[Pd2(η4-P7)2]4−: a Pd2 sandwiched by two anionic nortricyclane-type polyphosphides
Minghao
Deng
 ,
Xinyue
Wang
,
Xinyue
Wang
 ,
Yu
Tang
,
Yu
Tang
 * and
Fuxing
Pan
* and
Fuxing
Pan
 *
*
State Key Laboratory of Applied Organic Chemistry, Key Laboratory of Nonferrous Metal Chemistry and Resources Utilization of Gansu Province, College of Chemistry and Chemical Engineering, Lanzhou University, Lanzhou, 730000 China. E-mail: tangyu@lzu.edu.cn; panfx@lzu.edu.cn
First published on 8th December 2025
Abstract
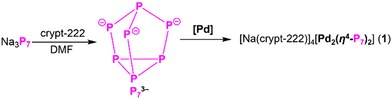
By introducing [Pd(PPh3)4] into a P73− solution, we have successfully synthesized and crystallized [Pd2(η4-P7)2]4− (1a) in [Na(crypt-222)]41a. As a phosphide congener of the reported [Pd2As14]4−, 1a can be viewed as a Pd2 sandwiched by two (η4-P7) units, representing the first organic-ligand–free Pd–P cluster. In addition, 1a provides a platform for investigating Pd–P alloys on the molecular level.
As an important phosphorus precursor in industry, white phosphorus (P4; Fig. 1a, left) has attracted great interest and has yielded tremendous applications in the fields of organo- and organometallic phosphorus chemistry.1–4 Conversely, its toxicity and metastable nature—being sensitive to temperature, air and light—make it difficult to handle during synthesis and often lead to the formation of Pnq species with varied nuclearities (n ≠ 4).5 As an alternative, the anionic nortricyclane-type polyphosphide P73− (light and temperature stable) serves as a closely related cage (Fig. 1a, right), but its chemistry remains largely underinvestigated.6–8 P73−, consisting of one basal three-membered ring, three bridging P atoms and one apical atom, can formally be regarded as adjacent P–P bonds of a P4 being inserted by three P−.5
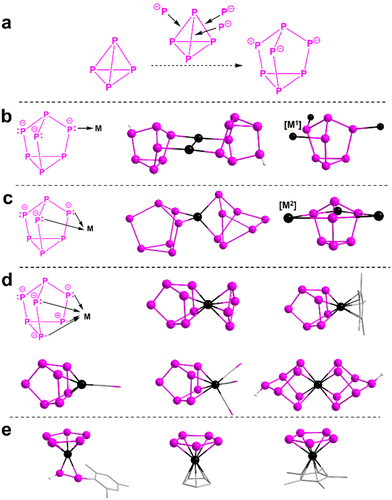 | ||
| Fig. 1 (a) Comparison of P4 (left) and P73− (right) structures; (b) 2-e–donor (η1-P7) unit on its own and in example compounds [M2(η1-:η1-HP7)2]2− (M = Ag, Au) and the neutral {[M1]3(η1-:η1-:η1-P7)} ([M1] = TtMe3, Tt = Ge, Sn, Pb; FeCp(CO)2, AuNHCdipp);10–13 (c) 4-e–donor (η2-P7) unit and example compounds [M(η2-P7)2]3− (M = Zn, Cd) and {[M2]3(η2-:η2-:η2-P7)} ([M2] = RE(NNfc)(THF)n, NNfc = 1,1-fc(NSitBuMe2)2, fc = ferrocenylene, RE/n = Sc/0, Y/1, La/1, Lu/1; Li(tmeda), Li(THF)2);14–20 (d) 6-e–donor (η4-P7) unit and example compounds [(η4-P7)Fe(η4-P4)]3−, [(C5Me5)Fe(η4-P7)]2−, [(η4-P7)Ni(CO)]3−, [(η4-P7)M(CO)3]3− (M = Cr, Mo, W) and [Fe(η4-HP7)2];2–19,21–25 and (e) example compounds resulting from fragmentations and rearrangements of P73−: [(η5-P5)Co{η2-P2H(Mes)}]2−, [(η5-P5)VCp]1−/2−, [(η5-P5)CrCp]− and [(η5-P5)Mn(C5Me5)]−.26,27 | ||
Upon bonding with s-/d-/f-p-block (semi)metal atom(s),9 P73− may remain intact and in the first situation: each of the two or three bridging P atoms serves as a two-electron (2-e) donor (η1; Fig. 1b), such as in the protonated [M2(η1-:η1-HP7)2]2− (M = Ag, Au) and the neutral {[M1]3(η1-:η1-:η1-P7)} ([M1] = TtMe3, Tt = Ge, Sn, Pb; FeCp(CO)2, AuNHCDipp),10–13 or in the second situation: act as a four-electron (4-e) donor (η2; Fig. 1c), such as in [(PPh3)HPt(η2-P7)]2−, [M(η2-P7)2]3− (M = Zn, Cd) and {[M2]3(η2-:η2-:η2-P7)} ([M2] = RE(NNfc)(THF)n, NNfc = 1,1-fc(NSitBuMe2)2, fc = ferrocenylene, RE/n = Sc/0, Y/1, La/1, Lu/1; Li(tmeda), Li(THF)2).14–20 On breaking one of the basal P–P bonds, two of the basal P atoms and two of the bridging P atoms together bond to one metal in a six-electron (6-e) donor η4 fashion (Fig. 1d), such as for [(η4-P7)Fe(η4-P4)]3−, [Fe(η4-HP7)2]2−, [(C5Me5)Fe(η4-P7)]2−, [(η4-P7)Ni(CO)]3− and [(η4-P7)M(CO)3]3− (M = Cr, Mo, W).19,21–25 Although the original {P7} unit tends to maintain its nortricyclane-type structure under various interactions, P73− does undergo fragmentations and rearrangements in certain situations, for example during chemical activation of P73− with [Co(PEt2Ph)2(Mes)2] yields [(η5-P5)Co{η2-P2H(Mes)}]2− containing a planar cyclo-{P5}.26 Very recently, a series of hybrid sandwich complexes with early transition metals at their centers ([(η5-P5)VCp]1−/2−, [(η5-P5)CrCp]− and [(η5-P5)Mn(C5Me5)]−; Fig. 1e) have been isolated by reacting P73− with the corresponding [MCp2] (M = V, Cr) and [Mn(C5Me5)2], respectively,27 representing another breakthrough as pioneered by the sandwich-type [(η5-P5)Fe(C5Me5)].28
Besides planar cyclo-P5, the aforementioned η4-type {P7} unit has proven to be another important candidate for constructing phosphorus-based sandwich compounds. In previously reported works,19,21–25 on cutting off one of the basal P–P bonds, the {P7} unit donates six electrons and forms σ-type delocalized interactions with the central metal atom (5c–2e bond). The {P7} unit shows a bonding nature isolobal with rectangularly distorted cyclobutadiene complexes–and, due to its flexibility, can accommodate metal ion(s). However, in the CCDC database (until Oct. 2025), examples of central metal atom(s) sandwiched by ligands that are 6-e donors {η4-P7} are remarkably rare.19,21–25
In this work, we report the synthesis, crystallization and characterization of salt compound 1–made up of a Pd2 centered anion [Pd2(η4-P7)2]4− (1a) and the cation [Na(crypt-222)]+ (crypt-222 = 4,7,13,16,21,24-hexaoxa-1,10-diazabicyclo[8.8.8]hexacosane)–denoted as [Na(crypt-222)]41a (1). 1 was synthesized by reacting Na3P729 with [Pd(PPh3)4]/crypt-222 in DMF (Scheme 1). This reaction mixture was stirred at room temperature (r.t.) for 3 hours, and the resulting dark brown solution was filtered and layered with THF. After 7 days crystallization time at r.t., dark red block-shaped crystals were isolated from the wall and bottom of the vial in a yield of ∼10% based on Na3P7. The obtained complex was characterized by carrying out single-crystal X-ray diffraction (SCXRD) and energy-dispersive X-ray spectroscopy (EDS) analyses. The detailed synthesis and structural characterization of 1 are provided in SI (Fig. S1–S3, Tables S1 and S2).
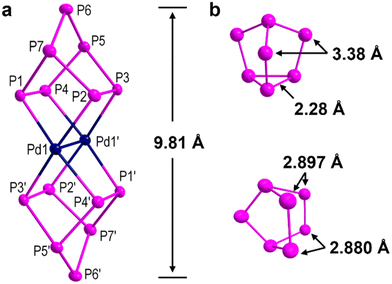
X-ray diffraction shows that compound 1 crystallizes in the triclinic space group P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) with half a positionally ordered 1a anion and two [Na(crypt-222)]+ counter cations in the asymmetric unit. As shown in Fig. 2a, cluster 1a is best described as a Pd2 sandwiched by two anionic nortricyclane-type polyphosphides (η4-P7) with an overall length of close to 1 nm (9.81 Å). One of the basal {P3} triangle P–P bonds in the P73− precursor, namely that showing a bond length of 2.28 Å (Fig. 2b, top),30 is broken in 1a, resulting in elongated P1/3⋯P2/4 distances (2.897/2.880 Å, calculated; Fig. 2b, bottom), each of which is far greater than the sum of the covalent single-bond radii for P2 (2.22 Å).31 The rest of the P–P bonds in the precursor show lengths in 1a within the normal range for unbroken bonds (2.1430(5)–2.2199(5) Å) and comparable with the reported ones.32–36 The distance between the bridging P atoms of the P73− precursor is 3.38 Å, and thus an obvious structural rearrangement occurs in order for these atoms to coordinate the central Pd2. The 1a represents, to the best of our knowledge, the first reported organic-ligand-free Pd-P compound. Note that the reported [(P2){Pd(PNP)}2] (2.33 Å; PNP = N(CHCHPtBu2)2),37 [(η2-LCP = PLC)M(PPh3)2]2+ (2.35–2.40 Å; LC = 4,5-dimethyl-1,3-diisopropyl-imidazol-2-yl),38 [CH3N(CH2CH2NPiPr2)2U(I)(μ-Pd)(μ-I)]2 (2.33 Å)39 and [L6Al6Pd3K2] (2.33 Å; L = [(CH3)2N(CH2)2NPiPr2]−)40 contain Pd–P bond lengths similar to those in 1a (2.3778(4)–2.4006(4) Å).
with half a positionally ordered 1a anion and two [Na(crypt-222)]+ counter cations in the asymmetric unit. As shown in Fig. 2a, cluster 1a is best described as a Pd2 sandwiched by two anionic nortricyclane-type polyphosphides (η4-P7) with an overall length of close to 1 nm (9.81 Å). One of the basal {P3} triangle P–P bonds in the P73− precursor, namely that showing a bond length of 2.28 Å (Fig. 2b, top),30 is broken in 1a, resulting in elongated P1/3⋯P2/4 distances (2.897/2.880 Å, calculated; Fig. 2b, bottom), each of which is far greater than the sum of the covalent single-bond radii for P2 (2.22 Å).31 The rest of the P–P bonds in the precursor show lengths in 1a within the normal range for unbroken bonds (2.1430(5)–2.2199(5) Å) and comparable with the reported ones.32–36 The distance between the bridging P atoms of the P73− precursor is 3.38 Å, and thus an obvious structural rearrangement occurs in order for these atoms to coordinate the central Pd2. The 1a represents, to the best of our knowledge, the first reported organic-ligand-free Pd-P compound. Note that the reported [(P2){Pd(PNP)}2] (2.33 Å; PNP = N(CHCHPtBu2)2),37 [(η2-LCP = PLC)M(PPh3)2]2+ (2.35–2.40 Å; LC = 4,5-dimethyl-1,3-diisopropyl-imidazol-2-yl),38 [CH3N(CH2CH2NPiPr2)2U(I)(μ-Pd)(μ-I)]2 (2.33 Å)39 and [L6Al6Pd3K2] (2.33 Å; L = [(CH3)2N(CH2)2NPiPr2]−)40 contain Pd–P bond lengths similar to those in 1a (2.3778(4)–2.4006(4) Å).
Organometallic complexes of sandwich-type dipalladium are not uncommon. The first structurally well-defined such compound with unsaturated hydrocarbon ligands was reported in 1965 ([LPd(C6H6)], L = (Al2Cl7)−; Fig. 3a)41,42 and then extended to monolayer palladium sheets, as for [Tr2Pd3Cl3]− (Tr = C7H7; Fig. 3b).43 The metallic sandwich-type clusters {(Ge9)2[η6-Ge(PdPPh3)3]}4− containing planar {Ge@Pd3} flanked by nonagermanide caps (Fig. 3c)44 and [Ge18Pd3(TtiPr3)6]2− (Tt = Si, Sn; Fig. 3d ) containing a central Pd3 triangle45,46 have also been reported. Dipalladium sandwiched by inorganic ligands, however, has only been previously observed in [Pd2As14]4− (Fig. 3e),47 featuring here a Pd–Pd bond length (2.714 Å) slightly longer than that in 1a (2.69687(19) Å). Thus, to the best of our knowledge, 1a represents the first organic-ligand–free Pd–P cluster and only the second example of a sandwich-type dipalladium inorganic compound. Recently, a few related reports containing {Asn} units such as [MM′As16]4− (M = Nb or Ta, M′ = Cu or Ag) and [Fe3(As3)3(As4)]3− have also been reported.48,49
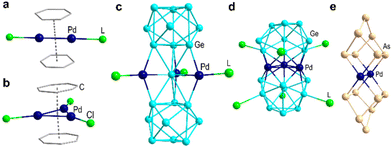 | ||
| Fig. 3 Examples of sandwich-type organometallic and inorganic complexes containing multinuclear palladium: (a) [LPd(C6H6)], L = (Al2Cl7)−;41,42 (b) [Tr2Pd3Cl3]− (Tr = C7H7);43 (c) {(Ge9)2[η6-Ge(PdPPh3)3]};4–44 (d) [Ge18Pd3(TtiPr3)6]2− (Tt = Si, Sn);45,46 (e) [Pd2As14].4–47 | ||
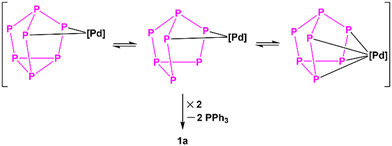
The reported [Pd2As14]4− was obtained via the reaction between As73− and [Pd(PCy3)2] (Cy = cyclohexyl) in ethylenediamine (en)—with en serving not only as solvent, but also as hydrogenation agent taking part in the redox process. “[As7PdH(PCy3)]2−” was proposed as an intermediate towards the formation of [Pd2As14]4−. We also tried to react P73− with [Pd(PPh3)4] in en but failed, indicative of the aprotic DMF as pure solvent being crucial for forming 1a, and of the [Pd(PPh3)4] precursor not proceeding through the hydrogenation pathway—and hence indicative of the mechanism of formation of 1a differing obviously from that of [Pd2As14]4−. Actually, one recent work described in a manuscript titled “Dynamic Behavior of the P73− Cluster and Its Derivatives with Main-Group and Transition Metal Fragments”,50 in combination with the aforementioned examples (e.g. [(η4-P7)Ni(CO)]3− and [(PPh3)HPt(η2-P7)]2−)19 may provide convincing ideas regarding the formation of 1a: the coordination type of {P7} in the intermediate Pd–P cluster is neither exactly η4- nor η2-, but rather in an equilibrium state in between, and the coupling of two such transition-state fragments form 1a (Scheme 2).
Conclusions
In conclusion, by introducing [Pd(PPh3)4] into P73− solution, we have successfully synthesized [Pd2(η4-P7)2]4− (1a) in [Na(crypt-222)]41a, representing the first organic-ligand–free Pd–P cluster and the second example of a sandwich-type dipalladium inorganic compound. 1a can be viewed as a Pd2 sandwiched by two (η4-P7) units—with each such unit resulting from breaking one of the basal P–P bonds of the P73− precursor in combination with a geometrical rearrangement upon coordinating with the Pd2. Our results prove that anionic nortricyclane-type polyphosphide can be used in the construction of sandwich-type clusters and provide a platform for investigating Pd–P alloys on the molecular level, and can finally be applied in catalytic or other fields.Conflicts of interest
There are no conflicts to declare.Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information (SI). Supplementary information: additional detailed synthesis, crystal photographs, X-ray diffraction analyses, EDS measurements. See DOI: https://doi.org/10.1039/d5dt02437d.CCDC 2493250 ([Na(crypt-222)4]1a) contains the supplementary crystallographic data for this paper.51
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 22471102, 22221001 and 22131007), Gansu Provincial Science and Technology Program under Grant No. 24ZD13GA015, the Fundamental Research Funds for the Central Universities (lzujbky-2024-03) and Gansu Province Higher Education Teachers’ Innovation Fund Project (2024B-002).References
- M. Scheer, G. Balázs and A. Seitz, Chem. Rev., 2010, 110, 4236–4256 CrossRef CAS PubMed.
- M. Caporali, L. Gonsalvi, A. Rossin and M. Peruzzini, Chem. Rev., 2010, 110, 4178–4235 CrossRef CAS PubMed.
- K. H. Whitmire, Coord. Chem. Rev., 2018, 376, 114–195 CrossRef CAS.
- M. Peruzzini, L. Gonsalvi and A. Romerosa, Chem. Soc. Rev., 2005, 34, 1038–1047 RSC.
- R. S. P. Turbervill and J. M. Goicoechea, Chem. Rev., 2014, 114, 10807–10828 CAS.
- L. Qiao, C. Zhang, X.-W. Zhang, Z.-C. Wang, H. Yin and Z.-M. Sun, Chin. J. Chem., 2020, 38, 295–304 CrossRef CAS.
- J. Du, P. J. Cobb, J. Ding, D. P. Mills and S. T. Liddle, Chem. Sci., 2023, 15, 13–45 RSC.
- B. van Ijzendoorn and M. Mehta, Dalton Trans., 2020, 49, 14758–14765 RSC.
- F. Pan, R. Ababei, W. Massa and S. Dehnen, Eur. J. Inorg. Chem., 2024, 27, e202300514 CrossRef CAS.
- D. Weber, C. Mujica and H. G. von Schnering, Angew. Chem., Int. Ed. Engl., 1982, 21, 863 CrossRef.
- G. Fritz, K. D. Hoppe, W. Hönle, D. Weber, C. Mujica, V. Manriquez and H. G. v. Schnering, J. Organomet. Chem., 1983, 249, 63–80 CrossRef CAS.
- M. Jo, J. Li, A. Dragulescu-Andrasi, A. Y. Rogachev and M. Shatruk, Dalton Trans., 2020, 49, 12955–12959 RSC.
- R. Ahlrichs, D. Fenske, K. Fromm, H. Krautscheid, U. Krautscheid and O. Treutler, Chem. – Eur. J., 1996, 2, 238–244 CrossRef CAS.
- C. Knapp, B. Zhou, M. S. Denning, N. H. Rees and J. M. Goicoechea, Dalton Trans., 2009, 39, 426–436 RSC.
- W. Huang and P. L. Diaconescu, Chem. Commun., 2012, 48, 2216–2218 RSC.
- W. Huang and P. L. Diaconescu, Eur. J. Inorg. Chem., 2013, 2013, 4090–4096 CrossRef CAS.
- W. Hönle, H. G. von Schnering, A. Schmidpeter and G. Burget, Angew. Chem., Int. Ed. Engl., 1984, 23, 817–818 CrossRef.
- S. Du, J. Hu, Z. Chai, W.-X. Zhang and Z. Xi, Chin. J. Chem., 2019, 37, 71–75 CrossRef CAS.
- S. Charles, J. C. Fettinger, S. G. Bott and B. W. Eichhorn, J. Am. Chem. Soc., 1996, 118, 4713–4714 CrossRef CAS.
- Q. You, J. Zhang, F. Zhang, J. Cai and X. Zhou, Chem. – Eur. J., 2023, 29, e202203679 CrossRef CAS PubMed.
- Z.-C. Wang, L. Qiao, Z.-M. Sun and M. Scheer, J. Am. Chem. Soc., 2022, 144, 6698–6702 CrossRef CAS PubMed.
- C. M. Knapp, J. S. Large, N. H. Rees and J. M. Goicoechea, Chem. Commun., 2011, 47, 4111–4113 RSC.
- E.-M. Schnöckelborg, J. J. Weigand and R. Wolf, Angew. Chem., Int. Ed., 2011, 50, 6657–6660 CrossRef PubMed.
- S. Charles, B. W. Eichhorn, A. L. Rheingold and S. G. Bott, J. Am. Chem. Soc., 1994, 116, 8077–8086 CrossRef CAS.
- M. V. Butovskiy, G. Balázs, M. Bodensteiner, E. V. Peresypkina, A. V. Virovets, J. Sutter and M. Scheer, Angew. Chem., Int. Ed., 2013, 52, 2972–2976 CrossRef CAS PubMed.
- C. M. Knapp, B. H. Westcott, M. A. C. Raybould, J. E. McGrady and J. M. Goicoechea, Angew. Chem., Int. Ed., 2012, 51, 9097–9100 CrossRef CAS PubMed.
- Z.-C. Wang, S. Wang, W.-J. Tian, Z.-Y. Guo, M. Scheer and Z.-M. Sun, J. Am. Chem. Soc., 2025, 147, 36530–36538 CrossRef CAS PubMed.
- O. J. Scherer and T. Brück, Angew. Chem., Int. Ed. Engl., 1987, 26, 59–59 Search PubMed.
- M. Cicač-Hudi, J. Bender, S. H. Schlindwein, M. Bispinghoff, M. Nieger, H. Grützmacher and D. Gudat, Eur. J. Inorg. Chem., 2016, 2016, 649–658 CrossRef.
- B. van Ijzendoorn, R. Lister-Roberts, N. Kaltsoyannis and M. Mehta, J. Am. Chem. Soc., 2025, 147, 30317–30325 CrossRef CAS PubMed.
- P. Pyykkö and M. Atsumi, Chem. – Eur. J., 2009, 15, 186–197 CrossRef PubMed.
- W. Fang, I. Douair, A. Hauser, K. Li, Y. Zhao, P. W. Roesky, S. Wang, L. Maron and C. Zhu, CCS Chem., 2021, 4, 2630–2638 CrossRef.
- Y. Zhao, Z. Chai, Z. Song, J. Wei and W.-X. Zhang, Organometallics, 2025, 44, 300–306 CrossRef CAS.
- X. Huangfu, Z. Wang, Y. Chen, J. Wei, W. Liu and W.-X. Zhang, Natl. Sci. Rev., 2024, 11, nwae162 CrossRef CAS PubMed.
- C. Yang, X. Jiang, Q. Chen, X. Leng, J. Xiao, S. Ye and L. Deng, J. Am. Chem. Soc., 2022, 144, 20785–20796 CrossRef CAS PubMed.
- Y. Mei, X.-Y. He, J. Li, M. Liu, Q. Liang, C. Yang and L. L. Liu, Nat. Commun., 2025, 16, 2321 CrossRef CAS PubMed.
- M. C. Neben, N. Wegerich, T. A. Al Said, R. R. Thompson, S. Demeshko, K. Dollberg, I. Tkach, G. P. I. Van Trieste, H. Verplancke, C. von Hänisch, M. C. Holthausen, D. C. Powers, A. Schnegg and S. Schneider, J. Am. Chem. Soc., 2025, 147, 5330–5339 CrossRef CAS PubMed.
- K. Schwedtmann, J. Haberstroh, S. Roediger, A. Bauzá, A. Frontera, F. Hennersdorf and J. J. Weigand, Chem. Sci., 2019, 10, 6868–6875 RSC.
- W. Fang, Y. Li, T. Zhang, T. Rajeshkumar, I. del Rosal, Y. Zhao, T. Wang, S. Wang, L. Maron and C. Zhu, Angew. Chem., Int. Ed., 2024, 63, e202407339 CrossRef CAS PubMed.
- P. Chen, J. Chen, Y. Zhao, J. Ma, J. Xie, C. Zhu, Z. Lin and Q. Zhu, J. Am. Chem. Soc., 2025, 147, 21153–21161 CrossRef CAS PubMed.
- G. Allegra, A. Immirzi and L. Porri, J. Am. Chem. Soc., 1965, 87, 1394–1395 CrossRef CAS.
- G. Allegra, G. T. Casagrande, A. Immirzi, L. Porri and G. Vitulli, J. Am. Chem. Soc., 1970, 92, 289–293 CrossRef CAS.
- T. Murahashi, M. Fujimoto, M. Oka, Y. Hashimoto, T. Uemura, Y. Tatsumi, Y. Nakao, A. Ikeda, S. Sakaki and H. Kurosawa, Science, 2006, 313, 1104–1107 CrossRef CAS PubMed.
- H.-L. Xu, N. V. Tkachenko, Z.-C. Wang, W.-X. Chen, L. Qiao, A. Muñoz-Castro, A. I. Boldyrev and Z.-M. Sun, Nat. Commun., 2020, 11, 5286 CrossRef CAS PubMed.
- L. G. Perla and S. C. Sevov, J. Am. Chem. Soc., 2016, 138, 9795–9798 CrossRef CAS PubMed.
- L. G. Perla, A. Muñoz-Castro and S. C. Sevov, J. Am. Chem. Soc., 2017, 139, 15176–15181 CrossRef CAS PubMed.
- M. J. Moses, J. Fettinger and B. Eichhorn, J. Am. Chem. Soc., 2002, 124, 5944–5945 CrossRef CAS PubMed.
- W.-Q. Zhang, H. W. T. Morgan, C.-C. Shu, J. E. McGrady and Z.-M. Sun, Inorg. Chem., 2022, 61, 4421–4427 CrossRef CAS PubMed.
- W.-Q. Zhang, Z.-S. Li, J. E. McGrady and Z.-M. Sun, Angew. Chem., Int. Ed., 2023, 62, e202217316 CrossRef CAS PubMed.
- K. Wang, Y. Wang, Z. Liu, L. Zeng, Z. Li, S. Albawardi, M. Mehta and J. E. McGrady, Inorg. Chem. DOI:10.1021/acs.inorgchem.5c03163.
- CCDC 2493250: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2ppff8.
| This journal is © The Royal Society of Chemistry 2026 |