Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
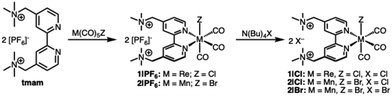
Pendant-cation assisted spontaneous axial halide substitution in group 7 M(L)(CO)3X complexes
Wesley W.
Kramer
 *ab,
Thomas R.
Sheridan†
*ab,
Thomas R.
Sheridan†
 b,
Jorge
Pérez-Vázquez‡
b,
Jorge
Pérez-Vázquez‡
 a,
Christophe A.
Boivin§
a,
Christophe A.
Boivin§
 a,
Amanda M.
Browne
a,
Amanda M.
Browne
 a,
Catherine G.
Logan
a,
Catherine G.
Logan
 a and
Brian C.
Sanders¶
a and
Brian C.
Sanders¶
 b
b
aDepartment of Chemistry, Hamilton College, Clinton, NY 13323, USA. E-mail: wkramer@hamilton.edu
bDivision of Chemistry and Chemical Engineering, California Institute of Technology, Pasadena, CA 91125, USA
First published on 27th February 2026
Abstract
The installation of cationic pendant groups proximal to catalyst active sites has resulted in improved performance for electrocatalytic CO2 reduction (CO2R) by molecular catalysts. It is not well understood how pendant cationic groups, designed to stabilize anionic CO2 reduction intermediates, interact with the axial halide ligands of M(diimine)(CO)3X (M = Re, Mn; X = Cl, Br), an important class of molecular CO2R electrocatalysts. We observe that in [M(tmam)(CO)3X]2+ complexes (1: M = Re, X = Cl; 2: M = Mn, X = Br, tmam2+ = 4,4′-methylenetrimethylammonium-2,2′-bipyridine) bearing the dicationic tmam2+ ligand, the cationic pendant groups facilitate the spontaneous exchange of the axial halide for solvent molecules (MeCN or H2O), a highly unusual behavior for this class of complexes. We present a study of the thermodynamics and kinetics of these reactions under aqueous and non-aqueous conditions. The extent to which this reaction occurs was found to depend on the identity of the metal, with substitution being more favorable for Mn than Re, and the solvent and halide substitution was more favorable in H2O than MeCN for both complexes. 1H NMR kinetics experiments for axial chloride substitution for 1 in D2O suggest a second-order dependence on complex concentration, indicating an intermolecular process. The halide-substitution reaction had little effect on the electrochemistry of the complexes in cyclic voltammetry experiments due to the reductive instability (deamination) of tmam2+. This study demonstrates that cationic pendant groups are noninnocent, actively modulating the coordination environment of the catalyst, which provides key insights into their potential roles in catalyst activation.
Introduction
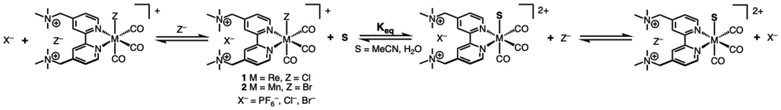
Carbon dioxide reduction (CO2R) is a critical reaction for the creation of carbon-neutral fossil fuels. However, due to the inert structure of carbon dioxide, large overpotentials are generally required for catalysis, making CO2 reduction at scale economically challenging. As one of the prototypical CO2 reduction (CO2R) electrocatalyst, group 7 M(bpy)(CO)3X (bpy = 2,2′-bipyridine)1–3 complexes have been useful models to test concepts in catalyst design, owing to their well-defined catalytically active sites and the readily modifiable bipyridine ligand scaffold. Ligand modification strategies have provided insights into how to influence and improve electrocatalytic CO2 reduction via modifications of the ligand electronic structure through the introduction of electron-donating or -withdrawing groups,4–6 introducing steric hindrance near the catalytic center,7 and groups that modify the secondary coordination sphere.8–11 One promising approach has shown that ligands modified with positively charged groups can reduce the overpotential required for CO2R through secondary-sphere electrostatic interactions that stabilize the negatively charged CO2 reduction intermediate.12–17 This electrostatic effect has been shown to significantly improve catalytic activity and lower overpotentials for CO2 reduction in both iron tetraphenylporphyrin12 and Re(bpy-R)(CO)3Cl13–17 derivatives using ligands modified with cationic pendant groups positioned near the metal center. Recent work by Thoi and coworkers sought to study the effects of the cationic groups of the tmam2+ (tmam2+ = 4,4′-methylenetrimethylammonium-2,2′-bipyridine) ligand on the electrocatalytic CO2R performance of rhenium tricarbonyl catalysts.18 This work found that the trimethyl ammonium functional groups are prone to reductive deamination of the pendant groups at potentials significantly positive of the onset of CO2R catalysis, making the study of their effect on CO2R impossible. Nonetheless, this work highlights both the promise and challenges of incorporating charged functional groups into ligand frameworks. In particular, the tmam2+ scaffold provides a valuable model for understanding how charged pendants can tune the electronic structure, coordination environment, and ultimately the catalytic behavior of transition metal complexes.An understudied aspect of cationic pendant groups in the secondary coordination sphere is how these groups affect the ligands in the primary coordination sphere. In the parent complex, Re(bpy)(CO)3Cl, the first 1-e− reduction is localized on the bipyridine ligand, resulting in the formation of [ReI(bpy˙−)(CO)3Cl]−, while the second 1-e− reduction is Re-centered and results in [Re0(bpy˙−)(CO)3]−. The dissociation of the axial Cl− is necessary to open a coordination site for CO2 binding. Several groups have shown that the use of bipyridine ligands functionalized with cationic groups can promote the dissociation of halide following the first reduction by stabilizing the release of the anion and lowering the onset potential of CO2R.13–17 The binding affinity between halide ions and the cationic ligand binding pockets has been shown to aid the dissociation of the axial halide of the group 7 tricarbonyl catalysts upon reduction.16 Anion binding with the tmam2+ ligand has been studied in some detail for a series of [Ru(tmam)n(bpy)3−n](2+2n)+ complexes.19–25 This work demonstrated that halides have a greater binding affinity with the cationic binding pockets of the tmam2+ ligands than the larger PF6− anions used to solubilize the complexes in non-aqueous solvents. The ligand's greater affinity for binding halides in its binding pocket, and proximity to the active site of the [M(tmam)(CO)3X]2+ complex may interact with the axial halide. However, the extent to which cationic groups of these complexes interact with the axial halide in their unreduced state has not been investigated.
To this end, we have synthesized a series of Re and Mn tricabonyl complexes with the tmam2+ ligand [M(tmam)(CO)3X](Y)2 (M = Mn, Re; X = Br, Cl; Y = PF6−, Cl−) to study their behavior in H2O and MeCN. Through 1H NMR, IR, and UV-vis spectroscopy, we have measured the effect of proximal positively charged groups on the lability of the axial halide. Interestingly, halide substitution by the solvent is observed for all complexes. We find that the extent of substitution depends on both the identity of the metal (with Mn being far more favorable to substitution than Re) and the solvent. The thermodynamics of this halide exchange process has been quantified using 1H NMR studies. Based on 1H NMR kinetics, a second order mechanism for halide dissociation involving two M(tmam)2+ complexes is proposed. Additionally, electrochemical studies in both aqueous and non-aqueous solvents were used to understand the influence of halide substitution chemistry on the electrochemical response of the complexes.
