Open Access Article
Open Access ArticleEffects of labile ligands and substituents in nickel enolate catalysts on ethylene/acrylate copolymerization activity: a DFT study
Ying
Wang
ab,
Xiaowei
Xu
a,
Yi
Luo
 a,
Sicong
Liu
a,
Zhuozheng
Wang
a,
Hao
Li
a,
Fan
Yang
a,
Sicong
Liu
a,
Zhuozheng
Wang
a,
Hao
Li
a,
Fan
Yang
 *b,
Xingxun
Li
*b and
Weisheng
Yang
*a
*b,
Xingxun
Li
*b and
Weisheng
Yang
*a
aPetroChina Petrochemical Research Institute, Beijing 102206, China. E-mail: yangweisheng@petrochina.com.cn
bState Key Laboratory of Heavy Oil Processing, China University of Petroleum-Beijing, Beijing 102249, China
First published on 13th January 2026
Abstract
In this study, density functional theory (DFT) was employed to investigate the copolymerization mechanism of ethylene with tert-butyl acrylate (tBA) catalyzed by neutral nickel enolate complexes featuring distinct substituents. It is computationally found that [2,6-(PhO)2C6H3]2PCHC(Ph)O–Ni (A) is more active than [2,6-(MeO)2C6H3]2PCHC(Ph)O–Ni (B), which is in line with experimental observations. Based on this agreement, it is demonstrated that the ethylene insertion into the tBA chain-end is the rate-determining step and the activity discrepancy between A and B is closely associated with the electronic effects of the substituents rather than the difference in sterics between the PhO group in A and the MeO in B. The natural population analysis (NPA) indicates that the phenoxy substituent can more effectively increase the positive charge on the Ni center, thereby enhancing its copolymerization activity. The influence of the labile ligand (L) (L = pyridine, PEt3 and PPh3) on the copolymerization activity of the more active A has been further investigated. The coordination strength of the labile ligand was found to significantly influence the catalytic performance. Specifically, a weaker coordinating labile ligand facilitates the ligand exchange between L and the monomer (tBA and ethylene) and enhances the efficiency of chain propagation. These mechanistic insights are helpful for the molecular design of copolymerization catalysts with high performance.
1. Introduction
The copolymerization of polar and non-polar olefins has become a prominent research direction in the field of polyolefins. The incorporation of polar monomers into polyolefins through coordination copolymerization enables better control of their microstructures and material properties, thereby widening the range of their potential applications in packaging materials, automotive components, and electronic materials.1–3 While early transition metal catalysts (e.g., group 4 metallocenes) exhibit high activity for ethylene polymerization, their intolerance to polar functional groups severely limits their application in copolymerization reactions with polar monomers.4–8 In contrast, late transition metal catalysts for the polymerization of polar olefins9–17 (Fig. 1a) have been extensively studied in both academia and industry due to their low oxophilicity and excellent functional group tolerance.18–25 However, highly efficient catalytic systems capable of addressing the copolymerization of polar and non-polar monomers on an industrial scale have been still limited. Among various catalytic options,2,26,27 nickel catalysts are more attractive for industrial applications owing to their higher economic advantages, greater abundant resources, and superior catalytic activity in the homopolymerization of ethylene.28,29 Notably, neutral phosphine–enolate nickel catalysts represent a versatile class of systems where steric and electronic modifications profoundly influence reactivity. Yet, the mechanistic basis of these effects, especially in acrylate copolymerization, has remained unclear, hindering rational catalyst design.To develop the field of late transition metal catalyzed olefin copolymerization, researchers have explored various factors influencing catalyst performance. These include electronic effects, steric hindrance, and metal center characteristics.30–39 Although notable progress has been achieved,40 considerable challenges still remain in this research field.41–43 A breakthrough was recently reported by Agapie et al.,44 through the development of neutral nickel phosphine–enolate complexes, viz., [2,6-(PhO)2C6H3]2PCHC(Ph)O–Ni (A) and [2,6-(MeO)2C6H3]2PCHC(Ph)O–Ni (B) (Fig. 1b). These complexes showed great potential in the coordination copolymerization of ethylene with tert-butyl acrylate (tBA). Remarkably, both catalytic activity and thermal stability were substantially improved by strategically incorporating steric hindrance on the phosphine side of the asymmetric ligands. In subsequent work, the effect of labile ligand L on the ethylene/acrylate copolymerization process was investigated.45 A 4–5 fold activity increase was observed when PEt3 was replaced by pyridine (Py). This ligand modification strategy, being more feasible than backbone restructuring, provides an efficient approach for catalyst optimization.
This study employs DFT calculations to elucidate the mechanism of ethylene and tBA copolymerization catalyzed by neutral nickel phosphine–enolate complexes. The theoretical calculations reveal the regulatory effects of different substituents on the ethylene/tBA copolymerization activity and the influence mechanism of labile ligand L on the copolymerization performance at the molecular level (Fig. 1b). By providing a detailed understanding of the catalytic mechanism, this study aims to guide the rational design of more efficient and robust nickel catalysts for the copolymerization of polar and non-polar monomers, thereby addressing the current challenges in this field.
2. Computational details
All the density functional theory (DFT) calculations were performed using the Gaussian 16 program.46 The TPSSTPSS47 functional together with the 6-31G(d) basis set for nonmetal atoms (C, H, O, N, and P) and the LANL2DZ48–50 basis set as well as associated pseudopotential for the metal atom (Ni) was used for geometry optimizations and subsequent frequency calculations. Such basis sets are denoted as BSI. Based on the TPSSTPSS/BSI geometries, single-point calculations were further performed at the higher level of theory by using the dispersion-corrected density functional method TPSSTPSS-D3BJ51 together with BSII. In BSII, Def2-TZVP52,53 was used for all the atoms. In these single-point calculations, the solvation effect of toluene (ε = 2.37) was considered through the SMD54 model. The energy profiles were constructed at the TPSSTPSS-D3BJ/BSII(SMD)//TPSSTPSS/BSI level, including Gibbs free energy corrections taken from frequency calculations in the gas-phase (298.15 K, 1 atm). After carrying out the above calculations, the noncovalent interaction (NCI) analysis55 was conducted for some important transition structures (TSs), which were shown using Multiwfn56,57 and VMD58 software. The optimized geometrical structures were plotted using CYLView.593. Results and discussion
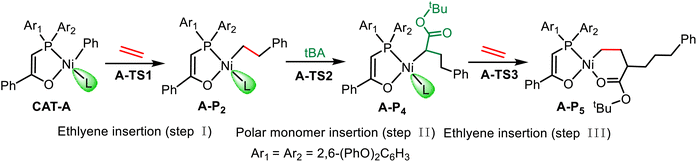
To comparatively investigate the substituent effect, both phosphine enolate based catalysts A and B are selected as computational models in this study, as shown in Chart 1, for a systematic mechanistic exploration of their coordination–insertion processes. The two catalysts exhibit distinct electronic and structural properties. The nickel center in catalyst A shows a higher NPA charge compared to that in catalyst B (+0.121 vs. +0.071), indicating stronger electron inadequacy. Structurally, catalyst A exhibits a larger dihedral angle (P2–Ni–O1–C1 = −10.1°) than catalyst B (−8.5°), suggesting greater flexibility. These differences might contribute to their distinct catalytic activities.443.1 Copolymerization mechanism of ethylene and tBA
Due to the electronic asymmetry of the ancillary ligand, the vinyl monomer may show two spatial orientations during coordination insertion: trans site (monomer in trans configuration to the phosphorus atom) and cis site (monomer in cis configuration to the phosphorus atom). Previous studies60–63 have demonstrated that the cis-site coordination is less stable than the trans-site (ΔGcis > ΔGtrans), but its insertion energy barrier is significantly lower than that of the trans-site (ΔG‡cis < ΔG‡trans). Based on this theoretical understanding, this study focuses on the cis-site insertion pathway to reveal the microscopic mechanism of the most favorable reaction pathway.As illustrated by the red pathway in Fig. 2, the chain initiation process catalyzed by A-CAT involves several critical elementary steps, beginning with ligand exchange:21 ethylene coordinates to the active nickel center in a trans-coordination mode, while the weakly coordinating triethylphosphine (PEt3) ligand dissociates from the metal center, yielding the trans-coordinated intermediate A-Ct1. This step requires overcoming an energy barrier of 27.5 kcal mol−1, and the resulting trans-configuration in A-Ct1 is stabilized by the combined electronic and steric effects of the nickel center, favoring initial coordination. Subsequently, A-Ct1 undergoes a configuration flip to form the cis-coordinated intermediate A-Cc1, a step requiring an isomerization barrier of 30.6 kcal mol−1. Ethylene then inserts through a four-center transition state A-TS1, with an activation barrier of 30.3 kcal mol−1, to afford the chain-initiation product A-P1. Additionally, more detailed calculations on the ligand exchange and isomerization processes are provided in Fig. S1 and S2, offering further insights into these mechanisms. Similarly, catalyst system B (blue pathway in Fig. 2) follows the sequence: B-CAT → B-ex-TS1 → B-Ct1 → B-iso-TS1 → B-Cc1 → B-TS1 → B-P1 → B-P2. In this pathway, the ligand-exchange barrier is 22.0 kcal mol−1, the trans/cis isomerization barrier is 27.7 kcal mol−1, and the ethylene insertion barrier is 31.3 kcal mol−1. Overall, although the two catalytic systems follow similar reaction pathways, A-CAT exhibits lower activation barriers during the chain-initiation stage (30.6 vs. 31.3 kcal mol−1).
 | ||
| Fig. 2 Energy profile for the chain initiation in the A and B systems. The energies are relative to the corresponding reactants. | ||
Afterward, the insertion of tBA and subsequent ethylene insertion are further calculated. In the polar monomer insertion process of the A system, there are two types of trans-site coordination: one is the coordination of the carbonyl oxygen of the functional group to the metal center, and the other is the coordination of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond of the monomer to the metal center. Given that the product formed upon the insertion of the first ethylene molecule is in the cis configuration, the trans position is preferentially occupied when the polar monomer coordinates. Consequently, in the whole process of insertion of polar monomers, the primary insertion modes involve carbonyl oxygen trans-coordination, carbon–carbon double bond trans-coordination, and carbon–carbon double bond cis-coordination. Additionally, based on regioselectivity, the polar monomer insertion modes are categorized into 1,2-insertion and 2,1-insertion. As shown in Table 1, in the case of the A system, the energy barrier for 2,1-insertion is 3.7 kcal mol−1 lower than that for 1,2-insertion (28.9 vs. 32.6 kcal mol−1), indicating a preference for the 2,1-insertion mode, which is common in such kind of systems.35,64–66 The similar is true for the catalyst B system (26.2 vs. 32.8 kcal mol−1). Table 1 indicates the relative energies of the relevant regioselectivities during the insertion of polar monomers, showing that for catalyst B, the 2,1-insertion mode is equally dominant for tBA monomer insertion. Compared to A-CAT, B-CAT exhibits a lower energy barrier for tBA insertion. This is because A-CAT undergoes significant geometric distortion due to steric effects during the reaction, and the resulting distortion energy becomes the dominant factor whose unfavorable impact cannot be compensated by electronic effects, as demonstrated by the distortion/interaction analysis (Fig. S3).
C bond of the monomer to the metal center. Given that the product formed upon the insertion of the first ethylene molecule is in the cis configuration, the trans position is preferentially occupied when the polar monomer coordinates. Consequently, in the whole process of insertion of polar monomers, the primary insertion modes involve carbonyl oxygen trans-coordination, carbon–carbon double bond trans-coordination, and carbon–carbon double bond cis-coordination. Additionally, based on regioselectivity, the polar monomer insertion modes are categorized into 1,2-insertion and 2,1-insertion. As shown in Table 1, in the case of the A system, the energy barrier for 2,1-insertion is 3.7 kcal mol−1 lower than that for 1,2-insertion (28.9 vs. 32.6 kcal mol−1), indicating a preference for the 2,1-insertion mode, which is common in such kind of systems.35,64–66 The similar is true for the catalyst B system (26.2 vs. 32.8 kcal mol−1). Table 1 indicates the relative energies of the relevant regioselectivities during the insertion of polar monomers, showing that for catalyst B, the 2,1-insertion mode is equally dominant for tBA monomer insertion. Compared to A-CAT, B-CAT exhibits a lower energy barrier for tBA insertion. This is because A-CAT undergoes significant geometric distortion due to steric effects during the reaction, and the resulting distortion energy becomes the dominant factor whose unfavorable impact cannot be compensated by electronic effects, as demonstrated by the distortion/interaction analysis (Fig. S3).
| Catalyst | P2 | Ct-O | 1,2-tBA Ct2 | 1,2-tBA Cc2 | 1,2-tBA TS2 | ΔG‡(1,2-tBA) | ΔΔG‡ |
|---|---|---|---|---|---|---|---|
| (2,1-tBACt2) | (2,1-tBACc2) | (2,1-tBATS2) | (ΔG‡(2,1-tBA)) | ||||
a
P2 denotes the insertion products with the ethylene chain-end. Ct-O denotes the intermediate with carbonyl-coordinating tBA at the trans-site. TS represents the insertion transition states. Ct2 and Cc2 represent the coordination of tBA via its vinyl (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C) at the trans-site and cis-site, respectively. ΔG‡ represents the insertion free-energy barrier. The energies of the stationary points are relative to the corresponding catalyst and monomer. C) at the trans-site and cis-site, respectively. ΔG‡ represents the insertion free-energy barrier. The energies of the stationary points are relative to the corresponding catalyst and monomer.
|
|||||||
| A | −3.7 | 11.5 | 12.2 | 14.8 | 28.9 | 32.6 | 3.7 |
| (12.5) | (14.6) | (25.2) | (28.9) | ||||
| B | −4.0 | 12.1 | 10.2 | 13.5 | 28.8 | 32.8 | 6.6 |
| (10.7) | (12.9) | (22.2) | (26.2) | ||||
The energy profiles for chain propagation are depicted in Fig. 3, with red traces representing the reaction pathways for catalyst A and blue traces for catalyst B. In system A, the ethylene insertion following tBA insertion is the rate-determining step in the chain growth process, with a reaction energy barrier 2.3 kcal mol−1 higher than that of the tBA insertion (31.2 vs. 28.9 kcal mol−1). The similar is true for system B (32.5 vs. 26.2 kcal mol−1). This phenomenon is primarily attributed to increased steric hindrance of the polymer chain and reduced nucleophilicity of the polymer chain resulting from the electron-withdrawing nature of the ester group.
As shown in Fig. 3, although catalyst B exhibits a lower energy barrier in the tBA insertion step compared with the A system, its higher energy barrier for subsequent ethylene insertion limits the overall reaction rate.67,68 Both catalysts show higher energy barriers for the insertion of ethylene into the tBA chain-end compared with the insertion of tBA, which might be the key factor leading to the differences in copolymerization reaction rates. To ensure structural integrity, the pathways for ligand exchange and trans/cis isomerization during the chain propagation process are presented in Fig. S4 and S5. Computational results confirm that these isomeric processes have lower energy barriers than the rate-determining step. Given that the ground-state energies of intermediates P5 and P4 are similar, we further investigated the ethylene insertion reaction starting from P5. The results indicate that this subsequent ethylene insertion does not lead to a significantly higher energy barrier (Fig. S6). To ensure the reliability of DFT results, this study employed multiple DFT methods to evaluate single-point energies for key intermediates in the rate-determining step. All methods consistently indicated that catalyst A exhibits higher activity than catalyst B (Table S1). Subsequent studies will further investigate the influence of catalyst structure on the reaction energy barrier by integrating molecular orbital theory and distortion/interaction analysis.
3.2 Analyses of the factors governing the copolymerization activity
The structural and electronic properties of the catalysts dictate both the reaction pathways and their associated energy profiles. Natural population analysis (NPA) shows that the Ni center in A-TS3 carries a higher positive charge than in B-TS3 (+0.235 vs. +0.228). Such a higher positive charge (electronic effect) dominantly contributed to the higher stability of A-TS3, as also demonstrated by the distortion/interaction analysis (Fig. 4). According to the frontier molecular orbital theory, the highest occupied molecular orbital (HOMO) of ethylene interacts with the lowest unoccupied molecular orbital (LUMO) of enolate nickel catalysts during the reaction process. This interaction facilitates the transfer of electrons from the reactants to the catalysts. The LUMO energies of catalysts A and B are −3.718 eV and −3.506 eV, respectively. Compared to the HOMO orbital energy of ethylene (−6.335 eV), catalyst A exhibits better orbital overlap and therefore higher reactivity. This electronic advantage directly accounts for the superior activity of catalyst A in ethylene/tBA copolymerization. However, catalyst A demonstrates a higher activation energy barrier for tBA insertion (28.9 kcal mol−1 in Avs. 26.2 kcal mol−1 in B), which can be attributed to the larger axial spatial steric hindrance. While this steric effect improves catalyst stability, it may also cause overcrowding at the metal center, potentially hindering the insertion of bulky polar monomers. This trade-off between electronic and steric effects highlights the complex interplay of factors influencing the catalytic performance.
 | ||
| Fig. 4 The distortion/interaction analysis (energy in kcal mol−1) and the optimized structures of (a) A-TS3 and (b) B-TS3. | ||
To further explore and explain the reasons for the differences in copolymerization activity between the two catalysts, a distortion/interaction analysis is performed on the rate-determining transition states A-TS3 and B-TS3 (Fig. 4). In the distortion/interaction analysis, the energies of the monomer fragment and the remaining metal complex fragment (two fragments) in the transition state geometry are obtained through single-point calculations. The interaction energy (ΔEint) in the transition state is calculated as the difference between the total energy of the transition state and the sum of the single-point energies of the two isolated fragments. The distortion energies of the two fragments, ΔEdist(cat) and ΔEdist(mono), are determined by comparing the energy of each fragment in the transition state geometry with its energy in its optimized geometry. Thus, the total electronic energy of the transition state is the sum of the interaction energy and the distortion energies, i.e., ΔETS = ΔEint + ΔEdist(cat) + ΔEdist(mono).
As shown in Fig. 4, the distortion energy of the catalyst part in the transition state is labeled in pink (ΔEdist(cat)), while the distortion energy of the monomer part is labeled in red (ΔEdist(mono)). The calculated results reveal that in A-TS3, the distortion energy of the monomer is 22.2 kcal mol−1, and that of the catalyst fragment is 53.7 kcal mol−1, yielding a total distortion energy of 75.9 kcal mol−1. In contrast, for B-TS3, the distortion energy of the monomer is 22.4 kcal mol−1, and that of the catalyst fragment is 54.1 kcal mol−1, resulting in a total distortion energy of 76.5 kcal mol−1. In comparison, the total distortion energy of A-TS3 is lower than that of B-TS3 (75.9 vs. 76.5 kcal mol−1), and this difference is reflected in the change of dihedral angle P2–Ni–O1–C1 between catalysts A and B. Catalyst A exhibits a smaller change in the P2–Ni–O1–C1 dihedral angle from −10.06° to −9.18°, whereas catalyst B undergoes a greater structural deformation (−8.52° → −5.33°), resulting in lower stability. This made catalyst B exhibit lower reactivity in catalyzing the copolymerization of tBA with ethylene. Furthermore, the stronger interaction between the monomer unit and the metal catalyst in A-TS3 (ΔEint values of −65.6 and −64.3 kcal mol−1 for A-TS3 and B-TS3, respectively) might originate from the weak van der Waals interactions between the benzene ring of the phenoxy substituent on the phosphorus and the alkyl chain or other benzene rings in catalyst A (Fig. S7). Such weak interactions, however, are absent in catalyst B, which might explain the weaker interaction between the two fragments in B compared to the one in catalyst A. A comparison of the contribution of deformation and interaction energies to the more stability of A-TS3 indicates that the interaction energy (electronic effect) is dominant (difference between A-TS3 and B-TS3: deformation energy of – 0.6 kcal mol−1vs. interaction energy of – 1.3 kcal mol−1). The above multiscale theoretical analyses indicate that catalyst A exhibits smaller deformation energy and stronger interaction energy, which ultimately results in a more stable electronic energy of the transition state (ΔETS of 10.3 vs. 12.2 kcal mol−1). These results indicate that catalyst A has higher activity in ethylene/tBA copolymerization. The strong agreement between these results and experimental observations provides a critical theoretical foundation for understanding the relationship between catalyst structure and performance.
DFT calculations revealed that the ligand-exchange process for different ligands L differs significantly, with the ligand-exchange energy ΔGex being 8.2 kcal mol−1 for pyridine (Py), 16.8 kcal mol−1 for triphenylphosphine (PPh3), and 18.3 kcal mol−1 for triethylphosphine (PEt3), as shown in Fig. 5. This establishes the stability trend in the order of Py < PPh3 < PEt3, which directly impacts catalytic behavior. Specifically, Py has a significantly higher propensity to undergo ligand exchange with ethylene than PEt3 and PPh3. This suggests that the Py ligand dissociates more easily from the metal center, thereby exposing the active site and reducing the energy barrier during the chain initiation stage (Fig. S8). To gain a deeper understanding of the mechanism by which the labile ligand influences the activity of catalyst A, we conducted frontier orbital analysis on three L-ligated catalyst systems: PEt3 (A-1), PPh3 (A-2), and pyridine (A-3). The results indicate that L significantly modulates the electronic structure, with HOMO–LUMO energy gaps of: A-1 (2.37 eV) > A-2 (2.11 eV) > A-3 (1.93 eV). According to the chemical hardness theory69,70 (η = (E(LUMO) − E(HOMO))/2), the A-3 system with pyridine coordination has the lowest η value, indicating higher reactivity and greater propensity to participate in chemical reactions. These results agree with Xiong et al.'s work, confirming Py's advantages in reducing initiation barriers.45 Furthermore, according to the NPA charge analysis, the charges on the metal center (Ni) in the three catalysts (A-1, A-2, and A-3) increase in the following order: A-1 (0.121) < A-2 (0.151) < A-3 (0.267). These results indicate that the nature of the ligand (L) significantly influences the electrophilicity of the metal center (Ni), thereby modulating its catalytic activity. Specifically, the aromatic Py ring features electron delocalization and exhibits electron-withdrawing effects, which may reduce the electron density at the metal center (Ni) and results in a more positive charge (0.267 for A-3). In contrast, PPh3 and PEt3 have P atoms as strong electron donors, resulting in relatively lower positive charges on the metal center (Ni). This difference in charge distribution reflects the role of the labile ligand in modulating the electronic structure of the metal center, which in turn affects the overall performance of the catalyst.
We further examined the chain propagation process of the catalyst A system with the coordination of different ligands (L). After the insertion of one ethylene molecule, the resulting intermediate binds with ligand L to form a more stable A-P2 complex, followed by the insertion of polar monomers. As shown in Table 2, A-3 shows the lowest insertion energy barrier at the rate-determining step (see Fig. S9 for the complete energy profile) due to the smallest exchange energy of Py. The smaller steric hindrance of the Py ligand allows tBA to more easily approach the metal center and insert, resulting in a lower energy barrier and a higher insertion rate of polar monomers in the case of A-3. In contrast, PPh3 and PEt3 exhibit larger steric hindrance, especially PPh3, whose significant steric hindrance impedes the insertion of bulky polar monomers (e.g., tBA), leading to the highest insertion energy barrier. As a result, A-2 exhibits the highest energy barrier for tBA insertion among the three cases (28.9, 30.3, and 23.4 kcal mol−1 for A-1, A-2, and A-3, respectively). Both theoretical and experimental results demonstrate that the Py ligated catalyst A-3 exhibits the highest copolymerization activity, while the PEt3-ligated catalyst A-1 and the PPh3-ligated catalyst A-2 show relatively lower activity due to their larger ligand-exchange energy. Although ligands with large steric hindrance (e.g., PPh3) can enhance catalyst stability, they might also hinder the insertion of bulky polar monomers. Therefore, when designing catalysts, the trade-off between steric hindrance of ligand L and catalytic activity could be necessary.
| CAT | L | A-TS1 | A-P2 | A_TS | ΔG‡tBA | A-P4 | A_TS3 | A-P5 | ΔG‡E |
|---|---|---|---|---|---|---|---|---|---|
| a A_TS1, A_TS2, and A_TS3 represent the insertion transition states. A-P2 and A-P4 denote the insertion products with coordination of L, and A-P5 represents the insertion product of ethylene into the tBA chain-end. ΔG‡tBA and ΔG‡E represent the insertion free-energy barriers for tBA and ethylene, respectively. The energies of the stationary points are relative to the corresponding reactants. The corresponding complete energy profile is presented in Fig. S8 and S9. | |||||||||
| A-1 | PEt3 | 30.3 | −3.7 | 25.2 | 28.9 | −10.9 | 20.3 | −11.4 | 31.2 |
| A-2 | PPh3 | 28.9 | −6.5 | 23.8 | 30.3 | −10.7 | 18.9 | −12.8 | 29.6 |
| A-3 | Py | 20.2 | −8.3 | 15.1 | 23.4 | −17.4 | 10.2 | −21.4 | 27.6 |
4. Conclusions
In summary, a comparative DFT study on the copolymerization mechanism of ethylene and tert-butyl acrylate (tBA) catalyzed by nickel enolate catalysts [2,6-(PhO)2C6H3]2PCHC(Ph)O–Ni (A) and [2,6-(MeO)2C6H3]2PCHC(Ph)O–Ni (B) is conducted, elucidating the influence mechanisms of the different substituents and labile ligands (L) on the copolymerization activity. It is found that the ethylene insertion into the tBA chain-end exhibits the highest reaction energy barrier, indicating that this step is the rate-determining step. The phenoxy substituent in catalyst A increases the positive charge on the Ni center dominantly through electronic effects, enhancing the interaction between the metal center and the monomer, thereby improving catalytic activity. The distortion/interaction analysis of the rate-determining step shows that catalyst A undergoes smaller geometric deformation in the transition state and exhibits stronger interaction, along with its weak interactions with surrounding atoms, which further stabilizes the transition state and reduces the reaction energy barrier. Based on the highly active catalyst A, the role of the labile ligands was investigated. In the catalytic process, monomers (ethylene or acrylates) must first replace ligand L and coordinate to the nickel center to undergo subsequent insertion reactions. The result reveals that the ligand exchange ability of the three labile ligands with monomers follows the order of Py > PPh3 > PEt3, which significantly influences the catalyst activity. Specifically, when the labile ligand is pyridine, catalyst A exhibits “fast initiation and slow growth” behavior. In contrast, with triethylphosphine (PEt3) as the ligand, its stronger coordination ability leads to “slow initiation and slow growth” behavior, and the influence of L persists even after the initiation step. Consequently, catalyst A exhibits the highest activity when pyridine is used as the labile ligand.Conflicts of interest
There are no conflicts to declare.Data availability
All the relevant data are within the manuscript and its supplementary information (SI). The energy profiles mentioned in the main text, analyses of energies and NCI, as well as the optimized coordinates of the stationary points are available in SI. Supplementary information is available. See DOI: https://doi.org/10.1039/d5dt02237a.References
- L. Guo, W. Liu and C. Chen, Mater. Chem. Front., 2017, 1, 2487–2494 RSC.
- R. Wu, W. K. Wu, L. Stieglitz, S. Gaan, B. Rieger and M. Heuberger, Coord. Chem. Rev., 2023, 474, 214844 CrossRef CAS.
- J. Y. Dong and Y. Hu, Coord. Chem. Rev., 2005, 250, 47–65 CrossRef.
- H. Wang, Y. Yang, M. Nishiura, Y. Higaki, A. Takahara and Z. Hou, J. Am. Chem. Soc., 2019, 141, 3249–3257 CrossRef CAS PubMed.
- H. Nsiri, I. Belaid, P. Larini, J. Thuilliez, C. Boisson and L. Perrin, ACS Catal., 2016, 6, 1028–1036 Search PubMed.
- J. Chen, Y. Gao and T. J. Marks, Angew. Chem., Int. Ed., 2020, 59, 14726–14735 CrossRef CAS PubMed.
- C. Wu, M. Ren, L. Hou, S. Qu, X. Li, C. Zheng, J. Chen and W. Wang, Engineering, 2023, 30, 93–99 Search PubMed.
- M. Atiqullah, M. Tinkl, R. Pfaendner, M. N. Akhtar and I. Hussain, Polym. Rev., 2010, 50, 178–230 Search PubMed.
- B. S. Xin, N. Sato, A. Tanna, Y. Oishi, Y. Konishi and F. Shimizu, J. Am. Chem. Soc., 2017, 139, 3611–3614 Search PubMed.
- M. Chen and C. Chen, Angew. Chem., Int. Ed., 2018, 57, 3094–3098 Search PubMed.
- X. Sui, S. Dai and C. Chen, ACS Catal., 2015, 5, 5932–5937 Search PubMed.
- B. P. Carrow and K. Nozaki, J. Am. Chem. Soc., 2012, 134, 8802–8805 Search PubMed.
- T. R. Younkin, E. F. Connor, J. I. Henderson, S. K. Friedrich, R. H. Grubbs and D. A. Bansleben, Science, 2000, 287, 460–462 Search PubMed.
- L. K. Johnson, C. M. Killian and M. Brookhart, J. Am. Chem. Soc., 1995, 117, 6414–6415 CrossRef CAS.
- E. Drent, R. van Dijk, R. van Ginkel, B. van Oort and R. I. Pugh, Chem. Commun., 2002, 744–745 Search PubMed.
- R. Nakano and K. Nozaki, J. Am. Chem. Soc., 2015, 137, 10934–10937 Search PubMed.
- W. Tao, R. Nakano, S. Ito and K. Nozaki, Angew. Chem., Int. Ed., 2016, 55, 2835–2839 Search PubMed.
- C. Chen, ACS Catal., 2018, 8, 5506–5514 CrossRef CAS.
- M. T. d. Costa, E. F. Lopes, R. S. Oliboni, D. S. Lüdtke, O. L. Casagrande and R. Stieler, Appl. Organomet. Chem., 2023, 37, e6984 Search PubMed.
- J. Ye, H. Mu, Z. Wang and Z. Jian, Organometallics, 2019, 38, 2990–2997 Search PubMed.
- M. M. Shoshani, S. Xiong, J. J. Lawniczak, X. Zhang, T. F. Miller and T. Agapie, Organometallics, 2022, 41, 2119–2131 Search PubMed.
- D. A. Park, S. Byun, J. Y. Ryu, J. Lee, J. Lee and S. Hong, ACS Catal., 2020, 10, 5443–5453 Search PubMed.
- S. Zhou and C. Chen, Sci. Bull., 2018, 63, 441–445 CrossRef CAS PubMed.
- L. Guo, S. Dai, X. Sui and C. Chen, ACS Catal., 2016, 6, 428–441 Search PubMed.
- C. Chen, S. Luo and R. F. Jordan, J. Am. Chem. Soc., 2008, 130, 12892–12893 CrossRef CAS PubMed.
- M. Khoshsefat, Y. Ma and W. Sun, Coord. Chem. Rev., 2021, 434, 213788 Search PubMed.
- D. Laws, C. D. Poff, E. M. Heyboer and S. B. Blakey, Chem. Soc. Rev., 2023, 52, 6003–6030 Search PubMed.
- D. Peng, H. He, W. Pang, S. Behzadi and M. Qasim, Polymer, 2023, 280, 126019 CrossRef CAS.
- Y. Zhang, H. Mu, L. Pan, X. Wang and Y. Li, ACS Catal., 2018, 8, 5963–5976 CrossRef CAS.
- G. Ren, R. Yuan, Q. Mahmood, Y. Zeng, Y. Wang, Z. Hu, S. Zou, T. Liang and W. Sun, J. Mol. Struct., 2024, 1316, 139037 CrossRef CAS.
- Z. Chen and M. Brookhart, Acc. Chem. Res., 2018, 51, 1831–1839 Search PubMed.
- F. Lin, M. Voccia, L. Odenwald, I. Göttker-Schnetmann, L. Falivene, L. Caporaso and S. Mecking, J. Am. Chem. Soc., 2023, 145, 27950–27957 CrossRef CAS PubMed.
- L. Min, W. Xingbao, L. Yi and C. Changle, Angew. Chem., Int. Ed., 2017, 56, 11604–11609 Search PubMed.
- Z. Lu, X. Xu, Y. Luo, S. He, W. Fan and S. Dai, ACS Catal., 2023, 13, 725–734 CrossRef CAS.
- J. Sun, M. Chen, G. Luo, C. Chen and Y. Luo, Organometallics, 2019, 38, 638–646 Search PubMed.
- M. Janeta, J. X. Heidlas, O. Daugulis and M. Brookhart, Angew. Chem., Int. Ed., 2020, 60, 4566–4569 Search PubMed.
- Q. H. Tran, M. Brookhart and O. Daugulis, J. Am. Chem. Soc., 2020, 142, 7198–7206 Search PubMed.
- Q. Li, H. Mu and Z. Jian, Polym. Chem., 2023, 14, 3196–3202 RSC.
- C. Chen, Nat. Rev. Chem., 2018, 2, 6–14 Search PubMed.
- S. Xiong, M. M. Shoshani, X. Zhang, H. A. Spinney, A. J. Nett, B. S. Henderson, T. F. Miller and T. Agapie, J. Am. Chem. Soc., 2021, 143, 6516–6527 Search PubMed.
- F. Ölscher, I. Göttker-Schnetmann, V. Monteil and S. Mecking, J. Am. Chem. Soc., 2015, 137, 14819–14828 Search PubMed.
- A. Michalak and T. Ziegler, J. Am. Chem. Soc., 2001, 123, 12266–12278 Search PubMed.
- H. Mu, G. Zhou, X. Hu and Z. Jian, Coord. Chem. Rev., 2021, 435, 213802 Search PubMed.
- S. Xiong, A. Hong, B. C. Bailey, H. A. Spinney, T. D. Senecal, H. Bailey and T. Agapie, Angew. Chem., Int. Ed., 2022, 61, e202206637 CrossRef CAS PubMed.
- S. Xiong, P. Ghana, B. C. Bailey, H. A. Spinney, B. S. Henderson, M. R. Espinosa and T. Agapie, ACS Catal., 2023, 13, 5000–5006 Search PubMed.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery Jr., J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman and D. J. Fox, Gaussian 16 revision A.03, Gaussian, Inc, Wallingford, CT.
- J. Tao, J. P. Perdew, V. N. Staroverov and G. E. Scuseria, Phys. Rev. Lett., 2003, 91, 146401 CrossRef PubMed.
- P. J. Hay and W. R. Wadt, J. Chem. Phys., 1985, 82, 270–283 Search PubMed.
- W. R. Wadt and P. J. Hay, J. Chem. Phys., 1985, 82, 284–298 Search PubMed.
- P. J. Hay and W. R. Wadt, J. Chem. Phys., 1985, 82, 299–310 CrossRef CAS.
- S. Grimme, S. Ehrlich and L. Goerigk, J. Comput. Chem., 2011, 32, 1456–1465 Search PubMed.
- F. Weigend, Phys. Chem. Chem. Phys., 2006, 8, 1057–1065 RSC.
- F. Weigend and R. Ahlrichs, Phys. Chem. Chem. Phys., 2005, 7, 3297–3305 RSC.
- A. V. Marenich, C. J. Cramer and D. G. Truhlar, J. Phys. Chem. B, 2009, 113, 4538–4543 CrossRef CAS PubMed.
- E. R. Johnson, S. Keinan, P. Mori-Sánchez, J. Contreras-García, A. J. Cohen and W. Yang, J. Am. Chem. Soc., 2010, 132, 6498–6506 CrossRef CAS PubMed.
- T. Lu and F. Chen, J. Comput. Chem., 2012, 33, 580–592 CrossRef CAS PubMed.
- T. Lu, J. Chem. Phys., 2024, 161, 082503 Search PubMed.
- W. Humphrey, A. Dalke and K. Schulten, J. Mol. Graphics, 1996, 14, 33–38 Search PubMed.
- C. Y. Legault, CYLview20, Université de Sherbrooke, 2020, https://www.cylview.org Search PubMed.
- S. Noda, A. Nakamura, T. Kochi, L. W. Chung, K. Morokuma and K. Nozaki, J. Am. Chem. Soc., 2009, 131, 14088–14100 Search PubMed.
- E. Rezabal, J. M. Ugalde and G. Frenking, J. Phys. Chem. A, 2017, 121, 7709–7716 CrossRef CAS PubMed.
- D. Guironnet, L. Caporaso, B. Neuwald, I. Göttker-Schnetmann, L. Cavallo and S. Mecking, J. Am. Chem. Soc., 2010, 132, 4418–4426 CrossRef CAS PubMed.
- N. Ryo, C. L. Wa, W. Yumiko, O. Yoshishige, O. Yoshikuni, I. Shingo, M. Keiji and N. Kyoko, ACS Catal., 2016, 6, 6101–6113 Search PubMed.
- Z. Song, S. Wang, R. Gao, Y. Wang, Q. Gou, G. Zheng, H. Feng, G. Fan and J. Lai, Polymers, 2023, 15, 4343 CrossRef CAS PubMed.
- Z. Chen, W. Liu, O. Daugulis and M. Brookhart, J. Am. Chem. Soc., 2016, 138, 16120 CrossRef CAS PubMed.
- S. Mecking, L. K. Johnson, L. Wang and M. Brookhart, J. Am. Chem. Soc., 1998, 120, 888–899 Search PubMed.
- W. Tian, H. Guan, W. Wang, X. Kong, W. Pang, Q. Wang, F. Wang and C. Zou, Appl. Organomet. Chem., 2025, 39, e70063 Search PubMed.
- J. Tan, J. Liu and X. Zhang, J. Org. Chem., 2025, 90, 2052–2061 Search PubMed.
- R. G. Pearson, Inorg. Chem., 1988, 27, 734–740 Search PubMed.
- P. Geerlings, F. De Proft and W. Langenaeker, Chem. Rev., 2003, 103, 1793–1874 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |