A comprehensive experimental, multivariate statistical, and DFT computational analysis of the catalytic oxidation of benzyl alcohols mediated by Mn(III) and Fe(III) metalloporphyrins
Larissa
Chimilouski
a,
Sheetal
Ranaut
b,
William Henrique
Slominski
a,
Patrícia Salvador
Tessaro
c,
Gilson
DeFreitas-Silva
c,
Edmar
Martendal
a,
Cláudio N.
Verani
 d,
Shivnath
Mazumder
d,
Shivnath
Mazumder
 *b and
Fernando Roberto
Xavier
*b and
Fernando Roberto
Xavier
 *a
*a
aDepartamento de Química, Universidade do Estado de Santa Catarina (UDESC), 89.219-710, Joinville, SC, Brazil. E-mail: fernando.xavier@udesc.br
bDepartament of Chemistry, Indian Institute of Technology Jammu (IIT Jammu), Jammu 181221, India
cDepartamento de Química, Universidade Federal de Minas Gerais (UFMG), 31270-901, Belo Horizonte, MG, Brazil
dDepartment of Chemistry and Biochemistry, University of Windsor, 401 Sunset Avenue, Windsor, Ontario N9B 3P4, Canada
First published on 30th January 2026
Abstract
The oxidation of p-substituted benzyl alcohols by metallated tetraphenylporphyrin catalysts [MnIII(TPP)Cl] (1) and [FeIII(TPP)Cl] (2) in the presence of the oxidizing agent (diacetoxyiodo)benzene (DAIB) was evaluated by means of an integrated approach considering the experimental results under the light of a three-level full factorial design (FFD) and density functional theory (DFT) analysis. Aiming to promote and maximize the formation of benzaldehyde and benzoic acid, the use of multivariate analysis allows us to study the interactivity and interplay between critical variables such as catalyst load (mol%) and time (minutes). On the one hand, benzaldehyde formation was optimized when 4.0 mol% of 1 catalyzed the reaction for 90 minutes or when 2.8 mol% of 2 catalyzed the reaction for 100 minutes. On the other hand, benzoic acid formation was favored when 5.0 mol% of 1 was used over 90 minutes or 4.8 mol% of 2 over 60 minutes. Several control tests were performed to verify the catalytic nature of the metalloporphyrins, including the use of butylated hydroxytoluene to confirm the presence of radical species involved in the catalytic process. The possible mechanisms as well as the electronic structures of the main reaction intermediates were assessed by means of DFT calculations and benchmarked against the experimental data and available literature. Catalysts 1 and 2 display subtle but distinct mechanistic pathways that explain the observed differences in chemical reactivity. Additionally, different linear free energy relationships associated with distinct spin densities of transition states were observed during the determinant step when p-substituted R-benzyl alcohol substrates containing R = OMe, H, Cl, NO2 were considered.
Introduction
The market value of benzaldehyde (BA), used in food and pharma industries, is nearly $610 million and is expected to grow to $970 million in the next 10 years.1 Therefore, the oxidation of benzyl alcohols (BnOH) into benzaldehyde (BA) prompts considerable interest, but shows limited selectivity with the co-formation of benzoic acid (BzA) and other carboxylic acid products.2Metalloporphyrins are versatile catalysts for the bromination3 and oxidation of hydrocarbons,4 production of solar fuels and degradation of pollutants,5 and reduction of CO2 and oxygen6 and have proven effective for alcohol oxidation in organic media7–12 with oxidants such as iodosylbenzene8 (PhIO), (diacetoxyiodo)benzene9 (DAIB), tert-butyl hydroperoxide13 (t-BuOOH), and hydrogen peroxide14 (H2O2), among others.7,15–17 This versatility of metalloporphyrins towards the oxidation of organic substrates18–20 is related to the formation of high-valent species that take advantage of strong σ-donation to empty metal orbitals increasing the ligand field strength, π-delocalization of radicals that spreads charge across the macrocycle, and synergistic metal–oxo bonding that combines σ- and π-interactions to support reactive M![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O species.21–24 Understanding how each of these contributions influences the reaction intermediates and products is key to the design of effective catalysts, and the selection of the metal, and by extension its electronic configuration, offers the most straightforward means of control. The 3d4 manganese(III) and 3d5 iron(III) ions, and to a lesser extent the 3d7 cobalt(II) ion, are often selected due to their ability to form M
O species.21–24 Understanding how each of these contributions influences the reaction intermediates and products is key to the design of effective catalysts, and the selection of the metal, and by extension its electronic configuration, offers the most straightforward means of control. The 3d4 manganese(III) and 3d5 iron(III) ions, and to a lesser extent the 3d7 cobalt(II) ion, are often selected due to their ability to form M![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds, and benzyl alcohol oxidation using the 3d4 system indeed showed dependence on the axial ligand present.9 The nature of the oxidant and the electronic structure of the organic substrate are additional factors that add complexity.25,26
O bonds, and benzyl alcohol oxidation using the 3d4 system indeed showed dependence on the axial ligand present.9 The nature of the oxidant and the electronic structure of the organic substrate are additional factors that add complexity.25,26
[MnIII(TPP)Cl] and [FeIII(TPP)Cl] (Scheme 1) are the simplest and cheapest metalloporphyrin catalysts studied by the scientific community. Furthermore, these complexes have already shown activity for the oxidation of benzyl alcohol.15,17 However, these studies were conducted using a univariate set of experiments that often fail to identify the ideal reaction conditions. Thus, the use of multivariate statistical analyses may be an interesting strategy to carry out the oxidation reaction of benzyl alcohol, aiming to obtain benzaldehyde with the highest possible yield.
Recently, collaborative work by the groups of Martendal, Peralta, and Xavier27 has yielded a series of successful examples for the application of multivariate statistical analyses such as Box–Behnken and Full Factorial designs28 to the oxidation of BnOH. These statistical methods use a response surface methodology to explore efficiently the relationships between multiple variables and a response. These designs allow for the estimation of linear, interaction, and quadratic effects, thereby enhancing experimental efficiency in the optimization of complex chemical processes. We have presented the first report27 on Box–Behnken design, optimizing the catalytic oxidation of cyclohexane and benzyl alcohol with 3d5 iron(III) and 3d9 copper(II) complexes. We expanded the approach in collaboration with Nordlander et al.29 to optimize the oxidation of cyclohexane by using a 3d6 iron(II) N4Py catalyst and with Verani et al.30 to elucidate the role of nuclearity in the oxidation of benzyl alcohol with mono and bimetallic copper(II) catalysts. The work on copper(II) catalysts was recently expanded by Martendal, Xavier et al.,31 successfully describing the benzyl alcohol oxidation in homo- and heterogeneous media.
With multivariate approaches, it is possible to understand how the conversion of a substrate into product(s) can be optimized by diverse catalysts, and when combined with density functional calculations, new opportunities arise to investigate their molecular mechanisms. Collaborative theoretical and experimental studies by the groups of Mazumder, Schlegel and Verani developed a robust body of work focused on the mechanisms of water splitting promoted by HS/LS3d7 cobalt(II),32–35LS3d6 cobalt(III),36,37 3d9 copper(II),38,39 3d8 nickel(II), and 4d6/3d8 ruthenium/nickel(II) molecular catalysts.40,41
This work integrates the statistical rigor of a three-level full factorial design with the mechanistic insight afforded by DFT calculations, delivering a comprehensive analysis of the oxidation of p-substituted benzyl alcohols to benzaldehydes and benzoic acids catalyzed by the established metalloporphyrin systems [MnIII(TPP)Cl] (1) and [FeIII(TPP)Cl] (2) in the presence of (diacetoxyiodo)benzene (DAIB) (Scheme 1).
Results and discussion
Both [MnIII(TPP)Cl] (1) and [FeIII(TPP)Cl] (2) are staple catalysts selected due to their ease of preparation and versatility. The oxidant DAIB was chosen because it is soluble in most organic solvents, safe, easy to handle, and commercially available, making it an appealing option for developing selective oxidation reactions.42–44 DCM was ultimately selected as the solvent due to its excellent solubility for the catalysts and because previous studies report its superior performance in metalloporphyrin-catalysed oxidation of benzyl alcohol.9 All substrates, including benzyl alcohol, p-nitrobenzyl alcohol, p-methoxybenzyl alcohol, p-chlorobenzyl alcohol, benzaldehyde, anisaldehyde, p-chlorobenzaldehyde, p-nitrobenzaldehyde, benzoic acid, p-methoxybenzoic acid, p-chlorobenzoic acid, and p-nitrobenzoic acid, were purchased from commercial sources.Catalytic oxidation of benzyl alcohol
The catalytic activity of [MnIII(TPP)Cl] (1) and [FeIII(TPP)Cl] (2) was initially evaluated using benzyl alcohol (BnOH) as the organic substrate, (diacetoxy)iodobenzene (DAIB) as the oxidizing agent and dichloromethane as the reaction solvent. The products quantified were benzaldehyde (BA) and benzoic acid (BzA). Preliminary tests were carried out with [MnIII(TPP)Cl] (1) or [FeIII(TPP)Cl] (2) to evaluate product formation over time. Three different catalyst loads were used (0.1 mol%, 5 mol% and their average amount of 2.45 mol%) over a time interval of 2–120 minutes (Fig. 1 and Fig. S1).For [MnIII(TPP)Cl] (1) (Fig. 1), the yield increased gradually, reaching saturation close to 120 minutes for BA and BzA. Also, when 2.45 mol% and 5.00 mol% of catalyst load were employed, the final yield values were similar to each other, indicating that a large amount of catalyst (5 mol%) will not greatly improve the BA production. However, when BzA is considered, the increments in the catalyst load are decisive. A similar pattern could be observed for complex 2 (Fig. S1), but with saturation occurring in a shorter time (approximately 30 minutes for 0.1 mol% and 5 mol% and 120 minutes for 2.45 mol%) with overall yields substantially lower than that for complex 1, for both BA and BzA.
Based on these data, the three-level full factorial design (FFD) was established as depicted in Table 1, with the yield of products calculated using the calibration curves previously built. The results generated were processed using Statistica 7 software, thus obtaining the response surfaces and evaluating the reaction conditions.
| Entry | Catalyst (mol%) | Time (min) | Yield (%) | |||
|---|---|---|---|---|---|---|
| 1 | 2 | |||||
| BA | BzA | BA | BzA | |||
| n.d. = not detected. Conditions: excess DAIB (equivalent to an average mass of 60 mg – 182.6 μmol), 20 μmol of substrate, and dichloromethane added to a final volume of 3 mL. The products (BA and BzA) were calculated using calibration curves based on data extracted from HPLC-DAD analysis. | ||||||
| 1 | 2.45 | 60 | 17.20 | 4.30 | 4.52 | 2.59 |
| 2 | 5.00 | 60 | 19.10 | 8.24 | 5.61 | 4.69 |
| 3 | 0.10 | 5 | 3.77 | n.d. | 0.67 | n.d. |
| 4 | 0.10 | 60 | 11.30 | 0.66 | 0.96 | 0.16 |
| 5 | 5.00 | 120 | 22.00 | 8.24 | 5.86 | 4.83 |
| 6 | 2.45 | 5 | 6.07 | 2.27 | 3.07 | 2.21 |
| 7 | 5.00 | 5 | 6.70 | 4.69 | 5.01 | 4.12 |
| 8 | 0.10 | 120 | 11.90 | 0.82 | 1.07 | 0.25 |
| 9 | 2.45 | 120 | 25.90 | 4.70 | 5.22 | 2.59 |
| 10 | 2.45 | 60 | 25.70 | 4.52 | 4.63 | 2.62 |
| 11 | 2.45 | 60 | 27.86 | 4.35 | 4.51 | 2.56 |
| 12 | 2.45 | 60 | 27.08 | 4.63 | 4.54 | 1.94 |
The value of the coefficient of determination (R2) found for the model was better than 0.98 for the formation of oxidation products.
These results indicate that surface models are adequate, as more than 98% of the variation in results can be explained by the mathematical model and residues (Fig. S2 and S3), which have a random distribution.
Fig. 2 shows the response surfaces generated for BA and BzA production for [MnIII(TPP)Cl] (1) and [FeIII(TPP)Cl] (2), obtained through the three-level full-factorial design. For all the variables herein studied, it was clearly possible to identify the regions of higher BA and BzA production.
For [MnIII(TPP)Cl] (1) (Fig. 2A and B), locally optimized values for BA yield were concentrated in the region of the maximum catalyst amount (4.00 mol%) and with a reaction time of 90 minutes, and for the formation of BzA, it was 5.00 mol% catalyst and 90 minutes of reaction. The ideal regions for the two products are very similar, although the selectivity is around 77% for the formation of BA (Table 2, entry 1). The Pareto chart (Fig. 3A and B) shows the significance of each one of the linear and quadratic terms of the mathematical model generated, and the catalyst load and the reaction time were significant (p > 0.05).
| Entry | Complex | Catalyst (mol%) | Time (min) | Yield % (±sd.) | TONf (103) | TOFg min−1 (103) | Selectivity % | |||
|---|---|---|---|---|---|---|---|---|---|---|
| BA | BzA | BA | BzA | BA | BzA | |||||
| a 4.00 mol% for BA and 5.00 mol% for BzA. b 2.75 mol% for BA and 4.75 mol% for BzA. c 100 min for BA and 60 min for BzA. d Iron salt, FeCl3·6H2O. e Benzaldehyde was used as the substrate. f TON, turnover number, refers to the ratio of mol of a product (BA/BzA)/mol of the catalyst. g TOF, turnover frequency, refers to TON/time (min−1); n.d. = not detected; sd = standard deviation (n = 3). Selectivity BA [= 100% × BA/(BA + BzA)]. h Control test without BHT used as a radical scavenger. i Control test with BHT used as a radical scavenger. | ||||||||||
| 1 | 1 | 4.00–5.00a | 90 | 23.8 ± 1.5 | 6.68 ± 0.42 | 194 | 40.8 | 2.2 | 0.4 | 78 |
| [25.9 ± 1.1] | [8.43 ± 0.14] | |||||||||
| 2 | 2 | 2.75–4.75b | 100–60c | 5.22 ± 0.10 | 4.39 ± 0.44 | 61.4 | 27.4 | 0.6 | 0.5 | 54 |
| [4.80 ± 0.06] | [4.39 ± 0.37] | |||||||||
| 3 | — | — | 60 | 0.61 ± 0.18 | n.d. | 100 | ||||
| 4 | S | 2.45 | 60 | 1.82 ± 0.03 | n.d. | 100 | ||||
| 5 | —e | — | 60 | — | 0.55 ± 0.05 | 100 | ||||
| 6 | 1 | 2.45 | 60 | — | 21.3 ± 1.3 | 100 | ||||
| 7 | 2 | 2.45 | 60 | — | 2.07 ± 0.19 | 100 | ||||
| 8 | 1 | 2.45 | 60 | 26.8 ± 1.5 | 4.50 ± 0.14 | 86 | ||||
| 9 | 2 | 2.45 | 60 | 4.52 ± 0.03 | 1.87 ± 0.16 | 71 | ||||
| 10 | 1 | 2.45 | 60 | 3.50 ± 0.42 | 0.160 ± 0.012 | 96 | ||||
| 11 | 2 | 2.45 | 60 | 0.76 ± 0.07 | 0.120 ± 0.012 | 86 | ||||
Analyzing Fig. 3A, it is possible to see that the linear and quadratic coefficients related to catalyst load are statistically higher than the experimental error, while the reaction time alone was not, but the interaction between reaction time and catalyst load had a significant influence. As observed in Fig. 3B, the linear term for the catalyst was the most influential, followed by the linear and quadratic terms involving the reaction time, confirming the shape of the response surface. The interaction between the two variables was also significant, confirming the need for a multivariate study of those variables. For this catalytic system, the two variables were shown to be prominent for both products.
When [FeIII(TPP)Cl] (2) was considered, the response surfaces are as illustrated in Fig. 2C and D. The optimum values can be attributed as 100 minutes of reaction and 2.8 mol% for BA. The surface shows a curvature, indicating that in this region the local optimum is found. For BzA, a surface without curvature is observed, indicating that the range studied included a local rather than a global maximum. However, catalyst loads higher than 5 mol% were not applied in this study. It was possible to delimit a local optimum in the red region of the graph, being 4.8 mol% of catalyst and a reaction time of 60 minutes. When analyzing the effect of reaction time in the range investigated (5–120 min), it proved to be statistically not important in the formation of BA and BzA for complex 2, whereas for complex 1, the reaction time had a greater influence on the formation of the products, as already discussed. Analyzing Fig. 3C and D, the linear and quadratic parameters relating to the catalyst are significant (p > 0.05) for BA, confirming the shape of the surface.
Fig. 2C shows that both linear and quadratic terms related to reaction time were relevant, which confirms the surface curvature. Additionally, the quadratic term of catalyst load was significant too, leading to a local maximum of this variable. For the formation of BzA (Fig. 3D), only the linear term of catalyst load was important, and no interaction between reaction time and catalyst load had an appreciable influence. Thus, this result suggests that the catalytic oxidation occurs sequentially, from BOH to BA and then from BA to BzA, as the response surface denotes that more than 5 mol% is needed to convert more BA into BzA.
Considering the data presented, the ranges of variables studied to maximize the amount of BA and BzA were delimited through multivariate analysis. As already mentioned, the catalyst and reaction time were obtained directly from the response surface shown in Fig. 2 for complexes 1 and 2. The maximized yield results are reported in Table 2. The values for the optimum region of 1 and 2 (Table 2, entries 1 and 2) were measured experimentally and the values in the brackets are the predicted values from the mathematical model. A good agreement between the predicted and experimental values is observed which indicates an excellent accuracy of our prediction model for the formation of the products using the two complexes. The observed selectivity was higher for complex 1 compared to 2, with 77% and 54%, respectively. The turnover number (TON) and turnover frequency (TOF) show the highest catalytic activity for 1, with a 3-fold increase in the number of cycles for BA formation. Both values demonstrate the better performance of the manganese complex 1 over the iron complex 2 in the formation of BA and BzA.
Additional experiments were carried out to gain a better understanding of the product yields found for [MnIII(TPP)Cl] (1) and [FeIII(TPP)Cl] (2), and Table 2 summarizes the tests carried out.
The first experiment (Table 2, entry 3) involved investigating the dependence of the catalyst on the reaction medium. Residual yields were quantified for BA and no BzA was quantified, indicating that the catalysts are essential for the oxidation of BnOH. To evaluate the role of the ligand in the complexes, an experiment was carried out in triplicate using FeCl3·6H2O (Table 2, entry 4) and its result was compared to that of the same experiment using complex 2. The yield of BA was found to drop by 65% in the presence of FeCl3·6H2O. This indicates the relevance of the ligand in stabilizing the active species during the catalytic process. Manganese salts (MnCl2·4H2O and MnSO4·H2O) were tested, but they were not soluble in the reaction medium studied. The free TPP ligand was also employed as the potential catalyst under the same experimental conditions as for the free metal ions (entry 4). No appreciable product formation within the quantification levels was observed.
Experiments were performed in which no catalyst was used for the oxidation of BA as a substrate to form BzA (Table 2, entry 5), where residual BzA amounts were quantified. This is a strong indication that even for this system, the use of a catalyst is necessary.
When the metal complexes were introduced using BA as a substrate, relevant results were observed. For complex 1 (Table 2, entry 6), around 21% yield of BzA was observed, while for complex 2 (Table 2, entry 7), only 2% yield was obtained. This fact reinforces the idea of higher catalytic activity of complex 1 over 2. Based on this set of experiments, it is possible to infer that each step of the sequential oxidation from BnOH → BA → BzA is mostly mediated by metal complexes 1 and 2.
In order to probe whether radical species were involved during the catalytic oxidation of BnOH promoted by complexes 1 and 2, parallel control tests were performed in the absence (entries 8 and 9) and presence (entries 10 and 11) of 2-tert-butyl-6-methyl-phenol (BHT), a classical radical scavenger. For both complexes 1 and 2, the production of BA and BzA was strongly affected where reaction yields dropped by 87% and 83% for BA as well as 96% and 94% for BzA. These results confirmed that a radical species is involved during the catalysis.
Considering the literature on catalytic oxidation involving metalloporphyrins, manganese porphyrins have been extensively used towards several organic substrates and generally higher yield values are reported compared to porphyrins with other metal centers. For example, Ahmad et al.45 carried out oxidation of styrene using manganese and iron porphyrin catalysts, where the conversion in the case of manganese was 98% and higher than that in the case of iron, which was 62%.
When evaluating the metalloporphyrin-catalyzed oxidation of benzyl alcohol, it is observed that the yield and selectivity of the products depend on the reaction conditions used (Table 3). Therefore, a direct comparison with the results obtained in our work is difficult. However, when comparing the results obtained with those reported by Karimipour et al.,9 a slightly higher yield is observed for the system presented in our work. It is important to highlight that many studies in the literature use co-catalysts to increase the reaction yield.9,16,17 However, this was not the focus of our work, although it shows a way to improve the catalytic activity of the complexes studied. Furthermore, it is observed that increasing the temperature may be an important variable to be evaluated in the oxidation of this substrate.
| Product yield (%) | Molar ratio Cat.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Subst. Subst.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Oxi. Oxi. |
Solvent | Oxidant | Temperature (°C) | Time (h) | Metalloporphyrin | Ref. | |
|---|---|---|---|---|---|---|---|---|
| BA | BzA | |||||||
| DCM = dichloromethane.a Room temperature (RT) was not specified in the article.b O2 bubbling (1 atm) and isobutyraldehyde (3 mmol) as a co-catalyst.c O2 bubbling (1 atm) and isobutyraldehyde (6 mmol) as a co-catalyst.d Co-metalloporphyrin was supported on silica nanoparticles; thus, the amount of catalyst was estimated by quantifying the cobalt through atomic absorption as indicated in the article. | ||||||||
| 26.80 | 4.50 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 41 41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 373 373 |
DCM | PhI(OAc)2 | 20 | 1 | [MnIII(TPP)Cl] | This work |
| 4.52 | 1.87 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 41 41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 373 373 |
DCM | PhI(OAc)2 | 20 | 1 | [FeIII(TPP)Cl] | This work |
| ∼20 | — | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 55 55 |
DCM | PhI(OAc)2 | RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a a |
1 | [MnIII(TPP)CN] | 9 |
| 43 | — | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 190 190 |
DCM | n-Bu4NHSO5 | 25 | 48 | [MnIII(TPP)OAc] | 7 |
| 33 | 12 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1000 1000![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) X X![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b b |
Benzotrifluoride | O2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b b |
60 | 0.5 | [MnIII(TPP)Cl] | 15 |
| 32 | — | [FeIII(TPP)Cl] | ||||||
| 42 | 58 | [CoIII(TPP)Cl] | ||||||
| 99 | — | [RuIII(TPP)Cl] | ||||||
| 94 | — | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
Water | n-Bu4NHSO5 | 60 | 0.25 | [FeIII(TPP)Cl] | 12 |
| 80.8 | — | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1000 1000![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) X X![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
o-Xylol | O2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
60 | — | [CuII(TPP)] | 46 |
| 48.7 | 1 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 935 935![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1402 1402![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) d d |
DCM | H2O2 | 70 | 5 | [CoIII(TPP)Cl] | 47 |
Electronic structures of the active species
For complexes [MnIII(TPP)Cl] (1) and [FeIII(TPP)Cl] (2), the electronic structures of their plausible active species were modeled by DFT studies. In this case, the active species [MnV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O(TPP)(Cl)]0 was proposed, with two possible spin configurations for the 3d2 MnV center, namely, low-spin singlet (S = 0) and high-spin triplet (S = 2/2). The singlet state is lower in energy (ΔG) compared to the triplet state by 2.0 kcal mol−1, giving rise to the low-spin ground state. The calculated result is in agreement with the diamagnetic ground state, reported by Nam et al.,48 for the species [MnV
O(TPP)(Cl)]0 was proposed, with two possible spin configurations for the 3d2 MnV center, namely, low-spin singlet (S = 0) and high-spin triplet (S = 2/2). The singlet state is lower in energy (ΔG) compared to the triplet state by 2.0 kcal mol−1, giving rise to the low-spin ground state. The calculated result is in agreement with the diamagnetic ground state, reported by Nam et al.,48 for the species [MnV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O(TPP)(Cl)]0. The spin density plot for the excited triplet state is shown in Fig. 4 (top). It is a bona fide metal-centered state with the MnV metal having a Mulliken spin density (MSD) value of 1.8. This result supports the presence of two unpaired electrons on the metal center in the triplet state.
O(TPP)(Cl)]0. The spin density plot for the excited triplet state is shown in Fig. 4 (top). It is a bona fide metal-centered state with the MnV metal having a Mulliken spin density (MSD) value of 1.8. This result supports the presence of two unpaired electrons on the metal center in the triplet state.
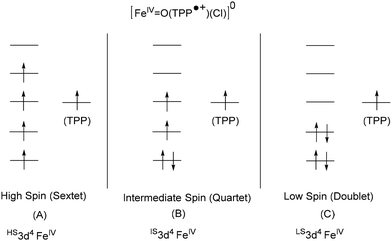
For [FeIII(TPP)Cl] (2), the iron(IV)-oxo porphyrin radical species [FeIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O(TPP˙+)(Cl)]0 was considered. This species can have three possible electronic states, as reported in Scheme 2. These are high-spin sextet (S = 5/2) with HS3d4 FeIV, intermediate-spin quartet (S = 3/2) with IS3d4 FeIV, and low-spin doublet (S = 1/2) with LS3d4 FeIV metal centers. Each of these states contains a porphyrin radical cation TPP˙+. DFT calculations reveal that the quartet state S = 3/2 is lowest in energy and is the ground electronic state. The sextet state S = 5/2 is significantly higher in energy (ΔG) than the quartet by 20.5 kcal mol−1 while the doublet state S = 1/2 is marginally higher than the quartet state by less than 1 kcal mol−1. This small difference does not allow us to rule out the doublet state but moderately favors the quartet state. These results are in agreement with the literature findings where the quartet state was confirmed to be the ground state.49,50 The spin density plot of the ground state is calculated and shown in Fig. 4 (bottom). The Mulliken spin density (MSD) values are 1.5 on the Fe metal, 0.8 on the O atom of the oxo fragment, and 0.6 on the porphyrin ligand framework. The MSD value on the O atom indicates a significant delocalization of the spin from the iron center to the oxo unit.
O(TPP˙+)(Cl)]0 was considered. This species can have three possible electronic states, as reported in Scheme 2. These are high-spin sextet (S = 5/2) with HS3d4 FeIV, intermediate-spin quartet (S = 3/2) with IS3d4 FeIV, and low-spin doublet (S = 1/2) with LS3d4 FeIV metal centers. Each of these states contains a porphyrin radical cation TPP˙+. DFT calculations reveal that the quartet state S = 3/2 is lowest in energy and is the ground electronic state. The sextet state S = 5/2 is significantly higher in energy (ΔG) than the quartet by 20.5 kcal mol−1 while the doublet state S = 1/2 is marginally higher than the quartet state by less than 1 kcal mol−1. This small difference does not allow us to rule out the doublet state but moderately favors the quartet state. These results are in agreement with the literature findings where the quartet state was confirmed to be the ground state.49,50 The spin density plot of the ground state is calculated and shown in Fig. 4 (bottom). The Mulliken spin density (MSD) values are 1.5 on the Fe metal, 0.8 on the O atom of the oxo fragment, and 0.6 on the porphyrin ligand framework. The MSD value on the O atom indicates a significant delocalization of the spin from the iron center to the oxo unit.
The corresponding molecular orbitals are calculated to confirm the nature of the electronic state, and their plots are shown in Fig. 5. Molecular orbital (MO) 169 is a 3dxy orbital on Fe and is doubly filled. The MOs 186α and 187α are singly occupied and can be considered as SOMOs. These SOMOs are an antibonding combination between 3dπ orbitals of Fe and 2pπ orbitals of the oxygen atom of the oxo fragment. The SOMOs are indicative of metal-to-oxo ligand π back-donation. Therefore, the calculated SOMOs are consistent with the spin density results supporting delocalization of charge from the metal center to the oxo moiety. The 3dx2−y2 (190α) and 3dz2 (191α) orbitals are unoccupied. The π* SOMO, 185α, is singly occupied and predominantly centered on the porphyrin ligand framework, supporting the presence of TPP˙ in the electronic structure.
Mechanistic insights
Mechanistic investigations like 18O-labelling, deuterium isotope effect, and linear free energy relationship analyses have indicated that the alcohol oxidation happens via the following steps: (a) α-H atom abstraction by the high-valent metal–oxo complex leading to the formation of the α-hydroxy radical intermediate, (b) an oxygen rebound step to the gem-diol, and (c) dehydration of the gem-diol to the corresponding carbonyl product.51It has been further reported that the rebound step (b) involving the α-hydroxy radical follows a barrierless pathway.51 Therefore, in the present study, we have concentrated on the first step (a), α-H atom abstraction by the high-valent metal–oxo complex. The detailed energetics of steps (b) and (c) can be found in Fig. S4. The calculated free energy profiles for the hydrogen abstraction event by [MnV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O(TPP)(Cl)]0 and [FeIV
O(TPP)(Cl)]0 and [FeIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O(TPP˙+)(Cl)]0 complexes are, respectively, shown in the left and right sides of Fig. 6.
O(TPP˙+)(Cl)]0 complexes are, respectively, shown in the left and right sides of Fig. 6.
The ground state (i) for the [MnV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O(TPP)(Cl)]0 complex is the low-spin singlet. The excited triplet state (ii) is only 2 kcal mol−1 higher in energy than the ground singlet state (Fig. 6, left). The calculated low singlet–triplet energy gap is consistent with a previous study by Harvey et al.52 The triplet electronic state (ii) can undergo a facile α-hydrogen abstraction from the substrate benzyl alcohol.16 The overall process, (ii) → (iii), can be conceptualized in the following way: a homolytic cleavage of the MnV
O(TPP)(Cl)]0 complex is the low-spin singlet. The excited triplet state (ii) is only 2 kcal mol−1 higher in energy than the ground singlet state (Fig. 6, left). The calculated low singlet–triplet energy gap is consistent with a previous study by Harvey et al.52 The triplet electronic state (ii) can undergo a facile α-hydrogen abstraction from the substrate benzyl alcohol.16 The overall process, (ii) → (iii), can be conceptualized in the following way: a homolytic cleavage of the MnV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond in triplet (S = 1) [MnV
O bond in triplet (S = 1) [MnV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O(TPP)(Cl)]0 (ii) leads to transient MnIV–O(˙) which subsequently reacts with the α-H of the benzyl alcohol substrate giving rise to the α-hydroxy radical and quartet (S = 3/2) [MnIV–OH(TPP)(Cl)]0 (iii). The high-spin 3d2 MnV center in triplet [MnV
O(TPP)(Cl)]0 (ii) leads to transient MnIV–O(˙) which subsequently reacts with the α-H of the benzyl alcohol substrate giving rise to the α-hydroxy radical and quartet (S = 3/2) [MnIV–OH(TPP)(Cl)]0 (iii). The high-spin 3d2 MnV center in triplet [MnV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O(TPP)(Cl)]0 (ii) gets reduced to high-spin 3d3 MnIV in quartet [MnIV–OH(TPP)(Cl)]0 (iii). The transition state for hydrogen atom abstraction (ii-TS) is located at only 5.2 kcal mol−1 and is thermally very accessible. The hydrogen activation step, (i) → (iii), is overall thermodynamically favorable by 12.4 kcal mol−1. A similar mechanism was proposed by Karimipour and co-workers9 where compatible conditions (dichloromethane and DAIB as the oxidant) were employed.
O(TPP)(Cl)]0 (ii) gets reduced to high-spin 3d3 MnIV in quartet [MnIV–OH(TPP)(Cl)]0 (iii). The transition state for hydrogen atom abstraction (ii-TS) is located at only 5.2 kcal mol−1 and is thermally very accessible. The hydrogen activation step, (i) → (iii), is overall thermodynamically favorable by 12.4 kcal mol−1. A similar mechanism was proposed by Karimipour and co-workers9 where compatible conditions (dichloromethane and DAIB as the oxidant) were employed.
The ground state (iv) for [FeIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O(TPP˙+)(Cl)]0 is a tri-radicaloid quartet state (S = 3/2) with two unpaired electrons on the FeIV center and one on the porphyrin moiety (Fig. 6, right). This quartet electronic state (iv) can abstract α-hydrogen from the substrate benzyl alcohol to form the complex [FeIV–OH(TPP)(Cl)]0 (v) traversing through the transition state (iv-TS). The transition state (iv-TS) has a barrier of 8.1 kcal mol−1. The conversion from (iv) to (v) can be outlined in the following manner: the homolytic cleavage of the FeIV
O(TPP˙+)(Cl)]0 is a tri-radicaloid quartet state (S = 3/2) with two unpaired electrons on the FeIV center and one on the porphyrin moiety (Fig. 6, right). This quartet electronic state (iv) can abstract α-hydrogen from the substrate benzyl alcohol to form the complex [FeIV–OH(TPP)(Cl)]0 (v) traversing through the transition state (iv-TS). The transition state (iv-TS) has a barrier of 8.1 kcal mol−1. The conversion from (iv) to (v) can be outlined in the following manner: the homolytic cleavage of the FeIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond in [FeIV
O bond in [FeIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O(TPP˙+)(Cl)]0 (iv) forms the transient FeIV–O(˙) species which subsequently reacts with the α-H of the benzyl alcohol substrate giving rise to the α-hydroxy radical and triplet (S = 1) [FeIV–OH(TPP)(Cl)]0 (v). The step, (iv) to (v), is downhill by 6.6 kcal mol−1 and it involves the reduction of the radical cation TPP˙+ in (iv) to the neutral TPP moiety in (v). The 3d2 FeIV center retains its oxidation state from (iv) to (v). Similar iron porphyrin-based catalytically active species have been described in the literature by Nam and co-workers.53
O(TPP˙+)(Cl)]0 (iv) forms the transient FeIV–O(˙) species which subsequently reacts with the α-H of the benzyl alcohol substrate giving rise to the α-hydroxy radical and triplet (S = 1) [FeIV–OH(TPP)(Cl)]0 (v). The step, (iv) to (v), is downhill by 6.6 kcal mol−1 and it involves the reduction of the radical cation TPP˙+ in (iv) to the neutral TPP moiety in (v). The 3d2 FeIV center retains its oxidation state from (iv) to (v). Similar iron porphyrin-based catalytically active species have been described in the literature by Nam and co-workers.53
A comparison of the calculated free energy profiles between the Mn-system (Fig. 6, left) and the Fe-system (Fig. 6, right) shows that the kinetic barrier for hydrogen abstraction by [MnV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O(TPP)(Cl)]0 is 2.9 kcal mol−1 lower than that for hydrogen abstraction by [FeIV
O(TPP)(Cl)]0 is 2.9 kcal mol−1 lower than that for hydrogen abstraction by [FeIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O(TPP˙+)(Cl)]0. In addition, the hydrogen abstraction step is thermodynamically more downhill by 5.8 kcal mol−1 for Mn than in the case of Fe. Thus, our calculated results indicate that α-H atom abstraction by the high-valent metal–oxo complex is both kinetically and thermodynamically favorable for Mn than for Fe. These results are in total agreement with the experimental findings.
O(TPP˙+)(Cl)]0. In addition, the hydrogen abstraction step is thermodynamically more downhill by 5.8 kcal mol−1 for Mn than in the case of Fe. Thus, our calculated results indicate that α-H atom abstraction by the high-valent metal–oxo complex is both kinetically and thermodynamically favorable for Mn than for Fe. These results are in total agreement with the experimental findings.
The MnV center in [MnV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O(TPP)(Cl)]0 (ii) gets reduced to MnIV in [MnIV–OH(TPP)(Cl)]0 (iii). The α-H atom abstraction is associated with MnV/MnIV reduction and the TPP ligand behaves as a redox innocent platform. This is in sharp contrast to the case of the Fe-system where the TPP˙+ radical cation in [FeIV
O(TPP)(Cl)]0 (ii) gets reduced to MnIV in [MnIV–OH(TPP)(Cl)]0 (iii). The α-H atom abstraction is associated with MnV/MnIV reduction and the TPP ligand behaves as a redox innocent platform. This is in sharp contrast to the case of the Fe-system where the TPP˙+ radical cation in [FeIV![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O(TPP˙+)(Cl)]0 (iv) gets reduced to a neutral TPP fragment in [FeIV–OH(TPP)(Cl)]0 (v). Therefore, the α-H atom abstraction is associated with TPP˙+/TPP reduction in the case of Fe. The TPP ligand behaves as a redox non-innocent moiety. The FeIV center is redox innocent since there is no change of the metal oxidation state in the hydrogen abstraction event. The MnV/MnIV reduction is more favorable than TPP˙+/TPP reduction, resulting in better catalytic activity of the Mn-system than that of Fe.
O(TPP˙+)(Cl)]0 (iv) gets reduced to a neutral TPP fragment in [FeIV–OH(TPP)(Cl)]0 (v). Therefore, the α-H atom abstraction is associated with TPP˙+/TPP reduction in the case of Fe. The TPP ligand behaves as a redox non-innocent moiety. The FeIV center is redox innocent since there is no change of the metal oxidation state in the hydrogen abstraction event. The MnV/MnIV reduction is more favorable than TPP˙+/TPP reduction, resulting in better catalytic activity of the Mn-system than that of Fe.
Furthermore, this detailed DFT investigation identified the effect of the acetate ion present in the solution on the catalytic activity of the metal complexes once these anions are generated in solution from the DAIB oxidant agent. Detailed energetics is shown in Fig. S5. The Fe-complex has a greater propensity towards binding an acetate ion than the Mn-complex, resulting in the formation of an unproductive intermediate which is detrimental to the catalytic turnover of the Fe-complex.
Catalytic oxidation of p-substituted benzyl alcohols
To expand the scope of the applicability of Fe(III) and Mn(III) porphyrins in catalytic oxidation studies, a series of benzyl alcohol substrates containing electron-donor and electron-withdrawing groups were investigated using DAIB as the oxidizing agent.Different optimized conditions were obtained for [MnIII(TPP)Cl] (1) and [FeIII(TPP)Cl] (2), by means of multivariate analysis. Therefore, the same experimental conditions were chosen for both metal complexes to better compare their catalytic activity. Table 4 (entries 1–4) shows the yields and selectivity for the substrates for [MnIII(TPP)Cl] (1). For p-methoxybenzyl alcohol, the yield of its aldehyde was the highest in the study (28.95 ± 0.77%), in contrast to p-nitrobenzyl alcohol which had the lowest aldehyde production (12.34 ± 2.83%). Benzyl alcohol and p-chlorobenzyl alcohol showed intermediate yields. As a result, the BnOH → BA substrate-to-product yield followed the order: OMe > H > Cl > NO2. The same trend was observed for the BA → BzA yield of each p-substituted benzyl alcohol, but in lower quantities (Table 4, entries 1–4). The p-methoxybenzyl alcohol seemed to be the most activated substrate, confirmed by the highest TON and TOF values when compared to the other substrates (Table 4, entry 1). However, as the electron-donating groups are replaced by electron-withdrawing groups, the selectivity for their respective aldehyde production substantially increased (OMe < H < Cl < NO2) where complex 1 seemed to be more specific to the p-nitrobenzaldehyde production (Table 4, entry 4).
| Entry | Complex | Substrate | Catalyst (mol%) | Time (min) | Yield % (±sd.) | TONa (103) | TOFb min−1 (103) | Selectivity % | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| BA | BzA | BA | BzA | BA | BzA | ||||||
| a TON, turnover number, refers to the ratio of mol of a product (BA/BzA)/mol of the catalyst. b TOF, turnover frequency, refers to TON/time (min−1); n.d. = not detected; sd = standard deviation (n = 3). Selectivity BA [= 100% × BA/(BA + BzA)]. | |||||||||||
| 1 | 1 | OCH3 | 2.45 | 60 | 28.9 ± 0.8 | 13.2 ± 0.45 | 385 | 169 | 6.4 | 2.8 | 69 |
| 2 | H | 2.45 | 60 | 26.8 ± 1.5 | 4.50 ± 0.14 | 366 | 59.1 | 5.6 | 0.9 | 85 | |
| 3 | Cl | 2.45 | 60 | 22.1 ± 0.5 | 2.56 ± 0.07 | 295 | 34.6 | 4.9 | 0.5 | 89 | |
| 4 | NO2 | 2.45 | 60 | 12.3 ± 2.8 | n.d. | 135 | n.d. | 2.2 | n.d. | 100 | |
| 5 | 2 | OCH3 | 2.45 | 60 | 3.83 ± 0.11 | n.d. | 50.9 | n.d. | 0.8 | n.d. | 100 |
| 6 | H | 2.45 | 60 | 4.52 ± 0.02 | 1.87 ± 0.16 | 50.8 | 22.3 | 0.8 | 0.4 | 71 | |
| 7 | Cl | 2.45 | 60 | 6.50 ± 0.27 | 2.26 ± 0.08 | 85.1 | 26.2 | 1.4 | 0.4 | 74 | |
| 8 | NO2 | 2.45 | 60 | 7.74 ± 0.36 | 0.97 ± 0.05 | 118.8 | 11.8 | 1.9 | 0.2 | 89 | |
| 9 | — | OCH3 | — | 60 | 0.95 ± 0.03 | n.d. | n.d. | n.d. | n.d. | n.d. | 100 |
| 10 | — | H | — | 60 | 0.610 ± 0.071 | n.d. | n.d. | n.d. | n.d. | n.d. | 100 |
| 11 | — | Cl | — | 60 | 0.710 ± 0.013 | 1.21 ± 0.04 | n.d. | n.d. | n.d. | n.d. | 38 |
| 12 | — | NO2 | — | 60 | n.d. | 0.93 ± 0.03 | n.d. | n.d. | n.d. | n.d. | — |
When [FeIII(TPP)Cl] (2) was considered (Table 4, entries 5–8), the opposite trend was observed for BA production. For p-methoxybenzyl alcohol (entry 5), the yield is the lowest among the substrates studied (3.83 ± 0.11%), while for p-nitrobenzyl alcohol (entry 8), the highest BA yield in the series was observed (7.74 ± 0.36%). Intermediate values were obtained for benzyl and p-chlorobenzyl alcohols. Thus, the product yield followed the order: OMe < H < Cl < NO2. Considering BA → BzA yields, a small amount of the product was observed in the case of each p-substituted benzaldehyde, and no activity trend could be detected. The TON and TOF parameters followed the same trend for BnOH → BA production, where the alcohol-to-aldehyde formation increases with the electron-withdrawing effect of the substituent (Table 4, entries 5–8). Once again, no clear trends were observed for the TON and TOF parameters when BA → BzA conversion is taken into account. As observed for the manganese-containing 1, the lower alcohol-to-aldehyde yields are compensated by higher aldehyde selectivity over BzA (100%), even though a clear trend in selectivity could not be observed during the catalytic oxidation of p-substituted alcohols promoted by 2.
Finally, tests were carried out in triplicate (Table 4, entries 9 to 12) in the absence of the catalysts to assess whether the oxidant (DAIB) is able to oxidize the substrates. Because only trace amounts of the product were observed, the crucial role of the catalysts was revealed.
Linear free-energy relationships and mechanistic insights
The Hammett parameter (σp) can qualitatively describe the electronic substituent effect of the structure on the reactivity of a site X in the series p-XC6H4Y for different functional groups Y.This effect is one of many such linear free-energy relationships (LFERs) that have been developed,54 and provides a simple measure of the electron-withdrawing (σp > 0) or donating (σp < 0) ability of the functional group relative to Y![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) H. In this sense, it was proposed to apply the BA yield values obtained from the catalytic activity studies as a function of σp for different R-groups present in the p-substituted benzyl alcohols. Based on that, a linear free-energy relationship could be observed between BA yield promoted by complexes [MnIII(TPP)Cl] (1) and [FeIII(TPP)Cl] (2) and σp, as depicted in Fig. 7. The catalytic activity of the Mn system is more effective for the electron-rich p-OMe substituted alcohol than for the electron-deficient p-NO2 substituted substrate. These results are not surprising. The p-OMe group can exert a strong electron donating resonance (+R) effect and the corresponding electron-rich substrate can stabilize the Mn(V) metal center. In contrast, the p-NO2 function fails to induce such stabilization since it can exert only electron withdrawing resonance (–R). The computed activation barriers also support these phenomena. The barrier for the Cα–H activation step is calculated to be lower, 4.2 kcal mol−1, for p-OMe substituted benzyl alcohol than the barrier, 6.1 kcal mol−1, calculated for p-NO2 substituted alcohol in the presence of a Mn(V) metal center. For the iron species, the catalytic activity is reported to be better for the electron-deficient p-NO2 substituted alcohol than for the electron-rich p-OMe substituted alcohol. A significant presence of the spin density is observed on the phenyl rings of the alcohol substrates in the Cα–H activation transition states in the case of Fe (Fig. 8).
H. In this sense, it was proposed to apply the BA yield values obtained from the catalytic activity studies as a function of σp for different R-groups present in the p-substituted benzyl alcohols. Based on that, a linear free-energy relationship could be observed between BA yield promoted by complexes [MnIII(TPP)Cl] (1) and [FeIII(TPP)Cl] (2) and σp, as depicted in Fig. 7. The catalytic activity of the Mn system is more effective for the electron-rich p-OMe substituted alcohol than for the electron-deficient p-NO2 substituted substrate. These results are not surprising. The p-OMe group can exert a strong electron donating resonance (+R) effect and the corresponding electron-rich substrate can stabilize the Mn(V) metal center. In contrast, the p-NO2 function fails to induce such stabilization since it can exert only electron withdrawing resonance (–R). The computed activation barriers also support these phenomena. The barrier for the Cα–H activation step is calculated to be lower, 4.2 kcal mol−1, for p-OMe substituted benzyl alcohol than the barrier, 6.1 kcal mol−1, calculated for p-NO2 substituted alcohol in the presence of a Mn(V) metal center. For the iron species, the catalytic activity is reported to be better for the electron-deficient p-NO2 substituted alcohol than for the electron-rich p-OMe substituted alcohol. A significant presence of the spin density is observed on the phenyl rings of the alcohol substrates in the Cα–H activation transition states in the case of Fe (Fig. 8).
 | ||
| Fig. 8 The total spin densities calculated on the phenyl ring of the p-OMe and p-NO2 substituted benzyl alcohols in the hydrogen abstraction transition states in the presence of the Fe metal. | ||
This spin density originates due to delocalization from the Cα radical which is formed as a result of the homolytic cleavage of the Cα–H bond in the transition states.
