Cooperative E–H bond insertions and activations by transition metal complexes (E = B, C, Si, and Al): hemilability at play
Suvam
Saha
,
Stutee
Mohapatra
and
Sundargopal
Ghosh
 *
*
Department of Chemistry, Indian Institute of Technology Madras, Chennai, India. E-mail: sghosh@iitm.ac.in
First published on 23rd February 2026
Abstract
The cooperative activation and insertion of E–H σ-bonds at transition metal centers represent a powerful strategy for synthetic chemistry and catalysis (E = B, C, Si, Al). In recent years, hemilabile ligand frameworks capable of dynamically switching between coordinated and dissociated states have emerged as key enablers of such reactivity. Their ability to modulate electronic and steric environments around the metal center promotes synergistic metal–ligand cooperation (MLC). This Frontier article highlights recent advances in E–H bond activation and insertion processes typically mediated by transition metal complexes bearing hemilabile donor sites, with particular emphasis on mechanistic insights, bonding features, and the design principles governing ligand flexibility. Also, particular attention is given to σ-borane and σ-borate intermediates and C–H and Si–H bond cleavage. The emerging understanding of reversible coordination, metal–ligand bond polarity and ligand-assisted proton/hydride shuttling highlight hemilability as a central tool in developing next-generation catalytic platforms for sustainable synthesis and energy-relevant transformations.
Introduction
Transition metal complexes play a pivotal role in the activation of bonds such as C–H, B–H, and Si–H, thereby advancing the broad field of homogeneous catalysis.1 Typically, such activations take place at the metal center through elementary processes such as oxidative addition, reductive elimination, and β-hydride elimination (Chart 1a). Although ligands bound to the metal help stabilize the complex and modulate its reactivity patterns through electronic and steric effects, they generally do not play an active role in the primary bond activation event. Interestingly, over the past few decades, extensive studies have focused on the cooperative involvement of both the metal and the ligand in E–H (E = H, C, B, Si, Al, and Sn) bond activation, a concept widely recognised as metal–ligand cooperation (MLC) (Chart 1a).2 Systems that involve MLC in the rudimentary steps of catalytic processes have provided greater efficiency in both reactivity and selectivity terms as compared to the conventional approaches. Nature offers precedents for this concept, particularly in enzymatic catalysis such as dehydrogenation by [FeFe]- and [FeNi]-hydrogenases.3 Fujiwara's pioneering study on palladium acetate-mediated C–H activation laid the foundation for the MLC concept in homogeneous catalysis.4 | ||
| Chart 1 (a) Methods for E–H bond activation; (b) inspiration from nature for MLC; (c) modes of MLC involving hemilabile ligands. | ||
Subsequently, the extensive application of this strategy in cooperative H2 activation paved the way for its extension to other small molecules such as boranes, silanes, and alumanes.2,5
In MLC systems, ligands can act as Lewis bases and Lewis acids or undergo aromatisation/de-aromatisation to facilitate E–H bond activation.6 Also, redox non-innocent ligands can serve as electron reservoirs to regulate the metal oxidation state throughout the process. Thus, the ligand design is the most crucial aspect of the MLC approach for the E–H bond activation. In general, ligands can coordinate to metals through one or multiple binding sites, engaging in L-, X-, or Z-type interactions as classified by the covalent bond classification (CBC) system proposed by Green and Parkin.7 Due to the chelate effect, polydentate ligands offer increased stability and a higher degree of coordination environment of a metal in catalysis compared to monodentate ligands. While any heterobidentate ligand features a hard and a soft donor site, it can create vacant sites at the metal center readily under catalytic conditions and can be classified as a hemilabile ligand. Thus, based on the hard–soft acid–base (HSAB) principle developed by Pearson,8 the reactivity of hemilabile ligand coordinated metal complexes can be postulated. For instance, group 3–4 transition metals of higher oxidation states as well as lanthanides can be considered hard sites and they preferentially bind to the harder donor site of a heterobidentate hemilabile ligand. This results in weakening of the softer donor site-metal bond and eventually it becomes more susceptible to E–H bond activation. In contrast, softer late transition metal complexes with hemilabile ligands tend to activate E–H bonds through the harder donor site. Therefore, the careful selection of metal and ligand is an essential requirement for MLC.
Among the hemilabile bidentate ligands, the MLC activation of E–H bonds by 1,3-coordinated and 1,4-coordinated transition metal complexes has been studied extensively.9 These ligand frameworks are likewise widespread in nature, where they play key roles in processes such as proton transfer and element–hydrogen bond activation, among others. The purpose of this Frontier article is to survey the influence of hemilabile heterobidentate ligands on early and late transition metal centers, with emphasis on their 1,3- and 1,4-chelation modes in facilitating E–H bond activation (E = B, C, Si) via MLC. The E–H bond activations dictated by the HSAB principle in such complexes and their catalytic applications will be discussed. Simultaneously, the E–H bond insertions into the metal–ligand bonds or the MLC bonding of boranes or silanes, generating several unique molecules which are very interesting in terms of exploring novel bonding motifs and their utilization in catalysis will be presented. The scope of this Frontier article is summarised in Chart 2.
B–H bond activation by MLC
Boranes are important in synthesis and are being explored as carbon-free energy carriers, driving strong interest in B–H bond activation.1e While oxidative addition to transition metals remains a key strategy, metal–ligand cooperativity (MLC) has recently emerged as a powerful alternative, enabling efficient B–H activation and insertion processes that support catalytic formation of organoboron and other valuable products.1,2 Cooperative B–H binding has also uncovered unique bonding motifs in low-boron transition-metal complexes, such as boryl, borylene, σ-borate, and diborane species.1b,10 This Frontier article highlights recent advances in MLC-enabled B–H activation using ligand hemilability, focusing on ligand effects and resulting catalytic applications.Early transition metals
In 2020, Wang et al. reported a half-sandwich molybdenum thiolate complex [Cp*Mo(1,2-Ph2PC6H4S)(η2-NCMe)] (1), which contains a Mo–P–C–C–S five membered ring and a η2-MeCN ligand (Scheme 1).11 Complex 1 was found to be an efficient catalyst for the transfer hydrogenation of nitriles using [BH3·NH3] at room temperature. To gain mechanistic insight into this transformation, complex 1 was treated with [BH3·NH3] under ambient conditions. An immediate colour change from yellow to brown was observed, and the reaction afforded the Mo(II) hydride complex 2. Single-crystal X-ray diffraction analysis of 2 confirmed heterolytic B–H bond cleavage via Mo–S cooperation. Complex 2 features both a terminal Mo–H hydride and a bridging Mo–H–B hydride, forming a well-defined four-membered Mo–S–B–H metallacycle. The 11B NMR resonance at δ = −24.2 ppm is consistent with a borane-like ligand environment. The 1H NMR spectrum of 2 displayed three sets of hydride signals at δ = −4.49, −4.65, and −7.15 ppm as broad peaks, which were assigned to Mo–H–B–H, Mo–H–B–H and Mo–H, respectively. Notably, in the presence of ammonia, complex 2 mediates the reduction of nitriles to the corresponding amines. The combined mechanistic experiments and computational studies indicate that, during nitrile reduction with [BH3·NH3], the NH3 moiety serves as the source of the amine N–H hydrogens, whereas the BH3 fragment provides the methylene (CH2) hydrogens incorporated into the final amine product. These observations support a mechanism in which cooperative B–H bond activation is the key elementary step. | ||
| Scheme 1 B–H bond activation by a 1,4-P,S-chelated molybdenum complex. Reproduced from ref. 11 with permission from American Chemical Society,11 copyright 2020. | ||
The donor atoms in the 2-phosphinothiolate ligand of 1 are predominantly soft in nature. Probably, the lone pair on sulfur plays a decisive role in stabilising the BH2 unit formed upon the heterolytic cleavage of the B–H bond. Motivated by these observations, we sought to investigate the reactivity of Mo(II) complexes supported by bidentate chelating ligands that feature a combination of both soft and hard donor sites. To synthesise such a complex, the salt elimination reaction between [Cp*Mo(CO)3Me] and [K(2-mercaptobenzothiazolyl)] was carried out, which afforded the complex [Cp*Mo(CO)2{κ2-N,S-(NC7H4S2)}] (3a) (Scheme 2).12a In 3a, the soft Mo(II) center is chelated by the 2-mercaptobenzothiazolyl ligand through a soft S and a harder N donor, generating a strained four-membered Mo–N–C–S metallacycle. The reactivity of 3a with [BH3·THF] was examined; however, this resulted in decomposition of 3a. In contrast, using a different method, we have synthesised a W analogue of 3a, [Cp*W(CO)2{κ2-N,S-(NC7H4S2)}] (3b), from the photolytic treatment of [Cp*W(CO)3Me] with borate salts.12b The reaction of 3a and b with bulkier boranes MesBH2 and 3,5-trifluoromethylbenzene borane led cleanly to the formation of [Cp*M(CO)2{κ2-S,H-(NBH2R)(NC7H4S2)}] (4a: M = Mo, R = Mes; 4b: M = Mo, R = ArF, and 4c: M = W, R = ArF) (Scheme 2).12 The crystal structure of 4a displays a nonplanar six-membered molybdenacycle (MoHBNCS) generated from a hemilabile ring opening of the Mo–N bond followed by the capture of H2BMes. Considering sulfur as a soft base, it binds more strongly to soft Mo(II) centers as compared to hard N. Therefore, the insertion of borane occurs via cleavage of the weaker Mo–N bond in 3a. There is an elongation of approximately 15% in the Mo–coordinated B–H bond relative to the corresponding free B–H bond, consistent with significant activation and partial weakening of the B–H linkage upon coordination to the Mo(II) centre. The 11B NMR resonance for 4a at δ = −15.5 ppm is consistent with a borane-like ligand environment. However, computational studies suggest a bonding situation with mixed borane/borate character. The reaction of 3b with a bulky borane yielded an analogous σ-borate complex of tungsten, 4c. Due to the electron-poor boron center in 4b and 4c, the 11B chemical shifts are more deshielded than that of 4a.
 | ||
| Scheme 2 B–H bond activation by a 1,3-N,S-chelated molybdenum complex.12 | ||
Leitner and co-workers have synthesised a Mn P–N–P pincer complex, [Mn(Ph2PCH2SiMe2)2NH(CO)2Br] (5) (Scheme 3), via a salt elimination reaction between [Mn(CO)5Br] and the 1,3-bis ((diphenyl-phosphino)methyl)tetramethyldi-silazane ligand.13 Complex 5 proved to be an excellent catalyst for the challenging reduction of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O units of carboxylic acids, carbonic acid derivatives and even CO2, utilising pinacolborane as a reducing agent. The mechanistic investigation showed that complex 5 converts to 5a containing a Mn
O units of carboxylic acids, carbonic acid derivatives and even CO2, utilising pinacolborane as a reducing agent. The mechanistic investigation showed that complex 5 converts to 5a containing a Mn![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond in the presence of a base. The addition of pinacolborane to 5a led to the formation of a manganese σ-borate complex 6, which contains a four-membered Mn–H–B–N ring. Complex 6 converts slowly to a Mn–H complex, evident from NMR experiments. These observations suggest a complete heterolytic cleavage of the B–H bond by metal–ligand cooperation.
N bond in the presence of a base. The addition of pinacolborane to 5a led to the formation of a manganese σ-borate complex 6, which contains a four-membered Mn–H–B–N ring. Complex 6 converts slowly to a Mn–H complex, evident from NMR experiments. These observations suggest a complete heterolytic cleavage of the B–H bond by metal–ligand cooperation.
 | ||
| Scheme 3 B–H bond activation by a Mn–N bond.14 | ||
Late transition metals
Oestreich and co-workers reported the synthesis of a coordinatively unsaturated ruthenium complex, [(DmpS)Ru(PR3)][BArF4] (Dmp = 2,6-dimesitylphenyl), 7, characterized by a strongly polarized Ru–S interaction (Scheme 4).14 When the reactivity of 7 toward a range of substituted boranes was examined using NMR techniques, the resulting saturated adducts, 8, exhibited upfield B–H signals, as compared to the uncoordinated boranes. Structural analysis of one of these adducts revealed an unusual arrangement in which boron binds to the sulfur donor while the corresponding hydrogen bridges to the Ru center, forming a four-membered H–Ru–B–S ring system. In this species, the B–H bond is significantly elongated (1.55 Å), approximately 30% longer than in a typical B–H bond, consistent with substantial activation. The complex can be interpreted as the product of σ-bond metathesis at the B–H unit, generating a ruthenium hydride in combination with a sulfur-stabilized borenium fragment. The same system was subsequently utilized as a catalyst for the dehydrogenative borylation of indoles and pyrroles, where the proposed reaction pathway involves initial electrophilic borylation at the heteroaromatic ring followed by deprotonation to yield the borylated products. | ||
| Scheme 4 B–H bond activation and borenium cation formation by a ruthenium–thiolate complex. Reproduced from ref. 14 with permission from American Chemical Society,14 copyright 2013. | ||
Several examples have been reported demonstrating the participation of metal carbene complexes in cooperative B–H bond activation.15,16 In this context, Gessner and co-workers reported the B–H bond activation of several boranes using the Ru–carbene complex 9 (Scheme 5).16b Upon reaction with catecholborane, complex 9 underwent B–H bond cleavage across the Ru![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond to afford complex 10. The molecular structure of 10 revealed a slightly pyramidalized boron center, with a sum of angles of 351.0°, indicating an interaction between the boron atom and the Ru–H bond, as further supported by natural bond orbital (NBO) analysis. Although the B⋯H distance (1.68 Å) in 10 is longer than that of a typical B–H bond, as well as those involved in agostic interactions with a metal center, it is consistent with a weak secondary interaction. Interestingly, treatment of 9 with [BH3·SMe2] led to the activation of two borane units, yielding complex 11. One B–H bond of a BH3 molecule was activated via coordination at the Ru–S bond, whereas a B–H bond of a second BH3 unit was fully cleaved, resulting in protonation of the carbene carbon atom and insertion of a BH2 moiety into the P–S bond of 9. Complex 11 can be described either as a ruthenium hydride bearing a boryl-substituted thioether ligand or as a thiolato complex featuring BH3 coordination along the Ru–S linkage. Notably, the reactivity of 9 toward pinacolborane was found to be similar to that observed with [BH3·SMe2].
C bond to afford complex 10. The molecular structure of 10 revealed a slightly pyramidalized boron center, with a sum of angles of 351.0°, indicating an interaction between the boron atom and the Ru–H bond, as further supported by natural bond orbital (NBO) analysis. Although the B⋯H distance (1.68 Å) in 10 is longer than that of a typical B–H bond, as well as those involved in agostic interactions with a metal center, it is consistent with a weak secondary interaction. Interestingly, treatment of 9 with [BH3·SMe2] led to the activation of two borane units, yielding complex 11. One B–H bond of a BH3 molecule was activated via coordination at the Ru–S bond, whereas a B–H bond of a second BH3 unit was fully cleaved, resulting in protonation of the carbene carbon atom and insertion of a BH2 moiety into the P–S bond of 9. Complex 11 can be described either as a ruthenium hydride bearing a boryl-substituted thioether ligand or as a thiolato complex featuring BH3 coordination along the Ru–S linkage. Notably, the reactivity of 9 toward pinacolborane was found to be similar to that observed with [BH3·SMe2].
 | ||
| Scheme 5 B–H bond activation by a ruthenium–carbene complex. Reproduced from ref. 16b with permission from John Wiley and Sons,16b copyright 2018. | ||
In 2009, the Stradiotto group synthesised a coordinatively unsaturated ruthenium complex, [Cp*Ru(κ2-P,O-L)] (L = 1-diisopropylphosphino-2-indanone), and utilised it in activation of various E–H bonds (E = H, B, and Si).17 Similarly, while exploring the role of hemilabile 1,3-N,S-chelating ligands in metal–ligand cooperative (MLC) transformations, we serendipitously isolated a rare Ru(III) complex, [PR3{κ2-N,S-(C7H4NS2)}2Ru{κ1-S-(C7H4NS2)}] (12), featuring two κ2-1,3-N,S-chelated rings, one pendant mercaptobenzothiazole unit, and a phosphine ligand (Scheme 6).18 Remarkably, treatment of complex 12 with [BH3·THF] afforded a bis-(σ-borate) species, [PPh3Ru{κ3-H,S,S-(NH2BSBH2N)(S2C7H4)2}] (13), whereas the reaction with the bulky MesBH2 yielded two unusual bis-(dihydroborate) complexes, [{κ3-S,H,H-(NH2BMes)Ru(S2C7H4)}2] (trans-14 and cis-14). In both transformations, dual-site borane insertion occurred across the two Ru–N bonds, accompanied by dissociation of the PPh3 ligand. The formation of the B2S core in 13 likely involves cleavage of the C–S bond of the dangling ligand in 12, releasing benzothiazole. On the other hand, complete dissociation of the 2-mercaptobenzothiazolyl ligand leads to the generation of isomers of complex 14. Notably, the 11B NMR resonance of 13 (δ = −10.1 ppm) is significantly more shielded than those of trans-14 (δ = 39.4 ppm) and cis-14 (δ = 31.2 ppm), indicating a higher σ-donation of B–H bond to the metal in 13.
 | ||
| Scheme 6 Dual site B–H bond activation by a 1,3-N,S-chelated ruthenium(III) complex.18 | ||
We have also synthesised bis-1,3-N,S-chelated Ru(II) and Os(II) complexes 15a and b, each containing two strained four-membered M–N–C–S metallacycles (Scheme 7).19 Upon reaction with [BH3·SMe2], both complexes undergo B–H bond activation across the polar M–N bonds, albeit leading to different products. In the case of the Ru complex (15a), the borane unit inserts into the polar Ru–N bond to afford a ruthenium σ-borate complex (16) that retains a four-membered Ru–N–C–S ring framework (Scheme 7).19b In contrast, the Os(II) analogue (15b) promotes heterolytic B–H bond cleavage upon reaction with [BH3·SMe2], resulting in the formation of an osmium(σ-borate) hydride complex (17).19c The reaction is proposed to proceed via initial B–H bond cleavage across one Os–N bond to generate an osmium hydride intermediate, followed by cleavage of the second Os–N bond to form a B–N bond. Subsequent coordination of one of the remaining B–H bonds of the [BH2L2] fragment (L = 2-mercaptobenzothiazolyl) to the osmium center forms complex 17. Although the Os–H hydride in 17 could not be observed in the solid-state X-ray diffraction analysis, its presence was confirmed by a sharp resonance at δ = −13.26 ppm in the 1H NMR spectrum, which remained unchanged upon 11B decoupling.
 | ||
| Scheme 7 B–H bond activations by bis-1,3-N,S-chelated ruthenium(II) and osmium(II) complexes.19 | ||
In contrast, the reaction of an analogous bis-κ2-N,S-chelated iron(II) complex, [(dppe)Fe{κ2-N,S-(mp)}2] (mp = 2-mercapto-pyridyl) (18), with [BH3·SMe2] afforded a rare dimeric iron bis(dihydridoborate) complex, [Fe{κ3-S,H,H-(H2BH(mp))}2]2, ΛΔ/ΔΛ-19, isolated as a pair of enantiomers (Scheme 8).20 Each enantiomer features an almost planar Fe1–S1–Fe2–S2 square core, with a C2 axis of symmetry passing through the centre of this plane. The NMR studies reveal the formation of a monomeric bis-(dihydroborate) intermediate, [Fe{κ3-S,H,H-(H2BH(C5H4NS))}2], which subsequently undergoes dimerization to furnish ΛΔ/ΔΛ-19. These findings clearly illustrate that subtle variations in metal identity and ligand environment can steer the same MLC-assisted B–H insertion pathway toward distinct structural outcomes, enabling access to novel architectures.
 | ||
| Scheme 8 Dual site B–H bond activation by a 1,3-N,S-chelated iron complex and dimerization of the dihydroborate complex.20 | ||
Daly et al. reported the synthesis of a Ru(II) complex 20 supported by a triaryl, redox-active S2N2 ligand constructed from o-phenylenediamine and thioanisole fragments (Scheme 9).21 Upon treatment of 20 with the reducing agent [NaBH4], the reaction afforded complex 21, featuring dual metal–ligand cooperative coordination of two BH3 units across the Ru–N bonds. Structural analysis revealed a notable elongation of the N–C bonds and a shortening of the NC–NC bonds in 21 relative to 20, consistent with the reduction of the diiminoquinone framework to the corresponding o-phenylenediamine form. The 11B NMR spectrum of 21 displayed a single resonance at δ = −9.3 ppm, while 1H NMR measurements suggested the presence of three stereoisomers, attributable to different spatial orientations of the two SMe2 substituents. In complexes 3a and b, 12, and 18, the polarity of the M–N bonds governs B–H activation, whereas the metal–ligand cooperative binding of BH3 in 21 is driven primarily by the reduction of the non-innocent ligand backbone, enabling coordination without cleavage of the M–N bonds.
 | ||
| Scheme 9 B–H bond activation by a Ru–N bond. Reproduced from ref. 21 with permission from American Chemical Society,21 copyright 2020. | ||
A similar metal–ligand cooperative binding of BH3 by a rigid tetradentate bisphosphine–bisamide [PNNP]2− ligand chelated Fe(II) complex (22) was reported by Thomas and co-workers (Scheme 10).22 Dual-MLC bound BH3 complex 23 shows a more shielded 11B chemical shift at δ = −17.1 ppm than complex 21. The 1H chemical shifts for the M–H–B protons for 23 (δ = −15.40 ppm) are more shielded than those of 21 (δ = −9.56, −9.35 and −8.96 ppm for three stereoisomers), indicating more metal hydride character. The molecular structures of both 21 and 23 show the B–H binding generated two M–N–B–H metallacycles with an elongation of the B–H bonds (1.334 Å (23), 1.310 Å (21)) coordinated to metal atoms compared to the terminal B–H hydrogens (1.104 Å (23), 1.112 Å (21)), indicating cooperative B–H bond activation. The structural assessment and NMR spectroscopic data support the assignment of fully cleaved B–H bonds with weak Lewis acid/base interactions between the metal hydrides and borane fragments. A few other examples of MLC binding of boranes (24–26) have also been reported (Chart 3).23
 | ||
| Scheme 10 B–H bond activation by an Fe–N bond. Reproduced from ref. 22 with permission from Royal Society of Chemistry,22 copyright 2020. | ||
 | ||
| Chart 3 Examples of MLC binding of BH3.23 | ||
Schafer and Love et al. synthesised 1,3-N,O-chelated Rh and Ir complexes [M{κ2-N,O-Xyl(N)P(O)(OEt)2}(η4-cod)] (27a: Rh, 27b: Ir) containing a M–O–P–N four-membered ring. The harder O-center and a relatively softer N donor are coordinated to the soft Rh/Ir metals (Scheme 11), forming a hemilabile framework. Upon treatment with bulky cyclohexylborane (HBCy2), the borane is captured by the M–O bonds of 27a and b generating six-membered M–N–P–O–B–H metallacycles in 28a and b.24 The 11B chemical shifts for 28a and b (δ = 13.0 ppm (28a) and δ = 14.6 ppm (28b)) are substantially shielded compared to those of free Cy2BH (δ = 59.0 ppm), consistent with Shimoi-type O → B bonding along with B–H σ-donation to the metal center. The B–H bond distances (1.310 Å (28a), 1.41 Å (28b)) are considerably longer than the free B–H bond distances, indicating B–H bond activation (Scheme 11).
 | ||
| Scheme 11 B–H bond insertion by 1,3-N,O-chelated Rh/Ir complexes.24 | ||
Complexes 28a and b promote chemoselective hydroboration of aldehydes even in the presence of an alkyne (Scheme 12). When free HBCy2 or the Lewis base-stabilised borane complex [(EtO)3P·BHCy3] is used, alkyne hydroboration proceeds, as observed with 1-octyne.24 In contrast, the use of complexes 28b results in inverse chemoselectivity, where aldehyde hydroboration dominates. The change in selectivity cannot be attributed solely to Lewis-base stabilization of the borane, thereby indicating a substantial role of MLC. The proposed mechanism for hydroboration involves activation through either M–H–B bond cleavage or P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O–B bond cleavage, enabling capture of the small molecule and generating boron-functionalized products. The hydroboration regenerates the initial complexes 27a and b, thereby closing the catalytic cycle.
O–B bond cleavage, enabling capture of the small molecule and generating boron-functionalized products. The hydroboration regenerates the initial complexes 27a and b, thereby closing the catalytic cycle.
 | ||
| Scheme 12 Chemoselective hydroboration of aldehydes via MLC. Reproduced from ref. 24 with permission from John Wiley and Sons,24 copyright 2016. | ||
Recently, a series of κ2-N,L-chelated Ir and Rh complexes, [Cp*M(κ1-E-C5H4NL)(κ2-N,L-C5H4NX)], (29a: M = Ir, L = S; 29b: M = Ir, L = Se; 29c: M = Rh, L = S; and 29d: M = Rh, L = Se) were synthesised by us through salt-metathesis reactions of [CpMCl2]2 with the corresponding thio- or seleno-pyridyl potassium salts (Scheme 13).25 These complexes feature one ligand bound in a κ1-L mode, while the second adopts a κ2-N,L chelation pattern, generating strained four-membered metallacycles with unusually small bite angles (≈68°). Exposure of the Ir derivatives (29a and b) to [BH3·THF] produces two distinct classes of products: monometallic diborane(5) complexes, [(Cp*IrIIIH){κ3-B,B,L-(B2H4-C5H4NL)}] (30a: E = S, 30b: E = Se), and Ir(I) dihydroborate complexes, [(η4-C5Me5H)IrI{κ2-H,H,L-(H3B-C5H4NL)}] (31a: L = S, 31b: L = Se).21 The diborane(5) species 30a and b stabilize a rare non-classical [B2H4(C5H4NL)]− fragment, plausibly formed through MLC-assisted B–H activation at the Ir–N site followed by dehydrogenative B–B coupling. We have earlier synthesised a bimetallic template based classical diborane(5) complex and a monometallic classical diborane(6) complex, in which the formation involves no ligand cooperation.26 In contrast, the Rh analogues (29c and d) undergo B–H insertion into the Rh–N bond to yield Rh(I) dihydroborate complexes, [(η4-C5Me5H)RhI{κ3-H,H,L-(H3B-C5H4NL)}] (31c: L = S, 31d: L = Se), accompanied by Cp* ring slippage. These divergent outcomes highlight the distinct reactivity profiles of Ir and Rh toward B–H activation within strained κ2-N,L ligand environments.
 | ||
| Scheme 13 B–H bond activation by MLC and dehydrogenative B–B homocoupling.25 | ||
Moving to Group 10 metals, Wang and co-workers synthesised half-sandwich Ni(II) complexes of the type [Cp*Ni(1,2-R2PC6H4X)] (32a: X = O, R = Ph; 32b: X = NH, R = Ph), featuring M–P–C–C–O/N five-membered metallacycles (Scheme 14).27 Complexes 32a and b readily undergo heterolytic cleavage of the B–H bond in HBPin to form the corresponding nickel hydride complexes 33a and b. Interestingly, B–H activation by the Ni–O complex 32a is reversible, whereas the Ni–N analogue 32b mediates irreversible activation. As a consequence, in the catalytic hydroboration of N-heterocycles, the Ni–O system 32a exhibits significantly superior performance compared to the Ni–N complex 32b.27
 | ||
| Scheme 14 B–H bond activation at Ni–N and Ni–O bonds.27 | ||
Recently Gunanathan et al. synthesised a stable pyridine based Cu(II) NNN pincer complex, 34, which utilizes ammonia borane as a hydrogen source to facilitate the 1,2-reduction of quinolines (Scheme 15).28 Based on the several experimental studies, they have proposed a plausible mechanism for this catalytic process. Complex 34 undergoes dehydrohalogenation in the presence of a base to form an amide complex, 35, which further undergoes disproportionation to yield an active catalyst 36. Complex 36 may have coordinated with [NH3·BH3] to form the intermediate I. This intermediate possibly transfers hydrides to the metal and ligand center simultaneously to form II by releasing borazine. Eventually, intermediate II may have been reduced with quinoline selectively to form 1,2-dihydroquinoline and regenerate the catalytically active species 36. The metal–ligand cooperative activation of B–H and N–H bonds is crucial for the catalytic transformation.
 | ||
| Scheme 15 B–H bond activation and utilization as a hydride source.28 | ||
Si–H bond activation by MLC
Metal-mediated and metal–ligand cooperative (MLC) Si–H bond activation processes have been studied extensively, owing to the relatively low bond dissociation energy of the Si–H bond, which is comparatively lower than that of H–H. This reduced bond strength renders the Si–H activation as an attractive pathway for accessing value-added organosilicon compounds. Although significant progress has been made, examples of Si–H bond activation via metal–ligand cooperation remain relatively uncommon. One of the earliest examples of MLC-driven Si–H activation was reported by Tilley and co-workers, who showed that a Ta![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond in tantalum imido complexes can heterolytically cleave Si–H bonds (Chart 4).29 Subsequent studies by Andersen, Clot, and others revealed analogous reactivity at Ti
N bond in tantalum imido complexes can heterolytically cleave Si–H bonds (Chart 4).29 Subsequent studies by Andersen, Clot, and others revealed analogous reactivity at Ti![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N and Ti
N and Ti![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S bonds.30 In this Frontier article, recent advances in Si–H bond cleavage, insertion processes, and their emerging catalytic applications will be highlighted.
S bonds.30 In this Frontier article, recent advances in Si–H bond cleavage, insertion processes, and their emerging catalytic applications will be highlighted.
 | ||
| Chart 4 Si–H activations by M–N or M–S bonds.29,30 | ||
As discussed earlier, based on the dual site B–H bond activation across the Fe–N (amide) linkage of the 18-electron low-spin bis(amido)bis(phosphine) Fe(II) complex 22, Thomas et al. have utilised it for the activation of the Si–H bonds of primary silanes (Scheme 16).31 Treatment of complex 22 with two equivalents of PhSiH3 affords the Fe–Hydride complex 43via Si–H bond activation followed by dehydrogenation. The molecular structure of 43 reveals that two Si–H bonds have been cleaved across Fe–N linkages by two independent units of 22 (Scheme 16). In the resulting complex, the Si–H bonds are elongated by at least 10% relative to those in free silanes. The absence of significant 1JSi–H coupling for the Fe-hydride signals (H1 and H2) indicates minimal M–H⋯Si interaction and supports a predominantly Fe–H character, consistent with typical Fe–H bond lengths. In contrast, H3 lies farther from the Fe centers. Computational analysis shows that H1 and H2 function as Fe hydrides involved in 3c–2e Fe–H–Si interactions, whereas Si–H3 behaves as a Si hydride engaged in a weaker, agostic-type 4c–2e interaction with the Fe centers. Interestingly, when excess silane was reacted with 22, it led to the isolation of a mixture of two trans and cis isomers of iron silyl complex 44 in a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 ratio (Scheme 16). The solid-state structure of one of the isomers, trans-44, revealed that two equivalents of PhSiH3 have undergone dehydrogenative coupling across the Fe–amide linkage to form a new Si–Si bond, accompanied by the release of two equivalents of H2. The Si–Si (2.307 Å) bond distance in trans-44 is also between those of a typical Si–Si and a Si
2 ratio (Scheme 16). The solid-state structure of one of the isomers, trans-44, revealed that two equivalents of PhSiH3 have undergone dehydrogenative coupling across the Fe–amide linkage to form a new Si–Si bond, accompanied by the release of two equivalents of H2. The Si–Si (2.307 Å) bond distance in trans-44 is also between those of a typical Si–Si and a Si![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Si bond. Traditionally, Si–Si bond formation has relied on Wurtz-type coupling of halosilanes using alkali metals; however, these reactions often require harsh conditions and generate undesirable by-products. This was the first example of a Si–Si bond formation via MLC binding of Si–H bonds under ambient conditions.
Si bond. Traditionally, Si–Si bond formation has relied on Wurtz-type coupling of halosilanes using alkali metals; however, these reactions often require harsh conditions and generate undesirable by-products. This was the first example of a Si–Si bond formation via MLC binding of Si–H bonds under ambient conditions.
 | ||
| Scheme 16 Si–H activation and silane coupling across an Fe–amide bond. Reproduced from ref. 31 with permission from American Chemical Society,31 copyright 2023. | ||
Rauchfuss and co-workers have described the synthesis of an iron(II) diphosphine–dialkoxide dicarbonyl complex [Fe(LP2O2)(CO)2] (45; P2O2 = (Ph2PC6H4CHO)22−) by the coupling of 2 equiv. of 2-diphenylphosphinobenzaldehyde in the presence of [Fe(CO)3bda] (bda = benzylideneacetone) (Scheme 17).32 Although complex 45 was unreactive towards H2, it takes part in heterolytic Si–H bond activation of Ph2SiH2 to produce an Fe hydride complex [HFe[P2O(OSiHPh2)](CO)2] (46), bearing an O-silylated phosphine–alkoxide arm (Scheme 17). The hemilability of the ligand backbone plays a crucial part in this case. IR and NMR spectroscopic data support the presence of an Fe–H unit alongside silylation localized at oxygen, confirming ligand-centered Si–H engagement rather than further reaction at the metal. X-ray structural analysis verified the endo orientation of the siloxy substituent, demonstrating retention of stereochemistry at the ligand backbone. Notably, the complex remains stable even in excess Ph2SiH2, as the second alkoxide group does not undergo silylation, and the Si–H bond within the silylated product is unreactive toward another equivalent of the starting Fe-complex, reflecting steric shielding and electronic saturation at the remaining alkoxide.
 | ||
| Scheme 17 Heterolytic Si–H cleavage across an Fe–O bond. Reproduced from ref. 32 with permission from American Chemical Society,32 copyright 2015. | ||
In the course of our recent efforts for B–H bond activation, we have isolated several κ2-N,S-chelated group 8 metal complexes. One of these complexes, 16 was thermolysed with 1.2 equiv. of Ph2SiH2 that led to the Si–H bond activation by the polar Ru–N bond yielding isomeric silyl complexes 47 and 47′ along with a trisilyl ruthenium hydride complex 48 in low yields (Scheme 18).33 At higher temperature, the yield of 48 increases. The upfield 1H chemical shift at δ = −10.56 ppm supports the presence of a Ru–H moiety. The 29Si{1H} NMR spectrum features a single resonance at δ = 55.8 ppm, which is in agreement with a symmetrical environment in 48. The solid-state X-ray structure of 48 clearly confirms that the ruthenium center is coordinated to three silyl groups in an octahedral fashion together with a capped hydride ligand (Ru–H) (Fig. 1).
 | ||
| Scheme 18 Si–H bond activation by a κ2-N,S-chelated ruthenium complex.33 | ||
 | ||
| Fig. 1 Molecular structure of 48.33 | ||
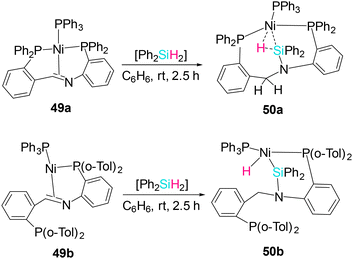
Moret et al. have reported the synthesis of multiple Ni(0) complexes 49a and b, supported by a pincer-type ligand platform containing an imine moiety bridged by two/one o-phenylene linkers with phosphine substituents (PCNP). Reaction of the Ni PCNP complex 49a with diphenylsilane in benzene yielded a single isolable product 50a, arising from formal C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N hydrosilylation (Scheme 19).34 N-SiPh2H remains Ni-bound via η2-Si–H σ-coordination, supported by 31P NMR (32.8, 17.1, 7.8 ppm, mutually coupled) and a hydridic 1H at −2.77 ppm showing 2J(H–Si) = 109 Hz, consistent with η2-Si–H binding rather than oxidative addition. To probe steric effects, o-tolyl Ni(0) complex 50b was treated with diphenylsilane in C6D6, heterolytic cleavage of Si–H occurred yielding 50b. DFT calculations suggest a cooperative Ni-mediated ligand-to-ligand hydrogen transfer as a key step. Interestingly, the degree of activation of the remaining Si−H bond in the product was found to strongly depend on the coordination number at Ni. Three-coordinate complex 50a is best described as an η2(Si,H) σ-complex. However, in 50b, the Si–H bond is activated to a larger extent, and it is better viewed as a Ni(II) center bearing a silyl and a hydride ligand engaging in a secondary interaction.
N hydrosilylation (Scheme 19).34 N-SiPh2H remains Ni-bound via η2-Si–H σ-coordination, supported by 31P NMR (32.8, 17.1, 7.8 ppm, mutually coupled) and a hydridic 1H at −2.77 ppm showing 2J(H–Si) = 109 Hz, consistent with η2-Si–H binding rather than oxidative addition. To probe steric effects, o-tolyl Ni(0) complex 50b was treated with diphenylsilane in C6D6, heterolytic cleavage of Si–H occurred yielding 50b. DFT calculations suggest a cooperative Ni-mediated ligand-to-ligand hydrogen transfer as a key step. Interestingly, the degree of activation of the remaining Si−H bond in the product was found to strongly depend on the coordination number at Ni. Three-coordinate complex 50a is best described as an η2(Si,H) σ-complex. However, in 50b, the Si–H bond is activated to a larger extent, and it is better viewed as a Ni(II) center bearing a silyl and a hydride ligand engaging in a secondary interaction.
Al–H bond activation by MLC
Coordination and activation of Al–H bonds exclusively at metal centres is well established. Nikonov and co-workers demonstrated the reversible oxidative addition of an Al–H bond to an Al(I) centre.35 Subsequently, the Aldridge group reported σ(Al–H) coordination to group 6 transition metal centers in both κ1 and κ2 binding modes. In contrast, coordination of a β-diketiminato-supported alane to a cobalt centre resulted in an unusually high degree of Al–H bond activation.36 Crimmin and colleagues described the coordination of hydroalumane to group 8 metals, leading to complexes that feature three-centre-two-electron (3c–2e) M–H–Al bonding motifs.37 In fact, the aluminium based ligand supported transition metal complexes have been utilised in a variety of small molecule activations.38,39 Despite these advances, examples of Al–H bond activation proceeding through metal–ligand cooperation are rare.One notable example was reported by Oestreich and co-workers, who investigated the reaction of complex 7 with DIBAL-H, which rapidly afforded the hydroalane adduct 51 (Scheme 20).5 In this complex, aluminium binds to the sulfur donor of the ligand framework, while the hydride is connected to the ruthenium centre. In a true sense, the hydroalane adduct (51) cannot be considered as a sulfur-stabilised aluminium ion.40 The Al–H bond in 51 is significantly elongated relative to that in free hydroalanes, consistent with heterolytic Al–H bond cleavage. However, computational studies suggest that the bonding in 51 is better described as a σ(Ru–H) → Al donor–acceptor interaction. In addition to the Ru–H and Al–S linkages, a weak Ru⋯Al interaction is also evident. Importantly, the Ru–S-activated DIBAL-H adduct 51 was shown to promote C(sp3)–F bond cleavage, and complex 7 proved to be a highly effective catalyst for hydrodefluorinative Friedel–Crafts alkylation of CF3-substituted arenes, delivering unsymmetrical diarylmethanes with a broad range of arene nucleophiles.
 | ||
| Scheme 20 Al–H bond activation by a polar Ru–S bond.5 | ||
C–H bond activation by MLC
C–H bond cleavage is one of the most important elementary reactions for several organic transformations. The chemistry of C–H activation has evolved significantly over the last 60 years, mainly utilizing sigma bond metathesis or concerted metalation deprotonation processes. The examples of metal–ligand cooperative C–H bond activation have emerged over the last decade. Ligand aromatization–dearomatization and metal–carbene bond mediated C–H bond activations are the most well known. However, the examples of hemilabile ligand participation in MLC C–H activation are quite limited and mostly the recent advances in the activation of C–H bonds of terminal alkynes will be discussed in this Frontier article. Notably, unlike the hydridic E–H (E = B, Al, and Si) bonds discussed above, the C–H bond of terminal alkynes is protic in nature.The 1,3-N,O-chelated phosphoramidate complex, [Ir{κ2-N,O-Xyl(N)P(O)(OEt)2}(η4-COD)] (52), was employed for C–H activation to utilize cooperation between Ir(III) and a hemilabile phosphoramidate ligand to develop a new chemical transformation: 1-alkyne O-phosphoramidation (Scheme 21).41 The goal was to overcome the preference of alkyne functionalization reactions for Markovnikov or N-functionalized products, thereby enabling the synthetically important anti-Markovnikov O-functionalization pathway. Achieving such selectivity provides access to vinyl organophosphates, compounds of notable biological and synthetic importance. The reaction of 52 with a terminal alkyne results in a change from a four- to a five-membered metallacycle to give the 16-electron (E)-vinyloxy iridium(III) complex, [Cp*Ir(κ2-N,C-(E)-Xyl(N)P(O–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(H)(Ph)) (OEt)2)]+ (53). Interestingly, under the mild acidic work-up conditions, complex 53 led to the isolation of (Z)-vinyloxy phosphoramidate 54 and a known aggregate [Cp*Ir(O2CCF3)2(H2O)]n. The hemilabile N,O-chelate plays an active role in the catalytic cycle, functioning as a proton shuttle (LAPS: ligand-assisted proton shuttle) and enabling reversible [(O2CCF3)2(H2O)]n. coordination dynamics that facilitate alkyne activation and promote selective C–O bond formation at the terminal carbon. In accordance with the HSAB principle, the alkyne insertion occurred through the cleavage of the relatively weaker bond between the soft Ir and hard O compared to the softer N. This study nicely illustrated how ligand design and cooperative reactivity can be harnessed to override intrinsic regioselectivity in alkyne functionalization.
C(H)(Ph)) (OEt)2)]+ (53). Interestingly, under the mild acidic work-up conditions, complex 53 led to the isolation of (Z)-vinyloxy phosphoramidate 54 and a known aggregate [Cp*Ir(O2CCF3)2(H2O)]n. The hemilabile N,O-chelate plays an active role in the catalytic cycle, functioning as a proton shuttle (LAPS: ligand-assisted proton shuttle) and enabling reversible [(O2CCF3)2(H2O)]n. coordination dynamics that facilitate alkyne activation and promote selective C–O bond formation at the terminal carbon. In accordance with the HSAB principle, the alkyne insertion occurred through the cleavage of the relatively weaker bond between the soft Ir and hard O compared to the softer N. This study nicely illustrated how ligand design and cooperative reactivity can be harnessed to override intrinsic regioselectivity in alkyne functionalization.
 | ||
| Scheme 21 Ligand-assisted proton shuttling by a 1,3-N,O-chelated phosphoramidate complex, 52. Reproduced from ref. 41 with permission from American Chemical Society,41 copyright 2016. | ||
Maseras and Sola groups have demonstrated the effect of a hemilabile acetate ligand in C–H bond activation by MLC as compared to the transition metal based activation. The five coordinate Ru hydride complex [Ru(Cl)H(CO)(PiPr3)2] can react with 1 equivalent of phenylacetylene to produce a phenylacetylide derivative.42a However, when a 6-coordinate derivative [Ru(κ2-acetate)H(CO)(PiPr3)2] (55) reacts with phenylacetylene, it doesn't stop at the alkenyl derivative (56) (Scheme 22).42b In fact, complex 56 reacts with one more equivalent of phenylacetylene to produce complex 57 with the release of styrene. Computational studies revealed that this pathway is assisted by one oxygen atom of the hemilabile acetate ligand. This is followed by proton transfer to the alkenyl ligand, again mediated by the same acetate –OH functionality through a ligand-assisted proton shuttle (LAPS) mechanism. This cooperative sequence ultimately leads to styrene formation and the alkynyl complex 57.
 | ||
| Scheme 22 Acetate assisted C–H bond activation and proton shuttle.42b | ||
The hydroborate complexes of Ru, [(k2-N,S-L)PR3Ru{κ3-H,S,S’-H2B(L)2}] (16) (R = Ph or Cy; L = C7H4NS2) containing a strained four membered Ru–N–C–S ring synthesised by us have been proven to show hemilabile characteristics for small molecule activation. When they are employed for the C(sp)–H bond activation of terminal alkynes containing electron-withdrawing groups, it leads to the insertion of the alkynes into the Ru–N as well as Ru–S bonds to form complexes 58 and 59 (R′ = CO2Me or C6H4NO2), respectively (Scheme 23).19a At room temperature or under photolytic or mild thermolytic conditions, complex 58 is the major product. Interestingly, at higher temperatures, complex 58 was converted to 59. This indicates that amination was favoured under milder conditions, while under harsher conditions thionation was more facile. Both complexes contain a five-membered ruthenacycle featuring an exocyclic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond. Guided by earlier reports on LAPS-type processes, we proposed that the alkyne first undergoes metal-mediated activation, followed by proton transfer from the alkyne to the basic N,S-benzothiazolyl fragment. This is then succeeded by an intramolecular anti-Markovnikov attack by either the nitrogen or sulfur atom of the benzothiazolyl unit onto the activated alkyne. Overall, the reactions are consistent with a ligand-assisted proton shuttle (LAPS) mechanism, in which the nucleophilic N or S site of the benzothiazolyl group in 16 serves as an internal base, facilitating both the C–H activation step and subsequent bond-forming events.
C bond. Guided by earlier reports on LAPS-type processes, we proposed that the alkyne first undergoes metal-mediated activation, followed by proton transfer from the alkyne to the basic N,S-benzothiazolyl fragment. This is then succeeded by an intramolecular anti-Markovnikov attack by either the nitrogen or sulfur atom of the benzothiazolyl unit onto the activated alkyne. Overall, the reactions are consistent with a ligand-assisted proton shuttle (LAPS) mechanism, in which the nucleophilic N or S site of the benzothiazolyl group in 16 serves as an internal base, facilitating both the C–H activation step and subsequent bond-forming events.
 | ||
| Scheme 23 Amination vs. thionation of alkynes by 1,3-N,S-chelated Ru–borate complexes.19a | ||
In 2018, Albéniz and coworkers showed that the arylation of pyridines proceeds via a ligand-assisted C–H bond activation process. They employed 6-hydroxy-2,2′-bipyridyl (L1) which is tautomeric with its ketone form, as a ligand to accomplish the efficient conversion of pyridine and 4-methyliodobenzene to 3-tolylpyridine successfully with good yield (Scheme 24).43 However, when the 4-hydroxy-2,2′-bipyridyl (L2) was utilised, the reaction didn't show satisfactory results. The theoretical studies show that the ligand-assisted concentrated deprotonation of pyridionate oxygen is a key step in the catalytic cycle. Therefore, the position of the keto group in the coordinating ligand in 60 is in the closest proximity to the Pd center, which facilitates C–H activation and subsequent palladation to form intermediate 61. The catalytic process operates via a Pd(0)/Pd(II) redox cycle. Notably, cleavage of the meta-C–H bond is energetically favoured, consistent with the observed regioselectivity in the product distribution.
 | ||
| Scheme 24 Ligand-assisted C–H bond activation and arylation of pyridines.43 | ||
Crimmin and co-workers reported the synthesis of a low-spin d6 iron dihydride complex (62) featuring an aluminium-based ligand that interacts directly with the hydride ligands (Scheme 25).39 Upon heating, complex 62 undergoes cooperative intermolecular C(sp3)–H bond activation to afford complex 63. Complex 62 was also competent in intermolecular C(sp2)–H bond activation. The reaction of 62 with pyridine proceeds rapidly and results in highly selective ortho-C–H activation of the heteroarene (64). Computational studies indicate that, in the case of C(sp3)–H activation, initial deprotonation by the Lewis basic iron center was followed by coordination of the Lewis acidic aluminium center to the resulting deprotonated CH2 fragment, leading to the formation of 63. In contrast, for pyridine activation, deprotonation at the iron centre was again the first step, after which aluminium initially coordinated to the pyridine nitrogen atom. A subsequent, energetically favourable rearrangement involving migration from an Al–N to an Al–C interaction was crucial for the formation of complex 64.
 | ||
| Scheme 25 Cooperative C(sp2)–H bond activation by an Fe–Al complex.39 | ||
Conclusions and outlook
As evident from this Frontier article, metal–ligand cooperation (MLC) has emerged as a powerful strategy for facile E–H (E = B, Si, C, Al) bond activation. The effectiveness of hemilabile ligands in MLC arises not only from their ability to promote heterolytic E–H bond cleavage, but also from the enhanced catalytic efficiency and product selectivity they provide. Fine-tuning the properties of hemilabile ligands, together with appropriate metal selection guided by the HSAB principle, is therefore crucial. Following heterolytic cleavage, insertion of E–H bonds into metal–ligand frameworks via MLC gives rise to a wide range of structurally intriguing σ-complexes featuring M–H–E three-centre-two-electron bonds, which have made significant contributions to synthetic chemistry. Although hemilabile ligand-assisted MLC in E–H (E = B, Si, and C) bond activation has grown substantially over the past few decades, main-group element-hemilabile ligand cooperation in bond activation and catalysis remains relatively underexplored.44 We sincerely hope that future developments in this area will lead to major advances in the field.Author contributions
The manuscript was written with contributions from all the authors. All authors have given their approval to the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included, and no new data were generated or analysed as part of this Frontier article.Acknowledgements
We acknowledge support from the Anusandhan National Research Foundation (ANRF), New Delhi, India, Grant No. CRG/2023/000189. S. S. thanks DST-INSPIRE for the research fellowship. S. M. thanks IIT Madras for the research fellowship.References
- (a) R. H. Crabtree, in The Organometallic Chemistry of the Transition Metals, Wiley-Interscience, New York, 6th edn, 2014 Search PubMed; (b) K. Saha, D. K. Roy, R. D. Dewhurst, S. Ghosh and H. Braunschweig, Acc. Chem. Res., 2021, 54, 1260–1273 CrossRef CAS PubMed; (c) J. H. Docherty, T. M. Lister, G. McArthur, M. T. Findlay, P. Domingo-Legarda, J. Kenyon, S. Choudhary and I. Larrosa, Chem. Rev., 2023, 123, 7692–7760 CrossRef CAS PubMed; (d) G. I. Nikonov, Si-H Bond Activation by Transition-Metal Lewis Acids, in Organosilicon Chemistry: Novel Approaches and Reactions, ed. T. Hiyama and M. Oestreich, Wiley-VCH, Weinheim, 2019, pp. 87–113 Search PubMed; (e) Z. Huang, S. Wang, R. D. Dewhurst, N. V. Ignat′ev, M. Finze and H. Braunschweig, Angew. Chem., Int. Ed., 2020, 59, 8800–8816 CrossRef CAS PubMed.
- (a) J. R. Khusnutdinova and D. Milstein, Metal-Ligand Cooperation, Angew. Chem., Int. Ed., 2015, 54, 12236–12273 CrossRef CAS PubMed; (b) T. Higashi, S. Kusumoto and K. Nozaki, Chem. Rev., 2019, 119, 10393–10402 CrossRef CAS PubMed; (c) L. Omann, C. D. F. Königs, H. F. Klare and M. Oestreich, Acc. Chem. Res., 2017, 50, 1258–1269 CrossRef CAS PubMed.
- M. D. Wodrich and X. Hu, Nat. Rev. Chem., 2017, 2, 1–7 Search PubMed.
- I. Moritani and Y. Fujiwara, Tetrahedron Lett., 1967, 8, 1119–1122 CrossRef.
- F. Forster, T. T. Metsänen, E. Irran, P. Hrobárik and M. Oestreich, J. Am. Chem. Soc., 2017, 139, 16334–16342 CrossRef CAS PubMed.
- (a) M. R. Elsby and R. T. Baker, Chem. Soc. Rev., 2020, 49, 8933–8987 RSC; (b) C. Gunanathan and D. Milstein, Acc. Chem. Res., 2011, 44, 588–602 CrossRef CAS PubMed.
- (a) M. L. H. Green, J. Organomet. Chem., 1995, 500, 127–148 CrossRef CAS; (b) M. L. H. Green and G. Parkin, J. Chem. Educ., 2014, 91, 807–816 CrossRef CAS; (c) A. Amgoune and D. Bourissou, Chem. Commun., 2011, 47, 859–871 RSC.
- R. G. Pearson, J. Chem. Educ., 1968, 581–587 CrossRef CAS.
- M. W. Drover, J. A. Love and L. L. Schafer, Chem. Soc. Rev., 2017, 46, 2913–2940 RSC.
- (a) K. Saha, R. Ramalakshmi, S. Gomosta, K. Pathak, V. Dorcet, T. Roisnel, J.-F. Halet and S. Ghosh, Chem. – Eur. J., 2017, 23, 9812–9820 CrossRef CAS PubMed; (b) U. Kaur, K. Saha, S. Gayen and S. Ghosh, Coord. Chem. Rev., 2021, 446, 214106 CrossRef CAS; (c) D. K. Roy, B. Mondal, R. S. Anju and S. Ghosh, Chem. – Eur. J., 2015, 21, 3640–3648 CrossRef CAS PubMed; (d) R. Borthakur, K. Saha, S. Kar and S. Ghosh, Coord. Chem. Rev., 2019, 399, 213021 CrossRef CAS; (e) S. K. Bose, D. K. Roy, P. Shankahari, K. Yuvaraj, B. Mondal, A. Sikder and S. Ghosh, Chem. – Eur. J., 2013, 19, 2337–2343 CrossRef CAS PubMed; (f) R. S. Anju, D. K. Roy, K. Geetharani, B. Mondal, B. Varghese and S. Ghosh, Dalton Trans., 2013, 42, 12828–12831 RSC; (g) K. Yuvaraj, D. K. Roy, K. Geetharani, B. Mondal, V. P. Anju, P. Shankhari, V. Ramkumar and S. Ghosh, Organometallics, 2013, 32, 2705–2712 CrossRef CAS.
- S.-F. Hou, J.-Y. Chen, M. Xue, M. Jia, X. Zhai, R.-Z. Liao, C.-H. Tung and W. Wang, ACS Catal., 2020, 10, 380–390 CrossRef CAS.
- (a) S. Saha, A. Haridas, F. Assanar, C. Bansal, P. K. S. Antharjanam and S. Ghosh, Dalton Trans., 2022, 51, 4806–4813 RSC; (b) S. Saha, S. Mohapatra, M. Kumar, D. Yadav and S. Ghosh, Inorg. Chem., 2025, 64, 2618–2629 CrossRef CAS PubMed.
- C. Erken, A. Kaithal, S. Sen, T. Weyhermüller, M. Hölscher, C. Werlé and W. Leitner, Manganese-catalyzed hydroboration of carbon dioxide and other challenging carbonyl groups, Nat. Commun., 2018, 9, 4521 CrossRef PubMed.
- T. Stahl, K. Mether, Y. Ohki, K. Tatsumi and M. Oestreich, J. Am. Chem. Soc., 2013, 135, 10978–10981 CrossRef CAS PubMed.
- C. C. Comanescua and V. M. Iluc, Chem. Commun., 2016, 52, 9048–9051 RSC.
- (a) K.-S. Feichtner and V. H. Gessner, Chem. Commun., 2018, 54, 6540–6553 RSC; (b) L. T. Scharf, J. Weismann, K. S. Feichtner, F. Lindl and V. H. Gessner, Chem. – Eur. J., 2018, 24, 3439–3443 CrossRef CAS PubMed.
- M. A. Rankin, K. D. Hesp, G. Schatte, R. McDonald and M. Stradiotto, Dalton Trans., 2009, 4756–4765 RSC.
- M. Zafar, A. Ahmad, S. Saha, R. Ramalakshmi, T. Roisnel and S. Ghosh, Chem. Sci., 2022, 13, 8567–8575 RSC.
- (a) M. Zafar, R. Ramalakshmi, K. Pathak, A. Ahmad, T. Roisnel and S. Ghosh, Chem. – Eur. J., 2019, 25, 13537–13546 CrossRef CAS PubMed; (b) A. Ahmad, S. Saha, M. Zafar, T. Roisnel, P. Ghosh and S. Ghosh, Eur. J. Org. Chem., 2023, e202201283 CrossRef CAS; (c) S. Gayen, F. Assanar, S. Shyamal, D. P. Dorairaj and S. Ghosh, Chem. Sci., 2024, 15, 15913–15924 RSC.
- U. Kaur, S. Gayen, H. Sharma, K. Vanka and S. Ghosh, Chem. – Eur. J., 2025, 31, e202404469 CrossRef CAS PubMed.
- K. D. Spielvogel, J. A. Luna, S. M. Loria, L. P. Weisburn, N. C. Stumme, M. R. Ringenberg, G. Durgaprasad, J. M. Keith, S. K. Shaw and S. R. Daly, Inorg. Chem., 2020, 59, 10845–10853 CrossRef CAS PubMed.
- G. P. Hatzis and C. M. Thomas, Chem. Commun., 2020, 56, 8611–8614 RSC.
- (a) S. Takemoto, T. Ito, Y. Yamazaki, M. Tsujita and H. Matsuzaka, J. Organomet. Chem., 2016, 812, 158–166 CrossRef CAS; (b) H. Song, K. Ye, P. Geng, X. Han, R. Liao, C.-H. Tung and W. Wang, ACS Catal., 2017, 7, 7709–7717 CrossRef CAS.
- M. W. Drover, L. L. Schafer and J. A. Love, Angew. Chem., Int. Ed., 2016, 55, 3181–3186 CrossRef CAS PubMed.
- S. Saha, P. Chakrabortty and S. Ghosh, Chem. – Eur. J., 2025, 31, e02125 CrossRef CAS PubMed.
- (a) K. Saha, S. Ghorai, S. Kar, S. Saha, R. Halder, B. Raghavendra, E. D. Jemmis and S. Ghosh, Angew. Chem., Int. Ed., 2019, 58, 17684–17689 CrossRef CAS PubMed; (b) S. Bairagi, S. Giri, G. Joshi, E. D. Jemmis and S. Ghosh, Angew. Chem., 2025, 137, e202417170 CrossRef.
- Z. Zhao, J. Liu, C. Tung and W. Wang, Chin. Chem. Lett., 2023, 34, 108293 CrossRef CAS.
- N. Hariharan and C. Gunanathan, Org. Lett., 2025, 27, 10348–10353 CrossRef CAS PubMed.
- T. I. Gountchev and T. D. Tilley, J. Am. Chem. Soc., 1997, 119, 12831–12841 CrossRef CAS.
- (a) Z. K. Sweeney, J. L. Polse, R. G. Bergman and R. A. Andersen, Organometallics, 1999, 18, 5502–5510 CrossRef CAS PubMed; (b) L. C. Stevenson, S. Mellino, E. Clot and P. Mountford, J. Am. Chem. Soc., 2015, 137, 10140–10143 CrossRef CAS PubMed; (c) J. Du, L. Wang, M. Xie and L. Deng, Angew. Chem., Int. Ed., 2015, 54, 12640–12644 CrossRef CAS PubMed; (d) C. P. Casey, S. W. Singer, D. R. Powell, R. K. Hayashi and M. Kavana, J. Am. Chem. Soc., 2001, 123, 1090–1100 CrossRef CAS PubMed.
- J. E. Stevens, C. E. Moore and C. M. Thomas, J. Am. Chem. Soc., 2023, 145, 794–799 CrossRef CAS PubMed.
- W.-Y. Chu, X. Zhou and T. B. Rauchfuss, Organometallics, 2015, 34, 1619–1626 CrossRef CAS.
- M. Zafar, R. Ramalakshmi, A. Ahmad, P. K. S. Antharjanam, S. Bontemps, S. Sabo-Etienne and S. Ghosh, Inorg. Chem., 2021, 60, 1183–1194 CrossRef CAS PubMed.
- D. G. A. Verhoeven, A. F. Orsino, R. L. M. Bienenmann, M. Lutz and M.-E. Moret, Organometallics, 2020, 39, 623–629 CrossRef CAS.
- T. Chu, I. Korobkov and G. I. Nikonov, J. Am. Chem. Soc., 2014, 136, 9195–9202 CrossRef CAS PubMed.
- J. A. B. Abdalla, I. M. Riddlestone, J. Turner, P. A. Kaufman, R. Tirfoin, N. Phillips and S. Aldridge, Chem. – Eur. J., 2014, 20, 17624–17634 CrossRef CAS PubMed.
- (a) T. N. Hooper, S. Lau, W. Chen, R. K. Brown, M. Garçon, K. Luong, N. S. Barrow, A. S. Tatton, G. A. Sackman, C. Richardson, A. J. P. White, R. I. Cooper, A. J. Edwards, I. J. Casely and M. R. Crimmin, Chem. Sci., 2019, 10, 8083–8093 RSC; (b) J. A. Zurakowski, B. Stadler, M. W. Drover and M. R. Crimmin, Angew. Chem., Int. Ed., 2025, 64, e202512684 CrossRef CAS PubMed.
- P. Zatsepin, T. Moriyama, C. Chen, S. Muratsugu, M. Tada and M. Yamashita, J. Am. Chem. Soc., 2024, 146, 3492–3497 CrossRef CAS PubMed.
- N. Gorgas, A. J. P. White and M. R. Crimmin, J. Am. Chem. Soc., 2022, 144, 8770–8777 CrossRef CAS PubMed.
- T. A. Engesser, M. R. Lichtenthaler, M. Schleep and I. Krossing, Chem. Soc. Rev., 2016, 45, 789–899 RSC.
- M. W. Drover, J. A. Love and L. L. Schafer, J. Am. Chem. Soc., 2016, 138, 8396–8399 CrossRef CAS PubMed.
- (a) M. A. Esteruelas, J. Herrero and L. A. Oro, Organometallics, 1993, 12, 2377–2379 CrossRef CAS; (b) A. de Aguirre, S. Díez-González, F. Maseras, M. Martín and E. Sola, Organometallics, 2018, 37, 2645–2651 CrossRef CAS.
- V. Salamanca, A. Toledo and A. C. Albeniz, J. Am. Chem. Soc., 2018, 140, 17851–17856 CrossRef CAS PubMed.
- L. Greb, F. Ebner, Y. Ginzburg and L. M. Sigmund, Eur. J. Inorg. Chem., 2020, 2020, 3030–3047 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |