Open Access Article
Open Access ArticleMolybdenum(VI) borano–imido complexes and their application as Lewis acid catalysts in homocoupling of diazo compounds
Karel Škoch *a,
Michaela Buziková
*a,
Michaela Buziková a,
Michał Jakubczyk
a,
Michał Jakubczyk a,
Krishna Pattanamcheril Anilkumar
a,
Krishna Pattanamcheril Anilkumar ab,
Jan Demel
ab,
Jan Demel a and
Miroslava Litecká
a and
Miroslava Litecká a
a
aInstitute of Inorganic Chemistry of the Czech Academy of Sciences, Husinec-Řež 1001, 250 68, Czech Republic. E-mail: skoch@iic.cas.cz
bDepartment of Inorganic Chemistry, Faculty of Science, Charles University, Hlavova 2030, 128 40, Prague, Czech Republic
First published on 26th February 2026
Abstract
We report the synthesis, structural characterization, and catalytic activity of a borano–imido molybdenum complex featuring a unique motif B–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) MoCl4(thf), prepared via the reaction of a phenylpyridine-based boron azide with [MoCl4(thf)2]. Spectroscopic and crystallographic analyses confirmed the formation of a linear B–N–Mo(VI) linkage. Reaction of the formed imido complex with various neutral donors resulted in solvent-assisted reduction and formation of several structurally diverse Mo(V) derivatives, showing the adaptability of the imido framework. The Mo(VI) imido complex acts as a strong Lewis acid and efficiently catalyses the homocoupling of diazo compounds under mild conditions. For diaryldiazomethanes, the product selectivity between tetraarylethylenes and azines is primarily governed by the electronic nature of the substituents: electron-donating groups favor the formation of tetraarylethylenes, whereas electron-withdrawing groups lead exclusively to 1,2-bis(diarylmethylene)hydrazines. The complex also mediated the transformation of 1-diazoindenes to afford either 1,1′-biindene or bis(1,2-indenylidene)hydrazines, with the selectivity controlled by steric effects. Furthermore, α-diazophenylacetates were efficiently coupled to yield mixtures of maleates and fumarates, accompanied by minor amounts of azine by-product, with negligible influence of solvent or substituent variation.
MoCl4(thf), prepared via the reaction of a phenylpyridine-based boron azide with [MoCl4(thf)2]. Spectroscopic and crystallographic analyses confirmed the formation of a linear B–N–Mo(VI) linkage. Reaction of the formed imido complex with various neutral donors resulted in solvent-assisted reduction and formation of several structurally diverse Mo(V) derivatives, showing the adaptability of the imido framework. The Mo(VI) imido complex acts as a strong Lewis acid and efficiently catalyses the homocoupling of diazo compounds under mild conditions. For diaryldiazomethanes, the product selectivity between tetraarylethylenes and azines is primarily governed by the electronic nature of the substituents: electron-donating groups favor the formation of tetraarylethylenes, whereas electron-withdrawing groups lead exclusively to 1,2-bis(diarylmethylene)hydrazines. The complex also mediated the transformation of 1-diazoindenes to afford either 1,1′-biindene or bis(1,2-indenylidene)hydrazines, with the selectivity controlled by steric effects. Furthermore, α-diazophenylacetates were efficiently coupled to yield mixtures of maleates and fumarates, accompanied by minor amounts of azine by-product, with negligible influence of solvent or substituent variation.
Introduction
Organoimido complexes represent a distinct class of compounds featuring a double bond between nitrogen and transition metals.1 The imido donor [RN]2− is formally a dianionic species that acts as both a σ- and π-donor2 and its electronic nature, general properties and even the geometry are influenced by the metal centre, its oxidation state, the ancillary ligands and the nature of the R group.3 These have played a key role in the development of olefin metathesis, for which Robert Schrock was ultimately awarded the Nobel prize.4 Related compounds can also be encountered as reactive species in diverse nitrogen transfer reactions5 including diverse aminations,6 aziridations7 or nitrogen heterocycle synthesis.8 Although the role of ancillary ligands in imido chemistry is well established, non-carbon-substituted imido complexes remain scarce. Aside from the more accessible silyl–9 and phosphanyl–imido10 species, borano–imido complexes are less explored. Notable examples represent complexes of Ti11 and Nb12 derived from tris(pentafluorophenyl)borane-capped nitrido complexes (C6F5)3B–N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) [M], together with low-valent Mo species formed via hydroboration (or chloroboration) of terminal nitrido complexes.13 These were systematically investigated and also isolated as key intermediates in molybdenum-mediated dinitrogen reduction.14
[M], together with low-valent Mo species formed via hydroboration (or chloroboration) of terminal nitrido complexes.13 These were systematically investigated and also isolated as key intermediates in molybdenum-mediated dinitrogen reduction.14
Recently, we have reported the synthesis of a remarkably robust phenylpyridine boron azide15 1, readily accessible from simple borane LNCBH2 (LNC = 2-phenylpyridine).16 With this work, we report our investigations on its reactivity towards molybdenum ions, synthesis and reactivity of one-of-a-kind borano–imido complexes and their application as Lewis acid catalysts in the homocoupling of diazo compounds.
Results and discussion
Following the general protocol,17 boron azide 1 was treated with [MoCl4(thf)2]18 at 75 °C for 5 h to give a deep red complex 2, which was isolated in 87% yield (Fig. 1). 1H NMR shows signals for the phenylpyridine and cyclopentyl fragments, along with two broad peaks for the coordinated tetrahydrofuran molecule (δH = 4.39 and 1.38 ppm). The 11B NMR signal at δ = 8.0 ppm is remarkably high for a tetracoordinate boron, indicating electron density delocalization toward the nitrogen atom. The structure of 2 was ultimately confirmed by single-crystal X-ray diffraction analysis, revealing a pseudo-octahedral arrangement on molybdenum, in which the imido fragment and tetrahydrofuran adopt a trans-configuration, whereas the axial positions are occupied by chloride anions. The imido-donor atom adopts an almost perfect linear geometry (B–N–Mo angle – 179.3(8)°), while the chloride ligands form a square arrangement slightly tilted toward the tetrahydrofuran molecule (N(2)–Mo–Cl angles ranging from 95° to 99°). The very short N–Mo bond in 2 (1.682(9) Å) lies roughly midway between the typical Ar–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Mo(VI) imido double bond (∼1.72 Å)19 and the N
Mo(VI) imido double bond (∼1.72 Å)19 and the N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Mo(VI) triple bond (∼1.65 Å),20 indicating a specific donor character of the borano–imido ligand. For the molecular structure, see Fig. 2.
Mo(VI) triple bond (∼1.65 Å),20 indicating a specific donor character of the borano–imido ligand. For the molecular structure, see Fig. 2.
We further explored the reactivity of the imido complex toward different donors. The reaction of 2 with bipyridine (bipy) afforded a grey paramagnetic Mo(V) complex 3, which confirmed the preservation of the linear geometry of the imido donor, resulting in a pseudo-octahedral geometry in a mer-type arrangement with the axial chloride donors once again tilting away from the imidoborane scaffold. A pronounced trans effect of the imido donor is observed in the crystal structure, with the Mo–N4 bond over 0.16 Å longer than the Mo–N3 bond. Treatment of 2 with bis(diphenylphosphino)ethane (dppe) gave a poorly soluble orange-red solid compound 4. Although the quality of SC-XRD data was insufficient for deposition to CCDC database due to the poor crystal quality, the structure clearly reveals the fac-isomeric arrangement of the ligands (for the crystal structure model and more details, see the SI). The reaction of 2 with triphenylphosphine, regardless of whether the [Mo]/PPh3 ratio was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 or 1
1 or 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, led to the formation of a brown complex 5, in which the pentacoordinate molybdenum forms a tetragonal pyramidal geometry with the imido donor in an apical position. Due to the general low solubility and the paramagnetic nature of Mo(V) (configuration d1), the obtained complexes were analysed only by EA, HRMS, FTIR and SC-XRD (for the depiction of the crystal structures, see Fig. 2; for details, see the SI).21
2, led to the formation of a brown complex 5, in which the pentacoordinate molybdenum forms a tetragonal pyramidal geometry with the imido donor in an apical position. Due to the general low solubility and the paramagnetic nature of Mo(V) (configuration d1), the obtained complexes were analysed only by EA, HRMS, FTIR and SC-XRD (for the depiction of the crystal structures, see Fig. 2; for details, see the SI).21
To gain a deeper insight into the electronic properties of the imido complex 2, we targeted its isoelectronic fluorene analogue 6. The detailed synthetic procedure is provided in the SI. Briefly, 9H-fluorenone was first reacted with cyclopentylmagnesium bromide, and the resulting alcohol was converted into the corresponding azide 6 using TMSN3, and subsequently, compound 6 was treated with [MoCl4(thf)2]. Unlike the smooth formation of 2, the reaction of the fluorenyl azide 6 with [MoCl4(thf)2] was not selective. Although we cannot rule out the transient formation of the targeted complex, the gradual colour change from reddish-brown to dark green suggested the reduction to Mo(V). Crystallization of the reaction mixture provided a small amount of green crystals of complex 7. SC-XRD analysis revealed the formation of a dimeric species, in which the pentavalent molybdenum atom coordinates both the imido ligand and a tetrahydrofuran molecule. Its octahedral coordination environment is completed by two terminal and two bridging chloride ligands (see Fig. 3). The resulting coordination environment is therefore analogous to that observed in dimeric molybdenum pentachloride.22 However, in direct comparison, the geometry of molecule 7 exhibits a noticeable flattening as the Mo–Mo distances are significantly elongated (3.825(4) Å for compound MoCl5 vs. 3.930(2) Å for compound 7) and the Mo–Cl–Mo angles defined by the bridging chloride bond are widened (98.36(7)° for MoCl5 vs. 100.44(2)° for 7).
Catalytic investigations
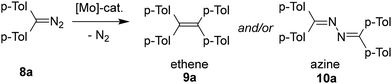
Having prepared complex 2, we next considered its potential application in catalysis. The catalytic chemistry of molybdenum complexes is remarkably broad,23 but we decided to focus on the fact that 2 features a tetrahydrofuran fragment prone to dissociation, efficiently masking a Lewis-acidic site. Motivated by our previous work on borenium-catalysed diazo homocoupling,24 we investigated whether 2 could mediate this transformation as efficiently and extend the substrate scope beyond what main-group Lewis acids allow. While related diazo-to-olefin (homo)couplings have been reported for late-transition metals such as rhodium,25 ruthenium,26 or copper,27 to our knowledge this reactivity has not been reported for any early transition metal.Indeed, under screening conditions (bis-p-tolyldiazomethane 8a as a substrate, 1 mol% catalyst, CDCl3 solvent, 60 min reaction time at room temperature), compound 2 proved catalytically active, affording a complete conversion to the corresponding tetraarylethylene, which was isolated in 95% yield (Table 1, entry 1). In contrast, when the reaction was conducted in aromatic solvents (d6-benzene), the dominant product was the corresponding azine (bis(p-tolylmethylene)hydrazine), isolated in 86% yield (entry 4). We have not previously observed this solvent-dependent selectivity in boron-based catalytic systems (entries 23 and 24). Whereas complexes 3 and 4 were virtually inactive under the screening conditions, complexes 5 and 7 promoted this transformation, albeit with low efficiency. The precursors MoCl5 and [MoCl4(thf)2] also catalysed the reaction; both exhibited inferior activity and selectivity compared to catalyst 2. For comparison, we prepared and evaluated a simple mesitylimido complex [MoCl4(NMes)(thf)] (see the SI for its synthesis and characterization). While this complex also displayed considerable catalytic activity, it did not exhibit the product selectivity observed for catalyst 2. The results are summarized in Table 1.
| Entry | Catalyst | Solvent | Conversiona (%) | |
|---|---|---|---|---|
| Ethene 9a | Azine 10a | |||
| Reaction conditions: 0.2 mmol reaction scale, 1 mol% of the catalyst, 1 mL of solvent, 60 minutes at room temperature.a Conversion estimated by 1H NMR (the isolated yields in parentheses are reported for the reaction performed on a 0.5 mmol scale).b 0.5 mol% of 7 was used. | ||||
| 1 | 2 | CDCl3 | 99(95) | 0 |
| 2 | 2 | CH2Cl2 | 55 | 45 |
| 3 | 2 | Tol | 10 | 90 |
| 4 | 2 | C6D6 | 5 | 95(86) |
| 5 | 2 | Et2O | <5 | 20 |
| 6 | 2 | MTBE | <5 | <5 |
| 7 | 2 | THF | 0 | 0 |
| 8 | 3 | CDCl3 | 0 | 0 |
| 9 | 3 | C6D6 | 0 | 0 |
| 10 | 4 | CDCl3 | 0 | 0 |
| 11 | 4 | C6D6 | 0 | 0 |
| 12 | 5 | CDCl3 | 25 | 0 |
| 13 | 5 | C6D6 | 0 | 20 |
| 14b | 7 | CDCl3 | 20 | 0 |
| 15b | 7 | C6D6 | 0 | 20 |
| 16 | [MoCl4(thf)2] | CDCl3 | 15 | 40 |
| 17 | [MoCl4(thf)2] | C6D6 | 0 | 70 |
| 18 | MoCl5 | CDCl3 | 60 | 40 |
| 19 | MoCl5 | C6D6 | 30 | 55 |
| 20 | [MoCl4(NMes)(thf)] | CDCl3 | 80 | 20 |
| 21 | [MoCl4(NMes)(thf)] | C6D6 | 10 | 90 |
| 22 | B(C6F5)3 | CDCl3 | 95 | 0 |
| 23 | B(C6F5)3 | C6D6 | 90 | 0 |
Encouraged by these results, we evaluated catalyst 2 in a broader substrate set comprising both electron-rich and -poor diaryldiazomethanes, diazoindenes and α-diazo esters. Although solvent effects were evident under catalyst screening conditions, the substrate scope showed that the selectivity is predominantly determined by the electronic nature of the diazo compound. Whereas electron-rich diaryldiazomethanes (8c, p-methoxyphenyl) cleanly afforded the corresponding tetraarylethylene 9 in both CDCl3 and C6D6, electron-poor analogues (8d, p-fluorophenyl and 8e, p-bromophenyl) reacted more slowly, required more forcing conditions (60 °C and extended reaction times) and produced the corresponding azines 10 exclusively. The very electron-poor 8f (3,5-bistrifluoromethylphenyl) did not show any signs of any homocoupling product formation.
We further explored the possibility of homocoupling of synthetically accessible 1-diazoindenes.28 The homocoupling reactions were carried out under more demanding conditions, employing 2 mol% of catalyst 2 and heating to 60 °C. Formation of 1,1′-biindenylidene 9h (ethylene-type coupling product) was observed only in the case of the simplest 1-diazoindene 8h; 9h was isolated as a mixture of E- and Z isomers in approximately 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio. In contrast, sterically encumbered 2-methyl- and 2-phenyl-substituted 1-diazoindenes afforded exclusively the corresponding azine products 10i and 10k, which were isolated in excellent yields up to 93% regardless of the solvent used.
1 ratio. In contrast, sterically encumbered 2-methyl- and 2-phenyl-substituted 1-diazoindenes afforded exclusively the corresponding azine products 10i and 10k, which were isolated in excellent yields up to 93% regardless of the solvent used.
The less nucleophilic methyl phenyldiazoacetates 8k–m exhibited different reactivity patterns and generally required harsh reaction conditions (2 mol% of 2, 5 h at 60 °C) to achieve a full conversion. The main product was an ethylene-type coupling product, giving rise to mixtures of the corresponding 2,3-diphenylmaleate (Z-9) and 2,3-diphenylfumarate (E-9) with a slight preference for the E-isomer, regardless of the presence of electron-donating (p-methoxy) or electron-withdrawing (p-bromo) substituents. NMR measurements also revealed the formation of a small amount of the corresponding azines 10k–m. In some cases, we were able to separate all individual reaction products by column chromatography. Similar to diaryldiazomethanes, the reaction performed in benzene under otherwise identical conditions proceeded worse and did not reach full conversion even after 20 h. On the other hand, the influence of the solvent on the reaction selectivity was marginal (for reaction summary and crystal structures of the selected substrates and products, see Fig. 4 and the SI).
When the catalytic results are compared with those observed for main-group borenium catalysts,18 it was seen that the borano–imido molybdenum Lewis acid 2 was not as active and required a higher catalyst loading for the homocoupling of diaryldiazomethanes (1% for 2 versus 0.1% for the best borenium catalyst). However, complex 2 appears to be more robust and capable of mediating the homocoupling of substrate classes such as α-diazo esters, which generate less nucleophilic carbocation according to Meyr's nucleophilicity scale,29 and were therefore unreactive in borenium-catalysed reactions.
Finally, we attempted the homocoupling of bis-diazoketone 11. In the presence of 2 mol% of 2, a nitrogen molecule was rapidly eliminated to give rise to 2,5-diphenyl-3,4-diazacyclopentadienone (azine-like) intermediate 12, which immediately underwent a double [3 + 2]-dipolar cyclization to provide a polycyclic trimer-like compound 13 as a sole product (see Fig. 5). It should however be noted that the same outcome was proposed as a product of thermolysis of 11 by Trost.30 The structure and head-to-tail arrangement of the polycyclic product 13 were ultimately confirmed by SC-XRD.
We expect that the reaction proceeds in a manner analogous to that we previously described for main-group Lewis acids.24 The initial step involves the interaction of the transition metal centre with the diazo compound, leading to the formation of a metal–carbene intermediate (Fig. 6). This transformation represents a common entry point into the chemistry of highly reactive carbene species, and in numerous cases, such intermediates have been successfully intercepted and characterized.31 The resulting carbene complex is then expected to undergo nucleophilic attack by a second molecule of the diazo compound. Depending on whether the nucleophile is the terminal nitrogen atom (N-nucleophile) or the carbon atom of the diazo compound (C-nucleophile), the reaction furnishes either an azine or an ethene derivative. The preference for C- versus N-nucleophilic attack is primarily governed by the electronic properties of the respective diazo compound, although to a certain degree the selectivity can be modulated by the polarity of the solvent.
Our attempts to perform the reaction under stoichiometric conditions and/or at lower temperatures did not result in the formation of a discrete carbene intermediate. In these experiments, either no reaction occurred or the homocoupling product was observed. These outcomes suggest that generation of a carbene intermediate represents the critical step, which is consistent with the thermodynamic data obtained in our previous work.24
Conclusions
The synthesis, structural characterization, and catalytic applications of the borano–imido molybdenum(VI) complex are presented, as obtained from the reaction of a phenylpyridine-based boron azide with [MoCl4(thf)2]. Spectroscopic and single-crystal X-ray analyses confirmed a linear B–N–Mo linkage with significant electronic delocalization within the borane–imido fragment and a pseudo-octahedral coordination geometry. Coordination studies with neutral donors (bipyridine, dppe and triphenylphosphine) yielded structurally diverse Mo(V) derivatives, highlighting the adaptability of the borano–imido framework.The borano–imido complex efficiently acts as a Lewis acid catalyst promoting the homocoupling of diazo compounds under mild conditions. For diaryldiazomethanes, product selectivity between tetraarylethylenes and azines was influenced primarily by the electronic nature of the substituents and, to a lesser extent, by the solvent. Electron-donating groups favored exclusive formation of tetraarylethylenes, whereas electron-withdrawing substituents led to selective azine formation. The complex also mediated the homocoupling of 1-diazoindenes and α-diazophenylacetates, with selectivity dictated by steric and electronic factors, yielding mixtures of ethylene-type and azine-type products.
Overall, this work establishes borano–imido molybdenum complexes as a new class of electronically distinctive and catalytically active imido species, expanding the scope of imido chemistry and offering new strategies for the homocoupling of diazo compounds.
Conflicts of interest
There are no conflicts to declare.Data availability
Data supporting this article have been included in the supplementary information (SI). Supplementary information: complete experimental procedures, characterisation of the prepared compounds, copies of the NMR spectra, crystallographic parameters and additional structure diagrams. See DOI: https://doi.org/10.1039/d6dt00469e.CCDC 2520309–2520319 contain the supplementary crystallographic data for this paper.32a–k
Acknowledgements
This work was supported by the Czech Science Foundation (project no. 23-06866S) and the Lumina Quaeruntur fellowship (project no. 200401) provided by the Czech Academy of Sciences. IR measurements were supported by the NanoEnviCz infrastructure (LM2023066), provided by the Ministry of Education, Youth and Sports of the Czech Republic. We thank Nina Hlávková for technical assistance with structure preparation.References
- D. E. Wigley, Organoimido Complexes of the Transition Metals, in Progress in Inorganic Chemistry, John Wiley & Sons, 1994, pp. 239–482 Search PubMed.
- (a) W. A. Nugent and B. L. Haymore, Coord. Chem. Rev., 1980, 31, 123–175 CrossRef CAS; (b) R. R. Schrock, Chem. Rev., 2002, 102, 145–180 CrossRef CAS PubMed; (c) R. R. Schrock, Chem. Rev., 2009, 109, 3211–3226 CrossRef CAS PubMed.
- P. Barrie, T. A. Coffey, G. D. Forster and G. Hogarth, J. Chem. Soc., Dalton Trans., 1999, 4519–4528 RSC.
- R. Schrock, Angew. Chem., Int. Ed., 2006, 45, 3748–3759 CrossRef CAS PubMed.
- K. Kawakita, B. F. Parker, Y. Kakiuchi, H. Tsurugi, K. Mashima, J. Arnold and I. A. Tonks, Coord. Chem. Rev., 2020, 407, 213118 CrossRef CAS PubMed.
- Y. Li, Y. Shi and A. L. Odorn, J. Am. Chem. Soc., 2004, 126, 1794–1803 CrossRef CAS PubMed.
- M. N. Cosio and D. C. Powers, Nat. Rev. Chem., 2023, 7, 424–438 Search PubMed.
- (a) K. Kawakita, E. P. Beaumier, Y. Kakuchu, H. Tsurugi, I. A. Tonks and K. Mashima, J. Am. Chem. Soc., 2019, 141, 4194–4198 CrossRef CAS PubMed; (b) T. Akiyama, A. Yamamoto, T. Kasahara, T. Kusamoto and H. Tsurugi, Organometallics, 2025, 44, 1047–1056 CrossRef CAS.
- (a) J. D. Lichtenman, S. C. Critchlow and N. M. Doherty, Inorg. Chem., 1990, 29, 439–442 CrossRef; (b) C. M. Jones, M. E. Lerchen, C. J. Church, B. M. Schomber and N. M. Doherty, Inorg. Chem., 1990, 29, 1679–1682 CrossRef CAS; (c) A. Schorm and J. Sundmeyer, Eur. J. Inorg. Chem., 2001, 2947–2955 CrossRef CAS.
-
(a) For the P–N
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) [Mo] bond, see: G. Zhang, T. Liu, J. Song, Y. Quan, L. Jin, M. Si and Q. Liao, J. Am. Chem. Soc., 2022, 144, 2444–2449 CrossRef CAS PubMed;
(b) For the P–N
[Mo] bond, see: G. Zhang, T. Liu, J. Song, Y. Quan, L. Jin, M. Si and Q. Liao, J. Am. Chem. Soc., 2022, 144, 2444–2449 CrossRef CAS PubMed;
(b) For the P–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) [W] bond, see: A. R. Cowley, J. R. Dilworth, A. K. Nairn and A. J. Robbie, Dalton Trans., 2005, 680–693 RSC.
[W] bond, see: A. R. Cowley, J. R. Dilworth, A. K. Nairn and A. J. Robbie, Dalton Trans., 2005, 680–693 RSC. - (a) A.-M. Fuller, W. Clegg, R. W. Harrington, D. L. Hughes and S. J. Lancaster, Chem. Commun., 2008, 5776–5778 RSC; (b) A.-M. Fuller, D. L. Hughes, G. A. Jones and S. J. Lancaster, Dalton Trans., 2012, 41, 5599–5609 RSC.
- C. Camp, L. N. Grant, R. G. Bergman and J. Arnold, Chem. Commun., 2016, 52, 5538–5541 RSC.
- (a) M. F. Espada, S. Bennaamane, Q. Liao, N. Saffon-Merceron, S. Massou, E. Clot, N. Nebra, M. Fustier-Boutignon and N. Mézailles, Angew. Chem., Int. Ed., 2018, 57, 12865–12868 CrossRef CAS PubMed; (b) T. Itabashi, K. Arashiba, H. Tanaka, K. Yoshizawa and Y. Nishibayashi, Organometallics, 2022, 41, 366–373 CrossRef CAS; (c) I. Benaissa, B. Rialland, S. Bennaamane, M. F. Espada, N. Saffon-Merceron, M. Fustier-Boutignon, E. Clot and N. Mézailles, Angew. Chem., Int. Ed., 2024, 63, e20242586 CrossRef PubMed.
- (a) S. Bennaamane, M. F. Espada, A. Mulas, T. Personeni, N. Saffon-Merceron, M. Fustier-Boutignon, C. Bucher and N. Mézailles, Angew. Chem., Int. Ed., 2021, 60, 2021–20214 CrossRef PubMed; (b) S. Suginom, A. Okochi, T. Nakamura, A. Konomi, H. Tanaka, K. Yoshizawa and Y. Nishibayashi, J. Am. Chem. Soc., 2025, 147, 26684–26692 CrossRef PubMed.
- K. Škoch, M. Buziková, D. Hnyk, M. Litecká, A. Vykydalová, D. Bavol, K. Lang and K. Kirakci, Inorg. Chem., 2025, 64, 18556–18566 CrossRef PubMed.
- K. Škoch, M. Buziková, D. Hnyk, M. Litecká, M. Kloda, K. Kirakci and K. Lang, Chem. – Eur. J., 2024, 30, e202403263 CrossRef PubMed.
- C. Y. Chou, J. C. Huffmann and E. A. Matta, J. Chem. Soc., Chem. Commun., 1984, 1184–1185 RSC.
- (a) L. Castellani and M. C. Gallazzi, Transition Met. Chem., 1985, 10, 194–195 CrossRef CAS; (b) F. Stoffelbach, D. Saurenz and R. Poli, Eur. J. Inorg. Chem., 2001, 2699–2703 CrossRef CAS.
- T. S. Pilyugina, R. R. Schrock, A. S. Hock and P. Müller, Organometallics, 2005, 24, 1929–1937 CrossRef CAS.
- D. Rütter, M. van Gastel, M. Leutzsch, N. Nöthling, D. SantaLucia, F. Neese and A. Fürstner, Inorg. Chem., 2024, 63, 8376–8389 CrossRef PubMed.
- C. Y. Chou, D. D. Devore, S. C. Huckett, E. A. Maatta, J. C. Huffman and F. Takusagawa, Polyhedron, 1986, 5, 301–304 CrossRef CAS.
- Several polymorphs of dimeric MoCl5 have been reported; for comparison, the β-MoCl5 polymorph was selected CrossRef; J. Beck and F. Wolf, Acta Crystallogr., Sect. B, 1997, 53, 895–903 CrossRef.
- (a) S. C. A. Sousa, I. Cabrita and A. C. Fernandes, Chem. Soc. Rev., 2012, 41, 5641–5653 RSC; (b) K. M. Dawood and K. Nomura, Adv. Synth. Catal., 2021, 363, 1970–1997 CrossRef CAS; (c) J.-L. Wang, J.-T. Li, G.-Y. Wu and C.-X. Zhuo, Trends Chem., 2024, 6, 587–501 Search PubMed.
- M. Buziková, J. Schulz, M. Litecká and K. Škoch, Inorg. Chem., 2026, 65, 1408–1418 CrossRef PubMed.
- (a) J. H. Hansen, B. T. Parr, P. Pelphrey, Q. Jin, J. Autschbach and H. M. L. Davies, Angew. Chem., Int. Ed., 2011, 50, 2544–2548 CrossRef CAS PubMed; (b) M. Cui, S. Lin, H. H. Y. Sung, I. D. Williams, Z. Lin and G. Jia, Organometallics, 2019, 38, 905–915 CrossRef CAS.
- (a) W. Baratta, A. Del Zotto and P. Rigo, Chem. Commun., 1997, 2163–2164 RSC; (b) M. Basato, C. Tubaro, A. Biffis, M. Bonato, G. Buscemi, F. Lighezzolo, P. Lunardi, C. Vianini, F. Benetollo and A. Del Zotto, Chem. – Eur. J., 2009, 15, 1516–1526 CrossRef CAS PubMed.
- (a) C. Zhu, G. Xu, D. Ding, L. Qiu and J. Sun, Org. Lett., 2015, 17, 4244–4247 CrossRef CAS PubMed; (b) P. K. Madarasi and C. Sivasankar, Appl. Organomet. Chem., 2022, 36, e6522 CrossRef.
- S. Xie, Z. Yan, Y. Li, Q. Song and M. Ma, J. Org. Chem., 2018, 83, 10916–10921 CrossRef CAS PubMed.
- T. Bug, M. Hartnagel, C. Schlierf and H. Mayr, Chem. – Eur. J., 2003, 9, 4068–4076 CrossRef CAS PubMed.
- B. M. Trost and P. J. Whitman, J. Am. Chem. Soc., 1974, 96, 7421–7429 CrossRef CAS.
- H. A. Khan, M. Szostak and C. Sivasankar, Org. Biomol. Chem., 2026, 24, 734–766 RSC.
- (a) CCDC 2520309: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qll97; (b) CCDC 2520310: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qllb8; (c) CCDC 2520311: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qllc9; (d) CCDC 2520312: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qlldb; (e) CCDC 2520313: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qllfc; (f) CCDC 2520314: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qllgd; (g) CCDC 2520315: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qllhf; (h) CCDC 2520316: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qlljg; (i) CCDC 2520317: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qllkh; (j) CCDC 2520318: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qlllj; (k) CCDC 2520319: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qllmk.
| This journal is © The Royal Society of Chemistry 2026 |