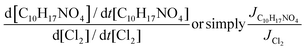Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Formation and photolysis of multifunctional organic nitrates from the reaction of limonene and NO3 radicals
Laura Wüsta,
Lasse Moormann ab,
Patrick Dewald
ab,
Patrick Dewald a,
Eunyeong Jin
a,
Eunyeong Jin a,
Jan Schuladena,
Jonathan Williamsa,
Frank Drewnickb,
Ulrich Pöschl
a,
Jan Schuladena,
Jonathan Williamsa,
Frank Drewnickb,
Ulrich Pöschl b,
Jos Lelievelda and
John N. Crowley
b,
Jos Lelievelda and
John N. Crowley *a
*a
aAtmospheric Chemistry Department, Max Planck Institute for Chemistry, Mainz, 55128, Germany. E-mail: john.crowley@mpic.de
bMultiphase Chemistry Department, Max Planck Institute for Chemistry, Mainz, 55128, Germany
First published on 5th May 2026
Abstract
Multifunctional organic nitrates (LIM-ONO2) were formed from the NO3-initiated oxidation of limonene in the Simulation Chamber for Atmospheric Reactions and Kinetics (SCHARK), detected quantitatively by thermal dissociation cavity ring-down spectroscopy (TD-CRDS) and identified by high-resolution time-of-flight chemical-ionization (iodide) mass spectrometry (HR-ToF-ICIMS). Based on HR-ToF-ICIMS signal intensities, the most abundant LIM-ONO2 were C10H17NO4 and C10H17NO5, together representing >60% of the total LIM-ONO2 signal. We developed a method for cold-trapping LIM-ONO2 from the chamber, enabling us to examine their photolysis in the absence of precursor chemicals after re-injection into the SCHARK. The photolytic loss frequency of C10H17NO4 in the chamber when irradiated with LEDs emitting at 370 ± 13 nm was (1.69 ± 0.06) × 10−4 s−1. By comparison to the photolysis frequency of a chemical actinometer (Cl2), we were able to gain insight into the quantum yield (0.3–0.8) and absorption cross section of C10H17NO4 at these wavelengths and make an estimate of its atmospheric lifetime with respect to photolysis.
1. Introduction
The oxidation of volatile organic compounds (VOCs) influences both air quality and climate through processes such as tropospheric ozone (O3) formation and secondary organic aerosol (SOA) production.1 Around 90% of all VOC emitted are biogenic, and the total global emissions of these biogenic volatile organic compounds (BVOCs) is estimated at ∼1000 Tg per year, with monoterpenes accounting for ∼5% of the total.2 Although the contribution of monoterpenes to total BVOC emissions is small, they are highly reactive towards key atmospheric oxidants (e.g. O3, OH and NO3) and potent producers of new particles. While OH-initiated oxidation dominates during the daytime, NO3 radicals (formed by the reaction of O3 with NO2, R1) play a central role in BVOC removal at night. These reactions lead to the formation of organic nitrates (RONO2, R2), which act as a temporary reservoir for reactive nitrogen.3–8 or can represent a permanent sink for NOx when organic nitrates partition into the particle phase and are removed via deposition9 or hydrolysis.10–12 During the day, NO3 undergoes rapid photolysis or reacts with NO to form NO26 which drastically reduces its lifetime. In environments (e.g. forested regions) with high levels of BVOCs, NO3 may contribute to their oxidation during the daytime.13–15| NO2 + O3 → NO3 + O2 | (R1) |
| NO3 + VOC + (O2) → RONO2 + products | (R2) |
There are several reaction steps between the initial formation of the nitrated organic radical (the product of electrophilic addition of NO3 to a double bond in the BVOC) and the formation of stable, multifunctional organic nitrates. The identity of the stable end-products depends on the fate of the nitrated peroxy radicals (RO2) formed when the nitrated organic radical reacts with O2. RO2 can react with NO, NO3, other peroxy radicals or undergo auto-oxidation. In our experiments, even though NO3 radicals initiate the oxidation of limonene, their mixing ratios are strongly suppressed by the high limonene mixing ratio and RO2 + NO3 reactions are not expected to play an important role. As there is no NO source in our chamber (under the dark conditions in which LIM-ONO2 are formed) and the reaction with NO2 forms thermally unstable peroxy nitrates that readily dissociate back to reactants, most products are expected to form in RO2 + RO2 type reactions. RO2 + HO2 reactions are expected to be of minor importance for our system.
The chemical mechanisms involved in monoterpene degradation have not yet been fully characterized for most commonly emitted monoterpenes and the available literature does not provide a comprehensive understanding of organic nitrate formation pathways or their contribution to SOA composition and NOx removal.3 This is particularly pronounced for limonene, a monoterpene prevalent in urban, forested and indoor environments.16,17 Limonene is highly reactive towards NO3 radicals, with a rate coefficient of 1.2 × 10−11 cm3 molecule−1 s−1 at 298 K.18 By comparison, the rate coefficients for NO3 + α-pinene (6.2 × 10−12 cm3 molecule−1 s−1 at 298 K) and NO3 + isoprene (6.5 × 10−13 cm3 molecule−1 s−1 at 298 K) are much lower.18 The reaction of NO3 with limonene leads to the formation of long-chain multifunctional organic nitrates, many of which can partition into the particle phase or contribute to SOA formation.19–22 Experimental studies have identified a variety of oxidation products from the limonene + NO3 reaction, including hydroxy nitrates, carbonyl nitrates and hydroperoxy nitrates, which have been detected in both the gas and particle phases.23–25
The photolysis of organic nitrates derived from biogenic trace gases such as isoprene and α-pinene has been identified as a potential loss process that directly affects the abundance of reactive nitrogen in the atmosphere. If photolysis occurs after the organic nitrates have undergone long distance transport, reactive nitrogen can be released in remote areas where ozone production efficiency is high. Many RONO2 species absorb dissociatively in the actinic region, releasing e.g. NO2 and multifunctional alkoxy radicals (R3) that lead to further, organic oxidation products.10,26–28 Experimental and theoretical studies have shown that the photochemical behaviour of organic nitrates strongly depends on their molecular structure. Measurements of Roberts and Fajer29 and Barnes et al.30 provided UV absorption cross sections for simple alkyl nitrates and a series of difunctional organic nitrates, including C3–C4 carbonyl nitrates and dinitrates. Barnes et al.30 demonstrated that the UV-absorption spectra of carbonyl-substituted (keto) nitrates are red-shifted relative to non-substituted alkyl nitrates and have a factor five higher absorption cross sections at wavelengths above 290 nm. Subsequent work by Muller et al.27 and Xiong et al.26 demonstrated that conjugated nitrooxy carbonyl compounds formed from isoprene oxidation exhibit strong absorption in the near-UV region and undergo rapid photolysis under atmospherically relevant conditions.
Muller et al.27 further showed that α- or β-nitrooxy carbonyl groups in isoprene derived nitrates enhance UV absorption and result in photolysis rates 3–20 times higher than those for OH loss. Xiong et al.26 studied the photolysis of the isoprene-derived 4,1-nitrooxy-enal, a conjugated carbonyl-nitrate, and showed that conjugation between the carbonyl and nitrate functional groups enhances absorption in the actinic region, resulting in efficient photolysis with daytime atmospheric lifetimes of less than one hour. These findings highlight that certain conjugated carbonyl nitrates originating from BVOCs are highly photolabile and that photolysis can represent a major daytime sink for this class of compounds. Recent work by Wang et al.31 has demonstrated that multifunctional organic nitrates (from α-pinene, β-pinene and limonene) can photolyse efficiently under actinic radiation, thereby reforming NOx.
| RONO2 + hv → NO2 + RO | (R3) |
Despite the potential importance of photolysis as a loss mechanism for atmospheric organic nitrates of biogenic origin, information regarding the photolytic lifetimes of such nitrates and their relative importance compared to e.g. deposition9,14 or hydrolysis32 remains sparse.
One factor that has hindered research into the photochemical degradation of atmospherically relevant organic nitrates is the difficulty of synthesizing the multifunctional nitrates involved. As a result, mainly surrogate species with similar structures have been examined so far.26,27,31,33,34 In this study, we generate atmospheric organic nitrates (LIM-ONO2) in the gas-phase via NO3 induced oxidation of limonene in a simulation chamber, isolate them from precursor molecules by sampling into a cold-trap and then re-inject them into a precursor-free chamber to examine their behaviour during photolysis. Our goal is to produce the first dataset on the photolysis of selected LIM-ONO2 species and to estimate their photodissociation quantum yields and atmospheric lifetimes.
2. Experimental
2.1. SCHARK chamber
The Simulation Chamber for Atmospheric Reactions and Kinetics (SCHARK) was used for the measurements described here. This cubic chamber (side length 1 m) is made of PFA film (0.13 mm thick) and has a volume of 1 m3. The volume surrounding the PFA film is permanently flushed with dry-zero air to minimize contamination from laboratory air and the permeation of water vapour. Gases entering the chamber pass through a circular loop of perforated PFA tubing (10 holes of 1 mm diameter) located at the bottom of the chamber. Together with a magnetically coupled Teflon-coated fan, the gas-jets thus created, result in complete mixing in 40 s.35 The chamber was operated at ambient pressure and temperature (298 ± 5 K) in dynamic-flow mode. The exchange constant (kexch) of the chamber was determined by following the exponential decrease in the concentration of O3 that had been injected into the chamber and is given by kexch (s−1) = 1.82 × 10−5 F, where F is the mass flow rate into the chamber in standard litres per minute (SLM).36 The uncertainty in kexch is 3% (2σ).A total of 26 evenly distributed light-emitting diodes (LEDs) with a central wavelength of around 370 nm and a width (full-width at half-maximum) of 13 nm were used to provide a homogeneous light flux through the chamber. The LEDs have the advantage over e.g. black-lamps that they result in less heating of the chamber and thus less desorption of organic nitrates adsorbed on the chamber walls, which could complicate the analysis of e.g. decays rates due to photolysis. An emission spectrum of the LEDs is given in the Results section.
2.2. HR-ToF-ICIMS
Measurements of organic nitrates, organic acids, alcohols and N2O5 were conducted with a high-resolution time-of-flight Iodide-chemical-ionization mass spectrometer (HR-ToF-ICIMS, Lee et al.,37 Aerodyne Research Inc, USA) with a resolution of 5000 at m/z 250. The primary iodide ions were generated from photoionization of 40 parts-per-million CH3I by volume (hereafter ppmv) in a flow of 1.0 L (STD) min−1 (hereafter SLM) N238,39 before entering the ion–molecule-reactor, which was kept at 60 °C and 170 mbar. The HR-ToF-ICIMS sampled a total flow of 2.1 SLM into the ion–molecule reactor with 25% originating from the SCHARK. The remaining 75% was purified air, 10% of which was humidified to ∼100% relative humidity at room temperature to maintain I− and I·H2O− reagent ions at a constant level and ratio. During data processing, ions were identified using Tofware V4.0.3 (Aerodyne Research Inc.). HR-ToF-ICIMS signals were normalized to the total reagent ion signal, corrected for inlet flow dilution, and baseline-subtracted to yield normalized counts per second (ncps). The limit of detection (LOD) was determined as three times the standard deviation (3σ) interval of the respective ion signal during zero-air measurement. In order to focus on the major products, only ions with signal intensities exceeding three times the LOD were included in the analysis. The HR-ToF-ICIMS was calibrated for N2O5 by comparison with a thermal-dissociation cavity-ringdown-spectrometer (TD-CRDS).39 No calibration standards are available for the limonene derived organic nitrates measured here; however, total multifunctional organic nitrates were also quantified using a TD-CRDS as described below. An average detection sensitivity of the HR-ToF-ICIMS for the organic nitrates is reported in Section 3.1.2.3. Five-channel cavity ring-down spectrometer
A five-channel cavity ring-down spectrometer40 was used to measure NO2, NOx, ∑PNs (peroxy nitrates, RO2NO2) and ∑ANs (multifunctional (“alkyl”) nitrates, RONO2). The “blue-channels” of the instrument used laser diodes emitting at 409 nm where NO2 absorbs. NO2 was measured directly after sampling through an unheated inlet whereas ∑PNs and ∑ANs were thermally dissociated to NO2 by heating their separate PFA and Quartz inlets36 to 448 and 648 K, respectively. The five-channel cavity ring-down spectrometer was operated in NOx rather than NO2 mode to minimize bias from secondary reactions when measuring ∑ANs and ∑PNs following thermal dissociation.35The cavity ring-down spectrometer was connected to the SCHARK via ∼160 cm of ¼-inch (0.635 cm ID) PFA tubing. Sample air entered each cavity at a flow rate of 2.1 SLM. Zeroing of the instrument was performed by overfilling the inlet line with dry zero air. Ring-down times were converted to mixing ratios using the effective absorption cross section σλ of NO2 at 409 nm. The value of σλ was calculated from the overlap between the laser emission and the NO2 absorption spectrum.41 The laser emission spectrum was recorded with a dedicated CCD spectrometer (Ocean Optics HR4000). The effective cross section was typically within a few percent of 6.4 × 10−19 cm2 molecule−1.
The LOD for NOx and ∑ANs was determined by measuring zero air over a period of one hour. The 2σ standard deviation for NOx and ∑ANs are 40 and 30 pptv, respectively (for an integration time of 1 s). Using these values the detection limits for NOx and ∑ANs are 40 and 50 pptv, respectively. Note that the LOD for ∑ANs is obtained by error propagation in both the NOx and ∑ANs channels. The total uncertainty of the NOx measurements includes the uncertainty associated with the absorption cross section41 and fluctuations in the laser emission spectrum, which together amount to 6.5%.42 The total uncertainty associated with the ∑ANs measurements depends on the total uncertainty of the NOx channel and amount to ∼9%. Mixing ratios attributed to ∑PNs were below the detection limit under our experimental conditions and are therefore not discussed further in this study.
2.4. Experimental procedure
Limonene nitrates were formed by the reaction of limonene with NO3 in dry air in the SCHARK chamber. NO3 was generated by thermal decomposition of N2O5, which was introduced into the chamber by passing ∼500 cm3 (STD) min−1 (hereafter sccm) dry zero air over N2O5 crystals held at 195 K (dry ice acetone bath). N2O5 was synthesized by flowing 150 sccm of NO (5% in N2, Westfalen) and 200 sccm of O2 with ∼4% O3 (generated via electrical discharge) through a 2 L glass reaction vessel.43 The resulting N2O5 was trapped as colourless crystals in a glass vessel held at 195 K. The N2O5 crystals were stored at 243 K until use (for a maximum of 2 days). This source of NO3 was preferred to the in situ reaction between NO2 and O3 as the latter also reacts with limonene. Limonene was introduced into the chamber via flow-controllers connected to a steel canister containing 430 ppmv limonene (96%, Sigma-Aldrich) in He (5.0, Westfalen). O3 was continuously monitored during the photolysis experiments using a commercial UV absorption instrument (2B Technologies Model 205), operating at 254 nm. The LOD of the O3 monitor was 2 ppbv for a 10 s averaging time.Each experiment had a duration of between 3 and 5 hours, after which the SCHARK chamber was cleaned by flushing first with humidified zero air (for 1–2 hours) to remove residual reactive nitrogen species, followed by dry zero air (overnight) to remove remaining humidity.
3. Results and discussion
3.1. Formation of limonene-nitrates (LIM-ONO2)
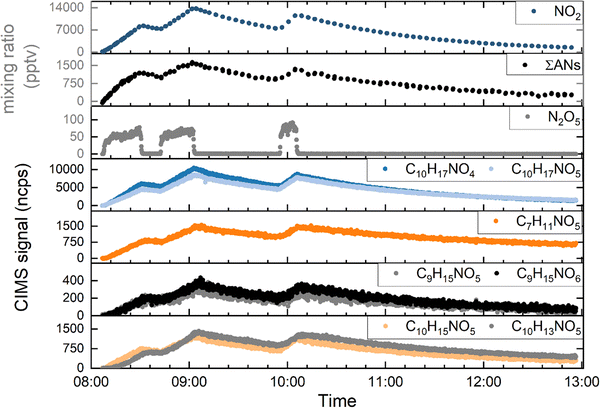
Fig. 1 shows the results of an experiment in which a flow of 2.8 sccm limonene (C10H16) in air (430 ppmv) in a total flow of 12.5 SLM zero-air was introduced into the chamber (prefilled with zero-air), resulting (after ∼20 min) in a limonene concentration of ∼20 ppbv. This was followed by three injections of N2O5 (at 08:07, 08:42 and 09:56) initiating the formation of various LIM-ONO2 that were detected using HR-ToF-ICIMS (lower 4 panels in Fig. 1). The mixing ratio of N2O5 that was injected was approximately 16 ppb, derived by numerical simulation of the measured mixing ratios in the SCHARK (Fig. S1).With one exception (C7H11NO5), the strongest signal intensities (molecules in bold in the following text) observed were from C10 nitrates clustered with iodide. These were observed at m/z 342.02 (C10H17NO4), 358.02 (C10H17NO5), (C10H15NO5) and 353.98 (C10H13NO5). Weaker signals were observed from C10H15NO6, C10H15NO7, C10H16N2O6 and C10H17NO6.
In addition to C10 nitrates we also observed C2H4N2O6, C2H5NO4, C3H5NO4, C3H5NO5, C4H7NO5, C4H8N2O6, C4H9NO4, C6H9NO5, C6H12N2O6, C7H9NO6, C7H11NO5, C7H11NO6, C8H13NO5, C8H13NO6, C8H15NO4, C9H13NO6, C9H13NO7, C9H15NO5, C9H15NO6. The m/z ratios, relative signal intensities and saturation mass concentration for all nitrates detected are listed in Table S1 and displayed in Fig. S2 which segregate the nitrates into low, semi and intermediate volatility VOCs (LVOC, SVOC and IVOC, respectively).44,45 The high abundance of IVOC compared to LVOC indicates that significant aging and partitioning into the particle phase has not occurred in our experiments. In Fig. 1 we plot the signal intensities for seven organic nitrates (those in bold-type above), selected either for their large signal intensities or because they are expected to be the main products of the reaction. The total multifunctional alkyl nitrate mixing ratio (ΣANs) as determined from the TD-CRDS measurement reached a maximum of 1.7 ppbv (at ∼09:00), coincident with the maximum HR-ToF-ICIMS signals and maximum NO2 mixing ratio (13 ppbv). With the exception of N2O5, the mixing ratios/signals of the trace-gases shown in Fig. 1 exhibit similar trends in production (as long as N2O5 is present) and loss (due to flow-out of the SCHARK) when N2O5 has depleted to zero. The time scale (∼60–90 s) for N2O5 to disappear after its flow into the SCHARK was ceased is commensurate with its thermal decomposition rate coefficient at 1 bar and 298 K (4.4 × 10−2 s−1) and the fact that each NO3 formed is rapidly scavenged (within 0.2 s) by limonene (20 ppbv).
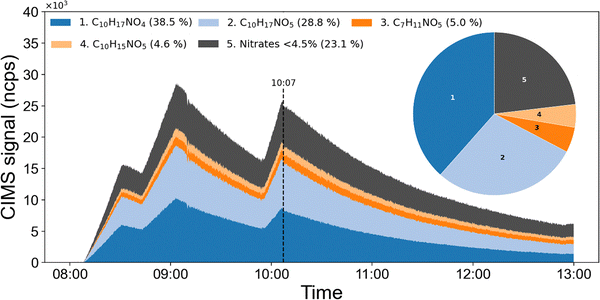
Fig. 2 shows the time-dependent contributions of the 4 dominant organic nitrates to the total signal, with the remaining signals summed and shown in grey. The relative composition does not change significantly with the number of N2O5 additions. The pie-chart inset indicates the contributions of each LIM-ONO2 signal at 10:07 after the three N2O5 injections. Note that the data are based on the HR-ToF-ICIMS signal intensities rather than mixing ratios of each nitrate during the gas-phase sampling period and the actual composition could be different. The largest signal is associated with C10H17NO4 which accounts for 38.5% of the total LIM-ONO2 signal. This is followed by C10H17NO5 (28.5%), C7H11NO5 (5%) and C10H15NO5 (4.6%). The remaining 22 nitrate species each contribute less than 4.5%, but together account for 23.1% of the total signal. No significant experiment-to-experiment variation in the fractional contribution of the 4 main organic nitrates was observed.
Table 1 lists the LIM-ONO2 species identified by the HR-ToF-ICIMS and indicates which ones could be cold-trapped and re-injected into the SCHARK before subject to photolysis. The likely functional groups are those previously reported in the literature (where available). While the exact structures remain uncertain, the LIM-ONO2 are likely formed via RO2 + RO2 reactions, since the high limonene concentration in the chamber suppresses reactions of RO2 with NO3. A simple numerical simulation indicates that, under high limonene conditions in the chamber, product formation from RO2 + RO2 reactions exceeds that from RO2 + NO3 reactions by a factor of ∼200 (Fig. S1). The proposed gas-phase mechanisms leading to the formation of some of the LIM-ONO2 species observed are shown in Fig. S3.
| LIM-ONO2 | Functional groups (excluding nitrate) | Ref. | Trapping behaviour | Photolysis behaviour | J (10−4 s−1) |
|---|---|---|---|---|---|
| Notes: Faxon et al.,24 Takeuchi et al.47 and Fry et al.19 reported particle-phase measurements. Additionally Fry et al.19 proposed a gas phase mechanism. + indicates an increase in the CIMS signal, − indicates a decrease in the CIMS signal.a indicates that the measured photolysis frequency was determined in the absence of cyclohexane. | |||||
| C10H17NO4 | Hydroxy (Fig. S3) | Mayorga et al.25 | Always | − | 1.69 ± 0.06 |
| Ayres et al.46 | |||||
| Devault et al.22 | 5.32 ± 0.07a | ||||
| Fry et al.19 | |||||
| C10H17NO5 | Hydroperoxy/carbonyl, hydroxy (Fig. S3) | Ayres et al.46 | Sometimes | + | — |
| Mayorga et al.25 | |||||
| Devault et al.22 | |||||
| C7H11NO5 | Hydroxy, carbonyl | Takeuchi et al.47 | Sometimes | + | — |
| C10H15NO5 | Dicarbonyl (Fig. S3) | Devault et al.22 | Always | − | — |
| Ayres et al.46 | |||||
| C10H13NO5 | — | Always | − | — | |
| C10H15NO6 | Faxon et al.24 | Always | + | — | |
| C10H15NO7 | Takeuchi et al.47 | Sometimes | + | — | |
| C9H15NO5 | Carbonyl, hydroxy (see Fig. S3) | Faxon et al.24 | Never | + | — |
| Mayorga et al.25 | |||||
| Fry et al.19 | |||||
| C9H15NO6 | Mayorga et al.25 | Never | + | — | |
| C9H13NO6 | Mayorga et al.25 | Never | + | — | |
| C9H13NO5 | Mayorga et al.25 | Never | + | — | |
| C10H17NO6 | Dicarbonyl, hydroxy (see Fig. S3) | Mayorga et al.25 | Never | + | — |
| Fry et al.19 | |||||
| Faxon et al.24 | |||||
| C10H16N2O6 | Dinitrate | — | Always | − | 1.22 ± 0.06 |
The main LIM-ONO2 species (C10H17NO4, C10H17NO5, C7H11NO5 and C10H15NO5) observed in our experiments are consistent with previous reports. Devault et al.22 identified C10H17NO4 and C10H17NO5 as the most intense contributors in the I-CIMS mass spectra, with additional detection of C10H15NO5. Mayorga et al.25 reported signals corresponding to C10H15,17NO4,5 and C9H13,15NO5-8 in both gas and particle phases, indicating that C10H15,17NO4,5 are likely first-generation products. Takeuchi et al.47 identified C7H11NO5 as a characteristic product in thermal desorption particle-phase analysis. HR-ToF-ICIMS measurements (Faxon et al.24) report dominant SOA species including C10H15NO6, C10H17NO6 and C9H13NO7.
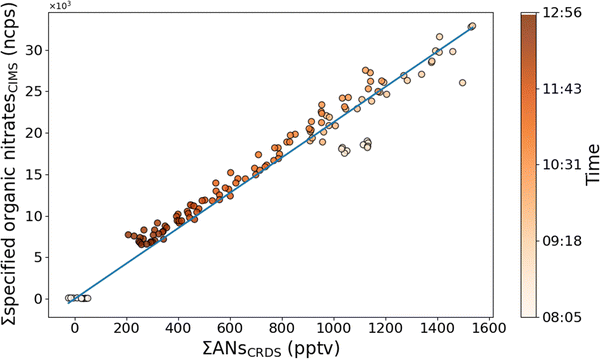
Fig. 3 shows a correlation between the total multifunctional alkyl nitrates (in pptv) measured by TD-CRDS and the summed HR-ToF-ICIMS signals (in normalised counts per second, ncps) of the individual organic nitrates in the absence of N2O5. The HR-ToF-ICIMS signals were multiplied by a factor 1 or 2 according to the number of nitrogen atoms in each species. A linear regression (blue line) was performed on the data obtained when N2O5 was absent (N2O5 can also be detected with the TD-CRDS at the TD-temperature for organic nitrates (648 K) and may therefore introduce artefacts in the data).
Assuming equal sensitivity of the HR-ToF-ICIMS to all detected LIM-ONO2, the regression slope provides an estimate of the HR-ToF-ICIMS sensitivity of 21 ncps pptv−1. An absolute sensitivity comparison to other CIMS instruments is hampered by various technical and operational factors.48,49 Therefore, a relative comparison with the sensitivity of commonly calibrated species for this CIMS appears to be most appropriate. A sensitivity of 21 ncps pptv−1 is broadly consistent with that observed for iodide clusters from N2O5 (∼30 ncps pptv−1) under identical operational conditions.39 As N2O5 is detected (as an iodide-cluster) with high sensitivity by HR-ToF-ICIMS, we conclude that reaction of I− with LIM-ONO2 (to form iodide-clusters) occurs at a similar rate coefficient.
3.2. LIM-ONO2 extraction/isolation
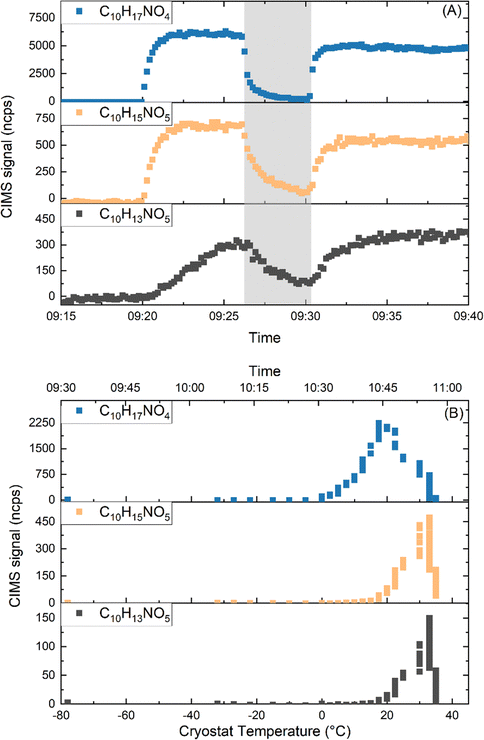
In order to extract and isolate LIM-ONO2 formed as described above, a flow of 5 SLM from the chamber was passed through a glass spiral (Fig. S4) submerged in a cooling liquid at −30 °C. To avoid further reaction in the condensed phase, the sampling was conducted only in periods when N2O5 (and thus NO3) was completely depleted. Between experiments, the glass spiral was cleaned with distilled water, followed by drying at 100 °C. Typically, LIM-ONO2 were collected over a period of ∼3 hours in this manner and subsequently stored in the glass-spiral at −78 °C (acetone/dry ice mixture) overnight, during which the SCHARK was flushed with humidified zero air in preparation for the next day's experiments.Fig. 4a shows the time-series signals of three specific LIM-ONO2 (C10H17NO4, C10H15NO5, C10H13NO5) measured by the HR-ToF-ICIMS during an experiment in which N2O5 had been injected to form LIM-ONO2 and products was sampled (after N2O5 had decayed to zero) via the glass-spiral at either room temperature (09:20 to 09:26 and 09:32 to 09:40) or when the glass-spiral was cooled to −30 °C (shaded grey). Note that (according to CPC measurements) no particles were present in the SCHARK during sampling into the cold trap.
The almost complete loss of the C10H17NO4 signal when sampling through the spiral at −30 °C indicates efficient trapping of these LIM-ONO2. In contrast, experiments using a GC-MS50 revealed that, at −30 °C, limonene was not trapped in the glass spiral to a significant extent at the typical mixing ratios used for the SCHARK experiments.
The fact that the LIM-ONO2 signals did not immediately go to zero when the glass-spiral was cooled, reflects the fact that we are dealing with molecules that have a high surface affinity (as would be expected for trace gases with large molecular weight and a high degree of substitution by polar groups) and that some desorption from the PFA-tubing between the glass-spiral and the HR-ToF-ICIMS takes place. The three different time-series of C10H17NO4, C10H15NO5 and C10H13NO5 indicate that they have different affinities for the tubing between the glass-spiral and the HR-ToF-ICIMS, with C10H17NO4 (hydroxy-nitrate, blue) being less “sticky” (i.e. the signal decays more rapidly) than C10H15NO5 (di-carbonyl-nitrate, orange) and C10H13NO5 (dark grey).
The temperature dependent desorption of LIM-ONO2 from the glass spiral was examined by passing a flow of 200 sccm zero-air through the glass-spiral prior to further dilution (5–10 SLM) and transport (via ∼2 m of PFA tubing) to the HR-ToF-ICIMS (i.e. bypassing the SCHARK). Fig. 4b shows the results obtained when the glass-spiral was continuously warmed from −78 °C to 35 °C over a period of ∼80 minutes. The first LIM-ONO2 to start desorbing was C10H17NO4 at a temperature of ∼0 °C with the maximum signal at 20 °C after which the signal decreased. A single (albeit broad) desorption feature is observed, indicating that the physical adsorption of C10H17NO4 on the glass-surface is limited to one type of site or that multiple adsorption sites have similar binding energies. Desorption of both C10H15NO5 and C10H13NO5 started at a temperature of ∼15 °C with the maximum signals obtained at 30 and 35 °C, respectively. As expected, the nitrates requiring higher temperatures to desorb from the glass surface are those that displayed the slowest decrease in Fig. 4a. This result is intuitive and related to the vapour pressure of the LIM-ONO2 and their affinity for surfaces. Similar plots illustrating the adsorption (trapping) and desorption of the other LIM-ONO2 are shown in Fig. S5 and S6. In Fig. S5 we see that the signals of nearly all the C10 LIM-ONO2 decrease when the sample is passed through the glass-spiral at −30 °C. Exceptions to this are C10H17NO6 and C9H15NO5 whose signals clearly increases during the trapping phase. As trapping only took place after N2O5 and NO3 were consumed, it is unlikely that these signals arise through condensed-phase chemistry involving NO3 or N2O5. However, we cannot rule out that the co-condensation of HNO3 in the trap (Fig. S6c) may have led to some chemical reactions in which e.g. C10H17NO6, and C9H15NO5 are formed. We also note that several C3 nitrates (e.g. C3H5NO4, C3H5NO5) and a C4-dinitrate (C4H8N2O8) were also trapped at −30 °C. The trapping behaviour of the most abundant limonene nitrates are summarized in Table 1. Trapping behaviour designated “always” means that the species could be effectively trapped in the glass-spiral, stored overnight (while the SCHARK was flushed) and transferred into the chamber by rapidly warming with warm water at ∼40–50 °C while passing 500 sccm of zero air through the glass-spiral in all experiments. Trapping behaviour designated “sometimes” means that we could trap the molecules in some of the experiments but not in all and “never” means that this LIM-ONO2 was not trapped. We have no definite explanation for the lack of reproducibility, which may be related to changes in composition of the mixture and effects related to competitive adsorption on the cold glass-surface.
3.3. Interaction of LIM-ONO2 with chamber walls in the SCHARK
As LIM-ONO2 compounds have a high affinity for surfaces, we expect that some fraction is lost to the walls of the SCHARK chamber. Such effects must be corrected for when deriving e.g. production rates through NO3 + limonene chemistry or loss rates due e.g. due photolysis. The ability to trap LIM-ONO2 and re-inject them into a clean chamber in which the production rate is zero, allows us to check for the wall losses of each individual LIM-ONO2.Wall loss rate coefficients (kwall) for LIM-ONO2 were quantified by injecting LIM-ONO2 from the glass-spiral into the SCHARK and monitoring their concentration decrease over time in the dark and in the absence of oxidants (e.g. O3 or NO3). Time dependent LIM-ONO2 signals measured by the HR-ToF-ICIMS decayed exponentially and the time series was fitted to a first-order expression (eqn (1)) to derive the species-specific wall-loss rate constant kwall.
| [LIM-ONO2]t = [LIM-ONO2]0 × exp − (kwall + kexch) t | (1) |
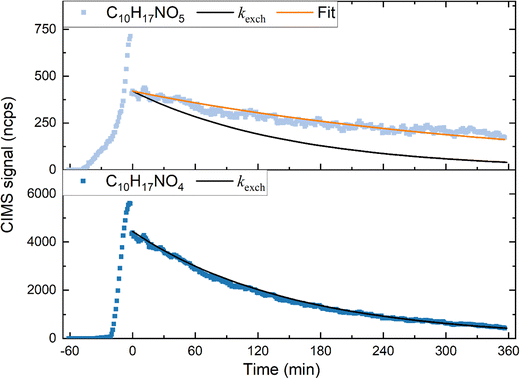
For C10H17NO4 (Fig. 5, lower panel) the observed exponential decay constant in the C10H17NO4 signal is similar to kexch (solid black line), indicating that this compound does not exhibit a measurable wall loss within the uncertainty of the experiment. The behaviour of C10H17NO5 is markedly different: the observed decay constant (kwall + kexch) is significantly smaller than the exchange rate. In the absence of chemical production of C10H17NO5 this can only be explained in terms of multi-phase equilibria, in which C10H17NO5 that has been adsorbed to the wall during the injection phase (i.e. before t = 0) desorbs back into the gas-phase, thereby reducing its overall decay rate. Close inspection of the signal during injection reveals a sharp decrease in signal as soon as the injection stops (e.g. from 700 to 460 ncps for C10H17NO5). This is not a mixing effect but clear evidence for uptake of the LIM-ONO2 to the chamber walls. For C10H17NO4 (lower panel of Fig. 5) the initial, fractional decrease after injection stops is smaller and the loss rate no longer significantly different from kexch. Fig. S7 shows that none of the major LIM-ONO2 species are totally free from wall effects as the decay rates are often lower than kexch. The LIM-ONO2 with the largest signal (C10H17NO4) shows a relatively small initial decrease in signal after injection into the “clean” SCHARK and its long-term behaviour is consistent with a trace gas whose only loss is due to flow out of the chamber. For these reasons, we now focus our analysis on C10H17NO4 for which auxiliary data such as absorption cross sections are available (see below).
3.4. Photolysis and lifetime of the dominant LIM-ONO2 (C10H17NO4)
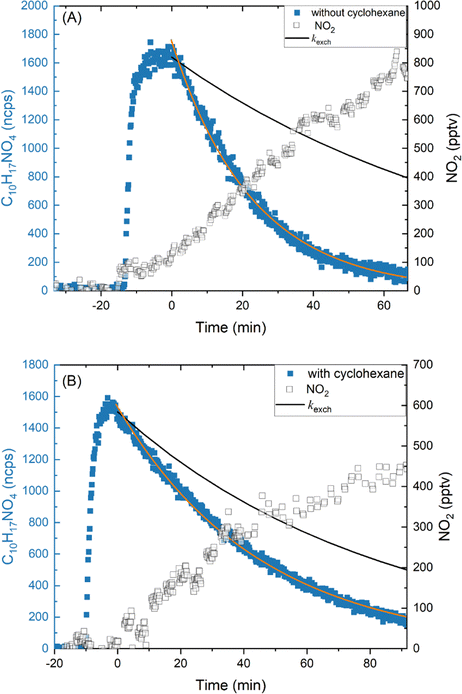
To investigate the behaviour of the various LIM-ONO2 species under irradiation (at a wavelength of 370 ± 13 nm), a cold-trapped sample was transferred from the glass-spiral into the SCHARK by warming rapidly using a water-bath at ∼40–50 °C while passing 500 sccm of zero air through the glass-spiral (see Section 3.2). To examine the potential formation of OH and assess its contribution to the overall loss of LIM-ONO2 during photolysis, experiments were carried out either with or without cyclohexane (120 ppmv) that serves as a OH scavenger.The time profiles for the measured signal intensity for C10H17NO4 under irradiation (from experiments both with and without added cyclohexane) are displayed in Fig. 6 (left y-axis). Following a short period (2 min) after injection of LIM-ONO2 to allow for mixing in the SCHARK, the LEDs were switched on to irradiate the gas-mixture. During irradiation, the NO2 mixing ratio increases as expected if the main process in LIM-ONO2 photolysis is cleavage of the O–NO2 bond. Note however, that NO2 is rapidly photolysed using the LEDs (JNO2 = 14.0 × 10−3 s−1) to form O(3P) and thus O3. No increase in the O3 concentration was observed, which is related to the 2 ppbv detection limit of the ozone-monitor. In addition, the NO2 formed also react with peroxy radicals generated during photolysis (and also from OH scavenging by cyclohexane) to form e.g. peroxy nitrates which will sequester some of the NOx. For this reason, the NO2 profile cannot be analysed to calculate e.g. the relative importance of O–NO2 bond fission in comparison to formation of other photochemical fragments.
A control experiment confirmed that no detectable nitrate-containing products were produced or released from the chamber walls when the LEDs were switched on without prior injection of LIM-ONO2 from the glass-spiral.
In the presence of cyclohexane, the overall loss of C10H17NO4 is by photolysis, flow out of the chamber and wall loss and is given by:
| [C10H17NO4]t = [C10H17NO4]0 × exp − (JC10H17NO4 + kwall + kexch) t | (2) |
This expression does not consider loss processes for C10H17NO4 through reaction with other trace gases. A potential exception to this is the reaction with O3. Although O3 is not present in the initial mixture injected into the SCHARK prior to photolysis, it will be generated via the photolysis of the primary photo-product NO2. The amount of O3 that can be generated per LIM-ONO2 photolysed is a complex function of the recycling of NO via reactions with peroxy radicals:
| LIM-ONO2 + hv → LIM-O + NO2 | (R4) |
| NO2 + hv → NO + O(3P) | (R5) |
| O(3P) + O2 + M → O3 + M | (R6) |
| LIM-O + O2 → RO2 | (R7) |
| RO2 + NO → LIM-O + NO2 | (R8) |
| LIM-ONO2 + O3 → products | (R9) |
For C10H17NO4, the slope of the regression from the experiment with added cyclohexane (JC10H17NO4 + kwall + kexch) was 3.69 × 10−4 s−1. Using the chamber exchange constant for the flow conditions of this experiment (kexch = 2.00 × 10−4 s−1) and the fact that no significant wall loss was observed (kwall + kexch ≈ kexch, see above) we derive JC10H17NO4 = (1.69 ± 0.06) × 10−4 s−1.
In the absence of cyclohexane, C10H17NO4 was lost much more rapidly due to reaction with photochemically formed OH. The loss frequency (after subtracting kexch) was 5.32 (±0.07) × 10−4 s−1. This indicates that a large fraction (0.75) of C10H17NO4 was lost via reaction with OH when cyclohexane was not added. A likely source of OH during irradiation is the photolysis of HONO which absorbs strongly at wavelengths of λ = 360–370 nm and has a SCHARK photolysis frequency of JHONO = 3.5 × 10−3 s−1. HONO was indeed detected (but not quantified) by HR-ToF-ICIMS and is presumably formed from the interaction of NO2 with the chamber surface as has often been documented for Teflon environmental chambers.51
The photolysis frequency of C10H17NO4 (JC10H17NO4) is a function of the photon flux through the SCHARK chamber, the absorption cross sections of C10H17NO4 over the LED emission spectrum (λ = 370 ± 13 nm), and the photodissociation quantum yield (ϕ) at these wavelengths. To put our observed loss frequency on a physical basis we performed chemical actinometry using Cl2 which absorbs at λ = 370 ± 13 nm with known cross sections and quantum yields.
| [Cl2]t = [Cl2]0 × exp − (JCl2 + kexch) t | (3) |
The relative loss rate of C10H17NO4 and Cl2 due to photolysis is given by:
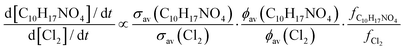 | (4) |
where Ji are photolysis frequencies (s−1). As the same operating parameters and number of LEDs were used to photolyse both Cl2 and C10H17NO4, the last terms on the right-hand side of eqn (4) effectively cancel, so that the relative decay rate of Cl2 and C10H17NO4 is only related to physical constants of the molecules (σ and ϕ). For such experiments, Cl2 represents an ideal actinometer as its absorption cross sections52 and photodissociation quantum yield (unity) are well established.18 However, it is important to avoid secondary chemistry including Cl2 re-combination or loss via reaction with reactive species such as alkyl fragments. For these reasons, experiments to determine JCl2 were conducted in the presence of ∼3 ppmv ethane, which reduces the Cl atom lifetime to ∼0.2 ms and prevents recombination. The C2H5 radicals thus formed were quasi-instantaneously scavenged by O2 (timescale of 30 ns for ∼200 mbar [O2]) to effectively prevent their reaction with Cl2. The C2H5O2 peroxy radicals formed do not react with Cl2.
| Cl2 + hv → 2Cl | (R10) |
| Cl + C2H6 → HCl + C2H5 | (R11) |
| C2H5 + O2 + M → C2H5O2 + M | (R12) |
A mixture of 500 sccm Cl2 (nominal 80 ppmv in N2) and 10 SLM dry zero air was introduced in the chamber (for ∼11 min), together with a single injection of ethane (6 mL of a 48% gas mixture in N2). Resulting in initial mixing ratios of ∼3 ppmv ethane (calculated from volumes and mixing ratio) and 290 ppbv Cl2 (measured by CRDS).
After switching off the Cl2 flow and waiting a few minutes for complete mixing, the LEDs were switched on and the Cl2 mixing ratio decreased exponentially as shown in Fig. S8. In the absence of chemical losses other than photolysis, the Cl2 loss is described by eqn (3). The sum (JCl2 + kexch) was derived by fitting the data shown in Fig. S8 to expression (3). JCl2 was then obtained by subtracting the chamber exchange constant (1.82 × 10−4 s−1), yielding an experimental, effective first-order photolysis frequency of JCl2 = (1.99 ± 0.01) × 10−3 s−1.
This is a factor ∼11.8 larger than the value of JC10H17NO4 = (1.69 ± 0.06) × 10−4 s−1 obtained under quasi-identical conditions (i.e. f was unchanged).
At this point in the analysis, we have derived:
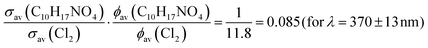 | (5) |
If we now assume that the photodissociation quantum yield for C10H17NO4 is unity at all wavelengths emitted by the LEDs (i.e. the same quantum yield as Cl2) the 2nd term on the left-hand side of eqn (5) disappears. In this case, the value of 0.085 represents the ratio of the absorption cross sections of C10H17NO4 and Cl2 weighted over the LED emission spectrum. The Cl2 absorption cross section averaged over the region of overlap with the LED emission is σav(Cl2) = 7.90 × 10−20 cm2 molecule−1, which yields a weighted average absorption cross section for C10H17NO4 over the same wavelength range (λ = 355–400 nm) of σav (C10H17NO4) = 6.72 × 10−21 cm2 molecule−1.
This cross section can be compared to the spectrum of C10H17NO4 reported by Wang et al.31 (blue datapoints in Fig. 7), but with some caveats. Firstly, the spectrum reported by Wang et al.31 was that of a sample dissolved in ether, which causes a solvatochromatic wavelength shift of unknown magnitude compared to the gas-phase. For this reason, Wang et al.31 used ethyl ether as solvent because of its low polarity, which reduces the magnitude of the wavelength shift. Secondly, the spectrum of Wang et al.31 covers the range λ = 200–367 nm and thus does not extend through the full range of wavelengths emitted by our LEDs (λ = 355–400 nm). To account for the second limitation, we have extrapolated the Wang et al.31 spectrum to longer wavelengths assuming an exponential decrease in cross section with increasing wavelength. Here we also consider two different scenarios. In the first, (Extrapolation 1) we extend the observed exponential decrease in cross section observed by Wang et al.31 between λ = 330 and 365 nm (dashed line in Fig. 7). As apparent from the log-plot, the discontinuation in the slope between 310 and 365 nm compared to shorter wavelengths may indicate a contribution from a different electronic transition. Alternatively, this could reflect a solvent effect or artefacts associated with low optical density and stray light at longer wavelengths. For this reason, in the second scenario (Extrapolation 2), we assume that the second feature in the absorption spectrum between ∼λ = 310 and 365 nm is an artefact and we instead extend the exponential decrease in cross section between 260 and 310 nm to longer wavelengths. This behaviour is described by the solid blue line in Fig. 7 and leads to significantly lower cross sections at the longer wavelengths where our LEDs emit.
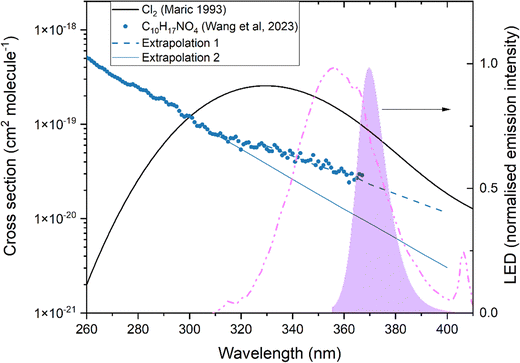 | ||
| Fig. 7 Absorption spectra of Cl252 and C10H17NO4 (dissolved in ether) reported by Wang et al.31 The solid and dashed lines are different extrapolations of the Wang et al. spectrum to longer wavelengths. The normalised LED emission spectra of the photolysis light sources used are also shown (right y-axis) with our LED emission spectrum the purple shaded area and the “black-lights” used by Wang et al. as a broken, purple line. The average cross-section, weighted by the LED emission spectrum in the same spectral region is either 2.27 × 10−20 cm2 molecule−1 (Extrapolation 1) or 8.57 × 10−21 cm2 molecule−1 (Extrapolation 2). | ||
Using either scenario we can calculate the average cross section, weighted by the emission of our LEDs. Using scenario Extrapolation 1 (dotted line), we obtain σav (355–400 nm) = 2.27 × 10−20 cm2 molecule−1, whereas under scenario Extrapolation 2 the cross sections are reduced to σav (355–400 nm) = 8.57 × 10−21 cm2 molecule−1. The latter value is in reasonable agreement (within 25%) with that derived from our photolysis experiments and assuming that the quantum yield is unity (σ = 6.72 × 10−21 cm2 molecule−1).
Alternatively, by inserting the (extrapolated) average cross sections of Wang et al. into eqn (6) we can derive the effective quantum yield (ϕav) over this wavelength range. Under scenario Extrapolation 1, we obtain ϕav (355–400 nm) = 0.30 and under scenario Extrapolation 2 we obtain ϕav (355–400 nm) = 0.78 at 1 bar and 298 K. The quantum yield reported by Wang et al.31 are much smaller with a value of 0.071 ± 0.035 and a chamber photolysis lifetime of C10H17NO4 of 21 h. In contrast, we measured a chamber photolysis lifetime due to photodissociation of 1.6 h for C10H17NO4.
In order to understand these differences in chamber lifetimes and derived quantum yields we first compare the emission spectra (normalised to λmax) of the lamps used to photolyse limonene nitrates in both studies, which are plotted in Fig. 7. We see that the emission of the “black-lights” used by Wang et al. is broader (extending from ∼320 to 400 nm) with a FWHM of ∼40 nm and thus (compared to the LEDs) has more extensive overlap with the absorption spectrum of C10H17NO4 at shorter wavelengths. As photodissociation quantum yields often decrease at longer wavelengths (lower photon energies) the difference in emission wavelengths cannot be the cause of the lower quantum yields observed by Wang et al. There are also large differences in the photon flux in both chambers. Wang et al.31 report (in their SI) a photolysis frequency for NO2 in their chamber of 2.1 × 10−3 s−1 which was obtained by photolysing NO2 and modelling its loss (which is partially defined by reformation in the NO + O3 reaction). We prefer to calculate JNO2 from the accurately measured value of JCl2 (1.99 × 10−3 s−1) and the relative overlap of the LEDs with the well-known spectra of Cl2 and NO2 and associated quantum yields (both unity over the entire wavelength range of our LEDs). Our value of JNO2 = 14.0 × 10−3 s−1 is a factor ∼7 larger than that in the Wang et al. chamber and helps explain why we see almost complete loss of C10H17NO4 within 80 min (Fig. 6) whereas Wang et al. observe a depletion of only 40% over a period of >4 hours (their Fig. 2), despite the better overlap between lamp emission and the UV absorption spectrum of C10H17NO4.
A further explanation for the derivation of such different quantum yields is that we are actually dealing with different molecules of the formula C10H17NO4 that are detected (in both Wang et al. and in this work) as clusters with I−. Should this be the case, our analysis, which uses the (extrapolated) cross-sections from Wang et al. is inappropriate. As the C10H17NO4 molecule in this work was generated in the reaction of NO3 with limonene and not by wet-synthetic methods as in Wang et al., we cannot rule this out. However, in the absence of a further analytical tool (e.g. gas chromatography) which to compare both samples, this remains purely speculative.
In summary, we observe that C10H17NO4 is a photolabile organic nitrate formed in the NO3-initiated oxidation of limonene, even when irradiated at the relatively long wavelengths emitted by our LEDs (λ = 355–400 nm) and that our data are compatible with a large quantum yield (possibly unity) if our extrapolation of the C10H17NO4 spectrum reported by Wang et al.31 is appropriate.
By convoluting the extrapolated C10H17NO4 spectrum (Scenario: Extrap 2) with the solar spectral photon flux (measured on a cloud free day (23. July) at a Latitude of 35° and Longitude 32°) and a quantum yield of 0.78 we can derive a diel profile for JC10H17NO4 (Fig. S9). The maximum photolysis frequency is ∼2.2 × 10−4 s−1 (at 10 UTC), which translates to a lifetime of 75 minutes. The average lifetime (over the entire diel profile) is 3.2 h and thus comparable to the deposition lifetimes of organic nitrates from BVOC.9,14 In Fig. S9 we also display the atmospheric photolysis frequency using our actinic flux measurements but using the Wang et al.31 cross sections (without extrapolation to longer wavelengths) and their quantum yield (0.071). The maximum photolysis frequency thus obtained is 2.8 × 10−5 s−1 which is very similar to their reported value (3.0 ± 1.5 × 10−5 s−1) obtained using the TUV model for a solar zenith angle of 28.14° (12:00 solar time, 1 August at 33.7° Latitude north (Atlanta, overhead O3 column 300 Dobson units and albedo 0.1). The factor 7.8 larger value obtained in this work is thus clearly related to the larger quantum yield (0.78 from this work and 0.071 by Wang et al.31).
3.5. Photolysis of C10H16N2O6
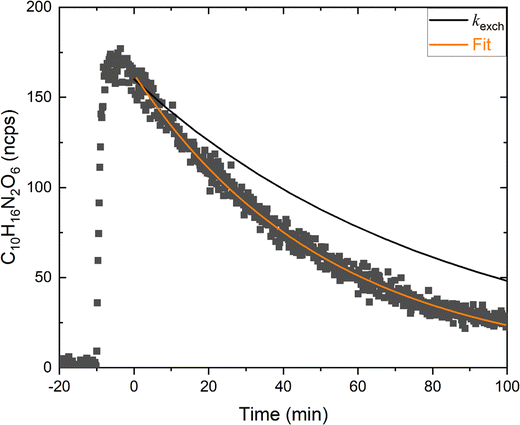
A simpler analysis was performed for the di-nitrate (C10H16N2O6), whose temporal evolution under irradiation is shown in Fig. 8. Similar to the analysis above, in the presence of cyclohexane, we assume that the loss of C10H16N2O6 occurs only by photolysis and chamber outflow and is described by:| [C10H16N2O6]t = [C10H16N2O6]0 × exp − (JC10H16N2O6 + kexch) t | (6) |
For C10H16N2O6, the fit to the data according to eqn (6) results in a loss constant in the presence of cyclohexane of 3.22 × 10−4 s−1. Using the chamber exchange constant (kexch = 2.00 × 10−4 s−1) we derive JC10H16N2O6 = (1.22 ± 0.06) × 10−4 s−1. This value is lower than that obtained for the mono-nitrate C10H17NO4 (JC10H17NO4 = (1.69 ± 0.06) × 10−4 s−1), indicating a slower photochemical loss (photolysis lifetime = 2.3 h) of the di-nitrate at the wavelengths emitted by our LEDs (λ = 355–400 nm). However, in the absence of an absorption spectrum for C10H16N2O6, the available data do not allow a more detailed quantitative interpretation of this difference and we can only conclude that, like C10H17NO4, the lifetime of C10H16N2O6 is strongly influenced by its photolysis.
3.6. Other LIM-ONO2
Fig. S10 shows a time series of 13 LIM-ONO2 species and NO2. During the irradiation period (yellow shaded area) in the presence of cyclohexane, we observe that the mixing ratios of the LIM-ONO2 species with the strongest HR-ToF-ICIMS signal (C10H17NO4) decrease, as does that of the di-nitrate C10H16N2O6, both of which are treated in detail above. In contrast to the photolytic loss of C10H17NO4 and C10H16N2O6, the mixing ratios of several shorter-chain nitrates (C2-9) increase during illumination as do those of some C10 nitrates (i.e. C10H13NO5, C10H15NO5, C10H17NO6). The formation of shorter-chain nitrates appears to be consistent with the observations of Takeuchi et al.53 who reported their production during photolysis of organic nitrates formed from α-pinene and β-pinene oxidation by the OH and NO3 radicals. The formation of shorter chain nitrates could be understood if dissociative absorption leads not only to O–NO2 bond fission but also to fission of a C(O)–C bond or if secondary reactions of the alkoxy radical formed in the primary photolysis step undergo further reactions that result in C–C bond fission. The increase in the signals of several C10-nitrates (notably C10H15NO5, C10H15NO6 and C10H15NO7) can be explained by reactions of an initially formed alkoxy radical with O2 to form a peroxy radical that can reform a nitrate via reaction with NO. Indeed, the main product of C10H17NO4 photolysis observed by Wang et al.31 was C10H17NO5. Which was attributed to formation of the C10H17O2 alkoxy radical and its decomposition and reaction with O2 in the presence of NO. The photolytic formation of C10-nitrates is potentially also explained if the RO2 + RO2 reactions that produce the nitrated products described above also form peroxides (C10 “dimers”) of the form ROOR (C20), which can dissociate by O–O bond fission. These ROOR have very low volatility and are likely to stick to the walls of the chamber or form particles rather than reside in the gas-phase. Previous studies have observed the formation of e.g. C20 dimers from the reaction of limonene with NO3 radicals, primarily as highly oxygenated organic molecules in both the gas and particle phases.23,24 The fact that we did not observe particles does not rule out the presence of very small particles formed by coagulation of ROOR type molecules and not detected by the CPC. ROOR dimers cannot be reliably identified with our HR-ToF-ICIMS due to the limited mass calibration range and the low sensitivity to C20 compounds. However, their low volatility means they would be collected efficiently in the cold-trap and re-injected into the SCHARK chamber. ROOR formed from NO3 + limonene are likely reservoirs of C10 nitrates in our experiments, and their photolysis (either in the gas-phase or adsorbed on the chamber walls) is a potential explanation for some of the observed formation of C10 nitrates under irradiation in the SCHARK chamber.4. Conclusions
In this study, we investigated the formation, isolation and photolysis of limonene-derived organic nitrates under controlled chamber conditions. Multifunctional organic nitrates formed from the reaction of limonene with NO3 were successfully trapped at −30 °C, without co-condensation of limonene, allowing their re-injection into the chamber for photochemical investigation. By comparing the total multifunctional alkyl nitrates (in pptv) measured by the TD-CRDS with the summed HR-ToF-ICIMS signals (in ncps) of the individual organic nitrates and assuming equal sensitivity of the HR-ToF-ICIMS to all detected LIM-ONO2, we estimated the HR-ToF-ICIMS sensitivity as 21 ncps pptv−1.Our experimental approach further enabled the determination of in-chamber (λ = 355–400 nm) photolysis frequencies of (1.69 ± 0.06) × 10−4 s−1 and (1.22 ± 0.06) × 10−4 s−1 for C10H17NO4 and C10H16N2O6, respectively. Photolysis of the re-injected limonene-derived organic nitrates led to the formation of shorter-chain nitrates (and an increase in NO2). Using a chemical actinometer (Cl2), with a photolysis frequency of (1.99 ± 0.007) × 10−3 s−1 under our irradiation conditions we derived JC10H17NO4/JCl2 = 0.085. From this ratio and assuming a quantum yield of unity, we derive a wavelength-averaged absorption cross section of 6.72 × 10−21 cm2 molecule−1 for C10H17NO4 which agrees to within 25% with the cross section extrapolated from the solution-phase spectrum reported by Wang et al.31 Alternatively, using different extrapolations of the Wang et al. cross-sections, we could estimate the quantum yield to be between 0.3 and 0.8. Our results demonstrate that C10H17NO4 and C10H16N2O6 are photolabile limonene-derived nitrates that undergo photochemical loss at near-UV wavelengths. The measured photolysis lifetimes of 1.6 h and 2.3 h for C10H17NO4 and C10H16N2O6, respectively, under chamber conditions indicates much faster degradation than previously reported for C10H17NO4.31 For ambient conditions we derived an averaged diel photolysis frequency of C10H17NO4 of 8.5 × 10−5 s−1 (corresponding to a photolysis lifetime of 3.2 h) which is comparable to other physical sinks (e.g. dry deposition).
Author contributions
Conceptualization: JNC. Data curation: LW, LM, EJ, JS. Formal analysis: LW. Funding acquisition: JL, UP, JNC. Methodology: LW, JNC. Project administration: JNC. Resources: FD, JW, UP, JNC, JL. Supervision: PD, JNC. Validation: JNC. Visualization: LW. Writing – original draft: LW. Writing – review & editing: LW, LM, PD, EJ, JS, JW, FD, UP, JL, JNC.Conflicts of interest
There are no conflicts to declare.Data availability
The data underlying the figures in this publication are available at the Max Planck repository (EDMOND) at https://doi.org/10.17617/3.NHP25R.Supplementary information is available. See DOI: https://doi.org/10.1039/d6cp00964f.
Acknowledgements
LM was funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) – TRR 301 “TPChange” (Project-ID 428312742). PD gratefully acknowledges funding from the Deutsche Forschungsgemeinschaft (project “MONOTONS”, project number: 522970430). Open Access funding provided by the Max Planck Society.References
- J. Williams, Environ. Chem., 2004, 1, 125 CrossRef CAS.
- A. B. Guenther, X. Jiang, C. L. Heald, T. Sakulyanontvittaya, T. Duhl, L. K. Emmons and X. Wang, Geosci. Model Dev., 2012, 5, 1471 CrossRef.
- N. L. Ng, S. S. Brown, A. T. Archibald, E. Atlas, R. C. Cohen, J. N. Crowley, D. A. Day, N. M. Donahue, J. L. Fry, H. Fuchs, R. J. Griffin, M. I. Guzman, H. Herrmann, A. Hodzic, Y. Iinuma, J. L. Jimenez, A. Kiendler-Scharr, B. H. Lee, D. J. Luecken, J. Mao, R. McLaren, A. Mutzel, H. D. Osthoff, B. Ouyang, B. Picquet-Varrault, U. Platt, H. O. T. Pye, Y. Rudich, R. H. Schwantes, M. Shiraiwa, J. Stutz, J. A. Thornton, A. Tilgner, B. J. Williams and R. A. Zaveri, Atmos. Chem. Phys., 2017, 17, 2103 CrossRef CAS PubMed.
- D. Mogensen, R. Gierens, J. N. Crowley, P. Keronen, S. Smolander, A. Sogachev, A. C. Nölscher, L. Zhou, M. Kulmala, M. J. Tang, J. Williams and M. Boy, Atmos. Chem. Phys., 2015, 15, 3909 CrossRef CAS.
- J. M. Liebmann, G. Schuster, J. B. Schuladen, N. Sobanski, J. Lelieveld and J. N. Crowley, Atmos. Meas. Tech., 2017, 10, 1241 CrossRef CAS.
- R. P. Wayne, I. Barnes, P. Biggs, J. P. Burrows, C. E. Canosa-Mas, J. Hjorth, G. Le Bras, G. K. Moortgat, D. Perner, G. Poulet, G. Restelli and H. Sidebottom, Atmos. Environ., Part A, 1991, 25A, 1 CrossRef CAS.
- P. O. Wennberg, K. H. Bates, J. D. Crounse, L. G. Dodson, R. C. McVay, L. A. Mertens, T. B. Nguyen, E. Praske, R. H. Schwantes, M. D. Smarte, J. M. St Clair, A. P. Teng, X. Zhang and J. H. Seinfeld, Chem. Rev., 2018, 118, 3337 CrossRef CAS PubMed.
- J. M. Roberts, Atmos. Environ., Part A, 1990, 24, 243 CrossRef.
- S. T. Andersen, R. Sander, P. Dewald, L. Wüst, T. Seubert, G. N. T. E. Türk, J. Schuladen, M. R. McGillen, C. Xue, A. Mellouki, A. Kukui, V. Michoud, M. Cirtog, M. Cazaunau, A. Bauville, H. Bouzidi, P. Formenti, C. Denjean, J. C. Etienne, O. Garrouste, C. Cantrell, J. Lelieveld and J. N. Crowley, Atmos. Chem. Phys., 2025, 25, 5893 CrossRef CAS.
- A. Zare, P. S. Romer, T. Nguyen, F. N. Keutsch, K. Skog and R. C. Cohen, Atmos. Chem. Phys., 2018, 18, 15419 CrossRef CAS.
- E. C. Browne, K. E. Min, P. J. Wooldridge, E. Apel, D. R. Blake, W. H. Brune, C. A. Cantrell, M. J. Cubison, G. S. Diskin, J. L. Jimenez, A. J. Weinheimer, P. O. Wennberg, A. Wisthaler and R. C. Cohen, Atmos. Chem. Phys., 2013, 13, 4543 CrossRef.
- J. A. Fisher, D. J. Jacob, K. R. Travis, P. S. Kim, E. A. Marais, C. Chan Miller, K. Yu, L. Zhu, R. M. Yantosca, M. P. Sulprizio, J. Mao, P. O. Wennberg, J. D. Crounse, A. P. Teng, T. B. Nguyen, J. M. Clair, R. C. Cohen, P. Romer, B. A. Nault, P. J. Wooldridge, J. L. Jimenez, P. Campuzano-Jost, D. A. Day, W. Hu, P. B. Shepson, F. Xiong, D. R. Blake, A. H. Goldstein, P. K. Misztal, T. F. Hanisco, G. M. Wolfe, T. B. Ryerson, A. Wisthaler and T. Mikoviny, Atmos. Chem. Phys., 2016, 16, 5969 CrossRef CAS PubMed.
- J. Liebmann, E. Karu, N. Sobanski, J. Schuladen, M. Ehn, S. Schallhart, L. Quéléver, H. Hellen, H. Hakola, T. Hoffmann, J. Williams, H. Fischer, J. Lelieveld and J. N. Crowley, Atmos. Chem. Phys., 2018, 3799 CrossRef CAS.
- J. Liebmann, N. Sobanski, J. Schuladen, E. Karu, H. Hellén, H. Hakola, Q. Zha, M. Ehn, M. Riva, L. Heikkinen, J. Williams, H. Fischer, J. Lelieveld and J. N. Crowley, Atmos. Chem. Phys., 2019, 19, 10391 CrossRef CAS.
- P. Dewald, T. Seubert, S. T. Andersen, G. N. T. E. Türk, J. Schuladen, M. R. McGillen, C. Denjean, J. C. Etienne, O. Garrouste, M. Jamar, S. Harb, M. Cirtog, V. Michoud, M. Cazaunau, A. Bergé, C. Cantrell, S. Dusanter, B. Picquet-Varrault, A. Kukui, C. Xue, A. Mellouki, J. Lelieveld and J. N. Crowley, Atmos. Chem. Phys., 2024, 24, 8983 CrossRef CAS.
- H. Hellén, A. P. Praplan, T. Tykkä, I. Ylivinkka, V. Vakkari, J. Bäck, T. Petäjä, M. Kulmala and H. Hakola, Atmos. Chem. Phys., 2018, 18, 13839 CrossRef.
- D. F. McGlynn, L. E. R. Barry, M. T. Lerdau, S. E. Pusede and G. Isaacman-VanWertz, Atmos. Chem. Phys., 2021, 21, 15755 CrossRef CAS.
- M. Ammann, R. A. Cox, J. N. Crowley, H. Herrmann, M. E. Jenkin, V. F. McNeill, A. Mellouki, M. J. Rossi, J. Troe and T. J. Wallington, IUPAC, IUPAC Task Group on Atmospheric Chemical Kinetic Data Evaluation, Last access March 2026, https://iupac.aeris-data.fr/.
- J. L. Fry, A. Kiendler-Scharr, A. W. Rollins, T. Brauers, S. S. Brown, H. P. Dorn, W. P. Dube, H. Fuchs, A. Mensah, F. Rohrer, R. Tillmann, A. Wahner, P. J. Wooldridge and R. C. Cohen, Atmos. Chem. Phys., 2011, 11, 3879 CrossRef CAS.
- M. Hallquist, I. Wangberg, E. Ljungstrom, I. Barnes and K. H. Becker, Environ. Sci. Technol., 1999, 33, 553 CrossRef CAS.
- A. Mutzel, Y. Zhang, O. Böge, M. Rodigast, A. Kolodziejczyk, X. Wang and H. Herrmann, Atmos. Chem. Phys., 2021, 21, 8479 CrossRef CAS.
- M. P. DeVault, A. C. Ziola and P. J. Ziemann, ACS Earth Space Chem., 2022, 6, 2076 CrossRef CAS.
- Y. Guo, H. Shen, I. Pullinen, H. Luo, S. Kang, L. Vereecken, H. Fuchs, M. Hallquist, I. H. Acir, R. Tillmann, F. Rohrer, J. Wildt, A. Kiendler-Scharr, A. Wahner, D. Zhao and T. F. Mentel, Atmos. Chem. Phys., 2022, 22, 11323 CrossRef CAS.
- C. Faxon, J. Hammes, M. Le Breton, R. K. Pathak and M. Hallquist, Atmos. Chem. Phys., 2018, 18, 5467 CrossRef CAS.
- R. Mayorga, Y. Xia, Z. Zhao, B. Long and H. Zhang, Environ. Sci. Technol., 2022, 56, 15337 CrossRef CAS PubMed.
- F. L. Z. Xiong, C. H. Borca, L. V. Slipchenko and P. B. Shepson, Atmos. Chem. Phys., 2016, 16, 5595 CrossRef CAS.
- J. F. Muller, J. Peeters and T. Stavrakou, Atmos. Chem. Phys., 2014, 14, 2497 CrossRef.
- K. C. Clemitshaw, J. Williams, O. V. Rattigan, D. E. Shallcross, K. S. Law and R. Anthony Cox, J. Photochem. Photobiol., A, 1997, 102, 117 CrossRef.
- J. M. Roberts and R. W. Fajer, Environ. Sci. Technol., 1989, 23, 945 CrossRef CAS.
- I. Barnes, K. H. Becker and T. Zhu, J. Atmos. Chem., 1993, 17, 353 CrossRef CAS.
- Y. Wang, M. Takeuchi, S. Wang, S. A. Nizkorodov, S. France, G. Eris and N. L. Ng, J. Phys. Chem. A, 2023, 127, 987 CrossRef CAS PubMed.
- Y. Wang, I. R. Piletic, M. Takeuchi, T. Xu, S. France and N. L. Ng, Environ. Sci. Technol., 2021, 55, 14595 CrossRef CAS PubMed.
- R. Suarez-Bertoa, B. Picquet-Varrault, W. Tamas, E. Pangui and J. F. Doussin, Environ. Sci. Technol., 2012, 46, 12502 CrossRef CAS PubMed.
- B. Picquet-Varrault, R. Suarez-Bertoa, M. Duncianu, M. Cazaunau, E. Pangui, M. David and J. F. Doussin, Atmos. Chem. Phys., 2020, 20, 487 CrossRef CAS.
- L. Wüst, P. Dewald, G. N. T. E. Türk, J. Lelieveld and J. N. Crowley, Atmos. Meas. Tech., 2025, 18, 1943 CrossRef.
- P. Dewald, R. Dörich, J. Schuladen, J. Lelieveld and J. N. Crowley, Atmos. Meas. Tech., 2021, 14, 5501 CrossRef CAS.
- B. H. Lee, F. D. Lopez-Hilfiker, C. Mohr, T. Kurten, D. R. Worsnop and J. A. Thornton, Environ. Sci. Technol., 2014, 48, 6309 CrossRef CAS PubMed.
- Y. Ji, L. G. Huey, D. J. Tanner, Y. R. Lee, P. R. Veres, J. A. Neuman, Y. Wang and X. Wang, Atmos. Meas. Tech., 2020, 13, 3683 CrossRef CAS.
- L. Moorman, J. N. Crowley, F. Fachinger and F. Drewnick, Environ. Sci.: Atmos., 2026, 6, 579 Search PubMed.
- N. Sobanski, M. J. Tang, J. Thieser, G. Schuster, D. Pöhler, H. Fischer, W. Song, C. Sauvage, J. Williams, J. Fachinger, F. Berkes, P. Hoor, U. Platt, J. Lelieveld and J. N. Crowley, Atmos. Chem. Phys., 2016, 16, 4867 CrossRef CAS.
- A. C. Vandaele, C. Hermans, P. C. Simon, M. Carleer, R. Colin, S. Fally, M. F. Merienne, A. Jenouvrier and B. Coquart, J. Quant. Spectrosc. Radiat. Transfer, 1998, 59, 171 CrossRef CAS.
- N. Sobanski, J. Schuladen, G. Schuster, J. Lelieveld and J. N. Crowley, Atmos. Meas. Tech., 2016, 9, 5103 CrossRef CAS.
- J. A. Davidson, A. A. Viggiano, C. J. Howard, I. Dotan, F. C. Fehsenfeld, D. L. Albritton and E. E. Ferguson, J. Chem. Phys., 1978, 68, 2085 CrossRef CAS.
- N. M. Donahue, S. A. Epstein, S. N. Pandis and A. L. Robinson, Atmos. Chem. Phys., 2011, 11, 3303 CrossRef CAS.
- Y. Li, U. Pöschl and M. Shiraiwa, Atmos. Chem. Phys., 2016, 16, 3327 CrossRef CAS.
- B. R. Ayres, H. M. Allen, D. C. Draper, S. S. Brown, R. J. Wild, J. L. Jimenez, D. A. Day, P. Campuzano-Jost, W. Hu, J. de Gouw, A. Koss, R. C. Cohen, K. C. Duffey, P. Romer, K. Baumann, E. Edgerton, S. Takahama, J. A. Thornton, B. H. Lee, F. D. Lopez-Hilfiker, C. Mohr, P. O. Wennberg, T. B. Nguyen, A. Teng, A. H. Goldstein, K. Olson and J. L. Fry, Atmos. Chem. Phys., 2015, 15, 13377 CrossRef CAS.
- M. Takeuchi, T. Berkemeier, G. Eris and N. L. Ng, Nat. Commun., 2022, 13, 7883 CrossRef CAS PubMed.
- S. Aggarwal, P. Bansal, Y. Wang, S. Jorga, G. Macgregor, U. Rohner, T. Bannan, M. Salter, P. Zieger, C. Mohr and F. Lopez-Hilfiker, Atmos. Meas. Tech., 2025, 18, 4227 CrossRef CAS.
- F. D. Lopez-Hilfiker, S. Iyer, C. Mohr, B. H. Lee, E. L. D'Ambro, T. Kurtén and J. A. Thornton, Atmos. Meas. Tech., 2016, 9, 1505 CrossRef CAS.
- E. Bourtsoukidis, F. Helleis, L. Tomsche, H. Fischer, R. Hofmann, J. Lelieveld and J. Williams, Atmos. Meas. Tech., 2017, 10, 5089 CrossRef CAS.
- F. Rohrer, B. Bohn, D. Brüning, F.-J. Johnen, A. Wahner and J. Kleffmann, ACP, 2005, 5, 2189 CrossRef CAS.
- D. Maric, J. P. Burrows, R. Meller and G. K. Moortgat, J. Photochem. Photobiol., A, 1993, 70, 205 CrossRef CAS.
- M. Takeuchi, Y. Wang and N. L. Ng, ACS EST Air, 2025, 2, 2445 CrossRef CAS PubMed.
| This journal is © the Owner Societies 2026 |