The total value of this spin density is calculated to be 0.45 and 0.39 on the p-OMe and p-NO2 substituted rings, respectively, in the case of Fe(IV). In contrast, in the case of the Mn(V) system, the total spin density is significantly lower, 0.14 and 0.22 on the p-OMe and p-NO2 substituted rings, respectively. The increased charge density present on the phenyl rings, in the case of Fe, is better stabilized by the p-NO2 than the p-OMe function due to the electron-withdrawing inductive (–I) and resonance (–R) effects of the nitro group. Consequently, the catalytic activity is better for the benzyl alcohol containing p-NO2 than for that with p-OMe.
Here it is noteworthy that the %yields for the alcohol oxidation products are 3.83 ± 0.11 and 7.74 ± 0.36 for p-OMe and p-NO2 substituted alcohols, respectively (Table 3). The difference in the %yield is small ∼3.91, indicating that only a very minute energy difference exists between the C–H activation transition states for the p-OMe and p-NO2 substituted systems in the case of species 2. Therefore, any analysis based on energy may be susceptible to DFT error and we have carefully refrained from such analysis in Fe systems.
Conclusions
Manganese(III) and iron(III) tetraphenylporphyrins were successfully used as catalysts for the oxidation of a series of benzyl alcohols aiming to achieve their respective products, benzaldehyde and benzoic acid. Through FFD multivariate analysis, it was possible to determine the best catalytic conditions in terms of catalyst load and reaction time, thus maximizing product formation. Based on the experimental data collected, the generated mathematical models were able to predict the conversion of the alcohol into benzaldehyde within the established experimental domain. Pareto charts revealed that catalyst load was the most significant variable for the [MnIII(TPP)Cl] (1) species, whereas reaction time seemed to have more impact when [FeIII(TPP)Cl] (2) was selected.For the catalytically active species generated in solution for each species, their electronic structure was modelled by DFT calculations. Experimentally observed differences in the reactivity were supported by DFT data, which enabled the inference of reaction mechanisms.
The contribution of electronic structures of the substrates, different p-substituted benzyl alcohols, was evaluated both experimentally and by DFT. Distinct linear free energy relationships (LFERs) were observed and explained, considering the spin density present in the transition state of the determining step in the oxidation reaction. The selectivity between benzaldehyde and benzoic acid was monitored, being inversely proportional to their reaction yields.
Author contributions
Conceptualization: F. R. X, E. M., G. F. S., C. N. V. and S. M.; synthesis: P. S. T. and G. F. S.; catalytic studies: L. C. and W. H. S; DFT studies: S. R. and S. M.; data curation: L. C., W. H. S., and S. R.; formal analysis: L. C., S. R., E. M., G. F. S., C. N. V., S. M. and F. R. X; funding acquisition: F. R. X; investigation: L. C., W. H. S., and S. R.; project administration: F. R. X.; resources: F. R. X, E. M., G. F. S, C. N. V. and S. M.; visualization: F. R. X, E. M., C. N. V. and S. M.; writing original draft: L. C., S. R., F. R. X. and E. M.; and writing – review & editing: F. R. X., E. M., G. F. S., C. N. V. and S. M.Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are available in the supplementary information (SI). Supplementary information: comparison between the calculated electronic structures by DFT herein described vs. data from the literature; calibration parameters, LOD and LOQ obtained from the HPLC-DAD analysis; residual graphs for the quadratic regressions obtained for complexes 1 and 2; reaction monitoring of the catalytic oxidation of BnOH to form BA and BzA mediated by 2; energetics of the oxygen rebound step and the subsequent dehydration pathway to the carbonyl product in the presence of Mn- and Fe-porphyrin systems; energetics of the acetate addition to Mn- and Fe-porphyrin and XYZ coordinates for the DFT calculated structures. See DOI: https://doi.org/10.1039/d5dt02798e.These data are available without specific restrictions, in accordance with the standard access policies of the journal.
Acknowledgements
The authors thankfully acknowledge the support from the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – CAPES (Finance Code 001), including financial support for L. C. (DS-scholarship) and the Fundação de Amparo à Pesquisa e Inovação do Estado de Santa Catarina – FAPESC 2024TR2628 to F. R. X.; from the U.S. Department of Energy – DOE-BES (grant DEFOA0002414) and the Natural Sciences and Engineering Research Council of Canada – NSERC (Discovery Grant RGPIN-2025-06312) to C. N. V.; and from the Royal Society of Chemistry – RSC (Research Fund grant R24-8999168020) to S. M.; F. R. X. and G. F. S. hold a Research Productivity Grant (306851/2022-9 and 311472/2022-2, respectively) from the Conselho Nacional de Desenvolvimento Científico e Tecnológico – CNPq; S. R. is supported as a JRF fellow of IIT Jammu. The authors also acknowledge access to the UDESC Multi-User Facility infrastructure at the Technological Sciences Center – CMU/CCT/UDESC, and the High-Performance Computing Facility AGASTYA at IIT Jammu.References
- Benzaldehyde Market Size, Share & Demand 2025 to 2035, https://www.futuremarketinsights.com/reports/benzaldehyde-market, (accessed October 30, 2025).
- B. K. Kundu, R. Ranjan, A. Mukherjee, S. M. Mobin and S. Mukhopadhyay, J. Inorg. Biochem., 2019, 195, 164–173 CrossRef CAS PubMed.
- Y. Xu, P. Wu, D. Xie, Y. Cui, Y. Zhang, B. Wang and M. Guo, Inorg. Chem. Front., 2025, 12, 4199–4212 RSC.
- S. Nakagaki, K. Castro, M. D. G. Neves, M. D. A. Faustino and Y. Iamamoto, J. Braz. Chem. Soc., 2019, 30, 2501–2535 CAS.
- F. Liu, I. Rincón, H. G. Baldoví, A. Dhakshinamoorthy, P. Horcajada, S. Rojas, S. Navalón and A. Fateeva, Inorg. Chem. Front., 2024, 11, 2212–2245 RSC.
- D. Zhang, L. E. Rosch, M. R. Crawley and T. R. Cook, Inorg. Chem. Front., 2024, 11, 5557–5565 RSC.
- A. Rezaeifard, M. Jafarpour, G. K. Moghaddam and F. Amini, Bioorg. Med. Chem., 2007, 15, 3097–3101 CrossRef CAS PubMed.
- C. Fabbri, C. Aurisicchio and O. Lanzalunga, Open Chem., 2008, 6, 145–153 CrossRef CAS.
- G. R. Karimipour, H. A. Shadegan and R. Ahmadpour, J. Chem. Res., 2007, 2007, 252–256 CrossRef.
- M. M. Pereira, L. D. Dias and M. J. F. Calvete, ACS Catal., 2018, 8, 10784–10808 CrossRef CAS.
- L. Que and W. B. Tolman, Nature, 2008, 455, 333–340 CrossRef CAS PubMed.
- A. Rezaeifard, M. Jafarpour and A. Naeimi, Catal. Commun., 2011, 16, 240–244 CrossRef CAS.
- A. Modak, J. Mondal and A. Bhaumik, Appl. Catal., A, 2013, 459, 41–51 CrossRef CAS.
- F. Adam and W.-T. Ooi, Appl. Catal., A, 2012, 445–446, 252–260 CrossRef CAS.
- H.-B. Ji, Q.-L. Yuan, X.-T. Zhou, L.-X. Pei and L.-F. Wang, Bioorg. Med. Chem. Lett., 2007, 17, 6364–6368 CrossRef CAS PubMed.
- R. Nasrollahi and S. Zakavi, Eur. J. Inorg. Chem., 2017, 2017, 2002–2010 CrossRef CAS.
- A. Rezaeifard, M. Jafarpour and A. Naeimi, Catal. Commun., 2011, 16, 240–244 CrossRef CAS.
- H. Srour, P. L. Maux and G. Simonneaux, Inorg. Chem., 2012, 51, 5850–5856 CrossRef CAS PubMed.
- M. Guo, Y.-M. Lee, M. S. Seo, Y.-J. Kwon, X.-X. Li, T. Ohta, W.-S. Kim, R. Sarangi, S. Fukuzumi and W. Nam, Inorg. Chem., 2018, 57, 10232–10240 CrossRef CAS PubMed.
- N. Amiri, P. Le Maux, H. Srour, H. Nasri and G. Simonneaux, Tetrahedron, 2014, 70, 8836–8842 CrossRef CAS.
- R. Zhang, S. Klaine, C. Alcantar and F. Bratcher, J. Inorg. Biochem., 2020, 212, 111246 CrossRef CAS PubMed.
- S. Sanfui, A. Roychowdhury, M. Usman, E. Garribba, C. J. Gómez-García and S. P. Rath, Inorg. Chem., 2024, 63, 5423–5431 CrossRef CAS PubMed.
- X. Li, H. Lei, L. Xie, N. Wang, W. Zhang and R. Cao, Acc. Chem. Res., 2022, 55, 878–892 CrossRef CAS PubMed.
- R. Kumar, B. Pandey, A. Sen, M. Ansari, S. Sharma and G. Rajaraman, Coord. Chem. Rev., 2020, 419, 213397 CrossRef CAS.
- K. W. Kwong, T.-H. Chen, W. Luo, H. Jeddi and R. Zhang, Inorg. Chim. Acta, 2015, 430, 176–183 CrossRef CAS.
- N. Anand and S. Yadava, J. Coord. Chem., 2018, 71, 3090–3098 CrossRef CAS.
- A. F. Vailati, R. D. Huelsmann, E. Martendal, A. J. Bortoluzzi, F. R. Xavier and R. A. Peralta, New J. Chem., 2020, 44, 2514–2526 RSC.
- G. E. P. Box and D. W. Behnken, Technometrics, 1960, 2, 455–475 CrossRef.
- S. Munshi, A. Sinha, S. Yiga, S. Banerjee, R. Singh, Md. K. Hossain, M. Haukka, A. F. Valiati, R. D. Huelsmann, E. Martendal, R. Peralta, F. Xavier, O. F. Wendt, T. K. Paine and E. Nordlander, Dalton Trans., 2022, 51, 870–884 RSC.
- L. Chimilouski, F. Morgan, D. Ekanayake, N. El-Harakeh, R. A. Peralta, E. Martendal, C. N. Verani and F. R. Xavier, J. Inorg. Biochem., 2023, 240, 112095 CrossRef CAS PubMed.
- L. Chimilouski, W. H. Slominski, A. I. Tillmann, D. Will, A. M. Dos Santos, G. Farias, E. Martendal, K. P. Naidek and F. R. Xavier, Molecules, 2024, 29, 2634 CrossRef CAS PubMed.
- C. A. Lucecki, N. Rani, K. K. Kpogo, J. Niklas, O. G. Poluektov, H. B. Schlegel and C. N. Verani, Inorg. Chem. Front., 2025, 12, 7411–7420 RSC.
- K. K. Kpogo, S. Mazumder, D. Wang, H. B. Schlegel, A. T. Fiedler and C. N. Verani, Chem. – Eur. J., 2017, 23, 9272–9279 CrossRef CAS PubMed.
- H. Baydoun, S. Mazumder, H. B. Schlegel and C. N. Verani, Chem. – Eur. J., 2017, 23, 9266–9271 CrossRef CAS PubMed.
- D. Basu, S. Mazumder, X. Shi, H. Baydoun, J. Niklas, O. Poluektov, H. B. Schlegel and C. N. Verani, Angew. Chem., Int. Ed., 2015, 54, 2105–2110 CrossRef CAS PubMed.
- D. Basu, S. Mazumder, K. K. Kpogo and C. N. Verani, Dalton Trans., 2019, 48, 14669–14677 RSC.
- D. Basu, S. Mazumder, J. Niklas, H. Baydoun, D. Wanniarachchi, X. Shi, R. J. Staples, O. Poluektov, H. B. Schlegel and C. N. Verani, Chem. Sci., 2016, 7, 3264–3278 RSC.
- D. M. Ekanayake, K. M. Kulesa, J. Singh, K. K. Kpogo, S. Mazumder, H. B. Schlegel and C. N. Verani, Dalton Trans., 2017, 46, 16812–16820 RSC.
- P. H. A. Kankanamalage, D. M. Ekanayake, N. Singh, A. C. P. De Morais, S. Mazumder, C. N. Verani, A. Mukherjee and M. Lanznaster, New J. Chem., 2019, 43, 12795–12803 RSC.
- N. El Harakeh, A. C. P. De Morais, N. Rani, J. A. G. Gomez, A. Cousino, M. Lanznaster, S. Mazumder and C. N. Verani, Angew. Chem., 2021, 133, 5787–5792 CrossRef.
- P. H. A. Kankanamalage, S. Mazumder, V. Tiwari, K. K. Kpogo, H. B. Schlegel and C. N. Verani, Chem. Commun., 2016, 52, 13357–13360 RSC.
- Z. Li, C.-G. Xia and C.-Z. Xu, Tetrahedron Lett., 2003, 44, 9229–9232 CrossRef CAS.
- J.-H. In, S.-E. Park, R. Song and W. Nam, Inorg. Chim. Acta, 2003, 343, 373–376 CrossRef CAS.
- V. S. Da Silva, L. I. Teixeira, E. Do Nascimento, Y. M. Idemori and G. DeFreitas-Silva, Appl. Catal., A, 2014, 469, 124–131 CrossRef.
- I. Ahmad, Shagufta and S. Rehman, Tetrahedron, 2022, 104, 132604 CrossRef CAS.
- R. Rahimi, E. Gholamrezapor and M. R. Naimi-jamal, Inorg. Chem. Commun., 2011, 14, 1561–1568 CrossRef CAS.
- F. Adam and W.-T. Ooi, Appl. Catal., A, 2012, 445–446, 252–260 CrossRef CAS.
- W. J. Song, M. S. Seo, S. D. B. George, T. Ohta, R. Song, M.-J. Kang, T. Tosha, T. Kitagawa, E. I. Solomon and W. Nam, J. Am. Chem. Soc., 2007, 129, 1268–1277 CrossRef CAS PubMed.
- M. Radoń, E. Broclawik and K. Pierloot, J. Chem. Theory Comput., 2011, 7, 898–908 CrossRef PubMed.
- B. Boso and G. Lang, J. Chem. Phys., 1983, 79, 1122 CrossRef CAS.
- J. H. Han, S.-K. Yoo, J. S. Seo, S. J. Hong, S. K. Kim and C. Kim, Dalton Trans., 2005, 402, 402–406 RSC.
- D. Ricciarelli, Q. M. Phung, L. Belpassi, J. N. Harvey and P. Belanzoni, Inorg. Chem., 2019, 58, 7345–7356 CrossRef CAS PubMed.
- W. Nam, Acc. Chem. Res., 2007, 40, 522–531 CrossRef CAS PubMed.
- M. B. Smith, March's Advanced Organic Chemistry Reactions, Mechanisms ad Structures, 2001 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |