Open Access Article
Open Access ArticleNano-structuring hydrogen-bonded organic frameworks: strategies, composites, and functional applications
Qiang Zhu
ab and
Antonio Fernandez
 *ac
*ac
aChemistry Department, Loughborough University, Loughborough LE11 3TU, UK
bState Key Laboratory of Natural Product Chemistry, College of Chemistry and Chemical Engineering, Lanzhou University, Lanzhou, Gansu 730000, China
cFacultad de Ciencias, Avenida Fuente nueva s/n, Universidad de Granada, Spain. E-mail: antoniofernandez@ugr.es
First published on 23rd April 2026
Abstract
Hydrogen-bonded organic frameworks are a rising class of porous materials that are now recognized for their high surface areas, mild synthesis, solution processability, and biocompatibility, making them attractive for a wide range of applications. However, their bulk forms often suffer from limitations such as poor electrical conductivity and insufficient structural stability, which can hinder practical implementation. To transcend the limitations of their bulk counterparts, a powerful strategy is their transformation into pristine nanostructures or nanocomposites, which enhances properties such as conductivity, stability, and porosity. These HOF nanomaterials leverage the synergistic effects of nanoconfinement and composite matrices, leading to enhanced performance in applications ranging from photocatalysis and energy storage to photothermal therapy. This review aims to provide an overview of recent developments, focusing on the classification, fabrication strategies, and diverse applications of HOF nanomaterials, thereby offering a perspective for future research in this evolving field.
1. Introduction
Hydrogen-bonded organic frameworks (HOFs) are an emerging class of porous molecular materials assembled via non-covalent hydrogen bonding, distinguishing them from covalent organic frameworks (COFs)1,2 and metal–organic frameworks (MOFs).3,4 The origin of HOFs can be tracked to 1969, when Duchamp and Marsh reported the crystal structure of trimesic acid, which forms a honeycomb-like network featuring interpenetrating hexagonal channels—a foundational prototype of a HOF.5 In the 1990s, Wuest and colleagues pioneered the field by introducing the ‘molecular tectonics’ strategy.6,7 They synthesized tetrahedral building blocks that self-assembled into three-dimensional (3D) diamondoid networks with guest-accessible voids, conceptualizing the design of hydrogen-bonded networks. However, the inherent weakness and flexibility of hydrogen bonds often resulted in structure collapse upon guest removal, hindering HOF development and causing the field to lag behind MOFs and COFs for years. A breakthrough came two decades later when Chen and co-workers demonstrated the first HOF with permanent porosity via gas sorption and formally coined the term ‘hydrogen-bonded organic frameworks’.8 This milestone ignited renewed interest in HOFs, and the field has since evolved rapidly, increasingly leveraging computation-assisted design and automation to accelerate the discovery of stable, functional materials.9–11 In 2012, Mastalerz et al. achieved a breakthrough by increasing the Brunauer–Emmett–Teller (BET) surface area of HOFs to 2796 m2 g−1 using a triptycene-based building block (TTBI).12 In 2015, Chen et al. reported a flexible HOF exhibiting high performance for CO2 capture.13 In 2017, guided by crystal structure prediction (CSP), Cooper, Day, and coworkers discovered three new polymorphs of TTBI, including one with a record-high surface area of 3425 m2 g−1.11 Later, Cao and Liu developed a widely used robust HOF, termed PFC-1, based on a pyrene core with an extended π-conjugated system.14 In 2019, Falcaro and Doonan et al. pioneered the use of HOFs for enzyme stabilization.15 Most recently, Bu, Chen, Li, and their teams have showcased the potential of flexible HOFs for gas separation, achieving an exceptional SO2/CO2 selectivity of 7331 and a high SO2 storage density of 3.27 g cm−3 under ambient conditions.16 Owing to their desirable properties, such as large BET surface areas, mild synthetic conditions, good solution processability, and excellent biocompatibility,17 HOFs have demonstrated great potential for practical applications, including gas separation, sensing, catalysis, and energy storage.18–20Nanomaterials, broadly defined as functional materials with at least one structural dimension confined to the nanoscale (typically <100 nm), often exhibit properties dramatically distinct from their bulk counterparts.21 A key advantage lies in their ability to function as composite matrices, integrating the merits of different components to achieve enhanced synergistic functionalities. Among various substrates, HOFs have emerged as particularly attractive candidates due to their excellent solution processability and high functional tunability.
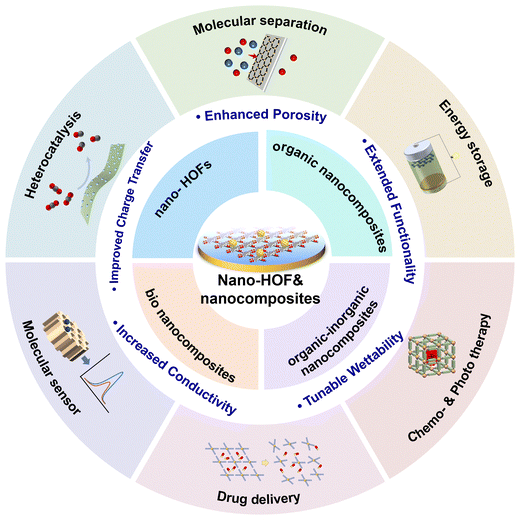
Transforming bulk HOFs into nanostructures and nanocomposites represents a promising strategy for optimizing their functional properties, as evidenced by several key enhancements. For instance, converting bulk HOFs into nanosheets or thin films increases their accessible pore surface area, thereby improving substrate accessibility. Additionally, integrating HOFs with conductive materials such as graphene oxide combines their efficient mass transfer properties with high electrical conductivity, making these composites attractive for electronic device applications. Combining HOFs with other framework materials, such as COFs, can also create heterojunctions that facilitate efficient charge separation, thereby boosting catalytic performance. This strategy also enables the tailoring of surface functionality. By coating substrates with HOFs, surface wettability can be precisely engineered, thereby optimizing reactant affinity, enhancing interfacial mass transport, and improving catalytic efficiency. Finally, employing HOFs as protective shells to encapsulate enzymes can not only stabilize them under harsh conditions but also introduces non-native functions through conformational modulation. These enhancements are expected to expand the materials' performance across a wide range of applications, including heterogeneous catalysis, energy storage, and photothermal therapy, among others (Fig. 1).
Despite a series of recent reviews on HOFs,22–27 a comprehensive summary of the current investigations in the field of HOF-based nanomaterials remains notably absent.28 Given this gap, this review aims to provide an overview of the recent developments in HOF-based nanomaterials. We will focus on their fabrication strategies, classification, and diverse applications, offering a promising perspective for future research in this field. To ensure clarity, this review categorizes the materials discussed into two distinct classes:
Pristine nano-HOFs
Materials that retain the intrinsic crystalline HOF structure but are fabricated showing effects. This includes nanosheets (NSs), nanoparticles (NPs), and nanorods (NRs).HOF nanocomposites
Hybrid materials where HOFs serve as a host matrix or template for incorporating functional nanomaterials (e.g., NPs, quantum dots). Here, the framework structure remains intact, but the functionality is enhanced by the guest species.2. Nano-structuring strategies
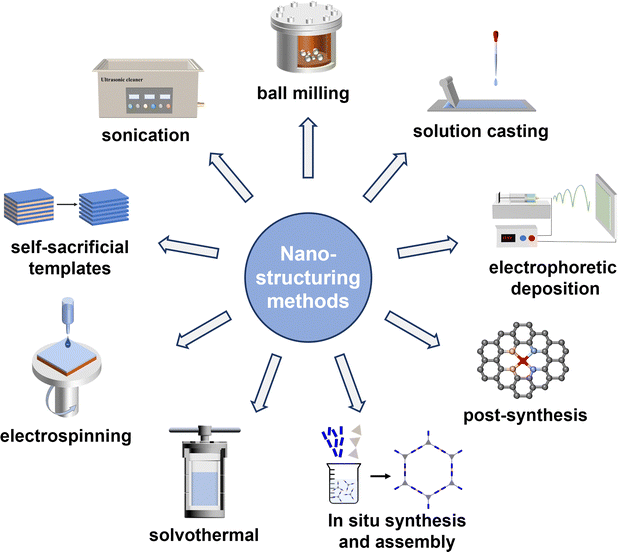
Nano-structuring strategies for HOFs and their nanocomposites fall into three categories based on the fabrication sequence: (i) in situ synthesis and assembly, (ii) in situ assembly, and (iii) post-conversion (Fig. 2).2.1. In situ synthesis and assembly
In situ synthesis and assembly integrate the in situ generation and assembly of building blocks, making it particularly effective for constructing HOFs from species with limited solubility. For instance, Wu and Wasielewski et al. reported a one-pot synthesis and crystallisation strategy employing naphthalene diimide (NDI) or perylene diimide (PDI)—each functionalized with two terminal guanine units—as electron-accepting cores. The assembly of the G2NDI and G2PDI frameworks was initiated by tert-butyloxycarbonyl (Boc) deprotection using trifluoroacetic acid (TFA) in CH2Cl2, followed by vapor diffusion of MeOH into the TFA/CH2Cl2 solution at room temperature. This process produced rod-like crystalline morphologies, resulting in G-quadruplex frameworks with high thermal stability.29 Similarly, during imidization, the molecular precursor (1,4,5,8-naphthalenetetracarboxylic dianhydride, NTCDA) spontaneously self-assembles into 2D HOF (HOF-DAT) nanosheets with a thickness of ∼1 nm and lateral sizes up to 10 µm.302.2. In situ assembly
In situ assembly represents a powerful approach for nanostructure fabrication, achieved by controlling the assembling process of HOF precursors or integrating diverse composite components. This approach typically includes, solvothermal methods, electrospinning, and self-templated assembly (Fig. 2).Solvothermal processing is a versatile strategy for the synthesis of HOFs at the nanoscale or nanocomposites (Fig. 2). This method leverages the inherent solution processability of HOF building blocks, allowing for precise control over crystal dimensions and morphologies by carefully modulating reaction parameters such as solvent polarity, temperature, and precursor concentration.31 For instance, by adjusting solvent polarity and reaction conditions, Cao and Liu et al. showed that the dimensions of PFC-1 crystals could be precisely tailored from micrometres down to nanometres (e.g., 100 × 300 nm).14 Pushing the boundaries of dimensionality, Wang et al. employed a hydrothermal polycondensation to prepare graphitic carbon nitride-based quantum dots (CNQDs, ∼2 nm). These units were then assembled via lateral hydrogen bonding into a porous, free-floating 2D film with an ultrathin thickness of approximately 2 nm.32
Electrospinning is a versatile and efficient technique that utilizes high-voltage electric fields to produce continuous fibres from polymer solutions. Li et al. fabricated antimicrobial nanofibers by electrospinning a precursor solution containing photoactive HOF building blocks and poly(vinylidene fluoride-co-hexafluoropropylene) (PVDF–HFP). During solvent evaporation, the polymer fibres solidified while simultaneously inducing the confined crystallisation of HOFs into uniform rod-like nanocrystals (∼60 nm in length) embedded within the fibre matrix, rather than merely adhering to the surface.33
Self-templated assembly of HOFs can be used to synthesise nanostructured materials (Fig. 2). For instance, Yamauchi and Fujita et al. demonstrated the assembly of MA-BTC HOF nanowires from melamine (MA) and benzene-1,3,5-tricarboxylic acid (BTC). Crucially, these HOF nanowires acted as both a sacrificial template and a precursor. A subsequent solvothermal treatment with various metal salts directly transformed them into MOF nanotubes, faithfully replicating and retaining the initial tubular structure.34
2.3. Post conversion
Post-conversion involves transforming bulk materials into nanomaterials or integrating them with other substrates to form nanocomposites through subsequent physical or chemical processes, such as sonication, ball milling, electrophoretic deposition, solution casting, and post-synthesis (Fig. 2).Sonication is a cost-effective and widely applicable post-conversion strategy for fabricating nano-HOFs or nanocomposites, particularly for exfoliation of bulk and layered HOFs into 2D H-bonded organic nanosheets (HONs) (Fig. 2). This method leverages mechanical energy to overcome the interlayer forces, yielding free-standing NSs or NRs that often retain the crystallinity and functionality of the parent material. White and Foster et al. exemplify the power of this approach by developing a top-down liquid exfoliation strategy for two-layered HOFs with a mild ultrasonic treatment (80 kHz, 12 h) in optimized solvents. This process produced highly stable, free-standing monolayer nanosheets (HON-1 and HON-2) with a uniform thickness of ∼0.8 nm and lateral dimensions of several micrometres. The resulting HONs exhibited exceptional colloidal and structural stability, maintaining their crystalline monolayer structure even after rigorous treatment in hot water.35
Ball milling is an efficient, economical, and safe mechanical approach for exfoliating HOFs into nanosheets or downsizing the bulk material into nanoparticles, serving as an alternative to conventional sonication (Fig. 2). For example, Xiao and Luo et al. prepared a layered 2D HOF (HOFs-8) from N1, N3, N5-tris(pyridine-4-yl)benzene-1,3,5-tricarboxamide (TPBTC). The bulk crystalline material was successfully delaminated into ultrathin nanosheets (2–5 nm) through ball milling, thereby enhancing ion accessibility.36 Beyond its use in post-conversion, mechanochemical synthesis also serves as a strategy for in situ assembly. Liu and Cao et al. demonstrated the versatility of mechanochemical synthesis by preparing eight structurally diverse nano-HOFs with predicted architectures, some of which are difficult to access via conventional solution-based methods. Notably, this approach enabled gram-scale production without loss of crystallinity, underscoring its potential to address a key scalability challenge in HOF fabrication. The strategy also facilitated the one-pot construction of functional composites; for instance, Pd@HOFs exhibited excellent catalytic activity toward CO oxidation.37
Solution casting is a technique that leverages the solution processability of HOFs to fabricate continuous crystalline thin films or nanofilm composites. Liu et al. demonstrated this method by dispersing bulk HOF-BTB crystals in N,N-dimethylformamide (DMF) to form a stable colloidal solution.38 Cryo-electron microscopy (Cryo-EM) coupled with three-dimensional electron diffraction (3D-ED) confirmed that the crystalline framework structure was preserved, with fragmented particles ranging from 20 to 200 nm. To fabricate the membrane, an anodic aluminum oxide (AAO) disk was immersed in the HOF-BTB casting solution at room temperature, followed by heating at 100 °C. Repeating this cycle yielded a continuous crystalline film with a thickness of approximately 126 nm. In contrast, membranes prepared via in situ assembly—by dissolving the BTB ligand directly in DMF—resulted in discontinuous and amorphous films. Moreover, this solution-based approach exhibits excellent substrate adaptability and generalizability to other HOF systems. Notably, Feng et al. first reported a microporous HOF membrane (UPC-HOF-6) grown on an Al2O3 substrate via in situ solution casting. This method promoted homogeneous nucleation and oriented crystal growth, achieving a polycrystalline membrane while its thickness is about 1 µm.39
Electrophoretic deposition (EPD) is a colloidal processing technique in which charged particles suspended in a liquid migrate under an electric field and deposit onto a conductive substrate to form uniform films or coatings. Liu and Cao et al. utilised the inherent negative surface charge of nano-PFC-1 crystals to achieve rapid EPD onto fluorine-doped tin oxide (FTO), forming dense, transparent electrochromic films (∼500 nm) in just two minutes (Fig. 4C).40 This approach was further used by Liu et al. for the fabrication of a heterostructured film by co-depositing PFC-45 and commercial Cu2O particles onto carbon paper, illustrating the adaptability of this technique for designing multicomponent HOF–oxide electrodes.41
Post-synthesis stands as a highly effective chemical strategy for diversifying the functionality of nano-HOFs (Fig. 2), although it typically requires robust frameworks where strong H-bonding interactions preserve the crystalline integrity during chemical processing.42 Zeng and Jiang et al. provided a compelling example of this strategy by constructing a robust biological HOF (HOF-25) from guanine-quadruplex building blocks, stabilized by Hoogsteen H-bonding within G-quartets and interlayer π–π stacking.43 Benefiting from its exceptional stability across a wide pH range (7–11) and in common organic solvents, HOF-25 was further functionalized with Ni2+ ions. This post-synthetic metalation introduced positive charges into the neutral supramolecular layers without compromising the hydrogen-bonded network or crystallinity. The electrostatic modification weakened interlayer π–π interactions, facilitating subsequent exfoliation via sonication. The resulting HOF-25-Ni ultrathin nanosheets, with a uniform thickness of ∼4.4 nm, were obtained in a yield of 56%—higher than that of the parent HOF-25 (45%). Post-chemical grafting can also be used to form HOF@polymers composites with core–shell structure. Du and Liu et al. reported an ‘armour-plating’ strategy where vinyl-terminated ionic liquids were grafted onto PFC-1 nanocrystals, followed by cross-linking polymerisation.33 This process yielded polymer-grafted HOFs (PG-HOFs) with a well-defined core–shell architecture, featuring a HOF core protected by a dense polymer shell approximately 5.12 nm thick, significantly enhancing stability.
Beyond the typical methods introduced above, Wu and Jiang et al. demonstrated that annealing treatment is crucial for improving the homogeneity and stability of HOF membranes. They found that this process reconstructs the membrane's topology from a loose lamellar structure into a more compact one by removing water molecules from adjacent nanosheets. This structural transformation results in a thinner membrane (from 5.9 to 5.3 µm), leading to enhanced overall performance.44
3. Classification and fabrication of HOF nanomaterials
As molecular crystals, HOFs offer excellent solution processability, enabling their facile fabrication into nanostructures such as NSs and NRs. Their exceptional compatibility further allows integration into diverse composite materials. Accordingly, the HOFs discussed in this review can be categorized into two main classes: nanoscale HOFs and HOF-based nanocomposites. Based on their composition, the latter can be further divided into organic nanocomposites, organic–inorganic (hybrid) nanocomposites, and bio-nanocomposites (Fig. 1).3.1. Nanoscale HOFs (nano-HOFs)
By modulating synthetic parameters and fabrication techniques, researchers have accessed a diverse library of nano-HOF architectures, ranging from zero-dimensional nanospheres to one-dimensional (1D) nanoribbons and two-dimensional (2D) nanosheets and nanofilms, with each morphology offering distinct advantages for specific applications.3.2. HOF organic nanocomposites
Beyond pristine nano-HOFs, bulk HOFs serve as versatile platforms for fabricating composites with organic materials like polymers, graphene oxide, HOFs and COFs.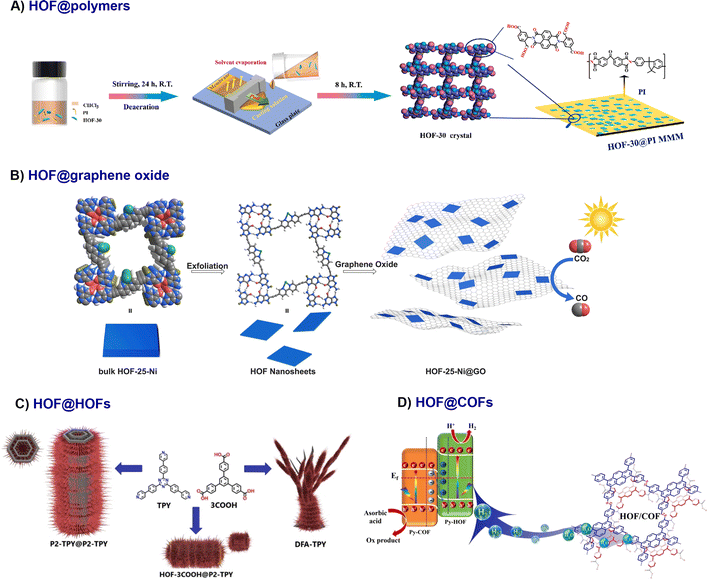 | ||
| Fig. 3 Schematic diagram of the fabrication of HOF organic composites. A) HOF-30@PI MMMs48 adapted with permission from ref. 48, Copyright 2021 Elsevier Inc; (B) HOF-25-Ni@GO,43 adapted with permission from ref. 43, Copyright 2022 Wiley-VCH; (C) Hierarchical HOF-on-HOF superstructures constructed through controlled sequential crystallisation, adapted with permission from ref. 51, Copyright 2024 Wiley-VCH. (D) All-organic HOF/COF S-scheme heterojunction enabling efficient interfacial charge transfer,52 adapted with permission from ref. 52, Copyright 2024 Wiley-VCH. | ||
In addition to post-mixing, HOFs can be integrated during polymer formation. Liu et al. employed interfacial polymerisation to fabricate a thin film nanocomposite (TFN) membrane: an aqueous suspension of Nano-PFC-1 and piperazine (PIP) was coated onto a polyether sulfone (PES) substrate and reacted with 1,3,5-benzenetricarbonyl trichloride (TMC) to form a polyamide (PA) active layer embedded with uniformly dispersed HOF NPs.49 In addition to nanofilms, nanofibers48 and core–shell structures33 were also observed as discussed in the fabrication section.
3.3. HOF organic–inorganic nanocomposites
The integration of metal species with HOFs enables the construction of functional organic–inorganic hybrid nanomaterials. These strategies not only modulate the morphology and porosity of HOF materials but also introduce highly active metal sites for electrocatalysis and energy storage.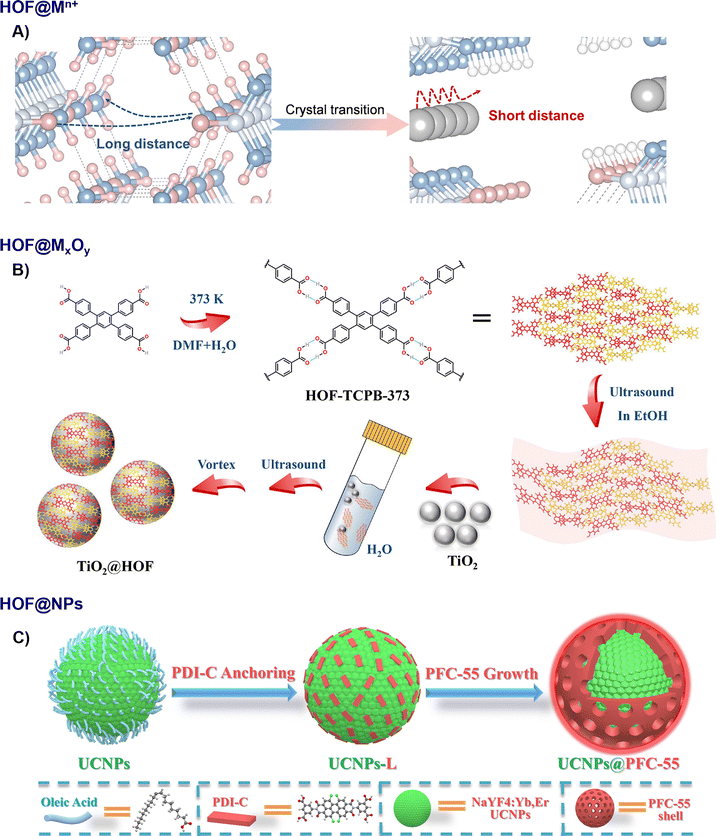 | ||
| Fig. 4 Schematic diagram of the fabrication of HOF organic–inorganic composites. (A) CAM-Ag, adapted with permission from ref. 54, Copyright 2025 Wiley-VCH; (B) HOF-25-Ni@GO45, adapted with permission from ref. 55, Copyright 2023 Elsevier Inc; (C) UCNP@PFC-55,57 adapted with permission from ref. 57, Copyright 2021 Wiley-VCH. | ||
For NPs confined within the HOF pores, Li and Farha et al. developed an in situ photo-deposition method to fabricate AgNPs@HOFs, where UV irradiation of a mixture containing HOF building blocks and AgNO3 led to the formation of ultra-small silver NPs (∼1 nm) uniformly distributed within the HOF pores. In contrast, conventional deposition onto pre-formed HOFs typically yielded larger AgNPs (10–20 nm) on the external surface, highlighting the potential of the in situ approach for achieving fine dispersion.62 For the last type configuration, Liu and Cao et al. reported a one-pot photochemical strategy that simultaneously reduced Ag+ ions to form Ag NPs (∼5 nm) on a HOF surface while oxidising the HOF framework to its radical form (Ag@oxHOFs), demonstrating the potential for concurrent nanoparticle formation and framework modification.63
3.4. HOF biocomposites
The inherent metal-free skeleton and excellent biocompatibility of HOFs position them as components for constructing functional biocomposites, particularly with enzymes and other biomolecules. Through co-crystallisation and biomaterial integration, HOF-based biocomposites offer new opportunities in biocatalysis, biosensing, biomedicine, and sustainable material design.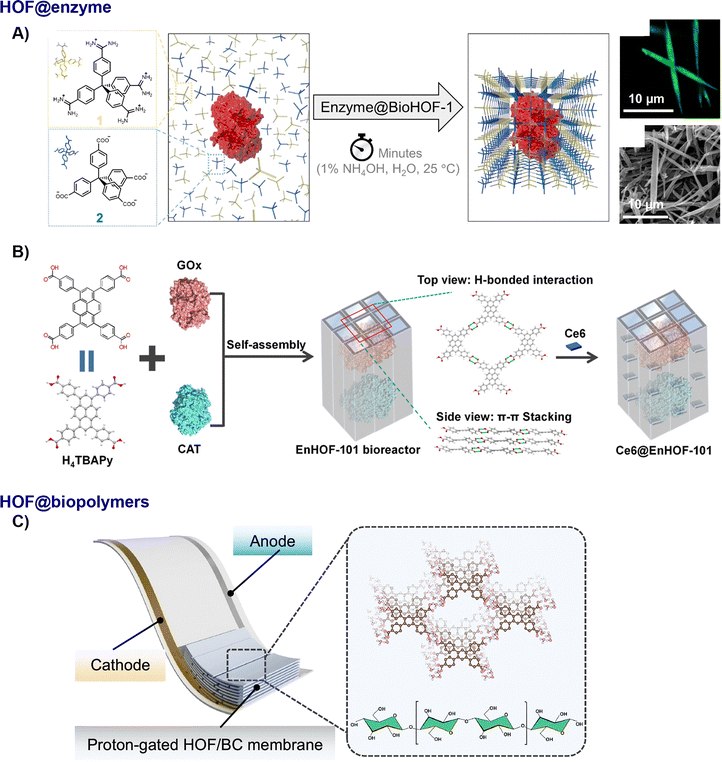 | ||
| Fig. 5 Schematic diagram of the fabrication of HOF biocomposites. (A) BioHOF-1,15 adapted with permission from ref. 15, Copyright 2019 ACS; (B) photobiocatalytic cascade nanoreactor Ce6@EnHOF-101,65 adapted with permission from ref. 65, Copyright 2022 ACS; (C) proton-gated HOF/BC membrane,74 adapted with permission from ref. 74, Copyright 2025 Springer Nature. | ||
4. Applications
4.1. Separation
HOF nanomaterials have demonstrated significant potential in separation applications, including gaseous, aqueous, and radionuclide capture processes. Their tuneable pore architectures, framework flexibility, and rich surface chemistry enable precise molecular recognition and selective transport.HOF-based membranes exhibit exceptional gas separation performance, often attributed to their unique structural responsiveness. Huang et al. demonstrated this by incorporating flexible HOF-30 into a PI matrix to fabricate a mixed-matrix membrane (MMM) (Fig. 6A–C).48 This integration yielded a HOF-30@PI MMM with a 10 wt% loading, which exhibited a high H2 permeability of 428.1 barer and an H2/CH4 separation factor of 61.7. This value is approximately 7.6 times higher than that of the pure PI membrane. Furthermore, the HOF-30@PI MMM displayed pressure-regulated H2/CH4 separation performance, a behaviour arising from the local dynamics of the flexible HOF structure. The potential of nano-HOF membranes was further demonstrated by the HOF-BTB@AAO system, which achieved high propylene/propane separation performance (C3H6 permeance ∼200 GPU, separation factor 14) via a molecular sieving mechanism.38 Beyond membrane separations, HOF nanocrystals also excel as adsorbents for gas purification. Janiak et al. reported a nanocrystalline CB6–H material with exceptional SO2 uptake capacity (6.18 mmol g−1 at 273 K) and selectivity over CO2 (IAST ≈ 120–142), ranking among the highest-performing organic porous materials for fluent gas desulfurization (Fig. 6D and E).31
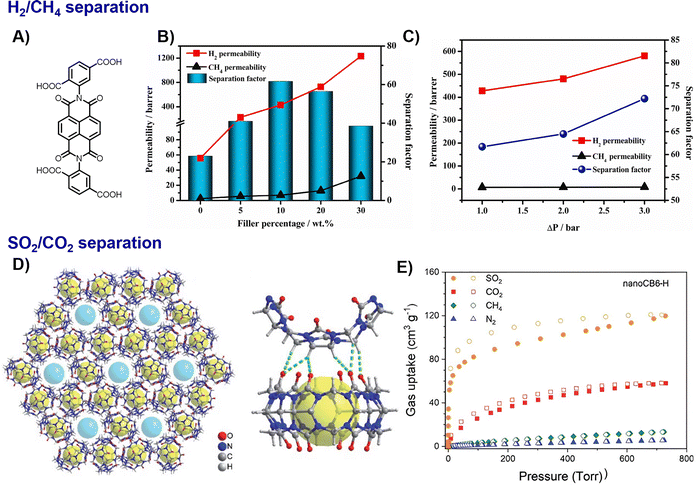 | ||
| Fig. 6 Properties of HOF-30 MMMs:48 (A) molecular building block, (B) permeability and separation factor of the HOF-30 MMMs with different HOF-30 loadings at 1 bar and 25 °C. (C) The H2/CH4 separation performance of HOF-30@PI MMM with 10 wt% HOF-30 loading at different pressure difference (ΔP), adapted with permission from ref.48, Copyright 2021 Elsevier B.V.; CB6-H used for SO2/CO2 separation:31 (D) view of the honeycomb-like structure of CB6-H along the c-axis and the intermolecular hydrogen bonds between neighbouring CB6 molecules, (E) single gas sorption isotherms measured for recycled nanoCB6-H at 293 K, adapted with permission from ref. 31, Copyright 2019 RSC. | ||
In liquid phase separations, HOF-based membranes show promise for water purification and resource recovery. The first demonstration of HOFs in liquid separation came from a thin film nanocomposite membrane (HOF-TFN-2) incorporating Nano-PFC-1 NPs within a polyamide layer. This membrane achieved high permeability and excellent dye rejection (>97% for rhodamine B, >95% for Congo red and Coomassie brilliant blue, >92% for calcein, and ∼84% for methyl blue) through precise molecular sieving.49 The inherent selectivity of HOFs has also been leveraged for strategic applications beyond simple separation. For instance, JZS-1, a porphyrin-based HOFs, exhibited preferential adsorption for cationic dyes in mixtures containing cationic methylene blue (MB+) along with either neutral or anionic dyes, despite not being designed for molecular separation. This adsorption property was leveraged to construct a host–guest system (MB@JZS-1), in which Förster resonance energy transfer (FRET) from the framework to the encapsulated MB significantly enhanced singlet oxygen generation. As a result, the material achieved good antimicrobial performance, with 99.9% inhibition of Staphylococcus aureus under red light irradiation.76
In addition to conventional molecular separation, HOF nanomaterials demonstrate exceptional capability in capturing valuable or hazardous elements. CSMRIHOF-1 film achieved a notable uranium extraction capacity of 17.9 mg g−1 from natural seawater over 30 days.47 Liu and Du et al. fabricated PG-HOFs that set new benchmarks for radioactive anion capture, with capacities of 699.0 mg g−1 for I− and 1.616 g g−1 for ReO4− (a TcO4− surrogate). PG-HOFs exhibited fast kinetics (>98% removal in 45 seconds), excellent selectivity, wide pH tolerance (3–13), and exceptional reusability over 30 cycles.77
4.2. Catalysis
HOFs have emerged as a versatile platform for designing advanced catalytic materials, and their based nanomaterials have demonstrated notable performance across diverse reaction systems, such as oxygen evolution reaction (OER), hydrogen evolution reaction (HER), CO2 conversion, environmental catalysis, and biocatalysis, etc.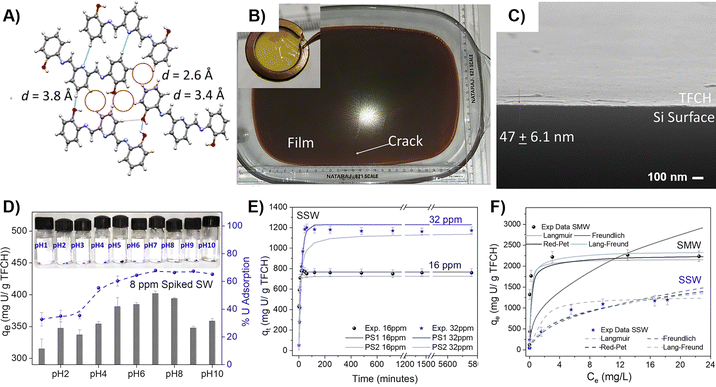 | ||
| Fig. 7 Large-area free-standing thin film of CSMCRIHOF-1, the TFCH, its characterization and adsorption experiments47 (A) crystal structure of CSMCRIHOF-1, (B) scalable preparation of TFCH, (C) cross-section SEM image of TFCH on Si wafer, (D) uranium uptake under different pH conditions, (E) adsorption kinetics of TFCH in 16 and 32 ppm spiked seawater, (F) adsorption isotherms with varied initial concentrations of uranium, adapted with permission from ref. 47, Copyright 2022 Elsevier Inc. | ||
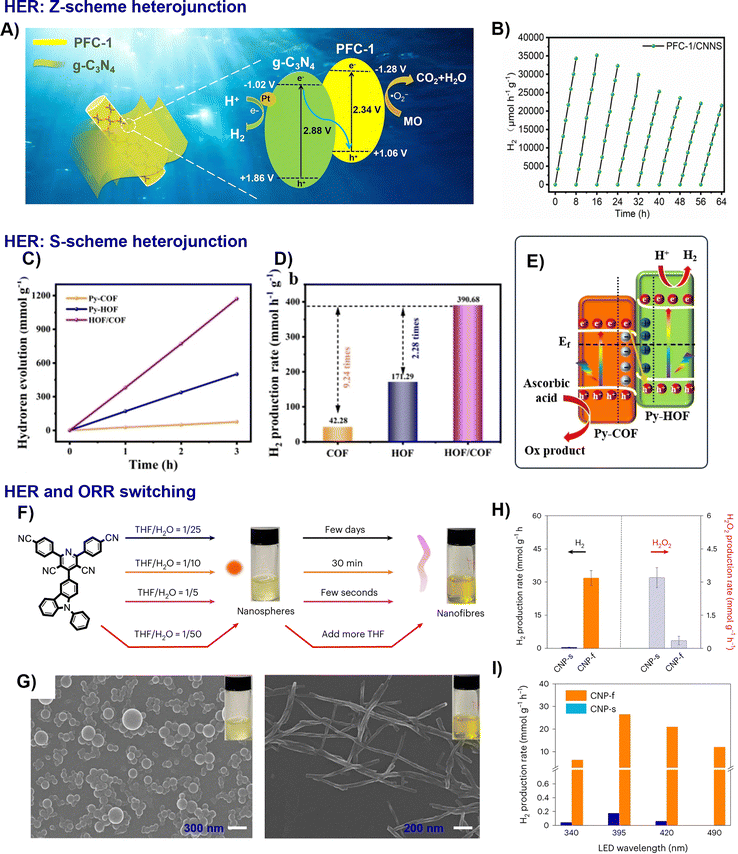 | ||
| Fig. 8 Representative examples of nano-HOFs and its composites used in HER: PFC-1/CNNS heterojunction system,53 (A) proposed carrier separation and photocatalytic processes under visible light, (B) HER cycling stability of PFC-1/CNNS, adapted with permission from ref. 53, Copyright 2022 Elsevier Inc; HOF/COF:52 (C) time profiles of photocatalytic hydrogen generation, (D) average hydrogen evolution rate, (E) photocatalytic reaction mechanism of S-scheme heterojunction, adapted with permission from ref. 52, Copyright 2024 Wiley-VCH. CNP,45 (F) summary of the morphology transformation, (G) SEM images of CNP-s and CNP-f, (H) photocatalytic H2 and H2O2 production, (I) photocatalytic H2 production, adapted with permission from ref. 45, Copyright 2023 Springer Nature. | ||
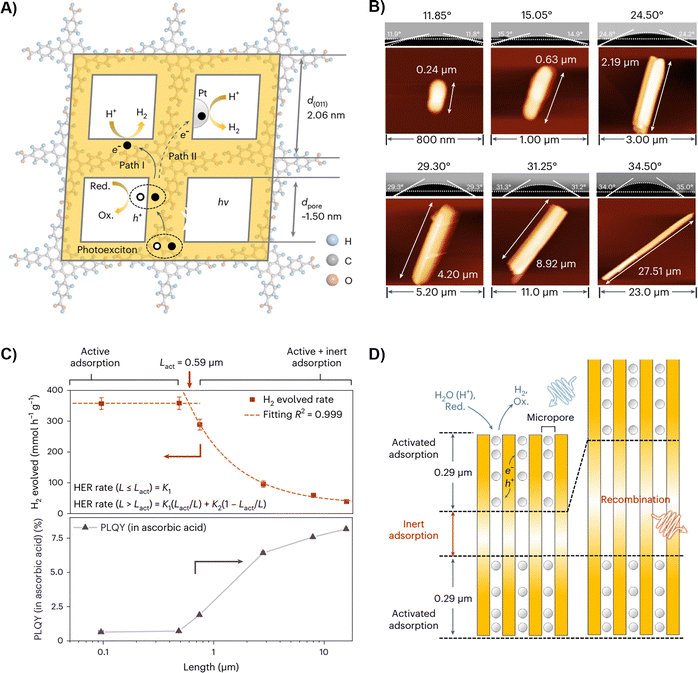 | ||
| Fig. 9 Micropore-confined exciton transfer in HOFs.80 (A) Schematic illustration of the possible migration behavior of excitons in the microporous framework. (Red.: reducing agent and Ox.: oxidation product.), (B) atomic force microscopy images of HOF-H4TBAPy nanorods of different lengths and their corresponding contact angles with water, (C) plot of maximum H2 evolution rate as a function of the mean length of the 1D microporous channel of the samples and the relationship between PLQY in ascorbic acid and different 1D channel lengths, (D) schematic diagram of HOF-H4TBAPy adsorbing water and hole scavenger. The length of the active adsorption region at both ends was determined to be 0.30 µm. Adapted with permission from ref. 80, Copyright 2023 Springer Nature. | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 323 µmol g−1 h−1 with 96.3% selectivity, among the highest for porous organic catalysts. The enhanced performance can be attributed to increased active site accessibility and facilitated electron transfer via the nanoscale engineering, where a HOF was exfoliated into ultrathin nanosheets hosting atomically dispersed Ni sites and supported on graphene oxide.43 A significant advancement toward practical application is the development of photocatalysts that operate without sacrificial agents. The PFC-45/Cu2O@CP film achieved a CO production rate of 11.81 µmol g−1 h−1 in pure water with nearly 100% selectivity. This performance was 2.4 and 3.2 times higher than that of PFC-45@CP and the bulk PFC-45/Cu2O, respectively. These results highlight the dual benefit of the heterojunction film architecture in promoting charge separation and scalability (Fig. 10D–G).41
323 µmol g−1 h−1 with 96.3% selectivity, among the highest for porous organic catalysts. The enhanced performance can be attributed to increased active site accessibility and facilitated electron transfer via the nanoscale engineering, where a HOF was exfoliated into ultrathin nanosheets hosting atomically dispersed Ni sites and supported on graphene oxide.43 A significant advancement toward practical application is the development of photocatalysts that operate without sacrificial agents. The PFC-45/Cu2O@CP film achieved a CO production rate of 11.81 µmol g−1 h−1 in pure water with nearly 100% selectivity. This performance was 2.4 and 3.2 times higher than that of PFC-45@CP and the bulk PFC-45/Cu2O, respectively. These results highlight the dual benefit of the heterojunction film architecture in promoting charge separation and scalability (Fig. 10D–G).41
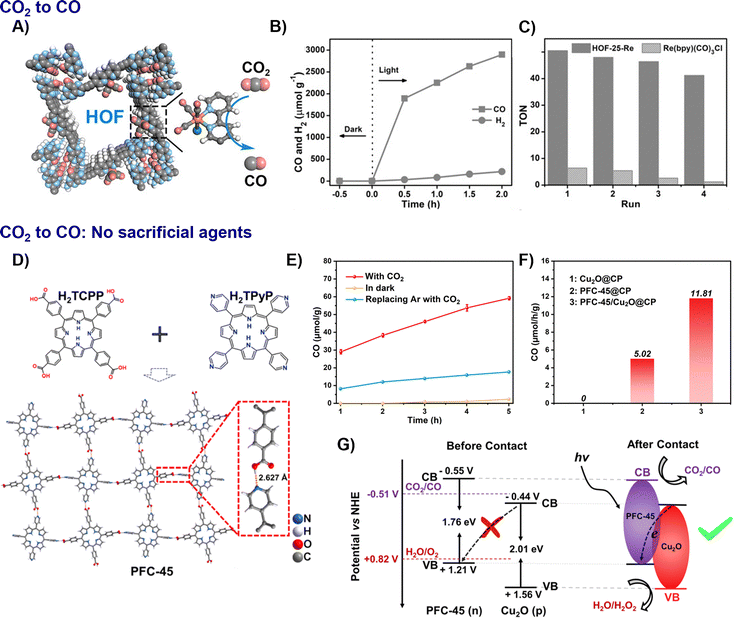 | ||
| Fig. 10 Representative examples of nano-HOFs and its composites used in CO2 conversion. HOF-25-Re:81 (A) proposed structure, (B) time-dependent CO and H2 evolution for the visible-light irradiated photocatalytic system, (C) the TON comparison of the CO2 photoreduction between HOF-25-Re and Re(bpy)(CO)3Cl, adapted with permission from ref. 81, Copyright 2021 Wiley-VCH; PFC-45/Cu2O:41 (D) structure of PFC-45; (E) control experiments of photocatalytic CO2 reduction under visible-light irradiation over PFC-45/Cu2O@CP, (F) CO production rate, (G) schematic diagram of charge transfer between p-type Cu2O and n-type PFC-45, adapted with permission from ref. 41, Copyright 2022 ACS. | ||
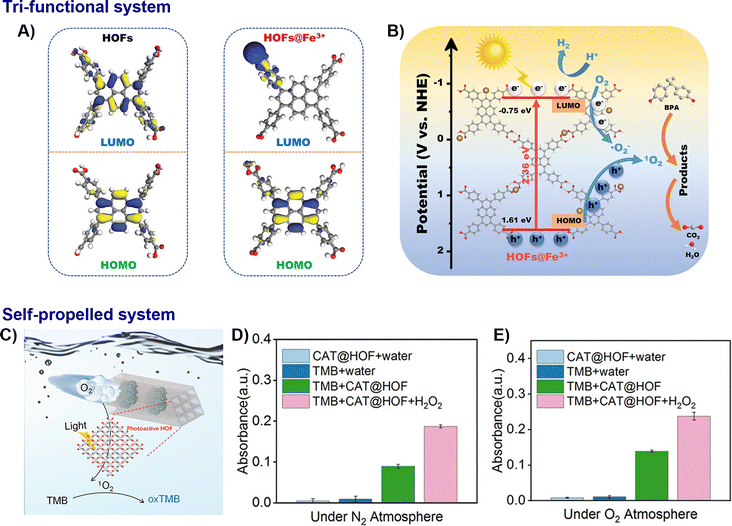 | ||
| Fig. 11 Representative examples of nano-HOFs and its composites used in environmental catalysis. HOF@Fe3+:82 (A) LUMO and HOMO orbitals distribution in HOFs and HOFs@Fe3+ from DFT calculation, (B) photocatalytic mechanism of HOFs@Fe3+, adapted with permission from ref. 82, Copyright 2023 Wiley-VCH; CAT@; HOF:67 (C) schematic presentation of the photocatalytic oxidation of TMB using CAT@HOF, (D) the enhancement of photocatalysis by self-propelling CAT@HOF under N2 atmosphere, (E) under O2 atmosphere, adapted with permission from ref. 67, Copyright 2023 Wiley-VCH. | ||
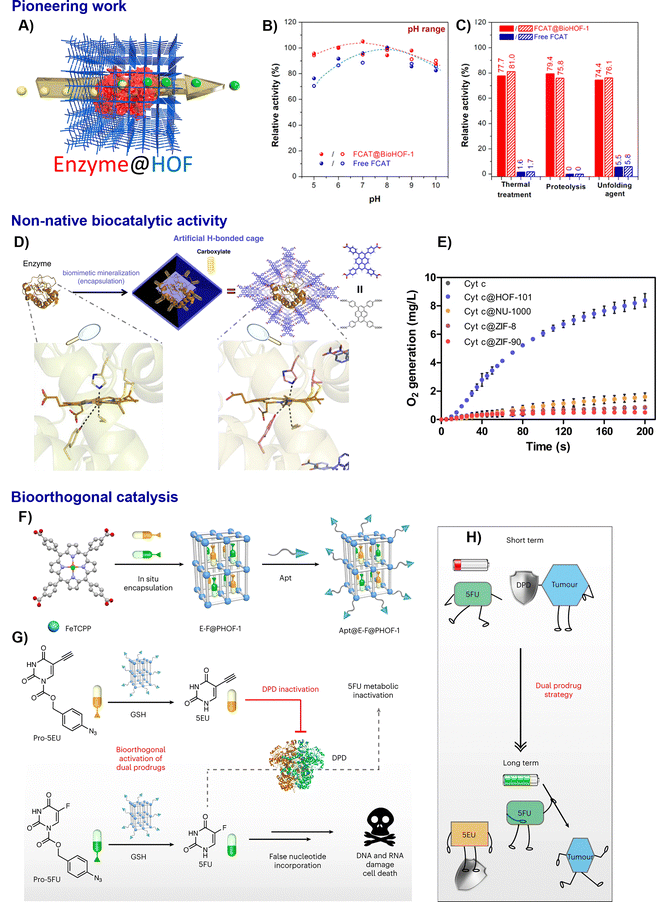 | ||
| Fig. 12 Representative examples of nano-HOFs and its composites used in biocatalysis. BioHOF-1:15 (A) schematic presentation of the enzyme encapsulation in HOFs, (B) relative activity of free FCAT and FCAT@BioHOF-1 at different Ph, (C) relative activity (%) of free FCAT and FCAT@BioHOF-1 after various treatment, adapted with permission from ref. 15, Copyright 2020 ACS; Non-native biocatalytic activity:83 (D) schematic illustration of the designed H-bonded nanocage for modulating the conformation of an enzyme, Cyt c (PDB: 6k9i), (E) CAT-like bioactivities, adapted with permission from ref. 83, Copyright 2022 Springer Nature; HOF-based biorthogonal prodrug activation for chemotherapy:85 (F) illustration of the synthesis of Apt@E-F@PHOF-1, (G) the chemotherapy mechanism of Apt@E-F@PHOF-1, (H) diagram of the dual drug strategy. Apt, aptamer AS1411; F, pro-5FU; E, pro-5EU, adapted with permission from ref. 85, Copyright 2023 Springer Nature. | ||
The excellent biocompatibility of HOFs enables their use for intracellular enzyme delivery. TA-HOFs were shown to deliver functional enzymes into cells with over 90% efficiency via clathrin-independent endocytosis. The encapsulated CAT (CAT@TA-HOFs) effectively reduced reactive oxygen species (ROS) in neuroblastoma cells under oxidative stress, improving cell viability from 35% to 60% and demonstrating the therapeutic potential of HOF-based enzyme delivery systems for neuroprotection.69
A breakthrough was achieved by using mesoporous HOFs simultaneously featuring small particle sizes (<100 nm) and large mesopores (>2 nm) to encapsulate enzymes. This design, exemplified by TaTb, effectively overcomes the mass transfer limitations of earlier carriers. When immobilising lactate dehydrogenase (LDH), the composite LDH@TaTb achieved a catalytic efficiency (Kcat/Km) approaching 99.7% of the free enzyme, a significant enhancement over ZIF-8 and BioHOF-1 benchmarks.68 Chen and Ouyang et al. found that HOF encapsulation can precisely manipulate enzyme function as well. Encapsulating cytochrome c (Cyt c) in HOF-101 induced a conformational switch from a native hexa-coordinated state to a non-native, catalase-like penta-coordinated state (Fig. 12D and E). This Cyt c@HOF-101 composite showed improved H2O2 decomposition activity, a functionality not present in the native enzyme and not achievable with MOF encapsulants, showcasing the unique ability of HOF scaffolds to confer novel catalytic properties.83
Recent advances demonstrate the power of protein-integrated HOFs in enabling sophisticated biorthogonal catalysis for targeted therapy. One strategy employs mitochondria-targeted HOFs (RuB-HOFs) that function as light-triggered photocatalysts. Co-encapsulation of CAT creates a dual-action platform for concurrent H2S release and reactive oxygen species scavenging, offering neuroprotection.84 To address the challenge of metabolic inactivation, a subsequent study designed a ferric porphyrin-based HOF that is intracellularly activated by glutathione. This platform simultaneously releases the anticancer drug 5-fluorouracil (5FU) and its inhibitor, 5-ethynyluracil (5EU), which protects the drug from degradation. This approach of co-releasing a drug and its protector enhanced therapeutic efficacy in the metastatic tumour model tested (Fig. 12F–H).85
4.3. Energy storage
HOFs and its composites have shown the ability to address key challenges across diverse electrochemical platforms, ranging from battery electrodes to ion-conducting membranes, highlighting their significant potential as a unifying materials platform for energy technologies.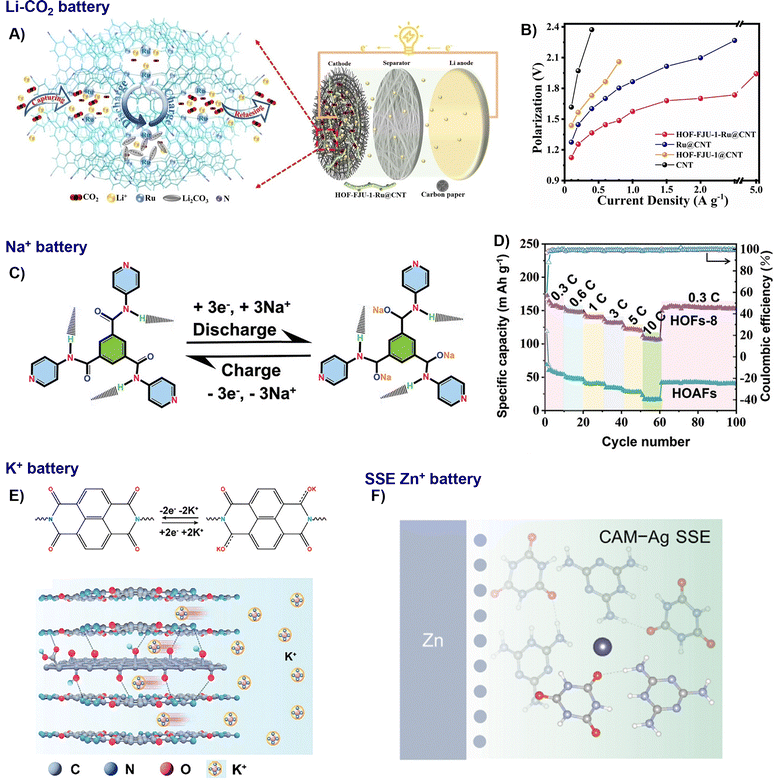 | ||
| Fig. 13 Representative examples of nano-HOFs and its composites used in batteries. Li–CO2 battery:60 (A) schematic diagram of a Li–CO2 battery, (B) battery overpotentials at various current densities, adapted with permission from ref. 60, Copyright 2023 Wiley-VCH; Na+ battery:30 (C) schematic illustration of the sodiation/desodiation process of the HOFs-8 electrode, (D) rate capabilities of HOFs-8 and HOAFs at different current rates from 0.3 to 10 C, adapted with permission from ref. 30, Copyright 2023 Wiley-VCH; K+ battery:86 (E) reaction mechanism of H-NDA-G and schematic representation of potassium storage in H-NDA-G based on interface decoupling, adapted with permission from ref. 86, Copyright 2024 Wiley-VCH; Zn+ battery:54 (F) schematic illustration of interface chemistry of Zn electrodes in CAM-Ag SSE. Adapted with permission from ref. 54, Copyright 2025 Wiley-VCH. | ||
In addition to Li+ batteries, the robust, multi-site H-bonding in frameworks like HOF-DAT was evaluated as a cathode material for Na-batteries. It delivered a high reversible capacity of 144 mAh g−1 at 50 mA g−1, excellent rate capability (109 mAh g−1 at 2 A g−1), and good cycling stability, retaining 73 mAh g−1 after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at 1 A g−1.30 Subsequent studies have built upon this by engineering HOFs with specific redox-active motifs. For instance, HOFs-8 utilizes its carbonyl (C
000 cycles at 1 A g−1.30 Subsequent studies have built upon this by engineering HOFs with specific redox-active motifs. For instance, HOFs-8 utilizes its carbonyl (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) groups as the primary redox-active centres for Na+ storage, while its flexible H-bonding network accommodates volume changes (Fig. 13C and D). This design delivered a high reversible capacity (172.9 mAh g−1) and notable rate performance, while maintaining its capacity 107.9 mAh g−1 even at 10 °C.36
O) groups as the primary redox-active centres for Na+ storage, while its flexible H-bonding network accommodates volume changes (Fig. 13C and D). This design delivered a high reversible capacity (172.9 mAh g−1) and notable rate performance, while maintaining its capacity 107.9 mAh g−1 even at 10 °C.36
Compared to Na+ and Li+, K+ has lower Lewis acidity, which benefits the enhancement of the migration rate and transference number for K+ in liquid electrolytes.86 Meng and Mai et al. fabricated an H-NDA-G cathode by in situ synthesis of H-NDA on GO, forming a 3D “HOF-GO-HOF” sandwich conductive network via hydrogen bonding and π–π interactions. The weak electronic coupling within this network establishes charge transfer channels and an interconnected conductive framework, promoting efficient K+ redox reactions. As a result, the electrode achieved a high capacity of 120 mAh g−1 at 0.1 A g−1.50
Solid-state electrolytes (SSE) are safer and more stable in contrast to the conventional aqueous analogues. Xu and Wang et al. engineered a HOF-based solid-state electrolyte (CAM-Ag) for zinc-ion batteries by introducing Ag–N coordination bonds to induce a crystal transformation (Fig. 13F). This strategy created shortened Zn2+ conduction pathways, yielding a high ionic conductivity of 1.14 × 10−4 S cm−1 and an exceptional Zn2+ transference number of 0.72. The material's abundant H-bonding network effectively suppresses hydrogen evolution and by-product formation, while promoting uniform ion deposition to prevent dendrites.54
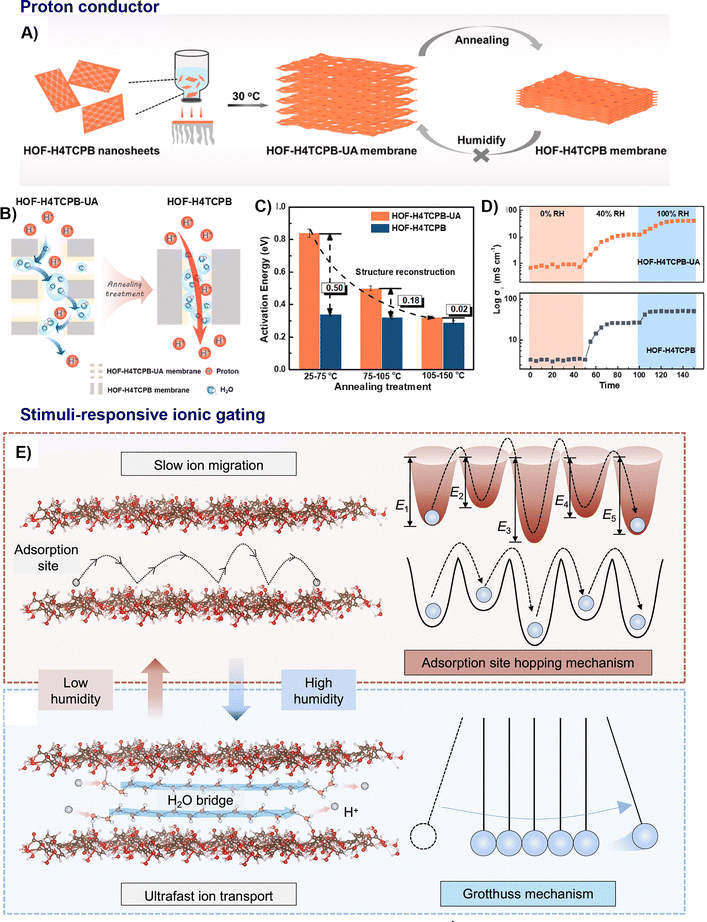 | ||
| Fig. 14 Representative examples of nano-HOFs and its composites used in proton conductor. HOF-H4TCPB membrane:44 (A) schematic preparation of HOF-H4TCPB membrane, (B) proton conduction in HOF-H4TCPB membrane at low humidity, (C) activation energy of HOF-H4TCPB membrane at various temperature range under 0% RH, (D) time-dependent vertical conductivity under different RH at 60 °C for 150 min, adapted with permission from ref. 44, Copyright 2022 Elsevier Inc, (E) the reversible formation and disruption of humidity-induced water bridges within HOFs facilitates the switching of proton transport mode from the adsorption site hopping of the Grotthuss mechanism, adapted with permission from ref. 74, Copyright 2025 Springer Nature. | ||
A stimuli-responsive ionic gating behaviour was observed on HOF-H4TCPB nanosheet with a record proton gating ratio of 5740, allowing its conductivity to switch by orders of magnitude in response to humidity (Fig. 14E). This dramatic change is driven by a shift from a slow, high-energy hopping mechanism at low humidity (2.06 eV barrier) to an ultrafast Grotthuss mechanism at high humidity (0.33 eV barrier), facilitated by the formation of water bridges. This property was further successfully harnessed for real-world applications, including soil moisture monitoring and wireless respiratory rate tracking, showcasing a novel approach for non-contact physical sensing.74
4.4. Sensor
The structural and functional versatility of HOFs allows them to be tailored for a vast sensing landscape: from detecting specific chemical and biological analytes with high precision to monitoring physical environmental changes. To date, HOF-based nanocomposites have demonstrated great potential for use in various sensing platforms, including optical, electrochromic, electrochemical, photoelectrochemical, and biological sensors. Recently, an optical sensor was fabricated, where two kinds of NH3 recognition sites, p-conjugated HOF and luminescent Pt2Cu4 nanocluster, were precisely assembled for ratiometric fluorescence sensing (Fig. 15A). The electron cloud interactions, enhanced by the nanoscopic quantum confinement effect of the metal clusters within the Pt2Cu4@HOF-101 composite, give rise to a distinctly different dual-fluorescence response signal toward NH3. This unique response enables accurate quantification of NH3 concentrations in the breath of liver and kidney disease patients, with a limit of detection (LOD) as low as 0.7 ppm. This combination of high sensitivity and selectivity demonstrates the potential of the system for non-invasive diagnosis through simple breath analysis.59 Analogously, the intrinsic redox activity of pyrene units in the nano-PFC-1 film forms the basis of an electrochromic sensor, exhibiting a reversible behaviour, ideal for visual sensing applications. Moreover, a multi-state electrochromic behaviour enables successive colour changes from yellow to green to blue-violet by the introduction of additional Fe3+/Fe2+ redox couples (Fig. 15B and C).40 White and Foster et al. developed HOF-based nanosheets which, due to their high surface area and exposed active sites, achieved a high Rhodamine B quenching efficiency (93–95%) significantly higher than their bulk counterparts (<5%).35 HOFs also provide an ideal matrix to enhance sensitivity in the realm of electrochemical and photoelectrochemical sensors. The plasmonic effect of AgNPs@HOF boosted the photocurrent, enabling a highly selective photoelectrochemical (PEC) sensor for CEES detection. This sensor achieved a LOD of 15.8 nmol L−1 with excellent discrimination against 13 potential interferents (Fig. 16). It is notable that AgNPs@HOF-101-10 achieved a photocurrent of 1.91 µA, over three times that of pristine HOF-101.62 For chemiresistive sensing, exfoliated 2D L-HOF-199 nanosheets demonstrate high sensitivity towards NO2 (152% to 5 ppm) and a low LOD of 15 ppb at room temperature. In contrast to the bulk crystals, the sensitivity and response speed were increased 3.25 times and more than twice, respectively.87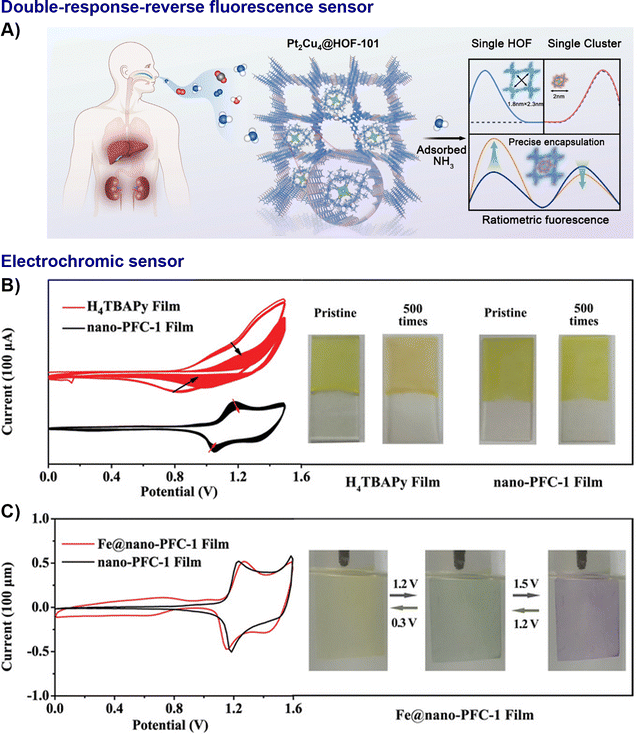 | ||
| Fig. 15 Representative examples of nano-HOFs and its composites used in optical sensor. (A) Schematic illustration of the construction of Pt2Cu4@HOF-101 for double-response-reverse NH3 fluorescence sensing,59 adapted with permission from ref. 59, Copyright 2025 Elsevier Inc; nano-PFC-1 film:40 (B) long-term CV scans (500 cycles) of the amorphous H4TBAPy film andnano-PFC-1 film at a scan rate of 20 mV s−1 and corresponding photographs of films in the pristine state and after 500 CV cycles, (C) CV curves of the Fe@nano-PFC-1 and nano-PFC-1 films at a scan rate of 5 mV s−1 and photographs of the Fe@nano-PFC-1 film during CV cycles, adapted with permission from ref. 40, Copyright 2020 Wiley-VCH. | ||
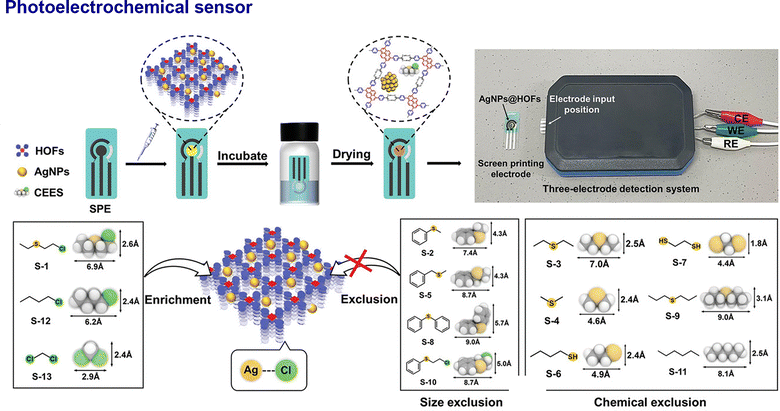 | ||
| Fig. 16 Representative examples of nano-HOFs and its composites used in sensoring. AgNPs@HOF-101-10:64 scheme for the fabrication of the CEES PEC sensor and molecular recognition steps to capture CEES, adapted with permission from ref. 64, Copyright 2022 Wiley-VCH. | ||
Furthermore, HOFs excel in biosensing. The large mesopores of MHOF-101 enable efficient multi-enzyme cascade reactions for detecting clinical biomarkers like glucose and uric acid in point-of-care biosensors.72
4.5. Chemo- and phototherapy
Chemotherapy relies on systemic pharmacological action, phototherapy enables localized light-mediated ablation, and chemo-phototherapy integrates both to achieve synergistic antitumor effects with reduced systemic toxicity. HOFs have recently emerged as promising candidates for photothermal therapy (PTT), photodynamic therapy (PDT) and chemo-photodynamic therapy (CPDT), owing to their tuneable photophysical properties, excellent biocompatibility, and versatile functionalization capabilities. Through rational structural design and composite engineering, researchers have developed a variety of high-performance HOF-based PTT and PDT agents that address key limitations of conventional materials, such as limited absorption range, low energy conversion efficiency, and shallow tissue penetration.To overcome the inherent limitation of most HOFs absorbing primarily in the UV-visible range, core–shell composite strategies have been successfully developed. For instance, the integration of UCNPs with HOFs via luminescence resonance energy transfer (LRET) has enabled deep-tissue NIR activation.57,58,88 A NIR-responsive photothermal and photodynamic synergistic platform, UCNPs@PFC-55, converts 808 nm NIR light to visible emissions, which then activate the HOF shell through LRET with 58.3% efficiency (Fig. 17A–D).57
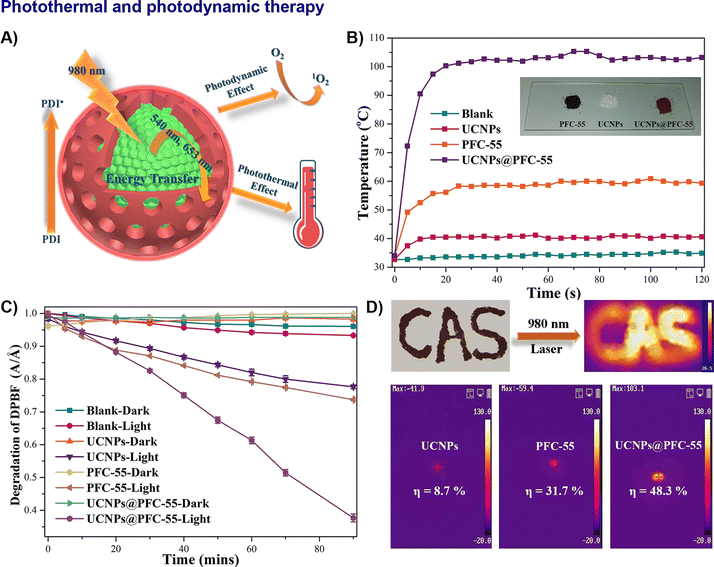 | ||
| Fig. 17 Representative example of nano-HOFs and its composites used in photothermal therapy. UCNPs@PFC-5:57 (A) RET process from UCNPs “core” to PFC-55 “shell” for achieving NIR-response photo-thermal and photodynamic effect, (B) photothermal conversion curves on quartzglass under NIR irradiation (980 nm, 1.5 W cm−2), (C) time-dependent absorbance of DPBF solution at 413 nm, (D) IR camera images of patterned letter made of UCNPs@PFC-55, adapted with permission from ref. 57, Copyright 2021 Wiley-VCH. | ||
HOFs can serve as versatile platforms for combination therapies. Early work by Liu and Cao et al. incorporated Doxo into photoactive Nano-PFC-1, enabling synergistic chemo-photodynamic therapy with reduced systemic toxicity.89 Subsequent studies, such as the exfoliation of 3D HOFs into 1D nanoribbons, further increased drug loading capacity and allowed triple-modal chemo-photodynamic-photothermal therapy under dual-wavelength irradiation.46 Enzyme-loaded systems like Ce6@EnHOF-101 have also been designed to execute synergistic photobiocatalytic cascades, modulating the tumour microenvironment for enhanced therapeutic outcomes.65
4.6. Other applications
Recent advances have underscored the potential of porous frameworks such as MOFs and HOFs in biomedical fields like drug delivery.89 In particular, HOFs offer a unique advantage: the dynamic nature of their hydrogen bonds facilitates stimuli-responsive drug release. For example, Chen and Wang et al. recently introduced HOFs as programmable platforms for focused ultrasound (FUS)-triggered drug activation, enabling precise control over cellular events in deep brain tissue. By systematically modulating hydrogen-bond density and the number of aromatic fused rings within the organic ligands, the authors established a predictive model that correlates molecular architecture with ultrasound activation thresholds. This rational design facilitated 'sono-chemogenetics,' where FUS-triggered scission of supramolecular interactions prompted the instantaneous release of neuromodulators from HOF nanocrystals. This strategy achieves spatiotemporal control of deep neural circuits with second-level latency, offering a versatile, non-invasive paradigm for targeted molecular delivery. (Fig. 18A–C).90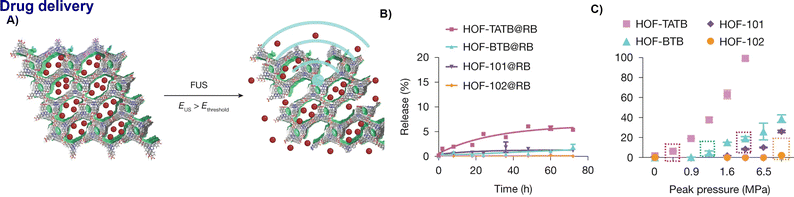 | ||
| Fig. 18 HOFs as ultrasound-programmable delivery platform:90 schematic of ultrasound-triggered drug release from HOF nanocrystals, with drug release occurring when EUS > Ethreshold, (B) The free dye release from HOF nanocrystals without ultrasound stimulation, (C) ultrasound-triggered dye release from HOF nanocrystals after 90 s of stimulus, adapted with permission from ref. 90, Copyright 2025 Springer Nature. | ||
Beyond drug delivery, antimicrobial activity was also explored in HOF composites. For example, HOF-101-F@PVDF-HFP nanofibers demonstrated good singlet oxygen (1O2) generation under green light (520 nm), achieving a 63 mmol g−1 of 1O2 in 20 minutes—nearly twice that of pure HOF-101-F microcrystalline powder (using 0.5 wt% HOF-101-F@PVDF-HFP formulation). These nanofibers exhibited rapid, broad-spectrum antimicrobial activity, inactivating >94% of Escherichia coli, among others, within 5 minutes under ambient light.33
In addition to phototherapy, HOF-based composites have shown potential in other photothermal applications, such as water evaporation. A notable example is Ag@oxPFC-1, which exhibits a photothermal conversion efficiency of 69.8% under 808 nm laser irradiation due to efficient non-radiative decay. This composite also exhibited great promise as an interfacial solar evaporator, which exhibited a water evaporation rate of 1.445 kg m−2 h−1 with 93.6% efficiency (Table 1).63
| Name | Composites | Preparation | Application | Ref. | |
|---|---|---|---|---|---|
| HOFs | Composite | ||||
| Separation | |||||
| HOF-30@PI | HOF-30 | Polyimide polymer | Solution-casting | H2/CH4 separation | J. Membr. Sci., 2022, 643, 120021 |
| UPC-HOF-6 | UPC-HOF-6 | Al2O3 | Solution-casting | H2/N2 separation | Angew. Chem. Int. Ed., 2020, 59, 3840 |
| CB6-1 | CB6-1 | — | Solvothermal | SO2/CO2 | J. Mater. Chem. A, 2020, 8, 19799 |
| HOF-BTB@AAO | HOF-BTB | Al2O3 | Solution-casting | C3H6/C3H8 separation | Nat. Commun., 2024, 15, 634 |
| PG-HOFs | PFC-1 | NH2-IL + bis-C2 | Post-synthesis | Radioactive anions adsorption (I−, IO3–, ReO4−) | Sci. China Chem., 2024, 67, 2958 |
| OH-IL + bis-C2 | |||||
| Olefin + bis-C2 | |||||
| HOF-TFN-2 | PFC-1 | PIP + PES + TMC | Post-synthesis | Dye separation | Nanoscale Adv., 2021, 3, 3441 |
| CSMCRIHOF2-P | CSMCRIHOF2-P | — | Solvothermal | Uranium extraction | Small, 2024, 20, 2306824 |
| TFCHs | CSMCRIHOF-1 | — | Solvothermal | Uranium extraction | Chem, 2022, 8, 2749 |
| DFA-TPY | TPY | DFA | Solvothermal | Oils, microplastics capture | Angew. Chem. Int. Ed., 2024, 63, e202404452 |
| Energy storage | |||||
| CAM-Ag | CAM | AgNO3 | Solvothermal + freeze-drying | Zn2+ battery | Adv. Mater., 2025, 37, 2500721 |
| HOF-FJU-1-Ru@CNT | HOF-FJU-1 | Ru@CNT | Solvothermal | CRR/CER/Li–CO2 battery | Angew. Chem. Int. Ed., 2023, 135, e202311480 |
| HOFs-8 | HOFs-8 | Acetylene black/cellulose sodium | Solvothermal | Na+ battery | Adv. Funct. Mater., 2024, 34, 2314851 |
| H-NDA-G | H-NDA | Graphene oxide | Solvothermal | K+ battery | Adv. Funct. Mater., 2025, 35, 2407452 |
| HOF-DAT | HOF-DAT | — | In situ synthesis and assembly | Na+ battery | Adv. Mater., 2021, 33, 2106079 |
| G2PDI | G2PDI | PVDF + CB | Solution-casting | Li+ battery | Nat. Chem., 2017, 9, 466 |
| HOF-H4TCPB | HOF-H4TCPB | — | Solvothermal | Proton conductor/hydrogen fuel cell | J. Membr. Sci., 2022, 664, 121118 |
| HOF-H4TCPB/BC | HOF-H4TCPB | Bacterial cellulose | Solvothermal | Proton gating/soil moisture monitor | Nat. Commun., 2025, 16, 754 |
| Catalysis | |||||
| TiO2@HOF-TCPB-373 | HOF-TCPB-373 | TiO2 | Milling/sonication | Photo-degradation | Appl. Surf. Sci., 2024, 644, 158770 |
| PFC-1/CNNS | PFC-1 | CNNS | Solvothermal | HER/photo-degradation | J. Photochem. Photobiol., A, 2023, 435, 114292 |
| LDH@TaTb | TaTb | LDH(E) | Solvothermal | Biocatalysis | Chem. Eng. J., 2023, 468, 143609 |
| Cyt c@HBF-1 | PFC-1 | Cyt-c | Solvothermal | Biocatalysis | Chem, 2021, 7, 2722 |
| Cyt c@HBF-2 | HBF-2 | ||||
| HOFs@Mn+ | PFC-1 | Fe3+, Cu2+, Zn2+ | Solvothermal | Photo-degradation/HER | Adv. Funct. Mater., 2023, 33, 2300954 |
| MHOF-101 | PFC-1 | β-Gal-GOx-HRP | Solvothermal | Biosensors/biocatalysis | Angew. Chem. Int. Ed., 2021, 60, 23608 |
| GOx-HRP | |||||
| HOF-25-Re | HOF-25 | Re(CO)5Cl | Solvothermal | CO2 reduction | Angew. Chem. Int. Ed., 2021, 60, 8983 |
| HOF-25-Ni@GO | HOF-25 | GO, Ni(ClO4)2·6H2O | Sonication+ solvothermal | CO2 reduction | Angew. Chem. Int. Ed., 2022, 61, e202211482 |
| CAT@RuB-HOFs | RuB-HOFs | CAT | Solvothermal | Biocatalysis | Angew. Chem. Int. Ed., 2023, 62, e202312784 |
| HOF/COF | PFC-1 | Py-COF | Solvothermal | HER | Angew. Chem. Int. Ed., 2025, 64, e202414229 |
| MTC@Co1−xFexS2 | MTC | Fe(NO3)3, Co(NO3)2, thiourea | Solvothermal | OER | Sustainable Energy Fuels, 2024, 8, 3174 |
| M.CA | MA + CA | — | Solvothermal | HER | ACS Omega, 2022, 7, 22440 |
| M.TC | MA + TC | ||||
| B.TC | BP + TC | ||||
| HOF-19⊃Pd(II) | HOF-19 | Pd2+ | Solvothermal | Organic catalysis | J. Am. Chem. Soc., 2019, 141, 8737 |
| protein@TA-HOFs | TA-HOFs | GFP/HRP/CAT/β-Gal | Solvothermal | Biocatalysis | Angew. Chem. Int. Ed., 2021, 60, 22315 |
| FAOx@BioHOF-1 | BioHOF-1 | FAOx | Solvothermal | Biocatalysis | J. Am. Chem. Soc., 2019, 141, 14298 |
| FCAT@ BioHOF-1 | FCAT | ||||
| HOF-102/cotton | HOF-102 | Cotton | Solution-casting | Photocatalysis | Cell Rep. Phys. Sci., 2020, 1, 100024 |
| PFC-45/Cu2O@CP | PFC-45 | Cu2O@CP | Sonication/electrophoretic deposition | CO2 reduction | ACS Appl. Mater. Interfaces, 2022, 14, 21050 |
| Cyt c@HOF-101 | PFC-1 | Cyt c | Solvothermal | Biocatalysis | Nat. Commun., 2022, 13, 4816 |
| CNP | CNP | Pt | Solvothermal | HER/H2O2 evolution | Nat. Nanotechnol., 2023, 18, 307 |
| H–S/TBAP-α | PFC-1 | [FeFe]-hydrogenases | Solvothermal | HER | ACS Catal., 2024, 14, 18603 |
| CAT@HOF | PFC-1 | CAT | Solvothermal | OER | Small Struct., 2023, 4, 2200346 |
| CNQD | MA + CA | — | Solvothermal | NRR/OER Zn–N2 battery | ACS Nano, 2022, 16, 9049 |
| HOF-H4TBAPy | PFC-1 | Pt | Solvothermal | HER | Nat. Catal., 2023, 6, 574 |
| Sensing | |||||
| HON-1 | HON-1 | — | Sonication | Fluorescence sensor | Chem. Sci., 2021, 12, 3322 |
| HON-2 | HON-2 | ||||
| Pt2Cu4@HOF-101 | PFC-1 | Pt2Cu4 | Solvothermal | Fluorescence sensor | Chem, 2025, 11, 102457 |
| L-HOF-199 | L-HOF-199 | — | Sonication | Chemiresistive NO2 sensor | Inorg. Chem., 2025, 64, 1258 |
| AgNPs@HOFs | PFC-1 | AgNPs | Solvothermal + photo-reduction | Photoelectrochemical sensor | Adv. Mater., 2022, 34, 2202287 |
| HOF-102 | |||||
| nano-PFC-1 | PFC-1 | FTO | Electrophoretic deposition | Electrochromic sensor | Angew. Chem. Int. Ed., 2020, 59, 22392 |
| Chemo-phototherapy | |||||
| CSS@P-P | PFC-55 | CSS + PAA | Solvothermal post-grafting | Cancer therapy | Nano Today, 2023, 48, 101751 |
| UCNPs@PFC-55 | PFC-55 | β-NaYF4:Yb,Er | Solvothermal+ post-synthesis | Bacterial inhibition | Angew. Chem. Int. Ed., 2021, 60, 25701 |
| UCNPs@PFC-73-Ni | PFC-73-Ni | UCNPs | Solvothermal | Antimicrobial therapy | Aggregate, 2024, 5, e481 |
| HOF-101-H@PVDF-HFP | PFC-1 | PVDF-HFP | Electrospinning | Antimicrobial | Adv. Funct. Mater., 2023, 33, 2214388 |
| HOF-101-F@PVDF-HFP | HOF-101-F | ||||
| Doxo@Nano-PFC-1 | PFC-1 | Doxorubicin | Solvothermal | Chemo-photodynamic therapy | Angew. Chem. Int. Ed., 2018, 57, 7691 |
| TCPP-1,3-DPP | TCPP-1,3-DPP | Doxorubicin | Sonication | Drug delivery/chemo and phototherapy | ACS Appl. Nano Mater., 2019, 2, 2437 |
| MB@JZS-1 | JZS-1 | Methylene blue | Solvothermal | Antimicrobial | ACS Appl. Mater. Interfaces, 2022, 14, 49875 |
| Ce6@EnHOF-101 | PFC-1 | GOx + CAT + Ce6 | Solvothermal | Bacterial inhibition/wound therapy | JACS Au, 2022, 2, 2048 |
| Apt@E-F@PHOF-1 | PHOF-1 | Apt + pro-5FU+ pro-5EU | Solvothermal | Cancer therapy | Nat. Catal., 2023, 6, 729 |
| Other applications | |||||
| HOF-TATB | HOF-TATB | Clozapine N-oxide | Solvothermal | Drug delivery/deep-brain neuromodulation | Nature, 2025, 638, 401 |
| Ag@oxPFC-1 | PFC-1 | AgNO3 | Post-synthesis | Solar-driven water evaporator | Sci. China Chem., 2025, 68, 1 |
| HOF-14 | |||||
5. Conclusions and perspective
In sum, this review has systematically outlined the methodologies for transforming bulk HOFs into nanostructured materials and nanocomposites. The transition to the nanoscale proves pivotal in terms of properties and versatility, enabling enhancements in pore accessibility, stability, and electronic conductivity that collectively improve active site accessibility, mass transport efficiency, and charge separation capability. These functional improvements establish HOF nanomaterials as promising platforms for advanced applications, including catalysis, energy storage, separations, and chemo- and phototherapy. Despite these advances, the rational design and practical of HOF nanomaterials remain constrained by several fundamental challenges, which also define key avenues for future research. Structural predictability: one major bottleneck lies in predictive materials design. Unlike reticular frameworks such as MOFs and COFs, where strong coordination or covalent bonds enable relatively reliable structure prediction, the dynamic and reversible nature of hydrogen bonding in HOFs fundamentally complicates the a priori design of stable and functional architectures. Subtle changes in assembly conditions can lead to divergent polymorphic outcomes, undermining rational design efforts. Recent progress in crystal structure prediction and high-throughput computational screening has demonstrated considerable promise in establishing energy–structure–function relationships. However, the accuracy of these predictions remains highly dependent on the quality of force fields and the completeness of conformational sampling, particularly for large, flexible building blocks. Experimental validation therefore remains essential to ground computational predictions in physical reality. Stability vs. processability: a fundamental dilemma: strategies enhancing framework robustness (interpenetration, crosslinking) often reduce solution processability, while high processability typically yields insufficient stability for practical applications. Although some HOFs exhibit high stability under specific conditions, this often comes at the cost of the very processability that distinguishes HOFs from other porous materials. HOFs with stimuli-responsive assembly/disassembly for processing might offer a promising direction to decouple these competing demands. Additionally, how to harness the benefits of HOF flexibility should be explored in the future. Defects and disorders: defects and disorder are intrinsic to the soft and dynamic nature of HOFs, yet their role in functionality remains underexplored and poorly understood. On one hand, disorder may disrupt long-range pore regularity, compromising size-exclusion selectivity and structural integrity. On the other hand, localized defects could introduce additional binding sites, facilitate charge separation, or create reactive centres for catalysis. This dual role presents both a challenge and an opportunity. A systematic characterization and theoretical modelling are urgently needed to correlate defect structures with functional outcomes. Reproducibility and scale-up: the sensitivity of supramolecular assembly to subtle environmental variations (e.g. solvents purity, ambient humidity, temperature), can lead to significant batch-to-batch variability in crystallinity and porosity. This reproducibility issue is critical for practical applications yet frequently overlooked in laboratory studies. Besides, developing continuous manufacturing approaches, such as flow chemistry, is needed for expanding to the large-scale practical use, since conventional batch synthesis methods (e.g., slow solvent evaporation) are inherently unscalable. Conductivity: most HOFs are electrical insulators due to limited π–π conjugation. This intrinsic limitation can be overcome through the rational design of conductive or redox-active building blocks—such as π-conjugated systems, heteroatoms, or metal centres—or through composite formation with conductive supports like carbon nanotubes and graphene oxide. The convergence of enhanced conductivity with solution-based processability positions HOF-based materials as promising candidates for flexible, wearable, and next-generation electronic devices. Nanoscale features vs. crystallinity: pursuing nanoscale morphologies to enhance kinetics often compromises crystallinity, while highly crystalline bulk HOFs suffer from slow diffusion. Therefore, an alternative strategy lies in engineering anisotropic interactions: strong bonding along one direction to maintain long-range order, and weak interactions in orthogonal directions to facilitate nanoscale processing. 1D or 2D HOFs might exemplify this concept, where the directional assembly preserves crystallinity while allowing isolation of nanoscale morphologies.Addressing these interconnected challenges demands a shift from empirical optimization toward integrated design frameworks. First, computationally guided design must evolve to capture not only ideal structures but also the dynamic disorder and condition-dependent behaviours that govern real-world HOF performance. Second, autonomous synthesis and continuous manufacturing should be leveraged not only to accelerate the discovery of functional materials but also to enhance reproducibility and scalability. Third, interfacial engineering, particularly for the formation of stable HOF-based nanocomposites, is essential for expanding HOF applications into emerging fields such as wearable electronics. In this context, transducing dynamic structural changes in response to external stimuli into readable signals represents an attractive direction that leverages the distinguishing flexibility of HOFs. Finally, advanced characterization must bridge the gap between nanoscale features and bulk crystallinity. The convergence of these complementary strategies will be pivotal in realizing HOFs as a versatile and practically relevant class of functional nanomaterials.
Author contributions
This review was conceptualized and coordinated by Dr Antonio Fernandez-Mato. The literature search and data curation were performed by Dr Qiang Zhu. Both authors contributed to the manuscript preparation.Conflicts of interest
The authors declare no conflict of interest.Abbreviations
| 1D | One-dimensional |
| 1O2 | Singlet oxygen |
| 2D | Two-dimensional |
| 3D | Three-dimensional |
| 3D-ED | Three-dimensional electron diffraction |
| 5EU | 5-Ethynyluracil |
| 5FU | 5-Fluorouracil |
| AAO | Anodic aluminum oxide |
| Apt | Aptamer as1411 |
| AQY | Apparent quantum yield |
| BC | Bacterial cellulose |
| BET | Brunauer–emmett–teller |
| bis-C2 | 3,3′-divinyl-1,1′-(1,2-ethanediyl)-diimidazolium dibromide |
| Boc | Tert-butyloxycarbonyl |
| BP | Bipyridine |
| BTC | Benzene-1,3,5-tricarboxylic acid |
| CA | Cyanuric acid |
| CAM | Cyanuric acid–melamine |
| CAT | Catalase |
| CB | Carbon black; |
| Ce6 | Chlorin e6 |
| CEES | 2-Chloroethyl ethyl sulfide |
| CEESO | 2-Chloroethyl ethyl sulfoxide |
| CER | CO2 evolution reaction |
| CNDQs | Carbon nitride-based quantum dots |
| CNNS | Carbon nitride nanosheets |
| CNP | 2,6-Bis(4-cyanophenyl)-4-(9-phenyl-9H-carbazol-3-yl)-pyridine-3,5-dicarbonitrile |
| CNT | Carbon nanotube |
| CP | Carbon paper |
| COFs | Covalent organic frameworks |
| CPDT | Chemo-photodynamic therapy |
| CRR | CO2 reduction reaction |
| Cryo-EM | Cryo-electron microscopy |
| CSP | Crystal structure prediction |
| CSS | NaYF4:Yb3+/Er3+@NaYF4:Nd3+@NaYF4 |
| Cyt c | Cytochrome c |
| DMF | N,N-dimethylformamide |
| Doxo | Doxorubicin |
| EPD | Electrophoretic deposition |
| FAOx | Fluorescein-tagged alcohol oxidase |
| FCAT | Fluorescein-tagged catalase |
| FRET | Förster resonance energy transfer |
| FTO | Fluorine-doped tin oxide |
| GFP | Green fluorescent protein |
| GO | Graphene oxide |
| GOx | Glucose oxidase |
| HBFs | H-bonded biohybrid frameworks |
| HER | Hydrogen evolution reaction |
| HRP | Horseradish peroxidase |
| HOFs | Hydrogen-bonded organic frameworks |
| HONs | 2D H-bonded organic nanosheets |
| ISSA | In situ synthesis and assembly |
| LDH | Lactate dehydrogenase |
| LOD | A limit of detection |
| LRET | Luminescence resonance energy transfer |
| MA | Melamine |
| MB+ | Cationic methylene blue |
| MMM | Mixed-matrix membrane |
| MNCs | Metal nanoclusters |
| MOFs | Metal–organic frameworks |
| MTC | Melamine–trithiocyanurate |
| MxOy | Metal oxides |
| NDI | Naphthalene diimide |
| NH3 | Ammonia |
| NH2-BDC | 2-Aminoterephthalic acid |
| NH2-IL | 1-Aminoethyl-3-vinylimidazolium bromide hydrobromide |
| NIR | Near-infrared |
| NPs | Nanoparticles |
| NRR | Nitrogen reduction reaction |
| NRs | Nanorods |
| NSs | Nanosheets |
| NTCDA | 1,4,5,8-Naphthalenetetracarboxylic dianhydride, |
| NTCDA | 1,4,5,8-Naphthalenetetracarboxylic dianhydride |
| OER | Oxygen evolution reaction |
| OH-IL | 1-(2-Hydroxyl-ethyl)-3-vinyl imidazolium bromide |
| olefin | 4-Penten-1-ol |
| PA | Polyamide |
| PAA | Polyacrylic acid |
| PDA | p-Phenylenediamine |
| PDI | Perylene diimide |
| PDT | Photodynamic therapy |
| PES | Polyethersulfone |
| PI | Polyimide |
| PIP | Piperazine |
| PTT | Photothermal therapy |
| PVDF-HFP | Poly(vinylidene fluoride-co-hexafluoropropylene) |
| PVP | Polyvinylpyrrolidone |
| RET | Resonance energy transfer |
| ROS | Reactive oxygen species |
| SSE | Solid-state electrolytes |
| TC | Trithiocyanuric acid |
| TFA | Trifluoroacetic acid |
| TFCHs | Thin film covalent hybrids |
| TFN | Thin film nanocomposite |
| TMC | 1,3,5-Benzenetricarbonyl trichloride |
| TPBTC | N1, N3, N5-tris(pyridine-4-yl)benzene-1,3,5-tricarboxamide |
| UCNPs | Upconversion nanoparticles |
| Uox-HRP | Urate oxidase xanthine |
Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work was partially supported by the Beatriz Galindo program (grant BG24/00084) from the Spanish Ministry of Science, Innovation and UniversitiesReferences
- K. Geng, T. He, R. Liu, S. Dalapati, K. T. Tan, Z. Li, S. Tao, Y. Gong, Q. Jiang and D. Jiang, Chem. Rev., 2020, 120, 8814–8933 CrossRef CAS PubMed.
- C. S. Diercks and O. M. Yaghi, Science, 2017, 355, eaal1585 CrossRef PubMed.
- H. Furukawa, K. E. Cordova, M. O’Keeffe and O. M. Yaghi, Science, 2013, 341, 1230444 CrossRef PubMed.
- A. M. Wright, M. T. Kapelewski, S. Marx, O. K. Farha and W. Morris, Nat. Mater., 2025, 24, 178–187 CrossRef CAS PubMed.
- D. J. Duchamp and R. E. Marsh, Acta Crystallogr., Sect. B, 1969, 25, 5–19 CrossRef CAS.
- M. Simard, D. Su and J. D. Wuest, J. Am. Chem. Soc., 1991, 113, 4696–4698 CrossRef CAS.
- X. Wang, M. Simard and J. D. Wuest, J. Am. Chem. Soc., 1994, 116, 12119–12120 CrossRef CAS.
- Y. He, S. Xiang and B. Chen, J. Am. Chem. Soc., 2011, 133, 14570–14573 CrossRef CAS PubMed.
- Q. Zhu, J. Johal, D. E. Widdowson, Z. Pang, B. Li, C. M. Kane, V. Kurlin, G. M. Day, M. A. Little and A. I. Cooper, J. Am. Chem. Soc., 2022, 144, 9893–9901 CrossRef CAS PubMed.
- B. Burger, P. M. Maffettone, V. V. Gusev, C. M. Aitchison, Y. Bai, X. Wang, X. Li, B. M. Alston, B. Li, R. Clowes, N. Rankin, B. Harris, R. S. Sprick and A. I. Cooper, Nature, 2020, 583, 237–241 CrossRef CAS PubMed.
- A. Pulido, L. Chen, T. Kaczorowski, D. Holden, M. A. Little, S. Y. Chong, B. J. Slater, D. P. McMahon, B. Bonillo, C. J. Stackhouse, A. Stephenson, C. M. Kane, R. Clowes, T. Hasell, A. I. Cooper and G. M. Day, Nature, 2017, 543, 657–664 CrossRef CAS PubMed.
- M. Mastalerz and I. M. Oppel, Angew. Chem., Int. Ed., 2012, 51, 5252–5255 CrossRef CAS PubMed.
- H. Wang, B. Li, H. Wu, T. L. Hu, Z. Yao, W. Zhou, S. Xiang and B. Chen, J. Am. Chem. Soc., 2015, 137, 9963–9970 CrossRef CAS PubMed.
- Q. Yin, P. Zhao, R. Sa, G. Chen, J. Lü, T. Liu and R. Cao, Angew. Chem., Int. Ed., 2018, 130, 7817–7822 CrossRef.
- W. Liang, F. Carraro, M. B. Solomon, S. G. Bell, H. Amenitsch, C. J. Sumby, N. G. White, P. Falcaro and C. J. Doonan, J. Am. Chem. Soc., 2019, 141, 14298–14305 CrossRef CAS PubMed.
- L. Li, X. Zhang, X. Lian, L. Zhang, Z. Zhang, X. Liu, T. He, B. Li, B. Chen and X. H. Bu, Nat. Chem., 2025, 17, 727–733 CrossRef CAS PubMed.
- H. Liaquat, M. Imran, Z. Saddique, S. Latif, K. M. Al-Ahmary, A. Sohail, H. Raza and M. Ahmed, J. Mol. Struct., 2025, 1320, 140221 CrossRef.
- X. Liu, G. Liu, T. Fu, K. Ding, J. Guo, Z. Wang, W. Xia and H. Shangguan, Adv. Sci., 2024, 11, 2400101 CrossRef CAS PubMed.
- B. Wang, R.-B. Lin, Z. Zhang, S. Xiang and B. Chen, J. Am. Chem. Soc., 2020, 142, 14399–14416 CrossRef CAS PubMed.
- R.-B. Lin, Y. He, P. Li, H. Wang, W. Zhou and B. Chen, Chem. Soc. Rev., 2019, 48, 1362–1389 RSC.
- A. C. Grimsdale and K. Müllen, Angew. Chem., Int. Ed., 2005, 44, 5592–5629 CrossRef CAS PubMed.
- C. Ma, L. Qin, T. Zhou and J. Zhang, Energy Environ. Sci., 2024, 17, 8992–9026 RSC.
- P. Soleimani Abhari, S. Gholizadeh, F. Rouhani, Y. L. Li, A. Morsali and T. F. Liu, Inorg. Chem. Front., 2023, 10, 6134–6159 RSC.
- C. Chen, L. Shen, H. Lin, D. Zhao, B. Li and B. Chen, Chem. Soc. Rev., 2024, 53, 2738–2760 RSC.
- Y. Zhang, M. Tian, Z. Majeed, Y. Xie, K. Zheng, Z. Luo, C. Li and C. Zhao, Separations, 2023, 10, 196 CrossRef CAS.
- D. Yu, H. Zhang, J. Ren and X. Qu, Chem. Soc. Rev., 2023, 52, 7504–7523 RSC.
- C. Halliwell, J. F. Soria and A. Fernandez, Angew. Chem., Int. Ed., 2023, 62, e202217729 CrossRef CAS PubMed.
- Y. Guo, C. Wang, G. Mo, Y. Wang, X. Song and P. Li, Cryst. Growth Des., 2023, 23, 7635–7646 CrossRef CAS.
- Y. L. Wu, N. E. Horwitz, K. S. Chen, D. A. Gomez-Gualdron, N. S. Luu, L. Ma, T. C. Wang, M. C. Hersam, J. T. Hupp, O. K. Farha, R. Q. Snurr and M. R. Wasielewski, Nat. Chem., 2017, 9, 466–472 CrossRef CAS PubMed.
- Y. Wu, X. Mao, M. Zhang, X. Zhao, R. Xue, S. Di, W. Huang, L. Wang, Y. Li and Y. Li, Adv. Mater., 2021, 33, 2106079 CrossRef CAS PubMed.
- J. Liang, S. Xing, P. Brandt, A. Nuhnen, C. Schlüsener, Y. Sun and C. Janiak, J. Mater. Chem. A, 2020, 8, 19799–19804 RSC.
- F. Wang, Q. Wang, S. Wang, K. Zhang, S. Jia, J. Chen and X. Wang, ACS Nano, 2022, 16, 9049–9061 CrossRef CAS PubMed.
- Y. Wang, R. Cao, C. Wang, X. Song, R. Wang, J. Liu, M. Zhang, J. Huang, T. You, Y. Zhang, D. Yan, W. Han, L. Yan, J. Xiao and P. Li, Adv. Funct. Mater., 2023, 33, 2214388 CrossRef CAS.
- Z. X. Cai, Y. Xia, Y. Ito, M. Ohtani, H. Sakamoto, A. Ito, Y. Bai, Z. L. Wang, Y. Yamauchi and T. Fujita, ACS Nano, 2022, 16, 20851–20864 CrossRef CAS PubMed.
- J. Nicks, S. A. Boer, N. G. White and J. A. Foster, Chem. Sci., 2021, 12, 3322–3327 RSC.
- C. Guo, Y. Gao, S. Li, Y. Wang, X. Yang, C. Zhi, H. Zhang, Y. Zhu, S. Chen, S. Chou, S. Dou, Y. Xiao and X. Luo, Adv. Funct. Mater., 2024, 34, 2314851 CrossRef CAS.
- W. Qin, D. Si, Q. Yin, X. Gao, Q. Huang, Y. Feng, L. Xie, S. Zhang, X. Huang, T. Liu and R. Cao, Angew. Chem., Int. Ed., 2022, 61, e202202089 CrossRef CAS PubMed.
- Q. Yin, K. Pang, Y.-N. Feng, L. Han, A. Morsali, X.-Y. Li and T.-F. Liu, Nat. Commun., 2024, 15, 634 CrossRef CAS PubMed.
- S. Feng, Y. Shang, Z. Wang, Z. Kang, R. Wang, J. Jiang, L. Fan, W. Fan, Z. Liu, G. Kong, Y. Feng, S. Hu, H. Guo and D. Sun, Angew. Chem., Int. Ed., 2020, 132, 3868–3873 CrossRef.
- J. Feng, T. Liu and R. Cao, Angew. Chem., Int. Ed., 2020, 59, 22392–22396 CrossRef CAS PubMed.
- A. A. Zhang, Y. L. Li, Z. Bin Fang, L. Xie, R. Cao, Y. Liu and T. F. Liu, ACS Appl. Mater. Interfaces, 2022, 14, 21050–21058 CrossRef CAS PubMed.
- B. Han, H. Wang, C. Wang, H. Wu, W. Zhou, B. Chen and J. Jiang, J. Am. Chem. Soc., 2019, 141, 8737–8740 CrossRef CAS PubMed.
- B. Yu, T. Meng, X. Ding, X. Liu, H. Wang, B. Chen, T. Zheng, W. Li, Q. Zeng and J. Jiang, Angew. Chem., Int. Ed., 2022, 61, e202211482 CrossRef CAS PubMed.
- Z. Yang, Y. Zhang, W. Wu, Z. Zhou, H. Gao, J. Wang and Z. Jiang, J. Membr. Sci., 2022, 664, 121118 CrossRef CAS.
- H. Yang, C. Li, T. Liu, T. Fellowes, S. Y. Chong, L. Catalano, M. Bahri, W. Zhang, Y. Xu, L. Liu, W. Zhao, A. M. Gardner, R. Clowes, N. D. Browning, X. Li, A. J. Cowan and A. I. Cooper, Nat. Nanotechnol., 2023, 18, 307–315 CrossRef CAS PubMed.
- X. T. He, Y. H. Luo, D. L. Hong, F. H. Chen, Z. Y. Zheng, C. Wang, J. Y. Wang, C. Chen and B. W. Sun, ACS Appl. Nano Mater., 2019, 2, 2437–2445 CrossRef CAS.
- A. Kaushik, K. Marvaniya, Y. Kulkarni, D. Bhatt, J. Bhatt, M. Mane, E. Suresh, S. Tothadi, K. Patel and S. Kushwaha, Chem, 2022, 8, 2749–2765 CAS.
- W. Li, Y. Li, J. Caro and A. Huang, J. Membr. Sci., 2022, 643, 120021 CrossRef CAS.
- X. T. Jiang, Q. Yin, B. T. Liu, J. Y. Chen, R. Wang and T. F. Liu, Nanoscale Adv., 2021, 3, 3441–3446 RSC.
- K. Han, C. Tan, S. Feng, Q. Zhang, X. Qiao, Z. Zhong, X. Wang, J. Meng and L. Mai, Adv. Funct. Mater., 2025, 35, 2407452 CrossRef CAS.
- C. A. Halliwell, K. Jolley, K. Yendall, M. R. J. Elsegood, G. N. Parkinson and A. Fernandez, Angew. Chem., Int. Ed., 2024, 63, e202404452 CrossRef CAS PubMed.
- R. Gao, R. Shen, C. Huang, K. Huang, G. Liang, P. Zhang and X. Li, Angew. Chem., Int. Ed., 2025, 64, e202414229 CrossRef CAS PubMed.
- H. Shi, D. Feng, H. Li, D. Yu and X. Chen, J. Photochem. Photobiol., A, 2023, 435, 114292 CrossRef CAS.
- J. Li, H. Wang, D. Guan, X. Wang, C. Miao and J. Xu, Adv. Mater., 2025, 37, 2500721 CrossRef CAS PubMed.
- X. Zhang, W. Liu, F. Han, L. Jiang and Z. Li, Appl. Surf. Sci., 2024, 644, 158770 CrossRef CAS.
- Z. Li, B. Yu, Y. Wang, B. Yan, J. Liu, Y. Liu, R. Wang, P. Rao and Y. Liu, ACS Appl. Mater. Interfaces, 2025, 17, 52981–52992 CrossRef PubMed.
- B. T. Liu, X. H. Pan, D. Y. Zhang, R. Wang, J. Y. Chen, H. R. Fang and T. F. Liu, Angew. Chem., Int. Ed., 2021, 60, 25701–25707 CrossRef CAS PubMed.
- J. Xiao, A. R. M. Shaheer, C. Liu, T. Liu and R. Cao, Aggregate, 2024, 5, e481 CrossRef CAS.
- Y. Wang, J. Yao, S. Wu, C. Zhi, L. Yin, Z. Song, J. Wang, L. Ling, Y. Ma, D. Zhang, J. Li, L. Li and B. Chen, Chem, 2025, 11, 102457 CAS.
- Z. Cheng, Y. Fang, Y. Yang, H. Zhang, Z. Fan, J. Zhang, S. Xiang, B. Chen and Z. Zhang, Angew. Chem., Int. Ed., 2023, 62, e202311480 CrossRef CAS PubMed.
- H. Naz, R. N. Ali, W. A. Qureshi, A. Ali, N. A. Albert and G. Zhu, Sustain. Energy Fuels, 2024, 8, 3174–3181 RSC.
- C. Wang, Y. Wang, K. O. Kirlikovali, K. Ma, Y. Zhou, P. Li and O. K. Farha, Adv. Mater., 2022, 34, 2202287 CrossRef CAS PubMed.
- J.-C. Liu, T. Li, P.-Y. Liao, Z.-Y. Ruan, Z.-H. Wang, J.-H. Jia and M.-L. Tong, Sci. China: Chem., 2025, 68, 4847–4855 CrossRef CAS.
- G. Chen, S. Huang, X. Ma, R. He and G. Ouyang, Nat. Protoc., 2023, 18, 2032–2050 CrossRef CAS PubMed.
- W. Huang, H. Yuan, H. Yang, L. Tong, R. Gao, X. Kou, J. Wang, X. Ma, S. Huang, F. Zhu, G. Chen and G. Ouyang, JACS Au, 2022, 2, 2048–2058 CrossRef CAS PubMed.
- X. Zhao, Q. Yin, X. Mao, C. Cheng, L. Zhang, L. Wang, T.-F. Liu, Y. Li and Y. Li, Nat. Commun., 2022, 13, 2721 CrossRef CAS PubMed.
- H. Yang, J. Fu, W. Huang, T. Wu, S. Huang, G. Chen and G. Ouyang, Small Struct., 2023, 4, 2200346 CrossRef CAS.
- W. Li, J. Shi, Y. Chen, X. Liu, X. Meng, Z. Guo, S. Li, B. Zhang and Z. Jiang, Chem. Eng. J., 2023, 468, 143609 CrossRef CAS.
- J. Tang, J. Liu, Q. Zheng, W. Li, J. Sheng, L. Mao and M. Wang, Angew. Chem., Int. Ed., 2021, 60, 22315–22321 CrossRef CAS PubMed.
- Y. Cai, J. Gao, J. Li, P. Liu, Y. Zheng, W. Zhou, H. Wu, L. Li, R. Lin and B. Chen, Angew. Chem., Int. Ed., 2023, 62, e202308579 CrossRef CAS PubMed.
- G. Chen, S. Huang, Y. Shen, X. Kou, X. Ma, S. Huang, Q. Tong, K. Ma, W. Chen, P. Wang, J. Shen, F. Zhu and G. Ouyang, Chem, 2021, 7, 2722–2742 CAS.
- Z. Tang, X. Li, L. Tong, H. Yang, J. Wu, X. Zhang, T. Song, S. Huang, F. Zhu, G. Chen and G. Ouyang, Angew. Chem., Int. Ed., 2021, 60, 23608–23613 CrossRef CAS PubMed.
- J. Yang, Q. Jiang, Y. Chen, Q. Wen, X. Ge, Q. Zhu, W. Zhao, O. Adegbite, H. Yang, L. Luo, H. Qu, V. Del-Angel-Hernandez, R. Clowes, J. Gao, M. A. Little, A. I. Cooper and L. N. Liu, ACS Catal., 2024, 14, 18603–18614 CrossRef CAS PubMed.
- D. Lei, Y. Wang, Q. Zhang, S. Wang, L. Jiang and Z. Zhang, Nat. Commun., 2025, 16, 754 CrossRef CAS PubMed.
- K. Ma, P. Li, J. H. Xin, Y. Chen, Z. Chen, S. Goswami, X. Liu, S. Kato, H. Chen, X. Zhang, J. Bai, M. C. Wasson, R. R. Maldonado, R. Q. Snurr and O. K. Farha, Cell Rep. Phys. Sci., 2020, 1, 100024 CrossRef.
- Y. J. Hou, S. Fang, X. Y. Zhang, J. Wang, Q. Ruan, Z. Xiang, Z. Wang and X. J. Zhu, ACS Appl. Mater. Interfaces, 2022, 14, 49875–49885 CrossRef CAS PubMed.
- H. R. Li, S. Wu, X. Jing, Z. Di, K. Liu, L. Wang, C. P. Li, Z. Liu and M. Du, Sci. China: Chem., 2024, 67, 2958–2967 CrossRef CAS.
- R. Li, X. Chen, Z. Bian, R. Yu, Y. Chen, J. Zhang, J. Wang and X. Feng, J. Alloys Compd., 2025, 1020, 179345 CrossRef CAS.
- L. Giri, B. Mohanty, R. Thapa, B. K. Jena and V. R. Pedireddi, ACS Omega, 2022, 7, 22440–22446 CrossRef CAS PubMed.
- Q. Zhou, Y. Guo and Y. Zhu, Nat. Catal., 2023, 6, 574–584 CrossRef CAS.
- B. Yu, L. Li, S. Liu, H. Wang, H. Liu, C. Lin, C. Liu, H. Wu, W. Zhou, X. Li, T. Wang, B. Chen and J. Jiang, Angew. Chem., Int. Ed., 2021, 60, 8983–8989 CrossRef CAS PubMed.
- L. Yang, J. Yuan, G. Wang, Q. Cao, C. Zhang, M. Li, J. Shao, Y. Xu, H. Li and J. Lu, Adv. Funct. Mater., 2023, 33, 2300954 CrossRef CAS.
- G. Chen, L. Tong, S. Huang, S. Huang, F. Zhu and G. Ouyang, Nat. Commun., 2022, 13, 4816 CrossRef CAS PubMed.
- J. Tang, J. Liu, Q. Zheng, R. Yao and M. Wang, Angew. Chem., Int. Ed., 2023, 62, e202312784 CrossRef CAS PubMed.
- C. Huang, C. Zhao, Q. Deng, H. Zhang, D. Yu, J. Ren and X. Qu, Nat. Catal., 2023, 6, 729–739 CrossRef CAS.
- P. Luo, C. Zheng, J. He, X. Tu, W. Sun, H. Pan, Y. Zhou, X. Rui, B. Zhang and K. Huang, Adv. Funct. Mater., 2022, 32, 2107277 CrossRef CAS.
- B. Yu, J. Xing, P. Zhang, R. Gao, S. Lin, K. Jiang and L. Zhang, Inorg. Chem., 2025, 64, 1258–1262 CrossRef CAS PubMed.
- Y. Liang, R. An, P. Du, P. Lei and H. Zhang, Nano Today, 2023, 48, 101751 CrossRef CAS.
- Y. Nie, D. Li, Y. Peng, S. Wang, S. Hu, M. Liu, J. Ding and W. Zhou, Int. J. Pharm., 2020, 585, 119513 CrossRef CAS PubMed.
- W. Wang, Y. Shi, W. Chai, K. W. K. Tang, I. Pyatnitskiy, Y. Xie, X. Liu, W. He, J. Jeong, J. C. Hsieh, A. R. Lozano, B. Artman, X. Shi, N. Hoefer, B. Shrestha, N. B. Stern, W. Zhou, D. W. McComb, T. Porter, G. Henkelman, B. Chen and H. Wang, Nature, 2025, 638, 401–410 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |