Open Access Article
Open Access ArticleA metal–metal quadruply bonded dimer of two pincer-ligated metal centers
Souvik Mandal a,
Ethan Y. Song
a,
Ethan Y. Song a,
Thomas J. Emge
a,
Thomas J. Emge a,
Faraj Hasanayn
a,
Faraj Hasanayn *b and
Alan S. Goldman
*b and
Alan S. Goldman *a
*a
aDepartment of Chemistry and Chemical Biology, Rutgers, The State University of New Jersey, New Brunswick, New Jersey 08854, USA. E-mail: alan.goldman@rutgers.edu
bDepartment of Chemistry, American University of Beirut, Beirut 1107 2020, Lebanon. E-mail: fh19@aub.edu.lb
First published on 11th May 2026
Abstract
Reduction of the pincer complex (PArNP)MoBr3 using Na/Hg under argon yields a quadruply bonded Mo–Mo dimer, (κ3-PArNP)BrMo![[quadruple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e003.gif) Mo(κ3-PArNP)Br. The complex represents a rare example of a metal–metal bonded species in which both metal centers bear a κ3-mer coordinated pincer ligand.
Mo(κ3-PArNP)Br. The complex represents a rare example of a metal–metal bonded species in which both metal centers bear a κ3-mer coordinated pincer ligand.
Complexes with metal–metal bonds comprise one of the most intensively studied classes of coordination compounds.1–4 The metal–metal quadruple bond, in particular, has fascinated chemists since first reported by Cotton in 1964.5,6
The field of pincer ligands has seen explosive growth over the past two to three decades. Interest stems from the great tunability that pincer ligands offer with respect to both steric and electronic factors, the ease and modularity of their syntheses, and the high stability of their complexes, attributable to multidentate bonding.7–9 These properties lead to numerous applications in diverse fields including catalysis and optoelectronics.10–15
Despite the breadth of these two classes of coordination complexes – metal–metal bonds and pincer-ligated systems – there has been little overlap between them. In particular there have been remarkably few reports of metal–metal bonded complexes in which both metals are κ3-pincer-ligated. To our knowledge, the only such examples (all reported quite recently) are Pt–Pt complexes in which each Pt center bears a planar κ3 NCN- or NNC-type ligand.16–18 These planar pincer ligands impose minimal steric hindrance, while the Pt–Pt bonds are quite long (>3.0 Å) and are supported by an ancillary ligand bridging the two Pt centers. Being the only examples of the (pincer)M–M(pincer) motif, these complexes might suggest that shorter metal–metal bonds and more “classical” pincer ligands19 (those with bulky terminal coordinating groups e.g. PR2 or NR2) would represent a highly unfavorable combination. Herein, however, we report a complex in which both Mo centers are ligated by a relatively bulky “classical” pincer, Ozerov's bis(2-(diisopropylphosphino)-4-methylphenyl)amide (PArNP),20 yet are linked by a short (2.17 Å) Mo–Mo quadruple bond.
There are over 1500 structurally characterized examples of metal–metal quadruply-bonded species reported in the 2025 Cambridge Structural Database,21 the majority of which (over 900) are Mo–Mo complexes. In all but about 80 cases, one or more bridging ligands are present and the non-bridging ligands are monodentate. Among the few complexes with tridentate ligands, in all but one case they are bridging, coordinating in a κ2 fashion to one Mo center and κ1 to the second (e.g. Scheme 1a).22–25 To date, only a single exception has been reported. In 2002, Walton and co-workers described a Mo![[quadruple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e003.gif) Mo complex in which a pyridine-based PNP ligand is κ3-coordinated to one of the two Mo centers (Scheme 1b).26
Mo complex in which a pyridine-based PNP ligand is κ3-coordinated to one of the two Mo centers (Scheme 1b).26
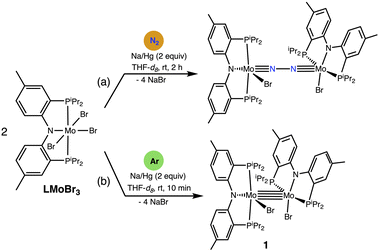
We recently reported the binding and splitting of dinitrogen by two (PArNP)MoX fragments by reaction of LMoBr3 (L = PArNP) with sodium amalgam under nitrogen atmosphere (Scheme 2a).27 When 2 equiv. sodium amalgam is added to a THF-d8 solution of LMoBr3 under argon atmosphere rather than under dinitrogen (Scheme 2b), upon vigorous shaking the color changes from green to dark purple and the 1H NMR spectrum reveals the complete loss of LMoBr3 with formation of a new diamagnetic product. This species is unreactive toward added N2, or even toward PMe3 or CO. The 31P{1H} NMR spectrum of the product mixture displays an AB doublet at 37.0 and 29.5 ppm with 2JPP = 104 Hz. Crystals were obtained by diffusion of pentane into a saturated THF solution of the product, and X-ray diffractometry reveals a Mo–Mo unit in which each Mo center bears a PArNP pincer ligand and a bromide, complex 1 (Scheme 2b; Fig. 1).
Table 1 gives selected X-ray crystallographic bond distances in 1 and compares them with those optimized using the M06 density functional in the gas phase, starting with the experimental structure. The non-metals carried the 6-311G(d,p) basis set, while molybdenum carried the SDD relativistic ECP and corresponding basis set (see the SI for computational details). The crystallographic Mo–Mo bond length in 1 is 2.172 Å, at the upper end of the normal range of Mo–Mo quadruple bond lengths, 2.06–2.17 Å.1,6 The calculated bond length, 2.166 Å, is in good agreement with the experimental value.
| Bond distances (Å) | 2 (XRD) | DFT optimized |
|---|---|---|
| Mo1–Mo2 | 2.172 | 2.166 |
| Mo1–N1 | 2.191 | 2.180 |
| Mo2–N2 | 2.164 | 2.198 |
| Mo1–P1 | 2.532 | 2.559 |
| Mo1–P2 | 2.521 | 2.582 |
| Mo2–P3 | 2.538 | 2.553 |
| Mo2–P4 | 2.551 | 2.561 |
| Mo1–Br1 | 2.569 | 2.623 |
| Mo2–Br2 | 2.544 | 2.625 |
The two i-Pr substituents on each pincer ligand that are syn to the Mo–Mo bond occupy essentially equatorial positions of the 5-membered metallacyclic rings. This arrangement minimizes steric crowding between the two units, while bringing the methine CH bonds to within approximately 2.7 Å of the bromo or amido ligand centers of the opposing metal center, suggesting some degree of hydrogen-bonding (Fig. 1b).28,29 The 1H NMR spectrum of 1 in THF indicates that the structural features of the ligands observed in the solid state are retained in solution. Specifically, four multiplets are observed at δ 4.65, 2.84, 2.23, and 1.50 ppm, corresponding to four i-Pr methine protons in very different environments.
An NBO analysis identifies one σ, two π, and one δ occupied MO in the Mo–Mo bond (Fig. 2) with predominantly metal d-character (>85%). Thus our experimental and computational results are fully consistent with a Mo–Mo quadruple bond in 1.1,6
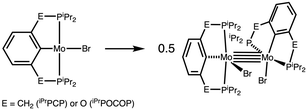
Given the unprecedented nature of a metal–metal bonded complex composed of two metal fragments with non-planar pincer ligands, it seemed surprising to find a system combining a relatively bulky pincer ligand with a particularly short M–M distance characteristic of a metal–metal quadruple bond. We considered whether features specific to the PArNP ligand, such as the H-bonding between metal centers discussed above, might strongly favor dimerization. To probe this possibility, hypothetical analogues with phenyl-based pincer ligands, iPrPCP and iPrPOCOP were investigated computationally. For this purpose, final electronic and solvation free energies were obtained by single point calculations in a polarisable continuum representing THF as solvent on the gas-phase geometries using the M06-D3 density functional and a def2 basis set as described in the SI. These calculations rule out a unique stabilizing role of PArNP: the computed Mo–Mo bond strengths in [(iPrPCP)MoBr]2 and [(iPrPOCOP)MoBr]2 (Scheme 3) are actually slightly greater than that calculated for 2 (see SI).†
Surprisingly, when the reduction of LMoBr3 under argon was carried out with 2 equiv. KC8 instead of Na/Hg, complex 1 was not formed. Instead, the 1H NMR spectrum indicated rapid formation of a paramagnetic species that was stable at room temperature for at least 20 days. The magnetic susceptibility of the product was determined by the Evans method to be 4.6μB, indicating a quintet ground spin state. Our initial experimental and computational investigations suggest this species is a THF adduct of the (PArNP)MoBr fragment LMoBr(THF)n, Scheme 4.
Attempts to trap the putative species LMoBr(THF)n support its formulation as a solvated form of the LMoBr fragment. Upon addition of 1 atm CO, a THF solution immediately changed from purple to red, and a diamagnetic species was observed by 1H and 31P{1H} NMR spectroscopy. When the reaction was performed with 13CO, the 31P{1H} and 13C{1H} NMR spectra clearly demonstrated the formation of a (PArNP)Mo species with three inequivalent CO ligands and two inequivalent PiPr2 groups (see SI). These spectra are readily assigned to the seven-coordinate complex LMoBr(CO)3 (Scheme 4), which is well precedented by Kirchner's halotricarbonyl molybdenum complexes.30,31 Attempts to crystallize this species were unsuccessful.
Addition of 2 equiv. of PMe3 to a solution of LMoBr(THF)n resulted in an immediate color change from purple to magenta. In contrast to the reaction with CO, the resulting 1H NMR spectrum was indicative of a paramagnetic species, assigned as LMoBr(PMe3)2 (Scheme 4). The magnetic susceptibility, measured by the Evans method, was 2.9μB, consistent with a triplet ground state.
Ligand binding to LMoBr was computationally investigated. Coordination of two THF molecules is calculated to be exothermic by 17.8 kcal mol−1, affording trans-LMoBr(THF)2 with a quintet ground state (although with an enthalpy only 1.0 kcal mol−1 lower than the corresponding triplet). Coordination of CO or PMe3 was calculated to be much more exergonic, yielding LMoBr(CO)3 and trans-LMoBr(PMe3)2 which are computed to have singlet and triplet ground states, respectively. These results (Table 2) are fully consistent with the experimental NMR and magnetic susceptibility data.
| Complex | Spin state | ΔH | ΔG |
|---|---|---|---|
| a Gibbs free energies and enthalpies computed at the M06-D3/def2 level in THF continuum and reported in kcal mol−1 with respect to the calculated energy of LMoBr (with the formation of 0.5 mol 2 indicated as 0.5 times the total values for 1.0 mol 2). Values of ΔG for formation of THF complexes are derived with a statistical correction to account for the concentration of THF solvent, 10.0 M (244.7 atm) and scaling of the rotational and translational entropy components by a factor of 0.5;32–34 see SI.b Attempts to optimize a quintet state of cis-LMoBr(THF)2 led to the decoordination of one THF ligand and to a complex LMoBr(THF)·THF in which the O atom of the non-coordinated THF molecule is H-bonded to an α-C–H bond of the coordinated molecule of THF. | |||
| LMoBr | Quintet | 0.0 | 0.0 |
| trans-LMoBr(THF)2 | Triplet | −16.8 | −10.7 |
| trans-LMoBr(THF)2 | Quintet | −17.8 | −12.6 |
| cis-LMoBr(THF)2 | Triplet | −13.3 | −8.3 |
| LMoBr(THF)·THFb | Quintet | −19.0 | −16.2 |
| trans-LMoBr(CO)2 | Singlet | −60.3 | −35.8 |
| cis-LMoBr(CO)2 | Singlet | −66.4 | −40.5 |
| LMoBr(CO)3 | Singlet | −81.2 | −44.0 |
| trans-LMoBr(PMe3)2 | Triplet | −39.2 | −22.5 |
| cis-LMoBr(PMe3)2 | Triplet | −38.0 | −21.3 |
| Dimerization: 0.5 1 | Singlet | 0.5(−51.6) | 0.5(−33.4) |
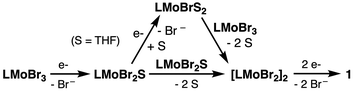
The assignment of LMoBr(THF)2 as the paramagnetic species formed upon reaction of LMoBr3 with KC8 has important mechanistic implications, namely that dimer 1 does not form via direct dimerization of monomeric LMoBr. Use of the highly effective reductant KC8 leads to rapid and quantitative reduction of LMoBr3 to give LMoBr(THF)2. While further mechanistic studies are required, we speculate that when the less effective reductant Na/Hg is employed, the reduction of LMoBr3 is sufficiently slow to allow reaction with the initially formed LMoBr(THF)2. An alternative possibility is that the presumed initial reduction product, LMoBr2(THF), is very rapidly reduced by KC8 to give LMoBr(THF)2, whereas reduction by Na/Hg is slow enough to permit LMoBr2(THF) to undergo dimerization. In either case, reaction of LMoBr3 with Na/Hg generates a complex with a Mo2Br4 core, which is subsequently reduced to form complex 1 (Scheme 5). At present we favor the former pathway, as DFT calculations indicate that reaction of LMoBr(THF)2 with LMoBr3 is thermodynamically much more favorable (ΔG = −0.6 kcal mol−1) than dimerization of LMoBr2(THF) (ΔG = 19.6 kcal mol−1).
In summary, a metal fragment bearing a typical (relatively bulky and non-planar) pincer ligand has been found to undergo dimerization to ultimately form a metal–metal quadruply bonded complex. Computational studies suggest that such dimerizations are thermodynamically favorable more generally. In light of the extensive interest in both metal–metal bonded complexes and pincer-ligated complexes, this points to a broad and unexplored class of bimetallic pincer complexes.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting the findings of this study are available within the article and its supplementary information (SI). Supplementary information: experimental procedures and characterizations, crystallographic data, NMR spectra, computational methods and results; optimized structures for calculated species (.mol format) (ZIP). See DOI: https://doi.org/10.1039/d6dt00920d.CCDC 2457509, 2459044, 2515783 and 2516137 contain the supplementary crystallographic data for this paper.35a–d
Acknowledgements
This work was supported through the National Science Foundation Chemical Catalysis program under Grant No. CHE-2247259. F. H. acknowledges support from the Mamdouha El-Sayed Bobst funds of AUB. Prof. Oleg Ozerov (Texas A&M University) is thanked for helpful discussions and a gift of PArNP synthetic precursor.References
- H. Vahrenkamp, Angew. Chem., Int. Ed. Engl., 1978, 17, 379–392, DOI:10.1002/anie.197803793.
- P. Buchwalter, J. Rosé and P. Braunstein, Chem. Rev., 2015, 115, 28–126, DOI:10.1021/cr500208k.
- J. F. Berry and C. C. Lu, Inorg. Chem., 2017, 56, 7577–7581, DOI:10.1021/acs.inorgchem.7b01330.
- R. H. Duncan Lyngdoh, H. F. Schaefer III and R. B. King, Chem. Rev., 2018, 118, 11626–11706, DOI:10.1021/acs.chemrev.8b00297.
- F. A. Cotton, N. F. Curtis, C. B. Harris, B. F. G. Johnson, S. J. Lippard, J. T. Mague, W. R. Robinson and J. S. Wood, Science, 1964, 145, 1305–1307, DOI:10.1126/science.145.3638.1305.
- Multiple bonds between metal atoms, ed. F. A. Cotton, C. A. Murillo and R. A. Walton, Springer Science & Business Media, New York, 2006 Search PubMed.
- G. van Koten and D. Milstein, Organometallic Pincer Chemistry, Springer GmbH, 2013 Search PubMed.
- E. Peris and R. H. Crabtree, Chem. Soc. Rev., 2018, 47, 1959–1968, 10.1039/C7CS00693D.
- Pincer Compounds: Chemistry And Applications, ed. D. Morales-Morales, Elsevier B.V., Amsterdam, 2018 Search PubMed.
- M. Albrecht and D. Morales-Morales, Iridium Complexes Org. Synth., 2009, 299–323 CAS.
- K. J. Szabo, Top. Organomet. Chem., 2013, 40, 203–242, DOI:10.1007/978-3-642-31081-2_7.
- The Privileged Pincer-Metal Platform: Coordination Chemistry & Applications, in Top. Organomet. Chem., ed. G. van Koten and R. A. Gossage, Springer GmbH, 2016, p. 54 Search PubMed.
- A. Kumar, T. M. Bhatti and A. S. Goldman, Chem. Rev., 2017, 117, 12357–12384, DOI:10.1021/acs.chemrev.7b00247.
- K. Das and A. Kumar, Adv. Organomet. Chem., 2019, 72, 1–57, DOI:10.1016/bs.adomc.2019.02.004.
- V. Arora, H. Narjinari, P. G. Nandi and A. Kumar, Dalton Trans., 2021, 50, 3394–3428, 10.1039/D0DT03593A.
- M. A. Esteruelas, S. Moreno-Blázquez, M. Oliván and E. Oñate, Inorg. Chem., 2024, 63, 14482–14494, DOI:10.1021/acs.inorgchem.4c01712.
- R. A. Shilov, I. S. Podkorytov, K. S. Kisel, E. E. Galenko, D. O. Karpitskaya, I. A. Rodionov, J. R. Shakirova and S. P. Tunik, Inorg. Chem., 2024, 63, 11194–11208, DOI:10.1021/acs.inorgchem.4c00984.
- A. Yu, W. Jiang, Y. Shen and Y. Zhang, Cryst. Growth Des., 2025, 25, 1872–1879, DOI:10.1021/acs.cgd.4c01747.
- M. Vogt and R. Langer, Eur. J. Inorg. Chem., 2020, 3885–3898, DOI:10.1002/ejic.202000513.
- L. Fan, B. M. Foxman and O. V. Ozerov, Organometallics, 2004, 23, 326–328, DOI:10.1021/om034151x.
- C. R. Groom and F. H. Allen, Angew. Chem., Int. Ed., 2014, 53, 662–671, DOI:10.1002/anie.201306438.
- F. A. Cotton and B. Hong, Inorg. Chem., 1993, 32, 2354–2359, DOI:10.1021/ic00063a026.
- C.-T. Lee, S.-F. Chiang, C.-T. Chen, J.-D. Chen and C.-D. Hsiao, Inorg. Chem., 1996, 35, 2930–2936, DOI:10.1021/ic951187b.
- C.-T. Lee, W.-K. Yang, J.-D. Chen, L.-S. Liou and J.-C. Wang, Inorg. Chim. Acta, 1998, 274, 7–14, DOI:10.1016/S0020-1693(97)05961-6.
- M.-C. Suen, S.-F. Chiang, J.-D. Chen, S.-S. Chern and C.-D. Hsiao, J. Chin. Chem. Soc., 1998, 45, 263–268, DOI:10.1002/jccs.199800042.
- H.-F. Lang, P. E. Fanwick and R. A. Walton, Inorg. Chim. Acta, 2002, 329, 1–8, DOI:10.1016/S0020-1693(01)00787-3.
- S. Mandal, S. Malakar, R. N. Allen, A. Tyryshkin, T. J. Emge, F. Hasanayn and A. S. Goldman, ChemRxiv, 2025, preprint. DOI:10.26434/chemrxiv-2025-40qzq.
- M. Domagała and S. J. Grabowski, J. Phys. Chem. A, 2005, 109, 5683–5688, DOI:10.1021/jp0511496.
- Y. Gu, T. Kar and S. Scheiner, J. Mol. Struct., 2000, 552, 17–31, DOI:10.1016/S0022-2860(00)00455-5.
- S. R. M. M. de Aguiar, B. Stöger, E. Pittenauer, M. Puchberger, G. Allmaier, L. F. Veiros and K. Kirchner, J. Organomet. Chem., 2014, 760, 74–83, DOI:10.1016/j.jorganchem.2013.12.018.
- D. Benito-Garagorri, E. Becker, J. Wiedermann, W. Lackner, M. Pollak, K. Mereiter, J. Kisala and K. Kirchner, Organometallics, 2006, 25, 1900–1913, DOI:10.1021/om0600644.
- T. D. Lohrey, A. Q. Cusumano, W. A. Goddard III and B. M. Stoltz, ACS Catal., 2021, 11, 10208–10222, DOI:10.1021/acscatal.1c02790.
- A. V. Finkelstein and J. Janin, Protein Eng., 1989, 3, 1–3, DOI:10.1093/protein/3.1.1.
- Y.-i. Izato, A. Matsugi, M. Koshi and A. Miyake, Phys. Chem. Chem. Phys., 2019, 21, 18920–18929, 10.1039/C9CP03226F.
- (a) CCDC 2457509: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2nh7hx; (b) CCDC 2459044: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2njv02; (c) CCDC 2515783: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qfw9c; (d) CCDC 2516137: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qg7q5.
Footnote |
| † Isodesmic reactions were calculated in which PH2 and PiPr2 groups were exchanged between complex 2 and the analogous RPCP- and RPOCOP-ligated dimeric complexes and analogues truncated with PH2 groups. The computed energies indicated that the PArNP backbone did not confer any particular relief (relative to RPCP or RPOCOP) of steric repulsion between the quadruply bonded metal centers (see SI). |
| This journal is © The Royal Society of Chemistry 2026 |