Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Ligand decorated nickel-based nanoparticles supported onto MXenes in catalytic hydrogenation of N-heterocycles
Judith
Medina-Vargas
 a,
Santiago
Martín
a,
Santiago
Martín
 bcd,
Iván
Sorribes
bcd,
Iván
Sorribes
 a,
Hermenegildo
García
a,
Hermenegildo
García
 *ae and
Jose A.
Mata
*ae and
Jose A.
Mata
 *a
*a
aInstitute of Advanced Materials (INAM), Universitat Jaume I, Avda. Sos Baynat s/n, 12071, Castellón, Spain. E-mail: jmata@uji.es
bInstituto de Nanociencia y Materiales de Aragón (INMA), CSIC-Universidad de Zaragoza, 50009, Zaragoza, Spain
cDepartamento de Química Física, Universidad de Zaragoza, 50009, Zaragoza, Spain
dLaboratorio de Microscopias Avanzadas (LMA), Universidad de Zaragoza, Edificio I+D+i., 50018, Zaragoza, Spain
eInstituto de Tecnología Química, Consejo Superior de Investigaciones Científicas-Universitat Politècnica de València, Avda. Los Naranjos s/n, 46022, Valencia, Spain
First published on 18th February 2026
Abstract
The development of efficient first-row transition metal catalysts is essential for advancing sustainable chemical processes. In this study, we report the synthesis of nickel-based nanoparticles (NiNPs) functionalized with N-heterocyclic carbene ligands and immobilized onto Ti3C2 MXene. Our convergent synthetic approach enables comprehensive and straightforward characterization of each component within the final hybrid material. The NiNPs are obtained through chemical reduction of a well-defined nickel organometallic complex, resulting in the formation of small nickel metal nanoparticles (3.0 ± 0.8 nm) that are rapidly oxidized to the corresponding NiO and Ni(OH)2 based nanoparticles containing surface NHC ligands. The hybrid catalyst exhibits high activity and selectivity in the hydrogenation of N-heterocycles under hydrogenation conditions, achieving quantitative yields at low catalyst loadings, particularly notable for a nickel-based system. Recycling studies revealed progressive catalyst deactivation, primarily due to sintering of NiNPs, which reduces the number of active surface sites. However, the catalytic activity can be fully restored through a mild regeneration treatment under reducing conditions. These findings underscore the potential of NiNP/MXene-based materials for selective hydrogenation reactions, and highlight the importance of addressing key challenges in sustainability such as the use of non-noble metals, catalyst stability and recyclability. Further design modifications aimed at preventing nanoparticle sintering may enhance the long-term viability of these systems in catalytic hydrogenation processes.
Introduction
Nickel nanoparticles (NiNPs) have emerged as valuable components in heterogeneous catalysis owing to the natural abundance and low cost of nickel, as well as the distinctive electronic and structural features that enable fine control over their catalytic behavior.1–5 Their catalytic versatility expands across a wide range of transformations including hydrogenation,6–8 cross coupling reactions,9–11 exchange processes,12 reforming13 and electrocatalysis.14–16 The catalytic performance of NiNPs is strongly linked to surface stabilizers such as surfactants and ligands, as well as interactions with their supports.17–19 The presence of surface modifiers confers stability and often induces selectivity,20–23 while the support provides a structural matrix that prevents sintering and promotes the dispersion of nanoparticles, thereby increasing the number of accessible active sites.24–28Traditional supports, including metal oxides (e.g., Al2O3, SiO2, and TiO2), carbon-based materials (e.g., graphene and derivatives, carbon nanotubes, and carbon nitride) and porous frameworks (e.g., MOFs and zeolites) have provided valuable platforms for metal nanoparticle immobilization. However, the application of MXenes as a support for metal nanoparticles in thermal catalysis is still relatively underexplored. MXenes are a family of two-dimensional transition metal carbides and nitrides with the general formula Mn+1Xn (M = early transition metal, X = C, N, n = 1–4).29 MXenes are promising materials for diverse applications30–33 including catalysis34–42 and as support of metal nanoparticles where they offer a suitable 2D morphology and strong metal–support interactions.
Among catalytic transformations, the hydrogenation of quinolines and related N-heterocycles is of particular significance. These reactions are key steps in fine-chemical and pharmaceutical synthesis,43–45 and play a pivotal role in liquid organic hydrogen carrier (LOHC) technology, where N-heterocycles act as reversible hydrogen storage media.46–48 Developing nickel-based catalysts that combine high activity, selectivity, and recyclability under mild conditions is therefore essential for sustainable hydrogenation chemistry.
In recent years, significant progress has been achieved in the development of nickel-based catalysts for the hydrogenation of N-heterocycles, with numerous systems demonstrating excellent activity (Table S1). For example, the transfer hydrogenation of quinolines using ammonia borane as a hydrogen source has been achieved under mild conditions (25 °C) employing Ni(II) complexes bearing amine ligands and nickel supported on carbonaceous materials.49,50 Several nickel-based catalysts have also enabled quantitative hydrogenation of quinolines under molecular hydrogen at pressures of 20–30 bar and moderate temperatures (120 °C). However, most of these systems require relatively high metal loadings (≥4 mol%).51–56 Moreover, along with the excellent activity of these materials, they also display good recyclability. However, the number of studies focusing on catalyst deactivation mechanisms and/or regeneration strategies remains scarce.
In contrast, the NiNPs/NHC@MXene hybrid catalyst developed in this work achieves 99% yield at moderate temperatures (120 °C, 30 bar H2) with low Ni loading (3.7 mol%), maintaining recyclability and recoverable activity upon reductive regeneration. This performance compares favorably to state-of-the-art systems while employing a non-precious metal and a 2D support. The design of this hybrid system integrates N-heterocyclic carbene (NHC) ligands with the MXene surface and stabilizes nickel species during nanoparticle formation. The presence of the NHC ligand not only controls nanoparticle nucleation and morphology but also enhances electronic stabilization at the metal–support interface.
Herein, we report the synthesis of NiNPs functionalized with N-heterocyclic carbene (NHC) ligands anchored onto the Ti3C2 MXene through a convergent approach. The catalytic properties of this hybrid material in the hydrogenation of quinolines have been evaluated. Catalyst performance, substrate scope and recyclability have been determined, with particular attention to deactivation pathways and potential regeneration strategies. Our methodology enables the independent characterization of all components within the hybrid material and provides opportunities for tuning the active site, paving the way for the design of more effective catalysts in future studies.
Experimental
Materials and methods
The ligand precursor (bromide of methyl-methylpyrene-imidazolium salt) and the nickel–NHC complex (complex 1) were synthesized according to previously reported procedures.57,58 All reagents and solvents were used as received unless otherwise specified. Solvents were dried using a solvent purification system (SPS).Nuclear magnetic resonance (NMR) spectra were recorded on Bruker spectrometers operating at 300 or 400 MHz (1H NMR) and 75 or 100 MHz (13C{1H} NMR), respectively, and referenced to SiMe4 (δ in ppm and J in Hz). NMR spectra were acquired at room temperature with the appropriate deuterated solvent.
Scanning electron microscopy (SEM) was performed using a JEOL 7001F microscope operating at 15 kV coupled with a Leica Zeiss LEO 440i system equipped with a 250 and 350 INCA (Oxford) software for the EDX analysis.
High-resolution transmission electron microscopy (HRTEM) images and high-angle annular dark-field images (HAADF-STEM) of the samples were acquired using a Jem-2100 LaB6 (JEOL) transmission electron microscope coupled with an INCA Energy TEM 200 (Oxford) energy dispersive X-Ray spectrometer (EDX) operating at 200 kV. Sample preparation: a drop of the methanol-dispersed sample was deposited onto a carbon-coated copper grid and air-dried.
X-ray diffraction (XRD) patterns were recorded with a D8 Advance diffractometer from Bruker-AXS, with a Bragg–Brentano θ/2θ geometry and Cu Ka radiation. The data were collected from 5° to 70°.
The nickel content in the hybrid material was determined by inductively coupled plasma mass spectrometry (ICP-MS). A 15 mg sample was digested using 69% nitric acid in a Mars 6 iWave microwave digestion system (CEM) under the following conditions: 1800 W power, 210 °C, and 25 minutes. After digestion, the sample was diluted to an appropriate volume (10 mL). ICP-MS measurements were performed in duplicate using an Agilent 7500 CX instrument.
X-ray photoelectron spectra (XPS) were acquired on a Kratos AXIS ultra-DLD spectrometer with a monochromatic Al Kα X-ray source (1486.6 eV) using a pass energy of 20 eV. All binding energies were calibrated with respect to the C 1s peak at 284.6 eV to ensure accurate referencing.
Gas chromatography (GC) analysis was performed on a Shimadzu GC-2010 apparatus equipped with a flame ionization detector (FID) and using a Teknokroma column (TRB-5MS; 30 m × 0.25 mm × 0.25 µm). Temperature program: (1) 80 °C, 10 °C min−1 until 150 °C, and 1 min. (2) 150 °C, 20 °C min−1 until 300 °C, and 2 min. Total time: 17.5 min (Fig. S1 & S2).
Hydrogen temperature programmed reduction (H2-TPR) studies were carried out on a Micromeritics AutoChem apparatus (2910) connected to a thermal conductivity detector (TCD). Before the test, samples were pretreated under an Ar atmosphere at room temperature for 15 min. H2-TPR analysis was carried out from 30 to 900 °C under a 10% H2/Ar flow (50 mL min−1) with a heating rate of 10 °C min.
Synthetic procedures
Catalytic procedures and experiments
Results and discussion
Synthesis and characterization
NiNPs decorated with NHCs supported on Ti3C2 MXene were synthesized via a convergent approach (Fig. 1). Ti3C2 MXene nanosheets were first prepared by etching the Al layer of the Ti3AlC2 MAX phase using HF. In parallel, the nickel complex 1 was synthesized via metallation of an NHC ligand precursor containing a pyrene moiety (Scheme S1). This NHC-pyrene ligand was selected based on prior studies demonstrating its effectiveness in anchoring metal complexes onto 2D materials61–63 and its role in stabilizing metal nanoparticles (Fig. S3 & S4).45,64–66 Metal complex 1 was then chemically reduced in a controlled manner using tBuNH2·BH3, resulting in the formation of NiNPs. These NiNPs, stabilized by NHC ligands, were in situ supported on the MXene surface, yielding the hybrid material NiNPs/NHC@MXene. This convergent synthetic strategy enables rational and precise control over the material formation, while also facilitating the characterization of both the MXene support and the active catalytic site.The Ti3AlC2 MAX phase, Ti3C2 MXene and MXene decorated with NiNPs (NiNPs/NHC@MXene), and the corresponding nickel complex were characterized using different microscopic, spectroscopic and analytical techniques. Detailed experimental procedures and additional materials are provided in the SI. Systematic characterization at each stage of synthesis enables a comprehensive understanding of the material properties and the structural and compositional changes occurring throughout the synthetic process.
Morphological analysis of Ti3C2 MXenevia SEM microscopy confirms the successful removal of the aluminum layer from the parent Ti3AlC2 (Fig. 2a). The initially compact laminar structure of Ti3AlC2 is transformed into a characteristic accordion-like morphology using HF (Fig. 2b). This morphological change provides direct evidence of the success of the etching procedure (Fig. S5–S7). This structural change is further corroborated by EDX analysis (Fig. S8). Comparison of the EDX analysis results of the MAX and MXene samples reveals a significant reduction in aluminum content and the appearance of fluorine on the MXene sample. The microscopic characterization of NiNPs/NHC@MXene required the use of HRTEM to observe the presence of NiNPs due to a low contrast and small size of the NiNPs. Despite these challenges, HRTEM images clearly confirm the formation of spherical NiNPs uniformly distributed on the MXene surface (Fig. 2c). Size distribution analysis was performed by analyzing ca. 200 NiNPs from different HRTEM images. The results indicate an average particle diameter of 3.0 ± 0.8 nm, with a relatively narrow distribution (Fig. 2d). EDX analysis further confirmed nickel as a major component in the nanoparticles (Fig. S9 & S10).
 | ||
| Fig. 2 Morphological characterization. (a) SEM image of the Ti3AlC2 MAX phase. (b) SEM image of Ti3C2 MXene. (c) HRTEM image of NiNPs/NHC@MXene and (d) size histogram of NiNPs/NHC@MXene (N = 200). | ||
Further evidence in the transformation of the Ti3AlC2 MAX phase into the Ti3C2 MXene and the successful incorporation of NiNPs to form NiNPs/NHC@MXene was obtained through comparative powder X-ray diffraction analysis (XRD) (Fig. 3). The Ti3AlC2 MAX phase exhibits sharp reflections of the hexagonal phase (space group P63/mmc) at 10°, 19° and 39° with Miller indices (002), (004) and (104), respectively (JCPDS 52-0875). These peaks are characteristic of the Ti3AlC2 MAX phase. The high crystallinity and narrow peaks indicate a well-ordered layered structure composed of Ti–C octahedral slabs interleaved with Al atomic layers.67 In contrast, the Ti3C2 MXene displays broad low-intensity peaks, characteristic of its lamellar structure and reduced crystallinity. A key distinction between the Ti3C2 MXene and the parent Ti3AlC2 MAX phase is the shift of the (002) peak to lower angles (from 10° to 7.5°), attributed to the removal of aluminum atoms. Additionally, the disappearance of the (104) peak at 39° confirms the complete elimination of the aluminum layer and verifies the formation of a pure Ti3C2 MXene phase.
The presence of NiNPs does not disturb the XRD pattern of the Ti3C2 MXene. A slight shift of the (002) peak of Ti3C2 MXene towards lower angles is observed (from 7.5° to 6.8°), indicating an increase in interlayer spacing which supports the insertion of NiNPs into the MXene layers. However, no distinct peaks corresponding to nickel-based nanoparticles (Ni metal, NiO and/or Ni(OH)2) are detected, probably due to their low concentration of nickel relative to the Ti3C2 MXene matrix (1/25 ratio in weight).
X-ray diffraction analysis confirms the transformation of the Ti3AlC2 MAX phase to the Ti3C2 MXene and provides indirect evidence on the intercallation of nickel-based nanoparticles into the MXene structure; however, this does not confirm the exact nature (Ni, NiO or Ni(OH)2) of these nickel-based nanoparticles.
Further insights into the chemical composition and oxidation states of elements in the hybrid material NiNPs/NHC@MXene were obtained by X-Ray Photoelectron Spectroscopy (XPS). The survey spectrum of MXene confirms the presence of Ti, C, O and F along with the disappearance of the Al signal observed in the Ti3AlC2 MAX phase (Fig. 4), clearly indicating the successful etching of the aluminum layer using HF. The high-resolution XPS spectrum peak of Ti 2p in Ti3C2 was deconvoluted into components corresponding to Ti–C, C–Ti–F and C–Ti–O (model shown in Fig. 5). The Ti–C component originates from the titanium-carbon bonds within the internal layers of MXene, while the C–Ti–F and C–Ti–O peaks confirm surface functionalization resulting from the etching process (Fig. 4).68
 | ||
| Fig. 4 Comparative XPS analysis of the Ti3AlC2 MAX phasevs. the Ti3C2 MXene. Survey spectrum and high-resolution core-level peaks corresponding to Ti 2p, C 1s and O 1s. | ||
 | ||
| Fig. 5 Model structures of the Ti3AlC2 MAX phase, Ti3C2 MXene and the NiNPs/NHC@MXene showing the main contributions observed by XPS analysis. | ||
Analysis of the high-resolution XPS C 1s spectrum supports the carbide nature of the material. The deconvoluted peaks at 281.9 eV and 283.2 eV are assigned to C–Ti bonding environments within the internal layers of the MXene. Peaks corresponding to C–T (T = F, O, OH) surface terminations are barely detectable due to their low relative abundance. A dominant graphitic C–C peak is observed at 284.6 eV, along with a small C–O peak at 286.1 eV, likely arising from partial oxidation by atmospheric oxygen.69,70
The high-resolution XPS data of the O 1s region reveal the presence of surface terminal oxygen atoms bonded to titanium. Additionally, the Ofcc component, indicative of oxygen atoms on the face-centered cubic (fcc) lattice sites, is typically associated with well-ordered metal oxides such as Al2O3 and TiO2 and may represent residual oxide impurities.71 Further details of XPS analysis can be found in the SI (Fig. S11 and S12).
XPS characterization was also performed on the NiNPs/NHC@MXene material (Fig. 6). The corresponding survey spectra and additional XPS details are provided in the SI (Fig. S13). The survey spectrum of the as-prepared material reveals the presence of titanium, nickel, carbon, nitrogen, fluorine, bromine, and oxygen species, verifying the coexistence of the MXene support, nickel-base nanoparticles, and nitrogen-containing NHC ligands. In the Ti 2p region, distinct peaks corresponding to Ti–C (Ti 2p3/2 at 454.5 eV) confirm the retention of the carbide structure of Ti3C2. Additional components at 456.0, 457.6, 460.1, 461.3, and 462.3 eV are attributed to Ti2+, Ti–O2, and higher oxidation state Ti3+/Ti4+, indicative of partial surface oxidation of Ti atoms during etching and post-synthesis air exposure. This is a common feature in MXenes, where surface Ti atoms readily form terminal –O and –OH groups. The Ti 2p spectrum of the NiNPs/NHC@MXene material shows similar components to the parent Ti3C2 MXene. The high-resolution Ni 2p core-level spectrum shows characteristic peaks at 856.4 eV and 859.3 eV in the Ni 2p3/2 region, corresponding to Ni–O and Ni–OH, respectively. The 856.4 eV peak may also indicate Ni–F bonding, which can arise from interactions between the nickel- and fluorine-terminated MXene surface. These interactions likely result from functional groups (O, OH, and F) formed on the MXene surface during the etching process and contribute to the stabilization of nickel species. The satellite Ni peaks are observed at 862.9 eV (Ni 2p3/2) and 881.1 eV (Ni 2p1/2), further confirming the oxidized state of nickel species. The absence of a distinct metallic Ni0 peak around 852.6 eV suggests that the surface of the Ni nanoparticles is largely oxidized to NiO/Ni(OH)2 under ambient conditions. The XPS provides surface information of the top few nanometers (penetration depth: 7–10 nm), and it could be argued that the nanoparticles could retain a metallic Ni core beneath this oxidized shell. However, this behavior is not expected in the NiNPs anchored on Ti3C2 MXene due to the small size of the nanoparticles (3.0 ± 0.8 nm).72–74 In the N 1s region, a prominent peak at 401.2 eV corresponds to carbene-type nitrogen from the NHC ligand bound to the Ni surface, evidencing its presence in the hybrid material. A secondary peak at 399.4 eV is assigned to adventitious or pyridinic nitrogen, likely arising from trace adsorbates.
 | ||
| Fig. 6 XPS analysis of NiNPs/NHC@MXene. Survey spectrum and high-resolution core-level peaks corresponding to Ti 2p, N 1s and Ni 2p. | ||
These features collectively confirm the successful anchoring of Ni nanoparticles functionalized with NHC ligands on the Ti3C2 MXene surface. The nature of these NiNPs is consistent with a NiO/Ni(OH)2 species containing NHC ligands.
Catalytic properties
Catalytic studies were initiated by optimizing the reaction conditions for the hydrogenation of quinoline (1D), selected as the model substrate. The reaction conditions were systematically optimized by varying key parameters, including the individual components of the hybrid catalytic material, temperature, hydrogen pressure and solvent. Control experiments conducted without any catalyst or using the Ti3AlC2 MAX phase or the Ti3C2 MXene alone demonstrated that these systems are not active in the hydrogenation of quinoline (1D) (Table 1, entries 1–3). In contrast, the hydrogenation of quinoline (1D) under homogeneous conditions using complex 1 provided 60% yield of the corresponding tetrahydroquinoline (entry 4). These control experiments confirm nickel as the primary active catalytic species in the hydrogenation of quinolines.| Entry | Cat. | Solvent | Temp. (°C) | P H2 (bar) | Conv.a (%) | Yieldb (%) |
|---|---|---|---|---|---|---|
| General reaction conditions: quinoline (1D) (0.1 mmol); catalyst loading (3.7 mol% based on Nisurf); and solvent (2 mL).a Conversion calculated by GC/FID using anisole as an internal standard.b Yield calculated by 1H NMR spectroscopy using 1,3,5-trimethoxybenzene as an external standard.c Catalyst loading: 20 mg.d Catalyst loading: 3.7 mol%. | ||||||
| 1 | — | EtOH | 150 | 30 | 0 | 0 |
| 2 | Ti3AlC2 MAX phase | EtOH | 150 | 30 | 1 | 0 |
| 3 | Ti3C2 MXene | EtOH | 150 | 30 | 3 | 2 |
| 4 | Complex 1d | EtOH | 150 | 30 | 63 | 60 |
| 5 | NiNPs@MXene | EtOH | 150 | 30 | 5 | 5 |
| 6 | Complex 2d | EtOH | 150 | 30 | 35 | 33 |
| 7 | NiNPs/Me2NHC@MXene | EtOH | 150 | 30 | 94 | 90 |
| 8 | NiNPs/NHC@MXene | EtOH | 150 | 30 | 96 | 95 |
| 9 | NiNPs/NHC@MXene | EtOH | 120 | 30 | 99 | 98 |
| 10 | NiNPs/NHC@MXene | EtOH | 100 | 30 | 27 | 12 |
| 11 | NiNPs/NHC@MXene | EtOH | 70 | 30 | 3 | 0 |
| 12 | NiNPs/NHC@MXene | EtOH | 120 | 20 | 56 | 41 |
| 13 | NiNPs/NHC@MXene | EtOH | 120 | 6 | 9 | 7 |
| 14 | NiNPs/NHC@MXene | EtOH | 120 | 1 | 3 | 3 |
| 15 | NiNPs/NHC@MXene | MeOH | 120 | 30 | 98 | 91 |
| 16 | NiNPs/NHC@MXene | 2-PrOH | 120 | 30 | 95 | 94 |
| 17 | NiNPs/NHC@MXene | Toluene | 120 | 30 | 4 | 1 |
| 18 | NiNPs/NHC@MXene | 1,4-Dioxane | 120 | 30 | 6 | 0 |
| 19 | NiNPs/NHC@MXene | o-DCB | 120 | 30 | 7 | 4 |
| 20 | NiNPs/NHC@MXene | p-Xylene | 120 | 30 | 0 | 0 |
| 21 | NiNPs/NHC@MXene | o-Xylene | 120 | 30 | 0 | 0 |
| 22 | NiNPs/NHC@MXene | Hexane | 120 | 30 | 7 | 6 |
We then studied the influence of the NHC ligand in the catalytic performance of the hybrid material. In a first set of experiments, a direct comparison required the preparation of analogous NiNPs supported on MXene, but without the NHC ligand. To achieve this, we followed the same synthetic protocol used for NiNPs/NHC@MXene, replacing the organometallic complex 1 with Ni(NO3)2·6H2O as the nickel source. This procedure resulted in the formation of NiNPs supported on MXene, designated as NiNPs/MXene, lacking the NHC ligand. Characterization by HRTEM microscopy revealed the formation of prismatic nanoobjects (Fig. S14 & S15). The particle size distribution of NiNPs/MXene was more challenging due to the less well-defined morphology. Nevertheless the average particle diameter was estimated to be approximately 4.1 nm (N = 100), which is comparable to that of NiNPs/NHC@MXene (3.0 nm) (Fig. S16). The different morphology obtained using different nickel sources confirmed the role of the NHC ligand in the formation/nucleation of NiNPs. The catalytic activity of NiNPs/MXene was then evaluated in the hydrogenation of quinoline (1D) under standard conditions. Notably, the material exhibited negligible catalytic activity (Table 1, entry 5). This result underscores the critical role of the NHC ligand in maintaining the activity and stability of the NiNPs.
We then explored the catalytic performance of NiNPs supported on MXene using an alternative NHC ligand with different structural properties (Me2NHC). We followed the same preparation method as for NiNPs/NHC@MXene. We synthesized complex 2 using the ligand Me2NHC and reduced with tBuNH2·BH3 to generate the new hybrid material, NiNPs/Me2NHC@MXene. Experimental details of synthetic procedures and characterization data are provided in the SI (section S9, Fig. S17–S21). HRTEM analysis revealed spherical NiNPs with an average diameter of 4.7 ± 2.4 nm. These NiNPs are relatively larger and more broadly distributed than those in NiNPs/NHC@MXene (3.0 ± 0.8 nm). Catalytic evaluation of complex 2 under homogeneous conditions showed lower activity than complex 1 (Table 1, entry 6), whereas the hybrid material NiNPs/Me2NHC@MXene exhibited catalytic performance comparable to that of NiNPs/NHC@MXene (Table 1, entry 7). These findings indicate that, although the NHC ligand plays a key role in nanoparticle formation, the subtle structural variations in the ligand have only a modest influence on the overall catalytic activity.
Hydrogenation of quinoline (1D) using the NiNPs containing the NHC ligand, NiNPs/NHC@MXene, under the standard conditions resulted in a 95% yield of tetrahydroquinoline (1H) (Table 1, entry 8). Temperature screening revealed that diminishing the reaction temperature from 150 °C to 120 °C maintained product formation within the quantitative range (Table 1, entry 9), while further decrease to 100 °C or below significantly reduced the yield (Table 1, entries 10–12). Different hydrogen pressures were tested to identify the most efficient one; however decreasing hydrogen pressure below 30 bar provided lower yields (Table 1, entries 12–14). A range of solvents were scrutinized to determine their influence on the hydrogenation of quinoline (1D) (Table 1, entries 15–22). Among them, alcohols provided the highest yields using either methanol or isopropanol as an alternative to ethanol. In contrast, other solvents gave poor results, indicating that solvent polarity plays a crucial role, likely due to enhanced charge separation in the transition state. The hydrogenation reactions were performed using a catalyst loading of 3.7 mol% nickel, calculated based on the dispersion factor that accounts exclusively for nickel atoms located on the surface of the nanoparticles. The number of surface-exposed nickel atoms was estimated under the assumption of a spherical nanoparticle morphology. Details of the calculation procedure are provided in the SI (Table S2).
Based on the systematic optimization studies, the optimal reaction conditions were identified as 120 °C, 30 bar hydrogen pressure and ethanol as solvent, resulting in a 98% yield using a catalyst loading of 3.7 mol% of nickel. With the optimal conditions, we screened the overall reaction trend by monitoring product formation and reagent disappearance by GC/FID until completion (Fig. 7). The reaction progress was monitored over time, showing a steady increase in the concentration of tetrahydroquinoline (1H) concurrent with a steady decrease in the concentration of quinoline (1D). The mass balance indicates the complete conversion of the reagent into the product without detectable formation of side products. These results confirm the high activity and selectivity of the NiNPs/NHC@MXene catalyst in the hydrogenation of quinoline (1D).
 | ||
| Fig. 7 Reaction monitoring in the hydrogenation of quinoline (1D). Conditions: quinoline (0.1 mmol), catalyst loading (3.7 mol% based on Nisurf), 130 °C, 30 bar H2 and EtOH (2 mL). | ||
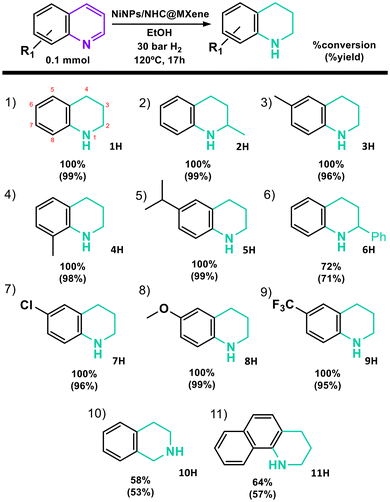
To evaluate the generality and limitations of the hydrogenation of N-heterocycles catalyzed by NiNPs/NHC@MXene, the substrate scope under the optimized conditions previously established was investigated. A range of substrates bearing various functional groups at different positions on the heterocyclic ring were tested. The reaction progress was independently assessed by monitoring substrate conversion and product yield. Conversion was determined by GC/FID using anisole as an internal standard, while the product yield was quantified by 1H NMR spectroscopy using 1,3,5-trimethoxybenzene as an external standard (Table 2).
The catalytic material NiNPs/NHC@MXene is active across a series of N-heterocycles, providing good to excellent yields regardless of the electronic nature or position of substituents (Table S3). Quinoline (1D) and its methyl-substituted derivatives at the 2- (2D), 6- (3D), or 8- (4D) positions were hydrogenated quantitatively (Table 2, entries 1–4). A bulky substituent at the 6-position did not significantly impact the yield (5D); however, a phenyl group at the 2-position (6D) resulted in reduced catalytic efficiency, yielding a lower conversion (Table 2, entries 5 and 6). The presence of electron-donating (MeO–) (8D) and electron-withdrawing (Cl– or CF3−) (7D and 9D) groups at the six position of the aromatic ring do not affect the reaction outcomes affording quantitative yields in all cases (Table 2, entries 7–9). Notably, more sterically or electronically complex substrates such as isoquinoline (10D) and benzo[h]quinoline (11D) also underwent successful hydrogenation, albeit with lower yields (53% and 57%, respectively) under the standard reaction conditions, likely due to slower reaction kinetics (Table 2, entries 10 and 11). Throughout the substrate scope study, no side products were detected. Only starting materials and the fully hydrogenated N-heterocycles were observed, as confirmed by 1H NMR analysis of the crude reaction mixtures (Fig. S22–S32), highlighting the high selectivity of the catalytic system. These results demonstrate that NiNPs functionalized with N-heterocyclic carbene ligands and supported on MXene represent an efficient and selective catalytic platform for the hydrogenation of N-heterocycles.
Recycling and reusability experiments
The reusability of NiNPs/NHC@MXene was evaluated through recycling experiments, in which the catalyst performance for quinoline (1D) hydrogenation was evaluated under identical reaction conditions across successive runs (Fig. 8). After each cycle, the solid catalyst was recovered by filtration, washed with EtOH (3 × 5 mL), air-dried, and reused in a new reaction with fresh reagents and solvent. The results showed that the catalytic performance of NiNPs/NHC@MXene rapidly decreased, going from 98% yield in the first run to a 21% yield in the fourth run. Catalyst deactivation is a major challenge in catalysis, often limiting efficiency and increasing operational costs. To investigate the deactivation mechanism and explore pathways for catalyst improvement, the spent catalyst was analyzed using XRD analysis and microscopic techniques to identify changes in the support and the active metal species. First, the morphology of the spent catalyst was analyzed by microscopy. HRTEM revealed a notable morphological transformation: while the freshly prepared NiNPs/NHC@MXene contained NiNPs averaging 3 nm in size (Fig. 2d), after four catalytic runs, the average size increased dramatically up to 80 nm (Fig. 9). Morphological analysis suggests that nanoparticle sintering is a major contributor to catalyst deactivation. | ||
| Fig. 8 Recycling and reusability properties of NiNPs/NHC@MXene. The yellow arrow indicates catalyst regeneration. | ||
 | ||
| Fig. 9 HRTEM characterization of NiNPs/NHC@MXene after 4 catalytic cycles at 1 μm (a) and 500 nm (b). | ||
The surface chemical composition and oxidation states of the NiNPs/NHC@MXene catalyst were analyzed by XPS before and after consecutive catalytic cycles (Fig. 10 and S33, S34). The as-prepared sample exhibits characteristic Ti 2p, Ni 2p, and C 1s signals consistent with Ti3C2 MXene decorated with Ni-based nanoparticles as previously described. In the Ti 2p region, the peaks at 454.5 eV and 456.0 eV correspond to Ti–C and Ti2+ species, respectively, whereas signals at 457.6–462.3 eV are attributed to surface Ti–Ox species (TiO2 and other oxides). These features remain nearly unchanged after the first catalytic run, indicating a certain stability of the MXene support under the reaction conditions. However, after the fourth run, there is a clear increase in the intensity of the peak attributed to surface Ti–Ox species, suggesting progressive surface oxidation of the MXene.
 | ||
| Fig. 10 Comparative XPS characterization of the NiNPs/NHC@MXene initial material and during the recycling experiments. High resolution core-level peaks corresponding to Ti 2p, Ni 2p and C 1s. | ||
The Ni 2p spectra of the as-prepared material show a main component as a doublet peak at 856.4 eV (Ni 2p3/2) and 874.3 eV (Ni 2p1/2) corresponding to NiO, along with their characteristic satellite features. The intensity of this NiO component gradually decreases in favor of the Ni(OH)2 species, and this transformation is evident after the first catalytic run. Following four catalytic cycles, the Ni 2p region is dominated by the doublet peak of Ni(OH)2 at 859.3 and 877.9 eV, evidencing the formation of hydroxylated nickel species. The C 1s spectra display a dominant C–C peak at 284.5 eV, with additional components assigned to C–O (286.1 eV), C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O (287.9 eV), and Ti–C (281.1 eV). The persistence of the Ti–C signal confirms the retention of the MXene carbide structure after the four catalytic runs. A clear increase in oxygenated carbon species is observed after successive runs as evidenced by the intensified C–O signal at 286.2 eV, which is attributed to surface oxidation. In the N 1s region, the peak at 401.2 eV, corresponding to carbene-type nitrogen from the NHC ligand, is clearly observed after the first catalytic run (Fig. S33). However, this signal is no longer visible after four catalytic cycles, suggesting that the NHC ligand detaches from the NiNP surface during repeated reactions (Fig. S34). These results indicate that the NHC ligand remains partially present after the first catalytic cycle but is completely lost after four cycles.
O (287.9 eV), and Ti–C (281.1 eV). The persistence of the Ti–C signal confirms the retention of the MXene carbide structure after the four catalytic runs. A clear increase in oxygenated carbon species is observed after successive runs as evidenced by the intensified C–O signal at 286.2 eV, which is attributed to surface oxidation. In the N 1s region, the peak at 401.2 eV, corresponding to carbene-type nitrogen from the NHC ligand, is clearly observed after the first catalytic run (Fig. S33). However, this signal is no longer visible after four catalytic cycles, suggesting that the NHC ligand detaches from the NiNP surface during repeated reactions (Fig. S34). These results indicate that the NHC ligand remains partially present after the first catalytic cycle but is completely lost after four cycles.
Overall, the XPS analysis during reusability experiments confirmed a higher oxidation degree of the NiNPs/NHC@MXene catalyst. Although catalytic reactions occur under reducing conditions, the observed oxidation likely results from the formation of highly reactive Ni surface sites during hydrogenation that subsequently oxidize upon exposure to air after each reaction cycle.
To further elucidate the redox behavior of NiNPs on MXene we performed H2 temperature programmed reduction (H2-TPR) analysis of the support Ti3C2 MXene, the as-prepared NiNPs/NHC@MXene catalyst and the corresponding used catalyst (Fig. 11). The H2-TPR profile of Ti3C2 MXene exhibits only weak hydrogen consumption below 400 °C and a broad high-temperature reduction feature above 700 °C, which can be attributed to stable Ti–O species, confirming the absence of readily reducible surface species under typical hydrogenation conditions.75 Upon deposition of nickel nanoparticles, a new and well-defined reduction peak appears at approximately 340 °C, which is assigned to the reduction of oxidized nickel species (NiO/Ni(OH)2). This behaviour indicates the presence of finely dispersed and readily reducible Ni nanoparticles on the MXene surface, in agreement with HRTEM analysis results (Fig. 2c & d). After catalytic hydrogenation, the TPR profile retains a nickel-related reduction feature at a similar temperature, although slightly shifted and narrowed, suggesting changes in nanoparticle dispersion consistent with partial sintering during catalysis. This interpretation is further supported by HRTEM analysis of the used catalyst (Fig. 9). Importantly, the high-temperature reduction feature associated with the MXene support remains essentially unchanged, demonstrating that the Ti3C2 framework is structurally stable under the applied reaction conditions. Given that the hydrogenation reactions are performed under significantly more reducing conditions (30 bar H2 at 120 °C for several hours) than those employed during H2-TPR measurements, it is reasonable to infer that NiO/Ni(OH)2 species are at least partially, and likely predominantly, reduced to metallic Ni during catalysis. This interpretation is consistent with the observed catalytic activity and with literature reports identifying Ni0 as the active phase in nickel-catalyzed hydrogenation reactions.76–78
Overall, the H2-TPR results indicate that catalyst deactivation is primarily related to nanoparticle agglomeration rather than irreversible oxidation of either the nickel species or the MXene support.
To address the observed deactivation, a regeneration strategy was explored involving a reductive treatment aimed at restoring nanoparticle dispersion. The spent catalyst was treated with tBuNH2·BH3 under stirring for 20 hours, following the same procedure used for the as-prepared catalyst. This treatment successfully recovered the catalytic performance to its initial level (Fig. 8, run 5). Further evidence of the successful regeneration process was experimentally obtained by XRD (Fig. 12). The characteristic diffraction peaks of NiNPs/NHC@MXene gradually broadened and eventually disappeared after four catalytic runs, indicating a loss of crystallinity and the formation of an amorphous phase. However, after the regeneration of the catalytic material, the XRD pattern was restored, displaying sharper and more defined reflections consistent with a material like the as-prepared catalyst. Further characterization of the regenerated material using HRTEM microscopy confirmed the disaggregation of NiNPs. The average particle size decreased from approximately 80 nm in the spent material to about 5 nm after regeneration (Fig. 13).
 | ||
| Fig. 12 XRD evolution of NiNPs/NHC@MXene during recycling and reusability experiments. XRD spectrum after run 1 (a), after run 4 (b) and after reactivation (c). | ||
 | ||
| Fig. 13 HRTEM characterization of NiNPs/NHC@MXene after the regeneration procedure at 100 nm (a) and 20 nm (b). | ||
The recycling and regeneration studies reveal that NiNPs/NHC@MXene suffers from deactivation due to nanoparticle sintering. Importantly, this deactivation can be reversed through a mild, room-temperature reduction treatment effectively restoring both the nanoparticle dispersion and the catalytic activity. This approach highlights the potential of NiNPs/NHC@MXene as a sustainable and reusable catalytic system whose performance is comparable to those of state-of-the-art nickel-based catalysts (Table S1).49–56,79
Conclusions
This study presents the development of a hybrid catalytic material consisting of nickel nanoparticles functionalized with NHC ligands and supported on Ti3C2 MXene (NiNPs/NHC@MXene). This material exhibits high catalytic efficiency and selectivity in the hydrogenation of quinolines, performing comparably to state-of-the-art nickel-based systems (Table S1). The role of the NHC ligand has been scrutinized, indicating the impact in the initial formation of metal nanoparticles on the material's surface by controlling the size and morphology.A distinctive feature of the NiNPs/NHC@MXene catalyst is its broad substrate scope. Recycling experiments revealed a gradual decrease in catalytic performance over successive cycles, primarily attributed to nanoparticle growth and agglomeration due to ligand disappearance. Notably, this deactivation can be reversed: a mild regeneration treatment under reducing conditions using tBuNH2·BH3 successfully restores the catalytic activity to its original level.
The hydrogenation activity and regenerability of the NiNPs/NHC@MXene system exemplify a sustainable strategy for designing efficient catalysts based on Earth-abundant metals. Moreover, these results underscore the versatility of Ti3C2 MXene as a suitable support for metal nanoparticles. Future efforts directed toward minimizing deactivation pathways and optimizing regeneration strategies will be crucial for extending catalyst lifetime and advancing the practical deployment of MXene-supported catalytic systems.
Author contributions
Judith Medina-Vargas, Santiago Martín and Iván Sorribes contributed to investigation, methodology, validation and data curation of results as well as writing – review and editing. Hermenegildo García and Jose A. Mata contributed to conceptualization, formal analysis, resources, writing original draft, writing – review and editing and funding acquisition. All authors have given approval to the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: additional characterization of materials by XPS, SEM and HRTEM. Further details of experimental procedures, NMR characterization of organic products and quantification analysis. See DOI: https://doi.org/10.1039/d5dt02759d.Acknowledgements
Thanks to the Spanish Ministry of Science and Innovation for financial support through the projects PID2021-126071OB-C21, PID2021-126071OB-C22 and CEX2023-001286-S, funded by MICIN/AEI/10.13039/501100011033/FEDER “Una manera de hacer Europa”. Generalitat Valenciana (MFA/2022/043) with funding from European Union NextGenerationEU and Universitat Jaume I (UJI-B2022-23 and GACUJIMB/2023/13) is also gratefully acknowledged. I. S. thanks the Grant CNS2022-136183 funded by MICIU/AEI/10.13039/501100011033 and by “European Union NextGenerationEU/PRTR”, and the Grant PID2022-143164OA-I00 funded by MICIU/AEI/10.13039/501100011033 and by “ERDF/EU”. I.S. also gratefully acknowledges the program ESGENT from Generalitat valenciana (CIESGT/2024/16). J. M.-V. thanks the program “Santiago Grisolia” (GrisoliaP2021/114). The authors thank ‘Servei Central d'Instrumentació Científica (SCIC) de la Universitat Jaume I′. S. M. is grateful to Gobierno de Aragón for the grant E31_23R with European Social Funds (Construyendo Europa desde Aragón).References
- D. Astruc, Nanoparticles and Catalysis, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, Germany, 2007 Search PubMed.
- N.-D. Jaji, H. L. Lee, M. H. Hussin, H. M. Akil, M. R. Zakaria and M. B. H. Othman, Advanced nickel nanoparticles technology: From synthesis to applications, Nanotechnol. Rev., 2020, 9, 1456–1480 CrossRef CAS.
- F. e Silva, V. Salim and T. Rodrigues, Controlled Nickel Nanoparticles: A Review on How Parameters of Synthesis Can Modulate Their Features and Properties, AppliedChem, 2024, 4, 86–106 CrossRef.
- M. R. Ahghari, V. Soltaninejad and A. Maleki, Synthesis of nickel nanoparticles by a green and convenient method as a magnetic mirror with antibacterial activities, Sci. Rep., 2020, 10, 12627 CrossRef CAS PubMed.
- Z. Ji, X. Shen, G. Zhu, H. Zhou and A. Yuan, Reduced graphene oxide/nickel nanocomposites: facile synthesis, magnetic and catalytic properties, J. Mater. Chem., 2012, 22, 3471–3477 RSC.
- M. G. Avello, S. Golling, L. Truong-Phuoc, L. Vidal, T. Romero, V. Papaefthimiou, N. Gruber, M. J. Chetcuti, F. R. Leroux, M. Donnard, V. Ritleng, C. Pham-Huu and C. Michon, (NHC-olefin)-nickel(0) nanoparticles as catalysts for the (Z)-selective semi-hydrogenation of alkynes and ynamides, Chem. Commun., 2023, 59, 1537–1540 RSC.
- P. Ryabchuk, A. Agapova, C. Kreyenschulte, H. Lund, H. Junge, K. Junge and M. Beller, Heterogeneous nickel-catalysed reversible, acceptorless dehydrogenation of N-heterocycles for hydrogen storage, Chem. Commun., 2019, 55, 4969–4972 RSC.
- M. D. de los Bernardos, S. Pérez-Rodríguez, A. Gual, C. Claver and C. Godard, Facile synthesis of NHC-stabilized Ni nanoparticles and their catalytic application in the Z-selective hydrogenation of alkynes, Chem. Commun., 2017, 53, 7894–7897 RSC.
- J.-F. Soulé, H. Miyamura and S. Kobayashi, Copolymer-Incarcerated Nickel Nanoparticles with N -Heterocyclic Carbene Precursors as Active Cross-Linking Agents for Corriu–Kumada–Tamao Reaction, J. Am. Chem. Soc., 2013, 135, 10602–10605 CrossRef.
- F. Alonso, P. Riente and M. Yus, Alcohols for the α–Alkylation of Methyl Ketones and Indirect Aza–Wittig Reaction Promoted by Nickel Nanoparticles, Eur. J. Org. Chem., 2008, 2008, 4908–4914 CrossRef.
- A. Charvieux, J. B. Giorgi, N. Duguet and E. Métay, Solvent-free direct α-alkylation of ketones by alcohols catalyzed by nickel supported on silica–alumina, Green Chem., 2018, 20, 4210–4216 RSC.
- D. Bouzouita, J. M. Asensio, V. Pfeifer, A. Palazzolo, P. Lecante, G. Pieters, S. Feuillastre, S. Tricard and B. Chaudret, Chemoselective H/D exchange catalyzed by nickel nanoparticles stabilized by N-heterocyclic carbene ligands, Nanoscale, 2020, 12, 15736–15742 RSC.
- G. Wu, C. Zhang, S. Li, Z. Han, T. Wang, X. Ma and J. Gong, Hydrogen Production via Glycerol Steam Reforming over Ni/Al2O3: Influence of Nickel Precursors, ACS Sustainable Chem. Eng., 2013, 1, 1052–1062 CrossRef CAS.
- S. Chen, J. Duan, J. Ran, M. Jaroniec and S. Z. Qiao, N-doped graphene film-confined nickel nanoparticles as a highly efficient three-dimensional oxygen evolution electrocatalyst, Energy Environ. Sci., 2013, 6, 3693–3699 RSC.
- L. Gu, C. Zhang, Y. Guo, J. Gao, Y. Yu and B. Zhang, Enhancing Electrocatalytic Water Splitting Activities via Photothermal Effect over Bifunctional Nickel/Reduced Graphene Oxide Nanosheets, ACS Sustainable Chem. Eng., 2019, 7, 3710–3714 CrossRef CAS.
- D. D. Kruger, J. J. Delgado, F. J. Recio, S. Goberna-Ferrón, A. Primo and H. García, Influence of surface terminal groups on the efficiency of two-electron oxygen reduction reaction catalyzed by iron single atoms on Ti3C2Tx (T = Cl, Br, NH) MXene, J. Mater. Chem. A, 2024, 12, 25291–25303 RSC.
- S. Ghosh, S. Gupta, M. Gregoire, T. Ourlin, P.-F. Fazzini, E. Abi-Aad, C. Poupin and B. Chaudret, Catalytic Sabatier Process under Thermally and Magnetically Induced Heating: A Comparative Case Study for Titania-Supported Nickel Catalyst, Nanomaterials, 2023, 13, 1474 CrossRef CAS PubMed.
- S. Navalon, A. Dhakshinamoorthy, M. Alvaro and H. Garcia, Metal nanoparticles supported on two-dimensional graphenes as heterogeneous catalysts, Coord. Chem. Rev., 2016, 312, 99–148 CrossRef CAS.
- D. Skoda, J. Vilcakova, R. S. Yadav, B. Hanulikova, T. Capkova, M. Jurca, M. Urbanek, P. Machac, L. Simonikova, J. Antos and I. Kuritka, Nickel nanoparticle–decorated reduced graphene oxide via one-step microwave-assisted synthesis and its lightweight and flexible composite with Polystyrene-block-poly(ethylene-ran-butylene)-block-polystyrene polymer for electromagnetic wave shielding application, Adv. Compos. Hybrid Mater., 2023, 6, 113 CrossRef CAS.
- P. Lara, O. Rivada-Wheelaghan, S. Conejero, R. Poteau, K. Philippot and B. Chaudret, Ruthenium Nanoparticles Stabilized by N-Heterocyclic Carbenes: Ligand Location and Influence on Reactivity, Angew. Chem., Int. Ed., 2011, 50, 12080–12084 CrossRef CAS PubMed.
- C. Pan, K. Pelzer, K. Philippot, B. Chaudret, F. Dassenoy, P. Lecante and M.-J. Casanove, Ligand-Stabilized Ruthenium Nanoparticles: Synthesis, Organization, and Dynamics, J. Am. Chem. Soc., 2001, 123, 7584–7593 CrossRef CAS PubMed.
- A. García-Zaragoza, C. Cerezo-Navarrete, A. Mollar-Cuni, P. Oña-Burgos, J. A. Mata, A. Corma and L. M. Martínez-Prieto, Tailoring graphene-supported Ru nanoparticles by functionalization with pyrene-tagged N-heterocyclic carbenes, Catal. Sci. Technol., 2022, 12, 1257–1270 RSC.
- C. Copéret, A. Comas-Vives, M. P. Conley, D. P. Estes, A. Fedorov, V. Mougel, H. Nagae, F. Núñez-Zarur and P. A. Zhizhko, Surface Organometallic and Coordination Chemistry toward Single-Site Heterogeneous Catalysts: Strategies, Methods, Structures, and Activities, Chem. Rev., 2016, 116, 323–421 CrossRef PubMed.
- L. Shen, J. Xu, M. Zhu and Y.-F. Han, Essential Role of the Support for Nickel-Based CO 2 Methanation Catalysts, ACS Catal., 2020, 10, 14581–14591 CrossRef CAS.
- J. Wang, M. Zhang, T. Miao, Y. Ling, Q. Wen, J. Zheng, J. Xu, T. Hayat and N. S. Alharbi, A facile self-template and carbonization strategy to fabricate nickel nanoparticle supporting N-doped carbon microtubes, Inorg. Chem. Front., 2018, 5, 844–852 RSC.
- R. J. White, R. Luque, V. L. Budarin, J. H. Clark and D. J. Macquarrie, Supported metal nanoparticles on porous materials. Methods and applications, Chem. Soc. Rev., 2009, 38, 481–494 RSC.
- C. Copéret, Single-Sites and Nanoparticles at Tailored Interfaces Prepared via Surface Organometallic Chemistry from Thermolytic Molecular Precursors, Acc. Chem. Res., 2019, 52, 1697–1708 CrossRef PubMed.
- M. Babucci, A. Guntida and B. C. Gates, Atomically Dispersed Metals on Well-Defined Supports including Zeolites and Metal–Organic Frameworks: Structure, Bonding, Reactivity, and Catalysis, Chem. Rev., 2020, 120, 11956–11985 CrossRef CAS PubMed.
- M. Naguib, M. W. Barsoum and Y. Gogotsi, Ten Years of Progress in the Synthesis and Development of MXenes, Adv. Mater., 2021, 33, 202103393 Search PubMed.
- N. Kitchamsetti and A. L. F. de Barros, Recent Advances in MXenes Based Composites as Photocatalysts: Synthesis, Properties and Photocatalytic Removal of Organic Contaminants from Wastewater, ChemCatChem, 2023, 15, e202300690 CrossRef CAS.
- V. Kamysbayev, A. S. Filatov, H. Hu, X. Rui, F. Lagunas, D. Wang, R. F. Klie and D. V. Talapin, Covalent surface modifications and superconductivity of two-dimensional metal carbide MXenes, Science, 2020, 369, 979–983 CrossRef CAS PubMed.
- B. Anasori, M. R. Lukatskaya and Y. Gogotsi, 2D metal carbides and nitrides (MXenes) for energy storage, Nat. Rev. Mater., 2017, 2, 16098 CrossRef CAS.
- A. Ebadi Amooghin, A. Arabi Shamsabadi, M. M. Moftakhari Sharifzadeh, A. Salehi, H. Sanaeepur, M. Dadashi Firouzjaei, V. Rad, H. Jafarian, M. A. Elliott and H. Garcia, Tailoring MXene composition and structure for High-Performance mixed matrix membranes in CO2 separation applications, Sep. Purif. Technol., 2025, 371, 133360 CrossRef CAS.
- Z. Li, L. Yu, C. Milligan, T. Ma, L. Zhou, Y. Cui, Z. Qi, N. Libretto, B. Xu, J. Luo, E. Shi, Z. Wu, H. Xin, W. N. Delgass, J. T. Miller and Y. Wu, Two-dimensional transition metal carbides as supports for tuning the chemistry of catalytic nanoparticles, Nat. Commun., 2018, 9, 5258 CrossRef CAS PubMed.
- Z. Li and Y. Wu, 2D Early Transition Metal Carbides (MXenes) for Catalysis, Small, 2019, 15, 1804736 CrossRef PubMed.
- T. K. Slot, V. Natu, E. V. Ramos-Fernandez, A. Sepúlveda-Escribano, M. Barsoum, G. Rothenberg and N. R. Shiju, Enhancing catalytic epoxide ring-opening selectivity using surface-modified Ti 3 C 2 T x MXenes, 2D Mater., 2021, 8, 035003 CrossRef CAS.
- J. D. Gouveia, Á. Morales-García, F. Viñes, F. Illas and J. R. B. Gomes, MXenes as promising catalysts for water dissociation, Appl. Catal., B, 2020, 260, 118191 CrossRef CAS.
- N. Li, X. Chen, W.-J. Ong, D. R. MacFarlane, X. Zhao, A. K. Cheetham and C. Sun, Understanding of Electrochemical Mechanisms for CO 2 Capture and Conversion into Hydrocarbon Fuels in Transition-Metal Carbides (MXenes), ACS Nano, 2017, 11, 10825–10833 CrossRef CAS PubMed.
- X. Zhang, C. Wu, Z. Shi, N. Wang and Y. Niu, Ni nanoparticles/NiFe LDHs/MXene ternary composites for catalytic hydrogenation of p-Nitrophenol, Chem. Phys. Lett., 2025, 866, 141931 CrossRef CAS.
- M. Bhosale, S. Thangarasu, N. Murugan, Y. A. Kim and T. Oh, Ti–MXene/α–Ni(OH) 2 Nanostructures as High–Performance Electrocatalyst for Oxygen Evolution Reaction, ChemSusChem, 2025, 18, e202402603 CrossRef CAS PubMed.
- M. Smirnova, B. Scheibe, R. Ramírez-Grau, H. García and A. Lewandowska-Andralojc, Synergistic effects of MXene support and cobalt salts in dye-sensitized photocatalytic hydrogen generation, Int. J. Hydrogen Energy, 2024, 88, 1098–1107 CrossRef CAS.
- A. Dhakshinamoorthy, R. Ramírez-Grau, H. Garcia and A. Primo, Opportunities of MXenes in Heterogeneous Catalysis: V2C as Aerobic Oxidation Catalyst, Chem. – Eur. J., 2024, 30, e202400576 CrossRef CAS PubMed.
- M. Puche, L. Liu, P. Concepción, I. Sorribes and A. Corma, Tuning the Catalytic Performance of Cobalt Nanoparticles by Tungsten Doping for Efficient and Selective Hydrogenation of Quinolines under Mild Conditions, ACS Catal., 2021, 11, 8197–8210 CrossRef CAS PubMed.
- I. Sorribes, L. Liu, A. Doménech-Carbó and A. Corma, Nanolayered Cobalt–Molybdenum Sulfides as Highly Chemo- and Regioselective Catalysts for the Hydrogenation of Quinoline Derivatives, ACS Catal., 2018, 8, 4545–4557 CrossRef CAS.
- A. Mollar-Cuni, S. Martín, G. Guisado-Barrios and J. A. Mata, Dual role of graphene as support of ligand-stabilized palladium nanoparticles and carbocatalyst for (de)hydrogenation of N-heterocycles, Carbon, 2023, 206, 314–324 CrossRef CAS.
- D. Teichmann, W. Arlt and P. Wasserscheid, Liquid Organic Hydrogen Carriers as an efficient vector for the transport and storage of renewable energy, Int. J. Hydrogen Energy, 2012, 37, 18118–18132 CrossRef CAS.
- D. Teichmann, W. Arlt, P. Wasserscheid and R. Freymann, A future energy supply based on Liquid Organic Hydrogen Carriers (LOHC), Energy Environ. Sci., 2011, 4, 2767–2773 RSC.
- A. F. Dalebrook, W. Gan, M. Grasemann, S. Moret and G. Laurenczy, Hydrogen storage: beyond conventional methods, Chem. Commun., 2013, 49, 8735–8751 RSC.
- V. Vermaak, H. C. M. Vosloo and A. J. Swarts, Fast and Efficient Nickel(II)–catalysed Transfer Hydrogenation of Quinolines with Ammonia Borane, Adv. Synth. Catal., 2020, 362, 5788–5793 CrossRef CAS.
- R. Yun, W. Ma, L. Hong, Y. Hu, F. Zhan, S. Liu and B. Zheng, Ni@PC as a stabilized catalyst toward the efficient hydrogenation of quinoline at ambient temperature, Catal. Sci. Technol., 2019, 9, 6669–6672 RSC.
- L. Niu, Y. An, X. Yang, G. Bian, Q. Wu, Z. Xia and G. Bai, Highly dispersed Ni nanoparticles encapsulated in hollow mesoporous silica spheres as an efficient catalyst for quinoline hydrogenation, Mol. Catal., 2021, 514, 111855 CAS.
- V. Goyal, T. Bhatt, A. Kuttasseri, A. Mahata, R. Zbořil, K. Natte and R. V. Jagadeesh, A biomass-derived nickel-based nanomaterial as a sustainable and reusable catalyst for hydrogenation of arenes and heteroarenes, RSC Sustainability, 2025, 3, 2235–2245 RSC.
- Z. Yuan, X. Li, G. Wang, Z. Zhu, Y. Liao, Z. Zhang and B. Liu, Efficient hydrogenation of N-heteroarenes into N-heterocycles over MOF-derived CeO2 supported nickel nanoparticles, Mol. Catal., 2023, 540, 113052 CAS.
- P. Ryabchuk, G. Agostini, M.-M. Pohl, H. Lund, A. Agapova, H. Junge, K. Junge and M. Beller, Intermetallic nickel silicide nanocatalyst—A non-noble metal–based general hydrogenation catalyst, Sci. Adv., 2018, 4, eaat0761 CrossRef PubMed.
- S. Mao, P. Ryabchuk, S. Dastgir, M. Anwar, K. Junge and M. Beller, Silicon-Enriched Nickel Nanoparticles for Hydrogenation of N-Heterocycles in Aqueous Media, ACS Appl. Nano Mater., 2022, 5, 5625–5630 CrossRef CAS.
- Y. Hu, M. Liu, S. Bartling, H. Lund, H. Atia, P. J. Dyson, M. Beller and R. V. Jagadeesh, A general and robust Ni-based nanocatalyst for selective hydrogenation reactions at low temperature and pressure, Sci. Adv., 2023, 9, eadj8225 CrossRef CAS PubMed.
- S. Sabater, J. A. Mata and E. Peris, Catalyst enhancement and recyclability by immobilization of metal complexes onto graphene surface by noncovalent interactions, ACS Catal., 2014, 4, 2038–2047 CrossRef CAS.
- J. Medina-Vargas, A. Mollar-Cuni, S. Martín, I. Sorribes and J. A. Mata, Graphene as a Suitable Support for Nickel Nanoparticles Functionalized with NHC Ligands in Dehydrogenation of Alcohols, ChemCatChem, 2025, 17, e202500490 CrossRef CAS.
- M. Naguib, M. Kurtoglu, V. Presser, J. Lu, J. Niu, M. Heon, L. Hultman, Y. Gogotsi and M. W. Barsoum, Two–Dimensional Nanocrystals Produced by Exfoliation of Ti3AlC2, Adv. Mater., 2011, 23, 4248–4253 CrossRef CAS PubMed.
- M. Alhabeb, K. Maleski, B. Anasori, P. Lelyukh, L. Clark, S. Sin and Y. Gogotsi, Guidelines for Synthesis and Processing of Two-Dimensional Titanium Carbide (Ti3C2Tx MXene), Chem. Mater., 2017, 29, 7633–7644 CrossRef CAS.
- D. Ventura-Espinosa, A. Carretero-Cerdán, M. Baya, H. García and J. A. Mata, Catalytic Dehydrogenative Coupling of Hydrosilanes with Alcohols for the Production of Hydrogen On-demand: Application of a Silane/Alcohol Pair as a Liquid Organic Hydrogen Carrier, Chem. – Eur. J., 2017, 23, 10815–10821 Search PubMed.
- D. Ventura-Espinosa, S. Sabater, A. Carretero-Cerdán, M. Baya and J. A. Mata, High Production of Hydrogen on Demand from Silanes Catalyzed by Iridium Complexes as a Versatile Hydrogen Storage System, ACS Catal., 2018, 8, 2558–2566 CrossRef CAS.
- A. Mollar-Cuni, P. Borja, S. Martin, G. Guisado-Barrios and J. A. Mata, A Platinum Molecular Complex Immobilised on the Surface of Graphene as Active Catalyst in Alkyne Hydrosilylation, Eur. J. Inorg. Chem., 2020, 2020, 4254–4262 CrossRef CAS.
- A. Mollar-Cuni, D. Ventura-Espinosa, S. Martín, Á. Mayoral, P. Borja and J. A. Mata, Stabilization of Nanoparticles Produced by Hydrogenation of Palladium-N-Heterocyclic Carbene Complexes on the Surface of Graphene and Implications in Catalysis, ACS Omega, 2018, 3, 15217–15228 CrossRef CAS PubMed.
- D. Ventura-Espinosa, S. Martín, H. García and J. A. Mata, Ligand effects in the stabilization of gold nanoparticles anchored on the surface of graphene: Implications in catalysis, J. Catal., 2021, 394, 113–120 CrossRef CAS.
- D. Ventura-Espinosa, S. Sabater and J. A. Mata, Enhancement of gold catalytic activity and stability by immobilization on the surface of graphene, J. Catal., 2017, 352, 498–504 CrossRef CAS.
- S. M. M. Raj, A. K. Sundramoorthy, R. Atchudan, D. Ganapathy and A. Khosla, Review—Recent Trends on the Synthesis and Different Characterization Tools for MXenes and their Emerging Applications, J. Electrochem. Soc., 2022, 169, 077501 CrossRef CAS.
- Y. Cao, Q. Deng, Z. Liu, D. Shen, T. Wang, Q. Huang, S. Du, N. Jiang, C.-T. Lin and J. Yu, Enhanced thermal properties of poly(vinylidene fluoride) composites with ultrathin nanosheets of MXene, RSC Adv., 2017, 7, 20494–20501 RSC.
- L.-Å. Näslund, P. O. Å. Persson and J. Rosen, X-ray Photoelectron Spectroscopy of Ti3AlC2, Ti3C2Tz, and TiC Provides Evidence for the Electrostatic Interaction between Laminated Layers in MAX-Phase Materials, J. Phys. Chem. C, 2020, 124, 27732–27742 CrossRef.
- M. Mičušík, M. Šlouf, A. Stepura, Y. Soyka, E. Ovodok, M. Procházka and M. Omastová, Aging of 2D MXene nanoparticles in air: An XPS and TEM study, Appl. Surf. Sci., 2023, 610, 155351 CrossRef.
- R. Ramírez-Grau, P. Garcia-Aznar, G. Sastre, S. Goberna-Ferrón, O. Pavel, A. Tirsoaga, B. Cojocaru, D. G. Popescu, V. I. Parvulescu, A. Primo and H. García, MXenes as Heterogeneous Thermal Catalysts: Regioselective Anti-Markovnikov Hydroamination of Terminal Alkynes with 102 h−1 Turnover Frequencies, J. Am. Chem. Soc., 2025, 147, 3315–3332 CrossRef PubMed.
- B. B. Nayak, S. Vitta, A. K. Nigam and D. Bahadur, Ni and Ni–nickel oxide nanoparticles with different shapes and a core–shell structure, Thin Solid Films, 2006, 505, 109–112 CrossRef CAS.
- P. Song, D. Wen, Z. X. Guo and T. Korakianitis, Oxidation investigation of nickel nanoparticles, Phys. Chem. Chem. Phys., 2008, 10, 5057–5065 RSC.
- V. Vonk, N. Khorshidi and A. Stierle, Structure and Oxidation Behavior of Nickel Nanoparticles Supported by YSZ(111), J. Phys. Chem. C, 2017, 121, 2798–2806 CrossRef CAS PubMed.
- J. Wang, Y. Wang, Y. Wang, X. Li, C. Wu, T. Zhang, B. Tian, L. Qian, Y. Guo, L. Wu, L. Zheng, Y. Li and X. Ding, Novel two-dimensional Ti3C2Tx MXene-supported Ni-M (M = Pd, Pt, Ru): A remarkable catalyst for efficient HDO of guaiacol under hydrothermal conditions: Performance, mechanism and kinetic studies, Fuel, 2024, 374, 132493 CrossRef CAS.
- S. Qiu, Q. Zhang, W. Lv, T. Wang, Q. Zhang and L. Ma, Simply packaging Ni nanoparticles inside SBA-15 channels by co-impregnation for dry reforming of methane, RSC Adv., 2017, 7, 24551–24560 RSC.
- J. Gao, Y. Cao, G. Luo, J. Fan, J. H. Clark and S. Zhang, High-efficiency catalytic hydrodeoxygenation of lignin-derived vanillin with nickel-supported metal phosphate catalysts, Chem. Eng. J., 2022, 448, 137723 CrossRef CAS.
- J. Zhou, Y. Zhang, S. Li and J. Chen, Ni/NiO Nanocomposites with Rich Oxygen Vacancies as High-Performance Catalysts for Nitrophenol Hydrogenation, Catalysts, 2019, 9, 944 CrossRef CAS.
- D. Ren, L. He, L. Yu, R.-S. Ding, Y.-M. Liu, Y. Cao, H.-Y. He and K.-N. Fan, An Unusual Chemoselective Hydrogenation of Quinoline Compounds Using Supported Gold Catalysts, J. Am. Chem. Soc., 2012, 134, 17592–17598 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |