Fluorinated B-phenylated scorpionates as tunable platforms for stabilizing thallium(I) and silver(I)–ethylene complexes
Anurag
Noonikara-Poyil
 a,
Vo Quang Huy
Phan
a,
Vo Quang Huy
Phan
 a,
Mukundam
Vanga
a,
Mukundam
Vanga
 a,
Andrii
Boretskyi
b,
Pavel
Mykhailiuk
a,
Andrii
Boretskyi
b,
Pavel
Mykhailiuk
 *bc and
H. V. Rasika
Dias
*bc and
H. V. Rasika
Dias
 *a
*a
aDepartment of Chemistry and Biochemistry, The University of Texas at Arlington, Arlington, Texas 76019, USA. E-mail: dias@uta.edu
bEnamine Ltd, Winston Churchill St 78, 02094, Kyiv, Ukraine
cChemistry Department, Taras Shevchenko National University of Kyiv, Volodymyrska 64, 01601 Kyiv, Ukraine. E-mail: Pavel.Mykhailiuk@gmail.com
First published on 3rd December 2025
Abstract
Fluorinated and B-phenylated bis(pyrazolyl)borate and bis(pyridyl)borate ligands have been utilized in thallium(I) and silver(I)–ethylene chemistry. These ligands contain electron-withdrawing trifluoromethyl or pentafluorosulfanyl groups that create a chemically inert half-rim and, together with a flanking phenyl group, provide steric protection at the metal site. Thallium(I) complexes display κ2-N,N ligand binding and noteworthy Tl–F coupling in 19F NMR, indicating proximity effects of the fluorinated substituents. The silver–ethylene complexes [Ph2B(3-(CF3)Pz)2]Ag(C2H4), [Ph2B(3-(SF5)Pz)2]Ag(C2H4), and [Ph2B(6-(CF3)Py)2]Ag(C2H4) feature trigonal-planar silver(I) sites, with NMR data showing upfield shifts for ethylene proton and carbon resonances relative to those of the free ethylene. The steric effects of fluorinated supporting ligands in thallium and silver complexes were evaluated using buried-volume calculations. In addition, C–H activation of alkanes via carbene insertion catalyzed by silver complexes is reported. Overall, the findings illustrate how fluorinated groups and N-heterocyclic donor arms influence metal–ligand bonding, the stability of coinage-metal π-complexes, and influence the catalytic efficiency and selectivity in alkane C–H functionalization.
Introduction
Bis(pyrazolyl)borates are useful monoanionic, nitrogen donor site-based ligands,1,2 which can be tuned electronically and sterically to support transition metals in various applications, such as catalysis, bioinorganic chemistry, and the development of luminescent materials.3 Bis(pyridyl)borates are a related ligand family, but they are based on larger, six-membered pyridyl heterocycles instead of five-membered pyrazolyl groups. Although bis(pyridyl)borates have been known since the 1990s,4,5 they have received less attention in metal coordination chemistry compared to their bis(pyrazolyl)borate counterparts.4,6–17 Moreover, bis(pyridyl)borates with pyridyl ring substituents near metal coordination sites were unknown until recently.11,12,18 We are interested in developing fluorinated versions of both these ligand classes,11,12,19–22 which belong to a family broadly known as scorpionates,2,23,24 and exploring their potential in coordination chemistry and metal-mediated processes. Fluorinated ligands are of interest because they act as weaker donors to metal ions and often endow different properties to metal complexes compared to their non-fluorinated, electron-rich equivalents.25–34 They typically increase Lewis acidity at metal centers and enhance the volatility, oxidation resistance, and fluorocarbon solubility of their metal adducts. Additionally, they have been shown to improve the activity of certain metal-catalyzed reactions.This paper describes structure and properties of thallium(I) and silver(I)–ethylene complexes supported by B-phenylated bis(pyrazolyl)borate ligands, [Ph2B(3-(CF3)Pz)2]− (1) and [Ph2B(3-(SF5)Pz)2]− (2), with CF3 and SF5 groups at the pyrazolyl ring 3-positions, and a B-phenylated bis(pyridyl)borate ligand [Ph2B(6-(CF3)Py)2]− (3) with CF3 groups at the pyridyl ring 6-positions (Fig. 1, Pz = pyrazolyl, Py = pyridyl). Fluorinated substituents on these ligands exhibit strong electron-withdrawing properties and shield the metal site by forming a chemically inert layer that protects parts of the periphery of the metal. The SF5 group has been gaining more attention recently as a fluorinated substituent. However, unlike the CF3 group,34–39 it is still less widely used in chemistry,40–46 and, in particular, in transition-metal chemistry.22,47–54 The SF5 group is often called the “super-trifluoromethyl group”, and exhibits higher electronegativity than CF3 (electronegativity parameters; χ = 3.65 [SF5] vs. χ = 3.36 [CF3]),40,55 and bulkier than CF3 with its size approaching that of tert-butyl group (van der Waals volumes; 61.4 Å3 [SF5] vs. 42.7 Å3 [CF3] vs. 76.4 Å3 [tBu]).56
Silver(I) complexes with ethylene are of both fundamental and practical interest;57 for example, silver plays a key role in ethylene epoxidation58–60 and olefin separation.61–63 Silver is also important in plant chemistry and horticulture because it has long been known to inhibit ethylene responses in plants.64–66 Although extensive spectroscopic and computational research has been conducted on silver(I) ethylene adducts,57,67,68 isolable silver(I) ethylene complexes are rare due to the weak σ-acceptor and poor π-donor properties of silver(I), which make them quite labile.57,69–71 We also present structural data for thallium(I) complexes of 1–3. They, in general, are good ligand transfer agents and can facilitate the separation and purification of scorpionate ligands from reaction mixtures.23,72–77 Thallium complexes are also of interest for their NMR properties because Tl has two spin 1/2 isotopes, 203Tl (29.5% natural abundance) and 205Tl (70.5% natural abundance), leading to interesting spin–spin coupling between NMR active nuclei.78–80
Results and discussion
The syntheses of [Ph2B(3-(CF3)Pz)2]Tl (4) and [Ph2B(3-(SF5)Pz)2]Tl (5) were reported earlier,22 but their molecular structures have not been reported. Here, we describe their X-ray crystal structures, highlight some interesting features, and present a higher-yield route (Scheme 1) to [Ph2B(3-(SF5)Pz)2]Tl (5).Compound 5, in the previous report, was synthesized by a solid-state reaction of NaBPh4 with the corresponding pyrazole, [3-(SF5)Pz]H, followed by the addition of TlOAc.22 This method, however, proved somewhat inefficient by affording the desired thallium complex in only 36% overall yield. The modest outcome can be attributed to the relatively mild reactivity of NaBPh4, which necessitated elevated reaction temperatures. Such conditions also required careful control to prevent the formation of by-products at higher temperatures (180 °C). To circumvent these limitations, a more reactive boron precursor, Ph2BH2Li,81 was employed in the presence of MeB(OiPr)2 as a Lewis acid catalyst, which is expected to coordinate to, and decrease the pKa of, the pyrazole and increase the rate of B–H bond protonolysis (Scheme 1).82 This approach facilitated the formation of the lithium salt of the bis(pyrazolyl)borate, which, upon treatment with TlOAc, produced the thallium complex [Ph2B(3-(SF5)Pz)2]Tl (5) in 64% overall yield, representing a significant improvement over the original protocol. Molecular structures of [Ph2B(3-(SF5)Pz)2]Tl (5) as well as that of the [Ph2B(3-(CF3)Pz)2]Tl (4) have now been confirmed by single-crystal X-ray diffraction (Fig. 2).
 | ||
| Fig. 2 Molecular structure of [Ph2B(3-(CF3)Pz)2]Tl (4, left) and [Ph2B(3-(SF5)Pz)2]Tl (5, right). Ellipsoids are shown at the 50% probability level. | ||
The related bis(pyridyl)borate complex [Ph2B(6-(CF3)Py)2]Tl (6) was obtained by metathesis of its potassium salt, [Ph2B(6-(CF3)Py)2]K11 with TlOAc in 79% yield (Scheme 2). The crystal structure of 6 is shown in Fig. 3. All three thallium complexes of fluorinated scorpionates (4–6) described in this study were isolated as air- and moisture-stable off-white powders, free of coordinated solvent or water molecules. In general, these features of thallium complexes and their facile metathesis chemistry, compared to their alkali metal counterparts, make them promising precursors (ligand transfer agent) for the synthesis of various metal complexes. Selected bond lengths and angles of [Ph2B(3-(CF3)Pz)2]Tl (4), [Ph2B(3-(SF5)Pz)2]Tl (5), and [Ph2B(6-(CF3)Py)2]Tl (6) together with data for several related systems, are summarized in Table 1. The thallium complexes 4–6 adopt the expected κ2-N,N coordination mode for the corresponding scorpionate. In compound 6, there is a close separation between the ipso-carbon of the flanking phenyl group and thallium (2.833(3) Å). Thallium(I)-π-arene interactions are known and are typically found in thallium complexes or salts of weakly coordinating anions.83–85 The arene-Tl separation in 6 falls within a region where weak but significant non-covalent interactions could occur. For comparison, it is within Bondi's Tl–C van der Waals contact distance of 3.66 Å,86 but longer than the sum of the covalent radii of Tl and Csp2 of 2.18 Å.87
 | ||
| Fig. 3 Molecular structures of [Ph2B(6-(CF3)Py)2]Tl (6). Ellipsoids are shown at the 50% probability level. | ||
| Compound | Tl–N (Å) | N–Tl–N (°) | Tl⋯B (Å) | Ref. |
|---|---|---|---|---|
| Abbreviations: Trip = 9-triptycyl, Mes = mesityl, Fc = ferrocenyl. Data for the second molecule in the asymmetric unit in italics. | ||||
| [Ph2B(3-(CF3)Pz)2]Tl (4) | 2.688(2), 2.688(2) | 63.87(9) | 3.565 | This work |
| [Ph2B(3-(SF5)Pz)2]Tl (5) | 2.668(2) 2.686(2) | 67.60(7) | 3.563 | This work |
| 2.682(2), 2.607(2) | 68.12(8) | 3.571 | ||
| [H2B(Pz)2]Tl | 2.799(7), 2.814(8) | 55.5(2) | 3.34 | 92 |
| [H2B(3,5-(CF3)2Pz)2]Tl | 2.7243(19), 2.7243(19) | 68.32(8) | 3.364 | 93 |
| [H2B(3,5-(tBu)2Pz)2]Tl | 2.632(12), 2.645(12) | 67.4(4) | 3.176 | 94 |
| [H2B(3-(tBu),5-(iPr)Pz)2]Tl | 2.628(11), 2.664(10) | 72.1(3) | 3.179 | 94 |
| [H2B(3-(tBu),5-(Me)Pz)2]Tl | 2.597(16), 2.59(3) | 73.6(6) | 3.118 | 94 |
| [H2B(3-(Fc),5-(CF3)Pz)2]Tl | 2.578(5), 2.593(5) | 76.54(15) | 3.504 | 95 |
| [H2B(3-(2-pyrazinyl)Pz)2]Tl | 2.644(4), 2.752(4) | 71.28(11) | 3.827 | 96 |
| [H2B(3-(Trip)Pz)2]Tl | 2.696(6), 2.705(6) | 78.23(19) | 3.313 | 97 |
| [Me(H)B(3-(Mes)Pz)2]Tl | 2.636(5), 2.636(5) | 79.1(2) | 3.578 | 98 |
| [Ph2B(6-(CF3)Py)2]Tl (6) | 2.670(2), 2.675(2) | 68.86(7) | 3.459 | This work |
A search in the Cambridge Structural Database (CSD)88 resulted in 12 entities for structurally characterized thallium bis(pyrazolyl)borates, and none for thallium bis(pyridyl)borates, highlighting the limited structural data available for thallium complexes of these ligands. Analysis of the structural metrics reveals no clear trend in Tl–N bond distances of 6 compared to 4, 5 and other bis(pyrazolyl)-based analogs, despite differences in the N-based heterocyclic donor arms (Table 1). These Tl–N bonds are longer than the sum of covalent radii (2.16 Å) but shorter than the van der Waals contact distance (3.51 Å).89 More notable differences are observed in the N–Tl–N angles. Complex 4, which has the least steric hindrance compared to complexes 5 and 6, shows a relatively small N–Tl–N angle of 63.87°, yet it is clearly wider than that observed for [H2B(Pz)2]Tl (55.5°), which is supported by the parent bis(pyrazolyl)borate ligand. This value N–Tl–N of 4 is smaller than those observed for complex 5 (67.60°) bearing the bulkier SF5 groups at the same position, and complex 6 (68.86°), which is further enlarged due to the bulky nature of pyridyl-based ligand. Another notable difference is that the boat-shaped six-membered Tl(NC)2B core in 6 is deeper than the Tl(NN)2B cores in 4 and 5, as evidenced by the shorter Tl⋯B separations in the former. There are no significant intermolecular interactions between thallium atoms in the solid state of complexes 4–6. For instance, the closest intermolecular Tl⋯Tl separation in 4, 5, and 6 are 6.02, 6.27, and 6.02 Å, respectively, well above the sum of the Bondi's van der Waals radii for two thallium atoms (3.92 Å), indicating the absence of significant Tl⋯Tl interaction.89–91
An intriguing feature of thallium complexes supported by these fluorinated ligands is the observation of four-bond Tl–F coupling, likely a through-space interaction. Fluorinated bis(pyrazolyl/pyridyl)borate thallium complexes are limited; thus, the coupling constants of 4–6 are compared with related bis- and tris(pyrazolyl/pyridyl)borate systems (Table S1). Complex 5, bearing both equatorial and axial fluorine atoms in the SF5-groups, exhibits two distinct coupling constants (4JTl–F = 777 and 76 Hz; see Fig. S18).22 The thallium complex 4 exhibits a smaller value of 576 Hz,22 whereas complex 6 shows a remarkably larger 4JTl–F of 1100 Hz. The coupling between thallium-203 and thallium-205 is not resolved in these signals, which is not unusual because their gyromagnetic ratios are similar, often making it impossible to distinguish the difference in 203Tl and 205Tl coupling constants.78 The enhanced coupling in complex 6 can be attributed to the closer proximity of thallium to the CF3 substituents at the 6-position of the pyridyl rings (with average F3C⋯Tl separation of 3.58 Å) relative to the 3-position of pyrazolyl substituents (with average F3C⋯Tl separation of 4.00 Å), allowing stronger through-space interaction with thallium. The Tl⋯C (of CF3) separations in complex 6 are also shorter than those observed in tris- and bis(pyrazolyl)borate thallium complexes with CF3-groups at the pyrazolyl ring 3-positions. However, we have recently reported a thallium complex of fluorinated tris(pyridyl)borate with relatively short F3C⋯Tl separations (average 3.45 Å) with much larger 4JTl–F (1208 Hz).75 Further analysis indicates that there is a strong correlation between the F3C⋯Tl separation and the Tl–F coupling constant (see Fig. S1), despite potential for complications in some cases.78,79
Space-filling models in Fig. 4 highlight the differing steric impacts of the B-phenylated and fluorinated ligand systems in complexes 4–6. Yet another and more quantitative approach involves buried volume (%VBur) calculations and steric maps using the SambVca 2.1 web application,99 in which the %VBur corresponds to the fraction of the volume of a sphere centered on the metal occupied by the coordinated ligand of focus, while the topographic steric map (Fig. 4) provides a graphical representation of the steric profile of a ligand using color-coded contour maps. For example, the %VBur values for complexes 4, 5, and 6 are 50.0%, 55.6% and 58.3%, respectively. Overall, the bis(pyridyl)borate framework in 6 affords the most protection around thallium, closely followed by the SF5-substituted ligand in 5, while the CF3-substituted analog in 4 provides the least protection around thallium.
Although sodium, potassium and other alkali metal salts are more widely used as a transfer reagent due to their lower toxicity, thallium complexes sometimes serve as better scorpionate transfer agents due to their ease of purification and good solubility in non-polar solvents.72,73,80Scheme 3 illustrates the use of thallium complexes with AgOTf and under ethylene atmosphere to generate silver(I)–ethylene adducts, [Ph2B(3-(CF3)Pz)2]Ag(C2H4) (7), [Ph2B(3-(SF5)Pz)2]Ag(C2H4) (8), and [Ph2B(6-(CF3)Py)2]Ag(C2H4) (9). These are crystalline, colorless solids. Compounds 8 and 9 lose coordinated ethylene easily under reduced pressure. The X-ray crystal structures of 7–9 are presented in Fig. 5, with selected bond lengths and angles summarized in Table 2. They are three-coordinate, trigonal planar silver species featuring an η2-bound C2H4. There is a close separation between the ipso-carbon of the flanking phenyl group of 9 and silver (2.6908(11) Å), which is within the Bondi's van der Waals contact separation of C and Ag (3.42 Å),86,89 but this interaction does not seem to have a notable effect on silver, as evidenced by the sum of angles at silver being 360° (and the retention of trigonal planarity). For comparison, the sum of the covalent radii of silver and sp2 carbon is 2.18 Å.87 The scorpionate ligands in 7–9 coordinate to silver in a κ2 fashion through the two nitrogen atoms from ligand arms, adopting the characteristic boat configuration. Most structural features are consistent across the three adducts; however, complexes 8 and 9 exhibit slightly longer Ag–C and Ag–N bond distances than those of complex 7. These observed metrical parameters are attributable to the steric influence of the supporting ligands in 8 and 9.
| Parameter | [Ph2B(3-(CF3)Pz)2]Ag(C2H4) (7)c | [Ph2B(3-(SF5)Pz)2]Ag(C2H4) (8)d | [Ph2B(6-(CF3)Py)2]Ag(C2H4) (9) |
|---|---|---|---|
| a The Ag⋯C(B) is the ipso-carbon separation between the Ag and flanking phenyl group. b ∑ at Ag represents the sum of angles at silver involving two nitrogen atoms bonded to Ag and the centroid of the ethylene carbons. c Values for the three molecules in the asymmetric unit. d Values for the two molecules in the asymmetric unit. | |||
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C C |
1.347(4) | 1.339(3) | 1.346(2) |
| 1.352(5) | 1.349(3) | ||
| 1.351(4) | |||
| Ag–N | 2.242(2), 2.266(2) | 2.2828(13), 2.2883(13) | 2.2767(10), 2.2732(10) |
| 2.243(2), 2.243(2) | 2.2827(13), 2.2707(13) | ||
| 2.236(2), 2.272(2) | |||
| Ag–C | 2.252(3), 2.262(3) | 2.2697(18), 2.2733(19) | 2.2710(13), 2.2701(13) |
| 2.262(3), 2.241(3) | 2.2724(19), 2.2689(19) | ||
| 2.254(3), 2.266(3) | |||
| ∠N–Ag–N | 84.45(7) | 79.01(5) | 84.46(4) |
| 85.75(7) | 82.55(5) | ||
| 85.94(7) | |||
| ∠C–Ag–C | 34.72(11) | 34.28(8) | 34.49(5) |
| 34.93(12) | 34.56(9) | ||
| 34.78(10) | |||
| Ag⋯C(B)a | 2.872 | 2.720 | 2.6908(11) |
| 2.862 | 2.763 | ||
| 2.841 | |||
| ∑ at Agb | 360.22 | 359.98 | 359.61 |
| 360.11 | 360.00 | ||
| 360.87 | |||
Percent buried volume (%VBur)99,100 of the scorpionate ligands in [Ph2B(3-(CF3)Pz)2]Ag(C2H4) (7), [Ph2B(3-(SF5)Pz)2]Ag(C2H4) (8), and [Ph2B(6-(CF3)Py)2]Ag(C2H4) (9), are 55.0%, 61.4%, and 64.5%, respectively, (Fig. 6). These values can be used to compare the steric bulkiness between the three supporting ligands, and indicate that [Ph2B(3-(CF3)Pz)2]− provides the smallest, while [Ph2B(6-(CF3)Py)2]− provides the highest steric protection to the silver–ethylene moiety in these molecules. This trend aligns with the values computed for the thallium complexes bearing the three ligands examined in this study (see above).
 | ||
Fig. 6 Computed steric maps using SambVca 2.1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 101 for supporting ligands in [Ph2B(3-(CF3)Pz)2]Ag(C2H4) (7, left), in [Ph2B(3-(SF5)Pz)2]Ag(C2H4) (8, middle), and [Ph2B(6-(CF3)Py)2]Ag(C2H4) (9, right), looking down the Ag⋯B axis. 101 for supporting ligands in [Ph2B(3-(CF3)Pz)2]Ag(C2H4) (7, left), in [Ph2B(3-(SF5)Pz)2]Ag(C2H4) (8, middle), and [Ph2B(6-(CF3)Py)2]Ag(C2H4) (9, right), looking down the Ag⋯B axis. | ||
In the 1H NMR spectrum at room temperature, the ethylene protons of 7 and 8 were observed at δ 4.67 and 4.73 ppm, respectively, while 9 shows its ethylene signal at δ 4.69 ppm. These protons likely experience the shielding cone of the B-phenyl group situated above the ethylene moiety, leading to larger than normal upfield shifts relative to the free C2H4 signal (δ 5.40 ppm).71,102 For example, [Me2B(3-(CF3)Py)2]Ag(C2H4), which has a methyl group above the ethylene moiety, exhibits its ethylene proton resonance at a significantly downfield position, δ 5.42 ppm.12 The ethylene carbons in 8 and 9 both appear as broad peaks at δ 103.9 ppm in their 13C{1H} NMR spectra, while in 7 the resonance is further upfield at δ 101.2 ppm. For comparison, the ethylene carbon peak of [Me2B(3-(CF3)Py)2]Ag(C2H4) appears in the same region at δ 102.8 ppm, while the resonance for free C2H4 was observed at δ 123.1 ppm.
Although ring current effects also could influence 13C NMR shifts,103 their through-space effects are generally smaller as they are further away from the flanking phenyl moiety. Thus, 13C NMR shifts of the ethylene carbons serve as a useful probe of bonding interactions. The ethylene 13C NMR resonance in the copper(I) complexes [Ph2B(3-(CF3)Pz)2]Cu(C2H4) and [Ph2B(3-(SF5)Pz)2]Cu(C2H4) have been observed at δ 82.7 and 86.4 ppm, respectively.22 The upfield shifts of metal-bound ethylene 13C resonances relative to free ethylene in these diamagnetic d10-systems are commonly attributed to the increased metal-to-ethylene π back-donation effect.57,71,104 The significantly higher upfield shifts in copper(I) complexes compared to their silver(I) counterparts 7 and 8 suggest that they exhibit greater π back-donation from the copper to ethylene. In fact, silver(I) in general forms the weakest interaction with ethylene among the group 11 family members.105–107 Moreover, because the silver atom is larger than the copper atom,87 replacing copper with silver in complexes bearing the same ligand and co-ligand results in smaller percent buried volumes. For instance, complexes 7 (55.0%) and 8 (61.4%) exhibit smaller buried volumes than their copper analogs [Ph2B(3-(CF3)Pz)2]Cu(C2H4) (59.5%)108 and [Ph2B(3-(SF5)Pz)2]Cu(C2H4) (65.6%),108 as the shorter Cu–ligand distances bring the ligand framework closer to the metal center (Fig. S2).
Silver–acetylene complexes are of considerable interest but remain rare, largely due to their instability toward decomposition.109–111 Without suitable supporting ligands, these complexes are often too unstable to isolate. In our previous work, the [Ph2B(3-(CF3)Pz)2]− (1) ligand enabled the successful isolation and structural characterization of a silver–acetylene complex, with 1H and 13C acetylene chemical shifts in CDCl3 at δ 2.12 and 70.9 ppm.111 Motivated by that result, we investigated the related [Ph2B(3-(SF5)Pz)2]− (2) and [Ph2B(6-(CF3)Py)2]− (3) ligands. The silver–acetylene complex [Ph2B(3-(SF5)Pz)2]Ag(C2H2) (10) was obtained by acetylene replacement of the corresponding ethylene adduct 8. The NMR spectra of 10 in CD2Cl2 confirmed the presence of coordinated acetylene resonances, as evidenced by the δ 2.38 ppm peak in 1H and the 68.6 ppm resonance in 13C, both shifted from those of free C2H2 signals (δ 1.80, 71.9 ppm). However, it was unstable even at −20 °C, decomposing over time into an insoluble material, thereby precluding crystallographic analysis to date. An analogous attempt with [Ph2B(6-(CF3)Py)2]− (3) resulted in rapid decomposition to a white solid and free ligand [Ph2B(6-(CF3)Py)2]H (3-H), as confirmed by 1H NMR, indicating the instability of the pyridyl-based system under such reaction conditions.
Finally, the catalytic behavior of CF3-group bearing bis(pyrazolyl)borate [Ph2B(3-(CF3)Pz)2]Ag(C2H4) (7) and bis(pyridyl)borate [Ph2B(6-(CF3)Py)2]Ag(C2H4) (9) complexes was examined in the context of alkane C–H functionalization through carbene transfer reactions from carbene precursor, ethyl diazoacetate, N2CHCO2Et (EDA). Previous studies from our group and Pérez's group have shown that silver complexes supported by weakly donating tris(pyrazolyl)borate ligands can enhance the electrophilicity of the metal center, thereby improving catalytic efficiency.57,112–119 Compared to copper(I), silver(I) can more effectively activate less reactive C–H bonds, such as those at primary C–H sites.114 Moreover, silver-based catalysts can often perform these reactions without requiring the slow addition of carbene precursors, a procedure commonly necessary for copper(I) systems to suppress the formation of undesired byproducts. Overall, the catalytic data reveal that the nature of the supporting ligand significantly influences both the activity and regioselectivity of the carbene insertion reaction. Catalysts 7 and 8, which contain more Lewis acidic silver centers, afford higher yields of C–H insertion products than catalyst 9, which features the more electron-donating pyridyl-based ligand (see Table 3 and Tables S8, S9 for more details). The greater electrophilicity of the metal center in 7 and 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11 likely facilitates the formation of the silver–carbene intermediate and promotes more efficient C–H bond insertion. In contrast, the increased electron density at the metal center in 9 may reduce its carbene electrophilicity, thereby lowering overall reactivity towards C–H moieties. In addition to these electronic effects, steric factors govern regioselectivity. Complex 9, supported by the bulkier pyridyl-based ligand, favors functionalization at primary C–H sites. This observation suggests that the increased steric congestion around the metal center limits the approach of secondary or tertiary C–H bonds, which are more hindered, and agrees with previous observations.118,120 These results illustrate that both electronic and steric features of the ligand framework can be strategically modulated to fine-tune the reactivity and selectivity of silver(I) carbene-transfer catalysts. Although the effectiveness achieved with some of these bis(pyrazolyl/pyridyl)borate-supported systems – especially the more electron-rich bis(pyridyl)borate-silver system – does not surpass that of more electrophilic and sterically encumbered tris(pyrazolyl)borate catalysts such as [HB(3,5-(CF3)2Pz)3]Ag(C2H4)113 or [HB(3,4,5-(Br)3Pz)3]Ag(THF),120 the data underscore the importance of balancing metal electrophilicity and ligand steric demand in optimizing C–H bond functionalization processes.
11 likely facilitates the formation of the silver–carbene intermediate and promotes more efficient C–H bond insertion. In contrast, the increased electron density at the metal center in 9 may reduce its carbene electrophilicity, thereby lowering overall reactivity towards C–H moieties. In addition to these electronic effects, steric factors govern regioselectivity. Complex 9, supported by the bulkier pyridyl-based ligand, favors functionalization at primary C–H sites. This observation suggests that the increased steric congestion around the metal center limits the approach of secondary or tertiary C–H bonds, which are more hindered, and agrees with previous observations.118,120 These results illustrate that both electronic and steric features of the ligand framework can be strategically modulated to fine-tune the reactivity and selectivity of silver(I) carbene-transfer catalysts. Although the effectiveness achieved with some of these bis(pyrazolyl/pyridyl)borate-supported systems – especially the more electron-rich bis(pyridyl)borate-silver system – does not surpass that of more electrophilic and sterically encumbered tris(pyrazolyl)borate catalysts such as [HB(3,5-(CF3)2Pz)3]Ag(C2H4)113 or [HB(3,4,5-(Br)3Pz)3]Ag(THF),120 the data underscore the importance of balancing metal electrophilicity and ligand steric demand in optimizing C–H bond functionalization processes.
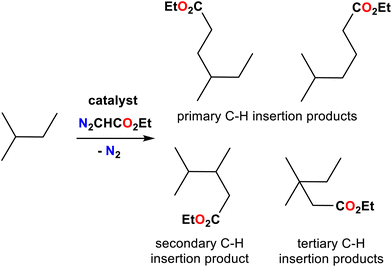
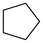
| Entry | Cat. | Substrate | Primary sites | Secondary sites | Tertiary sites | Yields (%) |
|---|---|---|---|---|---|---|
| a Values normalized for the relative number of C–H bonds each type, calculated from the observed carbene insertion product ratios and the number of C–H bonds of each type. | ||||||
| 1 | 7 |
|
— | 1 | — | 90 |
| 2 | 8 | — | 1 | — | 90 | |
| 3 | 9 | — | 1 | — | 61 | |
| 4 | 7 |

|
1 | 2.88 | — | 93 |
| 5 | 8 | 1 | 2.18 | — | 89 | |
| 6 | 9 | 1 | 1.94 | — | 56 | |
| 7 | 7 |
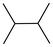
|
1 | — | 7.71 | 96 |
| 8 | 8 | 1 | — | 5.47 | 89 | |
| 9 | 9 | 1 | — | 3.50 | 38 | |
| 10 | 7 |
|
1 | 2.92 | 6.32 | 87 |
| 11 | 8 | 1 | 3.53 | 6.07 | 86 | |
| 12 | 9 | 1 | 2.10 | 3.60 | 56 | |
Conclusions
In summary, fluorinated and B-phenylated bis(pyrazolyl)borate and bis(pyridyl)borate ligands have been synthesized and evaluated as sterically and electronically tunable scorpionate platforms for stabilizing thallium(I) and silver(I) complexes, including rare isolable silver–ethylene adducts. The ligands [Ph2B(3-(CF3)Pz)2]− (1), [Ph2B(3-(SF5)Pz)2]− (2) and [Ph2B(6-(CF3)Py)2]− (3) incorporate strongly electron-withdrawing CF3 or SF5 substituents that introduce pronounced steric shielding; notably, SF5 substituents provide both greater electronegativity and steric bulk relative to CF3, and the bis(pyridyl)borate framework imparts still greater steric congestion around the metal center relative to bis(pyrazolyl)borates. Thallium(I) complexes exhibit characteristic κ2-N,N binding of the scorpionate ligand, along with measurable long-range thallium-203/205-fluorine-19 coupling that correlates directly with through-space Tl⋯F separations. These thallium complexes serve as efficient ligand-transfer reagents to silver, providing access to silver(I) ethylene complexes, in which the 1H and 13C NMR data reveal ethylene resonances shifted upfield relative to those of free C2H4, and their X-ray crystal structures show trigonal-planar silver sites. Comparative 13C NMR data indicate significantly weaker π-backbonding in Ag–ethylene compared to analogous Cu complexes, consistent with established trends in group 11 olefin binding. The use of silver catalysts with varying electronic and steric ligands strongly influences C–H functionalization of alkanes via carbene insertion. More electrophilic metal centers enhance reactivity and yields, while bulkier or more electron-rich ligands favor functionalization at less hindered primary sites. Overall, the work demonstrates how fluorinated substituents and heterocyclic donor arms in scorpionate ligands govern metal–ligand geometry, Tl–F coupling, steric environment, and the stability of labile silver π-complexes, and provides a versatile platform for developing catalysts for selective C–H functionalization.Experimental section
General information
All preparations and manipulations were carried out under an atmosphere of purified nitrogen using standard Schlenk techniques or in a vacuum atmosphere single-station dry box equipped with a −25 °C refrigerator. NMR solvents were purchased from Cambridge Isotopes Laboratories and used as received. Dichloromethane, toluene, and hexane were dried by passing HPLC-grade solvent through a solvent purification system (SPS, Innovative Technologies Inc.) and stored in Straus flasks. NMR spectra were recorded at 25 °C on a JEOL Eclipse 500 (1H, 500 MHz; 13C, 126 MHz; and 19F, 471 MHz), JEOL Eclipse 400 spectrometer (1H, 400 MHz; 13C, 100 MHz, 19F, 376 MHz), or JEOL Eclipse 300 (1H, 300 MHz; 13C, 75 MHz; and 19F, 273 MHz). 1H and 13C{1H} NMR spectra are referenced to the solvent peak (1H, CDCl3δ 7.26; 13C, CDCl3δ 77.16, CD2Cl2δ 54.00). 19F NMR values were referenced to external CFCl3. 1H, 13C{1H}, and 19F NMR chemical shifts are reported in ppm and coupling constants (J) are reported in hertz (Hz). Abbreviations used for signal assignments: Py = pyridyl, Ph = phenyl, s = singlet, d = doublet, t = triplet, q = quartet, pent = pentet, m = multiplet, br = broad, brs = broad singlet. Elemental analyses were performed using a PerkinElmer Model 2400 CHN analyzer. IR spectra were collected at room temperature on a Shimadzu IR Prestige-21 FTIR equipped with an ATR attachment using pure liquid or solid samples, with a resolution of 2 cm−1. Raman data were collected on a Thermo Scientific DXR3 Raman microscope using a 633 nm HeNe laser, with pure solid materials placed on a glass slide. [Ph2B(6-(CF3)Py)2]K,11 Ph2BH2Li,81 and MeB(OiPr)2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 121 were synthesized as previously reported. Compound [Ph2B(3-(CF3)Pz)2]Tl22 was prepared by a modified process as noted below. All other reactants and reagents were purchased from commercial sources or obtained as noted below.
121 were synthesized as previously reported. Compound [Ph2B(3-(CF3)Pz)2]Tl22 was prepared by a modified process as noted below. All other reactants and reagents were purchased from commercial sources or obtained as noted below.
Synthetic procedures
Warning: Thallium salts are toxic; therefore, these compounds should be handled with proper precautions.122 Care must be taken to prevent exposure through inhalation, accidental ingestion via contaminated hands or gloves, or skin contact. Waste products should be disposed of properly. Silver in combination with acetylene gas can lead to potentially explosive materials and should be prepared in small quantities and handled with appropriate precautions. While no difficulties were encountered with the complexes reported herein, caution should be exercised. Due care must also be taken when working with acetylene gas and handling acetylene tanks. It is known to produce explosive combinations with oxygen.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C). 19F NMR (CDCl3): δ (ppm) −61.5 (s).
C). 19F NMR (CDCl3): δ (ppm) −61.5 (s).
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 102.6 (Pz). 19F NMR (CDCl3): δ (ppm) 81.70 (pent, 2JF–F = 154.8 Hz, 2F), 64.53 (d, 2JF–F = 154.8 Hz, 8F).
C), 102.6 (Pz). 19F NMR (CDCl3): δ (ppm) 81.70 (pent, 2JF–F = 154.8 Hz, 2F), 64.53 (d, 2JF–F = 154.8 Hz, 8F).
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C). 19F NMR (CD2Cl2): δ (ppm) 81.5 (pent, 2JF–F = 154.8 Hz, 2F), 64.2 (d, 2JF–F = 154.8 Hz, 8F). Note: the compound readily loses acetylene under vacuum. The compound is not stable even at −20 °C for a long time. A white insoluble precipitate appears in methylene chloride. The complex decomposes in NMR solvent.
C). 19F NMR (CD2Cl2): δ (ppm) 81.5 (pent, 2JF–F = 154.8 Hz, 2F), 64.2 (d, 2JF–F = 154.8 Hz, 8F). Note: the compound readily loses acetylene under vacuum. The compound is not stable even at −20 °C for a long time. A white insoluble precipitate appears in methylene chloride. The complex decomposes in NMR solvent.
X-ray structure determinations
A suitable crystal covered with a layer of hydrocarbon/Paratone-N oil was selected and mounted on a Cryo-loop, and immediately placed in the low temperature nitrogen stream. The X-ray intensity data were measured at 100(2) K (unless otherwise noted) on a Bruker Smart ApexII or Bruker D8 Quest equipped with an Oxford Cryosystems 700 series cooler, a graphite monochromator, and a Mo Kα fine-focus sealed tube (λ = 0.71073 Å). Intensity data were processed, and absorption corrections were applied using the Bruker Apex program suite. Initial atomic positions were located by SHELXT,123 and the structures of the compounds were refined by the least-squares method using SHELXL124 within Olex2 GUI.125 Compounds [Ph2B(3-(SF5)Pz)2]Tl (5) and [Ph2B(3-(SF5)Pz)2]Ag(C2H4) (8) and [Ph2B(3-(CF3)Pz)2]Ag(C2H4) (7) crystallize with two, two, and three chemically identical but crystallographically different molecules of each type in the asymmetric unit, respectively. Fluorine atoms in one of the CF3 groups of [Ph2B(3-(CF3)Pz)2]Ag(C2H4) (7), and the equatorial fluorine atoms of one of the SF5 groups of [Ph2B(3-(SF5)Pz)2]Ag(C2H4) show positional disorder, but it was modeled satisfactorily. All the non-hydrogen atoms in 4–9 were refined anisotropically. Hydrogen atoms of the ethylene carbons of [Ph2B(6-(CF3)Py)2]Ag(C2H4) (9) were located on difference maps, included and refined freely. The remaining hydrogen atoms of 9 and those of 4–8 were included at calculated positions and refined using appropriate riding models. X-ray structural figures were generated using Olex2.Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information (SI): NMR spectroscopic data, steric volumes, additional figures, summaries of data collection and refinement, catalysis, and references. See DOI: https://doi.org/10.1039/d5dt02782a.CCDC 2498963–2498968 and 2504337 contain the supplementary crystallographic data for this paper.126a–g
Acknowledgements
This research was supported in part by the USDA REE, Agricultural Research Services (58-2040-3-017), and a grant from the American Floral Endowment, an industry-funded nonprofit dedicated to sustaining and advancing floriculture research. We also acknowledge the support from The University of Texas at Arlington.References
- S. Trofimenko, J. Am. Chem. Soc., 1966, 88, 1842–1844 CrossRef CAS.
- S. Trofimenko, Chem. Rev., 1993, 93, 943–980 CrossRef CAS.
- C. Pettinari and C. Santini, Polypyrazolylborate and scorpionate ligands, in Comprehensive Coordination Chemistry II: From Biology to Nanotechnology, ed. J. A. McCleverty and T. J. Meyer, Elsevier, Amsterdam, 2003, vol. 1, pp. 159–210 Search PubMed.
- T. G. Hodgkins, Inorg. Chem., 1993, 32, 6115–6116 CrossRef CAS.
- T. G. Hodgkins and D. R. Powell, Inorg. Chem., 1996, 35, 2140–2148 CrossRef CAS.
- B. L. Conley and T. J. Williams, J. Am. Chem. Soc., 2010, 132, 1764–1765 CrossRef CAS PubMed.
- B. L. Conley, D. Guess and T. J. Williams, J. Am. Chem. Soc., 2011, 133, 14212–14215 CrossRef CAS PubMed.
- Z. Lu, B. Malinoski, A. V. Flores, B. L. Conley, D. Guess and T. J. Williams, Catalysts, 2012, 2, 412–421 CrossRef CAS.
- J. A. Celaje, M. K. Pennington-Boggio, R. W. Flaig, M. G. Richmond and T. J. Williams, Organometallics, 2014, 33, 2019–2026 CrossRef CAS PubMed.
- M. K. Pennington-Boggio, B. L. Conley, M. G. Richmond and T. J. Williams, Polyhedron, 2014, 84, 24–31 CrossRef CAS PubMed.
- M. Vanga, A. Noonikara-Poyil, J. Wu and H. V. R. Dias, Organometallics, 2022, 41, 1249–1260 CrossRef CAS.
- B. T. Watson, M. Vanga, A. Noonikara-Poyil, A. Munoz-Castro and H. V. R. Dias, Inorg. Chem., 2023, 62, 1636–1648 CrossRef CAS PubMed.
- S. Pal, P. Y. Zavalij and A. N. Vedernikov, Organometallics, 2015, 34, 5183–5190 CrossRef CAS.
- E. Khaskin, P. Y. Zavalij and A. N. Vedernikov, J. Am. Chem. Soc., 2006, 128, 13054–13055 CrossRef CAS PubMed.
- V. A. Krylova, P. I. Djurovich, B. L. Conley, R. Haiges, M. T. Whited, T. J. Williams and M. E. Thompson, Chem. Commun., 2014, 50, 7176–7179 RSC.
- S. Pal and A. N. Vedernikov, Dalton Trans., 2012, 41, 8116–8122 RSC.
- A. Gogoi, P. Singh, S. Pal and M. Dixit, Inorg. Chem., 2022, 61, 10283–10293 CrossRef CAS PubMed.
- S. Stipurin and T. Strassner, Organometallics, 2024, 43, 1726–1735 CrossRef CAS.
- H. V. R. Dias and J. D. Gorden, Inorg. Chem., 1996, 35, 318–324 CrossRef CAS PubMed.
- T. T. Ponduru, Z. Sun, T. R. Cundari and H. V. R. Dias, ChemCatChem, 2019, 11, 4966–4973 CrossRef CAS.
- A. Noonikara-Poyil, S. G. Ridlen and H. V. R. Dias, Inorg. Chem., 2020, 59, 17860–17865 CrossRef CAS PubMed.
- A. Noonikara-Poyil, A. Munoz-Castro, A. Boretskyi, P. K. Mykhailiuk and H. V. R. Dias, Chem. Sci., 2021, 12, 14618–14623 RSC.
- C. Pettinari, Scorpionates II: Chelating Borate Ligands, World Scientific, 2008 Search PubMed.
- P. J. Fischer, in Comprehensive Coordination Chemistry III, ed. E. C. Constable, G. Parkin and L. Que Jr, Elsevier, Oxford, 2021, pp. 428–504, DOI:10.1016/B978-0-08-102688-5.00072-6.
- L. H. Doerrer and H. V. R. Dias, Dalton Trans., 2023, 52, 7770–7771 RSC.
- S. M. Park, M. Wei, J. Xu, H. R. Atapattu, F. T. Eickemeyer, K. Darabi, L. Grater, Y. Yang, C. Liu, S. Teale, B. Chen, H. Chen, T. Wang, L. Zeng, A. Maxwell, Z. Wang, K. R. Rao, Z. Cai, S. M. Zakeeruddin, J. T. Pham, C. M. Risko, A. Amassian, M. G. Kanatzidis, K. R. Graham, M. Grätzel and E. H. Sargent, Science, 2023, 381, 209–215 CrossRef CAS PubMed.
- J. C. Páez-Franco, M. R. Zermeño-Ortega, C. M. de la O-Contreras, D. Canseco-González, J. R. Parra-Unda, A. Avila-Sorrosa, R. G. Enríquez, J. M. Germán-Acacio and D. Morales-Morales, Pharmaceutics, 2022, 14, 402 CrossRef PubMed.
- R. Pedrazzani, S. Kiriakidi, M. Monari, I. Lazzarini, G. Bertuzzi, C. S. López and M. Bandini, ACS Catal., 2024, 14, 6128–6136 CrossRef CAS.
- W. Chen, L. Xu, Y. Hu, A. M. Banet-Osuna and J. Xiao, Tetrahedron, 2002, 58, 3889–3899 CrossRef CAS.
- S. Mishra and S. Daniele, Chem. Rev., 2015, 115, 8379–8448 CrossRef CAS PubMed.
- A. C. Sather, H. G. Lee, V. Y. De La Rosa, Y. Yang, P. Müller and S. L. Buchwald, J. Am. Chem. Soc., 2015, 137, 13433–13438 CrossRef CAS PubMed.
- S. Dehnen, L. L. Schafer, T. Lectka and A. Togni, Organometallics, 2021, 40, 3858–3864 CrossRef CAS.
- V. I. Saloutin, Y. O. Edilova, Y. S. Kudyakova, Y. V. Burgart and D. N. Bazhin, Molecules, 2022, 27, 7894 CrossRef CAS PubMed.
- M. A. García-Monforte, S. Martínez-Salvador and B. Menjón, Eur. J. Inorg. Chem., 2012, 2012, 4945–4966 CrossRef.
- P. K. Mykhailiuk, Chem. Rev., 2021, 121, 1670–1715 CrossRef CAS PubMed.
- O. A. Tomashenko and V. V. Grushin, Chem. Rev., 2011, 111, 4475–4521 CrossRef CAS PubMed.
- C. Alonso, E. Martínez de Marigorta, G. Rubiales and F. Palacios, Chem. Rev., 2015, 115, 1847–1935 CrossRef CAS PubMed.
- T. Furuya, A. S. Kamlet and T. Ritter, Nature, 2011, 473, 470–477 CrossRef CAS PubMed.
- H. Jia, A. P. Häring, F. Berger, L. Zhang and T. Ritter, J. Am. Chem. Soc., 2021, 143, 7623–7628 CrossRef CAS PubMed.
- P. R. Savoie and J. T. Welch, Chem. Rev., 2015, 115, 1130–1190 CrossRef CAS PubMed.
- Y. Yang, L. Han, L. Brettnacher, L. Canavero and A. Tlili, Chem. Rev., 2025, 125, 8426–8476 CrossRef CAS PubMed.
- J. M. W. Chan, J. Mater. Chem. C, 2019, 7, 12822–12834 RSC.
- H.-Y. Yang, H.-B. Du, Y.-P. Bai, Y.-H. Ding, Y.-F. Ma, G. Wu, N. Zhang, N. K. Szymczak and S. Guo, Sci. Adv., 2025, 11, eadw8408 CrossRef CAS PubMed.
- C. Ye, G. L. Gard, R. W. Winter, R. G. Syvret, B. Twamley and J. n. M. Shreeve, Org. Lett., 2007, 9, 3841–3844 CrossRef CAS PubMed.
- P. Paquin, N. DeGrâce, G. Bélanger-Chabot and J.-F. Paquin, J. Org. Chem., 2024, 89, 3552–3562 CrossRef CAS PubMed.
- L. C. Peyrical, L. Vinet, E. Azek and A. B. Charette, Org. Lett., 2024, 26, 10414–10418 CrossRef CAS PubMed.
- N. M. Shavaleev, G. Xie, S. Varghese, D. B. Cordes, A. M. Z. Slawin, C. Momblona, E. Ortí, H. J. Bolink, I. D. W. Samuel and E. Zysman-Colman, Inorg. Chem., 2015, 54, 5907–5914 CrossRef CAS PubMed.
- P. Kenyon and S. Mecking, J. Am. Chem. Soc., 2017, 139, 13786–13790 CrossRef CAS PubMed.
- M. Talavera, S. Hinze, T. Braun, R. Laubenstein and R. Herrmann, Molecules, 2020, 25, 3977 CrossRef CAS PubMed.
- R. D. W. Kemmitt, R. D. Peacock and J. Stocks, J. Chem. Soc. D, 1969, 554a–554a RSC.
- D. Langford, I. Göttker-Schnetmann, F. P. Wimmer, L. A. Casper, P. Kenyon, R. F. Winter and S. Mecking, Organometallics, 2019, 38, 2710–2713 CrossRef CAS.
- M. Vanga, B. T. Diroll, A. Muñoz-Castro, A. Boretskyi, P. Mykhailiuk and H. V. R. Dias, Inorg. Chem., 2025 DOI:10.1021/acs.inorgchem.5c03702.
- A. F. Henwood, J. Webster, D. Cordes, A. M. Z. Slawin, D. Jacquemin and E. Zysman-Colman, RSC Adv., 2017, 7, 25566–25574 RSC.
- H. R. A. Golf, H.-U. Reissig and A. Wiehe, J. Org. Chem., 2015, 80, 5133–5143 CrossRef CAS PubMed.
- L. J. Sæthre, N. Berrah, J. D. Bozek, K. J. Børve, T. X. Carroll, E. Kukk, G. L. Gard, R. Winter and T. D. Thomas, J. Am. Chem. Soc., 2001, 123, 10729–10737 CrossRef PubMed.
- R. Kordnezhadian, B.-Y. Li, A. Zogu, J. Demaerel, W. M. De Borggraeve and E. Ismalaj, Chem. – Eur. J., 2022, 28, e202201491 CrossRef CAS PubMed.
- H. V. R. Dias and C. J. Lovely, Chem. Rev., 2008, 108, 3223–3238 CrossRef CAS PubMed.
- A. Jalil, E. E. Happel, L. Cramer, A. Hunt, A. S. Hoffman, I. Waluyo, M. M. Montemore, P. Christopher and E. C. H. Sykes, Science, 2025, 387, 869–873 CrossRef CAS PubMed.
- T. Pu, H. Tian, M. E. Ford, S. Rangarajan and I. E. Wachs, ACS Catal., 2019, 9, 10727–10750 CrossRef CAS.
- M. O. Ozbek, I. Onal and R. A. van Santen, J. Catal., 2011, 284, 230–235 CrossRef CAS.
- D. J. Safarik and R. B. Eldridge, Ind. Eng. Chem. Res., 1998, 37, 2571–2581 CrossRef CAS.
- R. B. Eldridge, Ind. Eng. Chem. Res., 1993, 32, 2208–2212 CrossRef.
- Y. Ren, X. Liang, H. Dou, C. Ye, Z. Guo, J. Wang, Y. Pan, H. Wu, M. D. Guiver and Z. Jiang, Adv. Sci., 2020, 7, 2001398 CrossRef CAS PubMed.
- B. M. Binder, J. Biol. Chem., 2020, 295, 7710–7725 CrossRef CAS PubMed.
- E. M. Beyer Jr., Plant Physiol., 1976, 58, 268–271 CrossRef PubMed.
- M. E. Saltveit, K. J. Bradford and D. R. Dilley, J. Am. Soc. Hortic. Sci., 1978, 103, 472–475 CAS.
- T. Ziegler and A. Rauk, Inorg. Chem., 1979, 18, 1558–1565 CrossRef CAS.
- D. M. P. Mingos, J. Organomet. Chem., 2001, 635, 1–8 CrossRef CAS.
- H. V. R. Dias, D. Parasar, A. A. Yakovenko, P. W. Stephens, Á. Muñoz-Castro, M. Vanga, P. Mykhailiuk and E. Slobodyanyuk, Chem. Sci., 2024, 15, 2019–2025 RSC.
- I. Krossing and A. Reisinger, Angew. Chem., Int. Ed., 2003, 42, 5725–5728 CrossRef CAS PubMed.
- H. V. R. Dias and J. Wu, Eur. J. Inorg. Chem., 2008, 2008, 509–522 CrossRef.
- H. V. R. Dias, in Comprehensive Coordination Chemistry II, ed. J. A. McCleverty and T. J. Meyer, Pergamon, Oxford, 2003, pp. 383–463, DOI:10.1016/B0-08-043748-6/02003-X.
- M. M. Melero, Z. Kłosek, C. Ramírez de Arellano and A. Olmos, J. Org. Chem., 2023, 88, 9130–9135 CrossRef CAS PubMed.
- M. Kitamura, Y. Takenaka, T. Okuno, R. Holl and B. Wünsch, Eur. J. Inorg. Chem., 2008, 2008, 1188–1192 CrossRef.
- M. Vanga, V. Q. H. Phan, J. Wu, A. Munoz-Castro and H. V. R. Dias, Inorg. Chem., 2023, 62, 18563–18572 CrossRef CAS PubMed.
- J. H. W. LaFortune, B. F. Jacobs and C. L. B. Macdonald, in Comprehensive Coordination Chemistry III, ed. E. C. Constable, G. Parkin and L. Que Jr, Elsevier, Oxford, 2021, pp. 214–280, DOI:10.1016/B978-0-08-102688-5.00029-5.
- C. J. Allan and C. L. B. MacDonald, Comprehensive Inorganic Chemistry II: From Elements to Applications, 2nd edn, 2013, vol. 1, pp. 485–566 Search PubMed.
- P. Ghosh, P. J. Desrosiers and G. Parkin, J. Am. Chem. Soc., 1998, 120, 10416–10422 CrossRef CAS.
- R. Han, P. Ghosh, P. J. Desrosiers, S. Trofimenko and G. Parkin, J. Chem. Soc., Dalton Trans., 1997, 3713–3718 RSC.
- C. Janiak, Main Group Met. Chem., 1998, 21, 33–50 CrossRef CAS.
- M. R. Biscoe, C. Uyeda and R. Breslow, Org. Lett., 2004, 6, 4331–4334 CrossRef CAS PubMed.
- C. Chen and R. F. Jordan, Organometallics, 2010, 29, 3679–3682 CrossRef CAS.
- H. Schmidbaur, Angew. Chem., Int. Ed. Engl., 1985, 24, 893–904 CrossRef.
- M. Bochmann, Coord. Chem. Rev., 2009, 253, 2000–2014 CrossRef CAS.
- H. V. R. Dias, S. Singh and T. R. Cundari, Angew. Chem., Int. Ed., 2005, 44, 4907–4910 CrossRef CAS PubMed.
- S. Alvarez, Dalton Trans., 2013, 42, 8617–8636 RSC.
- B. Cordero, V. Gómez, A. E. Platero-Prats, M. Revés, J. Echeverría, E. Cremades, F. Barragán and S. Alvarez, Dalton Trans., 2008, 2832–2838 RSC.
- C. R. Groom, I. J. Bruno, M. P. Lightfoot and S. C. Ward, Acta Crystallogr., Sect. B, 2016, 72, 171–179 CrossRef CAS PubMed.
- A. Bondi, J. Phys. Chem., 1964, 68, 441–451 CrossRef CAS.
- A. L. Rheingold, L. M. Liable-Sands and S. Trofimenko, Chem. Commun., 1997, 1691–1692, 10.1039/a703349d.
- W. Uhl, Adv. Organomet. Chem., 2004, 51, 53–108 CrossRef CAS.
- P. Ghosh, A. L. Rheingold and G. Parkin, Inorg. Chem., 1999, 38, 5464–5467 CrossRef CAS.
- H. V. R. Dias and J. Thankamani, Acta Crystallogr., Sect. C: Cryst. Struct. Commun., 2013, 69, 959–962 CrossRef CAS PubMed.
- C. Dowling, P. Ghosh and G. Parkin, Polyhedron, 1997, 16, 3469–3473 CrossRef CAS.
- E. R. Sirianni, G. P. A. Yap and K. H. Theopold, Inorg. Chem., 2014, 53, 9424–9430 CrossRef CAS PubMed.
- K. L. V. Mann, J. C. Jeffery, J. A. McCleverty and M. D. Ward, Polyhedron, 1999, 18, 721–727 CrossRef CAS.
- T. Fillebeen, T. Hascall and G. Parkin, Inorg. Chem., 1997, 36, 3787–3790 CrossRef CAS PubMed.
- C. Chen, H. Lee and R. F. Jordan, Organometallics, 2010, 29, 5373–5381 CrossRef CAS.
- L. Falivene, Z. Cao, A. Petta, L. Serra, A. Poater, R. Oliva, V. Scarano and L. Cavallo, Nat. Chem., 2019, 11, 872–879 CrossRef CAS PubMed.
- L. Falivene, R. Credendino, A. Poater, A. Petta, L. Serra, R. Oliva, V. Scarano and L. Cavallo, Organometallics, 2016, 35, 2286–2293 CrossRef CAS.
- L. Falivene, Z. Cao, A. Petta, L. Serra, A. Poater, R. Oliva, V. Scarano and L. Cavallo, Nat. Chem., 2019, 11, 872–879 CrossRef CAS PubMed.
- H. V. R. Dias and J. Wu, Organometallics, 2012, 31, 1511–1517 CrossRef CAS.
- R. D. Vernet and V. Boekelheide, Proc. Natl. Acad. Sci. U. S. A., 1974, 71, 2961–2964 CrossRef CAS PubMed.
- A. B. Kazi, H. V. R. Dias, S. M. Tekarli, G. R. Morello and T. R. Cundari, Organometallics, 2009, 28, 1826–1831 CrossRef CAS.
- D. V. Karade, V. Q. H. Phan and H. V. R. Dias, Dalton Trans., 2024, 53, 10426–10433 RSC.
- B. T. Watson and H. V. R. Dias, Chem. Commun., 2024, 60, 4872–4889 RSC.
- C. Elschenbroich, Organometallics, WILEY-VCH, Weinheim, 3rd edn, 2016 Search PubMed.
- A. Noonikara-Poyil, A. Muñoz-Castro, A. Boretskyi, P. K. Mykhailiuk and H. V. R. Dias, Chem. Sci., 2024, 15, 1159–1161 RSC.
- H. V. R. Dias, Z. Wang and W. Jin, Inorg. Chem., 1997, 36, 6205–6215 CrossRef CAS.
- A. Reisinger, N. Trapp, I. Krossing, S. Altmannshofer, V. Herz, M. Presnitz and W. Scherer, Angew. Chem., Int. Ed., 2007, 46, 8295–8298 CrossRef CAS PubMed.
- A. Noonikara-Poyil, S. G. Ridlen, I. Fernández and H. V. R. Dias, Chem. Sci., 2022, 13, 7190–7203 RSC.
- M. M. Díaz-Requejo and P. J. Pérez, Chem. Rev., 2008, 108, 3379–3394 CrossRef PubMed.
- H. V. R. Dias, R. G. Browning, S. A. Richey and C. J. Lovely, Organometallics, 2004, 23, 1200–1202 CrossRef CAS.
- M. Álvarez, F. Molina and P. J. Pérez, J. Am. Chem. Soc., 2022, 144, 23275–23279 CrossRef PubMed.
- N. B. Jayaratna, D. B. Pardue, S. Ray, M. Yousufuddin, K. G. Thakur, T. R. Cundari and H. V. R. Dias, Dalton Trans., 2013, 42, 15399–15410 RSC.
- A. Caballero, E. Despagnet-Ayoub, M. M. Díaz-Requejo, A. Díaz-Rodríguez, M. E. González-Núñez, R. Mello, B. K. Muñoz, W.-S. Ojo, G. Asensio, M. Etienne and P. J. Pérez, Science, 2011, 332, 835–838 CrossRef CAS PubMed.
- R. Gava, A. Olmos, B. Noverges, T. Varea, E. Álvarez, T. R. Belderrain, A. Caballero, G. Asensio and P. J. Pérez, ACS Catal., 2015, 5, 3726–3730 CrossRef CAS.
- K. Rangan, M. Fianchini, S. Singh and H. V. R. Dias, Inorg. Chim. Acta, 2009, 362, 4347–4352 CrossRef CAS.
- C. J. Lovely, J. A. Flores, X. Meng and H. V. R. Dias, Synlett, 2009, 129–132 CAS.
- J. Urbano, T. R. Belderraín, M. C. Nicasio, S. Trofimenko, M. M. Díaz-Requejo and P. J. Pérez, Organometallics, 2005, 24, 1528–1532 CrossRef CAS.
- H. C. Brown and T. E. Cole, Organometallics, 1983, 2, 1316–1319 CrossRef CAS.
- S. Galván-Arzate and A. Santamaría, Toxicol. Lett., 1998, 99, 1–13 CrossRef PubMed.
- G. Sheldrick, Acta Crystallogr., Sect. A: Found. Adv., 2015, 71, 3–8 CrossRef PubMed.
- G. Sheldrick, Acta Crystallogr., Sect. C: Struct. Chem., 2015, 71, 3–8 Search PubMed.
- O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard and H. Puschmann, J. Appl. Crystallogr., 2009, 42, 339–341 CrossRef CAS.
- (a) CCDC 2498963: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2pwcqp; (b) CCDC 2498964: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2pwcrq; (c) CCDC 2498965: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2pwcsr; (d) CCDC 2498966: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2pwcts; (e) CCDC 2498967: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2pwcvt; (f) CCDC 2498968: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2pwcwv; (g) CCDC 2504337: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2q1z2v.
| This journal is © The Royal Society of Chemistry 2026 |