and W-based materials
4.1 Structural design
The design of Mo-based nanostructured materials can be achieved through a variety of methods, each with its own characteristics and advantages. The hydrothermal method is a commonly used method for preparing nanostructured Mo-based materials. Taking the preparation of nano-MoS2 as an example, the molybdenum source and the sulfur source react chemically in a high-temperature and high-pressure aqueous solution environment in the hydrothermal reaction system. The sulfur source decomposes to produce sulfur ions, which combine with molybdenum ions to gradually form MoS2 nanostructures. As shown in Fig. 4a, Nabilah Al-Ansi et al. prepared carbonized polymer dots (CPDs) by a hydrothermal method and annealing treatment using citric acid and ethylenediamine as raw materials. After hydrothermal treatment with Na2MoO4, thioacetamide and CPDs at 180 °C for 24 h and annealing at 700 °C under a H2/Ar atmosphere for 2 h, a MoS2@CPD material with a uniform 3D nanoflower-like microsphere structure was prepared (Fig. 4b). Its large specific surface area and rich pore structure enable it to maintain a practical capacity of 302.8 mAh g−1 after 500 cycles at a current density of 0.5 A g−1 (Fig. 4c).146 By precisely controlling the reaction conditions, the size and morphology of nano-MoS2 can be regulated. Specifically, MoS2 with a relatively uniform nanosheet structure can be obtained when the concentration ratio of the molybdenum source and the sulfur source is appropriate.
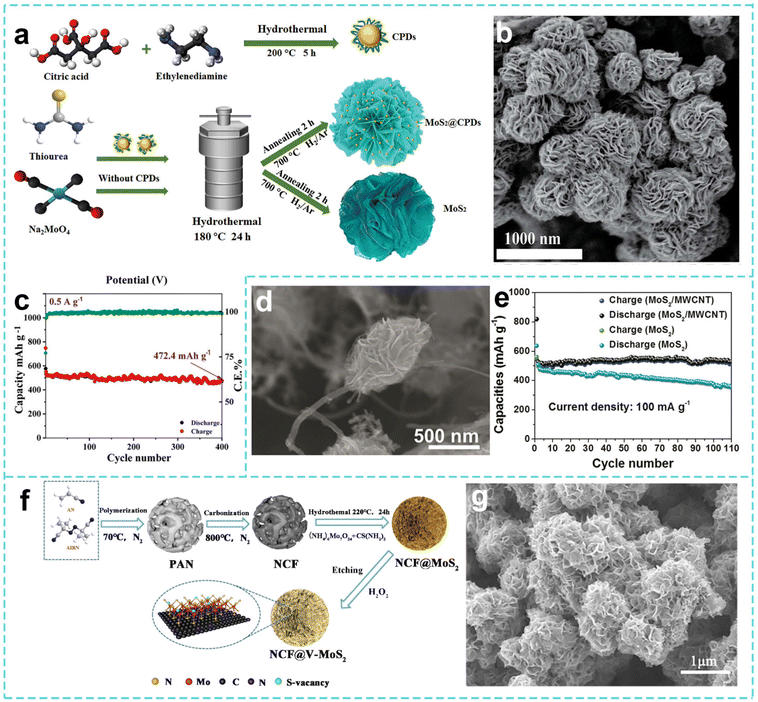 | ||
| Fig. 4 (a) Schematic demonstration of the synthesis of CPDs, MoS2, and MoS2@CPDs. (b) SEM image and (c) cycling performance of MoS2@CPDs-2 at 0.5 A g−1. Reproduced with permission from ref. 146. Copyright 2023, Wiley. (d) SEM image of the MoS2/MWCNT composite. (e) Cycling of the MoS2/MWCNT and MoS2 electrodes. Reproduced with permission from ref. 147. Copyright 2018, American Chemical Society. (f) Schematic of the synthesis process and (g) SEM image of NCF@V-MoS2. Reproduced with permission from ref. 148. Copyright 2022, Elsevier. | ||
Similarly, Bai et al. used Na2MoO4 and thiourea as raw materials with multi-walled carbon nanotubes (MWCNTs) through a hydrothermal reaction at 190 °C for 36 h to synthesize a nanoflower-shaped neuron-like MoS2/MWCNT composite material in one step (Fig. 4d). As shown in Fig. 4e, the nanoflower microspheres are connected by carbon nanotubes, which have both a large specific surface area and high conductivity. The discharge capacity is maintained at 527.7 mAh g−1 after 110 cycles at 100 mA g−1, and the capacity retention rate is about 100%.147 Hu et al. anchored MoS2 nanosheets with S vacancies on a 3D flower-shaped nitrogen-doped carbon framework (NCF@V-MoS2) by a similar hydrothermal method to prepare a nanoflower-shaped microsphere structure (Fig. 4f and g).148 In the experiment, ammonium molybdate was used as a molybdenum source and thiourea as a sulfur source, and the pre-prepared N-doped carbon flower was used as the supporting framework for a hydrothermal reaction at 220 °C for 24 h. The practical capacity of NCF@V-MoS2 remained at 413 mAh g−1 after 300 stable cycles at a current density of 1 A g−1.
In the study of the sodium storage mechanism and structural design of W-based sulfide compounds, Yang et al. adopted a solvothermal strategy to prepare a 3D hollow micro-flower-like WS2-based hybrid material (H-WS2@NC) by in situ interlayer modification of a nitrogen-doped carbon matrix using 2D WS2 nanosheets with extended interlayers as building blocks for bottom-up self-assembly (Fig. 5a). As shown in Fig. 5b, a 3D nano/micro-hollow structure was constructed with WS2 nanosheets with extended interlayers as building blocks, which provided mass transfer and electron transfer channels, an internal space to adapt to volume changes, and reduced ion diffusion energy barriers, so that the SIBs performed better (Fig. 5c).149 With continued improvement of the performance requirements for transition-metal dichalcogenide (TMD)-based electrode materials, Wang et al. made new attempts to expand the application scenarios and optimize the performance of WS2 materials. As shown in Fig. 5d and e, they developed a hybrid nanostructure of nitrogen-doped graphene quantum dots decorated with WS2 nanosheets and anchored with porous 3D carbon foam (NGQDs-WS2/3DCF). The porous structure of carbon foam is used to provide mechanical support and shorten the ion diffusion path. The strong interface effect of NGQDs is used to anchor the WS2 nanosheets to inhibit stacking and volume expansion, so that the material has a practical capacity of 268.4 mAh g−1 at 2000 mA g−1 and a capacity retention rate of 97.1% after 1000 cycles (Fig. 5f).150
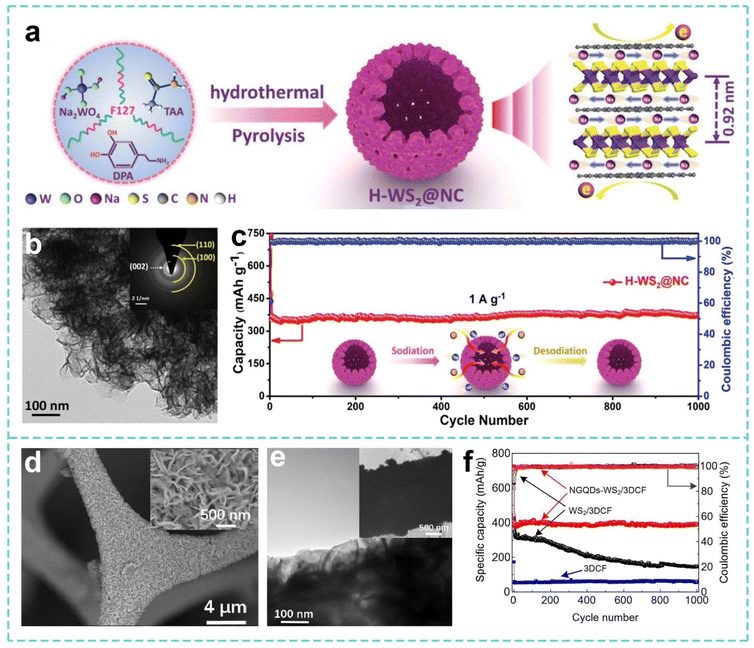 | ||
| Fig. 5 (a) Illustration of the synthesis process for H-WS2@NC. (b) TEM images of H-WS2@NC. (c) Long-term cycling performance of the H-WS2@NC electrode. Reproduced with permission from ref. 149. Copyright 2019, Wiley. (d) SEM image and (e) TEM image of the NGQD WS2/3DCF nanoarchitecture. (f) Cycling performance of the 3DCF, WS2/3DCF and NGQDs WS2/3DCF. Reproduced with permission from ref. 150. Copyright 2013, RSC. | ||
The aforementioned research systematically demonstrates the results of exploring Mo- and W-based nanomaterials in SIBs, covering various aspects from material structure design to innovative preparation methods. From the high number of active sites and short diffusion paths given by the nanostructure to the precise control of material morphology such as by means of a hydrothermal method and breakthroughs in structural design and mechanistic analysis of W-based materials, researchers continue to explore the potential for improving material performance through multi-dimensional exploration. Overall, nanoengineering has significantly improved the rate performance and cycling stability of Mo- and W-based anode materials, endowing them with more active sites, shorter Na+ diffusion pathways, and greater tolerance to volume changes. However, as research transitions from the laboratory to practical applications, the engineering feasibility urgently needs to be evaluated. Hydrothermal/solvothermal synthesis methods offer advantages in controlling material morphology and size, but large-scale preparation faces challenges such as uneven heat and mass transfer, long reaction times, and energy consumption and cost issues associated with high-temperature processing.151 Therefore, future nanostructure design should focus on scalable synthesis routes, simplified post-processing techniques, and compatibility with high-loading electrode manufacturing to achieve an effective transition from fundamental research to engineering applications.
In research on anode materials for SIBs, although the mesoporous structure is effective at improving the exposure of active sites, further enhancing the interfacial bonding between the active components and the carbon matrix to inhibit structural instability during the cycle remains an urgent problem to be solved. In response to this challenge, Wei et al. used a SiO2 template-assisted solvothermal reaction and subsequent high-temperature pyrolysis to synthesize a composite material that grew ultra-thin metal-phase MoS2 nanosheets on the inner surface of mesoporous hollow carbon spheres (M-MoS2@HCS) (Fig. 6a–c).157 The mesoporous hollow carbon spheres provide flexible support and ion diffusion channels to inhibit MoS2 aggregation and volume expansion, thus M-MoS2@HCS has a practical capacity of 401.3 mAh g−1 at 0.1 A g−1 for 100 cycles (Fig. 6d). Its composite structure significantly improves the reversibility of the sodium storage conversion reaction by synergistically accelerating electron transport, optimizing ion paths and buffering volume changes. On this basis, in view of the problems of poor conductivity, large volume changes and particle agglomeration of MoS2 as the anode of SIBs, Ma et al. adopted a template-free two-step dissolution thermal synthesis strategy to prepare a MoS2/C composite material of ultrathin nanosheets assembled on lignite-based carbon (Fig. 6e). As shown in Fig. 6f and g, the hollow structure alleviates volume expansion and shortens the Na+ diffusion path, and the strong interface between lignite-based carbon and MoS2 improves the conductivity and inhibits agglomeration. Thus, the practical capacity of the MoS2/C composite material is still 431 mAh g−1 after 2000 cycles at 20 A g−1 (Fig. 6h), and the sodium storage performance is optimized through the synergistic mechanism of “hollow structure buffering–carbon-based enhanced conductive dispersion”.158 With an in-depth understanding of the structure–performance relationship, researchers have further expanded the design of porous-hollow structures from single materials to multi-component composite systems. Cui et al. used a hard template method to synthesize a composite material of hollow porous double-shell N/S-doped carbon@WS2 nanospheres (NSC@WS2) (Fig. 6i and j).159 The constructed hollow porous double-shell structure effectively buffers the volume expansion during the cycle and shortens the Na+ diffusion path. The N/S-doped carbon inner shell improves the conductivity of the composite material and accelerates electron transfer. As shown in Fig. 6k, the reversible capacity of the material remains at 428 mAh g−1 after 400 cycles at 1 A g−1. The material improves the rate performance through the collaborative design of “hollow structure to buffer volume changes–doping carbon to improve conductivity–ultra-thin double shell to optimize ion transport”, overcoming the application bottleneck of WS2 (Fig. 6l).
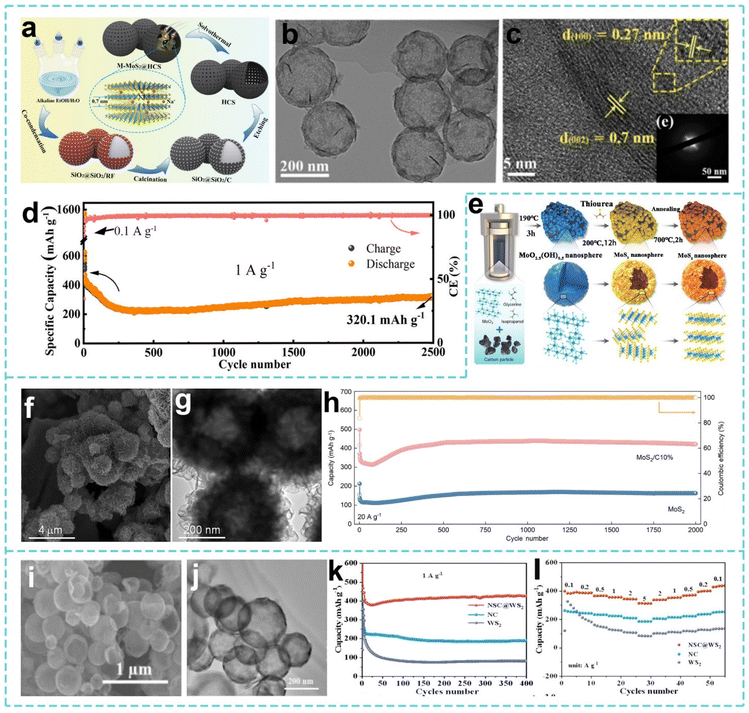 | ||
| Fig. 6 (a) Illustration of the synthesis process, (b) TEM image, and (c) HRTEM image of M-MoS2@HCS. (d) Cycling performance of the M-MoS2@HCS electrode at 1.0 A g−1. Reproduced with permission from ref. 157. Copyright 2021, American Chemical Society. (e) Illustration of the synthesis process, (f) SEM image, and (g) TEM image of MoS2/C. (h) Long cycle life of the MoS2 and MoS2/C electrodes at 20.0 A g−1. Reproduced with permission from ref. 158. Copyright 2025, Springer Nature. (i) SEM image and (j) TEM image of NSC@WS2. (k) Cycling performances and (l) rate performances of the NC, WS2, and NSC@WS2 electrodes. Reproduced with permission from ref. 159. Copyright 2025, Elsevier. | ||
4.2 Composite and hybrid design
A carbon coating, as a core composite strategy for SIB electrode materials, constructs a conductive network–mechanical barrier–interface regulation synergistic system by regulating the structure, composition and thickness of the carbon layer.160,161 Research has shown that when Mo- and W-based materials are hybridized with carbon, a carbon content of 5–10% can significantly enhance conductivity while maintaining a high specific capacity and good cycling stability. However, if the carbon content exceeds 15%, the specific capacity may decrease because the carbon layer occupies too many active sites. It is worth noting that rational composite and hybridization designs are also crucial for controlling the ICE. While the introduction of a carbon layer can improve conductivity and buffer volume changes, it may also consume a certain amount of sodium ions during the first cycle due to the formation of the SEI, thus affecting the ICE. By precisely controlling the thickness, crystallinity, and surface chemistry of the carbon layer, high ICE can be achieved. For example, by constructing a uniform and dense thin carbon coating, introducing appropriate surface functional groups, or performing pre-sodiation treatment, it is possible to promote the formation of a more stable and thinner SEI without significantly sacrificing active sites. This allows for the synergistic optimization of ICE by effectively reducing irreversible sodium loss while simultaneously leveraging the structural protection and conductivity enhancement effects of the carbon layer.162 In the study of structural–functional synergistic optimization of anode materials for SIBs, Tan et al. developed a 2D heterostructure composite material of carbon-coated MoSe2/MXene (MoSe2/Mo2CTx/C). From the unique structure of MXene-coated MoSe2 particles with loose carbon connections, the MXene layer enhances the surface activity of MoSe2, and the carbon layer provides rich pores, which synergistically improve the ion diffusion efficiency (Fig. 7a and b). As shown in Fig. 7c, there is a good specific capacity at different current densities after 2200 cycles. This heterostructure effectively inhibits MoSe2 agglomeration and enhances structural stability and ion conductivity through the synergistic effect of MXene interface activation and carbon layer pore optimization, as well as charge transfer regulation.163 Lu et al. further deepened the “interface-component” synergistic strategy and proposed an innovative path for strengthening the N-rich nitride substrate (Fig. 7d–f), which used the Mo–aniline skeleton as a precursor to prepare a composite material of carbon-coated N-rich MoxN modified few-layer MoSe2 nanosheets (MoSe2@MoxN/C-I) through calcination, selenization and nitridation processes. The carbon layer effectively enhances the structural stability and conductivity of MoxN, and nitrogen-rich MoxN and few-layer MoSe2 form a rich heterogeneous interface, providing a large number of active sites to accelerate Na+ diffusion and charge transfer. As shown in Fig. 7g, the reversible capacity is 178.6 mAh g−1 after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at 20 A g−1, and the capacity retention rate is 98.8%.164
000 cycles at 20 A g−1, and the capacity retention rate is 98.8%.164
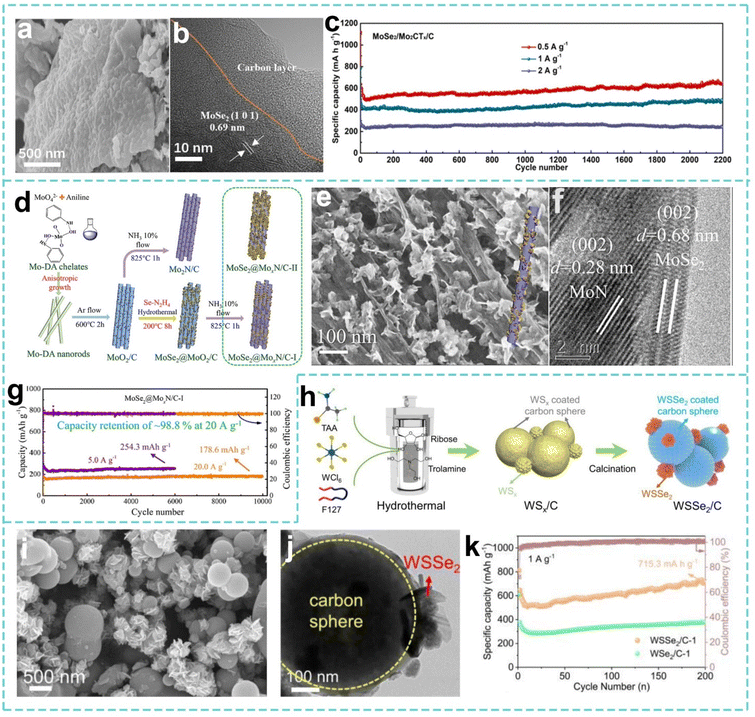 | ||
| Fig. 7 (a) SEM image and (b) HRTEM image of the MoSe2/Mo2CTx/C material. (c) Cycling performance at 0.5, 1, and 2 A g−1 of the MoSe2/Mo2CTx/C-containing cell. Reproduced with permission from ref. 163. Copyright 2022, Elsevier. (d) Illustration of the synthesis process for MoSe2@MoxN/C. (e) SEM image and (f) HRTEM image of MoSe2@MoxN/C-I. (g) Long cycling stabilities at 5.0 and 20.0 A g−1 of the MoSe2@MoxN/C-I electrode. Reproduced with permission from ref. 164. Copyright 2022, Elsevier. (h) Illustration of the synthesis process for WSSe2/C. (i) SEM image and (j) TEM image of WSSe2/C-1. (k) Short cycling performance at 1 A g−1 of the WSSe2/C-1 and WSe2/C-1 electrodes. Reproduced with permission from ref. 165. Copyright 2024, Elsevier. | ||
In the study of W-based carbon composites, Wang et al. prepared a carbon–uniformly encapsulated WSx precursor by a one-pot method combined with a high chloride hydrolysis method (Fig. 7h). Then selenization–co-carbonization were carried out to finally obtain a WSSe2/C composite material (Fig. 7i and j), which significantly enlarged the lattice spacing by constructing a built-in electric field, thereby ensuring fast and stable Na+ transport. The WSSe2/C-1 anode showed a high specific capacity of 715.3 mAh g−1 after completing 200 cycles at 1 A g−1 (Fig. 7k).165 This strategy of combining carbon encapsulation and a dual anion effect provides a reference for the development of high-power density W-based SIB anode materials.
In summary, the carbon coating strategy has built a multifunctional composite system for Mo- and W-based anode materials that integrates conductivity enhancement, structural stability, and interface optimization by precisely controlling the structure, composition, and dimensions of the carbon layer. From a single carbon layer coating to the multi-heterogeneous structure design, this strategy not only systematically solves the intrinsic electronic insulation, volume expansion, and interface instability problems of the material, but also achieves a cross-dimensional improvement in the sodium storage performance through the synergistic mechanism of conductive network construction, mechanical stress buffering and ion transport acceleration.
4.3 Defect and interface engineering
In the study of the structure–function synergistic design of anode materials for SIBs, MXene is an “all-around” two-dimensional material that combines high conductivity, hydrophilicity, excellent mechanical strength, and tunable surface chemistry. Therefore, Yu et al. synthesized a composite material (MoO3/MoS2/NC/MXene) with a N-doped carbon (NC) coating on MoO3/MoS2 modified on MXene nanosheets through gas-phase sulfidation and NC coating processes (Fig. 8a).171 As shown in Fig. 8b and c, the sulfurization treatment retains the MoO3 nanobelt structure to improve conductivity, the construction of the MoO3/MoS2 heterostructure accelerates ion/electron diffusion, while the introduction of NC and MXene enhances the conductivity and inhibits volume expansion. Thus, the composite material has a high discharge capacity of 464 mAh g−1 at a current density of 0.1 A g−1 and still maintains 202 mAh g−1 capacity at a high current density of 5 A g−1 (Fig. 8d). This work laid the foundation for subsequent interface defect regulation.
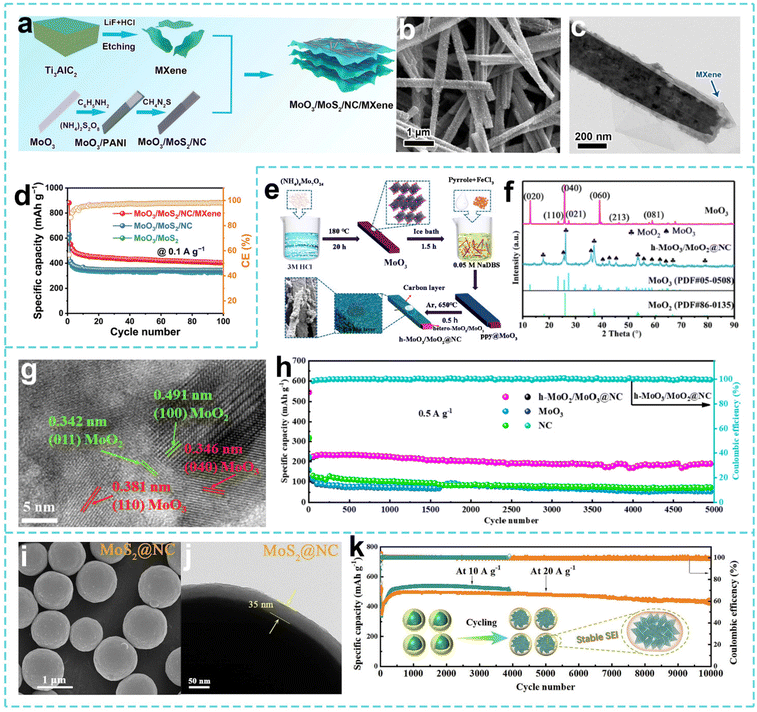 | ||
| Fig. 8 (a) Illustration of the synthesis process, (b) SEM image, (c) TEM image of MoO3/MoS2/NC/MXene. (d) Cycling performance of the MoO3/MoS2/NC/MXene electrodes. Reproduced with permission from ref. 171. Copyright 2024, Elsevier. (e) Illustration of the synthesis process for h-MoO3/MoO2@NC. (f) XRD patterns of MoO3 and the h-MoO3/MoO2@NC nanocomposite. (g) HRTEM image of h-MoO3/MoO2@NC. (h) Cycling performances of the h-MoO3/MoO2@NC, MoO3 and NC electrodes. Reproduced with permission from ref. 172. Copyright 2021, Elsevier. (i) SEM image and (j) TEM image of MoS2@NC. (k) Long-term cycling performance of the MoS2@NC electrodes. Reproduced with permission from ref. 173. Copyright 2023, Elsevier. | ||
Gao et al. converted the original MoO3 into heterogeneous MoO3/MoO2 and proposed a heterogeneous MoO3/MoO2@NC (Fig. 8e).172 As shown in Fig. 8f and g, the MoO3/MoO2 composite structure was successfully synthesized. The hybrid MoO3/MoO2 structure significantly improved the electronic conductivity and introduced oxygen defects, and the external N-doped carbon layer enhanced the structural stability. The MoO3/MoO2@NC material has a capacity of 190 mAh g−1 at 0.5 A g−1 after 5000 cycles, and shows an outstanding pseudocapacitive effect and wide temperature adaptability. Dong et al. proposed a spherical MoS2 block coated with a polydopamine-derived N-doped carbon sphere (MoS2@NC) material for SIBs.173 As shown in Fig. 8i and j, the MoS2@NC core–shell structure spontaneously reorganizes into ultrafine nanosheets during the cycle, improving material utilization and shortening the ion transmission distance. The outer flexible NC shell maintains the spherical structure of the electrode, inhibits agglomeration, and promotes the formation of a stable SEI layer. As shown in Fig. 8k, the material has been cycled for more than 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at a high current density of 20 A g−1, and the capacity still reaches 428 mAh g−1. The core–shell structure enhances the structural integrity and electronic conductivity and reduces the ion diffusion resistance through the synergistic mechanism of core structure reorganization and shell stability protection.
000 cycles at a high current density of 20 A g−1, and the capacity still reaches 428 mAh g−1. The core–shell structure enhances the structural integrity and electronic conductivity and reduces the ion diffusion resistance through the synergistic mechanism of core structure reorganization and shell stability protection.
Research on W-based materials presents a dual-track breakthrough of component innovation and bio-templating. For example, Fu et al. used a step-by-step method to prepare laurel leaf-shaped Co9S8/WS2@N doped carbon bimetallic sulfide (Co9S8/WS2@NC) materials (Fig. 9a).174 As shown in Fig. 9b and c, one can see the laurel leaf-shaped morphology of the Co9S8/WS2@NC material and the corresponding crystalline phase. The synergistic structure of large interlayer spacing WS2 and Co9S8 and the van der Waals force interface are used to inhibit WS2 aggregation, buffer volume changes and improve the electron transfer efficiency. The charge and discharge capacities of the Co9S8/WS2@NC anode are 510 and 406 mAh g−1, respectively, and the initial coulombic efficiency is 79.6% (Fig. 9d). As the current density increases from 0.1 A g−1 to 5.0 A g−1, it provides a practical capacity of 359 mAh g−1, and the capacity retention rate is 78.0% (Fig. 9e). Kang et al. took advantage of the bio-template and confined a small amount of layered WSe2 nanosheets in a composite material of N,P co-doped carbon (WSe2/N,P-C-2) derived from Chlorella (Fig. 9f and g).175 With the help of the bio-templating effect of Chlorella, the ultra-thin WSe2 nanosheets were uniformly dispersed in N,P co-doped biochar (Fig. 9h), forming a high activity carbon matrix confined nanosheet structure, which effectively inhibited the agglomeration and volume expansion of WSe2. As shown in Fig. 9i, the material has a good rate performance at different current densities, and has both wide applicability and ultra-stability. Besides, Liu et al. prepared Sn/N co-doped metallic phase tungsten selenide (1T-WSe2-Sn) materials using chitosan as a precursor and nanoreactor through a simple calcination technique.176 Sn doping induces WSe2 to transform from the 2H phase into the highly conductive 1T phase, while expanding the interlayer spacing and promoting electron/ion transport (Fig. 9j). N doping and chitosan-derived carbon layers improve the structural stability and form a conductive active phase stable carbon matrix synergistic structure.
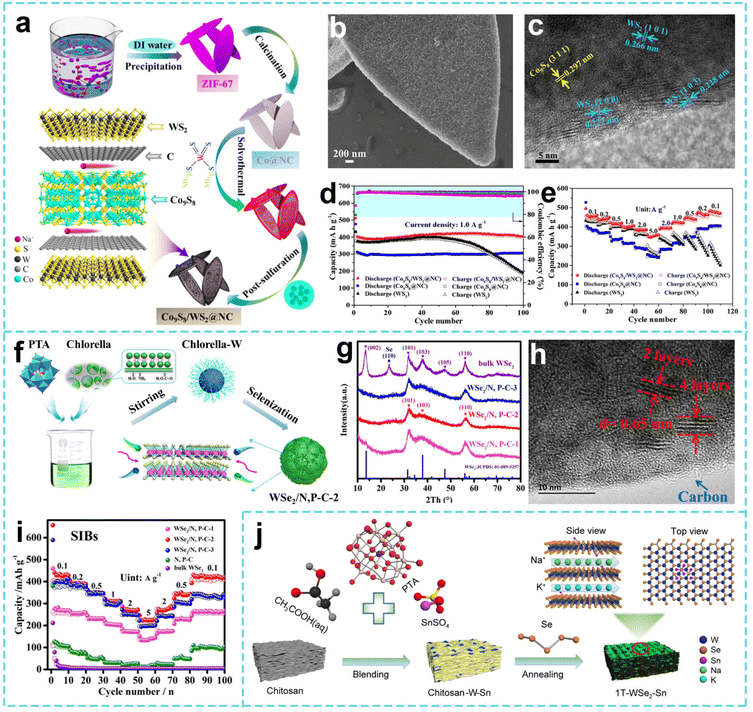 | ||
| Fig. 9 (a) Illustration of the synthesis process, (b) SEM image and (c) HRTEM image of Co9S8/WS2@N. (d) Cycling performance and (e) rate capability of the WS2, Co9S8@NC, Co9S8/WS2@NC electrodes. Reproduced with permission from ref. 174. Copyright 2021, Elsevier. (f) Schematic synthesis of the WSe2/N,P-C-2 composite. (g) XRD patterns of WSe2/N,P-C-1, WSe2/N,P-C-2, WSe2/N,P-C-3, bulk WSe2. (h) HRTEM image of the WSe2/N,P-C-2 composite. (i) The rate capability of electrodes at different current densities. Reproduced with permission from ref. 175. Copyright 2020, Elsevier. (j) Schematic illustration of the synthesis of the 1T-WSe2-Sn composite. Reproduced with permission from ref. 176. Copyright 2023, Springer Nature. | ||
In summary, the heteroatom doping strategy provides a systematic solution to the sodium storage problem of Mo- and W-based anode materials through atomic-level electronic structure regulation and multi-scale performance optimization. From the dual improvement of electron conduction and interface stability by N doping, to the phase structure transformation and interlayer spacing expansion induced by Sn doping and the reaction mechanism innovation brought about by P doping, researchers have gradually constructed a synergistic modification system of electronic structure optimization, ion transport acceleration, and mechanical stress buffering through single heteroatom doping, multi-heteroatom co-doping and bio-template integration. These studies not only reveal the structure–activity relationship between doping elements and material properties, but also achieve a leap from basic performance optimization to the practical application potential of materials through the combination of green synthesis processes and structural engineering.
Compared with 2H-MoS2, 1T-MoS2 is an attractive energy storage material due to its large interlayer spacing and excellent conductivity. The electron filling engineering of the Mo 4d orbital is the core idea to induce the efficient transformation of the 2H phase into the 1T phase. Based on this theory, Peng et al. successfully constructed uniform heterogeneous nanosheets (CuS@MoS2) by coprecipitation and a hydrothermal method (Fig. 10a–c).183 Electron-rich CuS acts as an electron donor, and part of the electrons at the heterogeneous interface are transferred from Cu to Mo, triggering rearrangement of the Mo 4d orbital and the formation of a strong built-in electric field at the interface, and causing the irreversible phase transition of MoS2 from 2H to 1T (Fig. 10d). The practical capacity of CuS@MoS2 remains at 464.18 mAh g−1 after 2700 cycles at a current density of 10 A g−1 (Fig. 10e). This work provides a new perspective for developing high-performance sodium storage anode materials based on 1T-rich MoS2. On this basis, She et al. focused on the structural design of anode materials for SIBs and constructed a 3D porous hollow heterostructure (MXene@MoS2) (Fig. 10f).184 The 3D flower-like structure promotes electrolyte penetration by virtue of its high specific surface area and porous channels (Fig. 10g), effectively inhibits the self-aggregation of MoS2 and buffers the volume expansion, while the solid heterogeneous interface formed by MXene and MoS2 accelerates electron transfer (Fig. 10h). Thus, the practical capacity of MXene@MoS2 remains at 494.4 mAh g−1 after 1000 cycles at 5 A g−1 (Fig. 10i). This study reveals the influence of the electrode material structure on the sodium storage mechanism through the strategy of template construction of the 3D structure and heterogeneous composite formation. Following the research context of structural optimization and interface regulation, Zhao et al. designed and prepared a composite material of layered MoS2/WS2 heterostructure nanostructures (HC@MoS2/WS2@NC) coated with double carbon layers to address the challenges of insufficient reaction kinetics and large volume changes (Fig. 10j).185 The double carbon constrained structure of the internal hard carbon core and the external nitrogen-doped carbon shell is used to anchor MoS2/WS2 to inhibit volume expansion, while the high conductivity of the double carbon layer is used to accelerate electron transport (Fig. 10k). Therefore, the HC@MoS2/WS2@NC material maintains a practical capacity of 333 mAh g−1 after 1000 cycles at 2 A g−1 (Fig. 10l and m).
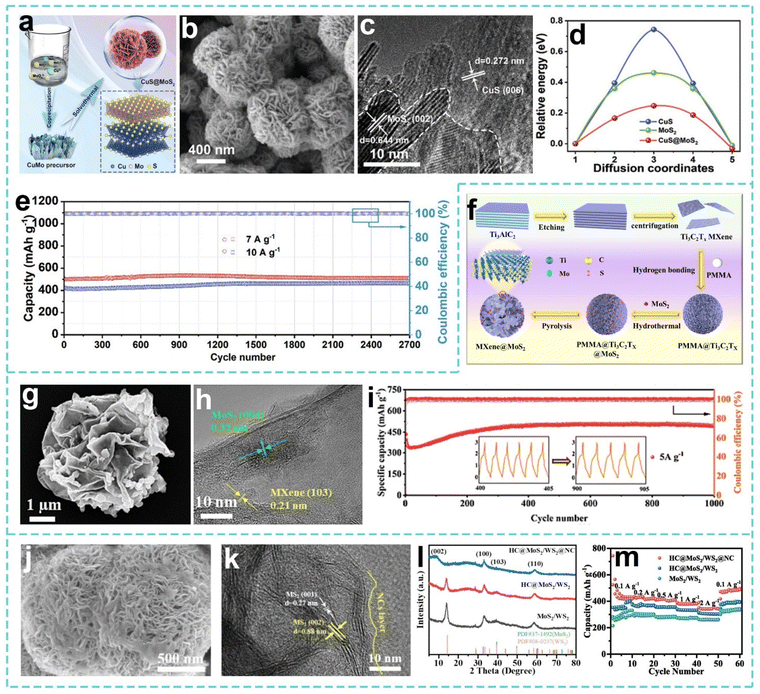 | ||
| Fig. 10 (a) Preparation process for the CuS@MoS2 heterostructure. (b) SEM image and (c) HRTEM image of CuS@MoS2. (d) Diffusion energy barriers of Na+ in CuS, MoS2, and CuS@MoS2. (e) Long-term cycling performance of CuS@MoS2. Reproduced with permission from ref. 183. Copyright 2025, Wiley. (f) Illustration of the synthesis process, (g) SEM image and (h) HRTEM image of MXene@MoS2. (i) Long cycling performance of the MXene@MoS2 electrode. Reproduced with permission from ref. 184. Copyright 2024, Elsevier. (j) SEM image and (k) HRTEM image of HC@MoS2/WS2@NC. (l) XRD patterns of MoS2/WS2, HC@MoS2/WS2, and HC@MoS2/WS2@NC. (m) Rate capability of MoS2/WS2, HC@MoS2/WS2, and HC@MoS2/WS2@NC at different current densities. Reproduced with permission from ref. 185. Copyright 2024, Elsevier. | ||
In summary, heterogeneous interface design has established a 3D performance improvement path of electronic structure optimization, ion transport acceleration, and structural stability enhancement for Mo- and W-based anode materials through a multi-dimensional synergistic mechanism across components and scales (Table 2). From the charge transfer regulation of two-component heterojunctions to the interface engineering innovation of 3D porous structures and core–shell composite systems, this strategy not only reveals the intrinsic relationship between interface effects and sodium storage performance, but also achieves precise control from atomic-level interface regulation to macroscopic structural design through preparation techniques such as the template method and in situ growth. In general, heterojunction interface engineering provides an effective approach to simultaneously tune the electronic structure, accelerate Na+ transport, and enhance the structural stability of Mo- and W-based anode materials. However, the complexity of their structure and composition also poses challenges for engineering feasibility. Multi-component heterostructures typically require precise control over the phase composition, interfacial contact, and spatial distribution of active regions, making their synthesis conditions sensitive and difficult to scale up.186 Furthermore, long-term interfacial stability under repeated sodiation/desodiation cycles remains a critical issue, especially when different components exhibit mismatched mechanical or electrochemical behavior. Therefore, future heterojunction engineering should focus on the systematic evaluation of simplified structures and scalable synthesis methods to ensure that the inherent advantages of interface tuning can be effectively translated into practically viable anode materials for SIBs.
| Materials | Cathode material | Current density (A g−1) | Capacity retention (mAh g−1) | ICE (half/full) | Capacity retention (%)/cycles | Ref. |
|---|---|---|---|---|---|---|
| MoS2@CPDs-2 | Na3V2(PO4)3F3 | 0.5 | 472.4 | 82.3% (full) | 62.5/500 | 146 |
| MoS2/MWCNT | — | 0.1 | 527.7 | 67.5% (half) | 100.0/110 | 147 |
| NCF@V-MoS2 | — | 1.0 | 413.0 | 64.8% (half) | —/300 | 148 |
| NGQDs-WS2/3DCF | Na0.44MnO2 | 2.0 | 268.4 | 65.1% (half) | 97.1/1000 | 150 |
| M-MoS2@HCS | — | 0.1 | 401.3 | 34.6% (half) | —/100 | 157 |
| MoS2/C | Na3Fe2(PO4)3 | 1.0 | 515.8 | 88.7% (full) | 94.3/1000 | 158 |
| NSC@WS2 | — | 5.0 | 234.8 | 71.2% (half) | 51.2/900 | 159 |
| MoSe2/Mo2CTx/C | — | 2.0 | 238.4 | 47.3% (half) | 86.3/2200 | 163 |
| MoSe2@MoxN/C-I | — | 20.0 | 178.6 | 42.9% (half) | 98.8/10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
164 |
| WSSe2/C-1 | AC | 1.0 | 715.3 | 56.4% (full) | 70.1/200 | 165 |
| H-MoO3/MoO2@NC | — | 0.5 | 190.0 | 38.9% (half) | 38.9/5000 | 172 |
| MoS2@NC | Na3V2(PO4)3 | 20.0 | 428.0 | 64% (full) | 98.7/10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
173 |
| Co9S8/WS2@N | — | 5.0 | 359.0 | 81.7% (half) | 78.0/120 | 174 |
| WSe2/N,P-C-2 | Na3V2(PO4)3 | 1.0 | 155.0 | 59% (full) | —/5300 | 175 |
| CuS@MoS2 | Na3V2(PO4)3 | 5.0 | 506.0 | 78% (full) | 98.2/3200 | 183 |
| MXene@MoS2 | Na3V2(PO4)3/C | 5.0 | 494.4 | 78.2% (full) | 79.9/1000 | 184 |
| HC@MoS2/WS2@C | Na3V2(PO4)3 | 2.0 | 333.0 | 64% (full) | 39.8/1000 | 185 |
5. Research progress on Mo- and W-based materials in SIB anodes
5.1 Mo-/W-based oxides
Metal oxides have attracted much attention in research on anode materials for SIBs due to their high theoretical specific capacity. Among them, Mo- and W-based oxides, as potential anode materials, exhibit unique physical and chemical properties and are expected to provide new solutions for improving the performance of SIBs.187 Mo-based oxide has a variety of oxidation states, such as MoO2, MoO3, etc. Its crystal structure and electronic structure enable it to undergo reversible redox reactions during the insertion and de-insertion of Na+, thereby achieving charge storage.188 W-based oxides, such as WO3, WO2, etc., also have rich crystal structures and chemical properties and show potential application value in the field of energy storage. Through the structural design, morphology control and compounding of Mo/W oxides with other materials, their electrochemical properties can be further optimized to improve the energy density, cycle life and rate performance of SIBs.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) C). Similarly, Li et al. constructed amorphous/crystalline MoO2 (a/c-MoO2) homojunction materials with multiple interfaces and defects in the a/c-MoO2 nanoclusters by a one-pot solvothermal method.190 Theoretical simulations show that electrons are redistributed at the uniform interface of a/c-MoO2 to form a built-in electric field effect, which can significantly improve the adsorption capacity of Na+ and reduce the diffusion energy barrier, promote the intercalation/deintercalation of Na+ in the material and improve the cycling stability. As a result, a/c-MoO2 achieves a practical capacity of 307 mAh g−1 at 0.1 A g−1, and can achieve 3000 stable cycles (Fig. 11d and e). At the same time, the in situ X-ray diffraction (XRD) test reveals its “adsorption–embedding–conversion” sodium storage mechanism (Fig. 11f). Based on the improvement in the performance of SIBs by interface defects, Zhou et al. designed a heterojunction material with a carbon-free layered structure (MoS2/MoO2).191 The HRTEM image confirms that the hybridization between MoS2 and MoO2 forms a large amount of heterogeneous interfaces (Fig. 11g). In addition, the heterojunction enhances the Na+ adsorption energy and reduces the Na+ diffusion energy barrier, promoting efficient ion transport (Fig. 11h). Thus, the capacity of MoS2/MoO2 is maintained at almost 100% after 100 cycles at 1 A g−1 (Fig. 11i).
C). Similarly, Li et al. constructed amorphous/crystalline MoO2 (a/c-MoO2) homojunction materials with multiple interfaces and defects in the a/c-MoO2 nanoclusters by a one-pot solvothermal method.190 Theoretical simulations show that electrons are redistributed at the uniform interface of a/c-MoO2 to form a built-in electric field effect, which can significantly improve the adsorption capacity of Na+ and reduce the diffusion energy barrier, promote the intercalation/deintercalation of Na+ in the material and improve the cycling stability. As a result, a/c-MoO2 achieves a practical capacity of 307 mAh g−1 at 0.1 A g−1, and can achieve 3000 stable cycles (Fig. 11d and e). At the same time, the in situ X-ray diffraction (XRD) test reveals its “adsorption–embedding–conversion” sodium storage mechanism (Fig. 11f). Based on the improvement in the performance of SIBs by interface defects, Zhou et al. designed a heterojunction material with a carbon-free layered structure (MoS2/MoO2).191 The HRTEM image confirms that the hybridization between MoS2 and MoO2 forms a large amount of heterogeneous interfaces (Fig. 11g). In addition, the heterojunction enhances the Na+ adsorption energy and reduces the Na+ diffusion energy barrier, promoting efficient ion transport (Fig. 11h). Thus, the capacity of MoS2/MoO2 is maintained at almost 100% after 100 cycles at 1 A g−1 (Fig. 11i).
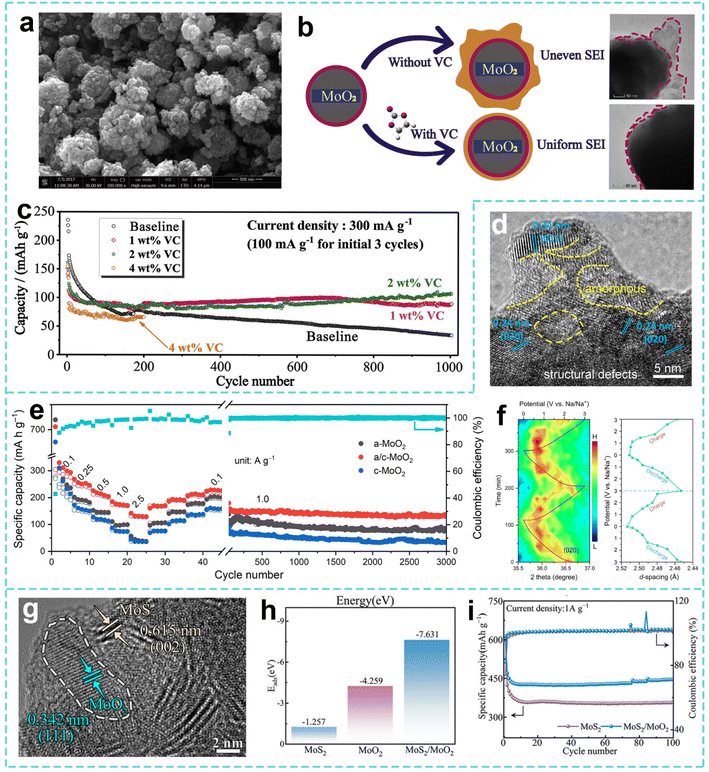 | ||
| Fig. 11 (a) SEM image of MoO2 powders. (b) Corresponding schematic illustration and TEM images of the MoO2 electrodes after 1000 cycles with baseline electrolyte and 2 wt% VC-containing electrolyte. (c) Comparison of the cycling stability of the MoO2 electrodes without and with VC additive. Reproduced with permission from ref. 189. Copyright 2020, Elsevier. (d) HRTEM image of a/c-MoO2. (e) Rate and cycling performance of the MoO2 electrodes. (f) Contour plot of the (020) peak evolution with the corresponding discharge–charge profiles in the initial two cycles and d(020)-spacing change as a function of applied potential. Reproduced with permission from ref. 190. Copyright 2023, Elsevier. (g) HRTEM image of MoS2/MoO2. (h) Calculated Na+ ion adsorption energy of MoO2, MoS2, and MoS2/MoO2. (i) Cycling performance of MoS2/MoO2. Reproduced with permission from ref. 191. Copyright 2025, Wiley. | ||
Compared with MoO2 materials, WO2 also has good application potential in SIB anode materials. For example, Xu et al. constructed a porous WO2/WS2-reduced graphene oxide (WO2/WS2–rGO) hetero-composite anode and P-doped biomass carbon cathode by a hydrothermal method combined with freeze drying and calcination processes.192 They also optimized the Na+ diffusion kinetics through heterogeneous interface synergy and 3D porous design, providing new ideas for low-cost, high-performance energy storage devices. As anode materials for SIBs, MoO2 and WO2 have made significant progress in structural design, interface engineering and electrolyte optimization. Through strategies such as nanostructuring, heterostructure construction, element doping and crystal surface regulation, the electron transport, ion diffusion and structural stability of the materials have been effectively improved. This will provide an important theoretical basis and technical inspiration for the development of high energy density and long life SIBs.
As an anode material for SIBs, MoO3 has made significant progress in recent years through multidimensional structural engineering strategies. Different researchers have collaboratively solved bottleneck problems such as poor conductivity, severe volume expansion and insufficient cycling stability from different angles. From the perspective of interlayer engineering, Wang et al. developed a two-step synthesis strategy based on partial reduction and organic molecular intercalation.196 By introducing bismuth thiol (DMcT) molecular intercalation, the interlayer spacing of MoO3 was extended from 6.92 Å to 10.40 Å (Fig. 12a), significantly improving the diffusion kinetics of Na+. This DMcT-MoO3 structure not only reduces the charge transfer resistance, but also enhances the interlayer stability through intermolecular forces (Fig. 12b and c), thereby improving the Na+ insertion–extraction kinetics. Furthermore, it has a long cycling stability and high rate performance (Fig. 12d). On this basis, Yang et al. further optimized the material structure through a nano-sizing strategy. As shown in Fig. 12e, the layered MoO3 nanoplates (MoO3 NPs) were prepared by pyrolysis of Mo-based metal organic frameworks, the unique 2D nanostructure of which effectively alleviated volume expansion during the charging and discharging process. Thanks to the shortened ion diffusion path and exposed active sites, the material maintained a practical capacity of 154 mAh g−1 after 1200 cycles at 50 mA g−1 (Fig. 12f).197 This nano-design approach complements interlayer engineering and jointly solves the problem of structural stability. In terms of mass loading, the MoO3 electrode in the assembled coin cell reportedly had a mass loading of 1–2 mg cm−2, and the electrolyte was NaClO4 in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of ethylene carbonate (EC) and diethyl carbonate (DEC). To further improve the electrochemical kinetics, Yu et al. synthesized a nanorod-shaped MoO3–MoS2 heterostructure by a two-step method (Fig. 12g). The built-in electric field of the heterojunction formed by interface engineering significantly accelerated charge transfer, allowing the material to maintain a practical capacity of 316 mAh g−1 at an ultra-high current density of 10 A g−1, and still maintain a practical capacity of 286 mAh g−1 after 2300 cycles at 5 A g−1. In situ XRD revealed that the high specific capacity of MoO3–MoS2 was attributed to the synergistic energy storage mechanism between the intercalation reaction of MoO3 and the conversion reaction of MoS2 (Fig. 12h and i). Additionally, the study reported a mass loading of 5.47 mg cm−2 for the MoO3–MoS2 electrodes prepared via 3D printing technology, demonstrating that the areal capacity increased with the mass loading, reaching up to 5.47 mg cm−2.198
1 mixture of ethylene carbonate (EC) and diethyl carbonate (DEC). To further improve the electrochemical kinetics, Yu et al. synthesized a nanorod-shaped MoO3–MoS2 heterostructure by a two-step method (Fig. 12g). The built-in electric field of the heterojunction formed by interface engineering significantly accelerated charge transfer, allowing the material to maintain a practical capacity of 316 mAh g−1 at an ultra-high current density of 10 A g−1, and still maintain a practical capacity of 286 mAh g−1 after 2300 cycles at 5 A g−1. In situ XRD revealed that the high specific capacity of MoO3–MoS2 was attributed to the synergistic energy storage mechanism between the intercalation reaction of MoO3 and the conversion reaction of MoS2 (Fig. 12h and i). Additionally, the study reported a mass loading of 5.47 mg cm−2 for the MoO3–MoS2 electrodes prepared via 3D printing technology, demonstrating that the areal capacity increased with the mass loading, reaching up to 5.47 mg cm−2.198
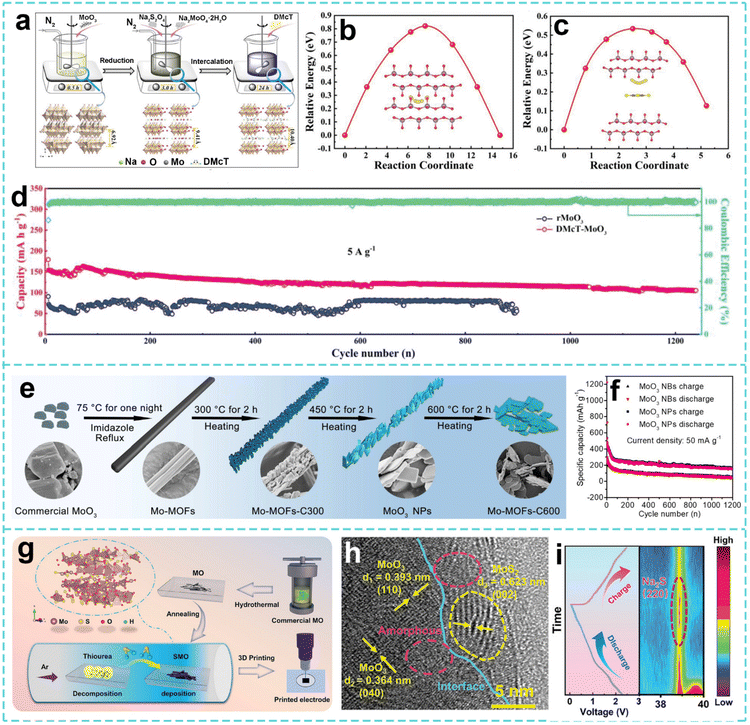 | ||
| Fig. 12 (a) Illustration of the synthesis process for DMcT-MoO3. Diffusion pathways and diffusion barriers of Na shuttles between (b) MoO3 and (c) DMcT-MoO3. (d) Long cycling performances of the rMoO3 and DMcT-MoO3 electrodes. Reproduced with permission from ref. 196. Copyright 2020, Wiley. (e) Diagram of the MoO3 NP synthesis process with increasing temperature. (f) Cycling performance of MoO3 NBs and MoO3 NPs at 50 mA g−1. Reproduced with permission from ref. 197. Copyright 2020, Springer Nature. (g) Illustration of the synthesis process, (h) HRTEM image of MoO3–MoS2. (i) In situ XRD evolution profiles of the MoO3–MoS2 electrode for the 1st discharge–charge process at 0.1 A g−1. Reproduced with permission from ref. 198. Copyright 2024, Wiley. | ||
These studies have systematically improved the sodium storage performance of MoO3-based anode materials through multi-dimensional innovations such as interlayer regulation, nanostructure design, heterogeneous interface engineering, morphology optimization and composite strategies. From the basic interlayer expansion to macroscopic structure design, and from single material optimization to multi-component synergy, it demonstrates the strong vitality of material engineering strategies in solving key material problems of SIBs, and provides an important technical route for the subsequent development of high-performance energy storage devices.
Unlike the rich layered structure of MoO3, WO3 has great application potential in the anode materials of SIBs because of its controllable porous structure and defects. García-García et al. prepared a WO3 film with a porous nanocolumn structure, the unique pores and surface properties of which significantly improved the Na+ diffusion efficiency and storage capacity. The separated nanocolumns and large number of pores allow Na+ to penetrate deeply and evenly into the film, improving the cycling stability of the material.199 Inspired by this, Zimmer et al. tried to make further breakthroughs from the structural dimension. They synthesized hollow WO3 microspheres constructed by self-assembling interconnected nanosheets through a hydrothermal method. Their unique interconnected hollow structure provided abundant active sites and volume expansion buffer space, achieving a high reversible capacity of 431 mAh g−1 at 100 mA g−1. The practical capacity recovered to 220 mAh g−1 after cycling, significantly optimizing the sodium ion diffusion kinetics and cycling stability.200
MoO3- and WO3-based materials have significantly optimized the sodium storage kinetics and cycling stability through interlayer engineering, heterogeneous structure design (such as MoO3–MoS2 interface electric field) and multi-dimensional composite systems (such as vertical nanosheet arrays and 3D hollow microspheres). This will be key in structural innovation and mechanistic exploration, providing theoretical support and technical pathways for the development of the next generation of low-cost, high-safety SIBs.
5.2 Mo-/W-based chalcogenides
Mo- and W-based chalcogenides typically possess a layered structure, with weak van der Waals forces between the layers, facilitating the intercalation and deintercalation of Na+, thereby enabling charge storage and release. Furthermore, Mo- and W-based chalcogenides exhibit multi-electron reaction activity, offering higher theoretical specific capacities. For example, MoS2 boasts a theoretical capacity of up to 670 mAh g−1, significantly higher than that of traditional graphite anode materials. Moreover, their electronic structure can be optimized by tuning their structure and composition, thereby improving the electrochemical performance and cycle life. Introducing defects, doping with other heteroatoms, or compositing with carbon-based materials can further enhance the conductivity and structural stability of these materials.201 In SIBs, the electrolyte plays a crucial role in the stability and performance of Mo- and W-based anodes. Compared to carbonate-based electrolytes, ether-based electrolytes generally exhibit better compatibility with Mo- and W-based layered sulfides and selenides. Ether solvents can form a more stable SEI layer on the surface of Mo- and W-based materials, protecting the anode during cycling and minimizing side reactions. However, the oxidative stability of ether-based electrolytes at high voltages remains a concern, as they are prone to decomposition under extreme conditions, potentially leading to undesirable side reactions. On the other hand, commonly used carbonate-based electrolytes also have limitations when paired with Mo- and W-based materials. While they tend to form a more stable and robust SEI layer, the high reactivity of unsaturated active sites on the Mo- and W-based surface with carbonate solvents leads to significant decomposition and the formation of gaseous byproducts, such as CO2 and CO. These byproducts not only increase internal resistance but also degrade the SEI layer, exacerbating capacity fading and shortening the cycle life.202 To improve the compatibility of electrolytes with Mo- and W-based anodes, future research should focus on developing electrolyte systems that are better matched to the unique surface chemistry and structural characteristics of these materials. This may include exploring novel high-concentration electrolytes to reduce side reactions and enhance SEI stability, as well as optimizing solvent selection to improve their oxidative stability at high voltages. Furthermore, solid-state electrolytes possess excellent chemical stability and minimal side reactions, offering a promising solution to address electrolyte compatibility issues. They can provide a stable interface, thereby improving the overall performance and lifespan of Mo- and W-based anodes in SIBs.Interlayer spacing regulation provides the basic conditions for Na+ transport and storage. Therefore, Tao et al. focused on optimizing the interlayer spacing of MoS2 by ion intercalation, and used density functional theory simulations to determine the ideal spacing range of 0.80 to 1.01 nm (Fig. 13a). At the same time, by regulating the mass ratio of 1-butyl-3-methylimidazolium acetate ([BMIm]Ac) ionic liquid to cellulose, the controllable intercalation of cellulose oligomers was achieved, and then porous carbon-supported interlayer expanded MoS2 was synthesized. As shown in Fig. 13b and c, this structure not only reduces the diffusion barrier of Na+ and shortens the Na+ diffusion path, but also limits the loss of sulfur in the cycle through the carbon matrix, thereby improving the cycle life, so that the material still maintains a practical capacity of 517.79 mAh g−1 after 250 cycles at 0.1 A g−1, highlighting the dual improvement of interlayer spacing optimization on sodium storage efficiency and structural stability (Fig. 13d and e).207 Based on the regulation of interlayer spacing, electronic structure optimization has become a key extension to improve the sodium storage activity. Fan et al. optimized the electronic structure by replacing atomic elements within the MoS2 layer, thereby changing its inherent physical properties. By reducing the band gap, the surface Na+ adsorption energy and electronic conductivity were enhanced, thereby effectively optimizing the electrochemical performance of sodium storage.208 Defect engineering breaks through the performance bottleneck of traditional structures at the atomic scale. As shown in Fig. 13f, Li et al. developed a hollow micro-cubic framework constructed of ultra-thin MoS2 nanosheets (HMF-MoS2) rich in molybdenum defects through a zeolite-like framework-driven strategy. The hollow micro-cubic framework can be clearly seen from the SEM and TEM images in Fig. 13g and h. This structure has a good practical capacity of 267 mAh g−1 after 125 cycles at a current density of 1 A g−1 (Fig. 13i).209 The abundant Mo vacancies in MoS2 can accelerate charge transfer and enhance the interaction between MoS2 and Na+, thereby promoting sodium storage. Sun et al. successfully prepared 1T phase ultrathin MoS2 nanosheets (1T MoS2) from 2H MoS2 by thermally driven sodium ion insertion assisted exfoliation. Compared with 2H MoS2, 1T MoS2 has a lower Na+ diffusion barrier, higher electronic conductivity, atomic mobility, and sodium affinity, as well as inhibiting the release of sulfur species and reducing phase changes during sodiation (Fig. 13j and k). Its capacity retention rate after 200 cycles at 1 A g−1 is as high as 94%, and it has good cycling stability (Fig. 13l).210
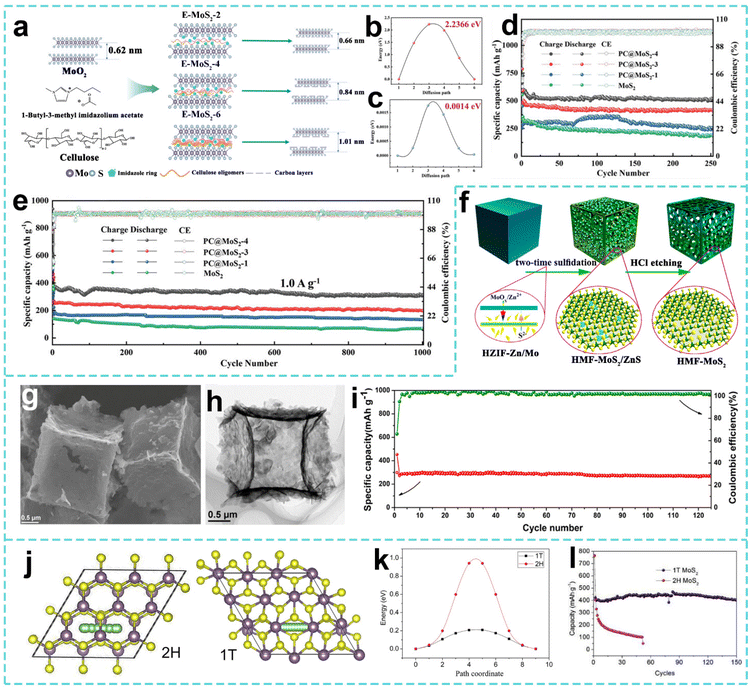 | ||
| Fig. 13 (a) Schematic illustration of the formation of different MoS2 interlayer distances. Migration energy barrier of (b) 0.62 nm and (c) 1.01 nm interlayer distances of MoS2. Cycling performances of PC@MoS2-4, PC@MoS2-3, PC@MoS2-1, and MoS2 at (d) 0.1 A g−1 and (e) 1 A g−1. Reproduced with permission from ref. 207. Copyright 2023, Wiley. (f) Schematic illustration of the formation of HMF-MoS2. (g) SEM image and (h) TEM image of HMF-MoS2. (i) Cycling performance of HMF-MoS2 at 1 A g−1. Reproduced with permission from ref. 209. Copyright 2019, American Chemical Society. (j) Corresponding crystal structures and (k) Na atom diffusion barriers of 2H and 1T MoS2. (l) Cycling performance of 2H and 1T MoS2 at 0.1 A g−1. Reproduced with permission from ref. 210. Copyright 2019, Elsevier. | ||
Similarly, in order to optimize the structural stability and electrochemical performance of WS2 in the anode material of SIBs, researchers have made important progress through innovative material design strategies. They conducted research from the dimensions of structural regulation, interface engineering and composite system construction, and gradually formed a technical path of multi-dimensional synergistic strengthening. Choi et al. prepared 3D reduced graphene oxide microspheres modified with layered WS2 nanosheets (WS2-3D rGO) by a two-step method of spray pyrolysis combined with sulfurization. In this design, the 3D rGO skeleton not only effectively inhibits the stacking of WS2 nanosheets, but also provides a buffer space for volume expansion during the intercalation/deintercalation of Na+, giving full play to the synergistic advantages of the high theoretical capacity of WS2 and the high conductivity of graphene.211 However, WS2 obtained by traditional chemical stripping still faced the problem of limited interlayer stripping efficiency, which prompted researchers to explore more efficient stripping techniques. Zhang et al. developed a molten salt electrolytic stripping method (Fig. 14a), using Na+ to uniformly intercalate WS2 under an electric field, and they successfully prepared a material with a few-layer lamellar structure (WS2–NaCl) (Fig. 14b). By optimizing the molten salt system and electrolysis parameters, the obtained WS2–NaCl-25 mA electrode still maintained a reversible capacity of 373 mAh g−1 after 100 cycles at 0.1 A g−1, and also showed a good cycling performance and rate performance. The mass loading of the WS2 material used as the anode in the SIB was 1.5 mg cm−2, and the electrolyte used was a NaClO4 solution dissolved in propylene carbonate (PC) with the addition of 5% fluoroethylene carbonate (FEC) (Fig. 14c).212 In terms of structural engineering, Lin et al. developed a different approach for constructing a hollow bimetallic heterojunction WS2@MnS material. Fig. 14d shows different views of the simulation model before and after Na+ insertion into the structure. WS2@MnS has a much lower Na+ diffusion energy than pure WS2 due to its unique heterostructure preparation; this successfully demonstrates that the construction of a functional heterostructure is beneficial for Na+ diffusion kinetics. The designed hollow structure both relieves cycling stress and increases the specific surface area (Fig. 14e). As shown in Fig. 14f, this synergistic effect enables the material to achieve 2000 ultra-long cycles at 0.1 A g−1.213 It is worth noting that the above-mentioned powder electrode materials still face the cumulative effect of volume expansion. For this reason, the Dey team pioneered the embedding of WS2 nanotubes (WS2NTs) into the (SiOC) ceramic fiber matrix. The 3D ceramic fiber network prepared by electrospinning combined with high-temperature pyrolysis not only effectively inhibited volume change to the WS2 nanotubes, but also blocked the dissolution of polysulfides through the chemical stability of the carbon oxide carbide matrix.214
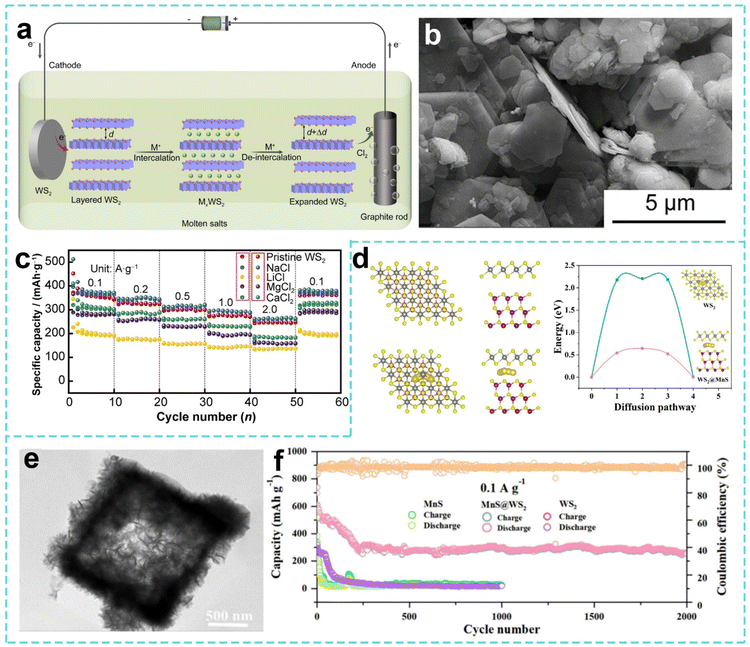 | ||
| Fig. 14 (a) Illustration of the electrochemical exfoliation process for WS2 in molten salt. (b) SEM image of WS2–NaCl-25 mA. (c) Rate performance at various current densities of pristine WS2 and WS2 treated with different molten salts. Reproduced with permission from ref. 212. Copyright 2023, Springer Nature. (d) Optimized model of WS2@MnS heterostructures before or after Na+ insertion and energy barriers of the diffusion behavior of Na+ in pure WS2 and WS2@MnS heterostructures. (e) TEM image of WS2@MnS heterostructures. (f) Cycling performances of pure WS2, pure MnS and WS2@MnS electrodes. Reproduced with permission from ref. 213. Copyright 2024, Elsevier. | ||
From the above research, although MoS2 and WS2 materials have application potential in SIBs, there are problems such as structural degradation, kinetic sluggishness and side reactions. Researchers achieved performance breakthroughs through in multi-scale modification strategies: (1) at the microscopic level, the interlayer spacing was precisely controlled with the help of DFT calculations, and the diffusion kinetics and electronic structure were optimized by combining ionic liquid intercalation technology and element substitution. (2) At the mesoscopic and macroscopic scales, hollow microcubic frameworks containing molybdenum vacancies and 1T/2H heterogeneous phase coral reef-like structures were constructed, and defect engineering and special structural design were used to provide sodium storage sites and alleviate volume expansion. At the same time, 3D graphene was composited and a conductive network was constructed by molten salt stripping to improve the conductivity and structural stability. These studies have solved material application problems from different angles and provided important technical references for the development of high-performance SIBs.
In the study of anode materials for SIBs, different research teams have conducted multi-faceted explorations of the key bottlenecks of layered transition metal disulfides. In response to the problem of selenium dissolution and kinetic limitations on the surface of layered transition metal sulfides (TMDs), Kang et al. proposed a heterogeneous MoSe2 nanoflower structure modified with surface amorphous MoO3 (MoSe2/MoO3) (Fig. 15a). Through a solvothermal reaction combined with a controllable oxidation strategy, a uniform amorphous MoO3 protective layer was constructed on the surface of the flower-like MoSe2 to form a unique heterogeneous interface (Fig. 15b and c). As shown in Fig. 15d and e, the material has a first-cycle discharge capacity of 729 mAh g−1 at a current density of 50 mA g−1, reaching 89% of the theoretical capacity, and has a good rate performance.217 The breakthrough in interface engineering laid the foundation for subsequent structural design. Liu et al. proposed the concept of 2D nanosheets combined with 3D porous carbon for collaborative design, and developed a composite material of 3D ordered macroporous carbon loaded with ultra-thin 2D MoSe2 nanosheets. This system shortens the Na+ diffusion path through the 0.76 nm ultra-large interlayer spacing of 2D MoSe2 and alleviates volume expansion with the ordered cavities of 3D ordered macroporous (3DOM) carbon.218 As the 3D structure design gradually matured, Li et al. shifted the focus of their research from macroscopic structure to microscopic synthesis mechanisms, exploring the regulatory effect of polymers on the growth of nanoparticles.
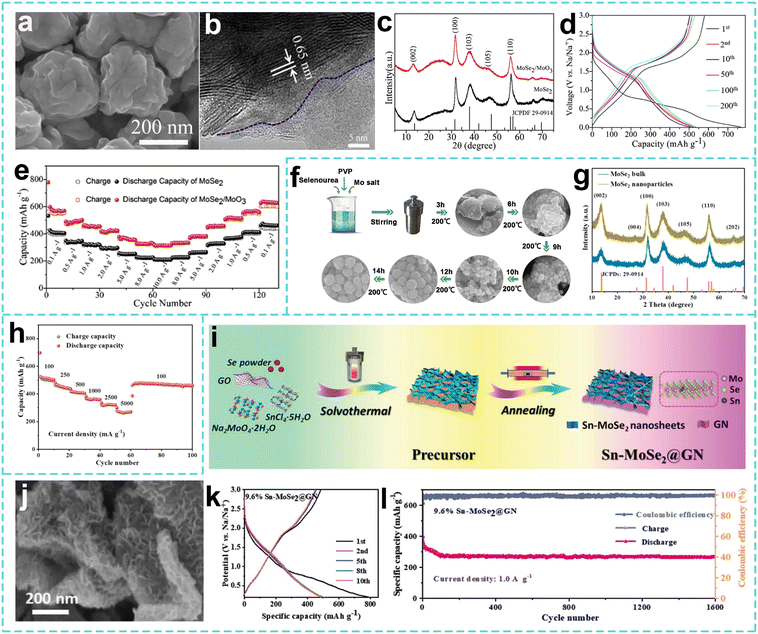 | ||
| Fig. 15 (a) SEM image and (b) HRTEM image of MoSe2/MoO3 composite. (c) XRD patterns of MoSe2 nanostructure and MoSe2/MoO3 composite. (d) Typical discharge/charge profiles of MoSe2/MoO3 composite anode. (e) Rate capability for MoSe2 and MoSe2/MoO3 composite. Reproduced with permission from ref. 217. Copyright 2018, Elsevier. (f) The detailed formation mechanism of the MoSe2 nanoparticles. (g) XRD patterns of MoSe2 nanoparticles and MoSe2 bulk. (h) Rate performance of the MoSe2 nanoparticles electrode. Reproduced with permission from ref. 219. Copyright 2022, Elsevier. (i) Schematic and the synthesis process, (j) SEM image of Sn-MoSe2@GN. (k) GCD profiles, (l) Long-term cycling performance of the Sn-MoSe2@GN electrode. Reproduced with permission from ref. 220. Copyright 2022, Wiley. | ||
As shown in Fig. 15f and g, by introducing polyvinyl pyrrolidone into a hydrothermal method, the excessive aggregation of MoSe2 nanosheets was suppressed by its steric hindrance effect, and the orderly assembly of MoSe2 nanosheets was guided to form uniform nanoparticles. The hierarchical structure formed optimized the electron transmission path and exposed more active sites. The reversible capacity of the material reached 374.9 mAh g−1 after 100 cycles at 500 mA g−1, and it also had a good rate performance. For the SIB application, the material loading of MoSe2 nanoparticles was 2.0 mg cm−2, and the standard electrolyte for SIBs was used (Fig. 15h).219 While the structural design and synthesis process are being continuously optimized, Hu et al. shifted their focus to the regulation of the intrinsic properties of the material. By doping tin into MoSe2 nanosheets grown in situ on a graphene (GN) substrate, they prepared an anode material (Sn-MoSe2@GN) (Fig. 15i and j).220 As shown in Fig. 15k, due to the formation of the SEI layer, the initial discharge/charge specific capacity of the material is 786.2/506.8 mAh g−1, and the initial coulombic efficiency is 64.46%. In addition, the practical capacity remains at 268.5 mAh g−1 after 1600 cycles at 1 A g−1 (Fig. 15l), providing a new paradigm for the combined application of chemical doping and electrochemical regulation.
Based on the optimization of a single energy storage function, Cho et al. prepared a mesoporous WSe2 reduced graphene oxide (WSe2–rGO) composite material by spray pyrolysis (Fig. 16a). As seen from Fig. 16b and c, the WSe2–rGO composite particles have both well-dispersed rGO nanosheets and many well-faceted WSe2 nanocrystals with folded edges. The discharge capacities of the WSe2–rGO composite particles and the bare WSe2 particles in the 100th cycle under Na+ storage at a current density of 0.5 A g−1 are 238 and 36 mAh g−1, respectively (Fig. 16d). Due to the synergistic effect of rGO and WSe2, the rGO network improves the electronic conductivity, the mesoporous structure shortens the ion diffusion path, and the wrinkled WSe2 nanocrystals expose abundant active sites, thereby improving its electrochemical performance.221 As the research deepened, Shinde et al. proposed an atomically thin (WO3/WSe2) heterostructure (Fig. 16e) to address the challenge of slow Na+ diffusion kinetics in aqueous sodium storage. As shown in Fig. 16f, an atomically thin WO3/WSe3 heterostructure was constructed by a hydrothermal method combined with chemical vapor deposition (CVD) and anchored on a carbon fiber substrate. The WO3/WSe3 electrode has a specific capacitance of 378.1 F g−1 at 1 A g−1, and its performance is stable over more than 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles (Fig. 16g), providing theoretical and experimental dual verification for the interface electronic regulation of 2D heterojunctions (Fig. 16h).222
000 cycles (Fig. 16g), providing theoretical and experimental dual verification for the interface electronic regulation of 2D heterojunctions (Fig. 16h).222
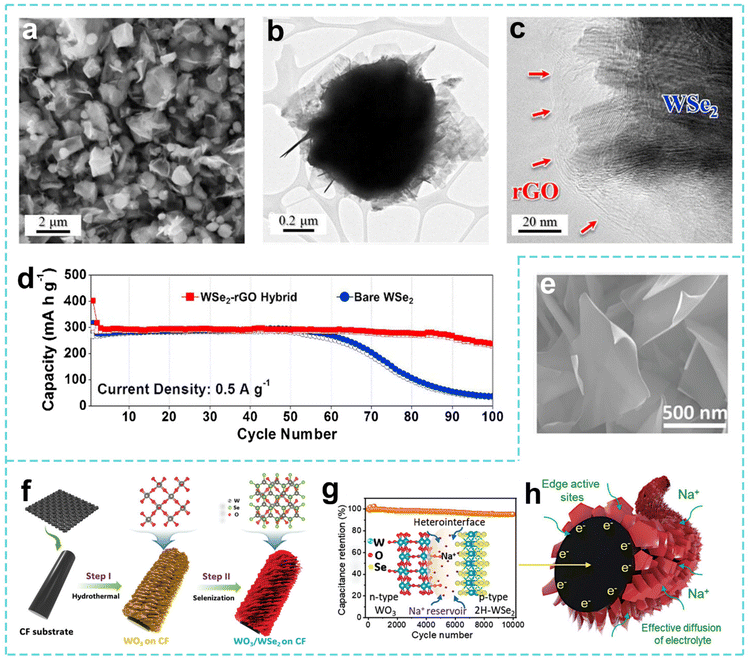 | ||
Fig. 16 (a) SEM image, (b) TEM image, and (c) HRTEM image of the WSe2–rGO composite. (d) Cycling performances of the WSe2–rGO electrode. Reproduced with permission from ref. 221. Copyright 2018, Elsevier. (e) SEM image of the WO3/WSe2 heterostructures. (f) Two-step synthesis protocol for the WO3/WSe2 heterostructures on CF. (g) Cycling stability measured for 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at 10 A g−1. The inset displays the schematics for the improved electrochemical performance of WO3/WSe2. (h) Schematic illustration of Na+ ion storage in the WO3/WSe2 heterostructure. Reproduced with permission from ref. 222. Copyright 2024, Wiley. 000 cycles at 10 A g−1. The inset displays the schematics for the improved electrochemical performance of WO3/WSe2. (h) Schematic illustration of Na+ ion storage in the WO3/WSe2 heterostructure. Reproduced with permission from ref. 222. Copyright 2024, Wiley. | ||
These studies revolve around the core bottleneck of layered TMDs in the anode application of SIBs, and innovations have been made at multiple levels, including surface engineering, 3D architecture, synthesis regulation, chemical bonding, doping optimization, and heterogeneous interfaces. They have constructed a theoretical system for the coordinated optimization of “structure–interface–function”, and formed a complete research chain from macroscopic design to microscopic mechanistic analysis. From the synergistic effect of flower-like nanostructures and surface amorphous layers to the spatial coupling of 3D porous carbon substrates and 2D nanosheets and the charge transfer optimization of heterogeneous interfaces, researchers have successfully achieved breakthroughs in the sodium storage dynamics of materials, structural stability, and versatility.
5.3 Mo- and W-based carbon composites
Mo- and W-based carbon composites combine the unique sodium storage mechanism of Mo- and W-based materials with the excellent conductivity and structural stability of carbon materials, showing great research value in the field of SIB anodes. In terms of sodium storage performance, Mo- and W-based materials combined with carbon materials provide adsorption sites and promote electron transfer to achieve a high specific capacity. In terms of cycling stability, carbon materials buffer the volume changes of Mo- and W-based materials, reduce structural damage, and improve cycling stability. In terms of conductivity, carbon materials construct electron transfer networks, make up for the conductivity defects of Mo- and W-based materials, and improve the rate performance.223 In terms of electrode density and volumetric energy density, Mo- and W-based carbon composite materials generally exhibit a higher packing density than that of traditional hard carbon materials, which helps to improve the volumetric energy density of batteries. Typical MoS2/WS2–carbon composites have a higher packing density than traditional hard carbon. This higher packing density not only increases the amount of active material per unit volume but also reduces the loss in volumetric energy density, thereby improving the overall energy density of the battery. By optimizing the carbon content and material structure, Mo- and W-based carbon composites can significantly increase the electrode density while maintaining the specific capacity, reducing the volume loss caused by low-density materials.224,225 In terms of electrolyte compatibility, carbon materials improve the interface compatibility, form a stable SEI membrane, and improve the coulombic efficiency and overall performance; this provides strong support for the development of SIBs.In the study of Mo-based carbon composite material systems, researchers have continuously broken through the performance limits of traditional materials through multi-dimensional structural design strategies. Based on this problem of slow interlayer diffusion and insufficient cycling stability of 2D MoS2-based SIB anode materials, Wang et al. constructed vertically oriented MoS2/C nanosheet array (MoS2/C NSA) materials on the surface of 3D carbon cloth through micro-area etching and functionalization strategies (Fig. 17a and b). The MoS2 interlayer spacing was expanded to 0.99 nm through heterogeneous interface engineering, and the Na+ diffusion coefficient was increased by 3 times by combining the surface contact advantages of the carbon cloth substrate, achieving a high sodium storage capacity and fast charge and discharge capabilities. The material maintained a practical capacity of 271 mAh g−1 after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at 3 A g−1. The material consists of MoS2/C heterostructures on carbon cloth, with a mass loading of 1.1 mg cm−2. The electrolyte used in this study was NaClO4 in a typical PC solution, with the addition of 5 wt% FEC as an electrolyte additive (Fig. 17c and d). This 3D heterogeneous interface system solves the bottleneck of interlayer diffusion kinetics.226
000 cycles at 3 A g−1. The material consists of MoS2/C heterostructures on carbon cloth, with a mass loading of 1.1 mg cm−2. The electrolyte used in this study was NaClO4 in a typical PC solution, with the addition of 5 wt% FEC as an electrolyte additive (Fig. 17c and d). This 3D heterogeneous interface system solves the bottleneck of interlayer diffusion kinetics.226
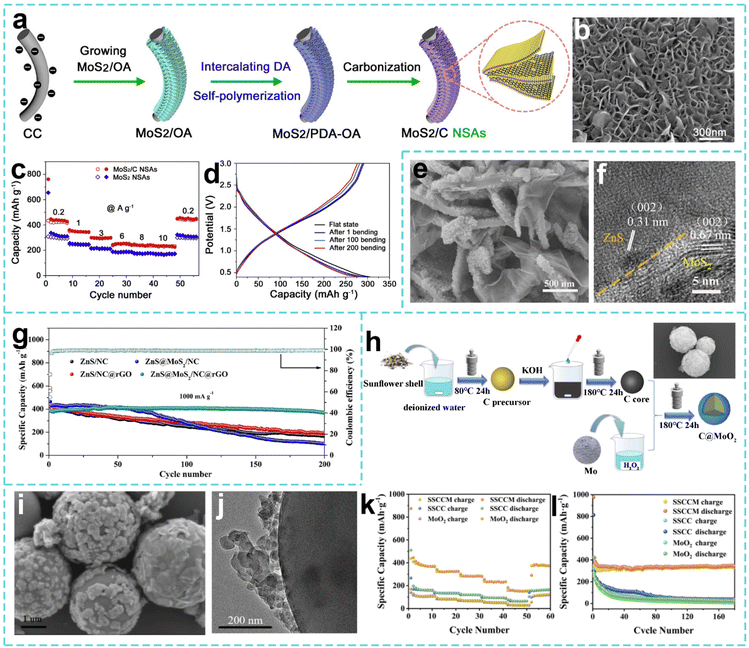 | ||
| Fig. 17 (a) Schematic illustration of the fabrication process and (b) SEM image of MoS2/C NSAs. (c) Rate performances of the MoS2/C NSAs and MoS2 NSAs electrodes. (d) The charge/discharge curves of the assembled MoS2/C NSAs flexible battery. Reproduced with permission from ref. 226. Copyright 2017, Elsevier. (e) SEM image and (f) HRTEM image of ZnS@MoS2/NC@rGO. (g) Cycling performance of different samples. Reproduced with permission from ref. 227. Copyright 2023, Elsevier. (h) Schematic description of the preparation of SSCCM. (i) SEM image and (j) HRTEM image of the SSCCM composite. (k) Rate behaviors and (l) cycling performances of SSCC, MoO2 and the SSCCM composite at 0.2C. Reproduced with permission from ref. 228. Copyright 2022, Elsevier. | ||
With the in-depth development of the concept of interface engineering, researchers began to explore the synergistic optimization of multi-composite systems and reaction mechanisms. Luo et al. fabricated a new type of hierarchical ZnS/MoS2 bimetallic sulfide through a sulfurization reaction, in which ZnS/MoS2 nanoparticles were encapsulated in a nitrogen-doped carbon framework derived from a Zn/Mo bimetallic imidazole framework (Zn/Mo BIFs) and covered with reduced graphene oxide (ZnS@MoS2/NC@rGO) (Fig. 17e and f). The nitrogen-doped carbon and rGO nanosheets obtained could improve the conductivity and accommodate the volume changes of ZnS and MoS2. At the same time, the ZnS–MoS2 heterostructure accelerates the diffusion kinetics in the interface and increases the charge transfer rate. The material consists of ZnS–MoS2 nanocomposites, with a mass loading of approximately 1.0 mg cm−2. The electrolyte used in this study is a mixture of NaClO4 in a carbonate solution of PC and EC, with 5 wt% FEC added (Fig. 17g).227
Zhao et al. developed a different approach, using biomass-derived carbon fibers and MoO2 composites, and designed a feasible core–shell structure C@MoO2 sunflower seed shell cellulose carbon sphere (C@MoO2 SSCCM) by coating MoO2 nanoparticles on the surface of biomass carbon spheres (Fig. 17h–j). This structure effectively inhibits volume changes during Na+ charging and discharging, making up for the low theoretical specific capacity of biomass carbon. Compared with pure MoO2 materials, the composite material has a better rate performance, with a practical capacity of 345.6 mAh g−1 after 180 cycles at 0.2C (Fig. 17k and l).228 To solve the common problem of high rate and long cycle life, Liu et al. prepared encapsulated MoSe2@hollow carbon nanosphere (MoSe2@HCNS) materials, that is, encapsulated multilayer MoSe2 nanosheets with an interlayer spacing extended to 1.02 nm in hollow carbon spheres using a spatial confinement strategy to construct a core–shell confinement and interlayer expansion synergistic system.229 The MoSe2@HCNS material has a practical capacity of 382 mAh g−1 at 10 A g−1 ultrafast charge and discharge, and a coulombic efficiency of over 99.5%. Innovations in preparation processes are also driving breakthroughs in material systems. The MoSe2 nanosheets used in this study had a loading of 1.1 mg cm−2, which was below the typical range of 2–3 mg cm−2. Zhang et al. synthesized self-supporting MoO2 nanosheets embedded in carbon nanofibers through electrospinning and sintering. The high specific capacity and cycling stability of MoO2 nanosheets are due to changes in the release volume of carbon nanofibers, which provide a 3D conductive diffusion network. The self-supporting structure can prevent particle shedding caused by polarization and improve the electrode stability. In this study, the MoO2 and carbon fiber material had a loading of 1.5 mg cm−2, which was below the 2–3 mg cm−2 range. The electrolyte used was a mixture of carbonates (PC and EC) with NaClO4, and 5% FEC was added.230
In the study of composite systems of tungsten-based sulfide and carbon materials, researchers have continuously broken through the limits of material performance through multi-dimensional structural design and interface engineering strategies. Liu et al. prepared a double-layer WS2/hollow carbon composite material by an in situ hydrothermal method, which involved growing WS2 on the surface of nanoscale hollow carbon spheres. The high specific surface area, excellent conductivity and unique structure of hollow carbon spheres provide a growth substrate for WS2, enhancing the conductivity and provide abundant active sites.231 Based on this structural advantage, Li et al. further innovated the interface design strategy and constructed a flower-like WS2/multi-walled carbon nanotube (WS2/MWCNT) hybrid structure. Through an in situ growth process, the petal-shaped WS2 nanosheets are tightly wrapped around the surface of 1D multi-walled carbon nanotubes to form a core–shell structure with strong interface bonding. This heterogeneous design makes the specific capacity of the composite material more than three times that of pure WS2 due to the synergistic effect of the 3D electron high-speed channels constructed by MWCNTs and the fast ion diffusion path of WS2 nanosheets. The material has a loading of 1.0 mg cm−2, which is below the 2–3 mg cm−2 threshold. The electrolyte used is based on NaClO4, with 5% FEC added.232 In response to the interface optimization problem of multi-component composite materials, Wang et al. proposed a partial selenization strategy and designed a composite material with a core–shell structure (MoWSe2/WO3/C) by a solvent-assisted method (Fig. 18a–c). This material forms a WSe2 heterojunction through controllable selenization of the WO3 surface, expanding the interlayer spacing between MoSe2 and WSe2 to 0.677 nm, and the carbon coating layer effectively improves the overall conductivity. As shown in Fig. 18d, the material still maintains a practical capacity of 384.3 mAh g−1 after 950 cycles at an ultra-high rate of 10 A g−1. Its performance advantage stems from the low diffusion barrier brought by the expanded layer structure and the fast charge transfer characteristics of the heterogeneous interface (Fig. 18e).233 In the in-depth exploration of the energy storage mechanism of 2D materials, Wei et al. constructed a 3D multi-level porous foam WS2/graphene (WS2/RGO) composite material by in situ anchoring of WS2 on the reduced graphene oxide skeleton (Fig. 18f and g).234 Its unique 3D interpenetrating porous structure effectively inhibits the stacking of WS2 layers and enhances the exposure of active sites. Combined with the high conductivity and flexible support of RGO, it exhibits a high reversible capacity of 419 mAh g−1 and excellent rate performance as an anode for SIBs at a current density of 100 mA g−1. The mass loading of WS2 in the composite material was less than 2–3 mg cm−2. The electrolyte used was a 1.0 M NaPF6 solution in EC/DMC, with the addition of 5% FEC (Fig. 18h). This material provides a reference for the multi-level pore and reversible transformation synergistic strategy for application in 2D metal sulfide-based energy storage materials and SIB anode designs.
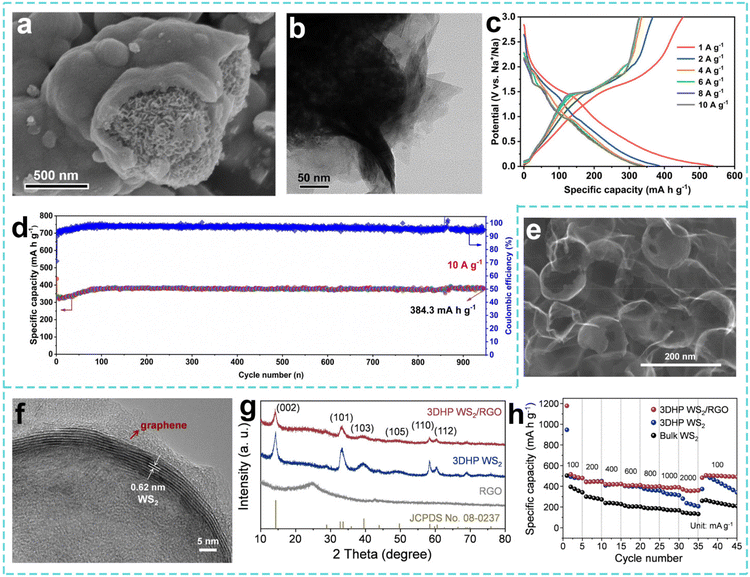 | ||
| Fig. 18 (a) SEM image and (b) TEM image of MoWSe2/WO3/C. (c) Charge–discharge voltage profiles, (d) long cycling performance and coulombic efficiency of MoWSe2/WO3/C at 10 A g−1. Reproduced with permission from ref. 233. Copyright 2023, Elsevier. (e) SEM image and (f) HRTEM image of 3DHP WS2/RGO. (g) XRD patterns of 3DHP WS2/RGO, 3DHP WS2 and RGO. (h) Rate capability of the 3DHP WS2/RGO, 3DHP WS2 and bulk WS2 electrodes at various current densities. Reproduced with permission from ref. 234. Copyright 2024, Elsevier. | ||
Through multi-composite strategies and innovative preparation processes, Mo- and W-based carbon composites have made significant progress in the application of anodes in SIBs. Techniques such as the hydrothermal method and in situ growth method effectively utilize the excellent conductivity and buffering properties of carbon materials, and cooperate with the sodium storage advantages of Mo- and W-based materials, to achieve a comprehensive improvement in cycling stability and rate performance. From expanding the interlayer spacing to promote ion embedding, to constructing a 3D porous structure to optimize the transmission path and from anchoring nanomaterials to enhance structural stability, to using heterogeneous interfaces to accelerate charge transfer, different studies explore the paths to improve the material performance from multiple dimensions.
5.4 Polymetallic molybdenum/tungstate
In the exploration of anode materials for SIBs, transition metal molybdates and tungstates stand out due to their unique structural advantages and electrochemical properties, while also facing some limitations. Polymetallic molybdate/tungstate exhibit significant advantages in Na+ storage, particularly due to their inherent structural buffering characteristics. The strong covalent bond network of the metal–oxygen polyhedra (MoO42−/WO42−) provides a stable framework structure, which helps mitigate volume expansion during cycling, thereby improving cycling stability. Furthermore, the synergistic effect of multiple cations in these materials can improve the electrochemical kinetics and overall Na+ storage performance. These materials typically possess layered or tunnel-like crystal structures, providing fast migration channels for Na+, resulting in an excellent rate performance. Their multi-electron reaction characteristics allow for theoretical capacities of 300–600 mAh g−1, significantly higher than those of traditional carbon-based anode materials. In addition, the strong covalent bond network of the metal–oxygen polyhedra (MoO42−/WO42−) effectively suppresses structural collapse during cycling, ensuring long-term stability.235 However, the use of polymetallic molybdate/tungstate also presents some limitations. Synthesis complexity is a major challenge, as the multi-metallic nature of these compounds requires more complex synthesis routes, leading to increased production costs and greater synthesis difficulty. Furthermore, the mass fraction of inactive components in these materials may be relatively high, which can reduce the overall efficiency of sodium-ion storage. For example, some phases in the multi-metallic system may not directly participate in Na+ storage, or their electrochemical activity may be lower than that of simpler MoO2 or MoS2 systems. Despite these challenges, the structural stability and multi-cation synergistic effects offered by polymetallic molybdate/tungstate make them promising candidates for future anode materials.236To solve the problem of volume expansion of antimony-based anodes caused by alloying reactions, Lu et al. constructed hierarchical Sb2MoO6 microspheres by a surfactant-free microwave-assisted hydrothermal method. The material is self-assembled from 1D nanobelts to form a 3D porous structure (Fig. 19a and b). The material has a good cycling performance at different current densities (Fig. 19c). The mass loading of the Sb2MoO6 material in this study was 1.0 mg cm−2, and the electrolyte used was a mixed solution of NaClO4 in PC and EC, with the addition of 5% FEC. The unique sodium storage mechanism originates from the in situ generated Na–Mo–O conductive buffer matrix (NaMo3O2 constructs an electronic network and NayMoVIOX inhibits volume expansion). This unique design offers a new direction for high-energy-density self-buffered anodes.237 In the field of cathode material development, to solve the bottleneck of kinetic performance optimization, Huu et al. innovatively used a low-temperature steam-assisted solid-phase method to synthesize orthogonal Fe2(MoO4)3 nanosheets. As a cathode material for SIBs, the Fe2(MoO4)3 nanosheets were subjected to electrochemical testing under a mass loading of approximately 1.1 mg cm−2, using a 1 M NaClO4 solution in PC as the electrolyte, with the addition of 5 wt% FEC. Through the synergistic effect of the 3D stacking morphology and surface pseudocapacitance effect, the capacity decay rate of this material after 1000 cycles at 1C is only 0.012%/time, and the capacity retention rate at 5C at −20–60 °C is 78%. This combination of a low-temperature green synthesis strategy and amorphous/crystalline phase interface regulation provides a new paradigm for the design of sodium superionic conductor cathodes suitable for all climates.238 In response to the structural stability challenge of molybdate anodes, Ali et al. developed a different approach and synthesized a nanocomposite material (Na2MoO4/C) by ball milling. This Na2MoO4/C nanocomposite was evaluated as an anode material for SIBs under a loading of approximately 3.5–3.8 mg cm−2, using a 1 M NaClO4 electrolyte dissolved in a mixed solvent of DEC, EC, and PC (volume ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). This process nanoscales the micron-sized particles to 50 nm and uniformly composites them with the carbon matrix to form a “nano-confinement–carbon coating” synergistic system (Fig. 19d and e). As shown in Fig. 19f, the capacity retention rate of the material after 1000 cycles is as high as 96%, and the volume strain is only 0.68%. In situ synchrotron radiation characterization confirmed the reversible coordination change mechanism of the Mo–O tetrahedron and the role of the rigid crystal framework in suppressing lattice distortion.239 To further improve the extreme performance of sodium superion conductor (NASICON) cathode materials, Sheng et al. developed a composite material of graphene-wrapped Fe2(MoO4)3 nanoparticles (Fig. 19g). The 3D conductive network constructed by the microemulsion method combined with the annealing process greatly improved the electronic conductivity. In this work, the performance of the graphene-wrapped Fe2(MoO4)3 nanoparticle composite material was evaluated with a cathode material mass loading of 1.5–3.0 mg cm−2 using 1 M NaClO4 dissolved in a mixed solvent of ethylene carbonate and dimethyl carbonate as the electrolyte. The material maintained a practical capacity of 64.1 mAh g−1 at a high rate of 100C, and the capacity retention rate reached 76% after 100 cycles at 10C (Fig. 19h). This “conductive network wrapping–nanostructure regulation” synergistic strategy successfully overcomes the dual defects of NASICON materials: long ion diffusion paths and poor electronic conductivity.240
1). This process nanoscales the micron-sized particles to 50 nm and uniformly composites them with the carbon matrix to form a “nano-confinement–carbon coating” synergistic system (Fig. 19d and e). As shown in Fig. 19f, the capacity retention rate of the material after 1000 cycles is as high as 96%, and the volume strain is only 0.68%. In situ synchrotron radiation characterization confirmed the reversible coordination change mechanism of the Mo–O tetrahedron and the role of the rigid crystal framework in suppressing lattice distortion.239 To further improve the extreme performance of sodium superion conductor (NASICON) cathode materials, Sheng et al. developed a composite material of graphene-wrapped Fe2(MoO4)3 nanoparticles (Fig. 19g). The 3D conductive network constructed by the microemulsion method combined with the annealing process greatly improved the electronic conductivity. In this work, the performance of the graphene-wrapped Fe2(MoO4)3 nanoparticle composite material was evaluated with a cathode material mass loading of 1.5–3.0 mg cm−2 using 1 M NaClO4 dissolved in a mixed solvent of ethylene carbonate and dimethyl carbonate as the electrolyte. The material maintained a practical capacity of 64.1 mAh g−1 at a high rate of 100C, and the capacity retention rate reached 76% after 100 cycles at 10C (Fig. 19h). This “conductive network wrapping–nanostructure regulation” synergistic strategy successfully overcomes the dual defects of NASICON materials: long ion diffusion paths and poor electronic conductivity.240
 | ||
| Fig. 19 (a) SEM image and (b) TEM image of Sb2MoO6 microspheres. (c) Rate performance of the Sb2MoO6 electrodes at 200 to 5000 mA g−1. Reproduced with permission from ref. 237. Copyright 2019, Elsevier. (d) SEM image and (e) TEM image of carbon composite with Na2MoO4. (f) Cycling test of the composite electrode at 0.05C. Reproduced with permission from ref. 239. Copyright 2021, American Chemical Society. (g) Schematic illustration of the formation of the Fe2(MoO4)3/graphene composite. (h) Cycling performance and coulombic efficiency of FMO-MG at a rate of 10C. Reproduced with permission from ref. 240. Copyright 2016, Elsevier. | ||
Turning to the multi-metal tungstate system in the field of layered oxide cathode interface engineering, Han et al. developed a one-step calcination grain boundary modification strategy to in situ construct a Na2WO4 network on nanomaterials (NM), and obtain micron-level spherical particles (NWO5@NM) by adjusting the molar ratio of Na![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ni
Ni![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Mn, which increased the volume density of the material by 18% (Fig. 20a). In this work, researchers evaluated a grain-boundary-modified O3-type nickel-rich layered cathode material with added Na2WO4 under SIB conditions. The mass loading was approximately 2.8–3.2 mg cm−2, and the electrolyte consisted of 1 M NaClO4 dissolved in a mixed solvent of EC, PC, and DEC (volume ratio of 1
Mn, which increased the volume density of the material by 18% (Fig. 20a). In this work, researchers evaluated a grain-boundary-modified O3-type nickel-rich layered cathode material with added Na2WO4 under SIB conditions. The mass loading was approximately 2.8–3.2 mg cm−2, and the electrolyte consisted of 1 M NaClO4 dissolved in a mixed solvent of EC, PC, and DEC (volume ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), with the addition of 5 wt% FEC. The practical capacity of NWO5@NM reached 126 mAh g−1 after 100 cycles at 1C (Fig. 20b). Mechanistic studies show that the Na2WO4 phase at the grain boundary has three functions: serving as a fast channel for Na+, reducing the charge transfer barrier, and inhibiting electrolyte side reactions through physical barriers (Fig. 20c). This coating-free in situ grain boundary ion conductor implantation technology has achieved the leap from bulk doping to precise interface control of the layered cathode.241 In response to the common problem of easy stacking of graphene composites, Wang et al. proposed an innovative solution: constructing a 3D FeWO4/graphene mesoporous composite material through an in situ self-assembly hydrothermal method (Fig. 20d). As shown in Fig. 20e and f, the heterogeneous nucleation of FeWO4 nanospheres and graphene forms an interpenetrating network structure, successfully expanding the interlayer spacing, and achieving a stable capacity of 377 mAh g−1 after 50 cycles at a current density of 20 mA g−1 for SIBs (Fig. 20g). This simple and scalable synthesis strategy provides a new idea for the development of universal energy storage materials compatible with multiple ions.242
1), with the addition of 5 wt% FEC. The practical capacity of NWO5@NM reached 126 mAh g−1 after 100 cycles at 1C (Fig. 20b). Mechanistic studies show that the Na2WO4 phase at the grain boundary has three functions: serving as a fast channel for Na+, reducing the charge transfer barrier, and inhibiting electrolyte side reactions through physical barriers (Fig. 20c). This coating-free in situ grain boundary ion conductor implantation technology has achieved the leap from bulk doping to precise interface control of the layered cathode.241 In response to the common problem of easy stacking of graphene composites, Wang et al. proposed an innovative solution: constructing a 3D FeWO4/graphene mesoporous composite material through an in situ self-assembly hydrothermal method (Fig. 20d). As shown in Fig. 20e and f, the heterogeneous nucleation of FeWO4 nanospheres and graphene forms an interpenetrating network structure, successfully expanding the interlayer spacing, and achieving a stable capacity of 377 mAh g−1 after 50 cycles at a current density of 20 mA g−1 for SIBs (Fig. 20g). This simple and scalable synthesis strategy provides a new idea for the development of universal energy storage materials compatible with multiple ions.242
 | ||
| Fig. 20 (a) SEM image of NaWO5@NM. (b) Capacity retention of NM and NaWO5@NM. (c) EIS spectra of NM and NaWO5@NM. Reproduced with permission from ref. 241. Copyright 2024, Elsevier. (d) Schematic illustration of the formation process of mesoporous FeWO4/graphene composites. (e) SEM image and (f) TEM image of the FeWO4/graphene nanosphere. (g) FeWO4/graphene nanosphere for SIB under different current densities over 50 cycles. Reproduced with permission from ref. 242. Copyright 2014, American Chemical Society. | ||
These breakthroughs systematically solve the core problems of volume expansion, ion diffusion kinetics and interface stability of electrode materials through a precise synthesis methodology (microwave hydrothermal, low-temperature solid phase, ball milling composite, etc.) and multi-scale structural regulation (graded microspheres, core–shell nanosheets, grain boundary modification, etc.). The molybdate system has made significant progress in the design of self-buffered anodes and the development of high-rate positive electrodes, while the tungstate system has broadened the application boundaries through structural evolution analysis and interface engineering innovation, jointly promoting the movement of SIBs toward high energy density, long cycle life and all-climate adaptability.
Overall, significant progress has been made in research on Mo- and W-based materials as anodes for high-performance sodium-ion batteries. Mo-/W-based oxides (such as MoO2, MoO3, WO3) typically provide a high theoretical capacity through conversion reactions, but their poor intrinsic conductivity and severe volume expansion lead to a poor rate performance and cycling stability. Mo-/W-based chalcogenides (such as MoS2, WS2, MoSe2) possess open layered structures, which facilitate Na+ intercalation and conversion reactions, exhibiting a high specific capacity and good rate potential; however, they are prone to interlayer stacking deactivation during cycling. To overcome these bottlenecks, Mo-/W-based carbon composites have become a research hotspot. Introducing a carbon matrix effectively improves the conductivity of the material and buffers volume changes, thereby significantly improving the cycle life and rate performance. In addition, multi-metallic molybdenum/tungsten compounds (such as molybdates) further optimize the sodium storage kinetics and structural stability through the synergistic effect between different metal components. Table 3 presents a comparison of the electrochemical properties of some Mo- and W-based materials that have been reported in recent years. In general, nanostructuring, composite materials, and structural design are key approaches for improving the electrochemical performance of such materials.
| Mo-/W-based materials | Cathode material | Half/full cells | Current density (A g−1) | Load mass (mg cm−2) | Capacity retention%/cycles | Ref. |
|---|---|---|---|---|---|---|
| VC-MoO2 | Na3V2(PO4)3 | Full | 0.3 | 1.02 | 96.5/1000 | 189 |
| a/c-MoO2 | — | Half | 1.0 | — | 86.0/3000 | 190 |
| MoS2/MoO2 | — | Half | 5.0 | — | 100.0/400 | 191 |
| DMcT-MoO3 | Na3V2(PO4)2O2F | Full | 0.2 | — | 99.2/100 | 196 |
| MoO3–MoS2 | Na3V2(PO4)3 | Full | 5.0 | 5.47 | 84.0/2300 | 198 |
| HMF-MoS2 | — | Half | 1.0 | — | 93.7/125 | 209 |
| 1T MoS2 | Na3V2(PO4)3 | Full | 1.0 | — | 94.0/200 | 210 |
| MoSe2/MoO3 | — | Half | 10 | — | 55.4/200 | 217 |
| MoSe2 | — | Half | 0.5 | 2.00 | 86.7/100 | 219 |
| Sn-MoSe2 | Na3V2(PO4)2F3/C | Full | 1.0 | — | 40.1/1600 | 220 |
| ZnS@MoS2/NCGO | Na3V2(PO4)3/C | Full | 0.2 | 1.00 | 43.5/100 | 227 |
| MoSe2@HCNS | — | Half | 10 | 1.10 | 99.5/1000 | 229 |
| Sb2MoO6 | Na3V2(PO4)3/C | Full | 2.0 | 1.00 | 98.7/450 | 237 |
| Fe2(MoO4)3 | MPC | Full | 0.091 | 1.10 | 100.0/1000 | 238 |
| Na2MoO4/C | — | Half | 0.05 | 3.5–3.8 | 96.0/1000 | 239 |
| Fe2(MoO4)3 | — | Half | 9.1 | 1.5–3.0 | 76.0/100 | 240 |
| WO2/WS2–rGO | PAC | Full | 5.0 | — | 79.0/6000 | 192 |
| WO3 | — | Half | 0.25 | — | 100.0/300 | 199 |
| WO3 | — | Half | 0.5 | — | 74.0/100 | 200 |
| WS2–NaCl | Na3V2(PO4)3 | Full | 0.1 | 1.50 | 81.4/100 | 212 |
| WS2@MnS | Na3V2(PO4)3 | Full | 0.1 | — | 100.0/2000 | 213 |
| WSe2–rGO | — | Half | 0.5 | — | 80.0/100 | 221 |
| WO2/WSe2 | MnSe/MnSe2 | Full | 1.0 | — | 92.9/10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
222 |
| WS2/hollow carbon | Na3V2(PO4)3/C | Full | 0.1 | — | 57.5/200 | 231 |
| MoWSe2/WO2/C | AC | Full | 10.0 | — | 50.3/950 | 233 |
| NWO5@NM | NaNi0.18Mn0.12O2 | Full | 0.2 | 2.8–3.2 | 65.6/100 | 241 |
6. Summary and outlook
In recent years, Mo- and W-based materials have significantly improved the sodium storage performance in research on SIB anodes through structural optimization and multi-mechanism synergy. Single-atom catalysis and dynamic in situ characterization technology provide atomic-level theoretical support for performance breakthroughs, and green processes and resource recycling promote sustainable development. However, volume effects, insufficient conductivity, and interface stability are still key bottlenecks. In the future, we need to focus on multi-mechanism material design, sodium storage mechanism research, electrolyte matching and interface optimization, synthesis methods and process improvements, etc., to promote the development of high-energy, long-life, low-cost SIBs, and help large-scale energy storage and new energy applications.With their multi-dimensional advantages, Mo- and W-based materials have shown broad and promising application prospects in the field of SIBs. Their high theoretical specific capacity characteristics provide core support for SIBs to achieve high energy density, and are expected to meet the needs of electric vehicles, energy storage power stations, etc. for large-capacity batteries. The unique sodium storage mechanism has opened a new path for performance improvement. With the in-depth study of the material structure and composition, its reversible specific capacity and cycling stability will be further optimized. In terms of structural design, nano-sizing, porosity and composite strategies give the material flexible designability, which can not only improve the charging and discharging efficiency by increasing the specific surface area and optimizing the ion transmission path, but also effectively alleviate the volume changes, enhance the structural stability, and ensure the long-term stable operation of the battery. Good electrolyte compatibility enables Mo- and W-based materials to form a stable interface with ether-based electrolytes, reduces resistance, inhibits side reactions, and significantly improves the overall performance of the battery. In addition, thanks to abundant and cheap sodium resources and the relatively considerable reserves of Mo and W elements, this type of material has outstanding cost advantages in large-scale applications, which meet the urgent needs of large-scale energy storage, distributed energy and other fields for low-cost energy storage technology.
When comparing Mo- and W-based materials with more widely used anode materials such as hard carbon, alloy anodes (e.g., Sn, Sb, P), and titanium (Ti)-based oxides, several factors should be considered, including cost, sustainability, safety, and scalability. Mo- and W-based materials generally have higher costs due to the need for specific processing methods and relatively expensive Mo and W resources. However, their potential for cost reduction through large-scale production and resource recycling, coupled with the abundance of sodium, make them competitive in the long term. In terms of sustainability, Mo- and W-based anodes are more environmentally friendly than lithium-based systems, benefiting from the availability of sodium and the ability to incorporate green synthesis methods. Safety is another strength, as Mo- and W-based materials offer strong structural stability, while alloy anodes face issues related to volume expansion and mechanical stress during cycling. Although Ti-based oxides offer excellent stability, their lower theoretical capacity limits their energy density. While Mo- and W-based materials face challenges of synthesis complexity, they show great potential for scalability due to advancements in green manufacturing processes. In general, Mo- and W-based materials are a promising alternative for high-performance, long-life, and low-cost SIBs, especially as advancements in material design and manufacturing processes continue. In the future, the main research focuses of Mo- and W-based materials in SIB anodes are as follows:
(1) Material structure and performance optimization. Even though nanoparticles can shorten the diffusion process of Na+, they still face the problem of agglomeration during the preparation process, and it is difficult to obtain an ideal structure. Therefore, researchers can try to prepare metal chalcogenide quantum dots to store sodium. The ultra-small size of quantum dots can significantly improve the utilization rate of electrode materials and increase active sites. Moreover, there are few studies in this area. Specifically, for Mo- and W-based anode materials, relevant research directions can be explored in the future. For example, quantum dots based on Mo/W compounds (i.e. MoS2, WS2) could be prepared through advanced synthesis techniques such as hydrothermal or solvothermal methods. The ultra-small size and high specific surface area of these quantum dots can enhance the sodium storage capacity and promote more efficient Na+ intercalation and deintercalation. In addition, coupling Mo- and W-based materials with other materials (such as carbon or metal oxides) to construct heterostructures further improves the electronic conductivity and stabilizes the interface. These structures can alleviate aggregation problems and optimize charge transfer, which is crucial for high-performance SIBs. In the future, these advancements will not only improve the sodium storage performance but also address key challenges such as capacity decay and stability in Mo- and W-based anode materials.
(2) Electrolyte matching and interface optimization. For future research on electrolyte systems, specific focus should be placed on pairing Mo- and W-based anodes with low-viscosity ether/carbonate blends to enhance ion conductivity and reduce resistance. Another promising avenue is interface design for high-concentration electrolytes, which can significantly reduce side reactions and improve the overall stability of SIBs. Furthermore, there is a need for operando characterization techniques, such as XRD, X-ray absorption spectroscopy (XAS), and tomography, which are currently underutilized but essential for providing real-time insights into structural and electrochemical changes during cycling. These advancements could play a crucial role in addressing the challenges Mo- and W-based anodes face in SIBs. In addition, researchers can explore solid electrolytes, known for their excellent chemical stability, which could further enhance the interface effect in SIBs, reduce side reactions, and alleviate volume expansion. This may become a key breakthrough in the next generation of sodium storage technology.
(3) Improved synthesis methods and processes. Since the reaction conditions for traditional preparation methods are difficult to control, researchers could use spray pyrolysis in the future to synthesize nanoparticles with uniform size and good dispersion in one step, and regulate the components and construct porous-hollow structures, which could significantly shorten the Na+ diffusion path and improve the specific capacity and rate performance of SIBs. Specifically, researchers can explore the optimization of precursor concentration and spray rate. By adjusting the precursor concentration and spray rate, the particle size distribution can be controlled, improving the uniformity of the nanoparticles. This will help improve dispersion and minimize aggregation, thus enhancing the electrochemical performance of Mo- and W-based materials. Furthermore, porous hollow structure design can be implemented. Spray pyrolysis can prepare porous and hollow nanostructures, which is beneficial for increasing the specific surface area and reducing the internal diffusion resistance of Na+. These structures can also mitigate volume expansion during cycling, thereby improving the cycling stability of the material. At present, there are few studies using this method, the design of which is worth the attention of researchers.
Mo- and W-based materials show strong application potential in the field of SIBs. At present, researchers have been able to inject new impetus into the technological upgrading of SIBs through structural optimization and multi-mechanism synergistic innovations. However, the development of such materials is still in the exploratory stage, and there are many challenges to be explored and solved. With the in-depth application of cutting-edge technologies such as single-atom catalysis and in situ characterization, as well as the large-scale promotion of green manufacturing processes, Mo- and W-based materials are expected to make the leap from laboratory research to industrialized applications. This will not only promote the application of SIBs toward the goals of achieving high energy density, long cycle life and low cost, but also provide solid technical support for the global energy structure transition and sustainable development strategy.
Author contributions
All authors contributed to the discussion of contents and the editing of the manuscript prior to submission.Conflicts of interest
There are no conflicts to declare.Data availability
This study did not generate any new datasets. All data analyzed are from publicly available sources, as cited in the manuscript.Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (22269020 and 42167068) and the Key Project of the Natural Science Foundation of Gansu Province (25JRRA004).References
- S. Dutta and R. B. Choudhary, Mater. Today Chem., 2025, 46, 102711 CrossRef CAS.
- C. Hakim, N. Sabi and I. Saadoune, J. Energy Chem., 2021, 61, 47–60 CrossRef CAS.
- Z. Sun, H. Liu, W. Li, N. Zhang, S. Zhu, B. Chen, F. He, N. Zhao and C. He, Prog. Mater. Sci., 2025, 149, 101401 CrossRef CAS.
- S. Gandi, V. S. C. S. Vaddadi, S. S. S. Panda, N. K. Goona, S. R. Parne, M. Lakavat and A. Bhaumik, J. Power Sources, 2022, 521, 230930 CrossRef CAS.
- Y. Zhang, G. Liu, Q. Sun, D. Qiao, J. Chen, L. Wen and M. Zhao, J. Energy Storage, 2024, 102, 114279 CrossRef CAS.
- M. Li, Z. Du, M. A. Khaleel and I. Belharouak, Energy Storage Mater., 2020, 25, 520–536 CrossRef.
- F. Wei, Q. Zhang, P. Zhang, W. Tian, K. Dai, L. Zhang, J. Mao and G. Shao, J. Electrochem. Soc., 2021, 168, 050524 CrossRef CAS.
- G. M. Tomboc, Y. Wang, H. Wang, J. Li and K. Lee, Energy Storage Mater., 2021, 39, 21–44 CrossRef.
- Z. Zeng, Y. Mao, Z. Hu, K. Chen, Q. Huang, Y. Song, Z. Wu, P. Zhang, T. Chen and X. Guo, Ind. Eng. Chem. Res., 2023, 62, 15343–15359 Search PubMed.
- M. Chen, Z. Wang, Y. Wang, Y. Li and Q. Chen, Appl. Mater. Today, 2021, 23, 100985 Search PubMed.
- L. Wang, H. Tian, X. Yao, Y. Cai, Z. Gao and Z. Su, ChemElectroChem, 2023, 11, e202300414 CrossRef.
- S. Dong, N. Lv, Y. Wu, Y. Zhang, G. Zhu and X. Dong, Nano Today, 2022, 42, 101349 CrossRef CAS.
- B. Liu, Y. Cao, H. Zhang, S. Wang, Q. Geng, Y. Li and F. Dong, Chem. Eng. J., 2022, 446, 136918 Search PubMed.
- J. Liang, C. Wei, D. Huo and H. Li, J. Energy Storage, 2024, 85, 111044 CrossRef CAS.
- M. Zhou, Y. Shen, J. Liu, L. Lv, X. Gao, Y. Zhang, X. Meng, X. Yang, Y. Zheng and Z. Zhou, Vacuum, 2022, 200, 111054 CrossRef CAS.
- L. Wang, G. Yang, S. Peng, J. Wang, W. Yan and S. Ramakrishna, Energy Storage Mater., 2020, 25, 443–476 CrossRef.
- M. He, S. Liu, J. Wu and J. Zhu, Prog. Solid State Chem., 2024, 74, 100452 CrossRef CAS.
- G. Sun, Z. Wei, N. Chen, G. Chen, C. Wang and F. Du, Chem. Eng. J., 2020, 388, 124305 Search PubMed.
- D. Choi, S. Lim and D. Han, J. Energy Chem., 2021, 53, 396–406 Search PubMed.
- S. Kumar, M. Singh, R. Mondal, M. Kumar, R. Prakash and P. Singh, Energy Fuels, 2022, 37, 1288–1296 CrossRef.
- Z. Qian, X. Wang, T. Liu, L. Zhang and J. Yu, J. Energy Storage, 2022, 51, 104522 CrossRef.
- C. Li, Q. Fu, K. Zhao, Y. Wang, H. Tang, H. Li, H. Jiang and L. Chen, Carbon, 2018, 139, 1117–1125 CrossRef CAS.
- M. I. Jamesh and A. S. Prakash, J. Power Sources, 2018, 378, 268–300 CrossRef CAS.
- C. Wang, S. Xue, X. Lei, J. Wen, X. Pan, F. Zhang, C. Zou and Y. Tang, Chem. Eng. J., 2023, 470, 144043 CrossRef CAS.
- X. He, X. Liu, Z. Yang, H. Zhang, L. Li, G. Xu, Y. Qiao, S. Chou and M. Wu, Electrochem. Commun., 2021, 128, 107067 CrossRef CAS.
- Q. Ruan, Y. Qian, M. Xue, L. Chen and Q. Zhang, J. Energy Chem., 2024, 89, 487–518 CrossRef CAS.
- J. Zhou, J. Chen, Y. Peng, Y. Zheng, A. Ze and X. Lin, Coord. Chem. Rev., 2022, 472, 214781 CrossRef CAS.
- Y. Jiang, J. Dong, S. Tan, Q. Wei, F. Xiong, W. Yang, Y. Shen, Q. Zhang, Z. Liu, Q. An and L. Mai, J. Energy Chem., 2021, 55, 295–303 Search PubMed.
- X. Yang, L. Gong, Y. Jin, T. Zheng, X. Wang, Q. Zhi, B. Yu, K. Wang and J. Jiang, CCS Chem., 2025, 7, 3063–3075 Search PubMed.
- Z. Liu, S. Tao, J. Han, Z. Gao, W. Xu, H. Min, X. Shen, H. Yang and J. Wang, Mater. Today Energy, 2023, 33, 101256 Search PubMed.
- M. R. Panda, A. Raj K, A. Ghosh, A. Kumar, D. Muthuraj, S. Sau, W. Yu, Y. Zhang, A. K. Sinha, M. Weyland, Q. Bao and S. Mitra, Nano Energy, 2019, 64, 103951 CrossRef CAS.
- Y. Wang, D. Wang, C. Bai, Y. Zhu, L. Xu, H. Xiao, Q. Shi, X. Li, X. Chen, H. Shao and G. Fang, Inorg. Chem. Front., 2024, 63, 15224–15235 CrossRef CAS PubMed.
- T. Song, N. Wu, X. Zheng, X. Zhang, S. Guo, Q. Yan, W. Yao and Y. Tang, CCS Chem., 2025, 7, 554–563 Search PubMed.
- J. Guo, J. Yang, J. Guan, X. Chen, Y. Zhu, H. Fu, Q. Liu, B. Wei and H. Geng, Chem. Eng. J., 2022, 450, 138007 CrossRef CAS.
- H. He, D. Sun, Y. Tang, H. Wang and M. Shao, Energy Storage Mater., 2019, 23, 233–251 Search PubMed.
- S. McArdle, F. Bauer, S. F. Granieri, M. Ast, F. Di Fonzo, A. T. Marshall and H. Radinger, ChemElectroChem, 2023, 11, e202300512 Search PubMed.
- H. Zheng, W. Yan and J. Zhang, Electrochem. Energy Rev., 2025, 8, 2401457 Search PubMed.
- X. Man, X. Min, Y. Yan, H. Gong, Y. Dai, T. Li, P. Xiao, Y. Sun, L. Yin and R. Wang, Energy Storage Mater., 2025, 75, 104076 CrossRef.
- S. Lin, H. Zhang, C. Shu, W. Hua, X. Wang, Y. Zhao, J. Luo, Z. Tang, Y. Wu and W. Tang, Adv. Funct. Mater., 2024, 34, 2409628 CrossRef CAS.
- Y. Li, X. Lu, X. Zhao, H. Wang and X. Hu, Prog. Nat. Sci.: Mater. Int., 2023, 33, 767–779 CrossRef CAS.
- L. Liu, Y. Gong, Y. Tong, H. Tian, X. Wang, Y. Hu, S. Huang, W. Huang, S. Sharma, J. Cui, Y. Jin, W. Gong and W. Zhang, CCS Chem., 2024, 6, 1255–1263 Search PubMed.
- X. Liu, Z. Pan, Y. Xu, H. Lu, B. Li, K. Yan, F. Tang, Q. Zhou and L. Kong, J. Energy Storage, 2025, 120, 116429 CrossRef CAS.
- S. Khammuang, T. Kaewmaraya, T. Hussain and K. Kotmool, J. Power Sources, 2026, 661, 238592 CrossRef CAS.
- Y. Jayamkondan, T. R. Penki and P. K. Nayak, Mater. Today Energy, 2023, 36, 101360 Search PubMed.
- C. I. Azambou, O. O. Obiukwu, P. K. Tsobnang, I. T. Kenfack, E. E. Kalu and E. E. Oguzie, J. Energy Storage, 2024, 94, 112506 Search PubMed.
- I. Ullah, T. R. Aldhafeeri, A. Haider, X. Wu, Z. Ullah, S. Chang, A. Innayat, N. Begum, M. A. Pope, F. Sher, H. U. Rehman and I. Hussain, ACS Appl. Energy Mater., 2025, 8, 1743–1751 Search PubMed.
- M. Ma, Y. Yao, Y. Wu and Y. Yu, Adv. Fiber Mater., 2020, 2, 314–337 Search PubMed.
- K. Xiong, T. Qi and X. Zhang, Electroanalysis, 2024, 37, e202400318 CrossRef.
- J. Wang, J. Cui, Z. Li, D. Zhang, H. Sun, H. Wang, Q. Wang, H. J. Woo, S. Ramesh and B. Wang, Chem. Eng. J., 2023, 464, 142764 CrossRef CAS.
- H. Alptekin, H. Au, A. C. S. Jensen, E. Olsson, M. Goktas, T. F. Headen, P. Adelhelm, Q. Cai, A. J. Drew and M. M. Titirici, ACS Appl. Energy Mater., 2020, 3, 9918–9927 CrossRef CAS.
- M. Kaur, P. Sharma, R. Mir, K. Kaur, R. K. Sharma and A. Kumar, FlatChem, 2025, 51, 100866 CrossRef CAS.
- K. Wang, H. Zhuo, J. Wang, F. Poon, X. Sun and B. Xiao, Adv. Funct. Mater., 2023, 33, 2212607 CrossRef CAS.
- Q. Ni, Y. Bai, F. Wu and C. Wu, Adv. Sci., 2017, 4, 1600275 CrossRef PubMed.
- J. Cai, C. Liu, S. Tao, Z. Cao, Z. Song, X. Xiao, W. Deng, H. Hou and X. Ji, Coord. Chem. Rev., 2023, 479, 214985 CrossRef CAS.
- Y. Chen, J. You, X. Zhao, M. Li, X. Han, H. Liu, H. Sun, X. Wang, H. Li, P. Wang and Z. Liu, Sci. China Mater., 2024, 67, 3637–3647 CrossRef CAS.
- F. Gao, D. Zhang, H. Zhang, C. Gao, G. Huang, Z. Zhang, Y. Liu, Y. Wang, M. Terrones and Y. Wang, Carbon, 2024, 229, 119481 CrossRef CAS.
- Y. Lin, S. Luo, W. Zhao, Q. Sun, J. Cong, P. Li, P. Li and S. Yan, J. Energy Chem., 2024, 98, 441–471 CrossRef CAS.
- X. Tang, C. Liu, H. Wang, L.-P. Lv, W. Sun and Y. Wang, Coord. Chem. Rev., 2023, 494, 215361 CrossRef CAS.
- T. Or, S. W. D. Gourley, K. Kaliyappan, Y. Zheng, M. Li and Z. Chen, Electrochem. Energy Rev., 2022, 5, 1600943 Search PubMed.
- J. Shi, L. Ding, Y. Wan, L. Mi, L. Chen, D. Yang, Y. Hu and W. Chen, J. Energy Chem., 2021, 57, 650–655 Search PubMed.
- A. Raj K, M. R. Panda, D. P. Dutta and S. Mitra, Carbon, 2019, 143, 402–412 CrossRef CAS.
- D. Alvira, D. Antorán and J. J. Manyà, Chem. Eng. J., 2022, 447, 137468 Search PubMed.
- J. Wu, X. Li, H. Chen, Z. Yuan, J. Huang, L. Tong, J. Long, M. Li, X. Chen and Y. Chen, CCS Chem., 2025, 7, 2742–2753 CrossRef.
- J. H. Kim, M. J. Jung, M. J. Kim and Y. S. Lee, J. Ind. Eng. Chem., 2018, 61, 368–380 CrossRef CAS.
- K. Mathiyalagan, D. Shin and Y.-C. Lee, J. Energy Chem., 2024, 90, 40–57 CrossRef CAS.
- Y. Zhao, Q. Liu, X. Zhao, D. Mu, G. Tan, L. Li, R. Chen and F. Wu, Mater. Today, 2023, 62, 271–295 CrossRef CAS.
- P. Ma, D. Fang, Y. Liu, Y. Shang, Y. Shi and H. Yang, Adv. Sci., 2021, 8, e2003185 CrossRef PubMed.
- T. Huang, B. Tian, J. Guo, H. Shu, Y. Wang and J. Dai, Mater. Sci. Semicond. Process., 2019, 89, 250–255 Search PubMed.
- M. Liu, W. Li, F. Liu and W. Zhang, Nano Lett., 2024, 24, 7180–7187 Search PubMed.
- H. Liu and B. Dong, Mater. Today Phys., 2021, 20, 100469 CrossRef CAS.
- Y. Liu, C. Sun, Y. Li, H. Jin and Y. Zhao, Energy Storage Mater., 2023, 57, 69–80 CrossRef.
- C. G. Real, E. H. N. S. Thaines, L. A. Pocrifka, R. G. Freitas, G. Singh and H. Zanin, J. Energy Storage, 2022, 52, 104793 CrossRef.
- Y. Lei, Z. Yan, W. Lai, S. Chou, Y. Wang, H. Liu and S. Dou, Electrochem. Energy Rev., 2020, 3, 766–792 CrossRef CAS.
- H. Yin, D. Han, X. Yu, M. Cao, Z. Hou, C. Li and M. Zhu, ACS Appl. Energy Mater., 2023, 6, 1155–1175 CrossRef CAS.
- M. Xiao, X. Dai and Y. Jiang, J. Energy Storage, 2025, 111, 115411 CrossRef CAS.
- L. Rakhymbay, N. Bazybek, K. Kudaibergenov, S. T. Myung, Z. Bakenov and A. Konarov, J. Power Sources, 2024, 602, 234347 CrossRef CAS.
- S. Nie, L. Liu, J. Liu, J. Xia, Y. Zhang, J. Xie, M. Li and X. Wang, J. Alloys Compd., 2019, 772, 314–323 CrossRef CAS.
- D. Zeng, J. Xie, K. Qi, F. Yang, S. Peng, L. Liu, L. Wu, Y. Chen, X. Xiong and Y. Qiu, J. Energy Storage, 2024, 80, 110339 CrossRef CAS.
- X. Lu, S. Li, Y. Li, F. Wu, C. Wu and Y. Bai, Adv. Mater., 2024, 36, e2407359 CrossRef PubMed.
- V. Palomares, N. Nieto and T. Rojo, Curr. Opin. Electrochem., 2022, 31, 100840 CrossRef CAS.
- Q. Cheng, X. Zhao, G. Yang, L. Mao, F. Liao, L. Chen, P. He, D. Pan and S. Chen, Energy Storage Mater., 2021, 41, 842–882 CrossRef.
- J. Do, I. Kim, H. Kim and Y. Jung, Energy Storage Mater., 2020, 25, 62–69 CrossRef.
- B. Ma, Y. Liang, X. Ma, W. Wang, C. Miao, G. Yu, Q. Wang, C. Xu and X. Cui, J. Colloid Interface Sci., 2025, 696, 137824 CrossRef CAS PubMed.
- X. Chen and Y. Zhang, Int. J. Energy Res., 2021, 45, 9753–9779 CrossRef CAS.
- J. Liang, L. Zhang, D. XiLi and J. Kang, Rare Met., 2020, 39, 1005–1018 CrossRef CAS.
- T. Qi, K. Xiong and X. Zhang, J. Power Sources, 2025, 626, 235721 CrossRef CAS.
- V. S. More and M. D. Yadav, Energy Fuels, 2024, 38, 20285–20313 CrossRef CAS.
- K. Mathiyalagan, R. Raja, D. Shin and Y. C. Lee, Prog. Mater. Sci., 2025, 151, 101425 CrossRef CAS.
- P. Zhang, Z. Wang, Z. He, D. Zhang, J. Wang and Y. Zhao, Composites, Part B, 2025, 303, 112564 Search PubMed.
- A. Song, Y. Li, D. Yuan, J. Wu, H. Gu, G. Zhang, A. Wu and J. Xu, Green Energy Environ., 2024, 38, 17146–17160 CAS.
- M. S. Balogun, Y. Luo, W. Qiu, P. Liu and Y. Tong, Carbon, 2016, 98, 162–178 CrossRef CAS.
- Y. Tian, Y. Chen, Y. Liu, H. Li and Z. Dai, Chem. Rec., 2022, 22, e202200123 CrossRef CAS PubMed.
- G. Zhang, T. Xiong, L. He, M. Yan, K. Zhao, X. Xu and L. Mai, J. Mater. Sci., 2017, 52, 3697–3718 CrossRef CAS.
- Z. Zhang, Y. Chen, S. Sun, K. Sun, H. Sun, H. Li, Y. Yang, M. Zhang, W. Li, S. Chou, H. Liu and Y. Jiang, J. Mater. Sci. Technol., 2022, 119, 167–181 CrossRef CAS.
- H. Liao, H. Liu, Q. Gou, R. Zeng, D. Zhao, X. Yuan, F. Chen, G. Xie and Y. Hou, J. Power Sources, 2025, 625, 235739 CrossRef CAS.
- X. Yang, D. Zhang, L. Zhao, C. Peng, K. Ren, C. Xu, P. Liu, Y. Zhou, Y. Lei, B. Yang, D. Xue and F. Liang, Adv. Energy Mater., 2024, 14, 2304365 CrossRef CAS.
- X. Lv, J. Song, Y. Lai, J. Fang, J. Li and Z. Zhang, J. Energy Storage, 2016, 8, 205–211 CrossRef.
- D. Zhou, C. Zeng, J. Xiang, T. Wang, Z. Gao, C. An and W. Huang, Ionics, 2022, 28, 2029–2040 CrossRef CAS.
- T. Sharmin, N. Hossain, F. T. Mohsin, M. A. Haque, M. M. Mashfy, T. A. Alvy and M. Nasim, Mater. Today Chem., 2024, 42, 102407 CrossRef CAS.
- S. Qi, B. Xu, V. T. Tiong, J. Hu and J. Ma, Chem. Eng. J., 2020, 379, 122261 CrossRef CAS.
- S. Zheng, Y. Tian, Y. Liu, S. Wang, C. Hu, B. Wang and K. Wang, Rare Met., 2020, 40, 272–289 CrossRef.
- W. Zhao, M. Wang, H. Lin, K. Kim, R. He, S. Feng and H. Liu, Prog. Nat. Sci.: Mater. Int., 2024, 34, 263–273 CrossRef CAS.
- M. A. U. Din, C. Li, L. Zhang, C. Han and B. Li, Mater. Today Phys., 2021, 21, 100486 CrossRef.
- Y. Gong, Y. Li, Y. Li, M. Liu, Y. Bai and C. Wu, Small, 2023, 19, e2206194 CrossRef PubMed.
- Y. Jiang, Z. Zhang, H. Liao, Y. Zheng, X. Fu, J. Lu, S. Cheng and Y. Gao, ACS Nano, 2024, 18, 7796–7824 CrossRef CAS PubMed.
- M. Lee, J. M. Kim, N. Lee, J. K. Park and S.-M. Paek, Chem. Eng. J., 2025, 519, 164931 CrossRef CAS.
- T. S. Tedla, N. W. Hlongwa, T. T. I. Nkambule and M. A. Kebede, Energy Rep., 2025, 14, 3175–3203 CrossRef.
- M. Xiao, J. Power Sources, 2026, 661, 238634 Search PubMed.
- L. Xu, Y. Li, Y. Xiang, C. Li, H. Zhu, C. Li, G. Zou, H. Hou and X. Ji, ACS Nano, 2025, 19, 14627–14651 CrossRef CAS PubMed.
- Y. Zhou, L. Zhou, Z. You, C. Zhao, J. Yang, S. Huang, M. Wang, Q. Zhong and J. Xiao, J. Energy Storage, 2025, 138, 118768 CrossRef CAS.
- J. Bai, L. Zhang, S. Li, H. Ren, Y. Liu and S. Guo, Chem. Eng. J., 2023, 452, 139111 CrossRef CAS.
- A. Cheng, H. Zhang, W. Zhong, Z. Li, Y. Tang and Z. Li, J. Electroanal. Chem., 2019, 843, 31–36 Search PubMed.
- J. Jiang, W. Yang, H. Wang, Y. Zhao, J. Guo, J. Zhao, M. Beidaghi and L. Gao, Electrochim. Acta, 2017, 240, 379–387 CrossRef CAS.
- H. Wu, N. Xu, Z. Jiang, A. Zheng, Q. Shi, R. Lv, L. Ni, G. Diao and M. Chen, Chem. Eng. J., 2022, 427, 131002 CrossRef CAS.
- B. Ye, L. Xu, W. Wu, Y. Ye, Z. Yang, Y. Qiu, Z. Gong, Y. Zhou, Q. Huang, Z. Shen, Z. Hong, Z. Meng, Z. Zeng, Z. Cheng, S. Ye, H. Hong, Q. Lan, F. Li, T. Guo and S. Xu, ACS Sustainable Chem. Eng., 2022, 10, 3166–3179 CrossRef CAS.
- G. Tian, Y. Song, X. Luo, Z. Zhao, F. Han, J. Chen, H. Huang, N. Tang and S. Dsoke, J. Alloys Compd., 2021, 877, 160299 CrossRef CAS.
- Q. Li, W. Zhang, J. Peng, W. Zhang, Z. Liang, J. Wu, J. Feng, H. Li and S. Huang, ACS Nano, 2021, 15, 15104–15113 CrossRef CAS PubMed.
- R. Wang, S. Wang, X. Peng, Y. Zhang, D. Jin, P. K. Chu and L. Zhang, ACS Appl. Mater. Interfaces, 2017, 9, 32745–32755 CrossRef CAS PubMed.
- Z.-Y. Song, Y.-D. Cao, L.-L. Fan, J. Song, Y. Feng, H. Liu, C.-L. Lv and G.-G. Gao, Rare Met., 2024, 44, 195–208 CrossRef.
- X. Xu, Y. Qiu, Z. Len, Z. Chen, W. Zhu, W. Zhao, Y. Dai, L. Cao and H. Geng, J. Colloid Interface Sci., 2024, 656, 252–261 CrossRef CAS PubMed.
- F. Zheng, W. Zhong, Q. Deng, Q. Pan, X. Ou, Y. Liu, X. Xiong, C. Yang, Y. Chen and M. Liu, Chem. Eng. J., 2019, 357, 226–236 CrossRef CAS.
- M. Liu, J. Zhang, S. Guo, B. Wang, Y. Shen, X. Ai, H. Yang and J. Qian, ACS Appl. Mater. Interfaces, 2020, 12, 17620–17627 CrossRef CAS PubMed.
- C. Wu, S. X. Dou and Y. Yu, Small, 2018, 14, e1703671 CrossRef PubMed.
- J. Guo, Z. Gu, M. Du, X. Zhao, X. Wang and X. Wu, Mater. Today, 2023, 66, 221–244 CrossRef CAS.
- G. Dong, Z. Yang, B. Gao, Z. Zhu, T. Zhou, L. Tao and W. Dong, J. Colloid Interface Sci., 2025, 695, 137722 CrossRef CAS PubMed.
- L. Wang, J. Światowska, S. Dai, M. Cao, Z. Zhong, Y. Shen and M. Wang, Mater. Today Energy, 2019, 11, 46–60 CrossRef CAS.
- K. S. Hong, D. H. Nam, S. J. Lim, D. Sohn, T. H. Kim and H. Kwon, ACS Appl. Mater. Interfaces, 2015, 7, 17264–17271 CrossRef CAS PubMed.
- M. Duan, Y. Meng, M. Xiao, W. H. Z. Ahmed, X. Wang, H. Gao, Y. Zhang and F. Zhu, J. Electroanal. Chem., 2021, 899, 115681 CrossRef CAS.
- X. Hu, M. Wang, R. Hu, L. Yang, Q. Li, Z. Yang, J. Yang, Q. Duan, J. Chen, B. Yu, B. Guo, Z. Ma, Y. Huang and X. Li, J. Energy Storage, 2024, 99, 113245 CrossRef CAS.
- P. Wang, S. Sun, Y. Jiang, Q. Cai, Y. H. Zhang, L. Zhou, S. Fang, J. Liu and Y. Yu, ACS Nano, 2020, 14, 15577–15586 CrossRef CAS PubMed.
- A. Roy, S. Dey and G. Singh, ACS Omega, 2024, 9, 24933–24947 CrossRef CAS PubMed.
- C. Zhao, C. Yu, M. Zhang, H. Huang, S. Li, X. Han, Z. Liu, J. Yang, W. Xiao, J. Liang, X. Sun and J. Qiu, Adv. Energy Mater., 2017, 7, 201602880 Search PubMed.
- W. Han, G. Liu, W. Seo, H. Lee, H. Chu and W. Yang, Carbon, 2021, 184, 534–543 CrossRef CAS.
- H. Liang, H. Zhang, L. Zhao, Z. Chen, C. Huang, C. Zhang, Z. Liang, Y. Wang, X. Wang, Q. Li, X. Guo and H. Li, Chem. Eng. J., 2022, 427, 131481 CrossRef CAS.
- Y. X. Wang, S. L. Chou, D. Wexler, H. K. Liu and S. X. Dou, Chemistry, 2014, 20, 9607–9612 CrossRef CAS PubMed.
- J. Li, Y. Sun, X. Zhang, Y. Sun and J. Guo, J. Energy Storage, 2023, 73, 108932 CrossRef.
- C. Wang, Z. Wang, D. Zhao, J. Ren, S. Liu, H. Tang, P. Xu, F. Gao, X. Yue, H. Yang, C. Niu, W. Chu, D. Wang, X. Liu, Z. Wang, Y. Wu and Y. Zhang, ACS Appl. Mater. Interfaces, 2021, 13, 55020–55028 Search PubMed.
- T. Yi, H. M. K. Sari, X. Li, F. Wang, Y. Zhu, J. Hu, J. Zhang and X. Li, Nano Energy, 2021, 85, 105955 CrossRef CAS.
- T. Yao, H. Wang, Y. Qin, J.-W. Shi and Y. Cheng, Composites, Part B, 2023, 253, 110557 CrossRef CAS.
- X. Hu, Y. Liu, J. Li, G. Wang, J. Chen, G. Zhong, H. Zhan and Z. Wen, Adv. Funct. Mater., 2019, 30, 201907677 Search PubMed.
- Y. F. Yuan, W. C. Zhao, D. Zhang, M. Zhu, S. M. Yin and S. Y. Guo, J. Alloys Compd., 2022, 895, 162681 CrossRef CAS.
- H. Lim, S. Yu, W. Choi and S. O. Kim, ACS Nano, 2021, 15, 7409–7420 CrossRef CAS PubMed.
- F. Zeng, L. Yang, Y. Pan, M. Xu, H. Liu, M. Yu, M. Guo and C. Yuan, Electrochim. Acta, 2019, 325, 134903 CrossRef CAS.
- B. Kang, Y. Wang, X. He, Y. Wu, X. Li, C. Lin, Q. Chen, L. Zeng, M. Wei and Q. Qian, Dalton Trans., 2021, 50, 14745–14752 Search PubMed.
- H. Zhang, J. Cheng, L. Qiu, W. Sun, Y. Bai, H. Y. Yang and C. M. Li, Adv. Funct. Mater., 2025, 10, 202425947 Search PubMed.
- N. Al-Ansi, A. Salah, Q. A. Drmosh, G. D. Yang, A. Hezam, A. Al-Salihy, J. Lin, X. L. Wu, L. Zhao, J. P. Zhang, S. L. Wang and H. Z. Sun, Small, 2023, 19, e2304459 CrossRef PubMed.
- Y. L. Bai, Y. S. Liu, C. Ma, K. X. Wang and J. S. Chen, ACS Nano, 2018, 12, 11503–11510 CrossRef CAS PubMed.
- J. Hu, J. Yuan, L. Zhao, G. Li, D. Chen, W. Han, Y. Chu, X. Cui, C. Li and Y. Zhang, J. Alloys Compd., 2022, 895, 162514 CrossRef CAS.
- W. Yang, J. Wang, C. Si, Z. Peng and Z. Zhang, Nano Res., 2017, 10, 2584–2598 CrossRef CAS.
- Y. Wang, D. Kong, S. Huang, Y. Shi, M. Ding, Y. Von Lim, T. Xu, F. Chen, X. Li and H. Y. Yang, J. Mater. Chem. A, 2018, 6, 10813–10824 RSC.
- F. Li, K. Ren, M. Hou, M. Lin, X. Yang, Y. Zhou, S. Xiong and F. Liang, ACS Energy Lett., 2024, 10, 195–204 CrossRef.
- M. Chen, S. Gu, J. Li, Y. Dai, Y. He, B. Sun, T. Gao, L. Xu and G. Zhou, Adv. Sci., 2025, e09997 CrossRef CAS PubMed.
- Y. Hao, Y. Cui, J. Wang, J. Hu, Q. Tang and S. Yang, Chem. Eng. J., 2025, 518, 164840 CrossRef CAS.
- G. Li, S. Sun, C. Chi, G. Ma, Q. Zhang, J. Li, H. Jin, S. Zhao, S. Wang and X. Bo, Chem. Eng. J., 2025, 519, 165176 CrossRef CAS.
- J. Liu, J. Li, Z. Yang, X. Zhao, D. Tu, W. Yang, J. Xu and Y. Yang, Chem. Eng. J., 2025, 512, 162394 CrossRef CAS.
- Y. Song, H. Cui, Y. Gan and W. Gao, Energy Storage Mater., 2025, 77, 104178 CrossRef.
- Z. Wei, P. Mao, C. Liu, G. Lan, M. Ahmad, R. Zheng, Z. Wang, H. Sun and Y. Liu, ACS Appl. Mater. Interfaces, 2021, 13, 58652–58664 CrossRef CAS PubMed.
- X. Ma, J. Zhao, R. Jiao, M. A. Mudassir, X. Xu, Y. Gao, Y. Liu, W. He, J. Cui and X. Zhao, Rare Met., 2025, 44, 4801–4814 CrossRef CAS.
- Y. Cui, L. Zhang, Q. Huo, J. Feng, C. Guo, M. Guo, G. Li, K. Wang and H. Chen, J. Energy Storage, 2025, 113, 115688 CrossRef CAS.
- D. Yang, Q. Guan, B. Wang, D. Zhang, K. Ren, H. Zhou, X. Li, Y. Zhou, Y. Cai, P. Liu, L. Zhao, M. Hou, B. Yang, D. Xue and F. Liang, Adv. Funct. Mater., 2024, 35, 2420014 CrossRef.
- H. Yin, H. Chen, N. Sun, X. Jian, A. Chen, B. Guo, L. Ji, Z. Yang, Y. Liu and B. Xu, Chin. Chem. Lett., 2025, 25, 111590 CrossRef.
- Y. Zheng, Z. Zhang, C. Liu, H. Liao, Z. Li, Y. Jiang, Y. Hou, L. Sun and Y. Gao, Chem. Eng. J., 2025, 510, 161745 CrossRef CAS.
- Y. Tan, M. Yi, Z. Zhu, X. Zhang, K. Qin, J. Zhang and Z. Rongshu, Mater. Today Commun., 2022, 31, 103740 CrossRef CAS.
- T. Lu, B. Liu, F. Zeng, G. Cheng, S. Chu, M. Xie, Z. Chen and Z. Hou, J. Energy Chem., 2022, 74, 332–340 CrossRef CAS.
- J. Wang, J. Liu, R. T. Subramaniam, D. Zhang, Z. Li, H. J. Woo and B. Wang, J. Power Sources, 2024, 614, 234991 CrossRef CAS.
- S. Cai, D. Deng, Y. Yuan, J. Liang, Y. Ye, H. Huang, L. Jin, Y. Zeng, G. Li, J. Weng, X. Fan, Y. Li and Q. Wu, J. Energy Storage, 2025, 134, 118239 CrossRef CAS.
- T. Wang, Z. Tian, X. Hu, Q. Liu, L. Wang, W. Liu, Y. Zhou, C. Ma, X. Li and R. Li, J. Power Sources, 2025, 648, 237369 CrossRef CAS.
- D. Chen, B. He, S. Jiang, X. Wang, J. Song, H. Chen, D. Xiao, Q. Zhao, Y. Meng and Y. Wang, Chem. Eng. J., 2025, 510, 161676 Search PubMed.
- Y. Wei, Z. Wang, P. Gao, Y. Zhu and X. Li, J. Power Sources, 2025, 646, 237285 CrossRef CAS.
- X. Xu, Y. Chu, Y. Mu, X. Wei, Q. Zhang, H. Rao, H. Gu, L. Pan, M. Han, Y. Wang, L. Zeng and L. Wei, ACS Nano, 2025, 19, 31395–31406 CrossRef CAS PubMed.
- Z. Yu, Q. Wang, K. Zhu, G. Wang, D. Cao and J. Yan, Appl. Surf. Sci., 2024, 652, 159294 CrossRef CAS.
- P. Gao, Q. Ru, Z. Pan, J. Zhang, W. Xu, F. C. C. Ling and L. Wei, J. Colloid Interface Sci., 2021, 599, 730–740 CrossRef CAS PubMed.
- G. Dong, H. Yu, L. Li, R. Zhang, X. Yang, K. Zhu, G. Wang and D. Cao, J. Colloid Interface Sci., 2023, 647, 395–405 Search PubMed.
- L. Fu, C. Kang, W. Xiong, P. Tian, S. Cao, S. Wan, H. Chen, C. Zhou and Q. Liu, J. Colloid Interface Sci., 2021, 595, 59–68 Search PubMed.
- B. Kang, X. Chen, L. Zeng, F. Luo, X. Li, L. Xu, M. Q. Yang, Q. Chen, M. Wei and Q. Qian, J. Colloid Interface Sci., 2020, 574, 217–228 CrossRef CAS PubMed.
- Y. Liu, Z. Lei, R. Liu, X. Li, P. Xiong, Y. Luo, Q. Chen, M. Wei, L. Zeng and Q. Qian, Rare Met., 2023, 42, 1557–1569 CrossRef CAS.
- B. Xu, D. Zhang, C. Peng, F. Liang, H. Zhao, B. Yang, D. Xue and Y. Lei, Adv. Energy Mater., 2023, 13, 2302325 CrossRef CAS.
- Y. A. Fesseha, K. K. Kuchayita, W. N. Su, C. W. Chiu and C. Cheng, Chem. Eng. J., 2025, 509, 161501 CrossRef CAS.
- Y. Jiang, M. Lian, J. Ma, Y. Long, X. Guo, Y. Sun, J. Lao and Z. Ye, Nano Lett., 2025, 25, 7241–7248 Search PubMed.
- H. G. Oh, S. Jang, H. R. Kim, D. Shin, J. Cho, H. Jang, J. K. Kim, I. Nam and S. K. Park, J. Energy Chem., 2025, 111, 462–473 CrossRef CAS.
- J. Song, Y. Ye, S. Cai, D. Deng, G. Li, Y. Zeng, J. Weng, X. Fan, Y. Li and Q. Wu, J. Power Sources, 2025, 657, 238221 Search PubMed.
- Q. Sun, W. Xie, B. Zheng, Y. Xu, F. Xie, T. Tian, J. Zhao, Y. Xiao, S. Lei and B. Cheng, J. Mater. Sci. Technol., 2026, 243, 181–191 Search PubMed.
- H. Peng, W. Miao, J. Zeng, Z. Wang, C. Yan, G. Ma and Z. Lei, Adv. Sci., 2025, 12, e2417288 CrossRef PubMed.
- K. She, Y. Huang, W. Fan, M. Yu, J. Zhang and C. Chen, J. Colloid Interface Sci., 2024, 656, 270–279 Search PubMed.
- B. Zhao, G. Suo, R. Mu, C. Lin, J. Li, X. Hou, X. Ye, Y. Yang and L. Zhang, J. Colloid Interface Sci., 2024, 668, 565–574 CrossRef CAS PubMed.
- P. Liu, F. Li, K. Ren, M. Hou, D. Yang, X. Yang, X. Wang, Y. Lei and F. Liang, Nano Energy, 2026, 148, 111637 CrossRef CAS.
- H. L. Andersen, O. K. Al Bahri, S. Tsarev, B. Johannessen, B. Schulz, J. Liu, H. E. A. Brand, M. Christensen and N. Sharma, Dalton Trans., 2018, 47, 1251–1260 Search PubMed.
- Z. Yin, Y. Bu, J. Ren, S. Chen, D. Zhao, Y. Zou, S. Shen and D. Yang, Chem. Eng. J., 2018, 345, 165–173 CrossRef CAS.
- W. Zhang, L. Xing, J. Chen, H. Zhou, S. Liang, W. Huang and W. Li, J. Alloys Compd., 2020, 822, 153530 CrossRef CAS.
- S. Li, W. Zhang, Y. Cui, J. Ma, H.-J. Peng, J. Li, X. Liu, D. H. L. Ng, X. Liu and J. Lian, J. Energy Chem., 2023, 78, 115–122 Search PubMed.
- J. Zhou, T. Ji, S. Xu, X. Wang, J. Wang, Y. Tang, Y. Li, W. Yin, H. Ji, S. Shi and G. Yang, Chem. – Eur. J., 2025, 27, 202500589 CrossRef PubMed.
- J. Xu, H. Guo, K. Huo and Y. Li, J. Electroanal. Chem., 2022, 920, 116598 Search PubMed.
- J. Cai, Y. Li, Z. Zhang, A. Cui, Z. Cai, Q. Xie and J. Wang, J. Power Sources, 2025, 638, 236572 Search PubMed.
- B. Pati, P. K. Samal, J. Panigrahy, S. Praharaj and D. Rout, J. Energy Storage, 2026, 141, 119187 Search PubMed.
- Y. Zhang, X. Wang, C. Shi, B. Han, C. Zhou, G. Wang, J. Li, R. Sun and K. Amine, ACS Nano, 2025, 19, 27270–27279 Search PubMed.
- B. Wang, E. H. Ang, Y. Yang, Y. Zhang, H. Geng, M. Ye and C. C. Li, Adv. Funct. Mater., 2020, 30, 202001708 Search PubMed.
- C. Yang, Q. Xiang, X. Li, Y. Xu, X. Wang, X. Xie, C. Li, H. Wang and L. Wang, J. Mater. Sci., 2020, 55, 12053–12064 CrossRef CAS.
- L. Yu, X. Tao, D. Sun, L. Zhang, C. Wei, L. Han, Z. Sun, Q. Zhao, H. Jin and G. Zhu, Adv. Funct. Mater., 2024, 34, 202311471 Search PubMed.
- F. J. García-García, J. Mosa, A. R. González-Elipe and M. Aparicio, Electrochim. Acta, 2019, 321, 134669 CrossRef.
- A. Zimmer, M. Tresse, N. Stein, D. Horwat and C. Boulanger, Electrochim. Acta, 2020, 360, 136931 CrossRef CAS.
- Z. Zhang, X. Zhao and J. Li, ChemNanoMat, 2016, 2, 196–200 CrossRef CAS.
- S. Li, H. Hou, Z. Huang, H. Liao, X. Qiu and X. Ji, Electrochim. Acta, 2017, 245, 949–956 CrossRef CAS.
- S. Gao, Y. He, K. Zhu, H. Yin, Z. Yang, G. Yue, J. Bai, Z. Cui, N. Wang, Q. Zhang and Y. Zhao, Chem. Eng. J., 2025, 523, 168326 Search PubMed.
- H. Ge, J. Liu, W. Shi, Y. Zhang and X. Wang, Chin. Chem. Lett., 2025, 25, 111972 CrossRef.
- C. Xiao, C. Wei, L. Qin, H. Cao, L. Yang, W. Deng, C. Qin, J. Wang and J. Guo, J. Energy Storage, 2025, 124, 116836 Search PubMed.
- Y. Yang, W. Hu, F. Liu, A. Li, D. Huang and R. Zou, Chem. Eng. J., 2025, 521, 166766 CrossRef CAS.
- W. Tao, J. Chen, C. Xu, S. Liu, S. Fakudze, J. Wang and C. Wang, Small, 2023, 19, 202207397 Search PubMed.
- B. Fan, H. Fan, X. Chen, X. Gao, S. Chen, Q. Tang, W. Luo, Y. Deng, A. Hu and W. Hu, ACS Appl. Mater. Interfaces, 2021, 13, 19894–19903 CrossRef CAS PubMed.
- Y. Li, R. Zhang, W. Zhou, X. Wu, H. Zhang and J. Zhang, ACS Nano, 2019, 13, 5533–5540 CrossRef CAS PubMed.
- D. Sun, D. Huang, H. Wang, G.-L. Xu, X. Zhang, R. Zhang, Y. Tang, D. Abd Ei-Hady, W. Alshitari, A. Saad Al-Bogami, K. Amine and M. Shao, Nano Energy, 2019, 61, 361–369 CrossRef CAS.
- S. H. Choi and Y. C. Kang, Nanoscale, 2015, 7, 3965–3970 RSC.
- B. Zhang, X. Chen, H. Zhao, H. Xie and H. Yin, Rare Met., 2023, 42, 1227–1237 CrossRef CAS.
- X. Lin, Y. Li, Z. Dong and J. Xu, J. Energy Storage, 2024, 92, 112212 Search PubMed.
- J. Han, W. Xu, Z. Liu, Z. Gao, S. Tao, H. Min, H. Yang and J. Wang, J. Alloys Compd., 2023, 957, 170282 CrossRef CAS.
- Y. Hu, Y. Bai, X. Wu, X. Wei, K. Wang and J. Chen, J. Alloys Compd., 2019, 797, 1126–1132 Search PubMed.
- M. B. Sreedhara, A. L. Santhosha, A. J. Bhattacharyya and C. N. R. Rao, J. Mater. Chem. A, 2016, 4, 9466–9471 Search PubMed.
- W. Kang, Y. Wang, D. Cao, Z. Kang and D. Sun, J. Alloys Compd., 2018, 743, 410–418 CrossRef CAS.
- W. Liu, M. Wei, L. Ji, Y. Zhang, Y. Song, J. Liao and L. Zhang, Chem. Phys. Lett., 2020, 741, 137061 CrossRef CAS.
- N. Li, Q. Zhou, J. Lin, Y. Lu, Z. Hou and Y. Qian, J. Alloys Compd., 2022, 922, 166306 CrossRef CAS.
- X. Hu, R. Zhu, B. Wang, X. Liu and H. Wang, Small, 2022, 18, e2200437 Search PubMed.
- J. S. Cho, S. K. Park, K. M. Jeon, Y. Piao and Y. C. Kang, Appl. Surf. Sci., 2018, 459, 309–317 Search PubMed.
- P. A. Shinde, V. Mahamiya, M. Safarkhani, N. R. Chodankar, M. Ishii, R. Ma, A. A. Ghaferi, L. K. Shrestha and K. Ariga, Adv. Funct. Mater., 2024, 34, 202406333 Search PubMed.
- H. Fan, Q. Zhang, Q. Gu, Y. Li, W. Luo and H. Liu, J. Mater. Chem. A, 2019, 7, 13736–13742 Search PubMed.
- X. Yue, J. Wang, Z. Xie, A. M. Patil, T. Yu, X. Du, Z. Wang, X. Hao, A. Abudula and G. Guan, J. Mater. Sci., 2020, 55, 14389–14400 Search PubMed.
- K. Wu, J. Zhan, G. Xu, C. Zhang, D. Pan and M. Wu, Nanoscale, 2018, 10, 16040–16049 Search PubMed.
- H. Wang, H. Jiang, Y. Hu, P. Saha, Q. Cheng and C. Li, Chem. Eng. Sci., 2017, 174, 104–111 CrossRef CAS.
- X. Luo, J. Shao, P. He, K. Li and W. Zhao, Appl. Surf. Sci., 2023, 607, 154821 Search PubMed.
- Q. Zhao, K. Yu, S. Wang and C. Liang, J. Alloys Compd., 2022, 927, 167004 CrossRef CAS.
- H. Liu, H. Guo, B. Liu, M. Liang, Z. Lv, K. R. Adair and X. Sun, Adv. Funct. Mater., 2018, 28, 201707480 Search PubMed.
- H. Zhang, Z. Han, X. Li, F. Kong, S. Tao and B. Qian, Ceram. Int., 2021, 47, 26839–26846 Search PubMed.
- Y. Liu, N. Wang, X. Zhao, Z. Fang, X. Zhang, Y. Liu, Z. Bai, S. Dou and G. Yu, J. Mater. Chem. A, 2020, 8, 2843–2850 Search PubMed.
- X. Li, J. Zhang, Z. Liu, C. Fu and C. Niu, J. Alloys Compd., 2018, 766, 656–662 CrossRef CAS.
- J. Wang, Y. Shao, F. Yuan, H. Sun, D. Zhang, Z. Li, S. Ramesh, H. J. Woo and B. Wang, J. Energy Chem., 2023, 80, 291–301 Search PubMed.
- Z. Wei, Y. Zhang, M. Li, C. Shu and D. Deng, J. Energy Storage, 2024, 81, 110115 Search PubMed.
- H. He, Y. Man, J. Yang, J. Xie and M. Xu, R. Soc. Open Sci., 2017, 4, 170892 Search PubMed.
- B. Chen, H. Lu, J. Zhou, C. Ye, C. Shi, N. Zhao and S. Z. Qiao, Adv. Energy Mater., 2018, 8, 201702909 Search PubMed.
- X. Lu, Z. Wang, K. Liu, J. Luo, P. Wang, C. Niu, H. Wang and W. Li, Energy Storage Mater., 2019, 17, 101–110 CrossRef.
- H. T. Huu, N. S. M. Viswanath, N. H. Vu, J.-W. Lee and W. B. Im, Nano Res., 2021, 14, 3977–3987 Search PubMed.
- G. Ali, A. J. Kiani, F. J. Iftikhar, S. A. Abbas Kazmi, M. Akbar, A. Rauf and K. Y. Chung, ACS Appl. Energy Mater., 2021, 4, 4638–4645 Search PubMed.
- J. Sheng, H. Zang, C. Tang, Q. An, Q. Wei, G. Zhang, L. Chen, C. Peng and L. Mai, Nano Energy, 2016, 24, 130–138 Search PubMed.
- S. Jeong, E. Cho and W. Choi, J. Power Sources, 2024, 617, 235141 CrossRef CAS.
- W. Wang, L. Hu, J. Ge, Z. Hu, H. Sun, H. Sun, H. Zhang, H. Zhu and S. Jiao, Chem. Mater., 2014, 26, 3721–3730 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |