Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Exploring pre-catalyst scope and selectivity in Negishi coupling of aryl iodides in deep eutectic solvents
L. Mauricio
Murillo-Herrera
,
Spencer A.
Frederick
,
Scott A.
Hicks
 ,
Simon B.
Duckett
,
Simon B.
Duckett
 and
John M.
Slattery
and
John M.
Slattery
 *
*
Department of Chemistry, University of York, Heslington, York YO10 5DD, UK. E-mail: john.slattery@york.ac.uk
First published on 17th February 2026
Abstract
Negishi coupling is a valuable synthetic protocol for the formation of C–C bonds. However, the use of highly reactive alkyl- or arylzinc reagents in these reactions usually requires them to be performed under strictly anhydrous conditions using Schlenk techniques. It has recently been demonstrated that using deep eutectic solvents (DES) as reaction media allows Negishi reactions to be performed without any protection from air or moisture. This paper explores pre-catalyst scope and regioselectivity in these reactions. This has led to new protocols using cheap and readily available Pd pre-catalysts, e.g. Pd3(OAc)6, whilst allowing coupling of some ortho-functionalised aryl iodides without the need for a supporting phosphine ligand. Unusual solvent-induced selectivity is demonstrated, which allows highly regioselective C–C bond formation to take place, even when multiple C–I bonds are present on the aryl iodide precursor. To further understand this, the activity and selectivity of a range of Pd pre-catalysts towards different iodoarenes has been explored, with the results helping inform the choice of catalyst for specific substrates and desired regioselectivity. The use of DES in these reactions promises to simplify their use, especially in environments where strict exclusion of air and moisture is not possible, whilst simultaneously providing access to regioselectivity that does not occur in conventional solvents under the same conditions.
Introduction
Negishi cross-coupling of organohalides or pseudo-halides with Zn organometallics, commonly catalysed by Ni or Pd, has found a great many applications in synthetic chemistry, natural product synthesis and medicinal chemistry.1–3 The biggest competitive advantage of this cross-coupling reaction, against other popular methodologies like Suzuki–Miyaura, Corriu–Kumada or Stille couplings, lies in the reactivity of the Zn organometallic used as the transmetalation reagent. As a moderately reactive metal, that is more electronegative than Li or Mg which are often used in organometallic nucleophiles, Zn exhibits high functional group tolerance. This enables the preparation of functionalized organozinc reagents (ester, cyanide, ether, amines, etc.), which successfully react with a wide range of functionalized organohalide electrophiles including ketones, nitriles, nitro-compounds, esters and amides.4 The reduced basicity of Zn organometallics, compared to related Mg and Li species, improves their cross-coupling performance by reducing Pd deactivation under catalytic conditions, which with more basic organometallics leads to organopalladate species.5,6 On the other hand, organozinc reagents are reactive enough to undergo cross-coupling reactions more rapidly than organoboron and organotin reagents, through rapid transmetalation steps, which allow for faster reactions with less-reactive substrates.7,8 Thus, Negishi couplings offer a good balance between selectivity, functional group tolerance and reactivity and as such are highly attractive for small-molecule synthesis.However, the Negishi coupling presents some important challenges: the presence of unwanted reactivity, leading to organozinc homocoupling and reductive dehalogenation of the electrophilic coupling partner, alongside the need to perform reactions in rigorously dried, non-protic solvents, with protection from atmospheric moisture during the reaction, to avoid protonolysis of the organozinc reagent. This latter limitation is particularly important in industrial settings and in laboratories lacking glovebox and Schlenk equipment. In addition, the relatively high reactivity of organozinc reagents poses challenges in controlling regioselectivity when multiple sites for C–C bond formation are present on the organohalide coupling partner.
Unproductive reactivity (leading to organozinc homocoupling and reductive dehalogenation) can be partially controlled by strategies that increase the rate of reductive elimination of the product relative to the unproductive pathways. Some examples are the employment of bulky and electron-rich phosphines like XPhos, the use of slightly hindered electrophiles, and the use of bulky organozinc reagents.9,10 The requirement for dry solvents and protective atmosphere is more challenging to address, due to the inherent reactivity of organozinc reagents towards water. It is here that some surprising recent developments in alternative solvent systems (vide infra) prove useful.
Recent years have seen a huge amount of research aiming to develop less harmful, more sustainable chemistry. In palladium catalysis, for example, the move towards using more sustainable solvents systems has been of particular interest.11 Amongst the many candidates being studied as alternatives to conventional volatile organic solvents, deep eutectic solvents (DES) have gathered significant momentum.12–20 DES are a diverse range of liquids formed from mixtures of Lewis basic components, or salts such as choline chloride, with Lewis acidic species or hydrogen bond donors, such as urea or glycerol.13,21 Eutectic mixtures of these species melt at considerably lower temperatures than their pure components (Scheme 1). They are related to ionic liquids (ILs), which have been used extensively in catalysis and as self-organised solvents for synthesis,22–30 since they often contain a high proportion of ions. However, they are generally cheaper and simpler to prepare, often composed of non-toxic and biodegradable components and many have the potential to be prepared from renewable resources, which makes them attractive alternatives to harmful volatile organic solvents, where there is an on-going drive for replacement. Compared to the related ILs, the simple, low E-factor synthetic routes to DES and their potentially reduced toxicity and environmental impact makes them particularly attractive from a green chemistry point of view. The application of DES in organic synthesis and catalysis has been reviewed recently.14–19,31
 | ||
| Scheme 1 Examples of (a) ionic or zwitterionic and (b) neutral components commonly found in DES. c) A schematic phase diagram for a DES. | ||
Remarkable recent studies have shown that ammonium-based DES can be used as solvents for reactions involving highly reactive organometallic reagents, despite the presence of protic functional groups and substantial amounts of water in the DES. In foundational work it was shown that in the presence of ammonium salts and Lewis bases, the Schlenk equilibrium of Grignard reagents is shifted towards the dialkylmagnesium and magnesium halide side.32 It has also been suggested that water, as well as other Lewis bases, prefer to interact with magnesium halides rather than alkylmagnesium species. Thus, magnesium halides can exert a water-scavenging effect that provides some protection for the organometallic reagent.33 It should be noted that in these studies, conditions somewhat similar to those in flow chemistry were used and the outcomes in batch conditions in solvents containing water or using “on water” conditions may be different. Indeed, a recent report using a range of techniques has suggested that some of these reactions may be “interface-driven”, with accumulation of reactants at the liquid surface.34 Another recent report has explored the addition of Li organometallics to ketones under “on water” conditions.35 These observations were followed shortly after by reports that electrophilic addition reactions involving organolithium and organomagnesium reagents could take place successfully in wet cholinium-based DESs in open air.36 It was also shown that coupling of organolithium reagents with aryl halides, catalysed by Pd complexes, can proceed in DES, although solvent-free conditions were even more effective in this study.37 Since then, more synthetic strategies involving air-sensitive organometallic reagents in DESs have been reported.38–46 The increased stability of polar-organometallic reagents in DESs, compared to volatile organic solvents, has been attributed to the participation of water molecules within the complex hydrogen-bonded network present in DES. This is thought to “deactivate” water, leading to a similar scavenging effect to the magnesium halides described above, slowing down the rate of hydrolysis of the organometallic. Indeed, some evidence for reduced water–water interactions, and increased water-DES interactions, have been seen in simulations of DES/water mixtures.47 It has also been proposed that the halide ions found in many DES can coordinate to the organometallic reagent to form anionic “ate”-complexes with enhanced nucleophilicity, while at the same time, the hydrogen-bond donor components of the DES can bind to and activate the electrophile, increasing the rate of the desired reaction.48
Using DES as solvents for cross-coupling reactions involving air-sensitive organometallic reagents therefore holds promise to circumvent the requirement for strictly air-sensitive conditions for Negishi couplings, allowing their use in a wider range of settings. DESs also present advantages in terms of the sustainability of the solvents. Recently, Capriati and co-workers reported cross-couplings between C(sp3) and C(sp2) lithium and zinc organometallic reagents and haloarenes in DES, or “on water” with NaCl as an additive, using Pd(PtBu3)2 as the catalyst (Scheme 2a).49,50 Cross coupling with the organolithium reagents did not proceed in DES, where only reductive dehalogenation of the substrate was observed. However, “on water” the reactions were successful and tolerant of haloarenes functionalized with both electron-withdrawing and donating functional groups. Organozinc reagents underwent cross-coupling satisfactorily in both DES and “on water”, but DES were more suitable for substrates bearing hydrogen-bond donors e.g., amines, carboxylic acids, and alcohols, since these haloarenes did not undergo coupling on water. The authors found that the reactions “on water” were very sensitive to the nature of the pre-catalyst, temperature and organozinc stoichiometry. In related work, Negishi coupling reactions between aryl bromides and C(sp3)–X (X = Br, I) halides have been reported “on water” with the assistance of micelles formed by non-ionic surfactants.51–54 Remarkably, in these systems it was possible to form the organozinc reagent in situ in the hydrophobic environment of the micelles, rather than the organometallic being formed ex situ and added at the start of the reaction. The synthesis of the Zn organometallic exhibited good functional group tolerance and the cross-coupling reaction was successful for aryl halides functionalized with ketones, esters and methoxy groups. Nitriles and alcohols were not tolerated. Since this publication, some additional cross-coupling reactions have been reported in DES, including Mizoroki–Heck from Dilauro et al. to give functionalised THF derivatives in good yield.55 D'Amico et al. reported Pd-catalysed cross-coupling of aryl bromides with functionalised thiophene derivaties in choline chloride/glycerol DES.56 Antenucci et al. reported Pd-catalysed coupling of arenediazonium o-benzenedisulfonimides and organolithium reagents in DES to give biaryl products.57 Niakan et al. have also reported Pd-free Suzuki and Sonogashira cross-coupling in DES, with immobilised Co nanoparticles acting as the catalyst.58 Most recently, Ramos-Martín et al. reported Friedel–Crafts benzylations in DES, catalysed by FeCl3.59
 | ||
| Scheme 2 Summary of previous methodologies, and that used in the present study, for Pd-catalysed cross-couplings with reactive polar organometallic reagents in DES. R3 is a range of substituents. | ||
Although DES have been shown to be promising alternative solvents for Negishi coupling,50 the role of the Pd pre-catalyst has not yet been reported. This has relevance for the potential application of these methodologies, where the use of cheaper, air-stable and readily available Pd pre-catalysts, like Pd3(OAc)6, would maximise their utility in a range of settings. An understanding of pre-catalyst activity towards different substrates, and selectivity in the presence of multiple carbon-halide or pseudohalide bonds in the substrate, is also important. This contribution will present methodologies that address both of these challenges, in addition to allowing a regioselective Negishi coupling in DES for the first time, whilst maintaining the ability to perform the cross-coupling reactions in wet solvents with no protective atmosphere (Scheme 2b).
Results and discussion
Developing Negishi coupling in DES using Pd3(OAc)6 as the pre-catalyst
Previous work demonstrated that the Pd(0) pre-catalyst Pd(PtBu3)2 allows highly effective Negishi coupling of a range of substrates in DES.50 However, this complex is air-, moisture- and temperature-sensitive and is ideally handled under an inert atmosphere and stored in a glovebox at low temperature. This negates some of the benefits of being able to perform these reactions in DES on the open bench, without protective conditions. Our aim was to develop a protocol that allows related reactions to take place using cheaper, air- and moisture-stable Pd(II) precursors based on ubiquitous Pd3(OAc)6. To develop appropriate reaction conditions we studied the reaction of nBuZnCl with methyl 2-iodobenzoate under similar conditions to those reported by Marder and Lei (Table 1).60 This reaction was chosen for several reasons: it was reported to proceed rapidly (less than 2 min for full conversion) and consideration of recent work with DES suggested that fast organometallic reactions were more likely to be compatible with the water and functional groups present in DES. Additionally, no phosphine ligand is required, which reduces the potential for ligand solubility problems in the DES and the high costs associated with some phosphines.61 In general, fewer additives is desirable in terms of sustainability, cost and convenience. Finally, the reaction is compatible with ammonium halides, which were used in the formation of the DES.| Entry | Solvent | Solvent wet/dry | Temp. (°C) | Pd3(OAc)6 (mol% Pd) | n BuZnCl (equiv.) | RZnCl rate of addition | Yield of 2a (%) | Yield of 3 (%) | |
|---|---|---|---|---|---|---|---|---|---|
| Typical conditions: 1 (1.0 mmol) and Pd3(OAc)6 were allowed to mix for 10 minutes in 5 cm3 solvent at the desired temperature. a in 2 cm3 dry THF was added (either dropwise or in one quick addition) while the reaction mixture was stirred at 1000 rpm, then the reaction was stirred for 5 minutes prior to workup (see SI for details).a NMR yields determined by 1H NMR spectroscopy.b GC yields determined by gas chromatography.c 1 equiv. [Bu4N]Br added as additive.d Reaction period was 12 h.e 10 cm3 solvent used. | |||||||||
| 1a,c | THF | Dry | 40 | 2 | 2 | Dropwise | 84 | 1 | |
| 2a | ChCl/urea | Dry | 40 | 1 | 1.6 | Dropwise | 88 | 10 | |
| 3a | ChCl/urea | Wet | 40 | 1 | 1.6 | Dropwise | 27 | 3 | |
| 4a | ChCl/urea | Dry | 40 | 2 | 1.6 | Dropwise | 90 | 6 | |
| 5a | ChCl/urea | Wet | 40 | 2 | 2.0 | Dropwise | 55 | 5 | |
| 6a | ChCl/urea | Dry | 40 | 2 | 2.0 | Fast, single | 31 | 10 | |
| 7a | ChCl/urea | Wet | 40 | 2 | 2.0 | Fast, single | 46 | 8 | |
| 8a | ChCl/urea | Dry | 60 | 1 | 2.0 | Dropwise | 87 | 12 | |
| 9a | ChCl/urea | Wet | 60 | 1 | 2.0 | Dropwise | 80 | 5 | |
| 10a | ChCl/urea | Wet | 60 | 2 | 2.0 | Dropwise | 97 | 0 | |
| 11a | ChCl/urea | Wet | 60 | 2 | 2.0 | Fast, single | 70 | 7 | |
| 12a | ChCl/urea | Dry | 60 | 0 | 2 | Dropwise | 0 | 0 | |
| 13a,d | ChCl/urea | Dry | 60 | 2 | 0 | — | 0 | 0 | |
| 14b | ChCl/DMU | Wet | 80 | 1.5 | 2.0 | Dropwise | 14 | 1 | |
| 15b | ChCl/AcNH | Wet | 80 | 1.5 | 2.0 | Dropwise | 14 | 1 | |
| 16a | ChCl/G | Wet | 40 | 0.5 | 2.0 | Dropwise | 6 | 0 | |
| 17a,d,e | ChCl/G | Wet | 40 | 0.5 | 2.0 | Dropwise | 0 | 0 | |
| 18a,d | ChCl/G | Dry | 40 | 1 | 1.6 | Dropwise | 0 | 0 | |
To explore solvent effects, five DES were prepared as described in the SI and shown in Scheme 3. Four were based on the hydrogen-bond acceptor (HBA) choline chloride (ChCl) with glycerol (Gly), urea, acetamide (AcNH2) and dimethylurea (DMU) as the hydrogen-bond donor (HBD) components (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 ratio of HBA
2 ratio of HBA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HBD). One was based on acetylcholine chloride (AcChCl) as the HBA, with urea as the HBD (1
HBD). One was based on acetylcholine chloride (AcChCl) as the HBA, with urea as the HBD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio). ChCl-based DES, with urea and glycerol, were chosen since these are the most widely used DES solvents for organic transformations and they are comparatively well studied and readily available. DES based on acetamide, dimethylurea and AcChCl were devised to reduce the protic nature of the HBD fraction and modify the solubility of the reagents used in the catalytic reactions. For ChCl/urea and ChCl/Gly, batches of DES were prepared under conditions designed to minimize their water content and these were stored under N2. These DES are denoted “dry” and typical water contents for these materials were around 400 ppm. In addition, batches of each DES were prepared directly from the components as received and stored in air. These DES are denoted “wet” and typical water contents for these were between 1 and 5 wt%, depending on the HBD used. Water contents for each DES, determined by Karl Fischer titrations, are given in the SI. Selected results from an initial screening study for the conversion of 1 into 2a and 3 as a function of solvent and reaction condition are shown in Table 1. A full set of data are given in the Table S1.
1 ratio). ChCl-based DES, with urea and glycerol, were chosen since these are the most widely used DES solvents for organic transformations and they are comparatively well studied and readily available. DES based on acetamide, dimethylurea and AcChCl were devised to reduce the protic nature of the HBD fraction and modify the solubility of the reagents used in the catalytic reactions. For ChCl/urea and ChCl/Gly, batches of DES were prepared under conditions designed to minimize their water content and these were stored under N2. These DES are denoted “dry” and typical water contents for these materials were around 400 ppm. In addition, batches of each DES were prepared directly from the components as received and stored in air. These DES are denoted “wet” and typical water contents for these were between 1 and 5 wt%, depending on the HBD used. Water contents for each DES, determined by Karl Fischer titrations, are given in the SI. Selected results from an initial screening study for the conversion of 1 into 2a and 3 as a function of solvent and reaction condition are shown in Table 1. A full set of data are given in the Table S1.
 | ||
| Scheme 3 Salts, hydrogen-bond donors, and their abbreviations, used to prepare the DES studied here. | ||
In THF, under an inert atmosphere of N2, the reaction proceeded smoothly (Table 1, entry 1). Formation of 2a is accompanied by dehalogenation of 1 to form 3, through a competing β-hydrogen elimination pathway. Although this is a relatively minor side reaction in THF, it can become more significant under some conditions in DES (vide infra). When comparing the results from different DES, it was found that those based on glycerol were not suitable reaction media, with yields of 2a of 6% at most and unreacted 1 recovered (Table 1, entries 16–18). 3 was not detected at the end of the reaction for either the “wet” or “dry” ChCl/Gly DES. In all cases, a white precipitate was formed during the reaction and there was no visible evidence of black Pd(0) particle formation, which occurs in THF. Although these DES have been shown to be compatible with other organometallic reactions,62 and when using Pd(PtBu3)2 as pre-catalyst for Negish couplings,50 in this case it appears that the alkylzinc reagent degrades more rapidly in the polyol-based DES than it takes part in the coupling reaction with Pd3(OAc)6 as pre-catalyst. Reasoning that this reactivity was likely to be promoted by the OH groups on the HBD in ChCl/Gly, which are both reactive and strong H-bond donors, the reaction was then studied in DES based on urea and related HBDs. These DES were found to support productive catalysis under a range of conditions.
Because of the rapid degradation of RZnX reagents by water, one might imagine that using “dry” DES would inherently be more successful than using the “wet” solvents. However, our results show a more complex picture for this reaction. At 40 °C, with dropwise addition of the nBuZnCl, a significantly improved yield of 2a is seen for the dry DES compared to the wet DES (Table 1, entries 2 to 5). However, when the organozinc reagent is added in a single, rapid addition the yield of 2a is similar between the dry and wet DES, with a somewhat higher yield for the wet solvent, although both show relatively poor yields under these conditions (Table 1, entries 6 and 7). Dilauro et al. have previously shown that a rapid, single addition of an organometallic reagent to reactions taking place “on water” or in DES promotes catalysis,49,50 but this seems less effective in our system, with dropwise addition generally producing better results.
By increasing the reaction temperature from 40 to 60 °C it was possible to circumvent the need for dry DES and protective N2 atmosphere (Table 1, entries 8 and 9). Further optimisation found that a slightly higher catalyst loading (2 mol% Pd) also increased the yield at both 40 and 60 °C in the wet solvent. As such, Table 1, entry 10 represents optimised conditions for this reaction, which allowed highly effective catalysis (up to 97% yield of 2a) in “wet” ChCl/urea (typically around 3 wt% water), without any precautions needed for the preparation or storage of the DES or any protection of the reaction from atmospheric water or O2. The reaction was repeated a number of times to give an average yield of 2a of 86% over 6 runs (Table S2, entries 14.1–14.6), demonstrating good reproducibility. A double-scale reaction gave an isolated yield of 2a, after workup and purification, of 80%, giving confidence that NMR/GC yields were representative of the isolated yields achievable following purification. Investigating the rate of addition of a under the optimised conditions, a fast, single addition of the organozinc reagent led to a degradation in performance (Table 1, entry 11). Control experiments under these optimised conditions, without addition of Pd, and without organozinc reagent (Table 1, entries 12 and 13) led to no formation of 2a, and no formation of dehalogenated side product 3, confirming that catalytic Pd is required and that in the absence of a, there is no dehalogenation of the substrate.
Exploration of additional DES as reaction media generally led to poor yields of 2a (Table 1, entries 14 and 15). This is interesting when the solubility of the reagents in the DES is considered, as there seems to be no simple correlation between the solubility of the reactant 1 and the success of the reaction. Pd3(OAc)6 has good solubility in all of the DES. 1 has only partial solubility in ChCl/urea, but the reaction proceeds very effectively under the optimised reaction conditions regardless. Moving to ChCl/AcNH2 and ChCl/DMU improves the solubility of the aryl iodide, but the yield of 2a is substantially reduced and evidence of reactions between 1 and the HBD are observed (Table S1). In AcChCl/urea the solubility of 1 and Pd3(OAc)6 is very poor, and so no catalytic experiments were carried out in this DES. When the above observations are coupled with the poor yields seen in DES based on Gly as the HBD, it is clear that the factors influencing the choice of DES for reactions such as this are complex and some trial-and-error testing is currently necessary to determine the optimal DES for the reaction, even amongst chemically similar types.
Finally, re-use of the DES/Pd mixture was tested by reaction of 1 with a to give 2a (conditions as per Table 1, entry 10), followed by extracting the organic products and unreacted starting material directly from the DES using dichloromethane. The results showed a decrease in the yield of 2a from 83% for the first cycle, to 64% for the second and 56% after a third cycle (see SI for details). There is clearly degradation in performance across these runs and so catalyst/solvent re-use is not attractive for this system.
Substrate scope and pre-catalyst optimisation
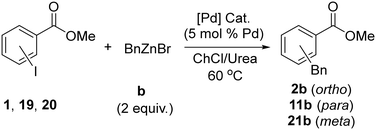
Expanding the substrate scope using the optimised reaction conditions above showed some interesting trends (see SI for full details). In general, iodoarenes with meta or para substituents exhibited little or no formation of the desired cross-coupled product using Pd3(OAc)6 as pre-catalyst. However, substrates with Lewis basic groups in the ortho positions, CO2R, C(O)R and to some extent OMe, which are capable of coordinating to the metals in the system, led to good yields of the desired products, whereas substrates with similar electronic properties (i.e. ortho-CF3-substituted) performed poorly. Based on these observations, the range of substrates with ortho-coordinating substituents on the iodoarene was expanded, as shown in Scheme 4. In addition, the pre-catalyst was re-optimised for para-substituted iodoarenes by the addition of 2 equiv. of the cheap and readily available phosphine PPh3. In total, 40 cross-coupling reactions are reported.The data in Scheme 4 shows that provided the correct Pd pre-catalyst is chosen, the Negishi couplings presented here are effective for a range of substrates and alkylzinc reagents. This includes primary and secondary alkylzinc halides (a, b, d) and an example bearing ester functionality (4-ethoxy-4-oxobutylzinc bromide, c). However, a nitrile-functionalised alkylzinc bromide (3-cyanopropylzinc bromide (e)) was found through competition experiments to poison the catalyst, leading to poor yields (<10%), potentially due to Pd-nitrile coordination (see SI for details). Reactions involving phenyl- and cyclopropylzinc halides produced low to no product formation and were not explored further. Expansion of the range of ortho-donor functionalised iodoarenes (pink box, Scheme 4) to include ether, amide and pyridyl groups alongside ester and ketone functionality gave good to excellent yields in most cases. An ortho-donor group appears to enhance the reaction, where Pd3(OAc)6 is the pre-catalyst, relative to alternative substitution geometries (see selectivity experiments below). The ester, ketone and amide-functionalised substrates showed the best yields of cross-coupled products. All of these have a carbonyl group at the ortho position of the iodoarene, which could coordinate Pd to generate a 5-membered palladacycle following oxidative addition (e.g.Scheme 5a). This has similarities to the directing group effects seen in related C–H functionalisation reactions.63
We tentatively propose that this type of stabilisation may facilitate pre-coordination of the substrate to Pd(0) and/or a faster oxidative addition in this system, which allows the rate of productive catalysis to outcompete the rate of hydrolysis of the organozinc reagent in the DES. This is at odds with previously measured kinetics for the oxidative addition of related ortho-donor functionalised aryl iodides to Pd(0), in the presence of phosphine ligands, where ortho-donors actually reduced the rate of oxidative addition.64 This was attributed primarily to steric effects. However, the presence of preferential Pd-phosphine interactions in the previous work may influence the observations and in the present study, where no phosphines are present, the ortho-donor interactions may be sufficiently stabilising to either allow pre-coordination of the substrate to Pd(0) or to otherwise facilitate oxidative addition. Interestingly, when iodo-2-phenylpyridine is the substrate, the yields are reduced for reactions with nBuZnCl as the nucleophile, while good yields are maintained with BnZnBr, despite the potential for formation of a related 5-membered palladacycle in both cases (18, Scheme 5b). In this case, the larger pyridyl substituent may begin to inhibit oxidative addition for steric reasons, as seen in the Pd(0) phosphine systems studied by Jutand.64 A subtle balance of competing effects is likely to be at play here.
To explore the potential role of palladacyclic intermediates, (18, Scheme 5b), a known and stable metallocycle was synthesised ex situ as a model for an intermediate of the reaction that yields product 10b.65 This was reacted with a stoichiometric amount of benzyl zinc bromide under similar conditions to the optimised catalytic procedure above (see SI for details). This led to formation of product 10b, albeit with only a modest yield of 17%, with a significant amount of the alkylzinc halide homocoupling product, bibenzyl, produced as a side product under these conditions. 18 was also used as a pre-catalyst for the coupling of 1 with BnZnBr, giving an overall yield of 89% 2a (see SI for details), which is comparable to when Pd3(OAc)6 is used as the pre-catalyst. These experiments demonstrate that palladacycles, such as 18, can convert to product under catalytically relevant conditions and are competent pre-catalysts, supporting their potential role as catalytic intermediates.
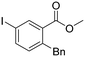
Reactions between 2-iodoanisole and a or b showed poor yields, despite the potential formation of a 4-membered palladacycle via coordination of OMe to Pd. This result suggests that these phosphine-free Negishi reaction conditions in ChCl/urea are not very effective for electron-rich iodoarenes, which are expected to undergo oxidative addition to Pd more slowly. Activity was also found with bromoarenes, albeit with only modest yields. Thus, when methyl 2-bromobenzoate reacted with b, the yield of product 2b was 44%. It was initially thought that bromoarenes would undergo oxidative addition to Pd too slowly and so productive catalysis would be unable to compete with the rate of hydrolysis of the organozinc reagent in the DES. However, it seems that this effect is not strong enough to completely suppress the catalytic reaction.
Addition of PPh3 to the system, by pre-formation of the Pd complex Pd(OAc)2(PPh3)2 and its use as pre-catalyst, allowed for good yields of para-substituted iodoarenes in most cases (Scheme 4, green box). However, reactions involving iodoarenes functionalised at the ortho position generally exhibited poor yields under these conditions, so this represents a complementary methodology to that using Pd3(OAc)6, as described above. With Pd(OAc)2(PPh3)2, the differences between electron-rich and electron-poor iodoarenes were no longer appreciable. Jutand and co-workers showed that this Pd(II) pre-catalyst is reduced in situ by an intermolecular cleavage of a phosphine and one of the acetate groups to yield a very reactive and unstable low-valent Pd species.66 Recently, Fairlamb and co-workers characterised this as a Pd(I) dimer bridged by one acetate and one phosphine ligand, which forms a very active trinuclear Pd species upon oxidative addition with haloarenes.67 A similar Pd(I) dimer, bridged by bromide, was prepared by Colacot et al. by reduction of Pd(COD)Br2 in the presence of 1 equivalent of PtBu3 and a base.68 The mechanism for the reduction of Pd(II) in this system was later explored in detail.69,70 Although Pd speciation in the DES is currently unknown, similar activation processes and formation of highly active Pd species derived from the pre-catalyst is possible and is consistent with the observed increase in reactivity towards para-substituted iodoarenes. However, in a chloride-rich environment, such as that provided by the DES, the formation of anionic Pd “ate” complexes is also possible.71–73 In addition, it has been proposed by Jutand and co-workers, that in chloride-rich environments, chloride can bind to Pd and narrow the P–Pd–P angle in bisphosphine complexes, which may also enhance rates of oxidative addition of iodoarenes to Pd.71,74
The effect of pre-catalyst on selectivity
The complementarity in the two methodologies shown in Scheme 4, where different pre-catalysts allow the use of aryl iodide substrates with different substitution patterns, inspired us to investigate the effect of Pd pre-catalyst towards ortho-, meta- and para-substituted methyl iodobenzoates 1, 19 (para) and 20 (meta) in more detail. Table 2 shows a summary of the % yield of the desired C–C coupled product 2b, 11b, and 21b (meta), from isomers of methyl iodobenzoate with BnZnBr in ChCl/urea using different pre-catalysts and reaction conditions.The results in Table 2 show significant variation in the amount of C–C coupled product that is formed depending on the substitution pattern of the substrate, the pre-catalyst and the conditions used. When Pd3(OAc)6 is used, higher yields for ortho-, rather than meta- or para-iodoarene, as described above, are clear. Pd(OAc)2(PPh3)2 is less active for these substrates, and more consistent in yield for the different substitution patterns, albeit with a preference for para-substituted substrates. Pd(PtBu3)2 is the most active catalyst for these reactions in ChCl/urea, but its activity depends on the reaction conditions used. Under the conditions reported by Dilauro et al.,50 good to excellent yields are seen for the para- and ortho/meta-substituted substrates respectively. If the optimised reaction conditions from the present study (Scheme 4) are used, which involve dropwise addition of the alkylzinc reagent, rather than a fast single addition, and a slightly higher catalyst loading, Pd(PtBu3)2 performs less effectively for the ortho-substituted iodoarene, but similarly for the others. This is the opposite of what is seen for Pd3(OAc)6 and highlights the subtle effects at play in these reactions.
As different pre-catalysts show differing activities towards aryl iodide substrates with different substitution patterns, this inspired us to investigate a regioselective Negishi coupling in DES where multiple C–X bonds are present in the electrophile. Control over the site of C–C bond formation in such substrates is highly desirable, allowing sequential functionalisation at different positions without the need for protection/activation, thus simplifying construction of complex molecular architectures. To this end we aimed to leverage the selectivity for C–C bond formation ortho to a coordinating functional group (e.g. ester, amide) when Pd3(OAc)6 is used as a pre-catalyst. Carboxylate-directed Suzuki–Miyaura coupling has been reported previously,75 and Pd(I) pre-catalysts have been shown to allow chemoselective Kumada and Negishi cross coupling for substrates containing two or more different halides/pseudohalides,76 but to the best of our knowledge donor-group-directed, regioselective Negishi reactions have not been described.
Methyl 2,3,5-triiodobenzoate (22), with one C–I bond ortho and two meta to the ester, was coupled with BnZnBr, under our optimised reaction conditions for ortho-coordinating functional groups in ChCl/urea (Scheme 6). More than twenty potential products, including regioisomers, are possible in this reaction via substitution of one or more I for Bn, and/or via dehalogenation. As such, this substrate represents a challenging test for selectivity in this reaction. Under these conditions with BnZnBr, 88% of the electrophile was consumed, resulting in 70% yield of the mono-substituted product 23i, where Bn has added to the position ortho to the ester. Only 2% yield was observed for the other mono-substituted isomers. Small amounts of di-substituted 24i–iii and tri-substituted 25 cross-coupled products were observed at 4% and 5% yield respectively. The mono-dehalogenated side products were present at 1%. Finally, a combination of partially substituted products with different degrees of dehalogenation were produced in 6% yield (Scheme S4, Table S6). Overall, the 70% selectivity of the reaction towards one, out of many potential products was very good. In order to confirm the connectivity of the major, monosubstituted product, 23i was isolated by column chromatography and analysed by NMR spectroscopy (Fig. S11–S16). A chemical shift of δ 8.37 ppm for the proton at the para position of the aromatic ring suggested that it is surrounded by two iodine atoms. The only connectivity pattern which could account for such downfield displacement is the one in which the new C–C bond is formed ortho to the ester. In addition, in the HMBC spectrum, a distinctive four-bond correlation between the methylene protons on the Bn group and the carbonyl carbon atom was detected. Both features are consistent with the proposed formulation of 23i.
 | ||
| Scheme 6 Regioselective Negishi coupling in ChCl/urea. Reactions performed at 60 °C in ChCl/urea, and at 40 °C in THF, with 1 equiv. of [NBu4]Br, as per ref. 60, as a control reaction. Selected products, not including dehalogentated species, and their compound numbers are shown. | ||
To explore the role of the DES in this regioselectivity, a control reaction was performed in THF using reaction conditions similar to those originally reported by Lei.60 In dry THF in the presence of 1 equiv. [NBu4]Br and 5 mol% Pd3(OAc)6, 22 was reacted with BnZnBr at 40 °C. In contrast to the DES, the reaction in THF resulted in a yield of 23i of only 10%, in addition to 28% of the other mono-substituted isomers. The di-substituted products were formed in 35% yield, while the tri-substituted product was present at 15%. Clearly the regioselectivity is poor in THF. Surprisingly, the degree of dehalogenation was also higher for this substrate in THF than in the DES, with a total of 13% of a range of dehalogenated products being formed.
The impact of pre-catalyst on the regioselectivity of this reaction in ChCl/urea was also explored. Using Pd(PtBu3)2, under the conditions optimised by Dilauro et al.,50 84% of the electrophile 22 was consumed, now resulting in 45% yield of ortho-substituted product 23i, with 2% yield of monosubstituted 23ii–iii. The tri-substituted 25 cross-coupled product was observed at 7% yield and di-substituted products 24i–iii at 11%, alongside some bis-dehalogenated (1, 20) side products at 3% (Scheme S4, Table S9). Using Pd(PtBu3)2 as pre-catalyst, but under the reaction conditions used for the Pd3(OAc)6 reaction above, led to even lower electrophile consumption and less selectivity (Scheme S4, Table S8). Finally, the reaction between 22 and BnZnBr was repeated with Pd(OAc)2(PPh3)2 as pre-catalyst, leading to 60% of the electrophile being consumed and 44% yield of 23i. Some formation of the tri-substituted product 25 was observed at 3% yield, alongside the mono-dehalogenated (26i–iii) and bis-dehalogenated (1, 20) side products at 5% and 7% respectively (Scheme S4, Table S7).
From this series of reactions, it can be seen that the reaction catalysed by Pd3(OAc)6 in DES shows the highest yield of, and best selectivity for, the ortho-monosubstituted product 23i. When PPh3 is present, with pre-catalyst Pd(OAc)2(PPh3)2, the yield is lower at a similar reaction time point, although some selectivity towards 23i is retained. Although this pre-catalyst was introduced to allow coupling of para-substituted iodoarenes under our conditions (Scheme 6), it proved to also be moderately competent for some of the listed ortho-substituted substrates. Pd(PtBu3)2 is a more active pre-catalyst for Negishi coupling reactions in DES, including those of meta-substituted haloarenes,50 but this activity now leads to much lower selectivity with electrophile 22, as a higher proportion of di- and tri-substituted products form than with the other pre-catalysts. Moving from the DES into THF leads to a loss of all selectivity in the reaction.
To further investigate selectivity, diiodoarene 26ii was synthesised, and the selectivity conferred by the three pre-catalysts investigated (Scheme 7). When Pd3(OAc)6 was used, 83% of the electrophile was consumed, with 64% yield of the ortho-substituted product 27ii and 16% yield of the disubstituted product 28ii alongside some dehalogenated products (Scheme S5, Table S10). With Pd(OAc)2(PPh3)2 as the pre-catalyst, 43% of the electrophile was consumed to give 20% of the ortho-substituted product 27ii and 5% yield of the meta-substituted product 27v. Disubstituted product 28ii was now present at 4% alongside some dehalogenated products (Scheme S5, Table S11). When the reaction was carried out with Pd(PtBu3)2 under the conditions referred to earlier in this work, 72% of the electrophile was consumed to give 16% ortho-substituted product 27ii and 47% disubstituted product 28ii, in addition to some partial dehalogenation products (Scheme S5, Table S12). When carried out under the conditions optimised by Dilauro et al. though, 84% of the electrophile was consumed to give 25% ortho-substituted product 27ii and 44% disubstituted product 28ii, alongside some dehalogenated products (Scheme S5, Table S13).
 | ||
| Scheme 7 Regioselective Negishi coupling in ChCl/urea. Reactions performed at 60 °C in ChCl/urea. Selected products, not including dehalogentated species, and their compound numbers are shown. | ||
Overall, these reactions with diiodoarene 26ii follow similar trends to those seen for triiodoarene 22, with Pd3(OAc)6 giving the highest selectivity for a single substitution, at the ortho-position. Pd(OAc)2(PPh3)2 led to very little yield, although no disubstitution was observed. When Pd(PtBu3)2 was used, a preference for disubstitution at both the ortho- and meta-positions was seen, which is consistent with its higher activity, which delivers lower selectivity as seen before. The results for both systems are summarised in Table 3.
| Substrate | Product | Pd3(OAc)6 | Pd(OAc)2(PPh3)2 | Pd(PtBu3)2 | |
|---|---|---|---|---|---|
| Dilauro et al. conditionsa50 | Conditions from this work | ||||
| [Pd] cat. at 5 mol%, BnZnBr at 2 equiv., dropwise addition.a [Pd] cat. at 2.5 mol% Pd. BnZnBr at 3 equiv., as shown in Scheme 4. GC yields determined by gas chromatography. | |||||

|
|
64 | 20 | 25 | 16 |

|
16 | 4 | 44 | 47 | |
| Starting material | 17 | 57 | 16 | 28 | |

|

|
70 | 45 | 45 | 20 |
| Polybenzylated products | 11 | 3 | 18 | 13 | |
| Starting material | 12 | 40 | 16 | 38 | |
Summary and conclusions
Novel methodologies for Negishi cross coupling of aryl iodides with alkylzinc halides in DES based on simple pre-catalysts, which are not temperature-, air- or moisture sensitive, have been developed. Two complementary methodologies, either phosphine-free with Pd3(OAc)6 as the pre-catalyst, or where PPh3 is added as a supporting ligand, allow cross coupling to take place with a range of aryl iodides. Substrates with donor groups ortho to the C–I bond, e.g. ester, amide, pyridyl, which can coordinate to metals present in the system, do not require a phosphine ligand to be added to achieve good yields. Palladacycle formation as part of the catalytic mechanism is proposed in these un-supported reactions, with both stoichiometric and catalytic testing of an isolated palladacycle supporting this. To functionalise substrates meta and para to the C–I bond requires the addition of PPh3 as a supporting ligand.The effect of Pd pre-catalyst on reactivity and selectivity towards iodoarenes with different substitution patterns was explored, using Pd3(OAc)6, Pd3(OAc)2(PPh3)2 and Pd(PtBu3)2 pre-catalysts. Pd(PtBu3)2 is the most active of these, and as demonstrated previously,50 achieves the widest substrate scope, but is the least selective when multiple cross-coupling sites exist on the iodoarene. Pd3(OAc)6 is active towards ortho-donor substituted substrates and allows highly selective cross-coupling when more than one C–I bond is present in the substrate. Pd3(OAc)2(PPh3)2 is the least active catalyst for ortho- and meta-substituted substrates, but competent for para-substitution, which allows some selectivity, albeit with relatively low yields in some cases.
The complementarity of these methodologies allowed us to develop regioselective Negishi coupling in ChCl using Pd3(OAc)6 as pre-catalyst, where C–C bond formation ortho to a coordinating group was strongly preferred, even when other C–I bonds were available on the aryl halide for functionalisation. A comparable reaction in THF led to a mixture of products with no selectivity. This is an unusual example of solvent-induced selectivity, whose origin appears to lie in the relative rates of productive catalysis vs. the rate of hydrolysis of the organozinc reagent in the DES. With Pd3(OAc)6 as pre-catalyst, the rate of C–C bond formation ortho to a coordinating group, such as an ester, is fast enough to out-compete hydrolysis of the organozinc reagent. However, cross coupling at the meta- and para-positions is slower than organozinc hydrolysis in the DES and these pathways are less favoured. Overall, here and in other recent studies, DES have been shown to be intriguing alternative solvents for organometallic chemistry.
Experimental
Full experimental details are given in the SI.Author contributions
L. Mauricio Murillo-Herrera: investigation, methodology, writing – original draft, writing – review & editing, funding acquisition. Spencer Frederick: investigation, methodology, writing – original draft, writing – review & editing. Scott Hicks: methodology, writing – review & editing. Simon Duckett: supervision, writing – review & editing, funding acquisition. John Slattery: conceptualization, supervision, writing – original draft, writing – review & editing, funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI).Supplementary information: experimental details. General methods. Preparation of all reagents. Procedures for all experiments. Characterisation data, including NMR spectra. GC methodology. Gas chromatograms. Additional raw data supporting this research is openly available from the research data repository of the University of York at https://doi.org/10.15124/a033ed32-ff01-4c85-a1de-8955d48b0891. See DOI: https://doi.org/10.1039/d5cy01526j.
Acknowledgements
We are grateful to Secretaría de Ciencia, Humanidades, Tecnología e Innovación (SECIHTI) Mexico for the award of a scholarship to Mauricio Murillo, to the EPSRC for a DTP studentship for Spencer Frederick and University of York for funding. We are grateful for the use of NMR and Gas Chromatography facilities supplied by the Department of Chemistry and supported by the technical team in the Department. We thank Prof. Anne Duhme-Klair and Prof. Tom Welton for useful discussions.References
- A. W. Dombrowski, N. J. Gesmundo, A. L. Aguirre, K. A. Sarris, J. M. Young, A. R. Bogdan, M. C. Martin, S. Gedeon and Y. Wang, Expanding the Medicinal Chemist Toolbox: Comparing Seven C(sp2)–C(sp3) Cross-Coupling Methods by Library Synthesis, ACS Med. Chem. Lett., 2020, 11, 597–604 CrossRef CAS PubMed.
- T. J. Sanderson, C. M. Black, J. W. Southwell, E. J. Wilde, A. Pandey, R. Herman, G. H. Thomas, E. Boros, A.-K. Duhme-Klair and A. Routledge, A Salmochelin S4-Inspired Ciprofloxacin Trojan Horse Conjugate, ACS Infect. Dis., 2020, 6, 2532–2541 CrossRef CAS PubMed.
- S. Tabassum, A. F. Zahoor, S. Ahmad, R. Noreen, S. G. Khan and H. Ahmad, Cross-coupling reactions towards the synthesis of natural products, Mol. Diversity, 2022, 26, 647–689 Search PubMed.
- D. Haas, J. M. Hammann, R. Greiner and P. Knochel, Recent Developments in Negishi Cross-Coupling Reactions, ACS Catal., 2016, 6, 1540–1552 Search PubMed.
- E.-i. Negishi, Magical Power of Transition Metals: Past, Present, and Future (Nobel Lecture), Angew. Chem., Int. Ed., 2011, 50, 6738–6764 Search PubMed.
- E.-i. Negishi, K. Akiyoshi and T. Takahashi, Inhibition of reductive elimination of diorganopalladium species by formation of tetraorganopalladates, J. Chem. Soc., Chem. Commun., 1987, 477–478 Search PubMed.
- L. Jin and A. Lei, Insights into the elementary steps in Negishi coupling through kinetic investigations, Org. Biomol. Chem., 2012, 10, 6817–6825 RSC.
- J. del Pozo, G. Salas, R. Álvarez, J. A. Casares and P. Espinet, The Negishi Catalysis: Full Study of the Complications in the Transmetalation Step and Consequences for the Coupling Products, Organometallics, 2016, 35, 3604–3611 CrossRef CAS.
- Q. Liu, Y. Lan, J. Liu, G. Li, Y.-D. Wu and A. Lei, Revealing a Second Transmetalation Step in the Negishi Coupling and Its Competition with Reductive Elimination: Improvement in the Interpretation of the Mechanism of Biaryl Syntheses, J. Am. Chem. Soc., 2009, 131, 10201–10210 CrossRef CAS PubMed.
- Y. Yang, N. J. Oldenhuis and S. L. Buchwald, Mild and General Conditions for Negishi Cross-Coupling Enabled by the Use of Palladacycle Precatalysts, Angew. Chem., Int. Ed., 2013, 52, 615–619 CrossRef CAS PubMed.
- J. Sherwood, J. H. Clark, I. J. S. Fairlamb and J. M. Slattery, Solvent effects in palladium catalysed cross-coupling reactions, Green Chem., 2019, 21, 2164–2213 Search PubMed.
- Q. H. Zhang, K. D. Vigier, S. Royer and F. Jerome, Deep eutectic solvents: syntheses, properties and applications, Chem. Soc. Rev., 2012, 41, 7108–7146 Search PubMed.
- E. L. Smith, A. P. Abbott and K. S. Ryder, Deep Eutectic Solvents (DESs) and Their Applications, Chem. Rev., 2014, 114, 11060–11082 Search PubMed.
- J. Garcia-Alvarez, Deep Eutectic Mixtures: Promising Sustainable Solvents for Metal-Catalysed and Metal-Mediated Organic Reactions, Eur. J. Inorg. Chem., 2015, 5147–5157 CrossRef CAS.
- J. Garcia-Alvarez, E. Hevia and V. Capriati, Reactivity of Polar Organometallic Compounds in Unconventional Reaction Media: Challenges and Opportunities, Eur. J. Org. Chem., 2015, 6779–6799 Search PubMed.
- P. Liu, J. W. Hao, L. P. Mo and Z. H. Zhang, Recent advances in the application of deep eutectic solvents as sustainable media as well as catalysts in organic reactions, RSC Adv., 2015, 5, 48675–48704 RSC.
- D. A. Alonso, A. Baeza, R. Chinchilla, G. Guillena, I. M. Pastor and D. J. Ramón, Deep Eutectic Solvents: The Organic Reaction Medium of the Century, Eur. J. Org. Chem., 2016, 2016, 612–632 Search PubMed.
- S. Khandelwal, Y. K. Tailor and M. Kumar, Deep eutectic solvents (DESs) as eco-friendly and sustainable solvent/catalyst systems in organic transformations, J. Mol. Liq., 2016, 215, 345–386 Search PubMed.
- X. Q. Xiong, Q. Han, L. Shi, S. Y. Xiao and C. Bi, Application of Deep-Eutectic Solvents in Green Organic Synthesis, Chin. J. Org. Chem., 2016, 36, 480–489 CrossRef CAS.
- A. P. Abbott, K. J. Edler and A. J. Page, Deep eutectic solvents-The vital link between ionic liquids and ionic solutions, J. Chem. Phys., 2021, 155, 5 Search PubMed.
- B. B. Hansen, et al., Deep Eutectic Solvents: A Review of Fundamentals and Applications, Chem. Rev., 2021, 121, 1232–1285 Search PubMed.
- V. I. Parvulescu and C. Hardacre, Catalysis in ionic liquids, Chem. Rev., 2007, 107, 2615–2665 Search PubMed.
- J. P. Hallett and T. Welton, Room-Temperature Ionic Liquids: Solvents for Synthesis and Catalysis. 2, Chem. Rev., 2011, 111, 3508–3576 Search PubMed.
- J. Li, S. Yang, W. Wu and H. Jiang, Recent Advances in Pd-Catalyzed Cross-Coupling Reaction in Ionic Liquids, Eur. J. Org. Chem., 2018, 2018, 1284–1306 Search PubMed.
- D. W. Bruce, Y. A. Gao, J. N. C. Lopes, K. Shimizu and J. M. Slattery, Liquid-Crystalline Ionic Liquids as Ordered Reaction Media for the Diels-Alder Reaction, Chem. – Eur. J., 2016, 22, 16113–16123 Search PubMed.
- Y. A. Gao, J. M. Slattery and D. W. Bruce, Columnar thermotropic mesophases formed by dimeric liquid-crystalline ionic liquids exhibiting large mesophase ranges, New J. Chem., 2011, 35, 2910–2918 RSC.
- J. D. Scholten, B. C. Leal and J. Dupont, Transition Metal Nanoparticle Catalysis in Ionic Liquids, ACS Catal., 2012, 2, 184–200 Search PubMed.
- H. P. Steinruck and P. Wasserscheid, Ionic Liquids in Catalysis, Catal. Lett., 2015, 145, 380–397 Search PubMed.
- R. L. Vekariya, A review of ionic liquids: Applications towards catalytic organic transformations, J. Mol. Liq., 2017, 227, 44–60 Search PubMed.
- F. T. U. Kohler, B. Morain, A. Weiß, M. Laurin, J. Libuda, V. Wagner, B. U. Melcher, X. Wang, K. Meyer and P. Wasserscheid, Surface-Functionalized Ionic Liquid Crystal–Supported Ionic Liquid Phase Materials: Ionic Liquid Crystals in Mesopores, ChemPhysChem, 2011, 12, 3539–3546 Search PubMed.
- S. E. Hooshmand, R. Afshari, D. J. Ramón and R. S. Varma, Deep eutectic solvents: cutting-edge applications in cross-coupling reactions, Green Chem., 2020, 22, 3668–3692 RSC.
- H. Zong, H. Huang, J. Liu, G. Bian and L. Song, Added-Metal-Free Catalytic Nucleophilic Addition of Grignard Reagents to Ketones, J. Org. Chem., 2012, 77, 4645–4652 Search PubMed.
- G. Osztrovszky, T. Holm and R. Madsen, Ultrafast Grignard addition reactions in the presence of water, Org. Biomol. Chem., 2010, 8, 3402–3404 Search PubMed.
- I. Manasi, M. Bortoli, D. T. Bowron, M. Campana, O. S. Hammond, T. F. Headen, J. Hooton, E. Hevia, M. Cascella, O. Eisenstein and K. J. Edler, Are Grignard Reactions in Deep Eutectic Solvents Interface-Driven?, Angew. Chem., Int. Ed., 2025, 64, e202513649 Search PubMed.
- S. D. Mador and A. Milet, Methyllithium “On-Water” Addition on Ketone: H-Bond Network and Mechanism, J. Comput. Chem., 2025, 46, e70200 Search PubMed.
- C. Vidal, J. Garcia-Alvarez, A. Hernan-Gomez, A. R. Kennedy and E. Hevia, Introducing Deep Eutectic Solvents to Polar Organometallic Chemistry: Chemoselective Addition of Organolithium and Grignard Reagents to Ketones in Air, Angew. Chem., Int. Ed., 2014, 53, 5969–5973 Search PubMed.
- E. B. Pinxterhuis, M. Giannerini, V. Hornillos and B. L. Feringa, Fast, greener and scalable direct coupling of organolithium compounds with no additional solvents, Nat. Commun., 2016, 7, 11698 Search PubMed.
- V. Mallardo, R. Rizzi, F. C. Sassone, R. Mansueto, F. M. Perna, A. Salomone and V. Capriati, Regioselective desymmetrization of diaryltetrahydrofurans via directed ortho-lithiation: an unexpected help from green chemistry, Chem. Commun., 2014, 50, 8655–8658 RSC.
- F. C. Sassone, F. M. Perna, A. Salomone, S. Florio and V. Capriati, Unexpected lateral-lithiation-induced alkylative ring opening of tetrahydrofurans in deep eutectic solvents: synthesis of functionalised primary alcohols, Chem. Commun., 2015, 51, 9459–9462 Search PubMed.
- C. Vidal, J. Garcia-Alvarez, A. Hernan-Gomez, A. R. Kennedy and E. Hevia, Exploiting Deep Eutectic Solvents and Organolithium Reagent Partnerships: Chemoselective Ultrafast Addition to Imines and Quinolines Under Aerobic Ambient Temperature Conditions, Angew. Chem., Int. Ed., 2016, 55, 16145–16148 CrossRef CAS PubMed.
- S. Ghinato, G. Dilauro, F. M. Perna, V. Capriati, M. Blangetti and C. Prandi, Directed ortho-metalation–nucleophilic acyl substitution strategies in deep eutectic solvents: the organolithium base dictates the chemoselectivity, Chem. Commun., 2019, 55, 7741–7744 RSC.
- M. Fairley, L. J. Bole, F. F. Mulks, L. Main, A. R. Kennedy, C. T. O'Hara, J. García-Alvarez and E. Hevia, Ultrafast amidation of esters using lithium amides under aerobic ambient temperature conditions in sustainable solvents, Chem. Sci., 2020, 11, 6500–6509 Search PubMed.
- A. F. Quivelli, G. D'Addato, P. Vitale, J. García-Álvarez, F. M. Perna and V. Capriati, Expeditious and practical synthesis of tertiary alcohols from esters enabled by highly polarized organometallic compounds under aerobic conditions in Deep Eutectic Solvents or bulk water, Tetrahedron, 2021, 81, 131898 Search PubMed.
- D. Elorriaga, B. Parra-Cadenas, A. Antiñolo, F. Carrillo-Hermosilla and J. García-Álvarez, Combination of air/moisture/ambient temperature compatible organolithium chemistry with sustainable solvents: selective and efficient synthesis of guanidines and amidines, Green Chem., 2022, 24, 800–812 RSC.
- S. E. García-Garrido, A. Presa Soto, E. Hevia and J. García-Álvarez, Advancing Air- and Moisture-Compatible s-Block Organometallic Chemistry Using Sustainable Solvents, Eur. J. Inorg. Chem., 2021, 2021, 3116–3130 Search PubMed.
- L. Cicco, A. Yousaf, P. Vitale, F. M. Perna and V. Capriati, A sustainable twist on the Ritter reaction: iron-based deep eutectic solvents as a green route to amide synthesis, RSC Sustainability, 2025, 3, 4079–4086 RSC.
- V. Alizadeh, F. Malberg, A. A. H. Pádua and B. Kirchner, Are There Magic Compositions in Deep Eutectic Solvents? Effects of Composition and Water Content in Choline Chloride/Ethylene Glycol from Ab Initio Molecular Dynamics, J. Phys. Chem. B, 2020, 124, 7433–7443 Search PubMed.
- J. García-Álvarez, E. Hevia and V. Capriati, The Future of Polar Organometallic Chemistry Written in Bio-Based Solvents and Water, Chem. – Eur. J., 2018, 24, 14854–14863 Search PubMed.
- G. Dilauro, A. Francesca Quivelli, P. Vitale, V. Capriati and F. M. Perna, Water and Sodium Chloride: Essential Ingredients for Robust and Fast Pd-Catalysed Cross-Coupling Reactions between Organolithium Reagents and (Hetero)aryl Halides, Angew. Chem., Int. Ed., 2019, 58, 1799–1802 CrossRef CAS PubMed.
- G. Dilauro, C. S. Azzollini, P. Vitale, A. Salomone, F. M. Perna and V. Capriati, Scalable Negishi Coupling between Organozinc Compounds and (Hetero)Aryl Bromides under Aerobic Conditions when using Bulk Water or Deep Eutectic Solvents with no Additional Ligands, Angew. Chem., Int. Ed., 2021, 60, 10632–10636 Search PubMed.
- A. Krasovskiy, C. Duplais and B. H. Lipshutz, Zn-Mediated, Pd-Catalyzed Cross-Couplings in Water at Room Temperature Without Prior Formation of Organozinc Reagents, J. Am. Chem. Soc., 2009, 131, 15592–15593 CrossRef CAS PubMed.
- C. Duplais, A. Krasovskiy and B. H. Lipshutz, Organozinc Chemistry Enabled by Micellar Catalysis. Palladium-Catalyzed Cross-Couplings between Alkyl and Aryl Bromides in Water at Room Temperature, Organometallics, 2011, 30, 6090–6097 CrossRef CAS PubMed.
- A. Krasovskiy, C. Duplais and B. H. Lipshutz, Stereoselective Negishi-like Couplings Between Alkenyl and Alkyl Halides in Water at Room Temperature, Org. Lett., 2010, 12, 4742–4744 Search PubMed.
- C. Duplais, A. Krasovskiy, A. Wattenberg and B. H. Lipshutz, Cross-couplings between benzylic and aryl halides “on water”: synthesis of diarylmethanes, Chem. Commun., 2010, 46, 562–564 Search PubMed.
- G. Dilauro, L. Cicco, P. Vitale, F. M. Perna and V. Capriati, Ligand-Free Pd-Catalyzed Reductive Mizoroki-Heck Reaction Strategy for the One-Pot Synthesis of Functionalized Oxygen Heterocycles in Deep Eutectic Solvents, Eur. J. Org. Chem., 2023, 26, e202200814 CrossRef CAS.
- F. D'Amico, C. Papucci, D. Franchi, G. Reginato, M. Calamante, L. Zani, A. Dessì and A. Mordini, Sustainable Pd-Catalyzed Direct Arylation of Thienyl Derivatives with (Hetero)aromatic Bromides under Air in Deep Eutectic Solvents, ACS Sustainable Chem. Eng., 2022, 10, 3037–3047 CrossRef.
- A. Antenucci and S. Dughera, Cross-Coupling Reactions between Arenediazonium-Benzenedisulfonimides and Organolithium Reagents in a Deep Eutectic Solvent, ChemistrySelect, 2023, 8, e202303046 CrossRef CAS.
- M. Niakan and M. Masteri-Farahani, An efficient clean and sustainable methodology for catalytic C-C coupling process over a Pd-free magnetically recoverable cobalt catalyst, J. Mol. Liq., 2022, 355, 118932 CrossRef CAS.
- M. Ramos-Martín, J. García-Alvarez and A. P. Soto, Access to Substituted 1,1-Diarylalkanes by Friedel-Crafts Benzylations Mediated by FeCl-based Deep Eutectic Solvents, ChemSusChem, 2025, 18, e202400892 CrossRef PubMed.
- J. Liu, Y. Deng, H. Wang, H. Zhang, G. Yu, B. Wu, H. Zhang, Q. Li, T. B. Marder, Z. Yang and A. Lei, Effective Pd-Nanoparticle (PdNP)-Catalyzed Negishi Coupling Involving Alkylzinc Reagents at Room Temperature, Org. Lett., 2008, 10, 2661–2664 CrossRef CAS PubMed.
- X. Marset, A. Khoshnood, L. Sotorríos, E. Gómez-Bengoa, D. A. Alonso and D. J. Ramón, Deep Eutectic Solvent Compatible Metallic Catalysts: Cationic Pyridiniophosphine Ligands in Palladium Catalyzed Cross-Coupling Reactions, ChemCatChem, 2017, 9, 1269–1275 CrossRef CAS.
- M. J. Rodríguez-Álvarez, J. García-Álvarez, M. Uzelac, M. Fairley, C. T. O'Hara and E. Hevia, Introducing Glycerol as a Sustainable Solvent to Organolithium Chemistry: Ultrafast Chemoselective Addition of Aryllithium Reagents to Nitriles under Air and at Ambient Temperature, Chem. – Eur. J., 2018, 24, 1720–1725 CrossRef PubMed.
- C. Sambiagio, D. Schönbauer, R. Blieck, T. Dao-Huy, G. Pototschnig, P. Schaaf, T. Wiesinger, M. F. Zia, J. Wencel-Delord, T. Besset, B. U. W. Maes and M. Schnürch, A comprehensive overview of directing groups applied in metal-catalysed C–H functionalisation chemistry, Chem. Soc. Rev., 2018, 47, 6603–6743 RSC.
- M. Alami, C. Amatore, S. Bensalem, A. Choukchou-Brahim and A. Jutand, Kinetics of the Oxidative Addition of ortho-Substituted Aryl Halides to Palladium(0) Complexes, Eur. J. Inorg. Chem., 2001, 2001, 2675–2681 Search PubMed.
- X. L. Wang, X. M. Ji, C. D. Shao, Y. Zhang and Y. H. Zhang, Palladium-catalyzed C-H alkylation of 2-phenylpyridines with alkyl iodides, Org. Biomol. Chem., 2017, 15, 5616–5624 RSC.
- C. Amatore, E. Carre, A. Jutand and M. A. M'Barki, Rates and Mechanism of the Formation of Zerovalent Palladium Complexes from Mixtures of Pd(OAc)2 and Tertiary Phosphines and Their Reactivity in Oxidative Additions, Organometallics, 1995, 14, 1818–1826 CrossRef CAS.
- N. W. J. Scott, M. J. Ford, C. Schotes, R. R. Parker, A. C. Whitwood and I. J. S. Fairlamb, The ubiquitous cross-coupling catalyst system ‘Pd(OAc)2’/2PPh3 forms a unique dinuclear PdI complex: an important entry point into catalytically competent cyclic Pd3 clusters, Chem. Sci., 2019, 10, 7898–7906 RSC.
- T. J. Colacot, M. W. Hooper and G. A. Grasa, UK Pat., PCT/GB2010/051232, 2011 Search PubMed.
- C. C. C. Johansson Seechurn, T. Sperger, T. G. Scrase, F. Schoenebeck and T. J. Colacot, Understanding the Unusual Reduction Mechanism of Pd(II) to Pd(I): Uncovering Hidden Species and Implications in Catalytic Cross-Coupling Reactions, J. Am. Chem. Soc., 2017, 139, 5194–5200 CrossRef CAS PubMed.
- C. Fricke, T. Sperger, M. Mendel and F. Schoenebeck, Catalysis with Palladium(I) Dimers, Angew. Chem., Int. Ed., 2021, 60, 3355–3366 CrossRef CAS PubMed.
- C. Amatore, M. Azzabi and A. Jutand, Role and effects of halide ions on the rates and mechanisms of oxidative addition of iodobenzene to low-ligated zerovalent palladium complexes Pd0(PPh3)2, J. Am. Chem. Soc., 1991, 113, 8375–8384 Search PubMed.
- C. Amatore and A. Jutand, Anionic Pd(0) and Pd(II) Intermediates in Palladium-Catalyzed Heck and Cross-Coupling Reactions, Acc. Chem. Res., 2000, 33, 314–321 Search PubMed.
- M. Kolter, K. Böck, K. Karaghiosoff and K. Koszinowski, Anionic Palladium(0) and Palladium(II) Ate Complexes, Angew. Chem., Int. Ed., 2017, 56, 13244–13248 CrossRef CAS PubMed.
- S. Kozuch, C. Amatore, A. Jutand and S. Shaik, What Makes for a Good Catalytic Cycle? A Theoretical Study of the Role of an Anionic Palladium(0) Complex in the Cross-Coupling of an Aryl Halide with an Anionic Nucleophile, Organometallics, 2005, 24, 2319–2330 CrossRef CAS.
- I. N. Houpis, C. Huang, U. Nettekoven, J. G. Chen, R. Liu and M. Canters, Carboxylate Directed Cross-Coupling Reactions in the Synthesis of Trisubstituted Benzoic Acids, Org. Lett., 2008, 10, 5601–5604 CrossRef CAS PubMed.
- I. Kalvet, G. Magnin and F. Schoenebeck, Rapid Room-Temperature, Chemoselective C-C Coupling of Poly(pseudo)halogenated Arenes Enabled by Palladium(I) Catalysis in Air, Angew. Chem., Int. Ed., 2017, 56, 1581–1585 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |