Open Access Article
Open Access ArticlePurine-derived N-heterocyclic carbene metal complexes: catalytic applications and reactivity
Alejandro Cervantes-Reyes
 *a,
Ricardo Malpica-Calderón
a,
Hugo Valdés
*b and
David Morales-Morales
*a,
Ricardo Malpica-Calderón
a,
Hugo Valdés
*b and
David Morales-Morales
 *a
*a
aInstituto de Química, Universidad Nacional Autónoma de México, 04510 Coyoacán, CDMX, Mexico. E-mail: alejandro.cervantes@iquimica.unam.mx; damor@unam.mx
bDepartamento de Química Orgánica y Química Inorgánica, Instituto de Investigación Química “Andrés M. del Río” (IQAR), Facultad de Farmacia, Universidad de Alcalá, Alcalá de Henares, 28805 Madrid, Spain. E-mail: hugo.valdes@uah.es
First published on 6th February 2026
Abstract
Caffeine and other purine bases are naturally occurring, inexpensive, and readily available heterocycles composed of a fused imidazole–pyrimidine framework, whose five-membered imidazole core corresponds to an N-heterocyclic carbene (NHC) precursor. These features make purine scaffolds ideal precursors for the design of metal-NHC ligands that combine biological relevance, structural diversity, and tunable electronic properties. Over the past two decades (2004–2025), an expanding family of purine-derived NHC complexes has been developed across the periodic table, including those of Ru, Ir, Rh, Ni, Pd, Pt, Cu, Ag and Au, revealing distinctive coordination modes, redox behaviors, and catalytic activities. This review provides a comprehensive overview of their synthetic strategies, structural features, and catalytic applications, emphasizing how the intrinsic anisotropy and multifunctionality of the purine framework enable unique metal–ligand interactions and reactivity patterns relevant to sustainable catalysis, photophysics, and bio-organometallic chemistry.
1. Introduction
The isolation of the first stable free carbene represents a seminal milestone in modern chemistry.1–4 This achievement opened the way for the design of functionalized imidazolylidene scaffolds, later known as N-heterocyclic carbenes (NHCs), which rapidly emerged as powerful ligands in transition-metal catalysis.5–9 Since then, NHC–metal complexes have become an essential class of organometallic compounds, underpinning advances in selectivity control, catalytic efficiency, and reaction design across organic and inorganic chemistry.10,11 Over the past decades, an extensive variety of structurally diverse NHC–metal architectures has been developed, fine-tuning steric and electronic properties to enable new reactivity pathways, enhance substrate scope, and provide cost-effective alternatives for industrially relevant transformations.12–26Purines constitute an especially appealing platform within this landscape. These aromatic heterocycles consist of a fused pyrimidine–imidazole core, and naturally occurring derivatives, such as adenine, guanine, hypoxanthine, xanthine, theobromine, theophylline, isoguanine, and caffeine, exhibit rich tautomeric behavior arising from keto–enol and amine–imine equilibria.27 Fig. 1 depicts the purine skeleton and its derivatives, highlighting the atom numbering scheme adopted throughout this review. The imidazole fragment within the purine structure inherently contains a pre-carbenic site (C8), which can be exploited for metal coordination, providing enhanced stability and tunable electronic environments in organometallic catalysis.28 In some cases, an additional pre-carbenic position may also be available on the pyrimidine ring (C2). Related fused NHC scaffolds based on pyridine–imidazole frameworks have also been described in the literature.29–60
Among these, xanthine derivatives have attracted growing attention due to their natural abundance, low cost, and ready availability. These compounds serve as bio-derived NHC precursors, enabling the development of functionalized NHC–metal complexes suitable for both homogeneous and heterogeneous catalysis.61–66 Their structural diversity allows for derivatization through straightforward N-alkylation or tether modification, yielding ligands with tailored solubility, steric bulk, and electronic profiles, and even surface-anchored catalysts when supported on materials such as graphene, silica, or polystyrene.
While several reviews have summarized the synthetic and biological aspects of purine-based NHC complexes,67–69 their catalytic reactivity has often been only briefly mentioned or treated tangentially. To the best of our knowledge, no comprehensive review has yet focused specifically on the catalytic applications and reactivity studies of NHC–metal complexes derived from purine frameworks.
In this contribution, we present an integrated overview (2004–2025) of the catalytic behavior of purine-derived NHC–metal complexes, encompassing Ru, Ir, Rh, Ni, Pd, Pt, Cu, Ag and Au systems. Special attention is given to their synthetic methodologies, ligand-transfer processes, ancillary-ligand exchange dynamics, and catalytic transformations, including cross-coupling, condensation, and hydrogen-transfer reactions. Through this analysis, we aim to establish purine-based NHC ligands as a sustainable and multifunctional platform in transition-metal catalysis, bridging the gap between bio-inspired design and advanced organometallic reactivity.
2. Purine-based NHC complexes of Group 8 metals
Group 8 of the periodic table comprises iron (Fe), ruthenium (Ru), and osmium (Os). In the literature, several Fe complexes incorporating a purine fragment have been reported.70–76 However, to the best of our knowledge, there are no examples of Fe complexes featuring purine-derived NHC ligands, highlighting a clear gap in this area. Purine-based NHC complexes of Os77 have been explored at the DFT level (B3LYP/6-311++G(d,p)//LANL2DZ), providing valuable theoretical insights into their bonding characteristics and electronic structure.78In contrast to Fe and Os, some purine-based Ru–NHC complexes have been described in the literature, together with their catalytic applications.79 In particular, xanthine-derived Ru(II)–NHC complexes 1a and 1b were evaluated in a series of transformations, including transfer hydrogenation, alkene oxidation, and the synthesis of vinyl ester compounds.79 The transfer hydrogenation reaction was performed in i-propanol, which served simultaneously as a solvent and hydrogen donor (Scheme 1). Both complexes exhibited comparable catalytic performance, affording yields between 59% and 99% across various substrates. Remarkably, an imine substrate was quantitatively reduced to the corresponding secondary amine. This behavior is consistent with previous reports describing Ru-complex-mediated hydrogen transfers.80
The oxidation of alkenes using 1a and 1b (1 mol%) in the presence of NaIO4 produced benzaldehyde and acetophenone selectively from styrene and methylstyrene, respectively, within 30–45 min (Scheme 2a). The formation of the corresponding carboxylic acids was not detected under these conditions. In contrast, oxidation of aliphatic substrates such as 1-hexene and cyclooctene afforded the corresponding carboxylic acids after 24 h in moderate to good yields (Scheme 2b).
 | ||
| Scheme 2 Caffeine-derived Ru–NHC complexes (1a and 1b) catalyze the oxidation of alkenes and the synthesis of vinyl esters, highlighting their versatility in redox and coupling transformations. | ||
Complexes 1a and 1b also catalyzed the formation of vinyl esters from benzoic acid and 1-hexyne under microwave irradiation. The reaction afforded mixtures of Markovnikov (using 1a, yield: 72%, and 1b, yield: 58%) and anti-Markovnikov (using 1a, yield: 28%, and 1b, yield: 42%) products. The distinct Z/E selectivities observed underscore marked differences in the reactivity and stereochemical behavior of the two Ru–NHC catalysts (Scheme 2c). Complex 1b, bearing the bulkier N-substituent on the NHC ligand, favored the formation of the less sterically hindered E-stereoisomer.
The alkylation of anilines via dehydrogenative coupling represents a green and atom-economical strategy for C–N bond formation.81 The methylation of aniline derivatives using methanol as a C1 source (Scheme 3) was accomplished through the in situ generation of a xanthine-based Ru–NHC complex (2).82 This catalytic system exhibited good functional-group tolerance, accommodating halogens, ethers, and nitro substituents. Reported yields ranged from moderate to excellent (62–99%), highlighting the efficiency and versatility of the Ru–NHC framework.
 | ||
| Scheme 3 NHC–Ru complex (2) catalyzes the dehydrogenative methylation of anilines with methanol, affording N-methylaniline derivatives under mild conditions. | ||
The reactivity of Ru cluster 3 was examined in the presence of triphenylphosphine (Scheme 4).77 At room temperature, cluster 3 reacted smoothly to afford the mono- and bis-phosphine-substituted derivatives [Ru3(CO)9(PPh3){μ,η1,κ1-C5N4(CH3)3O2}(μ-H)] (4a) and [Ru3(CO)8(PPh3)2 {μ,η1,κ1-C5N4(CH3)3O2}(μ-H)] (5) in 38% and 8% yields, respectively. When the same reaction was performed in toluene under similar conditions, complexes 4a and 5 were obtained in 41% and 21% yields, respectively. Both products are air-stable in the solid state, although they undergo slow decomposition in organic solution upon exposure to air. Attempts to obtain single crystals suitable for X-ray diffraction were unsuccessful. Cluster 4a can undergo isomerization in solution through several pathways. For instance, migration of the PPh3 ligand either to a second equatorial site on the same ruthenium center or to a different ruthenium center can generate a new isomer. Alternatively, hydride migration to another Ru–Ru edge may also lead to isomer formation. The two species likely differ by substitution at the nitrogen-bound ruthenium atom (major isomer) versus the carbon-bound ruthenium atom (minor isomer), as illustrated in Scheme 4b.
 | ||
| Scheme 4 Ligand exchange behavior of NHC–Ru complex 3 upon addition of PPh3 and postulated structures of the two isomeric forms of the complex (4a and 4b). | ||
3. Purine-based NHC complexes of Group 9 metals
Over the past three decades, the chemistry of Group 9 metals has gained considerable attention owing to their broad catalytic versatility. Cobalt complexes containing a purine fragment have been known since the 1950s.75,76,83–89 However, to date, no examples of purine-derived NHC complexes of cobalt have been reported, despite the existence of numerous Co–NHC complexes in the literature with well-established catalytic applications.90In contrast, several purine-based Ir–NHC complexes have been synthesized, characterized, and evaluated in catalytic transformations. The preparation of Ir(I)–NHC complexes of the type [(NHC)Ir(COD)Cl], where COD denotes 1,5-cyclooctadiene, has enabled their study from multiple perspectives. For example, the chloride ligand can be replaced by fluorinated arylthiolate (SArF) groups through ligand exchange with [Pb(SArF)2],91 which serves as a halide abstractor (Scheme 5). The driving force for this transformation is the formation of the insoluble [PbCl2] byproduct and the strong affinity of Pb(II) for halide ions. The resulting Ir–SArF complexes (7a and 7b) were isolated in 53% and 64% yields, respectively. Interestingly, incorporation of the SArF ligand enhanced cytotoxic activity but reduced selectivity; nevertheless, both complexes showed activity against several cancer cell lines, highlighting their potential as bioactive species.
 | ||
| Scheme 5 Chloride-to-fluorinated arylthiolate ligand exchange in Ir–NHC complexes, illustrating the formation of SArF-substituted derivatives. | ||
The COD ligand in complexes of the type [(NHC)Ir(COD)Cl] can be readily substituted by CO,92–94 affording the corresponding [(NHC)Ir(CO)2Cl] species (Scheme 6).66 This transformation is conveniently achieved by bubbling CO through a solution of the Ir–NHC precursor at 0 °C, a temperature that enhances CO solubility and promotes ligand exchange. The significance of the [(NHC)Ir(CO)2Cl] complexes lies in the fact that their C–O stretching frequencies (ν(CO)) provide a direct measure of the electronic properties of the NHC ligand.18,19,95–97 For complexes 8b–f, the average ν(CO) values were 2019 cm−1 (8a), 2020 cm−1 (8b and 8c), 2022 cm−1 (8d), and 2016 cm−1 (8e). Notably, the corresponding value for the well-known IMes derivative is 2023 cm−1,97 serving as a useful benchmark for comparison.
 | ||
| Scheme 6 Formation of dicarbonyl Ir–NHC complexes through CO substitution of the [(NHC)Ir(COD)Cl] precursor, illustrating ligand exchange from COD to CO. | ||
A characteristic feature of Ir(III) complexes is their ability to activate C(sp2)–H bonds, particularly when such activation enables the formation of five-membered metallacycles. This strategy has been extensively employed for the construction of bidentate ligands and their corresponding Ir(III) coordination compounds.98 In this context, two structural isomers, 9-mer and 9-fac, were isolated and fully characterized.99 Remarkably, the mer-isomer can be converted into the fac-isomer upon treatment with trifluoroacetic acid (TFA) in a CH2Cl2/EtOAc mixture at 65 °C (Scheme 7). The two isomers display distinct photophysical properties, with quantum yields of 0.99 (9-mer) and 0.77 (9-fac), respectively.
 | ||
| Scheme 7 Observed mer-to-fac isomerization of NHC–Ir complex 9 in acidic medium, illustrating reversible structural rearrangement under protonic conditions. | ||
Following a similar C(sp2)–H activation strategy, a series of Ir(III) and Rh(III) complexes bearing C^D-chelating ligands (C = C8 of the purine core; D = CNHC, NR2 or PR2) were synthesized.100–102 In these systems, coordination of the metal to the D ligand precedes the C–H activation of the neutral purine, leading to the formation of an azolyl-type complex.103–105 Subsequent protonation of the free nitrogen atom within the purine moiety affords access to the corresponding protic NHC (pNHC) species (Scheme 8).100,101
 | ||
| Scheme 8 Protonation of the free nitrogen atom within the purine moiety affords the corresponding protic NHC (pNHC) species. | ||
In the case of the xanthine–pyridine C^N chelate ligand (10), the 13C NMR spectra of both complexes exhibit nearly identical chemical shifts for the C8 carbon—174.7 ppm for the pNHC complex (11) and 173.9 ppm for the azolyl analogue (10). This observation indicates that the C8 carbon functions as a carbene donor in both species, irrespective of the protonation state of the adjacent nitrogen atom.100
Coordination through the C2 position of the purine ring is also feasible (Scheme 9).106 The reaction of caffeine with aqueous NaOH, followed by the sequential addition of concentrated HNO3 and triethyl orthoformate [HC(OEt)3], affords the corresponding azolium salt (14), which serves as a versatile precursor for the synthesis of Ir(I) and Rh(I) complexes. This azolium intermediate can also undergo C2–C2 dimerization, leading to the formation of a dimeric bis-NHC species (16), from which bis-NHC carbonyl Rh(I) complexes can be obtained (17). The Tolman electronic parameters (TEPs), determined from FT-IR spectra of the CO stretching bands, were 2057 cm−1 for the dimeric complex and 2058 cm−1 for the monomeric analogue, indicating comparable electron-donating abilities of the two NHC environments. Notably, these TEP values closely match those reported for NHC ligands bearing electron-withdrawing substituents directly attached to the heterocyclic backbone,18 which is consistent with the presence of the carbonyl groups in the purine framework.
When two xanthine-derived NHC ligands are coordinated to the Rh(COD) fragment (18), the COD ligand can be readily replaced by carbon monoxide to yield the corresponding dicarbonyl complex 19 (Scheme 10).107 The FT-IR spectrum displays two characteristic C–O stretching bands at ν(CO) = 2080 and 1962 cm−1, consistent with a cis-configuration of the CO ligands. These relatively low frequencies reflect the strong σ-donor ability of the NHC ligands, which enhances back-donation from the metal center to the carbonyl groups.
 | ||
| Scheme 10 Substitution of the COD ligand by CO in NHC–Rh complex 18, yielding the corresponding dicarbonyl Rh-NHC species. | ||
Iridium and rhodium complexes have been extensively investigated as catalysts in transfer hydrogenation, borylation, and hydroformylation reactions. In this context, some purine-based NHC complexes of Ir and Rh have been successfully applied in these transformations. Scheme 11 summarizes the reaction conditions and turnover numbers (TONs) reported for xanthine-derived Ir–NHC catalysts in transfer hydrogenation.100,108–110
 | ||
| Scheme 11 Transfer hydrogenation and benzene borylation catalyzed by xanthine-based Ir–NHC complexes. | ||
Complex 20, a caffeine-derived Ir–NHC species, exhibited notable catalytic activity in the transfer hydrogenation of ketones (Scheme 11a). The reaction, performed in the presence of KOH at 80 °C for 2 h, afforded TON values of up to 84.108 In contrast, reactions catalyzed by the chelating Ir–NHC complexes 21a and 21b required significantly longer reaction times (30–90 h), achieving TON values of 33 and 13.3, respectively.100
The bis-NHC Ir complex 22 catalyzed the borylation of benzene through C(sp2)–H activation, affording the corresponding product in a low yield (12%) (Scheme 11c).109 The limited catalytic efficiency is likely attributable to the weak σ-donor character of the caffeine-derived NHC ligands, which reduces metal–ligand electron density. Furthermore, the non-chelating nature of the bis-NHC framework appears to disfavor the formation of the active Ir species after the initial catalytic turnover, thereby diminishing overall activity.
Rhodium complexes are widely employed as catalysts in the hydroformylation of olefins. The xanthine-derived Rh–NHC complex 23 effectively catalyzed the hydroformylation of 1-octene under high-pressure conditions (50 bar, CO/H2 mixture) (Scheme 12).110 After 1 h of reaction, a substrate conversion of 39% was achieved, corresponding to a turnover frequency (TOF) of 2410 h−1. The linear-to-branched (l/b) aldehyde ratio at this stage was 1.5. As the reaction progressed and the conversion increased to 80%, the l/b ratio decreased to 0.5, indicating that the branched aldehyde was formed preferentially as the kinetic product.
4. Purine-based NHC complexes of Group 10 metals
Group 10 metals are well known for their pivotal role in cross-coupling catalysis, particularly nickel and palladium, which have been extensively employed in a wide range of C–C and C–heteroatom bond-forming reactions.111–120 Platinum, on the other hand, has found important applications in hydrosilylation of alkynes in aqueous media, where it acts as an active and recyclable catalyst, as well as in hydroamination reactions, which constitute key steps in the synthesis of nitrogen-containing heterocycles.121–123 In addition, several Pt complexes have been explored as anticancer metallodrugs, expanding the relevance of Group 10 chemistry beyond catalysis.124The Ni–NHC complexes (24a–c), stabilized by xanthine-derived ligands and a cyclopentadienyl ancillary group, were evaluated as catalysts in the Suzuki–Miyaura cross-coupling between aryl bromides and arylboronic acids (Scheme 13).125 The catalytic scope was assessed using complex 24a, which delivered 15 examples with yields ranging from 73% to 98%. Notably, 24a also exhibited good activity in the coupling of aryl chlorides—in place of aryl bromides—when PPh3 (5 mol%) was used as a co-ligand, affording biaryl products derived from pharmaceutical substrates such as chlorpromazine (81% yield), fenofibrate (73% yield) and indomethacin (82% yield).
Interestingly, complex 24c is among the few reported examples in which a phenyl group is directly bonded to the N atom of the NHC ligand. Its synthesis is not straightforward and is typically achieved via an Ullmann coupling.125,126 The presence of the phenyl substituent impacts the percent buried volume (%Vbur) of the NHC ligand. %Vbur is defined as the percentage of a sphere (r = 3.5 Å) centered at the metal that is occupied by the ligand.127 For NHCs bearing N-methyl substituents, %Vbur is 27.8% (as in 24a), whereas the introduction of an N-phenyl group increases this value to 31.2% (as in 24c). NHC ligands featuring N-benzyl substituents display a slightly higher %Vbur of 32.3% (as in 24b), consistent with the greater steric demand imposed by bulkier N-substituents around the metal center.
A graphene (GP)-supported, caffeine-derived Ni-NHC catalyst (25) efficiently promoted the arylation of benzoxazole with a broad range of arylboronic acids, affording a representative series of 2-arylbenzoxazoles (Scheme 14).128 Under optimized conditions, benzoxazole reacted with electron-deficient arylboronic acids, such as 4-nitrophenylboronic acid and 3,5-dichlorophenylboronic acid, to give the corresponding products, with yields of up to 78%. Reaction with neutral phenylboronic acid provided the corresponding product in 88% yield. Notably, arylation also proceeded efficiently with heteroarylboronic acids, including 3-thienylboronic acid and 2-furanylboronic acid, affording 87% and 84% yields, respectively.
 | ||
Scheme 14 Benzoxazole arylation reaction promoted by graphene-supported NHC–Ni complex (25). Arylboronic acid-dependent reaction times: a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 h, b 8 h, b![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8.5 h, c 8.5 h, c![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 h, d 9 h, d![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10.5 h, e 10.5 h, e![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9.5 h and f 9.5 h and f![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 h. 10 h. | ||
NHC–palladium complexes are among the most prominent catalysts in modern organic synthesis. In particular, Pd–NHC pre-catalysts are highly versatile and efficient for promoting C–C and C–N cross-coupling as well as carbopalladation reactions, making them fundamental tools in both synthetic chemistry and homogeneous catalysis. The strong Pd–carbene bond endows these complexes with remarkable thermal and chemical stability, ensuring sustained activity of the catalytically relevant Pd(0) species even at low catalyst loadings. Moreover, the rational design of NHC ligands provides the necessary stereoelectronic protection to the metal center, suppressing undesired off-cycle Pd(I) species and maintaining high turnover efficiency.111–113
Under optimized conditions, Pd–NHC complexes enabled the efficient synthesis of structurally diverse molecules, ranging from fine chemicals to pharmaceuticals. Indeed, the development of palladium-catalyzed cross-coupling reactions represents a milestone in catalysis—an achievement recognized by the 2010 Nobel Prize in Chemistry, awarded to Richard F. Heck, Ei-ichi Negishi, and Akira Suzuki.129
In this context, purine-derived NHC–Pd complexes have recently emerged as innovative and efficient catalytic systems. Scheme 15 illustrates xanthine-based Pd(II) complexes that have been employed as catalysts in Suzuki–Miyaura C–C cross-coupling reactions.130–136 Representative examples include reactions where an aryl halide or aryldiazonium salt serves as the electrophilic partner, while phenylboronic acid acts as the nucleophilic coupling reagent. Owing to the polar nature of xanthine ligands, most reported protocols utilize polar solvents—typically water, alcohols (i-propanol or methanol), or THF/water mixtures. The most commonly used bases are K2CO3 and KOH; however, when aryldiazonium salts are employed, the coupling can proceed efficiently under base-free conditions. Temperature also plays a key role, with most reactions being carried out between room temperature and 100 °C, depending on substrate reactivity and catalyst loading.
One of the main advantages of xanthine derivatives is their synthetic versatility, as they can be readily obtained from caffeine, theophylline, or theobromine. The N1, N7, and N9 positions of the xanthine core can be conveniently functionalized via nucleophilic substitution using alkyl halides, enabling straightforward ligand diversification. Complex 26 represents a bis-NHC species derived from caffeine, whereas in complexes 27a and 27b, the N1 and N9 nitrogen atoms were functionalized with methyl or benzyl (Bn) groups, respectively.
Such nitrogen functionalization provides an efficient means to tune the physicochemical properties of the resulting metal complexes or to anchor them onto supports. For instance, complex 31, bearing a sulfonate substituent, exhibits enhanced water solubility and can be further immobilized on a nano-magnetite support, allowing easy recovery using an external magnet. Another noteworthy example is complex 32, which was supported on a polystyrene matrix, demonstrating the versatility of xanthine-derived NHC frameworks for heterogeneous catalysis.
A wide variety of co-ligands can be incorporated around the metal center, providing additional stability and functional diversity. Typical examples include halides, acetates, heterocycles, phosphines, and allyl ligands. The water solubility of complexes 27a–c, for instance, was significantly enhanced by coordination of the sodium triphenylphosphine trisulfonate (TPPTS) ligand, a well-known hydrophilic phosphine. Moreover, the formation of PEPPSI (Pyridine Enhanced Precatalyst Preparation, Stabilization and Initiation)-type complexes is also feasible within this family. Such PEPPSI-NHC systems are generally recognized as highly active and robust catalysts, combining excellent air stability with efficient palladium(II) reduction under catalytic conditions.
A direct comparison of the catalytic activity of the various xanthine-based Pd–NHC complexes is not straightforward, as they have been evaluated under different reaction conditions. Nevertheless, several consistent trends can be identified. Complexes 26, 27, 28, 29, 31, and 30 displayed notable activity in the Suzuki–Miyaura coupling of a broad range of aryl bromides. Among them, the PEPPSI-type complexes exhibited the highest turnover numbers (TONs), reaching values of up to 380 for complex 28b. Substrates containing electron-withdrawing groups (EWGs), such as –NO2, –CHO, –COR, and –CF3, generally provided higher yields than those bearing electron-donating groups (EDGs), including –OH, –OMe, and –alkyl. This behavior reflects the facilitating influence of EWGs on the oxidative addition step, which promotes palladium insertion into the C–Br bond and thereby accelerates the catalytic cycle.
The nature of the aryl halide also exerts a significant influence on catalytic performance. As expected, aryl iodides generally exhibit higher reactivity than aryl bromides, consistent with the well-established order of oxidative addition rates (I > Br > Cl) for palladium-catalyzed cross-coupling reactions. Notably, among the xanthine-derived catalysts, only complex 31 exhibited measurable activity toward the more challenging aryl chlorides, delivering the biphenyl product in 80% yield.
In contrast, aryl diazonium salts undergo oxidative addition much more readily than halides, owing to nitrogen gas evolution, which acts as a strong thermodynamic driving force. Using complex 32a, cross-coupling products were obtained in 58–98% yields under base-free conditions and at room temperature, underscoring the excellent activity of purine-based Pd–NHC catalysts toward diazonium substrates.
Halogenated heterocycles were also evaluated as substrates in cross-coupling reactions. For instance, complex 27c efficiently catalyzed the Suzuki–Miyaura coupling of 2-bromothiophene, affording the desired product in 40% yield at 60 °C. Increasing the reaction temperature to 80 °C markedly improved the yield to 95%. In another example, complex 29a successfully promoted the coupling of 3-bromopyridine, delivering the corresponding product in 97% yield, thus demonstrating the broad substrate tolerance of xanthine-based Pd–NHC catalysts toward heteroaryl bromides.
Complexes 28a, 29a and 29b were also evaluated as catalysts in cross-coupling between N-benzoylglutarimide and arylboronic acids (Scheme 16).133 The reactions were conducted using 2 mol% of catalyst in 1,4-dioxane as the solvent, with K2CO3 as the base, at 120 °C for 15 h. All three complexes displayed comparable catalytic activity, affording the coupling products in 75% (28a), 76% (29a), and 81% (29b) yields, with corresponding TONs of 37.5, 38.0, and 40.5, respectively.
 | ||
| Scheme 16 Cross-coupling between N-benzoylglutarimide and arylboronic acids catalyzed by the Pd–NHC complex. | ||
Another representative transformation catalyzed by palladium complexes is the Heck-type reaction. Scheme 17 summarizes the catalysts that have been employed for this purpose and the corresponding reaction conditions.130,133,135,137 A wide range of solvents have been explored for this transformation, most of which are polar media. Typical examples include water, ethanol, i-propanol, N-methylpyrrolidone (NMP), and dimethylacetamide (DMA). The base of choice is generally K2CO3, although an example has been reported under base-free conditions. The reactions are typically conducted at temperatures between 90 and 100 °C.
Complexes 28a–c, 29a, 29b, and 30 efficiently catalyzed Heck-type coupling reactions using aryl bromide substrates, whereas complex 26 was active with aryl iodides at 90 °C. The corresponding turnover numbers (TONs) were 46 (26), 40 (28a), 90 (28b), 85 (28c), 41.5 (28a), 34.5 (29a), 33.5 (29b), and 100 (30), respectively. As expected, the use of aryl chlorides remains challenging. However, complex 30 was capable of catalyzing reactions involving aryl chlorides bearing electron-withdrawing groups (EWGs), which facilitate C–Cl bond activation. For example, the cross-coupling of methyl acrylate with 1-iodo-4-nitrobenzene afforded the Heck-type product in 20% yield, while the corresponding bromide or iodide substrates produced the product in quantitative yield (100%). In contrast, substrates containing electron-donating groups (EDGs) negatively affected the reaction efficiency; in the case of 4-bromoanisole catalyzed by complex 30, the yield decreased to 25%.
Complex 32a efficiently catalyzed the Heck-type reaction using aryl diazonium salts as substrates at room temperature, achieving a TON of 109. This methodology exhibited broad olefin tolerance, accommodating a wide variety of substituents such as –CO2Me, –CO2Et, –CO2nBu, –CONH2, –Ph, p-methoxyphenyl, and p-chlorophenyl groups.
Complexes 28a–c efficiently catalyzed the coupling of 1-methyl-2-pyrrolecarboxaldehyde and 3,5-dimethylisoxazole with a variety of aryl bromides.137 The reactions were carried out under conditions analogous to those employed in Heck-type couplings, using 1 mol% of catalyst, potassium acetate as the base, and DMA as the solvent at 120 °C for 4 h (Scheme 18).
 | ||
| Scheme 18 Cross-coupling of 1-methyl-2-pyrrolecarboxaldehyde and 3,5-dimethylisoxazole with aryl bromides catalyzed by NHC–Pd complexes 28a–c. | ||
When 1-methyl-2-pyrrolecarboxaldehyde was used as the substrate, the turnover numbers (TONs) reached 65 for 28a, 94 for 28b, and 85 for 28c. In contrast, reactions with 3,5-dimethylisoxazole afforded slightly lower TON values of 46 (28a), 61 (28b), and 69 (28c). The reaction conditions exhibited good functional-group tolerance, accommodating aldehyde (CHO), ketone, and methoxy (OMe) substituents without noticeable deactivation.
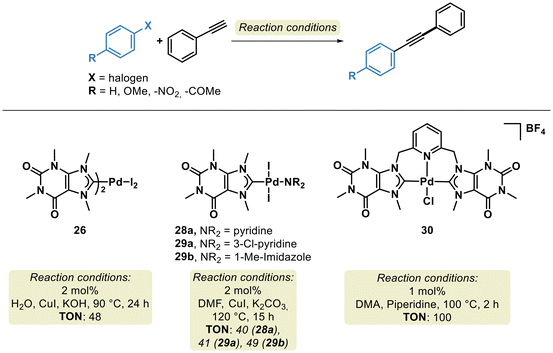
Xanthine-based Pd–NHC complexes also exhibit catalytic activity in the Sonogashira cross-coupling reaction. Complexes 26, 28a, 29a, 29b and 30 effectively catalyzed the transformation (Scheme 19) at temperatures between 90 and 100 °C.130,133,135 The TONs obtained for the different catalysts were 48 (26), 40 (28a), 41 (29a), 49 (29b), and 100 (30). As expected, pincer complexes are well-recognized for their high catalytic robustness and stability, which explains the superior performance of complex 30 in this transformation.
A series of caffeine-derived Pd–NHC catalysts were evaluated in the cyanation of aryl bromides (Scheme 20).138 Preliminary studies revealed that the zwitterionic complexes (33c, 33e, and 33g) were significantly more active, affording up to 99% yield after 4 h, compared to their chelated counterparts (33a, 33b, and 33d), which produced up to 50% yield. This enhanced reactivity is presumably due to the anionic substituent in the zwitterionic species, which improves the solubility of the active Pd–NHC intermediates in the aqueous reaction medium. Further exploration of the reaction scope using complexes 33f and 33g as catalysts afforded a broad range of products, with yields of 12–64% (33f) and 16–74% (33g) and corresponding TON values of 64 and 74, respectively, under similar conditions.
On the other hand, bis(pNHC) Pd(II) complexes can also be obtained.139 These species exhibit a dynamic equilibrium between the bis(pNHC) complex (34) and the ‘theophyllinate’ complex (35), in which the theophylline ligand coordinates through the N9 atom. In this system, the pNHC form corresponds to the kinetic product, whereas the ‘theophyllinate’ species represents the thermodynamic product, as illustrated in Scheme 21. This equilibrium is solvent-dependent: the ‘theophyllinate’ complex is favored in poorly coordinating solvents (e.g., CDCl3), where the carbene species is almost undetectable by 13C NMR spectroscopy. Conversely, in strongly coordinating solvents such as DMSO-d6, the NHC form is stabilized and can be clearly identified spectroscopically.
Purine-based NHC complexes of platinum have also been synthesized.105,140–142 However, to the best of our knowledge, no catalytic applications have been reported to date. Instead, these Pt–NHC systems have primarily been explored for their biological properties, including potential anticancer activity.67,140,142
5. Purine-based NHC complexes of Group 11 metals
The chemistry of Group 11 metals (Cu, Ag, and Au) is highly distinctive for each element. Copper, one of the most abundant transition metals in the Earth's crust, occurs mainly in mineral ores and is commonly commercialized as sulfate, carbonate, halide, or oxide salts. These compounds are bench-stable, inexpensive, and readily available—qualities that make copper an attractive metal feedstock for catalyst preparation and other industrial applications.118,143,144Cu–NHC complexes are particularly well established in σ-/π-activation reactions involving molecules with multiple bonds (e.g., olefins, alkynes, NH3, and CO2), typically enabling functionalization and heteroatom incorporation, or even C–H bond activation. A notable example is the copper-catalyzed alkyne–azide cycloaddition (“click reaction”), which affords 1,4-disubstituted 1,2,3-triazoles with complete atom economy.25,145–150
In contrast, silver and gold are far less abundant and are classified as precious transition metals. Although their catalytic applications have been known for decades, the last two to three decades have witnessed a resurgence of interest, particularly due to their ability to act as mild Lewis acid catalysts.151–156 The following sections summarize the coordination chemistry, catalytic behavior, and reactivity trends of Group 11 metal complexes bearing purine-derived NHC ligands.
The theophylline-based Cu(I)–NHC complex (36), incorporating a hydrophilic ammonium salt unit, was evaluated as a catalyst for regioselective alkyne–azide cycloaddition between alkynes and alkyl halides in aqueous medium (Scheme 22).64 Under these conditions, a representative library of 1,4-disubstituted 1,2,3-triazoles was obtained in yields of up to 93%.
The catalytic activity of complex 36 was also examined in the Glaser homocoupling of alkynes (Scheme 23),64 affording 1,3-diynes in moderate to good yields (73–92%). The catalyst exhibited good substrate tolerance, efficiently converting diversely substituted alkynes, although electron-donating substituents appeared to enhance reaction efficiency. Recyclability experiments revealed certain limitations in catalyst recovery and reuse; however, the use of a catalyst based on inexpensive copper and a naturally occurring theophylline-derived NHC ligand represents a valuable advance toward sustainable, water-mediated Cu catalysis.
The cellulose/alumina (C/A)-supported Cu complex (37), derived from a caffeine-based NHC ligand, efficiently promoted the Ullmann-type coupling for the synthesis of diaryl ethers (Scheme 24).157 The reaction employed either phenyliodide or phenylbromide as aryl halide partners, together with a library of phenols bearing substituents of varying electronic and steric characteristics. When phenyliodide was used as the substrate, the reaction afforded slightly higher yields (71–90%) compared to the bromide analogues (64–83%). Notably, the presence of electron-withdrawing substituents (e.g., R = NO2) had a negative effect on the reaction outcome, resulting in lower yields (64–69%).
Caffeine-based NHC complexes of silver (38a–c) and gold (39a–c and 40), featuring N-attached hydroxyethyl-substituted wingtips, were evaluated as catalysts in the synthesis of propargylamines (Scheme 25a) and the hydroamination of alkynes (Scheme 25b).158 In the propargylamine synthesis (15–99% yield), the catalytic activity showed a marked dependence on the nature of the aldehyde. For instance, the [Ag(NHC)OAc] complex (38a) afforded 95% yield when acetaldehyde was used as the substrate, whereas the yield dropped sharply to 15% with benzaldehyde, highlighting a strong aromatic substituent effect.
 | ||
| Scheme 25 Synthesis of propargylamines and hydroamination of alkynes catalyzed by Au– and Ag–NHC complexes. | ||
Similarly, the gold acetate analogue (40) provided 91% yield with acetaldehyde, but only 54% yield with benzaldehyde, a trend consistent across a series of Au–NHC complexes. Furthermore, catalyst 40 was successfully applied to the hydroamination of alkynes, producing four different imine derivatives in moderate yields (39–73%). These results emphasize the Lewis-acidic character of Ag(I) and Au(I) centers, which plays a crucial role in activating π-bonds and facilitating nucleophilic addition processes in such transformations.
Up to this point, we have discussed reactions catalyzed by Ag(I) in which the metal fragment acts primarily as a Lewis acid. More challenging catalytic transformations, where Ag participates in oxidative addition steps, have been reported for the caffeine-derived complex 41. The polymer-supported (PS) complex 41 was evaluated in the Sonogashira coupling between terminal alkynes and aryl halides (Scheme 26).159 The reaction scope revealed a representative library of non-symmetrical internal alkynes bearing various (hetero)aryl substituents, affording yields ranging from 54% to 92%. No clear trends related to electronic or steric effects were observed—for instance, R = H (63% yield), p-CHO (69%), p-OH (58%), and o-CHO (54%). This study represents one of the few examples in which oxidative addition at Ag(I) is achieved without the assistance of multidentate or fluorinated ligands,160–174 underscoring the unique reactivity of the caffeine-based Ag–NHC system.
 | ||
Scheme 26 Sonogashira reaction catalyzed by caffeine-based NHC complexes of Au. a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Iodides. b Iodides. b![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Bromides. c Bromides. c![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Chlorides. Chlorides. | ||
Collectively, these examples underscore that purine-derived coinage–metal NHC complexes can display reactivity that goes beyond simple Lewis-acid activation. In this context, the gold system below illustrates how Au–NHC connectivity may undergo slow, solvent-dependent reorganization under basic conditions. In the preparation of the NHC–Au complex (42), prolonged stirring of the bis-NHC proligand with [AuCl(SMe2)] and K2CO3 in acetonitrile led to the formation of complex 43 (Scheme 27).175 The synthesis of complex 42 proceeds at room temperature within 3 h, whereas the subsequent transformation into complex 43 requires 72 h for completion. The initial step corresponds to a ligand-transfer reaction, while the formation of 43 involves partial cleavage and reformation of the Au–NHC bond. This process appears to be solvent-dependent, as the coordinating ability of acetonitrile likely facilitates reorganization within the metal coordination sphere over time.
A key reactivity feature of NHC–Ag complexes lies in the lability of the C(NHC)–Ag bond, which can readily dissociate in solution to establish dynamic equilibria among various silver species, the counteranion (e.g., Cl− and OAc−), the free NHC ligand, and other [NHC–Ag]+n fragments. This ligand-transfer reactivity is widely exploited as a synthetic route to other NHC–metal complexes, as it avoids the need for strong deprotonating agents, contaminant bases, or the isolation of free carbenes. The irreversible precipitation of AgX serves as the thermodynamic driving force, enabling clean isolation of the desired NHC–metal complex after filtration and solvent removal.176–179
This silver-mediated transmetalation strategy has been widely employed to prepare various NHC complexes, including those derived from xanthine ligands. For instance, rhodium and ruthenium NHC complexes (44 and 1a) were synthesized via a one-pot, two-step procedure involving the in situ activation of the corresponding xanthium triflate or iodide salts with [Ag2O], followed by transmetallation of the in situ-generated silver NHC intermediates with [RhCl(COD)]2 or [RuCl2(p-cymene)]2, respectively, to afford the final NHC–Rh and NHC–Ru complexes (44 and 1a) (Scheme 28a and b).79,180,181 Notably, omitting the isolation of the intermediate silver NHC complex and directly introducing the target metal precursor into the reaction mixture proved equally effective, confirming the efficiency and general applicability of this ligand-transfer methodology.
The reaction of equimolar amounts of the NHC–Ag complex (45) and [Rh(COD)Cl]2 in DMSO afforded the corresponding cationic NHC–Rh complex (46) via a typical ligand-transfer process from silver (Scheme 28c). Interestingly, the presence of PF6− as the counterion in complex 45 leads to the formation of [AgPF6], which can act as a halide scavenger in the presence of coordinating solvents. Complex 45 is air-stable up to its melting point. In the 13C NMR spectrum, the carbene carbon resonates as a doublet at 188.46 ppm (1JC–Rh = 43.8 Hz).181
Silver–NHC complexes readily undergo ligand transfer to palladium, providing a versatile and reliable route for the synthesis of NHC-Pd derivatives.
Scheme 29 summarizes several complexes obtained through this strategy.182–184 The nature of the product depends strongly on the reaction conditions. When the reaction of [Ag(NHC)2]X with a palladium precursor (e.g., [PdCl(η3-allyl)]2, [(Me-Py-CH2SPh)Pd(η2-olefin)], or [PdC4(COOMe)]n) is performed in the presence of a neutral ligand (L)—such as phosphines or arsines—the resulting heteroleptic complex contains both the NHC and the neutral ligand. In contrast, under ligand-free conditions, bis(NHC)-Pd species are preferentially formed. The palladium complexes displayed significant cytotoxic activity toward selected cancer cell lines, suggesting potential for further bioorganometallic exploration.
 | ||
| Scheme 29 Influence of neutral ligand addition on the formation of NHC-Pd(II) complexes, highlighting the generation of heteroleptic versus bis(NHC) species. | ||
For instance, the reaction of [Ag(NHC)2]BF4 with [PdCl(η3-allyl)]2 in the presence of two equivalents of triphenylphosphine afforded the heteroleptic complex (47). During this process, [AgBF4] acts as a halide scavenger, removing the chloride ligand coordinated to palladium and generating a vacant coordination site that accommodates the phosphine ligand. Conversely, when the reaction is carried out in the absence of PPh3, a cationic bis(NHC)–Pd complex (48) is obtained. The addition of KI effectively removes residual silver from the reaction mixture, and the resulting cationic complexes are stable in air and in solution.
Similarly, cationic [Ag(NHC)2]AgCl2 reacts with equivalent amounts of the polymeric precursor [PdC4(COOMe)]n in the presence of triphenylphosphine to afford heteroleptic complexes (49a–c) in up to 85% yield. In the absence of triphenylphosphine, bis(NHC)–Pd complexes are formed in moderate to good yields (71–90%), provided that thermodynamic and kinetic parameters are carefully controlled to prevent the formation of bis(PPh3)-Pd or mixed-product distributions.
In a related approach, a library of NHC–Pd complexes (52a–h) was prepared by reacting equimolar amounts of [Ag(NHC)2]AgCl2 and PPh3 (or AsPh3) with [(Me-Py-CH2SPh)Pd(η2-olefin)] in CH2Cl2, where AgCl precipitation indicated reaction progress. This strategy takes advantage of the lability of the 2-methyl-6-((phenylthio)methyl)pyridine ligand, which is prone to dissociation due to chelate-ring distortion. The resulting heteroleptic complexes (52a–h) were isolated by ether precipitation or solvent evaporation, whereas bis(NHC)–Pd complexes (51) were obtained under ligand-free conditions.
Using [Ag(NHC)2]I (53) as a ligand-transfer reagent, the functionalized NHC–Pt complex (55)—bearing an all-nitrogen, methylated caffeine core ligand—was obtained indirectly via the intermediate formation of the NHC–Pt(dvtms) complex (54) (Scheme 30).140 The reaction between complex 53 and [Pt2(dvtms)3] first afforded complex 54, which subsequently underwent oxidative addition of I2 in the presence of the coordinating amine ligand CyNH2, leading to complex 27 in 39% isolated yield on a gram scale (up to 2 g).
When complex 56 was dissolved in ethanol, methanol, or water/ethyl acetate mixtures and the solvent was allowed to slowly evaporate, the [Ag(NHC)OAc] complex (57) was obtained (Scheme 31).185 Complex 57 is a water-soluble, light-stable solid, consistent with its formulation as an acetate derivative. Further 13C NMR analysis showed a carbene carbon resonance at 186.2 ppm, along with carbonyl and methyl signals from the acetate group at 176.2 and 23.1 ppm, respectively, confirming the formation of the expected [Ag(NHC)OAc] complex (57).
6. Future outlook
Throughout this review, we have highlighted the growing importance of purine-derived NHC ligands as a versatile class of scaffolds for the stabilization of transition-metal centers and the promotion of a wide range of catalytic transformations. The complexes reported so far encompass Cu, Ni, Pd, Ag, Au, Rh, Ru, Ir, and Pt, representing nearly every major block of transition-metal catalysis. Collectively, these systems demonstrate how the unique structural and electronic characteristics of the purine nucleus—particularly its multiple heteroatom donors, accessible protonation sites, and distinct C–H activation potential—can be strategically harnessed to modulate metal–ligand bonding, reactivity, and selectivity.From a synthetic perspective, the xanthine, theophylline, and caffeine backbones have proven to be the most accessible and tunable frameworks for generating purine-based NHCs. Their straightforward N-alkylation chemistry enables rapid post-functionalization to adjust steric and electronic properties, while the use of Ag-mediated transmetalation routes allows mild and selective access to a broad spectrum of M–NHC complexes without resorting to free carbene intermediates. Further advances in polymer- and graphene-supported systems highlight the compatibility of purine NHCs with heterogeneous catalysis, offering promising opportunities for catalyst recovery and recyclability, in line with green-chemistry principles.
Mechanistically, purine-derived NHC complexes reveal a striking range of behaviors across the periodic table. Group 8–10 metals (Fe, Ru, Os, Co, Rh, Ir, Ni, Pd, and Pt) typically engage in oxidative-addition/reductive-elimination cycles, benefitting from the strong σ-donor ability and tunable π-acceptor capacity of the purine carbene center. In particular, Ir(III) and Ru(II) complexes display rich C–H activation chemistry, accessible mer/fac isomerism, and notable photophysical properties, suggesting avenues toward photo-responsive or luminescent catalysts. In contrast, Group 11 metals (Cu, Ag, and Au) illustrate the complementary σ/π-activation and Lewis-acid pathways characteristic of late-d10 systems: Cu–NHC complexes excel in atom-economical C–N, C–O, and “click” transformations under mild conditions, whereas Ag– and Au–NHC complexes operate as soft Lewis acids in hydroamination and propargylamine synthesis, with Au uniquely capable of oxidative addition even in the absence of hemilabile co-ligands or external oxidants.
Looking ahead, the integration of purine-based NHCs into catalytic, photochemical, and biological contexts is poised to expand dramatically. The inherent biocompatibility and modular structure of purine ligands make them attractive not only for homogeneous catalysis but also for bio-organometallic and medicinal applications, particularly when combined with Pt, Au, and Ru centers. Likewise, the development of Earth-abundant metal complexes (e.g., Fe, Ni, Cu, and Co) featuring purine-derived NHCs will be central to advancing sustainable catalysis and C–H bond functionalization using renewable feedstocks.
In summary, purine-derived NHC ligands stand at the intersection of organic synthesis, coordination chemistry, and materials science, offering a rich platform for multifunctional catalyst design. Their electronic anisotropy, facile derivatization, and compatibility with diverse metals and supports ensure that future efforts will continue to uncover new reactivity paradigms, deepen our understanding of metal–carbene bonding, and foster environmentally responsible transformations driven by rational ligand design.
Author contributions
Conceptualization: D. M.-M., A. C.-R. and H. V.; writing – original draft preparation: A. C.-R. and H. V.; writing – review and editing: D. M.-M., A. C.-R., R. M.-C. and H. V.; visualization: D. M.-M., A. C.-R., R. M.-C. and H. V.; supervision: D. M.-M., A. C.-R. and H. V.; project administration: A. C.-R., H. V. and D. M.-M.; and funding acquisition: D. M.-M and H. V. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
The authors declare that they have no conflicts of interest.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This research was supported by DGAPA-PAPIIT-UNAM and CONACYT. A. C.-R. thankfully acknowledges DGAPA/UNAM for a postdoctoral fellowship. R. M.-C. gratefully acknowledges the postgraduate fellowship provided by SECITHI with CVU 1079016. H. V. acknowledges financial support from the Universidad de Alcalá (PIUAH25/CC-074).References
- H. W. Wanzlick, Angew. Chem., Int. Ed. Engl., 1962, 1, 75–80 CrossRef.
- H. W. Wanzlick and H. J. Schönherr, Angew. Chem., Int. Ed. Engl., 1968, 7, 141–142 CrossRef CAS.
- A. J. Arduengo III, R. L. Harlow and M. Kline, J. Am. Chem. Soc., 1991, 113, 361–363 CrossRef.
- A. J. Arduengo III, M. Kline, J. C. Calabrese and F. Davidson, J. Am. Chem. Soc., 1991, 113, 9704–9705 CrossRef.
- M. N. Hopkinson, C. Richter, M. Schedler and F. Glorius, Nature, 2014, 510, 485–496 CrossRef CAS PubMed.
- E. Peris, Chem. Rev., 2018, 118, 9988–10031 CrossRef CAS PubMed.
- P. de Frémont, N. Marion and S. P. Nolan, Coord. Chem. Rev., 2009, 253, 862–892 CrossRef.
- W. A. Herrmann, M. Elison, J. Fischer, C. Köcher and G. R. J. Artus, Angew. Chem., Int. Ed. Engl., 1995, 34, 2371–2374 CrossRef CAS.
- W. A. Herrmann, Angew. Chem., Int. Ed., 2002, 41, 1290–1309 CrossRef CAS PubMed.
- S. Budagumpi, R. S. Keri, G. Achar and K. N. Brinda, Adv. Synth. Catal., 2020, 362, 970–997 CrossRef CAS.
- A. Cervantes-Reyes, F. Rominger, M. Rudolph and A. S. K. Hashmi, Adv. Synth. Catal., 2020, 362, 2523–2533 CrossRef CAS.
- H. Clavier and S. P. Nolan, Chem. Commun., 2010, 46, 841–861 RSC.
- A. Cervantes-Reyes, F. Rominger, M. Rudolph and A. S. K. Hashmi, Chem. – Eur. J., 2019, 25, 11745–11757 CrossRef CAS PubMed.
- E. L. Kolychev, A. F. Asachenko, P. B. Dzhevakov, A. A. Bush, V. V. Shuntikov, V. N. Khrustalev and M. S. Nechaev, Dalton Trans., 2013, 42, 6859–6866 RSC.
- C. A. Urbina-Blanco, A. Leitgeb, C. Slugovc, X. Bantreil, H. Clavier, A. M. Z. Slawin and S. P. Nolan, Chem. – Eur. J., 2011, 17, 5045–5053 CrossRef CAS PubMed.
- A. Cervantes-Reyes, F. Rominger and A. S. K. Hashmi, Chem. – Eur. J., 2020, 26, 5530–5540 CrossRef CAS PubMed.
- H. V. Huynh, Y. Han, R. Jothibasu and J. A. Yang, Organometallics, 2009, 28, 5395–5404 CrossRef CAS.
- D. J. Nelson and S. P. Nolan, Chem. Soc. Rev., 2013, 42, 6723–6753 RSC.
- H. V. Huynh, Chem. Rev., 2018, 118, 9457–9492 CrossRef CAS PubMed.
- S. S. Bera, G. Utecht-Jarzyńska, S. Yang, S. P. Nolan and M. Szostak, Chem. Rev., 2025, 125, 5349–5435 CrossRef CAS PubMed.
- R. O. Pankov, D. O. Prima and V. P. Ananikov, Coord. Chem. Rev., 2024, 516, 215897 CrossRef CAS.
- A. Neshat, P. Mastrorilli and A. Mousavizadeh Mobarakeh, Molecules, 2022, 27, 95 CrossRef CAS PubMed.
- C. Costabile, S. Pragliola and F. Grisi, Symmetry, 2022, 14, 1615 CrossRef CAS.
- T. Zhou, G. Utecht-Jarzyńska and M. Szostak, Coord. Chem. Rev., 2024, 512, 215867 CrossRef CAS PubMed.
- A. Cervantes-Reyes, K. Farshadfar, M. Rudolph, F. Rominger, T. Schaub, A. Ariafard and A. S. K. Hashmi, Green Chem., 2021, 23, 889–897 RSC.
- V. Dragutan, I. Dragutan, L. Delaude and A. Demonceau, Coord. Chem. Rev., 2007, 251, 765–794 CrossRef CAS.
- A. Kumari, Sweet Biochemistry. Remembering Structures, Cycles, and Pathways by Mnemonics, Academic Press, 2018 Search PubMed.
- M. Legraverend, Tetrahedron, 2008, 64, 8585–8603 CrossRef CAS.
- T. Zhou, P. Gao, R. Lalancette, R. Szostak and M. Szostak, Nat. Chem., 2024, 16, 2025–2035 CrossRef CAS PubMed.
- L.Á Turcio-García, H. Valdés, S. Hernández-Ortega, D. Canseco-Gonzalez and D. Morales-Morales, New J. Chem., 2022, 46, 16789–16800 RSC.
- L.Á Turcio-García, H. Valdés, A. Arenaza-Corona, S. Hernández-Ortega and D. Morales-Morales, New J. Chem., 2023, 47, 2090–2095 RSC.
- L.Á Turcio-García, R. Parra-Unda, H. Valdés, S. Hernández-Ortega, G. G. Valenzuela-Ramirez, Y. P. Ahumada-Santos, Y. Sánchez-Lugo, V. Reyes-Márquez and D. Morales-Morales, in Pharmaceutics, 2025, pp. 973 Search PubMed.
- S. C. Scott, J. A. Cadge, G. K. Boden, J. F. Bower and C. A. Russell, Angew. Chem., Int. Ed., 2023, 62, e202301526 CrossRef CAS PubMed.
- P. Gao, J. Xu, T. Zhou, Y. Liu, E. Bisz, B. Dziuk, R. Lalancette, R. Szostak, D. Zhang and M. Szostak, Angew. Chem., Int. Ed., 2023, 62, e202218427 CrossRef CAS PubMed.
- G. Galdi and C. Costabile, Chem. – Eur. J., 2024, 30, e202402774 CrossRef CAS PubMed.
- M. Alcarazo, T. Stork, A. Anoop, W. Thiel and A. Fürstner, Angew. Chem., Int. Ed., 2010, 49, 2542–2546 CrossRef CAS PubMed.
- T. Scattolin, G. Andreetta, M. Mauceri, F. Rizzolio, N. Demitri, V. Canzonieri and F. Visentin, J. Organomet. Chem., 2021, 952, 122014 CrossRef CAS.
- C. Stoll, C. Besnard and C. Mazet, JACS Au, 2025, 5, 4681–4687 CrossRef CAS PubMed.
- T. Zhou, C. Zhao, S. Yang, E. Bisz, B. Dziuk, R. Lalancette, R. Szostak, X. Hong and M. Szostak, ACS Catal., 2025, 15, 13846–13859 CrossRef CAS PubMed.
- T. Sheikh Mohammad, P. Sakharov, S. Raje and G. de Ruiter, ACS Catal., 2025, 15, 5370–5377 CrossRef CAS.
- T. S. Mohammad, Y. Jin, S. Raje, K. Młodzikowska-Pieńko, Z.-X. Yu and G. de Ruiter, J. Am. Chem. Soc., 2025, 147, 15195–15204 CrossRef CAS PubMed.
- K. Azouzi, R. Pointis, R. Buhaibeh, P. H. Fernández, L. Pedussaut, M. Boundor, A. Bonfiglio, A. Bruneau-Voisine, D. Wei, T. Roisnel, C. Duhayon, M.Á Casado, D. A. Valyaev, Y. Canac, S. Bastin, C. Raynaud and J.-B. Sortais, J. Catal., 2024, 430, 115334 CrossRef CAS.
- J.-P. Chang, Y.-W. Zhang, L.-Y. Sun, L. Zhang, F. E. Hahn and Y.-F. Han, Angew. Chem., Int. Ed., 2024, 63, e202409664 CrossRef CAS PubMed.
- Y.-W. Zhang, Y. Lu, L.-Y. Sun, P. D. Dutschke, M.-M. Gan, L. Zhang, A. Hepp, Y.-F. Han and F. E. Hahn, Angew. Chem., Int. Ed., 2023, 62, e202312323 CrossRef CAS PubMed.
- R. Kumar, M. K. Pandey, A. Bhandari and J. Choudhury, ACS Catal., 2023, 13, 4824–4834 CrossRef CAS.
- V. K. Rawat, K. Higashida and M. Sawamura, ACS Catal., 2022, 12, 8325–8330 CrossRef CAS.
- S. Tang, Y. Zhao and K. Nozaki, J. Am. Chem. Soc., 2021, 143, 17953–17957 CrossRef CAS PubMed.
- S. Garhwal, A. Kaushansky, N. Fridman and G. de Ruiter, Chem Catal., 2021, 1, 631–647 CAS.
- S. Garhwal, A. Kaushansky, N. Fridman, L. J. W. Shimon and G. d. Ruiter, J. Am. Chem. Soc., 2020, 142, 17131–17139 CrossRef CAS PubMed.
- S. Byun, S. Park, Y. Choi, J. Y. Ryu, J. Lee, J.-H. Choi and S. Hong, ACS Catal., 2020, 10, 10592–10601 CrossRef CAS.
- Y.-W. Zhang, S. Bai, Y.-Y. Wang and Y.-F. Han, J. Am. Chem. Soc., 2020, 142, 13614–13621 CrossRef CAS PubMed.
- L. Li, F. Han, X. Nie, Y. Hong, S. Ivlev and E. Meggers, Angew. Chem., Int. Ed., 2020, 59, 12392–12395 CrossRef CAS PubMed.
- D.-A. Park, S. Byun, J. Y. Ryu, J. Lee, J. Lee and S. Hong, ACS Catal., 2020, 10, 5443–5453 CrossRef CAS.
- Y. Tang, I. Benaissa, M. Huynh, L. Vendier, N. Lugan, S. Bastin, P. Belmont, V. César and V. Michelet, Angew. Chem., Int. Ed., 2019, 58, 7977–7981 CrossRef CAS PubMed.
- C. B. Schwamb, K. P. Fitzpatrick, A. C. Brueckner, H. C. Richardson, P. H. Y. Cheong and K. A. Scheidt, J. Am. Chem. Soc., 2018, 140, 10644–10648 CrossRef CAS PubMed.
- H. Yasuda, R. Nakano, S. Ito and K. Nozaki, J. Am. Chem. Soc., 2018, 140, 1876–1883 CrossRef CAS PubMed.
- R. Nakano and K. Nozaki, J. Am. Chem. Soc., 2015, 137, 10934–10937 CrossRef CAS PubMed.
- C. T. Check, K. P. Jang, C. B. Schwamb, A. S. Wong, M. H. Wang and K. A. Scheidt, Angew. Chem., Int. Ed., 2015, 54, 4264–4268 CrossRef CAS PubMed.
- A. Schmidt, N. Grover, T. K. Zimmermann, L. Graser, M. Cokoja, A. Pöthig and F. E. Kühn, J. Catal., 2014, 319, 119–126 CrossRef CAS.
- M. Alcarazo, S. J. Roseblade, A. R. Cowley, R. Fernández, J. M. Brown and J. M. Lassaletta, J. Am. Chem. Soc., 2005, 127, 3290–3291 CrossRef CAS PubMed.
- H. Ashihara, K. Mizuno, T. Yokota and A. Crozier, Xanthine Alkaloids: Occurrence, Biosynthesis, and Function in Plants, in Progress in the Chemistry of Organic Natural Products 105, ed. A. D. Kinghorn, H. Falk, S. Gibbons and J. i. Kobayashi, Springer International Publishing, Cham, 2017, pp. 1–88 Search PubMed.
- A. Chaudhary, D. Mathur, R. Gaba, R. Pasricha and K. Sharma, RSC Adv., 2024, 14, 8932–8962 RSC.
- F. P. L. Lim and A. V. Dolzhenko, Eur. J. Med. Chem., 2014, 85, 371–390 CrossRef CAS PubMed.
- A. Szadkowska, S. Staszko, E. Zaorska and R. Pawłowski, RSC Adv., 2016, 6, 44248–44253 RSC.
- N. U. D. Reshi and J. K. Bera, Coord. Chem. Rev., 2020, 422, 213334 CrossRef CAS.
- A. Sánchez-Mora, L. González-Sebastián, J. A. Cruz-Navarro, A. Arenaza-Corona, T. Ramírez-Apan, A. Nieto-Camacho and D. Morales-Morales, New J. Chem., 2025, 49, 18258–18269 RSC.
- M. O. Dar, R. H. Mir, R. Mohiuddin, M. H. Masoodi and F. A. Sofi, J. Inorg. Biochem., 2023, 246, 112290 CrossRef CAS PubMed.
- G. Francescato, M. I. P. S. Leitão, G. Orsini and A. Petronilho, ChemMedChem, 2024, 19, e202400118 CrossRef CAS PubMed.
- H. Valdés, D. Canseco-González, J. M. Germán-Acacio and D. Morales-Morales, J. Organomet. Chem., 2018, 867, 51–54 CrossRef.
- S. A. J. Almahmoud, M. Cariello and A. O. Elzupir, Orient. J. Chem., 2023, 39, 222–230 CAS.
- H. Song and H.-B. Kraatz, Inorg. Chim. Acta, 2009, 362, 1365–1368 CrossRef CAS.
- M. Campitiello, A. Cremonini, M. A. Squillaci, S. Pieraccini, A. Ciesielski, P. Samorì and S. Masiero, J. Org. Chem., 2021, 86, 9970–9978 CrossRef CAS PubMed.
- N. J. Forrow, N. C. Foulds, J. E. Frew and J. T. Law, Bioconjugate Chem., 2004, 15, 137–144 CrossRef CAS PubMed.
- C. M. Mikulski, M. L. Bayne, S. Grossman, M. Gaul, A. Renn, D. L. Staley and N. M. Karayannis, J. Coord. Chem., 1989, 20, 185–191 CrossRef CAS.
- C. M. Mikulski, S. Grossman, M. L. Bayne, M. Gaul, D. Kanach, K. Udell and N. M. Karayannis, J. Coord. Chem., 1990, 22, 175–182 CrossRef CAS.
- C. M. Mikulski, S. Grossman, M. L. Bayne, M. Gaul, D. Kanach, K. Udell and N. M. Karayannis, Inorg. Chim. Acta, 1990, 173, 31–35 CrossRef CAS.
- M. M. Alam, M. J. Mia, M. A. A. Mamun, S. S. Rakhi, K. A. Azam and S. Ghosh, J. Organomet. Chem., 2021, 944, 121791 CrossRef CAS.
- K. Sayin and A. Üngördü, Spectrochim. Acta, Part A, 2018, 193, 147–155 CrossRef CAS PubMed.
- F. Mazars and L. Delaude, Organometallics, 2023, 42, 1589–1597 CrossRef CAS.
- J. Canivet and G. Süss-Fink, Green Chem., 2007, 9, 391–397 RSC.
- J. Gunasekaran, S. Muthuselvan, D. Annadurai, D. B. Christopher Leslee, N. Venkatesan, S. Murthy, B. Ramasamy, L. G. Alves, A. M. Martins and S. B. Kuppannan, J. Mol. Struct., 2024, 1309, 138179 CrossRef CAS.
- F. Pirani and H. Eshghi, Inorg. Chem. Commun., 2025, 181, 115297 CrossRef CAS.
- J. Liquier-Milward, Nature, 1951, 167, 1068–1069 CrossRef CAS PubMed.
- A. Singh, S. K. Maiti, H. P. Gogoi and P. Barman, Polyhedron, 2023, 230, 116244 CrossRef CAS.
- S. D. K. Alzamili, Res. J. Chem. Environ., 2022, 26, 129–141 CAS.
- H.-K. Zhao, H.-W. Yang, X.-G. Wang, B. Ding, Z.-Y. Liu, X.-J. Zhao and E.-C. Yang, Inorg. Chem. Commun., 2019, 107, 107483 CrossRef CAS.
- H. E. Hamdani, M. E. Amane and C. Duhayon, J. Mol. Struct., 2019, 1190, 125–134 CrossRef CAS.
- H. E. Hamdani and M. E. Amane, J. Mol. Struct., 2019, 1184, 262–270 CrossRef CAS.
- Ö. Altun and M. Şuözer, J. Mol. Struct., 2017, 1149, 307–314 CrossRef.
- S. S. Bera and M. Szostak, ACS Catal., 2022, 12, 3111–3137 CrossRef CAS PubMed.
- I. Eslava-Gonzalez, H. Valdés, M. Teresa Ramírez-Apan, S. Hernandez-Ortega, M. Rosario Zermeño-Ortega, A. Avila-Sorrosa and D. Morales-Morales, Inorg. Chim. Acta, 2020, 507, 119588 CrossRef CAS.
- Q. Zhao, G. Meng, S. P. Nolan and M. Szostak, Chem. Rev., 2020, 120, 1981–2048 CrossRef CAS PubMed.
- H. Buhl and C. Ganter, J. Organomet. Chem., 2016, 809, 74–78 CrossRef CAS.
- P. V. Simpson, K. Radacki, H. Braunschweig and U. Schatzschneider, J. Organomet. Chem., 2015, 782, 116–123 CrossRef CAS.
- D. G. Gusev and E. Peris, Dalton Trans., 2013, 42, 7359–7364 RSC.
- A. R. Chianese, X. Li, M. C. Janzen, J. W. Faller and R. H. Crabtree, Organometallics, 2003, 22, 1663–1667 CrossRef CAS.
- R. A. Kelly III, H. Clavier, S. Giudice, N. M. Scott, E. D. Stevens, J. Bordner, I. Samardjiev, C. D. Hoff, L. Cavallo and S. P. Nolan, Organometallics, 2008, 27, 202–210 CrossRef.
- B. Joshi and M. Shivashankar, ACS Omega, 2023, 8, 43408–43432 CrossRef CAS PubMed.
- A. Sebris, M. Guzauskas, M. Mahmoudi, D. Volyniuk, J. V. Grazulevicius, A. Mishnev, I. Novosjolova, M. Turks, G. Jonusauskas and K. Traskovskis, J. Mater. Chem. C, 2023, 11, 14608–14620 RSC.
- M. Bevilacqua, V. Giuso, M. Rancan, L. Armelao, C. Graiff, W. Baratta, V. Di Marco and A. Biffis, Eur. J. Inorg. Chem., 2022, 2022, e202200484 CrossRef CAS.
- R. N. Osorio Yáñez, A. Hepp, T. T. Y. Tan and F. E. Hahn, Organometallics, 2020, 39, 344–352 CrossRef.
- D. Brackemeyer, C. Schulte to Brinke, F. Roelfes and F. E. Hahn, Dalton Trans., 2017, 46, 4510–4513 RSC.
- S. Kuwata and F. E. Hahn, Chem. Rev., 2018, 118, 9642–9677 CrossRef CAS PubMed.
- S. Cepa, C. Schulte to Brinke, F. Roelfes and F. E. Hahn, Organometallics, 2015, 34, 5454–5460 CrossRef CAS.
- D. Brackemeyer, A. Hervé, C. Schulte to Brinke, M. C. Jahnke and F. E. Hahn, J. Am. Chem. Soc., 2014, 136, 7841–7844 CrossRef CAS PubMed.
- A. Makhloufi, W. Frank and C. Ganter, Organometallics, 2012, 31, 7272–7277 CrossRef CAS.
- J. Schütz and W. A. Herrmann, J. Organomet. Chem., 2004, 689, 2995–2999 CrossRef.
- S. C. Zinner, C. F. Rentzsch, E. Herdtweck, W. A. Herrmann and F. E. Kühn, Dalton Trans., 2009, 7055–7062 RSC.
- C. F. Rentzsch, E. Tosh, W. A. Herrmann and F. E. Kühn, Green Chem., 2009, 11, 1610–1617 RSC.
- M. Bortenschlager, J. Schütz, D. von Preysing, O. Nuyken, W. A. Herrmann and R. Weberskirch, J. Organomet. Chem., 2005, 690, 6233–6237 CrossRef CAS.
- E. A. B. Kantchev, C. J. O'Brien and M. G. Organ, Angew. Chem., Int. Ed., 2007, 46, 2768–2813 CrossRef CAS PubMed.
- A. Kumar, M. Kumar and A. K. Verma, J. Org. Chem., 2020, 85, 13983–13996 CrossRef CAS PubMed.
- S. B. Umabharathi, M. Neetha and G. Anilkumar, Top. Curr. Chem., 2024, 382, 3 CrossRef CAS PubMed.
- P. Ruiz-Castillo and S. L. Buchwald, Chem. Rev., 2016, 116, 12564–12649 CrossRef CAS PubMed.
- N. Selander and K. J. Szabó, Chem. Rev., 2011, 111, 2048–2076 CrossRef CAS PubMed.
- F.-S. Han, Chem. Soc. Rev., 2013, 42, 5270–5298 RSC.
- B. M. Rosen, K. W. Quasdorf, D. A. Wilson, N. Zhang, A.-M. Resmerita, N. K. Garg and V. Percec, Chem. Rev., 2011, 111, 1346–1416 CrossRef CAS PubMed.
- A. A. Danopoulos, T. Simler and P. Braunstein, Chem. Rev., 2019, 119, 3730–3961 CrossRef CAS PubMed.
- E. Rufino-Felipe, H. Valdés and D. Morales-Morales, Eur. J. Org. Chem., 2022, e202200654 CrossRef CAS.
- E. Marín-Carrillo, H. Valdés, S. Hernández-Ortega and D. Morales-Morales, Inorg. Chim. Acta, 2023, 548, 121365 CrossRef.
- S. Díez-González, N. Marion and S. P. Nolan, Chem. Rev., 2009, 109, 3612–3676 CrossRef PubMed.
- V. V. Sharutin and A. R. Zykova, Rev. Adv. Chem., 2023, 13, 111–151 CrossRef CAS.
- A. Jayaraj, A. V. Raveedran, A. T. Latha, D. Priyadarshini and P. C. A. Swamy, Coord. Chem. Rev., 2023, 478, 214922 CrossRef CAS.
- S. Zhao, Z. Yang, G. Jiang, S. Huang, M. Bian, Y. Lu and W. Liu, Coord. Chem. Rev., 2021, 449, 214217 CrossRef CAS.
- J. Zhang, M. M. Rahman, Q. Zhao, J. Feliciano, E. Bisz, B. Dziuk, R. Lalancette, R. Szostak and M. Szostak, Organometallics, 2022, 41, 1806–1815 CrossRef CAS PubMed.
- H. A. Mohamed, B. R. M. Lake, T. Laing, R. M. Phillips and C. E. Willans, Dalton Trans., 2015, 44, 7563–7569 RSC.
- A. Gómez-Suárez, D. J. Nelson and S. P. Nolan, Chem. Commun., 2017, 53, 2650–2660 RSC.
- T. Aalhusaini, D. Pore and G. Rashinkar, J. Organomet. Chem., 2025, 1034, 123637 CrossRef CAS.
- https://www.nobelprize.org/prizes/chemistry/2010/summary/.
- F.-T. Luo and H.-K. Lo, J. Organomet. Chem., 2011, 696, 1262–1265 CrossRef CAS.
- T. Scattolin, L. Canovese, F. Visentin, S. Paganelli, P. Canton and N. Demitri, Appl. Organomet. Chem., 2018, 32, e4034 CrossRef.
- F. Mazars, G. Zaragoza and L. Delaude, J. Organomet. Chem., 2022, 978, 122489 CrossRef CAS.
- M. M. Rahman, J. Zhang, Q. Zhao, J. Feliciano, E. Bisz, B. Dziuk, R. Lalancette, R. Szostak and M. Szostak, Organometallics, 2022, 41, 2281–2290 CrossRef CAS PubMed.
- E. Mohammadi and B. Movassagh, J. Organomet. Chem., 2016, 822, 62–66 CrossRef CAS.
- O. Bysewski, A. Winter and U. S. Schubert, Inorganics, 2023, 11, 164 CrossRef CAS.
- S. Khot, P. Dandge, S. Shikalgar, P. Ghule, D. Pore and G. Rashinkar, J. Organomet. Chem., 2025, 1041, 123831 CrossRef CAS.
- F. Mazars, K. S. Etsè, G. Zaragoza and L. Delaude, J. Organomet. Chem., 2024, 1003, 122928 CrossRef.
- Q. Teng, Y. Zhao, Y. Lu, Z. Liu, H. Chen, D. Yuan, H. V. Huynh and Q. Meng, Organometallics, 2022, 41, 161–168 CrossRef CAS.
- V. R. Landaeta, R. E. Rodríguez-Lugo, E. N. Rodríguez-Arias, D. S. Coll-Gómez and T. González, Transition Met. Chem., 2010, 35, 165–175 CrossRef CAS.
- M. Skander, P. Retailleau, B. Bourrié, L. Schio, P. Mailliet and A. Marinetti, J. Med. Chem., 2010, 53, 2146–2154 CrossRef CAS PubMed.
- J. J. Hu, S.-Q. Bai, H. H. Yeh, D. J. Young, Y. Chi and T. S. A. Hor, Dalton Trans., 2011, 40, 4402–4406 RSC.
- J.-J. Zhang, C.-M. Che and I. Ott, J. Organomet. Chem., 2015, 782, 37–41 CrossRef CAS.
- O. S. Wenger, J. Am. Chem. Soc., 2018, 140, 13522–13533 CrossRef CAS PubMed.
- F. Lazreg, F. Nahra and C. S. J. Cazin, Coord. Chem. Rev., 2015, 293–294, 48–79 CrossRef CAS.
- W.-J. Yoo, T. V. Q. Nguyen and S. Kobayashi, Angew. Chem., Int. Ed., 2014, 53, 10213–10217 CrossRef CAS PubMed.
- M. R. Uehling, A. M. Suess and G. Lalic, J. Am. Chem. Soc., 2015, 137, 1424–1427 CrossRef CAS PubMed.
- F. Sebest, J. J. Dunsford, M. Adams, J. Pivot, P. D. Newman and S. Díez-González, ChemCatChem, 2018, 10, 2041–2045 CrossRef CAS PubMed.
- N. K. Devaraj and M. G. Finn, Chem. Rev., 2021, 121, 6697–6698 CrossRef CAS PubMed.
- M. Meldal and C. W. Tornøe, Chem. Rev., 2008, 108, 2952–3015 CrossRef CAS PubMed.
- Widespread applications of copper-catalyzed click reactions guided profound advances in organic chemistry, drug discovery, biomaterials and agrochemical synthesis, among other research fields, which honored pioneering efforts of Carolyn R. Bertozzi, P. Morten and K. B. Sharpless awarded with the 2022 Nobel Prize in Chemistry, see: https://www.nobelprize.org/prizes/chemistry/2022/summary/.
- R. Dorel and A. M. Echavarren, Chem. Rev., 2015, 115, 9028–9072 CrossRef CAS PubMed.
- A. S. K. Hashmi, Chem. Rev., 2007, 107, 3180–3211 CrossRef CAS PubMed.
- R. P. Herrera and M. C. Gimeno, Chem. Rev., 2021, 121, 8311–8363 CrossRef CAS PubMed.
- E. Jiménez-Núñez and A. M. Echavarren, Chem. Rev., 2008, 108, 3326–3350 CrossRef PubMed.
- Z. Li, C. Brouwer and C. He, Chem. Rev., 2008, 108, 3239–3265 CrossRef CAS PubMed.
- C. Nevado, Chimia, 2010, 64, 247–251 CrossRef CAS PubMed.
- T. N. Aalhusaini, W. N. Al-darkazali, D. Pore and G. Rashinkar, J. Organomet. Chem., 2025, 1039, 123752 CrossRef CAS.
- A. Mariconda, D. Iacopetta, M. Sirignano, J. Ceramella, A. D'Amato, M. Marra, M. Pellegrino, M. S. Sinicropi, S. Aquaro and P. Longo, Int. J. Mol. Sci., 2024, 25, 2599 CrossRef CAS PubMed.
- T. Aalhusaini, D. Pore and G. Rashinkar, J. Organomet. Chem., 2025, 1023, 123406 CrossRef CAS.
- M. Font, F. Acuña-Parés, T. Parella, J. Serra, J. M. Luis, J. Lloret-Fillol, M. Costas and X. Ribas, Nat. Commun., 2014, 5, 4373 CrossRef CAS PubMed.
- L. Demonti, D. Joven-Sancho, M. Baya and N. Nebra, Chem. – Eur. J., 2025, 31, e202501606 CrossRef CAS PubMed.
- E. M. H. Larsen, T. Brock-Nannestad, J. Skibsted, A. Reinholdt and J. Bendix, Chem. Sci., 2024, 15, 18067–18075 RSC.
- L. Demonti, D. Joven-Sancho, N. Saffon-Merceron, M. Baya and N. Nebra, Chem. – Eur. J., 2024, 30, e202400881 CrossRef CAS PubMed.
- L. Demonti, D. Joven-Sancho and N. Nebra, Chem. Rec., 2023, 23, e202300143 CrossRef CAS PubMed.
- L. Demonti, H. Tabikh, N. Saffon-Merceron and N. Nebra, Eur. J. Inorg. Chem., 2023, 26, e202300042 CrossRef CAS.
- C. M. Lemon, D. C. Powers, M. Huynh, A. G. Maher, A. A. Phillips, B. P. Tripet and D. G. Nocera, Inorg. Chem., 2023, 62, 3–17 CrossRef CAS PubMed.
- L. Demonti, N. Saffon-Merceron, N. Mézailles and N. Nebra, Chem. – Eur. J., 2021, 27, 15396–15405 CrossRef CAS PubMed.
- D. Joven-Sancho, M. Baya, L. R. Falvello, A. Martín, J. Orduna and B. Menjón, Chem. – Eur. J., 2021, 27, 12796–12806 CrossRef CAS PubMed.
- M. Deuker, Y. Yang, R. A. J. O'Hair and K. Koszinowski, Organometallics, 2021, 40, 2354–2363 CrossRef CAS.
- X. Ribas, L. Capdevila and P. Font, 6.13 - High-Valent Cu, Ag, and Au Coordination Compounds, in Comprehensive Coordination Chemistry III, ed. E. C. Constable, G. Parkin and L. Que Jr., Elsevier, Oxford, 2021, pp. 474–516 Search PubMed.
- D. Joven-Sancho, M. Baya, A. Martín, J. Orduna and B. Menjón, Chem. – Eur. J., 2020, 26, 4471–4475 CrossRef CAS PubMed.
- L. Capdevila, E. Andris, A. Briš, M. Tarrés, S. Roldán-Gómez, J. Roithová and X. Ribas, ACS Catal., 2018, 8, 10430–10436 CrossRef CAS.
- S. Weske, R. A. Hardin, T. Auth, R. A. J. O'Hair, K. Koszinowski and C. A. Ogle, Chem. Commun., 2018, 54, 5086–5089 RSC.
- M. Font and X. Ribas, Pincerlike Cyclic Systems for Unraveling Fundamental Coinage Metal Redox Processes, in The Privileged Pincer-Metal Platform: Coordination Chemistry & Applications, ed. G. van Koten and R. A. Gossage, Springer International Publishing, Cham, 2016, pp. 269–306 Search PubMed.
- V. Stoppa, T. Scattolin, M. Bevilacqua, M. Baron, C. Graiff, L. Orian, A. Biffis, I. Menegazzo, M. Roverso, S. Bogialli, F. Visentin and C. Tubaro, New J. Chem., 2021, 45, 961–971 RSC.
- A. Cervantes-Reyes, T. Saxl, P. M. Stein, M. Rudolph, F. Rominger, A. M. Asiri and A. S. K. Hashmi, ChemSusChem, 2021, 14, 2367–2374 CrossRef CAS PubMed.
- G. Fang and X. Bi, Silver Complexes in Organic Transformations, in Silver Catalysis in Organic Synthesis, 2019, pp. 661–722 Search PubMed.
- K. Yamashita, S. Hase, Y. Kayaki and T. Ikariya, Org. Lett., 2015, 17, 2334–2337 CrossRef CAS PubMed.
- Y. Li, X. Chen, Y. Song, L. Fang and G. Zou, Dalton Trans., 2011, 40, 2046–2052 RSC.
- J.-J. Zhang, J. K. Muenzner, M. A. Abu el Maaty, B. Karge, R. Schobert, S. Wölfl and I. Ott, Dalton Trans., 2016, 45, 13161–13168 RSC.
- A. Kascatan-Nebioglu, M. J. Panzner, J. C. Garrison, C. A. Tessier and W. J. Youngs, Organometallics, 2004, 23, 1928–1931 CrossRef CAS.
- T. Scattolin, N. Pangerc, I. Lampronti, C. Tupini, R. Gambari, L. Marvelli, F. Rizzolio, N. Demitri, L. Canovese and F. Visentin, J. Organomet. Chem., 2019, 899, 120857 CrossRef CAS.
- T. Scattolin, S. Giust, P. Bergamini, I. Caligiuri, L. Canovese, N. Demitri, R. Gambari, I. Lampronti, F. Rizzolio and F. Visentin, Appl. Organomet. Chem., 2019, 33, e4902 CrossRef.
- T. Scattolin, I. Caligiuri, L. Canovese, N. Demitri, R. Gambari, I. Lampronti, F. Rizzolio, C. Santo and F. Visentin, Dalton Trans., 2018, 47, 13616–13630 RSC.
- A. Kascatan-Nebioglu, A. Melaiye, K. Hindi, S. Durmus, M. J. Panzner, L. A. Hogue, R. J. Mallett, C. E. Hovis, M. Coughenour, S. D. Crosby, A. Milsted, D. L. Ely, C. A. Tessier, C. L. Cannon and W. J. Youngs, J. Med. Chem., 2006, 49, 6811–6818 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |