 Open Access Article
Open Access ArticleMultistate coupled diabatic neural network potential for the quantum non-adiabatic photofragmentation of CH2+
Pablo del Mazo-Sevillano *a,
Susana Gómez-Carrasco
*a,
Susana Gómez-Carrasco a,
Alfredo Aguado
a,
Alfredo Aguado b and
Octavio Roncero
b and
Octavio Roncero *c
*c
aDepartamento de Química Física, Facultad de Ciencias Químicas, Universidad de Salamanca, 37008 Salamanca, Spain. E-mail: pablomazo@usal.es
bDepartamento de Química Física Aplicada (UAM), Unidad Asociada a IFF-CSIC, Facultad de Ciencias Módulo 14, Universidad Autónoma de Madrid, 28049 Madrid, Spain
cInstituto de Física Fundamental (IFF-CSIC), C.S.I.C., Serrano 123, 28006 Madrid, Spain. E-mail: octavio.roncero@csic.es
First published on 6th May 2026
Abstract
Tracking the complex non-adiabatic transitions in far-ultraviolet photodissociation demands highly accurate diabatic potential energy matrices (PEMs) across numerous excited states. To address this, we introduce a fully automated diabatization method that leverages artificial neural networks to fit PEMs. Our approach divides the PEM into a physically grounded zeroth-order diagonal term, which is then corrected by a neural network matrix to capture electronic couplings. By enforcing symmetry constraints on off-diagonal elements and sharing degenerate diabatic states between the A′ and A″ irreducible representations, the diabatization process becomes completely automatic. We validate this method using time-dependent wavepacket calculations to simulate the photodissociation of CH2+, incorporating relevant states up to ≈13.6 eV. Finally, we compute partial cross-sections for all fragmentation channels—including total and partial fragmentation yielding CH+, CH, H2, and H2+ diatoms—revealing a notably high cross-section for the formation of the CH radical.
1 Introduction
Photodissociation plays a fundamental role in outer atmospheres, on Earth and exo-planets, as well as in the borders of molecular clouds and protoplanetary disks,1–4 by controlling the abundance of small molecules. Those environments are irradiated by high-energy ultraviolet photons producing a large variety of radicals and ions, which in turn trigger the formation of more complex molecules. Hence, it is not only crucial to know the total photodissociation rates, but also the branching ratios for the formation of photo-products in different electronic states.The fragmentation dynamics at high energy is governed by non-adiabatic transitions among many excited electronic states. When these states cross, leading to conical intersections (CI), the dynamical simulations become complicated, since the non adiabatic couplings (NACs) present singularities.5,6 This problem is frequently overcome by a unitary transformation to a diabatic electronic representation7,8 where the kinetic adiabatic couplings vanish. However, this is in general not possible for all degrees of freedom,9 and the term quasi-diabatization is commonly used. Several diabatization procedures exist and they are classified as derivate-, property- or energy-based methods, depending on the information they use.10 One common approach is the regularization,11 i.e. the elimination of the singularities appearing at conical intersections.
Block diagonalization12,13 and effective Hamiltonian14 methods use reference geometries to generate a local description of diabatic Hamiltonians and are successfully applied to study photodissociation spectra. H3+,15 C6H6+ (ref. 16) and C4N2H417 are representative examples of diabatization, using derivative-based methods, of large systems including four or five electronic states, using normal coordinates thus allowing the accurate description of photoabsorption spectra using quantum time dependent methods, but not fully follow the dissociation dynamics. However, in many cases there are multiple crossings, not only in the Frank–Condon region but also close to products in different rearrangement channels, that need to be considered to properly describe the branching ratios in photodissociation. The problem becomes more complex when dealing with large energy intervals, as in astrochemistry, because a large number of electronic states are needed to describe the photofragmentation rate in all the UV radiation field.
The application of machine learning techniques to produce diabatic potential energy matrices (PEM) is being found to be extremely useful as has been already proven in different systems.18–21 For two-state systems fully automatic diabatization methods (meaning no diabatic information is used) can provide reasonable PEM from pure adiabatic information,22–24 leveraging the symmetry restrictions in the couplings of the PEM. As the number of states grows larger semi-automatic diabatization methods, as the family of diabatization by deep neural networks (DDNN25), exists, where minimal diabatic information is required.26,27 Other approaches use machine learning methods with clustering and regression techniques to generate smooth and well ordered diabatic states with property-based diabatization.28 In this work we present a fully automatic diabatization method applicable to many electronic states over a large energy interval for triatomic systems. For that we first construct an initial PEM model for describing the products in different rearrangements, which are correlated by symmetry considerations allowing to impose an ordering of the diabatic states. A many-body term expressed as an artificial neural network is added to the zeroth order model to correct the short distance region and include the couplings among the diabatic states with symmetry restrictions.
The diabatization method is applied to the study of the photodissociation of CH2+ cation, whose ground state presents important Renner–Teller effects due to its 2Π character29 and it has multiple ionic crossings in the CH+/CH and H2/H2+ products rearrangement channels, shown in Fig. 1, thus providing a good benchmark. This system is a missing stone in the hydrogenation chain CHn+ + H2 → CHn+1+ + H, giving rise to CH3+ which is considered the origin of hydrocarbons in the interstellar medium (ISM).30,31 CH+ is one of the first molecules observed in the ISM,32 and since then has been observed in a great variety of interstellar and circumstellar environments. On the other hand, CH3+ was observed very recently in a protoplanetary disk with the James Webb Space Telescope.33 Thus the missing detection of CH2+ is fundamental to corroborate the above mentioned hydrogenation chain as the formation mechanism of CH3+. Formation and destruction rates are needed to properly model the abundance of these ions. The photodissociation cross sections have been calculated for CH+ (ref. 34 and 35) and CH3+ (ref. 36) using quantum dynamical methods, while for CH2+ only a vertical excitation model from the ground equilibrium geometry is available.37
The final goal of this work is to study the non-adiabatic photofragmentation dynamics of CH2+ in a new diabatic model composed of 16 coupled electronic states, and it is organized as follows. First, the new diabatization method is presented and applied to CH2+. Next, the results on the photodissociation dynamics obtained with a quantum wave packet treatment are discussed. Finally, some conclusions are extracted.
2 Diabatization
Machine learning diabatization methods often rely on some diabatic information to impose the correct order of the different states. In this work we present a fully automatic diabatization method based on a many body expansion of the potential energy. It is applied to produce a diabatic potential energy matrix (PEM) of 16 states of the CH2+ system, 8 in each A′ and A″ irreducible representations of the Cs point group. With this number of states we properly include the Frank–Condon region of the CH2+ and possible dissociation channels up to ≈13.6 eV. While the diabatization method is automatic, it still requires a very detailed analysis of the electronic structure of the system.2.1 Diatomic fragments
The diatomic fragments in the [CH + H]+ and [C + H2]+ channels in Fig. 1 present a great variety of electronic states, bound and dissociative, in which the diatomic fragment can be neutral or ionized. When performing the calculations for the whole triatomic system in the A′ or A″ representations the adiabatic states present many crossings, related for instance with charge transfers between the distant partners. These adiabatic states are first diabatized by performing independent calculations of each diatomic fragment, with different charges and in different irreducible representations. Fig. 2 displays the diabatized diatomic curves for both CH/CH+ and H2/H2+ systems. | ||
| Fig. 2 Representation of the diabatic diatomic curves included in the U0 model and correlation with the total dissociation asymptotes. | ||
2.2 Correlation diagram
The different electronic states in all the rearrangement channels are connected with the lowest energy total fragmentation asymptotes: C+(2P) + H(2S) + H(2S), C(3P) + H(2S) + H+ and C(1D) + H(2S) + H+. A full description of the 22 states that correlate to these asymptotes would require a matrix size which is intractable and computationally very demanding to perform exact quantum dynamics simulations. Instead of including the full set of 10 electronic states correlating to the C(1D) asymptote, we have only considered the 4 states corresponding to the two Δ components. This truncation will yield an incomplete description of some asymptotic states as we will discuss later.Considering that the Λ quantum number and charge of each diatomic fragment are conserved across the diabatic states, a correlation between reactants and products can be done. On top of that, it is necessary to use three-body information to complete the correlation of states with the same Λ that tend to the same asymptote. The correlation diagram of the diatomic states is presented in Fig. 2.
The diabatic states are ordered as follows. The first two states are Σ and are different in the A′ and A″ diabatic manifolds. The next four states are of Π character and are degenerate in the A′ and A″ manifolds, correlating to the same products. The same occurs for the last two states, which are of Δ character. In the following we will use A′ and A″ to refer to adiabatic states and D′ and D″ for diabatic states. The states included in the model are enumerated in Table 1, together with the energies in the different minima and total dissociation.
| State | [CH + H]+ | ΔE/eV | [C + H2]+ | ΔE/eV | [H + H + C]+ | ΔE/eV |
|---|---|---|---|---|---|---|
| 1D′ | CH+(1Σ+) + H(2S) | 4.71 | H2(3Σu+) + C+(2P) | — | H + H + C+(2P) | 8.87 |
| 2D′ | CH+(3Σ+) + H(2S) | — | H2(1Σg+) + C+(2P) | 4.25 | H + H + C+(2P) | 8.87 |
| 1D″ | CH(2Σ−) + H+ | 11.17 | H2+(2Σu+) + C(3P) | — | H+ + H + C(3P) | 11.33 |
| 2D″ | CH+(3Σ−) + H(2S) | 9.49 | H2+(2Σg+) + C(3P) | 8.57 | H+ + H + C(3P) | 11.33 |
| 3D′, 3D″ | CH+(3Π) + H(2S) | 5.90 | H2(3Σu+) + C+(2P) | — | H + H + C+(2P) | 8.87 |
| 4D′, 4D″ | CH+(1Π) + H(2S) | 7.74 | H2(1Σg+) + C+(2P) | 4.25 | H + H + C+(2P) | 8.87 |
| 5D′, 5D″ | CH(2Π) + H+ | 7.88 | H2+(2Σu+) + C(3P) | — | H+ + H + C(3P) | 11.33 |
| 6D′, 6D″ | CH+(3Π) + H(2S) | — | H2+(2Σg+) + C(3P) | 8.57 | H+ + H + C(3P) | 11.33 |
| 7D′, 7D″ | CH(2Δ) + H+ | 10.86 | H2+(2Σu+) + C(1D) | — | H+ + H + C(1D) | 12.68 |
| 8D′, 8D″ | CH+(1Δ) + H(2S) | 11.33 | H2+(2Σg+) + C(1D) | 9.94 | H+ + H + C(1D) | 12.68 |
2.3 Potential energy matrix
Two independent PEMs are considered: for the A′ and A″ irreducible representations. The diagonal elements correspond to the diabatic states and the non-diagonal elements to their couplings. The Π and Δ subblocks are identical in both representations, as well as Π–Δ couplings, so the differences between the energies are only due to the Σ states and how they couple with the Π and Δ states in each representation.In our model we describe 16 electronic states, 8 in each A′ and A″ representations. The PEM is factored into a zeroth order diagonal matrix (U0), which accurately includes all the possible diatomic and total dissociation states, plus a three-body neural network matrix to incorporate corrections to the diabatic states and their couplings in the 3-body region (UNN),
| U = U0 + UNN | (1) |
The elements of U0 are expressed as:
| U0ii = V1B + V2B + Vlr | (2) |
The UNN matrix elements are computed from a permutationally invariant polynomial neural network (PIP–NN).38 Those associated to Π and Δ states are shared between the two PEM in order to maintain the degeneracy in linear configurations. The output of the neural network is multiplied by a damping function of the CH distances to make them zero as the system tends to a 2-body or total dissociation region. Additional constraints to the non-diagonal elements arise from the fact that for highly symmetric configurations (C∞v, D∞h) the PEM should resemble the adequate block structure. To impose these constraints, couplings between Σ–Π, Σ–Δ and Π–Δ are multiplied by a sine function of the H–C–H angle. The full diabatic matrices and further details on the neural network implementation are presented in the SI.
While in theory this method can be generalized to more than 3 particles, the main difficulty resides in the definition of U0. As the number of atoms increases, the number of relevant asymptotic states can be very large, which would lead to an intractable diabatic matrix. On the other hand, UNN can be easily generalized to other systems with a PIP representation.39–41
UNN is trained by minimizing the following loss function (![[script L]](https://www.rsc.org/images/entities/char_e144.gif) ) which, in contrast to other methods, only requires adiabatic information and hence makes the diabatization method automatic:
) which, in contrast to other methods, only requires adiabatic information and hence makes the diabatization method automatic:
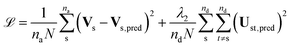 | (3) |
Two key aspects make this method different from similar PEM factorizations such as the PM-DDNN26 and turn the diabatization method into fully automatic: (1) sharing the degenerate diabatic states and couplings among the A′ and A″ irreducible representations and (2) constraining the non-diagonal couplings at highly symmetric configurations to resemble the adequate Σ, Π and Δ subblock structure in linear configurations.
2.4 Features of the PEM
The PEM has been trained with na = 5 and nd = 8 in eqn (3), meaning that the neural network term will modify the 8 diabatic states in each representation to finally reproduce accurately the 5 lowest adiabatic states of each representation. These states cover an energy up to 14 eV in the Frank–Condon region of the CH2+, which is the relevant energy range for astrochemical considerations. In the diatomic and total dissociation regions the number of accurate states is in general larger than five thanks to the U0 term. The ab initio calculations have been performed with the state averaged CASSCF level of theory followed by a MRCI calculation with the cc-pCVTZ basis set with ORCA 6.0.42–45 In all the calculations the core orbitals of the C atom are closed. A total of seven doublet states are computed for each representation of the Cs point group.U0 is responsible of reproducing the full PEM up to a rather small distance between the three atoms as depicted in Fig. 3, where a comparison between U0 and the ab initio data is presented. For R = 6 Å we find a good agreement with the exception of the states that are not included in the zeroth order model: some that correlate with C(1D) or the curve corresponding to H2 + C+(4P) which is visible in the right panels. As the third atom approaches to distances ≈3 Å, U0 is still rather accurate and only for very short distances the zeroth order model is clearly in disagreement. It is in this short range region where UNN acts, correcting the U0 diabatic energies and including the missing couplings.
Fig. 4 displays a path connecting the CH2+ in its linear configuration with the [CH + H]+ asymptote on the left and the [C + H2]+ asymptote in the right. In the top panel the adiabatic energies are compared with the calculated ab initio, represented with dots. For the 5 lowest adiabatic states we find a very good agreement along the path. In the bottom panel, the evolution of the diabatic states towards the photofragments is depicted. For instance, the minimum in the CH2+ configuration comes from the 5D′, 5D″ diabatic states which connect CH(2Π) + H+ and H2+(2Σu+) + C(3P) asymptotes, in accordance with our previous correlation diagram for this system.27 However, this is not evident from Fig. 3. In U0 the lowest diabatic Π state close to the CH2+ minimum are the 3D′, 3D″ states, which correlate with CH+(3Π) + H(2S) and H2(3Σu+) + C+(2P), so one could assume that UNN will try to preserve this order. Instead, UNN pushes the 3D′, 3D″ states up in energy while bringing down the 5D′, 5D″ to reproduce the minimum of the CH2+. Further comparisons of the adiabatic and ab initio results can be found in the SI.
The root mean squared errors (RMSE) of the different states are presented in Table 2. The largest error in the fifth state is due to the missing Σ and Π components of the C(1D) + H(2S) + H+ asymptote, which limits the capability of the model to describe the H2+(2Σg+) + C(1D) asymptote. Excluding [C + H2]+ geometries, the errors of the fifth adiabatic state up to 14 eV are 86.0 meV and 76.4 meV for the A′ and A″ irreducible representations, respectively.
| Irrep. | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|
| A′ | 35.0 (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 542) 542) |
41.3 (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 209) 209) |
46.1 (19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 557) 557) |
76.4 (18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 763) 763) |
523.8 (18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 331) 331) |
| A″ | 45.4 (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 421) 421) |
43.7 (19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 963) 963) |
41.2 (19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 239) 239) |
57.0 (18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500) 500) |
519.2 (17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 682) 682) |
More relevant to the quantum dynamics are the non-adiabatic coupling matrix elements (NACMEs) which can be analytically computed from the PEM and compared with ab initio calculations in regions close to a CI.46 Fig. 5 presents a comparison between those computed from the PEM and ab initio at a CASSCF level close to the CI between the first two adiabatic states in the CH+ + H exit.
3 Photodissociation dynamics
The photodissociation dynamics is studied with the quantum time-dependent MADWAVE3 code47 as described previously48–51 using the CH + H Jacobi coordinates, with r being the HC internuclear vector, R joining HC center-of-mass to the second H atom, and γ = r·R/rR. The rovibrational H2 products are analyzed using the reactant-product transformation method.52 The dynamics is performed in the 8 × 8 diabatic representations of the A′ and A″ symmetries independently.Here we consider the absorption from the ground vibrational states in the adiabatic ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2A′ and Ã2A″ states, described in the SI, corresponding to initial total angular momentum Ji = 0. The initial wave packet is built by multiplying the bound state by the transition electric dipole moment as described previously.48–51 The transition dipole moments have been calculated from the ground adiabatic states to the 5 (4) adiabatic excited electronic A′ (A″), as described in the SI. These adiabatic transition dipole moments are then transformed to the diabatic representation using the eigen-vectors obtained after diagonalization of the 8 × 8 diabatic matrix at each nuclear configuration. Thus, the simultaneous excitation of the first 5 (4) A′ (A″) adiabatic states is considered here.
2A′ and Ã2A″ states, described in the SI, corresponding to initial total angular momentum Ji = 0. The initial wave packet is built by multiplying the bound state by the transition electric dipole moment as described previously.48–51 The transition dipole moments have been calculated from the ground adiabatic states to the 5 (4) adiabatic excited electronic A′ (A″), as described in the SI. These adiabatic transition dipole moments are then transformed to the diabatic representation using the eigen-vectors obtained after diagonalization of the 8 × 8 diabatic matrix at each nuclear configuration. Thus, the simultaneous excitation of the first 5 (4) A′ (A″) adiabatic states is considered here.
We study the Ji = 0 → J = 1− rotational excitation for 4 different electronic transitions: from either ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2A′(0, 0, 0) or Ã2A″(0, 0, 0) initial states towards 8 electronically excited and coupled diabatic states, of either A′ or A″ symmetry.
2A′(0, 0, 0) or Ã2A″(0, 0, 0) initial states towards 8 electronically excited and coupled diabatic states, of either A′ or A″ symmetry.
The absorption spectra obtained from the autocorrelation function50,51 shows an excellent agreement with the total photodissociation flux over all fragments above the first dissociation limit, as displayed in Fig. 6. This serves not only to check to convergence of the calculation, but also to guarantee that all the flux over different fragmentation channels is properly collected.
The assignment of the bands is done by comparing these spectra to those obtained in the adiabatic representation, as described in the SI. The most intense peaks are those arriving to 5A′ and 4A″ electronic states, because these excited states have considerably larger transition dipole moments with ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2A′ and Ã2A″ states than the other electronic states at lower energies.
2A′ and Ã2A″ states than the other electronic states at lower energies.
The spectra to the A′ manifold of states obtained from the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) or the à states show significant differences, attributed to the different initial wave packets, formed from different initial bound states (specially in the angular coordinate) and different transition dipole moments (see SI).
or the à states show significant differences, attributed to the different initial wave packets, formed from different initial bound states (specially in the angular coordinate) and different transition dipole moments (see SI).
The transitions towards the A″ states show a drop around 10 eV, which is clearly attributed to the energy separation among the A″ states, less densely packed than the A′ states. The drop at 10 eV observed in the Ã2A″ → A′ transition is due to the reduction of the 1A″ → 3 and 4 A′ matrix elements of the dipole moments.
There is a great variety of fragmentation processes, as illustrated by the electronic states of the fragments shown in Fig. 2: total fragmentation
CH2+(![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2A′, Ã2A″) + hν → C+ + H + H 2A′, Ã2A″) + hν → C+ + H + H
| (4) |
| → C + H+ + H, |
CH2+(![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2A′, Ã2A″) + hν → CH+ + H 2A′, Ã2A″) + hν → CH+ + H
| (5) |
| → CH + H+, |
CH2+(![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2A′, Ã2A″) + hν → H2 + C+ 2A′, Ã2A″) + hν → H2 + C+
| (6) |
| → H2+ + C. |
These different fragmentation routes are distinguished by the analysis of different fluxes on each electronic state and on individual rovibrational states of H2/H2+ and CH/CH+ diatomic products.52 The total flux, obtained for the CH internuclear distance r = 6 Å, collects the total fragmentation, eqn (4), the formation of H2 or H2+, eqn (6), depending on the electronic state, and one half of CH or CH+, eqn (5), expressed as
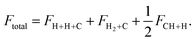 | (7) |
The fluxes for H2/H2+ and one of the two CH/CH+ rearrangement channels are obtained by evaluating the sum over all possible rovibrational diatomic eigen states in each channel for each electronic states, giving the quantities FH2+C and FCH+H/2. For the [CH + H]+ there are two equivalent rearrangement channels giving the same flux, FCH+H/2 each. In Fig. 7 the cross sections for each channel and final electronic states obtained after photodissociation of the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2A′(0, 0, 0) ground state towards the 8 lower A′ and A″ final electronic states, respectively, are shown with a similar qualitative behavior to those obtained from Ã2A″(0, 0, 0) initial state shown in the SI.
2A′(0, 0, 0) ground state towards the 8 lower A′ and A″ final electronic states, respectively, are shown with a similar qualitative behavior to those obtained from Ã2A″(0, 0, 0) initial state shown in the SI.
The main fluxes in the higher energy interval, E > 11 eV, are either to total fragmentation or to CH/CH+ diatomic fragments, with small proportion of H2/H2+ products. For the 5, 6, 7 and 8 diabatic D′ or D″ states, the total fragmentation yield neutral carbon atoms, in either 3P or 1D states and the charge is in the hydrogen atom. On the contrary, for the 3, 4 D′ or D″ states the charge is in the carbon atoms in the C+(2P) state. The Σ states in the A′ and A″ symmetry show different behavior: in the A′ manifold the charge is in the C+(2P) ion, while in the A″ manifold the charge is in the hydrogen atoms, leading neutral C(3P) atoms.
The most astonishing result is the high cross section to form neutral CH products, in either the ground X2Π (states 5D′ and 5D″) or A2Δ (states 7D′ and 7D″). CH can be formed in the neutral–neutral reaction C(3P) + H2, but is endothermic by ≈1 eV. Thus the neutral reaction can only occur when H2 is initially vibrationally excited in v = 2 or 3. Abundant highly vibrationally excited H2 is only found in strongly UV illuminated regions such as the borders of molecular clouds (photodissociation or PDR regions) or protoplanetary discs (PPDs). In such environments, the high UV flux will also induce the formation of neutral CH fragments by the photodissociation of CH2+.
The formation of CHn+ is attributed to the hydrogenation chain of reactions CHn+ + H2 → CHn+1+ + H, which stops in CH3+, which in turn reacts with many other molecules, being the origin of several chemical networks for the formation of many larger hydrocarbons. CH+ is one of the first observed molecules in the ISM32 and since then widely observed. CH3+ was recently observed33 in a protoplanetary disk (d203–506) illuminated by the strong far ultraviolet (FUV) radiation field from nearby massive stars in Orion's trapezium cluster. However, CH2+ has not been observed so far.
Thus the confirmation of this hydrogenation chain requires the detection of CH2+, and its abundance determination requires incorporating all the destruction pathways, such as photodissociation, dissociative recombination with electrons and hydrogenation.
The total photodissociation cross section of CH2+(![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) ) (summing the contributions towards A′ and A″ manifolds) is shown in Fig. 8 and compared to those of CH3+ from ref. 36 and CH+, which is recalculated in this work as described in the SI and is in agreement with that reported by Kirby et al.35 The photodissociation cross section of CH2+ is larger than that of CH3+ but smaller than that of CH+.
) (summing the contributions towards A′ and A″ manifolds) is shown in Fig. 8 and compared to those of CH3+ from ref. 36 and CH+, which is recalculated in this work as described in the SI and is in agreement with that reported by Kirby et al.35 The photodissociation cross section of CH2+ is larger than that of CH3+ but smaller than that of CH+.
 | ||
Fig. 8 Total photodissociation cross section for the ground vibrational state of CH2+ (![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2A′) (summed over all A′ and A″ states), compared to those of CH3+ from ref. 36, and CH+, recalculated here (see text) and very similar to that of Kirby et al.35 2A′) (summed over all A′ and A″ states), compared to those of CH3+ from ref. 36, and CH+, recalculated here (see text) and very similar to that of Kirby et al.35 | ||
The photodestruction is determined by the photodissociation rate obtained as the integral of the photodissociation cross section with the energy dependent radiation field. Using Draine's mean interstellar radiation field,53 the photodissociation rate of CH2+ is 6.45 × 10−11 s−1, while that of CH3+ is36 6.83 × 10−12 s−1 and for CH+ is 2.89 × 10−10 s−1. The rates obtained here for CH2+ and CH+ are slightly lower than the values reported for the ISRF field in ref. 3 of 1.4 and 3.3 × 10−10 s−1, respectively. One reason is that the ISRF flux is a modification of the Draine's field. The difference for CH2+ is more important because a simple vertical transition was assumed to calculate the photodissociation cross section.
The total photodissociation cross section to form neutral CH products is shown in Fig. 9, which are rather high specially for CH(X2Π) at higher photon energies, where the UV radiation field is not shielded. The photodissociation rates to form CH in the 2Π and 2Δ electronic states are 9.96 × 10−12 s−1 and 3.37 × 10−12 s−1, respectively, using Draine's mean interstellar radiation field.53 These values are small, but considering that the H2 + C neutral reaction is endothermic by 1 eV, it can be an alternative source of CH.
 | ||
Fig. 9 Total photodissociation cross section to form neutral CH products from the ground vibrational state of CH2+ (![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2A′ (summed over all A′ and A″ states). 2A′ (summed over all A′ and A″ states). | ||
4 Conclusions
The study of the branching ratios among the different photoproducts in photodissociation over large energy intervals, as needed in astrochemistry, requires the description of non-adiabatic dynamics, involving many electronic states. To overcome the singularities of non-adiabatic couplings at conical intersections, the most widely used method is a unitary transformation to a diabatic representation, which is not unique. There are many diabatization methods, but usually they are restricted to few states. In this work we have developed a diabatization method describing up to 16 electronic states, divided in two symmetry blocks. The method has been applied to the photodissociation of CH2+, not yet dynamically studied, and that present many photoproducts, bound and dissociative and with several charge distributions.The diabatization method consists in building a basis of electronic states to represent the possible products, with different charges and electronic excitation. A correlation among the rearrangement channels is done, imposing symmetry restrictions, such as the conservation of the projection of angular momentum, Λ, on the diatomics fragments. The full diabatic matrices are then factorized in two terms, a diagonal zero-order term and a full matrix built using a neural network method, VNN. The zero-order term allows to describe the diagonal interaction among the fragments up to intermediate distances, ≈3–4 Å, keeping the nature of the asymptotic states. The VNN matrix is build with an artificial neural network, imposing symmetry restrictions on the couplings at symmetric configurations and introducing a penalty function to force non-diagonal terms to be as small as possible. This process allowed an accurate description of the lowest 10 adiabatic eigenvalues in the short distance range and 16 in the asymptotes, separated in two symmetry blocks, covering energies up to 14 eV.
The diabatic model allows the quantum dynamics of the total photodissociation cross sections rate constants for CH2+ up to 13.6 eV, interval needed to astrochemical models. More important, it allows to describe the partial cross section for a great variety of fragmentation processes, with the products in different arrangement channels, charge and electronic states: total fragmentation, C+ + H + H and C + H + H+, CH+(X1Σ,) + H, CH(X2Π, 2Δ), H2(X1Σ,) + C+ and H2+(2Σg).
Here we focus in the formation of the CH radical, whose formation in the C + H2 reaction is endothermic. In strongly illuminated astronomical objects, the CH2+ photodissociation leads to significant of CH radical, and it is important to determine its contribution in PDRs and PPDs under different conditions to determine its contribution.
CH2+ is a prototype and this kind of studies are needed to search for alternative formation routes of many molecules and radicals in astronomical objects illuminated by UV radiation. For this purpose diabatization is a key important requirement, specially in cases where the fragments present crossings. At these crossings at long distance the non-adiabatic couplings vanish and surface hopping methods would adiabatically continue in one adiabatic state, corresponding to a mixture of two states each one assigned to one chemical product, as illustrated in Fig. 2 for the present case.
Author contributions
Pablo del Mazo-Sevillano: conceptualization (equal); methodology (equal); software (equal); writing – review & editing (equal). Alfredo Aguado: conceptualization (equal); funding acquisition (equal); methodology (equal); writing – review & editing (equal). Susana Gómez-Carrasco: conceptualization (equal); funding acquisition (equal); writing – review & editing (equal). Octavio Roncero: conceptualization (equal); funding acquisition (equal); methodology (equal); project administration (equal); software (equal); writing – review & editing (equal).Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information is available. See DOI: https://doi.org/10.1039/d6cp01221c.The total photodissociation cross sections calculated here for CH2+ and CH+ are available at ZENODO https://zenodo.org/records/19847742https://zenodo.org/records/19847742. The codes used for the calculations have been properly cited, and the details of the calculations are provided in the supplementary information (SI). Any other data that support the findings of this study are available from the authors, upon reasonable request.
Acknowledgements
This work has received funding from Ministerio de Ciencia, Innovación y Universidades, MICIU (Spain), under Grants No. PID2021-122549NB-C21, PID2021-122549NB-C22 and PID2024-156686NB-I00. Computational assistance was provided by the Supercomputer facilities of Lusitania founded by the CénitS and Computaex Foundation.Notes and references
- M. C. van Hemert and E. F. van Dishoeck, Chem. Phys., 2008, 343, 292 CrossRef CAS.
- E. F. van Dishoeck and R. Visser, Laboratory Astrophysics: from Molecules through Nanoparticles to Grains, Wiley-VCH, 2015, p. 1 Search PubMed.
- A. N. Heays, A. D. Bosman and E. F. van Dishoeck, Astron. Astrophys., 2017, 602, A105 CrossRef.
- H. R. Hrodmarsson and E. F. van Dishoeck, Astron. Astrophys., 2023, 675, A25 CrossRef CAS.
- D. R. Yarkony, Modern electronic structure theory, World Scientific, Singapore, 1995, p. 642 Search PubMed.
- D. R. Yarkony, Conical Intersections: electronic structure, dynamics and spectroscopy, Advanced series in Physical Chemistry, World Scientific Publishing Co., 2004, p. 175 Search PubMed.
- F. T. Smith, Phys. Rev., 1969, 179, 111 CrossRef.
- K. Ruedenberg and G. J. Atchity, J. Chem. Phys., 1993, 99, 3799 CrossRef CAS.
- C. A. Mead and D. G. Truhlar, J. Chem. Phys., 1982, 77, 6090 CrossRef CAS.
- H. Köppel, Conical Intersections: electronic structure, dynamics and spectroscopy, Advanced series in Physical Chemistry, World Scientific Publishing Co., 2004, p. 175 Search PubMed.
- A. Thiel and H. Köppel, J. Chem. Phys., 1999, 110, 9371 CrossRef CAS.
- T. Pacher, H. Köppel and L. S. Cederbaum, J. Chem. Phys., 1991, 95, 6668 CrossRef CAS.
- T. Pacher, L. S. Cederbaum and H. Köppel, Adv. Chem. Phys., 1993, 84, 293 CrossRef CAS.
- B. N. Papas, M. S. Schuurman and D. R. Yarkony, J. Chem. Phys., 2008, 129, 124104 CrossRef PubMed.
- S. Ghosh, S. Mukherjee, B. Mukherjee, S. Mandal, R. Sharma, P. Chaudhury and S. Adhikari, J. Chem. Phys., 2017, 147, 074105 CrossRef PubMed.
- S. Mukherjee, S. Ravi, K. Naskar, S. Sardar and S. Adhikari, J. Chem. Phys., 2021, 154, 094306 CrossRef CAS.
- S. Hazra, S. Mukherjee, S. Ravi, S. Sardar and S. Adhikari, ChemPhysChem, 2022, 23, e202200482 CrossRef CAS PubMed.
- Y. Guan, H. Guo and D. R. Yarkony, J. Chem. Phys., 2019, 150, 214101 CrossRef PubMed.
- Y. Guan, D. H. Zhang, H. Guo and D. R. Yarkony, Phys. Chem. Chem. Phys., 2019, 21, 14205–14213 RSC.
- Y. Guan, C. Xie, D. R. Yarkony and H. Guo, Phys. Chem. Chem. Phys., 2021, 23, 24962–24983 RSC.
- D. M. G. Williams and W. Eisfeld, J. Chem. Phys., 2018, 149, 204106 CrossRef PubMed.
- C. Xie, X. Zhu, D. R. Yarkony and H. Guo, J. Chem. Phys., 2018, 149, 144107 CrossRef PubMed.
- W. Li, Y. Liang, X. Niu, D. He, W. Xing and Y. Zhang, J. Chem. Phys., 2024, 161, 044310 CrossRef CAS PubMed.
- W. Li, B. Dong, X. Niu, M. Wang and Y. Zhang, J. Chem. Phys., 2024, 074302 CrossRef CAS PubMed.
- Y. Shu and D. G. Truhlar, J. Chem. Theory Comput., 2020, 16, 6456 CrossRef CAS PubMed.
- Y. Shu, F. B. Akher, H. Guo and D. G. Truhlar, J. Phys. Chem. A, 2024, 128, 1207–1217 CrossRef CAS PubMed.
- P. del Mazo-Sevillano, A. Aguado, F. Lique, R. A. Jara-Toro and O. Roncero, Phys. Chem. Chem. Phys., 2025, 27, 15775–15786 RSC.
- S. Sršeň, O. A. von Lilienfeld and P. Slavíček, Phys. Chem. Chem. Phys., 2024, 26, 4306 RSC.
- P. Jensen, M. Brumm, W. P. Kraemer and P. R. Bunker, J. Mol. Spectrosc., 1995, 172, 194 CrossRef CAS.
- J. H. Black and A. Dalgarno, Astrophys. J., 1976, 203, 132–142 CrossRef CAS PubMed.
- D. Smith, Chem. Rev., 1992, 92, 1473 CrossRef CAS.
- A. E. Douglas and G. Herzberg, Astrophys. J., 1941, 94, 381 CrossRef CAS.
- O. Berné, M.-A. Martin-Drumel and I. Schroetter, et al., Nature, 2023, 621, 56 CrossRef PubMed.
- R. P. Saxon, K. Kirby and B. Liu, J. Chem. Phys., 1980, 73, 1873 CrossRef CAS.
- K. Kirby, W. G. Roberge, R. P. Saxon and B. Liu, Astrophys. J., 1980, 855 CrossRef CAS.
- P. del Mazo-Sevillano, A. Aguado, J. R. Goicoechea and O. Roncero, J. Chem. Phys., 2024, 160, 184307 CrossRef PubMed.
- G. Theodorakopoulos and I. D. Petsalakis, J. Mol. Struct.:THEOCHEM, 1991, 230, 205 CrossRef.
- B. Jiang and H. Guo, J. Chem. Phys., 2013, 139, 054112 CrossRef PubMed.
- J. Li, Z. Varga, D. G. Truhlar and H. Guo, J. Chem. Theory Comput., 2020, 16, 4822–4832 CrossRef CAS PubMed.
- P. del Mazo-Sevillano, D. Félix-González, A. Aguado, C. Sanz-Sanz, D. Kwon and O. Roncero, Mol. Phys., 2024, e2183071 CrossRef.
- P. L. Houston, C. Qu, Q. Yu, P. Pandey, R. Conte, A. Nandi and J. M. Bowman, J. Chem. Theory Comput., 2024, 20, 3008–3018 CrossRef CAS PubMed.
- F. Neese, Wiley Interdiscip. Rev.:Comput. Mol. Sci., 2025, 15, e70019 Search PubMed.
- C. Kollmar, K. Sivalingam, B. Helmich-Paris, C. Angeli and F. Neese, J. Comput. Chem., 2019, 40, 1463–1470 CrossRef CAS PubMed.
- F. Neese, J. Comput. Chem., 2022, 44, 381 CrossRef PubMed.
- M. Ugandi and M. Roemelt, Int. J. Quantum Chem., 2023, 123, e27045 CrossRef CAS.
- C. Sanz-Sanz, A. Aguado, O. Roncero and F. Naumkin, J. Chem. Phys., 2015, 143, 234303 CrossRef PubMed.
- O. Roncero and P. del Mazo-Sevillano, Comput. Phys. Commun., 2025, 109471 CrossRef CAS.
- M. Paniagua, A. Aguado, M. Lara and O. Roncero, J. Chem. Phys., 1999, 111, 6712 CrossRef CAS.
- A. Aguado, M. Paniagua, C. Sanz-Sanz and O. Roncero, J. Chem. Phys., 2003, 119, 10088 CrossRef CAS.
- A. Chenel, O. Roncero, A. Aguado, M. Agúndez and J. Cernicharo, J. Chem. Phys., 2016, 144, 144306 CrossRef PubMed.
- A. Aguado, O. Roncero, A. Zanchet, M. Agúndez and J. Cernicharo, Astrophys. J., 2017, 838, 33 CrossRef.
- S. Gómez-Carrasco and O. Roncero, J. Chem. Phys., 2006, 125, 054102 CrossRef PubMed.
- B. T. Draine, Astrophys. J., Suppl. Ser., 1978, 36, 595 CrossRef CAS PubMed.
| This journal is © the Owner Societies 2026 |






