Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Hacking 3D printers as laboratory robots
Sander
Baas
a,
Nessa
Carson
 b and
Vittorio
Saggiomo
b and
Vittorio
Saggiomo
 *a
*a
aDepartment of BioNanoTechnology, Wageningen University and Research, The Netherlands. E-mail: Vittorio.saggiomo@wur.nl
bData Science & Modelling, Pharmaceutical Sciences, R&D, AstraZeneca, Macclesfield SK10 2NA, UK
First published on 26th March 2026
Abstract
The emergence of affordable and reliable 3D printers has enabled laboratories to optimize setups, print custom parts, accelerate research, and rapidly prototype. A new movement has emerged in the past decade, where 3D printers are repurposed as laboratory-specific robots. There are three distinct approaches in the 3D-printer-as-lab-robot approach: modifying the extruder for non-standard material printing, replacing the extruder with a third-party implement, such as a pipette, microscope, or slide holder, or deconstructing the printer completely and using it as a cheap and widely available parts kit for lab-built robots such as syringe pumps. New developments in printer hardware and software control, which enable the use of printers as laboratory robots, are also discussed.
Introduction
Although simple instrument automation, such as the use of autosampler devices, has become commonplace, the state of the art is continually pushing new boundaries in hardware and workflows, in turn pushing greater demand for laboratory robotics.1 In fields including chemistry, biology, and materials science, automation has seen a significant uprise in recent years,2–6 enabling scientists not only to perform human-centric work faster, but also to expand workflows and eliminate roadblocks, moving slow steps and reducing pain points during research and development.7–13Challenges involving the cost and ease of use of automation, which both vary from accessible to highly specialist, are driving some researchers to build their own robotic hardware rather than use commercial kits. 3D printers serve as a common basis for these homemade robots, due to the accessibility of their hardware and software, a strong ‘hacking’ (Open Hardware) community where designs and modifications can be shared as open-source, and often a low price point.14 Costs for consumer 3D printers have fallen in recent years due to increased market demand, the advent of entry-level fused deposition modeling printers (FDM, also known as fused filament fabrication, FFF), and platforms such as RepRap, a project to develop “self-replicating” 3D printers that can print many of their own plastic components.15–19
3D printers themselves have a variety of lab-based applications, from making vessel holders and equipment parts to bioprinting and printing solid dispersion drug formulations.14,20,21 There even exists a free, open library of 3D printable models specifically curated for scientific use by the NIH.22 However, modified 3D printer-based robots are now enabling a wide variety of additional scientific applications, including more specialized bioprinting, electrospinning, syringe pumps, battery applications, and microscopy.
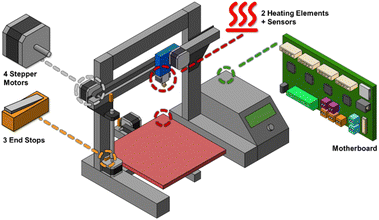
A 3D printer is itself a 3-axis robot. They typically involve a printhead motor that can be manipulated within a Cartesian XYZ space to deposit material in 3D space, thereby manufacturing items such as machine parts, consumer products, and decorations. The motorized XYZ axes are the most used parts for transforming a 3D printer into a functionally distinct 3-axis robot. FDM is now the most common printing technology, where a thermoplastic filament is heated and deposited in space, at which point it cools to form a part of the structure of the item, which can further be built on by depositing more of the thermoplastic filament. FDM printers are now fairly commonplace in laboratories and universities, in makerspaces for public use, in homes as consumer devices, and now as outsourced services.23,24 In addition to the four motors, FDM printers usually feature two heating units, one for the printhead and one for the printing bed. Each of the two heating units consists of a heating resistor and a thermistor for monitoring the temperature. The printhead can easily reach temperatures of up to 250 °C, while the bed typically reaches around 100 °C. Additionally, the motherboard monitors three end-stop switches to determine the printhead's home position. These switches and motherboard inputs can also be used for modifications.
Designs for 3D printers can be created using a variety of free or commercial software, including computer-aided design (CAD) programs, as well as ChemDraw for molecular structures.25 However, the instructions to execute these designs on the hardware are written with machine-specific instructions, which typically use the G-code programming language (standing for “geometric code”). G-code is a universal CNC (Computer Numerical Control) programming language that allows for the relative and absolute positioning and movement of the printhead. This is used in conjunction with M-code (“machine code”) for non-geometrical functions, such as turning the heater on and off and setting fan speeds.
Modifying 3D printers for laboratory applications can be accomplished in various ways, as described herein (Fig. 1 and Table 1). Part 1 describes robots in which the fourth motor, usually used as the filament feeder, is used for an alternative function (Fig. 2). In Part 2, we explore robots where the entire printhead has been exchanged for a different tool (Fig. 3). In Part 3, we discuss using other parts of the printer for laboratory purposes, and lastly, the software used for controlling these robots (Fig. 4).
| Setup name | Printer | Purpose | Device type/modification | Year | References |
|---|---|---|---|---|---|
| 3D-printer based slot-die coater | Felix 2.0 | Solar cell fabrication | Syringe extruder modification | 2014 | 26 |
| Open-source 3D bioprinter | Felix 3.0 | Bioprinting and tissue engineering | Syringe extruder modification | 2016 | 27 |
| Ultra-low-cost 3D bioprinter | Anet A8 | Bioprinting | Syringe extruder modification | 2016 | 28 |
| Liquid-phase 3D bioprinter | Ultimaker original | Printing gelatin alginate hydrogels | Syringe extruder modification | 2019 | 29 |
| NOSE | Prusa i3 | Open-source bioprinting platform | Syringe extruder modification | 2019 | 30 |
| FRESH | MakerBot replicator | Complex suspended hydrogel printing | Syringe extrusion & scaffold removal | 2015 | 31 |
| FRESH 2.0 | Printrbot simple metal, flashforge creator pro, makerbot replicator 2× | Collagen-based heart component production | Syringe extrusion & scaffold removal | 2019 | 32 |
| Replistruder | — | Universal syringe extruder | Syringe extruder head | 2015, 2022 | 33 and 35 |
| Custom ultral-low-cost 3D bioprinter | Anet A8 | Bio-ink deposition for 3D cell culture | Syringe extruder modification | 2020 | 36 |
| Enderstruder | Creality Ender-3 V2 | Syringe extruder | Syringe extruder modification | 2024 | 38 |
| 3D food printer with syringe pump | Rigid3D Zero2 | Syringe extruder food printer | Syringe extruder modification | 2023 | 40 |
| Dual simultaneous reagent dispenser | Hellbot magna 1 | Lateral flow assay production | Syringe extruder & 3-way valve combination | 2024 | 41 |
| NovaSpider | Not specified | 3D electrospinning | Syringe-fed electrospinning nozzle and electrode addition | 2021 | 43 |
| Hybrid bioprinter & electrospinner | Creality Ender-3 | Vascular tissue engineering | Syringe extruder, electrospinning head, & needle collector | 2021 | 44 |
| Electrospun polycaprolactone substrate fabricator | Not specified | Colorimetric assay substrate production | Syringe-fed needle tip extruder & silicon ground plate | 2024 | 45 |
| Xspin | Not specified | Dual fiber material electrospinning | Mixing 3D electrospinning setup | 2024 | 46 |
| MEWron | VORON 0.1 | Open-source melt electrowriting (MEW) | Filament & syringe MEW printheads, MEW collector | 2023 | 48 |
| Benchtop magnetic field mapper | Velleman K8200 | RF coil magnetic field mapper | Sniffer coil printhead replacement | 2016 | 49 |
| Automated platform for MALDI matrix deposition | Wanhao duplicator i3 | MALDI matrix deposition | Nebulizer printhead replacement | 2018 | 50 |
| Dual purpose electrode sprayer | Creality Ender-3 V2 Neo | Battery electrode deposition | Airbrush nozzle printhead replacement, heatbed upgrade | 2024 | 51 |
| Ender VX500 | Creality Ender-3 | Automated RNA extraction platform | Tube holder printhead replacement | 2021 | 52 |
| Lateral flow assay construction system | Not specified (pictures indicate creality Ender-3) | Construction of bioassay spots | Fountain pen dispenser printhead replacement | 2023 | 53 |
| Incubot | Tronxy X1 | Microscope for tissue culture imaging | Microscope hardware printhead replacement | 2021 | 54 |
| Enderscope | Creality Ender-3 | Microplastic detection with scanning microscope | Microscope hardware printhead replacement | 2024 | 55 |
| HistoEnder | Creality Ender-3 | Histology slide autostaining | Slide holder printhead add-on | 2022 | 56 |
| Automated dip coater | Creality Ender-3 | Open-source dip coating apparatus | Extruder motor gripper modification | 2023 | 57 |
| HistoEnder TEM grid stainer | Creality Ender-3 | Staining of electron microscopy grids | Addition of TEM grid holders | 2024 | 58 |
| 3D-printer-based fraction collector | Creality Ender-5 S1 | Automated fraction collection | Capillary holder printhead replacement & well plate holder | 2024 | 59 |
| Ender-3-based pipetting robot | Creality Ender-3 pro | Automated liquid handling | Pipette holder printhead replacement | 2024 | 60 |
| PALH | Creality Ender-3 | Automated liquid handling | Stepper motor controlled custom pipette printhead replacement | 2025 | 61 |
| Ender-3-based syringe pumps | Creality Ender-3 | Open-source hardware syringe pump system | 3D printer deconstructed & components reused | 2021 | 62 |
| 3D-printer-based lab material handler | Prusa i3 | Handling of magnetic structures | Magnetic ‘hand’ add-on | 2017 | 67 |
| Ultrasonic immersion setup | VORON V0 | Material characterization | Immersion tank & transducer holder | 2025 | 68 |
| Otto validator | Creality Ender-3 pro | Franz diffusion cell autosampler | Reversible add-on of peristaltic pumps, vial holders, & deposition needles | 2025 | 69 |
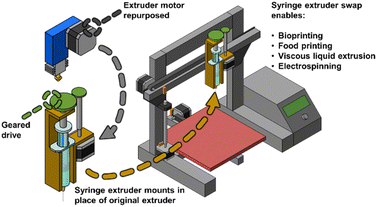
Part 1 – using the fourth motor
One of the easiest transformations, and one of the earliest to appear in literature, of the fourth motor is to reuse it to push a syringe plunger. Here, the rotational motion of the stepper motor is transformed into linear motion using gears and leadscrews. An early example of this approach was achieved by Vak's group in 2015.26 Vak and colleagues modified a 3D printer (Felix 2.0) to use a homemade, 3D-printed syringe holder. This 3D syringe pump setup was used to produce organic bulk heterojunction (BHJ) solar cells. The authors not only used the syringe to constantly push liquid through the nozzle but could also deposit a single drop of liquid, raise the nozzle, and then use the nozzle to slide the liquid over the build plate to fabricate a continuous 1D coating, showcasing the customizability of the setup.Following the initial proof of principle of the first syringe extruder, multiple 3D printers have subsequently been modified for use as 3D bioprinters.
Syringe extrusion: bioprinters, food printers, and more
An early example of a 3D printer printhead modified with a controllable syringe for bioprinting was from Bruno and Sachs's group in 2016.27 Here, a Felix 3.0 printer was modified for precise cell placement, using microcapillaries instead of standard syringe pumps. The group managed to reach the outstanding precision of a 1 nL extrusion, and hence was even able to reliably extrude single cells (each one suspended in a nanolitre of medium).In 2019, as 3D printers continued to become increasingly inexpensive, Gilbert's group modified an Anet A8 to make an ultra-low-cost 3D bioprinter.28 The printer itself is portable and can be used within a standard flow cabinet. In the same year, Reynaud and Rodriguez's groups modified an Ultimaker Original for use as a 3D bioprinter.29 The researchers optimized multiple variables of the printing process, such as extrusion and movement speed, to enhance the robot's capabilities for fabricating stable gel structures. In a further paper published in the same year, Brand-Saberi's group modified a Prusa i3 to be a bioprinter, which they named NOSE (Nydus One Syringe Extruder).30 Here, the authors used this modified printer for the FRESH (Freeform Reversible Embedding of Suspended Hydrogel) method of bioprinting. In this approach, a cross-linkable or gelatinizing liquid is printed not on a standard flatbed, but inside a gelatin bath. After printing, the bath can be heated to remove the gelatin, leaving the printed object behind. The original FRESH used a blended gelatin puck as a supporting bath,31 whereas FRESH 2.0 employed gelatin coacervate in its place.32 Using a suspension bath as the support enables the printing of complex and hollow structures, such as vascular systems, as well as those made from readily deformable materials like collagen.
It is important to note that the developers of the FRESH bioprinting system, the Feinberg group, are also the ideators of the Replistruder,33 one of the initial designs and still the most popular design of a syringe extruder. The Replistruder is now available in its 4th34 and 5th iterations.35
In 2020, Ioannidis and Taraviras's groups also used an Anet A8 to build a bioprinter.36 Compared to previous systems, their setup allowed for the fast swapping of syringes. The ability to rapidly switch between syringes with different diameters can be vital when dealing with time-sensitive bio-inks.
The Anet A8 was one of the cheapest commercially available 3D printers, but unfortunately suffered from reliability issues, being implicated in several fire accidents. At the end of 2018, another Chinese 3D printer company, Creality, began marketing the Ender 3, a sub-€200 printer that was simultaneously reliable and modifiable, and built on open hardware principles.37 In recent years, the Creality Ender 3 and its subsequent Ender models have proven to be low-cost and reliable.
For this reason, in 2024, Crosby's group used the Ender 3 as a base to build a bioprinter called the “Enderstruder”.38 Their research was supported by a full step-by-step guide to building and using the hardware and software.
For more information about 3D bioprinters, we recommend the recent review “Open-source extrusion 3D bioprinters: Trends and recommendations” from the same group.39
Food printers work on the same principle as bioprinters. A syringe is filled with food paste, and this modified 3D printer is used to print food such as chocolate, candy, or even in vitro meat. In 2023, Demircan and Özçelik successfully introduced this method by modifying a 3D printer to print three different food pastes.40 The researchers also propose using a cooling unit to blow cold air while printing, with the suggestion that this could improve the stability of materials printed in melted or softened forms.
In 2024, Siano's group expanded the use of the syringe by adding another stepper motor to a Hellbot Magna 1 3D printer.41 The motherboard of this printer allows for the addition of a fifth stepper motor, which the authors used for a second syringe pump. The group also modified the firmware by adding a “Mixing Extruder” option. With this, the two extrusion motors can be used either independently or with mixing in any ratio between them. This new mode enabled the simultaneous use of the two syringe pumps to dispense different amounts of liquids onto paper strips as test and control lines for lateral flow immunoassays (LFIA). This modified 3D printer enabled more controlled and reproducible fabrication of lateral flow assay equipment.
In general, the approach of switching the printing head with a syringe holder and using the fourth motor to push the plunger can be applied to any paste or highly viscous material, such as silicones or ceramic material.42
Electrospinning
If the syringe needle and printbed are attached to a high-voltage power supply, then 3D syringe printers can be transformed into 3D electrospinning printers, where electrical forces are used to stretch a liquid polymer to produce micron- or nano-sized fibers. Those fibers are then utilized for a multitude of applications, ranging from filtration to bioscaffolds. Typically, electrospinning produces only long fibers, which then need to be molded to form the final structure. A 3D printing electrospinning machine produces both nano- and microfibers simultaneously, depositing them in any desired 3D shape.This approach can be seen in the early work of Radacsi's group, where an Ultimaker printer was modified into an electrospinning setup, with which the authors were able to produce different sizes of fibers and structures.43
In 2021, the same group modified a 3D printer to function as both a bioprinter and an electrospinning machine. The bioprinting part was able to produce a 3D structure using bio-ink, while an external electrospinning head coated this biostructure with nanofibers for reinforcement.44 This was the first DIY hybrid bioprinting-electrospinning setup that was based on the modification of a commercially available 3D printer.
In 2023, Kim's group showed how to make electrospun polycaprolactone (PCL) membranes using a modified 3D printer.45 The use of the 3D printer allowed the researchers to quickly screen many variables, including pore size, percentage of PCL in the solution, and tensile strength. The material was then used as a substrate for bioassays.
In 2024, Maniruzzaman's group developed the “MaGIC” system (Multi-channeled and Guided Inner-Controlling printheads).46 This technology involves coupling two different syringes to the same electrospinning printhead. The new technique enabled the group to produce multicomponent nanofibers with different functionalizations. The two syringes can be used singularly, producing single nanofibers, or together in mixing mode, which produces either “alongside” (parallel sub-strands) or “mixed” (complex morphology) electrospun fibers.
For more on 3D printers and electrospinning, we suggest Ejiohuo's review “A perspective on the synergistic use of 3D printing and electrospinning to improve nanomaterials for biomedical applications”.47
Analogously to electrospinning, but using molten polymers, Melt Electrowriting Printers (MEW), such as the MEWron, have been used to print Nylon-12 microfibers.48
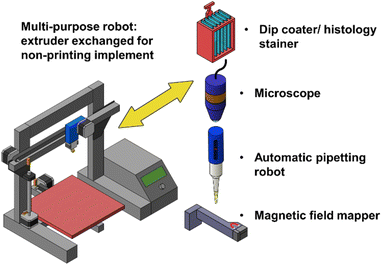
Part 2 – exchanging the printhead
Another, possibly easier, way to hack a 3D printer into a 3-axis robot is to replace the printhead with a different tool, eliminating the need to use the extruder motor (Fig. 3). In this way, the printer is used as a Cartesian robot that can move its new attachment in XYZ space. This approach also allows the use of other controllable units instead of the printhead. These units can be driven and synchronized by an external unit, such as a single-board computer (SBC) e.g., a Raspberry Pi, or connected to other inputs/outputs of the 3D printer board and controlled by G-code.In the remainder of this section, we will discuss various tools that have been used as alternatives to the printhead in 3D printer-based robots.
Magnetic field mapper
In 2016, Aletras's group modified a Velleman K8200 build-it-yourself 3D printer to make a magnetic field mapper for magnetic resonance imaging (MRI) machines.49 The group attached a sniffer coil to the printhead and moved it using the XYZ axes of the 3D printer to map the B1 magnetic field generated by an MRI radiofrequency (RF) coil.Spraying robots
A few years later, in 2018, Mackay's group modified a 3D printer to produce and optimize a matrix for matrix-assisted laser desorption-ionization mass spectrometry (MALDI MS).50 The printhead was replaced with a nebulizer for producing different matrices at varying speeds and pressures. Similarly, in 2024, Aivaliotis and Vernardou used the printhead as a spray setup to make thin films for batteries.51 The spray gun on the printhead enabled production of thin films with different properties.Sample preparation and lateral flow immunoassays
At the peak of the COVID-19 pandemic in 2020, the Stinear group modified a 3D printer for automatic RNA extraction.52 The printhead was exchanged with a tube holder and magnetic combs for moving RNA samples on magnetic particles through different washing steps before PCR amplification. This was a cost-effective method to automate sample preparation. On the same topic, the Kalogianni group also used a 3D printer robot to prepare LFIA.53 These authors modified the printhead to accommodate two simple pens for dispensing reagents onto the membranes of lateral flow devices, making them ready for use by consumers. This method is simple and reproducible, and can be modified with different reagent concentrations, amounts, line width, and placement on the lateral flow membrane.Automated microscopes
In 2021, the Pickering group modified a printer to hold a microscope, and used the XYZ axes for sample positioning. They demonstrated that the entire setup can be accommodated in an incubator to monitor cells in real-time and for long-term experiments.54Burke has also recently modified a 3D printer for reflection and fluorescence analyses of microplastics across the entire 20 × 20 cm print bed. The innovation lies in the ability to switch between microscope and 3D printing modes, and the different heads can be swapped in just a few minutes. The microscope can automatically switch between reflection and fluorescence by using a G-code command to switch filters mechanically.55 The authors also tested the positional error of the 3D printer by performing a series of repeated, oscillating 6 mm movements (29 movements in each direction, 58 movements in total) and determined that the positional deviation was ±4 µm in the x- and y-axes.
Staining and coating robots
Histology staining is a tedious procedure in which glass slides mounted with tissue need to be treated with different reagents at specific times. To automate this process, in 2022, Ponzetti and Saggiomo modified a 3D printer printhead to include a glass slide holder.56 The histology glass slides were then automatically placed in different reagent vats on the build plate, thereby completely automating the histology routine. One year later, in 2023, Rauh's group improved this methodology by transforming a 3D printer into a dip coating machine.57 In this case, the fourth motor was cleverly used as an automatic clamp for picking up and releasing a single glass slide (or other material). The dipping and lifting speed of the slide from containers with coating agents changed the coating density and quality. Soon afterwards, in 2025, Seifert used a similar approach, this time for automated electron microscopy grid staining.58Robotic samplers
In 2024, Mohammed's group used a 3D printer as a programmable fraction collector for samples as small as nanogram-scale.59 The printhead was replaced with a fraction collector, connected to a nano-LC equipped with a C-18 column, for nanogram-scale fractionation. In the same year, Wah Ng developed a cost-effective liquid handler by printing a holder for a standard automatic pipette to be mounted in place of the printhead.60 The fourth motor was used to push the plunger up and down, taking up and dispensing liquid. This approach, however, can only be used for a fixed volume of liquid, as there is no function for automatically rotating the pipette's adjustment knob to change volumes. To overcome this problem, in 2025, Tripathi's group improved the single automatic pipette system with a fully automated, 3D printer-based liquid handler, which they called “Personal Automated Liquid Handler” (PALH).61 In this system, the liquid volume can be adjusted, and the pipette tip can be discarded and changed automatically. This liquid handler is also capable of using the printer's two heating elements and incorporating magnetic separation. This cost-effective robot is hence comparable in function, if not better than, commercial systems that can cost up to ten times as much.Syringe pumps
Another way to use a 3D printer as a laboratory robot is not by modifying it, but using its components to construct a new electromechanical system. This is the approach that Saggiomo's group used in 2021, where most of the components inside a 3D printer box were used to build three syringe pumps.62 We used the linear movement of the 3D printer and electronics to 3D print some adaptors, thereby manufacturing three programmable syringe pumps.Part 3 – hardware and software
Hardware
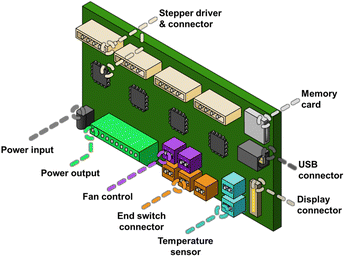
Recent years have seen the development of 3D printer motherboards with many controllable connections, beyond simple 3-axis motor and switch I/O. For instance, most 3D printer motherboards have at least two fan controllers. In the non-modified 3D printer, one fan is used to cool the heat sink in the print head, and the other to cool the material after extrusion (Fig. 4). These devices typically operate at 12 V or 24 V, and their speed can be controlled via PWM (Pulse Width Modulation). However, other electronics can also be attached to those connectors and controlled by the printing software. This approach was used by Aivaliotis and Vernardou, where a 24 V solenoid air valve was attached to a fan connector and controlled by the printer to initiate the spray coating process.51The motherboard also has at least three connectors intended for the end stops (switches) of the three axes. The switches are used to home the printhead to the (0,0,0) XYZ position. They are wired to input/output (I/O) ports, and can vary from physical switches to optical sensors. The ports are digital on/off and can, in principle, be used to set the printhead at a specific position, for example, in the case of microscopy. Hypothetically, these switches could also be exchanged with other 5 V sensors, although to our knowledge, this has not yet been reported.
The motherboard also requires connections for the power input, typically 12 V or 24 V DC. There is a connection for an LCD screen, which can be used to set up the printer/robot. Many printers have a USB port and an SD card reader as input options, which can be used to load G-code files to control the robot's movement.
These are the minimum required connections on a standard, low-level FDM 3D printer. Nowadays, more sophisticated printers and motherboards have increased connectivity, with some models supporting up to seven motors, multiple fans, additional connections and controls for LEDs, multiple heating units, and more, thereby opening up a wide range of robotic applications.
Although many 3D printers operate on a bare motherboard, their capabilities can be enhanced with more advanced motherboards, such as the Duet system.63 These open-source boards, and their expansion boards, can increase the number of possible motors and sensors. They also enable the use of RepRap firmware, which is more powerful than the standard Merlin firmware (see next section). One such system built on top of a Duet board is the Labm8,64 a commercially-available microfluidic platform.
It should be noted that although the stepper motors can move repeatedly and reproducibly by a few microns, the instrument may be composed of multiple parts, which can worsen its performance, and care should be taken during calibration of any new build.
Software
A robot would not be a robot without some form of control. Luckily, a 3D printer already comes with preinstalled firmware. The G-code line-by-line language was developed in the early 1960s and is still used today to control fabrication machines, including printers, CNC machines, and lathes. G-code is a relatively simple language that controls movement, input, and output from the motherboard through each line. Due to its simplicity, plain G-code lacks useful programming constructs such as logical operators and conditional statements. However, extensions to the language can be used to create looping and conditional statements, depending on the firmware used by the machine.One of the most common firmware types to date is Marlin, as it is lightweight and contains most of the standard and some more advanced 3D printing parameters, such as auto-bed leveling and filament runout sensing. However, the more powerful RepRap firmware allows the operator to use more advanced G-code commands and parameters. Similarly, the newly-developed Klipper firmware is gaining traction due to its expanded functionality. One example of improvement over Marlin is that both RepRap and Klipper support “meta” commands, which enable the construction of basic programming constructs such as conditionals, loops, variables, and parameters, thereby transforming G-code into a more complex and useful programming language.
A comprehensive list of G-code commands and their compatible firmware is available online.65
At this point, the G-code can be saved on the SD card and run locally on the printer, or the commands can be sent line by line from a computer over a serial connection (i.e., USB). This process can be simplified by attaching an SBC such as a Raspberry Pi for running the commands, as the SBC can also be interfaced with other hardware. There are multiple Python libraries designed for interfacing SBCs or computers to 3D printers/robots. For example, Aivaliotis and Vernardou wrote a Python graphical user interface (GUI) for controlling their robotic spray setup,51 and Rousseau has published a full Python library66 for controlling and using the Enderscope. Another approach is to use DIOS (Design of Inputs-Outputs with Sikuli), which uses the open-source Sikuli platform to control GUI components. This provides a general framework for designing automated experimental procedures, requiring no prior knowledge of programming or electronics.67
Conclusions and perspective
Although hacking 3D printers to be used as laboratory robots has demonstrated value in terms of both cost and customization, many untapped possibilities remain. Newer motherboards, along with improved firmware, are expanding the possibilities of incorporating more sensors and electronics onto a single board. For example, many new printers have a bed leveling option, which can be repurposed to check the height of glass slides, lateral flow assays, accessory holders, and other elements, facilitating the automatic positioning of these elements on the build plate relative to each other.Using an external SBC allows the addition of even more sensors and electronics, all orchestrated by a single unit. This is, for example, useful to connect a camera that can control the robot. The camera can easily be attached to the SBC, and its feed used automatically or autonomously to control the robot using computer vision. Further, an SBC can be used to control and link multiple 3D printers/robots to work in conjunction. This approach may be the easiest and fastest solution to democratic self-driving labs (SDLs), namely cost-effective robots that can be built and used without advanced knowledge of electronics or programming.
And a final word of warning: to enable the democratization of the SDL and improve the use of robotics in chemical laboratories, the hardware and software must be replicable and reproducible. Recently, we have seen a push to make software and raw data fully available with scientific journal publications. However, we cannot say the same for the hardware, which is typically briefly discussed in the experimental section at best, in many cases lacking a full guide on how to build/assemble it. This must change, and we call on the community of authors, reviewers, and editors to collectively work towards better reproducibility of hardware, thereby improving systems for all. A starting point would be to explicitly list all components, including 3D CAD files (such as STL and STEP file types), and provide more detailed assembly instructions in hardware-focused publications. This should be the first step in progressing open-source hardware.
During the writing of this review, some new 3D printer hacks have been published: using a 3D printer for an ultrasonic immersion test setup,68 a Franz diffusion cell autosampler.,69,70 a 3D printer for Aerogels.71 We also missed an HPLC fraction collector.72
Author contributions
Conceptualization: V. S. Literature review: S. B., N. C., V. S. Writing: V. S., N. C., S. B. Visualization: S. B.Conflicts of interest
There are no conflicts to declare.Data availability
This review does not contain any additional data.Acknowledgements
VS and SB gratefully acknowledge Open Science NL, Open Science Fund for funding.Notes and references
- C. W. Coley, N. S. Eyke and K. F. Jensen, Autonomous Discovery in the Chemical Sciences Part I: Progress, Angew. Chem., Int. Ed., 2020, 59(51), 22858–22893, DOI:10.1002/anie.201909987.
- X. Caldentey and E. Romero, High-Throughput Experimentation as an Accessible Technology for Academic Organic Chemists in Europe and Beyond, Chem. Methods, 2023, 3(5), e202200059, DOI:10.1002/cmtd.202200059.
- C. W. Coley, N. S. Eyke and K. F. Jensen, Autonomous Discovery in the Chemical Sciences Part II: Outlook, Angew. Chem., Int. Ed., 2020, 59(52), 23414–23436, DOI:10.1002/anie.201909989.
- M. Christensen, L. P. E. Yunker, P. Shiri, T. Zepel, P. L. Prieto, S. Grunert, F. Bork and J. E. Hein, Automation Isn't Automatic, Chem. Sci., 2021, 12(47), 15473–15490, 10.1039/D1SC04588A.
- K. Antonios, A. Croxatto and K. Culbreath, Current State of Laboratory Automation in Clinical Microbiology Laboratory, Clin. Chem., 2022, 68(1), 99–114, DOI:10.1093/clinchem/hvab242.
- Y. Xie, K. Sattari, C. Zhang and J. Lin, Toward Autonomous Laboratories: Convergence of Artificial Intelligence and Experimental Automation, Prog. Mater. Sci., 2023, 132, 101043, DOI:10.1016/j.pmatsci.2022.101043.
- S. Lo, S. G. Baird, J. Schrier, B. Blaiszik, N. Carson, I. Foster, A. Aguilar-Granda, S. V. Kalinin, B. Maruyama, M. Politi, H. Tran, T. D. Sparks and A. Aspuru-Guzik, Review of Low-Cost Self-Driving Laboratories in Chemistry and Materials Science: The “Frugal Twin” Concept, Digital Discovery, 2024, 3(5), 842–868, 10.1039/D3DD00223C.
- J. Y. Pan, Engineering Chemistry Innovation, ACS Med. Chem. Lett., 2019, 10(5), 703–707, DOI:10.1021/acsmedchemlett.9b00096.
- N. Carson, Rise of the Robots, Chem.–Eur. J., 2020, 26(15), 3194–3196, DOI:10.1002/chem.202000656.
- D. Plant, P. Harper, G. Holdgate, D. Murray, H. Plant, J. Vincent and M. Wigglesworth, Laboratory Automation in Early Drug Discovery, Drug Target Review, 2020, 7(1), 21–24 Search PubMed.
- W. Gao, P. Raghavan and C. W. Coley, Autonomous Platforms for Data-Driven Organic Synthesis, Nat. Commun., 2022, 13(1), 1075, DOI:10.1038/s41467-022-28736-4.
- J. Bai, L. Cao, S. Mosbach, J. Akroyd, A. A. Lapkin and M. Kraft, From Platform to Knowledge Graph: Evolution of Laboratory Automation, JACS Au, 2022, 2(2), 292–309, DOI:10.1021/jacsau.1c00438.
- G. Tom, S. P. Schmid, S. G. Baird, Y. Cao, K. Darvish, H. Hao, S. Lo, S. Pablo-García, E. M. Rajaonson, M. Skreta, N. Yoshikawa, S. Corapi, G. D. Akkoc, F. Strieth-Kalthoff, M. Seifrid and A. Aspuru-Guzik, Self-Driving Laboratories for Chemistry and Materials Science, Chem. Rev., 2024, 124(16), 9633–9732, DOI:10.1021/acs.chemrev.4c00055.
- V. Saggiomo, A 3D Printer in the Lab: Not Only a Toy, Adv. Sci., 2022, 9(27), 2202610, DOI:10.1002/advs.202202610.
- R. Jones, P. Haufe, E. Sells, P. Iravani, V. Olliver, C. Palmer and A. Bowyer, RepRap – the Replicating Rapid Prototyper, Robotica, 2011, 29(1), 177–191, DOI:10.1017/S026357471000069X.
- E. E. Petersen and J. Pearce, Emergence of Home Manufacturing in the Developed World: Return on Investment for Open-Source 3-D Printers, Technologies, 2017, 5(1), 7, DOI:10.3390/technologies5010007.
- RepRap contributors. RepRap, https://reprap.org/, accessed 2025-07-27 Search PubMed.
- K. Rajan, M. Samykano, K. Kadirgama, W. S. W. Harun and Md. M. Rahman, Fused Deposition Modeling: Process, Materials, Parameters, Properties, and Applications, Int. J. Adv. Manuf. Technol., 2022, 120(3), 1531–1570, DOI:10.1007/s00170-022-08860-7.
- H. Agrawaal and J. E. Thompson, Additive Manufacturing (3D Printing) for Analytical Chemistry, Talanta Open, 2021, 3, 100036, DOI:10.1016/j.talo.2021.100036.
- A. S. Pamidi, M. B. Spano and G. A. Weiss, A Practical Guide to 3D Printing for Chemistry and Biology Laboratories, Curr. Protoc., 2024, 4(10), e70036, DOI:10.1002/cpz1.70036.
- O. Jennotte, N. Koch, A. Lechanteur and B. Evrard, Three-Dimensional Printing Technology as a Promising Tool in Bioavailability Enhancement of Poorly Water-Soluble Molecules: A Review, Int. J. Pharm., 2020, 580, 119200, DOI:10.1016/j.ijpharm.2020.119200.
- NIH 3D. https://3d.nih.gov/, accessed 2025-09-05.
- T. Rayna, L. Striukova and J. Darlington, Co-Creation and User Innovation: The Role of Online 3D Printing Platforms, J. Eng. Technol. Manag., 2015, 37, 90–102, DOI:10.1016/j.jengtecman.2015.07.002.
- J. Cui, L. Ren, J. Mai, P. Zheng and L. Zhang, 3D Printing in the Context of Cloud Manufacturing, Robot. Comput.-Integr. Manuf., 2022, 74, 102256, DOI:10.1016/j.rcim.2021.102256.
- ChemDraw | Revvity Signals Software, https://revvitysignals.com/products/research/chemdraw, accessed 2025-09-05 Search PubMed.
- D. Vak, K. Hwang, A. Faulks, Y.-S. Jung, N. Clark, D.-Y. Kim, G. J. Wilson and S. E. Watkins, 3D Printer Based Slot-Die Coater as a Lab-to-Fab Translation Tool for Solution-Processed Solar Cells, Adv. Energy Mater., 2015, 5(4), 1401539, DOI:10.1002/aenm.201401539.
- J. A. Reid, P. A. Mollica, G. D. Johnson, R. C. Ogle, R. D. Bruno and P. C. Sachs, Accessible Bioprinting: Adaptation of a Low-Cost 3D-Printer for Precise Cell Placement and Stem Cell Differentiation, Biofabrication, 2016, 8(2), 025017, DOI:10.1088/1758-5090/8/2/025017.
- M. Kahl, M. Gertig, P. Hoyer, O. Friedrich and D. F. Gilbert, Ultra-Low-Cost 3D Bioprinting: Modification and Application of an Off-the-Shelf Desktop 3D-Printer for Biofabrication, Front. Bioeng. Biotechnol., 2019, 7, 184, DOI:10.3389/fbioe.2019.00184.
- M. Alruwaili, J. A. Lopez, K. McCarthy, E. G. Reynaud and B. J. Rodriguez, Liquid-Phase 3D Bioprinting of Gelatin Alginate Hydrogels: Influence of Printing Parameters on Hydrogel Line Width and Layer Height, Bio-Des. Manuf., 2019, 2(3), 172–180, DOI:10.1007/s42242-019-00043-w.
- N. Bessler, D. Ogiermann, M.-B. Buchholz, A. Santel, J. Heidenreich, R. Ahmmed, H. Zaehres and B. Brand-Saberi, Nydus One Syringe Extruder (NOSE): A Prusa I3 3D Printer Conversion for Bioprinting Applications Utilizing the FRESH-Method, HardwareX, 2019, 6, e00069, DOI:10.1016/j.ohx.2019.e00069.
- T. J. Hinton, Q. Jallerat, R. N. Palchesko, J. H. Park, M. S. Grodzicki, H.-J. Shue, M. H. Ramadan, A. R. Hudson and A. W. Feinberg, Three-Dimensional Printing of Complex Biological Structures by Freeform Reversible Embedding of Suspended Hydrogels, Sci. Adv., 2015, 1(9), e1500758, DOI:10.1126/sciadv.1500758.
- A. Lee, A. R. Hudson, D. J. Shiwarski, J. W. Tashman, T. J. Hinton, S. Yerneni, J. M. Bliley, P. G. Campbell and A. W. Feinberg, 3D Bioprinting of Collagen to Rebuild Components of the Human Heart, Science, 2019, 365(6452), 482–487, DOI:10.1126/science.aav9051.
- NIH 3D - Replistruder Syringe-Based Extruder, https://3d.nih.gov/entries/3DPX-002102, accessed 2025-11-13.
- J. W. Tashman, D. J. Shiwarski and A. W. Feinberg, A High Performance Open-Source Syringe Extruder Optimized for Extrusion and Retraction during FRESH 3D Bioprinting, HardwareX, 2021, 9, e00170, DOI:10.1016/j.ohx.2020.e00170.
- J. W. Tashman and D. J. Shiwarski, Replistruder 5 - Syringe Pump Extruder for 3D Printing, 2022, DOI:10.5281/zenodo.7135874.
- K. Ioannidis, R. I. Danalatos, S. Champeris Tsaniras, K. Kaplani, G. Lokka, A. Kanellou, D. J. Papachristou, G. Bokias, Z. Lygerou and S. Taraviras, A Custom Ultra-Low-Cost 3D Bioprinter Supports Cell Growth and Differentiation, Front. Bioeng. Biotechnol., 2020, 8, 580889, DOI:10.3389/fbioe.2020.580889.
- Creality Co. Ltd, Creality3DPrinting/Ender-3, 2021, https://github.com/Creality3DPrinting/Ender-3, accessed 2021-06-01.
- D. J. Cordova, A. A. Rodriguez, S. C. Woodward and C. O. Crosby, The Enderstruder: An Accessible Open-Source Syringe Extruder Compatible with Ender Series 3D Printers, HardwareX, 2024, 17, e00510, DOI:10.1016/j.ohx.2024.e00510.
- C. O. Crosby, Open-Source Extrusion 3D Bioprinters: Trends and Recommendations, Bioprinting, 2024, 38, e00336, DOI:10.1016/j.bprint.2024.e00336.
- E. Demircan and B. Özçelik, Development of Affordable 3D Food Printer with an Exchangeable Syringe-Pump Mechanism, HardwareX, 2023, 14, e00430, DOI:10.1016/j.ohx.2023.e00430.
- G. Siano, L. Peretti, J. M. Márquez, N. Pujato, L. Giovanini and C. Berli, 3D Printer-Controlled Syringe Pumps for Dual, Active, Regulable and Simultaneous Dispensing of Reagents. Manufacturing of Immunochromatographic Test Strips, Microchem. J., 2024, 201, 110652, DOI:10.1016/j.microc.2024.110652.
- S. Fu, M. Zhu and Y. Zhu, Organosilicon Polymer-Derived Ceramics: An Overview, J. Adv. Ceram., 2019, 8(4), 457–478, DOI:10.1007/s40145-019-0335-3.
- M. Vong, F. J. Diaz Sanchez, A. Keirouz, W. Nuansing and N. Radacsi, Ultrafast Fabrication of Nanofiber-Based 3D Macrostructures by 3D Electrospinning, Mater. Des., 2021, 208, 109916, DOI:10.1016/j.matdes.2021.109916.
- F. Fazal, F. J. Diaz Sanchez, M. Waqas, V. Koutsos, A. Callanan and N. Radacsi, A Modified 3D Printer as a Hybrid Bioprinting-Electrospinning System for Use in Vascular Tissue Engineering Applications, Med. Eng. Phys., 2021, 94, 52–60, DOI:10.1016/j.medengphy.2021.06.005.
- C. Xu, G. Bonfante, J. Park, V. Salles and B. Kim, Fabrication of an Electrospun Polycaprolactone Substrate for Colorimetric Bioassays, Biomed. Microdevices, 2024, 26(2), 32, DOI:10.1007/s10544-023-00673-z.
- A. Y. Darwesh, Y. Zhang, N. H. Aghda, F. Alkadi and M. Maniruzzaman, Advanced 3D Electrospinning “Xspin” System: Fabrication of Bifiber Floating Oral Pharmaceutical Scaffolds for Controlled Drug Delivery, Mol. Pharm., 2024, 21(2), 916–931, DOI:10.1021/acs.molpharmaceut.3c00982.
- O. Ejiohuo, A Perspective on the Synergistic Use of 3D Printing and Electrospinning to Improve Nanomaterials for Biomedical Applications, Nano Trends, 2023, 4, 100025, DOI:10.1016/j.nwnano.2023.100025.
- A. Reizabal, T. Kangur, P. G. Saiz, S. Menke, C. Moser, J. Brugger, P. D. Dalton and S. Luposchainsky, MEWron: An Open-Source Melt Electrowriting Platform, Addit. Manuf., 2023, 71, 103604, DOI:10.1016/j.addma.2023.103604.
- A. Vavoulas, N. Vaiopoulos, E. Hedström, C. G. Xanthis, H. G. Sandalidis and A. H. Aletras, Using a Modified 3D-Printer for Mapping the Magnetic Field of RF Coils Designed for Fetal and Neonatal Imaging, J. Magn. Reson., 2016, 269, 146–151, DOI:10.1016/j.jmr.2016.06.005.
- L. H. Tucker, A. Conde-González, D. Cobice, G. R. Hamm, R. J. A. Goodwin, C. J. Campbell, D. J. Clarke and C. L. Mackay, MALDI Matrix Application Utilizing a Modified 3D Printer for Accessible High Resolution Mass Spectrometry Imaging, Anal. Chem., 2018, 90(15), 8742–8749, DOI:10.1021/acs.analchem.8b00670.
- D. Aivaliotis and D. Vernardou, An Affordable Dual Purpose Spray Setup for Lithium-Ion Batteries Thin Film Electrode Deposition, Materials, 2024, 17(20), 5114, DOI:10.3390/ma17205114.
- K. Vandelannoote, A. H. Buultjens, L. Li, L. K. Sharkey, M. Herisse, S. J. Pidot, T. Hoang, B. P. Howden, I. R. Monk, T. Seemann, J. Y. H. Lee and T. P. Stinear, Accessible Platform for High-Throughput COVID-19 Molecular Diagnostics and Genome Sequencing Using a Repurposed 3D Printer for RNA Extraction, ACS Biomater. Sci. Eng., 2021, 7(9), 4669–4676, DOI:10.1021/acsbiomaterials.1c00775.
- P. M. Kalligosfyri, S. S. Tragoulias, P. Tsikas, E. Lamprou, T. K. Christopoulos and D. P. Kalogianni, Design and Validation of a Three-Dimensional Printer-Based System Enabling Rapid, Low-Cost Construction of the Biosensing Areas of Lateral Flow Devices for Immunoassays and Nucleic Acid Assays, Anal. Chem., 2024, 96(1), 572–580, DOI:10.1021/acs.analchem.3c04915.
- G. O. T. Merces, C. Kennedy, B. Lenoci, E. G. Reynaud, N. Burke and M. Pickering, The Incubot: A 3D Printer-Based Microscope for Long-Term Live Cell Imaging within a Tissue Culture Incubator, HardwareX, 2021, 9, e00189, DOI:10.1016/j.ohx.2021.e00189.
- N. Burke, G. Müller, V. Saggiomo, A. R. Hassett, J. Mutterer, P. Ó Súilleabháin, D. Zakharov, D. Healy, E. G. Reynaud and M. Pickering, EnderScope: A Low-Cost 3D Printer-Based Scanning Microscope for Microplastic Detection, Philos. Trans. R. Soc. A., 2024, 382(2274), 20230214, DOI:10.1098/rsta.2023.0214.
- M. Ponzetti, G. Chinna Rao Devarapu, N. Rucci, A. Carlone and V. Saggiomo, HistoEnder: A 3D Printer-Based Histological Slide Autostainer That Retains 3D Printer Functions, HardwareX, 2022, 12, e00370, DOI:10.1016/j.ohx.2022.e00370.
- F. Rauh, O. Bienek, I. D. Sharp and M. Stutzmann, Conversion of a 3D Printer for Versatile Automation of Dip Coating Processes, Rev. Sci. Instrum., 2023, 94(8), 083901, DOI:10.1063/5.0128116.
- P. Seifert, Adaptation of the HistoEnder, an Open-Source 3D Printer for Automated Transmission Electron Microscopy Grid Staining, J. Histotechnol., 2025, 1–5, DOI:10.1080/01478885.2024.2403872.
- E. S. Kitano, G. Nisbet, Y. Demyanenko, K. M. Kowalczyk, L. Iselin, S. Cross, A. Castello and S. Mohammed, Repurposed 3D Printer Allows Economical and Programmable Fraction Collection for Proteomics of Nanogram Scale Samples, Anal. Chem., 2024, 96(28), 11439–11447, DOI:10.1021/acs.analchem.4c01731.
- A. Kopyl, Y. Yew, J. W. Ong, T. Hiscox, C. Young, M. Muradoglu and T. W. Ng, Automated Liquid Handler from a 3D Printer, J. Chem. Educ., 2024, 101(2), 640–646, DOI:10.1021/acs.jchemed.3c00855.
- D. Naranbat, B. Phelps, J. Murphy and A. Tripathi, How to Convert a 3D Printer to a Personal Automated Liquid Handler for Life Science Workflows, SLAS Technol., 2025, 30, 100239, DOI:10.1016/j.slast.2024.100239.
- S. Baas and V. Saggiomo, Ender3 3D Printer Kit Transformed into Open, Programmable Syringe Pump Set, HardwareX, 2021, 10, e00219, DOI:10.1016/j.ohx.2021.e00219.
- Duet3D Ltd. GitHub. https://github.com/Duet3D, accessed 2025-11-13 Search PubMed.
- Business - Webflow HTML website template. https://www.labm8.io/, accessed 2025-11-13.
- G-code - RepRap, https://reprap.org/wiki/G-code, accessed 2025-09-09 Search PubMed.
- S. Gharbi, E. Poiraud, H. L. Guenno, E. Grandgirard, C. Rousseau, N. Burke, J. Mutterer and D. Rousseau, Enderscope.Py: A Library for Computational Imaging Using the EnderScope Automated Microscope, SoftwareX, 2025, 31, 102210, DOI:10.1016/j.softx.2025.102210.
- G. G. Siano, M. Montemurro, M. R. Alcaráz and H. C. Goicoechea, Open-Source Assisted Laboratory Automation through Graphical User Interfaces and 3D Printers: Application to Equipment Hyphenation for Higher-Order Data Generation, Anal. Chem., 2017, 89(20), 10667–10672, DOI:10.1021/acs.analchem.7b02758.
- H. K. Adepu, C. M. O'Donnell, J. Y. Kim and L. D. Sotelo, A Practical and Frugal Approach to Building an Ultrasonic Immersion Test Setup, JASA Express Lett., 2025, 5(9), 092801, DOI:10.1121/10.0039221.
- H. K. Y. Chan, L. Archbold, W. M. Lau and K. W. Ng, Validating Otto: A Franz Diffusion Cell Autosampler to Automate in Vitro Permeation Studies, J. Pharm. Sci., 2025, 114(7), 103837, DOI:10.1016/j.xphs.2025.103837.
- K. W. Ng, L. Archbold and W. Man Lau, Building Otto: An open-source Franz diffusion cell autosampler for automating in vitro skin permeation studies, HardwareX, 2026 DOI:10.1016/j.ohx.2025.e00735.
- J. L. Okkabaz, S. Darvishi, I. S. Akgün, Ş. B. Barım, E. Özgönül, D. Kuduğ, Ç. Parmaksızoğlu, E. Şenses, C. Erkey and A. Kiraz, A New Framework for 3D Printing Aerogels witt Additives: Hardware and Ink Development, ACS Omega, 2026, 11(1), 340–348, DOI:10.1021/acsomega.5c02676.
- M. C. Carvalho and J. M. Oakes, Turning a 3D Printer into a HPLC Fraction Collector: A Tool for Compound-Specific Stable Isotope Measurements, Hardware, 2023, 1, 29–53, DOI:10.3390/hardware1010004.
| This journal is © The Royal Society of Chemistry 2026 |