Results and discussion
Synthesis and characterization
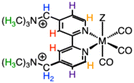
Dicationic [M(tmam)(CO)3X](PF6)2 complexes were prepared by refluxing a solution of [tmam](PF6)2 and the appropriate M(CO)5X (M = Re, X = Cl; M = Mn, X = Br) in acetone as shown in Scheme 1. Acetone was chosen as the solvent for this reaction to promote the solubility of the tmam2+ ligand. The products, [Re(tmam)(CO)3Cl](PF6)2 (1|PF6) and [Mn(tmam)(CO)3Br](PF6)2 (2|PF6), were precipitated from the reaction mixture by the addition of Et2O. The H2O-soluble salts, [Re(tmam)(CO)3Cl](Cl)2 (1|Cl) and [Mn(tmam)(CO)3Br](Cl)2 (2|Cl), were precipitated from the acetone reaction mixture by the addition of excess TBACl. The various salts of these metal complexes were obtained in 76–92% yields. [Mn(tmam)(CO)3Br](Br)2 (2|Br) was prepared by treating a concentrated acetone solution of 2|PF6 with excess TBABr to afford 41% yield. The lower yield of 2|Br stems from its increased solubility in acetone, leading to loss of product during purification. The identity and purity of all complexes were confirmed using 1H and 13C NMR, FTIR, mass spectrometry, and elemental analysis. 1H NMR and FTIR data for 1|PF6 agree with previously published spectroscopic data for [Re(tmam)(CO)3Br] [PF6]2.18 The MeCN adducts of 1 and 2 were prepared by treating MeCN solutions of complexes 1|PF6 and 2|PF6, respectively, with AgPF6.X-ray quality crystals of 1 and 2 were grown by slow diffusion of Et2O into MeCN. Interestingly, crystals of the tricationic MeCN adduct [Re(tmam)(CO)3(MeCN)](PF6)3 (1-MeCN+) were grown by slow diffusion of Et2O into a MeCN solution containing 1|PF6 and excess NH4PF6. Typically, Ag salts are required to remove the chloride from these Re complexes.26 X-ray quality crystals of 1|Cl, 2|Cl, and 2|Br could not be grown. X-ray crystal structures of 2|PF6 are shown in Fig. 1, and those of 1|PF6 and 1-MeCN+ are shown in Fig. S1 and S2. The solid-state structures of 1|PF6 and 2|PF6 show the pendant trimethylammonium groups oriented opposite the bipyridine plane from the axial halide. M–X bond distances (1|PF6: Re–Cl = 2.487 Å, 2|PF6: Mn–Br = 2.534 Å) agree with those of comparable Re and Mn tricarbonyl complexes.3,27,28 In all three structures, a PF6− anion is situated in the dicationic binding pocket of the tmam2+ ligand. This close pairing of the PF6− is typically observed in the crystal structures of tmam2+ complexes.18,20,25
 | ||
| Fig. 1 ORTEP structures (50% thermal probability for all non-hydrogen atoms) of [Mn(TMAM)(CO)3(Br)](PF6)2 (2|PF6) showing PF6− in the cationic pocket. | ||
Spectroscopic characterization
The ATR FT-IR spectra of 1|X and 2|X complexes are shown in Fig. S3 and S4 and the CO stretching frequencies are listed in Table 1. The solid-state FT-IR spectra of 1|PF6 and 1|Cl show three dominant CO stretches as expected for a fac tricarbonyl complex. However, the lower energy CO stretches are significantly broadened, and in the spectrum of 1|Cl, both peaks display clear shoulders. The ATR spectra of the 2|X complexes still show three major peaks, but the spectra are complicated by additional CO stretches around the low energy CO. The presence of these broadened spectra and additional CO peaks may indicate that the complexes have adopted multiple packing arrangements in the solid state.29 The IR spectra of 1|PF6, 1-MeCN+, 2|PF6, and 2-MeCN+ exhibit a P–F stretch at 823 cm−1, and this signal is absent from the spectra of 1|Cl, 2|Cl, and 2|Br, confirming complete salt metathesis. The solution phase FTIR spectra of 1|PF6 and 2|PF6 in MeCN (Fig. S5) are comparable to the solid-state spectra but are significantly sharper. The FTIR spectrum of 1|PF6 shows three sharp peaks.| Complex | ν CO (cm−1) | λ/nm (ε/M−1 cm−1) |
|---|---|---|
| a Spectra collected in DMSO. b Spectra collected in MeCN. | ||
| 1|PF6 | 2018, 1919, 1891, (2025, 1923, 1904)b | 302 (14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000), 390 (3800) 000), 390 (3800) |
| 1|Cl | 2023, 1910, 1873 | 303 (16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000), 390 (3400) 000), 390 (3400) |
1-MeCN+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b b |
2039, 1919br | 249 (18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000), 284 (15 000), 284 (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000), 312 (13 000), 312 (13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000), 326 (13 000), 326 (13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000), 359 (4300) 000), 359 (4300) |
| 2|PF6 | 2023, 1944, 1931, 1911, (2030, 1941, 1929)b | 305 (23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000), 437 (3400) 000), 437 (3400) |
| 2|Cl | 2024, 1934, 1924, 1901 | 305 (21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000), 433 (3200) 000), 433 (3200) |
| 2|Br | 2023, 1934(sh), 1907 | 305 (22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000), 437 (3300) 000), 437 (3300) |
2-MeCN+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b b |
2049, 1941 br | 246 (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000), 296 (20 000), 296 (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000), 389 (3700) 000), 389 (3700) |
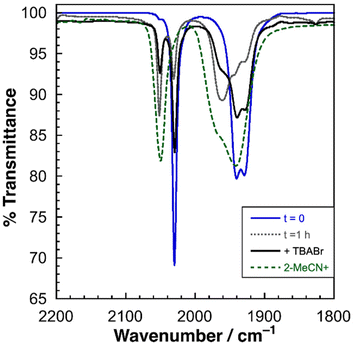
For 2|PF6, more complex behavior is observed in the solution phase. The FTIR spectrum of freshly prepared solutions shows three CO stretches at 2030, 1941, and 1929 cm−1. However, over time, additional peaks emerge at 2051 and 1961 cm−1. As shown in Fig. 2, these new peaks correspond to the presence of 2-MeCN+ in solution. Addition of TBABr to the sample shifts the spectrum back towards the original, suggesting a ligand substitution equilibrium in which the axial Br− ligand is displaced by MeCN. This phenomenon is discussed in more detail below.
The UV-vis spectra of 1|X and 2|X were obtained in DMSO and are shown in Fig. 3. Peak maxima are given in Table 1. For both 1|X and 2|X, the identity of the counterion X− had only minor effects on the absorption spectrum of the complex. All salts of compound 1 display an MLCT absorption at 390 nm and a tmam2+ π–π* transition at 303 nm with a shoulder at 326 nm. For compound 2 salts, the MLCT band of 2|Cl (433 nm) is blue-shifted by 4 nm compared to 2|PF6 and 2|Br which both appear at 437 nm. All three compounds exhibit a tmam2+ π–π* transition at 305 nm. Though the effect is small, the identity of the counterion primarily affects the molar absorptivity of the complexes. The near uniformity of the absorption spectra of the 1|X and 2|X salts suggests that ion pairing with the tmam2+ ligand has little effect on the electronic structure of the complexes. Minor changes in the absorption spectrum of [Ru(tmam)n(L)m](2+2n)+ complexes upon anion exchange have been observed.20 The UV-vis spectra of 1-MeCN+ and 2-MeCN+ in MeCN are shown in Fig. S6 and peak data are summarized in Table 1. For 1-MeCN+, the MLCT peak appears at 359 nm, and the π–π* transitions are split into several distinct peaks. The UV-vis spectrum of 2-MeCN+ also displays a blue-shifted MLCT absorption at 389 nm and a broadened π–π* transition centered at 296 nm with a shoulder at 324 nm (not observed in the spectrum of 2|PF6).
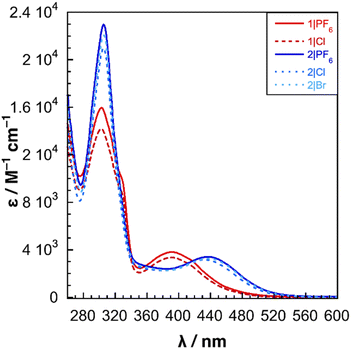 | ||
| Fig. 3 UV-vis spectra of complexes 1|X and 2|X obtained in DMSO. Molar absorptivity of each complex was calculated from a linear fit derived from absorbance at 5 different concentrations. | ||
The 1H NMR spectra of all 1|X and 2|X complexes in deuterated dimethylsulfoxide (DMSO-d6) are shown in Fig. S7–S11 and chemical shifts are tabulated in Table 2. All complexes in DMSO-d6 show a single species consistent with the crystal structures. For all complexes, the methylene protons of the tmam2+ ligand appear in a roofed doublet pattern (Fig. S7–S11). This is consistent with the presence of an anion in the tmam2+ binding pocket. This locks the pendant arms in place and renders the two methylene protons inequivalent.20 While the identity of the anion did not have a significant impact on the UV-vis spectra of these complexes, the NMR spectra are more sensitive to electrostatic interactions between the anion and the binding pocket and are, therefore, more sensitive to changes in the counterion. For both 1 and 2, substitution of PF6− for Cl− results in a large downfield shift for the 3,3′ protons of the bipyridine. While the 5,5′ peak shifts slightly upfield, the 6,6′ protons are unaffected by anion exchange. Smaller downfield shifts were also observed for the protons of the pendant groups. Similar downfield shifts are observed for 2|Br in comparison with 2|PF6, though the shifts are smaller than those observed for 2|Cl. The extent of the downfield shift is correlated with the strength of the interaction between the anion and the electropositive 3,3′ protons.20–22 The trend in the position of the 3,3′ peaks of the 2|X complexes, Cl− (9.27 ppm) > Br− (9.11 ppm) > PF6 (8.66 ppm), correlates with increasing anion sizes. This behavior is consistent with observations of [Ru(tmam)(L)2]4+ complexes titrated with TBAI or TBACl.20–22
Halide substitution equilibria
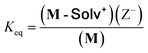 using the average normalized peak areas for the aromatic protons of the parent complex (M), M = 1 or 2, and the solvato species (M-Solv+), Solv = MeCN or H2O. The concentration of the liberated axial halide (Z− = Cl− or Br−) is assumed to be [Z−] = [M-Solv+]. Keq = 7.94 ± 0.84 × 10−4 for the equilibrium between 1 and 1-MeCN+. Equilibrium constants and thermodynamic data for these halide substitution reactions are given in Table 3. This behavior is rare for Re(L)(CO)3X complexes.30 Preparation of solvato complexes for this class of compounds generally requires the use of Ag salts. We hypothesize that the cationic pendant groups of the tmam2+ ligand enable the halide substitution chemistry observed for 1via their capacity to stabilize the release of the halide into the dicationic binding pocket. Qualitatively similar behavior was noted for a recently reported [Re(L2+)(CO)3Cl]2+ (L2+ = 6,6′-(2-((trimethylamonium)-methyl)phenyl)-2,2′-bipyridine) complex, which showed a slow evolution of its 1H NMR spectrum that was attributed to interconversion between rotamers of the large pendant groups.13 However, in light of our observations, it is possible that the authors were observing a slow chloride substitution reaction in MeCN-d3.
using the average normalized peak areas for the aromatic protons of the parent complex (M), M = 1 or 2, and the solvato species (M-Solv+), Solv = MeCN or H2O. The concentration of the liberated axial halide (Z− = Cl− or Br−) is assumed to be [Z−] = [M-Solv+]. Keq = 7.94 ± 0.84 × 10−4 for the equilibrium between 1 and 1-MeCN+. Equilibrium constants and thermodynamic data for these halide substitution reactions are given in Table 3. This behavior is rare for Re(L)(CO)3X complexes.30 Preparation of solvato complexes for this class of compounds generally requires the use of Ag salts. We hypothesize that the cationic pendant groups of the tmam2+ ligand enable the halide substitution chemistry observed for 1via their capacity to stabilize the release of the halide into the dicationic binding pocket. Qualitatively similar behavior was noted for a recently reported [Re(L2+)(CO)3Cl]2+ (L2+ = 6,6′-(2-((trimethylamonium)-methyl)phenyl)-2,2′-bipyridine) complex, which showed a slow evolution of its 1H NMR spectrum that was attributed to interconversion between rotamers of the large pendant groups.13 However, in light of our observations, it is possible that the authors were observing a slow chloride substitution reaction in MeCN-d3.
| K eq | ΔGrxn/kJ mol−1 | ΔHrxn/kJ mol−1 | ΔSrxn/J mol−1 K−1 | |
|---|---|---|---|---|
| a The concentration of salt in these samples was 100 mM (10 equivalents vs. complex). | ||||
| 1|PF6 | 7.94 ± 0.84 × 10−4 | 17.7 ± 2.7 | — | — |
| “wet” | 1.36 ± 0.32 × 10−3 | 16.4 ± 0.6 | — | — |
+TBAPF6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a a |
5.12 + 0.84 × 10−3 | 13.1 ± 0.4 | — | — |
+NH4PF6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a a |
8.50 ± 0.44 × 10−2 | 5.78 ± 0.20 | — | — |
| 1|Cl | 2.21 ± 0.10 | −2.00 ± 0.12 | −4.32 ± 0.26 | −8.28 ± 0.88 |
| 2|PF6 | 4.51 ± 0.94 × 10−2 | 7.71 ± 0.52 | — | — |
| “wet” | 1.42 ± 0.05 × 10−1 | 4.83 ± 0.09 | — | — |
+TBAPF6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a a |
1.63 ± 0.05 × 10−1 | 4.50 ± 0.07 | — | — |
| 2|Cl | 12.4 ± 0.4 | −6.23 ± 0.08 | — | — |
| 2|Br | 14.5 ± 0.6 | −6.62 ± 0.10 | −10.4 ± 1.0 | −12.7 ± 3.3 |
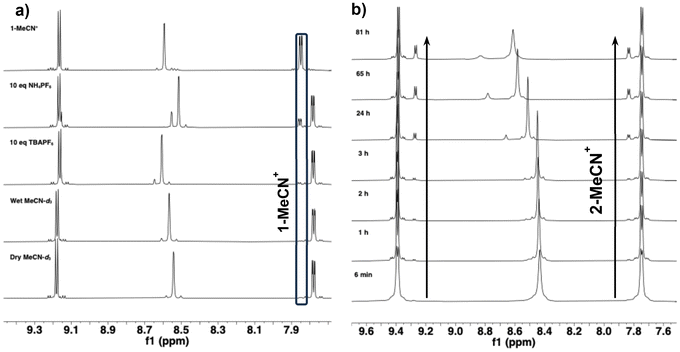
Complex 2|PF6 also displays halide substitution behavior in MeCN, but with a larger Keq compared to 1|PF6. The initial 1H NMR spectrum of 2|PF6 in MeCN-d3 showed significantly broadened peaks. Over the course of one hour, the peaks sharpened and peaks corresponding to a second Mn(tmam) species grew. The concentration of the new species continued to increase over the course of several days until equilibrium was reached as shown in Fig. 4. The 5,5′ peak of the new species is shifted upfield of the corresponding peak of 2. All other resonances, including the 6,6′ peak which was insensitive to the identity of the counterion in DMSO, appear downfield of peaks belonging to the parent complex. This behavior is consistent with partial dissociation of the axial Br− from the complex to generate a mixture of 2 and 2-MeCN+. The equilibrium constant for the reaction of 2 with MeCN is Keq = 4.51 ± 0.94 × 10−2, more than two orders of magnitude greater than that of 1|PF6. Comparison of the equilibrated sample of 2|PF6 to the spectrum of an independently prepared sample of 2-MeCN+ (Fig. S14) supports the assignment of the new species as the product of a halide substitution reaction. The positions of the 6,6′ protons of the pure 2-MeCN+ sample is identical to those of the new species (7.83 ppm), and the 5,5′ protons that appear are shifted by less than 0.01 ppm. The 3,3′ protons in the pure 2-MeCN+ sample are significantly upfield of both 3,3′ peaks in the equilibrated sample of 2|PF6 due to the absence of free Br− in solution. The increasing concentration of Br− in solution results in a downfield shift of the peaks associated with protons in the cationic binding pocket. As the reaction progressed towards equilibrium a large downfield shift in the position of the 3,3′ peaks from both species was observed. The magnitude of the shift was greater for 2-MeCN+ (Δδ2|PF6 = 0.20 ppm, Δδ2-MeCN+ = 0.34 ppm) implying that more of the liberated Br− end up associated with the cationic binding pockets of the more highly charged 2-MeCN+ complexes.
For both 1 and 2, an increase in the equilibrium constant was observed when the spectra were obtained in MeCN-d3 saturated with atmospheric H2O (Keq,1 = 1.36 ± 0.32 × 10−3, Keq,2 = 1.42 ± 0.05 × 10−1) compared to samples prepared using dry solvents in an N2-filled glovebox. In DMSO-d6, the spectra of the 1|X and 2|X complexes are unaffected by the presence of ambient H2O as shown in Fig. S15 and S16. In wet solvent, the methylene protons of the halide and solvent-bound complexes appear as a singlet rather than the roofed doublet observed in DMSO-d6, or the weaker splitting observed in dry MeCN-d3, indicating weaker ion pairing between the tmam2+ and PF6− ions, possibly due to stronger ion–dipole interactions between the counterion and the H2O in solution. The observation of increased Keq alongside weaker tmam-halide ion pairing suggests that the dicationic binding pockets are better able to participate in halide dissociation in wet samples. This combined with the faster halide substitution in wet solvent suggests that halide dissociation is rate limiting in the substitution reaction.
The equilibrium between 2 and 2-MeCN+ was observed using UV-vis spectroscopy in anhydrous MeCN over a period of eight hours. The reaction proceeds very slowly, but subtle changes in the UV-vis spectrum were apparent. A spectrum recorded 65 h after sample preparation shows that the substitution continued to progress slowly. The changes over time in the UV-vis spectrum of 2|PF6 map neatly to those observed in the UV-vis spectrum of 2-MeCN+ (Fig. 5). This is shown more clearly in the difference spectrum of 2|PF6 (Fig. 5, inset), which is qualitatively identical to the difference between the initial spectrum of 2|PF6 (t = 1 min) and the spectrum of 2-MeCN+, further supporting the proposed halide substitution reaction. In agreement with the smaller substitution Keq for 1 in MeCN observed by NMR, the UV-vis spectrum of 1|PF6 in MeCN (Fig. S17) was stable over time and is comparable to the absorption spectrum of 1|PF6 in DMSO.
The 1H NMR spectra of 1 and 2 were collected in MeCN-d3 with 10 equivalents (100 mM) of TBAPF6 (Fig. 4 and S18, respectively) to examine the effect of excess PF6− on Keq. The added TBAPF6 had very little effect on the 1H NMR spectrum of 1, though a minor upfield shift of the protons in the tmam2+ binding pocket was observed, likely due to greater [PF6−] increasing the occupancy of the tmam2+ binding pocket. For 2, the 3,3′ peak of the parent complex still shifts downfield (Δδ = 0.17 ppm) as the sample reaches equilibrium, but at equilibrium the 3,3′ peak of 2 in the 100 mM TBAPF6 solution appears slightly upfield (by 0.02 ppm) of the same peak in neat MeCN-d3. This difference is more dramatic for the 3,3′ protons of 2-MeCN+ which shift 0.12 ppm upfield after the addition of TBAPF6. A minor increase in Keq was observed for both 1|PF6 and 2|PF6 compared to samples without TBAPF6 in wet MeCN-d3 (Table 3). Despite the small Keq measured for the halide substitution reaction of 1|PF6 in MeCN, crystals of 1-MeCN+ were grown from an MeCN solution with excess NH4PF6, suggesting that the presence of the electrolyte shifts the equilibrium towards 1-MeCN+. The 1H NMR spectra of 1|PF6 in MeCN-d3 with 100 mM NH4PF6 initially showed similar concentrations of 1-MeCN+ to samples with TBAPF6. However, after sitting at room temperature in the dark for 12 d, the sample containing NH4PF6 displayed significantly higher concentrations of 1-MeCN+, whereas with TBAPF6 no increase in [1-MeCN+] was observed after eighty days, as shown in Fig. 4. Under these conditions, Keq = 9.72 ± 0.80 × 10−2 is two orders of magnitude greater than Keq for 1|PF6 in wet MeCN-d3. Weaker ion pairing was observed in samples containing NH4PF6 as an upfield shift of the 3,3′ protons compared to the complex in neat MeCN-d3 despite much higher [Cl−]. The increase in Keq and the weaker ion pairing can both be attributed to the formation of NH4Cl (which has limited solubility in MeCN) upon release of Cl−, pushing the substitution equilibrium towards 1-MeCN+.
To determine the reversibility of the bromide substitution of 2|PF6, TBABr was added to equilibrated samples of 2|PF6 in MeCN-d3 with and without TBAPF6. As expected, the addition of TBABr caused the equilibrium between 2 and 2-MeCN+ to shift back towards 2. As shown in Fig. S19, addition of one equivalent of TBABr (10 mM) results in a decrease in 2-MeCN+ in both samples, and a second equivalent (20 mM TBABr) almost completely reverses the dissociation reaction. The presence of additional Br− in solution results in a large downfield shift of the 3,3′ proton resonances due to strong ion pairing between tmam2+ and the halide. Notably, the presence of excess TBAPF6 in solution slowed the shift, suggesting competitive binding between PF6− and Br− and the binding pocket. Increasing the concentration of TBABr to 5 eq. in the 100 mM TBAPF6 sample led to the precipitation of 2|Br. Without excess PF6−, 2|Br began to precipitate with only two equivalents of TBABr.
 | ||
| Fig. 6 1H NMR spectra showing the changes to the tmam2+ ligand resonances of (a) 1|Cl at equilibrium with various amounts of added KCl and (b) 2|Br at equilibrium with various amounts of added NaBr. | ||
Equilibrated NMR samples of 1|Cl and 2|Br were titrated with halide salts to evaluate the reversibility of the halide substitution reaction. KCl was added to a sample of 1|Cl in D2O at equilibrium. As expected, the addition of one equivalent of KCl increased the concentration of 1 over the course of 90 minutes as the sample reached an equilibrium. Subsequent additions of KCl further shifted the equilibrium concentration of 1 as shown in Fig. 6. For 2 the Br− salt of the complex was used in titration experiments to avoid complications that could arise from the difference in the ion pairing strengths of Cl− and Br−. Increasing [Br−] with additions of NaBr resulted in an increase in the concentration of 2 compared to that of 2-D2O+, but 2-D2O+ remained the dominant species in solution up to 20 eq. of Br− (Fig. 6). For both 1 and 2, increasing [X−] in the solution resulted in downfield shifts of the 3,3′, and N(CH3)3 resonances in the tmam2+ binding pockets. The ability to shift the concentration of the equilibrium species confirms that a halide–solvent exchange reaction is in fact the equilibrium being observed. As a control experiment, TBABr was added in aliquots to 2|Br in DMSO-d6 (Fig. S23). Outside of the expected downfield shift of the protons in the binding pocket, no other changes were observed with increasing [Br−].
Variable temperature (VT) 1H NMR experiments were conducted to determine the enthalpic and entropic contributions to ΔG for the halide displacement reactions. VT experiments for 1|PF6 and 2|PF6 were frustrated by the slow interconversion between the parent and solvato complexes in MeCN. For both 1|Cl and 2|Br, the concentration of the halide-bound complex increased with increasing temperatures. The spectra and van't Hoff plots for 1|Cl and 2|Br are displayed in Fig. S24 and S25, respectively. Halide substitution is slightly exothermic for both complexes and larger for 2versus1 (ΔHrxn,1 = −4.32 ± 0.26 kJ mol−1; ΔHrxn,2 = −10.4 ± 1.0 kJ mol−1). The more favorable ΔHrxn for 2 can be rationalized by considering the greater hard–soft mismatch between Mn(I) and Br− than between Re(I) and Cl−. Both reactions display negative entropy of reaction (ΔSrxn,1 = −8.28 ± 0.88 J mol−1 K−1; ΔSxn,2 = −10.4 ± 3,3 J mol−1 K−1) suggesting an entropic penalty associated with solvation of the tricationic metal complexes and liberated halide, and the greater entropy cost for 2 is consistent with the larger solvation shell needed for the larger Br− anion.
The halide substitution equilibrium was also observed using UV-vis spectroscopy. Fig. 7 shows the progression of the absorption spectrum following injection of a DMSO stock solution of 1|Cl into H2O over the first two hours of the reaction. Isosbestic points are observed at 385 nm, 312, and 287 nm. Over time, the spectra show a blue-shift of the MLCT and peak π–π* absorptions and increasing prominence of the shoulders in tmam2+ π–π* transitions. The UV-vis spectrum of 2|Cl in H2O (Fig. S26) shows an MLCT absorption at 400 nm, which is consistent with other [Mn(diimine)(CO)3(H2O)]+ species in the literature, demonstrating complete conversion of 2 to 2-H2O+.32,34
The kinetics of the chloride substitution reaction between 1|Cl and D2O was monitored using 1H NMR. A time series of spectra is shown in Fig. 8a. Typically, under reaction conditions in which [1] ≪ [D2O], the forward rate should only depend on [1], and we expect a first-order dependence on [1]. However, the data were better fit by plotting [1]−1vs. t (Fig. 8b), indicating that the halide dissociation is second order in [1] and the involvement of a second equivalent of 1 in the halide substitution. One potential explanation for this second-order dependence is a bimolecular interaction shown in Fig. 8d. Such 1–1 dimers may be supported by favorable π-stacking interactions between the bpy core of the tmam2+ ligand and electrostatic interactions between the halides and the tmam2+ binding pockets. In this configuration, the pendant ammonium arms of the tmam2+ ligand of one complex could stabilize the release of the Cl− from the other. This hypothesis was based on the observation of a similar interaction between complexes of 1 in a crystal structure of 1|Cl grown in H2O with excess NaCl (Fig. 8c). However, disorder in cocrystallized H2O and NaCl prevented adequate refinement of the data, and this is only circumstantial evidence for the proposed mechanism. Furthermore, the NMR experiments were performed at a single concentration (10 mM), and it is possible that this cooperative reactivity is concentration dependent, and the second order mechanism only dominates at higher concentrations. The observation of halide substitution in UV-vis experiments—in which [1] ranged from 0.051 to 0.009 mM—shows that the reaction proceeds at much lower concentrations. Additional experimental work is required to provide a more complete analysis of this substitution mechanism. At this point, we cannot rule out alternative halide substitution mechanisms.
Electrochemical characterization
The CV profile of 1|PF6 (Fig. 9a) shows a broad irreversible reduction with Epc = −1.38 V and a second partially reversible reduction with E1/2 = −1.86 V. The use of the term “partially reversible” here indicates the observation of some return current on the reverse scan, but the redox couple is not strictly electrochemically reversible. Two, smaller reductions appear at more negative potentials; the first of these is irreversible (Epc = −2.05), and the second is partially reversible (E1/2 = −2.26 V). Return scans show a small anodic peak (Epc = −0.50 V) corresponding to the Re0–Re0 dimer.18,36 The reduction at −1.38 V has been previously assigned to be a 1-electron reduction of the tmam2+ ligand and results in ligand degradation. However, there is a cathodic shoulder of the main peak at −1.45 V which is visible at 100 mV s−1 and the peak height of the combined feature is ∼2 times that of the subsequent peak at all scan rates (50–1600 mV s−1). At scan rates > 400 mV s−1, the shoulder becomes the dominant peak, providing additional support for the assignment of the peak as two overlapping reductions. This behavior likely arises from sequential EC processes in which reduction (electrochemical step, E) localized on the tmam2+ ligand leads to reductive deamination of the tmam2+ ligand (chemical step C) to generate a monocationic ligand that can undergo a second EC process (generating a second equivalent of NMe3) at more negative potentials due to the reduced positive charge on the ligand. At low scan rates (ν ≤ 200 mV s−1), the voltammograms show little change when the voltage range is swept a second time, but when ν ≥ 400 mV s−1, the magnitude of the first reduction feature is significantly smaller in the second cycle (Fig. S30). Peak heights for the other features do not change with multiple scans. This is consistent with the assignment of the first cathodic feature as a ligand degradation process. At faster scan rates, the deaminated complexes produced in the first sweep do not have time to fully diffuse away from the electrode surface, and there is a lower concentration of 1 in the diffusion layer leading to decreased current for the reduction associated with the reductive deamination.
In CVs of Re(bpy)(CO)3Cl, substitution of the axial Cl− for MeCN produces a large positive shift in the Re-centered reduction, from −2.14 to −1.78 V, but the ligand-centered reduction is less sensitive to axial ligand substitution.36–38 For 1-MeCN+ (Fig. 9a), a small positive shift of the tmam-centered reductions is observed (Epc = −1.36 V). A new irreversible reduction with Epc = −1.69 V vs. Fc+/0 appears between the tmam2+ reductions, which may be a ReI/0 feature. A reduction at −1.84 V appears at very similar potentials to the corresponding reduction of 1. The peak at −2.23 V is consistent between CVs of 1 and 1-MeCN+, but the reduction at −2.05 V is no longer observed for 1-MeCN+. The conservation of the reduction at −1.84 V indicates that this is not a Re centered reduction. Instead, it is likely a reduction of the neutral, deaminated bipyridine ligand, and its reduction potential is very close to the ligand centered reduction of Re(dmb)(CO)3Cl (E1/2 = −1.89 V).5,38 This assignment agrees with the ligand degradation pathway proposed by Thoi et al. However, unambiguous assignment of the catalytically relevant redox events of 1 is made difficult by those reductions occurring at potentials cathodic of the reductive degradation of the ligand. Additionally, because the reductive deamination of the tmam2+ ligand may be accompanied by halide dissociation, we are unable to make claims regarding the effect of the ligand substitution equilibrium on the electrochemical behavior of these complexes, unless the identity of the axial ligand results in notable differences in the deamination events.
The CVs of 2|PF6 are shown in Fig. 9b. Cathodic scans reveal an irreversible, 2-electron reduction with Epc = −1.40 V, which is attributed to the tmam2+ reductive deamination. In this case, there is no apparent potential difference between the first and second EC steps. As with 1|PF6, a decrease in peak current is observed in subsequent scans following the first voltage sweep (Fig. S31). At more negative potentials, there is a broad, irreversible, cathodic feature (Epc = −1.66) and a partially reversible 1-electron reduction (E1/2 = −1.92 V). In the return scan, the dimer oxidation is observed at −0.69 V. Previous studies have established the reductive electrochemistry of Mn(dmb)(CO)3Br (dmb = 4,4′-dimethyl-2,2′-bipyridine). The complexes undergo an irreversible, 1-e−, L(π*)-localized reduction at Epc = −1.73 V, which induces rapid dissociation of that axial bromide, producing a Mn0(dmb)(CO)3 metalloradical species that then forms a Mn0–Mn0 dimer. Subsequently, the dimer is reduced in an irreversible, 2-e− process (Epc = −1.90 V), fragmenting the dimer into two [Mn(dmb)(CO)3]− species.39 The reported electrochemical behavior of [Mn(dmb)(CO)3(MeCN)]+ is nearly identical to its neutral analogue, (Mn(dmb)(CO)3Br).39 The first reduction is shifted 140 mV more positive, but the potential of the dimer reduction is unchanged. Similarly, voltammograms in Fig. 9b show that the electrochemical behavior of 2-MeCN+ is, broadly, like that of 2. The CVs of 2-MeCN+ (Fig. 9b) show two irreversible reductions near the potential at which the first tmam2+ reductions were observed for 2. Interestingly, in the reductive deamination, events are split in the solvato complex. The first reduction is shifted more positive than the 2-electron feature of 2 (Epc = −1.27 V), and the second reduction is at nearly the same potential as the corresponding reduction of 2 (Epc = −1.38 V). The irreversible reduction at −1.65 V vs. Fc+/0 is still present in the CVs of 2-MeCN+. A complicated mixture of overlapping reductions between −1.88 V and −2.05 V vs. Fc+/0 is observed at similar potentials to the Mn-centered, third reduction of 2. The splitting of this reduction into several new signals with similar potentials implies the presence of multiple Mn species in the diffusion layer.
The changes in the 2-electron reductive deamination of tmam2+ between the 2 and 2-MeCN+ complexes allowed for the observation of the halide substitution equilibrium on the electrochemical behavior of 2. The CVs of samples of 2 that were stored in electrolyte solution in the dark for 2 days prior to data collection were obtained and compared to those CVs taken immediately after sample preparation. Qualitatively, the CVs of the two samples are very similar; however, CVs of the equilibrated sample (Fig. 9b) show a small anodic shoulder at the foot of the 2-electron tmam2+ reduction (−1.27 V), which reveals the presence of 2-MeCN+. Indeed, this feature is often observed in samples of 2|PF6 within one hour of solution preparation. In agreement with observations via1H NMR and IR, this shoulder was not observed in samples that were allowed to reach an equilibrium in the presence of two equivalents of TBABr (Fig. S32). As noted above, further analysis of the effects of the ligand exchange on the electrochemistry of 2 is prevented by the irreversibility of the ligand degradation process.
The CV 2|Cl in N2 purged pH 4.75, 0.1 M NaClO4 (Fig. 10b) shows two overlapping, irreversible reductions at −1.01 V and −1.08 V. These peaks appear similar to peaks assigned to tmam2+ reductions for 1|PF6 in MeCN. At a scan rate of 100 mV s−1, no other reductions are visible on the first scan, but scans at 50 mV s−1 (Fig. S36) reveal an additional broad reduction at −1.22 V; this is likely a metal centered reduction. Unlike with 1, aqueous CVs of 2 show a clear dimer oxidation peak at −0.26 V. Significant changes in the voltammogram are observed on a second scan of the voltage range (Fig. 10b). In accordance with observations for 1, the tmam2+ reductions show diminished peak height after the first cycle, which is consistent with ligand degradation. Additionally, the Mn centered reduction becomes significantly more prominent and is shifted, cathodically, to −1.28 V in the second cycle. This behavior may arise from the conversion of multiple Mn species—which would result in multiple smaller peaks that could be lost in the baseline current—to a single, more well-defined species. This would also explain the observation of the more well-defined Mn-centered reduction at slower scan rates. CVs of 2 were also collected in 0.1 M NaClO4 with 5 mM and 20 mM of added NaBr. As with 1, the addition of NaBr had little effect on the CVs of 2 in aqueous solution (Fig. S37).
Conclusions
Complexes 1|X and 2|X have allowed us to investigate the effect of proximal positively charged groups and their counterions on the electronic, structural, and electrochemical characteristics of Re and Mn tricarbonyl complexes. Using the 1H NMR shifts of bipyridine protons as a measurement of anion binding strength with the cationic binding pockets of tmam2+, we observe that the size of the ion is a major factor in binding strength. The identity of the anion has little effect on the electronic structure of the complexes as evidenced by the lack of changes in the UV-vis spectra of the complexes. However, the cationic pendant groups of tmam2+ facilitate spontaneous exchange of the axial halide for solvent molecules in an equilibrium process. Equilibrium constants determined using 1H NMR spectroscopy show that the favorability of the exchange depends on the metal and the solvent. Halide exchange was only observed in MeCN and H2O, but not in DMSO or MeOH. Weaker anion-tmam ion-pairing in MeCN than in DMSO was observed in 1H NMR, and anion binding was still weaker in D2O. This is the inverse of the trend in halide lability for these complexes. The lower ion-pairing strength is also indicative of the stability of the solvated anion. Solvents that can better support the free anion pull the counterion away from the tmam2+, which may then drive the substitution in part to stabilize the empty cationic binding pockets.The strength of the M–X bond is also a key factor in halide substitution. Thermodynamic parameters obtained from VT-NMR studies of 1|Cl and 2|Br in H2O demonstrate that halide substitution is more enthalpically favored for 2 than 1. The greater hard–soft mismatch in Mn–Br likely contributes to the more favorable halide dissociation observed for Mn than Re. Additional studies using a series of Re–X complexes (X = Cl, Br, I) would be required to better understand this aspect of chemistry. Time course 1H NMR data for 1 in D2O suggest that the ligand substitution is second-order in 1, which suggests an intermolecular process in which halide dissociation from one complex is stabilized by the dicationic binding pocket of a separate complex. Our hypothesis is based on the observation of this interaction in low-quality X-ray crystal structures of 1. A more detailed study would be required to confirm this hypothesis.
The observed halide substitution reaction had little effect on the observed electrochemical response of complexes 1 and 2 in both MeCN and H2O. This is due to the reductive instability of the tmam2+ ligand, which is observed to undergo reductive deamination at potentials anodic of the ligand- and metal-centered reductions of the catalyst. If halide dissociation occurs alongside the reductive deamination (producing a solvato complex), one would expect the potential of subsequent reductions to have little dependence on the identity of the axial ligand in the bulk sample. An alternative ligand framework is required to study the potential effects of pendant group-assisted ligand substitution on the electrochemical and electrocatalytic behavior of these catalysts.
The installation of pendant functional groups that can participate in catalytic reactions through the secondary coordination sphere has been fruitful in improving the performance of molecular electrocatalysts for a variety of reactions. This study shows that these functional groups can produce unexpected reactivity. Our examination into the effects of the cationic pendant groups of the tmam2+ ligand on the coordination chemistry of Re and Mn M(DI)(CO)3X complexes has shown that these groups are noninnocent in their interactions with the X-type halide ligand. Understanding these interactions can suggest how these functional groups support catalytic reactions by participating in catalyst activation and their ability to stabilize charged catalytic intermediates.
Author contributions
WK, BS, and TS contributed to the conceptualization and supervision of this work. They also contributed to the methodology employed in this work. WK, BS, TS, and JPV contributed as investigators and validators for the synthesis and spectroscopic characterization of compounds reported herein. WK, TS, JPV, CB, AB, and CL contributed as investigators for the electrochemical experiments. WK wrote the original draft of this manuscript. WK, BS and TS contributed to reviewing and editing the written manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information (SI): experimental details, spectroscopic data used for characterization of complexes (UV-vis, FTIR, 1H NMR), 1H NMR data demonstrating halide substitution for all complexes as detailed in the text, and additional electrochemical characterization for all complexes. See DOI: https://doi.org/10.1039/d5dt02433a.All experimental data are available upon request from the corresponding author: (wkramer@hamilton.edu).
CCDC 2494313–2494315 contain the supplementary crystallographic data for this paper.46a–c
Acknowledgements
This project was conceived and initial work was performed in the lab of Prof. Harry B. Gray and funded by NSF CCI Solar Fuels (CHE-130124). Additional support was provided at Caltech and a Dr and Mrs Daniel C. Harris SURF Fellowship. The authors acknowledge Harry B. Gray for his unwavering support and exceptional mentorship. The authors also acknowledge Brendon J. McNicholas for his helpful contributions during the writing of this manuscript, and we thank Larry Henling and Michael Takase for their assistance with X-ray crystallography experiments.References
- J. Hawecker, J. M. Lehn and R. Ziessel, Helv. Chim. Acta, 1986, 69, 1990–2012 CrossRef CAS.
- J. Hawecker, J.-M. Lehn and R. Ziessel, J. Chem. Soc., Chem. Commun., 1984, 328–330 RSC.
- J. M. Smieja, M. D. Sampson, K. A. Grice, E. E. Benson, J. D. Froehlich and C. P. Kubiak, Inorg. Chem., 2013, 52, 2484–2491 CrossRef CAS PubMed.
- L. Rotundo, E. Azzi, A. Deagostino, C. Garino, L. Nencini, E. Priola, P. Quagliotto, R. Rocca, R. Gobetto and C. Nervi, Front. Chem., 2019, 7, 417 CrossRef CAS PubMed.
- M. L. Clark, P. L. Cheung, M. Lessio, E. A. Carter and C. P. Kubiak, ACS Catal., 2018, 8, 2021–2029 CrossRef CAS.
- G. F. Manbeck, J. T. Muckerman, D. J. Szalda, Y. Himeda and E. Fujita, J. Phys. Chem. B, 2015, 119, 7457–7466 CrossRef CAS PubMed.
- M. D. Sampson, A. D. Nguyen, K. A. Grice, C. E. Moore, A. L. Rheingold and C. P. Kubiak, J. Am. Chem. Soc., 2014, 136, 5460–5471 CrossRef CAS PubMed.
- F. Franco, C. Cometto, L. Nencini, C. Barolo, F. Sordello, C. Minero, J. Fiedler, M. Robert, R. Gobetto and C. Nervi, Chem. – Eur. J., 2017, 23, 4782–4793 CrossRef CAS PubMed.
- F. Franco, C. Cometto, F. F. Vallana, F. Sordello, E. Priola, C. Minero, C. Nervi and R. Gobetto, Chem. Commun., 2014, 50, 14670–14673 RSC.
- L. Rotundo, C. Garino, E. Priola, D. Sassone, H. Rao, B. Ma, M. Robert, J. Fiedler, R. Gobetto and C. Nervi, Organometallics, 2018, 38, 1351–1360 CrossRef.
- I. Siewert, Acc. Chem. Res., 2022, 55, 473–483 CrossRef CAS PubMed.
- I. Azcarate, C. Costentin, M. Robert and J.-M. Savéant, J. Am. Chem. Soc., 2016, 138, 16639–16644 CrossRef CAS PubMed.
- L. Rotundo, S. Ahmad, C. Cappuccino, D. E. Polyansky, M. Z. Ertem and G. F. Manbeck, Inorg. Chem., 2023, 62, 7877–7889 CrossRef CAS PubMed.
- L. Rotundo, S. Ahmad, C. Cappuccino, A. J. Pearce, H. Nedzbala, S. R. Bottum, J. M. Mayer, J. F. Cahoon, D. C. Grills, M. Z. Ertem and G. F. Manbeck, J. Am. Chem. Soc., 2024, 146, 24742–24747 CrossRef CAS PubMed.
- S. Sung, X. Li, L. M. Wolf, J. R. Meeder, N. S. Bhuvanesh, K. A. Grice, J. A. Panetier and M. Nippe, J. Am. Chem. Soc., 2019, 141, 6569–6582 CrossRef CAS PubMed.
- S. Sung, D. Kumar, M. Gil-Sepulcre and M. Nippe, J. Am. Chem. Soc., 2017, 139, 13993–13996 CrossRef CAS PubMed.
- N. R. Crudo, A. Sonea, L. M. Karn, D. B. Leznoff and J. J. Warren, ACS Catal., 2025, 15, 14293–14304 CrossRef CAS.
- S. S. Saund, M. A. Siegler and V. S. Thoi, Inorg. Chem., 2021, 60, 13011–13020 CrossRef CAS PubMed.
- L. Troian-Gautier, S. A. M. Wehlin and G. J. Meyer, Inorg. Chem., 2018, 57, 12232–12244 CrossRef CAS PubMed.
- W. B. Swords, G. Li and G. J. Meyer, Inorg. Chem., 2015, 54, 4512–4519 CrossRef CAS PubMed.
- S. A. M. Wehlin, L. Troian-Gautier, R. N. Sampaio, L. Marcélis and G. J. Meyer, J. Am. Chem. Soc., 2018, 140, 7799–7802 CrossRef CAS PubMed.
- S. A. M. Wehlin, L. Troian-Gautier, G. Li and G. J. Meyer, J. Am. Chem. Soc., 2017, 139, 12903–12906 CrossRef CAS PubMed.
- L. Troian-Gautier, W. B. Swords and G. J. Meyer, Acc. Chem. Res., 2018, 52, 170–179 CrossRef PubMed.
- A. M. Deetz, M. J. Goodwin, E. A. Kober and G. J. Meyer, Inorg. Chem., 2023, 62, 11414–11425 CrossRef CAS PubMed.
- L. Troian-Gautier, E. E. Beauvilliers, W. B. Swords and G. J. Meyer, J. Am. Chem. Soc., 2016, 138, 16815–16826 CrossRef CAS PubMed.
- J. V. Caspar and T. J. Meyer, J. Phys. Chem., 1983, 87, 952–957 CrossRef CAS.
- J. M. Smieja and C. P. Kubiak, Inorg. Chem., 2010, 49, 9283–9289 CrossRef CAS PubMed.
- M. D. Sampson and C. P. Kubiak, J. Am. Chem. Soc., 2016, 138, 1386–1393 CrossRef CAS PubMed.
- M. Jakonen, L. Oresmaa and M. Haukka, Cryst. Growth Des., 2007, 7, 2620–2626 CrossRef CAS.
- M. A. Bento, N. A. G. Bandeira, H. N. Miras, A. J. Moro, J. C. Lima, S. Realista, M. Gleeson, E. J. Devid, P. Brandão, J. Rocha and P. N. Martinho, Inorg. Chem., 2024, 63, 18211–18222 CrossRef CAS PubMed.
- T. Pivarcsik, J. Kljun, S. C. Rodriguez, D. C. Alcaraz, U. Rapuš, M. Nové, E. F. Várkonyi, J. Nyári, A. Bogdanov, G. Spengler, I. Turel and E. A. Enyedy, ACS Omega, 2024, 9, 44601–44615 CrossRef CAS PubMed.
- J. J. Walsh, G. Neri, C. L. Smith and A. J. Cowan, Organometallics, 2019, 38, 1224–1229 CrossRef CAS.
- C. P. Kelly, C. J. Cramer and D. G. Truhlar, J. Phys. Chem. B, 2007, 111, 408–422 CrossRef CAS PubMed.
- T. N. Twala, M. Schutte-Smith, A. Roodt and H. G. Visser, Dalton Trans., 2015, 44, 3278–3288 RSC.
- D. C. Grills, M. Z. Ertem, M. McKinnon, K. T. Ngo and J. Rochford, Coord. Chem. Rev., 2018, 374, 173–217 CrossRef CAS.
- B. P. Sullivan, C. M. Bolinger, D. Conrad, W. J. Vining and T. J. Meyer, J. Chem. Soc., Chem. Commun., 1985, 1414–1416 RSC.
- M. McKinnon, K. T. Ngo, S. Sobottka, B. Sarkar, M. Z. Ertem, D. C. Grills and J. Rochford, Organometallics, 2018, 38, 1317–1329 CrossRef.
- A. I. Breikss and H. D. Abruña, J. Electroanal. Chem. Interfacial Electrochem., 1986, 201, 347–358 CrossRef CAS.
- M. Bourrez, F. Molton, S. Chardon-Noblat and A. Deronzier, Angew. Chem., Int. Ed., 2011, 50, 9903–9906 CrossRef CAS PubMed.
- S. Sinha, A. Sonea, W. Shen, S. S. Hanson and J. J. Warren, Inorg. Chem., 2019, 58, 10454–10461 CrossRef CAS PubMed.
- S. Sinha, A. Sonea, C. A. Gibbs and J. J. Warren, Dalton Trans., 2020, 49, 7078–7083 RSC.
- E. Gala, G. C. D. Bandomo, M. Vettori, S. Royuela, M. Martínez-Fernández, J. I. Martínez, E. Salagre, E. G. Michel, F. Zamora, J. Lloret-Fillol and J. L. Segura, J. Mater. Chem. A, 2025, 13, 1142–1152 RSC.
- A. Zhanaidarova, S. C. Jones, E. Despagnet-Ayoub, B. R. Pimentel and C. P. Kubiak, J. Am. Chem. Soc., 2019, 141, 17270–17277 CrossRef CAS PubMed.
- C. Das, S. Ghosh, R. Biswas, G. K. Lahiri and A. Dutta, Chem. Commun., 2024, 60, 10492–10495 RSC.
- A. Nakada and O. Ishitani, ACS Catal., 2017, 354–363 Search PubMed.
- (a) CCDC 2494313: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pqjqp; (b) CCDC 2494314: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pqjrq; (c) CCDC 2494315: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pqjsr.
Footnotes |
| † Current institution: Sandia National Laboratory, Albuquerque, NM 87123, USA. |
| ‡ Current institution: Department of Chemistry, University of Michigan, Ann Arbor, MI 48109, USA. |
| § Current Institution: Department of Chemistry, University of Rochester, Rochester, NY 14627, USA. |
| ¶ Current institution: Biosciences Division, Oak Ridge National Laboratory, Oak Ridge, TN 37830, USA. |
| This journal is © The Royal Society of Chemistry 2026 |