Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
The PPP model – a minimum viable parametrisation of conjugated chemistry for modern computing applications
Marcel D.
Fabian
 *a,
Nina
Glaser
*a,
Nina
Glaser
 ab and
Gemma C.
Solomon
ab and
Gemma C.
Solomon
 ab
ab
aNNF Quantum Computing Programme, Niels Bohr Institute, University of Copenhagen, Denmark. E-mail: marcel.fabian@nbi.ku.dk
bNano-Science Center and Department of Chemistry, University of Copenhagen, Denmark
First published on 15th January 2026
Abstract
The semi-empirical Pariser–Parr–Pople (PPP) Hamiltonian is reviewed for its ability to provide a minimal model of the chemistry of conjugated π-electron systems, and its current applications and limitations are discussed. Since its inception, the PPP Hamiltonian has helped in the development of new computational approaches in instances where compute is constrained due to its inherent approximations that allow for an efficient representation and calculation of many systems of chemical and technological interest. The crucial influence of electron correlation on the validity of these approximations is discussed, and we review how PPP full configuration interaction-type calculations have enabled a deeper understanding of conjugated polymer systems. More recent usage of the PPP Hamiltonian includes its application in high-throughput screening activities to the inverse design problem, which we illustrate here for two specific fields of technological interest: singlet fission and singlet–triplet inverted energy gap molecules. Finally, we conjecture how utilizing the PPP model in quantum computing applications could be mutually beneficial.
1 Introduction
“The underlying physical laws necessary for the mathematical theory of a large part of physics and the whole of chemistry are thus completely known, and the difficulty is only that the exact application of these laws leads to equations much too complicated to be soluble. It therefore becomes desirable that approximate practical methods of applying quantum mechanics should be developed, which can lead to an explanation of the main features of complex atomic systems without too much computation.”, P. A. M. Dirac, 1929.The famous lines by Dirac from the introductory paragraph of his publication on quantum mechanics of many-electron systems1 offer a fascinating glimpse into the early formative years of quantum mechanics. Optimism regarding the veracity of the formalism was met by the realisation of the daunting nature of solving any chemically relevant multi-electron system. Before the invention of the digital computer, computation was a manual human endeavour and quantum mechanical description of many electron systems was beyond the scope of what was achievable. In the years that followed, theoretical chemistry has benefited tremendously from the introduction of computers and the steadily increasing hardware resources, following Moore's law, which eventually allowed the description of molecules and other systems of ever-increasing complexity. Nonetheless, the nature of these “complex atomic systems” means that an exact description is still outside the realms of computational feasibility.
The Pariser–Parr–Pople (PPP) Hamiltonian was devised as such an “approximate practical method” for the electronic structure problem, and in this perspective we follow its historic development and explore the future prospects in modern computing. We describe the approximations that have made the PPP Hamiltonian practical for chemistry, and why it remains relevant even as increasing computational capabilities removed earlier constraints. Finally, we give an outlook on how the PPP Hamiltonian might be impactful for quantum computing, which currently faces similar resource constraints to those seen in classical computing 70 years ago.
2 The origins of the PPP model
2.1 The Hückel model
The origin of the PPP methodology traces back to the beginnings of quantum mechanics itself. In 1931 Hückel formulated the molecular orbital (MO) theory for conjugated molecules,2–5 now known as the Hückel MO (HMO) model. It was later extended by Lennard-Jones, Coulson,6 and Longuet-Higgins7–13 to the general theory of π-electrons for unsaturated and aromatic molecules.14 With very limited computational effort, the HMO theory provided a qualitative understanding of π-electron conjugated systems and thus the ability for organic chemists to predict and plan experiments.14 At that time, this essentially meant calculations were done with pen and paper. While its use in state-of-the-art computation today has greatly diminished, HMO theory remains very valuable in teaching chemical concepts such as the chemical bond or the MO-LCAO approach (molecular orbitals from linear combinations of atomic orbitals) and is included in most chemistry textbooks.15The HMO model relies on several assumptions15 that are also relevant for our introduction to the PPP model, and we will summarize them here. The most consequential assumption is the distinction between two different types of one-electron functions that are called σ- and π-MOs, respectively. In the case of planar molecules, the σ-MOs are symmetric with respect to the molecular plane and are linear combinations of atomic orbitals (AOs) centred on the various atoms of the molecule. The σ-MOs define the structural backbone of the conjugated molecule, are assumed to be localized and transferable between the same atom types. Importantly, the σ-MOs are not treated explicitly in HMO theory.
The π-MOs are conceptually linear combinations of 2pz-orbitals that are antisymmetric with respect to the molecular plane (see Fig. 1). There are up to 2 electrons in each delocalized π-MO, that can move through the bonds between neighbouring atoms, defined by the resonance integral. The overlap between 2pz-orbitals is assumed to be orthonormal in the zero differential overlap (ZDO) approximation.16 This requires replacement of the original resonance integral, with a reduced resonance integral,15,17 which is treated in HMO theory as a semi-empirical parameter. Finally, the HMO model is a one-electron theory, which means that the electrons do not explicitly interact through the Hamiltonian.
 | ||
| Fig. 1 Schematic illustration of orbitals that can form σ-bonds (blue) and π-bonds (purple) in benzene. | ||
The omission of electron–electron interactions leads to one of the fundamental shortcomings of HMO theory.18,19 The electron–electron interaction terms were (and still can be) challenging to calculate: first, they are generally non-analytic, unless the AO basis is chosen to be represented by Gaussians, as suggested later by Boys.20 Furthermore, when treated naively, the number of terms grows as O(N4), with N being the number of orbitals. Due to these challenges, electron–electron interactions were typically not treated explicitly in quantum chemical calculations up to the early 1960s21 for anything but the smallest systems. As anecdotal evidence of the constrained computational power, we highlight that up to 1960 a grand total of 80 full ab initio calculations had been performed on molecules with 3 or more electrons.21,22 The first capable workstations and mainframes started to emerge at that time23 and quickly grew in capabilities. It was, however, clear that experimental chemists were interested in much larger molecules and their spectroscopic properties, which were inaccessible even with these growing computational resources. Hence, there was a large need for an approximate treatment of electron–electron interaction before an explicit inclusion of these terms was computationally feasible. This novel model would have to retain the simplicity and computability of the HMO theory, but through the inclusion of some form of electron–electron interaction, it would hopefully enable a more quantitative agreement with experimental results.
2.2 The PPP model
Such an extension to the HMO theory was indeed proposed in 1953 by Pariser and Parr,24,25 and separately also by Pople.26 Due to the similarity of the underlying assumptions, these methods are unified as the Pariser–Parr–Pople (PPP) model. The key insight for both proposals was that the ZDO approximation (see Section 3.2.2), already invoked in the HMO for the overlap matrix, would also simplify and reduce the number of electron–electron interaction integrals from O(N4) to a more manageable number of O(N2).18 These integrals were parametrised based on experimentally available data, which is why both the Hückel and PPP model belong to the class of semi-empirical approaches (see Section 2.3).Pariser and Parr started from the Hückel MOs and performed a configuration interaction (CI) calculation.24,25 This calculation could run on desk calculation machines available at the time23 by virtue of the reduction of integral terms that had to be evaluated. Pople also started from an initial Hückel guess and then self-consistently solved the Roothaan–Hall equation while invoking the ZDO approximation.26,27 The original suggestion of Pople was therefore tailored towards the ground state, whereas Pariser and Parr targeted the first few excited states.23 In later years, the combination of an initial self-consistent treatment with a subsequent limited CI calculation (such as, e.g., CIS)27 became very popular by virtue of its success in describing both ground states and electronic excitation spectra of organic molecules.
2.3 Beyond PPP: general semi-empirical and ab initio methods
While the ZDO approximation greatly reduces the number of non-zero electron–electron interaction terms, the remaining integrals still have to be determined somehow. Instead of calculating these integrals explicitly as in ab initio methods, Pariser and Parr suggested a semi-empirical approach, where the integrals are treated as parameters and fitted to certain experimental values.24,25 This calibration also helps compensate for neglected terms.28The initial limitation of the ZDO approximation to planar π-electron systems was lifted when Pople introduced the so-called complete neglect of differential overlap (CNDO) and neglect of diatomic differential overlap (NDDO) approximations that could treat general three-dimensional systems with σ-electrons explicitly.29 Based on these initial schemes, many semi-empirical methods have been proposed with increasing sophistication and generally also a larger number of parameters.16,28,30–33 The parametrisation for these methods followed different philosophies, with either fitting to experimental data or to ab initio Hartree–Fock (HF) results and targeting either ground state or excited state properties.28,34,35 More recently there have been efforts to improve parametrisations through machine learning approaches, for example for the application of the PPP Hamiltonian to organic radicals.36 This approach is potentially useful for additional heteroatom parametrisation as well, where there is less consensus on a standard compared to the carbon atom parametrisation.37–40
We want to mention the great contention over the correct parametrisation in specific semi-empirical methods41–45 here because it highlights the strengths and weaknesses of semi-empirical approaches in general and specifically in relation to ab initio approaches. Most semi-empirical models are built on the MO ab initio framework, but through the neglect or parametrisation of certain integrals can treat much larger systems.28 This is reliable within the limitations of the parametrisation, but can fail drastically outside. It is not necessarily clear a priori how well the parameters transfer between different systems when no reference data are available. Here, we mention an early version of the MINDO semi-empirical method as an example where the parametrisation failed, resulting in large errors in the heats of atomization/formation for highly strained molecules.23
Broadly, semi-empirical treatments lack the generality of a fully ab initio approach, where the path to improve results is known, namely through a higher level of theory or larger basis sets. At the same time, however, semi-empirical treatments might result in better agreement with experiments than ab initio calculations, while also being much faster and applicable to larger systems when limited computational resources are available. In terms of speed and generality, semi-empirical methods can be placed in-between a full ab initio treatment and molecular mechanics (MM) models.
3 PPP as the MVP of chemistry
The PPP Hamiltonian has been remarkably successful in computational chemistry applications, especially in the description and determination of excited-state properties of conjugated π-electron systems. In the following section, we will highlight why the PPP model is the minimal viable parametrisation (MVP) to describe many chemically relevant systems through comparison with other model Hamiltonians. We will then describe how many of the empirical approximations of the PPP Hamiltonian have been theoretically explained and validated once accurate ab initio calculations became computationally feasible.3.1 Comparison with other models
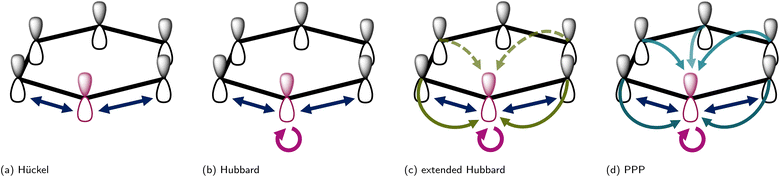
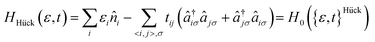 | (1) |
 creates (annihilates) an electron with spin σ in the pz-AO located on atom i and
creates (annihilates) an electron with spin σ in the pz-AO located on atom i and 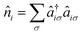 counts the total number of electrons on atom i. The onsite orbital energy εi, also known as the core resonance integral α, is often discarded as it only accounts for a constant energy shift when all atoms are equivalent. This is true for idealized geometries, (i.e. equal bond lengths) that do not contain any heteroatoms such as nitrogen or oxygen.46 The second term, tijHück is the kinetic energy or hopping integral, also known as the resonance integral β in the chemistry literature. This sum only runs over 〈i,j〉, which are connected atoms i and j (i.e., directly bonded, also termed nearest-neighbour atoms). An illustration of this interaction for benzene is shown in Fig. 2a. In solid-state physics, eqn (1) is also known as the tight-binding model.
counts the total number of electrons on atom i. The onsite orbital energy εi, also known as the core resonance integral α, is often discarded as it only accounts for a constant energy shift when all atoms are equivalent. This is true for idealized geometries, (i.e. equal bond lengths) that do not contain any heteroatoms such as nitrogen or oxygen.46 The second term, tijHück is the kinetic energy or hopping integral, also known as the resonance integral β in the chemistry literature. This sum only runs over 〈i,j〉, which are connected atoms i and j (i.e., directly bonded, also termed nearest-neighbour atoms). An illustration of this interaction for benzene is shown in Fig. 2a. In solid-state physics, eqn (1) is also known as the tight-binding model.
The Hückel Hamiltonian is a one-particle Hamiltonian that can also comprise the non-interacting part H0 of a more general interacting Hamiltonian, with a specific parameter set {ε,t}Hück. This Hamiltonian does not explicitly include electron–electron interactions, either between electrons on the same atom i, with different spin σ, or between electrons on different atoms. This omission limits the model's ability to accurately describe polar bonding46 and to predict the correct singlet–triplet energy splitting for a given electron configuration,21 both of which are chemically significant.
In polymers, these limitations are also evident: the Hückel Hamiltonian fails to reproduce experimentally observed features such as negative spin densities in linear polyenes,47,48 topological solitons in odd polyenes,47 and nonlinear optical properties in conjugated polyenes.49 Nevertheless, the Hückel Hamiltonian has been successful in providing a realistic description of charge mobility in conjugated polymer systems,50 after the inclusion of electron–phonon coupling terms, leading to the Holstein model. Overall, however, it has to be noted that the neglect of explicit electron–electron interaction in the Hückel model is often too drastic an approximation to answer chemically relevant questions.
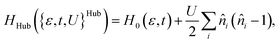 | (2) |
The Hubbard model has been widely applied in solid-state physics, but has also seen a more limited use in chemical contexts.15 The most common application of Hubbard-type models in chemistry is the DFT+U method, a density functional theory (DFT) approach, where the U parameter is included in the functional form for strongly correlated systems. As certain regimes of the Hubbard Hamiltonian (e.g., the half-filled 1D Hubbard model) have an analytical solution,53 the model can also be used as a benchmark for the development of novel quantum chemical methods for strongly correlated systems.
In conjugated π-electron systems, long-range electron–electron interactions often play a crucial role,54 which are not described by the Hubbard model. This has qualitatively significant consequences, for instance, the standard Hubbard model cannot describe the correct excited-state ordering in linear polyenes55 or a bound exciton in polymers (a bound state of an electron and a hole) due to the restriction to local, same-atom electron–electron interactions characterized by U.48 To describe bound excitons with the Hubbard model, some form of inter-atomic electronic interaction is necessary and one usually defines an extended Hubbard Hamiltonian (Fig. 2c)
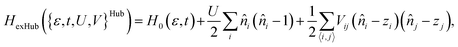 | (3) |
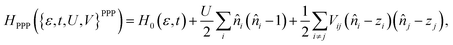 | (4) |
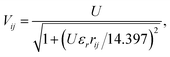 | (5) |
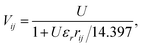 | (6) |
When comparing the different models, we can say that in the weak coupling limit, where U/t ≪ 1, all model Hamiltonians become Hückel-like. In this regime the description with a Hückel Hamiltonian can be very effective with a single parameter t or t/U. In the intermediate coupling range, the explicit parameter U yields both the (extended) Hubbard and PPP Hamiltonian. The main difference is in the functional form of the electronic interaction V: whereas V in the extended Hubbard model is a tunable but generally constant parameter for all nearest-neighbour or next-nearest neighbours, it is distance-dependent for the PPP Hamiltonian. A detailed comparison between the PPP and Hubbard Hamiltonians for finite graphene and polycyclic aromatic hydrocarbons (PAH) found that the PPP Hamiltonian succeeds in accounting for long-range interactions. These interactions effectively screen ionic charges, whereas the standard and extended Hubbard Hamiltonian can fail to capture this behaviour.51 In conclusion, the PPP model has been found to be generally better suited for describing the electronic properties of PAHs and related conjugated systems than the Hubbard models or even simpler Hückel Hamiltonians51 and therefore constitutes the minimum viable parametrisation for these molecular systems.
3.2 Validation
The PPP Hamiltonian is semi-empirical in its nature and in theory less general than a full ab initio treatment. In practice, the much more involved and computationally expensive ab initio methods often struggled to match the agreement with experiment as obtained through simpler and faster PPP model calculations.60 Of course, one could argue that this is unsurprising, given that semi-empirical PPP parameters represent a fit to experimental data. However, this argument falls short because the PPP parameters for a specific small molecular system (e.g. ethene or benzene) have been shown to transfer well to other systems far outside the original parametrisation regime. Explaining this success of the PPP model from a theoretical standpoint has posed a great challenge. In general, the theoretical justifications for the various approximations have been given much later, well after their ad hoc introduction and demonstrated success. One of the reasons for this delay has been that detailed ab initio calculations were computationally demanding, and only once they became feasible it was possible to mimic these approximations within the ab initio framework. Here we present the most consequential approximations invoked in the PPP model, namely π-electron treatment, zero differential overlap, and semi-empirical parameters, and discuss their theoretical justification and limitations.A theoretical validation for reducing the full electronic Hamiltonian to a valence π-electron Hamiltonian is given by the effective valence shell Hamiltonian  approach.35,60–65 Here the complete molecular electronic Hamiltonian is cast into a formally exact effective Hamiltonian which acts solely within the valence space.66 This approach can mimic the approximations made for the PPP Hamiltonian, such as the π-electron approximation, through an ab initio Hamiltonian and evaluate their merit. Within this approach, it has been validated that, although not explicitly included in the PPP Hamiltonian, σ- and π-orbital relaxation, σ–σ, π–π and σ–π-correlations are included through adjustment of parameters when accounting for correlation effects.64 Furthermore, the π-electron approximation within the ab initio effective Hamiltonian
approach.35,60–65 Here the complete molecular electronic Hamiltonian is cast into a formally exact effective Hamiltonian which acts solely within the valence space.66 This approach can mimic the approximations made for the PPP Hamiltonian, such as the π-electron approximation, through an ab initio Hamiltonian and evaluate their merit. Within this approach, it has been validated that, although not explicitly included in the PPP Hamiltonian, σ- and π-orbital relaxation, σ–σ, π–π and σ–π-correlations are included through adjustment of parameters when accounting for correlation effects.64 Furthermore, the π-electron approximation within the ab initio effective Hamiltonian  is exact and generally also holds for the semi-empirical Hamiltonian. This is true as long as complicated effective n-body interaction terms with n ≥ 3 can be neglected for the ab initio Hamiltonian, or alternatively included in an averaged fashion in the PPP Hamiltonian.60,64,67 While the π-electron approximation in general relies on the planarity of the molecule, this can be extended to include non-planar conformations through a simple cosine functional dependency with the torsional angle for the kinetic energy/hopping term.34
is exact and generally also holds for the semi-empirical Hamiltonian. This is true as long as complicated effective n-body interaction terms with n ≥ 3 can be neglected for the ab initio Hamiltonian, or alternatively included in an averaged fashion in the PPP Hamiltonian.60,64,67 While the π-electron approximation in general relies on the planarity of the molecule, this can be extended to include non-planar conformations through a simple cosine functional dependency with the torsional angle for the kinetic energy/hopping term.34
 calculations on benzene and cyclobutadiene show that in the case where correlation contributions are included correctly most two-electron resonance, hybrid, and exchange matrix integrals become very small. Furthermore, almost all three- and four-centre two-electron integrals can be considered negligible, corroborating the ZDO approximation for the PPP Hamiltonian.65 For the kinetic energy integrals, it is found that non-nearest neighbour integrals can be non-negligible.65,66
calculations on benzene and cyclobutadiene show that in the case where correlation contributions are included correctly most two-electron resonance, hybrid, and exchange matrix integrals become very small. Furthermore, almost all three- and four-centre two-electron integrals can be considered negligible, corroborating the ZDO approximation for the PPP Hamiltonian.65 For the kinetic energy integrals, it is found that non-nearest neighbour integrals can be non-negligible.65,66
 35,61–65 They showed that for
35,61–65 They showed that for  calculations of ethylene, trans-butadiene, cyclobutadiene, hexatriene and benzene, remarkable transferability of correlated integrals could be obtained.64,65 However, this transferability critically depends on the correct inclusion of correlation interaction. The importance of correlation for the transferability of the parameters had already been noted earlier, specifically for the parametrisation of electron–electron interaction.68
calculations of ethylene, trans-butadiene, cyclobutadiene, hexatriene and benzene, remarkable transferability of correlated integrals could be obtained.64,65 However, this transferability critically depends on the correct inclusion of correlation interaction. The importance of correlation for the transferability of the parameters had already been noted earlier, specifically for the parametrisation of electron–electron interaction.68
From a computational perspective, the need to include considerable electron correlation for the approximations underlying the PPP Hamiltonian to hold and thereby reach accurate results poses a significant challenge. A full configuration interaction (FCI, also called exact diagonalisation in physics) calculation will quickly become unfeasible due to its exponential scaling, even for a reduced Hilbert space such as with the PPP Hamiltonian. Even a more restricted inclusion of correlation, where excitations in the configurations of a reference wave function are considered to some order (single, double, triple excitation etc.) will become intractable for larger systems due to their generally high polynomial scaling. Still, for FCI a complete description of electron correlation within the model is reached, and thus any remaining discrepancy between experimental data and the FCI results originates from neglected effects outside the scope of the model or its parameters and can inform an updated model description. For early examples of this exact model diagonalisation approach, see for instance ref. 47,49,69–74.
4 PPP then
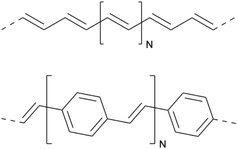
In this section, we will illustrate why and how the PPP Hamiltonian has remained relevant over so many years, even after its original intended use in small conjugated molecules was subsequently taken over by more general full ab initio methods. We focus on conjugated polymers to showcase this development. Conjugated polymers are quasi-one-dimensional systems where the PPP Hamiltonian has been widely applied with great success. Two exemplary polymers, trans-polyacetylene and poly(para-phenylene-vinylene), are shown in Fig. 3.Increased theoretical interest in polymers arose after the successful synthesis of thin polyacetylene films75 and following the discovery of conductance for doped polyacetylene,76,77 which was awarded the Nobel Prize in Chemistry in 2000. These findings opened up a large field of technical applications such as plastic field-effect transistors, electromagnetic shielding, nonlinear optical devices, photovoltaic devices and light-emitting devices,78 in particular, following the discovery of electroluminescence in phenyl-based polymers, such as poly(para-phenylene-vinylene).79
The theoretical understanding of such polymers can be challenging due to the extended size of these systems. Furthermore, electron–electron interactions are only weakly screened (due to the low dimensionality of the system) and therefore electron correlation has to be considered.78 The interaction strength is commonly characterized as weak in phenyl-based polymers such as poly(para-phenylene-vinylene) and as intermediate, in between the weak and strong electron–electron interaction limits, for polyenes such as trans-polyacetylene.78
While an ab initio treatment with correlated methods would be the preferred choice in the case of strong electron–electron interaction, this becomes very challenging due to the large system sizes one has to reach to extrapolate to the infinite system size limit.72 The simpler independent particle description with single-determinant ab initio methods such as RHF, UHF or DFT will not be sufficient for the intermediate-coupling regime. It has therefore been advantageous to revert to different model Hamiltonians, where we will discuss here specifically results with the PPP Hamiltonian. Our primary focus is the FCI approach because it unambiguously showcases the strengths and limitations of a chosen model and basis, separated from any potential approximations invoked in the method used to solve it.80 Finally, we will also discuss an approximate FCI-type solver, the density matrix renormalization group (DMRG) method, and how it benefitted uniquely from the PPP Hamiltonian in describing polymeric systems.
4.1 FCI studies
We have already seen in Section 3.2 that electron correlation has to be considered for the fundamental approximations of the PPP Hamiltonian to hold. A valid question one can ask then is how much correlation needs to be included to obtain reliable results? An early comparison between various levels of perturbation theory and FCI calculations on the electronic structure of benzene found correlation to be important, and the convergence in the perturbation series towards the FCI result to be slow in terms of excitations.69 Additional evidence for the importance of electron correlation was established for linear polyenes through experimental observation81 and subsequent theoretical confirmation.82,83Based on these findings, another key point was raised regarding the influence of the interaction strength on the necessary level of electron correlation:68 Depending on the interaction strength in a system, either of the two respective reference basis representations (MO and valence bond (VB) basis) can become inadequate to resolve polyene spectra, unless solved for FCI, where both approaches become equivalent.83 As already discussed earlier, polyenes usually fall in the intermediate interaction regime48,78 and an FCI treatment can therefore give reassurance that interactions are adequately described.
While FCI is size extensive, this property is lost if the CI expansion is truncated, as is the case with configuration interaction singles and doubles (CISD).72 There are, however, other widely used correlated methods such as coupled cluster (CC) that are size extensive and have been employed with the PPP Hamiltonian to calculate infinite-chain properties from cyclic polyenes by extrapolating from smaller chain lengths.85 For these systems, single-reference CC calculations break down, when correlation effects become sufficiently strong in larger polyenes.86 This is especially true when a mean-field solution is taken as the reference state. Extensions to the CC approach with a valence bond reference,87–91 on the other hand, have been shown to work well, also in the highly correlated limit. Another successful description of strong correlation within the coupled cluster method has been developed recently with the PPP Hamiltonian and the pair Coupled Cluster Doubles (pCCD) approach.92
The original parametrisation predated extensive CI calculations93 and was based on experimental data and either SCF or limited CI calculations (e.g., CIS).74 The standard parameters were considered to demonstrate robustness and transferability74 because they hold up well once electron correlation is accounted for. The proper inclusion of electron correlation in excited state calculations is thus preferable over reparametrisation of the model as previously undertaken for mean-field approaches.52
Later, screened parameters were proposed94 that better describe the high-energy excited states of phenyl-based polymers95 and were successfully used in oligoacenes as well.96 The screening was originally suggested as an environmental effect in the condensed phase, which reduces the effective charge of π-electrons over larger distances. Further FCI calculations on phenyl systems59 concluded, however, that the screening parameters had to be included even in the gas phase. The screening effect was thus attributed to the screening effect of the σ-electrons rather than external environment effects.59
As a concrete example, we mention symmetry-adapted FCI calculations of the anthracene molecule, essentially three fused benzene molecules along one axis, where a range of effects74 prohibited the PPP model description from reaching sub-0.1 eV accuracy. The first of the effects is symmetry-breaking, which is not captured by the symmetry-adapted basis that is utilized. Another effect is vibronic coupling or nonadiabatic coupling, which is neglected in the PPP model due to the underlying Born–Oppenheimer (BO) approximation. In the BO approximation, the motion of atomic nuclei is assumed to be decoupled from the motion of electrons and only affects the electrons parametrically. This is usually an excellent approximation, but can fail when electronic states become degenerate or nuclear motion relative to the electronic timescale cannot be neglected.97
An additional source of potential discrepancy between FCI studies and experimental data can be the relaxation of the molecular structure in the excited state from the ground state geometry. While this effect was considered in the anthracene study,74 the authors caution that for accurate relaxation energies the difference between solid-state experimental data and gas-phase molecular calculations has to be considered as a solid-state shift in energy of up to 0.2–0.3 eV.
While every single effect described here can potentially be addressed and corrected for in a model, solving the augmented model computationally becomes harder and harder. The hope then is to understand a model and its limitations confidently and exactly for smaller systems, so that the approximations can be made with reassurance in bigger systems. Researchers can differ significantly in their perception of such models, which also influences how they are employed and benchmarked. Some believe that exploring the model itself holds value, and in this context FCI calculations are particularly interesting. There are also others who view the model as simply a tool for understanding the experimental reality, thus being more interested in the reproduction of experimental results. In this case, FCI can provide the benchmark data to support the use of more approximate methods to focus on questions of experimental interest.
4.2 Beyond FCI studies
In an extensive body of work, Paldus and coworkers showed that the CCSD approach breaks down for annulenes (strongly correlated cyclic polyenes) and conceived an approximate coupled-pair method (ACPQ) that could deliver close to FCI results for these systems.86 Later, the valence bond coupled cluster approach with singly and doubly excited cluster amplitudes (VB-CCSD) was developed and tested by the same group for the PPP Hamiltonian on π-electron systems, especially for strongly correlated systems. In a severely resource-constrained compute environment, the PPP Hamiltonian offered a way to check exact solutions and furthermore allowed the exploration of the entire range of correlation effects, simply by tuning the hopping term/resonance integral.90 Together, these features meant that the PPP Hamiltonian played a significant role in the methodological development of the coupled cluster approach.111
For conjugated polyenes and more complex polymer systems, we have seen throughout this chapter that accurate treatment of correlation is essential. Furthermore, there was considerable interest in reaching much larger system sizes than what could be afforded by an FCI treatment. Consequently, the adaptation of the density matrix renormalization group method (DMRG)112 for chemistry in the context of model Hamiltonians in the 1990s marked a great opportunity to tackle conjugated systems beyond what was previously possible.48
There are several reasons why DMRG using the PPP Hamiltonian is particularly useful for the treatment of conjugated polymers. DMRG is size extensive for one dimension,113 variational, and therefore in theory systematically improvable until FCI accuracy. Furthermore, the computational scaling is reduced due to the sparsity of the PPP Hamiltonian.114 An additional advantage for DMRG is the particle–hole symmetry of the PPP Hamiltonian, originally described for the HMO theory by the Coulson–Rushbrooke theorem115 and discussed for the PPP Hamiltonian by Pariser.116 This symmetry is also known under the name of alternacy symmetry or pseudoparity and has been exploited together with spatial and spin-flip symmetries in DMRG-calculations to target high-lying excited states.114 For these reasons, unprecedented lengths of polymer chains containing 100 carbon atoms could be accurately investigated using DMRG.48 This answered long-standing questions in excited state ordering, exciton binding energies, and solved problems in linear and nonlinear spectroscopy of conjugated polymer systems. It also conclusively confirmed the usefulness of DMRG for chemistry, which was then later also extended to ab initio Hamiltonians.48,117–125
5 PPP today
In the previous section, we have shown how the relatively simple PPP model has helped to gain theoretical understanding of experimentally relevant electronic and optical properties in conjugated polymers. This process can be viewed as gaining understanding from a given structure (molecule, polymer, etc.) through electronic structure calculations and is shown in the top of Fig. 4.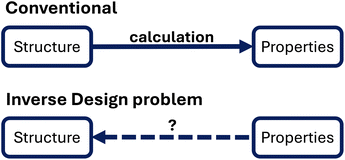 | ||
| Fig. 4 Inverse design problem, adapted from ref. 126, with permission from AIP Publishing. | ||
The results of these calculations are properties of the system of interest, such as the electronic structure, optical spectra or similar ones. These properties can be compared with experimental data and may be refined through more accurate calculations or a better model. While this process has been successful in gaining insight, it can also be relatively slow because it involves many calculations.
There is also no guarantee that the calculated properties are useful or even insightful for experimental chemists searching for new systems of interest. These molecular engineering problems could, for example, include the search for a better catalyst or more efficient photovoltaic materials. Ideally, calculations could also deliver insight for the opposite problem, as indicated in Fig. 4: from some defined properties to a new and improved structure. This is known as the inverse design problem126–128 and has been discussed extensively in the context of high-throughput screening.
One of the challenges inherent in the inverse design problem is the vastness of chemical space, by some estimates consisting of 1060 molecules.128 Often, researchers do not feel completely in the dark and believe that chemical intuition can reduce this space considerably to some of the most relevant regions. Even if this is true, the question is how human chemical intuition can be translated into actionable instructions for a computer. One strategy that has been employed is the use of design rules to significantly reduce the chemical space before the computationally intensive search using virtual screening, machine learning, and genetic algorithms.126 These design rules, or simply chemical intuition, have a long history in organic chemistry. One of the early examples is Hückel's rule, which is derived from HMO theory and predicts compounds with (4n + 2) π-electrons to be aromatic.4
The PPP Hamiltonian has been discussed in this context as a tool to derive a cheap scoring function129 and has been used as a tool to formulate and verify design rules.126,130–134 The PPP Hamiltonian has been employed in the construction of design rules, especially for its ability to treat larger system sizes than otherwise would be possible with ab initio multi-configurational methods.130 Furthermore, the particle-hole symmetry of the PPP Hamiltonian has proved useful in the determination of the energetic ordering and form of molecular orbitals in radical emitter systems, but particle-hole symmetry is broken by chemical substitution.131 Proposals include specifically tailored design rules for systems such as acenes,130 radical-based organic light-emitting diodes (OLEDs),132 magnetic molecules,133 photo-induced spin-polarisation facilitated by spin–orbit coupling134 and a more generalized framework description of design rules for the PPP Hamiltonian.126
We focus our discussion in the following sections on two relevant application fields, singlet fission and the search for molecules with an inverted singlet–triplet energy gap (InveST). These constitute two very challenging inverse design problems, and we will describe the challenges in modelling and how the PPP Hamiltonian specifically has been used to gain insight and formulate design rules.
5.1 Singlet fission
The study of singlet fission in molecules is largely motivated by the desire to develop photovoltaics with increased efficiency. The overall efficiency in solar cells is limited to around 30% (Shockley–Queisser limit135) with the assumption that one absorbed photon can yield one electron–hole pair.136 This limit could be circumvented if one photon can generate multiple electron–hole pairs. For dye-sensitized solar cells it was suggested137 that this could be achieved through the process of singlet fission, where in most discussed cases an organic chromophore in an excited singlet state transfers energy to a neighbouring ground-state chromophore or chromophores, to produce two triplet excited states138 (for a schematic illustration see also Fig. 5a). The efficiency for an ideal solar cell with this process rises to nearly 50%.137,138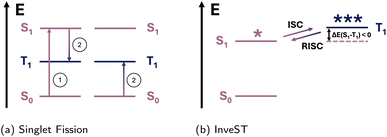 | ||
| Fig. 5 Schematic illustration of the SF process ((a), adapted from ref. 138 with permission from the American Chemical Society, copyright 2010) and the InveST mechanism ((b), adapted from ref. 144 with permission from Elsevier, copyright 2021). | ||
The inverse design problem to find the right chromophores is complicated by the fact that singlet fission appears to be highly system-specific, with different mechanisms proposed.136 The search for suitable chromophores therefore cannot be limited to finding the optimal electronic structure of the chromophores, but must eventually also include the dynamic evolution139 of the two triplet states. Furthermore, environmental effects may need to be included explicitly, as polar solvents can cause the electronic energy levels of the chromophores to fluctuate in the same order of magnitude as the original isolated chromophores' level splitting.136
The PPP Hamiltonian has been used in the context of singlet fission in different capacities, and here we can only give a small selection. One study found design rules based on PPP computed spectra that predict the spectra of acene-based molecules, including oligomers with unusual bonding geometries and heteroatom substitutions.130 Other use cases include FCI calculations to engineer chromophore properties through substitution with heteroatoms or substituents for a more efficient singlet fission. Investigated systems range from pyrene with inter-molecular singlet fission140 to diphenylpolyenes that support endoergic to isoergic singlet fission with increasing chain length.141
Another focus is on the adaption of the PPP model to include additional physical effects such as electron–phonon interactions in polyenes that affect the singlet fission process.142 The resulting Hamiltonian is called the PPP-Peierls (PPPP) model, and it accounts for the larger flexibility in polyenes compared to rigid structures such as solids or even acenes. The PPP model was also adapted to include an intermolecular interaction component between two different chromophores, where only a minimal enhancement in performance due to singlet fission was found for a pentacene–C60 solar cell.143
5.2 Inverted singlet–triplet energy gap
Another very challenging inverse design problem is the discovery of more efficient organic light-emitting diode (OLED) materials. There are several factors that limit the internal quantum efficiency (IQE), which is defined as the ratio between the number of generated photons to the number of injected electrons.145 While singlet excitons in OLEDs are fluorescent, triplet excitons are normally non-emissive and decay non-radiatively to the ground state.146 Additionally, the desired singlet excitons are only generated in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 ratio relative to the triplet excitons, due to the spin statistics of recombining charge carriers.147
3 ratio relative to the triplet excitons, due to the spin statistics of recombining charge carriers.147
The final impediment is embodied in Hund's multiplicity rule that places the first excited singlet state S1 of an organic closed-shell molecule higher in energy than the corresponding first excited triplet state T1. Defining the singlet–triplet energy gap as ΔEST = S1 − T1, molecules that obey Hund's rule therefore have a positive value for ΔEST. This indicates that energetically population transfers from the singlet to the triplet state are favoured in a process called intersystem crossing (ISC).
The focus in the development of new OLED materials has therefore been to address these limitations through molecular engineering. The efficiency in OLEDs has been improved by introducing phosphorescent emitters that make the triplet state bright as well as by reducing ΔEST, so that thermally activated delayed fluorescence (TADF), due to a reverse ISC (RISC), becomes possible.
The theoretical discovery of two stable organic molecules with an inverted singlet–triplet energy gap146,148 (InveST), where ΔEST < 0, marked an important achievement, which has significant potential to improve OLED efficiency. For InveST molecular systems, the triplet excitons avoid thermalization and are instead converted through an energetically favourable RISC to the fluorescent singlet exciton.149 A schematic illustration of the InveST mechanism is shown in Fig. 5b.
Finding molecules with InveST properties has been challenging, which is somewhat unsurprising given the dearth of examples of Hund's rule violations either experimentally or theoretically before 2019.144 There has been a large effort to theoretically predict new InveST molecules and to understand the underlying process to create useful design rules.150 Experimentally, there has also been confirmation for some InveST molecules.147,148,151–153 A great challenge in treating InveST molecules theoretically has been the inadequacy of uncorrelated excited-state methods and linear response TD-DFT to describe the InveST phenomenon.146,154
It has been shown155–157 that the inclusion of accurate electron correlation is crucial to reproduce inverted gaps. This is due to one of the characteristic features of InveST molecules: a minimal exchange integral between the frontier orbitals (HOMO and LUMO), which in turn elevates the importance of the spin-polarization,155 an otherwise smaller effect. Spin-polarization can then even reverse the sign of ΔEST. To describe spin-polarization effects, electron correlation with at least double excitations from an HF reference is necessary.154
The inverse design problem for InveST molecules is a very active research field, where theoretical efforts have taken a prominent role and are complemented by experiment.150 Here again, we will focus on just the PPP Hamiltonian, its specific benefits for the study of InveST molecules and how it has been applied so far.
Some computational challenges, such as the need for higher-order electron correlation treatment, can be more easily overcome with the PPP Hamiltonian. Furthermore, even the smallest proposed InveST systems are at the edge of what standard correlated ab initio methods can reach.144 The PPP Hamiltonian can therefore help to make larger systems accessible and reveal general trends.158 The absolute value of ΔEST can be rather small159 and is in the same order as the error of the computational methods.150 Chemical accuracy is therefore required to determine the sign of ΔEST with confidence, and a PPP FCI calculation could help identify critical effects such as environment, correlation contribution etc. A very early PPP Hamiltonian study, predating the renewed interest in InveST molecules by decades, found molecules such as propalene, pentalene and heptalene in violation of Hund's rule for D2h symmetry, but in agreement with it for a relaxed C2h symmetry.156
The PPP Hamiltonian has been discussed in the context of high-throughput screening for InveST molecules as a scoring function, to filter out the most promising InveST candidates, that can be investigated more closely by higher-level ab initio methods.129 One of the attractive features of the PPP Hamiltonian as a scoring function is the cheap and simple description relative to other approaches, while still capturing the most important physics.129 In that case, the electronic structure was solved with configuration interaction singles and perturbative double excitations for a very efficient virtual screening. The authors mention two main concerns with their findings: the inability to correctly predict oscillator strengths and restriction to π-electron transitions. It seems to be an open challenge to strike the balance between a cheap scoring function on the one hand and a sufficiently accurate description, potentially requiring more costly inclusion of correlation interactions.
For triangulene systems with the PPP Hamiltonian, it was shown that triple excitations of the HF reference are non-negligible for the description of the excited states when the transition energy is compared with FCI calculations.160 Further work by some of the same authors found that within the PPP model space, a negative ΔEST can be explained by a network of alternating electron-donor and acceptor groups in the molecular rim, rather than the triangular molecular structure itself.158
6 PPP tomorrow
As detailed above, the PPP Hamiltonian can be viewed as a “minimum viable parametrisation of conjugated chemistry”, with a long and influential history in the development of new theoretical methods. It is ‘minimal’ in that it offers a highly reduced representation of the full electronic structure problem, explicitly treating only the π-electrons, yet still providing an insightful description for many molecules of chemical or technological interest. Over the past seven decades, the PPP model was able to provide insights into extended systems that were otherwise unreachable, and it has propelled the development of novel computational methods in quantum chemistry.As the PPP model is well established nowadays and many smaller systems have been calculated accurately with existing methods, it can be used as a valuable reference to benchmark novel methods. While the PPP approach is rooted in the same molecular orbital framework as most ab initio methods, it replaces the cumbersome integral evaluation with a more lightweight parametrisation of the electronic interactions. Due to this model character, it is easy to tune the parameters and explore specific regimes directly, such as the weak and strong coupling limits, which allows for a straightforward testing of new computational approaches across different correlation regimes. There has also been substantial progress in clarifying the scope and limits of the model itself, and different parametrisation techniques have been developed to maximize the predictive power of the PPP model.
Meanwhile, quantum computing is emerging as a promising alternative to mitigate the unfavourable scaling of traditional quantum chemistry methods by leveraging quantum mechanical principles to represent and manipulate quantum states more efficiently than classical methods. While current noisy intermediate-scale quantum (NISQ)-era devices lack the error correction and scalability required for chemically accurate simulations of large systems, rapid hardware progress suggests that the first fault-tolerant quantum computations for chemistry are soon within reach.161 However, these early fault-tolerant quantum computers will offer only very limited computational resources, thus requiring reduced problem descriptions and optimal utilization of the available resources to obtain meaningful results.162,163
Here, we propose the PPP Hamiltonian as an ideal candidate for insightful calculations in such a resource-constrained compute environment, since it can be considered the minimum viable model of organic molecules that still captures the essential chemistry. The π-electron approximation of the PPP model allows for a drastic reduction of the Hilbert space size and requires significantly fewer resources compared to an ab initio calculation, even when considering only a minimal basis set. To illustrate this on a simple example, we compare an ab initio treatment of benzene with the corresponding PPP model Hamiltonian. For the ab initio description, a minimal STO-3G basis set for the 6 carbon and 6 hydrogen atoms results in a total of 72 spin orbitals, while the PPP Hamiltonian only requires two spin orbitals per carbon atom, thus describing benzene with a total of 12 spin orbitals. For quantum computing applications, the reduction in the number of spin orbitals translates directly into an equivalent reduction of the number of qubits needed to represent the system. This makes PPP a prime candidate for calculations on early quantum computers that possess only a limited number of (logical) qubits.
Another advantage of the PPP model can be found in the ZDO approximation, which leads to a very sparse Hamiltonian matrix. In the atomic orbital representation that we have used in eqn (4) for the PPP Hamiltonian, all terms are diagonal, except for the nearest-neighbour hopping terms tij. The same is true for the real-space valence-bond description of the PPP Hamiltonian,164 whereas the molecular orbital representation of the PPP Hamiltonian is less sparse and has more terms.158,165 The AO representation of the PPP model will have a formal scaling of O(N2) in the number of Hamiltonian terms without any further screening, where N is the number of orbitals. In contrast, the MO representation of the PPP Hamiltonian165 and general ab initio Hamiltonians166 formally scale as O(N4). The actual number of terms for the MO PPP Hamiltonian will still be greatly lower than an ab initio Hamiltonian due to the neglect of three- and four-site two-electron integrals in the ZDO approximation. For quantum computing applications, sparser Hamiltonians require fewer terms to be encoded on the device, thereby reducing the number of gate operations needed to represent a given Hamiltonian in a quantum circuit. Another potential advantage of the PPP model in that context is that many interaction parameters will be identical, especially for idealized molecular structures, such that these terms can be grouped and implemented more efficiently.167–169 The advantages of the PPP model for quantum computing are summarized schematically in Fig. 6.
 | ||
| Fig. 6 Scheme illustrating the mutual benefits of employing the PPP model in quantum computing applications for chemistry. | ||
While the PPP Hamiltonian is ideally suited as an early test bed for developing quantum computing applications, the reverse is also true, as illustrated in Fig. 6. The PPP model, conceived in a resource-constrained environment and therefore often used in more approximate treatments of systems, benefits greatly when more correlation contributions can be considered, ideally solving the system exactly. This is particularly true when calculating excited-state properties, as these may necessitate a multi-reference treatment of the wave function while capturing multiple, potentially close-by states. Furthermore, some approximations discussed before for the PPP model are only truly valid when considerable electron correlation is included. The pursuit of exact solutions has been prohibitive beyond modest system sizes within the classical computing paradigm due to the exponential scaling of the underlying problem.
Here, the quantum computing equivalent of an FCI calculation, the quantum phase estimation algorithm (QPE), offers a more favourable polynomial scaling compared to the exponentially scaling classical FCI computation. The minimal description of the π-electron PPP Hamiltonian provides an additional advantage to reach larger systems efficiently with limited quantum resources, especially in the calculation of spectroscopic properties that are classically unfeasible. The combined quantum semi-empirical approach (QPE + PPP) will also be more efficient than a minimal quantum-ab initio approach (QPE + STO-3G, as discussed before for the benzene molecule). Strong correlation effects will be better and more efficiently described in the minimal semi-empirical calculation than the minimal ab initio calculation, due to their implicit inclusion in the parametrisation.35,64 Moreover, the included correlation interaction in the QPE calculation will ensure that the approximations of the PPP Hamiltonian (π-electron, ZDO, transferability of the semi-empirical parameters) are truly valid analogous to the classical FCI studies.
In the same spirit as in the classical case, the PPP model need not only be applied in FCI-type calculations such as QPE, but can also serve as a benchmark model to validate more approximate quantum algorithms. Similarly to its use in the development of approximate correlated classical methods, particularly for CC-variants and DMRG applications in chemistry, the PPP Hamiltonian could be used as a resource-efficient test system for early fault-tolerant algorithms that only capture a limited degree of correlation, such as quantum subspace methods, filtering techniques, and statistical approaches.170
For these reasons, we conjecture that the PPP Hamiltonian is ideally suited for early fault-tolerant quantum computing applications and vice versa. Within this framework, different avenues and possibly new tradeoffs might present themselves which were traditionally not considered. As an example, we return to the previously mentioned choice of representing the PPP model in three different bases (AO/MO/VB). It is not clear if the MO representation, which is predominant in classical applications, is also the optimal choice for a quantum computer. For the PPP Hamiltonian, the AO representation offers the most compact representation and also the least resource demands. The traditional drawback of the AO representation against the two other approaches, MO and VB, has been the difficulty to truncate this basis. Therefore, AO representations of the PPP Hamiltonian commonly necessitate an FCI calculation, something that comes naturally with the QPE algorithm.
The largest classical FCI calculation to date was for the propane molecule, C3H8, with a minimal STO-3G basis which required the description of 26 electrons in 23 spatial orbitals (equivalent to 46 spin orbitals).171 This limitation of classical FCI to a double-digit number of spin orbitals highlights the potential utility of quantum computers already with a comparable number of logical qubits, particularly when employing model Hamiltonians such as the PPP model. By pairing QPE with PPP, one can achieve a chemically meaningful yet highly efficient description in terms of qubit resources which requires only very limited circuit depth thanks to the sparsity of the PPP Hamiltonian. Hence, this combination is a particularly promising candidate for early impactful applications of fault-tolerant quantum computing in chemistry.
Author contributions
MDF, NG, GCS: conceptualization, writing – original draft, writing – review and editing.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work is supported by the Novo Nordisk Foundation, Grant number NNF22SA0081175, NNF Quantum Computing Programme. This work is partly funded by Innovation Fund Denmark (IFD) under File no. 4356-00009B. We thank Matthew Teynor for helpful comments on the manuscript.Notes and references
- P. A. M. Dirac, Proc. R. Soc. Lond. A, 1929, 123, 714–733 CAS.
- E. Hückel, Z. Phys., 1931, 70, 204–286 CrossRef.
- E. Hückel, Z. Phys., 1931, 72, 310–337 CrossRef.
- E. Hückel, Z. Phys., 1932, 76, 628–648 CrossRef.
- E. Hückel, Z. Phys., 1933, 83, 632–668 Search PubMed.
- J. E. Lennard-Jones and C. A. Coulson, Trans. Faraday Soc., 1939, 35, 811–823 RSC.
- C. A. Coulson and H. Longuet-Higgins, Proc. R. Soc. Lond. A, 1947, 191, 39–60 CAS.
- H. C. Longuet-Higgins and G. W. Wheland, Annu. Rev. Phys. Chem., 1950, 1, 133–150 CrossRef CAS.
- H. C. Longuet-Higgins, Nature, 1950, 166, 139–141 CrossRef CAS PubMed.
- H. C. Longuet-Higgins, J. Chem. Phys., 1950, 18, 283–291 CrossRef CAS.
- H. C. Longuet-Higgins, J. Chem. Phys., 1950, 18, 275–282 CrossRef CAS.
- H. C. Longuet-Higgins, J. Chem. Phys., 1950, 18, 265–274 CrossRef CAS.
- H. Longuet-Higgins and J. Pople, Proc. Phys. Soc. A, 1955, 68, 591–600 CrossRef.
- N. Trinajstić, Int. J. Quantum Chem., 1977, 12, 469–477 CrossRef.
- W. Kutzelnigg, J. Comput. Chem., 2007, 28, 25–34 Search PubMed.
- M. C. Zerner, Reviews in Computational Chemistry, Wiley, 1st edn, 1991, vol. 2, pp. 313–365 Search PubMed.
- R. S. Mulliken, J. Chim. Phys., 1949, 46, 497–542 CrossRef CAS.
- K. Jug, Int. J. Quantum Chem., 1990, 37, 403–414 CrossRef CAS.
- J. A. Pople, J. Phys. Chem., 1957, 61, 6–10 CrossRef CAS.
- S. Boys, Proc. R. Soc. Lond. A, 1950, 200, 542–554 CAS.
- R. G. Parr, The Quantum Theory of Molecular Electronic Structure: A Lecture-Note and Reprint Volume, Benjamin, Reading, Mass, 3rd edn, 1972 Search PubMed.
- L. C. Allen and A. M. Karo, Rev. Mod. Phys., 1960, 32, 275–285 Search PubMed.
- M. J. S. Dewar, The Molecular Orbital Theory of Organic Chemistry, McGraw-Hill, New York, 1969 Search PubMed.
- R. Pariser and R. G. Parr, J. Chem. Phys., 1953, 21, 767–776 Search PubMed.
- R. Pariser and R. G. Parr, J. Chem. Phys., 1953, 21, 466–471 CrossRef CAS.
- J. A. Pople, Trans. Faraday Soc., 1953, 49, 1375 RSC.
- J. A. Pople, Proc. Phys. Soc. A, 1955, 68, 81–89 Search PubMed.
- W. Thiel, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2014, 4, 145–157 CAS.
- J. A. Pople, D. P. Santry and G. A. Segal, J. Chem. Phys., 1965, 43, S129–S135 Search PubMed.
- G. Klopman and R. C. Evans, Semiempirical Methods of Electronic Structure Calculation, Springer US, Boston, MA, 1977, pp. 29–67 Search PubMed.
- J. J. P. Stewart, Reviews in Computational Chemistry, Wiley, 1st edn, 1990, vol. 1, pp. 45–81 Search PubMed.
- W. Thiel, Tetrahedron, 1988, 44, 7393–7408 CrossRef CAS.
- W. Thiel, Theory and Applications of Computational Chemistry, Elsevier, 2005, pp. 559–580 Search PubMed.
- J. N. Murrell and A. J. Harget, Semi-Empirical Self-Consistent-Field Molecular Orbital Theory of Molecules, Wiley-Interscience, London, New York, 1972 Search PubMed.
- G. Segal, Semiempirical Methods of Electronic Structure Calculation: Part A: Techniques, Springer, New York, NY, 1977 Search PubMed.
- J. Shen, L. E. Walker, K. Ma, J. D. Green, H. Bronstein, K. T. Butler and T. J. H. Hele, Chem. Sci., 2025, 16, 17356–17368 RSC.
- K. Ohno, Theoret. Chim. Acta, 1964, 2, 219–227 CrossRef CAS.
- J. Koutecký, J. Chem. Phys., 1967, 47, 1501–1511 CrossRef.
- J. Hinze and D. L. Beveridge, J. Am. Chem. Soc., 1971, 93, 3107–3114 CrossRef CAS.
- F. A. Van-Catledge, J. Org. Chem., 1980, 45, 4801–4802 CrossRef CAS.
- T. Clark, J. Mol. Struct.:THEOCHEM, 2000, 530, 1–10 CrossRef CAS.
- J. A. Pople, J. Am. Chem. Soc., 1975, 97, 5306–5308 CrossRef CAS.
- W. J. Hehre, J. Am. Chem. Soc., 1975, 97, 5308–5310 CrossRef CAS.
- M. J. S. Dewar, J. Am. Chem. Soc., 1975, 97, 6591 CrossRef CAS.
- M. J. S. Dewar, J. Phys. Chem., 1985, 89, 2145–2150 CrossRef CAS.
- W. Kutzelnigg, Einführung in die Theoretische Chemie, Wiley-VCH Verlag, Weinheim, 2002 Search PubMed.
- S. Ramasesha and Z. G. Soos, J. Chem. Phys., 1984, 80, 3278–3287 Search PubMed.
- H. Ma, U. Schollwöck and Z. Shuai, Density Matrix Renormalization Group (DMRG)-Based Approaches in Computational Chemistry, Elsevier, Amsterdam, 2022 Search PubMed.
- S. Ramasesha and P. K. Das, Chem. Phys., 1990, 145, 343–353 CrossRef CAS.
- L. Berencei, A. Grout-Smith, J. E. Poole and W. Barford, J. Chem. Phys., 2019, 151, 064120 CrossRef.
- G. Chiappe, E. Louis, E. San-Fabián and J. A. Vergés, J. Phys.: Condens. Matter, 2015, 27, 463001 CrossRef CAS PubMed.
- T. A. Skotheim, Electroresponsive Molecular and Polymeric Systemsvol. 1, CRC Press, Boca Raton, 1st edn, 1988 Search PubMed.
- S. Ramasesha, S. K. Pati, Z. Shuai and J. Brédas, Advances in Quantum Chemistry, Elsevier, 2000, vol. 38, pp. 121–215 Search PubMed.
- K. Gundra and A. Shukla, Topological Modelling of Nanostructures and Extended Systems, Springer Netherlands, Dordrecht, 2013, pp. 199–227 Search PubMed.
- S. Dallakyan, M. Chandross and S. Mazumdar, Phys. Rev. B, 2003, 68, 075204 CrossRef.
- Z. Shuai, S. K. Pati, W. P. Su, J. L. Brédas and S. Ramasesha, Phys. Rev. B, 1997, 55, 15368–15371 CrossRef CAS.
- Z. Shuai, J. L. Brédas, S. K. Pati and S. Ramasesha, Phys. Rev. B, 1998, 58, 15329–15332 CrossRef CAS.
- N. Mataga, K. Nishimoto and Z. Für, Phys. Chem., 1957, 13, 140–157 CAS.
- C. W. M. Castleton and W. Barford, J. Chem. Phys., 2002, 117, 3570–3582 Search PubMed.
- B. H. Brandow, Int. J. Quantum Chem., 1979, 15, 207–242 CrossRef CAS.
- C. H. Martin, R. L. Graham and K. F. Freed, J. Chem. Phys., 1993, 99, 7833–7844 CrossRef CAS.
- C. H. Martin and K. F. Freed, J. Chem. Phys., 1994, 101, 5929–5941 CrossRef CAS.
- C. H. Martin and K. F. Freed, J. Chem. Phys., 1994, 101, 4011–4027 Search PubMed.
- C. H. Martin and K. F. Freed, J. Chem. Phys., 1994, 100, 7454–7470 CrossRef CAS.
- C. H. Martin and K. F. Freed, J. Phys. Chem., 1995, 99, 2701–2716 CrossRef CAS.
- C. H. Martin and K. F. Freed, J. Chem. Phys., 1996, 105, 1437–1450 CrossRef CAS.
- B. H. Brandow, Quantum Theory of Polymers, Springer Netherlands, Dordrecht, 1978, pp. 137–167 Search PubMed.
- K. Schulten, I. Ohmine and M. Karplus, J. Chem. Phys., 1976, 64, 4422–4441 CrossRef CAS.
- P. B. Visscher and L. M. Falicov, J. Chem. Phys., 1970, 52, 4217–4223 CrossRef CAS.
- I. R. Ducasse, T. E. Miller and Z. G. Soos, J. Chem. Phys., 1982, 76, 4094–4104 CrossRef CAS.
- Z. G. Soos and S. Ramasesha, J. Chem. Phys., 1989, 90, 1067–1076 Search PubMed.
- S. Ramasesha, I. Albert and B. Sinha, Mol. Phys., 1991, 72, 537–547 CrossRef CAS.
- I. D. L. Albert, D. Pugh, J. O. Morley and S. Ramasesha, J. Phys. Chem., 1992, 96, 10160–10165 Search PubMed.
- S. Ramasesha, D. S. Galvao and Z. G. Soos, J. Phys. Chem., 1993, 97, 2823–2829 Search PubMed.
- T. Ito, H. Shirakawa and S. Ikeda, J. Polym. Sci. Polym. Chem. Ed., 1974, 12, 11–20 CrossRef CAS.
- H. Shirakawa, E. J. Louis, A. G. MacDiarmid, C. K. Chiang and A. J. Heeger, J. Chem. Soc., Chem. Commun., 1977, 578 Search PubMed.
- C. K. Chiang, C. R. Fincher, Y. W. Park, A. J. Heeger, H. Shirakawa, E. J. Louis, S. C. Gau and A. G. MacDiarmid, Phys. Rev. Lett., 1977, 39, 1098–1101 CrossRef CAS.
- W. Barford, Electronic and Optical Properties of Conjugated Polymers, Oxford university press, Oxford, 2nd edn, 2013 Search PubMed.
- J. H. Burroughes, D. D. C. Bradley, A. R. Brown, R. N. Marks, K. Mackay, R. H. Friend, P. L. Burns and A. B. Holmes, Nature, 1990, 347, 539–541 Search PubMed.
- D. Baeriswyl, D. K. Campbell and S. Mazumdar, Conjugated Conducting Polymers, Springer Berlin Heidelberg, Berlin, Heidelberg, 1992, vol. 102, pp. 7–133 Search PubMed.
- B. Hudson and B. Kohler, Chem. Phys. Lett., 1972, 14, 299–304 Search PubMed.
- K. Schulten and M. Karplus, Chem. Phys. Lett., 1972, 14, 305–309 CrossRef CAS.
- B. S. Hudson, B. E. Kohler and K. Schulten, Excited States, Elsevier, 1982, vol. 6, pp. 1–95 Search PubMed.
- M. Timár, G. Barcza, F. Gebhard, L. Veis and Ö. Legeza, Phys. Chem. Chem. Phys., 2016, 18, 18835–18845 Search PubMed.
- J. Paldus, M. Takahashi and R. W. H. Cho, Phys. Rev. B, 1984, 30, 4267–4291 CrossRef CAS.
- R. Podeszwa, S. A. Kucharski and L. Z. Stolarczyk, J. Chem. Phys., 2002, 116, 480–493 CrossRef CAS.
- J. Paldus and X. Li, Isr. J. Chem., 1991, 31, 351–362 Search PubMed.
- J. Paldus and J. Planelles, Theoret. Chim. Acta, 1994, 89, 13–31 Search PubMed.
- J. Planelles, J. Paldus and X. Li, Theoret. Chim. Acta, 1994, 89, 33–57 Search PubMed.
- J. Planelles, J. Paldus and X. Li, Theoret. Chim. Acta, 1994, 89, 59–76 Search PubMed.
- J. Paldus and X. Li, Pauling's Legacy: Modern Modelling of the Chemical Bond, Elsevier, 1999, vol. 6, pp. 481–501 Search PubMed.
- Z. Karimi, S. Ahmadkhani, K. Boguslawski and P. Tecmer, Efficient and Reliable Modeling of Large $π$-Electron Systems with the Pariser–Parr–Pople Hamiltonian and pCCD-based Methods, 2025 Search PubMed.
- Z. Soos and S. Ramasesha, Chem. Phys. Lett., 1983, 101, 34–37 Search PubMed.
- M. Chandross and S. Mazumdar, Phys. Rev. B, 1997, 55, 1497–1504 Search PubMed.
- A. Shukla, H. Ghosh and S. Mazumdar, Phys. Rev. B, 2003, 67, 245203 CrossRef.
- P. Sony and A. Shukla, Phys. Rev. B, 2007, 75, 155208 Search PubMed.
- W. Barford, R. J. Bursill and M. Y. Lavrentiev, Phys. Rev. B, 2002, 65, 075107 CrossRef.
- V. Chuiko, A. D. S. Richards, G. Sánchez-Díaz, M. Martínez-González, W. Sanchez, G. B. Da Rosa, M. Richer, Y. Zhao, W. Adams, P. A. Johnson, F. Heidar-Zadeh and P. W. Ayers, J. Chem. Phys., 2024, 161, 132503 CrossRef CAS PubMed.
- J. Čížek, J. Paldus and L. Šroubková, Int. J. Quantum Chem., 1969, 3, 149–167 Search PubMed.
- J. Paldus, J. Čižek and I. HubaČ, Int. J. Quantum Chem., 1974, 8, 293–303 Search PubMed.
- J. Paldus and M. J. Boyle, Int. J. Quantum Chem., 1982, 22, 1281–1305 CrossRef CAS.
- M. Takahashi, J. Paldus and J. Čížek, Int. J. Quantum Chem., 1983, 24, 707–727 Search PubMed.
- J. Paldus, M. Takahashi and B. W. H. Cho, Int. J. Quantum Chem., 1984, 26, 237–244 Search PubMed.
- M. Takahashi and J. Paldus, Phys. Rev. B, 1985, 31, 5121–5142 Search PubMed.
- P. Piecuch and J. Paldus, Theoret. Chim. Acta, 1990, 78, 65–128 CrossRef CAS.
- P. Piecuch, S. Zarrabian, J. Paldus and J. Čižek, Phys. Rev. A, 1990, 42, 5155–5167 Search PubMed.
- P. Piecuch, S. Zarrabian, J. Paldus and J. Čížek, Phys. Rev. B, 1990, 42, 3351–3379 CrossRef PubMed.
- P. Piecuch and J. Paldus, Int. J. Quantum Chem., 1991, 40, 9–34 CrossRef.
- J. Paldus and P. Piecuch, Int. J. Quantum Chem., 1992, 42, 135–164 CrossRef CAS.
- P. Piecuch, J. Čížek and J. Paldus, Int. J. Quantum Chem., 1992, 42, 165–191 CrossRef CAS.
- J. Paldus, Theory and Applications of Computational Chemistry, Elsevier, 2005, pp. 115–147 Search PubMed.
- S. R. White, Phys. Rev. Lett., 1992, 69, 2863–2866 CrossRef PubMed.
- S. Weatherly and T. Van Voorhis, J. Chem. Theory Comput., 2025, 21, 7818–7829 Search PubMed.
- R. J. Bursill and W. Barford, J. Chem. Phys., 2009, 130, 234302 Search PubMed.
- C. A. Coulson and G. S. Rushbrooke, Math. Proc. Camb. Phil. Soc., 1940, 36, 193–200 CrossRef CAS.
- R. Pariser, J. Chem. Phys., 1956, 24, 250–268 Search PubMed.
- G. K.-L. Chan, J. Chem. Phys., 2004, 120, 3172–3178 Search PubMed.
- Ö. Legeza, R. Noack, J. Sólyom and L. Tincani, Computational Many-Particle Physics, Springer Berlin Heidelberg, Berlin, Heidelberg, 2008, vol. 739, pp. 653–664 Search PubMed.
- K. H. Marti, M. Reiher and Z. Für, Phys. Chem., 2010, 224, 583–599 CAS.
- G. K.-L. Chan and S. Sharma, Annu. Rev. Phys. Chem., 2011, 62, 465–481 Search PubMed.
- U. Schollwöck, Ann. Phys., 2011, 326, 96–192 Search PubMed.
- Y. Kurashige, Mol. Phys., 2014, 112, 1485–1494 CrossRef CAS.
- S. Wouters and D. Van Neck, Eur. Phys. J. D, 2014, 68, 272 Search PubMed.
- T. Yanai, Y. Kurashige, W. Mizukami, J. Chalupský, T. N. Lan and M. Saitow, Int. J. Quantum Chem., 2015, 115, 283–299 Search PubMed.
- A. Baiardi and M. Reiher, J. Chem. Phys., 2020, 152, 040903 CrossRef CAS PubMed.
- J. D. Green, E. G. Fuemmeler and T. J. H. Hele, J. Chem. Phys., 2022, 156, 180901 Search PubMed.
- B. Sanchez-Lengeling and A. Aspuru-Guzik, Science, 2018, 361, 360–365 CrossRef CAS PubMed.
- S. Bennett, A. Tarzia, M. A. Zwijnenburg and K. E. Jelfs, Machine Learning in Chemistry, The Royal Society of Chemistry, 2020, pp. 280–310 Search PubMed.
- K. Jorner, R. Pollice, C. Lavigne and A. Aspuru-Guzik, J. Phys. Chem. A, 2024, 128, 2445–2456 CrossRef CAS PubMed.
- T. J. H. Hele, E. G. Fuemmeler, S. N. Sanders, E. Kumarasamy, M. Y. Sfeir, L. M. Campos and N. Ananth, J. Phys. Chem. A, 2019, 123, 2527–2536 CrossRef CAS PubMed.
- M. Dubbini, F. Bonvini, L. Savi and F. Di Maiolo, J. Phys. Chem. C, 2024, 128, 18158–18169 Search PubMed.
- J. D. Green and T. J. H. Hele, J. Chem. Phys., 2024, 160, 164110 CrossRef CAS PubMed.
- A. Valentim, G. A. Bocan, J. D. Fuhr, D. J. García, G. Giri, M. Kumar and S. Ramasesha, Phys. Chem. Chem. Phys., 2020, 22, 5882–5892 RSC.
- L. Savi, M. T. Barreca, M. Bedogni and F. Di Maiolo, J. Chem. Theory Comput., 2025, 5c01571 CrossRef PubMed.
- W. Shockley and H. J. Queisser, J. Appl. Phys., 1961, 32, 510–519 CrossRef CAS.
- P. E. Teichen and J. D. Eaves, J. Phys. Chem. B, 2012, 116, 11473–11481 Search PubMed.
- M. C. Hanna and A. J. Nozik, J. Appl. Phys., 2006, 100, 074510 CrossRef.
- M. B. Smith and J. Michl, Chem. Rev., 2010, 110, 6891–6936 CrossRef CAS PubMed.
- D. Casanova, Chem. Rev., 2018, 118, 7164–7207 CrossRef CAS PubMed.
- R. Patra and M. Das, J. Phys. Chem. A, 2024, 128, 7375–7383 CrossRef CAS PubMed.
- N. Karmakar and M. Das, Comput. Theor. Chem., 2022, 1217, 113918 CrossRef CAS.
- D. J. Valentine, D. Manawadu and W. Barford, Phys. Rev. B, 2020, 102, 125107 CrossRef CAS.
- K. Aryanpour, J. A. Muñoz and S. Mazumdar, J. Phys. Chem. C, 2013, 117, 4971–4979 CrossRef CAS.
- R. Pollice, P. Friederich, C. Lavigne, G. D. P. Gomes and A. Aspuru-Guzik, Matter, 2021, 4, 1654–1682 CrossRef CAS.
- F. Wang, X.-K. Liu and F. Gao, Advanced Nanomaterials for Solar Cells and Light Emitting Diodes, Elsevier, 2019, pp. 1–35 Search PubMed.
- P. de Silva, J. Phys. Chem. Lett., 2019, 10, 5674–5679 CrossRef CAS PubMed.
- N. Aizawa, Y.-J. Pu, Y. Harabuchi, A. Nihonyanagi, R. Ibuka, H. Inuzuka, B. Dhara, Y. Koyama, K.-i. Nakayama, S. Maeda, F. Araoka and D. Miyajima, Nature, 2022, 609, 502–506 CrossRef CAS PubMed.
- J. Ehrmaier, E. J. Rabe, S. R. Pristash, K. L. Corp, C. W. Schlenker, A. L. Sobolewski and W. Domcke, J. Phys. Chem. A, 2019, 123, 8099–8108 CrossRef CAS PubMed.
- M. Anwer and S. Yin, Org. Electron., 2025, 144, 107282 CrossRef CAS.
- Á. J. Pérez-Jiménez, Y. Olivier and J. C. Sancho-García, Adv. Opt. Mater., 2025, 13, 2403199 CrossRef.
- K. D. Wilson, W. H. Styers, S. A. Wood, R. C. Woods, R. J. McMahon, Z. Liu, Y. Yang and E. Garand, J. Am. Chem. Soc., 2024, 146, 15688–15692 Search PubMed.
- D. Blasco, R. T. Nasibullin, R. R. Valiev, M. Monge, J. M. López-de-Luzuriaga and D. Sundholm, Phys. Chem. Chem. Phys., 2024, 26, 5922–5931 RSC.
- Y. Kusakabe, K. Shizu, H. Tanaka, K. Tanaka and H. Kaji, Appl. Phys. Express, 2024, 17, 061001 CrossRef CAS.
- A. L. Sobolewski and W. Domcke, J. Phys. Chem. Lett., 2021, 12, 6852–6860 CrossRef CAS PubMed.
- H. Kollmar and V. Staemmler, Theoret. Chim. Acta, 1978, 48, 223–239 CrossRef CAS.
- S. Koseki, T. Nakajima and A. Toyota, Can. J. Chem., 1985, 63, 1572–1579 Search PubMed.
- W. T. Borden, H. Iwamura and J. A. Berson, Acc. Chem. Res., 1994, 27, 109–116 Search PubMed.
- M. Bedogni and F. Di Maiolo, J. Chem. Theory Comput., 2024, 20, 8634–8643 Search PubMed.
- A. Dreuw and M. Hoffmann, Front. Chem., 2023, 11, 1239604 Search PubMed.
- M. Bedogni, D. Giavazzi, F. Di Maiolo and A. Painelli, J. Chem. Theory Comput., 2024, 20, 902–913 CrossRef CAS PubMed.
- R. Acharya, D. A. Abanin, L. Aghababaie-Beni, I. Aleiner, T. I. Andersen, M. Ansmann, F. Arute, K. Arya, A. Asfaw, N. Astrakhantsev, J. Atalaya, R. Babbush, D. Bacon, B. Ballard, J. C. Bardin, J. Bausch, A. Bengtsson, A. Bilmes, S. Blackwell, S. Boixo, G. Bortoli, A. Bourassa, J. Bovaird, L. Brill, M. Broughton, D. A. Browne, B. Buchea, B. B. Buckley, D. A. Buell, T. Burger, B. Burkett, N. Bushnell, A. Cabrera, J. Campero, H.-S. Chang, Y. Chen, Z. Chen, B. Chiaro, D. Chik, C. Chou, J. Claes, A. Y. Cleland, J. Cogan, R. Collins, P. Conner, W. Courtney, A. L. Crook, B. Curtin, S. Das, A. Davies, L. De Lorenzo, D. M. Debroy, S. Demura, M. Devoret, A. Di Paolo, P. Donohoe, I. Drozdov, A. Dunsworth, C. Earle, T. Edlich, A. Eickbusch, A. M. Elbag, M. Elzouka, C. Erickson, L. Faoro, E. Farhi, V. S. Ferreira, L. F. Burgos, E. Forati, A. G. Fowler, B. Foxen, S. Ganjam, G. Garcia, R. Gasca, É. Genois, W. Giang, C. Gidney, D. Gilboa, R. Gosula, A. G. Dau, D. Graumann, A. Greene, J. A. Gross, S. Habegger, J. Hall, M. C. Hamilton, M. Hansen, M. P. Harrigan, S. D. Harrington, F. J. H. Heras, S. Heslin, P. Heu, O. Higgott, G. Hill, J. Hilton, G. Holland, S. Hong, H.-Y. Huang, A. Huff, W. J. Huggins, L. B. Ioffe, S. V. Isakov, J. Iveland, E. Jeffrey, Z. Jiang, C. Jones, S. Jordan, C. Joshi, P. Juhas, D. Kafri, H. Kang, A. H. Karamlou, K. Kechedzhi, J. Kelly, T. Khaire, T. Khattar, M. Khezri, S. Kim, P. V. Klimov, A. R. Klots, B. Kobrin, P. Kohli, A. N. Korotkov, F. Kostritsa, R. Kothari, B. Kozlovskii, J. M. Kreikebaum, V. D. Kurilovich, N. Lacroix, D. Landhuis, T. Lange-Dei, B. W. Langley, P. Laptev, K.-M. Lau, L. Le Guevel, J. Ledford, J. Lee, K. Lee, Y. D. Lensky, S. Leon, B. J. Lester, W. Y. Li, Y. Li, A. T. Lill, W. Liu, W. P. Livingston, A. Locharla, E. Lucero, D. Lundahl, A. Lunt, S. Madhuk, F. D. Malone, A. Maloney, S. Mandrà, J. Manyika, L. S. Martin, O. Martin, S. Martin, C. Maxfield, J. R. McClean, M. McEwen, S. Meeks, A. Megrant, X. Mi, K. C. Miao, A. Mieszala, R. Molavi, S. Molina, S. Montazeri, A. Morvan, R. Movassagh, W. Mruczkiewicz, O. Naaman, M. Neeley, C. Neill, A. Nersisyan, H. Neven, M. Newman, J. H. Ng, A. Nguyen, M. Nguyen, C.-H. Ni, M. Y. Niu, T. E. O'Brien, W. D. Oliver, A. Opremcak, K. Ottosson, A. Petukhov, A. Pizzuto, J. Platt, R. Potter, O. Pritchard, L. P. Pryadko, C. Quintana, G. Ramachandran, M. J. Reagor, J. Redding, D. M. Rhodes, G. Roberts, E. Rosenberg, E. Rosenfeld, P. Roushan, N. C. Rubin, N. Saei, D. Sank, K. Sankaragomathi, K. J. Satzinger, H. F. Schurkus, C. Schuster, A. W. Senior, M. J. Shearn, A. Shorter, N. Shutty, V. Shvarts, S. Singh, V. Sivak, J. Skruzny, S. Small, V. Smelyanskiy, W. C. Smith, R. D. Somma, S. Springer, G. Sterling, D. Strain, J. Suchard, A. Szasz, A. Sztein, D. Thor, A. Torres, M. M. Torunbalci, A. Vaishnav, J. Vargas, S. Vdovichev, G. Vidal, B. Villalonga, C. V. Heidweiller, S. Waltman, S. X. Wang, B. Ware, K. Weber, T. Weidel, T. White, K. Wong, B. W. K. Woo, C. Xing, Z. J. Yao, P. Yeh, B. Ying, J. Yoo, N. Yosri, G. Young, A. Zalcman, Y. Zhang, N. Zhu and N. Zobrist, Google Quantum AI and Collaborators, Nature, 2025, 638, 920–926 Search PubMed.
- Y. Alexeev, V. S. Batista, N. Bauman, L. Bertels, D. Claudino, R. Dutta, L. Gagliardi, S. Godwin, N. Govind, M. Head-Gordon, M. Hermes, K. Kowalski, A. Li, C. Liu, J. Liu, P. Liu, J. M. Garcia-Lustra, D. Mejia-Rodriguez, K. Mueller, M. Otten, B. Peng, M. Raugus, M. Reiher, P. Rigor, W. Shaw, M. van Schilfgaarde, T. Vegge, Y. Zhang, M. Zheng and L. Zhu, A Perspective on Quantum Computing Applications in Quantum Chemistry Using 25–100 Logical Qubits, 2025 Search PubMed.
- Y. Yoshida, N. Takemori and W. Mizukami, J. Chem. Phys., 2024, 161, 084303 Search PubMed.
- Z. G. Soos and S. Ramasesha, Phys. Rev. B, 1984, 29, 5410–5422 Search PubMed.
- S. Mukhopadhyay, B. J. Topham, Z. G. Soos and S. Ramasesha, J. Phys. Chem. A, 2008, 112, 7271–7279 CrossRef CAS PubMed.
- T. Helgaker, P. Jørgensen and J. Olsen, Molecular Electronic-Structure Theory, Wiley, Chichester New York Weinheim, Repr edn, 2004 Search PubMed.
- A. J. Bay-Smidt, F. R. Klausen, C. Sünderhauf, R. Izsák, G. C. Solomon and N. S. Blunt, PRX Quantum, 2025, 6, 030348 Search PubMed.
- C. Gidney, Quantum, 2018, 2, 74 CrossRef.
- I. D. Kivlichan, C. Gidney, D. W. Berry, N. Wiebe, J. McClean, W. Sun, Z. Jiang, N. Rubin, A. Fowler, A. Aspuru-Guzik, H. Neven and R. Babbush, Quantum, 2020, 4, 296 Search PubMed.
- A. Katabarwa, K. Gratsea, A. Caesura and P. D. Johnson, PRX Quantum, 2024, 5, 020101 CrossRef.
- H. Gao, S. Imamura, A. Kasagi and E. Yoshida, J. Chem. Theory Comput., 2024, 20, 1185–1192 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |