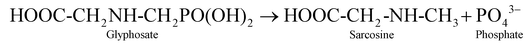DOI:
10.1039/D5DT02596F
(Paper)
Dalton Trans., 2026,
55, 2200-2223
Environmental applications of an ultrasonically synthesized and PEG assisted Yb2O3/ZnFe2O4 nanocomposite for dye and herbicide degradation and electrochemical sensing of catechol
Received
29th October 2025
, Accepted 30th December 2025
First published on 19th January 2026
Abstract
This study effectively generated zinc ferrite (ZnFe2O4) and ytterbium oxide (Yb2O3) nanoparticles (NPs) using the co-precipitation method. Using the synthesized NPs, a binary nanocomposite Yb2O3/ZnFe2O4@PEG supported by polyethene glycol (PEG) was formed via ultrasonication. The morphological, structural, functional, and optical features of the produced NPs and the binary nanocomposite were examined using scanning electron microscopy (SEM), X-ray diffraction (XRD), Fourier-transform infrared spectroscopy (FT-IR), and UV-visible spectroscopy. The photocatalytic activity of the NPs and the nanocomposite against the pesticide glyphosate and the Brilliant Blue FCF dye was investigated under the influence of visible light. In contrast to the individual NPs, the Yb2O3/ZnFe2O4@PEG nanocomposite had noticeably better photocatalytic activity. It achieved 86% and 91% degradation of the dye and pesticide, respectively, under 90 minutes, which may be explained on the basis of enhanced charge separation, synergistic interactions, and increased surface activity. Moreover, the Yb2O3/ZnFe2O4@PEG nanocomposite was tested for catechol sensing through the electrochemical method. The cyclic voltammetry analysis showed that the bare GCE produced a negligible current response, whereas the nanocomposite-modified electrode displayed pronounced redox peaks in the presence of catechol, confirming its strong electrocatalytic activity. The sensor exhibited a linear response for catechol in the concentration range of 40–100 µM, with a sensitivity of 0.367 µA µM−1 cm−2 and a limit of detection of 11.52 µM, confirming its suitability for sensitive electrochemical monitoring. The resulting binary nanocomposite can be used as a photocatalyst to effectively break down hazardous dyes and pesticides, hence enabling environmental cleanup applications.
1. Introduction
Nanotechnology has been an esteemed research area since the last century. Particulate materials with at least one dimension less than 100 nm are referred to as nanoparticles.1 A nanocomposite is a type of material made up of many phases, at least one of which has dimensions of one, two, or three nanometres. The objective of synergy between various components is accomplished by nanoscale phase processes. NPs, nanofibers, nanoclays, and any other moiety that may be organic, inorganic, or biological are examples of nanomaterials (NMs) that may be present as components of nanocomposites.2 By combining metallic, polymeric, and non-metallic materials using various methods, the nanocomposite materials offer the added benefit of preserving key characteristics to get around flaws and express some new ones. Such a material is an example of a multiphase crossover between reinforcing and matrix materials. The reinforcing material is a dispersed phase that is usually made up of fibrous elements like glass fibre, organic fibre, etc., whereas the matrix material is a continuous phase that includes metallic, inorganic, non-metallic, and polymer matrix materials.3 Generally speaking, binary nanocomposites are made up of exactly two separate components, whereas ternary nanocomposites are made up of three different constituents. When compared to single-material systems, binary and ternary nanocomposites are often utilised to leverage synergistic interactions between multiple materials, resulting in enhanced functional properties such as mechanical strength, electrical conductivity, or catalytic activity.4 All kinds of nanocomposites produce a notable improvement in characteristics when compared to their counterparts, according to experimental work. As a result, they can be used in a wide range of systems, including sensors, battery cathodes, nanowires, lightweight composites, and optics.5 Because of their special properties, which include high strength, high stiffness, high durability, low density, high resistance, corrosion resistance, gas barrier, and heat resistance, nanocomposites have several advantages over other materials.6 In order to create nanocomposites with special qualities, researchers worldwide are developing new methods for their synthesis. Due to their numerous practical uses, transition metal oxide-based nanocomposites have garnered a lot of interest as a flexible and advantageous material in practically every scientific discipline. Many transition metal oxide-based nanocomposites have been reported in the literature for their significant practical applications.7 A recent study, for example, reports conjugating plant-based CuO NPs to multiple thiosemicarbazone ligands using ultrasonication. The nanoconjugates demonstrated enhanced antibacterial, antioxidant, and photocatalytic activities. Among the conjugates, CuO/3-APTSC exhibited the highest scavenging activity of all the nanoconjugates and 84% degradation of malachite green dye.8 The same research group also reported the conjugation of TSCs with ZnO NPs, resulting in conjugates with enhanced antibacterial, antioxidant and photocatalytic activity.9 In a similar vein, another work by Chaba et al. described the use of a zinc oxide-coated carbon nanofiber composite (ZnO@CNF) as an adsorbent that was successfully synthesized by the hydrothermal technique to remove amoxicillin from environmental water matrices. Furthermore, it was found that the carbon nanofiber nanocomposite coated with ZnO may be reused for up to fifteen cycles.10 Another recent work used the colloid solution approach with polyvinyl alcohol (PVA) as a surfactant to produce a series of air filters based on the TiO2/Pd nanophotocatalyst. The resulting nanophotocatalysts (1.5 wt% of PVA) had a superior N2 adsorption ability than TiO2 and other catalyst samples with a larger surface area, according to the nitrogen adsorption–desorption isotherms of the catalysts. In investigations involving the photocatalytic degradation of toluene in a gaseous phase under visible light illumination, the photocatalytic activity of the nanophotocatalyst filter was examined. The 10 wt% nanophotocatalyst filter showed the greatest photocatalytic capacity to break down toluene out of all the filters. For the elimination of toluene, the photocatalytic effectiveness of the nanophotocatalyst containing 1.5 wt% PVA was considerably increased. The presence of Pd NPs on the surface of the TiO2 nanocatalyst and the electron transfer between the conduction band/defect level and Pd NPs may have improved the adsorption capacity and enhanced the electron–hole pair separation, which in turn may have contributed to the enhanced photodegradation of toluene.11 Noble metal-based nanocomposites, particularly those containing Au, Ag, or Pt NPs, have also been thoroughly studied for their unique optical, catalytic, and biological properties in addition to transition metal-based nanocomposites. The plasmonic characteristics, high stability, and adaptable surface chemistry of these noble metal-based systems frequently contribute to their improved performance in fields like sensing, catalysis, and bioapplications.12 A recent work, for instance, investigated the use of silk sericin-based silver NPs (SS AgNPs) for food coating and antibacterial applications. Food coating experiments on tomatoes dramatically decreased weight loss and microbial contamination, while appreciable antibacterial activity was observed against Pseudomonas sp. and Staphylococcus sp. The safety of SS-AgNPs for applications in biomedicine and food was confirmed by additional biocompatibility testing using hemolysis and MTT assays.13 In order to improve capabilities in fields like energy storage, sensing, and biomedicine, researchers have recently focused more on studying rare-earth element (REE)-based nanocomposites. This is because REEs have special electrical, luminescent, and redox properties. In order to achieve synergistic effects and open up new application domains, the current research trend is moving toward the construction of rare-earth nanocomposites, such as rare-earth-doped nanophosphors and REE-based metal–organic frameworks.14 A relatively recent study synthesized a nanocomposite of lanthanum ferrite and antimony oxide heterojunction (LaFe2O3/Sb2O3) using a straightforward hydrothermal technique to enable the photocatalytic degradation of MG dye in the presence of visible light. The photocatalytic results show good redox behavior and efficient charge transfer kinetics across the heterojunction interface. The binary nanocomposite's increased surface area and decreased charge transfer resistance have led to the effective degradation of 98% of the organic dye in 88 minutes. The MG dye photodegradation mechanism was shown to involve hydroxyl radicals (OH) in scavenging studies, while recycling research proved the material's dependability and long-term application as a photocatalyst.15 GO/g-C3N4/Fe2O3 composites based on cerium and ytterbium are reported in another investigation. Super-capacitance and EDLC characteristics were measured using electrochemical methods (cyclic voltammetry, galvanic charge discharge, and electrochemical impedance spectroscopy). According to the results, at a current density of 1 A g−1 and a scan rate of 2 mV s−1, the gravimetric capacitance computed from GCD results is 219 F g−1 for ytterbium-based nanocomposites and 169 F g−1 for cerium-based nanocomposites. The ytterbium-based nanocomposite's calculated specific capacitance is 189 F g−1, while the cerium-based material's is 125 F g−1. The results of the EIS indicated that cerium-based nanocomposites provided more resistance than ytterbium. According to the results, GO/g-C3N4/Fe2O3 based on ytterbium oxide is a more effective electrocatalyst than cerium-based nanocomposites. The degradation efficiency of ytterbium oxide-based GO/g-C3N4/Fe2O3 was higher than that of cerium-based GO/g-C3N4/Fe2O3, indicating that photocatalytic activity correlated with the electrochemical data.16
Ytterbium oxide (Yb2O3) NPs possess unique optical, redox, and electronic properties arising from the 4f-orbital configuration of ytterbium. Their high chemical stability, oxygen vacancy formation tendency, and efficient electron-transfer capability have led to their increasing use in photocatalysis, pollutant degradation, and electrochemical detection platforms. Recent studies have reported Yb-based nanostructures demonstrating enhanced visible-light-driven catalytic activity as well as improved electrochemical response due to efficient charge transport mechanisms.17
Zinc ferrite (ZnFe2O4), a spinel-type ferrite NP, has become more and more popular because of its many uses, which include gas sensing, visible-light photocatalysis, and high-capacity energy storage. These uses are made possible by its advantageous bandgap, magnetic behaviour, and chemical stability. Because of its low toxicity, biocompatibility, and adjustable magnetic properties, zinc ferrite NPs are being investigated more and more for biomedical and environmental applications outside of energy, such as medicine delivery, pollutant degradation, and magnetic hyperthermia.18 ZnFe2O4 is well known for its narrow bandgap, strong magnetic behaviour, structural stability, and environmental compatibility. These attributes make ZnFe2O4 an attractive candidate for visible-light photocatalysis, dye degradation, and electrochemical sensing applications. Blended and doped ZnFe2O4 systems have been shown to significantly enhance charge carrier mobility, generate more active sites, and improve photocatalytic degradation efficiencies towards organic dyes and pollutants.19 As per a recent study by Keerthana et al., for instance, ZnFe2O4 NPs doped with samarium were synthesized using a straightforward co-precipitation technique. The host performs better in dye degradation because of the small bandgap of 2% Sm-doped ZnFe2O4. ZnFe2O4 NPs doped with 2% Sm effectively broke down the dye to 65%. Doping the zinc ferrite NPs with Sm improved their ability to degrade the MB dye. The 2% Sm-doped ZnFe2O4 material demonstrated reusability for real-world applications by withstanding three cycles with a comparable efficiency of 65%.20 Earlier, copper ferrite NPs (CuFe2O4) synthesized from Cissus rotundifolia plant extract were tested for their antibacterial and photocatalytic activity, showing 82% degradation of the MB dye under UV-light.21 Roostaei et al. reported ZnFe2O4/ZnO/CeO2 ternary magnetic nanocomposites using the sol–gel auto-combustion process, showing excellent efficiency under visible light. In aqueous solutions, it was then used to photodegrade the cationic dye methyl violet and the anionic dye erythrosine. After 120 minutes of exposure to visible light, ZnFe2O4/ZnO/CeO2 demonstrated superior photocatalytic performance against an anionic dye. The nanocomposite showed good recyclability after the fifth cycle with minimal loss of photocatalytic activity because of synergistic characteristics, large surface area, enhanced active sites, and recyclability, etc.22 A synergistic effect was also obtained in the case of an earlier reported nanocomposite (B-CuO/rGO), which showed improved photocatalytic activity against the MB dye (98%) as compared to B-CuO alone (85%) in 90 minutes.23
In addition to dyes, pesticides have also emerged as potent pollutants. The products formed by the transformation of pesticides have gained attention because their high concentration can lead to toxicity.24 NPs have been used to degrade pesticides like dichlorophenyltrichloroethane (DDT) in a short time.25 Under acidic conditions, Fenton oxidation of DDT was achieved following pseudo-first-order kinetics.26 Bimetallic systems of NPs like Mg/Pd and Ni/Fe have been used to remove high concentrations of DDT.27 Zero-valent FeNPs have been used to degrade lindane, generating g-3,4,5,6-tetrachlorocyclohexane.28 It was also degraded under visible light by TiO2 doped with nitrogen.29 Iron sulphide NPs, stabilized by biopolymers, also degraded lindane up to 94% in 8 h.30 Glyphosate (N-phosphonomethyl glycine) is a non-selective, organophosphate herbicide, affecting human health by accumulating in the food chain. CeO2 NPs, capped with citric acid, have been used to degrade glyphosate from water at pH = 4, following pseudo-kinetics. Citric acid may play a role in the initiation of electron transfer to regenerate Ce(III) sites as electron donors, enhancing photocatalytic activity through ligand-to-metal charge transfer.31 In another study, calcium peroxide NPs (nCPs) were fabricated, generating hydroxyl radicals (˙OH) with Fe(II), and were used as a catalyst forming a Fenton-based system. At pH = 3, the nCPs achieved 99.60% removal of total phosphorus and 75.10% chemical oxygen demand (COD), proving to be efficient for glyphosate removal from aqueous systems, following the Behnajady–Modirshahla–Ghanbery (BMG) kinetics model.32 In another study, ZnO NPs were evaluated for the removal of glyphosate to obtain suspensions of glyphosate-ZnO NPs. It was found that ZnO NPs removed approximately 70–90% herbicide from the aqueous system.33
Phenols are commonly utilised in cosmetics, tanning, medicines, photography, etc.; however, some of them also get released into the environment and contaminate rivers and groundwater. They have been efficiently detected by electrochemical methods owing to their high sensitivity, quick response and easy output. Graphene hybrids have been employed to assess organic pollutants owing to their large surface area and conductivity.34 Catechol is an important environmental contaminant, and has been found to be removed through reduced graphene oxide nanosheets and CuO modified GEC.35 The CuO/rGO nanocomposite was also prepared for the sensing of catechol.36 The NiO/CNT nanocomposite fabricated by atomic layer deposition was tested for its electrochemical performance against hydroquinone and catechols. The redox peak currents of both compounds were found to be enhanced and the oxidation peaks were also well separated.37 Another graphene-based sensor doped with nitrogen and palygorskite (Pal) was developed for the simultaneous detection of hydroquinone and catechol. NGE improved the poor conductivity of Pal and served as an excellent matrix for adherence. The modified electrode showed enhanced redox peak currents for both pollutants.38 Among the various analytical techniques developed for catechol detection, electrochemical sensing has emerged as a promising approach owing to its high sensitivity, rapid response, cost-effectiveness, and ease of miniaturization. Electrochemical methods allow for the direct and selective oxidation of catechol, enabling accurate quantification even in complex matrices. Recent advances in nanotechnology have significantly improved the performance of electrochemical sensors. Incorporating nanomaterials into sensor platforms can enhance electron transfer rates, increase the surface area, and promote catalytic activity. Metal oxide-based nanocomposites in particular have drawn a lot of interest due to their stability, conductivity, and tunable surface properties. For example, a study conducted by Karthika A. et al. reported a nanocomposite material consisting of SrWO4 (SrW), zinc phthalocyanine (ZnP) and Pd–carbon (Pd–C) synthesised via a simplistic sonochemical process for serotonin (ST) sensing. Under the optimized conditions, the SrW/ZnP/Pd–C nanocomposite modified glassy carbon electrode (GCE) shows a higher electrocatalytic activity towards serotonin (ST) oxidation compared to SrW and SrW/ZnP modified GCEs in 0.05 M phosphate buffered saline (PBS) (pH 7) due to enhanced electrochemical activity of the catalyst owing to better electron transfer efficiency, good conductivity and high surface area of the nanocomposite material. The fabricated ST sensor displayed excellent repeatability, reproducibility and stability towards the detection of ST in 0.05 M PBS. Furthermore, the SrW/ZnP/Pd–C@GCE sensor for ST was tested on real-world blood serum, urine and blood plasma samples and exhibited an excellent recovery percentage.39 Similarly, another study reported a novel polyvinyl alcohol supported cupric oxide nanocomposite (CuO/PVA) modified glassy carbon electrode (GCE), developed for the determination of mercury(II) ions by electrochemical methods. The CuO/PVA nanocomposite was synthesized by the sonochemical method. The CuO/PVA nanocomposite modified GCE for Hg(II) ion detection provides a wide linear response range (10–70 μM) and a lower detection limit (0.42 nM), respectively. The CuO/PVA GCE showed good selectivity towards Hg(II) ion detection even in the presence of interferents such as arsenic, cadmium, lead, chromium and nickel. The proposed fabricated CuO/PVA GCE sensor exhibited good reproducibility for the selective determination of mercury in river and tap water and obtained good recovery results.40 Thus, such materials can effectively facilitate redox reactions and improve the sensitivity and selectivity of pollutants and other chemical substances like catechol detection.41 Polyethylene glycol (PEG) has recently emerged as one of the most effective polymeric stabilizers and structure-directing agents for NP synthesis due to its ability to control nucleation, restrict particle agglomeration, and enhance surface functionalization. PEG-assisted nanostructures often exhibit improved dispersion, enhanced charge separation, and better catalytic and electrochemical activity, making PEG an excellent additive in photocatalytic and sensing nanocomposites.42 Recent research has demonstrated that combining rare-earth oxides with spinel ferrites creates heterojunction interfaces capable of improving light harvesting and accelerating charge separation. Such binary nanocomposites display superior photocatalytic and electrochemical performance compared to individual components due to synergistic band alignment and reduced electron–hole recombination. Therefore, blending Yb2O3 with ZnFe2O4 provides a promising route to design efficient multifunctional nanocomposites for environmental remediation and electrochemical sensing.
In the present work, Yb2O3 NPs and ZnFe2O4 NPs were synthesized via a simple co-precipitation method, and they were both conjugated to form a binary nanocomposite through a sonication process. The morphological and structural parameters of the NPs and the nanocomposite were analyzed employing different analytical techniques. Visible light irradiation was used to examine the photocatalytic activity of the nanocomposite and electrochemical sensing applications were also studied for the detection of catechol.
2. Materials and methods
All the chemicals employed in this work were of analytical grade, and no additional purification was required before use. Ytterbium nitrate pentahydrate (Yb(NO3)3·5H2O) with 99.9% purity, zinc nitrate hexahydrate (Zn(NO3)2·6H2O) with 98% purity, and ferric nitrate nonahydrate (Fe(NO3)3·9H2O) with 98% purity were procured from Sisco Research Laboratories (SRL) Pvt. Ltd. PEG 1500, sodium hydroxide (NaOH), ethanol and Brilliant Blue FCF dye were procured from SRL as well. Ammonium salt of glyphosate (71% SG) was obtained from Exylon. A NEUATION iFUGE D08 centrifuge machine was used for centrifuging the reaction mixtures and recovering the precipitates. For the formation of a binary nanocomposite, an ultrasonic cleaner (sonicator) of the LMUC series (Model No.: LMUC3D) was used. UV–Visible absorption spectra were recorded between 200 and 800 nm using an Agilent Cary UV-Vis Compact Peltier R3.15AL250V spectrophotometer. The crystallinity and phase identification purity of the synthesised precursors and the binary nanocomposite were detected by using a diffractometer (Model No.: D8 Advance Eco, Make: Bruker, Germany). FT-IR spectra were obtained using a spectrophotometer (Model No.: Nicole 6700, Make: Thermo-Scientific, USA). The morphology of the nanostructures was determined using a scanning electron microscope (Model: JSM 6490 LV, Make: JEOL, Japan). Electrochemical sensing was carried out using the CHI-6054F electrochemical workstation.
3. Experimental
3.1 Synthesis of Yb2O3 NPs
Yb2O3 NPs were prepared via co-precipitation (eqn (1) and (2)).43 A 0.1 M solution of Yb(NO3)3·5H2O and 0.3 M solution of NaOH were prepared in 100 mL of distilled water each. NaOH solution was added dropwise to the former solution with constant stirring at room temperature until the pH became 9 and white coloured ytterbium hydroxide was completely precipitated. The mixture was stirred for one hour after complete precipitation. The precipitate was recovered through centrifugation and was washed thoroughly multiple times with ethanol and water to remove extra base and any other impurities, and was dried in an oven at 110 °C for 5–6 hours. Using a mortar and pestle, the residue was ground into a fine powder, which was then calcined in a muffle furnace for four hours at 500 °C to produce Yb2O3 NPs (Fig. 1).
 |
| | Fig. 1 Schematic representation of the synthesis of Yb2O3 NPs. | |
Precipitation:
| |  | (1) |
Calcination at 500 °C:
| | | 2Yb(OH)3 → Yb2O3 + 3H2O | (2) |
3.2 Synthesis of ZnFe2O4 NPs
ZnFe2O4 NPs were also prepared through the co-precipitation reaction (eqn (3) and (4)).44 0.1 mol of Zn(NO3)2·6H2O, and 0.2 mol of Fe(NO3)3·9H2O were dissolved in 100 mL of distilled water with continuous stirring to form a uniform precursor solution. To this solution, 10% NaOH was added dropwise with continuous stirring until the medium turned basic (pH = 9) and complete precipitation occurred. After recovering the precipitate by centrifugation, it was properly cleaned with ethanol and water to get rid of any excess base and any contaminants. It was then dried for 5–6 hours at 110 °C in an oven. Using a mortar and pestle, the residue was ground into a fine powder, which was then calcined in a muffle furnace for 4 hours at 500 °C to produce ZnFe2O4 NPs (Fig. 2).
 |
| | Fig. 2 Schematic representation of the synthesis of ZnFe2O4 NPs. | |
Precipitation:
| | 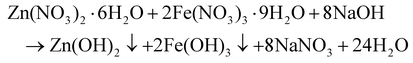 | (3) |
Calcination at 500 °C:
| | | Zn(OH)2 + 2Fe(OH)3 → ZnFe2O4 + 4H2O | (4) |
3.3 Synthesis of the binary nanocomposite Yb2O3/ZnFe2O4@PEG
For the synthesis of the binary nanocomposite, the ultrasonication method was utilized (eqn (5)).45 100 mg of PEG was dissolved in 50 mL of distilled water while heating. To this solution, 50 mg each of Yb2O3 and ZnFe2O4 NPs were added and stirred for 5–10 minutes. At room temperature, this mixture was sonicated for 2 hours and then stirred for 6–7 hours. After recovering the residue by centrifugation, it was extensively cleaned of contaminants using ethanol and water and dried for 4–5 hours at 110 °C in an oven (Fig. 3). During this ultrasonication-assisted synthesis, PEG was used as a stabilizing agent because it prevents aggregation by coating the surface of NPs, providing steric stabilization. Although PEG-free control experiments could not be incorporated in the present study, the role of PEG in the composite is supported by multiple lines of evidence. FT-IR analysis (Fig. 4) clearly shows characteristic C–O–C stretching vibrations, confirming successful PEG binding on the nanoparticle surface. PEG is known to act as a steric stabilizer and dispersing agent, improving nanoparticle distribution and preventing agglomeration. The PEG-assisted composite also exhibits a higher photocatalytic degradation efficiency and improved optical absorption compared to the individual components, indirectly indicating enhanced interfacial charge transfer. These effects are consistent with PEG-mediated surface modification, which introduces oxygen-containing functional groups that facilitate electron mobility at the heterojunction interface.
 |
| | Fig. 3 Schematic representation of the synthesis of the binary nanocomposite Yb2O3/ZnFe2O4@PEG. | |
 |
| | Fig. 4 FT-IR spectra of Yb2O3 NPs, ZnFe2O4 NPs, and the binary composite. | |
Ultrasonication:
| | 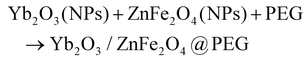 | (5) |
3.4 Photocatalytic activity evaluation
The photocatalytic activity of NPs was assessed using visible light against the herbicide glyphosate and the Brilliant Blue FCF dye. 50 mg of NPs were combined with 50 mL of dye solution (10 ppm). Prior to the solution being exposed to light, the mixture was magnetically agitated for 30 minutes in the dark to reach desorption/absorption equilibrium. A UV-Visible spectrophotometer (200 nm to 800 nm) was used to measure the absorbance of the degraded dye solution after 3 mL of the sample was removed every 15 minutes. The percentage degradation of the dye was calculated using eqn (6).| | | % degradation = (A0 − At)/A0 | (6) |
where A0 represents the dye absorbance at the beginning, and At represents the dye absorbance at time t. Additionally, the kinetics of Brilliant Blue FCF degradation were examined. Plotting ln(Ct) versus time allowed for the evaluation of the degradation kinetics. The kinetic study was performed using the pseudo-first-order kinetic equation (7).
Here, C0 represents the initial concentration, Ct denotes the concentration at time t, and k is the rate constant. The slope of the plot of ln(C0/Ct) against time gives a rate constant, k.
3.5 Electrochemical sensing
The electrochemical workstation consisted of three electrodes with a glassy carbon electrode (GCE) as the working, a Pt wire as the counter and Ag/AgCl as the reference electrodes. The electrode materials were prepared by dissolving 5 mg of the binary nanocomposite in 5 mL of ethanol and ultrasonically treating it for 20 minutes. 6 µL of this suspension was drop-cast onto the surface of GCE and dried under a bulb for 30 minutes. All the electrochemical measurements were performed in 0.1 M phosphate buffer having pH = 7.
4. Results and discussion
4.1 FT-IR analysis
The FT-IR spectra (Fig. 4) were recorded in the range of 4000–400 cm−1 to investigate the functional groups and confirm the formation of metal–oxygen bonds. In Yb2O3 NPs, a broad absorption band observed around 3390.24 cm−1 and 3210.89 cm−1 corresponds to the O–H stretching vibrations, indicating the presence of adsorbed water or surface hydroxyl groups. The bands at 1536.99 cm−1 and 1376.92 cm−1 were assigned to the bending vibrations of water molecules.46 Most importantly, the strong peaks at 578.54 cm−1 and 416.55 cm−1 confirmed Yb–O stretching vibrations, characteristic of metal–oxygen bonding in rare earth metal oxides.47 These results affirm the successful formation of Yb2O3 NPs, with typical vibrational features of metal–oxygen lattices and minor contributions from surface-adsorbed species. The FT-IR spectra of the synthesised ZnFe2O4 NPs, observed between 4000 and 400 cm−1, indicate the presence of distinctive functional groups and the spinel ferrite structure. Wide absorption bands at 3386 cm−1 and 3230 cm−1 indicate the presence of adsorbed water or surface hydroxyl groups. The bands at 1695 cm−1 and 1378 cm−1 correspond to H–O–H bending and C–H deformation vibrations, respectively. Sharp bands at 769 cm−1, 543 cm−1, and 455 cm−1 indicate Fe–O and Zn–O stretching vibrations in the spinel lattice, which correspond to metal–oxygen bonds in tetrahedral and octahedral sites. The successful formation of the spinel ZnFe2O4 was confirmed by these bands. Surface interactions may modify metal–oxygen vibrations, resulting in additional small peaks between 1149 and 956 cm−1. FT-IR findings indicate the formation of phase-pure ZnFe2O4 NPs.48 The FT-IR spectrum of the PEG-supported binary nanocomposite exhibited several absorption bands, indicating the presence of functional groups and metal–oxygen vibrations. Adsorbed water molecules and surface hydroxyl groups exhibit a broad absorption band at about 3455 cm−1 and a lesser shoulder near 3872 cm−1, indicating O–H stretching vibrations. The signal at 2915 cm−1 is attributable to C–H stretching vibrations, which may originate from capping agents or leftover solvents used during synthesis. The bands at 1160 cm−1 and 975 cm−1 correspond to stretching and bending vibrations, suggesting the presence of polymeric residual PEG.49 The fingerprint region shows absorption bands at 647 cm−1 and 420 cm−1, indicating metal–oxygen (M–O) vibrations. The band at ∼647 cm−1 is assigned to Fe–O stretching vibrations in the tetrahedral site of the spinel structure of ZnFe2O4. The lower frequency band at 420 cm−1 corresponds to the stretching modes of Yb–O bonds in the Yb2O3 lattice. These bands confirm the creation of a binary composite while retaining the metal oxide framework. The existence of these distinctive vibrations suggests the effective synthesis of the Yb2O3/ZnFe2O4@PEG binary nanocomposite.50
4.2 XRD analysis
The XRD pattern (Fig. 5a) of the synthesized Yb2O3 NPs exhibits distinct peaks at 2θ values of 29.50°, 34.22°, 49.24°, and 58.53°, corresponding to the (222), (400), (440), and (622) planes, respectively, of the cubic Yb2O3 phase (JCPDS card no. 43-1037). A high degree of crystallinity in the prepared NPs is indicated by the crisp and strong peaks. The absence of additional peaks suggests high phase purity with no detectable secondary phases or impurities. The most intense peak at 2θ ≈ 29.50° corresponds to the (222) reflection plane of cubic Yb2O3 NPs. The broadening of the peaks, especially around 29–35°, suggests the nanoscale size of the crystallites with an average crystallite size of 10.513 nm (Table 1).46 The XRD pattern of the synthesized ZnFe2O4 NPs (Fig. 5b) revealed distinct and sharp peaks at 2θ values of approximately 29.77°, 35.04°, 42.63°, 56.44°, and 62.07°, which correspond to the (220), (311), (400), (511), and (440) crystal planes of the cubic spinel structure, respectively, as per JCPDS card no. 22-1012.51 The peak at 35.04° shows the presence of the typical (311) plane of ZnFe2O4, indicating the successful development of the spinel ferrite phase. The absence of impurity peaks contributes to the sample's purity and phase stability.52 The broad breadth of the peaks suggests the nanocrystalline structure of the material, which can be further confirmed by using the Scherrer equation to estimate crystallite size. The average crystallite size came out to be 10.206 nm (Table 2).53 The XRD spectrum of the synthesized binary nanocomposite (Fig. 5c) exhibits distinct diffraction peaks at 2θ values of approximately 29.36°, 34.08°, 49.09°, and 58.38°. These peaks are well-defined and sharp, indicating the crystalline nature of the composite material. The peak at 29.36° corresponds to the (222) plane of Yb2O3 NPs, matching with standard JCPDS card no. 43-1037 for cubic ytterbium oxide.54 The peaks at 34.08°, 49.09°, and 58.38° are consistent with the (311), (511), and (440) planes of ZnFe2O4, in agreement with JCPDS card no. 22-1012 for the cubic spinel ferrite phase.55 The average crystallite size was found to be 9.147 nm (Table 3). The presence of Yb2O3 and ZnFe2O4 peaks suggests that no structural interference or phase change occurred during composite synthesis. The retention of both components’ primary peaks indicates that the composite is physically blended or has weak structural interactions, without generating a new crystalline phase. The peak broadening, particularly at lower angles, indicates a nanocrystalline structure with small crystallite sizes, as expected for composites made by co-precipitation or ultrasonication processes.
 |
| | Fig. 5 XRD spectra of (a) Yb2O3 NPs, (b) ZnFe2O4 NPs, and (c) Yb2O3/ZnFe2O4@PEG. | |
Table 1 XRD data of Yb2O3 NPs
|
K
|
λ (Å) |
Peak position 2θ (°) |
FWHM βsize (°) |
L (nm) |
| 0.94 |
1.54178 |
29.50907 |
0.82405 |
10.420 |
| 0.94 |
1.54178 |
34.214 |
0.79794 |
10.888 |
| 0.94 |
1.54178 |
49.23898 |
0.86838 |
10.519 |
| 0.94 |
1.54178 |
58.52044 |
0.93099 |
10.224 |
|
|
|
|
Average crystallite size = 10.513 nm |
Table 2 XRD data of ZnFe2O4 NPs
|
K
|
λ (Å) |
Peak position 2θ (°) |
FWHM βsize (°) |
L (nm) |
| 0.94 |
1.54178 |
29.72493 |
0.80906 |
10.619 |
| 0.94 |
1.54178 |
35.05864 |
1.10845 |
7.856 |
| 0.94 |
1.54178 |
42.64213 |
0.80743 |
11.040 |
| 0.94 |
1.54178 |
56.48093 |
0.8064 |
11.689 |
| 0.94 |
1.54178 |
62.04735 |
0.98611 |
9.826 |
|
|
|
|
Average crystallite size = 10.206 nm |
Table 3 XRD data of Yb2O3/ZnFe2O4@PEG
|
K
|
λ (Å) |
Peak position 2θ (°) |
FWHM βsize (°) |
L (nm) |
| 0.94 |
1.54178 |
29.36721 |
0.85113 |
10.086 |
| 0.94 |
1.54178 |
34.43209 |
1.55889 |
5.577 |
| 0.94 |
1.54178 |
49.08477 |
0.85916 |
10.625 |
| 0.94 |
1.54178 |
58.37889 |
0.92322 |
10.303 |
|
|
|
|
Average crystallite size = 9.147 nm |
The average crystallite size was estimated using the Scherrer equation (eqn (8)).56
| | 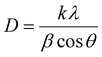 | (8) |
where
k = 0.94 is the Scherrer constant,
λ = incident X-ray wavelength = 1.542 Å,
β = full width at half maximum (FWHM) of the diffraction peak, and
θ = diffraction angle.
4.3 UV-Vis analysis
The UV-Vis absorption spectrum (Fig. 6a) of the produced Yb2O3 NPs reveals a significant absorption peak at 368 nm, attributable to the intrinsic electronic transitions from the valence to the conduction band. This absorption trend is typical of rare-earth metal oxides and indicates a broad band gap, consistent with Yb2O3's semiconducting properties. The band gap calculated through the Tauc plot was found to be 4.91 eV.46 Two strong peaks were visible in the ZnFe2O4 NP UV–Vis absorption spectra at around 221 and 342 nm. The sharp absorption at 221 nm is assigned to O2− → Fe3+ charge transfer transitions within the spinel lattice, while the absorption at 342 nm corresponds to band gap excitation. Compared to bulk ZnFe2O4 (band gap ∼1.9–2.2 eV), the observed blue shift indicates a higher band gap, which is a characteristic of nanoparticles due to quantum confinement effects, confirming the nanoscale nature of the synthesized ZnFe2O4 NPs. The measured band gap was 4.61 eV (Fig. 6b).53 The UV-Visible absorption spectra of the Yb2O3/ZnFe2O4@PEG binary nanocomposite showed strong peaks at 312 nm, 335 nm, and 366 nm, which correspond to the distinctive charge transfer transitions in ZnFe2O4 and interband transitions of Yb(III) ions. The conjugation between the two components decreases the band gap to 3.99 eV. The spectrum demonstrates that the absorbance gradually decreases as the wavelength increases, which is consistent with semiconductor behaviour. The existence of numerous absorption patterns suggests an interaction between Yb2O3 and ZnFe2O4 NPs, leading to a modified band structure and synergistic optical characteristics. Such properties may increase the material's suitability for photocatalytic and optoelectronic applications.50 Based on the optical band gaps obtained from the Tauc plots (Yb2O3: 4.91 eV, ZnFe2O4: 4.61 eV, and Yb2O3/ZnFe2O4@PEG: 3.99 eV), the conduction band (CB) and valence band (VB) edge potentials were calculated using the Mulliken electronegativity (χ) method. The CB edge was determined using ECB = χ − EC − 0.5Eg, while the VB edge was obtained from EVB = ECB + Eg, where EC = 4.5 eV. The calculated band-edge positions were: Yb2O3 (CB = −1.41 eV, VB = +3.51 eV), ZnFe2O4 (CB = −0.91 eV, VB = +3.71 eV), and Yb2O3/ZnFe2O4@PEG (CB = −0.75 eV, VB = +3.25 eV). These values clearly reveal a staggered Type-II heterojunction, which allows photogenerated electrons to migrate from the CB of ZnFe2O4 to that of Yb2O3, while holes transfer from the VB of Yb2O3 to the VB of ZnFe2O4, thereby enhancing charge separation and photocatalytic efficiency.
 |
| | Fig. 6 (a) UV-Vis spectra, and (b) band gaps of Yb2O3 NPs, ZnFe2O4 NPs, and Yb2O3/ZnFe2O4@PEG. | |
4.4 SEM and EDX analysis
The EDX spectra of the precursor NPs and the binary nanocomposite (Fig. 7a–c) confirmed the presence of their respective components. The elemental composition in Yb2O3 NPs was found to be: O (63.30%) and Yb (36.70%), whereas in ZnFe2O4, it was found to be O (47.49%), Fe (28.64%), and Zn (23.87%), and in the binary nanocomposite, it was found to be C (58.27%), O (16.57%), Fe (26.64%), Zn (7.04%), and Yb (8.0%).
 |
| | Fig. 7 EDX of (a) Yb2O3 NPs, (b) ZnFe2O4 NPs, and (c) Yb2O3/ZnFe2O4@PEG. | |
The SEM micrograph of Yb2O3 NPs (Fig. 8(a and b)) shows the formation of irregularly shaped agglomerates composed of fine NPs. The particle distribution appears heterogeneous, with smaller NPs aggregated together to form larger clusters. The surface morphology suggests that the particles are mostly granular with a rough texture. Larger secondary aggregates are also visible, which is commonly observed due to the high surface energy of rare-earth oxides leading to agglomeration. This morphology is consistent with previous observations in the literature. There have been reports of similar agglomerated rare-earth oxide NP formations in the literature. For instance, almost spherical particles aggregated into bigger clusters with pore spaces between agglomerates were seen in doped Yb2O3 NPs produced by wet chemical techniques, which is consistent with the high surface energy effects characteristic of rare-earth oxides.54 Mouattah et al. studied mixed rare-earth oxide systems including Yb1.4Y0.6O3 and found that agglomeration of particles increased with precipitation and thermal treatment, a phenomenon driven by high surface energy and thermal coalescence.57 The SEM images of ZnFe2O4 NPs (Fig. 9(a and b)) show needle-like morphology with a network-like arrangement. The particles appear elongated and intertwined, forming clusters rather than discrete spherical grains. The surface texture looks rough and porous, which is characteristic of ferrite nanomaterials synthesized through chemical routes. Similar agglomerated morphologies have been reported for ZnFe2O4 NPs synthesized via green and hydrothermal methods.53 For ZnFe2O4 NPs produced chemically and hydrothermally, similar morphologies have been extensively documented in the literature. For example, Liu et al. reported porous and elongated ZnFe2O4 nanorods that formed interconnected network-like structures. The high surface energy of ferrite NPs and magnetic interactions caused the rod-like units to entwine and aggregate. Similar to the current observation, the authors attributed the rough and porous surface roughness to gas development and crystal growth during calcination.58 In another study, Wang et al. used a hydrothermal method to synthesize ZnFe2O4 nanostructures, and instead of isolated grains, needle-like and rod-shaped particles grouped into clusters were obtained. Rough surfaces and interparticle porosity, which were thought to be typical characteristics of chemically generated spinel ferrites, were seen in the SEM images.59 The SEM micrograph of the binary nanocomposite (Fig. 10(a and b)) reveals a heterogeneous morphology consisting of irregularly shaped agglomerates and interconnected clusters. The remaining SEM images are given in the SI as Fig. S1–S3. The surface appears rough and porous, with both fine NPs and larger aggregates clearly visible. Compared to the individual ZnFe2O4 and Yb2O3 NPs’ SEM images, the nanocomposite displays a more densely packed network, suggesting successful mixing and integration of the two components. The morphology indicates that ultrasonic treatment effectively facilitated the combination of ZnFe2O4 and Yb2O3 NPs, leading to intimate contact between the two phases. The observed agglomeration is expected due to the high surface energy and magnetic/electrostatic interactions of NPs. The rough, porous structure of the nanocomposite is advantageous, as it may provide a higher surface area, promoting enhanced catalytic and adsorption properties. Similar morphological traits—such as enhanced porosity and intimate interfacing—were observed in ferrite–graphene oxide composites, which promoted catalytic performance.50
 |
| | Fig. 8 SEM images of Yb2O3 NPs at (a) 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000× magnification and (b) 30 000× magnification and (b) 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000× magnification. 000× magnification. | |
 |
| | Fig. 9 SEM images of ZnFe2O4 NPs at (a) 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000× magnification and (b) 30 000× magnification and (b) 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000× magnification. 000× magnification. | |
 |
| | Fig. 10 SEM images of the binary nanocomposite at (a) 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000× magnification and (b) 30 000× magnification and (b) 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000× magnification. 000× magnification. | |
4.5 Photocatalytic degradation of dyes
Brilliant Blue FCF (also known as FD&C Blue no. 1, E133) is a synthetic anionic triphenylmethane dye (Fig. 16) that is commonly used as a colouring agent in the food, cosmetics, textile, leather, paper, and pharmaceutical sectors. It has become one of the most widely used dyes due to its bright colour, excellent water solubility, and stability in the presence of light and heat. However, its widespread application causes large discharge into aquatic habitats via industrial effluents and domestic wastewater.60 Brilliant Blue remains in water bodies due to its chemical stability and resistance to biodegradation, contributing to environmental pollution. As a result, metal oxide nanoparticles and composites were used to degrade the dye.61 In the present study, the photocatalytic degradation of Brilliant Blue FCF was studied using Yb2O3 NPs, ZnFe2O4 NPs and their binary nanocomposite (Fig. 13(a, b and c)). The dye degradation with Yb2O3 NPs was found to be 83.09% (Fig. 14) and follows pseudo-first order kinetics with a rate constant of 1.602 × 10−2 min−1 (R2 = 0.99068) as shown in Fig. 15(a) while ZnFe2O4 NPs’ photodegradation was found to be 77%, also following pseudo-first order kinetics and the rate constant was found to be 1.577 × 10−2 min−1 (R2 = 0.9909) as shown in Fig. 15(b). Similarly, the photodegradation of the nanocomposite Yb2O3/ZnFe2O4@PEG following pseudo-first order kinetics was found to be 86.38% and the rate constant was found to be 2.09 × 10−2 min−1 (R2 = 0.98185) as shown in Fig. 15(c). The enhanced superior photocatalytic degradation efficiency exhibited by the binary nanocomposite toward Brilliant Blue dye compared to the individual NPs can be attributed to synergistic effects. Ultrasonically combining Yb2O3 and ZnFe2O4 NPs resulted in the formation of a heterojunction at the contact, reducing the band gap energy. This enables the effective separation of photogenerated electron–hole pairs. This inhibits the recombination of charge carriers, increasing the number of active species available for dye degradation. The heterostructure encourages the formation of reactive oxygen species, which play an important role in breaking down large Brilliant Blue dye molecules into smaller, less hazardous intermediates and, eventually, mineralizing them. The ultrasonically produced nanocomposite provides strong interfacial contact between ZnFe2O4 and Yb2O3. This increases quick charge transfer while reducing electron–hole recombination, a frequent disadvantage of single-component photocatalysts. Thus, the binary nanocomposite's better performance can be attributed to synergistic heterojunction creation, improved charge separation, and increased production of reactive species, all of which jointly boost degradation efficiency when compared to individual nanoparticles. Previous research has also demonstrated the photocatalytic capability of these nanocomposites. For instance, a recent study reported the production of a novel ZnFe2O4–ZnO–CeO2 based magnetically separable ternary nanophotocatalyst. This ternary photocatalyst combines the oxygen storage capacity and photocatalytic qualities of CeO2, the durability and catalytic effectiveness of ZnO, and the magnetic features of ZnFe2O4. More than 90% of the erythrosine dye was degraded by the photocatalyst, demonstrating exceptional photocatalytic activity. Because of its synergistic qualities, large surface area, improved active sites, improved charge dynamics, and potential for reuse, ZnFe2O4, ZnO, and CeO2 together boost photocatalytic performance.22 Another recent study revealed the production of visible-light-responsive lanthanum-doped copper ferrite/graphitic carbon nitride composites (La-CuFe2O4/g-C3N4). This heterojunction photocatalyst demonstrated exceptional photodegradation of 97.35% against rhodamine B when compared to pure individual components under constant reaction conditions. This nanocomposite photocatalyst improved photodegradation of the RhB dye by increasing the surface area and active sites, and lowering bandgap energy.62 In a recent research, ZnFe2O4 NPs were conjugated with polyaniline (PAni) and AgNPs to create the Ag@ZnFe2O4-PAni nanocomposite. It was discovered that adding AgNPs to ZnFe2O4 together with PAni increases the rate of electron–hole generation when exposed to light by red shifting its band gap energy and boosting its absorption power. The Ag@ZnFe2O4-PAni material's charge transfer mechanisms were enhanced by the conjugated double bonds of PAni and the surface plasmon resonance effect of AgNPs, which encouraged charge separation during the photocatalytic process and enhanced the photodegradation of ZnFe2O4 NPs. The produced Ag@ZnFe2O4-PAni nanostructures were used to photodegrade the MB dye under simulated sunshine illumination. The results showed that ternary nanostructures have a greater photocatalytic efficiency than pure ZnFe2O4. After 60 minutes of light illumination, Ag@ZnFe2O4-PAni plasmonic nanostructures decomposed 99.6% of MB dye.63 Some examples of nanocomposites exhibiting dye degradation are discussed in Table 4. A general mechanism of dye degradation is given in Fig. 11. The reactions of the photocatalytic degradation pathway64 are listed below as eqn (9)–(16).
 |
| | Fig. 11 Probable mechanism of Brilliant Blue FCF dye and glyphosate pesticide degradation. | |
 |
| | Fig. 12 Calculated conduction band (CB) and valence band (VB) positions of Yb2O3, ZnFe2O4, and the Yb2O3/ZnFe2O4@PEG composite, illustrating the Type-II band alignment and the charge-transfer pathway. | |
 |
| | Fig. 13 Time-dependent UV-Vis spectra of Brilliant Blue dye treated with (a) Yb2O3 NPs, (b) ZnFe2O4 NPs, and (c) Yb2O3/ZnFe2O@PEG. | |
 |
| | Fig. 14 % degradation of Brilliant Blue FCF dye treated with Yb2O3 NPs, ZnFe2O4 NPs and Yb2O3/ZnFe2O4@PEG. | |
 |
| | Fig. 15 Pseudo-first-order kinetic plot of ln(C0/Ct) vs. time of (a) Yb2O3 NPs, (b) ZnFe2O4 NPs, and (c) Yb2O3/ZnFe2O4@PEG. | |
 |
| | Fig. 16 Structure of Brilliant Blue FCF dye. | |
Table 4 Some examples of nanocomposites exhibiting dye degradation
| S. no. |
Nanocomposite |
Dye |
Time (minutes) |
Source |
% degradation |
Ref. |
| 1 |
ZnO/g-C3N4 |
MB and acid blue 113 |
90 and 120 |
Visible light |
97 and 83 |
67
|
| 2 |
Gd2O3@CA, CdS/Gd2O3@CA, and CdS/Gd2O3/GO@CA |
MB |
60 |
Visible light |
41.02, 54.71, and 71.42 |
68
|
| 3 |
CuO/ZnFe2O4 |
Congo Red |
120 |
Visible light |
91.13 |
69
|
| 4 |
g-C3N4/Zn2V2O7 |
RB and MB |
180 |
Visible light |
94 for RB and 97 for MB |
70
|
| 5 |
g-C3N4/Nb2O5/Ag |
MB |
80 |
Visible light |
92 |
71
|
| 6 |
Ag/ZnO/S-g-C3N4 |
MB |
60 |
Sunlight |
98 |
72
|
| 7 |
Cu2O/α-Fe2O3 |
Congo Red |
60 |
Mercury lamp |
95.57 |
73
|
| 8 |
ZnO-ZnFe2O4 |
Congo Red |
180 |
Solar light |
97.3 |
74
|
| 9 |
Co0.5Cu0.25Zn0.25YxFe2−xO4 |
MB |
60 |
|
95 |
75
|
| 10 |
Zinc–copper–chromium (ZCCF) spinel nanoferrites |
MB |
120 |
Visible light |
96.93 |
76
|
Activation:
| | | Catalyst + hν → e− + h+ | (9) |
ROS generation:
| | | HO2˙ + HO2˙ → H2O2 + O2 | (13) |
| | | e− + H2O2 → ˙OH + OH− (or H2O2 + hν → 2 ˙OH) | (14) |
Brilliant Blue FCF degradation:
| |  | (15) |
Mineralization:
| |  | (16) |
The photocatalyst experiences photoexcitation when exposed to light, producing holes in the valence band and electrons in the conduction band. While the electrons convert dissolved oxygen to superoxide radicals (˙O2−), the photogenerated holes react with hydroxide ions or surface-adsorbed water molecules to produce highly oxidative hydroxyl radicals (˙OH). These reactive oxygen species also take part in a sequence of events that produce more ˙OH radicals and hydrogen peroxide. These reactive species and photogenerated holes attack the Brilliant Blue dye molecules adsorbed on the catalyst surface, causing the chromophoric groups responsible for colour to cleave. Effective dye mineralization is demonstrated by the continued oxidation of the broken intermediates into carbon dioxide, water, and inorganic ions.65
The proposed charge-transfer mechanism (Fig. 11) for Brilliant Blue FCF degradation is derived from the experimentally measured band gaps (Fig. 12) of Yb2O3 (4.91 eV), ZnFe2O4 (4.61 eV), and the Yb2O3/ZnFe2O4@PEG composite (3.99 eV), together with the band-edge positions calculated using electronegativity theory. The calculated conduction and valence band potentials were −1.41 eV/+3.51 eV for Yb2O3, −0.91 eV/+3.71 eV for ZnFe2O4, and −0.75 eV/+3.25 eV for the composite. These values confirm the formation of a staggered Type-II heterojunction between Yb2O3 and ZnFe2O4. In this configuration, photogenerated electrons migrate from the CB of ZnFe2O4 (−0.91 eV) to the more negative CB of Yb2O3 (−1.41 eV), while holes move from the VB of Yb2O3 (+3.51 eV) to the VB of ZnFe2O4 (+3.71 eV). This directional carrier separation suppresses recombination and increases the generation of reactive oxygen species (˙OH and ˙O2−). The enhanced photocatalytic performance of the composite – 86.38% degradation and a rate constant of 2.09 × 10−2 min−1, compared to 83.09% (1.602 × 10−2 min−1) for Yb2O3 and 77% (1.577 × 10−2 min−1) for ZnFe2O4 – corroborates this mechanism. The improved kinetics indicates that the heterojunction facilitates faster charge transfer and prolongs charge-carrier lifetimes, explaining the superior degradation efficiency of the Yb2O3/ZnFe2O4@PEG nanocomposite.66
4.6 Photocatalytic pesticide degradation
Glyphosate [N-(phosphonomethyl)glycine] (Fig. 17), a systemic herbicide that is broad-spectrum and non-selective, has been widely employed in agriculture since the 1970s.77 Because of its great efficacy and compatibility with genetically modified glyphosate-resistant crops, it has become one of the most widely used herbicides globally. However, there are serious environmental and health risks associated with glyphosate and its main metabolite, aminomethylphosphonic acid (AMPA), which are frequently found in soil, surface water, groundwater, and even food products. While glyphosate is a widely used herbicide, its persistence and toxic implications need the development of effective remediation solutions, including photocatalysis employing advanced nanomaterials, which offer a sustainable and economical alternative.78 In the present study, the photocatalytic degradation of glyphosate was studied using Yb2O3 NPs, ZnFe2O4 NPs and their binary nanocomposite (Fig. 18(a, b and c)). The photocatalytic pesticide degradation with Yb2O3 NPs followed pseudo-first order kinetics and was found to be 71.54% (Fig. 19), with a rate constant of 1.328 × 10−2 min−1 (R2 = 0.99047) as given in Fig. 20(a), while with ZnFe2O4 NPs, photodegradation was found to be 55% and also followed pseudo-first order kinetics with a rate constant of 8.63 × 10−3 min−1 (R2 = 0.99305) as shown in Fig. 20(b). Similarly, the photodegradation of the Yb2O3/ZnFe2O4@PEG nanocomposite followed pseudo-first order kinetics and was found to be 91.28% and the rate constant was found to be 2.49 × 10−2 min−1 (R2 = 0.94784) (Fig. 20(c)). This enhanced performance can be attributed to several synergistic factors arising from heterojunction formation between the two semiconductor type NPs. The involvement of ytterbium's 4f orbital acts as an electron trap, minimizing electron–hole recombination. Zinc ferrite, a spinel semiconductor, improves visible light absorption and charge mobility. Their combination forms a heterojunction, which improves charge separation and carrier lifetimes, increasing photocatalytic efficiency. Ultrasonication increases homogeneous dispersion and close interaction among NPs. It increases the surface area and active sites, which improves interaction with glyphosate molecules. The composite structure increases chemical stability. The binary system speeds up the creation of reactive oxygen species, which are essential for breaking down glyphosate.79 The binary nanocomposite's better glyphosate degradation ability may be due to its optimized electronic structure, increased light absorption, and shape caused by ultrasonication. These properties work together to promote photocatalytic activity, making it a good candidate for long-term pesticide cleanup. Previous research has found that various nanocomposites improve photocatalytic degradation efficiency in similar ways. For example, a recent work described the synthesis of BiOBr/Cu2O heterojunction photocatalysts. The improved nanocomposite completely degraded glyphosate under 10 minutes of light irradiation and maintained good stability after five photocatalytic cycles. The increased activity is ascribed to the high surface area, distinctive shape, and S-scheme heterojunction of the composite material, which facilitates the separation of charge carriers and boosts the redox potential of electrons and holes.80 Another work discussed the production of NiO–ZnO photocatalysts. The degradation of the Bentazon herbicide under UV light was used to investigate the NiO–ZnO nanocomposite's photocatalytic activity. The degradation efficiency of the photocatalyst was determined to be 70% after 100 minutes of exposure to a UV light source. The light absorption range of the nanocomposite is from UV to natural light because it has a lower band gap energy (2.6 eV) than the pure NiO and ZiO components.81 Similarly, a study reported the formation of a ZnO/rGO composite, which was then employed for the photocatalytic degradation of dimethoate. The ZnO/rGO nanocomposite photodegraded dimethoate at a rate and efficiency that were 4 and 1.5 times higher than that of bare ZnO, respectively, and the nanocomposite had a large surface area. The enhanced photocatalytic activity was linked to the presence of rGO's effective carrier transport and reduction in the electron–hole recombination rate.82 Some other examples of nanocomposites exhibiting pesticide degradation are given in Table 5. The reactions generally involved in the photocatalytic degradation pathway83 are given as eqn (17)–(34).
 |
| | Fig. 17 Structure of glyphosate pesticide. | |
 |
| | Fig. 18 Time-dependent UV-Vis spectra of glyphosate pesticide treated with (a) Yb2O3 NPs, (b) ZnFe2O4 NPs, and (c) Yb2O3/ZnFe2O4@PEG. | |
 |
| | Fig. 19 % degradation of the pesticide via Yb2O3 NPs, ZnFe2O4 NPs and Yb2O3/ZnFe2O4@PEG. | |
 |
| | Fig. 20 Pseudo-first-order kinetic plot of ln(C0/Ct) vs. time of (a) Yb2O3 NPs, (b) ZnFe2O4 NPs, and (c) Yb2O3/ZnFe2O4@PEG. | |
Table 5 Some examples of nanocomposites exhibiting pesticide degradation
| S. no. |
Composite |
Pesticide |
Time (minutes) |
Source |
% degradation |
Ref. |
| 1 |
TaSe2/g-C3N4 |
Terbufos |
90 |
Visible light |
98 |
84
|
| 2 |
GO/ZnO |
Chlorpyrifos |
90 |
Visible light |
93.58 |
85
|
| 3 |
CuO–ZnO |
Profenofos |
80 |
Visible light |
100 |
86
|
| 4 |
GO–TiO2 |
Dichlorvos and malathion |
90 |
UV light |
80 and 90 |
87
|
| 5 |
ZnO/WO2.72 |
Dichlorvos |
80 |
Visible light |
86.6 |
88
|
Activation:
| | | Catalyst + hν → e− + h+ | (17) |
ROS generation:
| | | H2O2 + e− → ˙OH + OH− | (23) |
Pathway 1: C–P bond cleavage:
| | 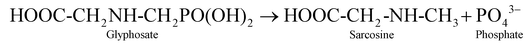 | (24) |
| |  | (25) |
| |  | (26) |
| |  | (27) |
Pathway 2: C–N bond cleavage:
| |  | (28) |
| |  | (29) |
| |  | (30) |
Pathway 3: Direct C–N bond cleavage:
| |  | (31) |
| |  | (32) |
| |  | (33) |
| | | HOOC-CHO + ˙OH → CO2 + H2O | (34) |
4.7 Recyclability of the nanocomposite
The recyclability of the nanocomposite Yb2O3/ZnFe2O4@PEG was evaluated for both Brilliant Blue dye and glyphosate pesticide degradation. For dye degradation, the fresh NPs exhibited 86.38% efficiency, while after the first cycle, the activity slightly decreased to 86.01% (Fig. 21(a)). Similarly, in pesticide degradation, the fresh nanoparticles achieved 91.28% efficiency, which reduced marginally to 91.00% in the first cycle (Fig. 21(b)). These results indicate that the nanocomposite retains excellent recyclability and stability, as the degradation efficiency remains nearly unchanged after reuse. The minor decrease (<0.5%) demonstrates that the nanocomposite can be effectively reused without significant loss of catalytic activity. The recyclability performance of the Yb2O3/ZnFe2O4@PEG nanocomposite was evaluated after successive cycles of dye and pesticide degradation. The catalyst retained a high percentage of activity in the second cycle, with only a slight decrease in degradation efficiency. This indicates that the material possesses strong durability and stability under repeated use. This is further supported by the post-recyclability XRD graphs (Fig. 22) and the fact that the characteristic peaks of the composite remained intact, suggesting that the structural framework of the catalyst was not altered during photocatalytic reactions. The stability of peaks also demonstrates that no significant phase transformation occurred during recycling. Both dye and pesticide degradation experiments confirmed comparable recovery values, emphasizing the robustness of the nanocomposite. The synergistic effect of Yb2O3 and ZnFe2O4 within the PEG matrix appears to prevent catalyst deactivation. Thus, the nanocomposite not only exhibits excellent initial activity but also maintains its efficiency after multiple cycles. These results highlight its strong potential for sustainable and practical wastewater treatment applications.
 |
| | Fig. 21 Recyclability graphs of Yb2O3/ZnFe2O4@PEG with (a) Brilliant Blue FCF and (b) glyphosate. | |
 |
| | Fig. 22 XRD spectra of the recovered Yb2O3/ZnFe2O4@PEG after the second photocatalytic cycle. | |
4.8 Electrochemical sensing
Catechol (1,2-dihydroxybenzene) is an important dihydroxybenzene derivative widely used in the chemical, pharmaceutical, and agricultural industries. However, due to its high toxicity, poor biodegradability, and potential to cause harmful effects on human health and the environment, its detection and quantification in various samples have become a critical area of research. Even at low concentrations, catechol can interfere with biological processes and contaminate water sources, making its monitoring essential.89 In this study, a Yb2O3/ZnFe2O4@PEG nanocomposite synthesized via ultrasonication is explored as an efficient sensing material for the electrochemical detection of catechol. The electrochemical performance of the Yb2O3/ZnFe2O4@PEG modified electrode towards catechol was systematically investigated. Cyclic voltammetry revealed that the bare GCE produced negligible current response, whereas the nanocomposite-modified electrode displayed pronounced redox peaks in the presence of catechol (Fig. 23), confirming its strong electrocatalytic activity. Scan rate-dependent CVs (10–100 mV s−1) (Fig. 24(a)) showed that both anodic and cathodic peak currents increased proportionally with the scan rate, and the linear relationship between the peak current and the square root of scan rate (R2 = 0.9875) indicated a diffusion-controlled process (Fig. 24(b)). Concentration-dependent linear sweep voltammetry (40–100 µM catechol) (Fig. 25(a)) further demonstrated a steady rise in the anodic peak current with increasing analyte concentration, yielding a calibration slope of 0.0609 µA µM−1 and excellent linearity (R2 = 0.9865). Based on this slope and the electroactive surface area (0.166 cm2), the sensitivity was calculated as 0.367 µA µM−1 cm−2. With a background noise of approximately 0.234 µA, the limit of detection, which was calculated by dividing the slope by three times the blank's standard deviation, was 11.52 µM (Fig. 25(b)). These findings confirm that Yb2O3/ZnFe2O4@PEG provides a stable and diffusion-driven electron transfer platform with good analytical sensitivity for catechol detection in neutral phosphate buffer.
 |
| | Fig. 23 CV curves of (a) the bare GCE with, (b) Yb2O3/ZnFe2O4@PEG without, and (c) Yb2O3/ZnFe2O4@PEG with 100 µM catechol in 0.1 M PB (pH = 7) at a fixed scan rate of 50 mV s−1. | |
 |
| | Fig. 24 (a) CV curves at different applied scan rates of Yb2O3/ZnFe2O4@PEG for 100 µM catechol in 0.1 M PB (pH = 7) and (b) calibration plot between the electrooxidation current value and the square root of the scan rate. | |
 |
| | Fig. 25 (a) LSV curves of Yb2O3/ZnFe2O4@PEG for different concentrations of 100 µM catechol in 0.1 M PB (pH = 7) at a fixed scan rate 50 mV s−1 and (b) calibration plot between the electrooxidation current value and the concentration of catechol. | |
Similar studies have been reported in the literature showing that recent research has demonstrated that electrocatalytic hydrogen peroxide production has advanced significantly, offering a cleaner and more energy-efficient alternative to the anthraquinone process. Improved catalyst systems including carbon materials, metal-based catalysts, and macrocyclic complexes have shown higher H2O2 selectivity and efficiency. Progress in understanding ORR pathways and optimizing operating conditions has further strengthened catalyst performance, while new reactor designs have enhanced stability and production rates. Remaining challenges include reducing catalyst cost and improving long-term durability, but overall findings indicate that electrocatalysis is becoming a promising and sustainable route for future large-scale H2O2 generation.90 In another study, a nanozyme-based sensing material was synthesized to develop a colorimetric sensor array capable of intelligently identifying multiple pesticide residues. The material consists of three Cu–amino acid nanozymes (Cu-Leu, Cu-Ile, and Cu-Phe), whose laccase-like activity can be finely adjusted through controlled coordination between copper ions and different amino acids. Their simple and cost-effective synthesis improves activity regulation and overall sensor reliability. When paired with 2,4-dichlorophenol and 4-aminoantipyrine, the nanozymes produce distinct colorimetric responses that enable clear discrimination of seven pesticides using LDA and HCA. The platform demonstrates excellent sensitivity, achieving detection limits below 0.0012 μM. By integrating the YOLOv8 deep learning model to automatically interpret the LDA patterns, the system reaches a validation mAP of 0.99 and a testing confidence of 0.93, significantly accelerating detection and enhancing accuracy for real-world pesticide monitoring.91
To assess the analytical significance of the developed sensor, the obtained limit of detection (LOD) was compared with values reported for recently developed catechol sensors. Although the LOD achieved in this work is moderate relative to highly conductive graphene-based systems, it is still comparable to many metal–oxide-based sensors reported in the literature, such as ZnFe2O4-modified electrodes. The moderately high LOD can be attributed to the absence of conductive carbon supports, which typically enhance electron transport and signal amplification. Incorporating materials such as rGO or CNTs in future designs may further lower the LOD and enhance overall performance.
4.9 Reproducibility, repeatability and stability
The reproducibility and repeatability are important factors for any sensing device. The reproducibility of the fabricated sensor was examined using 3 different GCEs with Yb2O3/ZnFe2O4@PEG under similar conditions; the obtained LSV responses are summarised in Fig. 26. The result obtained depicts good reproducibility of the fabricated sensor with an RSD value of 3%. The repeatability of the Yb2O3/ZnFe2O4@PEG sensor was examined by taking 5 independent LSV responses and the results obtained show decent repeatability with an RSD value of 7.5%. The long-term stability is an essential parameter for any sensing material. The stability was examined (Fig. 27) using 30 cycles of cv and the current shows 80% of the initial current response signifying good stability of the Yb2O3/ZnFe2O4@PEG nanocomposite.
 |
| | Fig. 26 Scan rate of the Yb2O3/ZnFe2O4@PEG GCE at 50 mV s−1 for (a) reproducibility and (b) repeatability for catechol. | |
 |
| | Fig. 27 Cycling stability of the Yb2O3/ZnFe2O4@PEG GCE for 100 µM catechol at a scan rate of 50 mV s−1 for 30 cycles. | |
For any sensor, to achieve high selectivity is a really challenging task, but it is desirable for its practical utility. The selectivity of the Yb2O3/ZnFe2O4@PEG sensor was tested by first running LSV of 100 µM catechol only at a scan rate of 50 mV s−1. Then a mixture of 100 µM catechol with common interferents such as resorcinol, hydroquinone, and p-nitrophenol at a concentration of 1000 µM was tested and the obtained LSV response (Fig. 28) showed negligible interference in the current response, signifying the high selectivity of the fabricated sensor.
 |
| | Fig. 28 LSV response of the Yb2O3/ZnFe2O4@PEG GCE for 100 µM catechol and 100 µM catechol + 1000 µM interfering compounds at a scan rate of 50 mV s−1. | |
Real sample analysis was also carried out using the LSV technique. The standard addition method was used to investigate real samples. The real sample was taken from Gomti river. First of all, the river water was filtered using Whatman cellulose filter paper to discard unwanted impurities. After that, different amounts of catechol (0 µM, 50 µM, and 100 µM) were spiked into a real water system and their LSV response was investigated. The results are shown in Table 6. The fabricated Yb2O3/ZnFe2O4@PEG sensor exhibited good real sample response. The findings are also compared with some previously reported nanocomposites for catechol sensing as given in Table 7.
Table 6 Real sample analysis
| River water |
Added (µM) |
Found (µM) |
Recovery (%) |
| Catechol |
0 |
0 |
0 |
|
|
50 |
52.28 |
104.26 |
|
|
100 |
96.86 |
96.86 |
Table 7 Some studies for catechol sensing nanocomposites
| S. no. |
Nanocomposite/electrode |
Target analyte(s) |
Detection technique |
Linear range |
Limit of detection (LOD) |
Results |
Ref. |
| 1 |
Graphene oxide and 5,10,15-tri(4-tert-butylphenyl)-20-(4-phenol)-porphyrin (GO–Por) modified electrode |
Catechol |
CV/DPV |
5–250 µM |
0.063 µM |
GO–Por composite enhanced catechol oxidation peak; excellent sensitivity. |
92
|
| 2 |
Graphene oxide–PAMAM (G3) + ionic liquid carbon paste electrode |
Catechol & resorcinol |
CV/DPV |
1–600 µM |
0.47 µM |
Enabled simultaneous detection with well-separated peaks; high stability due to GO–PAMAM synergy. |
93
|
| 3 |
MOF-derived CuO/NiO nanohybrids |
Catechol (in tea samples) |
DPV |
0.5–700 µM |
0.12 µM |
Metal–oxide nanohybrids showed strong electrocatalytic activity and excellent real-sample performance. |
94
|
| 4 |
Co@SnO2-polyaniline (Co@SnO2-PANI) composite |
Catechol & hydroquinone |
DPV |
1–300 µM |
0.32 µM |
Provided well-resolved peaks, enabling simultaneous determination in real water samples. |
95
|
| 5 |
PEDOT/CNT–graphene oxide composite |
Hydroquinone, catechol & nitrite |
DPV |
0.001–500 µM |
3.8 nM |
Ultra-low LOD and excellent stability; effective in wastewater testing. |
96
|
| 6 |
Electro-reduced graphene oxide (ERGO)-modified MWCNT paste electrode |
Catechol & hydroquinone |
CV/DPV |
0.4–400 µM |
0.083 µM |
ERGO–MWCNTPE enabled well-resolved peaks for CC & HQ; good sensitivity; validated in tap water and urine. |
97
|
| 7 |
Ultrafine-layer CoP–NiCoP heterostructure on graphene |
Catechol & hydroquinone |
CV/DPV |
0.5–300 µM (typical reported) |
Low-µM to sub-µM |
CoP–NiCoP/graphene showed strong electrocatalytic oxidation and stable simultaneous detection in real river water. |
98
|
| 8 |
Yb2O3/ZnFe2O4@PEG-modified GCE |
Catechol |
CV, LSV |
40–100 µM |
11.52 µM |
Modified electrode shows strong redox peaks |
Present study |
4.10 Mechanism of detection of catechol
Catechol is detected electrochemically at the Yb2O3/ZnFe2O4@PEG modified GCE by a reversible redox process that involves oxidizing catechol to o-benzoquinone and then reducing it back to catechol. When a potential is applied, catechol experiences a two-electron, two-proton oxidation according to eqn (35).| | 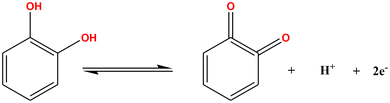 | (35) |
At the electrode–electrolyte interface, the Yb2O3/ZnFe2O4@PEG nanocomposite exhibits a high density of electroactive sites and speeds up electron transfer. In comparison with the bare GCE, the synergistic interaction between Yb2O3 and ZnFe2O4 NPs reduces the activation energy for catechol oxidation, leading to increased anodic and cathodic peak currents. The increased surface area, faster charge transfer kinetics, and improved stability of the redox species at the modified electrode surface are the main factors responsible for enhanced sensing performance. Similar redox behavior and electrocatalytic increase have been documented for metal–oxide-based nanocomposite modified electrodes, where synergistic interactions of the oxide component greatly boost electron transfer kinetics and sensing performance.99 The presence of mixed metal oxides, which offer a large number of electroactive sites and speed up electron transport, is responsible for the increased anodic and cathodic peak currents seen at the modified electrode. For multi-metal oxide composite modified GCEs, comparable gains in charge transfer resistance, diffusion-controlled kinetics, and two-electron–two-proton catechol oxidation have been documented.100
5. Electrochemical impedance spectroscopy (EIS)
One essential tool for differentiating between modified and unmodified electrodes in terms of impedance or conductivity of redox processes is EIS. It also makes the kinetics of charge transfer easier to comprehend.100 EIS was used to examine the interfacial charge transfer characteristics of the Yb2O3/ZnFe2O4@PEG modified GCE. The measurements were conducted in 5 mM potassium ferrocyanide [Fe(CN)6]3− with 0.1 M KCl as the supporting electrolyte across a frequency range of 100 kHz to 1 Hz, at an applied potential of 0.24 V. The Nyquist plot of the Yb2O3/ZnFe2O4@PEG GCE displays a clear semicircle in the high-frequency region, followed by a linear segment in the low-frequency region. The semicircle represents the charge transfer resistance (Rct) at the interface of the electrode and electrolyte, while the linear section relates to Warburg impedance, signifying diffusion-controlled transport of electrons. The modest semicircle diameter indicates minimal charge transfer resistance, affirming effective interfacial electron transfer kinetics at the Yb2O3/ZnFe2O4@PEG GCE interface. This behavior can be attributed to the synergistic interplay between Yb2O3 and ZnFe2O4, which promotes improved electrical conductivity and efficient charge transport at the electrode interface. The enhanced charge transfer properties seen in the EIS analysis confirm the outstanding electrochemical performance of the Yb2O3/ZnFe2O4@PEG composite, positioning it as a potential electrode material for electrochemical sensing applications, such as catechol detection.101,102 Furthermore, the outcome demonstrates that the Yb2O3/ZnFe2O4@PEG GCE exhibits enhanced electronic conductivity and expedites the kinetics of electron transfer (Fig. 29).
 |
| | Fig. 29 Nyquist plot of Yb2O3/ZnFe2O4@GCE in 5 mM potassium ferrocyanide in 0.1 M KCl. Low frequency – 1 Hz. | |
6. Conclusion
Yb2O3 and ZnFe2O4 NPs were produced by co-precipitation, and a PEG-supported binary nanocomposite was created through ultrasonication. Structural and morphological analysis validated the materials’ crystalline character and uniform surface features, while optical studies demonstrated their potential for photocatalytic use. The photocatalytic degradation studies revealed significant differences in performance. For the Brilliant Blue FCF dye, the degradation efficiencies were 76.42% with Yb2O3 NPs and 77% with ZnFe2O4 NPs, whereas it was 86.38% with the binary nanocomposite. For glyphosate pesticide, the degradation values were 71.54%, 55%, and 91.28%. The findings show that the Yb2O3/ZnFe2O4@PEG nanocomposite outperforms the individual NPs. The superior activity is ascribed to the synergistic interaction between Yb2O3 and ZnFe2O4, improved charge carrier separation, and the stabilizing/dispersing effect of PEG. Overall, this study demonstrates the utility of binary nanocomposites as efficient, reusable, and environmentally acceptable photocatalysts for the degradation of toxic dyes and pesticides, thereby contributing to effective environmental remediation efforts. Also, the sensor exhibited excellent sensitivity, a wide linear detection range, a low limit of detection, and good selectivity towards catechol, even in the presence of common interfering substances. The improved sensing performance can be attributed to the synergistic effect between Yb2O3 and ZnFe2O4 NPs, which enhances electron transfer kinetics and provides more active sites for catechol oxidation. Additionally, the ultrasonication method proved to be a simple, eco-friendly, and efficient approach for fabricating high-performance nanocomposites for sensing applications. During ultrasonication-assisted synthesis, PEG serves as an essential steric stabilizer, enclosing all the components of the nanocomposite in a polymeric shell that prevents aggregation and supports the binary nanocomposite's long-term structural integrity.
Author contributions
Nashra Fatima – investigation, formal analysis, interpretation, and writing – original and final draft; Sristhi Dwivedi – characterization; Pratibha Bansal – electrochemical sensing; Ekhlakh Veg – analysis, interpretation and writing; Vishal Mishra – electrochemical sensing; Tahmeena Khan – conceptualization, supervision, and writing – final draft and proof-reading.
Conflicts of interest
There are no conflicts to declare.
Data availability
All the data have been included in the paper.
Acknowledgements
The authors acknowledge the R&D cell of Integral University, Lucknow, for providing the Manuscript Communication Number (IU/R&D/2025-MCN0003919). Dr Tahmeena Khan extends gratitude to Integral University for the support extended through the Outstanding Researcher Award for the year 2024, and Dr Pratibha Bansal acknowledges the support received through SERB (SUR/2022/004896) for providing an electrochemical workstation used for sensing catechol. The authors also acknowledge the supported extended by the Central Instrumentation Facility (CIF), Integral University, Lucknow.
References
- K. S. I. Khan and I. Khan, Arabian J. Chem., 2019, 12, 908–931 CrossRef.
- E. Omanović-Mikličanin, A. Badnjević, A. Kazlagić and M. Hajlovac, Health Technol., 2020, 10, 51–59 CrossRef.
-
R. M. Wang, S. R. Zheng and Y. P. Zheng, Polymer Matrix Composites and Technology, Woodhead Publishing Limited and Science Press Limited, 2011 Search PubMed.
- J. Zia and M. S. S. R. Tejaswini, RSC Adv., 2025, 15, 9055–9080 RSC.
- T. Hassan, A. Salam, A. Khan, S. U. Khan, H. Khanzada, M. Wasim and I. S. Kim, J. Polym. Res., 2021, 28, 36 CrossRef CAS.
- T. Naseem and M. Waseem, Int. J. Environ. Sci. Technol., 2022, 19, 2221–2246 CrossRef CAS.
- S. Yadav, N. Rani and K. Saini, IOP Conf. Ser.: Mater. Sci. Eng., 2022, 1225, 012004 CrossRef CAS.
- E. Veg, A. Raza, P. Bansal, S. Rai, S. Sharma, S. Dwivedi and T. Khan, Surf. Interfaces, 2025, 47, 107271 CrossRef.
- E. Veg, A. Raza, P. Bansal, S. Rai, S. Sharma, R. Gupta, S. Dwivedi, S. Joshi, A. R. Khan and T. Khan, Inorg. Chem. Commun., 2025, 160, 115265 CrossRef.
- J. M. Chaba and P. N. Nomngongo, Emerging Contam., 2019, 5, 143–149 CrossRef.
- S. Badkoobeh Hezaveh, M. T. Ranjbar and B. Nabavi, Colloid Nanosci. J., 2024, 2, 228–237 CrossRef.
- E. E. Elemike, D. C. Onwudiwe, L. Wei, C. L. Chaogang and Z. Zhiwei, Sol. Energy Mater. Sol. Cells, 2019, 201, 110106 CrossRef CAS.
- S. Shaw, R. Mondal, P. Dam, A. Mandal, R. Acharya, S. Manna and A. K. Mandal, RSC Adv., 2024, 14, 33068–33079 RSC.
- R. Kiran, N. Kamath, M. I. Sayyed, A. H. Almuqrin and S. D. Kamath, RSC Adv., 2025, 15, 20040–20060 RSC.
- S. Jabeen, A. S. Ganie, N. Ahmad, S. Hijazi, S. Bala, D. Bano and T. Khan, Inorg. Chem. Commun., 2023, 152, 110729 CrossRef CAS.
- N. Farooq, R. Luque, M. M. Hessien, A. M. Qureshi, F. Sahiba, M. A. Nazir and A. Rehman, Appl. Sci., 2021, 11, 9000 CrossRef CAS.
- T. Saravanan, P. Anandan, M. Shanmugam, M. Azhagurajan, M. Mohamed Ismail, M. Arivanandhan and R. Jayavel, Polym. Bull., 2020, 77, 3891–3906 CrossRef CAS.
- M. Bhattu, R. Acevedo and A. H. Shnain, E3S Web Conf., 2024, 588, 02014 CrossRef CAS.
- S. Sharma, V. Dutta, P. Raizada, A. Hosseini-Bandegharaei, V. Thakur, V. H. Nguyen and P. Singh, J. Environ. Chem. Eng., 2021, 9, 105812 CrossRef.
- S. P. Keerthana, R. Yuvakkumar, P. S. Kumar, G. Ravi and D. Velauthapillai, Environ. Res., 2021, 197, 111047 CrossRef CAS PubMed.
- S. Jabeen, V. U. Siddiqui, S. Sharma, S. Rai, P. Bansal, S. Bala, A. Raza, M. I. Ahmad, A. R. Khan and T. Khan, J. Alloys Compd., 2024, 984, 174020 CrossRef CAS.
- S. Roostaei, H. Ansarinejad, E. A. Dawi, F. H. Alsultany, S. K. Issa, M. Ahmadi-Kashani and M. Salavati-Niasari, Appl. Water Sci., 2025, 15, 1–19 CrossRef.
- S. Jabeen, V. U. Siddiqui, S. Rastogi, S. Srivastava, S. Bala, N. Ahmad and T. Khan, Mater. Today Chem., 2023, 33, 101712 CrossRef CAS.
- A. Nawab, A. Aleem and A. Malik, Bioresour. Technol., 2003, 88, 41–46 CrossRef CAS PubMed.
- H. Tian, J. Li, Z. Mu, L. Li and Z. Hao, Sep. Purif. Technol., 2009, 66, 84–89 CrossRef CAS.
- R. Boussahel, D. Harik, M. Mammar and S. Lamara-Mohamed, Desalination, 2006, 369–372 Search PubMed.
- S. K. Gautam and S. Suresh, J. Hazard. Mater., 2007, 139, 146–153 CrossRef CAS PubMed.
- D. W. Elliott, H. L. Lien and W. X. Zhang, J. Environ. Eng., 2009, 135, 317–324 CrossRef CAS.
- J. Senthilnathan and L. Philip, Chem. Eng. J., 2010, 161, 83–92 CrossRef CAS.
- K. M. Paknikar, V. Nagpal, A. V. Pethkar and J. M. Rajwade, Sci. Technol. Adv. Mater., 2005, 6, 370–374 CrossRef CAS.
- H. Wu, Q. Sun, J. Chen, G.-Y. Wang, D. Wang, X.-F. Zeng and J.-X. Wang, Chem. Eng. J., 2021, 425, 130640 CrossRef CAS.
- F. Li, T. S. Y. Choong, L. C. Abdullah, S. N. A. Md Jamil and N. N. Amerhaider Nuar, Polymers, 2023, 15, 775 CrossRef CAS PubMed.
- M. Rodríguez Páez, Y. Ochoa-Muñoz and J. E. Rodriguez-Páez, Biocatal. Agric. Biotechnol., 2019, 22, 101434 CrossRef.
- H. Yin, Q. Zhang, Y. Zhou, Q. Ma, L. Zhu and S. Ai, Electrochim. Acta, 2011, 56, 2748–2754 CrossRef CAS.
- Y. Wang, Y. Shao, D. W. Matson, J. Li and Y. Lin, ACS Nano, 2010, 4, 1790–1798 CrossRef CAS PubMed.
- Y. Zhao, X. Song, Q. Song and Z. Yin, CrystEngComm, 2012, 14, 6710–6719 RSC.
- L. Zhao, J. Yu, S. Yue, L. Zhang, Z. Wang, P. Guo and Q. Liu, J. Electroanal. Chem., 2018, 808, 245–251 CrossRef CAS.
- Y. Wu, W. Lei, M. Xia, F. Wang, C. Li, C. Zhang, Q. Hao and Y. Zhang, Appl. Clay Sci., 2018, 162, 38–45 CrossRef CAS.
- A. Karthika, P. Karuppasamy, M. K. Shetty and M. Subramaniam, Next Res., 2025, 100884 CrossRef.
- A. Karthika, V. R. Raja, P. Karuppasamy, A. Suganthi and M. Rajarajan, Microchem. J., 2019, 145, 737–744 CrossRef CAS.
- M. Madadelahi, F. O. Romero-Soto, R. Kumar, U. B. Tlaxcala and M. J. Madou, Biosens. Bioelectron., 2025, 117099 CrossRef CAS PubMed.
- C. O. Ehi-Eromosele, B. I. Ita and E. E. J. Iweala, Dig. J. Nanomater. Biostruct., 2016, 11, 7–14 Search PubMed.
- M. Rahimi-Nasrabadi, S. M. Pourmortazavi, M. Aghazadeh, M. R. Ganjali, M. S. Karimi and P. Norouzi, J. Mater. Sci.: Mater. Electron., 2017, 28, 9478–9488 CrossRef CAS.
- F. Hoseini, M. Farahamndjou and T. P. Firoozabadi, J. Fundam. Appl. Sci., 2016, 8, 738–745 CrossRef CAS.
- F. Khoerunnisa, M. Nurhayati, H. Herlini, Q. A. A. Adzkia, F. Dara, H. Hendrawan and J. Lim, J. Water Process Eng., 2023, 52, 103556 CrossRef.
- S. Kumar, G. R. Chaudhary and S. Chaudhary, J. Mol. Liq., 2022, 355, 118929 CrossRef CAS.
- A. Sarilmaz, Turk. J. Chem., 2022, 46, 1694–1701 CrossRef CAS PubMed.
- T. Sarkar, S. Kundu, G. Ghorai, P. K. Sahoo, V. R. Reddy and A. Bhattacharjee, Appl. Phys. A, 2025, 131, 1–24 CrossRef.
- M. Brogly, S. Bistac and D. Bindel, Polymers, 2024, 16, 1244 CrossRef CAS PubMed.
- M. Rahimi-Nasrabadi, M. Rostami, F. Ahmadi, A. F. Shojaie and M. D. Rafiee, J. Mater. Sci.: Mater. Electron., 2016, 27, 11940–11945 CrossRef CAS.
- K. Hatami Kahkesh, Z. Baghbantaraghdari, D. Jamaledin, F. Dabbagh Moghaddam, N. Kaneko and M. Ghovvati, Mater. Chem. Horiz., 2023, 2, 49–56 Search PubMed.
- S. Nasiri, M. Rabiei, A. Palevicius, G. Janusas, A. Vilkauskas, V. Nutalapati and A. Monshi, NanoTrends, 2023, 3, 100015 Search PubMed.
- S. Husain, M. Yusup, N. H. Haryanti, M. Saukani, S. Arjo and A. Riyanto, IOP Conf. Ser.: Earth Environ. Sci., 2021, 758, 012001 Search PubMed.
- F. Unal and K. Kazmanli, Mater. Sci. Forum, 2021, 1034, 117–122 Search PubMed.
- W. A. Sami and Z. S. Sadeq, J. Phys.: Conf. Ser., 2021, 1999, 012060 CrossRef CAS.
- S. Patar, B. K. Bhuyan, M. Konwar, B. Mahanta, P. Saikia, A. K. Guha and L. J. Borthakur, ChemistrySelect, 2022, 7, e202201936 CrossRef CAS.
- D. Mouattah, S. Belhadj, M. B. Taouti and D. Benbertal, J. Nanostruct., 2021, 11, 377–387 CAS.
- T. Liu, J. Liu, Q. Liu, L. Rumin, H. Zhang, X. Jing and J. Wang, Sens. Actuators, B, 2017, 250, 111–120 CrossRef CAS.
- R. Jia, K. Zhang, S. Kang and C. Wang, J. Alloys Compd., 2025, 183416 CrossRef CAS.
- A. T. Bişgin, J. AOAC Int., 2020, 103, 1478–1485 CrossRef PubMed.
- A. Sidorowicz, G. Fais and F. Desogus,
et al.
, Environ. Sci. Pollut. Res., 2024, 31, 57765–57777 CrossRef CAS.
- A. Ahmed, R. Alabada, M. Usman, A. A. Alothman, M. K. Tufail, S. Mohammad and Z. Ahmad, Diamond Relat. Mater., 2024, 141, 110630 CrossRef CAS.
- S. S. Jaafar, R. I. Faeq, A. M. Naji, O. A. Nief and M. K. Mohammed, RSC Adv., 2024, 14, 26066–26076 RSC.
- A. S. Al-Shehri, Z. Zaheer, A. M. Alsudairi and S. A. Kosa, ACS Omega, 2021, 6, 27510–27526 CrossRef CAS PubMed.
- P. Karuppasamy, N. R. N. Nisha, A. Pugazhendhi, S. Kandasamy and S. Pitchaimuthu, J. Environ. Chem. Eng., 2021, 9, 105254 CrossRef CAS.
- B. Zhao, N. Shao, X. Chen, J. Ma, Y. Gao and X. Chen, Colloids Surf., A, 2023, 663, 131072 CrossRef CAS.
- M. Gayathri, M. Sakar and E. Satheeshkumar,
et al.
, J. Mater. Sci.: Mater. Electron., 2022, 33, 9347–9357 CrossRef CAS.
- D. Abdrabou, M. Ahmed, A. Hussein and T. El-Sherbini, Environ. Sci. Pollut. Res., 2023, 30, 99789–99808 CrossRef CAS PubMed.
- D. Ravichandran, M. M. Abdullah, V. Vasanthakumar, R. Ranjith, S. Suganya, R. Meena and S. Murugesan, Colloids Surf., A, 2024, 689, 133701 CrossRef.
- G. Nagaraju, K. R. Pooja, G. R. Suma, N. G. Palan, R. Kiran, R. Puttegowda and J. Manjanna, Inorg. Chem. Commun., 2025, 176, 114143 CrossRef CAS.
- S. Ramar, P. Elango, A. Velusamy, B. Athinarayanan, V. K. Jothi, D. Pattappan and Y. T. Lai, J. Mol. Struct., 2024, 1317, 139127 CrossRef CAS.
- S. Iqbal, A. Bahadur, M. Javed, O. Hakami, R. M. Irfan, Z. Ahmad, A. AlObaid, M. M. Al-Anazy, H. B. Baghdadi and H. S. M. Abd-Rabboh,
et al.
, Mater. Sci. Eng., B, 2021, 272, 115320 CrossRef CAS.
- Z. Li, X. Chen, M. Wang, X. Zhang, L. Liao, T. Fang and B. Li, Desalin. Water Treat., 2021, 215, 222–231 CrossRef CAS.
- R. A. Rahmayeni, Y. Stiadi, N. Jamarun and A. S. Emriadi, J. Mater. Environ. Sci., 2017, 8, 1634–1643 Search PubMed.
- M. A. Abdo, S. F. Mansour, N. S. Al-Bassami and N. I. Abu-Elsaad, Ceram. Int., 2022, 48, 15314–15326 CrossRef CAS.
- A. A. Alghamdi, A. Maatouk, R. M. Almotawa, M. S. Sadeq, E. Abdel-Fattah and M. A. Abdo, Radiat. Phys. Chem., 2024, 225, 112111 CrossRef CAS.
- A. H. C. van Bruggen, M. M. He, K. Shin, V. Mai, K. C. Jeong, M. R. Finckh and J. G. Morris, Sci. Total Environ., 2018, 616–617, 255–268 CrossRef CAS PubMed.
- J. P. Myers, M. N. Antoniou and B. Blumberg,
et al.
, Environ. Health, 2016, 15, 19 CrossRef PubMed.
- S. Ali, A. Khan and Z. U. H. Khan,
et al.
, Chemosphere, 2023, 312, 136543 Search PubMed.
- L. Cao, D. Ma, Z. Zhou, C. Xu, C. Cao, P. Zhao and Q. Huang, Chem. Eng. J., 2019, 368, 212–222 CrossRef CAS.
- S. Yasmeen, L. Burratti, L. Duranti, E. Sgreccia and P. Prosposito, Nanomaterials, 2024, 14, 470 CrossRef CAS PubMed.
- Z. Zhu, F. Guo, Z. Xu, X. Di and Q. Zhang, RSC Adv., 2020, 10, 11929–11938 RSC.
- F. D. J. Silerio-Vázquez, M. García-Roig, L. A. González-Burciaga, C. M. Nuñez-Nuñez and J. B. Proal-Nájera, J. Water Process Eng., 2024, 66, 105948 CrossRef.
- T. P. Dao, T. H. Vu, L. Gnanasekaran, T. M. Aminabhavi, Y. Vasseghian and S. W. Joo, Chem. Eng. J., 2024, 501, 157469 CrossRef.
- S. K. Sumantrao, P. Kariyajjanavar, A. H. Shridhar, S. C. Ghagane, S. S. Chigari, G. Bonageri and A. S. Alsubaie, J. Environ. Chem. Eng., 2025, 13, 115437 CrossRef CAS.
- S. Ullah, S. A. B. Bukhari, H. Nasir, T. Akhtar, S. Mahboob and M. Zahid, J. Photochem. Photobiol., A, 2024, 455, 115787 CrossRef CAS.
- R. Kumar, L. George, Z. Jun and S. Mukherji, Chemosphere, 2022, 308, 136402 CrossRef CAS PubMed.
- W. Li, Z. Wu, A. Meng, Z. Li, X. Chen, J. Zhang and Z. Li, Mater. Lett., 2025, 382, 137867 CrossRef CAS.
- S. P. de Oliveira, T. C. de Oliveira Cândido, A. C. Pereira and D. N. da Silva, Analytica, 2024, 5, 250–262 CrossRef CAS.
- Z. Fang, C. Peng, Q. Zhou and Z. Liu, Chem. Rec., 2025, 25, e202500066 CrossRef CAS PubMed.
- D. Li, J. Yin, Z. Yu, Z. Gao, N. Xu and L. Meng, Sens. Actuators, B, 2025, 138493 CrossRef CAS.
- T. Ö. Varol, O. Hakli and U. Anik, New J. Chem., 2021, 45, 1734–1742 RSC.
- F. Garkani Nejad, H. Beitollahi and I. Sheikhshoaie, Diagnostics, 2023, 13, 632 CrossRef CAS PubMed.
- T. Iftikhar, A. Aziz, G. Ashraf, Y. Xu, G. Li, T. Zhang, M. Asif, F. Xiao and H. Liu, Food Chem., 2022, 395, 133642 CrossRef CAS PubMed.
- Q. Saleem, S. Shahid, M. Javed, S. Iqbal, A. Rahim, S. Mansoor, A. Bahadur, N. S. Awwad, H. A. Ibrahium, R. S. Almufarij and E. B. Elkaeed, RSC Adv., 2023, 13, 10017–10028 RSC.
- Y. M. Ahmed, M. A. Eldin, A. Galal and N. F. Atta, Sci. Rep., 2024, 14, 5654 CrossRef CAS PubMed.
- T. Chen, C. Liu, X. Liu, C. Zhu and D. Zheng, Int. J. Mol. Sci., 2024, 25, 9829 CrossRef CAS PubMed.
- T. Chen, C. Liu, X. Liu, C. Zhu and D. Zheng, Int. J. Mol. Sci., 2024, 25, 9829 CrossRef CAS PubMed.
- T. Iftikhar, A. Aziz, G. Ashraf, Y. Xu, G. Li, T. Zhang and H. Liu, Food Chem., 2022, 395, 133642 CrossRef CAS PubMed.
- A. S. Murali and B. Saraswathyamma, Ceram. Int., 2025, 51, 18492–18504 CrossRef CAS.
- M. Anitha, S. Mahalingam, J. Kim, M. Arivanandhan and P. Anandan, J. Inorg. Organomet. Polym. Mater., 2024, 34, 1835–1849 CrossRef CAS.
- Y. Gao, T. Xie, H. Wang, J. Yang and H. Lin, J. Mater. Sci.: Mater. Electron., 2024, 35, 1719 CrossRef CAS.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  *a
*a

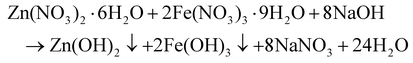
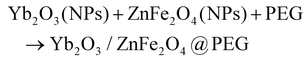
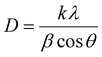

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000× magnification and (b) 30
000× magnification and (b) 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000× magnification.
000× magnification.