 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Design of Pt-based pseudo-binary intermetallic catalysts for dry reforming of propane using CO2
Feilong
Xing†
 a,
Fang
Zhang
b,
Ken-ichi
Shimizu
a,
Fang
Zhang
b,
Ken-ichi
Shimizu
 a and
Shinya
Furukawa‡
a and
Shinya
Furukawa‡
 *a
*a
aInstitute for Catalysis, Hokkaido University, N21, W10, Sapporo 001-0021, Japan. E-mail: furukawa@chem.eng.osaka-u.ac.jp; Tel: +81 06 6879 7808
bDivision of Applied Chemistry, Graduate School of Engineering, Osaka University, Osaka 565-0871, Japan
First published on 3rd March 2026
Abstract
The utilization of CO2 along with commercial light alkane dehydrogenation processes has attracted much attention due to the high demand for the reduction of CO2 in the atmosphere caused by human activities and the shale gas revolution. Compared with the widely studied dry reforming of methane, converting propane to syngas using CO2 is more beneficial in terms of easy handling, energy saving and CO2 utilization efficiency. However, the design of an efficient and stable catalyst remains a challenge due to the difficulty of activating CO2 and propane simultaneously. Herein, we report a multifunctional catalyst with a pseudo-binary alloy structure, Pt2CoIn3/CeO2, which exhibits high activity for C–C bond cleavage and efficient CO2 activation. As a result, it delivers high CO selectivity and strong coke resistance, maintaining stable performance over 100 h of reaction. Detailed X-ray analyses revealed that the intermetallic structure varies with the Co content, which in turn tunes both the C3H6 adsorptivity and CO2 activation capability. By combining intermediate surface-reaction studies with density functional theory calculations, we identify the difference in the energy barriers for C3H6 decomposition and C3H6 desorption as a key descriptor governing product selectivity. These findings provide a new design concept for multimetallic catalysts that can be extended to a broad range of selective conversion reactions.
Introduction
Syngas (a mixture of H2 and CO) production is an important industrial process for the conversion of natural gas into liquid fuels and valuable chemicals.1 In this regard, thermochemical reforming has attracted much attention as a technology for obtaining syngas.2–4 Conventional syngas production via steam reforming often yields products with H2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CO > 3, which is higher than that required for further downstream processing of olefins (Fischer–Tropsch reaction) or the synthesis of oxygenated compounds. Therefore, as an alternative, the dry reforming of methane with CO2 is highly important and thus has been extensively studied.5–8 Recently, the dry reforming of light hydrocarbons such as ethane and propane has also received considerable attention due to the high demand for the reduction of CO2 in the atmosphere caused by human activities and the shale gas revolution.9–13 Compared with methane, the use of ethane and propane can consume more CO2 stoichiometrically, thereby improving the CO2 utilization efficiency and yielding higher H2/CO2 ratios,14 which enhances their suitability for subsequent industrial applications and therefore offers great potential for reducing greenhouse gas emissions. In particular, propane is more easily activated than methane at lower reaction temperatures. Propane is generally produced by oil refinery or natural gas separation and can also be liquefied for facile storage and transportation.13 However, due to the higher number of carbon atoms in propane than in methane, its catalytic conversion is typically prone to coking and rapid catalyst deactivation, posing challenges for the design of efficient catalysts.
CO > 3, which is higher than that required for further downstream processing of olefins (Fischer–Tropsch reaction) or the synthesis of oxygenated compounds. Therefore, as an alternative, the dry reforming of methane with CO2 is highly important and thus has been extensively studied.5–8 Recently, the dry reforming of light hydrocarbons such as ethane and propane has also received considerable attention due to the high demand for the reduction of CO2 in the atmosphere caused by human activities and the shale gas revolution.9–13 Compared with methane, the use of ethane and propane can consume more CO2 stoichiometrically, thereby improving the CO2 utilization efficiency and yielding higher H2/CO2 ratios,14 which enhances their suitability for subsequent industrial applications and therefore offers great potential for reducing greenhouse gas emissions. In particular, propane is more easily activated than methane at lower reaction temperatures. Propane is generally produced by oil refinery or natural gas separation and can also be liquefied for facile storage and transportation.13 However, due to the higher number of carbon atoms in propane than in methane, its catalytic conversion is typically prone to coking and rapid catalyst deactivation, posing challenges for the design of efficient catalysts.
There have been many previous reports on the catalytic dry reforming of propane (DRP) using precious metals such as Pt, Ru, Rh, and Re.9,15–17 Precious metals exhibit high catalytic activity and selectivity in thermochemical reforming processes, but their high cost has limited their application in industry. Transition metal-based catalysts such as Ni are cost-effective alternatives to precious metal-based catalysts, and many researchers have studied the activity of Ni-based catalysts for dry reforming of methane reactions.13,18–22 However, Ni-based catalysts are rapidly deactivated by sintering and coke formation,19–25 and this is even more pronounced when Ni-based catalysts are used in DRP. Catalyst design based on multimetallic alloys is a possible approach to developing an efficient coke-resistant catalyst while reducing costs in large-scale processes.26–29 Usually, it is challenging to activate both molecules simultaneously due to the different properties of propane and carbon dioxide. However, this could be overcome by combining an appropriate metal element for each molecule as a component of the alloy.28 Recently, we reported that Pt1Co1In2/CeO2 exhibited remarkably high catalytic activity and CO2 utilization efficiency in the oxidative dehydrogenation of propane using CO2 (CO2-ODP).27 The roles of Pt, Co, and In in the catalysis of CO2-ODP were C3H8 dehydrogenation, CO2 reduction, and enhancing C3H6 selectivity by the ensemble effect, respectively.
In this study, we discovered that changing the Pt![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co
Co![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) In ratio in the ternary system drastically switched the product distribution from CO2-ODP to DRP: the Co-rich PtCo2In3/CeO2 worked as a highly efficient catalyst for DRP with high CO selectivity and long-term stability of over 100 h. By controlling the amount of Co-doping, the catalytic activity and stability of the Pt–Co–In/CeO2 catalyst can be largely improved. The origin of the “switching catalysis” was investigated by a mechanistic study based on some characterization techniques such as X-ray powder diffraction (XRD), transmission electron microscopy (TEM), and temperature-programmed-mass spectrometry measurements (TP-MS). Here, we report not only a highly efficient catalyst for DRP with high selectivity, coke resistance, and thermal stability, but also the high tunability of catalyst design based on the multi-metallic alloys.
In ratio in the ternary system drastically switched the product distribution from CO2-ODP to DRP: the Co-rich PtCo2In3/CeO2 worked as a highly efficient catalyst for DRP with high CO selectivity and long-term stability of over 100 h. By controlling the amount of Co-doping, the catalytic activity and stability of the Pt–Co–In/CeO2 catalyst can be largely improved. The origin of the “switching catalysis” was investigated by a mechanistic study based on some characterization techniques such as X-ray powder diffraction (XRD), transmission electron microscopy (TEM), and temperature-programmed-mass spectrometry measurements (TP-MS). Here, we report not only a highly efficient catalyst for DRP with high selectivity, coke resistance, and thermal stability, but also the high tunability of catalyst design based on the multi-metallic alloys.
Results
Structure characterization of the catalysts
The Pt–Co–In/CeO2 (Pt: 3 wt%, Pt![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co
Co![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) In = 1
In = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 and 2
3 and 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, hereafter, PtCo2In3 and Pt2CoIn3) catalysts were prepared by a conventional impregnation method using CeO2 as a support. The catalyst was first characterized by high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) and energy dispersive X-ray (EDX) analyses (Fig. 1a and S1). The elemental maps of Pt, Co, and In confirmed that these three elements were highly dispersed on the support and comprised the nanoparticles with a diameter of ∼5 nm. Other Co species were also present apart from those in the alloy nanoparticles. To confirm whether Co was doped into the nanoparticles, X-ray diffraction (XRD) analysis (Fig. 1b and S2a) was performed. The referenced PtIn/CeO2 showed two intense peaks assignable to the 110 and 102 diffractions of intermetallic Pt2In3 (P
3, hereafter, PtCo2In3 and Pt2CoIn3) catalysts were prepared by a conventional impregnation method using CeO2 as a support. The catalyst was first characterized by high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) and energy dispersive X-ray (EDX) analyses (Fig. 1a and S1). The elemental maps of Pt, Co, and In confirmed that these three elements were highly dispersed on the support and comprised the nanoparticles with a diameter of ∼5 nm. Other Co species were also present apart from those in the alloy nanoparticles. To confirm whether Co was doped into the nanoparticles, X-ray diffraction (XRD) analysis (Fig. 1b and S2a) was performed. The referenced PtIn/CeO2 showed two intense peaks assignable to the 110 and 102 diffractions of intermetallic Pt2In3 (P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1, ICSD-197672) at 39.7° and 40.0°, respectively (Fig. S2a). These peaks slightly shifted to lower angles when a small amount of Co was added (39.6° and 39.9°, Fig. 1b, Pt2CoIn3/CeO2), indicating the lattice shrinkage of Pt2In3 upon Co-doping and the formation of the (Pt1−xCox)2In3 pseudo-binary alloy structure. When the Co ratio was further increased (PtCo2In3/CeO2), diffraction peaks assignable to the PtIn2-like (Fm
m1, ICSD-197672) at 39.7° and 40.0°, respectively (Fig. S2a). These peaks slightly shifted to lower angles when a small amount of Co was added (39.6° and 39.9°, Fig. 1b, Pt2CoIn3/CeO2), indicating the lattice shrinkage of Pt2In3 upon Co-doping and the formation of the (Pt1−xCox)2In3 pseudo-binary alloy structure. When the Co ratio was further increased (PtCo2In3/CeO2), diffraction peaks assignable to the PtIn2-like (Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m, ICSD-59496) structure appeared at 40.0° and 40.1°, which were slightly lower than those of the corresponding pure PtIn2 (40.1° and 40.2°, respectively). This indicates that Co had been doped into the lattice of PtIn2 intermetallic structures, forming a (Pt1−xCox)In2 pseudo binary alloy phase. The crystal structure of Pt–In intermetallics changed depending on the Co content. To determine the crystallographic site of Co, X-ray absorption fine structure (XAFS) analysis was carried out. The Pt LIII-, Co K- and In K-edge X-ray adsorption near-edge structure (XANES) spectra of the Pt-based catalysts showed white line intensities similar to those of the reference foil (Fig. 1c), which confirmed that these metals were in the metallic states. The Pt LIII-edge adsorption edges shifted to high energy (Fig. S3a), which could contribute to the electron transfer by alloying with Co and In. The electronic state of surface Pt was further examined by XPS (Fig. S2b). The Pt 4f peak of monometallic Pt was located at ∼71.0 eV, consistent with the reported binding energy of metallic Pt.30–32 In contrast, the PtIn intermetallic catalysts exhibited a positive shift to higher binding energy, attributable to alloying-induced electronic modification, in agreement with the XANES results. Moreover, increasing Co-doping resulted in a further slight positive shift of the Pt 4f peaks, indicating the progressive modulation of the Pt electronic environment, which may influence reactant adsorption. A small fraction of In2O3 was observed in the In K-edge XANES (Fig. S3d) and extended XAFS (EXAFS) spectra (Fig. S3e; the Fourier transform of the EXAFS (FT-EXAFS) is shown in Fig. S3f), probably due to the high-oxophilicity with the lattice oxygen of CeO2. Fig. 1d shows the Pt LIII-edge EXAFS raw oscillation features of Pt foil and the corresponding catalysts. PtCo2In3/CeO2 and Pt2CoIn3/CeO2 exhibited similar features to those of PtIn/CeO2, but were different from the reference Pt foil having an fcc structure. Similar trends were also shown in the Co K-edge and In K-edge spectra (Fig. S3b and e). These results indicate that the locations of the transition metal (Pt and Co) and the typical metal (In) atoms in the Pt–Co–In/CeO2 catalysts were crystallographically altered, which is consistent with the XRD results (Fig. 1b). Fig. 1e shows the Pt LIII-edge FT-EXAFS spectra of the Pt-based catalysts. The peak position of Pt–In scattering in Pt–Co–In/CeO2 was shorter than that in PtIn/CeO2, suggesting that the lattice distortion resulted from Co-doping. Importantly, the EXAFS curve-fitting was successfully done, considering the Pt–In (In–Pt) and Co–In (In–Co) scatterings, whereas no reliable fitting was obtained when Pt–Co (Co–Pt) scatterings were considered (Fig. S4 and Table S1). This strongly supports the site-selective doping of Co into the Pt sites and demonstrates the formation of the pseudo-binary alloy structure on the CeO2 support. Co–Co scattering was also observed, obtained in the Co K-edge, which may be attributed to monometallic Co species that did not participate in the alloy formation; this is consistent with the observation in the HAADF-STEM-EDX analysis.
m, ICSD-59496) structure appeared at 40.0° and 40.1°, which were slightly lower than those of the corresponding pure PtIn2 (40.1° and 40.2°, respectively). This indicates that Co had been doped into the lattice of PtIn2 intermetallic structures, forming a (Pt1−xCox)In2 pseudo binary alloy phase. The crystal structure of Pt–In intermetallics changed depending on the Co content. To determine the crystallographic site of Co, X-ray absorption fine structure (XAFS) analysis was carried out. The Pt LIII-, Co K- and In K-edge X-ray adsorption near-edge structure (XANES) spectra of the Pt-based catalysts showed white line intensities similar to those of the reference foil (Fig. 1c), which confirmed that these metals were in the metallic states. The Pt LIII-edge adsorption edges shifted to high energy (Fig. S3a), which could contribute to the electron transfer by alloying with Co and In. The electronic state of surface Pt was further examined by XPS (Fig. S2b). The Pt 4f peak of monometallic Pt was located at ∼71.0 eV, consistent with the reported binding energy of metallic Pt.30–32 In contrast, the PtIn intermetallic catalysts exhibited a positive shift to higher binding energy, attributable to alloying-induced electronic modification, in agreement with the XANES results. Moreover, increasing Co-doping resulted in a further slight positive shift of the Pt 4f peaks, indicating the progressive modulation of the Pt electronic environment, which may influence reactant adsorption. A small fraction of In2O3 was observed in the In K-edge XANES (Fig. S3d) and extended XAFS (EXAFS) spectra (Fig. S3e; the Fourier transform of the EXAFS (FT-EXAFS) is shown in Fig. S3f), probably due to the high-oxophilicity with the lattice oxygen of CeO2. Fig. 1d shows the Pt LIII-edge EXAFS raw oscillation features of Pt foil and the corresponding catalysts. PtCo2In3/CeO2 and Pt2CoIn3/CeO2 exhibited similar features to those of PtIn/CeO2, but were different from the reference Pt foil having an fcc structure. Similar trends were also shown in the Co K-edge and In K-edge spectra (Fig. S3b and e). These results indicate that the locations of the transition metal (Pt and Co) and the typical metal (In) atoms in the Pt–Co–In/CeO2 catalysts were crystallographically altered, which is consistent with the XRD results (Fig. 1b). Fig. 1e shows the Pt LIII-edge FT-EXAFS spectra of the Pt-based catalysts. The peak position of Pt–In scattering in Pt–Co–In/CeO2 was shorter than that in PtIn/CeO2, suggesting that the lattice distortion resulted from Co-doping. Importantly, the EXAFS curve-fitting was successfully done, considering the Pt–In (In–Pt) and Co–In (In–Co) scatterings, whereas no reliable fitting was obtained when Pt–Co (Co–Pt) scatterings were considered (Fig. S4 and Table S1). This strongly supports the site-selective doping of Co into the Pt sites and demonstrates the formation of the pseudo-binary alloy structure on the CeO2 support. Co–Co scattering was also observed, obtained in the Co K-edge, which may be attributed to monometallic Co species that did not participate in the alloy formation; this is consistent with the observation in the HAADF-STEM-EDX analysis.
Catalytic performance in DRP
The prepared PtCo2In3/CeO2 and Pt2CoIn3/CeO2 catalysts were then tested in CO2-assisted propane conversion with a C3H8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CO2 molar ratio of 1
CO2 molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 at 550 °C. Fig. 2a–d show the time course of C3H8 conversion, CO2 conversion and the product distribution of both catalysts, respectively. The Pt2CoIn3/CeO2 showed a C3H6-rich product distribution that consisted of 60% C3H6, 39% CO, and the other C1–2 products, and C3H6 selectivity remained at 52% after a 10 h reaction. In contrast, the PtCo2In3/CeO2 catalyst was CO-dominant; CO selectivity was 89% at the initial stage and increased to 95% after a 10 h reaction. This suggests that PtCo2In3/CeO2 has a higher dry reforming activity, likely due to its strong propensity for C–C bond cleavage and subsequent oxygenation, whereas Pt2CoIn3/CeO2 preferentially produces C3H6via C–H bond cleavage and facile desorption. Differences were also observed in the C3H8 conversion trend over time. Although the initial C3H8 conversion was similar for PtCo2In3/CeO2 and Pt2CoIn3/CeO2 (∼16%, Fig. 2a), C3H8 conversion increased after 10 h of reaction and stabilized at ∼26% for PtCo2In3/CeO2, whereas it gradually decreased to ∼10% for Pt2CoIn3/CeO2. To understand the individual effect of each second metal, we also performed control experiments using the corresponding monometallic (Pt/CeO2) and bimetallic (PtCo/CeO2, Co-rich PtCo3/CeO2, PtIn/CeO2, and In-rich PtIn3/CeO2) catalysts (Fig. S5). C3H8 conversion for Pt/CeO2, PtCo/CeO2, and PtIn/CeO2 was 40%, 33%, and 28%, while the CO2 conversion was 38%, 40%, and 16%, respectively. The Co-rich PtCo3/CeO2 exhibited high initial conversions of both C3H8 and CO2, likely due to the strong C–C cleavage ability of the Co-rich PtCo alloy and/or the large amount of remaining Co species. In contrast, In-rich PtIn3/CeO2 showed low conversions of C3H8 and CO2, which may be attributed to the blocking of the active sites by unalloyed In species. These results show that alloying Pt with Co or In decreases C3H8 conversion due to the ensemble effect, while adding a certain amount of Co increases CO2 conversion. Furthermore, Pt2CoIn3/CeO2 displayed a similar product distribution to that of PtIn/CeO2, indicating that adding only a small amount of Co did not significantly affect the inherent product distribution of PtIn/CeO2. All the catalysts except PtCo2In3/CeO2 were rapidly deactivated, highlighting that PtCo2In3/CeO2 not only showed superior activity in the dry reforming reaction but also had excellent stability. To identify the alloying effect of PtCo2In3/CeO2, we further tested the control experiments (Fig. S6) using physical mixtures of the binary and monometallic catalysts (Pt/CeO2 + Co–In/CeO2) and tandem catalytic systems with two sequential catalyst beds (Pt/CeO2 → glass wool → Co–In/CeO2 and CoIn/CeO2 → glass wool → Pt/CeO2). The physical mixture test showed lower catalytic activity than PtCo2In3/CeO2, while the tandem system exhibited lower stability, suggesting that the ternary alloy based on the pseudo-binary structure is essential for high catalytic performance. Then, we examined the long-term stability of PtCo2In3/CeO2, which showed a remarkably high stability: the conversion of C3H8 and CO2 did not decrease, even at 100 h, while maintaining ∼95% CO selectivity and ∼87% net CO selectivity. In contrast to conventional systems that require high temperatures or high CO2/propane ratios for considerable conversion (Table S2), the developed catalyst achieved a high propane consumption rate under mild, energy-efficient conditions. It maintained a high reaction rate while exhibiting excellent durability and coke resistance, addressing the challenges of low-temperature dry reforming. To verify the reason for the long-term stability, temperature-programmed oxidation (TPO, Fig. S7) and XRD analyses (Fig. S8) of the spent catalysts after 50 h catalytic run were conducted. As verified by O2-TPO of the fresh monometallic Pt, bimetallic PtIn, and PtCo2In3 catalysts (Fig. S7c), the O2 consumption of the fresh samples was below the detection limit, indicating that the O2 uptake and the corresponding CO2 evolution observed for the spent catalysts mainly reflect the combustion of deposited coke and are not dominated by the oxidation of intrinsic defects from the supports. The amount of coke shown in Fig. 3a was quantified by integrating the CO2 signal (Fig. S7b) throughout the entire TPO run. For clear comparison, the catalyst exhibiting the largest integrated TPO peak area (PtIn/CeO2) was defined as 100, and the integrated areas of the other catalysts were normalized to this value. The relative coke amount on PtCo2In3/CeO2 was much lower than that on Pt/CeO2, PtCo/CeO2, and PtIn/CeO2, even after 50 h reaction (Fig. 3a). On the other hand, the physical mixture of the binary and monometallic catalysts (Pt/CeO2 + Co–In/CeO2) also exhibited a quite low coke amount, which was only slightly higher than that of PtCo2In3/CeO2. On combining these results, we conclude that the close contact of Pt, Co, and In at an atomic level is necessary to minimize coke accumulation. A possible explanation is that the proximity of the three metal elements Pt–Co–In allows facile coupling of C and O atoms generated by C–C scission on the Pt–Co sites33 and by CO2 activation on the Co–In sites,27 respectively, at their interface.
3 at 550 °C. Fig. 2a–d show the time course of C3H8 conversion, CO2 conversion and the product distribution of both catalysts, respectively. The Pt2CoIn3/CeO2 showed a C3H6-rich product distribution that consisted of 60% C3H6, 39% CO, and the other C1–2 products, and C3H6 selectivity remained at 52% after a 10 h reaction. In contrast, the PtCo2In3/CeO2 catalyst was CO-dominant; CO selectivity was 89% at the initial stage and increased to 95% after a 10 h reaction. This suggests that PtCo2In3/CeO2 has a higher dry reforming activity, likely due to its strong propensity for C–C bond cleavage and subsequent oxygenation, whereas Pt2CoIn3/CeO2 preferentially produces C3H6via C–H bond cleavage and facile desorption. Differences were also observed in the C3H8 conversion trend over time. Although the initial C3H8 conversion was similar for PtCo2In3/CeO2 and Pt2CoIn3/CeO2 (∼16%, Fig. 2a), C3H8 conversion increased after 10 h of reaction and stabilized at ∼26% for PtCo2In3/CeO2, whereas it gradually decreased to ∼10% for Pt2CoIn3/CeO2. To understand the individual effect of each second metal, we also performed control experiments using the corresponding monometallic (Pt/CeO2) and bimetallic (PtCo/CeO2, Co-rich PtCo3/CeO2, PtIn/CeO2, and In-rich PtIn3/CeO2) catalysts (Fig. S5). C3H8 conversion for Pt/CeO2, PtCo/CeO2, and PtIn/CeO2 was 40%, 33%, and 28%, while the CO2 conversion was 38%, 40%, and 16%, respectively. The Co-rich PtCo3/CeO2 exhibited high initial conversions of both C3H8 and CO2, likely due to the strong C–C cleavage ability of the Co-rich PtCo alloy and/or the large amount of remaining Co species. In contrast, In-rich PtIn3/CeO2 showed low conversions of C3H8 and CO2, which may be attributed to the blocking of the active sites by unalloyed In species. These results show that alloying Pt with Co or In decreases C3H8 conversion due to the ensemble effect, while adding a certain amount of Co increases CO2 conversion. Furthermore, Pt2CoIn3/CeO2 displayed a similar product distribution to that of PtIn/CeO2, indicating that adding only a small amount of Co did not significantly affect the inherent product distribution of PtIn/CeO2. All the catalysts except PtCo2In3/CeO2 were rapidly deactivated, highlighting that PtCo2In3/CeO2 not only showed superior activity in the dry reforming reaction but also had excellent stability. To identify the alloying effect of PtCo2In3/CeO2, we further tested the control experiments (Fig. S6) using physical mixtures of the binary and monometallic catalysts (Pt/CeO2 + Co–In/CeO2) and tandem catalytic systems with two sequential catalyst beds (Pt/CeO2 → glass wool → Co–In/CeO2 and CoIn/CeO2 → glass wool → Pt/CeO2). The physical mixture test showed lower catalytic activity than PtCo2In3/CeO2, while the tandem system exhibited lower stability, suggesting that the ternary alloy based on the pseudo-binary structure is essential for high catalytic performance. Then, we examined the long-term stability of PtCo2In3/CeO2, which showed a remarkably high stability: the conversion of C3H8 and CO2 did not decrease, even at 100 h, while maintaining ∼95% CO selectivity and ∼87% net CO selectivity. In contrast to conventional systems that require high temperatures or high CO2/propane ratios for considerable conversion (Table S2), the developed catalyst achieved a high propane consumption rate under mild, energy-efficient conditions. It maintained a high reaction rate while exhibiting excellent durability and coke resistance, addressing the challenges of low-temperature dry reforming. To verify the reason for the long-term stability, temperature-programmed oxidation (TPO, Fig. S7) and XRD analyses (Fig. S8) of the spent catalysts after 50 h catalytic run were conducted. As verified by O2-TPO of the fresh monometallic Pt, bimetallic PtIn, and PtCo2In3 catalysts (Fig. S7c), the O2 consumption of the fresh samples was below the detection limit, indicating that the O2 uptake and the corresponding CO2 evolution observed for the spent catalysts mainly reflect the combustion of deposited coke and are not dominated by the oxidation of intrinsic defects from the supports. The amount of coke shown in Fig. 3a was quantified by integrating the CO2 signal (Fig. S7b) throughout the entire TPO run. For clear comparison, the catalyst exhibiting the largest integrated TPO peak area (PtIn/CeO2) was defined as 100, and the integrated areas of the other catalysts were normalized to this value. The relative coke amount on PtCo2In3/CeO2 was much lower than that on Pt/CeO2, PtCo/CeO2, and PtIn/CeO2, even after 50 h reaction (Fig. 3a). On the other hand, the physical mixture of the binary and monometallic catalysts (Pt/CeO2 + Co–In/CeO2) also exhibited a quite low coke amount, which was only slightly higher than that of PtCo2In3/CeO2. On combining these results, we conclude that the close contact of Pt, Co, and In at an atomic level is necessary to minimize coke accumulation. A possible explanation is that the proximity of the three metal elements Pt–Co–In allows facile coupling of C and O atoms generated by C–C scission on the Pt–Co sites33 and by CO2 activation on the Co–In sites,27 respectively, at their interface.
Mechanistic study
Next, a mechanistic study was conducted to elucidate the origin of the difference in selectivity and stability between PtCo2In3/CeO2 and Pt2CoIn3/CeO2 catalysts. The apparent activation energy (Fig. 3c and d) was evaluated using Arrhenius-type plots. Compared with the Pt2CoIn3/CeO2, the activation energies of PtCo2In3/CeO2 for C3H8 and CO2 decreased from 121.9 to 100.1 kJ mol−1 and 129.5 to 117.5 kJ mol−1, respectively. This suggests that adjusting the amount of Co in the PtIn crystal structure could reduce the activation barrier, which is consistent with the activity trend in the DRP reaction. Then, we performed a kinetic study on the CO2-DRP reaction over PtCo2In3/CeO2 at 550 °C within the Arrhenius temperature window. The reaction order of PC3H8 is positive for both rC3H8 and rCO2 (0.69 and 1.31; Fig. S9c and d), whereas that of PCO2 is negative for rCO2 (−0.47; Fig. S9a) and near-zero for rC3H8 (−0.02; Fig. S9b). This near-zero to negative dependence suggests that increasing PCO2 does not accelerate the rate in the investigated range and can even suppress CO2 consumption, which is consistent with competitive adsorption by CO2-derived surface species. This behavior suggests that the overall rate is primarily governed by steps associated with propane activation/dehydrogenation, whereas the influence of CO2 partial pressure on propane consumption rate is negligible under these conditions. To further determine the reason for the different selectivities of these two catalysts, temperature-programmed surface reactions (TPSRs) were conducted under various conditions, such as C3H6 desorption (C3H6-TPD), propylene oxidation by CO2 (C3H6 + CO2), and reverse water-gas shift reaction (RWGS, H2 + CO2). From the C3H6-TPD results (Fig. 3b), PtCo2In3 exhibited a broad desorption feature between −40 and 40 °C, whereas Pt2CoIn3 showed a single sharp peak at around −30 °C and almost complete desorption by this temperature. The broader and slightly higher-temperature desorption on PtCo2In3 suggests the stronger and more heterogeneous adsorption of C3H6, while Pt2CoIn3 binds C3H6 more weakly and thus favors the facile desorption of propylene once it is formed from propane dehydrogenation. TPD reflects adsorption stability under inert conditions; stronger C3H6 adsorption on PtCo2In3 therefore implies longer surface residence and a higher probability for subsequent surface reactions, whereas rapid desorption on P2tCoIn3 limits further transformation. To further elucidate the effect of Co content on the subsequent transformation of reaction intermediates, C3H6 + CO2 TPSR experiments were performed. C3H6 starts to be consumed at around 350 °C over PtCo2In3, whereas a higher temperature is required over Pt2CoIn3. This indicates that the strongly adsorbed C3H6 species on PtCo2In3 are more readily activated toward subsequent oxidation/reforming, leading to the higher formation of CO, H2O, and H2, which aligns well with the C3H6 selectivity trend. Moreover, the easier transformation of C3H6 over PtCo2In3 is accompanied by enhanced CO2 activation, as evidenced by the larger decrease in the m/z = 44 signal. Confirmed by the following H2 + CO2 TPSR experiments, PtCo2In3 shows slightly higher consumption of H2 and CO2 and higher production of CO and H2O than Pt2CoIn3 (Fig. 4b), pointing to a more efficient RWGS pathway. Overall, PtCo2In3 exhibits higher activity and selectivity for dry reforming than Pt2CoIn3, which can be attributed to the kinetically more favorable activation/dehydrogenation of C3H6 due to the stronger C3H6 adsorption capability, and the more efficient RWGS reaction by verifying the Co-content.Density functional theory (DFT) calculations were conducted to obtain atomic-scale insight into the selectivity switching trend between Pt2CoIn3 and PtCo2In3 catalysts depending on their specific surface structure. The Pt2In3(012) and PtIn2(111) planes were selected as the models of Pt2CoIn3 and PtCo2In3, respectively, because they are the most stable surfaces having high surface atomic densities, hence the strong diffraction as shown in Fig. 1b. The reaction environments of the Co-doped pseudo-binary alloy catalysts (Pt2CoIn3 and PtCo2In3) were modeled as Pt2In3(012)–Co and PtIn2(111)–Co, respectively, by replacing surface Pt atoms of the Pt2In3 (012) and PtIn2(111) slab models with Co atoms. Fig. 5a shows the transition state (TS) of the C3H6 decomposition to C3H5 + H on Pt2In3(012)–Co (left) and on PtIn2(111)–Co (right) surfaces. The difference in the energy barriers (ΔE = Ea − Ed) of C3H6 decomposition (Ea) and desorption (Ed) was used as a scale for evaluating the selectivity trend (Fig. 5b). Pt2In3(012)–Co exhibited high Ea and low Ed, thus providing large ΔE and favoring the dehydrogenation step. In contrast, PtIn2(111)–Co showed very low ΔE due to moderate Ea and high Ed, which easily induced the decomposition process leading to dry reforming. This trend agrees well with the experimental selectivity trend. The change in the surface structure originating from the bulk crystal structures plays a crucial role in determining the fate of C3H6—whether it desorbs to dehydrogenation or decomposes to reforming. The stronger adsorption capability of the PtCo2In3 surface suggests that dry reforming primarily occurs via C3H6 without changing the main reaction mechanism, while just altering the product distribution.
Conclusion
Overall, we have designed a highly efficient ternary PtCo2In3/CeO2 catalyst for the DRP reaction with high selectivity and stability, even for a 100 h reaction. The results showed that the catalytic behavior of PtCo2In3/CeO2 and Pt2CoIn3/CeO2 catalysts is highly sensitive to the doping amount of Co. Comprehensive structural characterization (X-ray techniques and TEM) showed that increasing the Co content drives an intermetallic phase evolution from Pt2In3 (P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1) to PtIn2 (Fm
m1) to PtIn2 (Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m). By controlling the amount of Co-doping, the catalytic properties of Pt–Co–In/CeO2 can be drastically switched from “CO2-ODP-dominant” to “DRP-specific”. TPSR experiments and DFT calculations further revealed that, compared with Pt2CoIn3/CeO2, PtCo2In3/CeO2 exhibits stronger adsorptivity of C3H6 and lower activation energy of CO2, thus facilitating the subsequent decomposition of propylene into CO. Herein, we report an excellent catalyst for DRP that opens up a new avenue for catalyst design concepts based on multi-metallic catalysts. The obtained insights and technology will contribute to the carbon-neutralization of industrial processes for light alkane conversion.
m). By controlling the amount of Co-doping, the catalytic properties of Pt–Co–In/CeO2 can be drastically switched from “CO2-ODP-dominant” to “DRP-specific”. TPSR experiments and DFT calculations further revealed that, compared with Pt2CoIn3/CeO2, PtCo2In3/CeO2 exhibits stronger adsorptivity of C3H6 and lower activation energy of CO2, thus facilitating the subsequent decomposition of propylene into CO. Herein, we report an excellent catalyst for DRP that opens up a new avenue for catalyst design concepts based on multi-metallic catalysts. The obtained insights and technology will contribute to the carbon-neutralization of industrial processes for light alkane conversion.
Experiments
Catalyst preparation
Pt/CeO2, Pt–Co/CeO2, Pt–In/CeO2, and Pt–Co–In/CeO2 (Pt: 3 wt%) were prepared by a conventional impregnation method using H2PtCl6 (aqueous solution, Kojima Chemicals, Pt 8.77 wt%), In(NO3)3·3H2O, and Co(NO3)2·6H2O, as metal precursors. The CeO2 support (JRC-CEO-2, SBET = 123.1 m2g−1) was added to a vigorously stirred aqueous solution (50 mL H2O per gram of CeO2) containing Pt and the corresponding second and/or third metal precursor(s) (Pt![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co = 1
Co = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, Pt
1, Pt![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) In = 1
In = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and Pt
1, and Pt![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co
Co![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) In = 1
In = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 or 2
3 or 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), followed by stirring for 3 h at room temperature. The mixture was dried under reduced pressure at 50 °C using a rotary evaporator, followed by calcination under flowing air at 500 °C for 1 h and reduction under flowing H2 (50 mL min−1) at 600 °C for 1 h. Co/CeO2, CoIn/CeO2, and In/CeO2 were prepared by a similar method, where the amounts of Co and In were adjusted to be equal to those included in Pt–Co–In/CeO2.
3), followed by stirring for 3 h at room temperature. The mixture was dried under reduced pressure at 50 °C using a rotary evaporator, followed by calcination under flowing air at 500 °C for 1 h and reduction under flowing H2 (50 mL min−1) at 600 °C for 1 h. Co/CeO2, CoIn/CeO2, and In/CeO2 were prepared by a similar method, where the amounts of Co and In were adjusted to be equal to those included in Pt–Co–In/CeO2.
Catalytic test
DRP was performed in a quartz fixed-bed reactor with an internal diameter of 6 mm under atmospheric pressure. The catalysts (0.10 g) were diluted with sea sand (0.90 g, Miyazaki Chemical, 99.9%) and treated under flowing hydrogen (10 mL min−1) at 550 °C for 0.5 h prior to the catalytic reactions. The catalysts were then evaluated by feeding the reactant gas mixture (C3H8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CO2
CO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) He = 1
He = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3
3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) with a total flow rate of 30 mL min−1. The 100 h long-time stability reaction was test by feeding reactant gas mixture (C3H8
2) with a total flow rate of 30 mL min−1. The 100 h long-time stability reaction was test by feeding reactant gas mixture (C3H8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CO2
CO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) He = 1
He = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3
3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 26) at the same total flow rate. The gas phase was analyzed and quantified using an online thermal conductivity detection gas chromatograph (Shimadzu GC-8A, column: Gaskuropack 54) equipped downstream. For all the catalysts, C3H6, C2H4, C2H6, CH4, CO, CO2, H2O, and H2 were detected as reaction products in outlet gas.
26) at the same total flow rate. The gas phase was analyzed and quantified using an online thermal conductivity detection gas chromatograph (Shimadzu GC-8A, column: Gaskuropack 54) equipped downstream. For all the catalysts, C3H6, C2H4, C2H6, CH4, CO, CO2, H2O, and H2 were detected as reaction products in outlet gas. | (1) |
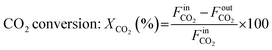 | (2) |
In this reaction, CO can be generated from CxHy species via dry reforming as well as from the oxidative dehydrogenation of propane. To distinguish between these pathways, we have defined two separate expressions for CO selectivity.
Product selectivity, including CO selectivity, is defined on a carbon number basis with respect to product-derived carbon:
 | (3) |
 | (4) |
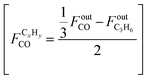 .
.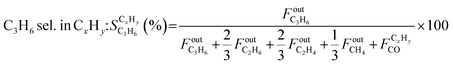 | (5) |
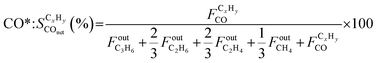 | (6) |
 | (7) |
Characterization
The crystal structure of the prepared catalyst was examined via powder X-ray diffraction (XRD) using a Rigaku MiniFlex II/AP diffractometer with Cu Kα radiation. High-angle annular dark field scanning transmission electron microscopy (HAADF-STEM) was carried out using a JEOL JEM-ARM200 M microscope equipped with an energy dispersive X-ray (EDX) analyzer (EX24221M1G5T). STEM analysis was performed at an accelerating voltage of 200 kV. To prepare the TEM specimen, all samples were sonicated in ethanol and then dispersed on a Mo grid supported by an ultrathin carbon film.The TPO experiment was conducted using BELCAT II (MicrotracBEL) to quantify the amount of coke deposited on the spent catalysts after 50 h of DRP at 550 °C (0.1 g of the catalyst with 0.9 g quartz sand). The spent catalyst was placed in a quartz tube reactor and treated under flowing He (30 mL min−1) at 150 °C for 30 min, followed by cooling to room temperature. The catalyst bed temperature was then increased (40–900 °C, ramping rate: 5 °C min−1) under flowing O2/He (2%, 50 mL min−1). The amount of CO2 in the outlet gas was quantified using an online mass spectrometer. C3H6 and CO2 temperature-programmed desorption were performed. Prior to the C3H6-TPD, the as-reduced catalyst (100 mg) was heated to 600 °C under a 5%H2/Ar gas mixture (20 mL min−1) with a ramping rate of 20 °C min−1, then kept at the same temperature for 0.5 h. After the reduction, the catalyst was cooled to −35 °C using CATCryo-II under a He flow (20 mL min−1), kept at −35 °C for 0.5 h, and subsequently exposed to 5% C3H6/He gas mixture (20 mL min−1) at the same temperature for 5 min. The catalyst was then purged under He (50 mL min−1) at −35 °C for 1.5 h, and subsequently heated from −35 °C to 300 °C with a ramping rate of 2 °C min−1. The outlet gas (C3H6; m/z = 41) was analyzed online using a quadrupole mass spectrometer (BELMASS). For C3H6 + CO2-TPSR, the catalyst was pre-treated under a 5%H2/Ar gas mixture (20 mL min−1) at 600 °C, purged under He (50 mL min−1) at 50 °C for 0.5 h, and subsequently heated from 50 °C to 600 °C at a ramping rate of 5 °C min−1 with C3H6 + CO2 gases (5 + 15 mL min−1) flowing downstream of the catalyst bed. The outlet gas (CO2; m/z = 44, C3H6; m/z = 41, CO; m/z = 28, H2O; m/z = 18, and H2; m/z = 2) was analyzed online by a quadrupole mass spectrometer (MicrotracBEL, BELMASS).
XPS (Kratos-ULTRA2, Shimadzu) analysis was conducted using an air-isolated sample vessel and an apparatus equipped with a charge neutralization system. The binding energy was corrected against the signal of the C 1s orbital (C 1s = 284.6 eV). Before the measurement, the catalyst was pretreated with H2 (30 mL min−1) at 550 °C for 2 h in a quartz tube, followed by cooling to room temperature, purging with Ar (10 mL min−1) and storing in the glovebox. X-ray absorption fine structure (XAFS) spectra of the prepared catalysts were obtained at the BL01B14 beamline of SPring-8, Japan Synchrotron Radiation Research Institute (JASRI), using Si(111) (for Co K- and Pt LIII-edges) and Si(311) (for In K-edge) double-crystals as monochromators. Prior to the measurement, the catalyst was pelletized (ca. 150 mg with a diameter of 10 mm) and pretreated with H2/N2 (20%, 40 mL min−1) at 550 °C for 0.5 h in an in situ quartz cell, followed by cooling to room temperature with N2 purge (50 mL min−1). The XAFS spectra were recorded in transmission (In K-edge) and fluorescence (Pt LIII-, and Co K-edge: using a 19-element Ge solid-state detector) modes at room temperature. Athena and Artemis software ver. 0.9.25, implemented in the Demeter package, were used for the analysis of the obtained XAFS spectra. FEFF8 was used for the calculation of the back-scattering amplitude and phase shift functions.34 We defined the R-factor (R2) for curve-fitting as follows: 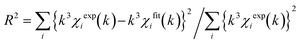 .
.
Computational details
Periodic DFT calculations were performed using the CASTEP code35 with Vanderbilt-type ultrasoft pseudopotentials, as well as the revised version of the Perdew–Burke–Ernzerhof exchange–correlation functional based on the generalized gradient approximation.36 At a kinetic energy of 360 eV, the plane-wave basis set was truncated. A 0.1 eV Fermi smearing was utilized. The Tkatchenko–Scheffler method was used to analyze dispersion correlations with a scaling coefficient of sR = 0.94 and a damping parameter of d = 20.37 The reciprocal space was sampled using a k-point mesh with a spacing of typically 0.04 Å−1, as generated by the Monkhorst–Pack scheme.38 Geometry optimizations and transition state (TS) searches were performed on supercell structures using periodic boundary conditions. The surfaces were modeled using metallic slabs with a thickness of four atomic layers with 13 Å of vacuum spacing. Geometry optimizations were performed using the two-point steepest descent (TPSD) algorithm.39 We chose Pt2In3(012) and PtIn2(111) as the most stable surfaces (Pt2CoIn3 and PtCo2In3, respectively), which have high surface atom density and diffraction intensity.40,41 Pt2In3(012)–Co and PtIn2(111)–Co were constructed as models of the surface pseudo binary alloy by replacing one of the surface Pt atoms of Pt2In3(012) and PtIn2(111)–Co. The unit cell sizes of the bulk materials (Pt2In3(012)–Co and PtIn2(111)–Co) were first optimized, followed by modeling the slab structure and surface relaxation with the size of the supercell fixed. The convergence criteria for structure optimization and energy calculation were set to (a) an SCF tolerance of 1.0 × 10−6 eV per atom, (b) an energy tolerance of 1.0 × 10−5 eV per atom, (c) a maximum force tolerance of 0.05 eV Å−1, and (d) a maximum displacement tolerance of 1.0 × 10−3 Å.The adsorption energy was defined as follows: Ead = EA-S − (ES + EA), where EA-S is the energy of the slab together with the adsorbate, EA is the total energy of the free adsorbate, and ES is the total energy of the bare slab. The adsorption energy for an oxygen-preadsorbed slab was calculated using ESH, which is the total energy of the oxygen-adsorbed slab, instead of using ES.
The transition state (TS) search was performed using the complete linear synchronous transit/quadratic synchronous transit (LST/QST) method.42,43 Linear synchronous transit maximization was performed, followed by energy minimization in the directions conjugate to the reaction pathway. The approximated TS was used to perform QST maximization with conjugate gradient minimization refinements. This cycle was repeated until a stationary point was found. Convergence criteria for the TS calculations were set to root-mean-square forces on an atom tolerance of 0.05 eV Å−1.
Conflicts of interest
The authors declare no competing interests.Data availability
All data supporting the findings of this study are available within the article and its supplementary information (SI).Supplementary information is available. See DOI: https://doi.org/10.1039/d6cy00231e
Acknowledgements
This work was supported by JSPS KAKENHI (Grant Numbers 22KJ0077, 22J11748, 17H01341, 17H04965, 25K23522, and 20H02517) and JST PRESTO (JPMJPR19T7). The XAFS analysis was performed with the approval of JASRI (No. 2021B1962 and 2022A0302). We appreciate the technical staffs of the Faculty of Engineering, Hokkaido University, and of the Research Institute for Electronic Science, Hokkaido University, for their assistance with HAADF-STEM observation.References
- D. Pakhare and J. Spivey, A Review of Dry (CO2) Reforming of Methane over Noble Metal Catalysts, Chem. Soc. Rev., 2014, 43(22), 7813–7837 RSC.
- L. Zhong, F. Yu, Y. An, Y. Zhao, Y. Sun, Z. Li, T. Lin, Y. Lin, X. Qi, Y. Dai, L. Gu, J. Hu, S. Jin, Q. Shen and H. Wang, Cobalt Carbide Nanoprisms for Direct Production of Lower Olefins from Syngas, Nature, 2016, 538(7623), 84–87 CrossRef CAS PubMed.
- S. Ma, S. d. Huang and Z. P. Liu, Dynamic Coordination of Cations and Catalytic Selectivity on Zinc–Chromium Oxide Alloys during Syngas Conversion, Nat. Catal., 2019, 2(8), 671–677 CrossRef CAS.
- F. Jiao, J. Li, X. Pan, J. Xiao, H. Li, H. Ma, M. Wei, Y. Pan, Z. Zhou, M. Li, S. Miao, J. Li, Y. Zhu, D. Xiao, T. He, J. Yang, F. Qi, Q. Fu and X. Bao, Selective Conversion of Syngas to Light Olefins Downloaded From, Science, 2016, 351(6277), 1065 CrossRef CAS PubMed.
- C. Palmer, D. C. Upham, S. Smart, M. J. Gordon, H. Metiu and E. W. McFarland, Dry Reforming of Methane Catalysed by Molten Metal Alloys, Nat. Catal., 2020, 3(1), 83–89 CrossRef CAS.
- Y. Song, E. Ozdemir, S. Ramesh, A. Adishev, S. Subramanian, A. Harale, M. Albuali, B. A. Fadhel, A. Jamal, D. Moon, S. H. Choi and C. T. Yavuz, Dry Reforming of Methane by Stable Ni–Mo Nanocatalysts on Single-Crystalline MgO, Science, 2020, 367(6479), 777–781 CrossRef CAS PubMed.
- M. Akri, S. Zhao, X. Li, K. Zang, A. F. Lee, M. A. Isaacs, W. Xi, Y. Gangarajula, J. Luo, Y. Ren, Y. T. Cui, L. Li, Y. Su, X. Pan, W. Wen, Y. Pan, K. Wilson, L. Li, B. Qiao, H. Ishii, Y. F. Liao, A. Wang, X. Wang and T. Zhang, Atomically Dispersed Nickel as Coke-Resistant Active Sites for Methane Dry Reforming, Nat. Commun., 2019, 10, 5181 CrossRef PubMed.
- F. Cheng, X. Duan and K. Xie, Dry Reforming of CH4/CO2 by Stable Ni Nanocrystals on Porous Single-Crystalline MgO Monoliths at Reduced Temperature, Angew. Chem., Int. Ed., 2021, 60(34), 18792–18799 CrossRef CAS PubMed.
- F. Solymosi, P. Tolmacsov and T. S. Zakar, Dry Reforming of Propane over Supported Re Catalyst, J. Catal., 2005, 233(1), 51–59 CrossRef CAS.
- B. Yan, X. Yang, S. Yao, J. Wan, M. N. Z. Myint, E. Gomez, Z. Xie, S. Kattel, W. Xu and J. G. Chen, Dry Reforming of Ethane and Butane with CO2 over PtNi/CeO2 Bimetallic Catalysts, ACS Catal., 2016, 6(11), 7283–7292 CrossRef CAS.
- X. Li, B. Yan, S. Yao, S. Kattel, J. G. Chen and T. Wang, Oxidative Dehydrogenation and Dry Reforming of N-Butane with CO2 over NiFe Bimetallic Catalysts, Appl. Catal., B, 2018, 231, 213–223 CrossRef CAS.
- E. Gomez, Z. Xie and J. G. Chen, The Effects of Bimetallic Interactions for CO2-Assisted Oxidative Dehydrogenation and Dry Reforming of Propane, AIChE J., 2019, 65(8), 1–12 CrossRef.
- L. B. Råberg, M. B. Jensen, U. Olsbye, C. Daniel, S. Haag, C. Mirodatos and A. O. Sjåstad, Propane Dry Reforming to Synthesis Gas over Ni-Based Catalysts: Influence of Support and Operating Parameters on Catalyst Activity and Stability, J. Catal., 2007, 249(2), 250–260 CrossRef.
- J. Dong, Y. Song, S. Huang, P. Chang and Z. Lai, Dry Reforming of Propane over PtNi Bimetallic Catalysts to Produce Syngas, ChemistrySelect, 2025, 10(34), e03739 CrossRef CAS.
- F. Solymosi, P. Tolmacsov and K. Kedves, CO2 Reforming of Propane over Supported Rh, J. Catal., 2003, 216(1–2), 377–385 CrossRef CAS.
- D. Sutton, J.-F. Moisan and J. R. H. Ross, Kinetic Study of CO2 Reforming of Propane over Ru/Al2O3, Catal. Lett., 2001, 75(3–4), 175–181 CrossRef CAS.
- J. Niu, Y. Wang, S. E. Liland, S. K. Regli, J. Yang, K. R. Rout, J. Luo, M. Rønning, J. Ran and D. Chen, Unraveling Enhanced Activity, Selectivity, and Coke Resistance of Pt-Ni Bimetallic Clusters in Dry Reforming, ACS Catal., 2021, 11(4), 2398–2411 CrossRef CAS.
- A. Siahvashi and A. A. Adesina, Synthesis Gas Production via Propane Dry(CO2) Reforming: Influence of Potassium Promotion on Bimetallic Mo-Ni/Al2O3, Catal. Today, 2013, 214, 30–41 CrossRef CAS.
- A. Siahvashi, D. Chesterfield and A. A. Adesina, Propane CO2 (Dry) Reforming over Bimetallic Mo-Ni/Al2O3 Catalyst, Chem. Eng. Sci., 2013, 93, 313–325 CrossRef CAS.
- A. Olafsen, C. Daniel, Y. Schuurman, L. B. Råberg, U. Olsbye and C. Mirodatos, Light Alkanes CO2 Reforming to Synthesis Gas over Ni Based Catalysts, Catal. Today, 2006, 115(1–4), 179–185 CrossRef CAS.
- M. S. P. Sudhakaran, M. M. Hossain, G. Gnanasekaran and Y. S. Mok, Dry Reforming of Propane over γ-Al2O3 and Nickel Foam Supported Novel SrNiO3 Perovskite Catalyst, Catalysts, 2019, 9(68), 1–18 Search PubMed.
- K. Liu, F. Xing, Y. Xiao, N. Yan, K. I. Shimizu and S. Furukawa, Development of a Highly Stable Ternary Alloy Catalyst for Dry Reforming of Methane, ACS Catal., 2023, 13(6), 3541–3548 CrossRef CAS.
- M. B. Jensen, L. B. Råberg, A. Olafsen Sjåstad and U. Olsbye, Mechanistic Study of the Dry Reforming of Propane to Synthesis Gas over a Ni/Mg(Al)O Catalyst, Catal. Today, 2009, 145(1–2), 114–120 CrossRef CAS.
- F. Barzegari, M. Rezaei, M. Kazemeini, F. Farhadi and A. R. Keshavarz, Effect of Rare-Earth Promoters(Ce, La, Y and Zr) on the Catalytic Performance of NiO-MgO-SiO2 Catalyst in Propane Dry Reforming, Mol. Catal., 2022, 522(112235), 1–13 Search PubMed.
- M. Ahadzadeh, S. M. Alavi, M. Rezaei and E. Akbari, Propane Dry Reforming over Highly Active NiO-MgO Solid Solution Catalyst for Synthesis Gas Production, Mol. Catal., 2022, 524(112325), 1–11 Search PubMed.
- F. Xing, J. Jeon, T. Toyao, K. I. Shimizu and S. Furukawa, A Cu-Pd Single-Atom Alloy Catalyst for Highly Efficient NO Reduction, Chem. Sci., 2019, 10(36), 8292–8298 RSC.
- F. Xing, Y. Nakaya, S. Yasumura, K. Shimizu and S. Furukawa, Ternary Platinum–Cobalt–Indium Nanoalloy on Ceria as a Highly Efficient Catalyst for the Oxidative Dehydrogenation of Propane Using CO2, Nat. Catal., 2022, 5(1), 55–65 CrossRef CAS.
- F. Xing and S. Furukawa, Metallic Catalysts for Oxidative Dehydrogenation of Propane Using CO2, Chem. – Eur. J., 2023, 29(3), e202202173 CrossRef CAS PubMed.
- Y. Nakaya and S. Furukawa, Catalysis of Alloys: Classification, Principles, and Design for a Variety of Materials and Reactions, Chem. Rev., 2023, 123(9), 5859–5947 CrossRef CAS PubMed.
- J. Dong, D. Li, Y. Zhang, P. Chang and Q. Jin, Insights into the CeO2 Facet-Depended Performance of Propane Oxidation over Pt-CeO2 Catalysts, J. Catal., 2022, 407, 174–185 CrossRef CAS.
- J. Dong, Y. Zhang, D. Li, A. Adogwa, S. Huang, M. Yang, J. Yang and Q. Jin, Reaction-Driven Evolutions of Pt States over Pt-CeO2 Catalysts during CO Oxidation, Appl. Catal., B, 2023, 330, 122662 CrossRef CAS.
- H. Lei, N. Zhang, S. Hu, F. Peng, J. Zhou, J. He, L. Zhang, H. Wang, C. Ma, H. Yan, K. Shimizu and J. Zeng, Thermally Triggered Redox Flexibility of Pt/CeO2 Cluster Catalyst Against In-Situ Atomic Redispersion, Angew. Chem., 2025, 64(31), e202509239 CrossRef CAS PubMed.
- Á. Szamosvölgyi, Á. Pitó, A. Efremova, K. Baán, B. Kutus, M. Suresh, A. Sápi, I. Szenti, J. Kiss, T. Kolonits, Z. Fogarassy, B. Pécz, Á. Kukovecz and Z. Kónya, Optimized Pt-Co Alloy Nanoparticles for Reverse Water-Gas Shift Activation of CO2, ACS Appl. Nano Mater., 2024, 7(9), 9968–9977 CrossRef PubMed.
- A. Ankudinov and B. Ravel, Real-Space Multiple-Scattering Calculation and Interpretation of x-Ray-Absorption near-Edge Structure, Phys. Rev. B: Condens. Matter Mater. Phys., 1998, 58(12), 7565–7576 CrossRef CAS.
- M. D. Segall, P. J. D. Lindan, M. J. Probert, C. J. Pickard, P. J. Hasnip, S. J. Clark and M. C. Payne, First-Principles Simulation: Ideas, Illustrations and the CASTEP Code, J. Phys.: Condens. Matter, 2002, 14, 2717–2744 CrossRef CAS.
- B. Hammer, L. B. Hansen and J. K. Nørskov, Improved Adsorption Energetics within Density-Functional Theory Using Revised Perdew-Burke-Ernzerhof Functionals, Phys. Rev. B: Condens. Matter Mater. Phys., 1999, 59(11), 7413–7421 CrossRef.
- A. Tkatchenko and M. Scheffler, Accurate Molecular van Der Waals Interactions from Ground-State Electron Density and Free-Atom Reference Data, Phys. Rev. Lett., 2009, 102(7), 073005 CrossRef PubMed.
- K. Hu, M. Wu, S. Hinokuma, T. Ohto, M. Wakisaka, J. I. Fujita and Y. Ito, Boosting Electrochemical Water Splitting: Via Ternary NiMoCo Hybrid Nanowire Arrays, J. Mater. Chem. A, 2019, 7(5), 2156–2164 RSC.
- J. Barzilai and J. M. Borwein, Two-Point Step Size Gradient Methods, IMA J. Numer. Anal., 1988, 8, 141–148 CrossRef.
- R. Jana and S. C. Peter, One-Pot Solvothermal Synthesis of Ordered Intermetallic Pt2In3 as Stable and Efficient Electrocatalyst towards Direct Alcohol Fuel Cell Application, J. Solid State Chem., 2016, 242, 133–139 CrossRef CAS.
- J. Ye, C. Liu and Q. Ge, A DFT Study of Methanol Dehydrogenation on the PdIn(110) Surface, Phys. Chem. Chem. Phys., 2012, 14(48), 16660–16667 RSC.
- N. Govind, M. Petersen, G. Fitzgerald, D. King-Smith and J. Andzelm, A Generalized Synchronous Transit Method for Transition State Location, Comput. Mater. Sci., 2003, 28(2), 250–258 CrossRef CAS.
- T. A. Halgren and W. N. Lipscomb, The Synchronous-Transit Method for Determining Reaction Pathways and Locating Molecular Transition States, Chem. Phys. Lett., 1977, 49(2), 225–232 CrossRef CAS.
Footnotes |
| † Present address: MDX Research Center for Element Strategy, Institute of Integrated Research, Institute of Science Tokyo, Yokohama 226-8503, Japan. |
| ‡ Present address: Division of Applied Chemistry, Graduate School of Engineering, Osaka University, Osaka 565-0871, Japan. |
| This journal is © The Royal Society of Chemistry 2026 |





