Fe3O4-based magnetic nanoparticles for the removal of water contaminants
Ali
Moeini
 a,
Iman
Khezri
a,
Iman
Khezri
 a,
Reza
Ahadi Dolatsara
a,
Reza
Ahadi Dolatsara
 a,
Maryam
Hajiebrahimi
a,
Maryam
Hajiebrahimi
 b,
Hamed
Beydaghi
b,
Hamed
Beydaghi
 a,
Aziz
Shahraki
a,
Aziz
Shahraki
 a,
Adrine
Malek Khachatourian
a,
Adrine
Malek Khachatourian
 *a,
Muhammet S.
Toprak
*a,
Muhammet S.
Toprak
 c and
Rajender S.
Varma
c and
Rajender S.
Varma
 *d
*d
aDepartment of Materials Science and Engineering, Sharif University of Technology, Tehran, Iran. E-mail: khachatourian@sharif.edu
bFaculty of Materials and Metallurgical Engineering, Semnan University, Semnan, Iran
cDepartment of Applied Physics, KTH-Royal Institute of Technology, SE10691 Stockholm, Sweden
dCentre of Excellence for Research in Sustainable Chemistry, Department of Chemistry, Federal University of São Carlos, 13565-905 São Carlos, SP, Brazil. E-mail: rajvarma@hotmail.com
First published on 2nd January 2026
Abstract
Anthropogenic activities generate industrial wastewater, posing the greatest threat to the environment and requiring cleanup to mitigate environmental impacts and human health risks. Because of their remarkable characteristics, Fe3O4-based magnetic nanoparticles (MNPs) have recently gained popularity as powerful tools for wastewater treatment. To alleviate the qualitative strain on water supplies, MNPs have found newer applications owing to their inherent characteristics. Herein, the physical, chemical, and biological/green approaches for the synthesis and assembly of MNPs are deliberated, with biologically derived options found to be environmentally benign and sustainable in the long term. The efficacy of MNPs in the elimination or recovery of various wastewater contaminants, such as heavy metals, inorganic pollutants, dyes, biological pollutants, and microalgae, is presented. Moreover, the advantages and disadvantages of MNPs in their usage for wastewater treatment, the removal mechanism via coagulation, and the manufacture of MNPs are appraised in detail. Finally, the potential of Fe3O4-based MNPs for deployment in environmental cleanup and their future applications are addressed.
1. Introduction
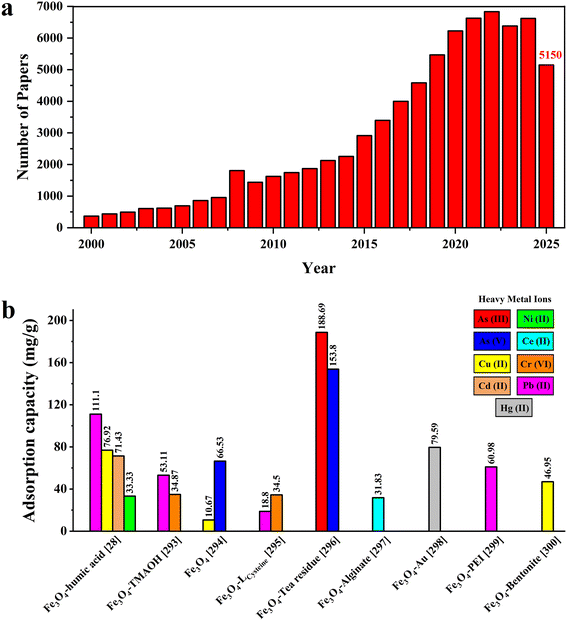
Water is the primary resource for human life, and consequently, its contamination has become one of the most pressing environmental challenges of our day. The rising demand for water has significantly compromised its quality. Rapid industrialization, agricultural production, and urbanization have resulted in the deterioration of the environment, wherein pollutants adversely affect the quality of vital water resources (rivers and oceans) and eventually compromise human health and sustainable social development.1–3 Ecosystems and human health are seriously threatened by water pollution from various sources, namely heavy metals, pesticides, viruses, and synthetic chemicals. A large percentage of global industrial and urban wastewater is discharged into the environment without treatment, thus jeopardizing human health and ecosystems. In less developed nations with inadequate wastewater treatment and sanitation infrastructure, this proportion is even greater. These issues are exacerbated by poor disposal of industrial waste, inadequate wastewater treatment, and agricultural runoff, which lead to the deterioration of water quality, loss of biodiversity, and public health risks, including dangerous diseases such as cancer, a cause of many human deaths each year.4–8 Researchers have increasingly focused on water pollution studies via various methods pertaining to the removal of assorted pollutants to improve the quality of drinking water.9 This interest is signified in the rising trend in publications on water pollutant removal over the past decades (Fig. 1), highlighting the significance of research in this field.Publication analysis shows that there have been three separate epochs of study on Fe3O4-based magnetic nanoparticles (MNPs) and their biomedical and environmental applications. Research before 2010 primarily focused on nanoparticle (NP) production, particles’ physicochemical characteristics, and the fundamentals of coagulation and flocculation. As a result of the focus on fundamental and methodological publications largely aimed at determining the mechanisms of action of NPs, their practical applications were confined to laboratory research and straightforward heavy-metal and pollutant separation. There has been a dramatic uptick in the number of articles published between 2010 and 2020, with most of the new studies focusing on environmental applications or the elimination of heavy metals, dyes, pharmaceuticals, or new forms of pollution and emerging contaminations. During this time, there was a surge in basic research on practical and integrated environmental and medical applications, as well as consideration of the biomedical, antibacterial, and nanocomposite uses for functionalized NPs to improve their performance in real-world settings. From 2020 onwards, studies have primarily focused on post-placement technologies that are environmentally friendly, have multifunctional applications, and being centered on versatile and greener technologies. Special consideration is given to environmental concerns, sustainability, material recycling, and the creation of eco-friendly NPs. These particles are used for microplastic removal, antiviral systems, photocatalysis, and a wide range of chemical applications.
The general trend indicates that research on Fe3O4 and similar NPs has shifted its focus from fundamental and preliminary synthetic investigations to practical, ecological, and biological implications, and the industrialization of these NPs including their pilot usage. This field has matured and moved on from theoretical research to multipurpose and sustainable applications, as is apparent from the explosive growth of papers from 2010 to 2020, with the expansion of practical applications and functionalization, and increased emphasis on sustainability and green technology after 2020. This development is indicative of the growing significance of MNPs in many fields, including medicine, the environment, and fundamental research that makes use of applied technology.
The social, economic, and environmental repercussions of water pollution underscore the importance of deploying efficient water purification technology to safeguard water supplies and advance sustainable development. Undeniably, two major issues facing communities worldwide are water pollution and the supply of clean drinking water. Traditional water treatment techniques for eliminating contaminants from water sources include chlorination and filtration.10,11 Microplastics and pharmaceutical pollutants are increasingly becoming issues in water treatment that traditional treatment facilities cannot eliminate. Pharmaceuticals and their metabolically-derived chemical entities infiltrate the water sources, causing hormonal imbalances and antibiotic resistance, while microplastics are created by the breakdown of abundant plastics. The use of modern and sustainable technologies is essential to protect the environment and human health, given the ineffectiveness of conventional approaches for eliminating harmful contaminants. Through the technologies deploying ozone, membrane filtration, and ultraviolet (UV) disinfection,12 there are optimistic future possibilities for the successful management of mounting pollutants and improving the water treatment efficiency.13
Water purification is one of the most important processes for maintaining human and environmental health, wherein pollutants and harmful substances in water are removed using various physical, chemical, and biological methods.14 Common water purification methods such as filtration, reverse osmosis, chlorination, UV radiation, distillation, coagulation, and ion exchange each have advantages and disadvantages. They should be deployed in combination based on the type of contamination, water source, and environmental conditions.15 Filtration, one of the simplest methods, removes suspended particles and some contaminants by passing water through porous layers, improving taste and odor. However, due to its limited removal of dissolved substances and slow filtration speed, it usually requires being combined with other methods.16 Reverse osmosis is advanced technology that uses semipermeable membranes to remove ions and dissolved contaminants extensively, but its weaknesses include high energy consumption and high water waste. On the other hand, methods such as distillation purify water through evaporation and redistillation, which are very effective but energy-consuming and costly.17,18 Chemical methods include processes such as coagulation, chlorination, and disinfection, which aim to remove colloids, microorganisms, and pathogens. Coagulation is typically achieved by adding chemicals to water, which converts suspended particles into flocs that can be easily separated. Chlorination and UV radiation are also primary disinfection methods; however, chlorination can produce hazardous byproducts, and UV radiation is ineffective at removing suspended particles without prior treatment.19,20 Biological processes are mainly used in wastewater treatment to purify water by breaking down organic matter with microorganisms.21Table 1 lists standard water purification methods, their advantages, disadvantages, and usages.
| Purification method | Advantages | Disadvantages | Applications | Ref. |
|---|---|---|---|---|
| Filtration | Low installation and maintenance costs, improved taste and odor, and removal of volatile organic compounds | Relatively low filtration speed. Need for periodic filter changes in case of high contamination load | When high-quality outlet water is required (drinking water, sensitive industrial applications) | 16 |
| Reverse osmosis (RO) | Removal of a wide range of ions and dissolved salts, and improves taste | High water and energy consumption. Removal of beneficial mineral salts, and maintenance costs | Drinking water production, seawater desalination, industrial wastewater treatment, water recycling, ultrapure water production for the pharmaceutical and electronics industries, and removal of chemical and microbial contaminants | 17 |
| Distillation | Produces water of the highest purity. No need for filters or chemicals | Slow and costly process. High thermal energy consumption | Removal of dissolved salts and minerals, destruction of microorganisms and bacteria, removal of volatile organic compounds, and production of pure, contaminant-free water for laboratory, medical, and drinking purposes | 18 |
| Adsorption | Ability to absorb organic pollutants and heavy metals. Selective design capability, and possibility of recovery | Requires large amounts of adsorbent. Saturation and need for regeneration or replacement, and cost of adsorbent | When the concentration of pollutants is relatively low, or as a supplementary step | 15 |
| Ion exchange | High efficiency at removing specific ions (nitrate, arsenic). It can be combined with other methods | Medium to high initial cost. Limited resin capacity, and need for frequent reduction | Removing water hardness by removing calcium and magnesium ions, reducing the concentration of heavy metals such as lead and cadmium, removing nitrates and sulfates, recycling resins in industrial systems, and producing ultrapure water for pharmaceutical, power plant, and electronic uses | 22 |
| Disinfection by chlorine | Cheap and available. Effective at inactivating bacteria and viruses | Production of dangerous by-products (DBPs), ineffective against some parasites | For ultimate microbial assurance in drinking water systems | 20 |
| UV radiation | A physical process without chemicals. Effective against most viruses and bacteria | No residual protective effect. Requires pretreatment to remove high amounts of suspended particles | Inactivation of microorganisms, bacteria, viruses, and fungi without the need for chemicals, prevention of algae and pathogenic agents, disinfection of drinking water and treated wastewater, and use as the final disinfection stage in advanced treatment systems | 23 |
| Coagulation/chemical flocculation | Rapid adsorption and separation of suspended particles and ions. Reduction of sludge volume and chemical residues. Possibility of recycling NPs | Need to adjust pH and optimal dosage for better performance | Often used as a primary step in municipal wastewater or surface water treatment | 24 |
| Advanced oxidation processes (AOPs) | Decomposition of persistent organic compounds, without adding excessive chemicals. Capable of reducing micropollutant compounds | High energy cost. Low selectivity in some cases. Production of intermediate products | For the removal of resistant organics, pharmaceuticals, and complex compounds | 25 |
| Combined methods (e.g. coagulation + adsorption) | Combining the advantages of methods, improves efficiency, and reduces problems such as clogging or sludge | Higher design and implementation costs. Process control complexity | In systems where the inlet water is difficult or requires a higher outlet quality | 26 |
Although conventional water treatment methods can remove many contaminants, each method has its own challenges, including high energy consumption, sludge production, chemical residues, and inefficiency at removing certain compounds. The use of coagulation, especially with the aid of MNPs, can improve water quality more effectively by rapidly adsorbing and separating suspended particles and ionic contaminants. Due to their high specific surface areas and magnetic properties, MNPs can rapidly adsorb metal ions and organic materials and subsequently be easily separated by applying a magnetic field. Also, compared to conventional coagulants, MNPs can be separated after adsorption without the need for additional chemicals, thereby significantly reducing wastewater generation. Surface coating of these MNPs increases the colloidal stability of NPs and prevents their aggregation. Moreover, MNPs can be removed from the system using a magnetic field and returned to the treatment cycle after washing and regeneration. The rapid magnetic separation and high recycling capacity of these MNPs make this technology a desirable and sustainable option for treating contaminated water. This process can be deployed as a low-cost, effective technology in industrial and municipal water treatment.27–29
Coagulation is an essential process in water treatment, removing suspended particles, colloids, and organic substances from raw water to enhance its quality. This approach entails the usage of a coagulant, often a metal salt like aluminum sulfate or ferric chloride, to destabilize and agglomerate particles via floc formation. By neutralizing particle charges, coagulants enable collisions to form larger aggregates that can settle preferentially, allowing them to be easily removed from the water.30 Frequently, the coagulation process is followed by flocculation, in which mild mixing promotes the growth of the floc, enhancing its settling characteristics and facilitating its removal in subsequent sedimentation or filtration operations. Several factors affect the efficacy of coagulation, including the type and amount of coagulant deployed, pH, temperature, and the properties of the water being treated.31 Conventional water treatment facilities depend on coagulation, which also helps to remove turbidity, color, bacteria, and other contaminants from raw water supplies. In addition to its main purpose of eliminating particles, coagulation also eliminates pathogens, dissolved organic compounds, and certain heavy metals by complexing with coagulant species or adsorbing onto floc surfaces.32 Advanced coagulation techniques, including pre-oxidation with ozone or chlorine, can enhance the elimination of organic pollutants and the overall efficacy of water treatment. Coagulation-based water treatment systems are versatile and adaptable to many water sources and processing objectives, thus rendering them essential in sustainable water management strategies designed to deliver safe and clean drinking water to global populations.33
Ceramic NPs based on iron oxide find widespread usage in several technological areas,34,35 including catalysis,36,37 energy storage,38–40 biomedical,41–43 and water treatment;44,45 popular varieties are maghemite (γ-Fe2O3), hematite (α-Fe2O3), and magnetite (Fe3O4). More recently, the assembly and application of iron oxide-based nanocomposites with innovative attributes and capabilities have been extensively investigated.46 This attention is in view of their nanoscale size, high surface area-to-volume ratio, and superparamagnetic properties.47 Superparamagnetism is a form of magnetism that occurs in small magnetic NPs, where magnetization can indiscriminately flip direction due to temperature fluctuation, leading to non-magnetic behavior in the absence of an external magnetic field.
Through simple preparations, modifications, and coatings, along with the capability of atomic-scale manipulation of matter, unique magnetic features can be realized.48,49 Additionally, Fe3O4 nanocomposites are highly compatible with biotechnology, showing great potential due to their low toxicity, chemical stability, and high biocompatibility.50,51 Studies have shown that preparative protocols and coatings on the surface of materials play a crucial function in regulating the morphology, size, surface chemistry, and magnetic attributes of the ensuing NPs.52–54,55 To date, many researchers have developed proficient physical and chemical protocols for synthesizing Fe3O4 nanocomposites.56 Recently, various approaches have been introduced for producing high-quality nanostructures, such as nano-ovals,57 nanobelts,58 nanorings,59 nanoclusters,60 and other nanoscale structures.61,62 To better understand the behavior of colloidal iron oxide particles and to improve their performance, scrutinizing their stability is of utmost importance. Iron oxide NPs tend to aggregate in aqueous and natural environments. The surface of Fe3O4 NPs is hydrophilic due to the presence of hydroxyl groups, which enhances interparticle attractive forces around neutral pH, leading to aggregation. To prevent this, the electrostatic and steric stability of the NPs must be enhanced. The presence of hydroxyl groups on their surface enables chemical bonding with various functional groups, and through surface modification techniques, the stability of these particles can be improved while introducing new functionalities. Separating contaminants using an exterior magnetic field, along with the straightforward recovery of Fe3O4 NPs for reuse, makes this approach a cost-effective and efficient solution for removing toxic heavy metals from water. In general, nanomaterials must be stabilized to prevent aggregation and to ensure low sedimentation rates, thereby maintaining their reactivity and functionality in various environments.63–65 However, it has been described that nanomaterials tend to aggregate.66 Most often, electrostatic and van der Waals (vdW) forces determine the colloidal stability of the particles.67 One promising strategy is altering nanomaterials with varied functionalities. The deployment of surfactants, polymers, and stabilizers has been widely utilized to create stable NPs with specific chemical properties and targeted functionalities.68–74
A comparative analysis of the properties, applications, advantages, and limitations of different NPs is presented in Table 2. Table 2 indicates that many NPs possess unique qualities, uses, and limits that influence their efficacy for water purification. Fe3O4-based MNPs are recognized for their facile recovery and reusability, while silver NPs are proficient at eliminating biological pollutants owing to their antibacterial properties. Photocatalytic materials like TiO2 excel at degrading persistent organic compounds, whereas graphene materials provide excellent adsorption of pollutants owing to their large specific surface areas. Bio-inspired NPs, synthesized from biological materials, offer a sustainable alternative to conventional wastewater treatment methods. Their tailored design enhances the efficiency of the removal of pollutants, while their multifunctionality and integration with traditional technologies yield superior treatment outcomes. Although bio-inspired NPs offer many benefits, industrial manufacturing still faces issues in terms of repeatability and scalability. If these limitations can be circumvented, these NPs could improve filtration and oxidation; however, in mass production, they lack advantages over Fe3O4-based MNPs and are mostly considered complementary options for water purification.75,76
| NP types | Properties | Applications | Advantages | Disadvantages | Ref. |
|---|---|---|---|---|---|
| Fe3O4-based MNPs | Magnetic characteristics, easy recovery, and good reusability | Heavy metal removal, organic pollutant adsorption, and simple separation | Reusable because of its excellent adsorption capability and magnetic recovery, easily scalable | Lack of stability in acidic environments and susceptibility to aggregation, releasing iron ions | 77 and 78 |
| Ag NPs | High contact area and antimicrobial properties | Degradation of organic contaminants, removal of microbiological pollutants | Very efficient at removing a wide variety of bacteria | Expensive, aggregation problems, and possible toxicity to aquatic life and environments | 79 and 80 |
| ZnO | High photocatalytic activity in the degradation of organic pollutants and microbial inactivation | Photocatalytic processes related to water purification, although dependence on UV light limits widespread use | Low to moderate material costs | Moderate toxicity risk | 81 |
| TiO2 | Photocatalytic efficacy and stability under UV radiation | Degradation of drugs, colors, and organic contaminants | Sustainability, reusability when exposed to ultraviolet light | Expensive, limited action in visible light; activation needs UV light | 82 and 83 |
| CuO | Good efficiency for microbial removal and effective adsorption capability | Widespread use and common production; scalable for a variety of purification applications | Low cost; cost-effective materials and synthesis routes | Risk of copper ion leakage and environmental and health hazards | 84 and 85 |
| Graphene-based materials | Excellent adsorption capacity due to high surface area | Adsorption of organic contaminants and heavy metal elimination | Excellent adsorption capacity, amenable for functionalization for targeted removal | High manufacturing costs and possible long-term environmental effects | 86 and 87 |
| Mn3O4 | Strong oxidation potential, photocatalytic activity in visible and near-visible light | Adsorption of heavy metals, dyes, and drugs, catalytic degradation of organic pollutants, and antibiotics | Environmentally friendly with minimal toxic byproducts, capable of removing a wide range of contaminants | Lower adsorption rates for some contaminants, the possibility of leaching and surface degradation, and limited selectivity | 88 |
| Bio-inspired NPs | Biocompatibility, a wide range of functions | Multi-functional elimination of organic and inorganic contaminants | Minimal toxicity, eco-friendly synthesis techniques | Large-scale manufacturing difficulties and inconsistent synthesis results | 89 |
Nanoscale materials comprising the Fe3O4 phase possess distinctive magnetic characteristics. This magnetic feature of Fe3O4 NPs makes it easier to manage and separate particles; this has led to great interest in their possible deployment in coagulation-based water purification processes.90,91 The large surface area-to-volume ratio of Fe3O4 NPs, which typically range from a few nanometers to several hundred nanometers in size, enables them to interact with water pollutants, thereby improving the effectiveness of the coagulation process. These NPs serve as a substitute coagulating agent, helping eliminate contaminants such as heavy metals, organic compounds, and microbes from water sources during purification protocols.92 They also help efficiently deposit and separate pollutants from water by encouraging the formation of larger particle agglomerates, enabling simple filtrations or sedimentation processes for their removal.93 Fe3O4 NPs are a better option for use in sustainable and effective water filtration systems because of their capacity to selectively adsorb specific pollutants and their simplicity of post-processing removal via magnetic separation. Although additional scientific studies on Fe3O4 NPs for water purification coagulation have been conducted, a thorough examination of this topic is still warranted and requires scrutiny. Herein, important new perspectives on the synthesis, mechanisms, and applications of MNPs in coagulation-based water purification systems are presented. Furthermore, the benefits, drawbacks, and opportunities of iron oxide-based MNPs are documented; these may help identify areas of study gaps for further investigations.94
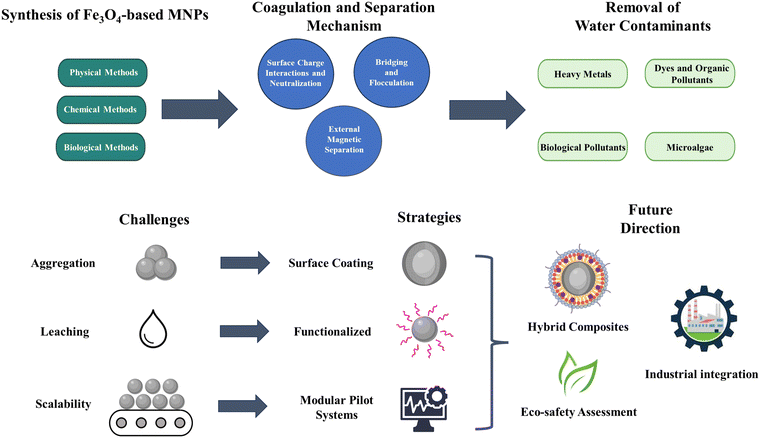
Fig. 2 presents a roadmap of the challenges, improvement strategies, and future development paths for Fe3O4-based MNPs in water purification. The roadmap is designed with an analytical and forward-looking approach and depicts the relationship between fundamental problems and technological solutions in a chain. In the challenges section, three main obstacles are identified: particle aggregation, which reduces the active surface area and decreases the adsorption efficiency; ion leaching, which threatens chemical and environmental stability; and limited scalability, which hinders the commercialization and industrial application of these nanostructures. In response to these challenges, key strategies are proposed, including coating and surface modification to prevent aggregation and enhance colloidal stability, chemical functionalization to improve selectivity and reduce the risk of ion release, and the design of modular, tunable systems at the pilot scale to enhance industrial-scale applicability. Future directions focus on developing Fe3O4 hybrid composites with higher adsorption efficiency and stability, by conducting a comprehensive eco-safety assessment to ensure the sustainable use of these NPs, and integrating the technology into large-scale industrial processes. By providing an integrated viewpoint from the perspectives of materials science, environmental engineering, and nanotechnology, this graphic map presents a coherent framework to better understand the current challenges and guide future research in the field of Fe3O4-based MNPs. Furthermore, this can be used as a strategic reference for the development of novel and sustainable water treatment processes.
2. Fe3O4-based MNP synthesis methods
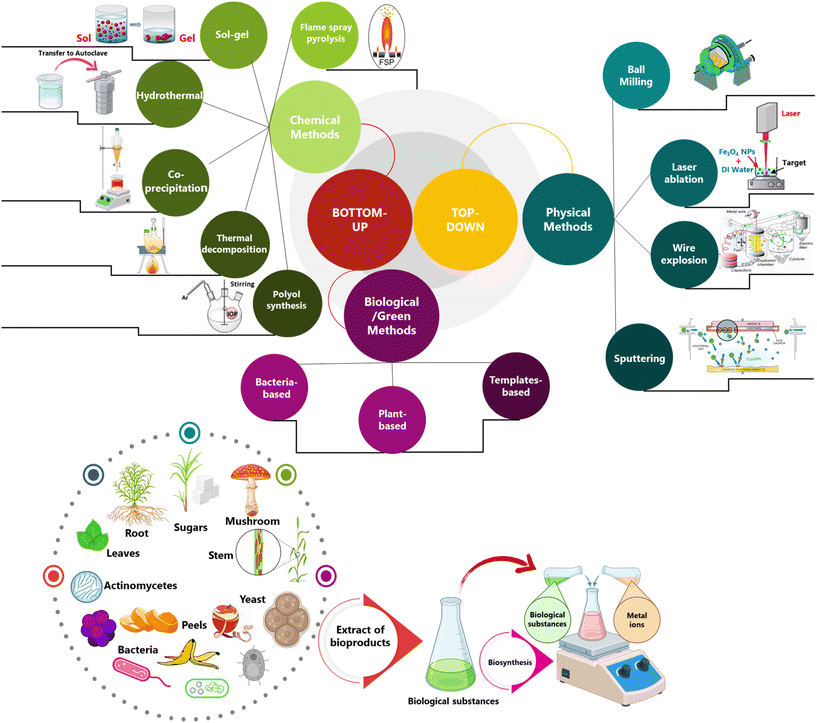
Several routes have been deployed for synthesizing Fe3O4 NPs (Fig. 3), including chemical, physical, and biological techniques,95 each with its benefits and drawbacks depending on the application. The term “physical approach” refers to a broad category of methods deployed in the synthesis of Fe3O4 NPs, including the use of electrical, radiation, and thermal energy.96 Some examples of physical approaches include ball milling, laser ablation, wire explosion, sputtering, and flame spray pyrolysis. The ball milling procedure involves crushing iron particles with a grinding agent to generate small particles. This technique is also a popular method for mechanical alloying and the fabrication of composite materials.97,98 The laser ablation technique involves directing high-energy laser pulses onto a solid iron or iron oxide target submerged in a liquid. This causes the material to evaporate and cool rapidly, ultimately leading to the formation of Fe3O4 NPs.99 During the electrical explosion of a wire, an elevated-density current pulse (exceeding the binding energy) is passed through the wire in a liquid or gas atmosphere. Consequently, the material heats up in a gust, a vivid light blazes, a mix of superheated aerosol and simmering droplets of the shattering wire material, while a shockwave disperses to the ambient atmosphere. The explosion of a base metal wire, such as Fe, generates a mixture of oxides and the metal.100 In the sputtering technique, a target is bombarded with intense ions, usually from a plasma, which ejects atoms or clusters. These atoms or clusters subsequently deposit onto a substrate in the form of a thin film or Fe3O4 NPs.101 In a vapor-phase synthesis method, often referred to as flame spray pyrolysis, precursors are burned off in the gas phase, thereby generating high-purity nanocrystals, including iron oxides.102Chemical synthesis methods are commonly used for synthesizing Fe3O4 NPs.103 The co-precipitation approach is a commonly utilized process wherein the formation of Fe3O4 precipitates is accomplished. This operation involves the hydrolysis of iron compounds, primarily FeCl2 and FeCl3, in a basic aqueous solution, often achieved by adding ammonia or sodium hydroxide.104 Another method is the hydrothermal method, which exploits superheated water in high-pressure autoclaves to synthesize Fe3O4 NPs. Under high temperature and pressure conditions, reactants become more soluble and reactive, enabling the formation of highly crystalline particles.105 Other techniques include reduction methods, using solvents such as polyalcohols (also known as polyol) to partially reduce Fe3+ precursor to produce Fe3O4 NPs.106 In the thermolysis process, typically a metal–organic precursor is decomposed in an organic solvent at elevated temperatures to form the desired NPs.107 The sol–gel method for Fe3O4-based MNPs involves reacting chemical solution precursors to form NPs through hydrolysis and condensation of the precursors, which can be performed in both aqueous and non-aqueous solvents.108 The ambient-air fabrication of MNPs entails their synthesis under ordinary atmospheric conditions, eliminating the need for controlled conditions such as under vacuum or inert gases. Benefits of this approach include decreased costs and more production scalability.109 This approach is more straightforward and cost-effective than techniques that require a controlled environment, and sol–gel synthesis in air can be one of these methods.110
Another group of Fe3O4 NP synthesis techniques is classified as biological or greener methods. Live organisms or byproducts of biological processes are essential in the biological processes deployed to generate Fe3O4 NPs. This approach is considered more eco-friendly because it can produce NPs with the desired sizes and properties without deploying hazardous chemicals.111 Using energy-efficient, environmentally friendly processes and abundant materials is central to the green synthesis method for producing Fe3O4 NPs, which aims to reduce negative impacts on the environment and human health.112
Table 3 presents a comparative analysis of different synthesis methods for the fabrication of MNPs, and for each method, a brief description of the synthesis mechanism, along with key parameters affecting the process, is provided. Biological and environmental conditions, particle size and degree of crystallinity, shape controllability, and scalability are among the indicators that have been considered for a more comprehensive analysis of these methods. Also, the advantages and disadvantages of each method have been identified separately to enable their critical evaluation. Finally, the potential applications of each method are listed according to the characteristics of the final product. Table 3 provides a comprehensive overview of the strengths and weaknesses of each technique and can be used as a good reference for selecting a synthesis method tailored to specific needs in different fields, especially environmental applications.
| Synthesis methods | Brief explanation | Important parameters/biological–environmental conditions | Particle size/crystallinity | Shape control/scalability | Advantages | Disadvantages | Potential applications | Ref. |
|---|---|---|---|---|---|---|---|---|
| Co-precipitation | Reaction of iron salts in an alkaline environment and at controlled temperature | pH, temperature, Fe2+–Fe3+ ion ratio, and stirring speed/eco-friendly | 5–20 nm/medium | Low/high | Simple, cheap, mass-produced | Less precise control of NPs size | Medicine, catalysis, magnetic storage | 113 and 114 |
| Hydrothermal | Reaction in an autoclave under high pressure and temperature | Temperature, pressure, time, concentration of precursors/eco-friendly | 10–50 nm/high | Good/medium | NPs with high crystallinity, shape control | Requires autoclave, time-consuming | Medicine, catalysis, sensors | 115 and 116 |
| Thermal decomposition | Decomposition of organoiron precursors at high temperatures in the presence of surfactants | Temperature, time, type, and concentration of precursors, type of surfactant/need for organic solvents | 5–15 nm/high | Good/medium | Precise size and shape control, uniform NPs | Higher cost, special equipment required | Medicine, magnetic data acquisition, sensors | 117 and 118 |
| Sol–gel | Gel formation from chemical precursors and then heating | pH, temperature, gelation time, type of precursor/chemically dependent | 5–30 nm/medium to high | Good/low | Uniform production, ability to control the composition | Time-consuming, may require calcination | Catalysis, coatings, medicine | 119 and 120 |
| Microemulsion synthesis | Formation of small emulsion (water/oil) droplets that act as nano-reactors | Type of emulsion, ratio of different phases, reaction time/eco-friendly | 3–10 nm/medium | Very good/low | Very good control of size and distribution | Less scalability, higher cost | Medicine, nanomedicine, catalysis | 121 and 122 |
| Electrochemical synthesis | Iron oxide deposition on the electrode by applying an electric current | Electrical current, time, electrolyte concentration/depends on the electrolyte | 5–20 nm/medium | Medium/medium | Good thickness control, simple | Need for electrochemical equipment | Magnetic films, catalysis | 123 and 124 |
| Microwave-assisted synthesis | Using microwave heating to accelerate reactions | Microwave power, time, temperature/depending on the type of material | 5–20 nm/medium to high | Good/medium | High reaction speed, uniformity | Special equipment | Medicine, catalysts, sensors | 125 and 126 |
| Flame spray pyrolysis | Spraying the precursor solution into a very high-temperature flame | Temperature, flame speed, precursor type, spray speed/dependent on flame fuel | 5–50 nm/medium to high | Low/very high | Fast production, suitable for industrial scale | Challenging size control | Industrial production, medicine, energy storage | 127 and 128 |
| Ball milling (mechanical milling) | Mechanical grinding of powders to produce NPs | Grinding time, grinding speed, ball size, ball to powder ratio/depends on the grinding conditions | 20–200 nm/medium | Low/high | Simple, cheap, mass-produced | Pollution, irregular size, high energy | Industrial production, magnetic materials, catalysis | 129 and 130 |
| Sputtering | Vapor deposition of atoms from a metal target onto a substrate by ion bombardment | Gas pressure, power, target–substrate distance/requires a vacuum and special equipment | 1–10 nm (thin film)/high | Good (film)/low | High-quality thin films | High cost, special equipment | Manufacturing of magnetic films, sensors | 131 and 132 |
| Spray pyrolysis | Precursor solution spraying and rapid heating | Temperature, spray speed/dependent on precursors | 20–200 nm/medium | Low/high | Scalable, rapid production | Less size control | Industrial production, catalysis, sensors | 133 and 134 |
| Ultrasonic-assisted synthesis | Using ultrasound to enhance the reaction | Ultrasound intensity, time/a relatively consistent environment | 5–30 nm/medium | Good/low | Better size distribution | Low scalability | Catalyst, nano-drug, nanostructures | 135 and 136 |
| Laser ablation | High-energy laser irradiation of a metal target in liquid to produce NPs | Laser power, pulse time, liquid type/chemical-free | 5–30 nm/high | Good/low | Chemical-free, good control | Expensive equipment, small scale | Medicine, catalysis, electronics | 99 and 137 |
| Thermal plasma synthesis | Using plasma for evaporation and the formation of NPs | Temperature, pressure, and carrier gas/dependent on plasma conditions | 10–100 nm/high | Medium/high | Rapid production of NPs | High cost | Industrial production, energy storage, catalysis | 138 and 139 |
| Wire explosion | Electric explosion of metal wire in a gaseous or liquid environment to produce NPs | Explosion current, environment, wire/dependent on the explosion environment | 5–50 nm/medium to high | Medium/medium | Rapid production of uniform NPs | Need for specialized equipment | Industrial production, energy storage | 140 and 141 |
| Polyol synthesis | Synthesis in a polyol solvent (such as ethylene glycol) as the reducing agent and reaction medium | Temperature, time, type of polyol, precursor concentration/eco-friendly | 5–20 nm/high | Good/low | Size control, good dispersion, and simple | Long reaction time | Nanomedicine, catalysis | 142 and 143 |
| Biosynthesis | Using bacteria or plant extracts to reduce ions | Type of microorganism, culture conditions/very eco-friendly | 10–50 nm/medium | Low/low | Eco-friendly, environmentally friendly | Less control over size and shape | Medicine, pharmacy | 144 and 145 |
Bio-reduction allows some microbes, such as fungi and bacteria, to reduce Fe compounds into iron oxide NPs. Alternatively, some plant extracts or enzymes might be employed as reducing agents during the MNP synthesis process.146–148 Also, the use of plant extracts or natural components as stabilizing and reducing agents is a common component of this approach during the synthesis of MNPs.149,150
As presented in Table 4, biosynthesized Fe3O4 NPs can be divided into two main groups, namely microbial synthesis and phytosynthesis. The general steps for the synthesis of Fe3O4 NPs using the microbial method entail the selection of the appropriate microorganism, cultivation of the microorganism in a medium enriched with metal ions, bio-reduction of ions, extraction and purification of NPs, followed by analysis and identification of NPs.151,152 The advantages of the microbial method include biocompatibility and non-toxicity, a lower energy requirement than physical and chemical methods, the production of NPs with uniform size and shape, and the ability to control the surface coating of the NPs (utilizing biological materials produced by cells).153 Additionally, among the broader applications of MNPs synthesized by microbial methods, targeted drug delivery, MRI imaging, magnetic hyperthermia (in cancer treatment), and the removal of environmental pollutants (such as heavy metals) can be mentioned.154–157
| Microbial synthesis | Phytosynthesis | ||||||
|---|---|---|---|---|---|---|---|
| Intracellular | Extracellular | Plant extract (beans, roots, fruits, leaves, seaweed, seeds) | Plant wastes | ||||
| Magnetospirillum sp. AMB-1 | 162 | Geobacter metallireducens GS-15 | 163 | Annona muricata hot water extract | 164 | Musa acuminata peel ash hot water extract | 165 |
| Aquaspirillum magnetotacticum | 166 | Thermophilic bacteria TOR-39 | 167 | Mimosa pudica hot water extract | 168 | Banana peel ash and Colocasia esculenta leaf water extract | 169 |
| Pseudomonas aeruginosa Kb1 | 170 | Lactobacillus casei cytoplasmic extract | 171 | Platanus orientalis L. hot water extract | 172 | Watermelon rind water extract | 173 |
| Magnetospirillum magnetotacticum | 174 | Clostridium sp. | 175 | Carob hot water extract | 176 | Plantain peel extract | 177 |
| Magnetospirillum magneticum AMB-1 | 161 | Verticillum sp. | 178 | Carob extract | 179 | Aqueous extract of spent tea waste | 180 |
| Acidithiobacillus ferrooxidans | 159 | Actinobacter spp. | 181 | Kappaphycus alvarezii aqueous extract | 182 | Syzygium cumini seed extract hot water extract | 183 |
| Magnetospirillum gryphiswaldense | 160 | Spirogyra sp. | 184 | Aloe vera water extract | 185 | Chinese cabbage (Brassica rapa subsp. pekinensis) outer leaves extract | 186 |
| Bacillus cereus strain HMH1 | 187 | Cicer arietinum L. water extract | 188 | Citrus unshiu tangerine peel water extract | 189 | ||
| Geobacter sulphurreducens | 190 | Padina pavonica hot water extract | 191 | Pomegranate, lemon, orange, and apple peels in hot water extract | 192 | ||
| Spirulina sp. | 184 | Shanghai white tea (Camellia sinensis) | 193 | Corn silky hair (Zea mays L.) water extract | 186 | ||
| Fusarium oxysporum | 178 | Brown Sargassum muticum water extract | 95 | ||||
| Thermoanaerobacter ethanolicus TOR-39 | 194 | Stevia fresh water extract | 195 | ||||
| Clostridium sp. | 196 | Urea and gram-bean (GBE, Cicer arietinum L.) extract | 180 | ||||
| Shewanella sp. | 197 | Desmodium gangeticum hot water extract | 198 | ||||
| Protoparmeliopsis muralis | 199 | Carica papaya hot water extract | 185 | ||||
| Passiflora tripartita methanol/water extract | 200 | ||||||
| Neem leaf hot water extract | 201 | ||||||
| Rhus coriaria hot water extract | 202 | ||||||
| Amaranthus spinosus water extract | 203 | ||||||
| Psidium guajava and Moringa oleifera hot water extract | 204 | ||||||
| Chromolaena odorata extract | 205 | ||||||
| Hordeum vulgare water extract | 206 | ||||||
| Sargassum acinarium hot water extract | 191 | ||||||
| Azadirachta indica hot water extract | 207 | ||||||
| Soya bean sprouts | 208 | ||||||
| Albizia adianthifolia hot water extract | 209 | ||||||
| Flax extract | 179 | ||||||
| Andean blackberry hot water extract | 210 | ||||||
| Fig (Ficus carica) ethanolic extract | 211 | ||||||
| Eichhornia crassipes hot water extract | 212 | ||||||
In the synthesis of MNPs via microbial means, there are two main sites for the formation of NPs: intracellular and extracellular synthesis. In intracellular synthesis, NPs are formed inside the microorganism cell; metal ions enter the cell and are reduced by enzymes or metabolic pathways to generate NPs.158 Wu et al.159 investigated the intracellular synthesis of Fe3O4 NPs using the bacterium Acidithiobacillus ferrooxidans (Fig. 4A). Results revealed that the addition of sulfur to a medium containing divalent iron delayed microbial growth and iron oxidation. TEM micrographs showed that iron was an essential energy source for NP synthesis in cells and that varying the iron-to-sulfur ratios did not significantly affect the bacterial appearance or NP size, whereas in the absence of iron, MNPs were not formed. Furthermore, the observed morphology was club-shaped under ferrous iron and rod-shaped under elemental sulfur. Lang and Schuler160 examined the process of magnetosome biomineralization in magnetotactic bacteria (Magnetospirullum gryphiswaldense). Magnetosomes are nano-sized organelles that are membrane-enclosed, form magnetic iron crystals, and are arranged in a regular chain structure within the cell. This process is genetically controlled and involves the accumulation of iron, crystal deposition in a specific compartment, and organization and alignment of the particle chains. Contrary to what is expected from most inorganic systems, the Fe3O4 crystals produced by these bacteria have uniform morphology and size. Yoshino et al.161 synthesized thermo-responsive MNPs using a magnetosome display system (Fig. 4B). The results revealed that bio-nanoparticles were synthesized from magnetotactic bacteria (Magnetospirillum magneticum AMB-1) and a decamer peptide called ELP10, which became hydrophobic above its transition temperature, thereby immobilizing on the NPs. Similarly, at 60 °C, the hydrophobicity of the NP surface increased compared to that at 20 °C, leading to particle aggregation via hydrophobic and magnetic interactions. Size distribution analysis confirmed that with increasing temperature up to 60 °C, the immobilization effect of ELP10 caused more particle aggregation, resulting in faster collection of MNPs under an external magnetic field at 60 °C.
 | ||
| Fig. 4 (A) TEM images of individual cells collected from media with various ferrous/sulfur element concentrations (g per 100 mL) in the growth medium: (a) 4.47/—, (b) 4.47/0.8, (c) 4.47/1.6, and (d) —/1,159 reproduced from ref. 159 with permission from MDPI, copyright 2025; (B) procedure for the production of ELP-MNPs by the magnetosome display system,161 reproduced from ref. 161 with permission from American Chemical Society, copyright 2025; (C) (a) SEM image of Haejae-2 and (b) TEM image of the cell's cross section showing precipitated Fe3O4 NPs adjoining the cell exterior,175 reproduced from ref. 175 with permission from MDPI, copyright 2025. | ||
On the other hand, in extracellular synthesis, NPs are synthesized in the environment outside the cell (e.g., culture medium or supernatant solution). Microorganisms secrete enzymes, proteins, or metabolites into the environment that reduce the metal ions to form NPs.213–215 Fatemi et al.187 succeeded in the biosynthesis (extracellular) of magnetic iron oxide NPs using the Bacillus cereus strain HMH1. The results showed that the NPs produced had nearly monodisperse spherical morphology with an average diameter of around 29 nm. In another study by Kim and Roh,175 the microbial growth conditions were investigated to control the size of Fe3O4 NPs synthesized by Fe(III)-reducing bacteria, isolated from sediments of intertidal areas of South Korea (Haejae-2 mixed culture bearing Clostridium sp.) (Fig. 4C). The results showed that these bacteria were capable of producing Fe3O4 NPs with diameters ranging from 2 nm to 10 nm. Furthermore, MNPs produced through microbial processes exhibited superparamagnetic behavior, rendering them highly valuable for various biotechnological and medical applications.
Another biosynthesis process for Fe3O4 NPs is the phytosynthesis method, which is a greener biological means to generate NPs wherein plant extracts (from beans, roots, fruits, leaves, seaweed, and seeds) or residual plant wastes are deployed to reduce metal ions and convert them into metal or metal oxide NPs.216Table 4 highlights some of the research conducted via this phytosynthesis strategy. Plant extracts such as aloe vera,185 green tea leaf,203,217 and soybeans208 have been successfully employed to synthesize MNPs. Some examples of beneficial secondary metabolites found in plant extracts include polyphenols, which are present in both green tea and grape seed extracts. Stabilization and agglomeration of MNPs are two crucial roles that these metabolites perform. By conducting the synthesis at moderate temperatures and pressures, this approach can eliminate the use of organic solvents and hazardous compounds, which are detrimental to the environment. This approach not only produces high-quality MNPs but also promotes sustainability by enabling their use in various areas, such as water purification, waste treatment, heavy metal removal, and biomedical applications. Prodigious adoption of this methodology has the potential to accelerate the development of nanotechnology and its potential applications to improve people's lives worldwide by advancing technologies that are less harmful to the environment and more sustainable.218–220
3. Coagulation mechanism for Fe3O4-based MNPs
The coagulation mechanism generally removes organic substances through three primary mechanisms: negatively charged organic colloids aggregate with positively charged metal ions; organic molecules form precipitates with metal ions; or adsorption onto the surface of alum, leading to coagulation. In recent years, the use of MNPs has gained significant attention in view of their exclusive magnetic attributes, large specific exterior area to volume ratios, and relatively easy recovery through the application of magnetic fields. The coagulation mechanism involving MNPs can operate through various physical or chemical processes, enabling the removal of heavy metals, suspended pollutants, and other water impurities. The coagulation methods for each mechanism via the deployment of Fe3O4 NPs are described in the following sections.2213.1. Surface charge interactions and neutralization
The mechanism of electrostatic interactions and charge neutralization involves several steps. Techniques include zeta potential assessment, which is used to evaluate the surface charge of particles, thereby providing insights into their stability and inclination to assemble. This helps to manage electrostatic interactions between the NPs. Theoretical models, such as DLVO theory, provide a systematic framework for understanding how vdW and electrostatic forces interact to determine the stability or instability of colloidal systems during coagulation.222,223 Industries may maximize operations, such as flocculation in wastewater treatment or the generation of stable dispersions in pharmaceutical formulations, by altering these interactions via pH adjustment or the addition of electrolytes. A schematic for the neutralization of MNPs is depicted in Fig. 5a. Initially, Fe3O4 molecules dissociate in water and release Fe2+ and Fe3+ ions, which have the opposite charge to that of the colloidal particles. These ions are adsorbed onto the surface of the negatively charged colloids, reducing their surface electrostatic charge and, consequently, inducing electrostatic repulsion between them. This phenomenon initiates the flocculation process. In addition to reducing electrostatic charges, Fe3O4 coagulants also function as a bridging agent through vdW forces, facilitating further binding between colloidal particles and promoting the formation of larger aggregates. As the repulsive forces diminish and the bridging effect of the coagulant increases, the colloidal particles readily adhere to one another, forming larger flocs that can either settle under the effect of gravity or be removed via simple filtration.224–226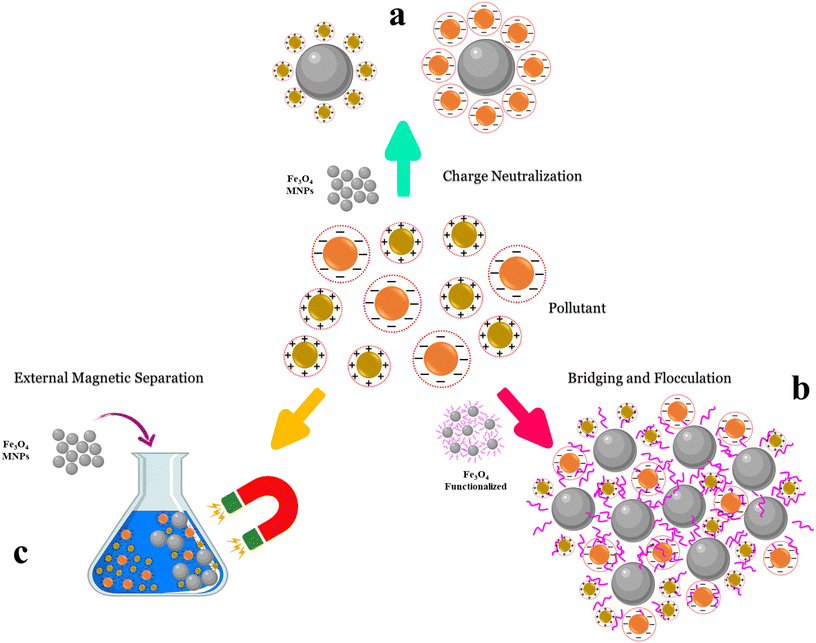 | ||
| Fig. 5 Schematic of the coagulation mechanism using Fe3O4 NPs: (a) charge neutralization, (b) bridging and flocculation, and (c) external magnetic separation. | ||
Among the factors influencing the efficiency of coagulation via this mechanism, pH is a key parameter, along with the coagulant dose, which must be precisely controlled to achieve optimal pollutant removal. pH affects not only the ionization of coagulants but also the behavior, or speciation, of contaminants dissolved in water. An optimal procedure for pollutant removal via Fe3O4-based MNP coagulation requires careful pH control. Assorted pollutants require different pH ranges for effective coagulation.227,228 For example, neutral pH is generally suitable for inorganic particles, whereas an acidic pH is preferred for the removal of humic organic substances via charge neutralization mechanisms.229 Moreover, lower pH increases the positive charge on coagulant species, thus enhancing the neutralization of negatively charged colloids.229,230 However, extremely low or high pH levels may reduce the efficiency of the coagulation process, either by decreasing the reactivity of the coagulant or by altering the physicochemical characteristics of the particles in solution.229
Studies have demonstrated the significant impact of pH on removal efficiency for pollutants. For example, as reported by El-Nemr et al.,231 the removal percentage and equilibrium adsorption capacity of substances like Pea-B and Pea-BO-NH2 tend to increase with rising pH values. By increasing the pH (from 2 to 7), the removal efficiency and the amount of adsorbed Cu2+ ions increased significantly for both biochars, because at low pH, H+ ions competed with Cu2+ ions for adsorption sites. Still, at higher pH, the biochar surface functional groups are deprotonated and acquire a more negative charge, thereby enhancing the electrostatic adsorption of Cu2+. In the context of Fe3+ ion removal with activated carbon generated from horse chestnut shells, the removal percentage begins at around 75% at pH = 2, ascends to about 95% at pH = 4, and thereafter declines to around 85% at pH = 12. The equilibrium adsorption capacity for Fe3+ ions fluctuates with pH, reaching a minimum at pH 6 and increasing at both lower and higher pH values.232 In conclusion, pH and coagulant dosage are critical factors for optimizing the coagulation performance of Fe3O4-based MNPs. These parameters influence the surface charge of both the coagulant and contaminants, thereby affecting the primary coagulation mechanisms, including charge neutralization and adsorption.233,234
3.2. Bridging and flocculation
The bridging and flocculation mechanism plays a key role in the coagulation process. It involves the interaction between colloidal particles and coagulant molecules, resulting in the formation of larger flocs that can be easily settled or filtered. Initially, in the bridging mechanism, polymeric coagulant molecules form bridges between colloidal particles. Polymers such as polyacrylamides or polysaccharides physically adsorb multiple colloidal particles, forming connections that lead to particle aggregation. Unlike the charge neutralization mechanism, which relies on electrostatic forces to neutralize surface charges, the bridging and flocculation mechanism relies on the physical adsorption of polymer chains onto colloidal particles. This brings the particles closer together, thereby promoting the formation of larger aggregates. These interactions may occur through chemical bonds or electrostatic forces, leading to the formation of initial clusters known as microflocs (Fig. 5b).During the flocculation stage, due to water flow and agitation, microflocs interact and connect, forming larger and denser flocs. This process is governed by vdW, electrostatic, and hydrophobic forces between adjacent particles. The growth of flocs during flocculation largely depends on Brownian motion, collision kinetics, and the adhesiveness between microflocs.235–237 However, the nature and strength of the interparticle forces determine whether these collisions result in effective attachment and growth of the floc. As flocs grow, their density increases, facilitating their sedimentation or flotation and allowing their removal from the water. The sedimentation or flotation behavior of the flocs depends on the specific gravity of the surrounding liquid, which serves as a regulatory factor. Finally, sedimentation under gravity and subsequent filtration are employed for separation. This process is critical for achieving clean water suitable for drinking, industrial, and agricultural applications. Temperature has a significant impact on reaction rates and molecular mobility in solution.221,238,239 Generally, higher temperatures can enhance coagulation and flocculation rates by increasing the kinetic energy of particles in solution.240 However, it should be noted that the effect of temperature is not uniform and depends on the type of coagulant deployed and the specific reaction conditions. Therefore, adjusting the temperature based on these operational parameters is crucial for achieving optimal performance in the coagulation process.241 Studies have shown that higher temperatures lead to higher adsorption by the coagulant, and the mechanism of colloidal particle adsorption may shift from physical to chemical.94
3.3. External magnetic separation
Contaminants such as heavy metals and organic compounds often carry electric charges but lack magnetic properties, so they cannot be directly attracted by magnets. Therefore, one effective approach to remove colloidal particles and pollutants is to use magnetic intermediaries that enable magnetic field-based separation. In the external magnetic separation mechanism, MNPs are used for pollutant removal and can be dispersed in water; their mechanism is illustrated schematically in Fig. 5c. First, these MNPs are surface-functionalized and bestowed with an electric charge. These charged NPs then adsorb oppositely charged pollutants via surface adsorption, thereby attaching to them. Once the MNPs bind to specific contaminants in water, such as heavy metals, organic pollutants, or microorganisms, an external magnetic field is applied to the water sample. The magnetic field attracts MNPs, and since the pollutants are attached to them, the entire NP–pollutant complex is influenced by the magnetic field. Ultimately, this process facilitates the aggregation of pollutants into larger clusters in a specific area of the water sample. After magnetic separation, the pollutants can be easily removed from the water using magnets or magnetic separation techniques.27,242,243 This rapid process eliminates contaminants without requiring complex filtration systems or chemicals, making it an affordable and environmentally friendly method for water purification. Achieving the ideal efficiency in the separation process depends critically on the MNP dose (i.e. the quantity of MNPs added to the solution). Generally speaking, increasing the MNP dosage improves the process efficiency, as more material becomes available to adsorb dissolved particles and form larger flocs.244Overdosing with MNPs can result in chemical waste. Moreover, the ensuing numerous encounters between MNPs can promote aggregation and clumping, thereby lowering their efficiency. Furthermore, excess MNP application might increase the electrostatic repulsion in the solution, thereby separating colloidal particles and inhibiting their aggregation. These factors could together cause the MNP's total adsorption capacity to drop. Consequently, a key phase in the design and running of water or wastewater treatment systems is the adjustment of the MNP dosage.94,245
4. Removal of water contaminants by MNPs
Contaminants found in wastewater are among the most problematic, as they do not biodegrade in most cases. Examples of these pollutants include heavy metals, organic pollutants, microalgae, bacteria, and viruses, which are responsible for causing significant health issues in living organisms.246 This section discusses the elimination of water pollutants utilizing iron oxide-based MNPs.4.1. Heavy metal removal
The crust, soil, and water on our planet contain a variety of heavy metals. Anthropogenic demands, such as the use of pesticides and herbicides, as well as industrial waste and the combustion of fossil fuels, cause these substances to be present at greater quantities than they normally occur. Industrial runoff containing heavy metal ions has been more prevalent over the last many decades.247 Heavy metal ions are dangerous to humans, animals, and the environment, even at low concentrations, and therefore are a major problem for freshwater supplies.248 Mental disorders, musculoskeletal discomfort, gastrointestinal issues, visual difficulties, chronic weariness, and a heightened vulnerability to fungal infections are some of the chronic degenerative illnesses that may be caused by heavy metal poisoning.249–251The heavy elements mentioned are usually introduced into the environment through human activities such as mining, fossil fuel combustion, industrial waste disposal, and wastewater treatment. Although these elements are used in many areas, such as the production of electronic equipment, paints, batteries, and advanced alloys, their uncontrolled release into the ecosystem can cause severe biological effects. These effects include cytotoxicity, neurological disorders, damage to the kidneys and immune system, and even the occurrence of chronic diseases such as cancer. Therefore, understanding the sources, uses, and effects of these elements is essential for pollution control and environmental risk assessment.35,36 In this regard, the advancement of nanotechnology, especially in the field of NP development, has provided new solutions for removing heavy metals from water. MNPs play a key role in the purification of water contaminated with heavy metals due to their high specific surface areas, easy recovery, and high adsorption efficiency. Table 5 examines the heavy elements from various perspectives and categorizes them by source of release into the environment, industrial applications, and biological effects.
| Heavy metals | Sources of release to the environment | Applications | Biological effects | Ref. |
|---|---|---|---|---|
| Aluminum | Natural and industrial resources, exchange with magnesium and calcium | Food, pharmaceutical, and construction industries | Effects on biological systems, binding to citrate, transport in blood, storage in bone, accumulation in the brain, involvement in Alzheimer's disease, bone weakness, and oxidative stress | 252 and 253 |
| Antimony | Batteries, flame retardants, ceramics, alloys | Coal combustion, plastics industry, flame arresters | Skin and eye irritation, respiratory toxicity, cardiac disorders, and potential carcinogenicity | 254 and 255 |
| Arsenic | Gold mining, natural erosion, geological processes, biological activity, volcanic eruptions, and fossil fuels | Car batteries, paints and pigments, pesticides, textiles, medical waste, semiconductors, electronics industry, food additives, cement, and the glass sector | Skin damage, hyperkeratosis, skin cancer, lung, liver, kidneys, bladder, digestive problems, high blood pressure, reproductive problems | 222 and 256 |
| Cadmium | Weathering of rocks, forest fires, volcanic eruptions, waste incineration, metal plating, fertilizers, PVC products, cadmium alloys, batteries | Industrial products, batteries, alloys, and plastics | Accumulation in the body causes damage to the nervous system, liver, lungs, kidneys, heart, carcinogenicity, mutagenicity, pulmonary fibrosis, severe digestive problems, and death | 257 and 258 |
| Chromium | Industrial activities, leather, paints, stainless steel | Chromic acid production, dyes and pigments, wood preservation, tanning, paint and printing, oil refining, stainless steel, textiles, pulp | Cancer, kidney and liver damage, lung tumors, severe diarrhea, skin allergies, breathing problems, internal bleeding | 259 and 260 |
| Cobalt | Paint industry, rechargeable batteries, special alloys, and mining | Automotive, metalworking, paint, and electronics industries | Low blood pressure, vomiting, nausea, heart disease, vision problems, loss of appetite, thyroid damage, hair loss, bleeding, diarrhea, genetic changes | 261 and 262 |
| Copper | Submarine sediments, wiring, plumbing, alloys, and dietary supplements, using contaminated water for irrigation, garbage disposal | Electrical wiring, plumbing, electronics, white goods, brass and bronze alloys, dietary supplements, oil refining, mining, tanning | Diarrhea, vomiting, stomach pain, dermatitis, asthma, bronchitis, anemia, kidney and liver damage | 263 and 264 |
| Iron | Natural, insoluble at normal pH, water sediments, iron pipes, municipal and industrial wastewater, erosion of rocks and soil | Body needs, enzymes, hemoglobin, metallurgy, energy storage | Excess iron leads to oxidative stress, liver damage, fibrosis, cirrhosis, and damage to cell membranes | 265 and 266 |
| Lead | Batteries, paints, paper, galena ore (PbS), mining, papermaking, tanning, metal smelting, pesticides | Batteries, paint, soundproofing materials, metal industry, agriculture | Decreased IQ in children, mental disorders, high blood pressure, anemia, migraines, muscle pain, memory disorders, Alzheimer's, and bone formation disorders | 267 and 268 |
| Mercury | Mining, thermometer and barometer production, electroplating, catalysts, fluorescent lamps, batteries, electric switches, pharmaceuticals | Thermometers, medicines, and solar cell industry | Tremors, insomnia, memory loss, neurological disorders | 269 and 270 |
| Nickel | Waste incineration, mining, fuel, coal, ash, boilers, cement, pesticides, and electroplating | Ceramic industry, plating, glazing, and power plants | Lung and bone cancer, chest pain, shortness of breath, dermatitis, skin allergies, cyanosis, headache | 271 and 272 |
| Selenium | Chemical fertilizers, oil refining, and power plants | Dietary supplements (in low doses), semiconductors | High doses cause hair loss, brittle nails, nervousness, and nerve damage | 273 and 274 |
| Platinum | Platinum group elements in the Earth's crust, automotive catalysts, jewelry, anticancer drugs, road dust | Jewelry, anti-cancer drug, car catalyst, automotive and pharmaceutical industries | Allergies, asthma, nausea, hair loss, miscarriage, eczema | 275 and 276 |
| Tellurium | Industrial waste, heavy metal purification | Alloys, electronics industry, and solar energy | Garlic breath odor, fatigue, digestive disorders, liver toxicity | 277 and 278 |
| Tin | Metal containers, alloys, and preservatives | Cans, kitchenware, polyvinyl | Chronic toxicity, kidney damage, abdominal pain, nervous irritation | 279 and 280 |
| Vanadium | Earth's crust, shock-resistant alloys, therapeutic supplements, ceramic pigment | Ceramic industry, complementary therapy, spring steel, and additives | Throat, nose, and eye irritation, nosebleeds, dizziness, headache, nausea, skin rash, nervous disorders, liver and kidney bleeding, tremors, paralysis, behavioral changes, heart disease | 281 and 282 |
| Zinc | Paint, sulfate fertilizers, battery, and plating industries | Paint, pigments, rubber, and chemical industries, batteries, manufacturing, electroplating, needed by the body, enzymes, nervous system | Gastroenteritis, peritonitis, growth retardation, stomach pain, skin irritation, diarrhea, vomiting, anemia, shock, death | 283 and 284 |
As shown in Fig. 6a, the number of publications on heavy metal removal from water has risen significantly from 2000 to 2022. This trend has continued steadily since then, indicating that this problem is becoming more prevalent. Over the last several years, there has been a growing emphasis on the use of inexpensive alternative nanomaterials as prospective sorbents for the elimination of heavy metals. To enhance the exterior adsorption capacity and to form stable complexes with aqueous-based metal ion species, it is desirable to have high porosity and a large surface area among small particles.285 The use of Fe3O4 NPs for the adsorption and deposition of metal ions, as well as for magnetic separation, is an essential method for managing waste and recovering valuable components from metal-containing waste.286 The interactions between metal ions and functional groups present on the surface of Fe3O4 NPs enable the sorption of metal ions from a medium onto the surface of the MNPs, an adsorption process.287,288 The surfaces of Fe3O4 NPs carry specific charges, such as carboxylate (COO−) or hydroxyl (OH−) groups, and may serve as sites for metal ion adsorption.289,290 Metal ions may undergo adsorption and then precipitate out as bigger solids; these can then be separated by magnetic separation methods. Metals may be more efficiently and effectively removed from solutions or debris with this method.291,292Fig. 6b presents some of the recent research on the usage of Fe3O4 NPs to remove heavy metals.28,293–300
Fe3O4-based MNPs are promising adsorbents and demonstrate exceptional reusability for the elimination of aqueous metal ions in wastewater treatment processes. Rajput et al.293 produced Fe3O4 MNPs via the chemical co-precipitation method for the removal of aqueous Cr6+ and Pb2+ (Fig. 7a). The study examined the impact of pH, revealing that maximum removal of Cr6+ and Pb2+ occurred at pH 2.0 and 5.0, respectively (Fig. 7b). Xue et al.28 synthesized Fe3O4 MNPs modified with oxidized humic acid (HA-O/Fe3O4) by chemical amendment to enhance the adsorption capacity and investigated its adsorption of Ni2+, Cu2+, Pb2+, and Cd2+ ions (Fig. 7c). HA-O/Fe3O4 demonstrated accelerated kinetic performance for the removal of these ions, adhering to the pseudo-second-order kinetics equation, while the adsorption isotherm aligns with the Langmuir model, exhibiting the highest adsorption capacities of 111.10, 76.92, 71.43, and 33.33 mg g−1, respectively. The interaction among metal ions and the functionalities on the adsorbent was further examined, revealing that adsorption occurred via complexation at high pH and cation exchange at low pH (Fig. 7d).
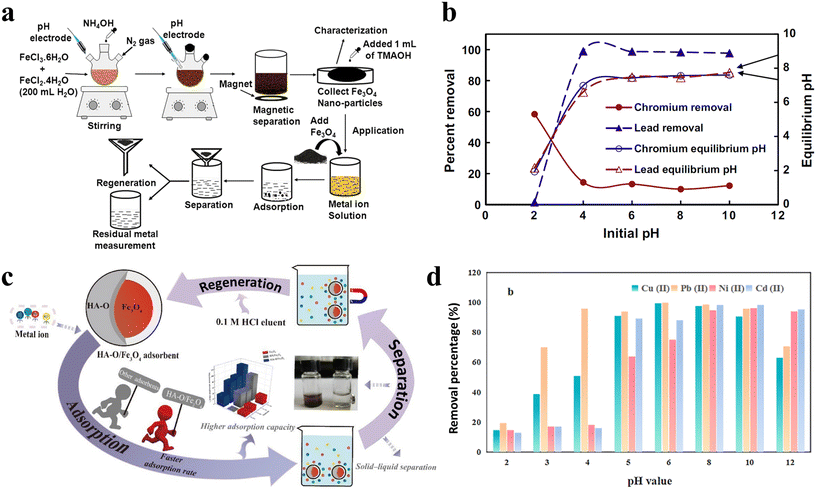 | ||
| Fig. 7 (a) Schematic of the synthesis process and heavy metal removal steps for Fe3O4 MNPs, (b) effect of pH on chromium (Cr6+) and lead (Pb2+) adsorption of Fe3O4 MNPs,293 reproduced from ref. 293 with permission from Elsevier, copyright 2025; (c) schematic of heavy metal removal steps and separation of HA-O/Fe3O4 MNPs, (d) effect of pH on heavy metal adsorption of HA-O/Fe3O4 MNPs,28 reproduced from ref. 28 with permission from Elsevier, copyright 2025. | ||
One important part of waste management is magnetic separation, which is both efficient and environmentally safe, with the key component being the materials’ response to magnetic fields.301 The process of metal removal involves exposing materials containing metal to a magnetic field, thus enabling their separation from non-magnetic material.302 Sophisticated automated magnetic separation equipment is often used to accomplish this partition, thereby improving processing efficiency and allowing capacity enhancement.303 Adsorption and deposition of metal ions onto MNPs, combined with magnetic separation methods, enhance the elimination of metal and metal ions from waste and solutions. Magnetic precipitation and separation allow for more complete separation of metals from solution or waste materials while also separating them from other materials.304 This technique improves metal removal efficiency, minimizes environmental impact, and reduces the need for additional raw materials.305
4.2. Removal of dyes and organic pollutants
In the context of our day-to-day operations, dyes and other organic pollutants are encountered to a significant extent due to their widespread usage in textiles, food, medicines, medical diagnostics, cosmetics, and other industries.306–308 The occurrence of these substances in aquatic effluent, even at very low dilutions, is exceedingly hazardous and undesirable.309 The projected yearly average total amount of dyes deployed worldwide by enterprises is deemed considerable.310 Consequently, studies conducted by academic researchers on the elimination of organic pollutants and dyes have shown considerable growth (Fig. 8a). Various metal oxide-based NPs are utilized for the removal of dyes from wastewater.311–314 Among different metal oxides, the elimination of dyes from such marine liquids is a common application of Fe3O4 NPs, whether they are pure, doped, or in composite form.315,316Organic substances may be adsorbed onto Fe3O4 NP surfaces from solutions, wherein hydrogen or vdW bonds can form between organic molecules and functional groups.317 Surfaces of MNPs with varying charges may reduce the concentration of liquid organic pollutants by acting as adsorption sites for these compounds. In addition to adsorption, Fe3O4 NPs can oxidize or undergo photocatalytic processes, both of which degrade organic contaminants.318 Numerous recent studies have used Fe3O4 NPs for the adsorption and degradation of dyes and organic chemicals, as depicted in Fig. 8b.319–327
The synthesis of the Fe3O4@MnO2 core–shell nanocomposite for the removal of methylene blue and As5+, two major pollutants, from water (Fig. 9a) has been described.320 As shown in Fig. 9b, Fe3O4 cores are coated with a MnO2 shell. The Fe3O4@MnO2 core–shell nanocomposite exhibited ferromagnetic behavior and a maximum adsorption capacity of 146.1 mg g−1 for methylene blue and 16.6 mg g−1 for As5+; it has the potential to be an effective adsorbent for the elimination of methylene blue and As5+ from aquatic media.328
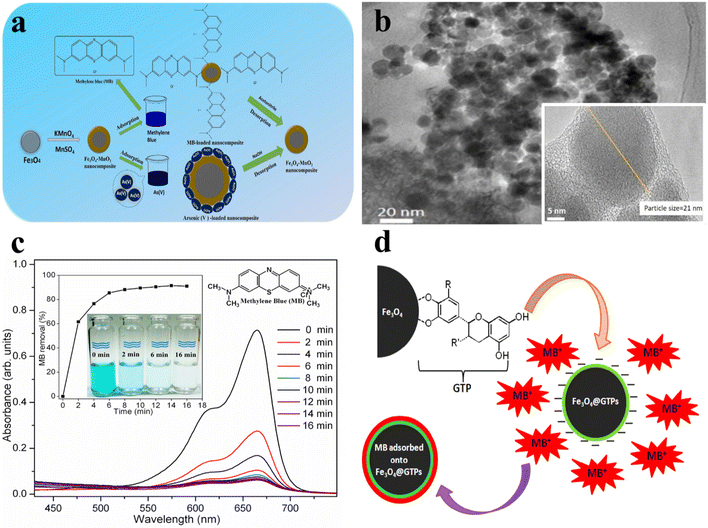 | ||
| Fig. 9 (a) Schematic of the synthesis process and organic pollution steps of Fe3O4@MnO2 MNPs, (b) TEM images of the Fe3O4–MnO2 magnetic nanocomposite,320 reproduced from ref. 320 with permission from Elsevier, copyright 2025; (c) UV-vis spectra of methylene blue adsorption in the presence of Fe3O4@GTPs MNPs at varying time intervals, and (d) schematic of the adsorption mechanism of methylene blue on the Fe3O4@GTP MNPs,327 reproduced from ref. 327 with permission from Elsevier, copyright 2025. | ||
Using a green and safe wet-chemical process, Singh et al.327 produced superparamagnetic Fe3O4 NPs coated with polyphenols from green tea (Fe3O4@GTP MNPs). Due to the hydrophilic coating of GTPs, the MNPs produced were highly stable and well dispersed in an aqueous medium. The cationic dye methylene blue could be rapidly adsorbed onto Fe3O4@GTPs due to the high concentration of negatively charged polyphenolic hydroxyls (single bond OH) on the surface of these MNPs. These groups could form an effective complex with the cationic dye methylene blue via electrostatic interactions, thereby significantly improving its adsorption. Fig. 9c depicts a schematic representation of the methylene blue adsorption process on the Fe3O4@GTP MNPs, which are ideal for this investigation due to their broad surface area and excellent dispersibility in aqueous medium. The methylene blue percentage removal rate was significantly elevated during the initial adsorption phase, followed by a considerably slower elimination rate, ultimately reaching an equilibrium state after 16 min of interaction. Up to ∼60% methylene blue was removed within the initial 2 min of contact, while over 95% removal could be achieved in the subsequent 16 min of interaction (Fig. 9d). In wastewater treatment, these MNPs demonstrated a high methylene blue dye removal capacity (7.25 mg g−1). Magnetic separation also makes it easy to remove the MNPs from the liquid, enabling multiple recycling cycles.
To enhance the removal efficiency of contaminants, particularly organic chemicals, surface functionalization of Fe3O4 NPs is crucial.329 The catalytic activity and affinity of Fe3O4 NPs can be enhanced for specific contaminants by altering their surface characteristics through the addition of specific functional groups, such as amino or carboxylic groups. For example, amino groups may promote the adsorption of organic molecules at an acidic pH, while carboxylic groups can do the opposite.330 Because of this unique attribute, Fe3O4 NPs can degrade organic molecules in solutions more efficiently, leading to better overall waste processing.331
4.3. Elimination of biological pollutants
The employment of antibacterial compounds has essential relevance in multiple applications, which extend from medical to industrial sectors, owing to the compelling need to combat bacterial infections and maintain product longevity.332 Fe3O4-based MNPs have promising biological and environmental usages due to their antibacterial and antiviral characteristics.333 Several kinds of bacteria and viruses are susceptible to MNPs’ antimicrobial activity, which may limit the proliferation of harmful microbes and decrease the transmission of illnesses caused by microbial contamination.334 Breaching microbial membranes and rendering pathogens inactive is the principal mechanism by which MNPs exhibit antibacterial and antiviral activities;335 they display this action primarily by disrupting microbial membranes. Interactions between MNPs and bacterial cell membranes may cause structural damage, thus compromising membrane integrity.336 The death of bacteria may result from this damage, leading to cell membrane leakage and the loss of intracellular contents.337 Additionally, MNPs can damage the viral membrane, thereby preventing the virus from infecting host cells and replicating, and thus preventing it from infecting other people.338In addition to their antiviral effects, MNPs can inactivate pathogens. By preventing the virus from spreading, attaching to, or penetrating the host cells, MNPs may interrupt the viral life cycle.339 Furthermore, MNPs can hinder the progression of infection by interfering with the activity of enzymes that are essential for viral replication. There are good prospects for limiting the spread of infectious diseases and preserving a clean environment by deploying MNPs for pathogen inactivation.340 Accordingly, Fe3O4-based MNPs have been deployed for antibacterial applications against S. aureus, B. subtilis, E. coli, K. pneumonia, C. albicans, and S. mutans bacteria.29,341–343
In addition to the antibacterial properties of MNPs, antifungal applications of MNPs are additional options for these NPs. Azadi et al.344 evaluated the cytotoxicity and antifungal activity of a novel nano-antifungal agent targeting drug-resistant C. species, the Fe3O4@SiO2/Schiff-base combination with Cu2+ MNPs. There is an immediate need for novel treatment approaches, as fungal infections are becoming more common and resistant to conventional antifungals. These MNPs were synthesized from Schiff-base ligands, known for their broad-spectrum antibacterial activity. C. tropicalis, C. glabrata, C. dubliniensis, C. krusei, C. parapsilosis, and Candida albicans were the six Candida species assessed by deploying the broth microdilution procedure to determine the antifungal capability. With a minimum inhibitory concentration of 8 μg mL−1 against C. parapsilosis, the findings showed robust antifungal activity in the 8–64 μg mL−1 range. C. albicans was identified as the most prevalent cause of infection, with a minimum inhibitory concentration of 32 μg mL−1. Fungal cell wall and membrane breakdown, combined with enhanced reactive oxygen species formation and cell death, is likely the antifungal mechanism (Fig. 10). These NPs show great promise for the management of serious fungal infections, as well as infections that may occur in healthcare settings. These MNPs have also been exploited in the treatment of medicinal and antibiotic wastes.345–348
 | ||
| Fig. 10 (a) Cell structure schematic of a fungal cell, (b) antifungal action of Fe3O4@SiO2/Schiff-base/Cu(II) MNPs, and plausible cell structure disruption,344 reproduced from ref. 344 with permission from Nature, copyright 2025. | ||
4.4. Recovery or elimination of microalgae
An interesting example of integrating water treatment with sustainable agriculture is the Fe3O4-based technology deployed in the coagulation process for microalgae cultivation.349 Herein, MNPs minimize algae overgrowth and enhance microalgae culture efficiency across a variety of appliances, including biofuel generation, wastewater treatment, and nutritional supplement production.350 To facilitate harvesting microalgal cells, MNPs can be added to the growth media, which causes cells to aggregate and recover, thus simplifying the harvesting process.351 By using a magnetic field to separate aggregated microalgae masses from the culture medium, these particles’ magnetic characteristics make harvesting easier and more resource- and energy-efficient.352While solving the problem of excessive algal growth in water resources, this innovative method also paves the way for the sustainable usage of microalgae in other biotech applications.353 Microalgae cultivation that incorporates MNP coagulation technology could significantly advance eco-friendly farming practices. Compared to other chemical methods, this one is safer for the environment and may lessen negative effects on the planet.354 Several obstacles could limit this method's broader adoption, including the high cost of producing MNPs and worries about the environmental implications of their manufacture.355 In addition, restoring ecological balance and improving water quality may be achieved by removing excess microalgae from water resources susceptible to algal blooms.356,357 Recently, in the application of removing these microalgae, researchers investigated MNPs, including Fe3O4, Zn-doped Fe3O4, and Fe3O4–PACI for Microcystis aeruginosa358–360 and Fe3O4–CPAM for Chlamydomonas sp.361 Research has also been conducted on their recovery, including Fe3O4–PEI for Scenedesmus dimorphus,362Chlorella vulgaris, Chlorella ellipsoidea, Microcystis aeruginosa, and Auxenochlorella protothecoides,363 and Fe3O4–SiO2 for Chlorella sorokiniana364 (Fig. 11).
5. Challenges in the application and development of Fe3O4-based MNPs
A wide range of treatment methods and procedures, comprising membrane separation, ion exchange, solvent extraction, evaporation, precipitation, and reverse osmosis, have been utilized to remove metal ions from wastewater. On the other hand, many of these techniques have limitations, namely significant operational and capital expenses, particularly in the management and removal of the metal sludge produced.365–367 Given this, efforts have been focused on developing inexpensive materials capable of eliminating pollutants from aqueous solutions. An interesting and potentially game-changing possibility is the utilization of MNPs for water purification, notably via the exploitation of coagulation processes. In light of ongoing nanotechnology development, these NPs offer a novel approach for addressing water pollution worldwide. MNPs can improve the effectiveness of coagulation processes, permitting more precise removal of contaminants and impurities from water. This is because of the unique magnetic qualities that they possess and the high specific surface areas that they encompass. This technology not only promises to enhance water quality but also has the potential to facilitate the development of more cost-effective and sustainable treatment systems. Nevertheless, to make MNPs more widely applicable, it is necessary to address their cost and sustainability. This calls for an integrated strategy that includes developing synthesis techniques that are both energy-efficient and environmentally friendly, improving manufacturing efficiency to lower prices, and promoting innovation through collaboration among governments, companies, and research institutions.94,368–373The utilization of MNPs for the adsorption of heavy metals has been the subject of a significant number of recent studies. For instance, the elimination and retrieval of hexavalent chromium (Cr6+) has been accomplished by employing maghemite NPs. The results from this experiment demonstrated that the adsorption capacity of these NPs for Cr6+ was superior to that of conventional adsorbents, such as activated carbon and clay.374,375 A further investigation conducted by the same group revealed that the adsorption of Cr6+ onto γ-Fe2O3 that had been coated with γ-FeOOH was primarily controlled by outer-sphere complexation.376 The researchers also demonstrated that surface-modified jacobsite NPs could effectively remove Cr6+ from simulated model wastewater. The adsorption process, which is then followed by magnetic separation, is what makes this technology possible for the elimination of hazardous ions in a quick, efficient, and cost-effective manner.377 In other studies, it has been demonstrated that MNPs comprising structured Fe3O4 are capable of effectively adsorbing metal ions. Within the scope of this discussion, the utilization of both biosorption and magnetic separation offers synergistic benefits comprised of operational adaptability, environmental friendliness, and cost-effectiveness.378 The removal of Cr6+ was also shown to be effective by the use of iron electrodes in electrocoagulation, as reported more recently.379 Despite these benefits, the application of Fe3O4 NPs in the purification of water is now confronted with several obstacles. The tendency of these particles to clump together is a primary concern, as it can reduce their effective surface area and adsorption capacity. In addition, iron ions may leak into the water under some circumstances, potentially generating secondary pollution. There is a possibility that the lifespan of these NPs may decrease with frequent use and recycling, which highlights the necessity of approaches that include effective regeneration or replacement. Additionally, although magnetic separation is typically effective, it may require expensive equipment and may not be effective enough for treating large quantities of water involving magnetic separation. Fig. 12 presents a brief overview of the applications, merits, and demerits of Fe3O4-based MNPs for water purification.94 In the following section, these challenges are examined in detail to assess their various dimensions, including iron ion leakage, the effects of particle agglomeration, limitations of magnetic separation, reduced lifetime due to recycling, and industrialization.
 | ||
| Fig. 12 Description of the applications, merits, and demerits of Fe3O4-based MNPs in water purification. | ||
5.1. Iron ion leaching in Fe3O4-based MNPs in water treatment
As noted, one challenge that may be encountered during Fe3O4-based MNP treatment is the release or potential leaching of Fe (Fe2+/Fe3+) ions. Some factors, such as partial dissolution of the iron oxide structure, surface phase changes, oxidation or erosion, inadequate surface coating or performance, pH conditions, heat, contact time, and corrosive environments, can affect this challenge. Fe3O4-based MNPs (composite Fe2+/Fe3+) used in water as an adsorbent, coagulant, or coagulant aid, under certain conditions, release some of the iron contained in their structure due to reaction with water/oxidant/acid/base.380 For example, in a study by Jiali Yi et al.,381 deploying a heterogeneous photo-Fenton system with Fe3O4/ZSM-5/Schwertmannite, the amount of leached Fe was reported to be between 0.18 and 0.23 mg L−1 after six cycles (30 mg of catalyst was added to 50 mL of solution, corresponding to a loading of 0.6 g L−1). Additionally, the surface of Fe3O4-based MNPs might be affected by environmental factors (highly acidic or alkaline pH, the presence of chloride ions, oxidants, and salts, and electrolyte flow), potentially leading to corrosion, oxidation, or surface dissolution, which could result in the release of iron ions.380,382 An active surface on the NPs and the transformation of iron from a solid to a soluble state are both possible outcomes in the absence of an adequate protective coating.383Consider the scenario when MNPs are not completely removed from water after the treatment process. In that case, the remaining particles can gradually undergo oxidation, surface corrosion, or partial dissolution, releasing Fe2+ and Fe3+ ions into the environment. This can not only reduce the quality of treated water, but if these ions or particles enter natural environments, they may have long-term environmental impacts, including causing changes in pH, increasing iron deposition, and having possible impacts on microorganisms and aquatic ecosystems. For this reason, it is essential to adopt a multi-level and comprehensive approach for controlling and monitoring NPs’ behavior. These solutions include designing and coating the NP surfaces to prevent oxidation and release of iron (Fe2+/Fe3+), implementing a complete magnetic recycling process to effectively remove particles from water, conducting multi-cycle durability tests to assess the stability of the coatings, maintaining magnetic properties and pollutant adsorption efficiency during repeated use, and carefully monitoring the amount of iron released during operation cycles. In addition, the determination and compliance of the permissible iron release limit should be consistent with legal requirements and local and international standards related to drinking water and wastewater discharge to ensure human and environmental health. Ultimately, the combination of these engineering and management measures, along with the safe and sustainable design of NPs, can significantly increase long-term reusability, reduce environmental risks, and improve the safety of the water treatment process with MNPs.
5.2. Stability, fate, and reusability of Fe3O4-based MNPs in water treatment
Furthermore, one of the fundamental challenges in the widespread application of MNPs is the comprehensive assessment of their chemical and physical stability after purification and the determination of their final fate across various recycling stages and natural environments. This issue not only affects the overall efficiency of the purification process but is also of vital importance from the perspective of environmental safety, long-term sustainability of the technology, and prevention of unwanted effects on ecosystems.A significant benefit of MNPs is their facile separation from aqueous media using a magnetic field. This feature enables the rapid recovery of NPs post-contaminant adsorption, allowing for reuse in successive cycles after washing or surface regeneration.384 Several studies indicate that Fe3O4 NPs with various surface coatings (including silica, polymers, and organic ligands) can be deployed for multiple cycles.325,385
In a study by Ya Pang et al.,386 PEI-coated MNPs exhibited stability in both acidic and basic solutions at various concentrations, with no notable dissolution of iron detected within the 0.2–1 M range. The NPs exhibited sufficient magnetic strength for post-treatment separation and retained their structure, unlike uncoated Fe3O4 NPs, which completely disintegrated in strong acid. The MNPs exhibited only 9% reduction in Cr6+ removal efficiency across six successive adsorption–desorption cycles, demonstrating commendable repeatability and stability of adsorption capacity over repeated applications. The recycling of NPs is economically significant, since the expense of manufacturing new NPs substantially exceeds that of cleaning and renewing used particles. The regeneration technique, particularly the use of washing solutions such as HCl or NaOH, or complexing agents such as EDTA, must be carefully selected to prevent structural deterioration or excessive oxidation of the NP surface.387
Notwithstanding the potential for recycling, research indicates that the efficacy of MNPs may progressively diminish throughout repeated cycles. This phenomenon is primarily attributed to three factors, including surface oxidation, deterioration of the surface coating, and aggregation or agglomeration.388 In the presence of dissolved oxygen or at acidic pH, the magnetite phase (Fe3O4) progressively converts into maghemite (γ-Fe2O3) and, in further phases, into hematite (α-Fe2O3). This process diminishes magnetic characteristics, complicating magnetic recycling.389 On the other hand, damage to or removal of the protective layer (such as silica or polymer) leaves the active Fe2+/Fe3+ surface vulnerable to corrosion or dissolution. Finally, repeated cycles of vdW forces and magnetic interactions among particles lead to aggregation, diminishing the active surface area and altering the particle size distribution. This process results in a decreased adsorption capacity in subsequent iterations.390
Upon collecting the MNPs, several techniques are employed to restore efficiency and facilitate reproduction, including chemical washing with acidic or alkaline solutions to eliminate adsorbed contaminants, chemical reduction using reducing agents such as sodium borohydride (NaBH4) or ascorbic acid to convert Fe3+ into Fe2+, and re-coating to reinstate the protective layer. Keerthana et al.391 demonstrated that increasing the amount of the reducing agent NaBH4 led to the synthesis of smaller, well-arranged, and more functional Fe3O4 NPs, which significantly enhanced the photocatalytic efficiency and increased pollutant removal to 97%. Whenever NPs lose their functionality due to significant oxidation, irreversible adsorption of contaminants, or the loss of magnetic properties, they are disposed of as solid waste. In this instance, stabilization and decontamination techniques, including encapsulation in cement, glass, or ceramic matrices, have been suggested to prevent the re-leakage of iron or related contaminants.392,393
5.3. Environmental behavior and lifecycle management of Fe3O4-based MNPs in water treatment
If some MNPs remain in the environment for any reason, their behavior and stability are determined by several factors. In natural aquatic environments, pH, ionic strength, natural organic matter (NOM), and oxidation–reduction potential (Eh) all play critical roles.394,395 Fe3O4 NPs oxidize and dissolve in acidic pH and in the presence of chloride or nitrate ions, but aggregate and precipitate in alkaline pH and in the presence of phosphate or silicate ions. From an environmental perspective, iron released from NPs at low concentrations is usually non-toxic. However, the presence of stable or aggregated NPs in aquatic sediments may affect microorganisms and biofilms. In reducing environments such as sludge or anaerobic soils, Fe3+ can be reduced to Fe2+, altering the chemical behavior of NPs. Fe3O4-based MNPs, owing to their large active surface area and the release of Fe2+/Fe3+ ions at low pH, can generate reactive oxygen species (ROS) and induce oxidative stress upon direct contact with aquatic cells, including algae and bacteria.396From a sustainability perspective, the recycling and reuse of MNPs, contingent on regulating iron ion release and preventing surface oxidation, constitute a significant advantage of this technology.397 The development of stable and corrosion-resistant coatings, including silicate or polymer coatings enhanced with functional ligands, can markedly prolong the lifespan of NPs. Nonetheless, life cycle assessment (LCA) indicates that improper recycling processes or NP leakage into the environment may pose potential risks, including alterations in aquatic sediment quality and bioaccumulation in organisms.
Overall, the stability of MNPs after treatment is determined by their initial particle design (structure and coating), the operating conditions of the system, and the recycling method. While Fe3O4-based MNPs are generally biocompatible, their gradual oxidation or dissolution can release iron ions into the environment. Thus, selecting the appropriate coating, monitoring surface changes during each usage cycle, and employing principled recovery and stabilization methods are critical for achieving sustainable and safe water treatment technology.
5.4. Pilot-scale and industrialization of Fe3O4-based MNPs in water treatment
A crucial bridge between laboratory-scale research and industrial implementation is the successful deployment of Fe3O4-based MNPs at the pilot scale for water purification. The practical usage of Fe3O4-based MNPs in water treatment systems relies on data from pilot-scale investigations and assessments using real wastewater matrices, which present significantly greater operational complexity than controlled experimental settings. Due to their distinctive characteristics, such as magnetic responsiveness and quick, selective separation, Fe3O4-based MNPs are an attractive choice for massive, economically viable water treatment projects. To improve their adsorption capability for pollutants, including phosphates, heavy metals, and organic compounds, these flexible NPs may be functionalized with a variety of materials like La(OH)3 and g-C3N4. Acceptable synthesis techniques, such as co-precipitation and low-temperature precipitation, enable the manufacture of NPs at the low temperatures required for pilot-scale applications. Magnetic separation devices, such as MagNERD, may be deployed in pilot-scale applications to collect these NPs after they have adsorbed pollutants from a suspension. Research in the lab has shown that Fe3O4/La(OH)3 composites are highly effective for phosphate removal, and that functionalized composites such as g-C3N4@Fe3O4 can remove microplastics and persistent organic pollutants with efficiencies over 90%. Also, heavy metals such as Pb2+ and Cd2+ may be chelated with high capacity by water-dispersible Fe3O4 NPs.398–401In an example of a real-world application, Kim et al.402 investigated the use of iron oxide NP–chitosan composites for phosphate removal from natural water collected from the Seoho stream in Suwon (Republic of Korea). Under dynamic flow conditions, the composites were able to remove phosphate in fixed-bed column experiments. An adsorption tower and dissolved air flotation/chemical reactor were built next to the Seoho stream as part of a factory-scale pilot test. The findings demonstrated a phosphorus removal rate of approximately 52.3% and a capacity of 0.059 mg phosphorus per gram of composite, which affirmed that MNPs could effectively remove phosphate in both laboratory and field settings.
Nevertheless, pilot-scale applications are accompanied by challenges, including ensuring uniformity of particle size and surface properties on a large scale, optimizing and minimizing synthesis costs, and efficient recovery of NPs after treatment. To overcome these constraints, one may use magnetic recovery devices, renewable bioreaction routes, and greener synthesis methods. Reducing NP discharge into aquatic environments by encapsulating or coating them with biocompatible polymers is another environmental factor. To advance this technology, more research ought to be conducted to determine which pollutants can be effectively removed, how to improve continuous filtration systems at the pilot scale, how to ensure compliance with environmental protocols and regulations, and how to assess the operational and long-term effectiveness of these systems in real-world water treatment applications.398,400,403–405
6. Conclusion
The increasing demand for potable water and ever-growing industrial developments have resulted in water pollution from various sources, including heavy metals, pesticides, viruses, and synthetic chemicals. As a result, organizations and industries have expressed their tremendous interest in developing and manufacturing materials for purifying water and eliminating harmful impurities. MNPs are considered one of these materials for the efficient removal of water contaminants. This work has examined a considerable proportion of newly published research on the preparation and beneficial attributes of MNPs. Special focus has been on the synthesis and mechanism of coagulation for Fe3O4 NPs, including surface charge interactions and neutralization, bridging and floc formation, external magnetic separation, and the removal of water contaminants such as heavy metals, dyes, and organic pollutants, as well as biological pollutants, and the recovery or elimination of microalgae. Water contamination and the shortage of clean water are global crises, but stakeholders may help find solutions by concentrating on the aforementioned areas. Aiming to develop cutting-edge water purification technology, Fe3O4 NPs might be a game-changer, helping to better and sustainably treat polluted water resources worldwide.Author contributions
Ali Moeini: writing – original draft, writing – review & editing, methodology, data curation, visualization, Iman Khezri: writing – original draft, data curation, visualization, Reza Ahadi Dolatsara: writing – original draft, data curation, Maryam Hajiebrahimi: writing – original draft, data curation, Hamed Beydaghi: writing – original draft, data curation, Aziz Shahraki: writing – original draft, data curation, A. Malek Khachatourian: writing – review & editing, conceptualization, methodology, supervision, M. S. Toprak: writing – review & editing, conceptualization, supervision, R. S. Varma: writing – review & editing, conceptualization, supervision.Conflicts of interest
The authors have no competing interests to declare that are relevant to the content of this article.Data availability
No primary research results, software or code have been included and no new data were generated or analyzed as part of this review.References
- X. Xu, H. Yang and C. Li, Theoretical model and actual characteristics of air pollution affecting health cost: a review, Int. J. Environ. Res. Public Health, 2022, 19, 3532 CrossRef
.
- N. Morin-Crini, E. Lichtfouse, G. Liu, V. Balaram, A. R. L. Ribeiro and Z. Lu,
et al., Worldwide cases of water pollution by emerging contaminants: a review, Environ. Chem. Lett., 2022, 20, 2311–2338 CrossRef
.
- A. H. Afzali, M. Hajiebrahimi, M. M. Hosseinieh Farahani, S. Alamdari and O. Mirzaee, Exploring the Potential of ZnS/CdS Dual Nanocomposites in Photocatalytic Degradation for Water Cleanup, Adv. Ceram. Prog., 2024, 10, 5–14 Search PubMed
.
- I. R. Abubakar, K. M. Maniruzzaman, U. L. Dano, F. S. AlShihri, M. S. AlShammari and S. M. S. Ahmed,
et al., Environmental sustainability impacts of solid waste management practices in the global South, Int. J. Environ. Res. Public Health, 2022, 19, 12717 CrossRef PubMed
.
- M. J. L. Juncal, P. Masino, E. Bertone and R. A. Stewart, Towards nutrient neutrality: A review of agricultural runoff mitigation strategies and the development of a decision-making framework, Sci. Total Environ., 2023, 874, 162408 CrossRef
.
-
S. Mishra, R. N. Bharagava, N. More, A. Yadav, S. Zainith and S. Mani, et al., Heavy metal contamination: an alarming threat to environment and human health, Environ. Biotechnol. Sustain. Futur, Springer, 2018, pp. 103–125 Search PubMed
.
- L. Lin, H. Yang and X. Xu, Effects of water pollution on human health and disease heterogeneity: a review, Front. Environ. Sci., 2022, 10, 880246 CrossRef
.
- P. Babuji, S. Thirumalaisamy, K. Duraisamy and G. Periyasamy, Human health risks due to exposure to water pollution: a review, Water, 2023, 15, 2532 CrossRef CAS
.
- A. Saravanan, P. S. Kumar, R. V. Hemavathy, S. Jeevanantham, P. Harikumar and G. Priyanka,
et al., A comprehensive review on sources, analysis and toxicity of environmental pollutants and its removal methods from water environment, Sci. Total Environ., 2022, 812, 152456 CrossRef CAS
.
- H. A. Hasan, M. H. Muhammad and N. Ismail, A review of biological drinking water treatment technologies for contaminants removal from polluted water resources, J. Water Proc. Eng., 2020, 33, 101035 CrossRef
.
- D. P. W. Sheng, M. R. Bilad and N. Shamsuddin, Assessment and optimization of coagulation process in water treatment plant: A review, ASEAN J. Sci. Eng., 2023, 3, 79–100 CrossRef
.
- J. F. J. R. Pesqueira, M. F. R. Pereira and A. M. T. Silva, Environmental impact assessment of advanced urban wastewater treatment technologies for the removal of priority substances and contaminants of emerging concern: a review, J. Cleaner Prod., 2020, 261, 121078 CrossRef CAS
.
- A. I. Stefanakis, The role of constructed wetlands as green infrastructure for sustainable urban water management, Sustainability, 2019, 11, 6981 CrossRef CAS
.
- I. Ali, New generation adsorbents for water treatment, Chem. Rev., 2012, 112, 5073–5091 CrossRef CAS
.
- S. Bolisetty, M. Peydayesh and R. Mezzenga, Sustainable technologies for water purification from heavy metals: review and analysis, Chem. Soc. Rev., 2019, 48, 463–487 RSC
.
- S. Aziz, A. R. Mazhar, A. Ubaid, S. M. H. Shah, Y. Riaz and T. Talha,
et al., A comprehensive review of membrane-based water filtration techniques, Appl. Water Sci., 2024, 14, 169 CrossRef
.
- Z. Yang, Y. Zhou, Z. Feng, X. Rui, T. Zhang and Z. Zhang, A review on reverse osmosis and nanofiltration membranes for water purification, Polymers, 2019, 11, 1252 CrossRef CAS
.
- D. Curto, V. Franzitta and A. Guercio, A review of the water desalination technologies, Appl. Sci., 2021, 11, 670 CrossRef CAS
.
- V. Gitis and N. Hankins, Water treatment chemicals: Trends and challenges, J. Water Proc. Eng., 2018, 25, 34–38 CrossRef
.
- A. A. Lanrewaju, A. M. Enitan-Folami, S. Sabiu and F. M. Swalaha, A review on disinfection methods for inactivation of waterborne viruses, Front. Microbiol., 2022, 13, 991856 CrossRef
.
- M. F. Hanafi and N. Sapawe, A review on the current techniques and technologies of organic pollutants removal from water/wastewater, Mater. Today: Proc., 2020, 31, A158–A165 CAS
.
- K. Vaaramaa and J. Lehto, Removal of metals and anions from drinking water by ion exchange, Desalination, 2003, 155, 157–170 CrossRef CAS
.
- L. Afriani and T. Trijoko, The ultraviolet light (UV) technology as a disinfection of drinking water (a literature study), Int. J. Health Educ. Soc., 2020, 3, 1–13 Search PubMed
.
- J. Ayach, W. El Malti, L. Duma, J. Lalevée, M. Al Ajami and H. Hamad,
et al., Comparing conventional and advanced approaches for heavy metal removal in wastewater treatment: an in-depth review emphasizing filter-based strategies, Polymers, 2024, 16, 1959 CrossRef CAS
.
- Y. Deng and R. Zhao, Advanced oxidation processes (AOPs) in wastewater treatment, Curr. Pollut. Rep., 2015, 1, 167–176 CrossRef CAS
.
- F. Bu, B. Gao, Q. Yue, C. Liu, W. Wang and X. Shen, The combination of coagulation and adsorption for controlling ultra-filtration membrane fouling in water treatment, Water, 2019, 11, 90 CrossRef CAS
.
- M. H. Mohamed Noor, S. Wong, N. Ngadi, I. M. Inuwa and L. A. Opotu, Assessing the effectiveness of magnetic nanoparticles coagulation/flocculation in water treatment: a systematic literature review, Int. J. Environ. Sci. Technol., 2022, 19, 6935–6956 CrossRef CAS
.
- S. Xue, Y. Xiao, G. Wang, J. Fan, K. Wan and Q. He,
et al., Adsorption of heavy metals in water by modifying Fe3O4 nanoparticles with oxidized humic acid, Colloids Surf., A, 2021, 616, 126333 CrossRef CAS
.
- B. A. Eldeeb, W. M. A. El-Raheem and S. Elbeltagi, Green synthesis of biocompatible Fe3O4 magnetic nanoparticles using Citrus Sinensis peels extract for their biological activities and magnetic-hyperthermia applications, Sci. Rep., 2023, 13, 19000 CrossRef CAS PubMed
.
-
L. K. Wang and M.-H. S. Wang, A new wave of flotation technology advancement for wastewater treatment, Environ. Flotat. Eng, 2021, pp. 143–166 Search PubMed
.
- H. Wei, B. Gao, J. Ren, A. Li and H. Yang, Coagulation/flocculation in dewatering of sludge: a review, Water Res., 2018, 143, 608–631 CrossRef CAS PubMed
.
- H. N. P. Dayarathne, M. J. Angove, R. Aryal, H. Abuel-Naga and B. Mainali, Removal of natural organic matter from source water: Review on coagulants, dual coagulation, alternative coagulants, and mechanisms, J. Water Proc. Eng., 2021, 40, 101820 CrossRef
.
- D. Yan, Z. Sun, J. Wang, L. Wang, R. Pan and Q. Wu,
et al., Enhanced coagulation with Mn(III) pre-oxidation for treatment of micro-polluted raw water, Water, 2019, 11, 2302 CrossRef CAS
.
-
R. S. Varma and B. Banerjee, Magnetic Nanocatalysis: Synthetic Applications, Walter de Gruyter GmbH & Co KG, 2022 Search PubMed
.
-
R. S. Varma and B. Banerjee, Magnetic Nanocatalysis: Industrial Applications, Walter de Gruyter GmbH & Co KG, 2022 Search PubMed
.
- R. Hudson, Y. Feng, R. S. Varma and A. Moores, Bare magnetic nanoparticles: sustainable synthesis and applications in catalytic organic transformations, Green Chem., 2014, 16, 4493–4505 RSC
.
- R. B. N. Baig and R. S. Varma, Magnetically retrievable catalysts for organic synthesis, Chem. Commun., 2013, 49, 752–770 RSC
.
- L. Lv, M. Peng, L. Wu, Y. Dong, G. You and Y. Duan,
et al., Progress in iron oxides based nanostructures for applications in energy storage, Nanoscale Res. Lett., 2021, 16, 138 CrossRef CAS
.
- Z. Abasali Karaj Abad, A. Nemati, A. Malek Khachatourian and M. Golmohammad, Synthesis and characterization of rGO/Fe2O3 nanocomposite as an efficient supercapacitor electrode material, J. Mater. Sci.: Mater. Electron., 2020, 31, 14998–15005 CrossRef CAS
.
- Z. Abasali Karaj Abad, A. Malek Khachatourian, M. Golmohammad and A. Nemati, High-Performance Hybrid Asymmetric Supercapacitors with rGO/α-Fe2O3 and rGO/TiO2 Nanocomposites, Iran. J. Mater. Sci. Eng., 2025, 22(1), 99–106 Search PubMed
.
- A. K. Gupta and M. Gupta, Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications, Biomaterials, 2005, 26, 3995–4021 CrossRef CAS
.
- A. Paryab, M. Saghatchi, B. Zarin, S. Behsam, S. Abdollahi and A. Malek Khachatourian,
et al., Magnetic particles–integrated microfluidics: from physical mechanisms to biological applications, Rev. Chem. Eng., 2024, 40, 1023–1072 CrossRef CAS
.
- B. Azadpour, N. Aharipour, A. Paryab, H. Omid, S. Abdollahi and H. M. Hosseini,
et al., Magnetically-assisted viral transduction (magnetofection) medical applications: An update, Biomater. Adv., 2023, 154, 213657 CrossRef CAS
.
- R. B. N. Baig and R. S. Varma, Organic synthesis via magnetic attraction: benign and sustainable protocols using magnetic nanoferrites, Green Chem., 2013, 15, 398–417 RSC
.
- M. Nasrollahzadeh, M. Sajjadi and R. S. Varma, Magnetically recoverable nanocatalyst based on N-heterocyclic ligands: Efficient treatment of environmental pollutants in aqueous media, Clean Technol. Environ. Policy, 2020, 22, 423–440 CrossRef CAS
.
- Y. Monga, P. Kumar, R. K. Sharma, J. Filip, R. S. Varma and R. Zbořil,
et al., Sustainable synthesis of nanoscale zerovalent iron particles for environmental remediation, ChemSusChem, 2020, 13, 3288–3305 CrossRef
.
- B. Pan, H. Qiu, B. Pan, G. Nie, L. Xiao and L. Lv,
et al., Highly efficient removal of heavy metals by polymer-supported nanosized hydrated Fe(III) oxides: behavior and XPS study, Water Res., 2010, 44, 815–824 CrossRef PubMed
.
- C. Boyer, M. R. Whittaker, V. Bulmus, J. Liu and T. P. Davis, The design and utility of polymer-stabilized iron-oxide nanoparticles for nanomedicine applications, NPG Asia Mater., 2010, 2, 23–30 CrossRef
.
- A. Dias, A. Hussain, A. S. Marcos and A. C. A. Roque, A biotechnological perspective on the application of iron oxide magnetic colloids modified with polysaccharides, Biotechnol. Adv., 2011, 29, 142–155 CrossRef PubMed
.
- M. Bystrzejewski, K. Pyrzyńska, A. Huczko and H. Lange, Carbon-encapsulated magnetic nanoparticles as separable and mobile sorbents of heavy metal ions from aqueous solutions, Carbon, 2009, 47, 1201–1204 CrossRef
.
- L. Dong, G. Chen, G. Liu, X. Huang, X. Xu and L. Li,
et al., A review on recent advances in the applications of composite Fe3O4 magnetic nanoparticles in the food industry, Crit. Rev. Food Sci. Nutr., 2024, 64, 1110–1138 CrossRef PubMed
.
- U. Jeong, X. Teng, Y. Wang, H. Yang and Y. Xia, Superparamagnetic colloids: controlled synthesis and niche applications, Adv. Mater., 2007, 19, 33–60 CrossRef
.
- L. Machala, R. Zboril and A. Gedanken, Amorphous iron(III) oxide a review, J. Phys. Chem. B, 2007, 111, 4003–4018 CrossRef PubMed
.
- G. M. Saladino, B. Hamawandi, C. Vogt, G. K. Rajarao and M. S. Toprak, Click chemical assembly and validation of bio-functionalized superparamagnetic hybrid microspheres, Appl. Nanosci., 2020, 10, 1861–1869 CrossRef
.
- N. Aharipour, A. M. Khachatourian and A. Nemati, Synthesis of Fe3O4@ m-SiO2 Nanocomposites Using Rice Husk Ash Derived SiO2, Iran. J. Mater. Sci. Eng., 2024, 21(4), 119–128 Search PubMed
.
- A. Hassanjani-Roshan, M. R. Vaezi, A. Shokuhfar and Z. Rajabali, Synthesis of iron oxide nanoparticles via sonochemical method and their characterization, Particuology, 2011, 9, 95–99 CrossRef
.
- J. Zhong and C. Cao, Nearly monodisperse hollow Fe2O3 nanoovals: Synthesis, magnetic property and applications in photocatalysis and gas sensors, Sens. Actuators, B, 2010, 145, 651–656 CrossRef
.
- H. Fan, T. Zhang, X. Xu and N. Lv, Fabrication of N-type Fe2O3 and P-type LaFeO3 nanobelts by electrospinning and determination of gas-sensing properties, Sens. Actuators, B, 2011, 153, 83–88 CrossRef
.
- M. Gotić, G. Dražić and S. Musić, Hydrothermal synthesis of α-Fe2O3 nanorings with the help of divalent metal cations, Mn2+, Cu2+, Zn2+ and Ni2+, J. Mol. Struct., 2011, 993, 167–176 CrossRef
.
- J. Vidal-Vidal, J. Rivas and M. A. López-Quintela, Synthesis of monodisperse maghemite nanoparticles by the microemulsion method, Colloids Surf., A, 2006, 288, 44–51 CrossRef
.
- G. M. Saladino, R. Kakadiya, S. R. Ansari, A. Teleki and M. S. Toprak, Magnetoresponsive fluorescent core–shell nanoclusters for biomedical applications, Nanoscale Adv., 2023, 5, 1323–1330 RSC
.
- M. D. Garrido, B. Hamawandi, J. F. Serrano-Claumarchirant, G. M. Saladino, A. B. Ergül and M. D. Marcos,
et al., A rapid synthesis of magnetic-core mesoporous silica-shell nanostructures–as potential theranostic agents–by means of microwave irradiation and the atrane method, Nanoscale, 2025, 17, 6539–6549 RSC
.
- S. R. Kanel, D. Nepal, B. Manning and H. Choi, Transport of surface-modified iron nanoparticle in porous media and application to arsenic(III) remediation, J. Nanopart. Res., 2007, 9, 725–735 CrossRef
.
- A. Tiraferri, K. L. Chen, R. Sethi and M. Elimelech, Reduced aggregation and sedimentation of zero-valent iron nanoparticles in the presence of guar gum, J. Colloid Interface Sci., 2008, 324, 71–79 CrossRef
.
- C.-L. Lin, C.-F. Lee and W.-Y. Chiu, Preparation and properties of poly (acrylic acid) oligomer stabilized superparamagnetic ferrofluid, J. Colloid Interface Sci., 2005, 291, 411–420 CrossRef
.
- K. L. Chen, S. E. Mylon and M. Elimelech, Enhanced aggregation of alginate-coated iron oxide (hematite) nanoparticles in the presence of calcium, strontium, and barium cations, Langmuir, 2007, 23, 5920–5928 CrossRef PubMed
.
- T. Hyeon, S. S. Lee, J. Park, Y. Chung and N. H. Bin, Synthesis of highly crystalline and monodisperse maghemite nanocrystallites without a size-selection process, J. Am. Chem. Soc., 2001, 123, 12798–12801 CrossRef PubMed
.
- Y. K. Sung, B. W. Ahn and T. J. Kang, Magnetic nanofibers with core (Fe3O4 nanoparticle suspension)/sheath (poly ethylene terephthalate) structure fabricated by coaxial electrospinning, J. Magn. Magn. Mater., 2012, 324, 916–922 CrossRef
.
- G. U. I. Sheng, S. Xiaodong and L. I. N. Benlan, Surface organic modification of Fe3O4 nanoparticles by silane-coupling agents, Rare Met., 2006, 25, 426–430 CrossRef
.
- L. Zhu, C. Li, J. Wang, H. Zhang, J. Zhang and Y. Shen,
et al., A simple method to synthesize modified Fe3O4 for the removal of organic pollutants on water surface, Appl. Surf. Sci., 2012, 258, 6326–6330 CrossRef
.
- A. Alaghmandfard, O. Sedighi, N. T. Rezaei, A. A. Abedini, A. M. Khachatourian and M. S. Toprak,
et al., Recent advances in the modification of carbon-based quantum dots for biomedical applications, Mater. Sci. Eng., C, 2021, 120, 111756 CrossRef
.
- A. Moeini, H. Anabestani, H. R. Madaah Hosseini and A. Malek Khachatourian, Luminescent properties of graphene quantum dots (GQDs) functionalized with LCysteine, J. Ultrafine Grained Nanostruct. Mater., 2023, 56, 121–128 Search PubMed
.
- S. Liu, B. Yu, S. Wang, Y. Shen and H. Cong, Preparation, surface functionalization and application of Fe3O4 magnetic nanoparticles, Adv. Colloid Interface Sci., 2020, 281, 102165 CrossRef
.
- F. Mohammadzadeh, M. Gharivi, M. Hekmati, F. Montazeri, A. S. Mirmohammadali and A. M. Khachatourian, Design and Characterization of Nitrogen-Doped Reduced Graphene Oxide-Enhanced Silk Fibroin/Gum Tragacanth Hydrogels for Wound Healing, Results Eng., 2025, 107813 CrossRef
.
- A. Jena, P. K. Sahoo, A. Ghosal and N. K. Sahoo, Bioinspired synthesis of iron-based nanomaterials and nanocomposite: For environmental remediation, Mater. Today: Proc., 2022, 67, 1090–1096 Search PubMed
.
- S. Malik, A. Dhasmana, S. Preetam, Y. K. Mishra, V. Chaudhary and S. P. Bera,
et al., Exploring microbial-based green nanobiotechnology for wastewater remediation: a sustainable strategy, Nanomaterials, 2022, 12, 4187 CrossRef
.
- M. Kaleem, L. A. Minhas, M. Z. Hashmi, H. M. U. Farooqi, R. Waqar and K. Kamal,
et al., Biogenic synthesis of iron oxide nanoparticles and experimental modeling studies on the removal of heavy metals from wastewater, J. Saudi Chem. Soc., 2024, 28, 101777 CrossRef
.
- A. G. Leonel, A. A. P. Mansur and H. S. Mansur, Advanced functional nanostructures based on magnetic iron oxide nanomaterials for water remediation: a review, Water Res., 2021, 190, 116693 CrossRef
.
- F. W. Lee, K. P. Wai and C. H. Koo, Enhancing contaminant removal in sewage treatment plant effluent with PES/Ag membrane, J. Appl. Membr. Sci. Technol., 2024, 28, 15–26 CrossRef
.
- G. Palani, H. Trilaksana, R. M. Sujatha, K. Kannan, S. Rajendran and K. Korniejenko,
et al., Silver nanoparticles for waste water management, Molecules, 2023, 28, 3520 CrossRef PubMed
.
- O. H. Aremu, C. O. Akintayo, E. B. Naidoo, S. M. Nelana and O. S. Ayanda, Synthesis and applications of nano-sized zinc oxide in wastewater treatment: a review, Int. J. Environ. Sci. Technol., 2021, 18, 3237–3256 CrossRef
.
- Z. Bielan, A. Kubiak, J. Karczewski, J. Ryl, K. Załęski and E. Coy,
et al., Organic pollutants photodegradation increment with use of TiO2 nanotubes decorated with transition metals after pulsed laser treatment, Mater. Sci. Semicond. Process., 2024, 177, 108378 CrossRef
.
- H. Hamrayev, S. Korpayev and K. Shameli, Advances in synthesis techniques and environmental applications of TiO2 nanoparticles for wastewater treatment: a review, J. Res. Nanosci. Nanotechnol., 2024, 12, 1–24 CrossRef
.
- B. D. Harishchandra, M. Pappuswamy, G. Shama, V. A. Arumugam, T. Periyaswamy and R. Sundaram, Copper nanoparticles: a review on synthesis, characterization and applications, Asian Pac. J. Cancer Biol., 2020, 5, 201–210 CrossRef
.
- K. Simeonidis, S. Mourdikoudis, E. Kaprara, M. Mitrakas and L. Polavarapu, Inorganic engineered nanoparticles in drinking water treatment: a critical review, Environ. Sci.: Water Res. Technol., 2016, 2, 43–70 RSC
.
-
A. Jamil, M. Anwar-ul-Haq, R. Ahmed, A. Rizwan, M. Umair and M. Zia-ur-Rehman, Potential applications of carbon nanotubes for the removal of organic and inorganic pollutants from wastewater, Water Treat. Using Eng. Carbon Nanotub, Elsevier, 2024, pp. 307–329 Search PubMed
.
- S. D. Priyadharshini, S. Manikandan, R. Kiruthiga, U. Rednam, P. S. Babu and R. Subbaiya,
et al., Graphene oxide-based nanomaterials for the treatment of pollutants in the aquatic environment: Recent trends and perspectives–A review, Environ. Pollut., 2022, 306, 119377 CrossRef PubMed
.
- P. Yadav, A. Bhaduri and A. Thakur, Manganese oxide nanoparticles: an insight into structure, synthesis and applications, ChemBioEng Rev., 2023, 10, 510–528 CrossRef
.
- B. Rajalakshmi, N. Singh, A. Madhavi, I. Khan, A. A. Hameed and S. Singh, et al., Bio-Inspired Nanomaterial's for Energy Harvesting and Storage: A Green Approach, E3S Web Conf., EDP Sciences, 2024, vol. 552, pp. 1122.
- N. Ajinkya, X. Yu, P. Kaithal, H. Luo, P. Somani and S. Ramakrishna, Magnetic iron oxide nanoparticle (IONP) synthesis to applications: present and future, Materials, 2020, 13, 4644 CrossRef PubMed
.
-
N. Tian, Y. Nie, X. Tian and Y. Wang, Current water treatment technologies: an introduction, Handb Nanomater Nanocomposites Energy Environ Appl, 2021, pp. 2033–2066 Search PubMed
.
- K. Q. Jabbar, A. A. Barzinjy and S. M. Hamad, Iron oxide nanoparticles: Preparation methods, functions, adsorption and coagulation/flocculation in wastewater treatment, Environ. Nanotechnol., Monit. Manage., 2022, 17, 100661 Search PubMed
.
- V. Kumar, E. Singh, S. Singh, A. Pandey and P. C. Bhargava, Micro-and nano-plastics (MNPs) as emerging pollutant in ground water: Environmental impact, potential risks, limitations and way forward towards sustainable management, Chem. Eng. J., 2023, 459, 141568 CrossRef
.
- D. Arrisujaya, Y. Yulizar and A. Saefumillah, Accelerated water purification with magnetite (Iron (II, III) Oxide) nanoparticles: Coagulation applications, J. Chem. Rev., 2025, 7(1), 25–62 Search PubMed
.
- S. Majidi, F. Zeinali Sehrig, S. M. Farkhani, M. Soleymani Goloujeh and A. Akbarzadeh, Current methods for synthesis of magnetic nanoparticles, Artif. Cells, Nanomed., Biotechnol., 2016, 44, 722–734 CrossRef
.
- S. Khizar, N. M. Ahmad, N. Zine, N. Jaffrezic-Renault, A. Errachid-el-salhi and A. Elaissari, Magnetic nanoparticles: From synthesis to theranostic applications, ACS Appl. Nano Mater., 2021, 4, 4284–4306 CrossRef
.
- A. Erwin, S. Salomo, P. Adhy, N. Utari, W. Ayu and Y. Wita,
et al., Magnetic iron oxide particles (Fe3O4) fabricated by ball milling for improving the environmental quality, IOP Conf. Ser.:Mater. Sci. Eng., 2020, 845, 12051 Search PubMed
.
- M. R. Akbarpour, F. Gazani, H. M. Mirabad, I. Khezri, A. Moeini and N. Sohrabi,
et al., Recent advances in processing, and mechanical, thermal and electrical properties of Cu-SiC metal matrix composites prepared by powder metallurgy, Prog. Mater. Sci., 2023, 101191 CrossRef
.
- F. S. Abdulwahid, A. J. Haider and S. Al-Musawi, Effect of laser parameter on Fe3O4 NPs formation by pulsed laser ablation in liquid, AIP Conf. Proc., 2023, 2769, 020039 CrossRef
.
- Y. A. Kotov, Electric explosion of wires as a method for preparation of nanopowders, J. Nanopart. Res., 2003, 5, 539–550 CrossRef
.
- M. S. Ansari, M. H. D. Othman, M. O. Ansari, S. Ansari and H. Abdullah, Reactively sputtered half-metallic Fe3O4 thin films at room temperature on polymethyl methacrylate: a perspective for flexible spintronics, Ceram. Int., 2020, 46, 19302–19310 CrossRef
.
- F. Meierhofer and U. Fritsching, Synthesis of metal oxide nanoparticles in flame sprays: review on process technology, modeling, and diagnostics, Energy Fuels, 2021, 35, 5495–5537 CrossRef
.
- S. Mishra and M. D. Yadav, Magnetic nanoparticles: A comprehensive review from synthesis to biomedical frontiers, Langmuir, 2024, 40, 17239–17269 CrossRef PubMed
.
- M. M. Ba-Abbad, A. Benamour, D. Ewis, A. W. Mohammad and E. Mahmoudi, Synthesis of Fe3O4 nanoparticles with different shapes through a co-precipitation method and their application, JOM, 2022, 74, 3531–3539 CrossRef
.
- G. Niraula, J. A. H. Coaquira, E. O. P. Reyes, Y. Javed, E. Reguera and J. Garcia,
et al., Hydrothermally synthesized Fe3O4 microparticles: Structural, magnetic, Mössbauer and magneto-hyperthermia properties, Results Chem., 2025, 102066 CrossRef CAS
.
- J. Wan, W. Cai, X. Meng and E. Liu, Monodisperse water-soluble magnetite nanoparticles prepared by polyol process for high-performance magnetic resonance imaging, Chem. Commun., 2007, 5004–5006 RSC
.
- O. V. Kharissova, B. I. Kharisov, C. M. Oliva González, Y. P. Méndez and I. López, Greener synthesis of chemical compounds and materials, R. Soc. Open Sci., 2019, 6, 191378 CrossRef PubMed
.
- A. Mustafa, U. Ali, A. Iqbal, M. Qayyum, I. U. Haq and F. Islam, Synthesis and Characterization of Fe3O4 Nanoparticles by Sol-Gel Method Using Water as a Solvent, Adv. Nanopart., 2025, 14, 1–11 CrossRef
.
- H. Rahbari, R. Ahadi Dolatsara, A. Moeini, S. A. Zargar, A. Shahraki and M. H. Siadati,
et al., Ambient-Air Fabrication of Triple-Cation Mixed-Halide Perovskite Cells with High-Speed Doctor-Blade Printing by PEG as Ink Additive and Triton X-100 as ETL Surface Modifier, ACS Appl. Electron. Mater., 2025, 7(3), 964–982 CrossRef CAS
.
- M. Islam, M. S. I. Sarker, M. K. R. Khan and M. N. I. Khan, Facile Sol–gel Synthesis of CoPt Nanopowder in Air Ambient: A Study for Soft Magnetic Device Applications, Bangladesh J. Phys., 2024, 31, 43–51 CrossRef
.
- G. M. Nair, T. Sajini and B. Mathew, Advanced green approaches for metal and metal oxide nanoparticles synthesis and their environmental applications, Talanta Open, 2022, 5, 100080 CrossRef
.
- S. Saif, A. Tahir and Y. Chen, Green synthesis of iron nanoparticles and their environmental applications and implications, Nanomaterials, 2016, 6, 209 CrossRef
.
- P. B. Balakrishnan, N. Silvestri, T. Fernandez–Cabada, F. Marinaro, S. Fernandes and S. Fiorito,
et al., Exploiting unique alignment of cobalt ferrite nanoparticles, mild hyperthermia, and controlled intrinsic cobalt toxicity for cancer therapy, Adv. Mater., 2020, 32, 2003712 CrossRef CAS PubMed
.
- A. Moeini, A. Ghiasi, M. D. M. Abadi, A. M. Khachatourian, H. R. M. Hosseini and M. Malek, Zn-doped superparamagnetic iron oxide nanoparticles–LCysteine functionalized@ N-doped graphene quantum dots as multifunctional contrast agents for dual-model imaging (MRI & FI), Mater. Des., 2025, 114269 CrossRef CAS
.
- S. Fayazzadeh, M. Khodaei, M. Arani, S. R. Mahdavi, T. Nizamov and A. Majouga, Magnetic properties and magnetic hyperthermia of cobalt ferrite nanoparticles synthesized by hydrothermal method, J. Supercond. Novel Magn., 2020, 33, 2227–2233 CrossRef CAS
.
- S. Gyergyek, D. Makovec, M. Jagodič, M. Drofenik, K. Schenk and O. Jordan,
et al., Hydrothermal growth of iron oxide NPs with a uniform size distribution for magnetically induced hyperthermia: Structural, colloidal and magnetic properties, J. Alloys Compd., 2017, 694, 261–271 CrossRef CAS
.
- D. Amara, I. Felner, I. Nowik and S. Margel, Synthesis and characterization of Fe and Fe3O4 nanoparticles by thermal decomposition of triiron dodecacarbonyl, Colloids Surf., A, 2009, 339, 106–110 CrossRef CAS
.
- T. K. O. Vuong, T. L. Le, D. V. Pham, H. N. Pham, T. H. Le Ngo and H. M. Do,
et al., Synthesis of high-magnetization and monodisperse Fe3O4 nanoparticles via thermal decomposition, Mater. Chem. Phys., 2015, 163, 537–544 CrossRef CAS
.
- F. Baino, E. Fiume, M. Miola, F. Leone, B. Onida and F. Laviano,
et al., Fe-doped sol-gel glasses and glass-ceramics for magnetic hyperthermia, Materials, 2018, 11, 173 CrossRef
.
-
C. J. Brinker and G. W. Scherer, Sol-gel science: the physics and chemistry of sol-gel processing, Academic press, 2013 Search PubMed
.
- T. Lu, J. Wang, J. Yin, A. Wang, X. Wang and T. Zhang, Surfactant effects on the microstructures of Fe3O4 nanoparticles synthesized by microemulsion method, Colloids Surf., A, 2013, 436, 675–683 CrossRef
.
- G. Asab, E. A. Zereffa and T. A. Seghne, Synthesis of silica–coated Fe3O4 nanoparticles by microemulsion method: characterization and evaluation of antimicrobial activity, Int. J. Biomater., 2020, 2020, 4783612 Search PubMed
.
- N. Rahimdad, A. Khalaj, G. Azarian and D. Nematollahi, Electrochemical device for the synthesis of Fe3O4 magnetic nanoparticles, J. Electrochem. Soc., 2019, 166(2), E1–E6 CrossRef
.
- T. Marín, P. Montoya, O. Arnache and J. Calderón, Influence of surface treatment on magnetic properties of Fe3O4 nanoparticles synthesized by electrochemical method, J. Phys. Chem. B, 2016, 120, 6634–6645 CrossRef
.
- A. Sathya, S. Kalyani, S. Ranoo and J. Philip, One-step microwave-assisted synthesis of water-dispersible Fe3O4 magnetic nanoclusters for hyperthermia applications, J. Magn. Magn. Mater., 2017, 439, 107–113 CrossRef
.
- Y.-J. Liang, F. Fan, M. Ma, J. Sun, J. Chen and Y. Zhang,
et al., Size-dependent electromagnetic properties and the related simulations of Fe3O4 nanoparticles made by microwave-assisted thermal decomposition, Colloids Surf., A, 2017, 530, 191–199 CrossRef
.
- R. Strobel and S. E. Pratsinis, Direct synthesis of maghemite, magnetite and wustite nanoparticles by flame spray pyrolysis, Adv. Powder Technol., 2009, 20, 190–194 CrossRef
.
- Z. Wang, G. Liu, J. Zhou, X. Zhao and J. Cai, Flame spray pyrolyzed carbon-encapsulated Au/Fe3O4 nanoaggregates enabled efficient photothermal therapy and magnetic hyperthermia of esophageal cancer cells, Front. Bioeng. Biotechnol., 2024, 12, 1400765 CrossRef
.
- C. Suryanarayana, Mechanical alloying and milling, Prog. Mater. Sci., 2001, 46, 1–184 CrossRef
.
- Z. Jalil, A. Rahwanto, M. Mustanir, H. Akhyar and E. Handoko, Magnetic behavior of natural magnetite (Fe3O4) extracted from beach sand obtained by mechanical alloying method, AIP Conf. Proc., 2017, 1862, 30023 CrossRef
.
- H. Liu, E. Y. Jiang, R. K. Zheng and H. L. Bai, Structure and magnetic properties of polycrystalline Fe3O4 films deposited by reactive sputtering at room temperature, Phys. Status Solidi, 2004, 201, 739–744 CrossRef
.
- L. Pan, G. Zhang, C. Fan, H. Qiu, P. Wu and F. Wang,
et al., Fabrication and characterization of Fe3O4 thin films deposited by reactive magnetron sputtering, Thin Solid Films, 2005, 473, 63–67 CrossRef
.
- A. Gaona-Esquivel, D. S. Hernández-Manzo, P. J. Sánchez-Trujillo, O. E. Cigarroa-Mayorga and M. Meléndez-Lira, Hyperthermia effect on phantom of breast carcinoma tissue induced by Mn3O4 nanoparticles synthesized by one-step spray pyrolysis method, Key Eng. Mater., 2020, 853, 56–60 Search PubMed
.
- A. Inukai, N. Sakamoto, H. Aono, O. Sakurai, K. Shinozaki and H. Suzuki,
et al., Synthesis and hyperthermia property of hydroxyapatite–ferrite hybrid particles by ultrasonic spray pyrolysis, J. Magn. Magn. Mater., 2011, 323, 965–969 CrossRef
.
- J. Feng, J. Mao, X. Wen and M. Tu, Ultrasonic-assisted in situ synthesis and characterization of superparamagnetic Fe3O4 nanoparticles, J. Alloys Compd., 2011, 509, 9093–9097 CrossRef
.
- Z. Bonyadi, F. S. Khatibi and F. Alipour, Ultrasonic-assisted synthesis of Fe3O4 nanoparticles-loaded sawdust carbon for malachite green removal from aquatic solutions, Appl. Water Sci., 2022, 12, 221 CrossRef
.
- M. L. Paramês, Z. Viskadourakis, M. S. Rogalski, J. Mariano, N. Popovici and J. Giapintzakis,
et al., Magnetic properties of Fe3O4 thin films grown on different substrates by laser ablation, Appl. Surf. Sci., 2007, 253, 8201–8205 CrossRef
.
- V. Subramanian, R. Baskaran and H. Krishnan, Thermal plasma synthesis of iron oxide aerosols and their characteristics, Aerosol Air Qual. Res., 2009, 9, 172–186 CrossRef
.
- S. Samal, Thermal plasma technology: The prospective future in material processing, J. Cleaner Prod., 2017, 142, 3131–3150 CrossRef
.
- I. V. Beketov, A. P. Safronov, A. I. Medvedev, J. Alonso, G. V. Kurlyandskaya and S. M. Bhagat, Iron oxide nanoparticles fabricated by electric explosion of wire: focus on magnetic nanofluids, AIP Adv., 2012, 2(2), 022154 CrossRef
.
- K. Song, C.-Y. Suh, K.-S. Ko, J.-H. Bang, W. Kim and I.-J. Shon, The effect of phases in nanoparticles produced by electrical wire explosion on Arsenic(III) removal, Mater. Trans., 2012, 53, 739–744 CrossRef
.
- A. H. Oh, H.-Y. Park, Y.-G. Jung, S.-C. Choi and G. S. An, Synthesis of Fe3O4 nanoparticles of various size via the polyol method, Ceram. Int., 2020, 46, 10723–10728 CrossRef
.
- M. Gunay, H. Kavas and A. Baykal, Simple polyol route to synthesize heptanoic acid coated magnetite (Fe3O4) nanoparticles, Mater. Res. Bull., 2013, 48, 1296–1303 CrossRef
.
- Y. P. Yew, K. Shameli, M. Miyake, N. B. B. A. Khairudin, S. E. B. Mohamad and T. Naiki,
et al., Green biosynthesis of superparamagnetic magnetite Fe3O4 nanoparticles and biomedical applications in targeted anticancer drug delivery system: A review, Arabian J. Chem., 2020, 13, 2287–2308 CrossRef
.
- H. Zhu, S. Jia, T. Wan, Y. Jia, H. Yang and J. Li,
et al., Biosynthesis of spherical Fe3O4/bacterial cellulose nanocomposites as adsorbents for heavy metal ions, Carbohydr. Polym., 2011, 86, 1558–1564 CrossRef
.
- M. Bokolia, D. Baliyan, A. Kumar, R. Das, R. Kumar and B. Singh, Biogenic synthesis of nanoparticles using microbes and plants: Mechanisms and multifaceted applications, Int. J. Environ. Anal. Chem., 2024, 1–29 Search PubMed
.
- D. Hebbalalu, J. Lalley, M. N. Nadagouda and R. S. Varma, Greener techniques for the synthesis of silver nanoparticles using plant extracts, enzymes, bacteria, biodegradable polymers, and microwaves, ACS Sustainable Chem. Eng., 2013, 1, 703–712 CrossRef
.
- J. Virkutyte and R. S. Varma, Green synthesis of metal nanoparticles: biodegradable polymers and enzymes in stabilization and surface functionalization, Chem. Sci., 2011, 2, 837–846 RSC
.
- H. Singh, M. F. Desimone, S. Pandya, S. Jasani, N. George and M. Adnan,
et al., Revisiting the green synthesis of nanoparticles: uncovering influences of plant extracts as reducing agents for enhanced synthesis efficiency and its biomedical applications, Int. J. Nanomed., 2023, 4727–4750 CrossRef
.
- R. Mohammadinejad, S. Karimi, S. Iravani and R. S. Varma, Plant-derived nanostructures: types and applications, Green Chem., 2016, 18, 20–52 RSC
.
- R. K. Das, V. L. Pachapur, L. Lonappan, M. Naghdi, R. Pulicharla and S. Maiti,
et al., Biological synthesis of metallic nanoparticles: plants, animals and microbial aspects, Nanotechnol. Environ. Eng., 2017, 2, 1–21 CrossRef CAS
.
- R. T. Kapoor, M. R. Salvadori, M. Rafatullah, M. R. Siddiqui, M. A. Khan and S. A. Alshareef, Exploration of microbial factories for synthesis of nanoparticles–a sustainable approach for bioremediation of environmental contaminants, Front. Microbiol., 2021, 12, 658294 CrossRef PubMed
.
- S. Muhammadi, M. Afzal and S. Hameed, Bacterial polyhydroxyalkanoates-eco-friendly next generation plastic: production, biocompatibility, biodegradation, physical properties and applications, Green Chem. Lett. Rev., 2015, 8, 56–77 CrossRef
.
- M. Nadeem, R. Khan, N. Shah, I. R. Bangash, B. H. Abbasi and C. Hano,
et al., A review of microbial mediated iron nanoparticles (IONPs) and its biomedical applications, Nanomaterials, 2021, 12, 130 CrossRef PubMed
.
- S. Naidu, I. K. Singh and A. Singh, Microbial synthesis of magnetic nanoparticles for plant science and agriculture, Plant Nano Biol., 2023, 4, 100036 CrossRef
.
- B. Koul, A. K. Poonia, D. Yadav and J.-O. Jin, Microbe-mediated biosynthesis of nanoparticles: Applications and future prospects, Biomolecules, 2021, 11, 886 CrossRef CAS PubMed
.
- A. Moeini, T. Hassanzadeh Chinijani, A. Malek Khachatourian, M. Vinicius Lia Fook, F. Baino and M. Montazerian, A critical review of bioactive glasses and glass–ceramics in cancer therapy, Int. J. Appl. Glass Sci., 2023, 14, 69–87 CrossRef CAS
.
-
M. M. Ramadan, H. Almoammar and K. A. Abd-Elsalam, Microbially Synthesized Biomagnetic Nanomaterials, Magn Nanostructures Environ Agric Appl, 2019, pp. 49–75 Search PubMed
.
- L. Wu, B. Yang, X. Wang, B. Wu, W. He and M. Gan,
et al., Effects of single and mixed energy sources on intracellular nanoparticles synthesized by Acidithiobacillus ferrooxidans, Minerals, 2019, 9, 163 CrossRef CAS
.
- C. Lang and D. Schüler, Biogenic nanoparticles: production, characterization, and application of bacterialmagnetosomes, J. Phys.: Condens. Matter, 2006, 18, S2815–28 CrossRef CAS
.
- T. Yoshino, T. Shimada, Y. Ito, T. Honda, Y. Maeda and T. Matsunaga,
et al., Biosynthesis of thermoresponsive magnetic nanoparticles by magnetosome display system, Bioconjugate Chem., 2018, 29, 1756–1762 CrossRef CAS PubMed
.
- T. Matsunaga and H. Takeyama, Biomagnetic nanoparticle formation and application, Supramol. Sci., 1998, 5, 391–394 CrossRef CAS
.
- D. R. Lovley, J. F. Stolz, G. L. Nord Jr and E. J. P. Phillips, Anaerobic production of magnetite by a dissimilatory iron-reducing microorganism, Nature, 1987, 330, 252–254 CrossRef CAS
.
- A. S. Athithan, J. Jeyasundari, D. Renuga and Y. B. A. Jacob, Annona muricata fruit mediated biosynthesis, physicochemical characterization of magnetite (Fe3O4) nanoparticles and assessment of its in vitro antidiabetic activity, Rasayan J. Chem., 2020, 13, 1759–1766 CrossRef
.
- N. N. Nassar, A. Hassan and P. Pereira-Almao, Metal oxide nanoparticles for asphaltene adsorption and oxidation, Energy Fuels, 2011, 25, 1017–1023 CrossRef
.
- R. P. Blakemore, K. A. Short, D. A. Bazylinski, C. Rosenblatt and R. B. Frankel, Microaerobic conditions are required for magnetite formation within Aquaspirillum magnetotacticum, Geomicrobiol. J., 1985, 4, 53–71 CrossRef
.
- C. Zhang, H. Vali, C. S. Romanek, T. J. Phelps and S. V. Liu, Formation of single-domain magnetite by a thermophilic bacterium, Am. Mineral., 1998, 83, 1409–1418 CrossRef
.
- V. V. Makarov, S. S. Makarova, A. J. Love, O. V. Sinitsyna, A. O. Dudnik and I. V. Yaminsky,
et al., Biosynthesis of stable iron oxide nanoparticles in aqueous extracts of Hordeum vulgare and Rumex acetosa plants, Langmuir, 2014, 30, 5982–5988 CrossRef
.
- S. Thakur and N. Karak, One-step approach to prepare magnetic iron oxide/reduced graphene oxide nanohybrid for efficient organic and inorganic pollutants removal, Mater. Chem. Phys., 2014, 144, 425–432, DOI:10.1016/j.matchemphys.2014.01.015
.
- H. Kabary, M. F. Eida, M. M. Attia, N. Awad and S. M. Easa, Magnetotactic characterization and environmental application P. aeruginosa kb1 isolate, Annu. Res. Rev. Biol., 2017, 20, 1–10 CrossRef
.
- P. Torabian, F. Ghandehari and M. Fatemi, Biosynthesis of iron oxide nanoparticles by cytoplasmic extracts of bacteria lactobacillus casei, Asian J. Green Chem., 2018, 2, 171–280 Search PubMed
.
- G. M. Sulaiman, A. T. Tawfeeq and A. S. Naji, Biosynthesis, characterization of magnetic iron oxide nanoparticles and evaluations of the cytotoxicity and DNA damage of human breast carcinoma cell lines, Artif. Cells, Nanomed., Biotechnol., 2018, 46, 1215–1229 CrossRef PubMed
.
- C. Prasad, S. Gangadhara and P. Venkateswarlu, Bio-inspired green synthesis of Fe3O4 magnetic nanoparticles using watermelon rinds and their catalytic activity, Appl. Nanosci., 2016, 6, 797–802 CrossRef
.
- A. P. Philipse and D. Maas, Magnetic colloids from magnetotactic bacteria: chain formation and colloidal stability, Langmuir, 2002, 18, 9977–9984 CrossRef
.
- Y. Kim and Y. Roh, Effects of microbial growth conditions on synthesis of magnetite nanoparticles using indigenous Fe(III)-reducing bacteria, Minerals, 2018, 8, 212 CrossRef
.
- A. M. Awwad and N. M. Salem, A green and facile approach for synthesis of magnetite nanoparticles, Nanosci. Nanotechnol., 2012, 2, 208–213 CrossRef
.
- S. Venkateswarlu, Y. S. Rao, T. Balaji, B. Prathima and N. V. V. Jyothi, Biogenic synthesis of Fe3O4 magnetic nanoparticles using plantain peel extract, Mater. Lett., 2013, 100, 241–244, DOI:10.1016/j.matlet.2013.03.018
.
- A. Bharde, D. Rautaray, V. Bansal, A. Ahmad, I. Sarkar and S. M. Yusuf,
et al., Extracellular biosynthesis of magnetite using fungi, Small, 2006, 2, 135–141 CrossRef PubMed
.
- H. M. Fahmy, Oxidative impact of carob leaf extract–synthesized iron oxide magnetic nanoparticles on the kidney, liver, testis, and spleen of wistar rats, Bionanoscience, 2020, 10, 54–61 CrossRef
.
- A. Azizi, Green synthesis of Fe3O4 nanoparticles and its application in preparation of Fe3O4/cellulose magnetic nanocomposite: a suitable proposal for drug delivery systems, J. Inorg. Organomet. Polym. Mater., 2020, 30, 3552–3561 CrossRef
.
- A. Bharde, A. Wani, Y. Shouche, P. A. Joy, B. L. V. Prasad and M. Sastry, Bacterial aerobic synthesis of nanocrystalline magnetite, J. Am. Chem. Soc., 2005, 127, 9326–9327 CrossRef PubMed
.
- Y. P. Yew, K. Shameli, M. Miyake, N. Kuwano, N. B. A. Khairudin and S. E. Mohamad,
et al., Green synthesis of magnetite (Fe3O4) nanoparticles using seaweed (Kappaphycus alvarezii) extract, Nanoscale Res. Lett., 2016, 11, 1–7 CrossRef PubMed
.
- S. Venkateswarlu, B. Natesh Kumar, C. H. Prasad, P. Venkateswarlu and N. V. V. Jyothi, Bio-inspired green synthesis of Fe3O4 spherical magnetic nanoparticles using Syzygium cumini seed extract, Phys. B, 2014, 449, 67–71, DOI:10.1016/j.physb.2014.04.031
.
- H. J. S. Hawezy, K. H. Sdiq, V. A. Qadr, S. S. Anwer and S. J. Salih, Biosynthesis of magnetite-nanoparticles using microalgae (Spirulina sp. and Spirogyra sp.), Plant Arch., 2020, 20, 1023–1027 Search PubMed
.
- R. Rahmani, M. Gharanfoli, M. Gholamin, M. Darroudi, J. Chamani and K. Sadri,
et al., Plant-mediated synthesis of superparamagnetic iron oxide nanoparticles (SPIONs) using aloe vera and flaxseed extracts and evaluation of their cellular toxicities, Ceram. Int., 2020, 46, 3051–3058 CrossRef
.
- A. Khataee, B. Kayan, D. Kalderis, A. Karimi, S. Akay and M. Konsolakis, Ultrasound-assisted removal of Acid Red 17 using nanosized Fe3O4-loaded coffee waste hydrochar, Ultrason. Sonochem., 2017, 35, 72–80, DOI:10.1016/j.ultsonch.2016.09.004
.
- M. Fatemi, N. Mollania, M. Momeni-Moghaddam and F. Sadeghifar, Extracellular biosynthesis of magnetic iron oxide nanoparticles by Bacillus cereus strain HMH1: Characterization and in vitro cytotoxicity analysis on MCF-7 and 3T3 cell lines, J. Biotechnol., 2018, 270, 1–11 CrossRef
.
- R. R. Koli, M. R. Phadatare, B. B. Sinha, D. M. Sakate, A. V. Ghule and G. S. Ghodake,
et al., Gram bean extract-mediated synthesis of Fe3O4 nanoparticles for tuning the magneto-structural properties that influence the hyperthermia performance, J. Taiwan Inst. Chem. Eng., 2019, 95, 357–368 CrossRef
.
- M. H. Ehrampoush, M. Miria, M. H. Salmani and A. H. Mahvi, Cadmium removal from aqueous solution by green synthesis iron oxide nanoparticles with tangerine peel extract, J. Environ. Health Sci. Eng., 2015, 13, 1–7 CrossRef
.
- J. M. Byrne, N. D. Telling, V. S. Coker, R. A. D. Pattrick, G. Van Der Laan and E. Arenholz,
et al., Control of nanoparticle size, reactivity and magnetic properties during the bioproduction ofmagnetite by Geobacter sulfurreducens, Nanotechnology, 2011, 22, 455709 CrossRef CAS
.
- H. Y. El-Kassas, M. A. Aly-Eldeen and S. M. Gharib, Green synthesis of iron oxide (Fe3O4) nanoparticles using two selected brown seaweeds: Characterization and application for lead bioremediation, Acta Oceanol. Sin., 2016, 35, 89–98 CrossRef CAS
.
- S. Bano, S. Nazir, A. Nazir, S. Munir, T. Mahmood and M. Afzal,
et al., Microwave-assisted green synthesis of superparamagnetic nanoparticles using fruit peel extracts: surface engineering, T2 relaxometry, and photodynamic treatment potential, Int. J. Nanomed., 2016, 3833–3848 CrossRef CAS
.
- B. Kumar, K. Smita, L. Cumbal, A. Debut, S. Galeas and V. H. Guerrero, Phytosynthesis and photocatalytic activity of magnetite (Fe3O4) nanoparticles using the Andean blackberry leaf, Mater. Chem. Phys., 2016, 179, 310–315 CrossRef CAS
.
- L. W. Yeary, J.-W. Moon, L. J. Love, J. R. Thompson, C. J. Rawn and T. J. Phelps, Magnetic properties of biosynthesized magnetite nanoparticles, IEEE Trans. Magn., 2005, 41, 4384–4389 CAS
.
- N. Madubuonu, S. O. Aisida, A. Ali, I. Ahmad, T. Zhao and S. Botha,
et al., Biosynthesis of iron oxide nanoparticles via a composite of Psidium guavaja-Moringa oleifera and their antibacterial and photocatalytic study, J. Photochem. Photobiol., B, 2019, 199, 111601 CrossRef CAS PubMed
.
- Y. Kim and Y. Roh, Microbial synthesis and characterization of superparamagnetic Zn-substituted magnetite nanoparticles, J. Nanosci. Nanotechnol., 2015, 15, 6129–6132 CrossRef CAS
.
- H. Jung, J.-W. Kim, H. Choi, J.-H. Lee and H.-G. Hur, Synthesis of nanosized biogenic magnetite and comparison of its catalytic activity in ozonation, Appl. Catal., B, 2008, 83, 208–213 CrossRef CAS
.
- A. Meyyappan, S. A. Banu and G. A. Kurian, One step synthesis of iron oxide nanoparticles via chemical and green route–an effective comparison, Int. J. Pharm. Pharm., 2014, 7, 70–74 Search PubMed
.
- M. Alavi, N. Karimi and T. Valadbeigi, Antibacterial, antibiofilm, antiquorum sensing, antimotility, and antioxidant activities of green fabricated Ag, Cu, TiO2, ZnO, and Fe3O4 NPs via protoparmeliopsis muralis lichen aqueous extract against multi-drug-resistant bacteria, ACS Biomater. Sci. Eng., 2019, 5, 4228–4243 CrossRef CAS PubMed
.
- B. Kumar, K. Smita, L. Cumbal and A. Debut, Biogenic synthesis of iron oxide nanoparticles for 2-arylbenzimidazole fabrication, J. Saudi Chem. Soc., 2014, 18, 364–369 CrossRef
.
- M. Khatami, H. Q. Alijani, B. Fakheri, M. M. Mobasseri, M. Heydarpour and Z. K. Farahani,
et al., Super-paramagnetic iron oxide nanoparticles (SPIONs): Greener synthesis using Stevia plant and evaluation of its antioxidant properties, J. Cleaner Prod., 2019, 208, 1171–1177 CrossRef CAS
.
- A. A. Barzinjy, D. A. Abdul, F. H. S. Hussain and S. M. Hamad, Green synthesis of the magnetite (Fe3O4) nanoparticle using Rhus coriaria extract: a reusable catalyst for efficient synthesis of some new 2-naphthol bis-Betti bases, Inorg. Nano-Met. Chem., 2020, 50, 620–629 CrossRef CAS
.
-
S. Shojaee and M.M Shahri, Green synthesis and characterization of iron oxide magnetic nanoparticles using Shanghai white tea (Camelia sinensis) aqueous extract, 2016 Search PubMed
.
- K. Parekh, J. Nair and A. Bhardwaj, Biosynthesis of magnetite nanoparticles: an eco-friendly and scalable approach, Adv. Nat. Sci.: Nanosci. Nanotechnol., 2020, 11, 35014 CAS
.
- E. C. Nnadozie and P. A. Ajibade, Green synthesis and characterization of magnetite (Fe3O4) nanoparticles using Chromolaena odorata root extract for smart nanocomposite, Mater. Lett., 2020, 263, 127145 CrossRef CAS
.
- H. Muthukumar and M. Matheswaran, Amaranthus spinosus leaf extract mediated FeO nanoparticles: physicochemical traits, photocatalytic and antioxidant activity, ACS Sustainable Chem. Eng., 2015, 3, 3149–3156 CrossRef CAS
.
- G. Jagathesan and P. Rajiv, Biosynthesis and characterization of iron oxide nanoparticles using Eichhornia crassipes leaf extract and assessing their antibacterial activity, Biocatal. Agric. Biotechnol., 2018, 13, 90–94 CrossRef
.
- Y. Cai, Y. Shen, A. Xie, S. Li and X. Wang, Green synthesis of soya bean sprouts-mediated superparamagnetic Fe3O4 nanoparticles, J. Magn. Magn. Mater., 2010, 322, 2938–2943 CrossRef CAS
.
- N. I. Taib, F. A. Latif, Z. Mohamed and N. D. S. Zambri, Green synthesis of iron oxide nanoparticles (Fe3O4-NPs) using Azadirachta indica aqueous leaf extract, Int. J. Eng. Technol., 2018, 7, 9–13 CAS
.
- M. Nurbas, H. Ghorbanpoor and H. Avci, An eco-friendly approach to synthesis and characterization of magnetite (Fe3O4) nanoparticles using Platanus orientalis L. leaf extract, Dig. J. Nanomater. Biostruct., 2017, 12(4), 993–1000 Search PubMed
.
- D. Itsaon, Green synthesis of superparamagnetic iron oxide nanoparticle from Ficus carica fruit extract, characterization studies and its application on dye degradation studies, Asian J. Pharm. Clin. Res., 2017, 10, 125–128 Search PubMed
.
- N. D. S. Zambri, N. I. Taib, F. A. Latif and Z. Mohamed, Utilization of neem leaf extract on biosynthesis of iron oxide nanoparticles, Molecules, 2019, 24, 3803 CrossRef CAS PubMed
.
- K. B. Narayanan and N. Sakthivel, Biological synthesis of metal nanoparticles by microbes, Adv. Colloid Interface Sci., 2010, 156, 1–13 CrossRef CAS PubMed
.
- N. I. Hulkoti and T. C. Taranath, Biosynthesis of nanoparticles using microbes—a review, Colloids Surf., B, 2014, 121, 474–483 CrossRef CAS
.
- W. Qin, C. Wang, Y. Ma, M. Shen, J. Li and K. Jiao,
et al., Microbe–mediated extracellular and intracellular mineralization: environmental, industrial, and biotechnological applications, Adv. Mater., 2020, 32, 1907833 CrossRef CAS PubMed
.
- A. Husen and K. S. Siddiqi, Phytosynthesis of nanoparticles: concept, controversy and application, Nanoscale Res. Lett., 2014, 9, 1–24 CrossRef CAS
.
- P. Plachtová, Z. Medrikova, R. Zboril, J. Tucek, R. S. Varma and B. Maršálek, Iron and iron oxide nanoparticles synthesized with green tea extract: differences in ecotoxicological profile and ability to degrade malachite green, ACS Sustainable Chem. Eng., 2018, 6, 8679–8687 CrossRef
.
- P. Mondal, A. Anweshan and M. K. Purkait, Green synthesis and environmental application of iron-based nanomaterials and nanocomposite: A review, Chemosphere, 2020, 259, 127509 CrossRef CAS PubMed
.
- V. Kumar, N. K. Kaushik, S. K. Tiwari, D. Singh and B. Singh, Green synthesis of iron nanoparticles: Sources and multifarious biotechnological applications, Int. J. Biol. Macromol., 2023, 253, 127017 CrossRef CAS PubMed
.
- D. D. Suppiah, N. M. Julkapli, S. Sagadevan and M. R. Johan, Eco-friendly green synthesis approach and evaluation of environmental and biological applications of Iron oxide nanoparticles, Inorg. Chem. Commun., 2023, 152, 110700 CrossRef CAS
.
- H. Cui, X. Huang, Z. Yu, P. Chen and X. Cao, Application progress of enhanced coagulation in water treatment, RSC Adv., 2020, 10, 20231–20244 RSC
.
- A. Nath, A. Mishra and P. P. Pande, A review natural polymeric coagulants in wastewater treatment, Mater. Today: Proc., 2021, 46, 6113–6117 CAS
.
-
R. G. dos Santos, Forces Acting in Colloidal Systems, Fundam. Surf. Thermodyn. Phase Behav. Its Relat. Prop, Springer, 2024, pp. 33–56 Search PubMed
.
- S. Shrestha, B. Wang and P. Dutta, Nanoparticle processing: Understanding and controlling aggregation, Adv. Colloid Interface Sci., 2020, 279, 102162 CrossRef CAS
.
-
B. Rezai and E. Allahkarami, Wastewater treatment processes—techniques, technologies, challenges faced, and alternative solutions, Soft Comput. Tech. solid waste wastewater Manag, Elsevier, 2021, pp. 35–53 Search PubMed
.
- D. Cruz, M. Pimentel, A. Russo and W. Cabral, Charge neutralization mechanism efficiency in water with high color turbidity ratio using aluminium sulfate and flocculation index, Water, 2020, 12, 572 CrossRef CAS
.
- K. Saxena, U. Brighu and A. Choudhary, Parameters affecting enhanced coagulation: a review, Environ. Technol. Rev., 2018, 7, 156–176 CrossRef
.
- R. J. C. Go, H.-L. Yang, C.-C. Kan, D. C. Ong, S. Garcia-Segura and M. D. G. de Luna, Natural organic matter removal from raw surface water: Benchmarking performance of chemical coagulants through excitation-emission fluorescence matrix spectroscopy analysis, Water, 2021, 13, 146 CrossRef CAS
.
- J. Naceradska, L. Pivokonska and M. Pivokonsky, On the importance of pH value in coagulation, J. Water Supply: Res. Technol, 2019, 68, 222–230 CrossRef
.
- A. Nowacka, M. Włodarczyk-Makuła and B. Macherzyński, Comparison of effectiveness of coagulation with aluminum sulfate and pre-hydrolyzed aluminum coagulants, Desalin. Water Treat., 2014, 52, 3843–3851 CrossRef CAS
.
- M. A. El-Nemr, N. M. Abdelmonem, I. M. A. Ismail, S. Ragab and A. El Nemr, Ozone and ammonium hydroxide modification of biochar prepared from Pisum sativum peels improves the adsorption of copper(II) from an aqueous medium, Environ. Processes, 2020, 7, 973–1007 CrossRef CAS
.
- U. Imtiyaz and M. A. Rather, Batch adsorption of Fe3+ ions from aqueous solutions using activated carbon derived from Horse Chestnut Shells found in Northwestern Himalayas: Equilibrium, kinetic and thermodynamic studies, J. Sol-Gel Sci. Technol., 2024, 112, 533–552 CrossRef CAS
.
- M. A. Inam, R. Khan, M. Akram, S. Khan and I. T. Yeom, Effect of water chemistry on antimony removal by chemical coagulation: Implications of ζ-potential and size of precipitates, Int. J. Mol. Sci., 2019, 20, 2945 CrossRef
.
- Y. Yue, G. An, L. Liu, L. Lin, R. Jiao and D. Wang, Pre-aggregation of Al13 in optimizing coagulation for removal of humic acid, Chemosphere, 2021, 277, 130268 CrossRef
.
-
B. D. Johnson, K. Kranck and D. K. Muschenheim, Physicochemical factors in particle aggregation, Biol. Part. Aquat. Syst, CRC Press, 2nd edn, 2020, pp. 75–96 Search PubMed
.
- K. Saxena and U. Brighu, Comparison of floc properties of coagulation systems: Effect of particle concentration, scale and mode of flocculation, J. Environ. Chem. Eng., 2020, 8, 104311 CrossRef
.
-
N. Tambo and K. Ogasawara, Particles and particle separation basics, IWA Publ, 2020 Search PubMed
.
- M. Khiadani, R. Kolivand, M. Ahooghalandari and M. Mohajer, Removal of turbidity from water by dissolved air flotation and conventional sedimentation systems using poly aluminum chloride as coagulant, Desalin. Water Treat., 2014, 52, 985–989 CrossRef
.
- V. R. Moreira, R. N. Guimarães, P. B. Moser, L. V. S. Santos, E. C. de Paula and Y. A. R. Lebron,
et al., Restrictions in water treatment by conventional processes (coagulation, flocculation, and sand-filtration) following scenarios of dam failure, J. Water Proc. Eng., 2023, 51, 103450 CrossRef
.
- A. Khazaie, M. Mazarji, B. Samali, D. Osborne, T. Minkina and S. Sushkova,
et al., A review on coagulation/flocculation in dewatering of coal slurry, Water, 2022, 14, 918 CrossRef
.
- M. S. S. Abujazar, S. U. Karaağaç, S. S. A. Amr, M. Y. D. Alazaiza and M. J. K. Bashir, Recent advancement in the application of hybrid coagulants in coagulation-flocculation of wastewater: A review, J. Cleaner Prod., 2022, 345, 131133 CrossRef
.
- H. Kristianto, Recent advances on magnetic natural coagulant: a mini review, Environ. Technol. Rev., 2021, 10, 255–270 CrossRef
.
- M. Soylak, O. Ozalp and F. Uzcan, Magnetic nanomaterials for the removal, separation and preconcentration of organic and inorganic pollutants at trace levels and their practical applications: A review, Trends Environ. Anal. Chem., 2021, 29, e00109 Search PubMed
.
- V. Saritha, N. Srinivas and N. V. Srikanth Vuppala, Analysis and optimization of coagulation and flocculation process, Appl. Water Sci., 2017, 7, 451–460 CrossRef
.
- A. T. Nair and M. M. Ahammed, The reuse of water treatment sludge as a coagulant for post-treatment of UASB reactor treating urban wastewater, J. Cleaner Prod., 2015, 96, 272–281 CrossRef
.
- P. Saharan, G. R. Chaudhary, S. K. Mehta and A. Umar, Removal of water contaminants by iron oxide nanomaterials, J. Nanosci. Nanotechnol., 2014, 14, 627–643 CrossRef PubMed
.
- A. Baghel, B. Singh, P. Pandey, R. K. Dhaked, A. K. Gupta and K. Ganeshan,
et al., Adsorptive removal of water poisons from contaminated water by adsorbents, J. Hazard. Mater., 2006, 137, 396–400 CrossRef PubMed
.
- A. T. Paulino, F. A. S. Minasse, M. R. Guilherme, A. V. Reis, E. C. Muniz and J. Nozaki, Novel adsorbent based on silkworm chrysalides for removal of heavy metals from wastewaters, J. Colloid Interface Sci., 2006, 301, 479–487 Search PubMed
.
- A. Chaabouni, Z. Marzougui, B. Elleuch, M. M. Eissa and A. Elaissari, Aminodextran magnetic colloidal particles for heavy metals removal, Sci. Adv. Mater., 2013, 5, 854–864 CrossRef
.
- E. Prato, F. Biandolino and C. Scardicchio, Test for acute toxicity of copper, cadmium, and mercury in five marine species, Turk. J. Zool., 2006, 30, 285–290 Search PubMed
.
- J. W. Hamilton, R. C. Kaltreider, O. V. Bajenova, M. A. Ihnat, J. McCaffrey and B. W. Turpie,
et al., Molecular basis for effects of carcinogenic heavy metals on inducible gene expression, Environ. Health Perspect., 1998, 106, 1005–1015 Search PubMed
.
- J. Briffa, E. Sinagra and R. Blundell, Heavy metal pollution in the environment and their toxicological effects on humans, Heliyon, 2020, 6, e04691 CrossRef PubMed
.
- I. Krupińska, Aluminium drinking water treatment residuals and their toxic impact on human health, Molecules, 2020, 25, 641 CrossRef PubMed
.
- H. Tang, G. Meng, J. Xiang, A. Mahmood, G. Xiang and S. Ullah,
et al., Toxic effects of antimony in plants: Reasons and remediation possibilities—A review and future prospects, Front. Plant Sci., 2022, 13, 1011945 Search PubMed
.
- C. J. Boreiko and T. G. Rossman, Antimony and its compounds: Health impacts related to pulmonary toxicity, cancer, and genotoxicity, Toxicol. Appl. Pharmacol., 2020, 403, 115156 CrossRef PubMed
.
- J. O. Fatoki and J. A. Badmus, Arsenic as an environmental and human health antagonist: A review of its toxicity and disease initiation, J. Hazard. Mater. Adv., 2022, 5, 100052 Search PubMed
.
-
G. A. Engwa, P. U. Ferdinand, F. N. Nwalo and M. N. Unachukwu, Heavy Metal Toxicity in Humans, Poisoning Mod World New Tricks an Old Dog?, 2019, vol. 77 Search PubMed
.
- H. Azar and M. F. Vajargah, Investigating the effects of accumulation of lead and cadmium metals in fish and its impact on human health, J. Aquac. Mar. Biol., 2023, 12, 209–213 CrossRef
.
- H. Hossini, B. Shafie, A. D. Niri, M. Nazari, A. J. Esfahlan and M. Ahmadpour,
et al., A comprehensive review on human health effects of chromium: insights on induced toxicity, Environ. Sci. Pollut. Res., 2022, 29, 70686–70705 CrossRef
.
- M. Iyer, U. Anand, S. Thiruvenkataswamy, H. W. S. Babu, A. Narayanasamy and V. K. Prajapati,
et al., A review of chromium (Cr) epigenetic toxicity and health hazards, Sci. Total Environ., 2023, 882, 163483 CrossRef
.
- K. K. Lawal, I. K. Ekeleme, C. M. Onuigbo, V. O. Ikpeazu and S. O. Obiekezie, A review on the public health implications of heavy metals, World J. Adv. Res. Rev., 2021, 10, 255–265 CrossRef
.
- L. Leyssens, B. Vinck, C. Van Der Straeten, F. Wuyts and L. Maes, Cobalt toxicity in humans—A review of the potential sources and systemic health effects, Toxicology, 2017, 387, 43–56 CrossRef CAS PubMed
.
- J. Y. Uriu-Adams and C. L. Keen, Copper, oxidative stress, and human health, Mol. Aspects Med., 2005, 26, 268–298 CrossRef CAS
.
- N. Karim, Copper and human health-a review, J. Bahria Univ. Med. Dent. Coll., 2018, 8, 117–122 CrossRef
.
- T. Kawabata, Iron-induced oxidative stress in human diseases, Cells, 2022, 11, 2152 Search PubMed
.
- R. Gozzelino and P. Arosio, Iron homeostasis in health and disease, Int. J. Mol. Sci., 2016, 17, 130 CrossRef PubMed
.
- D. Nayeri and S. A. Mousavi, A comprehensive review on the coagulant recovery and reuse from drinking water treatment sludge, J. Environ. Manage., 2022, 319, 115649 Search PubMed
.
- K. Jomova, S. Y. Alomar, E. Nepovimova, K. Kuca and M. Valko, Heavy metals: toxicity and human health effects, Arch. Toxicol., 2025, 99, 153–209 Search PubMed
.
- M. M. Al-Sulaiti, L. Soubra and M. A. Al-Ghouti, The causes and effects of mercury and methylmercury contamination in the marine environment: A review, Curr. Pollut. Rep., 2022, 8, 249–272 CrossRef CAS
.
- F. Li, C. Ma and P. Zhang, Mercury deposition, climate change and anthropogenic activities: A review, Front. Earth Sci., 2020, 8, 316 CrossRef
.
- W. Begum, S. Rai, S. Banerjee, S. Bhattacharjee, M. H. Mondal and A. Bhattarai,
et al., A comprehensive review on the sources, essentiality and toxicological profile of nickel, RSC Adv., 2022, 12, 9139–9153 RSC
.
- A. Mustafa, U. Zulfiqar, M. Z. Mumtaz, M. Radziemska, F. U. Haider and J. Holatko,
et al., Nickel (Ni) phytotoxicity and detoxification mechanisms: A review, Chemosphere, 2023, 328, 138574 CrossRef CAS
.
- U. Tinggi, Selenium toxicity and its adverse health effects, Rev. Food Nutr. Toxic., 2005, 4, 29–56 CAS
.
- I. Zwolak and H. Zaporowska, Selenium interactions and toxicity: a review: selenium interactions and toxicity, Cell Biol. Toxicol., 2012, 28, 31–46 CrossRef CAS
.
- L. Savignan, S. Faucher, P. Chéry and G. Lespes, Platinum group elements contamination in soils: Review of the current state, Chemosphere, 2021, 271, 129517 CrossRef CAS
.
-
F. Zereini and C. L. S. Wiseman, Platinum metals in the environment, 2015 Search PubMed
.
- M. W. Ashraf, S. I. Haider, A. R. Solangi and A. F. Memon, Toxicity of tellurium and its compounds, Phys. Sci. Rev., 2023, 8, 4375–4390 Search PubMed
.
- D. Sári, A. Ferroudj, D. Semsey, H. El-Ramady, E. C. Brevik and J. Prokisch, Tellurium and Nano-Tellurium: Medicine or Poison?, Nanomaterials, 2024, 14, 670 CrossRef
.
- F. Cima, Tin: environmental pollution and health effects, Encycl. Environ. Health, 2011, 10, 351–359 Search PubMed
.
- S. G. Schäfer and U. Femfert, Tin—a toxic heavy metal? A review of the literature, Regul. Toxicol. Pharmacol., 1984, 4, 57–69 Search PubMed
.
- D. J. White and L. S. Levy, Vanadium: environmental hazard or environmental opportunity? A perspective on some key research needs, Environ. Sci.: Processes Impacts, 2021, 23, 527–534 Search PubMed
.
- D. Rehder, Vanadium in health issues, ChemTexts, 2018, 4, 1–7 CrossRef
.
- D. Nayeri and S. A. Mousavi, A comprehensive review on the recent development of inorganic nano-adsorbents for the removal of heavy metals from water and wastewater, Environ. Dev. Sustainability, 2024, 26, 33–88 CrossRef
.
- C. T. Chasapis, P.-S. A. Ntoupa, C. A. Spiliopoulou and M. E. Stefanidou, Recent aspects of the effects of zinc on human health, Arch. Toxicol., 2020, 94, 1443–1460 CrossRef CAS
.
- C. L. Warner, W. Chouyyok, K. E. Mackie, D. Neiner, L. V. Saraf and T. C. Droubay,
et al., Manganese doping of magnetic iron oxide nanoparticles: tailoring surface reactivity for a regenerable heavy metal sorbent, Langmuir, 2012, 28, 3931–3937 CrossRef CAS
.
- S. Mahanty, S. Chatterjee, S. Ghosh, P. Tudu, T. Gaine and M. Bakshi,
et al., Synergistic approach towards the sustainable management of heavy metals in wastewater using mycosynthesized iron oxide nanoparticles: Biofabrication, adsorptive dynamics and chemometric modeling study, J. Water Proc. Eng., 2020, 37, 101426 CrossRef
.
- L. S. Ganapathe, M. A. Mohamed, R. Mohamad Yunus and D. D. Berhanuddin, Magnetite (Fe3O4) nanoparticles in biomedical application: From synthesis to surface functionalisation, Magnetochemistry, 2020, 6, 68 CrossRef CAS
.
- X. Zeng, G. Zhang, J. Zhu and Z. Wu, Adsorption of heavy metal ions in water by surface functionalized magnetic composites: a review, Environ. Sci.: Water Res. Technol., 2022, 8, 907–925 RSC
.
- F. I. El-Dib, D. E. Mohamed, O. A. A. El-Shamy and M. R. Mishrif, Study the adsorption properties of magnetite nanoparticles in the presence of different synthesized surfactants for heavy metal ions removal, Egypt J. Pet., 2020, 29, 1–7 CrossRef
.
- D. Arrisujaya, N. S. Utami, T. Mulyawati, D. Rahmalisa, S. Wati and H. Hidayat, Chromium(VI) and lead(II) adsorption using Mangifera kemanga leaves, Water Pract. Technol., 2023, 18, 2785–2796 CrossRef
.
- S. K. Gunatilake, Methods of removing heavy metals from industrial wastewater, Methods, 2015, 1, 14 Search PubMed
.
- M. Hemmati, M. Rajabi and A. Asghari, Magnetic nanoparticle based solid-phase extraction of heavy metal ions: a review on recent advances, Microchim. Acta, 2018, 185, 1–32 CrossRef CAS
.
- S. Rajput, C. U. Pittman Jr and D. Mohan, Magnetic magnetite (Fe3O4) nanoparticle synthesis and applications for lead (Pb2+) and chromium (Cr6+) removal from water, J. Colloid Interface Sci., 2016, 468, 334–346 CrossRef CAS
.
- S. L. Iconaru, R. Guégan, C. L. Popa, M. Motelica-Heino, C. S. Ciobanu and D. Predoi, Magnetite (Fe3O4) nanoparticles as adsorbents for As and Cu removal, Appl. Clay Sci., 2016, 134, 128–135 CrossRef CAS
.
- Y. Bagbi, A. Sarswat, D. Mohan, A. Pandey and P. R. Solanki, Lead and chromium adsorption from water using L-cysteine functionalized magnetite (Fe3O4) nanoparticles, Sci. Rep., 2017, 7, 7672 CrossRef
.
- S. Lunge, S. Singh and A. Sinha, Magnetic iron oxide (Fe3O4) nanoparticles from tea waste for arsenic removal, J. Magn. Magn. Mater., 2014, 356, 21–31 CrossRef CAS
.
- M. A. Serunting, R. Rusnadi, D. A. Setyorini and B. S. Ramadan, An effective cerium(III) ions removal method using sodium alginate-coated magnetite (Alg-Fe3O4) nanoparticles, J. Water Supply: Res. Technol., 2018, 67, 754–765 CrossRef
.
- L. F. O. Maia, M. S. Santos, T. G. Andrade, R. de Carvalho Hott, M. C. da Silva Faria and L. C. A. Oliveira,
et al., Removal of mercury(II) from contaminated water by gold-functionalised Fe3O4 magnetic nanoparticles, Environ. Technol., 2020, 959–970 CrossRef CAS PubMed
.
- Y. Tao, C. Zhang, T. Lü and H. Zhao, Removal of Pb(II) ions from wastewater by using polyethyleneimine-functionalized Fe3O4 magnetic nanoparticles, Appl. Sci., 2020, 10, 948 CrossRef CAS
.
- A. A. Mohammed and S. S. Isra'a, Bentonite coated with magnetite Fe3O4 nanoparticles as a novel adsorbent for copper(II) ions removal from water/wastewater, Environ. Technol. Innovation, 2018, 10, 162–174 CrossRef
.
- M. Iranmanesh and J. Hulliger, Magnetic separation: its application in mining, waste purification, medicine, biochemistry and chemistry, Chem. Soc. Rev., 2017, 46, 5925–5934 RSC
.
- L. M. Rossi, N. J. S. Costa, F. P. Silva and R. Wojcieszak, Magnetic nanomaterials in catalysis: advanced catalysts for magnetic separation and beyond, Green Chem., 2014, 16, 2906–2933 RSC
.
- M. Trobe and M. D. Burke, The molecular industrial revolution: automated synthesis of small molecules, Angew. Chem., Int. Ed., 2018, 57, 4192–4214 CrossRef CAS
.
- E. Aghaei, R. D. Alorro, A. N. Encila and K. Yoo, Magnetic adsorbents for the recovery of precious metals from leach solutions and wastewater, Metals, 2017, 7, 529 CrossRef
.
- C. F. Carolin, P. S. Kumar, A. Saravanan, G. J. Joshiba and M. Naushad, Efficient techniques for the removal of toxic heavy metals from aquatic environment: A review, J. Environ. Chem. Eng., 2017, 5, 2782–2799 CrossRef CAS
.
- A. Rane and S. J. Joshi, Biodecolorization and biodegradation of dyes: A review, Open Biotechnol. J., 2021, 15, 97–108 CrossRef
.
- M. Wainwright, Dyes in the development of drugs and pharmaceuticals, Dyes Pigm., 2008, 76, 582–589 CrossRef CAS
.
- Z. Abdi, S. A. Zargar, O. Bagheri, R. A. Dolatsara and A. M. Khachatourian, Preparation of rod-like Ni-doped ZnO anchored onto N-doped graphene nanosheets for pharmaceutical pollutant removal and evaluation of phytotoxicity, Mater. Sci. Semicond. Process., 2025, 188, 109205 CrossRef CAS
.
- S. A. Zargar, M. Gharivi, O. Bagheri, A. M. Khachatourian and A. Hashemi, Green synthesis of Z-scheme N-doped g-C3N4/Nd-doped ZnO heterostructure by pomegranate waste peel with enhanced photocatalytic performance for organic pollutants removal and antibacterial activity, FlatChem, 2024, 48, 100736 CrossRef CAS
.
- A. Mittal, J. Mittal, A. Malviya and V. K. Gupta, Adsorptive removal of hazardous anionic dye “Congo red” from wastewater using waste materials and recovery by desorption, J. Colloid Interface Sci., 2009, 340, 16–26 CrossRef PubMed
.
- M. Kasbaji, I. Ibrahim, M. Mennani, M. M. Mohamed, T. M. Salama and I. A. Moneam,
et al., Future trends in dye removal by metal oxides and their Nano/Composites: A comprehensive review, Inorg. Chem. Commun., 2023, 158, 111546 CrossRef
.
- M. Z. Esfahani, E. Soroush, S. Mohammadnejad, M. Helli, A. M. Khachatourian and M. S. Toprak,
et al., Copper oxide/graphene-based composites: Synthesis methods, appliances and recent advancements, FlatChem, 2024, 100716 CrossRef
.
- Z. Abdi, P. A. Masouleh and A. M. Khachatourian, Fabrication of NiFe2O4@ PEI and NiFe2O4@ PPy nanospheres for adsorptive and photocatalytic removal of organic dyes, Inorg. Chem. Commun., 2023, 110833 CrossRef
.
- Z. Abdi, O. Bagheri, M. Kazemzadehhojaghan and A. M. Khachatourian, In situ synthesis of polypyrrole/Nd-doped ZnO nanocomposite with enhanced visible light photocatalytic performance for the degradation of organic dyes, Mater. Chem. Phys., 2023, 296, 127248 CrossRef
.
- A. B. Mapossa, W. Mhike, J. L. Adalima and S. Tichapondwa, Removal of organic dyes from water and wastewater using magnetic ferrite-based titanium oxide and zinc oxide nanocomposites: a review, Catalysts, 2021, 11, 1543 CrossRef
.
- A. A. Wani, M. Shahadat, S. W. Ali, S. Z. Ahammad and M. K. Uddin, Recent advances and future perspectives of polymer-based magnetic nanomaterials for detection and removal of radionuclides: A review, J. Mol. Liq., 2022, 365, 119976 CrossRef
.
- S. P. Schwaminger, P. F. García, G. K. Merck, F. A. Bodensteiner, S. Heissler and S. Günther,
et al., Nature of interactions of amino acids with bare magnetite nanoparticles, J. Phys. Chem. C, 2015, 119, 23032–23041 CrossRef
.
- A. M. Vindedahl, J. H. Strehlau, W. A. Arnold and R. L. Penn, Organic matter and iron oxide nanoparticles: aggregation, interactions, and reactivity, Environ. Sci.: Nano, 2016, 3, 494–505 RSC
.
- X. Liu, J. Tian, Y. Li, N. Sun, S. Mi and Y. Xie,
et al., Enhanced dyes adsorption from wastewater via Fe3O4 nanoparticles functionalized activated carbon, J. Hazard. Mater., 2019, 373, 397–407 CrossRef
.
- K. K. Arun Viswan and D. Gangadharan, Adsorptive remediation of organic pollutant and arsenic(V) ions from water using Fe3O4-MnO2 nanocomposite, Nano-Struct. Nano-Objects, 2022, 29, 100837 CrossRef
.
- Y. Vicente-Martínez, M. Caravaca, A. Soto-Meca, M.Á Martín-Pereira and M. d. C. García-Onsurbe, Adsorption studies on magnetic nanoparticles functionalized with silver to remove nitrates from waters, Water, 2021, 13, 1757 CrossRef
.
- K. Zare, H. Sadegh, R. Shahryari-ghoshekandi, M. Asif, I. Tyagi and S. Agarwal,
et al., Equilibrium and kinetic study of ammonium ion adsorption by Fe3O4 nanoparticles from aqueous solutions, J. Mol. Liq., 2016, 213, 345–350 CrossRef
.
- A. Muhammad, A.-H. A. Shah, S. Bilal and G. Rahman, Basic Blue dye adsorption from water using Polyaniline/Magnetite (Fe3O4) composites: Kinetic and thermodynamic aspects, Materials, 2019, 12, 1764 CrossRef PubMed
.
- T. Chen, Q. Wang, J. Lyu, P. Bai and X. Guo, Boron removal and reclamation by magnetic magnetite (Fe3O4) nanoparticle: An adsorption and isotopic separation study, Sep. Purif. Technol., 2020, 231, 115930 CrossRef
.
- P. Wang, X. Wang, S. Yu, Y. Zou, J. Wang and Z. Chen,
et al., Silica coated Fe3O4 magnetic nanospheres for high removal of organic pollutants from wastewater, Chem. Eng. J., 2016, 306, 280–288 CrossRef
.
- L. Lai, Q. Xie, L. Chi, W. Gu and D. Wu, Adsorption of phosphate from water by easily separable Fe3O4@ SiO2 core/shell magnetic nanoparticles functionalized with hydrous lanthanum oxide, J. Colloid Interface Sci., 2016, 465, 76–82 CrossRef PubMed
.
- K. K. Singh, K. K. Senapati and K. C. Sarma, Synthesis of superparamagnetic Fe3O4 nanoparticles coated with green tea polyphenols and their use for removal of dye pollutant from aqueous solution, J. Environ. Chem. Eng., 2017, 5, 2214–2221 CrossRef
.
- S. R. Kanel, T. K. Das, R. S. Varma, S. Kurwadkar, S. Chakraborty and T. P. Joshi,
et al., Arsenic contamination in groundwater: Geochemical basis of treatment technologies, ACS Environ. Au, 2023, 3, 135–152 CrossRef PubMed
.
- A. Wu, X. Zhao, J. Wang, Z. Tang, T. Zhao and L. Niu,
et al., Application of solid-phase extraction based on magnetic nanoparticle adsorbents for the analysis of selected persistent organic pollutants in environmental water: A review of recent advances, Crit. Rev. Environ. Sci. Technol., 2021, 51, 44–112 CrossRef
.
- A. A. Nayl, A. I. Abd-Elhamid, A. A. Aly and S. Bräse, Recent progress in the applications of silica-based nanoparticles, RSC Adv., 2022, 12, 13706–13726 RSC
.
- S. Zahmatkesh, M. Hajiaghaei-Keshteli, A. Bokhari, S. Sundaramurthy, B. Panneerselvam and Y. Rezakhani, Wastewater treatment with nanomaterials for the future: A state-of-the-art review, Environ. Res., 2023, 216, 114652 CrossRef
.
- M. M. H. Farahani, M. Hajiebrahimi, S. Alamdari, A. Najafzadehkhoee, G. M. Khounsaraki and M. Agheb,
et al., Synthesis and antibacterial activity of silver doped zinc sulfide/chitosan bionanocomposites: A new frontier in biomedical applications, Int. J. Biol. Macromol., 2024, 280, 135934 CrossRef
.
- S. D. Anderson, V. V. Gwenin and C. D. Gwenin, Magnetic functionalized nanoparticles for biomedical, drug delivery and imaging applications, Nanoscale Res. Lett., 2019, 14, 188 CrossRef PubMed
.
- V. Novickij, R. Stanevičienė, I. Vepštaitė-Monstavičė, R. Gruškienė, T. Krivorotova and J. Sereikaitė,
et al., Overcoming antimicrobial resistance in bacteria using bioactive magnetic nanoparticles and pulsed electromagnetic fields, Front. Microbiol., 2018, 8, 2678 CrossRef
.
- E. Parra-Ortiz and M. Malmsten, Photocatalytic nanoparticles–from membrane interactions to antimicrobial and antiviral effects, Adv. Colloid Interface Sci., 2022, 299, 102526 CrossRef PubMed
.
- S. K. Modi, S. Gaur, M. Sengupta and M. S. Singh, Mechanistic insights into nanoparticle surface-bacterial membrane interactions in overcoming antibiotic resistance, Front. Microbiol., 2023, 14, 1135579 CrossRef
.
- C. Mao, Y. Xiang, X. Liu, Y. Zheng, K. W. K. Yeung and Z. Cui,
et al., Local photothermal/photodynamic synergistic therapy by disrupting bacterial membrane to accelerate reactive oxygen species permeation and protein leakage, ACS Appl. Mater. Interfaces, 2019, 11, 17902–17914 CrossRef PubMed
.
-
R. Kumar, N. Kumari and G. C. Sahoo, Iron Oxide-Based Nanoparticles in Modern Antimicrobial and Antiviral Applications, Nanoparticles Mod. Antimicrob. Antivir. Appl, Springer, 2024, pp. 289–303 Search PubMed
.
- R. G. Kerry, S. Malik, Y. T. Redda, S. Sahoo, J. K. Patra and S. Majhi, Nano-based approach to combat emerging viral (NIPAH virus) infection, Nanomedicine, 2019, 18, 196–220 CrossRef PubMed
.
- S. K. Jat, H. A. Gandhi, J. Bhattacharya and M. K. Sharma, Magnetic nanoparticles: an emerging nano-based tool to fight against viral infections, Mater. Adv., 2021, 2, 4479–4496 RSC
.
- Y. Haldorai, D. Kharismadewi, D. Tuma and J.-J. Shim, Properties of chitosan/magnetite nanoparticles composites for efficient dye adsorption and antibacterial agent, Korean J. Chem. Eng., 2015, 32, 1688–1693 CrossRef
.
- G. Darabdhara, P. K. Boruah, N. Hussain, P. Borthakur, B. Sharma and P. Sengupta,
et al., Magnetic nanoparticles towards efficient adsorption of Gram positive and Gram negative bacteria: An investigation of adsorption parameters and interaction mechanism, Colloids Surf., A, 2017, 516, 161–170 CrossRef
.
- S. Singh, K. C. Barick and D. Bahadur, Inactivation of bacterial pathogens under magnetic hyperthermia using Fe3O4−ZnO nanocomposite, Powder Technol., 2015, 269, 513–519 CrossRef
.
- S. Azadi, E. Azizipour, A. M. Amani, A. Vaez, Z. Zareshahrabadi and A. Abbaspour,
et al., Antifungal activity of Fe3O4@ SiO2/Schiff-base/Cu(II) magnetic nanoparticles against pathogenic Candida species, Sci. Rep., 2024, 14, 5855 CrossRef PubMed
.
- S. Altaf, R. Zafar, W. Q. Zaman, S. Ahmad, K. Yaqoob and A. Syed,
et al., Removal of levofloxacin from aqueous solution by green synthesized magnetite (Fe3O4) nanoparticles using Moringa olifera: Kinetics and reaction mechanism analysis, Ecotoxicol. Environ. Saf., 2021, 226, 112826 CrossRef PubMed
.
- S. Aydin, M. E. Aydin, F. Beduk and A. Ulvi, Removal of antibiotics from aqueous solution by using magnetic Fe3O4/red mud-nanoparticles, Sci. Total Environ., 2019, 670, 539–546 CrossRef PubMed
.
- A. Mohseni-Bandpi, T. J. Al-Musawi, E. Ghahramani, M. Zarrabi, S. Mohebi and S. A. Vahed, Improvement of zeolite adsorption capacity for cephalexin by coating with magnetic Fe3O4 nanoparticles, J. Mol. Liq., 2016, 218, 615–624 CrossRef
.
- W. Cai, M. Guo, X. Weng, W. Zhang and Z. Chen, Adsorption of doxorubicin hydrochloride on glutaric anhydride functionalized Fe3O4@ SiO2 magnetic nanoparticles, Mater. Sci. Eng., C, 2019, 98, 65–73 CrossRef PubMed
.
- K. F. Ngulube, A. Abdelhaleem, A. I. Osman, L. Peng and M. Nasr, Advancing sustainable water treatment strategies: harnessing magnetite-based photocatalysts and techno-economic analysis for enhanced wastewater management in the context of SDGs, Environ. Sci. Pollut. Res., 2024, 1–37 Search PubMed
.
- P. Agarwal, R. Gupta and N. Agarwal, Advances in synthesis and applications of microalgal nanoparticles for wastewater treatment, J. Nanotechnol., 2019, 2019, 7392713 Search PubMed
.
- R. Pahariya, A. Chauhan, A. Ranjan, R. K. Basniwal, S. Upadhyay and S. K. Thakur,
et al., A critical review on the efficacy and mechanism of nanoparticle-based flocculants for biodiesel feedstock production from microalgae, BioEnergy Res., 2024, 17, 1065–1079 CrossRef
.
- A. Kucmanová and K. Gerulová, Microalgae harvesting: a review, Ved. Pr. Mater. Fak. Slov. Tech. Univ. Bratislave Trnave, 2019, 27, 129–143 Search PubMed
.
- A. Ahmad, F. Banat, H. Alsafar and S. W. Hasan, Algae biotechnology for industrial wastewater treatment, bioenergy production, and high-value bioproducts, Sci. Total Environ., 2022, 806, 150585 CrossRef PubMed
.
- Y. Yang, X. Fan, J. Zhang, S. Qiao, X. Wang and X. Zhang,
et al., A critical review on the interaction of iron-based nanoparticles with blue-green algae and their metabolites: from mechanisms to applications, Algal Res., 2022, 64, 102670 CrossRef
.
- O. P. Fuentes, D. M. Trujillo, M. E. Sánchez, A. L. Abrego-Perez, J. F. Osma and J. C. Cruz, Embracing sustainability in the industry: a study of environmental, economic, and exergetic performances in large-scale production of magnetite nanoparticles, ACS Sustainable Chem. Eng., 2024, 12, 760–772 CrossRef
.
- A. S. Nwankwegu, Y. Li, Y. Huang, J. Wei, E. Norgbey and L. Sarpong,
et al., Harmful algal blooms under changing climate and constantly increasing anthropogenic actions: the review of management implications, 3 Biotech, 2019, 9, 1–19 CrossRef PubMed
.
- M. Pelaez, B. Baruwati, R. S. Varma, R. Luque and D. D. Dionysiou, Microcystin-LR removal from aqueous solutions using a magnetically separable N-doped TiO2 nanocomposite under visible light irradiation, Chem. Commun., 2013, 49, 10118–10120 RSC
.
- L. Chen, X. Wu, C. Liu, Y. Zhao and Q. Huang, Study on removal of Microcystis aeruginosa and Cr(VI) using attapulgite-Fe3O4 magnetic composite material (MCM), Algal Res., 2021, 60, 102501 CrossRef
.
- J. Qi, H. Lan, R. Liu, H. Liu and J. Qu, Efficient Microcystis aeruginosa removal by moderate photocatalysis-enhanced coagulation with magnetic Zn-doped Fe3O4 particles, Water Res., 2020, 171, 115448 CrossRef PubMed
.
- B. Zhang, D. Jiang, X. Guo, Y. He, C. N. Ong and Y. Xu,
et al., Removal of Microcystis aeruginosa using nano-Fe3O4 particles as a coagulant aid, Environ. Sci. Pollut. Res., 2015, 22, 18731–18740 CrossRef
.
- J. Ma, W. Xia, X. Fu, L. Ding, Y. Kong and H. Zhang,
et al., Magnetic flocculation of algae-laden raw water and removal of extracellular organic matter by using composite flocculant of Fe3O4/cationic polyacrylamide, J. Cleaner Prod., 2020, 248, 119276 CrossRef
.
- S. Ge, M. Agbakpe, Z. Wu, L. Kuang, W. Zhang and X. Wang, Influences of surface coating, UV irradiation and magnetic field on the algae removal using magnetite nanoparticles, Environ. Sci. Technol., 2015, 49, 1190–1196 CrossRef
.
- K. Gerulová, A. Kucmanová, Z. Sanny, Z. Garaiová, E. Seiler and M. Čaplovičová,
et al., Fe3O4-PEI nanocomposites for magnetic harvesting of chlorella vulgaris, chlorella ellipsoidea, microcystis aeruginosa, and auxenochlorella protothecoides, Nanomaterials, 2022, 12, 1786 CrossRef PubMed
.
- D. T. C. Van, L. Van Toan, N. T. P. Dung, T. D. Thuan, D. T. Cuc and D. T. Mai,
et al., Recovery of Chlorella biomass using Fe3O4 and Fe3O4@ SiO2 magnetic nanoparticles: isotherm and thermodynamic characteristics of adsorption, Vietnam J. Chem., 2023, 61, 116–125 CrossRef
.
- M. K. Jha, V. Kumar, L. Maharaj and R. J. Singh, Studies on leaching and recycling of zinc from rayon waste sludge, Ind. Eng. Chem. Res., 2004, 43, 1284–1295 CrossRef
.
- S. E. Kentish and G. W. Stevens, Innovations in separations technology for the recycling and re-use of liquid waste streams, Chem. Eng. J., 2001, 84, 149–159 CrossRef CAS
.
- M. K. Jha, R. R. Upadhyay, J. Lee and V. Kumar, Treatment of rayon waste effluent for the removal of Zn and Ca using Indion BSR resin, Desalination, 2008, 228, 97–107 CrossRef
.
- S. Sun, C. B. Murray, D. Weller, L. Folks and A. Moser, Monodisperse FePt nanoparticles and ferromagnetic FePt nanocrystal superlattices, Science, 2000, 287, 1989–1992 CrossRef
.
- J. Xie, C. Xu, N. Kohler, Y. Hou and S. Sun, Controlled PEGylation of monodisperse Fe3O4 nanoparticles for reduced non–specific uptake by macrophage cells, Adv. Mater., 2007, 19, 3163–3166 CrossRef
.
- Q. A. Pankhurst, J. Connolly, S. K. Jones and J. J. J. Dobson, Applications of magnetic nanoparticles in biomedicine, J. Phys. D: Appl. Phys., 2003, 36, R167–181 CrossRef
.
- S. Sun and H. Zeng, Size-controlled synthesis of magnetite nanoparticles, J. Am. Chem. Soc., 2002, 124, 8204–8205 CrossRef
.
- S. Si, A. Kotal, T. K. Mandal, S. Giri, H. Nakamura and T. Kohara, Size-controlled synthesis of magnetite nanoparticles in the presence of polyelectrolytes, Chem. Mater., 2004, 16, 3489–3496 CrossRef
.
- S. Wan, J. Huang, H. Yan and K. Liu, Size-controlled preparation of magnetite nanoparticles in the presence of graft copolymers, J. Mater. Chem., 2006, 16, 298–303 RSC
.
- J. Hu, G. Chen and I. M. C. Lo, Removal and recovery of Cr(VI) from wastewater by maghemite nanoparticles, Water Res., 2005, 39, 4528–4536 CrossRef PubMed
.
- J. Hu, I. M. C. Lo and G. Chen, Comparative study of various magnetic nanoparticles for Cr(VI) removal, Sep. Purif. Technol., 2007, 56, 249–256 CrossRef
.
- J. Hu, I. M. C. Lo and G. Chen, Performance and mechanism of chromate (VI) adsorption by δ-FeOOH-coated maghemite (γ-Fe2O3) nanoparticles, Sep. Purif. Technol., 2007, 58, 76–82 CrossRef
.
- J. Hu, I. M. C. Lo and G. Chen, Fast removal and recovery of Cr(VI) using surface-modified jacobsite (MnFe2O4) nanoparticles, Langmuir, 2005, 21, 11173–11179 CrossRef PubMed
.
- H. Li, Z. Li, T. Liu, X. Xiao, Z. Peng and L. Deng, A novel technology for biosorption and recovery hexavalent chromium in wastewater by bio-functional magnetic beads, Bioresour. Technol., 2008, 99, 6271–6279 CrossRef PubMed
.
- I. Heidmann and W. Calmano, Removal of Cr(VI) from model wastewaters by electrocoagulation with Fe electrodes, Sep. Purif. Technol., 2008, 61, 15–21 CrossRef
.
- H. Guo and A. S. Barnard, Naturally occurring iron oxide nanoparticles: morphology, surface chemistry and environmental stability, J. Mater. Chem. A, 2013, 1, 27–42 RSC
.
- J. Yi, J. Xu, J. Liu, Y. Zheng and Q. Wang, High-Efficient Elimination of Spiramycin by Fe3O4/ZSM-5/Sch via Heterogeneous Photo-Fenton Oxidation at Neutral pH, Sustainability, 2023, 15, 12343 CrossRef
.
- A. E. Chastukhin, A. D. Izotov, I. G. Gorichev and A. M. Kutepov, Analysis of Fe2O3 and Fe3O4 dissolution kinetics in terms of the chain mechanism model, Theor. Found. Chem. Eng., 2003, 37, 398–406 CrossRef
.
- S. Guo, C. D. Powell, D. Villagrán and M. S. Wong, Magnetic In–Pd catalysts for nitrate degradation, Environ. Sci.: Nano, 2020, 7, 2681–2690 RSC
.
- M. M. Falinski, R. S. Turley, J. Kidd, A. W. Lounsbury, M. Lanzarini-Lopes and A. Backhaus,
et al., Doing nano-enabled water treatment right: sustainability considerations from design and research through development and implementation, Environ. Sci.: Nano, 2020, 7, 3255–3278 RSC
.
- M. B. Gawande, P. S. Branco and R. S. Varma, Nano-magnetite (Fe3O4) as a support for recyclable catalysts in the development of sustainable methodologies, Chem. Soc. Rev., 2013, 42, 3371–3393 RSC
.
- Y. Pang, G. Zeng, L. Tang, Y. Zhang, Y. Liu and X. Lei,
et al., Preparation and application of stability enhanced magnetic nanoparticles for rapid removal of Cr(VI), Chem. Eng. J., 2011, 175, 222–227 CrossRef
.
- T. Sithole, A review on regeneration of adsorbent and recovery of metals: adsorbent disposal and regeneration mechanism, S. Afr. J. Chem. Eng., 2024, 50, 39–50 Search PubMed
.
- D. Xiao, T. Lu, R. Zeng and Y. Bi, Preparation and highlighted
applications of magnetic microparticles and nanoparticles: a review on recent advances, Microchim. Acta, 2016, 183, 2655–2675 CrossRef
.
- X. Zhang, Y. Niu, X. Meng, Y. Li and J. Zhao, Structural evolution and characteristics of the phase transformations between α-Fe2O3, Fe3O4 and γ-Fe2O3 nanoparticles under reducing and oxidizing atmospheres, CrystEngComm, 2013, 15, 8166–8172 RSC
.
- S. Moosavi, C. W. Lai, S. Gan, G. Zamiri, O. Akbarzadeh Pivehzhani and M. R. Johan, Application of efficient magnetic particles and activated carbon for dye removal from wastewater, ACS Omega, 2020, 5, 20684–20697 CrossRef
.
- S. P. Keerthana, S. Gayathri, R. Yuvakkumar, L. Kungumadevi, G. Ravi and A. G. Al-Sehemi,
et al., Conversion and reducing agent effect on zero valent iron into Fe3O4 for photocatalytic degradation under UV light irradiation, Environ. Res., 2022, 214, 113959 CrossRef PubMed
.
- N. Sarfraz, M. Ashraf, S. Ali and I. Khan, Magnetism-driven iron oxide nanocomposites for energy and environmental solutions: harnessing magnetism, Mater. Today Sustainability, 2023, 24, 100589 CrossRef
.
- Y. Iqbal, W. Ma, X. Li, S. Fu, A. Shahzad and I. Ahmad,
et al., A brief outlook on the preparation and magnetic properties of iron oxide nanoparticles for water remediation, Water, Air, Soil Pollut., 2025, 236, 687 CrossRef
.
- L. Chekli, S. Phuntsho, L. D. Tijing, J. L. Zhou, J.-H. Kim and H. K. Shon, Stability of Fe-oxide nanoparticles coated with natural organic matter under relevant environmental conditions, Water Sci. Technol., 2014, 70, 2040–2046 CrossRef PubMed
.
- R. Zhang, X. Zheng, W. Fan, X. Wang, T. Zhao and X. Zhao,
et al., Fate models of nanoparticles in the environment: a critical review and prospects, Environ. Sci.: Nano, 2025, 12, 3394–3412 RSC
.
- A. G. Kicheeva, E. S. Sushko, L. S. Bondarenko, K. A. Kydralieva, D. A. Pankratov and N. S. Tropskaya,
et al., Functionalized magnetite nanoparticles: characterization, bioeffects, and role of reactive oxygen species in unicellular and enzymatic systems, Int. J. Mol. Sci., 2023, 24, 1133 CrossRef PubMed
.
-
M. Singh, S. Dhiman, N. Debnath and S. Das, Magnetic nanoparticles and their application in sustainable environment, Sustain. Nanotechnol. Environ. Remediat, Elsevier, 2022, pp. 457–483 Search PubMed
.
- S. Ahmed, Y. Zhang, B. Wu, Z. Zheng, C.-F. Leung and T. Choy,
et al., Scaled-up development of magnetically recyclable Fe3O4/La(OH)3 composite for river water phosphate removal: From bench-scale to pilot-scale study, Sci. Total Environ., 2021, 791, 148281 CrossRef PubMed
.
- R. Indhur, A. Kumar, F. Bux and S. Kumari, Efficient Microplastic Removal from Wastewater using Fe3O4 Functionalized g-C3N4 and BNNS: A Comprehensive Study, J. Environ. Chem. Eng., 2025, 117145 CrossRef
.
- C. D. Powell, A. J. Atkinson, Y. Ma, M. Marcos-Hernandez, D. Villagran and P. Westerhoff,
et al., Magnetic nanoparticle recovery device (MagNERD) enables application of iron oxide nanoparticles for water treatment, J. Nanopart. Res., 2020, 22, 48 CrossRef
.
- L. Wang, J. Li, Q. Jiang and L. Zhao, Water-soluble Fe3O4 nanoparticles with high solubility for removal of heavy-metal ions from waste water, Dalton Trans., 2012, 41, 4544–4551 RSC
.
- J.-H. Kim, S.-B. Kim, S.-H. Lee and J.-W. Choi, Laboratory and pilot-scale field experiments for application of iron oxide nanoparticle-loaded chitosan composites to phosphate removal from natural water, Environ. Technol., 2018, 39, 770–779 CrossRef CAS
.
- P. Beigi and M. Babamoradi, Preparation of Fe3O4@ pectin nanocomposite hydrogel with high heating efficiency for hyperthermia applications, Phys. B, 2023, 670, 415360 CrossRef CAS
.
- S. Hsieh, B. Y. Huang, S. L. Hsieh, C. C. Wu, C. H. Wu and P. Y. Lin,
et al., Green fabrication of agar-conjugatedFe3O4 magnetic nanoparticles, Nanotechnology, 2010, 21, 445601 CrossRef CAS
.
- C. Verma, D. K. Verma, E. Berdimurodov, I. Barsoum, A. Alfantazi and C. M. Hussain, Green magnetic nanoparticles: a comprehensive review of recent progress in biomedical and environmental applications, J. Mater. Sci., 2024, 59, 325–358 CrossRef CAS
.
| This journal is © The Royal Society of Chemistry 2026 |