Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Polystyrene-bound thioxanthone – a heterogeneous photocatalyst for alcohol oxidation via singlet oxygen production
Max
Schmallegger
 *a,
Luis
Herbst
ab,
Renata
Raptová
a,
Mathias
Wiech
a,
Dana
Dvoranová
c,
Dmytro
Neshchadin
*a,
Luis
Herbst
ab,
Renata
Raptová
a,
Mathias
Wiech
a,
Dana
Dvoranová
c,
Dmytro
Neshchadin
 a and
Michal
Zalibera
a and
Michal
Zalibera
 c
c
aInstitute of Physical and Theoretical Chemistry, Graz University of Technology, Stremayrgasse 9, 8010 Graz, Austria. E-mail: schmallegger@tugraz.at
bTH-Köln; Fakultät 11, Campus Leverkusen, Campusplatz 1, 51379 Leverkusen, Germany
cInstitute of Physical Chemistry and Chemical Physics, Slovak University of Technology in Bratislava, Radlinského 9, SK-81237 Bratislava, Slovak Republic
First published on 2nd February 2026
Abstract
We have synthesized a photocatalyst in which thioxanthone is covalently attached on a polystyrene (TX@PS) polymer and investigated its activity in the photocatalytic oxidation of alcohols to aldehydes. Upon LED irradiation (405 nm), benzyl alcohol is converted to benzaldehyde at a yield of 73% and hydroxymethylfurfural forms formic acid at 99%. Employing the photocatalyst for consecutive runs (up to three) results in product yields of 51% for the oxidation of benzaldehyde and 98% for hydroxymethylfurfural. EPR provides evidence that the oxidation proceeds via a singlet oxygen route. This proof-of-concept study shows that TX@PS can straightforwardly be prepared. Using this catalyst provides yields reflecting comparable systems and allows a convenient isolation of the products.
Introduction
Photocatalysis has emerged as a pivotal technology for addressing global environmental and energy challenges. By utilizing light energy to drive chemical reactions, photocatalysis offers a sustainable and efficient pathway for applications such as water purification, air detoxification, hydrogen production, and CO2 reduction.1–4 Unlike conventional catalytic processes that necessitate high temperatures and pressures, photocatalysis operates under mild conditions, thereby reducing energy consumption and environmental impact.In heterogeneous catalysis, a solid photocatalyst facilitates reactions in liquid or gaseous media. This technique offers several advantages, including facile catalyst recovery, high stability, and suitability for large-scale applications.5,6
Despite its advantages, photocatalysis faces challenges such as low quantum efficiency, rapid charge recombination, and limited visible light absorption. The development of novel photocatalysts with enhanced stability, efficiency, and selectivity is critical for expanding the practical applications of this technology.7–9
This study presents an approach that uses polymer-bound thioxanthone (TX) as a photosensitizer for heterogeneous photocatalysis. Thioxanthone functions as an effective electron transfer (ET) agent and triplet energy (EnT) photosensitizer.10–13 It has been employed as a photocatalyst in cycloadditions, photoinduced rearrangements, alkene isomerization, and other transformations.13–15 In many cases, TX has been investigated in bulk solutions, but TX moieties have also been incorporated into supramolecular structures such as metal–organic frameworks (MOFs), covalent organic frameworks (COFs), covalent organic cages (COCs), metal–organic cages (MOCs), and (porous) polymers (PPs). The reactivity of organic molecules in confined spaces differs from that in isotropic solutions. For example, ground-state reactions within the cavities of supramolecular systems, like MOFs, COFs, COCs, MOCs, and PPs, have shown accelerated reaction rates, enhanced regioselectivity and stereoselectivity, and in some cases, different reaction mechanisms than in the bulk solution.16–21
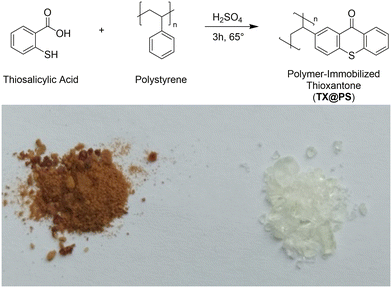
Polymers offer several advantages as a platform for photocatalysis: they can be synthesized from a range of monomers with different properties and varying pore sizes, providing flexibility to fine-tune interactions with guest-molecules.22,23 Such polymers can be examined in the solid state or when dissolved, allowing for reactivity and analysis in both liquid and solid states. To study TX incorporated into (porous) polymers, this research utilizes the possibility of directly grafting TX moieties onto polystyrene via an in situ process (Scheme 1). The photosensitizing moiety thus becomes covalently attached to the polymer backbone (TX@PS).10,24,25
This approach produces a polymer with distinct photochemical properties and provides precise control over the photocatalytic oxidation of alcohols under mild conditions. The strategic placement of TX within the polymer matrix ensures stability and minimizes the risk of removal by solvents, thereby enhancing the overall efficacy of the system.
Experimental section
Materials
All chemicals employed in this study were purchased from commercial suppliers and used without further purification.Preparation of polymer-bound thioxanthone
Polymer-bound thioxanthone was prepared using a two-step procedure. First, we prepared polystyrene using a photochemical approach: 170 mg of the bisacylphosphane oxide photoinitiator Irgacure 819 (BAPO) were dissolved in 5 ml of methanol and mixed with 5 ml of a styrene/divinylbenzene crosslinker mixture (volume ratios 2.8 mL/2.2 mL, 3.3 mL/1.7 mL, 3.8 mL/1.2 mL and 4.3 mL/0.7 mL, respectively). The solutions were irradiated at 405 nm for one hour. The obtained polymers were washed with ethanol and dried overnight at 60 °C.In the second step, thioxanthone moieties were synthesized on the backbone of polystyrene via a reaction with thiosalicylic acid (Scheme 1). For this, 1 g of the polymer was added to a 20 mM solution (2 ml) of thiosalicylic acid in concentrated sulfuric acid. The mixture was stirred at 65 °C for 3 h. The resulting polymer was washed with ethanol and dried at 60 °C overnight.
Characterization of the polymer-bound thioxanthone catalyst
Evaluation of singlet oxygen production by electron paramagnetic resonance (EPR) spectroscopy
EPR experiments were conducted using a Miniscope MS300 X-band EPR spectrometer (Magnettech). An aqueous solution of the sterically hindered amine 4-oxo-2,2,6,6-tetramethylpiperidine (TEMP; 50 mM) and 10 mg of the TX@PS photocatalyst was irradiated with a high-power LED (λmax = 405 nm) under an ambient O2 atmosphere for 30 min.Polystyrene-bound TX as a heterogeneous photocatalyst for alcohol oxidation
10 mg of the TX@PS photocatalyst were added to a 10 mM solution (benzyl alcohol in CH3CN-d3 or 5-HMF in H2O-d2, respectively; volume = 1 mL). The solution was bubbled with O2 for 10 minutes and irradiated with a high-power LED (λmax = 405 nm) for 18 h. Afterwards, the catalyst was separated from the reaction mixture by centrifugation, washed with ethanol and dried at 60 °C overnight for subsequent use. The reaction mixture was directly used for product analysis with 1H NMR.Results and discussion
Photochemical formation of polystyrene
 | ||
| Fig. 1 Normalized optical spectra of solid polystyrene (black, dotted line), polystyrene-immobilized thioxanthone (red, solid line) and TX in acetonitrile solution (black, dashed line). | ||
For the reactants to access the catalytically active species efficiently, it is critical that the composite provides a sufficiently high surface area. BET measurements revealed that TX@PS is mesoporous, with a surface area of approximately 5.4 m2 g−1 (see SI, Fig. S10).
Chemical stability of TX@PS
The chemical stability of heterogeneous photocatalysts is critical to ensure catalytic activity over prolonged periods and avoid impurities in the final product. To evaluate the chemical stability of the TX@PS system and its potential application under different solvent conditions, it was dispersed under three distinct solute conditions: nonpolar aprotic (toluene), polar aprotic (acetonitrile), and polar protic (water) conditions. NMR spectra were recorded for the solutions after storing them at room temperature and 70 °C for 24 h. No signals attributable to dissolved TX@PS are detected in water and acetonitrile, whereas clear signals corresponding to the dissolved polymer are detected in toluene (see SI, Fig. S4). While crosslinked polystyrene (PS) is generally insoluble in toluene,26 the detection of dissolved fractions indicates the presence of non-crosslinked or lightly crosslinked polystyrene chains, showing the potential limitations of this approach for application in nonpolar aprotic solvents, while still rationalizing its use in the other two solvent systems.Polystyrene-bound TX for photochemical singlet oxygen production
The ability of the polystyrene-bound thioxanthone to produce singlet oxygen was evaluated using electron paramagnetic resonance (EPR) spectroscopy. The photocatalyst was dispersed in an oxygen-saturated aqueous solution of the EPR-sterically hindered amine TEMP. Upon irradiation, the polystyrene-bound moieties undergo excitation and intersystem crossing to the triplet state. Subsequently, this excited triplet state undergoes energy transfer to molecular oxygen, generating singlet oxygen.13 This singlet oxygen further reacts with TEMP to form a persistent TEMPO radical, which can be conveniently detected by EPR (Scheme 2).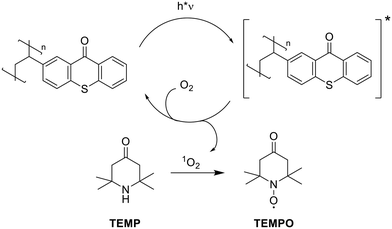 | ||
| Scheme 2 Singlet oxygen production via energy transfer from photoexcited polystyrene-bound TX and subsequent oxidation of TEMP, resulting in the formation of the persistent TEMPO radical. | ||
Fig. 2 shows the corresponding EPR spectra before and after 30 min of continuous irradiation at 405 nm. We employed 405 nm for irradiation, as thioxanthone exhibits significant absorbance at this wavelength, whereas polystyrene does not demonstrate absorbance (see Fig. 1). This ensures that the incident light is predominantly absorbed by the photo-active thioxanthone. Before irradiation, no signals are detected, whereas a characteristic triplet signal, attributable to the persistent TEMPO radical with a nitrogen hyperfine coupling constant of aN = 1.596 mT is observed after light exposure. This further supports the formation of photoactive TX moieties on the polystyrene backbone and illustrates its potential as a heterogeneous photocatalyst (see also SI, Fig. S9).
 | ||
| Fig. 2 EPR spectra of an oxygen-saturated aqueous solution of TEMP before (red) and after (black) irradiation for 30 minutes with a high-power LED (λmax = 405 nm). | ||
Assessing polystyrene-bound TX as a heterogeneous photocatalyst for alcohol oxidation
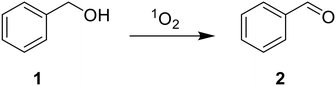
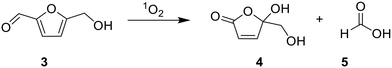
As a proof-of-concept, we employ TX@PS as a heterogeneous photocatalyst for two model reactions: the oxidation of benzyl alcohol (1) to benzaldehyde (2) and the oxidation of hydroxymethylfurfural (5-HMF; 3) to 5-(hydroxymethyl)-5-hydroxyfuran-2(5H)-one (4) and formic acid (5). The reactions were chosen to demonstrate the potential of the photocatalyst to work both in organic solvents (e.g. acetonitrile: Table 1) and under aqueous conditions (Table 2) and proceed via singlet oxygen: upon excitation of TX@PS and subsequent energy transfer leading, the singlet oxygen acts as an oxidant either via hydrogen atom transfer or direct insertion reaction.10,13| Modifications of reaction conditionsa | Product yieldb [%] |
|---|---|
| a Reagents and conditions: benzyl alcohol (10 mmol), TX@PS (50 mg) in acetonitrile (1 ml) at RT; 405 nm LED irradiation under O2 atmosphere for 18 h. b Determined by 1H NMR spectroscopy. | |
| None | 73 |
| Catalyst re-used; second run | 60 |
| Catalyst re-used; third run | 51 |
| 4-hour irradiation | 4 |
| No light | 1 |
| No catalyst | 0 |
| No light, T = 70 °C | 1 |
| Modifications of reaction conditionsa | Conversionb [%] | Product ratio 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5b [%] 5b [%] |
|---|---|---|
| a Reagents and conditions: hydroxymethylfurfural (10 mmol), TX@PS (50 mg) in water (1 ml) at RT; 405 nm LED irradiation under O2 for 18 h. b Determined by 1H NMR spectroscopy. | ||
| None | >99 | 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 76 76 |
| Catalyst re-used; second run | >99 | 27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 73 73 |
| Catalyst re-used; third run | >99 | 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70 70 |
| 4-hour irradiation | 59 | 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 50 |
| No light | 1 | — |
| No catalyst | 2 | — |
| No light, T = 70 °C | 1 | — |
Product yields for the oxidation of benzyl alcohol (1) to benzaldehyde (2) reach 73% in the first run and decrease to 51% upon subsequent reuse of the same catalyst (Table 1). This reduction in catalytic efficiency is potentially due to photobleaching of the chromophore upon prolonged irradiation.27–29 Reducing the irradiation time from 18 to 4 h significantly reduces the reaction yield to only 4%. This illustrates the necessity of long-term irradiation. Virtually no formation of 2 was detected without the photocatalyst or when irradiation was omitted, rationalizing the reaction pathway displayed in Scheme 2, where TX@PS produces singlet oxygen via energy transfer upon irradiation (SI, Fig. S5 and S6). Whilst under our reaction conditions, the solution heated up to a maximum of only 35 °C, it is still important to evaluate the influence of elevated temperature on the catalytic activity.30 Therefore, we performed the experiment in the presence of TX@PS at elevated temperature (T = 70 °C), without light irradiation. Again, no product is detected, underpinning that light irradiation is necessary for catalytic activity.
The oxidation of 5-HMF (3) is well documented and can afford multiple products, such as 5-hydroxymethyl-2-furancarboxylic acid, 2,5-diformylfuran, 2,5-furandicarboxylic acid, 5-(hydroxymethyl)-5-hydroxyfuran-2(5H)-one, and formic acid, depending on the reaction conditions.31–33 Under our conditions, only 5-(hydroxymethyl)-5-hydroxyfuran-2(5H)-one (4) and formic acid (5) were detected as products (SI, Fig. S7). The oxidation of 5-HMF (3) proceeded quantitatively (>99%), even after catalyst reuse. When the irradiation time was shortened from 18 h to 4 h, the conversion of 3 decreased substantially to 59%. In the absence of either the photocatalyst or light, no significant educt conversion was observed, even at elevated temperatures of 70 °C (2%, 1% and 1%, respectively), confirming that TX@PS functions as a heterogeneous photocatalyst for singlet oxygen generation (SI, Fig. S7–S9). To determine the relative amounts of 4 and 5 formed, we compared the integrals of characteristic NMR resonances (3.71 ppm (2H) for 4; 8.08 ppm (1H) for 5).33 In all three catalytic runs, 5 was the major product; however, the ratio of 4 to 5 shifted upon catalyst reuse (from 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 76 in the first run to 30
76 in the first run to 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70 in the third; Table 2 and SI, Fig. S7). This trend again suggests changes in the catalytically active species, consistent with our observations for the oxidation of 1 (see above).
70 in the third; Table 2 and SI, Fig. S7). This trend again suggests changes in the catalytically active species, consistent with our observations for the oxidation of 1 (see above).
The product yields obtained from our model reactions using TX@PS as a heterogeneous photocatalyst are comparable to those reported in previous studies:34–36 specifically, the oxidation of benzyl alcohol to benzaldehyde achieved a 73% product yield with our heterogeneous system, whereas a comparable homogeneous approach using TX in solution yielded up to 82%.36 Despite the slightly lower yield, our method offers significant practical advantages, including a simplified work-up process and catalyst reusability, owing to the heterogeneous nature of the TX@PS photocatalyst. For the oxidation of 5-HMF, TX@PS exhibits high product yields even after repeated reuse of the heterogeneous photocatalyst, consistent with results reported for other organic dyes.33
Conclusions
Herein, we present a proof-of-concept study on the grafting and immobilization of TX on PS. The material exhibits activity as a heterogeneous photocatalyst for alcohol oxidation via a singlet oxygen pathway. For the oxidation of benzyl alcohol in an organic solvent (acetonitrile) and hydroxymethylfurfural in an aqueous solution, product yields of 73% and 99% are achieved, respectively. Tables 3 and 4 show a comparison of TX@PS with different photo-catalytic systems employed for the oxidation of benzyl alcohol and 5-HMF, respectively: For the oxidation of benzyl alcohol to benzaldehyde, TX@PS achieves product yields of 73%; different systems reported in literature show yields ranging from 36–99% (Table 3). Therefore, our photo-catalyst demonstrates photocatalytic performance comparable to or exceeding that of as established systems reported in literature.| Catalyst | Reaction conditions | Yield/% | Reaction runs | Ref. |
|---|---|---|---|---|
| TX@PS | 18 h, 405 nm, CH3CN, O2, RT | 73 | 3 runs | This work |
| Bi4Ti3O12 nanosheets | 5 h, 360 nm, benzo-trifluoride, atm, RT | 36 | — | 37 |
| BiOBr/g-C3N4 | 5 h, Xe lamp, in bulk, atm, RT | 36 | 4 runs | 38 |
| Photoactive MOF | 16 h, white light EtOH/H2O/H2O2, atm, RT | 75 | 3 runs | 39 |
| CsPbBr3/Ni4P2 | 6 h, >400 nm, in CH3CN, O2, RT | 99 | 3 runs | 40 |
| Nano-TiO2 | 6 h, >365 nm, in CH3CN, atm, 30 °C | 76 | — | 41 |
| Catalyst | Reaction conditions | Yield/% | Reaction runs | Ref. |
|---|---|---|---|---|
| a Main product: 5-formyl-2-furancarboxylic acid. b Main product: 2,5-diformylfuran. | ||||
| TX@PS | 18 h, 405 nm, H2O, O2, RT | 99 | 3 runs | This work |
| In–SnS2 | 2 h, 450 nm, CH3CN, atm, RT | 73a | 3 runs | 42 |
| g-C3N4/NaNbO3 | 8 h, Xe lamp, H2O, O2, RT | 36b | 5 runs | 43 |
| (Cu2O)/TiO2 | 90 min, Xe lamp, H2O, O2, RT | 23a | 5 runs | 44 |
| SGH-TiO2 | 4 h, 515 nm, CH3CN, atm, RT | 52a | 5 runs | 45 |
For the oxidation of 5-HMF, TX@PS achieves conversion and product yields of exceeding 99%; different systems reported in literature also show quantitative educt conversion and product yields ranging from 23–73% (Table 4). Importantly, is has to be noted that depending on the catalyst and reaction conditions, different main products (5-formyl-2-furancarboxylic acid or: 2,5-diformylfuran) are reported.
In addition to high product yields, TX@PS exhibits chemical stability towards the solvents employed, facilitates a simple work-up procedure and has high reusability. Our findings suggest that the integration of organic dyes with polystyrene provides a straightforward platform for heterogeneous photocatalysis.
Conflicts of interest
There are no conflicts of interest to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI).Supplementary information is available. See DOI: https://doi.org/10.1039/d5cy01464f.
Acknowledgements
The authors are grateful for the support of Austria's Agency for Education and Internationalisation (OeAD) Aktion Österreich–Slowakei (Project SK 05/2024), Slovak Research and Development Agency (Project SK-AT-23-0004), and Scientific Grant Agency (VEGA) (1/0392/24) and (1/0422/24). This research was funded in whole, or in part, by the Austrian Science Fund (FWF) https://doi.org/10.55776/I5710. For the purpose of open access, the author has applied a CC BY public copyright licence to any Author Accepted Manuscript version arising from this submission. The authors thank NAWI Graz for financial support. We are indebted to Christoph Neubauer and Prof. Heidrun Gruber-Woelfler, TU Graz, for their help with the BET measurements.References
- A. Fujishima and K. Honda, Nature, 1972, 238, 37–38 Search PubMed.
- M. N. Chong, B. Jin, C. W. K. Chow and C. Saint, Water Res., 2010, 44, 2997–3027 CrossRef CAS.
- S. G. Kumar and L. G. Devi, J. Phys. Chem. A, 2011, 115, 13211–13241 Search PubMed.
- M. A. Fox and M. T. Dulay, Heterogeneous Photocatalysis, 1993, vol. 93 Search PubMed.
- J.-M. Herrmann, Catal. Today, 1999, 53, 115–129 CrossRef CAS.
- M. R. Hoffmann, S. T. Martin, W. Choi and D. W. Bahnemann, Chem. Rev., 1995, 95, 69–96 CrossRef CAS.
- J. Zhang, Q. Xu, Z. Feng, M. Li and C. Li, Angew. Chem., Int. Ed., 2008, 47, 1766–1769 Search PubMed.
- A. Kudo and Y. Miseki, Chem. Soc. Rev., 2009, 38, 253–278 RSC.
- F. E. Osterloh, Chem. Mater., 2008, 20, 35–54 CrossRef CAS.
- V. P. Demertzidou, E. Skolia and C. G. Kokotos, ChemCatChem, 2025, 17, e00760 Search PubMed.
- A. Vega-Peñaloza, J. Mateos, X. Companyó, M. Escudero-Casao and L. Dell'Amico, Angew. Chem., Int. Ed., 2020, 1082–1097 Search PubMed.
- L. Marzo, S. K. Pagire, O. Reiser and B. König, Angew. Chem., 2018, 130, 10188–10228 CrossRef.
- N. F. Nikitas, P. L. Gkizis and C. G. Kokotos, Org. Biomol. Chem., 2021, 19, 5237–5253 Search PubMed.
- A. Vega-Peñaloza, J. Mateos, X. Companyó, M. Escudero-Casao and L. Dell'Amico, Angew. Chem., Int. Ed., 2021, 60, 1082–1097 CrossRef PubMed.
- S. Dutta, J. E. Erchinger, F. Strieth-Kalthoff, R. Kleinmans and F. Glorius, Chem. Soc. Rev., 2024, 53, 1068–1089 RSC.
- Y. Okamoto and T. R. Ward, in Comprehensive Supramolecular Chemistry II, Elsevier, 2017, pp. 459–510 Search PubMed.
- Y. Xue, X. Hang, J. Ding, B. Li, R. Zhu, H. Pang and Q. Xu, Coord. Chem. Rev., 2021, 430, 213656 CrossRef CAS.
- Y. Fang, J. A. Powell, E. Li, Q. Wang, Z. Perry, A. Kirchon, X. Yang, Z. Xiao, C. Zhu, L. Zhang, F. Huang and H.-C. Zhou, Chem. Soc. Rev., 2019, 48, 4707–4730 RSC.
- A. B. Grommet, M. Feller and R. Klajn, Nat. Nanotechnol., 2020, 15, 256–271 CrossRef CAS.
- D. J. Cram, Nature, 1992, 356, 29–36 Search PubMed.
- D. J. Cram, Angew. Chem., Int. Ed. Engl., 1988, 27, 1009–1020 CrossRef.
- D. Wu, F. Xu, B. Sun, R. Fu, H. He and K. Matyjaszewski, Chem. Rev., 2012, 112, 3959–4015 CrossRef CAS.
- X. Mu, T. Bertron, C. Dunn, H. Qiao, J. Wu, Z. Zhao, C. Saldana and H. J. Qi, Mater. Horiz., 2017, 4, 442–449 RSC.
- M. Aydin, N. Arsu and Y. Yagci, Macromol. Rapid Commun., 2003, 24, 718–723 CrossRef CAS.
- K. Fukumori, Y. Akiyama, M. Yamato and T. Okano, ChemNanoMat, 2016, 2, 454–460 CrossRef CAS.
- A. Charlesby, J. Polym. Sci., 1953, XI, 513–520 Search PubMed.
- A. Jiménez-Almarza, A. López-Magano, R. Mas-Ballesté and J. Alemán, ACS Appl. Mater. Interfaces, 2022, 14, 16258–16268 Search PubMed.
- S. Freeburne, J. L. Sacco, E. W. Gomez and C. W. Pester, Macromol. Chem. Phys., 2023, 224, 1–9 CrossRef.
- L. S. Herculano, L. C. Malacarne, V. S. Zanuto, G. V. B. Lukasievicz, O. A. Capeloto and N. G. C. Astrath, J. Phys. Chem. B, 2013, 117, 1932–1937 CrossRef CAS PubMed.
- W. Wang, Y. Liu, X. Shao, Y. Zheng, B. Zhu, J. Tang, Q. Pan and Q. Ke, Mol. Catal., 2026, 591, 115689 CAS.
- Y. Y. Gorbanev, S. Kegnæs and A. Riisager, in Topics in Catalysis, 2011, vol. 54, pp. 1318–1324 Search PubMed.
- M. Ventura, F. Lobefaro, E. de Giglio, M. Distaso, F. Nocito and A. Dibenedetto, ChemSusChem, 2018, 11, 1305–1315 CrossRef CAS PubMed.
- T. S. A. Heugebaert, C. V. Stevens and C. O. Kappe, ChemSusChem, 2015, 8, 1648–1651 CrossRef CAS.
- E. Tacchi, G. Rossi and A. Sartorel, Chem. – Eur. J., 2025, 31, e202501124 CrossRef CAS PubMed.
- S. Najafishirtari, K. Friedel Ortega, M. Douthwaite, S. Pattisson, G. J. Hutchings, C. J. Bondue, K. Tschulik, D. Waffel, B. Peng, M. Deitermann, G. W. Busser, M. Muhler and M. Behrens, Chem. – Eur. J., 2021, 27, 16809–16833 CrossRef CAS.
- N. F. Nikitas, D. I. Tzaras, I. Triandafillidi and C. G. Kokotos, Green Chem., 2020, 22, 471–477 RSC.
- M. Shen, Y. Shi, Z. Wang, T. Wu, L. Hu and L. Wu, J. Colloid Interface Sci., 2022, 608, 2529–2538 CrossRef CAS.
- W. Wang, X. Cong, X. Zhang, W. Xu, J. Diao, Y. Bao and F. Guo, J. Environ. Chem. Eng., 2025, 13, 115552 Search PubMed.
- C. Si, X. Liu, T. Zhang, J. Xu, J. Li, J. Fu and Q. Han, Inorg. Chem., 2023, 62, 4210–4219 CrossRef CAS.
- Y. Dong, Y. Feng, Z. Li, H. Zhou, H. Lv and G. Y. Yang, ACS Catal., 2023, 13, 14346–14355 CrossRef CAS.
- D. A. Giannakoudakis, A. Qayyum, M. Barczak, R. F. Colmenares-Quintero, P. Borowski, K. Triantafyllidis and J. C. Colmenares, Appl. Catal., B, 2023, 320, 121939 CrossRef CAS.
- A. Putta Rangapppa, W. Xue, A. Chowdhury, Y. Liu, J. M. Lee and J. Zhao, ChemSusChem, 2025, 18, e202402197 CrossRef CAS PubMed.
- Y. Zhu, Y. Zhang, L. Cheng, M. Ismael, Z. Feng and Y. Wu, Adv. Powder Technol., 2020, 31, 1148–1159 CrossRef CAS.
- Q. Zhang, H. Zhang, B. Gu, Q. Tang, Q. Cao and W. Fang, Appl. Catal., B, 2023, 320, 122006 CrossRef CAS.
- A. Khan, M. Goepel, A. Kubas, D. Łomot, W. Lisowski, D. Lisovytskiy, A. Nowicka, J. C. Colmenares and R. Gläser, ChemSusChem, 2021, 14, 1351–1362 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |