Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Correction: Chromium-activated phosphors: from theory to applications
Shengqiang Liuaf,
Leipeng Liab,
Bing Chen*ac,
Quanlin Liud and
Feng Wang*ae
aDepartment of Materials Science and Engineering, City University of Hong Kong, Kowloon, Hong Kong SAR 999077, China. E-mail: fwang24@cityu.edu.hk
bCollege of Physics Science and Technology, Hebei University, Baoding, 071002, China
cCollege of Electronic and Optical Engineering and College of Flexible Electronics (Future Technology), Nanjing University of Posts and Telecommunications, Nanjing 210023, China. E-mail: bchen@njupt.edu.cn
dBeijing Municipal Key Laboratory of New Energy Materials and Technologies, School of Materials Sciences and Engineering, University of Science and Technology Beijing, Beijing 100083, China
eHong Kong Institute for Clean Energy, City University of Hong Kong, Kowloon, Hong Kong SAR 999077, China
fKey Laboratory of Optoelectronic Devices and Systems of Ministry of Education and Guangdong Province, College of Physics and Optoelectronic Engineering, Shenzhen University, Shenzhen 518060, China
First published on 13th May 2026
Abstract
Correction for ‘Chromium-activated phosphors: from theory to applications’ by Shengqiang Liu et al., Chem. Soc. Rev., 2026, 55, 1954–1998, https://doi.org/10.1039/D5CS00957J.
The authors regret that there were several errors in the original article.
Page 1955:
• the biography of Quanlin Liu should say “Since 2005, he has been a full professor in Materials Science at the University of Science and Technology Beijing (USTB).
Page 1957:
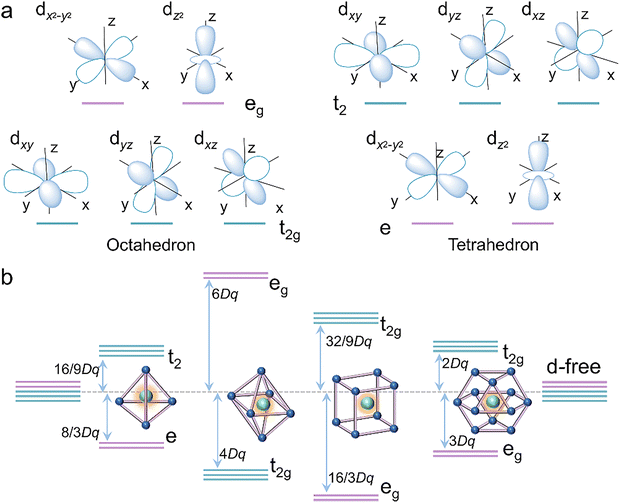
• section 2.1 should start “The 3d orbital with an angular quantum number of 2 comprises…”
• the sentence beginning “Besides the octahedron” should read “Besides the octahedron, there are some other regular polyhedra, such as 4-coordinated tetrahedron (Td symmetry), 8-coordinated cube (Oh symmetry), and 12-coordinated cuboctahedron (Oh symmetry).”
• eqn (1) should read
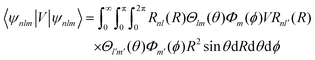 | (1) |
| ψnlm = Rnl(r)Ylm(θ, ϕ) | (2) |
Page 1958:
• the sentences near the end of section 2.1 beginning “In a tetrahedral coordination”, should read “In a tetrahedral coordination, the t2 orbitals point more directly toward the ligands, resulting in significant electrostatic interaction with higher splitting energy, while the e orbitals are directed toward the inter-ligand gaps (Fig. 2a). Therefore, the t2 orbitals of the tetrahedron lie above the e orbitals in energy.”
• the second sentence of section 2.2 should end “and L is the total orbital angular momentum quantum number.”
• “Hundt’s rule” should read “Hund’s rule”.
Page 1961:
• the sentences beginning “The anomalous luminescence” should read “The anomalous luminescence due to AFMC has been documented in inorganic matrices heavily doped with Mn2+ or Cr3+, whereas the FMC has only been experimentally observed in limited cases involving Mn2+ activated sulfide.8,57-59
Fig. 6b and c display the schematic presentations of the energy level splitting of the Cr3+–Cr3+ pair due to AFMC.”
• eqn (4) should read
| E(S) = −J[S(S + 1) − SA(SA + 1) − SB(SB + 1)] | (4) |
Page 1963:
• in the sentence beginning “Due to high coordination energy and charge balance”, the radius of Ge4+ in six-coordinated surroundings should read 0.53 Å.
Page 1965:
• eqn (7) should read
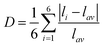 | (7) |
Fig. 2, 3 and 8 were incorrect in the original article. The correct figures are shown here.
References 1 and 342 were incorrect in the original article. The correct references are shown below as ref. 1 and 2, respectively.
The Royal Society of Chemistry apologises for these errors and any consequent inconvenience to authors and readers.
References
- G. Liu, W. Chen, Z. Xiong, Y. Wang, S. Zhang and Z. Xia, Nat. Photonics, 2024, 18, 562–568 CrossRef CAS.
- R. A. Sharrock, Genome Biol., 2008, 9, 230 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |