Open Access Article
Open Access ArticleIn or out? Adaptive metal binding by a diphosphine-based Zr metal–organic cage
Jiehye
Shin
 and
Casey R.
Wade
and
Casey R.
Wade
 *
*
Department of Chemistry and Biochemistry, The Ohio State University, 100 West 18th Ave, Columbus, OH 43210, USA. E-mail: wade.521@osu.edu
First published on 31st March 2026
Abstract
A lantern-type Zr metal–organic cage (MOC), Zr–P2N2, has been synthesized using ditopic carboxylate linkers based on the 1,5-diaza-3,7-diphosphacyclooctane (P2N2) ligand platform. Single-crystal X-ray diffraction shows that the diphosphine groups of the P2N2 linkers point into the confined MOC cavity. Postsynthetic metalation of the diphosphine groups with Pd and Pt precursors generates the heterobimetallic cages Zr–P2N2–PdCl2 and Zr–P2N2–PtCl2. Structural characterization reveals that the linkers flip to accommodate metal chelation, resulting in outward facing metal diphosphine groups. Conversely, reaction of Zr–P2N2 with AgPF6 results in formation of Zr–P2N2–Ag, containing a hexasilver core supported by phosphine complexation within the cage interior. This work highlights the adaptive structural behavior of Zr–P2N2 which provides new opportunities for designing responsive MOCs.
Introduction
Metal–organic cages (MOCs) have attracted increasing interest for applications in catalysis and molecular separation owing to their chemical tunability and inherent porosity.1–3 MOCs are considered molecular analogues of metal–organic frameworks (MOFs).4 Both are assembled from organic linkers and metal ions or clusters and exhibit well-defined cavities or pores. However, unlike MOFs, the molecular nature of MOCs confers solubility and solution-state processability, providing unique opportunities for heterogeneous and homogeneous catalyst design.5A common strategy for designing MOC-based catalysts involves using linkers with secondary ligand groups for catalytic metal binding.6–8 Hard–soft acid–base principles are often exploited to impart orthogonal reactivity between the primary donor groups necessary for cage self-assembly and the secondary ligand groups. For example, the combination of hard Lewis acidic metal ions such as Zr4+ with hard carboxylate ligands can facilitate cage assembly while softer Lewis basic ligands remain available for catalyst binding. Catalytically active metal fragments can be selectively complexed at the secondary donor sites, either via postsynthetic reactions or cage assembly with metallolinkers.9,10 This general strategy has been used to design a variety of heterometallic MOCs with nitrogen-based secondary ligand groups such as bipyridines.11–14
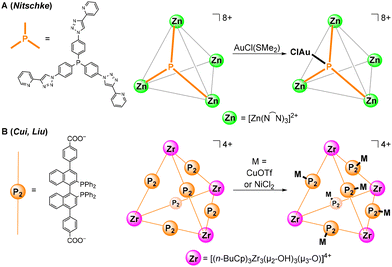
Phosphine ligands are widely used in homogeneous catalysis owing to their steric and electronic versatility and ability to support catalytically active late transition metal species.15–18 However, there have been relatively few reports of MOCs containing phosphines as secondary ligand groups. Nitschke and co-workers reported a Zn-based MOC (Scheme 1A) assembled from triarylphosphine-based linkers.19 The phosphine groups were found to react with AuCl(SMe2), resulting in binding of four AuCl units at the exterior of the tetrahedral cage. Surprisingly, the phosphine MOC exhibited no reaction with other late transition metal precursors such as [Pd(MeCN)4](BF4)2 and [Cu(MeCN)4]OTf. More recently, Cui, Liu, and co-workers reported the synthesis of a tetrahedral Zr MOC (Scheme 1B) using linkers based on chiral 2,2′-bis(diphenylphosphino)-1,1′-binaphthyl (BINAP) ligands.20 The BINAP groups were metalated with CuOTf and NiCl2 to generate heterometallic MOCs that catalyze asymmetric alkylation and alkynyl amination reactions as homogeneous catalysts.
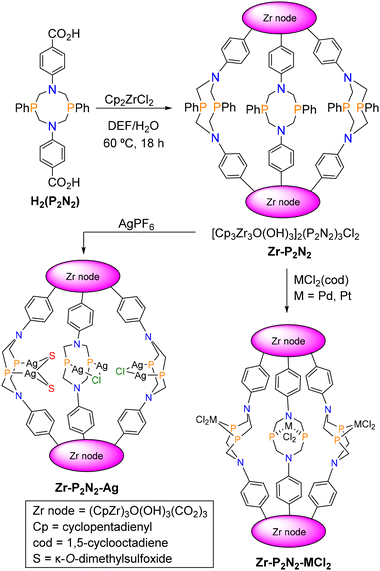
Herein, we describe the synthesis and characterization of a novel, lantern-type Zr MOC, Zr–P2N2, assembled from diphosphine-based linkers (Scheme 2). The ditopic linkers are based on the 1,5-diaza-3,7-diphosphacyclooctane (P2N2) ligand platform, which has been widely studied in homogeneous catalyst design.21–23 The new MOC undergoes postsynthetic metalation with PdCl2(cod), PtCl2(cod), and AgPF6, revealing that metal chelation at the diphosphine sites requires marked structural changes including an outward flip of the linker groups.
Results and discussion
Synthesis and characterization of Zr–P2N2
The H2(P2N2) linker was synthesized following a reported procedure.24Zr–P2N2 was obtained as colourless crystals by solvothermal reaction of H2(P2N2) with Cp2ZrCl2 in a N,N-diethylformamide (DEF) and H2O solvent mixture (Scheme 2). Single crystal X-ray diffraction (SCXRD) revealed formation of a lantern-type cage comprised of a pair of [(CpZr)3(OH)3O]4+ clusters bridged by three (P2N2)2− linkers (Fig. 1a, Table S1). The 8-membered P2N2 heterocycle adopts a crown conformation with the lone pairs of the phosphine groups facing inward to the cage. Previous studies have shown that this is the most energetically favourable ligand conformation, and it is commonly observed in solid state structures of P2N2 ligands.25 The sum of the bond angles around the N atoms (ave. 119.95°) reflects a nearly trigonal planar geometry arising from delocalization of the N lone pair into the aromatic ring of the benzoate group (Table S2). These features result in formation of a trigonal bipyramidal cage with a solvent-accessible cavity that is ∼10 Å in diameter. Disordered DEF solvent molecules residing in the cage and interstitial space could not be adequately modelled in the structure refinement. The associated electron density was removed from the calculation using SQUEEZE (3715 electrons, 64 DEF molecules per unit cell).26 The electron density map and 1H NMR data indicate the presence of approximately one DEF molecule inside the cavity of each cage (Fig. S1).The packing diagram of Zr–P2N2 viewed along the b-axis shows a slipped-stacking arrangement with adjacent molecules displaced by approximately half a molecular length (Fig. 1c). Disordered chloride anions were located in pockets created by the µ-OH groups of the Zr clusters, supporting an empirical formula of {[Cp3Zr3O(OH)3]2(P2N2)3}Cl2 (Fig. 1b). Although the structure contains DEF solvent molecules in the interstitial space between the cages, Zr–P2N2 does not retain permanent, gas accessible porosity after desolvation, and N2 adsorption isotherm measurements showed no measurable gas uptake. The absence of gas-accessible porosity in Zr–P2N2 is consistent with other lantern-type Zr cages.2
Zr–P2N2 is slightly soluble in DMSO, EtOH and MeOH, but is insoluble in H2O and most other polar organic solvents including DMF, acetone, and acetonitrile. The poor solubility of Zr–P2N2 is at least partly attributed to strong hydrogen bonding interactions between the hydroxyl groups of the Zr clusters and the chloride counteranions. The 31P NMR spectrum measured in DMSO-d6 shows a single resonance at −40.9 ppm, which is shifted slightly downfield from the free linker (−47.4 ppm) (Fig. S2 and S3). The 1H NMR spectrum exhibits all expected resonances for the η5-Cp groups and (P2N2)2− linkers. The linker resonances experience very subtle changes in chemical shift compared to H2(P2N2). The µ-OH groups also appear as a sharp singlet at 9.76 ppm, supporting retention of the lantern cage structure in solution.3 The 1H DOSY NMR spectrum of Zr–P2N2 in DMSO-d6 shows an average diffusion coefficient of 7.98 × 10−11 m2 s−1, which is consistent with other reported lantern-type Zr MOCs (Fig. S4).3,27
ESI-MS data obtained for Zr–P2N2 in MeOH solution show that the lantern cage structure is retained, but the phosphine groups undergo partial oxidation (Fig. S5). A mass signal corresponding to the parent cage (z = +2) is observed at m/z = 1346.04 and accompanied by a progression of minor species corresponding to sequential oxidation of the three diphosphine groups. The 31P NMR spectrum of Zr–P2N2 in MeOD-d4 exhibits a single resonance at −43 ppm, even after exposure to ambient conditions for several days (Fig. S6). This result suggests that phosphine oxidation occurs in situ during the ESI-MS experiment rather than during sample preparation. Signals corresponding to the +1 (–H+), +3 (+H+), and +4 (+2H+) forms of Zr–P2N2 are also observed. Importantly, the isotopic distributions of these signals are consistent with the lantern-type structure, and no mass signals are observed in the m/z = 1789–1806 range, which would indicate the presence of a tetrahedral cage impurity with z = +3.
Synthesis and characterization of Zr–P2N2–MCl2 (M = Pd, Pt)
The inward facing diphosphine groups of Zr–P2N2 are not well poised to accommodate metal chelation. In addition to the small pore size (10 Å diameter, 589 Å3 volume), the phosphine lone pairs diverge, precluding metal chelation without a significant distortion of the cage. Thus, we hypothesized that postsynthetic metalation would require either a linker flip such that the diphosphine groups point outward from the cage or restructuring to give a tetrahedral cage with more sterically accessible phosphine sites. Accordingly, suspensions of Zr–P2N2 in MeOH were treated with 3 equiv. of PdCl2(cod) (cod = η4-1,5-cyclooctadiene) or PtCl2(cod) and gently stirred for 16 h at room temperature. After isolation and washing, the solid products, Zr–P2N2–PdCl2 and Zr–P2N2–PtCl2, were analysed by 31P and 1H NMR spectroscopy in DMSO-d6. The 31P NMR spectra showed a single resonance at +9.5 and −7.4 ppm for Zr–P2N2–PdCl2 and Zr–P2N2–PtCl2, respectively (Fig. S7 and S8). The presence of a single, downfield shifted signal indicates formation of symmetric cages containing equivalent P2N2–MCl2 metallolinkers (Fig. S9 and S10). The 1H NMR spectra also exhibit signals consistent with symmetric, metalated linkers.The postsynthetic metalation was further investigated by titrating Zr–P2N2 solutions in DMSO-d6 with up to 4 equiv. of MCl2(cod) (M = Pd, Pt) and monitoring the reactions by 1H and 31P NMR spectroscopy (Fig. S11 and S12). Upon addition of 1–2 equiv. of the metal precursors, the 31P NMR spectra show the appearance of product resonances at +10 ppm (Zr–P2N2–PdCl2) and −6.8 ppm (Zr–P2N2–PtCl2) with a concomitant decrease in the parent phosphine resonance at −41 ppm. The product signals appear abruptly and without the appearance of any discernible intermediate species.
We have been unable to obtain single crystals of Zr–P2N2–PdCl2 and Zr–P2N2–PtCl2 suitable for X-ray diffraction using the postsynthetic metalation approach. Consequently, H2(P2N2–PdCl2) and H2(P2N2–PtCl2) metallolinkers were prepared (Fig. S9 and S10) and used for direct assembly of Zr–P2N2–PdCl2 and Zr–P2N2–PtCl2. The 31P and 1H NMR spectra of the products match those obtained from postsynthetic metalation reactions, supporting the formation of the same products in both cases (Fig. S13 and S14). The direct assembly gave single crystals suitable for X-ray diffraction, revealing formation of isostructural, lantern-shaped MOCs with P2N2–MCl2 metallolinkers. As predicted, the linkers undergo a flip, resulting in square planar PdCl2 and PtCl2 diphosphine groups that point outward from the cage (Fig. 2, Tables S3 and S4, Fig. S15 and S16). The heterocyclic P2N2 rings adopt chair–boat conformations with two of the MCl2 units pointed up and the third pointed downward with respect to the primary axis of the lantern cage.25 The presence of a single resonance in the 31P NMR spectra indicates that the metallolinkers are dynamic in solution, undergoing rapid exchange of the up/down orientations of the MCl2 units. The change in conformation of the P2N2 linkers from crown-type in Zr–P2N2 to chair–boat in the Zr–P2N2–MCl2 structures also has a pronounced effect on the shape of the cages. The Zr⋯Zr distances between the metal nodes in Zr–P2N2–MCl2 (17.6 Å) are significantly longer than for the parent cage (15.1 Å) (Fig. S17). A concomitant decrease in the equatorial diameter diminishes the internal volume of the Zr–P2N2–MCl2 cages. Residual electron density in the interstitial spaces between the Zr–P2N2–MCl2 cages, attributed to disordered solvent molecules, was removed from the structure refinement using SQUEEZE. The calculation indicated 1839 electrons for Zr–P2N2–PdCl2 and 1596 electrons for Zr–P2N2–PtCl2 within the masks, corresponding to ∼32 and ∼28 DEF molecules per unit cell, respectively. The electron density maps suggest that no DEF molecules are present within the cavities of the cages, consistent with their decreased pore size compared to Zr–P2N2. Overall, the SCXRD data reveal significant structural changes, including a linker flip to generate outward facing Pd/Pt-diphosphine groups and a notable increase in the Zr⋯Zr intercluster distances.
 | ||
| Fig. 2 Structures of (a) Zr–P2N2–PdCl2 and (b) Zr–P2N2–PtCl2. (Hydrogen atoms are omitted for clarity. Cyan: Zr, red: O, gray: C, green: Cl, magenta: Pd, orange: P, blue: N, yellow: Pt.) | ||
The 1H DOSY NMR spectra of Zr–P2N2–MCl2 in DMSO-d6 show average diffusion coefficients of 8.89 × 10−11 m2 s−1 for Pd and 8.61 × 10−11 m2 s−1 for Pt (Fig. S18 and S19). These values are slightly larger than those of the parent Zr–P2N2 (7.98 × 10−11 m2 s−1), indicating that the elongated structures of the Zr–P2N2–MCl2 cages reduces their effective hydrodynamic radius (Table S5).27,28
Zr–P2N2–PdCl2 and Zr–P2N2–PtCl2 exhibit limited solubility in MeOH and DMSO and are insoluble in water and all other polar organic solvents tested. Moreover, the 1H and 31P NMR spectra measured in methanol-d4 showed obvious decomposition, as indicated by the appearance of PhPH2 (31P: −124 ppm) and multiple new 1H NMR resonances in the 6–9 ppm region (Fig. S20 and S21). This solvent sensitivity and limited solubility precluded characterization of the Zr–P2N2–MCl2 cages using ESI-MS. Attempts to improve their solubility via PF6−/Cl− salt metathesis were largely unsuccessful due to increased solvent sensitivity (see SI for additional details). Consequently, Zr–P2N2–PdCl2 and Zr–P2N2–PtCl2 were analysed by MALDI-TOF mass spectrometry to verify that the lantern-type cage is maintained in bulk samples (Fig. S22 and S23). The MALDI spectrum of Zr–P2N2–PdCl2 displayed two mass signals (z = +2, 1612.29 amu and 1593.77 amu) that correspond to the parent lantern-type cage and the same ion with loss of an HCl equivalent from the Zr–OH and PdCl2 groups. The MALDI spectrum of Zr–P2N2–PtCl2 showed only a single mass signal (z = +2, 1745.39 amu) associated with the parent ion. No trace signals corresponding to tetrahedral cages were observed for either sample. These results indicate that Zr–P2N2–PdCl2 and Zr–P2N2–PtCl2 exclusively form lantern-type cages, consistent with the structures determined by SCXRD.
Synthesis and characterization of Zr–P2N2–Ag
The solid-state structures of Zr–P2N2–PdCl2 and Zr–P2N2–PtCl2 raised the question of whether Zr–P2N2 could accommodate metal coordination at the diphosphine groups in the cage interior without undergoing a linker flip. To test this possibility, we considered silver phosphine species which present a minimal steric profile. Accordingly, a suspension of Zr–P2N2 in DMSO was treated with 6 equiv. of AgPF6. Colourless crystals of Zr–P2N2–Ag suitable for X-ray diffraction were obtained by ethyl acetate vapor diffusion into the reaction solution. However, the structure solution and refinement were limited by weak diffraction and significant disorder of the guest solvent molecules and counteranions, resulting in high R-factors (R1 = 0.2131, wR2 = 0.5733, see SI for additional details). The resulting data preclude detailed analysis of bond metrics but establish connectivity of the core structure. Zr–P2N2–Ag adopts a lantern-type structure resembling the parent Zr–P2N2 but with dinuclear Ag species complexed at each of the diphosphine ligands in the interior of the cage (Fig. 3, Fig. S30, and Tables S6 and S7). The cage is bisected by a mirror plane in the Pbcm space group, resulting in crystallographically inequivalent linkers. Two of the diphosphine ligands are equivalent and support unsymmetric, dinuclear species (Ag2 and Ag3) capped by a µ-Cl ligand originating from the Zr–P2N2 counteranion. The remaining diphosphine ligand is bisected by the mirror plane and supports a symmetric dinuclear Ag species with two bridging κ-O-DMSO ligands. As a result, each Ag1 atom is three-coordinate and adopts a distorted Y-shaped geometry. The presence of two chloride ligands in the hexanuclear Ag core suggests that each Zr–P2N2–Ag molecule should bear an overall charge of +6, which is balanced by disordered PF6− or Cl− ions residing outside of the cage.The Zr⋯Zr distance between metal nodes in Zr–P2N2–Ag (ave. 15.7 Å) is comparable to that of the parent Zr–P2N2 (15.1 Å), indicating that the cage does not require significant distortion to accommodate complexation of the Ag species. The preferential accumulation of the three Ag2 units inside the cage appears to arise from a combination of steric and geometric factors inherent to the P2N2 ligands and the cage structure. The intraligand P⋯P distances are relatively short for the chelate species in Zr–P2N2–MCl2 (ave. 3.039 Å for Pd and 3.029 Å for Pt) and much longer for the dinucleating ligands in Zr–P2N2–Ag (ave. 3.912 Å) owing to the diverging metal–phosphine bond vectors. The different coordination modes affect the relative orientation of the phenyl substituents, which splay toward the P2MCl2 chelate groups in Zr–P2N2–MCl2 but bend away from the P2Ag2 units in Zr–P2N2–Ag. As a result, the inward-facing P2Ag2 groups alleviate unfavorable steric interactions that would occur if they were outward-facing.
Crystalline samples of Zr–P2N2–Ag were dissolved in DMSO-d6 and analysed by 1H and 31P NMR spectroscopy. The 31P NMR spectrum is consistent with the presence of the three crystallographically unique silver-phosphine sites observed in the solid-state structure (Fig. 3b). The three phosphine resonances centered around −19, −23, and −28 ppm each appear as two doublets owing to spin–spin coupling with 107Ag (I = 1/2, ∼52% abundance) and 109Ag (I = 1/2, ∼48% abundance) isotopes (Fig. 4). The 31P NMR spectrum was also measured at a higher magnetic field strength to clearly resolve the individual signals and coupling constants of the isotopologues (Fig. S34). The 1J(107Ag–31P) and 1J(109Ag–31P) coupling constants were assigned based on the relative gyromagnetic ratios of the isotopes (γ(109Ag)/γ(107Ag) ≈ 1.15).29 The 1J(107Ag–31P) (582–617 Hz) and 1J(109Ag–31P) (676–710 Hz) values are comparable to those reported for other diphosphine bridged, dinuclear Ag complexes.30,31 However, given the similarity in the Ag coordination environments and NMR chemical shifts, the resonances cannot be unambiguously assigned to the corresponding phosphine groups in the structure of Zr–P2N2–Ag. The 1H NMR spectrum displays three Zr–OH group resonances and overlapping multiplets in both the aromatic and aliphatic regions, which is consistent with the lower symmetry of Zr–P2N2–Ag relative to the parent cage (Fig. S31). Zr–P2N2–Ag was also analyzed by mass spectrometry using MALDI-TOF and ESI-MS in acetonitrile solution (Fig. S35–S37). Both spectra show signals for z = +2 ions corresponding to the hexasilver parent cage with solvent exchange or adduct formation with the MALDI matrix. In addition, inductively-coupled plasma optical emission spectroscopy (ICP-OES) provided a Zr![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ag ratio of 1
Ag ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.98 which is consistent with the expected stoichiometry (6 Ag and 6 Zr per cage) for Zr–P2N2–Ag (Fig. S38).
0.98 which is consistent with the expected stoichiometry (6 Ag and 6 Zr per cage) for Zr–P2N2–Ag (Fig. S38).
 | ||
| Fig. 4 31P{1H} NMR spectrum of Zr–P2N2–Ag (DMSO-d6, 162 MHz) showing the Ag–P coupling and chemical shifts of the doublets. | ||
Conclusions
In summary, we report the synthesis and structural characterization of a new lantern-type MOC, Zr–P2N2, comprised of trinuclear Zr metal nodes and linkers based on the known P2N2 ligand motif. There are only two previously reported MOCs with phosphine-functionalized linkers, and both contain sterically accessible phosphine groups poised for metal coordination.19,20 However, in Zr–P2N2, the diphosphine donor groups point into the small cavity of the lantern-type cage. The limited pore space and divergent phosphine lone pairs preclude diphosphine chelation of metal fragments inside the cage. Consequently, reaction of Zr–P2N2 with MCl2(cod) (M = Pd, Pt) induces a linker flip, resulting in outward-facing (P2N2)PdCl2 and (P2N2)PtCl2 groups. This adaptive behaviour points to dynamic linker exchange processes reminiscent of those observed for related Zr MOFs.32,33 In contrast, metalation of Zr–P2N2 with AgPF6 affords a hexasilver cage, Zr–P2N2–Ag, with dinuclear Ag species complexed in the interior of the cage. Retention of the inward-facing diphosphine groups is enabled by the smaller steric profile of the dinuclear silver fragments along with the bridging, rather than chelating, mode of the diphosphine ligands. The adaptive structure of Zr–P2N2 offers possibilities for designing switchable catalysts, sensors, or selective metal capture systems. Our ongoing efforts are focused on improving the solubility and stability of Zr–P2N2 and related MOCs to further explore these applications.Experimental
Materials and methods
Bis(hydroxymethyl)phenylphosphine, PdCl2(cod), and PtCl2(cod) were prepared following reported procedures.34–36 4-aminobenzoic acid (Alfa Aesar, 99%), trans-2-[3-(4-tert-butylphenyl)-2-methyl-2-propenylidene]malononitrile (DCTB, Fisher, 98+%), N,N-dimethylformamide (DMF, Fisher Chemical), N,N-diethylformamide (DEF, TCI America), acetone (Fisher Chemical), ethyl acetate (Fisher Chemical), and dimethyl sulfoxide (VWR Chemicals) were used as received. Cp2ZrCl2 (Thermo Scientific, 98%) was used as received and stored in a N2-filled glovebox. Phenylphosphine (Beantown Chemical) and AgPF6 (TCI America) were used as received and stored at −20 °C in a N2-filled glovebox. All other solvents and reagents were of reagent grade and used as received.Solution-state NMR spectra were collected using a Bruker DPX 400 or AVIII 600 MHz spectrometer. For 1H NMR spectra, the solvent resonance was referenced as an internal standard. For 31P{1H} NMR spectra, 85% H3PO4 was used as an external standard (0 ppm). For 1H DOSY NMR spectroscopy, saturated solutions of the analyte in DMSO-d6 or MeOD-d4 were analysed using a Bruker AVIII 600 MHz spectrometer. The μ-OH resonances were excluded for average diffusion coefficients (D) calculations due to their potential exchange with H2O.
Single crystal X-ray diffraction analyses were performed using a Bruker Kappa Photon III Diffractometer equipped with Mo Kα radiation (λ = 0.71073 Å). Each crystal was mounted on a MiTeGen micromount with paratone oil. The analyses were conducted under N2 flow at temperatures of 100 K or 240 K. The structures were solved using ShelXT and refined with SHELXL implemented in the Olex2 1.5 software package.37–39
ICP-OES analyses were performed using an Agilent 5100 ICP-OES spectrometer. Zr–P2N2–Ag samples were digested in a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) mixture of H2SO4 (ThermoSci, 99.999% metal-basis) and H2O2 (Ricca Chemical). Calibration curves were prepared using commercial ICP standards (Ag, VWR Chemicals; Zr, Inorganic Ventures). Powder X-ray diffraction patterns were collected using a Rigaku Miniflex 600 Diffractometer equipped with nickel-filtered Cu Kα radiation (λ = 1.5418 Å).
1 (v/v) mixture of H2SO4 (ThermoSci, 99.999% metal-basis) and H2O2 (Ricca Chemical). Calibration curves were prepared using commercial ICP standards (Ag, VWR Chemicals; Zr, Inorganic Ventures). Powder X-ray diffraction patterns were collected using a Rigaku Miniflex 600 Diffractometer equipped with nickel-filtered Cu Kα radiation (λ = 1.5418 Å).
Mass spectrometry
Electrospray ionization (ESI) mass spectrometry data were collected with a ThermoFisher Orbitrap Exploris MX and Vanquish HPLC mass spectrometer. MALDI-TOF mass spectrometry data were collected using a Bruker Microflex LRF in reflection mode. Saturated solutions of analyte and matrix (DCTB) were prepared separately in 100 µL of DMSO. The solutions were then combined in an Eppendorf tube, and the resulting mixture was spotted onto a Bruker MSP 96 polished steel BC plate. After drying completely, the spots formed light yellow solids. ESI-MS and MALDI-TOF MS spectra were simulated using the molecular mass calculator and “Soup of Peaks” tools available via the open-access EPFL Mass Spectrometry Toolbox.40Synthetic procedures
NMR titration procedures
Author contributions
J. S.: investigation, formal analysis, writing; C. R. W.: conceptualization, formal analysis, supervision, writing.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: NMR spectral data, mass spectrometry data, crystallographic data, PXRD data, ICP-OES data, additional experimental details. See DOI: https://doi.org/10.1039/d6dt00536e.CCDC 2535025–2535029 (Zr–P2N2, Zr–P2N2–PdCl2, Zr–P2N2–PtCl2, Zr–P2N2–Ag (masked counteranions) and Zr–P2N2–Ag) contain the supplementary crystallographic data for this paper.41a–e
Acknowledgements
This research was supported by the U.S. National Science Foundation under Grant No. CHE-2044904. We acknowledge resources from the Campus Chemical Instrumentation Center Nuclear Magnetic Resonance Facility and Mass Spectrometry and Proteomics Facility at The Ohio State University. These facilities are supported in part by funding from OSU's Enterprise for Research, Innovation and Knowledge.References
- S. Lee, J. H. Lee, J. C. Kim, S. Lee, S. K. Kwak and W. Choe, ACS Appl. Mater. Interfaces, 2018, 10, 8685–8691 CrossRef CAS PubMed.
- A. J. Gosselin, G. E. Decker, B. W. McNichols, J. E. Baumann, G. P. A. Yap, A. Sellinger and E. D. Bloch, Chem. Mater., 2020, 32, 5872–5878 CrossRef CAS.
- M. G. Sullivan, H. K. Welgama, M. R. Crawley, A. E. Friedman and T. R. Cook, Chem. Mater., 2024, 36, 567–574 CrossRef CAS.
- B. S. Pilgrim and N. R. Champness, ChemPlusChem, 2020, 85, 1842–1856 CrossRef CAS PubMed.
- E.-S. M. El-Sayed, Y. Di Yuan, D. Zhao and D. Yuan, Acc. Chem. Res., 2022, 55, 1546–1560 CrossRef CAS PubMed.
- C. García-Simón, R. Gramage-Doria, S. Raoufmoghaddam, T. Parella, M. Costas, X. Ribas and J. N. H. Reek, J. Am. Chem. Soc., 2015, 137, 2680–2687 CrossRef PubMed.
- S. Chen, K. Li, F. Zhao, L. Zhang, M. Pan, Y.-Z. Fan, J. Guo, J. Shi and C.-Y. Su, Nat. Commun., 2016, 7, 13169 CrossRef CAS PubMed.
- J. Guo, Y. Xu, K. Li, L. Xiao, S. Chen, K. Wu, X. Chen, Y. Fan, J. Liu and C. Su, Angew. Chem., Int. Ed., 2017, 56, 3852–3856 CrossRef CAS PubMed.
- J. Jiao, C. Tan, Z. Li, Y. Liu, X. Han and Y. Cui, J. Am. Chem. Soc., 2018, 140, 2251–2259 CrossRef CAS PubMed.
- M. T. Yong, O. M. Linder-Patton and W. M. Bloch, Inorg. Chem., 2022, 61, 12863–12869 CrossRef CAS PubMed.
- G. Liu, M. Zeller, K. Su, J. Pang, Z. Ju, D. Yuan and M. Hong, Chem. – Eur. J., 2016, 22, 17345–17350 CrossRef CAS PubMed.
- H. S. Lee, S. Jee, R. Kim, H.-T. Bui, B. Kim, J.-K. Kim, K. S. Park, W. Choi, W. Kim and K. M. Choi, Energy Environ. Sci., 2020, 13, 519–526 RSC.
- C. Ji, W. Wang, E.-S. M. El-Sayed, G. Liu, Y. Si, K. Su, Z. Ju, F. Wu and D. Yuan, Appl. Catal., B, 2021, 285, 119782 CrossRef CAS.
- S. Kim, S. Jee, K. M. Choi and D.-S. Shin, Nano Res., 2021, 14, 486–492 CrossRef CAS.
- C. A. Tolman, Chem. Rev., 1977, 77, 313–348 CrossRef CAS.
- M. L. Clarke and J. J. R. Frew, in Organometallic Chemistry, ed. I. J. S. Fairlamb and J. M. Lynam, Royal Society of Chemistry, Cambridge, 2009, vol. 35, pp. 19–46 Search PubMed.
- D. Setiawan, R. Kalescky, E. Kraka and D. Cremer, Inorg. Chem., 2016, 55, 2332–2344 CrossRef CAS PubMed.
- W. Chen, P. Cai, H. Zhou and S. T. Madrahimov, Angew. Chem., Int. Ed., 2024, 63, e202315075 CrossRef CAS PubMed.
- C. T. McTernan, T. K. Ronson and J. R. Nitschke, J. Am. Chem. Soc., 2019, 141, 6837–6842 CrossRef CAS PubMed.
- X. Zhao, M. Sun, Y. Li, H. Jiang, Y. Cui and Y. Liu, J. Am. Chem. Soc., 2026, 148, 1141–1152 CrossRef CAS PubMed.
- E. S. Wiedner, A. M. Appel, S. Raugei, W. J. Shaw and R. M. Bullock, Chem. Rev., 2022, 122, 12427–12474 CrossRef CAS PubMed.
- Z. Chen, E. S. Isbrandt and S. G. Newman, Org. Lett., 2024, 26, 7723–7727 CrossRef CAS PubMed.
- E. S. Isbrandt, D. E. Chapple, N. T. P. Tu, V. Dimakos, A. M. M. Beardall, P. D. Boyle, C. N. Rowley, J. M. Blacquiere and S. G. Newman, J. Am. Chem. Soc., 2024, 146, 5650–5660 CrossRef CAS PubMed.
- G. N. Nikonov, A. S. Balueva, O. A. Erastov and B. A. Arbuzov, Bull. Acad. Sci. USSR, Div. Chem. Sci., 1989, 38, 1223–1226 CrossRef.
- S. Latypov, A. Strelnik, A. Balueva, Y. Spiridonova, A. Karasik and O. Sinyashin, Eur. J. Inorg. Chem., 2016, 2016, 1068–1084 CrossRef CAS.
- A. L. Spek, Acta Crystallogr., Sect. C: Struct. Chem., 2015, 71, 9–18 CrossRef CAS PubMed.
- G. A. Taggart, A. Guliyeva, K. Kim, G. P. A. Yap, D. J. Pochan, T. H. Epps and E. D. Bloch, J. Phys. Chem. C, 2023, 127, 2379–2386 CrossRef CAS.
- P. Groves, Polym. Chem., 2017, 8, 6700–6708 RSC.
- C.-W. Burges, R. Koschmieder, W. Sahm and A. Schwenk, Z. Naturforsch., A, 1973, 28, 1753–1758 CAS.
- M. Ohkouchi, D. Masui, M. Yamaguchi and T. Yamagishi, J. Mol. Catal. A: Chem., 2001, 170, 1–15 CrossRef CAS.
- K. Plevová, L. J. Prieto Pabon, M. Gaysinski, S. Poulain-Martini and V. Michelet, ChemPlusChem, 2022, 87, e202200217 CrossRef PubMed.
- M. Kim, J. F. Cahill, Y. Su, K. A. Prather and S. M. Cohen, Chem. Sci., 2012, 3, 126–130 RSC.
- L. Feng, G. S. Day, K.-Y. Wang, S. Yuan and H.-C. Zhou, Chem, 2020, 6, 2902–2923 CAS.
- K. Peng, V. Mawamba, E. Schulz, M. Löhr, C. Hagemann and U. Schatzschneider, Inorg. Chem., 2019, 58, 11508–11521 CrossRef CAS PubMed.
- C. Janot, J.-B. Chagnoleau, N. R. Halcovitch, J. Muir and C. Aïssa, J. Org. Chem., 2020, 85, 1126–1137 CrossRef CAS PubMed.
- A. F. Greene, J. Shin and C. R. Wade, Organometallics, 2024, 43, 2434–2442 CrossRef CAS.
- O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard and H. Puschmann, J. Appl. Crystallogr., 2009, 42, 339–341 CrossRef CAS.
- G. M. Sheldrick, Acta Crystallogr., Sect. C:Struct. Chem., 2015, 71, 3–8 Search PubMed.
- G. M. Sheldrick, Acta Crystallogr., Sect. A:Found. Adv., 2015, 71, 3–8 CrossRef PubMed.
- MS ToolS (EPFL). Online Mass Spectrometry Tools – EPFL MSToolbox. https://ms.epfl.ch/ (accessed 2024–2025).
- (a) CCDC 2535025: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2r2x0s; (b) CCDC 2535026: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2r2x1t; (c) CCDC 2535027: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2r2x2v; (d) CCDC 2535028: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2r2x3w; (e) CCDC 2535029: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2r2x4x.
| This journal is © The Royal Society of Chemistry 2026 |