Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
A change in ligand hapticity promotes Lewis base dissociation
Payton A.
Fortuna†
a,
Leslie S. G.
Kelley†
a,
Yipeng
Sun
b,
Jiabin
Xu
a,
Zhiqiang
Wang
 a,
Paul D.
Boyle
a,
Paul D.
Boyle
 a,
Tsun-Kong
Sham
a,
Tsun-Kong
Sham
 *a,
Viktor N.
Staroverov
*a,
Viktor N.
Staroverov
 *a and
Johanna M.
Blacquiere
*a and
Johanna M.
Blacquiere
 *a
*a
aDepartment of Chemistry, The University of Western Ontario, London, N6A 3K7, Canada. E-mail: tsham@uwo.ca; vstarove@uwo.ca; johanna.blacquiere@uwo.ca
bDepartment of Mechanical and Materials Engineering, The University of Western Ontario, London, N6A 5B9, Canada
First published on 22nd January 2026
Abstract
We report a phosphine 1-azaallyl ligand L2 [2-(diphenylphosphino)-2′-(3,3-dimethyl-1-azaallyl)-1,1′-biphenyl], which enables reversible coordination of Lewis bases through changes in ligand hapticity. In the palladium methyl complex [Pd(CH3)(L2)], L2 adopts a κ1-P;η3-NCC coordination mode. Pyridine binding induces reorganization to a κ2-PN mode, while reversion to the κ1-P;η3-NCC mode facilitates pyridine dissociation. These interconversions were established by 13C{1H} and 1H–31P HMBC NMR spectroscopy, X-ray absorption spectroscopy, and density-functional theory calculations. The results highlight a ligand-controlled mechanism for reversible substrate coordination based on tunable binding modes.
Introduction
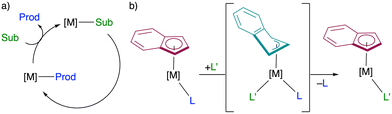
Ligand substitution is a fundamental reaction step that is common among nearly all mechanisms of transition metal catalysis.1,2 Catalyst turnover requires that product molecules generated at the metal centre be displaced by an incoming substrate (Fig. 1a). Mechanistically, this can occur through either associative or dissociative pathways.3 In some cases, this step is rate-limiting, and strong product binding can inhibit catalytic activity. Consequently, ligand features that promote efficient product dissociation without compromising substrate binding are highly desirable in the catalyst design.Several ligand characteristics are known to facilitate substitution reactions. For example, dissociation can be enhanced through electronic effects associated with strong trans-effect ligands or through steric congestion imposed by bulky ancillary ligands.1,2 Hemilabile ligands have also been widely explored as a strategy to promote substitution chemistry, as low barrier isomerization between different coordination modes can reversibly open and close coordination sites at the metal centre.4–7 Classical hemilabile ligands involve changes in denticity (e.g. κ2 ↔ κ1, κ3 ↔ κ2), but other strategies to alter metal coordination properties include changes in hapticity or switches in donor type arising from distinct ligand resonance forms.4,8–11 A canonical example of the former is the indenyl ligand, which facilitates associative substitution reactions via ring slippage (i.e., η5 ↔ η3; Fig. 1b).12–14
The 1-azaallyl ligand has a four-electron delocalized π-system, across an anionic NCC fragment, that engenders a variety of coordination modes, with one or two coordination sites at a metal.15 Lappert demonstrated that Ni(II) and Pd(II) bis(1-azaallyl) complexes exhibit η3-NCC ligand coordination, while chemical exchange of the ligand substituents was rationalized based on an isomerization via an η1-N (i.e., κ1-N) coordination mode.16 This change in hapticity means the ligand readily toggles between coordination to one and two sites on the metal. To take advantage of this versatile coordination chemistry, we have integrated the 1-azaallyl moiety into a ligand structure that also includes a monodentate phosphine donor (L1, Fig. 2).17 The ligand readily binds to Ru(II) or Pd(II) metal fragments through a bidentate mode (i.e., κ2-PN; Fig. 2, e.g. Pd(II), A).17,18 This mode was enforced through the metal coordination geometry and the presence of excess donor ligands (pyridine molecules). In the absence of donor ligands, the 1-azaallyl fragment stabilizes a Pd–Me moiety by µ-N bridging in complex B.18 Ligation of L1 to a Ru–Cp* fragment (Cp* = 1,2,3,4,5-pentamethylcyclopentadienyl) afforded C, which does not have a placeholder ligand, and the 1-azaallyl group exhibited rapid exchange ascribed to an η3-NCC ↔ κ1-N interconversion.19 While the κ1-P;η3-NCC coordination mode of L1 was supported by electronic structure calculations, it had not been observed spectroscopically. Presumably, the relatively small P–M–N bite angle is preferrable for κ2-PN, rather than κ1-P;η3-NCC, coordination.
 | ||
| Fig. 2 Previously reported phosphine 1-azaallyl ligand L1, corresponding complexes A–C, and the new ligand L2 reported herein. | ||
Herein, we report preparation of a new phosphine 1-azaallyl ligand (L2), which could more readily access a κ1-P;η3-NCC coordination mode. We targeted a Pd(II) complex, both since this metal is frequently used as a model system for substitution chemistry and because it has broad relevance in catalysis. The coordination chemistry of L2 was evaluated through a range of spectroscopic and computational means. The impact of the coordination mode on substitution chemistry was probed, and experiments reveal ligand L2 promotes Lewis base dissociation.
Results and discussion
Synthesis of ligands and complexes
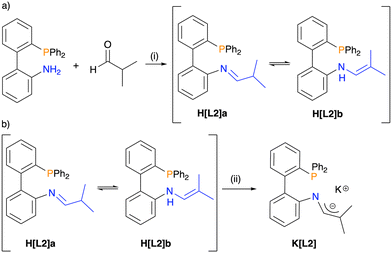
A condensation reaction between 2′-(diphenylphosphanyl)-[1,1′-biphenyl]-2-amine and isobutyraldehyde was performed to afford H[L2], as a white solid with an average yield of 87% (Scheme 1a). The 31P{1H} NMR spectrum of the isolated product in CDCl3 has two distinct singlets at δP = –12.4 and −13.3 in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 ratio. The 1H NMR spectrum also exhibits two sets of signals for product H[L2]. One set includes a doublet at δH = 7.55 consistent with an imine proton, as well as a doublet of septets at δH = 2.39 and a broad multiplet in the range of δH = 0.97–0.89 that were assigned to an iso-propyl group. These signals support the presence of the expected imine tautomer, H[L2]a. The second set of signals includes two singlets at δH = 1.69 and 1.36, assigned to two inequivalent methyl groups, and two doublets at δH = 6.08 and 4.94 correspond to vinyl and N–H protons, respectively. This pattern is consistent with the enamine tautomer, H[L2]b.
2 ratio. The 1H NMR spectrum also exhibits two sets of signals for product H[L2]. One set includes a doublet at δH = 7.55 consistent with an imine proton, as well as a doublet of septets at δH = 2.39 and a broad multiplet in the range of δH = 0.97–0.89 that were assigned to an iso-propyl group. These signals support the presence of the expected imine tautomer, H[L2]a. The second set of signals includes two singlets at δH = 1.69 and 1.36, assigned to two inequivalent methyl groups, and two doublets at δH = 6.08 and 4.94 correspond to vinyl and N–H protons, respectively. This pattern is consistent with the enamine tautomer, H[L2]b.
Deprotonation of H[L2] with K[HMDS] was performed to afford the phosphine 1-azaallyl ligand K[L2], as an orange solid in a 76% yield (Scheme 1b). The 31P{1H} NMR spectrum of K[L2] has one singlet at δP = –13.6, confirming deprotonation of both tautomers to give one species. Successful deprotonation is further confirmed by the 1H NMR spectrum of K[L2], where the methine proton of H[L2]a and the N–H proton of H[L2]b are no longer observed.
The tautomeric mixture of the neutral ligand H[L2]a/H[L2]b was coordinated to [PdCl(CH3)(COD)], affording complex 1 in 67% isolated yield (Scheme 2). The 31P{1H} NMR spectrum of the product shows two singlets at δP = 36.2 and 35.8 in a 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (CDCl3). The 1H NMR spectrum also displays patterns for both imine and enamine moieties, indicating that the two species correspond to the two tautomeric forms 1a and 1b, respectively. The diagnostic doublets for the Pd-bound methyls for 1a (0.47 ppm) and 1b (0.61 ppm) have coupling constants of 3JHP = 3.4 and 3.6 Hz, respectively, which are consistent with a cis orientation of the phosphine and methyl for both tautomeric products.18,20–22
1 ratio (CDCl3). The 1H NMR spectrum also displays patterns for both imine and enamine moieties, indicating that the two species correspond to the two tautomeric forms 1a and 1b, respectively. The diagnostic doublets for the Pd-bound methyls for 1a (0.47 ppm) and 1b (0.61 ppm) have coupling constants of 3JHP = 3.4 and 3.6 Hz, respectively, which are consistent with a cis orientation of the phosphine and methyl for both tautomeric products.18,20–22
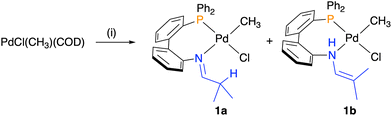 | ||
Scheme 2 Synthesis of the tautomeric mixture of palladium complexes 1a/1b. Conditions: (i) 1.2 equiv. tautomeric mixture of ligand H[L2], CH2Cl2, rt, 24 h. Product ratio 1a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1b = 17 1b = 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. 1. | ||
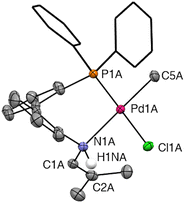
Single crystals were grown via slow vapour diffusion of pentane into a solution of 1 in a 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of toluene and CH2Cl2 at −20 °C. X-ray diffraction revealed only the enamine tautomer 1b (Fig. 3). The crystal structure contains two symmetry-independent molecules in the asymmetric unit, which are enantiomers and are designated as complex A and complex B. The structural parameters of only complex A will be discussed here since A and B have identical connectivity and very similar bond lengths and angles. The structure confirms a square planar geometry at the palladium centre, with very minimal distortion23 according to the τ4 value of 0.07. A cis orientation of the phosphine and methyl substituents is observed, which is consistent with the NMR spectroscopic data. The assignment of the structure as the enamine tautomer 1b is supported by the bond lengths of N(1A)–C(1A) = 1.440(2) Å and C(1A)–C(2A) = 1.332(3) Å, which are characteristic of a C–N single bond and a C–C double bond, respectively. The P(1A)–Pd–N(1A) bite angle was determined to be 95.18(5)° (for B = 95.97(5)°), which is significantly wider than the angle of 78.48° found for the analogous complex with the monoaryl linked phosphine imine ligand H[L1].4 Therefore, H[L2] imparts less conformational strain on the metal centre and may allow for isolation of a Pd phosphine 1-azaallyl species in which the anionic ligand L2 is coordinated in a κ1-P;η3-NCC mode.
1 mixture of toluene and CH2Cl2 at −20 °C. X-ray diffraction revealed only the enamine tautomer 1b (Fig. 3). The crystal structure contains two symmetry-independent molecules in the asymmetric unit, which are enantiomers and are designated as complex A and complex B. The structural parameters of only complex A will be discussed here since A and B have identical connectivity and very similar bond lengths and angles. The structure confirms a square planar geometry at the palladium centre, with very minimal distortion23 according to the τ4 value of 0.07. A cis orientation of the phosphine and methyl substituents is observed, which is consistent with the NMR spectroscopic data. The assignment of the structure as the enamine tautomer 1b is supported by the bond lengths of N(1A)–C(1A) = 1.440(2) Å and C(1A)–C(2A) = 1.332(3) Å, which are characteristic of a C–N single bond and a C–C double bond, respectively. The P(1A)–Pd–N(1A) bite angle was determined to be 95.18(5)° (for B = 95.97(5)°), which is significantly wider than the angle of 78.48° found for the analogous complex with the monoaryl linked phosphine imine ligand H[L1].4 Therefore, H[L2] imparts less conformational strain on the metal centre and may allow for isolation of a Pd phosphine 1-azaallyl species in which the anionic ligand L2 is coordinated in a κ1-P;η3-NCC mode.
Coordination of L2 with palladium
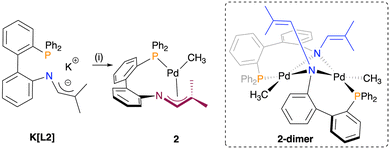
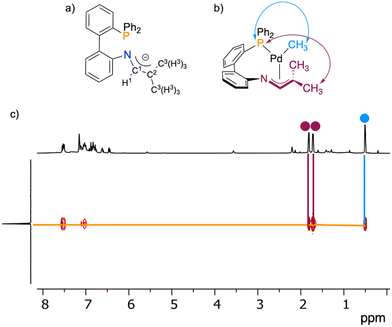
An initial strategy to access a phosphine 1-azaallyl complex involved deprotonation of the tautomeric mixture of the phosphine imine/enamine complexes 1a/1b. However, these attempted deprotonation reactions produced complex reaction mixtures as evidenced by multiple signals in the 31P{1H} NMR spectrum, inconsistent with a well-defined product.We therefore pursued an alternative route, in which the deprotonated ligand salt K[L2] was reacted with [PdCl(CH3)(COD)] to afford an orange product 2 in a 70% yield (Scheme 3). The 31P{1H} NMR spectrum of isolated 2 has one singlet at δP = 25.7, consistent with phosphine coordination and formation of a single product. Several attempts were made to afford X-ray quality crystals of 2, but without success. However, the combination of NMR spectroscopy and X-ray absorption spectroscopy supported a κ1-P;η3-NCC coordination mode of L2, which corresponds to mononuclear 2. This supporting evidence is as follows.
The chemical shifts of the carbon atoms of the 1-azaallyl fragment are highly diagnostic of metal coordination mode.17 A combination of 1H–13C HSQC and 1H–13C HMBC allowed assignment of C1 and C2 of 2 at δC = 129.4 and 88.4, respectively (see Fig. 4a for atom numbering). By comparison, in complexes of L1 where the 1-azaallyl group coordinates to the metal exclusively through nitrogen, the corresponding C1 and C2 resonances appear in the ranges 135–142 and 115–119 ppm, respectively.17–19 The observed values for 2 fall well outside these ranges.
Among these signals, C2 is particularly sensitive to the coordination environment. Its pronounced upfield shift in 2 is indicative of direct metal coordination at this carbon,16,24 consistent with a κ1-P;η3-NCC binding mode. Furthermore, the resonances for C1, C2, and C3 are all observed as doublets due to coupling to phosphorus, whereas the corresponding signals in the previously reported dinuclear B are singlets.
Additional support for the κ1-P;η3-NCC mode was obtained from 1H–31P HMBC NMR spectroscopy (Fig. 4b and c). In this binding mode, protons H3/H3′ are within four bonds to the phosphine donor, which would be expected to give rise to an observable correlation. Such a correlation is indeed present for 2. A conceivable alternative product of the reaction of K[L2] with [PdCl(CH3)(COD)] is 2-dimer, a complex analogous to B18 (see Fig. 2). However, 2-dimer would place H3/H3′ six bonds away from phosphorus, which would not give rise to a 1H–31P HMBC NMR correlation. Consistent with this expectation, dinuclear B shows no correlation between H3/H3′ and P in the 1H–31P HMBC NMR spectrum.18 For the same reasons, a mononuclear THF solvate, [Pd(CH3)(κ2-PN-L2)(THF)], is also inconsistent with the observed spectroscopic data. Collectively, the NMR spectroscopic data supports assignment of L2 as adopting a κ1-P;η3-NCC coordination mode, corresponding to assignment as the mononuclear structure 2.
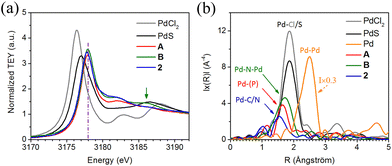
We pursued analysis of 2 by X-ray absorption spectroscopy (XAS) to further corroborate the assignment of L2 coordination as κ1-P;η3-NCC, and to identify any unique properties of the palladium centre due to this coordination mode (Fig. 5). In addition to complex 2, two other comparator Pd(II) complexes, A and B (see Fig. 2), coordinated by phosphine 1-azaallyl ligand L1 were also studied. These compounds were selected since they exhibit distinct coordination modes of the 1-azaallyl group, in which A has κ1-N and B has µ-N binding. The Pd L3-edge XANES, the Pd K-edge EXAFS and the P K-edge XANES have been recorded and analyzed; the results are highlighted in Fig. 5, Fig. S52 and 53.
The Pd L3-edge XANES shows sharp peaks for 2, A and B around 3178 eV due to a 2p3/2 → 4d transition (Fig. 5a). As expected, these peaks are found at a higher energy than those for the Pd(II) reference samples PdCl2 and PdS, in which the transition promotes an electron into the conduction band of the solid-state materials. Compounds A and B have experimentally indistinguishable energy (3178.0 eV), while that for 2 is slightly lower (3177.9 eV). This suggests 2 is slightly more electrophilic, which may be a consequence of Pd coordination to the π-system of the 1-azaallyl fragment. Compound B exhibits an additional broad peak at ∼3186 eV (marked by a green arrow), higher in relative energy to the 2p3/2 → 4d transition. This energy range is typical for transitions due to multiple scattering from close proximity high-Z elements, which would be expected for the dinuclear Pd–N–Pd motif. The absence of a similar peak for 2 further discounts a bridging coordination mode for L2 in 2 that would lead to a dinuclear structure.
The Pd K-edge XANES and EXAFS were acquired for 2, A, B and references, and the Fourier transformed (FT) data was plotted in radial space (Fig. 5b). The Pd K-edge XANES is less sensitive than the Pd L3-edge because the K edge has a much shorter core-hole lifetime than the L edge and has corresponding broader linewidths in XANES. However, the Pd K-edge EXAFS can still provide useful information on the local structure of Pd atoms. The dominant local bonding interaction is labelled for each sample. It should be noted that C and N have similar backscattering amplitude (y axis) and cannot be distinguished in the FT, as are P, S and Cl (the backscattering amplitude increases as the atomic number increases).
The known bonding environments for A and B both include phosphine, methyl and two N donors. They differ in that the N donors in A are Pd–N(amido) and Pd–N(pyridine), while in B they are two bridging amidos (i.e., Pd–N(amido)–Pd). Thus, the compounds give similar signatures, but the signal for B is observed at a slightly longer distance and higher amplitude. This could be due to the bridging nitrogens acting as lenses. As compared to A and B, the signal for 2 is notably at a shorter distance, has a lower amplitude, and has a shoulder as compared to A or B. A signal like that of B would be expected if 2 was also a bridging dinuclear complex, so the observed signal with dominant contribution from Pd–C/N further supports the assignment of 2 as a mononuclear compound supported by η3-NCC coordination mode of the 1-azaallyl fragment.
Substitution chemistry with complex 2
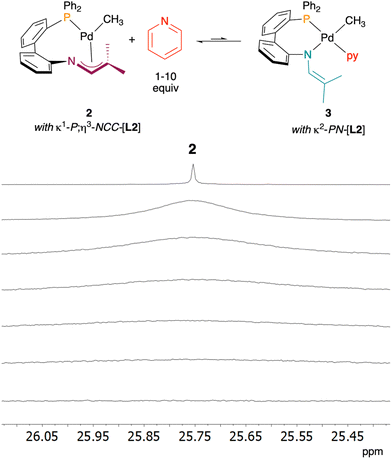
Our prior work with the Ru(II) phosphine 1-azaallyl complex C revealed that L1 engages in a dynamic equilibrium between κ2-PN and κ1-P;η3-NCC coordination modes (see Fig. 2).19 This reversibility permitted facile coordination of Lewis basic donors to afford isolable coordinatively saturated complexes of the type Ru(Cp*)(κ2-PN-L1)L (L = pyridine, PPh3). Similarly, the dinuclear palladium complex B with κ1-P;µ-N-(L1) readily reacts with pyridine to give the stable mononuclear adduct A.18 Based on these prior studies, we hypothesized that Lewis basic donors would bind to 2, and this would be accommodated by a change in L2 coordination mode from κ1-P;η3-NCC to κ2-P,N.Complex 2 was treated with pyridine, and reactions were analyzed by 31P{1H} and 1H NMR spectroscopy (Fig. 6, Fig. S2 and 3). The addition of 1 equivalent resulted in the broadening of the 31P signal for 2 as well as many of the 1H signals, indicative of equilibrium binding of pyridine. Increasing the pyridine equivalents up to 10 resulted in further broadening of both 1H and 31P signals. While signal broadening is consistent with chemical dynamics, the specific behaviour in this case is somewhat atypical. The broad signal is consistent with the fast exchange regime (i.e. relatively low-barrier exchange), for which the observed chemical shift should be a weighted average of the two species participating in the exchange.25 In this case, we observe no notable difference in chemical shift between that of 2 and the broadened signal for 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 2 and pyridine. We hypothesize that this is due to a very small Keq value that strongly favours the reactants, and therefore the weighted average chemical shift is dominated by the value for 2. This hypothesis is consistent with the observation that even 10 equiv. pyridine does not drive the equilibrium sufficiently to the proposed product 3. Nevertheless, we considered that the disappearance of a 31P signal could be due to formation of a paramagnetic product. An Evans analysis of a 1
1 2 and pyridine. We hypothesize that this is due to a very small Keq value that strongly favours the reactants, and therefore the weighted average chemical shift is dominated by the value for 2. This hypothesis is consistent with the observation that even 10 equiv. pyridine does not drive the equilibrium sufficiently to the proposed product 3. Nevertheless, we considered that the disappearance of a 31P signal could be due to formation of a paramagnetic product. An Evans analysis of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of 2 and pyridine did not reveal signal shifting due to paramagnetism (Fig. S4). Additionally, to confirm that 2 is an operative species in the observed equilibrium we designed an experiment to remove pyridine from the equilibrium mixture to regenerate 2. A 1
1 mixture of 2 and pyridine did not reveal signal shifting due to paramagnetism (Fig. S4). Additionally, to confirm that 2 is an operative species in the observed equilibrium we designed an experiment to remove pyridine from the equilibrium mixture to regenerate 2. A 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of 2 and pyridine was treated with excess B(C6F5)3 to sequester pyridine in a Lewis acid base adduct (py → B(C6F5)3). Sequestration was effective and 2 was regenerated, which confirmed that 2 is involved in the operative equilibrium (Fig. S6).
1 mixture of 2 and pyridine was treated with excess B(C6F5)3 to sequester pyridine in a Lewis acid base adduct (py → B(C6F5)3). Sequestration was effective and 2 was regenerated, which confirmed that 2 is involved in the operative equilibrium (Fig. S6).
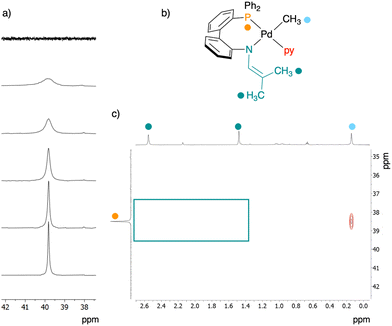
Evidence for the expected pyridine adduct 3 was obtained by variable temperature NMR spectroscopy. Dissolution of 2 in pyridine-d5 at 25 °C resulted in a 31P{1H} spectrum with no observable signal (Fig. 7a, top spectrum). The 1H NMR spectrum revealed a set of signals in which those of the 1-azaallyl fragment were noticeably broadened. Cooling the sample to −30 °C sharpened the 1H signals, and a singlet emerged in the 31P{1H} NMR spectrum at 39.8 ppm (Fig. 7, Fig. S7 and 8). Low temperature 1H and 2D NMR (1H–31P HMBC, 1H–1H COSY, 1H–13C HSQC, and 1H–13C HMBC) spectroscopy data were consistent with the formation of the pyridine adduct 3. Notably, no correlation was observed between phosphorus and the protons of the 1-azaallyl methyl substituents H3/H3′ (Fig. 7b and c). Both C1 and C2 were shifted downfield relative to their location in 2 (3: 142 and 92 ppm, respectively). Taken together, this data indicates that L2 is in the κ2-PN coordination mode and that pyridine occupies a coordination site on the metal (i.e.3). The signal at δP = 39, assigned to 3, was also observed below 0 °C for a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 mixture of 2 and pyridine in toluene-d8 (Fig. S13). To confirm that the low temperature product is the pyridine adduct 3, and not 2-dimer, VT NMR spectra of 2 in toluene-d8 were collected. No signal at 39 ppm emerged in the temperature range of 25 to −80 °C (Fig. S15 and S16).
4 mixture of 2 and pyridine in toluene-d8 (Fig. S13). To confirm that the low temperature product is the pyridine adduct 3, and not 2-dimer, VT NMR spectra of 2 in toluene-d8 were collected. No signal at 39 ppm emerged in the temperature range of 25 to −80 °C (Fig. S15 and S16).
The fact that pyridine adduct 3 is only directly observed at low temperatures and in the presence of excess pyridine is remarkable given that isolation of the related pyridine adduct [PdCH3(BINAP)(py)]OTf is possible (BINAP = 2,2′-bis(diphenylphosphino)-1,1′-binaphthyl).26 The κ2 bite angles of BINAP and L2 are close (BINAP = 93°,27 H[L2] = 95°), so similar stability of the pyridine adducts could be expected. The key difference between BINAP and L2 is the capacity of the latter to access the κ1-P;η3-NCC mode, which is evidently much more stable than the bidentate mode, and this induces pyridine dissociation from 3.
The experimental studies of the reaction of 2 with pyridine as reported above suggest that the energetic landscape of the various L2 binding modes is relatively shallow, a feature that is often advantageous in catalysis. To support this interpretation, we used density-functional theory methods to examine the reaction 2+ py ⇌3 (Fig. 6) in benzene solution. In principle, this reaction can proceed by either (a) an associative mechanism involving a transition state (TS) or (b) a dissociative mechanism in which the κ1-P;η3-NCC coordination mode of 2 shifts to κ2-PN to give a three-coordinate intermediate (2-open), followed by addition of pyridine. Formation of 2-dimer is also a possible side process. To assess these pathways, we computed zero-point-energy-corrected electronic energies (ΔE0) and Gibbs free energies (ΔG) of the species involved.
The calculations were carried out using the PBE1PBE hybrid functional in combination with the def2-TZVP basis set, Grimme's D4 empirical dispersion correction, and the SMD implicit solvation model, as implemented in the Gaussian Development Version.28 The chosen level of theory provides a well-established balance of accuracy and efficiency for transition-metal complexes of similar complexity. The results are summarized in Table 1. The reported values are not claimed to be highly accurate, but they lead to the same qualitative conclusions as the PBEh-3c![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 29 and B97h-3c
29 and B97h-3c![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 composite methods (Tables S2 and S3), as well as PBE1PBE calculations with a smaller basis set (Table S4).
30 composite methods (Tables S2 and S3), as well as PBE1PBE calculations with a smaller basis set (Table S4).
| Structure | ΔE0 | ΔG243 | ΔG298 |
|---|---|---|---|
| a Method: PBE1PBE/def2-SVP EmpiricalDispersion = GD4 SCRF (SMD, solvent = benzene). All data are referenced to the ideal-gas standard state of 1 atm for each species, whether solvated or not. b Relative to 2 per mole of monomer | |||
| 2 + py | 0.0 | 0.0 | 0.0 |
| TS | 0.2 | 36.9 | 46.2 |
| 3 | –40.5 | 0.0 | 9.0 |
| 2-open + py | 45.5 | 43.0 | 42.2 |
| 2-dimer | –49.9 | −21.7 | −15.6 |
According to Table 1, complex 3 is more stable than 2+ py at low temperatures, while 2-dimer is always more stable than both 3 and 2+ py. Thermodynamics alone would therefore suggest that dimer formation should be preferred. However, the kinetic landscape is markedly different.
An explicit TS was located for the 2+ py →3 pathway (Fig. S54), and it lies only 0.2 kJ mol−1 above the reactants on the potential energy surface (with zero-point-energy corrections). Therefore, the formation of 3 would have practically no barrier at 0 K; even at −30 °C, the Gibbs energy of the TS is only ΔG‡243 = +36.9 kJ mol−1 relative to the reactants. Because of the limitations of the harmonic oscillator/ideal-gas approximation, thermal corrections in this system are almost certainly overestimated, which means than the true ΔG‡243 could be even lower. This relatively low barrier is consistent with facile formation of 3 at reduced temperature.
On the other hand, even though the activation barrier for the dimer formation was not computed, it necessarily exceeds 43 kJ mol−1 relative to 2 (assuming the dimerization proceeds through 2-open), and is likely substantially higher because of entropic penalties. The higher activation barrier for 2-dimer supports the observation that the dimerization pathway is not observed.
As for the dissociative path from 2 to 3, the putative intermediate 2-open lies consistently higher than 2, with ΔE0 = +45.5 kJ mol−1 and ΔG243 = +43.0 kJ mol−1. Since we did not attempt to locate the transition state between 2 and 2-open, the possibility of the competing dissociative substitution pathway from 2 to 3 cannot be excluded, but this does not affect the above conclusion that 2-dimer is kinetically disfavoured.
Taken together, the data of Tables 1 and S2–S4 provide a consistent mechanistic explanation: 3 is the kinetically accessible product at low temperature, whereas dimer formation is apparently rendered negligible by unfavourable entropic factors.
The relatively small Gibbs energy difference between 2+ py and 3 at 243–298 K shows that L2 binding can readily change to accommodate different numbers of exogenous ligands on the metal. The preference for L2 binding via the κ1-P;η3-NCC mode promotes dissociation of the Lewis base pyridine. Thus, the dynamic coordination chemistry of L2 offers a low-energy pathway for substitution reactions at Pd.
Conclusions
A new ligand L2 with phosphine and 1-azaallyl donor groups was prepared that has a wider κ2-PN bite angle than a previous ligand derivative (i.e., L1). This characteristic permitted access to the κ1-P;η3-NCC coordination mode within a Pd(II)-methyl complex, 2. The mode was confirmed through characterization of 2 by NMR and X-ray absorption spectroscopies. The Pd L3-edge XANES measurements revealed that the κ1-P;η3-NCC coordination mode may lead to enhanced electrophilicity of Pd in 2, which is desirable for Lewis base coordination. Attempts to prepare a Lewis acid–base adduct of 2, in which L2 would adopt a κ2-PN mode to accommodate the new ligand, resulted in dynamic mixtures. The adduct 3 was observed, but only in the presence of an excess of pyridine and at low temperatures. Electronic structure calculations confirmed the energetic proximity of 2 and the pyridine adduct 3, which facilitates the observed reversible attachment of the Lewis base ligand.The capacity of the ligand to facilitate reactions with shallow energic pathways is a generally desirable feature of catalysts. For example, 2 may readily permit facile substrate coordination (i.e., Lewis base coordination) and product dissociation (i.e., Lewis base dissociation), without catalyst inhibition. We are currently exploring applications of 2 and related compounds in catalysis.
Author contributions
The manuscript was written through contributions of all authors.Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information (SI): NMR spectroscopy, IR spectroscopy, UV-vis spectroscopy, mass spectrometry, X-ray absorption spectroscopy, and computational (optimized XYZ coordinates) data. See DOI: https://doi.org/10.1039/d5dt02402a.CCDC 2487843 (1b) contains the supplementary crystallographic data for this paper.31
Acknowledgements
The authors acknowledge the support of the Natural Sciences and Engineering Research Council (NSERC) of Canada Discovery Grants Program (V. N. S., T. K. S., and J. M. B.). Part of the research described in this paper was performed at the Canadian Light Source, a national research facility of the University of Saskatchewan, which is supported by the Canada Foundation for Innovation (CFI), the Natural Sciences and Engineering Research Council (NSERC), the National Research Council (NRC), the Canadian Institutes of Health Research (CIHR), the Government of Saskatchewan, and the University of Saskatchewan.References
- J. F. Hartwig, Organotransition Metal Chemistry: From Bonding to Catalysis, University Science Books Sausalito, CA, 2010 Search PubMed.
- R. H. Crabtree, The Organometallic Chemistry of the Transition Metals, Wiley-Interscience, Hoboken, N.J, 2005 Search PubMed.
- R. B. Jordan, Reaction Mechanisms of Inorganic and Organometallic Systems, Oxford University Press, New York, 3 edn, 2007 Search PubMed.
- J. M. Blacquiere, ACS Catal., 2021, 11, 5416–5437 Search PubMed.
- P. Braunstein and F. Naud, Angew. Chem., Int. Ed., 2001, 40, 680–699 Search PubMed.
- C. S. Slone, D. A. Weinberger and C. A. Mirkin, in Prog. Inorg. Chem, ed. K. D. Karlin, John Wiley& Sons, Inc., 1999, vol. 48, pp. 233–350 Search PubMed.
- M. Bassetti, Eur. J. Inorg. Chem., 2006, 2006, 4473–4482 Search PubMed.
- G. M. Adams and A. S. Weller, Coord. Chem. Rev., 2018, 355, 150–172 Search PubMed.
- D. G. A. Verhoeven and M.-E. Moret, Dalton Trans., 2016, 45, 15762–15778 RSC.
- A. Beaufils, P. Melle, N. Lentz and M. Albrecht, Inorg. Chem., 2024, 63, 2072–2081 CrossRef CAS PubMed.
- K. D. Sipps, W. A. Gibbs, E. R. Sayfutyarova and J. L. Kuo, ACS Catal., 2024, 14, 18045–18054 CrossRef CAS.
- B. M. Trost and M. C. Ryan, Angew. Chem., Int. Ed., 2017, 56, 2862–2879 CrossRef CAS PubMed.
- M. J. Calhorda, C. C. Romão and L. F. Veiros, Chem. – Eur. J., 2002, 8, 868–875 CrossRef CAS PubMed.
- D. Zargarian, Coord. Chem. Rev., 2002, 233–234, 157–176 Search PubMed.
- C. F. Caro, M. F. Lappert and P. G. Merle, Coord. Chem. Rev., 2001, 219–221, 605–663 CrossRef CAS.
- A. G. Avent, P. B. Hitchcock, M. F. Lappert, R. Sablong and J. R. Severn, Organometallics, 2004, 23, 2591–2600 CrossRef CAS.
- J. M. Stubbs, K. F. Firth, B. J. Bridge, K. J. Berger, R. J. Hazlehurst, P. D. Boyle and J. M. Blacquiere, Dalton Trans., 2017, 46, 647–650 RSC.
- K. M. K. Jackman, B. J. Bridge, E. R. Sauvé, C. N. Rowley, C. H. M. Zheng, J. M. Stubbs, P. D. Boyle and J. M. Blacquiere, Organometallics, 2019, 38, 1677–1681 CrossRef CAS.
- M. B. Kindervater, V. N. Staroverov, K. M. K. Jackman, A. A. Fogh, L. S. G. Kelley, L. Lim, S. A. Sirohey, P. D. Boyle and J. M. Blacquiere, Dalton Trans., 2023, 52, 10744–10750 RSC.
- X. Zhou, K.-C. Lau, B. J. Petro and R. F. Jordan, Organometallics, 2014, 33, 7209–7214 Search PubMed.
- N. D. Contrella, J. R. Sampson and R. F. Jordan, Organometallics, 2014, 33, 3546–3555 CrossRef CAS.
- T. Kochi, S. Noda, K. Yoshimura and K. Nozaki, J. Am. Chem. Soc., 2007, 129, 8948–8949 Search PubMed.
- L. Yang, D. R. Powell and R. P. Houser, Dalton Trans., 2007, 955–964 RSC.
- P. B. Hitchcock, M. F. Lappert and M. Layh, Z. Anorg. Allg. Chem., 2000, 626, 1081–1086 CrossRef CAS.
- N. E. Jacobsen, in NMR Spectroscopy Explained, ed. N. E. Jacobsen, John Wiley & Sons, Inc., 2007, pp. 408–488, DOI:10.1002/9780470173350.ch10.
- A. M. Johns, J. W. Tye and J. F. Hartwig, J. Am. Chem. Soc., 2006, 128, 16010–16011 CrossRef CAS PubMed.
- M.-N. Birkholz, Z. Freixa and P. W. N. M. van Leeuwen, Chem. Soc. Rev., 2009, 38, 1099–1118 RSC.
- M. J. Frisch, G. W. Trucks, G. Scalmani, J. R. Cheeseman, X. Li, J. Bloino, B. G. Janesko, A. Marenich, J. Zheng, F. Lipparini, A. Jenkins, A. Liu, H. Liu, H. B. Schlegel, G. E. Scuseria, M. A. Robb, V. Barone, G. A. Petersson, H. Nakatsuji, B. Mennucci, C. Adamo, N. Rega, M. Caricato, H. P. Hratchian, J. V. Ortiz, F. Pawlowski, A. F. Izmaylov, H. Hu, C. Liao, J. L. Sonnenberg, D. Williams-Young, F. Ding, R. Gomperts, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery Jr., J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, M. Cossi, J. M. Millam, M. Klene, R. Cammi, R. L. Martin, O. Farkas, J. B. Foresman and D. J. Fox, Gaussian Development Version, Revision J.28, Gaussian, Inc., Wallingford, CT, 2023 Search PubMed.
- S. Grimme, J. G. Brandenburg, C. Bannwarth and A. Hansen, J. Chem. Phys., 2015, 143, 054107 CrossRef PubMed.
- J. G. Brandenburg, C. Bannwarth, A. Hansen and S. Grimme, J. Chem. Phys., 2018, 148, 064104 CrossRef PubMed.
- CCDC 2487843: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pht01.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |