 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Towards an understanding of photoluminescence in lead-free Cs2AgxNa1−xBiyIn1−yCl6 double perovskites by machine learning prediction from density functional theory ground state properties
Marina S.
Günthert
 *ab,
Larry
Lüer
*ab,
Larry
Lüer
 a,
Oleksandr
Stroyuk
a,
Oleksandr
Stroyuk
 c,
Oleksandra
Raievska
c,
Oleksandra
Raievska
 c,
Christian
Kupfer
c,
Christian
Kupfer
 ac,
Andres
Osvet
ac,
Andres
Osvet
 a,
Bernd
Meyer
a,
Bernd
Meyer
 b and
Christoph J.
Brabec
b and
Christoph J.
Brabec
 acd
acd
aFriedrich-Alexander-Universität Erlangen-Nürnberg, Materials for Electronics and Energy Technology (i-MEET), 91058 Erlangen, Germany
bFriedrich-Alexander-Universität Erlangen-Nürnberg, Interdisciplinary Center for Molecular Materials (ICMM), Computer Chemistry Center (CCC), 91052 Erlangen, Germany. E-mail: bernd.meyer@chemie.uni-erlangen.de
cForschungszentrum Jülich GmbH, Helmholtz-Institut Erlangen-Nürnberg für Erneuerbare Energien (HI ERN), 91058 Erlangen, Germany
dForschungszentrum Jülich GmbH, Institute of Energy Technologies (IET), Institute of Energy Materials and Devices (IMD), Photovoltaic (IMD-3), 52428 Jülich, Germany
First published on 26th January 2026
Abstract
Halide double perovskites are an emerging class of lead-free materials for optoelectronic and photovoltaic applications. Their properties can be tuned by changing ion ratios on different sublattices. An example is Cs2AgxNa1−xBiyIn1−yCl6 (CANBIC), which shows impressive photoluminescence for a small range of compositions. In this work, we combine perfect experimental composition control in high-throughput synthesis with density functional theory (DFT) calculations and machine learning to identify the subspace of optimal ion ratios. For the example of CANBIC, we demonstrate that important excited state parameters determining photoluminescence can be successfully predicted by using only high-throughput DFT ground state data in a two-step machine learning algorithm. This approach reveals the relevant ground state features for the observed photoluminescence and is in accordance with the self-trapped exciton mechanism.
1 Introduction
Lead-halide perovskites have undergone extraordinary development over recent years in the field of optoelectronics and particularly photovoltaics (PV), with champion PV cells demonstrating power conversion efficiencies above 25%.1,2 Nonetheless, they face two major problems: toxicity and stability. Therefore, the need to investigate lead-free alternatives has arisen, for example, through the substitution of Pb2+ in CsPbX3 with two heterovalent ions: one monovalent and one trivalent in an alternating manner. This leads to a class of lead-free halide double perovskites with the general formula A2B(I)B(III)X6. The most popular representatives of this material class are Cs2AgBiX6,3–6 Cs2NaBiX6,7 Cs2AgInX6,8–10 and Cs2NaInX6,11 with X = Cl, Br, I.Double perovskites have revealed unprecedented compositional flexibility due to the feasibility of independent variation of all four components as well as of alloying of two (and more) components on the B(I), B(III), and X sites.12–14 The latter approach appeared to be especially fruitful for the design of new optoelectronic materials because alloying on B(I) and B(III) sites breaks the symmetry of the perovskite lattice, resulting in drastic changes in the absorption and emission of light as well as in the dynamics of photo-physical processes.15
A particular example of such compositional design is Cs2AgxNa1−xBiyIn1−yCl6 (abbreviated as CANBIC by the first letters of constituent elements). Previous experiments have shown that the variation of the cation ratio at both the B(I) and B(III) sites in CANBIC results in a large variety of double perovskite compositions showing solid solution behavior rather than phase separation.13,14 The most interesting feature of these materials is the existence of very strong photoluminescence under UV irradiation as reported previously.13,16–18 The photoluminescence in CANBIC stems from a self-trapped exciton (STE). A STE is generally attributed to a strongly localized exciton (=trapped) in a lattice distortion. Therefore, it occurs in materials with a soft lattice.19
In the quest for high-performance, nontoxic PV materials, we need to maximize both energy efficiency (electrical output Wout per optical input Win), which requires minimizing non-radiative losses, and external quantum efficiency (electrons out per photons in), which balances photon utilization and contributes to integral power efficiency. Achieving high external quantum efficiency requires a high absorption coefficient and efficient separation of primary photo-excited electron–hole pairs into mobile charge carriers.
To do justice to the broad range of possible compositions, a high-throughput screening approach is needed for finding the best ion ratios in the two B-sublattices. Given the quasi-infinite compositional and processing space, computational pre-screening is mandatory to identify subspaces of high chance to encounter materials with the desired properties. However, the quantum-chemical prediction of the required excited-state properties, such as charge separation and recombination rates, is extremely resource consuming and therefore not apt for high-throughput screening.
For this reason, we explore in this work whether this gap can be bridged using a machine learning model, trained solely on easily accessible ground state data, which we collect from high-throughput density functional theory (DFT) calculations, to predict the excited-state properties of interest measured experimentally (see Fig. 1 for the overall workflow). We choose three target properties that are guaranteed to correlate with PV performance but can be obtained without actually building the devices, thereby strongly accelerating experimental screening of the subspaces identified by computational pre-screening: (i) the radiative recombination rate kr is linked to the absorption coefficient via the Strickler/Berg relation and can be obtained by combining steady-state photoluminescence (PL) and transient photoluminescence (TRPL) measurements; (ii) from the same experiments, the non-radiative recombination rate knr is also available, controlling energy losses; (iii) from comparing steady-state PL and UV-Vis measurements, the Stokes shift Es can be obtained.
These three target properties, for which high quality experimental data are available, are predicted by applying Gaussian Process Regression (GPR) as a machine learning method on a large set of ground state DFT data, spanning various structural, mechanical, electronic, and dynamical properties. To avoid overfitting, only such properties are included in the predictive model, which provide additional explanation of variance (mRMR – minimum Redundancy Maximum Relevance).20 The combination of GPR and mRMR has been used successfully to identify the minimum feature set carrying the essential information to predict degradation in organic photovoltaics.21 Combining the data driven machine learning (ML) approach with data sets based on experiments and DFT provides a holistic picture of the combinatorial possibilities in a material like CANBIC. We expect this algorithm to be of a general nature for STE-emissive materials and applicable to different compositions and classes of lead-free perovskites.
2 Methodology
2.1 Experimental
• Sol 1: 1 M InCl3 in 4 M HCl.
• Sol 2: 1 M BiCl3 in 4 M HCl.
• Sol 3: 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 2-propanol
1 2-propanol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 M HCl.
12 M HCl.
• Sol 4: 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 2-propanol
1 2-propanol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13.4 M HCl.
13.4 M HCl.
• Sol 5: 1 M AgNO3 in water.
• Sol 6: water.
• Sol 7: 7.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5
2.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 water
6 water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 M NaAc
4 M NaAc![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 M CsAc.
4 M CsAc.
The 6 × 8 well plate geometry gave rise to the idea of the nominal x and y ratios described in Table 1. In the first step, two separate well plates were synthesized, one with varying x ratios for columns 1–8 and one with varying y ratios for rows A–F. The corresponding volumes of the individual solutions present in precursors I and II are shown in Table 1 as well.
| Amounts and ratios | ||||||||
|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | |
| Sol 1 | 100 | 95 | 90 | 75 | 50 | 25 | 10 | 0 |
| Sol 2 | 0 | 5 | 10 | 25 | 50 | 75 | 90 | 100 |
| Sol 3 | 600 | 600 | 600 | 600 | 600 | 600 | 600 | 600 |
| y | 0.00 | 0.05 | 0.10 | 0.25 | 0.50 | 0.75 | 0.90 | 1.00 |
| Amounts and ratios | ||||||
|---|---|---|---|---|---|---|
| A | B | C | D | E | F | |
| Sol 4 | 500 | 500 | 500 | 500 | 500 | 500 |
| Sol 5 | 0 | 10 | 30 | 50 | 80 | 100 |
| Sol 6 | 100 | 90 | 70 | 50 | 20 | 0 |
| Sol 7 | 160 | 160 | 160 | 160 | 160 | 160 |
| x | 0.00 | 0.10 | 0.30 | 0.50 | 0.80 | 1.00 |
In the second step, the two plates are mixed well by well under constant shaking and left overnight for crystallization. After crystallization, the residual solvent was decanted and the particles were resuspended in 1 ml of 2-propanol and centrifuged for 2 min at room temperature and 1000 rpm using a Hermle Z 32 HK. Afterwards, the residual solvent was removed again and the procedure was repeated another time.
The synthesis relies mainly on sodium salts, which results in an overall minimum percentage of sodium in all double perovskites of 15% even after purification (visible from EDX data; see Table S1 in the SI). After the synthesis, the samples for optical and structural analysis were produced by drop-casting of the fresh precipitates suspended in 0.20 ml of 2-propanol on glass (or adhesive carbon tape, respectively). After drying for 2–5 min, a dense and homogeneous layer forms.
The XRD patterns were registered using a Panalytical X'Pert powder diffractometer with filtered Cu Kα radiation (λ = 1.54178 Å) and with an X'Celerator solid-state stripe detector in the Bragg–Brentano geometry with an angular range of 2θ = 5–100° and a step rate of 0.05° per min. Analysis was performed with MAUD software to carry out Rietveld refinement. The samples were drop-cast as suspensions in 2-propanol on glass substrates and dried under ambient conditions.
After excitation with a 75 W xenon lamp from Thorlabs, the reflectance spectra were recorded using a Lack Comet spectrometer from StellarNet Inc. An optical Y-fiber probe with an identical geometry for the sample and the ultra-pure BaSO4 reference from Alfa-Aesar was used for the registration of the spectra. Absorption spectra were then obtained by the division of the reflectance spectra of the sample and the reference, followed by subtracting the baseline.
The same spectrometer used for the reflectance spectra was used for the photoluminescence spectra in the range of 190–1000 nm with a UV LED from Thorlabs (360–370 nm) as the excitation source. For the kinetic curves of PL decay, a custom-designed setup based on a FluoTime300 luminescence spectrometer from PicoQuant GmbH was used. The setup is equipped with a 402 nm LDH-P-C-405B laser, which was used to excite the samples via an optical fiber. The PL signal was collected in the range of 420–800 nm with excitation and emission slits set to 4 nm.
By combining stationary measurements of the photoluminescence quantum yield (PLQY) from Stroyuk et al.16 with the kinetic data on PL lifetime τ, the values of the rate constants of the radiative recombination kr and the non-radiative recombination knr can be calculated using the following relationships:16
 | (1) |
2.2 Density functional theory
DFT calculations were performed using the periodic plane-wave code PWscf of the Quantum Espresso software package (version 7.0).23,24 The gradient-corrected exchange–correlation functional of Perdew, Burke and Ernzerhof revised for solids (PBEsol),25 Vanderbilt ultrasoft pseudopotentials,26 and a plane-wave basis set with an energy cutoff of 30 Ry were used. In the calculations of optical properties, the ultrasoft pseudopotentials were replaced with SG15 optimized norm-conserving Vanderbilt (ONCV) pseudopotentials and the plane-wave energy cutoff was increased to 60 Ry.27,28 The RPA frequency-dependent complex dielectric function is calculated using the PBEsol eigenvalues and eigenstates.23,24All parent compounds of CANBIC, i.e., Cs2AgBiCl6 (CABC), Cs2NaBiCl6 (CNBC), Cs2AgInCl6 (CAIC), and Cs2NaInCl6 (CNIC), have a face-centered cubic (fcc) crystal structure belonging to the space group Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m (225).29 The primitive and conventional double perovskite unit cells are shown in Fig. 2. For the calculations of various ground-state properties of CANBIC with different ion ratios x and y, we doubled the primitive fcc unit cell in all crystallographic directions to get an 80 atom (2 × 2 × 2) fcc supercell (see Fig. 2). This supercell has eight B(I) and eight B(III) sites, thus allowing us to sample altogether 81 different CANBIC compositions. Structure optimization was done by relaxing all atomic positions and the cubic lattice constant using a (2, 2, 2) Monkhorst–Pack k-point mesh for the supercell, i.e., the crystal structure was constrained to the fcc Bravais lattice, but no constraints were imposed on the atomic positions. The atoms were allowed to locally break the cubic symmetry by, for example, random off-centering or octahedra tilts and rotations. The density of the k-point mesh was increased to (4, 4, 4) in the calculations of elastic and optical properties and to (6, 6, 6) in density-of-states (DOS) calculations.
m (225).29 The primitive and conventional double perovskite unit cells are shown in Fig. 2. For the calculations of various ground-state properties of CANBIC with different ion ratios x and y, we doubled the primitive fcc unit cell in all crystallographic directions to get an 80 atom (2 × 2 × 2) fcc supercell (see Fig. 2). This supercell has eight B(I) and eight B(III) sites, thus allowing us to sample altogether 81 different CANBIC compositions. Structure optimization was done by relaxing all atomic positions and the cubic lattice constant using a (2, 2, 2) Monkhorst–Pack k-point mesh for the supercell, i.e., the crystal structure was constrained to the fcc Bravais lattice, but no constraints were imposed on the atomic positions. The atoms were allowed to locally break the cubic symmetry by, for example, random off-centering or octahedra tilts and rotations. The density of the k-point mesh was increased to (4, 4, 4) in the calculations of elastic and optical properties and to (6, 6, 6) in density-of-states (DOS) calculations.
2.3 Machine learning
The minimum Redundancy Maximum Relevance (mRMR) embedded Gaussian Process Regression (GPR) used in this work has previously been introduced by Liu et al.21 It is capable of finding the essential predictors for a target quantity from a large number of features. For the evaluation of best predictors, the hierarchical mRMR algorithm was given the following ground state DFT properties to choose from as features:• Structural features: lattice constant a [Å], unit cell volume V [Å3], unit cell mass M [u], and density ρ [u Å−3].
• Elastic constants: c11 [GPa], c12 [GPa], and c44 [GPa].
• Derived elastic properties: bulk modulus B [GPa], shear modulus G [GPa], Young's modulus Y [GPa], and Poisson ratio ν.
• Electronic properties: bandgap Eg [eV] and valence band shift VBshift [eV].
• Dynamical properties: Debye temperature ΘD [K].
The evaluated target properties are the three crucial parameters: Stokes shift Es and radiative and non-radiative recombination rates kr and knr, respectively.
The optimization is performed on the basis of known mRMR implementations,30–32 but it does not use a dedicated redundancy measure but instead aims for the improvement of the root mean square error. This limits the number of possible predictors to a few hundred due to computational cost, which is still sufficient for the present study.
In addition to predictive uncertainty, model robustness is assessed using a bootstrap-like resampling procedure. For each GPR configuration, 50 random train/test splits (75/25) are performed, and only surrogate models with RMSE within 0.8–1.2 times the mean RMSE are retained, thereby excluding failed or degenerate fits. Hyperparameters are optimized using automatic relevance determination (ARD), with a lower bound imposed on standardized length scales to favor monotonic trends.
3 Results and discussion
For a good prediction of the experimentally measured photoluminescence properties by ML from the DFT ground state features, two criteria have to be fulfilled: first, the DFT results have to be in good accordance with experimental results, and second, data have to be continuous functions.For the prediction of the photoluminescence criteria, the DFT calculations were performed to evaluate different ground-state properties of CANBIC perovskites, including structural properties, in particular the lattice parameter, elastic parameters (elastic constants, bulk, shear, and Young's moduli, and Poisson ratio), optical properties (dielectric function, reflectivity, energy loss function, absorption coefficient, refractive index, optical conductivity, and extinction coefficient), electronic properties (band structure and density-of-states), and dynamical properties (Debye temperature).
3.1 Density functional theory
The high-throughput DFT calculations were done with the 80 atom fcc supercell. This allows for eight B(I) and eight B(III) exchange positions and a total of 81 different compositions. For all compositions, the ground-state properties listed above were calculated and the results are presented in 2D colormaps with the B(I) ratio on one and the B(III) ratio on the other axes. Each dot in the map represents one distinct evaluated composition (the same goes for the presentation of the experimental data).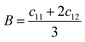 | (2a) |
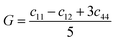 | (2b) |
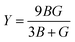 | (2c) |
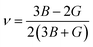 | (2d) |
The bulk and shear moduli together with the density ρ of the material determine the transversal and longitudinal velocities of sound vt and vl, respectively, which can be used to derive the Debye temperature ΘD using a simple Debye model:34,35
 | (3a) |
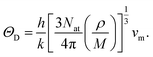 | (3b) |
Fig. 4 depicts both the 2D colormaps for the bulk, shear, and Young's modulus with the corresponding distortion schemes and the 2D colormap for the Debye temperature. The derived elastic and dynamic constants (Fig. 4) show similarly smooth trends like the lattice parameter. The values obtained for the four parent compounds match with literature values using the same technique.36–38 Both the bulk and shear moduli in Fig. 4a and b are almost invariant in terms of the Ag fraction. The opposite is visible for the Debye temperature, which is almost invariant to the Bi fraction (Fig. 4d). Interestingly, the Young's modulus displays a diagonal shift decreasing with almost the same slope on the x- and y-axes (Fig. 4c). Values for the individual elastic constants and the Poisson ratio can be found in Fig. S1 of the SI. All calculated elastic properties, both the elastic constants and the derived moduli, fulfill the continuity criterion.
 | (4a) |
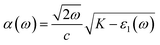 | (4b) |
 | (4c) |
 | (4d) |
 | (4e) |
 | (4f) |
 | (4g) |
A full set of the derived optical functions for the CANBIC parent compounds is given in Fig. 5. Only the Ag-containing compounds CABC and CAIC show non-zero ε2 values in the visible range (1.5–3.1 eV). Clearly ε1(0) increases from 2.33 to 3.74 eV for CNIC < CNBC < CAIC < CABC. For energies higher than 7.5 eV, all parent compounds show almost the same course for ε1 and ε2. The refractive index in the visible range is between 1.5 and 2.0 for all materials. The absorption coefficient shows a steep decrease at ≈10.0 eV for all compositions. Each of the compounds has at least two more absorption peaks (Table 2). From the absorption spectra, the bandgap can also be estimated by evaluating the absorption onset (Fig. 6c).
| CNIC | CABC | CAIC | CNBC | |
|---|---|---|---|---|
| Peak 1 | 4.69 | 3.91 | 3.55 | 5.83 |
| Peak 2 | 8.60 | 8.60 | 9.02 | 8.24 |
| Peak 3 | 13.59 | 13.53 | 13.65 | 13.47 |
 | ||
| Fig. 6 Band structures of (a) CAIC and (b) CNBC with total and atom-resolved projected DOSs (calculated with the primitive 10 atom fcc unit cell). 2D maps of the electronic properties of CANBIC: (c) absorption onset from Fig. 5d, (d) bandgap from DFT band structure calculations, (e) experimental bandgap from the indirect Tauc plot. (f) Absorbance data of samples with varying Bi content at x(Ag) = 0.5 adapted for indirect Tauc plot analysis. | ||
Along with the bandgap, additional information can be derived from the electronic structure calculations. To demonstrate the whole variance of the compositional space, two parent compounds with opposing compositions are shown in Fig. 6a and b: CAIC and CNBC. In particular, the data presented here show that the exchange of the B-site ions leads to a significant change in the valence bandwidth from almost 5.0 eV for CAIC to 2.5 eV for CNBC (Fig. 7a). This can be explained by analyzing the atom-resolved projected density-of-states (DOS) diagrams. For CAIC, the valence band is almost entirely composed of Ag- and Cl-related states. For CNBC, this Ag contribution obviously vanishes but a small Bi contribution very close to the valence band edge emerges. Another but less prominent feature is the composition of the conduction band. In both cases, the conduction band is made up of B(III) states, but for Bi there are significantly more unoccupied states than for In. More details on the elemental fractions of the conduction and valence bands over the whole material composition range can be found in Fig. S2 and S3 of the SI.
 | ||
| Fig. 7 (a) 2D map of the valence bandwidth. (b) 2D map of the valence band edge shift VBshift. (c) Comparison between valence band shift against vacuum from XPS and DFT-calculated VB shift for varying Bi content at x(Ag) = 0.4 (experimental data from Stroyuk et al.16). | ||
The position of the Cs-related peak was found to shift from ≈6.8 eV for CAIC to ≈6.2 eV for CNBC with respect to the valence band edge (Fig. 6a and b). The Cs peak originates from deep, almost core-like states, which are not affected by the composition. The observed shift can therefore directly be attributed to the change in the valence band edge due to the compositional variation. This gives us the opportunity to evaluate the valence band position in correlation to x and y (Fig. 7b). The calculated shifts of the valence band edge can be compared with experimental results from XPS measurements (Fig. 7c).16 In the experiments, only a small number of compositions were evaluated, allowing a comparison only for a subset of the whole compositional space. However, this comparison shows a perfect alignment of the experimental (black) and theoretical (red) results. Therefore, the graph in Fig. 7c can be used as a calibration to determine the VB position with respect to the vacuum level from the calculated VB edge shifts for the missing experimental data points.
3.2 Photoluminescence data
The experimental photoluminescence data are summarized in Fig. 8. The luminescence is visible for compositions with non-zero Ag and Bi contents. Its bright luminescence under UV illumination is probably the most investigated feature of CANBIC in previous work.12,16 The photoluminescence spectra (Fig. 8a) show a single broad emission peak that shifts to lower energies for increasing Bi content but is almost invariant for changing Ag content. The width of the emission peak is almost constant (Fig. 8b and c).For a potential application of CANBIC, it is essential to understand how the luminescence features change with the composition. Therefore, three features, the Stokes shift Es, the radiative recombination rate kr and the non-radiative recombination rate knr, are investigated.
In Fig. 8g, the kinetic luminescence decay curves of different compositions with varying Bi content are shown. With increasing Bi content, the luminescence lifetime decreases from ≈1300 ns to ≈150 ns. The luminescence lifetime τ was evaluated by using a stretched exponential with exponent b to fit the time t dependent decay curves:22,39,40
| f(t; τ, b) = e−(t/τ)1/b. | (5) |
The results are shown in Fig. 8e. The radiative and non-radiative recombination rate constants kr and knr are then determined by using eqn (1) (Fig. 8h and i).
Fig. 8d shows the combined PL and absorption spectra of samples with varying Bi content. All display a Stokes shift between 0.6 eV and 1.1 eV, which is quite stable for the compositional exchange. Both the PL peak and the absorption edge move to lower energies for increasing Bi content, but the PL movement is more dominant.
CANBIC's highest luminescence intensity is found for x = 0.4 and y = 0.05 (Fig. 8b). The same composition also possesses the highest luminescence lifetime and radiative recombination rate (Fig. 8e and h). CANBIC is known to be a self-trapped exciton material, which is in accordance with the high PLQY and lifetime observed and explains the prominent Stokes shift.
In CANBIC, the ideal recombination rates (low non-radiative, high radiative) are found for compositions with high Ag (85%) and low (but nonzero!) Bi (10%) content. Unfortunately, at this composition, the Stokes shift is rather high, making exciton dissociation into mobile carriers an inefficient process. As shown in Fig. 8f, minimization of the Stokes shift would require maximizing the Bi content, which however has adverse effects on non-radiative recombination (see Fig. 8i). This means that there exists no composition at which CANBIC will be a good PV material. Using ML, we hope to identify orthogonal DFT predictors for the Stokes shift and the recombination properties, respectively; these would allow us to identify new double perovskites with promising excited state properties as PV materials by computational pre-screening.
3.3 Machine learning: mRMR and GPR
For the prediction of this multi-objective target, multiple steps are necessary. Primarily, the suitable and most relevant DFT-calculated identifiers for the photoluminescence parameters have to be found using a minimum redundancy maximum relevance (mRMR) algorithm. Afterwards, the predictors identified in this way are used to generate a predictive model via a regression algorithm, specifically Gaussian Process Regression (GPR).20 The outcome of this predictive model is a possible indicator of the physics behind the photoluminescence.To enhance the probability for generalization beyond the available dataset, we restricted the shape of the machine learned surrogate function to monotonous trends. A simple example is displayed schematically in Fig. 8b: in our experimental dataset, PLQY shows a local maximum for a certain Ag/Na ratio. If we allow local maxima in the surrogate function along any given predictor, then mRMR will “happily” select the experimental Ag/Na ratio itself as a good predictor for PLQY. Even if we exclude experimental parameters from the predictor list, mRMR will still look for DFT properties that are strictly proportional to the Ag/Na ratio. Although this single predictor will correctly predict the Ag/Na ratio of maximum PLQY, it is improbable that it will generalize beyond the CANBIC class. However, if we disallow local extrema, then we force mRMR to identify the individual counter-players causing the local PLQY maximum, namely a single predictor that increases PLQY and another one that decreases it. These predictors have a higher chance to generalize beyond a specific material class.
Feature selection is performed by sequential addition of predictors to the GPR by a greedy algorithm, thereby allowing mRMR to be extended to non-linear correlations: first, GPR is run on all single predictors, and the one with the highest R2 value is retained. Out of the remaining predictors, the one bringing the strongest increase in R2 is added. The procedure ends when no further predictor increases R2 significantly. To test the stability of the resulting surrogate function, each GPR is run 50 times in a bootstrap scheme.
The list of best predictors for each target is given in Table 3. These predictors can now be used in a regression algorithm to predict the target properties. Fig. 9a–c show the corresponding surrogate models (colored isocontour) together with the experimental values (symbols). Color matching between symbols and isosurfaces shows the high quality of the prediction, as well as the absence of hidden parameters.
| Target | Best predictors |
|---|---|
| Stokes shift Es | Density ρ |
| Elastic constant c12 | |
| Radiative rate kr | Debye temperature ΘD |
| Valence band shift VBshift | |
| Non-radiative rate knr | Mass M |
| Debye temperature ΘD |
It is apparent that the dataset is not fully independent and isotropically distributed; specifically, there is some correlation between the elastic constant c12 and the density ρ in Fig. 9a and between the valence band shift VBshift and the Debye temperature ΘD in Fig. 9b. However, the spreading of the datapoints across the two-dimensional space is sufficient to quantify individual trends along both dimensions. This can be shown more clearly in one-dimensional intersections through the surrogate function. An example is displayed in Fig. 9d for the Stokes shift Es; the corresponding plots for kr and knr can be found in Fig. S5 of the SI. In Fig. 9d, the surrogate model is shown by a blue line. Light blue regions indicate the 95% confidence interval of a single prediction, as reported by GPR, while dark blue regions identify the 95% confidence interval for the trend. Fig. 9d shows that despite the correlation between c12 and ρ, the trends along both dimensions can be assessed with low uncertainty.
The set of decisive and sufficient features in Table 3 exhibits the desired orthogonality, allowing the optimization of one target without penalizing the other. However, with the current dataset, we cannot demonstrate the generalization of these predictors beyond CANBIC. For this reason, we apply domain knowledge to assess their potential for generalization.
3.4 Evaluation of the model
To gain physical insight from a numerical model, a closer look at the chosen best predictors is necessary.For the Stokes shift in Fig. 9a, the most relevant predictors are the density and one of the elastic constants. Both correlations are monotonic with the elastic constant having a far stronger influence on the target variable. With CANBIC being a self-trapped exciton material and self-trapping being strongly correlated with localized lattice distortions, this correlation is expected but nevertheless gratifying.
The non-radiative recombination rate is almost purely dependent on the molecular mass of the system with a slight deviation depending on the Debye temperature. A smaller molecular mass in this prediction indicates a lower non-radiative recombination rate. On the other hand, the compounds with the smallest molecular weight of the synthesis procedure could not be evaluated in this prediction, as they are not luminescent. Therefore, no rate constants could be determined in this work.
The final target property (the radiative recombination rate) is described again by the Debye temperature and additionally the VB edge shift. This is of special interest, as for none of the targets the PBEsol calculated bandgap was chosen as a predictor by the algorithm, but instead another characteristic of the calculated electronic structure. The accuracy of this parameter has already been shown by the XPS data comparison (Fig. 7c). Again, similar to the non-radiative recombination rate, the Debye temperature is a weaker predictor.
A high radiative and low non-radiative recombination rate originates either from a low number of trap states or a high defect tolerance of the material, mostly attributed to shallow trap states. CANBIC is known to have PLQY ≥ 90%, which already indicates a high defect tolerance or shallow traps. This observation is in accordance with CANBIC being a self-trapped exciton material, which is in general highly defect tolerant.
4 Conclusions
Overall, we demonstrated the possibility to investigate lead-free halide double perovskites with interchangeable cation ratios by high-throughput DFT calculations of a broad range of structural, elastic, optical, electronic and dynamical ground state properties. The DFT results are in very good accordance with the available experimental results and at the same time broaden the knowledge with data that could not be obtained experimentally due to the synthesis procedure or the amount of material consumption.For identifying subspaces of compositions with optimized ion ratios we developed a rapid pre-screening method of potential photovoltaic candidates for double perovskites. By using a GPR with embedded feature selection, we selected the relevant, non-redundant ground state DFT properties for the prediction of fluorescent properties. The model yields high-quality values for the Stokes shift and the radiative and non-radiative recombination rate without the use of sophisticated computational methods, which would be needed to determine excited-state properties.
Finally, we conclude that the Stokes shift is related to the lattice stiffness as expected for a self-trapped exciton material and therefore with a less stiff lattice the charge carrier mobility could be increased. Combining this information and the correlations for the recombination rates, a full set of proxies for the portfolio of requirements for a photovoltaic material is defined.
Author contributions
M. S. Günthert: investigation and writing – original draft; L. Lüer: investigation, methodology, and writing – review and editing; O. Stroyuk: conceptualization and writing – review and editing; O. Raievska: investigation and methodology; C. Kupfer: methodology; A. Osvet: writing – review and editing; B. Meyer: conceptualization, writing – review and editing, and project administration; C. J. Brabec: conceptualization, project administration, and funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
The ML code as well as the corresponding dataset is available on GitHub at https://github.com/larryluer/mrmr-gpr (building predictive models in a materials science context) and on Zenodo at https://doi.org/10.5281/zenodo.18301315. The experimental data are archived on Zenodo at https://doi.org/10.5281/zenodo.6992560 and the DFT data are published on Zenodo at https://doi.org/10.5281/zenodo.17800198.Supplementary information (SI): further characterization data of CANBIC, the ML training data, the mRMR training data, and the list of GPR predictions. See DOI: https://doi.org/10.1039/d5dd00152h.
Acknowledgements
The authors gratefully acknowledge financial support for this work by the Deutsche Forschungsgemeinschaft (DFG) under GRK 2495/2 (project number 399073171, projects E and L) and the German Federal Ministry for Economic Affairs and Climate Action (project Pero4PV, FKZ: 03EE1092A).Notes and references
- H.-S. Kim, C.-R. Lee, J.-H. Im, K.-B. Lee, T. Moehl, A. Marchioro, S.-J. Moon, R. Humphry-Baker, J.-H. Yum, J. E. Moser, M. Grätzel and N.-G. Park, Sci. Rep., 2012, 2, 591 Search PubMed.
- F. Qiu, J. Sun, Z. Zhang, T. Shen, H. Liu and J. Qi, Mater. Today Energy, 2021, 21, 100837 Search PubMed.
- H. Absike, N. Baaalla, R. Lamouri, H. Labrim and H. Ez-zahraouy, Int. J. Energy Res., 2022, 46, 11053–11064 CrossRef CAS.
- H. C. Sansom, G. Longo, A. D. Wright, L. R. V. Buizza, S. Mahesh, B. Wenger, M. Zanella, M. Abdi-Jalebi, M. J. Pitcher, M. S. Dyer, T. D. Manning, R. H. Friend, L. M. Herz, H. J. Snaith, J. B. Claridge and M. J. Rosseinsky, J. Am. Chem. Soc., 2021, 143, 3983–3992 CrossRef CAS.
- D. Gill, P. Bhumla, M. Kumar and S. Bhattacharya, J. Phys.: Mater., 2021, 4, 025005 CAS.
- F. Lv, T. Zhong, Y. Qin, H. Qin, W. Wang, F. Liu and W. Kong, Nanomaterials, 2021, 11, 1361 Search PubMed.
- R.-I. Biega, Y. Chen, M. R. Filip and L. Leppert, Nano Lett., 2023, 23, 8155–8161 CrossRef CAS.
- M. Kumar, M. Jain, A. Singh and S. Bhattacharya, Appl. Phys. Lett., 2021, 118, 021901 CrossRef CAS.
- Y. Liu, I. J. Cleveland, M. N. Tran and E. S. Aydil, J. Phys. Chem. Lett., 2023, 14, 3000–3006 CrossRef CAS PubMed.
- M. Liang, W. Lin, Q. Zhao, X. Zou, Z. Lan, J. Meng, Q. Shi, I. E. Castelli, S. E. Canton, T. Pullerits and K. Zheng, J. Phys. Chem. Lett., 2021, 12, 4965–4971 CrossRef CAS PubMed.
- Y. Yu, W. Zhou, C. Li, P. Han, H. Li and K. Zhao, Nanomaterials, 2023, 13, 549 CrossRef CAS.
- J. Luo, X. Wang, S. Li, J. Liu, Y. Guo, G. Niu, L. Yao, Y. Fu, L. Gao, Q. Dong, C. Zhao, M. Leng, F. Ma, W. Liang, L. Wang, S. Jin, J. Han, L. Zhang, J. Etheridge, J. Wang, Y. Yan, E. H. Sargent and J. Tang, Nature, 2018, 563, 541–545 CrossRef CAS PubMed.
- K. Dave, W.-T. Huang, T. Leśniewski, A. Lazarowska, D. Jankowski, S. Mahlik and R.-S. Liu, Dalton Trans., 2022, 51, 2026–2032 RSC.
- D. Manna, J. Kangsabanik, T. K. Das, D. Das, A. Alam and A. Yella, J. Phys. Chem. Lett., 2020, 11, 2113–2120 CrossRef CAS.
- W. Meng, X. Wang, Z. Xiao, J. Wang, D. B. Mitzi and Y. Yan, J. Phys. Chem. Lett., 2017, 8, 2999–3007 Search PubMed.
- O. Stroyuk, O. Raievska, A. Barabash, C. Kupfer, A. Osvet, V. Dzhagan, D. R. T. Zahn, J. Hauch and C. J. Brabec, Mater. Adv., 2022, 3, 7894–7903 RSC.
- F. Locardi, E. Sartori, J. Buha, J. Zito, M. Prato, V. Pinchetti, M. L. Zaffalon, M. Ferretti, S. Brovelli, I. Infante, L. De Trizio and L. Manna, ACS Energy Lett., 2019, 4, 1976–1982 Search PubMed.
- Y. Gao, Y. Pan, F. Zhou, G. Niu and C. Yan, J. Mater. Chem. A, 2021, 9, 11931–11943 Search PubMed.
- S. Li, J. Luo, J. Liu and J. Tang, J. Phys. Chem. Lett., 2019, 10, 1999–2007 CrossRef CAS.
- L.-Y. Bi, Y.-Q. Hu, M.-Q. Li, T.-L. Hu, H.-L. Zhang, X.-T. Yin, W.-X. Que, M. S. Lassoued and Y.-Z. Zheng, J. Mater. Chem. A, 2019, 7, 19662–19667 Search PubMed.
- C. Liu, L. Lüer, V. M. L. Corre, K. Forberich, P. Weitz, T. Heumüller, X. Du, J. Wortmann, J. Zhang, J. Wagner, L. Ying, J. Hauch, N. Li and C. J. Brabec, Adv. Mater., 2024, 36, 2300259 CrossRef CAS PubMed.
- O. Stroyuk, O. Raievska, A. Barabash, M. Batentschuk, A. Osvet, S. Fiedler, U. Resch-Genger, J. Hauch and C. J. Brabec, J. Mater. Chem. C, 2022, 10, 9938–9944 RSC.
- P. Giannozzi, S. Baroni, N. Bonini, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, G. L. Chiarotti, M. Cococcioni, I. Dabo, A. Dal Corso, S. de Gironcoli, S. Fabris, G. Fratesi, R. Gebauer, U. Gerstmann, C. Gougoussis, A. Kokalj, M. Lazzeri, L. Martin-Samos, N. Marzari, F. Mauri, R. Mazzarello, S. Paolini, A. Pasquarello, L. Paulatto, C. Sbraccia, S. Scandolo, G. Sclauzero, A. P. Seitsonen, A. Smogunov, P. Umari and R. M. Wentzcovitch, J. Phys.: Condens. Matter, 2009, 21, 395502 Search PubMed.
- P. Giannozzi, O. Andreussi, T. Brumme, O. Bunau, M. Buongiorno Nardelli, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, M. Cococcioni, N. Colonna, I. Carnimeo, A. Dal Corso, S. de Gironcoli, P. Delugas, R. A. DiStasio, A. Ferretti, A. Floris, G. Fratesi, G. Fugallo, R. Gebauer, U. Gerstmann, F. Giustino, T. Gorni, J. Jia, M. Kawamura, H.-Y. Ko, A. Kokalj, E. Küçükbenli, M. Lazzeri, M. Marsili, N. Marzari, F. Mauri, N. L. Nguyen, H.-V. Nguyen, A. Otero-de-la Roza, L. Paulatto, S. Poncé, D. Rocca, R. Sabatini, B. Santra, M. Schlipf, A. P. Seitsonen, A. Smogunov, I. Timrov, T. Thonhauser, P. Umari, N. Vast, X. Wu and S. Baroni, J. Phys.: Condens. Matter, 2017, 29, 465901 CrossRef CAS.
- J. P. Perdew, A. Ruzsinszky, G. I. Csonka, O. A. Vydrov, G. E. Scuseria, L. A. Constantin, X. Zhou and K. Burke, Phys. Rev. Lett., 2008, 100, 136406 Search PubMed.
- D. Vanderbilt, Phys. Rev. B: Condens. Matter Mater. Phys., 1990, 41, 7892–7895 Search PubMed.
- D. R. Hamann, Phys. Rev. B: Condens. Matter Mater. Phys., 2013, 88, 085117 Search PubMed.
- M. Schlipf and F. Gygi, Comput. Phys. Commun., 2015, 196, 36–44 CrossRef CAS.
- Y. Cai, W. Xie, Y. T. Teng, P. C. Harikesh, B. Ghosh, P. Huck, K. A. Persson, N. Mathews, S. G. Mhaisalkar, M. Sherburne and M. Asta, Chem. Mater., 2019, 31, 5392–5401 CrossRef CAS.
- Y. Zhu, Y. Zhu, X. Huang, J. Chen, Q. Li, J. He and P. Jiang, Adv. Energy Mater., 2019, 9, 1901826 CrossRef.
- Y. Wei, Z. Chen, G. Lu, N. Yu, C. Li, J. Gao, X. Gu, X. Hao, G. Lu, Z. Tang, J. Zhang, Z. Wei, X. Zhang and H. Huang, Adv. Mater., 2022, 34, 2204718 Search PubMed.
- Z. Zhou, S. Xu, J. Song, Y. Jin, Q. Yue, Y. Qian, F. Liu, F. Zhang and X. Zhu, Nat. Energy, 2018, 3, 952–959 Search PubMed.
- M. J. Mehl, J. E. Osburn, D. A. Papaconstantopoulos and B. M. Klein, Phys. Rev. B: Condens. Matter Mater. Phys., 1990, 41, 10311–10323 Search PubMed.
- M. Faghihnasiri, M. Izadifard and M. E. Ghazi, J. Phys. Chem. C, 2017, 121, 27059–27070 CrossRef CAS.
- O. L. Anderson, J. Phys. Chem. Solids, 1963, 24, 909–917 CrossRef CAS.
- M. N. Islam and J. Podder, Heliyon, 2022, 8, e10032 CrossRef PubMed.
- S. Choudhary, S. Tomar, D. Kumar, S. Kumar and A. S. Verma, East Eur. J. Phys., 2021, 74–80 Search PubMed.
- A. A. Sholagberu, W. A. Yahya and A. A. Adewale, Phys. Scr., 2022, 97, 085824 CrossRef.
- D. Zhu, J. Zito, V. Pinchetti, Z. Dang, A. Olivati, L. Pasquale, A. Tang, M. L. Zaffalon, F. Meinardi, I. Infante, L. De Trizio, L. Manna and S. Brovelli, ACS Energy Lett., 2020, 5, 1840–1847 CrossRef PubMed.
- L. Zdražil, S. Kalytchuk, M. Langer, R. Ahmad, J. Pospíšil, O. Zmeškal, M. Altomare, A. Osvet, R. Zbořil, P. Schmuki, C. J. Brabec, M. Otyepka and Š. Kment, ACS Appl. Energy Mater., 2021, 4, 6445–6453 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |







