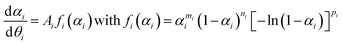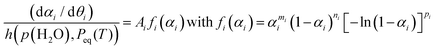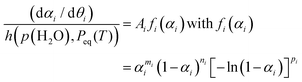Open Access Article
Open Access ArticleMultistep kinetics of the thermal decomposition of β-nickel hydroxide: extraction of the primary reaction steps and the effect of water vapor pressure
Kazuki Arima,
Yuna Kodani,
Mito Hotta and
Nobuyoshi Koga *
*
Department of Science Education, Graduate School of Humanities and Social Sciences, Hiroshima University, 1-1-1 Kagamiyama, Higashi-Hiroshima 739-8524, Japan. E-mail: nkoga@hiroshima-u.ac.jp; Tel: +81-82-424-7092
First published on 16th April 2026
Abstract
The thermal decomposition of nickel hydroxide (Ni(OH)2) was investigated using thermoanalytical techniques with a specific focus on the multistep kinetic behavior and the effect of the partial pressure of water vapor (p(H2O)). The thermal decomposition process was modeled as a four-step kinetic process, comprising the dehydration of absorbed or included water, a two-step primary reaction process yielding nickel oxide (NiO), and the evolution of the trapped water molecules as the crystal growth of NiO progressed. The kinetic characteristics of the individual reaction steps in the primary reaction process were revealed using advanced kinetic analysis methodologies for multistep reactions. A distinctive retardation effect of atmospheric water vapor pressure (p(H2O)ATM) was evidenced by systematically tracing the reaction process at varying p(H2O)ATM values. Combining the kinetic analysis methodologies for multistep reactions and universal kinetic description across different p(H2O) values, the individual reaction steps in the primary reaction process were described as a function of temperature, degree of reaction, and p(H2O)ATM. This approach was further extended to incorporate the effect of the self-generated water vapor pressure, thereby enabling the universal kinetic description covering all kinetic data in a stream of dry and wet N2 gases. The kinetic results indicated the initial reaction step in the primary reaction process as being regulative of the primary reaction process in the context of the physico-geometrical kinetic behavior and the effect of p(H2O). The novel kinetic findings are expected to provide the necessary information to refine the thermal processing of Ni(OH)2, yielding NiO with the desired properties and morphologies.
1. Introduction
Nickel hydroxide (Ni(OH)2) has a variety of applications in modern battery technology.1 The diverse functional properties of this material have significant implications for research in synthesis and characterization. This material is also a precursor to nickel oxide (NiO), which has a variety of applications, including as photocatalysts, gas sensors, electrochromics, and supercapacitors.2–8 Novel physical, chemical, and bio-inspired green mediated processes have been developed for the synthesis of nano-sized and nano-morphological Ni(OH)2 and NiO with enhanced functionalities.1,9–12 The preparation of NiO via the thermal decomposition of nickel compounds is a well-established method. However, it can also be regarded as innovative when a highly reactive precursor with preferable size and morphology is synthesized and the thermal decomposition process is meticulously controlled. It is also noteworthy that the formation of a nickel compound and its thermal decomposition process are relevant component processes in other modern synthetic techniques. Nevertheless, regulation of the thermal decomposition process poses significant empirical challenges in numerous synthetic techniques, necessitating the selection of optimal processing conditions through a process of trial and error, which is generally observed for a range of metal oxide processing techniques.13 Therefore, the kinetic characterization of the thermal decomposition process of the precursor materials poses a continuous challenge to gain further insight into the reaction process controlled by complex physico-geometrical constraints. Furthermore, it is necessary to obtain the relevant information to control the reaction process in order to obtain metal oxides with specific properties for practical applications.The thermal decomposition of Ni(OH)2 with a variety of morphological characteristics has been studied in terms of variations in the reaction behavior and the morphology of the solid product, NiO.14–21
| Ni(OH)2(s) → NiO(s) + H2O(g) | (1) |
Another concern regarding the kinetics of the thermal decomposition of Ni(OH)2 pertains to the effect of the partial pressure of water vapor (p(H2O)), which is involved in the reaction atmosphere (p(H2O)ATM) and is self-generated by the reaction (p(H2O)SG), on the kinetic behavior of the reaction. To the best of our knowledge, available information on the thermal decomposition of Ni(OH)2 is limited. The effect of p(H2O) on the kinetics is unavoidable, particularly in the context of reversible thermal dehydration/dehydroxylation processes.27 Recently, we reported the distinctive retardation effect of p(H2O) on the thermal decomposition of Ca(OH)2, Mg(OH)2, and Cu(OH)2.28–30 The analogous effect of the gaseous species of the product in the reaction atmosphere has been observed for the thermal dehydration of inorganic hydrates31–36 and metal carbonates.33,37,38 Besides, the thermal decomposition of Zn(OH)2 exhibited no distinguishable changes in the kinetic behavior in response to the variation in p(H2O)ATM.39 Therefore, a systematic investigation into the effect of p(H2O) on the thermal decomposition of Ni(OH)2 is imperative to elucidate the kinetic characteristics. Furthermore, the universal kinetic description of the reversible thermal dehydration and dehydroxylation processes as a function of temperature (T), degree of reaction (α), and p(H2O) can be recognized as one of the most distinguishable recent achievements in the field of the kinetics of the thermal decomposition of solids.40,41 Such an advanced kinetic description is expected to be applicable to the thermal decomposition of Ni(OH)2, thereby unveiling hitherto unexplored aspects of the kinetics.
This study demonstrates an advanced kinetic approach to the thermal decomposition of Ni(OH)2 to form NiO, utilizing state-of-the-art methodologies, namely kinetic analysis for the multistep process and universal kinetic description as a function of T, α, and p(H2O). The objective of this study was to reveal the physicochemical and physico-geometrical characteristics of each component reaction step in the multistep reaction, based on the outcomes of these advanced kinetic approaches. Furthermore, it elucidates the effect of p(H2O) on each reaction step, providing a universal kinetic description of the primary thermal decomposition process in the multistep reaction across a range of T and p(H2O) values. The novel physicochemical findings of the kinetics are expected to contribute to further advanced control of the thermal decomposition process of Ni(OH)2, resulting in the formation of NiO with the desired physical properties and morphologies.
2. Experimental
2.1. Sample preparation and characterization
Stock solutions of 0.5 M-Ni(NO3)2(aq) and 1.0 M-KOH(aq) were prepared by dissolving Ni(NO3)2·6H2O (special grade, Sigma-Aldrich Japan) and KOH (special grade, Kishida Chem.) into deionized and distilled water, respectively. In a similar manner, a solution of 10 wt%-NH3(aq) was prepared by diluting 25%-NH3(aq) (special grade, FUJIFILM Wako). 100 cm3 of 0.5 M Ni(NO3)2 was dropwise titrated with 70 cm3 of 10%-NH3(aq) to obtain a solution of [Ni(NH3)4](NO3)2(aq). The ammonia complex solution of Ni2+ was maintained at approximately 323 K, and a total of 100 cm3 of 1.0 M-KOH(aq) was titrated at a rate of 2 cm3 min−1. During the titration process, the solution was subjected to mechanical stirring. The precipitate resulting from the KOH(aq) titration was filtered and subsequently washed repeatedly with deionized and distilled water. The separated precipitate was then dried at 323 K for 24 h in an ambient air atmosphere using an electric oven (DK240S, Yamato). Thereafter, the desiccated precipitate was pulverized using an agate mortar and pestle, and used as the sample for this study. It is acknowledged that, in preparation for the present study, a series of Ni(OH)2 and basic nickel salts were prepared by systematically varying mother liquors and precipitation conditions. The Ni(OH)2 sample prepared utilizing the aforementioned method was selected on the basis of its elevated degree of crystallinity and minimal absorbed water content, in order to facilitate the thermoanalytical tracking of the primary stage of the thermal decomposition process. According to the findings of Ramesh et al.,42,43 Ni(OH)2 with elevated crystallinity has been observed to precipitate within the pH range of ammonia solutions, as observed in this study.The sample was characterized using powder X-ray diffractometry (XRD) and Fourier transform infrared spectroscopy (FTIR). The sample was press-fitted onto a sample holder plate and subsequently subjected to XRD measurement using a diffractometer (RINT-2200V, Rigaku). The XRD pattern was measured by irradiating with Cu-Kα (40 kV, 20 mA) and scanning 2θ values from 5° to 60° at a scanning speed of 4° min−1 in steps of 0.02°. For the FTIR measurement, the sample was diluted with KBr through grinding with an agate mortar and pestle, and the IR spectrum was recorded using a spectrometer (FT-IR 8600, Shimadzu) employing the diffuse reflectance method. For the morphological characterization, surface textures of the sample particles were observed using a scanning electron microscope (SEM, JSM-6510, JEOL) after the sample was coated with a thin Pt layer by sputtering (JFC-1600, JEOL, 30 mA, 30 s). The specific surface area (SBET) was determined by the Brunauer–Emmett–Teller (BET) single point method using a Flow Sorb II 2300 instrument (Micromeritics). The particle size distribution (PSD) was analyzed using an SALD-300 V, Shimadzu, instrument via a laser diffraction method.
2.2. Thermal behavior
The thermal behavior of the Ni(OH)2 sample was traced using simultaneous thermogravimetry (TG)–differential thermal analysis (DTA) coupled with mass spectroscopy (MS) for the evolved gas (TG/DTA–MS) and high temperature XRD (HTXRD). The TG/DTA–MS measurements were conducted with an instrument constructed using a TG–DTA (TG8120, Rigaku) and MS (M200-QA, Anelva), which were connected via a transfer line of the evolved gas. The 5.0 mg sample was weighed into a Pt sample pan (5 mm in diameter and 2.5 mm in depth). The TG–DTA measurement was carried out by heating the sample from 300 to 873 K at a heating rate (β) of 5 K min−1 in a stream of helium at a flow rate (qv) of 200 cm3 min−1. During the TG–DTA measurement, a portion of the outlet gas was continuously transferred to the MS via a silica capillary tube (inner diameter: 75 µm; length: 750 mm) that had been heated at 573 K. The MS measurements were then repeated in a m/z range of 10–50 in the electron impact mode (70 eV) with the secondary electron multiplier (SEM; 1.0 mA, 1.0 kV).For the HTXRD measurements, the sample was press-fitted to a Pt sample holder plate and the XRD patterns were recorded using an RINT-2200V diffractometer by additionally equipping it with a programmable heating chamber. The sample was heated according to two distinct temperature programs in a stream of dry N2 (qv = 100 cm3 min−1). The XRD measurements were performed at various programmed temperatures or times during the execution of these heating programs. In the stepwise isothermal heating mode, the sample was heated at a β of 5 K min−1 from room temperature to 773 K, during which isothermal holding sections for each 15 min were inserted every 25 K in a temperature range from 323 to 773 K. The XRD patterns were recorded during each isothermal holding section. In the isothermal heating mode, the sample was heated to 523 K at a β of 10 K min−1 and maintained at that temperature for 10 h. During the isothermal holding section, diffraction measurements were repeated a total of 40 times. The XRD measurement conditions were identical to those at room temperature for the sample characterization described above.
Approximately 300 mg of the Ni(OH)2 sample was weighed into an alumina boat (SSA-S #6A, length: 100 mm, width: 14 mm, height: 10 mm). Using an electric tube furnace, the sample was heated to various temperatures in a range of 448–773 K at a β of 5 K min−1 in a stream of dry N2 at a qv of 100 cm3 min−1. The heat-treated sample was recovered after cooling to room temperature in the tube furnace, and subsequently subjected to SBET measurement.
2.3. Tracking of the thermal decomposition process in a stream of dry N2
The thermal decomposition process of the Ni(OH)2 sample in a stream of dry N2 was tracked using simultaneous TG–DTA (STA7300, Hitachi High Tech.). Before the measurement of a series of TG–DTA curves for the kinetic calculation, test runs were conducted to select the appropriate measurement conditions including sample mass (m0) and the qv value of dry N2. The sample of varying m0 values (1.0 ≤ m0/mg ≤ 10.0) was weighed into a Pt pan (diameter: 5.0 mm; depth: 2.5 mm), and TG–DTA measurements were carried out during the heating of the sample from 300 to 873 K at a β of 5 K min−1 in a stream of dry N2 (qv = 300 cm3 min−1). In an alternative series of TG–DTA measurements, the m0 value was fixed to be 5.00 ± 0.05 mg and the sample was heated from 300 to 873 K at a β of 5 K min−1 in a stream of dry N2 with varying qv values (50 ≤ qv/cm3 min−1 ≤ 500).Based on the results of the preliminary TG–DTA measurements, the m0 and qv values for the TG–DTA measurement to obtain the kinetic data were fixed to be 5.00 ± 0.05 mg and 300 cm3 min−1, respectively. The TG–DTA measurements were conducted in three different heating program modes including linear nonisothermal, isothermal, and stepwise isothermal modes, in which the stepwise isothermal mode is categorized into the sample controlled thermal analysis technique.24,44 For the TG–DTA measurements under linear nonisothermal conditions, the sample was heated from 300 to 873 K at varying β values (0.5 ≤ β/K min−1 ≤ 10). The TG–DTA measurements under isothermal conditions were conducted by linearly heating the sample from 300 K to different preset temperatures (479 ≤ T/K ≤ 507) at a β of 10 K min−1 and then maintained at these temperatures until the mass loss process was completed. In the measurement of the stepwise isothermal mode, the sample was basically heated from 300 to 873 K at a β of 2 K min−1, whereas, when the mass loss rate reached the programmed threshold value (C), the temperature program mode was switched to the isothermal mode. The opposite was applied when the mass loss rate was reduced to the C value during the isothermal section, switching to the linear nonisothermal mode at a β of 2 K min−1. The measurements in the stepwise isothermal mode were conducted at varying C values in the range of 5–20 µg min−1. It is noteworthy that the repeatability of the TG–DTA curves was confirmed for the measurements at selected measurement conditions for each measurement under linear nonisothermal, isothermal, and stepwise isothermal modes. Furthermore, systematic shifts in the thermoanalytical curves with the heating parameters of β, T, and C were investigated as an indicator of reproducibility.
In the course of the TG–DTA measurements of the Ni(OH)2 sample (m0 = 5.0 mg) under linear nonisothermal conditions at a β of 5 K min−1 in a stream of dry N2 (qv = 300 cm3 min−1), the measurements were terminated at various temperatures in the temperature range of the mass loss process to obtain the partially decomposed sample at different α values. Subsequently, the heat-treated samples were subjected to SEM observations.
2.4. Tracking of the thermal decomposition process in a stream of wet N2
The thermal decomposition process of Ni(OH)2 was also traced in a stream of wet N2 characterized by different p(H2O)ATM values using a humidity-controlled TG (HUM-TG) system, which was constructed with a TG–DTA (TG8120, Rigaku) and a humidity controller (HUM-1, Rigaku). The construction and operation of the HUM-TG system were detailed in some previous publications.35,36 Using the 5.0 mg sample weighed into a Pt pan (diameter: 5.0 mm; depth: 2.5 mm), a series of TG–DTA measurements were conducted in the linear nonisothermal mode at a fixed β of 5 K min−1 in a stream of wet N2 (qv = 300–400 cm3 min−1) with varying p(H2O)ATM values in a range of 0.8–10.2 kPa. Notably, the qv value of the wet N2 flow was varied among the measurements depending on the controlled p(H2O)ATM value of the wet N2, in order to regulate the p(H2O)ATM value by mixing a wet and dry N2 flow at different flow rates. Furthermore, by selecting three different p(H2O)ATM values of 0.8, 3.6, and 9.2 kPa as the reaction condition, kinetic data under different temperature conditions were obtained using the TG–DTA measurements in the linear nonisothermal mode at varying β values and the isothermal mode at varying preset temperatures at each p(H2O)ATM value. In the linear nonisothermal mode, the sample was heated from 300 to 973 K at different β values (0.5 ≤ β/K min−1 ≤ 10). In the isothermal mode, the sample was heated to different preset temperatures at a β of 10 K min−1 and subsequently maintained at that temperature until the reaction was complete. The temperature range of the measurements in the isothermal mode was varied with the p(H2O)ATM value, which was 504 ≤ T/K ≤ 523, 512 ≤ T/K ≤ 532, and 516 ≤ T/K ≤ 535 for the measurements in a stream of wet N2 with p(H2O)ATM values of 0.8, 3.6, and 9.2 kPa, respectively.3. Results and discussion
3.1. Sample characterization
Fig. S1 shows the XRD pattern and FTIR spectrum of the sample. The XRD pattern agreed with that of β-Ni(OH)2 (hexagonal, S.G. = P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1(164), a = b = 0.31300 nm, c = 0.46300 nm, α = β = 90.000°, γ = 120.000°, ICDD 01-074-2075).45 The FTIR spectrum exhibited specific IR absorption peaks at 3639, 542, and 459 cm−1, which are attributed to the O–H stretching of non-hydrogen bonded OH groups, an in-plane bending of Ni–O–H, and Ni–O stretching, respectively.46,47 Fig. S2 shows the SEM image of the Ni(OH)2 sample. The sample was characterized as an agglomerate of nano-micrometer sized particles with the SBET value of 28.0 ± 0.1 m2 g−1. The sample particles exhibited a size distribution (Fig. S3), which was characterized by the average diameter size of 0.685 µm with the standard deviation of 0.274 in logarithmic scale along the log normal distribution function.
m1(164), a = b = 0.31300 nm, c = 0.46300 nm, α = β = 90.000°, γ = 120.000°, ICDD 01-074-2075).45 The FTIR spectrum exhibited specific IR absorption peaks at 3639, 542, and 459 cm−1, which are attributed to the O–H stretching of non-hydrogen bonded OH groups, an in-plane bending of Ni–O–H, and Ni–O stretching, respectively.46,47 Fig. S2 shows the SEM image of the Ni(OH)2 sample. The sample was characterized as an agglomerate of nano-micrometer sized particles with the SBET value of 28.0 ± 0.1 m2 g−1. The sample particles exhibited a size distribution (Fig. S3), which was characterized by the average diameter size of 0.685 µm with the standard deviation of 0.274 in logarithmic scale along the log normal distribution function.
3.2. Thermal behavior
Fig. 1 shows the results of TG/DTA–MS measurements. The TG curve demonstrated a modest mass loss below 400 K, a pronounced mass loss between 450 and 570 K, and a gradual mass loss extending to approximately 750 K. The primary mass loss process observed between 450 and 570 K was accompanied by a significant endothermic effect as observed in the DTA curve. Only the evolution of water vapor was detected by the peak of m/z 18 (H2O+) and its fragments in the MS thermograms during the entire temperature range of the TG/DTA–MS measurement. | ||
| Fig. 1 Results of TG/DTA–MS measurement for the thermal decomposition of Ni(OH)2 (m0 = 5.00 mg) at a β of 5 K min−1 in a stream of He at a qv of 200 cm3 min−1. | ||
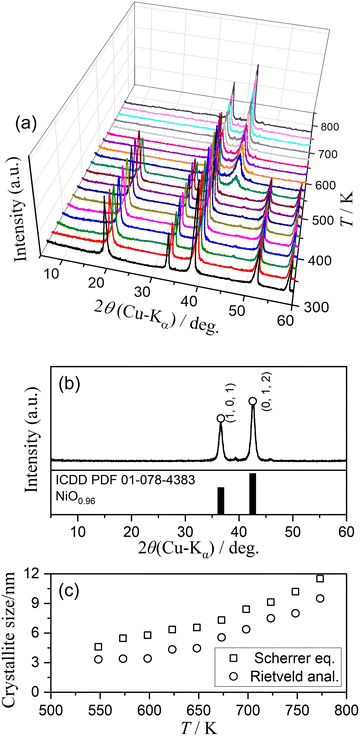
Fig. 2 shows the results of HTXRD measurements during heating the sample in the stepwise isothermal mode at a β of 5 K min−1 with isothermal holding sections at various temperatures in a stream of dry N2 (qv = 100 cm3 min−1). The XRD pattern of the original sample was maintained up to 523 K (Fig. 2(a)). Therefore, the initial mass loss observed below 400 K can be attributed to the dehydration of absorbed or included water in the original Ni(OH)2 sample. The change in the XRD pattern was observed in the temperature range from 523 to 573 K, which covers the temperature range of the primary mass loss step of the thermal decomposition observed in TG. The product phase was identified as NiO (Fig. 2(b); trigonal, S.G.: R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m(166), a = b = 0.29895 nm, c = 0.73210 nm, α = β = 90.000°, γ = 120.000°, ICDD 01-078-4383).48 After the transformation, the intensity of the XRD peaks attributed to NiO increased gradually as the temperature increased from 573 to 773 K. This was accompanied by the gradual increase in the crystallite size of NiO during heating from 573 to 773 K (Fig. 2(c)). It was thus anticipated that the gradual mass loss observed in the corresponding temperature range of the growth of XRD peaks of NiO can be attributed to the removal of the water molecules trapped in the NiO particles and these agglomerates. As illustrated in Fig. S4, at a constant temperature of 523 K, the gradual attenuation of the XRD peaks of Ni(OH)2 and compensative growth of the XRD peaks of NiO occurred without exhibiting any evidence of the formation of intermediate crystalline solids. The crystallite size of NiO obtained by the isothermal treatment for 600 min was calculated to be 3.2 nm using the Rietveld refinement analysis, which was slightly smaller than that calculated for the NiO at 548 K during the stepwise isothermal heating (Fig. 2(c)). As illustrated in Fig. S5, the SBET value of the sample exhibited a substantial increase to 146.0 m2 g−1 within the temperature range of the primary thermal decomposition process, i.e., 523–573 K, followed by a decline upon further heating to 773 K. However, the appearance of the sample particles as an agglomerate of nanoparticles remained relatively unchanged during the thermal decomposition process, as shown in Fig. S6. The findings indicated that the primary mass loss process occurring within the temperature range from 523 to 573 K can be treated as the thermal decomposition process of Ni(OH)2 to form NiO.
m(166), a = b = 0.29895 nm, c = 0.73210 nm, α = β = 90.000°, γ = 120.000°, ICDD 01-078-4383).48 After the transformation, the intensity of the XRD peaks attributed to NiO increased gradually as the temperature increased from 573 to 773 K. This was accompanied by the gradual increase in the crystallite size of NiO during heating from 573 to 773 K (Fig. 2(c)). It was thus anticipated that the gradual mass loss observed in the corresponding temperature range of the growth of XRD peaks of NiO can be attributed to the removal of the water molecules trapped in the NiO particles and these agglomerates. As illustrated in Fig. S4, at a constant temperature of 523 K, the gradual attenuation of the XRD peaks of Ni(OH)2 and compensative growth of the XRD peaks of NiO occurred without exhibiting any evidence of the formation of intermediate crystalline solids. The crystallite size of NiO obtained by the isothermal treatment for 600 min was calculated to be 3.2 nm using the Rietveld refinement analysis, which was slightly smaller than that calculated for the NiO at 548 K during the stepwise isothermal heating (Fig. 2(c)). As illustrated in Fig. S5, the SBET value of the sample exhibited a substantial increase to 146.0 m2 g−1 within the temperature range of the primary thermal decomposition process, i.e., 523–573 K, followed by a decline upon further heating to 773 K. However, the appearance of the sample particles as an agglomerate of nanoparticles remained relatively unchanged during the thermal decomposition process, as shown in Fig. S6. The findings indicated that the primary mass loss process occurring within the temperature range from 523 to 573 K can be treated as the thermal decomposition process of Ni(OH)2 to form NiO.
3.3. Thermal decomposition in a stream of dry N2
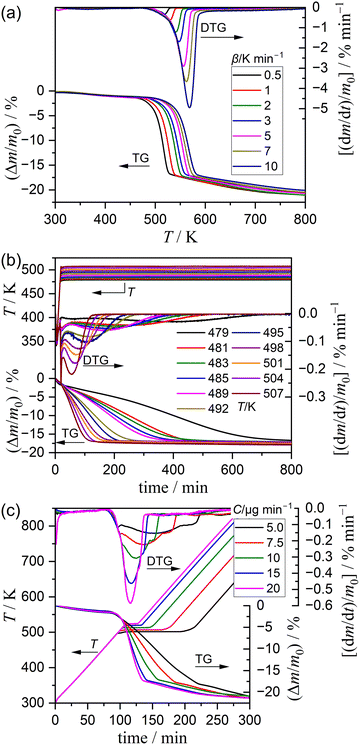
Fig. S7 and S8 illustrate the variations of the TG–derivative TG (DTG)–DTA curves with the measurement parameters of m0 and qv, respectively. The thermoanalytical (TA) curves exhibited systematic shifts to higher temperatures with increasing m0 (Fig. S7). The observed phenomena indicated that the reaction rate is retarded by the effects of self-generated water vapor and self-cooling. As m0 increases, the thickness of the sample bed also increases, thereby hindering diffusional removal of water vapor evolved by the reaction in the bed. Under such conditions, the reaction progresses from the upper surface of the sample bed to the bottom.49–51 Additionally, the self-cooling effect, attributable to the endothermic thermal decomposition, results in a deviation of the sample temperature variation from the linearly increasing temperature that has been programmed for the measurement.52,53 This effect is particularly pronounced for the larger m0 value. Conversely, the qv value demonstrated no significant impact on the TA curves, as demonstrated in Fig. S8. This observation indicates that the inert gas flow may not be adequately effective in removing the evolved water vapor from the sample bed in this reaction. While recognizing the inevitable influence of mass and heat transfer phenomena on the experimental TA curves, the m0 and qv values for the TG–DTA measurements in a stream of dry N2 were selected to be 5.00 mg and 300 cm3 min−1, respectively, by considering the stability and signal/noise ratio of the measured TA curves.Fig. 3 shows the TG–DTG or TG–DTG–temperature profile curves for the thermal decomposition of Ni(OH)2 in a stream of dry N2, recorded with different temperature program modes. Under linear nonisothermal conditions (Fig. 3(a)), the TG–DTG curves exhibited the three distinguishable mass loss steps. The initial step with slight mass loss (Δm1 = 1.2 ± 0.3%) was followed by a primary mass loss step (Δm2 = 16.1 ± 0.4%), and a subsequent step with slow mass loss (Δm3 = 3.3 ± 0.3%). The total mass loss of 20.6 ± 0.4% observed during the heating of the sample from 300 to 873 K was slightly larger than the calculated mass loss value of 19.4% according to eqn (1). The sum of the mass loss ratios attributed to the second and third mass loss steps, with reference to the sample mass observed after the first mass loss process was completed (m0′ = m0 − Δm1), was calculated to be 19.6 ± 0.4%, corresponding well with the calculated value for the thermal decomposition of Ni(OH)2. An increase in β caused a systematic shift in the TG–DTG curves of the primary mass loss step to higher temperatures. In contrast, the initial mass loss step did not demonstrate the variation with β value. The third mass loss step exhibited a shift of the TG–DTG curves to higher temperatures with increasing β values. However, the shift width was found to be more restricted in comparison with that observed for the second mass loss step.
The TG–DTG curves recorded in the isothermal mode (Fig. 3(b)) exhibited two mass loss steps. The initial mass loss step (Δm1 = 1.2 ± 0.2%) occurred during the linear heating of the sample from 300 K to the preset temperature for the subsequent isothermal holding. The second mass loss step was observed under isothermal conditions (Δm2 = 15.9 ± 0.5%). The mass loss ratios of the first and second mass loss steps were consistent with those observed under linear nonisothermal conditions. The third mass loss step observed in the linear nonisothermal mode did not occur under isothermal conditions examined in this study, due to the limited crystal growth of NiO at these temperatures. It is imperative to note that the presence of the two reaction stages was anticipated during the second mass loss step under isothermal conditions, evidenced by an inflection in the mass loss curve midway through the TG curves and two distinguishable DTG peaks appeared during the second mass loss step. The TG–DTG curves during the second mass loss process demonstrated a systematic prolongation along the time axis with decreasing T value. The TG–DTG curves recorded in the stepwise isothermal mode (Fig. 3(c)) clearly represented three mass loss steps occurring during the initial temperature increasing, isothermal holding, and subsequent temperature increasing sections. The total mass loss ratio of 21.1 ± 0.5% was equivalent to that observed under linear nonisothermal conditions within the standard deviation. The mass loss ratios of the individual mass loss steps also corresponded to those determined under linear nonisothermal conditions: Δm1 = 1.4 ± 0.2%, Δm2 = 16.2 ± 0.4%, and Δm3 = 3.4 ± 0.1%. The temperature profile during the second mass loss step systematically shifted to higher temperatures with increasing C value. The DTG curves under stepwise isothermal conditions also indicated the presence of two reaction stages in the second mass loss step, which exhibited two distinguishable DTG peaks.
In summary, the thermal decomposition of Ni(OH)2 occurred via a three-step mass loss process under linear nonisothermal and stepwise isothermal conditions. A similar mass loss behavior comprising the initial step of the dehydration of absorbed or included water molecules in the Ni(OH)2 agglomerate, followed by the primary step of the thermal decomposition of Ni(OH)2 and subsequent evolution of trapped water accompanied by the crystal growth of NiO has generally been observed for the thermal decomposition of Ni(OH)2 samples with various morphological characteristics, reported previously.14–21 In order to grasp the multistep thermal decomposition behavior from the kinetic standpoint, the overall thermal decomposition process including three mass loss steps was subjected to the isoconversional kinetic analysis using the Friedman plot.54 For kinetic analysis, the TG–DTG curves recorded under linear nonisothermal and stepwise isothermal conditions were converted to the kinetic data of a series of data points comprising time (t), T, α, and the normalized reaction rate (dα/dt), for which the α value was calculated with reference to the total mass loss of the three-step process.
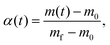 | (2) |
The procedure and results of the isoconversional kinetic analysis as a preliminary kinetic approach are detailed in Section S2-3 of the SI, with the Friedman plots at various α values represented in Fig. S9. In summary, the preliminary kinetic analysis suggested that the thermal decomposition of Ni(OH)2 is composed of four partially overlapping reaction steps, including the desorption of absorbed or included water molecules, the thermal decomposition of Ni(OH)2 via two reaction steps, and the evolution of the trapped water molecules in the NiO product. In addition, it was confirmed that the kinetic data obtained from the TG–DTG curves provided relevant kinetic relationships for the primary mass loss step, thereby, it is interpreted that the primary mass loss step is the actual thermal decomposition process of Ni(OH)2.
Consequently, the extraction of the kinetic data for the primary mass loss step, composed of two reaction steps, from the overall kinetic data was subjected as the subsequent kinetic approach. The DTG curves recorded under linear nonisothermal conditions were deconvoluted into individual peaks corresponding to the component reaction steps using statistical functions (F(t)) (mathematical deconvolution analysis (MDA)25,55–58).
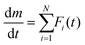 | (3) |
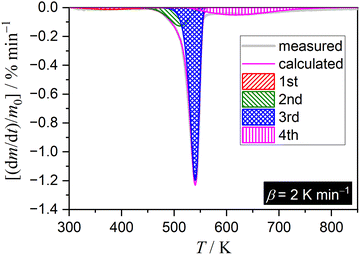 | ||
| Fig. 4 Typical fitting result of the overall DTG curve for the thermal decomposition of Ni(OH)2 at a β of 2 K min−1 in a stream of dry N2 via MDA using the Weibull function. | ||
In addition, each series of DTG peaks of individual reaction steps at varying β values were obtained as the result of MDA (Fig. S12). In each reaction step, the kinetic curves shifted systematically to higher temperatures with increasing β value. The kinetic data for individual reaction steps were formally analyzed via the isoconversional method (Friedman plot)54 and subsequent master plot method.59–64 The formal kinetic analysis procedures and results are detailed in Section S3-4 of the SI. In summary, the primary reaction process composed of the second and third reaction steps was characterized by individual constant Ea values during each reaction step, while the first and fourth reaction steps exhibited a systematic variation in Ea value as the reaction progressed (Fig. S13). The experimental master plots for the second and third reaction steps exhibited the maximum reaction rate midway through each reaction step. In contrast, the rate behavior of the first and fourth reaction steps was characterized by experimental master plots with a concave deceleration curve, indicative of a diffusion-controlled process. These observations for the first and fourth reaction steps are explainable when the reaction steps are regarded as the removal of included or trapped water molecules from the Ni(OH)2 and NiO product. The kinetic parameters for each reaction step determined by the formal kinetic analysis for the kinetic data separated via MDA are enumerated in Table S1.
The kinetic parameters for each reaction step determined by the formal kinetic analysis of the kinetic data obtained via MDA (Table S1) were further optimized based on the cumulative kinetic equation (kinetic deconvolution analysis (KDA)25,55,56,65).
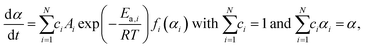 | (4) |
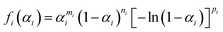 | (5) |
The SB model with three kinetic exponents was utilized to ensure the precise extraction of the kinetic curves for each component reaction step. This model demonstrates a higher degree of flexibility in comparison with the truncated SB model that incorporates two kinetic exponents. The kinetic parameters listed in Table S1 were set using eqn (4) as the initial values. Subsequently, all kinetic parameters were simultaneously optimized to minimize the square sum of the residues when fitting the overall experimental kinetic curve with the calculated curve.
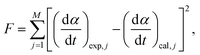 | (6) |
The typical results of KDA are illustrated in Fig. 5. Initially, all kinetic parameters for individual reaction steps under linear nonisothermal conditions at various β values were optimized via KDA, yielding a statistically significant fit to the experimental curves, irrespective of β (Fig. 5(a) and Fig. S14). The optimized kinetic parameters for each experimental kinetic curve were practically invariant among those at different β values. The average values of the optimized kinetic parameters are enumerated in Table S2. Subsequently, the experimental kinetic curves under isothermal and stepwise isothermal conditions were also subjected to KDA using the optimized kinetic parameters for the reactions under linear nonisothermal conditions (Table S2) as the initial values. Notably, the experimental kinetic curves under isothermal conditions were analyzed as composed of the partially overlapping three reaction steps, because the fourth reaction step observed under linear nonisothermal and stepwise isothermal conditions was not traced under isothermal conditions (Fig. 3). The experimental kinetic curves under isothermal conditions were successfully fitted via KDA as the three-step process irrespective of the measurement temperature (Fig. 5(b) and Fig. S15). The optimized kinetic parameters at each constant temperature exhibited minimal variations with the measurement temperature, indicating a limited value of standard deviation for the average values (Table S3). In addition, the optimized kinetic parameters for the first to third reaction steps under isothermal conditions were comparable to those under linear nonisothermal conditions. The analogous KDA results were attained for the four-step thermal decomposition process under stepwise isothermal conditions at various C values (Fig. 5(c) and Fig. S16 and Table S4). The KDA results demonstrate that the kinetic behavior of the individual reaction steps of the overall thermal decomposition of Ni(OH)2 remains constant across a range of heating conditions and program modes.
The refinement of the multistep kinetic description via KDA for the reactions under different heating modes provided us with a series of kinetic curves for the second and third reaction steps that constitute the primary reaction process of the thermal decomposition. The resulting kinetic curves are illustrated in Fig. S17 and S18 for the second and third reaction steps, respectively. The c2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c3 ratios of the extracted kinetic curves were determined to be 0.18 ± 0.02
c3 ratios of the extracted kinetic curves were determined to be 0.18 ± 0.02![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.82 ± 0.02. Each separated reaction step was expected to be able to be described as the single-step kinetic process controlled by a rate-determining step with a specific physico-geometrical constraint. Therefore, the formal kinetic approach based on the isoconversional and master plot methods was reexamined for the second and third reaction steps over different heating modes including linear nonisothermal, isothermal, and stepwise isothermal modes. The results of the formal kinetic analysis are demonstrated in Fig. 6. Based on the fundamental kinetic equation (eqn (S1)), the Friedman plots at various αi values were examined for each reaction step i.
0.82 ± 0.02. Each separated reaction step was expected to be able to be described as the single-step kinetic process controlled by a rate-determining step with a specific physico-geometrical constraint. Therefore, the formal kinetic approach based on the isoconversional and master plot methods was reexamined for the second and third reaction steps over different heating modes including linear nonisothermal, isothermal, and stepwise isothermal modes. The results of the formal kinetic analysis are demonstrated in Fig. 6. Based on the fundamental kinetic equation (eqn (S1)), the Friedman plots at various αi values were examined for each reaction step i.
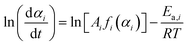 | (7) |
For each reaction step characterized by the individual Ea,i values, the hypothetical reaction rate (dαi/dθi) at infinite temperature can be calculated at various αi values.59–64
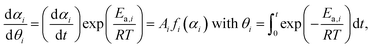 | (8) |
| Reaction step, i | ci/(c2 +c3)a | Ea,i/kJ mol−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b b |
|||||
|---|---|---|---|---|---|---|---|
| Ai/s−1 | mi | ni | pi | R2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
|||
| a Averaged over different heating conditions.b Averaged over 0.10 ≤ αi ≤ 0.90.c Determination coefficient of the nonlinear least-squares analysis. | |||||||
| 2 | 0.18 ± 0.02 | 115.3 ± 3.7 | (1.37 ± 0.01) × 109 | 1.89 ± 0.03 | 0.60 ± 0.01 | –1.69 ± 0.03 | 0.9999 |
| 3 | 0.82 ± 0.02 | 131.4 ± 1.9 | (1.93 ± 0.01) × 1010 | –0.14 ± 0.01 | 0.92 ± 0.01 | 0.63 ± 0.01 | 0.9999 |
As illustrated in Fig. S19, the experimental master plots of individual reaction steps were reanalyzed using physicochemical or physico-geometrical kinetic model functions. The experimental master plot for the second reaction step exhibited linear deceleration behavior after the brief initial acceleration stage. The observed rate behavior corresponds to the first-order kinetic model (F1): f(α) = 1 − α (Fig. S19(a)). Because the second reaction step is characterized as the initial stage of the primary thermal decomposition process with a limited contribution (c2/(c2 + c3) = 0.18), the kinetic obedience to F1 can be interpreted as the consumption of the selected reactive sites of the sample, which are presumably presented on the surface of the Ni(OH)2 agglomerates. Besides, the experimental master plot of the third reaction step, which is the major step of the primary thermal decomposition process (c3/(c2 + c3) = 0.82), exhibited the maximum reaction rate midway through the reaction step. Assuming homogeneous-like rate behavior, the rate behavior comprising the initial acceleration and subsequent deceleration is explained by an autocatalytic reaction as expressed by the extended Prout–Tompkins model (ePT(m, n)).69
| ePT(m, n): f(α) = αm(1 − α)n | (9) |
The experimental master plot of the third reaction step was satisfactorily fitted with ePT(0.52, 0.70) (Fig. S19(b)), which is comparable to SB(m, n, p). Considering heterogeneous characteristics of the physico-geometrical constrains of the reaction mechanisms, two distinct models are worth examination to describe the rate behavior with the maximum reaction rate occurring midway through the reaction. One is the nucleation–growth-type model described by the Johnson–Mehl–Avrami–Erofeev–Kolmogorov model (JMA(m)).70–75
| JMA(m): f(α) = m(1 − α)[−ln(1 − α)]1−1/m | (10) |
| GH(n): f(α) = 2n(1 − α)1−1/n[1 − (1 − α)1/n]1/2 | (11) |
3.4. Thermal decomposition in a stream of wet N2 with various atmospheric water vapor pressures
As illustrated in Fig. 7, the primary mass loss process of the thermal decomposition of Ni(OH)2 is influenced by p(H2O)ATM. The TG–DTG curves under linearly increasing temperature conditions exhibited a systematic shift to higher temperatures with increasing p(H2O)ATM value, indicating the retardation effect due to p(H2O). It is important to note that this phenomenon is characteristic of the reversible thermal decomposition process. In the context of thermal decomposition processes of this nature, a more pronounced shift in the thermoanalytical curves towards higher temperatures with an increase in p(H2O)ATM is generally observed, particularly within a reduced p(H2O) range. The retardation effect of p(H2O) on the kinetics of the thermal decomposition of solids should be considered with reference to the equilibrium pressure of the gaseous product (Peq(T)). The changes in the Peq(T) value with temperature are demonstrated in Fig. S20, which was calculated using the literature values of the thermodynamic parameters of the reaction system.82 The extended kinetic equation that describes the reaction as a function of T, α, and p(H2O) has been obtained by introducing an accommodation function (AF; h(p(H2O), Peq(T))) into the fundamental kinetic equation.29,40,41
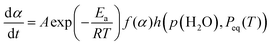 | (12) |
Fig. 8 shows the TG–DTG curves recorded under linear nonisothermal conditions at different β values and isothermal conditions at different T values in a stream of wet N2 with a p(H2O)ATM value of 3.6 kPa. The analogous TG–DTG curves at p(H2O)ATM values of 0.8 and 9.2 kPa are represented in Fig. S21 and S22, respectively. Irrespective of the p(H2O)ATM value, the TG–DTG curves exhibited three mass loss processes and systematically shifted to higher temperatures with increasing β value ((a) in Fig. 8 and Fig. S21 and S22). In the measurement using the isothermal mode, the initial mass loss step was observed during linear heating to the preset isothermal temperature. Thereafter, the primary mass loss process occurred at constant temperature ((b) in Fig. 8 and Fig. S21 and S22). The mass loss rate of the primary mass loss process increased systematically with increasing T. The mass loss behavior and its variation with the measurement temperature conditions were comparable to those previously observed for the reaction in a stream of dry N2 (Fig. 3).
It is important to note that the primary mass loss process occurred at significantly higher temperatures and at lower p(H2O) values with reference to the Peq(T) curve (Fig. S20), calculated according to eqn (13).
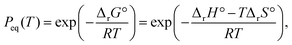 | (13) |
The extracted kinetic data of the second and third reaction steps under linear nonisothermal and isothermal conditions at each p(H2O)ATM value were simultaneously subjected to formal kinetic analysis, in which the effect of p(H2O) was ignored. The results are detailed in Section S4-3 of the SI, with Fig. S35, S36, and Table S8 for the second reaction step and Fig. S37, S38, and Table S9 for the third reaction step. Irrespective of the reaction steps, the formal kinetic analysis yielded apparent kinetic parameters that were distinct for the reactions at different p(H2O)ATM values. Consequently, the consideration of AF in eqn (12) is necessary for a universal kinetic description across different p(H2O)ATM values. Recently, an analytical form of AF was derived based on the classical theory of surface and interfacial processes, which comprise consecutive/concurrent elementary steps, while assuming one rate-determining step and steady-state conditions for the other steps.29,40,41
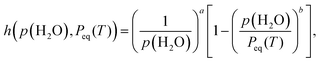 | (14) |
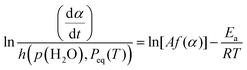 | (15) |
Initially, the extended Friedman plot was applied to each reaction step by assuming (a, b) = (0, 1) and (1, 1), while the p(H2O)ATM value was set for p(H2O). In both the second and third reaction steps, the results did not reveal a universal isoconversional relationship, yielding individual linear correlations at different p(H2O)ATM values (Fig. S39 and S40, respectively). Therefore, the most appropriate a = b value was optimized through the extended Friedman plot. The results of extended kinetic analysis with the optimized a = b are illustrated in Fig. 9. In both reaction steps, a universal isoconversional relationship was established with the optimized a = b values of 0.53 and 0.29 for the second and third reaction steps, respectively (Fig. 9(a) and (b)). These universal isoconversional relationships were established at various αi values (Fig. S41). Theoretically, the AF with a = b values has been derived for the surface and interfacial processes controlled by the formation of the product crystalline phase.29,40,41 However, the slope of the extended Friedman plot varied as the reaction progressed in each step. In the second reaction step, the Ea,2 value initially increased and subsequently converged to a constant value in the major reaction stage (Fig. 9(c)). In contrast, a gradual decline of the Ea,3 value was observed as the reaction progressed in the third reaction step; however, the observed variation was limited within 10 kJ mol−1 (Fig. 9(d)).
Assuming that the average Ea,i value over the major reaction stage (0.10 ≤ αi ≤ 0.90) is representative for the reaction across different p(H2O) values, the extended experimental master plot can be constructed by calculating (dαi/dθi)/h(p(H2O), Peq(T)) values at various αi values.32,33,37,38,40,41
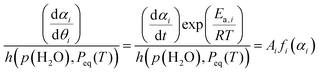 | (16) |
The extended experimental master plot for the second reaction step exhibited the maximum reaction rate at the initial stage of the reaction step and subsequently decelerated as the reaction progressed (Fig. 9(c)). The shape of the experimental master plot resembles that of the second reaction step in a stream of dry N2 (Fig. 6(c)); however, the deceleration stage is characterized by a convex shape. The extended experimental master plot for the third reaction step exhibited the maximum reaction rate midway through the reaction at α3 = 0.37 (Fig. 9(d)). The rate behavior comprising the initial acceleration and subsequent deceleration is equivariant to that in a stream of dry N2 (Fig. 6(d)). The extended experimental master plots were almost perfectly fitted using the SB(m, n, p) function, yielding constant Ai values for individual reaction steps.
Table 2 enumerates the apparent kinetic parameters for the second and third reaction steps determined in the context of universal kinetic description across different p(H2O)ATM values with the optimized a = b values. As was observed by the comparison of the shapes of the conventional and extended experimental master plots, the rate behavior under isothermal conditions, characterized by the kinetic exponents in SB(m, n, p), exhibited a trend resembling that between the reactions in a stream of dry and wet N2 in each reaction step. On the other hand, the apparent Arrhenius parameters determined by considering the effect of p(H2O)ATM were significantly larger than those determined by the conventional kinetic analysis for the reactions in a stream of dry N2 (Table 1). One characteristic of the extended Friedman plot should be addressed to correlate the apparent Arrhenius parameters (Ea, A) to the intrinsic Arrhenius parameters (Ea,int, Aint). The extended Friedman plot simultaneously examines the temperature dependences of reaction rate and Peq(T), which are explained by the Arrhenius and van’t Hoff equations, respectively. Comparing the kinetic equations of the conventional and extended Friedman plots (eqn (7) and (15)), the following relationships between the apparent Arrhenius parameters determined based on the extended kinetic equation (eqn (12)) and the intrinsic Arrhenius parameters were derived.31,32,40,41
| Ea,int ≈ Ea − bΔrH° | (17) |
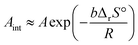 | (18) |
| Reaction step, i | a = b | Ea,i/kJ mol−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a a |
Intrinsic Arrhenius parametersa | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Ai/s−1 | mi | ni | pi | R2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b b |
Ea,i,int/kJ mol−1 | Ai, int/s−1 | |||
| a Averaged over 0.10 ≤ αi ≤ 0.90.b Determination coefficient of the nonlinear least-squares analysis. | |||||||||
| 2 | 0.53 | 209.5 ± 4.6 | (1.38 ± 0.01) × 1017 | 0.42 ± 0.03 | 0.78 ± 0.01 | –0.24 ± 0.03 | 0.9999 | 184.2 ± 4.6 | (2.12 ± 0.02) × 1013 |
| 3 | 0.29 | 183.4 ± 2.0 | (4.93 ± 0.01) × 1014 | 0.54 ± 0.03 | 0.93 ± 0.01 | 0.01 ± 0.03 | 0.9999 | 169.3 ± 2.0 | (3.64 ± 0.03) × 1012 |
The intrinsic Arrhenius parameters, calculated according to eqn (17) and (18) using literature values of 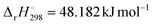 and
and 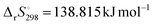 for the thermal decomposition of Ni(OH)2,82 exhibited a reduction of the values from the apparent values in both reaction steps, as listed in Table 2. However, the intrinsic Arrhenius parameters were still larger than those determined for the reactions in a stream of dry N2 (Table 1).
for the thermal decomposition of Ni(OH)2,82 exhibited a reduction of the values from the apparent values in both reaction steps, as listed in Table 2. However, the intrinsic Arrhenius parameters were still larger than those determined for the reactions in a stream of dry N2 (Table 1).
3.5 Universal kinetic description considering atmospheric and self-generated water vapor pressures
Another issue that must be addressed to achieve a rigorous kinetic description is the effect of p(H2O)SG on the kinetic behavior, as expected from the systematic shift in the TG–DTG curves with varying m0 values. This issue is relevant to both the kinetic results for the reactions in a stream of dry and wet N2 gases, as described in the previous sections. However, the p(H2O)SG value during the thermal decomposition process is unmeasurable. Recently, an empirical procedure was examined to consider both p(H2O)SG and p(H2O)ATM in the universal kinetic description across different T and p(H2O) values. The effective p(H2O)EF is empirically defined as the sum of the contributions of p(H2O)SG and p(H2O)ATM, where the p(H2O)SG value is assumed to be proportional to the reaction rate.33,37,40,41
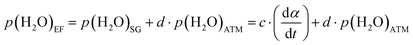 | (19) |
Fig. S42 and S43 demonstrate the extended kinetic analysis considering both p(H2O)SG and p(H2O)ATM, as applied to the second and third reaction steps across various p(H2O)ATM values. In both reaction steps, an enhancement of the linearity of the extended Friedman plots was accomplished through the optimization of the coefficients (c, d) (Fig. S42(a) and S43(a)). The linearity of the extended Friedman plots was guaranteed at various αi values (Fig. S42(b) and S43(b)). The d values for p(H2O)ATM optimized to achieve the best linearity of the plot were approximately unity during each reaction step, while the optimized c values for p(H2O)SG varied as the reaction progressed in both reaction steps (Fig. S42(c) and S43(c)). However, the variation trends of the c value were distinct between the second and third reaction steps.
In the second reaction step, the optimized c value was negligible in the initial stage and subsequently increased exponentially in the latter stage (Fig. S42(c)). The variation trend of the c value indicates that the effect of p(H2O)SG is negligible in the initial stage of the second reaction step, but increases in the latter stage. The anticipated change in the effect of p(H2O)SG as the reaction progressed is explainable considering the physico-geometrical constraint of the surface reaction process, for which the initial reaction site is on the surface of the reactant particles, where the evolved water vapor is immediately removed because the site is exposed to the atmosphere. However, the reactant surface is gradually covered by the solid product layer as the reaction progresses. Under such physico-geometrical conditions, diffusion of the gaseous product through the surface product layer becomes the necessary process, which induces the increase in the p(H2O)SG at the reaction site covered by the product layer. Besides, the c value for the third reaction step exhibited an approximately constant value during the primary stage of the third reaction step (Fig. S43(c)). Because the third reaction step was characterized by the initial acceleration and subsequent deceleration with the maximum reaction rate midway through in both the reactions in a stream of dry and wet N2, a noticeable increase in p(H2O)SG is anticipated during the primary stage. The constant c value during the primary stage is indicative of the significant effect of p(H2O)SG.
In both reaction steps, the apparent Ea,i values exhibited minimal variation from those determined in the previous analysis step (Table S10), with the effect of p(H2O)SG being ignored. As the a = b values in each reaction step were maintained unchanged from the previous analysis step, the Ea,i,int values calculated according to eqn (17) also exhibited a minimal change from the previous evaluation. However, the extension of the kinetic analysis by incorporating the effect of p(H2O)SG enabled the empirical evaluation of the effect of p(H2O)SG and its variation as the reaction progressed in each reaction step.
It is imperative to acknowledge that the effect of p(H2O)SG is an inevitable factor for the rigorous kinetic description, even for the reaction occurring in a stream of inert gas with negligible p(H2O)ATM. The conventional kinetic analysis for the reaction in a stream of dry N2, as demonstrated in the previous section, can be upgraded in the context of the extended kinetic analysis, considering both p(H2O)SG and p(H2O)ATM. All kinetic data for the reactions in a stream of dry N2 were incorporated into the extended kinetic analysis of each reaction step. Fig. 10 and 11 illustrate the results of extended kinetic analysis, incorporating the kinetic data in a stream of dry N2, for the second and third reaction steps, respectively. In both reaction steps, the extended Friedman plot exhibited a statistically significant linear correlation including the data points for the reactions in a stream of dry N2 (Fig. 10(a) and 11(a)), which was achieved by optimizing the exponents a = b in AF (eqn (14)) and the coefficients (c, d) in p(H2O)EF (eqn (19)). The comparable results of linear plot were observed at various αi values (Fig. 10(b) and 11(b)).
Extended isoconversional relationships were established with the optimized a = b values of 0.48 and 0.30 for the second and third reaction steps, respectively. The coefficient d for p(H2O)ATM was estimated to be approximately unity during each reaction step. The trend of the variation in the coefficient c for p(H2O)SG in the second reaction step (Fig. 10(c)), exhibiting negligible value in the initial stage and an exponential increase in the latter stage, was equivalent to that evaluated without incorporating the kinetic data in a stream of dry N2 (Fig. S42(c)). Conversely, the c values for the third reaction step demonstrated significantly larger values during the reaction step, in comparison with those evaluated without incorporating the kinetic data in a stream of dry N2 (Fig. S43(c)). This result underscores the substantial effect of p(H2O)SG on the third reaction step, which is characterized by a rapid increase and subsequent decrease in the reaction rate.
The apparent Ea,2 values for the second reaction step exhibited an initial escalation, as in the previous estimation (Fig. S42(c)); however, approximately constant values were evaluated in the subsequent primary stage to the end of the reaction step (Fig. 10(c)). Concerning the third reaction step, a slightly decreasing trend of the Ea,3 values was observed as the reaction progressed, in accordance with the previous estimation (Fig. S43(c)). In both reaction steps, the Ea,i values demonstrated a decline in comparison with the previous estimation that did not incorporate the kinetic data in a stream of dry N2. This also led in a decline of the intrinsic Ea,i values calculated according to eqn (17) (Fig. 10(c) and 11(c)). The extended experimental master plots of the individual reaction steps demonstrated an analogous form with those evaluated in the previous kinetic analyses for the reactions in a stream of wet N2 with varying p(H2O)ATM values (Fig. 10(d) and 11(d)). However, the Ai values, determined by fitting the experimental master plots for the individual reaction steps using SB(m, n, p), were found to be lower than those determined in the previous estimation. Consequently, the intrinsic Ai values calculated according to eqn (18) also demonstrated a decline in both the reaction steps.
Table 3 enumerates the kinetic parameters that universally describe the kinetics of the second and third reaction steps of the thermal decomposition of Ni(OH)2 across a range of p(H2O)ATM values, including those under negligible p(H2O)ATM conditions, determined by considering the effects of p(H2O)SG and p(H2O)ATM. The intrinsic Ea,2 value was found to be marginally larger than the intrinsic Ea,3 value, while the intrinsic A2 and A3 values were comparable within the range of the standard error. The magnitude relationships between the intrinsic Arrhenius parameters of the second and third reaction steps are indicative of the second reaction step being a more difficult kinetic process. Consequently, the sequence of the reaction steps is predicted to be regulated by a physico-geometrical constraint of the reaction. In addition, the a = b value in AF indicated the larger value for the second reaction step, denoting the larger retardation effect of p(H2O)ATM. Therefore, the retardation of the primary thermal decomposition process of Ni(OH)2 with increasing p(H2O)ATM is interpreted to be controlled by the second reaction step.
| Reaction step, i | a = b | Ea,i/kJ mol−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b b |
Intrinsic Arrhenius parametersb | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Ai/s−1 | mi | ni | pi | R2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
Ea,i,int/kJ mol−1 | Ai, int/s−1 | |||
| a Coefficient c was optimized at individual αi values, while d was fixed to be unity.b Averaged over 0.10 ≤ αi ≤ 0.90.c Determination coefficient of the nonlinear least-squares analysis. | |||||||||
| 2 | 0.48 | 195.9 ± 1.1 | (6.10 ± 0.06) × 1015 | –1.30 ± 0.18 | 1.36 ± 0.07 | 1.42 ± 0.17 | 0.9987 | 172.6 ± 1.1 | (9.52 ± 0.82) × 1011 |
| 3 | 0.30 | 180.4 ± 2.0 | (2.48 ± 0.01) × 1014 | 0.46 ± 0.02 | 0.97 ± 0.01 | 0.11 ± 0.02 | 0.9999 | 165.8 ± 2.0 | (1.83 ± 0.03) × 1012 |
The rate behaviors of each reaction step under isothermal conditions can be further assessed based on the extended experimental master plots or the SB(mi, ni, pi) functions, by considering the reaction sequence and the contributions of the second and third reaction steps. As illustrated in Fig. 12, the extended experimental master plots are fitted using physicochemical and physico-geometrical kinetic model functions. The second reaction step was interpreted as the preparatory process within the primary thermal decomposition process of Ni(OH)2, with a limited contribution c2/(c2 + c3) ≈ 0.2. The rate behavior of the second reaction step in a stream of dry N2 was characterized by a linear deceleration following the initial short acceleration stage (Fig. S19(a)), which was interpreted as exhibiting the characteristics of the F1 model. One potential explanation for this phenomenon is that it is due to the consumption of the reactive sites on the surfaces of the Ni(OH)2 agglomerates. The extended experimental master plot, which universally describe the rate behavior across a range of p(H2O)EF values, exhibited the deceleration behavior with a convex shape (Fig. 12(a)), following a short acceleration stage. In accordance with the anticipated physico-geometrical reaction scheme of the consumption of the active sites in the surface, the rate behavior was described by the JAM(m) model with m = 1.31 ± 0.01.
The extended experimental master plot of the third reaction step (Fig. 12(b)) exhibited a limited variation from that of the conventional experimental master plot for the reactions in a stream of dry N2 (Fig. S19(b)), which was characterized by the swift acceleration and deceleration with the contribution of c3/(c2 + c3) ≈ 0.8. The rate behavior of the third reaction step in a stream of dry N2 was described by JMA(2.39) or ePT(0.52, 0.70) with equivalent statistical significance (Fig. S19(b)), while ePT(m, n) with m = 0.57 ± 0.01 and n = 0.93 ± 0.01 yielded better fits to the extended experimental master plot than that with JMA(m) with m = 1.91 ± 0.01. However, the random nucleation and the expansion of the volume of the product phase in the Ni(OH)2 matrix, as described by the JMA(m) model, is one possible physico-geometrical model for describing the autocatalytic behavior of the third reaction step.
4. Conclusions
The thermal decomposition of Ni(OH)2 exhibiting a three-step mass loss process was interpreted as comprising the sequential processes of the dehydration of absorbed or included water molecules in the Ni(OH)2 agglomerate, followed by the primary step of the thermal decomposition of Ni(OH)2 and subsequent evolution of trapped water accompanied by the crystal growth of NiO. The primary reaction step was identified as the actual thermal decomposition process of Ni(OH)2 to form NiO, accompanied by a dramatic increase in the specific surface area. A kinetic modeling of a four-step kinetic process was proposed based on the formal kinetic analysis and subsequent MDA and KDA procedures as applied to the thermal decomposition under various temperature variation conditions in a stream of dry N2. In this model, the primary mass loss process was considered as comprising a partially overlapping two-step process. The initial step of the primary reaction process was considered as the preparatory process with a small contribution occurring due to the consumption of reactive sites on the surfaces of Ni(OH)2 agglomerates. The subsequent step exhibited an autocatalytic rate behavior, which was interpreted as regulated by the nucleation and expansion of the solid product phase in the Ni(OH)2 matrix. A distinctive retardation effect of p(H2O)ATM on the primary reaction process of the thermal decomposition of Ni(OH)2 was evidenced by systematic TG measurements in a stream of wet N2 with controlled p(H2O)ATM. The effect of p(H2O)ATM on the individual reaction steps in the primary reaction process was parameterized in the context of an extended kinetic equation incorporating an AF describing universally the kinetic behavior as a function of T, α, and p(H2O)ATM. The extended kinetic description was able to further extend by incorporating the effect of p(H2O)SG, providing a finding of the significant effect of p(H2O)SG during the latter reaction step in the primary reaction process of the thermal decomposition of Ni(OH)2. After considerable efforts, the universal kinetic description of the individual reaction steps was finally achieved, encompassing all kinetic behavior, including those traced in a stream of dry N2 and wet N2 with varying p(H2O)ATM. The apparent kinetic parameters determined through the extended kinetic approach to the individual reaction steps can be utilized to simulate the reaction behavior at a selected temperature and p(H2O)ATM values. The final kinetic results indicated that the initial reaction step in the primary reaction process is regulative of the kinetics of the primary thermal decomposition process. In addition, the effect of p(H2O)ATM is more distinguishable in the initial reaction step. These novel kinetic findings concerning the primary thermal decomposition process of Ni(OH)2 indicate that controlling the initial reaction step can alter the overall kinetics and the reaction conditions of NiO formation. This may be achieved by controlling the properties of Ni(OH)2 precursors and the reaction conditions of its thermal decomposition process.Conflicts of interest
There are no conflicts of interest to declare.Data availability
Data will be made available on request.Supplementary information (SI): S1. Sample preparation and characterization (Fig. S1–S3), S2. Thermal behavior (Fig. S4–S6), S3. Thermal decomposition in a stream of dry N2 (Fig. S7–S19; Tables S1–S4), S4. Thermal decomposition in a stream of wet N2 with various atmospheric water vapor pressures (Fig. S20–S41; Tables S5–S9), S5. Universal kinetic description considering atmospheric and self-generated water vapor pressures (Fig. S42 and S43; Table S10). See DOI: https://doi.org/10.1039/d6cp00975a.
Acknowledgements
The present work was supported by JSPS KAKENHI Grant Numbers 23K22282, 24K05970, 24K21457, and 25KJ1876.References
- D. S. Hall, D. J. Lockwood, C. Bock and B. R. MacDougall, Proc. R. Soc. A, 2015, 471, 20140792 CrossRef PubMed.
- A. Azens, Solid State Ionics, 1998, 113–115, 449–456 CrossRef CAS.
- I. Hotovy, J. Huran, L. Spiess, S. Hascik and V. Rehacek, Sens. Actuators, B, 1999, 57, 147–152 CrossRef CAS.
- K. Fominykh, J. M. Feckl, J. Sicklinger, M. Döblinger, S. Böcklein, J. Ziegler, L. Peter, J. Rathousky, E. W. Scheidt, T. Bein and D. Fattakhova-Rohlfing, Adv. Funct. Mater., 2014, 24, 3123–3129 CrossRef CAS.
- R. Kumar, R. K. Singh, R. Savu, P. K. Dubey, P. Kumar and S. A. Moshkalev, RSC Adv., 2016, 6, 26612–26620 RSC.
- M. A. Nasseri, F. Ahrari and B. Zakerinasab, Appl. Organomet. Chem., 2016, 30, 978–984 CrossRef CAS.
- D.-L. Sun, B.-W. Zhao, J.-B. Liu, H. Wang and H. Yan, Ionics, 2017, 23, 1509–1515 CrossRef CAS.
- Y. Abdelbaki, A. de Arriba, R. Issaadi, R. Sánchez-Tovar, B. Solsona and J. M. López Nieto, Fuel Process. Technol., 2022, 229, 107182 CrossRef CAS.
- M. Imran Din and A. Rani, Int. J. Anal. Chem., 2016, 2016, 1–14 CrossRef PubMed.
- J. Mou, Y. Ren, J. Wang, C. Wang, Y. Zou, K. Lou, Z. Zheng and D. Zhang, Microfluid. Nanofluid., 2022, 26, 25 CrossRef CAS.
- S. S. Narender, V. V. S. Varma, C. S. Srikar, J. Ruchitha, P. A. Varma and B. V. S. Praveen, Chem. Eng. Technol., 2022, 45, 397–409 CrossRef CAS.
- W. Ahmad and A. Rawat, Nat., Environ. Pollut. Technol., 2023, 22, 1353–1362 CrossRef CAS.
- K. Arima, Y. Aoki, M. Hotta and N. Koga, Ind. Eng. Chem. Res., 2025, 64, 7298–7312 CrossRef CAS.
- I. F. Hazell and R. J. Irving, J. Chem. Soc. A, 1966, 669–673 RSC.
- J. M. Fernández Rodfríguez, J. Morales and J. L. Tirado, J. Mater. Sci., 1986, 21, 3668–3672 CrossRef.
- Z.-H. Liang, Y.-J. Zhu and X.-L. Hu, J. Phys. Chem. B, 2004, 108, 3488–3491 CrossRef CAS.
- X. Zhang, W. Shi, J. Zhu, W. Zhao, J. Ma, S. Mhaisalkar, T. L. Maria, Y. Yang, H. Zhang, H. H. Hng and Q. Yan, Nano Res., 2010, 3, 643–652 CrossRef CAS.
- V. Logvinenko, V. Bakovets and L. Trushnikova, J. Therm. Anal. Calorim., 2011, 107, 983–987 CrossRef.
- A. Tang, X. Li, Z. Zhou, J. Ouyang and H. Yang, J. Alloys Compd., 2014, 600, 204–209 CrossRef CAS.
- R. Yahyaoui and K. Nahdi, Inorg. Chem.: Indian J., 2016, 11, 1–9 CAS.
- N. M. Zali, T.-F. Choo, N. U. Saidin and K.-Y. Kok, AIP Conf. Proc., 2024, 2925, 020001 CrossRef CAS.
- R. M. Gabr, A. N. El-Naimi and M. G. Al-Thani, Thermochim. Acta, 1992, 197, 307–318 CrossRef CAS.
- C. S. Carney, R. E. Chinn, Ö. N. Doğan and M. C. Gao, J. Alloys Compd., 2015, 644, 968–974 CrossRef CAS.
- O. T. Sørensen and J. Rouquerol, Sample Controlled Thermal Analysis, Kluwer, Dordrecht, 2003 Search PubMed.
- S. Vyazovkin, A. K. Burnham, L. Favergeon, N. Koga, E. Moukhina, L. A. Perez-Maqueda and N. Sbirrazzuoli, Thermochim. Acta, 2020, 689, 178597 CrossRef CAS.
- N. Koga, S. Vyazovkin, A. K. Burnham, L. Favergeon, N. V. Muravyev, L. A. Pérez-Maqueda, C. Saggese and P. E. Sánchez-Jiménez, Thermochim. Acta, 2023, 719, 179384 CrossRef CAS.
- S. Vyazovkin, Int. Rev. Phys. Chem., 2020, 39, 35–66 Search PubMed.
- S. Kodani, S. Iwasaki, L. Favergeon and N. Koga, Phys. Chem. Chem. Phys., 2020, 22, 13637–13649 RSC.
- N. Koga, L. Favergeon and S. Kodani, Phys. Chem. Chem. Phys., 2019, 21, 11615–11632 RSC.
- M. Fukuda, L. Favergeon and N. Koga, J. Phys. Chem. C, 2019, 123, 20903–20915 CrossRef CAS.
- S. Fukunaga, Y. Zushi, M. Hotta and N. Koga, Phys. Chem. Chem. Phys., 2025, 27, 3384–3400 RSC.
- M. Hotta, Y. Zushi, S. Iwasaki, S. Fukunaga and N. Koga, Phys. Chem. Chem. Phys., 2023, 25, 27114–27130 RSC.
- M. Hotta, L. Favergeon and N. Koga, J. Phys. Chem. C, 2023, 127, 13065–13080 CrossRef CAS.
- Y. Zushi, S. Iwasaki and N. Koga, Phys. Chem. Chem. Phys., 2022, 24, 29827–29840 RSC.
- Y. Yamamoto, L. Favergeon and N. Koga, J. Phys. Chem. C, 2020, 124, 11960–11976 CrossRef CAS.
- S. Fukunaga, M. Hotta and N. Koga, J. Phys. Chem. C, 2026, 130, 1526–1538 CrossRef CAS.
- M. Hotta, T. Tone, L. Favergeon and N. Koga, J. Phys. Chem. C, 2022, 126, 7880–7895 CrossRef CAS.
- N. Koga, Y. Sakai, M. Fukuda, D. Hara, Y. Tanaka and L. Favergeon, J. Phys. Chem. C, 2021, 125, 1384–1402 CrossRef CAS.
- Y. Kodani, K. Arima, S. Fukunaga, M. Hotta and N. Koga, J. Phys. Chem. C, 2025, 129, 20207–20222 CrossRef CAS.
- M. Hotta and N. Koga, Netsu Sokutei, 2025, 52, 2–10 Search PubMed.
- M. Hotta and N. Koga, Thermochim. Acta, 2024, 733, 179699 CrossRef CAS.
- T. N. Ramesh, R. S. Jayashree and P. Vishnu Kamath, J. Electrochem. Soc., 2003, 150, A520–524 CrossRef CAS.
- T. N. Ramesh, J. Phys. Chem. B, 2009, 113, 13014–13017 CrossRef CAS PubMed.
- O. T. Sørensen, J. Therm. Anal., 1978, 13, 429–437 CrossRef.
- A. Szytula, A. Murasik and M. Balanda, Phys. Status Solidi B, 1971, 43, 125–128 CrossRef CAS.
- J. Tientong, S. Garcia, C. R. Thurber and T. D. Golden, J. Nanotechnol., 2014, 2014, 1–6 CrossRef.
- K. Lawson, S. P. Wallbridge, A. E. Catling, C. A. Kirk and S. E. Dann, J. Mater. Chem. A, 2023, 11, 789–799 RSC.
- E. Konysheva, E. Suard and J. T. S. Irvine, Chem. Mater., 2009, 21, 5307–5318 CrossRef CAS.
- N. Koga and H. Tanaka, J. Therm. Anal., 1993, 40, 1173–1179 CrossRef CAS.
- S. Iwasaki, Y. Zushi and N. Koga, Phys. Chem. Chem. Phys., 2021, 23, 22972–22983 RSC.
- Y. Zushi, S. Iwasaki and N. Koga, Phys. Chem. Chem. Phys., 2022, 24, 15736–15748 RSC.
- H. Tanaka and N. Koga, J. Therm. Anal., 1990, 36, 2601–2610 CrossRef CAS.
- S. Vyazovkin, K. Chrissafis, M. L. Di Lorenzo, N. Koga, M. Pijolat, B. Roduit, N. Sbirrazzuoli and J. J. Suñol, Thermochim. Acta, 2014, 590, 1–23 CrossRef CAS.
- H. L. Friedman, J. Polym. Sci., Part C, 1964, 6, 183–195 CrossRef.
- N. Koga, Y. Goshi, S. Yamada and L. A. Pérez-Maqueda, J. Therm. Anal. Calorim., 2013, 111, 1463–1474 CrossRef CAS.
- N. Koga, in Handbook of Thermal Analysis and Calorimetry, ed S. Vyazovkin, N. Koga and C. Schick, Elsevier, Amsterdam, 2nd edn, 2018, vol. 6, ch. 6, pp. 213–251 Search PubMed.
- A. Perejón, P. E. Sánchez-Jiménez, J. M. Criado and L. A. Pérez-Maqueda, J. Phys. Chem. B, 2011, 115, 1780–1791 CrossRef PubMed.
- R. Svoboda and J. Málek, J. Therm. Anal. Calorim., 2013, 111, 1045–1056 CrossRef CAS.
- T. Ozawa, Bull. Chem. Soc. Jpn., 1965, 38, 1881–1886 CrossRef CAS.
- T. Ozawa, Thermochim. Acta, 1986, 100, 109–118 CrossRef CAS.
- J. Málek, Thermochim. Acta, 1992, 200, 257–269 CrossRef.
- N. Koga, Thermochim. Acta, 1995, 258, 145–159 CrossRef CAS.
- F. J. Gotor, J. M. Criado, J. Malek and N. Koga, J. Phys. Chem. A, 2000, 104, 10777–10782 CrossRef CAS.
- J. M. Criado, L. A. Perez-Maqueda, F. J. Gotor, J. Malek and N. Koga, J. Therm. Anal. Calorim., 2003, 72, 901–906 CrossRef CAS.
- P. E. Sánchez-Jiménez, A. Perejón, J. M. Criado, M. J. Diánez and L. A. Pérez-Maqueda, Polymer, 2010, 51, 3998–4007 CrossRef.
- J. Šesták and G. Berggren, Thermochim. Acta, 1971, 3, 1–12 CrossRef.
- J. Šesták, J. Therm. Anal., 1990, 36, 1997–2007 CrossRef.
- J. Šesták, J. Therm. Anal. Calorim., 2011, 110, 5–16 CrossRef.
- E. G. Prout and F. C. Tompkins, Trans. Faraday Soc., 1944, 40, 488–498 RSC.
- W. A. Johnson and K. F. Mehl, Trans. Am. Inst. Min. Metall. Eng., 1939, 135, 416–458 Search PubMed.
- M. Avrami, J. Chem. Phys., 1939, 7, 1103–1112 CrossRef CAS.
- M. Avrami, J. Chem. Phys., 1940, 8, 212–223 CrossRef CAS.
- M. Avrami, J. Chem. Phys., 1941, 9, 177–184 CrossRef CAS.
- B. V. Erofeyev, CR Acad. Sci. URSS, 1946, 52, 511–514 Search PubMed.
- N. N. Kolmogorov, Bull. Acad. Sci. USSR, Math. Ser. 1, 1937, 355–359 Search PubMed.
- A. K. Galwey and W. J. Hood, J. Phys. Chem., 1979, 83, 1810–1815 CrossRef CAS.
- T. Wada, M. Nakano and N. Koga, J. Phys. Chem. A, 2015, 119, 9749–9760 CrossRef CAS PubMed.
- K. L. Mampel, Z. Phys. Chem., Abt. A, 1940, 187, 43–57 Search PubMed.
- K. L. Mampel, Z. Phys. Chem., Abt. A, 1940, 187, 235–249 Search PubMed.
- H. Ogasawara and N. Koga, J. Phys. Chem. A, 2014, 118, 2401–2412 CrossRef CAS PubMed.
- L. Favergeon, M. Pijolat and M. Soustelle, Thermochim. Acta, 2017, 654, 18–27 CrossRef CAS.
- D. D. Wagman, W. H. Evans, V. B. Parker, R. H. Schumm, I. Halow, S. M. Bailey, K. L. Churney and R. L. Nuttall, The NBS Tables of Chemical Thermodynamic Properties. Selected Values for Inorganic and C1 and C2 Organic Substances in SI Units, American Chemical Society, Washington, DC, 1982 Search PubMed.
| This journal is © the Owner Societies 2026 |