 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Mechanism of glass transition temperature enhancement in multicomponent epoxy resins incorporating triazine rings
Yuuki Kinugawaa,
Yoshiaki Kawagoe *b,
Keigo Matsumotoc,
Masashi Ohnoc,
Shoko Mishima
*b,
Keigo Matsumotoc,
Masashi Ohnoc,
Shoko Mishima a,
Takahiko Kawaib and
Tomonaga Okabe
a,
Takahiko Kawaib and
Tomonaga Okabe ade
ade
aDepartment of Aerospace Engineering, Tohoku University, 6-6-01, Aoba, Aramaki, Aoba-ku, Sendai, Miyagi 980-8579, Japan
bResearch Center for Green X-Tech, 6-6-11, Aoba, Aramaki, Aoba-ku, Sendai, Miyagi 980-8579, Japan. E-mail: kawagoe@tohoku.ac.jp
cChemical Research Laboratories, Nissan Chemical Corporation, 10-1, Tsuboi-Nishi 2-chome, Funabashi, Chiba 274-8507, Japan
dDepartment of Materials Science and Engineering, University of Washington, BOX 352120, Seattle, WA 98195-1750, USA
eResearch Center for Structural Materials, Polymer Matrix Hybrid Composite Materials Group, National Institute for Materials Science, 1-2-1 Sengen, Tsukuba, Ibaraki 305-0047, Japan
First published on 15th May 2026
Abstract
Epoxy resins are widely used in electronic and structural applications. However, their use at high temperatures is limited by their glass transition temperature (Tg). Although the addition of multifunctional resins increases Tg via crosslink density, the molecular-scale contribution of specific intermolecular interactions, such as π–π stacking, remains poorly understood in multicomponent systems. Here, we investigated the molecular-level mechanism of Tg enhancement in a multicomponent epoxy resin system incorporating a triazine-based epoxy resin, tris(2-epoxypropyl)isocyanurate (TEPIC), into a conventional epoxy resin. The experimental results revealed a non-monotonic dependence of Tg on TEPIC content, with an initial decrease at low concentration followed by a pronounced increase at higher concentration, which cannot be explained solely by changes in crosslink density. Structural analysis using wide-angle X-ray scattering and molecular dynamics (MD) simulations showed that the amorphous halo originating from intermolecular packing splits into two peaks, suggesting that one of these peaks includes a contribution from π–π stacking interactions. MD simulations showed that ring pairs with centre-of-mass distances of 3.4–5.8 Å form stable π–π stacking structures, and that the population of these interactions correlates strongly with the observed variation in Tg. Quantum chemical calculations further demonstrated that benzene–triazine stacking interactions introduced by TEPIC are significantly stronger than benzene–benzene stacking. These results indicate that the enhancement of Tg arises not only from an increase in crosslink density but also from localized intermolecular constraints induced by strong stacking interactions, providing a molecular-level design guideline for high-performance epoxy resins.
1. Introduction
Epoxy resins are widely used in advanced electronic materials, such as adhesives and semiconductor encapsulants, owing to their high mechanical strength and excellent heat resistance, electrical insulation, and adhesion properties. In recent years, the continual miniaturization of electronic devices and the increasing demand for high-performance devices have led to a significant increase in power density and heat generation inside these devices. Therefore, the resin materials used for insulation, protection, and mechanical support in electronic devices must operate under high-temperature conditions. Epoxy resins exhibit a marked degradation in their thermal and mechanical properties at temperatures above their glass transition temperature (Tg),1–3 so they are usually limited to application temperatures below Tg. Therefore, it is necessary to increase the Tg of epoxy resins to enable further improvements in the performance of miniaturized devices.Diglycidyl ether of bisphenol A (DGEBA) is widely used owing to its relatively low cost and ease of handling; however, its relatively low Tg limits its use under demanding thermal conditions. To overcome this drawback, DGEBA is often modified by incorporating multifunctional epoxy resins to increase its crosslink density and hence, the Tg of the composite system.4–9 Despite the wide range of strategies proposed for increasing Tg, the fundamental details of the resulting crosslinked network structures remain insufficiently understood because the direct experimental observation of epoxy networks at the molecular level is inherently challenging. Therefore, many studies have indirectly inferred changes in the internal structure from macroscopic properties, such as the Tg and elastic modulus.
X-ray scattering methods are effective techniques for evaluating the periodicity of crosslinked structures and intermolecular interactions within epoxy resins.10–15 Ogawa et al.10 investigated the structural periodicity of epoxy resins using medium-angle X-ray scattering and revealed that a periodic structure at q = 1.2 nm−1 centered around the curing agent is formed as the epoxy–amine curing reaction proceeds. Dwyer et al.11 analyzed the polymer network structure of epoxy resins by wide-angle X-ray scattering (WAXS) and reported that the distance between π–π stacking rings increases upon heating.
To obtain a more detailed understanding of the relationship between the crosslinked structure and macroscopic properties, structural analysis at the molecular level is essential. Molecular dynamics (MD) simulations are a powerful technique for reproducing the curing process and network structure of epoxy resins at the molecular scale. In recent years, MD techniques have been increasingly used in combination with experimental techniques such as WAXS.10,16–22 Li et al.23 investigated the relationship between π–π stacking and the Tg of uncured silicon-containing arylacetylene resins using MD simulations. They demonstrated that increasing the fraction of aromatic rings enhances π–π interactions, which in turn contribute to increasing the Tg and viscosity. Yamamoto et al.15 investigated the correlation between crosslinked structures and physical properties in epoxy resins with various stoichiometric ratios using WAXS and MD simulations. They reported that, as the amine content increases beyond the stoichiometric ratio, the expansion of the distance between phenyl groups during the curing reaction is suppressed, providing steric resistance during deformation, leading to an increase in Young's modulus. Kawagoe et al.17 calculated scattering intensities using MD simulations and showed, through comparison with WAXS measurements, that the amorphous halos and peaks emerging during curing were successfully reproduced by the simulations. Furthermore, Zhao et al.8 used MD simulations to demonstrate that a higher tetraglycidyl diamino diphenyl methane (TGDDM) content in the DGEBA/TGDDM/4,4′-diaminodiphenyl sulfone (4,4′-DDS) system increases the number of ring structures, thereby improving the thermomechanical properties.
Most previous studies primarily interpreted the thermomechanical properties of cured epoxy resins in terms of network formation, via parameters such as the crosslink density and segmental rigidity (e.g., aromatic versus aliphatic linkages between junctions). In contrast, fewer studies have leveraged the richer structural information accessible by WAXS, including short-range intermolecular ordering, and combined it with MD simulations to establish structure–property relationships beyond network connectivity. Therefore, to deepen the understanding of structure–property relationships beyond network connectivity, we investigated a multicomponent epoxy resin system in which a triazine-based epoxy resin, tris(2-epoxypropyl)isocyanurate (TEPIC), is incorporated into a conventional DGEBA matrix. TEPIC has been widely used as an additive to enhance thermomechanical properties.24–26 In this study, the internal structure and thermomechanical properties were predicted by MD simulations, and the validity of the simulations was examined through comparison with WAXS measurements. This study aimed to elucidate the mechanism of Tg enhancement induced by the introduction of triazine rings based on the correlation between the internal structure and thermomechanical properties.
2. Experiments
2.1. Preparation
DGEBA (Mitsubishi Chemical Corporation) and TEPIC (Nissan Chemical Corporation) epoxy resins and 4,4′-DDS (Tokyo Chemical Industry Co., Ltd) curing agent were used. The molecular structures of these materials are shown in Fig. 1. Specimens were prepared by varying the TEPIC content in the DGEBA/TEPIC/4,4′-DDS resin system (0, 5, 10, 20, and 30 wt%), which were experimentally analyzed using thermomechanical and WAXS techniques.The cured epoxy resins were prepared as follows. First, DGEBA and 4,4′-DDS were stirred at 120 °C for 45 min. Subsequently, TEPIC preheated to 130 °C was added, and the mixture was stirred for an additional 10 min under vacuum using a rotation–revolution vacuum mixer (ARV-310P, Thinky) to obtain a homogeneous resin mixture. The 0 wt% mixture was prepared using the exact same procedure, simply omitting TEPIC. For all component formulations, the mixtures were carefully formulated to maintain a stoichiometric ratio of 1.0 between the total number of epoxy groups (from DGEBA and TEPIC) and the amine groups (from 4,4′-DDS). The mixture was poured into a mold made of glass plates covered with release film, and cured in an oven by heating from room temperature to 180 °C at a rate of 1.7 °C min−1, followed by holding at 180 °C for 2 h. The specimen thickness was controlled to 2 mm using polytetrafluoroethylene spacers. Various test specimens were prepared by cutting the cured plates to the required dimensions.
2.2. Thermomechanical properties
Two methods, thermomechanical analysis (TMA) (TMA7300, Hitachi High-Tech) and dynamic mechanical analysis (DMA) (DMA7100, Hitachi High-Tech), were used to determine the Tg values of the cured resins. In the TMA measurements, the specimens were heated from room temperature to a temperature above the Tg at a rate of 2 °C min−1 under compression mode with an applied load of 1 mN. The specimen size was 10 mm × 10 mm × 2 mm, and Tg was determined from the intersection of the linear fits to the temperature–thermal strain relationship in the glassy and rubbery regions along the 10 mm length direction. For DMA, the specimens were heated from room temperature to a temperature above Tg, and Tg was evaluated from the peak position of the loss modulus (E″). E″ was selected over the storage modulus (E′) and loss tangent because it provided superior sensitivity and resolution for distinguishing the multiple glass transitions observed in the multicomponent systems. The measurements were conducted in tensile mode with a heating rate of 2 °C min−1 and a frequency of 1 Hz. The specimen dimensions were 30 mm × 10 mm × 2 mm. The gauge length was set to 15 mm, and the displacement was set to 7.5 µm.2.3. Wide-angle X-ray scattering measurements
WAXS measurements were employed to quantitatively evaluate the molecular structure inside the resin. WAXS measurements were conducted using BL08W at the NanoTerasu synchrotron facility (Miyagi, Japan). 2D-WAXS patterns were recorded using a 2D hybrid photon counting detector Eiger2 R (Bruker K.K., Japan). WAXS measurements were performed using a camera length of 80 mm at room temperature and over the range of 80–280 °C in 20 °C increments.3. Numerical methods
3.1. Quantum-chemical reaction-path calculations
In the DGEBA/TEPIC/4,4′–DDS resin system, the epoxy–amine network can be formed through six elementary reaction types: (i) DGEBA reacting with primary amines; (ii) DGEBA reacting with secondary amines generated after DGEBA addition (DGEBA/secondary amine/DGEBA); (iii) DGEBA reacting with secondary amines generated after TEPIC addition (DGEBA/secondary amine/TEPIC); (iv) TEPIC reacting with primary amines; (v) TEPIC reacting with secondary amines generated after DGEBA addition (TEPIC/secondary amine/DGEBA); and (vi) TEPIC reacting with secondary amines generated after TEPIC addition (TEPIC/secondary amine/TEPIC). However, because these six reaction types occur simultaneously during curing, it is difficult to experimentally evaluate the relative magnitudes of the activation energies associated with each reaction. Therefore, only the reactions that could be evaluated by differential scanning calorimetry (DSC) measurements were considered in this study, i.e., DGEBA/primary amine, DGEBA/secondary amine/DGEBA, TEPIC/primary amine, and TEPIC/secondary amine/TEPIC. The activation energies and heats of formation, which characterize the interactions between the base resin and curing agent, are key factors governing both the curing process and the properties of the cured resin.27,28 These reaction parameters were determined using the global reaction route mapping (GRRM) method29–32 combined with density functional theory (DFT) calculations at the B3LYP/6-31G(d) level.Table 1 summarizes the activation energies and heats of formation obtained from the GRRM calculations. Based on the order of the activation energies, the reaction between TEPIC and primary amines proceeds first, followed by reactions between DGEBA and primary amines and between DGEBA and secondary amines, and finally the reaction between TEPIC and secondary amines occurs. This reaction sequence is in good agreement with the results obtained from separately conducted DSC measurements. These parameters were used as input data for the MD simulations of epoxy curing. For the DGEBA/secondary amine/TEPIC reaction, the reaction heat and activation energy of the DGEBA/secondary amine/DGEBA reaction were used, while for the TEPIC/secondary amine/DGEBA reaction, the corresponding values of the TEPIC/secondary amine/TEPIC reaction were adopted. The values for the DGEBA/primary amine and DGEBA/secondary amine/DGEBA reactions were taken from previous studies.8
3.2. Molecular dynamics simulations
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000. Periodic boundary conditions were imposed in all directions, and the simulation box was isothermally compressed at 300 K until the system density reached 0.9 g cm−3. Subsequently, a 300 ps NPT simulation was performed at 300 K and 1 atm to obtain the initial uncured structure.
000. Periodic boundary conditions were imposed in all directions, and the simulation box was isothermally compressed at 300 K until the system density reached 0.9 g cm−3. Subsequently, a 300 ps NPT simulation was performed at 300 K and 1 atm to obtain the initial uncured structure.Curing MD simulations were then performed by applying the following reaction model to the initial structure. Two criteria were applied to the reaction model. The first criterion was a distance condition, which determines candidate reactive pairs. When the distance between the representative atoms of two functional groups falls below the reaction cutoff distance Rc, the pair is considered a reaction candidate. In this work, the terminal C atom of the epoxy group and the N atom of the amine group were defined as the representative atoms, and the cutoff distance was set to Rc = 5.64 Å. The second criterion was the reaction probability condition, which determines whether a candidate pair will react. The reaction probability p for pairs satisfying the distance condition was calculated using the Arrhenius equation:
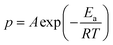 | (1) |
If the calculated p exceeds a uniformly generated random number a in the range from 0 to 1, a crosslinking reaction is assumed to occur between the corresponding functional groups. After each reaction event, structural relaxation was performed using the conjugate gradient method, followed by an NPT simulation (15 ps, 1 atm) and an NVT simulation (1 ps). Finally, the atomic velocities in the reaction region were scaled so that the kinetic energy satisfied Kafter = Kbefore + Hf, where Kbefore and Kafter are the total kinetic energies of the reactive atoms before and after the reaction, respectively, and Hf is the heat of formation obtained from the GRRM. The curing simulation was repeated until no further reactions occurred.
Before evaluating the thermomechanical properties of the cured models obtained from the curing simulations, annealing was performed following the methodology reported in previous studies.35,40 The annealing process consisted of an NVT simulation in which the temperature was cyclically varied between 300 and 1000 K, followed by an NPT simulation for equilibration. All MD simulations in this study were performed using the LAMMPS MD simulator,41 and the crosslink-formation procedure was implemented using in-house code. Interatomic interactions were described using the DREIDING force field,42 and partial charges were assigned using the charge equilibrium (QEq) method43 at each cycle. The QEq parameters reported by Zhang et al.44 and Demir and Walsh45 were employed. The van der Waals interactions were modeled using a Lennard–Jones potential with a cutoff distance of 12 Å, and electrostatic interactions were calculated using the particle–particle–particle–mesh method.46 The r-RESPA algorithm47 was used for time integration, with different timesteps assigned to specific interactions: 0.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) fs for bond and angle interactions, and 1.0
fs for bond and angle interactions, and 1.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) fs for dihedral, van der Waals, and electrostatic interactions. For both NVT and NPT simulations, temperature and pressure were controlled using the Nosé–Hoover thermostat and barostat, respectively.48
fs for dihedral, van der Waals, and electrostatic interactions. For both NVT and NPT simulations, temperature and pressure were controlled using the Nosé–Hoover thermostat and barostat, respectively.48
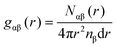 | (2) |
The coherent scattering intensity was then evaluated from the Fourier transformation of gαβ(r). Because the transformation was performed over a finite range (0 ≤ r ≤ rmax), a window function was applied to suppress termination ripples. In this study, the Lorch window function,
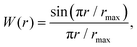 | (3) |
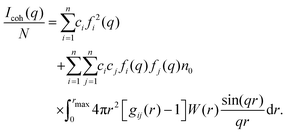 | (4) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sin
sin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ/λ is the magnitude of the scattering vector, where λ is the wavelength of the incident X-ray radiation and 2θ is the diffraction angle.
θ/λ is the magnitude of the scattering vector, where λ is the wavelength of the incident X-ray radiation and 2θ is the diffraction angle.
3.3. Quantum-chemical evaluation of fragment interaction energies
Quantum-chemical calculations were performed using the Gaussian program package to evaluate interaction energies between selected fragment pairs. Geometry optimization steps were performed at the MP2/6-311G(d,p) level. Single-point interaction energies were then computed at the MP2/aug-cc-pVDZ level with basis set superposition error correction using the counterpoise method,49 and the resulting interaction energies are presented in the subsequent sections.4. Results and discussion
4.1. Thermal properties
Fig. 2 shows the results of the DMA and TMA measurements and summarizes the Tg values for each resin determined from these measurements and MD simulations. The DMA results show that, compared with the unmodified system (0 wt%), adding 5 wt% TEPIC decreases Tg, which then increases markedly when the TEPIC content exceeds 10 wt%. Similarly, the TMA results indicate lower Tg values for the 5 wt% and 10 wt% TEPIC systems compared to that of the unmodified system, whereas a pronounced increase in Tg is observed upon the addition of 20 wt% or more TEPIC. Thus, both DMA and TMA measurements consistently reveal a characteristic behavior in which a decrease in Tg is observed with the addition of low TEPIC concentrations. This trend is also supported by the Tg values obtained from MD simulations. In contrast, an examination of E′ measured by DMA at temperatures above the Tg shows that the crosslink density increases monotonically with increasing TEPIC content. In general, the incorporation of resins with higher density of functional groups increases the crosslink density, thereby increasing Tg;26 however, this effect alone cannot sufficiently explain the observed decrease in Tg at low TEPIC additions. Supporting this interpretation, Downey et al.50 showed that adding an aliphatic trifunctional epoxy without benzene rings to a DGEBA/amine curing agent system led to a decrease in Tg with increasing additive content despite increased crosslinking. Therefore, the mechanism underlying this behavior was further investigated by analyzing temperature-dependent WAXS patterns and their correlation with the simulated internal structures. | ||
| Fig. 2 Thermal properties of each resin. (a) Temperature dependence of the storage modulus (E′) and loss modulus (E″) measured by DMA. Markers on the E″ indicate Tg. (b) Temperature dependence of the thermal strain obtained by TMA. Markers indicate Tg. (c) Tg as a function of TEPIC concentration measured by DMA, TMA, and MD simulations. For the 0 wt% sample, TMA data measured by Odagiri et al.36 are shown. | ||
Furthermore, the DMA results indicate that some compositions exhibit two Tg values. In particular, the system containing 20 wt% TEPIC shows the coexistence of clearly distinguishable rigid and soft regions. Importantly, this behaviour is unlikely to originate from mesoscopic phase separation into DGEBA–rich and TEPIC–rich phases: despite the TEPIC content being at most 30 wt%, the lower-temperature peak significantly weakens when the TEPIC content is increased from 20 to 30 wt%, rather than persisting as would be expected for stable phase coexistence. Instead, the results are consistent with network heterogeneity, i.e., coexistence of (i) three-component regions (DGEBA/TEPIC/4,4′-DDS) where TEPIC effectively contributes to local motion restriction and (ii) two-component regions (DGEBA/4,4′-DDS) where the constraint effect is weaker. This interpretation is further supported by the MD results in the following section, which show the same qualitative tendency without indications of large-scale demixing. Furthermore, separate small-angle X-ray scattering measurements showed no scattering peaks indicative of phase separation.
4.2. Internal structure indicated by WAXS
Fig. 3 shows experimental and MD-simulated WAXS patterns. For all resin systems, the experimental WAXS patterns show four major peaks, which are henceforth called peak 1, peak 2, peak 3, and peak 4, in order from the low-angle side. Fig. 3(c) shows that the MD simulations successfully reproduced three of the four peaks (peaks 2–4) observed in the experimental WAXS patterns. In addition, the simulations captured both the temperature-induced shift of peak 2 and the attenuation of the shoulder with increasing temperature. The good agreement between the experimental and simulated patterns indicates the reliability of the simulated structures for interpreting the molecular origin of peaks 2–4. The absence of peak 1 in the simulated patterns is likely due to the limited simulation box size (approximately 70 × 70 × 70 Å), which is insufficient to represent the larger-scale correlations (approximately 18 Å) that give rise to low-angle scattering. Previous studies have reported that peak 4 originates from the average C–C distance,11 while peaks 2 and 3 correspond to the amorphous halo originating from intermolecular correlations13,17,51 or π–π stacking.11 For the material system employed in this study, peak 1 is thought to originate from the distance between the sulfur atoms of 4,4′-DDS molecules connected by a DGEBA unit (see SI Section S1).Peaks 2 and 3 overlap, making it difficult to directly observe how each peak responds to changes in composition or temperature. Therefore, in this study, peak separation was performed using Gaussian functions in the amorphous halo region, allowing the two peaks to be deconvoluted, as shown in Fig. 4. Fig. 5 shows the temperature dependence of the structural periods calculated from the position corresponding to the peak maximum as d = 2π/q for peaks 2 and 3. Both peaks 2 and 3 exhibit an increase in the corresponding structural periodicity with increasing temperature, and the slope of the temperature dependence shows an inflection point near Tg. This is in good agreement with the linear thermal expansion behavior observed from the TMA measurements, suggesting that these peaks reflect structural information originating from intermolecular correlations. Fig. 6 shows the temperature dependence of the area ratio of peak 3 relative to the total area of the amorphous halo. The area of peak 3 normalized by the total amorphous halo area decreases with increasing temperature. This decrease becomes more pronounced around Tg. This suggests that some of the structures contributing to peak 3 may weaken as molecular mobility increases at higher temperatures.
4.3. π–π stacking
The WAXS results suggest the presence of ordered structures that weaken with increasing temperature. In epoxy systems containing aromatic rings, such thermally unstable ordered structures are often associated with π–π interactions. Therefore, MD simulations were used to examine the presence and quantify the number of π–π stacking pairs for each composition. The structural reliability of the MD models was verified in the previous section through comparison with experimental WAXS. In the present resin system, π–π interactions can occur between benzene rings and between electron-rich benzene rings and electron-deficient triazine rings.52–54For each composition, NPT simulations were performed for 400 ps and trajectories were recorded every 10 ps. From the obtained trajectories, ring pairs were considered to be in a π–π stacked state if they satisfy the criteria of a center-of-mass distance d ≤ 6 Å and an angle α ≤ 30° between the normal vectors of the two ring planes (see Fig. 7). Only pairs that met these criteria throughout the entire 400 ps trajectory were counted as stacked pairs. The analysis results were averaged over N = 10 independent simulations.
Fig. 7 shows the total number of stacking pairs, as well as the numbers of benzene–benzene and benzene–triazine stacking pairs, for each TEPIC content. The number of pairs was normalized by the total number of rings within the simulation box to enable the comparison of systems with different compositional ratios. Fig. 8 shows simulation boxes in which only π–π stacked ring structures are shown for each TEPIC content.
For compositions containing ≥10 wt% TEPIC, both benzene–triazine and benzene–benzene stacking were observed. In contrast, triazine–triazine stacking was not identified based on our criteria for any of the compositions investigated. Interestingly, although the concentration of benzene rings in the system decreases with increasing TEPIC content, the number of benzene–benzene stacking pairs does not decrease. This behavior is attributed to the addition of the relatively short-chain TEPIC molecules, which facilitate configurations in which benzene rings can approach each other more closely, thereby promoting stacking interactions. Fig. 9 shows the distribution of the center-of-mass distances for stacked ring pairs. The stacked-pair center-of-mass distances span a range of 3.4–5.8 Å, which is comparable to the real-space correlation lengths derived from peak 3.
 | ||
| Fig. 9 Histograms of center-of-mass distances for the identified π–π stacking pairs: for (a) 0 wt% and (b) 30 wt% TEPIC systems. | ||
Furthermore, the composition dependence of the total number of stacking pairs reveals an initial decrease from 0 wt% to 5 wt%, followed by a monotonic increase for TEPIC contents of ≥10 wt%. This trend agrees well with the composition dependence of Tg, suggesting that, in addition to network connectivity (crosslink density), π–π stacking provides local intermolecular constraints that contribute to the observed behavior of the Tg of the epoxy network. The minimum Tg observed at 5 wt% TEPIC addition is attributed to the low TEPIC content, which is insufficient to form benzene–triazine stacking or promote closer approaches between benzene rings. Therefore, the introduction of 5 wt% TEPIC simply reduces the number of benzene–benzene stacking interactions.
4.4. Evaluation of π–π interaction energies
Finally, quantum-chemical calculations were performed for benzene–benzene and benzene–triazine ring fragments to evaluate their π–π stacking interaction energies. In Fig. S3 of the SI, benzene–benzene stacking adopts a slipped configuration, whereas benzene–triazine stacking forms a face-to-face configuration. Table 2 summarizes the interaction energies, center-of-mass distances, and configuration for each stacking structure. Benzene–triazine stacking exhibits a stronger interaction energy than benzene–benzene stacking, suggesting that benzene–triazine stacking is more resistant to thermal disruption and more effectively constrains the molecular motion of rings. The stacking distances between ring pairs obtained from quantum chemical calculations are shorter than those obtained from MD simulations. This is attributed to steric hindrance induced by the crosslinked structures in the MD simulations, which prevents the rings from approaching as closely as they would intrinsically prefer.15| Type | Energy [kcal mol−1] | CoM distance [Å] | Configuration |
|---|---|---|---|
| Benzene–benzene | −4.03 | 3.38 | Slip |
| Benzene–triazine | −7.26 | 3.28 | Face-to-face |
The MD simulation results indicate that a higher number of π–π stacking pairs correlates with an increase in Tg. Furthermore, the quantum chemical calculations reveal that benzene–triazine stacking introduced by TEPIC addition suppresses molecular motion more strongly than benzene–benzene stacking. Taken together, these findings suggest that the increase in Tg upon sufficient TEPIC addition is not governed solely by crosslink density, but also by strong, localized intermolecular interactions associated with ring stacking within the network structure.
5. Conclusions
A multicomponent epoxy resin system was investigated in which a triazine-based epoxy resin, TEPIC, was added to a DGEBA/4,4′-DDS system. By combining WAXS, thermomechanical analysis, and MD simulations, the relationship between the internal structure and the glass transition behavior was systematically examined.Experimental thermal characterization revealed a characteristic decrease in Tg with the addition of a low TEPIC concentration, followed by a steady increase in Tg at higher TEPIC contents. This non-monotonic behavior cannot be explained solely by an increase in crosslink density induced by the addition of TEPIC.
The origin of the peaks observed in the WAXS measurements was clarified by comparison with MD simulation results. The amorphous halo originating from intermolecular packing was found to split into two peaks, suggesting that one of these peaks includes a contribution from π–π stacking interactions. Detailed MD simulations revealed that a subset of ring pairs with center-of-mass distances in the range of 3.4–5.8 Å participates in π–π stacking. Furthermore, the total number of such stacking pairs showed a trend consistent with the observed variation in Tg. Accordingly, π–π stacking interactions play a key role in determining the glass transition behavior of the epoxy network.
At high TEPIC contents, stacking between benzene and triazine rings occurs, and quantum chemical calculations revealed that benzene–triazine stacking exhibits stronger stacking energies than benzene–benzene stacking. These strong stacking interactions remain relatively stable against thermal fluctuations and strongly restrict the motion of rings. Such suppression of molecular mobility throughout the network induced by stacking interactions is considered to be responsible for the increase in Tg observed at high TEPIC contents.
In conclusion, the mechanism underlying the enhancement of Tg upon the introduction of triazine rings is attributed to both an increase in crosslink density and the introduction of localized intermolecular constraints arising from stacking interactions. This study presents a molecular-scale mechanism that rationally explains the experimentally observed characteristic Tg behavior and clarifies the role of π–π interactions in determining the thermal properties of epoxy resins. These findings provide a molecular-level design guideline for high-performance epoxy resins.
Author contributions
Yuuki Kinugawa: writing – original draft, writing – review & editing, validation, investigation, formal analysis. Yoshiaki Kawagoe: writing – review & editing, methodology, validation, investigation, formal analysis. Keigo Matsumoto: writing – review & editing, validation, investigation. Masashi Ohno: writing – review & editing, validation, investigation. Shoko Mishima: writing – review & editing, validation, investigation. Takahiko Kawai: writing – review & editing, validation, investigation. Tomonaga Okabe: writing – review & editing, methodology, funding acquisition, supervision.Conflicts of interest
There are no conflicts to declare.Data availability
The code for LAMMPS and curing python code can be found at https://www.lammps.org/#gsc.tab=0 and http://www.cosmic.plum.mech.tohoku.ac.jp/english.html, respectively.The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6cp01039c.
Acknowledgements
This study was partially supported by JST, K Program (Grant Number JPMJKP24W1), and by JSPS KAKENHI (Grant Number 23K25987). Numerical simulations were performed using the AFI-NITY supercomputer system at the Advanced Fluid Information Research Center, Institute of Fluid Science, and the AOBA supercomputer system at the Cyberscience Center, Tohoku University, Japan. We are grateful to the staff of QST NanoTerasu Center and PhoSIC for their support during our experiments.References
- R. Rohini and S. Bose, Phys. Chem. Chem. Phys., 2015, 17, 7907–7913 RSC.
- A. T. Sunny, R. Adhikari, S. Mathew and S. Thomas, et al., Phys. Chem. Chem. Phys., 2016, 18, 19655–19667 RSC.
- K. Ryuzono, S. Koo, Y. Hoshikawa, Y. Kawagoe and T. Okabe, Composites, Part A, 2025, 196, 108963 CrossRef CAS.
- A. Bajpai, J. R. Davidson and C. Robert, Appl. Mech., 2021, 2, 419–430 CrossRef.
- G. Cicala, S. Mannino, A. Latteri, G. Ognibene and G. Saccullo, Adv. Polym. Technol., 2018, 37, 1868–1877 CrossRef CAS.
- J. Anurangi, S. Jayalath, S. R. Senevirathna, M. Herath and J. Epaarachchi, Polym. Compos., 2024, 45, 10898–10910 CrossRef CAS.
- D. J. Hourston, J. M. Lane and H. X. Zhang, Polym. Int., 1997, 42, 349–355 CrossRef CAS.
- Y. Zhao, G. Kikugawa, K. Shirasu, Y. Kawagoe and T. Okabe, Polymer, 2024, 297, 126817 CrossRef CAS.
- X. Zhang, Q. Chi, C. Tang, H. Li, C. Zhang, Z. Li and T. Zhang, J. Mater. Sci.: Mater. Electron., 2023, 34, 638 CrossRef CAS.
- H. Ogawa, M. Aoki, S. Ono, Y. Watanabe, S. Yamamoto, K. Tanaka and M. Takenaka, Langmuir, 2022, 38, 11432–11439 CrossRef CAS PubMed.
- D. B. Dwyer, S. Isbill, Z. E. Brubaker, J. K. Keum, W. Bras and J. L. Niedziela, ACS Appl. Polym. Mater., 2023, 5, 5961–5971 CrossRef CAS.
- J. Liu, X. Liu, X. Cui, J. Qin, M. Wu, L. Fu, M. Shi, D. Wang and L. Liang, Polym. Chem., 2023, 14, 1665–1679 RSC.
- S. Kumar and W. W. Adams, Polymer, 1987, 28, 1497–1504 CrossRef CAS.
- R. Lovell and A. Windle, Polymer, 1990, 31, 593–601 CrossRef CAS.
- S. Yamamoto, N. T. Phan, K. Kihara, A. Shundo and K. Tanaka, Polym. J., 2025, 57, 357–366 CrossRef CAS.
- Q. Yang, X. Yang, X. Li, L. Shi and G. Sui, RSC Adv., 2013, 3, 7452–7459 RSC.
- Y. Kawagoe, Y. Kinugawa, K. Matsumoto, M. Ohno, N. Kishimoto, T. Kawai and T. Okabe, Phys. Chem. Chem. Phys., 2024, 26, 24250–24260 RSC.
- K. Hackenstrass, N. T. Jonasson, M. Hartwig-Nair, T. Rosén, S. Florisson and M. Wohlert, Faraday Discuss., 2026, 263, 52–64 RSC.
- R. Huo, Z. Zhang, N. Athir, Y. Fan, J. Liu and L. Shi, Phys. Chem. Chem. Phys., 2020, 22, 19735–19745 RSC.
- K. Yamaguchi, D. Kawaguchi, N. Miyata, T. Miyazaki, H. Aoki, S. Yamamoto and K. Tanaka, Phys. Chem. Chem. Phys., 2022, 24, 21578–21582 RSC.
- J. Kang, C. Wang, D. Li, G. He and H. Tan, Phys. Chem. Chem. Phys., 2015, 17, 16519–16524 RSC.
- J. Zhu, L. Wang, J. Lin, L. Du and Q. Zhuang, Phys. Chem. Chem. Phys., 2021, 23, 14027–14036 RSC.
- H. Li and W. Zhu, J. Mol. Liq., 2024, 409, 125499 CrossRef CAS.
- J. Li, C. Ren, K. S. Moon and C. P. Wong, 2019 IEEE 69th Electronic Components and Technology Conference (ECTC), 2019, pp. 2296–2301 Search PubMed.
- D. Xie, Q. Pang, Y. Zhao, Y. Li, F. Li and H. He, J. Appl. Polym. Sci., 2022, 139, 52940 CrossRef.
- D. Guzmán, X. Ramis, X. Fernández-Francos and A. Serra, Polymers, 2015, 7, 680–694 CrossRef.
- T. Okabe, Y. Oya, K. Tanabe, G. Kikugawa and K. Yoshioka, Eur. Polym. J., 2016, 80, 78–88 CrossRef CAS.
- Y. Oya, M. Nakazawa, K. Shirasu, Y. Hino, K. Inuyama, G. Kikugawa, J. Li, R. Kuwahara, N. Kishimoto, H. Waizumi, M. Nishikawa, A. Waas, N. Odagiri, A. Koyanagi, M. Salviato and T. Okabe, Chem. Phys. Lett., 2021, 762, 138104 CrossRef CAS.
- S. Maeda and K. Ohno, J. Phys. Chem. A, 2005, 109, 5742–5753 CrossRef CAS PubMed.
- S. Maeda, K. Ohno and K. Morokuma, Phys. Chem. Chem. Phys., 2013, 15, 3683–3701 RSC.
- K. Ohno and S. Maeda, Chem. Phys. Lett., 2004, 384, 277–282 CrossRef CAS.
- K. Ohno and S. Maeda, Phys. Scr., 2008, 78, 058122 CrossRef.
- T. Okabe, T. Takehara, K. Inose, N. Hirano, M. Nishikawa and T. Uehara, Polymer, 2013, 54, 4660–4668 CrossRef CAS.
- Y. Kawagoe and T. Okabe, Comput. Mater. Sci., 2023, 228, 112333 CrossRef CAS.
- T. Watanabe, Y. Kawagoe, K. Shirasu and T. Okabe, Int. J. Solids Struct., 2023, 283, 112489 CrossRef CAS.
- N. Odagiri, K. Shirasu, Y. Kawagoe, G. Kikugawa, Y. Oya, N. Kishimoto, F. S. Ohuchi and T. Okabe, J. Appl. Polym. Sci., 2021, 138, 50542 CrossRef CAS.
- Y. Kinugawa, Y. Kawagoe, K. Oine, K. Ryuzono, Y. Hoshikawa and T. Okabe, Adv. Composite Mater., 2024, 33, 1420–1432 CrossRef.
- Y. Kinugawa, Y. Kawagoe, K. Shirasu, K. Ryuzono and T. Okabe, Int. J. Mech. Sci., 2025, 306, 110779 CrossRef.
- T. Watanabe, Y. Kawagoe, Y. Hoshikawa, Y. Nakai, K. Ryuzono and T. Okabe, Int. J. Solids Struct., 2025, 308, 113130 CrossRef CAS.
- Y. Kawagoe, K. Kawai, Y. Kumagai, K. Shirasu, G. Kikugawa and T. Okabe, Mech. Mater., 2022, 170, 104332 CrossRef.
- S. Plimpton, J. Comput. Phys., 1995, 117, 1–19 CrossRef CAS.
- S. L. Mayo and B. D. Olafson, J. Phys. Chem., 1990, 94, 8897 CrossRef CAS.
- A. K. Rappe and W. A. Goddard III, J. Phys. Chem., 1991, 95, 3358–3363 CrossRef CAS.
- L. Zhang, S. V. Zybin, A. C. V. Duin, S. Dasgupta, W. A. Goddard and E. M. Kober, J. Phys. Chem. A, 2009, 113, 10619–10640 CrossRef CAS PubMed.
- B. Demir and T. R. Walsh, Soft Matter, 2016, 12, 2453–2464 RSC.
- R. W. Hockney and J. W. Eastwood, Computer simulation using particles, CRC Press, 2021 Search PubMed.
- M. Tuckerman, B. J. Berne and G. J. Martyna, J. Chem. Phys., 1992, 97, 1990–2001 CrossRef CAS.
- W. Shinoda, M. Shiga and M. Mikami, Phys. Rev. B: Condens. Matter Mater. Phys., 2004, 69, 134103 CrossRef.
- M. S. Gordon, J. M. Mullin, S. R. Pruitt, L. B. Roskop, L. V. Slipchenko and J. A. Boatz, J. Phys. Chem. B, 2009, 113, 9646–9663 CrossRef CAS PubMed.
- M. A. Downey and L. T. Drzal, Polymer, 2014, 55, 6658–6663 CrossRef CAS.
- K.-F. Lin, W.-Y. Shu and T.-L. Wey, Polymer, 1993, 34, 277–288 CrossRef CAS.
- T. Vialon, H. Sun, G. J. Formon, P. Galanopoulo, C. Guibert, F. Averseng, M. N. Rager, A. Percot, Y. Guillaneuf, N. J. V. Zee and R. NicolaÃ, J. Am. Chem. Soc., 2024, 146, 2673–2684 CrossRef CAS PubMed.
- H. Su, S. A. Jansen, T. Schnitzer, E. Weyandt, A. T. Rösch, J. Liu, G. Vantomme and E. W. Meijer, J. Am. Chem. Soc., 2021, 143, 17128–17135 CrossRef CAS PubMed.
- F. Ugozzoli and C. Massera, CrystEngComm, 2005, 7, 121–128 RSC.
| This journal is © the Owner Societies 2026 |







