 Open Access Article
Open Access ArticleImportance of consistent and well-dispersed catalyst datasets for machine learning in oxidative coupling of methane
Hinata Sudo a,
Yoshiki Hasukawa
a,
Yoshiki Hasukawa a,
Rensuke Koiwai
a,
Rensuke Koiwai a,
Fernando Garcia-Escobar
a,
Fernando Garcia-Escobar a,
Shun Nishimura
a,
Shun Nishimura b,
Lauren Takahashi
b,
Lauren Takahashi *a and
Keisuke Takahashi
*a and
Keisuke Takahashi *ac
*ac
aDepartment of Chemistry, Hokkaido University, North 10, West 8, Sapporo 060-0810, Japan. E-mail: lauren.takahashi@sci.hokudai.ac.jp; keisuke.takahashi@sci.hokudai.ac.jp
bGraduate School of Advanced Science and Technology, Japan Advanced Institute of Science and Technology, Nomi, Ishikawa 923-1292, Japan
cList Sustainable Digital Transformation Catalyst Collaboration Research Platform, Institute for Chemical Reaction Design and Discovery, Hokkaido University, Sapporo 001-0021, Japan
First published on 13th April 2026
Abstract
The role of highly uniform, diverse experimental data in catalyst informatics is examined using an oxidative coupling of methane dataset measured by a single researcher under consistent devices and conditions. Broad compositional coverage and minimized experimental variability enable machine learning to capture composition–performance relationships using simple one-hot encoding. Inverse analysis of the compositional space identifies promising catalysts for experimental validation. These results demonstrate that carefully curated, well-distributed datasets, even if relatively small, enable machine learning to effectively capture composition–performance relationships.
Introduction
Catalyst informatics aims to accelerate catalyst design by identifying trends and patterns embedded in catalyst datasets.1,2 Among the available approaches, machine learning has emerged as a powerful tool for extracting structure–performance relationships and guiding catalyst discovery, provided that sufficiently large and informative datasets are available.3–7 However, the application of machine learning to heterogeneous catalysis faces several fundamental challenges. The first is the limited availability and quality of catalyst data. This issue has been addressed through the compilation of literature datasets, the use of high-throughput experimentation to generate large-scale catalyst data, or the development of methods tailored to small datasets.8–11 Each strategy, however, has inherent limitations. Literature-derived datasets are often poorly dispersed, as researchers preferentially report successful results, while unsuccessful or so-called negative data are rarely disclosed. This lack of data diversity is a bottleneck not only for methane activation, but also across the broader landscape of carbon utilization, where high-quality data are essential for optimizing carbon efficiency in various thermochemical processes.12 Furthermore, as the field matures, machine-learning workflows are increasingly being integrated with techno-economic and life cycle assessments to ensure that catalyst discovery aligns with broader sustainability goals.13 Under these circumstances, high-throughput experimentation can mitigate both data scarcity and poor dispersion by systematically exploring broad catalyst spaces.14,15 Nevertheless, high-throughput approaches also have intrinsic drawbacks, as catalysts are typically evaluated under uniform experimental conditions, which may obscure catalyst-specific optimal operating windows and mask intrinsic structure–performance relationships. A second critical challenge lies in the definition of catalyst descriptors for supervised machine learning. The construction of effective catalyst descriptors remains a long-standing problem, as there is no universally accepted representation of heterogeneous catalysts.16,17 One-hot encoding, a commonly used representation, is particularly problematic for poorly dispersed or unevenly distributed datasets, as it treats chemically related compositions as independent categories and prevents the model from learning meaningful trends for sparsely sampled catalysts. An alternative strategy is to represent catalysts using physically meaningful quantities derived from periodic table properties, which has been shown to improve both predictive performance and interpretability.18 Consequently, both the quality of catalyst datasets and the choice of catalyst descriptors are central to the successful application of machine learning in catalyst informatics.In this work, the impact of uniformly generated and well-dispersed catalyst datasets produced by a single researcher and same experimental devices and environment is systematically investigated to elucidate how such data quality influences machine-learning performance. The oxidative coupling of methane (OCM) reaction is selected as a prototypical system. OCM aims at the direct conversion of methane into C2 hydrocarbons, primarily C2H4 and C2H6.19–21 The dataset used in this study is derived from previous work in which all catalysts are evaluated by a single researcher under strictly controlled experimental conditions, including identical experimental setups and standardized operating procedures.22 By minimizing experimental variability while maintaining broad compositional diversity, this dataset provides an ideal platform for catalyst informatics. Using this dataset, comprehensive data analysis and supervised machine-learning approaches are applied to uncover structure–performance relationships and to guide catalyst design for the OCM reaction.
Data and methods
Published OCM data are used in this work.22 The dataset is generated by a single researcher using identical experimental devices and a consistent experimental environment. It consists of 2928 data points, including non-catalytic conditions, support-only catalysts, and binary and ternary oxide catalysts. In detail, the dataset includes 25 elements as active catalyst components (Li, Na, Mg, K, Ca, Ti, Mn, Zn, Rb, Sr, Y, Zr, Mo, Sn, Cs, Ba, La, Ce, Nd, Sm, Eu, Hf, W, Pb and Bi) and 8 oxides as supports (BaO, CaO, MgO, SiO2, TiO2, ZnO, Y2O3 and La2O3). Reaction temperature, the CH4/O2 gas flow ratio and furnace length are also included as experimental variables. The C2 yield, defined as the sum of C2H4 and C2H6 yields, is used as the objective variable. Unless otherwise stated, the CH4/O2 ratio is fixed at 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and the furnace length is fixed at 270 mm. Furthermore, for comparison, preprocessed literature data are also ultilized.22 The physical quantities used for the catalyst descriptors are from the XenonPy library.23
1, and the furnace length is fixed at 270 mm. Furthermore, for comparison, preprocessed literature data are also ultilized.22 The physical quantities used for the catalyst descriptors are from the XenonPy library.23
Data preprocessing is performed prior to data analysis and machine learning. Data points exhibiting negative O2 or CH4 conversion values are removed. In addition, data points with selectivities exceeding 100% for H2, CO, CO2, C2H4, or C2H6 are excluded due to physical inconsistency and experimental noise. To eliminate the influence of varying experimental conditions and to focus on catalyst and support effects, data collected at 700 °C with a CH4/O2 ratio of 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and a furnace length of 270 mm are extracted for machine-learning analysis. Furthermore, only binary and ternary catalyst compositions are retained. As a result, the number of data points is reduced to 2124. The catalyst composition and support are represented using one-hot encoding.
1 and a furnace length of 270 mm are extracted for machine-learning analysis. Furthermore, only binary and ternary catalyst compositions are retained. As a result, the number of data points is reduced to 2124. The catalyst composition and support are represented using one-hot encoding.
Supervised machine learning is performed using random forest regression (RFR) implemented in the scikit-learn.24 The random state is fixed, and the number of trees is set to 100. Model performance is evaluated by cross-validation, in which the dataset is randomly split into 80% training and 20% test sets. The reported performance corresponds to the average R2 score of the test data obtained from 10 independent train–test splits.
Catalyst synthesis
The catalysts listed in Table 1 are prepared by the impregnation method. Aqueous solutions containing metal precursors are impregnated onto a support, followed by drying and calcination to disperse the metal species on the support surface. Lanthanum oxide (La2O3, ≥99.5%, Junsei Chemical Co., Ltd.) is used as the catalyst support. Sodium nitrate (NaNO3, ≥99.0%), magnesium nitrate hexahydrate (Mg(NO3)2·6H2O, ≥99.9%), calcium nitrate tetrahydrate (Ca(NO3)2·4H2O, ≥98.5%), barium nitrate (Ba(NO3)2, ≥99.0%), and lanthanum nitrate hexahydrate (La(NO3)3·6H2O) (FUJIFILM Wako Pure Chemical Corporation) are used as metal precursors.| Catalyst | Support | M1 | M2 | M3 |
|---|---|---|---|---|
| NaBaMg/La2O3 | La2O3 | NaNO3 | Mg(NO3)2·6H2O | Ba(NO3)2 |
| NaCaBa/La2O3 | La2O3 | NaNO3 | Ca(NO3)2·4H2O | Ba(NO3)2 |
| NaBaLa/La2O3 | La2O3 | NaNO3 | Ba(NO3)2 | La(NO3)3·6H2O |
For catalyst preparation, 2 g of La2O3 is added to 100 mL of deionized water. Metal nitrate salts are dissolved in 50 mL of deionized water so that the total molar fraction of the added metals is 3%. The metal precursor solution is added to the La2O3 suspension in the order M1, M2, and M3 at 5 min intervals under stirring, followed by stirring for 60 min and aging overnight at room temperature. The mixture is then heated under stirring to remove water. After drying at 80 °C for 8 h, the obtained solid is ground into a fine powder and calcined at 800 °C for 3 h, with heating and cooling rates of 800 °C h−1. A reference catalyst consisting of La2O3 only is prepared following the same procedure.
Catalyst testing
Oxidative coupling of methane is carried out in a continuous-flow fixed-bed reactor. An alumina tubular reactor (length: 370 mm, inner diameter: 6.0 mm) is used, and 75 mg of catalyst is placed between two layers of quartz wool (1 mg each). The reactor temperature is monitored using a K-type thermocouple located near the catalyst bed. Before the reaction, the reactor is purged with N2 at 200 °C for 30 min and then cooled to room temperature.Catalytic performance is evaluated using a CH4/O2/N2 gas mixture with flow rates of 8.0/4.0/16.0 mL min−1 at 600, 650, 675, 700, 725, 750, 800, and 850 °C. At each temperature, the reaction is conducted for 10 min before gas sampling. Reaction products are analyzed using a Shimadzu GC-2014 gas chromatograph equipped with a SHINCARBON ST 50/80 column. Conversions of CH4 and O2, as well as yields of CO, CO2, C2H4, C2H6, and C2 products and C2 selectivity, are calculated using N2 as an internal standard according to eqn (1)–(4). In eqn (1) and (2), in and out represent the inlet feed and outlet effluent streams, respectively, which are used to calculate the conversion of reagents (R) such as CH4 and O2. In eqn (2), n is equal to 1 for CO and CO2 and 2 for C2H4 and C2H6.
 | (1) |
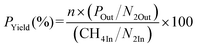 | (2) |
| C2yield = C2H4Yield + C2H6Yield | (3) |
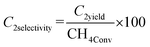 | (4) |
Results and discussion
OCM data are statistically analyzed. In particular, the distributions of catalyst elements, supports, and C2 yield are visualized in Fig. 1. Fig. 1 demonstrates that the catalyst composition and support materials are broadly and uniformly distributed across the dataset, while the C2 yield spans from 0% to high-yield regimes. Such wide coverage in both chemical composition and performance spaces is essential for machine-learning modeling, as it minimizes sampling bias, reduces extrapolation risk, and enables robust learning of composition–performance relationships. The well-dispersed nature of the dataset ensures that the model is trained on diverse catalytic environments, thereby improving generalizability and predictive reliability.As shown in Fig. 2(a), a pairwise correlation map is constructed between CH4 conversion, C2 yield, and selectivity, and the presence of catalyst elements and supports at 700 °C. Additionally, the feature importance of these catalyst elements and supports toward C2 yield is evaluated. Fig. 2(a) indicates that the La2O3 support exhibits a positive correlation with both CH4 conversion and C2 yield, suggesting that La2O3 is an active support under OCM reaction conditions. In a similar manner, certain alkaline earth elements, such as Sr and Ca, show weak but positive correlations with C2 yield, implying a potential promoting effect on C2 formation. Note that while Fig. 2(b) and (c) present the feature importance obtained from the random forest model, a high importance score does not necessarily imply a positive impact on the target variable. Random forest importance analysis of elements and supports is also performed for C2 yield as shown in Fig. 2(b) and (c), respectively. Fig. 2(b) reveals that Ca has the most predominant importance for C2 yield, followed by other alkaline earth metals such as Sr and Ba. Combining these results with the pairwise correlations in Fig. 2(a), it can be inferred that these specific alkaline earth elements exert a beneficial effect on C2 formation. Furthermore, although W also exhibits relatively high importance, its negative correlation in Fig. 2(a) suggests a deleterious effect on C2 yield. Similarly, Fig. 2(c) shows that La2O3 possesses the highest importance, which, consistent with Fig. 2(a), indicates its significant role in enhancing catalytic performance. While MgO also shows relatively high importance and a certain trend in Fig. 2(a), the underlying factors behind its contribution are discussed in detail in the following section in conjunction with the results shown in Fig. 3.
To evaluate the overall influence of catalyst elements and supports on C2 yield, violin plots are constructed. The distributions of C2 yield as a function of individual elements and supports are shown in Fig. 3. Fig. 3 indicates that catalysts containing Sr and Ca tend to exhibit higher C2 yields, consistent with the positive correlations observed in the pairwise correlation map as shown in Fig. 2. In addition, elements such as La and Mg are associated with relatively high C2 yields, whereas Cs, Bi, and Pb are generally linked to lower C2 yields. Notably, the interpretation is nontrivial because catalytic performance strongly depends on elemental combinations. As shown in Fig. 3(a), elements such as K, Na, Mn, and W are associated with both high and low C2 yields, depending on their pairing with other elements. This highlights the importance of combination effects rather than single-element contributions. Similarly, Fig. 3(b) shows that La2O3 supports tend to result in higher C2 yields, consistent with the analysis in Fig. 2. The catalytic performance may be attributed to the formation of subsurface peroxide species acting as active oxygen centers, which remain stable even at high temperatures.25 According to microkinetic analysis, these stable surface oxygen sites are suggested to promote methane dissociation and subsequent methyl radical formation, thereby facilitating gas-phase C2 coupling reactions.26 In a similar manner, Fig. 3(b) also indicates that MgO supports lead to enhanced C2 yields, which supports the trends observed in Fig. 2(a) and (c). This performance may be attributed to the presence of surface defects, particularly steps, which serve as active centers for the oxidative activation of methane. Kinetic studies suggest that these step sites facilitate a surface-mediated coupling process, contributing to high initial C2 selectivity.27 However, the support effect is also strongly coupled with the choice of active elements, as support performance varies substantially depending on elemental pairing.
Machine learning modeling is performed to predict highly active OCM catalysts. One-hot encoded representations of catalyst elements and supports are used as descriptor variables, while the objective variable is set to the C2 yield. Because experimental conditions strongly affect catalytic performance and can obscure composition performance relationships in machine learning analysis, the reaction temperature and CH4/O2 ratio are fixed at 700 °C and 3, respectively. The comparison between predicted and experimentally measured C2 yields is shown in Fig. 4(a). The model achieves a cross-validated coefficient of determination (R2) of 0.74, indicating good predictive performance under these constrained conditions. For comparison, 58 physical quantities in XenonPy are used, which result in an R2 of 0.74 as shown in Fig. 4(b); however, the MAE is a slightly better score in the one hot encoding case. Furthermore, literature data are also evaluated with one hot encoding where the temperature and CH4/O2 ratio are fixed at 700 °C and 3, respectively, and collected in Fig. 4(c). Fig. 4(c) shows that inconsistent data result in poor machine learning performance, and thus consistent data are quite important. For comparison, 58 physical quantities in XenonPy are used, which result in an R2 of 0.74 as shown in Fig. 4(b); however, the MAE is a slightly better score in the one-hot encoding case. Furthermore, literature data are also evaluated with one-hot encoding where the temperature and CH4/O2 ratio are fixed at 700 °C and 3, respectively, and collected in Fig. 4(c). Fig. 4(c) shows that inconsistent data result in poor machine learning performance, and thus consistent data are quite important. It should be noted that the well-diverse dataset, generated by a single researcher under identical devices and conditions, carries rich information about the catalysts, as the broad coverage of compositional space ensures that even simple one-hot encoding enables the model to extract relationships between catalyst composition and performance directly from the data. This work demonstrates that the quality and consistency of the dataset are critical. Collecting such data is labor intensive but reduces experimental noise and variability. Carefully curated, well-dispersed datasets can be more effective for machine learning than larger but heterogeneous datasets compiled from multiple sources.
 | ||
| Fig. 4 True and predicted C2 yield using (a) one hot encoding descriptors, (b) physical quantity descriptors, and (c) one hot encoding with literature data. | ||
An inverse analysis is performed to identify promising OCM catalysts. A total of 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 hypothetical binary and ternary catalyst combinations are generated from 25 elements (Na, Li, Mn, K, Sr, La, Ti, Ca, Ba, Mg, Rb, Y, Sm, Ce, Zn, Mo, Zr, Eu, Cs, Nd, Sn, W, Bi, Hf, and Pb) and 8 supports (BaO, CaO, La2O3, MgO, SiO2, TiO2, Y2O3, and ZnO). Each element–support combination is converted into a one-hot encoded representation and used as input for a trained random forest regression (RFR) model. The model is then applied to screen the full compositional space, and the top 3 predicted high C2 yield catalysts are summarized in Table 2.
800 hypothetical binary and ternary catalyst combinations are generated from 25 elements (Na, Li, Mn, K, Sr, La, Ti, Ca, Ba, Mg, Rb, Y, Sm, Ce, Zn, Mo, Zr, Eu, Cs, Nd, Sn, W, Bi, Hf, and Pb) and 8 supports (BaO, CaO, La2O3, MgO, SiO2, TiO2, Y2O3, and ZnO). Each element–support combination is converted into a one-hot encoded representation and used as input for a trained random forest regression (RFR) model. The model is then applied to screen the full compositional space, and the top 3 predicted high C2 yield catalysts are summarized in Table 2.
| Support | M1 | M2 | M3 | Predicted C2y |
|---|---|---|---|---|
| La2O3 | Na | Ba | Mg | 15.914 |
| La2O3 | Na | Ca | Ba | 15.896 |
| La2O3 | Na | Ba | La | 15.869 |
Based on machine learning predictions, three catalyst compositions, Na–Ba–Mg/La2O3, Na–Ca–Ba/La2O3, and Na–Ba–La/La2O3, as shown in Table 2, are selected for experimental validation. The catalytic performances of these catalysts, together with La2O3 as a reference, are shown in Fig. 5. All experiments are independently repeated twice to confirm reproducibility. As shown in Fig. 5, all three machine-learning-guided catalysts exhibit substantially higher C2 yields than the La2O3 reference, demonstrating the effectiveness of the inverse design strategy. The maximum C2 yields achieved by each catalyst are 19.2% at 750 °C for Na–Ba–Mg/La2O3, 19.2% at 750 °C for Na–Ca–Ba/La2O3, and 18.6% at 725 °C for Na–Ba–La/La2O3, respectively.
Conclusion
In summary, the role of data quality in catalyst informatics is systematically examined using a uniformly generated OCM catalyst dataset. Statistical analysis confirms that catalyst elements, supports, and C2 yield are well dispersed, providing a favorable foundation for machine learning. When experimental conditions are fixed, supervised learning using simple one-hot encoding successfully captures composition–performance relationships, achieving reliable predictive accuracy. Inverse analysis further enables efficient screening of a large compositional space, leading to the experimental validation of previously unreported high-performance OCM catalysts. Although generating these datasets by a single researcher is labor-intensive, the resulting consistency and broad compositional coverage substantially enhance the effectiveness of data-driven catalyst design. This work demonstrates that carefully curated, well-dispersed experimental datasets can enable meaningful machine learning even without large-scale high-throughput experiments, highlighting data quality as a key factor in advancing catalyst informatics.Conflicts of interest
There are no conflicts to declare.Data availability
All data are included in the manuscript.Acknowledgements
This work was funded by the Japan Science and Technology Agency (JST) ERATO Grant Number (JPMJER1903), PRESTO Grant Number (JPMJPR24T5), the JST Mirai Program Grant Number (JP-MJMI25G1), and the JSPS KAKENHI Grant-in-Aid for Scientific Research (B) Grant Numbers (JP23H01762) and (24K01241).References
- K. Takahashi, L. Takahashi, I. Miyazato, J. Fujima, Y. Tanaka, T. Uno, H. Satoh, K. Ohno, M. Nishida, K. Hirai, J. Ohyama, T. N. Nguyen, S. Nishimura and T. Taniike, The rise of catalyst informatics: towards catalyst genomics, ChemCatChem, 2019, 11, 1146–1152 CrossRef CAS.
- K. Takahashi, J. Ohyama, S. Nishimura, J. Fujima, L. Takahashi, T. Uno and T. Taniike, Catalysts informatics: paradigm shift towards data-driven catalyst design, Chem. Commun., 2023, 59, 2222–2238 RSC.
- J. R. Kitchin, Machine learning in catalysis, Nat. Catal., 2018, 1, 230–232 CrossRef.
- W. Yang, T. T. Fidelis and W.-H. Sun, Machine learning in catalysis, from proposal to practicing, ACS Omega, 2019, 5, 83–88 CrossRef PubMed.
- P. Schlexer Lamoureux, K. T. Winther, J. A. Garrido Torres, V. Streibel, M. Zhao, M. Bajdich, F. Abild-Pedersen and T. Bligaard, Machine learning for computational heterogeneous catalysis, ChemCatChem, 2019, 11, 3581–3601 CrossRef CAS.
- T. Toyao, Z. Maeno, S. Takakusagi, T. Kamachi, I. Takigawa and K.-I. Shimizu, Machine learning for catalysis informatics: recent applications and prospects, ACS Catal., 2019, 10, 2260–2297 CrossRef.
- J. A. Esterhuizen, B. R. Goldsmith and S. Linic, Interpretable machine learning for knowledge generation in heterogeneous catalysis, Nat. Catal., 2022, 5, 175–184 CrossRef.
- J. S. Hummelshoj, F. Abild-Pedersen, F. Studt, T. Bligaard and J. K. Norskov, Cat-App: a web application for surface chemistry and heterogeneous catalysis, Angew. Chem., Int. Ed., 2012, 51, 272–274 CrossRef CAS PubMed.
- A. J. Medford, M. R. Kunz, S. M. Ewing, T. Borders and R. Fushimi, Extracting knowledge from data through catalysis informatics, ACS Catal., 2018, 8, 7403–7429 CrossRef CAS.
- K. T. Winther, M. J. Hoffmann, J. R. Boes, O. Mamun, M. Bajdich and T. Bligaard, Catalysis-Hub.org, an open electronic structure database for surface reactions, Sci. Data, 2019, 6, 75 CrossRef PubMed.
- R. Tran, J. Lan, M. Shuaibi, B. M. Wood, S. Goyal, A. Das, J. Heras-Domingo, A. Kolluru, A. Rizvi, N. Shoghi, A. Sriram, F. Therrien, J. Abed, O. Voznyy, E. H. Sargent, Z. Ulissi and C. L. Zitnick, The Open Catalyst 2022 (OC22) dataset and challenges for oxide electrocatalysts, ACS Catal., 2023, 13, 3066–3084 CrossRef CAS.
- H. Shahbeik, S. Hu, S. Motamedi, S. A. F. S. A. Tajuddin, M. Tabatabaei and M. Aghbashlo, A comprehensive review of CO2 integration in thermochemical biomass conversion for enhanced biofuel production, Renewable Sustainable Energy Rev., 2026, 226, 116442 CrossRef CAS.
- M. Golvirdizadeh, S. A. G. Nia, H. Shahbeik, A. Shafizadeh, H. Hosseinzadeh-Bandbafha, M. Kiehbadroudinezhad, S. A. Seyedalikhani, M. Ahmadi, A. Hajiahmad, S. A. F. S. A. Tajuddin, M. Tabatabaei and M. Aghbashlo, Machine learning–guided CO2 methanation: From catalyst design optimization to techno-economic and life cycle assessment analyses, Energy, 2025, 139708 Search PubMed.
- T. N. Nguyen, T. T. P. Nhat, K. Takimoto, A. Thakur, S. Nishimura, J. Ohyama, I. Miyazato, L. Takahashi, J. Fujima, K. Takahashi and T. Taniike, High-throughput experimentation and catalyst informatics for oxidative coupling of methane, ACS Catal., 2019, 10, 921–932 CrossRef.
- T. Taniike and K. Takahashi, The value of negative results in data-driven catalysis research, Nat. Catal., 2023, 6, 108–111 CrossRef.
- Z. Yang and W. Gao, Applications of machine learning in alloy catalysts: rational selection and future development of descriptors, Adv. Sci., 2022, 9, 2106043 CrossRef CAS.
- L.-H. Mou, T. Han, P. E. Smith, E. Sharman and J. Jiang, Machine learning descriptors for data-driven catalysis study, Adv. Sci., 2023, 10, 2301020 CrossRef CAS PubMed.
- S. Ishioka, A. Fujiwara, S. Nakanowatari, L. Takahashi, T. Taniike and K. Takahashi, Designing catalyst descriptors for machine learning in oxidative coupling of methane, ACS Catal., 2022, 12, 11541–11546 CrossRef CAS.
- G. Keller and M. Bhasin, Synthesis of ethylene via oxidative coupling of methane: I. Determination of active catalysts, J. Catal., 1982, 73, 9–19 CrossRef CAS.
- G. Hutchings, M. Scurrell and J. Woodhouse, Oxidative coupling of methane using oxide catalysts, Chem. Soc. Rev., 1989, 18, 251–283 RSC.
- J. H. Lunsford, The catalytic oxidative coupling of methane, Angew. Chem., Int. Ed. Engl., 1995, 34, 970–980 CrossRef CAS.
- S. Nishimura, X. Li, J. Ohyama and K. Takahashi, Leveraging machine learning engineering to uncover insights into heterogeneous catalyst design for oxidative coupling of methane, Catal. Sci. Technol., 2023, 13, 4646–4655 RSC.
- S. Wu, G. Lambard, C. Liu, H. Yamada and R. Yoshida, iQSPR in XenonPy: a bayesian molecular design algorithm, Mol. Inf., 2020, 39, 1900107 CrossRef CAS PubMed.
- F. Pedregosa, G. Varoquaux, A. Gramfort, V. Michel, B. Thirion, O. Grisel, M. Blondel, P. Prettenhofer, R. Weiss, V. Dubourg, J. Vanderplas, A. Passos, D. Cournapeau, M. Brucher, M. Perrot and E. Duchesnay, Scikit-learn: Machine learning in Python, J. Mach. Learn. Res., 2011, 12, 2825–2830 Search PubMed.
- X. Zhou, Y. Pang, Z. Liu, E. I. Vovk, A. P. van Bavel, S. Li and Y. Yang, Active oxygen center in oxidative coupling of methane on La2O3 catalyst, J. Energy Chem., 2021, 60, 649–659 CrossRef CAS.
- Z. Xiong, J. Guo, Y. Deng, B. Liu, H. Lou, M. Zeng, Z. Wang, Z. Zhou, W. Yuan and F. Qi, Elucidating the mechanism for oxidative coupling of methane catalyzed by La2O3: experimental and microkinetic modeling studies, ACS Catal., 2024, 14, 1267–1280 CrossRef CAS.
- P. Schwach, W. Frandsen, M.-G. Willinger, R. Schlögl and A. Trunschke, Structure sensitivity of the oxidative activation of methane over MgO model catalysts: I. Kinetic study, J. Catal., 2015, 329, 560–573 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |




