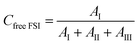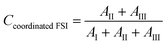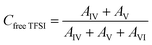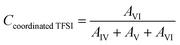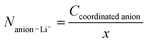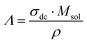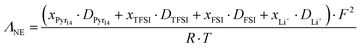The impact of anion mixing on Li+ coordination and transport properties in pyrrolidinium-based ionic liquid electrolytes
Received
30th January 2026
, Accepted 22nd April 2026
First published on 23rd April 2026
Abstract
A series of pyrrolidinium-based ionic liquid electrolytes were investigated to explore the potential benefits of incorporating mixed anions, such as bis(fluorosulfonyl)imide and bis(trifluoromethylsulfonyl)imide used in this study. The effect of mixing anions on fundamental physicochemical properties was investigated, with particular emphasis on transport properties and lithium-ion coordination. More specifically, the electrolytes were systematically characterized in terms of phase behavior, density, viscosity, transport properties (ionic conductivity and self-diffusion), and intermolecular interactions. The results show that mixing anions modifies the nature of intermolecular interactions, which, in turn, alters the coordination of Li+ with its counterions. In the case of the (LiFSI)0.2(Pyr14TFSI)0.8 sample, this resulted in improved thermal and transport properties, anticipating an improved performance in lithium-ion batteries.
1 Introduction
Ionic liquids (ILs) are a unique class of compounds that consist of (ideally) only ions and have a melting point below 100 °C.1 Notably, by selecting a specific combination of anions and cations, the physico-chemical properties of an ionic liquid can be tuned to suit specific applications.2 Furthermore, compared to conventional solvents, ILs exhibit broader electrochemical stability windows, good ionic conductivity, high thermal stability, non-flammability and low volatility. Consequently, they have been extensively studied for various applications, such as catalysis,3 gas separation,4–6 and energy storage devices, including lithium-ion batteries.7–9
Among the vast variety of ionic liquids, those based on pyrrolidinium have been extensively studied for lithium-ion batteries due to their capacity to support lithium electrochemistry and promote the formation of a stable solid electrolyte interphase (SEI).10,11 However, a key challenge in using ionic liquids in Li-ion batteries is their limited transport properties (i.e., slow Li+ transport) and strong Li+–anion interactions. Several strategies have been considered to address this issue, mainly aiming to enhance the diffusivity of the lithium ions. These include the use of molecular additives,12 eutectic mixtures,13,14 and binary/ternary ionic liquid systems.15–19 Appetecchi et al. investigated a mixture of N-methyl-N-propyl pyrrolidinium bis(fluorosulfonyl)imide (Pyr13FSI) and N-butyl-N-propyl pyrrolidinium bis(trifluoromethanesulfonyl)imide (Pyr14TFSI) ILs with 0.3 M of either lithium hexafluorophosphate (LiPF6) or lithium bis(trifluoromethanesulfonyl)imide (LiTFSI) salts. Their findings demonstrated enhanced ionic conductivity in ternary systems compared to binary ones, with a stronger effect at low temperatures.20 Zhou et al. examined the phase behavior of the same systems, reporting the absence of phase transitions beyond a molar ratio of 0.3 with either salt.21,22 Moreover, Bayley et al. studied ternary systems having Pyr13 as the common cation, paired with either FSI or TFSI anions over a broad compositional range in the presence of a lithium salt (0.5 m). Their results showed increased diffusivity of Li and TFSI ions upon addition of FSI.23 Nádherná et al. investigated the electrochemical performance, in Li-ion batteries, of binary systems containing 0.7 M LiFSI or LiTFSI in Pyr14TFSI. The authors reported higher charge/discharge capacities and improved electrochemical stability in the LiFSI/Pyr14TFSI mixed anion system.24 Similarly, Lahiri et al. observed comparable results in a 1 M LiTFSI/Pyr14FSI system.25
To strengthen the understanding of the molecular interactions and transport mechanisms in these mixed-anion ionic liquid electrolytes, a multi-technique approach was employed to investigate mixtures of Pyr14-based ionic liquids and lithium salts. An in-depth analysis of their phase behavior, transport properties, Li-ion coordination and intermolecular interactions was conducted, providing a side-by-side explanation of how Li+ mobility is related to Li-ion coordination.
2 Experimental details
2.1 Chemicals
The lithium bis(trifluoromethylsulfonyl)imide (LiTFSI) (99.9%) and lithium bis(fluorosulfonyl)imide (LiFSI) (99.9%) salts, along with 1-butyl-1-methylpyrrolidinium bis(fluorosulfonyl)imide (Pyr14FSI) (99.9%) and 1-butyl-1-methylpyrrolidinium bis(trifluoromethylsulfonyl)imide (Pyr14TFSI) (99.9%) ionic liquids, were purchased from Solvionic. The molecular structures of the ions found in the ionic liquids and in the salts are shown in Fig. 1. All compounds were dried under vacuum in a Büchi oven at 353 K for 12 hours and then stored in an argon-filled glovebox, before mixing and investigations.
 |
| | Fig. 1 Molecular structure of the 1-butyl-1-methylpyrrolidinium (Pyr14) and lithium (Li) cations and the bis(fluorosulfonyl)imide (FSI) and the bis(trifluoromethylsulfonyl)imide (TFSI) anions. | |
2.2 Preparation
Samples were prepared by mixing ionic liquids and Li salts in a 0.2LiA1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.8Pyr14A2 molar ratio, where A1 and A2 represent the FSI or the TFSI anion. All samples were prepared and stored in an argon-filled glovebox with O2 and H2O levels below 1 ppm. Throughout the following text, the samples are named (LiA1)0.2(Pyr14A2)0.8.
0.8Pyr14A2 molar ratio, where A1 and A2 represent the FSI or the TFSI anion. All samples were prepared and stored in an argon-filled glovebox with O2 and H2O levels below 1 ppm. Throughout the following text, the samples are named (LiA1)0.2(Pyr14A2)0.8.
2.3 Methods
2.3.1 Differential scanning calorimetry (DSC). DSC measurements were performed using a DSC 250 from TA Instruments. Samples were placed in hermetic aluminum pans and sealed in an argon-filled glovebox. DSC thermograms were collected under helium flow (50 mL min−1), over the temperature range of 123 K to 373 K with heating and cooling rates of 5 K min−1. Two scans were recorded for each measurement. The glass transition temperatures (Tg) were extracted from the second heating scan curves using sigmoid functions and the identification of the inflection points.
2.3.2 Viscosity and density. Viscosity and density measurements were carried out using an Anton Paar DMA 4500 M oscillation U-tube densitometer over the temperature range of 293 K to 353 K in 10 K increments. The density accuracy is about ±0.0001 g cm−3. The viscosity was determined using a Lovis 2000 ME rolling ball viscometer module, using which the dynamic viscosity (μ) was estimated from the density of the fluid (ρ) and the velocity of the falling ball (ν), as μ = ρ·ν. The viscosity measurement has an accuracy of up to 0.1% and a repeatability of 0.5%. The temperature accuracy is ±0.02 K with a repeatability of ±0.005 K.
2.3.3 Dielectric spectroscopy. Conductivity data were collected using a novocontrol GmBH broadband dielectric spectrometer, a parallel plate sample cell with stainless steel electrodes and a 3 mm spacer. Measurements were conducted in the frequency range of 10−2–107 Hz and the temperature range of 173–373 K. Data were collected every 10 K with a stabilization time of 180 s at each temperature. From three repeated measurements of the same solution, a value for the standard deviation has been estimated.
2.3.4 Nuclear magnetic resonance diffusometry. NMR experiments were performed using an AVANCE III HD Bruker NMR spectrometer, operating at 14.1 T and equipped with a Diff30 probe connected to a 60 A gradient amplifier. RF coil inserts of a 5 mm 1H/2H double coil, a 5 mm 19F single coil, and a 5 mm 7Li single coil configuration were used. The self-diffusion coefficients of the different species, i.e., Pyr14 (1H), FSI/TFSI (19F) and Li cation (7Li), were determined at five different temperatures in the range of 298–353 K. A 5 mm NMR tube was filled (∼1.5 cm) with the liquid sample and sealed in the glovebox using PTFE sealing tape. A regular stimulated echo NMR sequence (diffste) was used at room temperature. However, a double stimulated echo NMR sequence (diffDste) was employed at higher temperatures to ensure that thermal convection did not affect the results. An 18 µs 90-degree pulse, a 2 s acquisition time, a 5 s recycling delay, a 100 ms diffusion delay (Δ) and a 1 ms gradient pulse duration (δ) were used. The maximum gradient strength (g) varied in the range of 100–1200 Gauss per cm while keeping k constant; k is defined as| | |
k = (δ·γ·g)2(Δ − δ/3)
| (1) |
where Δ is the diffusion delay time, δ is the gradient pulse duration, γ is the gyromagnetic ratio of the studied nucleus and g is the gradient strength.The gradient strengths were calibrated using reference samples with known self-diffusion coefficients; the 1HDO trace signals in the D2O reference sample, hexafluorobenzene (C6F6) and a standard 1 M LiCl in the H2O sample were recorded for 1H, 19F and 7Li, respectively.26 To determine the intensity attenuation, the average of all 1H NMR signals was considered for the Pyr14 cation. For the Li cation, the signal at −0.4 ppm in the 7Li spectrum was utilized, while the signals at −80 ppm and 50 ppm in the 19F spectrum were used for the TFSI and FSI anions, respectively.
2.3.5 Raman spectroscopy. Raman spectra were recorded using a Bruker MultiRAM Fourier transform Raman spectrometer equipped with a Nd:YAG laser (1064 nm) and a liquid nitrogen-cooled Ge diode detector. The nominal laser power was set to 720 mW, with a spectral resolution of 2 cm−1 (full width at half-maximum). A Blackman–Harris three-term window function was applied for apodization, and the spectra were acquired as the average of 1000 scans. In order to estimate the integrated areas under selected peaks, a multi-peak fitting procedure based on a linear background and Voigt profiles was applied using the Origin lab software. The spectral region 680–780 cm−1 was deconvoluted into six contributions, corresponding to spectroscopically free FSI (I, separated from cations), weakly coordinated FSI (II, contact ion pairs), strongly coordinated FSI–Li+ (III, ion aggregates), the two conformers (cisoid (IV) and transoid (V)) of spectroscopically free TFSI, and coordinated TFSI–Li+ (VI). In the fitting procedure, peak positions, full widths at half maximum (FWHM), and Voigt profiles were fixed, while heights were left as a free fit parameter. The population of the “free” and “coordinated” anions was estimated from the relative intensities of the six components, as expressed below:| |
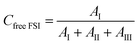 | (2) |
| |
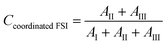 | (3) |
| |
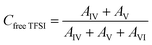 | (4) |
| |
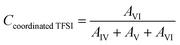 | (5) |
where AI, AII, AIII, AIV, AV, and AVI are the integrated areas of contributions I, II, III, IV, V and VI, respectively. These calculations are valid assuming that the Raman activities of the different TFSI and FSI conformations are equivalent,27,28 so the integrated areas directly reflect their relative populations. Then, the average number of FSI and TFSI anions coordinating with Li+ (Nanion–Li+) was determined using the following equation:| |
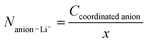 | (6) |
where x is the molar fraction of Li+ ions.
3 Results and discussion
For a comprehensive understanding of the transport properties of the electrolytes at focus in this work, their phase behavior, viscosity, density, ionic conductivity and self-diffusion, as well as intermolecular interactions, with a focus on coordination, were investigated.
3.1 Phase behavior
The phase behavior was investigated using DSC, and the DSC thermograms are shown in Fig. 2. Both neat ionic liquids, Pyr14FSI and Pyr14TFSI, show melting peaks and glass transition from which the glass transition temperature (Tg) was determined. The Tg of Pyr14FSI is found around 165 K, while that of Pyr14TFSI is slightly higher, observed at about 184 K.29 In addition, the DSC curves of the neat ionic liquids reveal cold crystallization and solid–solid transitions. However, the phase behavior is significantly altered with the addition of Li salts. Either salt in the Pyr14FSI-based systems inhibits recrystallization and stabilizes the liquid phase down to the Tg, which is found at 175 K. For Pyr14TFSI with added LiTFSI, recrystallization and melting are still observed; however, these transitions are suppressed upon mixing anions, i.e., with the addition of LiFSI. Interestingly, the glass transition temperature increases by 11 K when LiTFSI is added to Pyr14TFSI, whereas a Tg similar to that of the neat IL is observed for the mixed-anion system (LiFSI added to Pyr14TFSI), see also the values in Table 1. Thus, the incorporation of LiFSI into a TFSI-based ionic liquid has a more pronounced effect compared to the reversed scenario. Interestingly, an opposite trend was observed for imidazolium-based electrolytes.30
 |
| | Fig. 2 DSC thermograms of Pyr14FSI (a) and Pyr14TFSI (b) based samples. | |
Table 1 Glass transition temperatures (Tg) of all ionic liquid electrolytes, extracted from the second heating scan of the DSC curve
| Sample |
Tg (K) |
| Pyr14FSI |
165 |
| (LiFSI)0.2(Pyr14FSI)0.8 |
175 |
| (LiTFSI)0.2(Pyr14FSI)0.8 |
175 |
| Pyr14TFSI |
184 |
| (LiTFSI)0.2(Pyr14TFSI)0.8 |
195 |
| (LiFSI)0.2(Pyr14TFSI)0.8 |
184 |
3.2 Viscosity and density
Viscosity and density values, measured as a function of temperature, are shown in Fig. 3a and b. Corresponding values are summarized in Table S1. In Pyr14FSI-based electrolytes, the addition of either lithium salt leads to an increase in both density and viscosity. For the electrolytes based on Pyr14TFSI, the addition of LiTFSI salt leads to an increase in density and viscosity compared to the neat ionic liquid, whereas, upon the addition of LiFSI, these increases are less pronounced, resulting in lower density and viscosity compared to those of the LiTFSI-containing electrolytes. Overall, Pyr14FSI-based electrolytes exhibit lower viscosity and density compared to the Pyr14TFSI-based electrolytes, as previously reported in the literature.31 These results suggest that anion mixing in Pyr14TFSI-based electrolytes significantly modifies the intermolecular interactions, with possible effects on the Li+–anion coordination.
 |
| | Fig. 3 Viscosity (a), density (b) and ionic conductivity (c) values measured as a function of temperature. The dashed lines are guides to the eyes. The error bars are smaller than the plotted symbols and are therefore not visible. | |
3.3 Transport properties
The ionic conductivity was measured in the temperature range 193–373 K, using a broadband dielectric spectrometer (BDS), and the measured values are plotted in Fig. 3c. The ionic conductivity values are summarized in Table S2. For all electrolytes, the ionic conductivity increases with increasing temperature. The neat Pyr14FSI exhibits higher ionic conductivity than Pyr14TFSI, in agreement with the viscosity trend above and with the literature.31 In general, the addition of a Li salt to the neat ionic liquids leads to a decrease in ionic conductivity, which can be attributed to the corresponding increase in viscosity discussed above.
The measured density, viscosity and conductivity data were used to create a Walden plot with the aim of highlighting differences in ionic association and related transport mechanisms. The Walden plot is generally based on the Walden equation described by Angell et al.:32
where
Λ is the molar conductivity,
η is the viscosity,
α is an adjustable parameter and
C is a temperature dependent constant.
The Walden plots of all electrolytes are shown in Fig. 4. In this plot, the diagonal line represents the behavior expected from an aqueous solution of 0.01 M KCl, in which all ions are assumed to be dissociated and to diffuse independent of each other. A negative deviation from the reference line, also known as the ideal line, can be seen as a measure of ion association within the ionic liquid system, a rationale for the conductivity being lower than expected from the mobility of free ions.32 The data of all investigated ionic liquids fall below the ideal line, with deviations ranging from −0.140 to −0.343, increasing from the neat ionic liquids to the electrolytes (Table S3). This implies, according to Angell et al., the presence of ionic interactions that prevent full ion dissociation.32 Additionally, upon the addition of a salt, the Walden plots of the Pyr14TFSI-based ionic liquids show a slightly greater deviation from the ideal line than those of Pyr14FSI-based ionic liquids. The slopes of all lines, reflecting the temperature dependence of ion association, are approximately 0.90, consistent with values typically reported for ionic liquids.32–35
 |
| | Fig. 4 Walden plot for Pyr14FSI and Pyr14TFSI based samples. | |
The ionic conductivity values of the mixed-anion electrolytes were also plotted on a Tg-scaled Arrhenius plot (an Angell's plot), normalizing the temperature scale to each material's glass transition temperature, Fig. 5. The Tg values used in this approach are those obtained from the DSC data discussed above, also reported in Fig. 1. This representation allows extending the discussion of ionic conductivity to the concept of fragility; fragility is a fundamental property of glass-forming liquids that describes how rapidly the dynamics change upon heating from the glass transition temperature Tg, or vice versa. In an Angell's plot, fragile liquids display a strong curvature, whereas stronger liquids exhibit a closer to linear behavior. In Fig. 5, the ionic conductivity values for the two samples converge onto a single curve, revealing similar fragility and indicating that their temperature-dependent conductivities evolve in a comparable manner relative to Tg, despite differences in composition and glass transition temperatures. Consequently, their dynamics change at a similar rate near Tg, where rapid changes in dynamics facilitate higher mobilities at elevated temperatures, an advantageous behavior for practical applications.36
 |
| | Fig. 5 Tg-scaled plot (Angell plot) of the ionic conductivity of (LiTFSI)0.2(Pyr14FSI)0.8 and (LiFSI)0.2(Pyr14TFSI)0.8. The dashed lines are guides to the eyes. The error bars are smaller than the plotted symbols and are therefore not visible. | |
To selectively distinguish Li+ ion transport, which cannot be resolved by BDS, the self-diffusion coefficients of all involved ionic species, i.e., Pyr14, Li+, FSI and TFSI, were estimated using NMR diffusometry, Fig. 6a–f. The extracted self-diffusion coefficient values are summarized in Table S4. While for Pyr14FSI, the anion exhibits higher diffusivity than the cation, the opposite trend is observed for Pyr14TFSI. This finding aligns with earlier studies and may be attributed to the size difference between the FSI and TFSI anions.37–40 When salts are added, the self-diffusion coefficients of all ions decrease due to increased viscosity, in agreement with the viscosity and conductivity data discussed above. Interestingly, the addition of LiFSI to Pyr14TFSI leads to a smaller decrease in diffusion coefficient than the addition of LiTFSI (especially at lower temperatures). This difference may be attributed to the smaller size of the FSI anion, favoring fluidity. Importantly, in FSI-based systems, the self-diffusion coefficients of all ions, including the Li+, remain higher than those of TFSI-based systems, consistent with previously reported results.15,39 The relative contribution of Li+ ions to the overall conductivity can be estimated by calculating the apparent transference number (tLi+) from the diffusion data. High Li+ transference numbers are essential for efficient charge–discharge performance in Li-ion batteries. Across all studied electrolytes, the tLi+ remains approximately 0.07 with a negligible temperature dependence (Table S5). These values are consistent with the literature and indicate that the Li+ contribution to the conductivity remains similar across all four electrolytes.
 |
| | Fig. 6 The three upper panels and the three bottom panels show the self-diffusion coefficients estimated for the Pyr14FSI based (a)–(c) and the Pyr14TFSI based (d)–(f) samples, respectively. The first (a) and (d), second (b) and (e) and third (c) and (f) columns show self-diffusion values measured from the 1H, 19F and 7Li nuclei. Dashed lines are guides to the eyes. The error bars are smaller than the plotted symbols and are therefore not visible. | |
To gain deeper insight into the degree of ion dissociation, the ratio of molar conductivity Λ/ΛNE, also known as ionicity, was estimated.41–43 Λ is the molar conductivity obtained from impedance measurements (eqn (8)), while ΛNE is the molar conductivity calculated from the self-diffusion coefficients using the Nernst–Einstein equation (eqn (9)):
| |
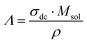 | (8) |
with
σdc being the ionic conductivity measured by BDS,
Msol the molar mass of the solution =
χIL·
MIL +
χsalt·
Msalt and
ρ the density of the solution,
| |
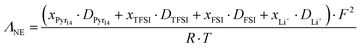 | (9) |
where
F is the Faraday constant,
R is the universal gas constant and
xi is the molar ratio of the
ith component. The molar conductivities and ionicity values are summarized in Table S6.
Fig. 7a and b show that
Λ is smaller than
ΛNE in all systems and at all temperatures. Consequently, the ionicity
Λ/
ΛNE shows a value ≤1 in all studied liquids, as shown in
Fig. 7c, indicating that some ionic species diffuse in pairs or clusters, hence not contributing to the macroscopically measured ionic conductivity, in line with the trend observed in the Walden plot (
Fig. 4). The ionicity decreases in both systems with the addition of Li salt, in particular, when anions are mixed, highlighting overall stronger interactions and aggregate formation in the electrolytes.
 |
| | Fig. 7 Molar conductivities obtained from NMR and impedance measurements for Pyr14FSI (a) and Pyr14TFSI (b) based samples. Estimated ionicity values as a function of temperature (c). | |
3.4 Ion coordination
Further insight into the nature of intermolecular interactions can be obtained by Raman spectroscopy. Room temperature Raman spectra were recorded for both the neat ionic liquids and the electrolytes, Fig. 8. In Pyr14TFSI-based systems, the strongest vibrational mode occurs around 743 cm−1, corresponding to the expansion-contraction mode of the TFSI anion (νs S–N–S and νs CF3).44 The sensitivity of this mode to the coordination of the anion has been thoroughly investigated using both experimental and theoretical methods.27,45 In the neat Pyr14TFSI, TFSI exhibits two coexisting, weakly coordinated conformers (cis and trans). The two conformers give rise to two closely spaced bands in the Raman spectrum between 740 and 744 cm−1. With the addition of LiTFSI, a new Raman band appears at higher frequencies, around 748 cm−1, attributed to TFSI anions coordinating with the Li+ cation.46–48 The average number of TFSI anions around a Li+ anion was calculated to be 2.17 (Table 2).48 Similarly, FSI in Pyr14FSI exhibits two conformations (C1 and C2) that coexist at room temperature, associated with two (theoretically calculated) closely spaced Raman modes at 725 and 732 cm−1. However, a single broader Raman band is typically observed experimentally around 730 cm−1. Upon addition of LiFSI, an additional Raman mode has been reported in the literature at approximately 744 cm−1, corresponding to FSI coordinated with Li+.49,50 The calculated average number of FSI anions surrounding Li+ is 3.33 (Table 2), which is higher than the corresponding number of TFSI in the (LiTFSI)0.2(Pyr14TFSI)0.8 sample (2.17). This suggests that Li+ coordination involves more FSI than TFSI anions, consistent with the results discussed above.
 |
| | Fig. 8 Experimentally recorded Raman spectra (symbols), fitting peaks (colored Voigt profiles) and fit results (black lines) for all Pyr14-based solutions. Peaks are labeled consistently across all six panels as follows: spectroscopically free FSI (I), weakly coordinated FSI (II), strongly coordinated FSI–Li+ (III), cis (IV) and trans (V) conformers of spectroscopically free TFSI, and Li+-coordinated TFSI (VI). | |
Table 2 Average number of Li+-coordinated FSI and TFSI anions (Nanion–Li+) estimated from peak fitting the Raman spectra
| Samples |
NFSI–Li+ |
NTFSI–Li+ |
| (LiTFSI)0.2(Pyr14TFSI)0.8 |
— |
2.17 |
| (LiFSI)0.2(Pyr14FSI)0.8 |
3.33 |
— |
| (LiFSI)0.2(Pyr14TFSI)0.8 |
0.73 |
1.78 |
| (LiTFSI)0.2(Pyr14FSI)0.8 |
2.70 |
0.54 |
However, mixing anions results in a more complex Li+ solvation structure. Six Raman modes were identified and fitted, corresponding to two conformers of weakly coordinated TFSI, one Li+-coordinated TFSI, one free FSI, one weakly coordinated FSI (contact ion pairs) and one strongly FSI coordinated Li+ (ion aggregates). The calculated average numbers of FSI and TFSI anions coordinating with Li+ were 0.73 and 1.78 for the (LiFSI)0.2(Pyr14TFSI)0.8 sample and 2.70 and 0.54 for the (LiTFSI)0.2(Pyr14FSI)0.8 sample. Consequently, the average number of anions around each Li+ in the (LiFSI)0.2(Pyr14TFSI)0.8 sample, with mixed anions, is 2.51 slightly higher than that in the single-anion electrolyte (LiTFSI)0.2(Pyr14TFSI)0.8 (2.17). This can be explained by the higher ability of FSI to coordinate with Li+. In contrast, the average number of anions around Li+ remains nearly unchanged in the Pyr14FSI-based samples.
The total number of anions coordinated with Li ions in the IL system plays a significant role in determining the strength of the Li-ion solvation structure, which, in turn, influences the transport properties of the Li ion. We find that the order of this coordination across the four samples is as follows: (LiTFSI)0.2(Pyr14TFSI)0.8 < (LiFSI)0.2(Pyr14TFSI)0.8 < (LiTFSI)0.2(Pyr14FSI)0.8 ≤ (LiFSI)0.2(Pyr14FSI)0.8.
These results are consistent with the higher Li+ self-diffusion coefficients observed for Pyr14FSI-based systems compared to their counterparts. Fig. 9 illustrates the correlation between the average number of Li-coordinated anions (Nanion–Li+) and Li+ mobility (DLi+), showing that an increase in Nanion–Li+ is associated with an increase in DLi+.
 |
| | Fig. 9 The Li+ self-diffusion coefficient (DLi+) as a function of the average number of Li+-coordinated anions Nanion–Li+). The dashed line is a guide to the eyes. | |
Conclusions
In summary, mixtures of pyrrolidinium-based ionic liquids and 0.2 mole fraction of lithium salts with either identical or mixed anions were investigated with a focus on their physicochemical properties, particularly Li-ion diffusion and coordination. The incorporation of lithium salt generally leads to increased viscosity, higher glass transition temperature (Tg), reduced ionic conductivity and slower ion diffusivity compared to those of neat ionic liquid systems. Notably, mixing anions shows a more pronounced effect in Pyr14TFSI-based systems, where the addition of FSI anions significantly lowers Tg and enhances both conductivity and ion diffusivity. This behavior is likely due to the smaller size and greater Li-coordinating ability of the FSI anion compared to those of TFSI, which alters the intermolecular interactions within the system. Despite the positive impact of the mixed-anion approach, the larger size of the TFSI anion has a dominant impact reducing ion transport and increasing viscosity, relative to the systems rich in FSI. Although some observed effects, such as the increased viscosity and decreased conductivity, may be expected to persist at other Li salt concentrations, as reported in the literature, further studies are necessary to confirm these trends in the herein studied systems.30,35 Among the mixed-anion solutions investigated in this work, (LiTFSI)0.2(Pyr14FSI)0.8 emerges as a promising candidate for lithium-ion battery electrolytes, due to its favorable balance of transport properties and thermal behavior.
Author contributions
Nicole Abdou: data curation, formal analysis, investigation, methodology, visualization, writing – original draft, and writing – review and editing. Quan Wu: data curation, formal analysis, investigation, methodology, and writing – review and editing. Lars Evenäs: investigation, validation, and writing – review and editing. Anna Martinelli: funding acquisition, supervision, validation, and writing – review and editing. Aleksandar Matic: conceptualization, funding acquisition, resources, supervision, validation, and writing – review and editing.
Conflicts of interest
There are no conflicts to declare.
Data availability
The numerical data used in this manuscript, i.e., values for density, viscosity, ionic conductivity, self-diffusion coefficients, apparent Li+ transference numbers, molar conductivities, and ionicity are reported in the supplementary information (SI). See DOI: https://doi.org/10.1039/d6cp00353b.
Acknowledgements
Nicole Abdou and Anna Martinelli kindly thank the Areas of Advance Materials Science and Energy at Chalmers University of Technology for the financial support. Quan Wu and Aleksandar Matic thank the Swedish Electricity Storage and Balancing Centre (SESBC). This project is financed through the Swedish Electricity Storage and Balancing Centre (SESBC). The centre is funded by the Swedish Energy Agency together with five academic and twenty-eight non-academic partners. The authors also thank the Swedish NMR Centre in Gothenburg for the allocated NMR time.
Notes and references
- Z. Lei, B. Chen, Y.-M. Koo and D. R. MacFarlane, Chem. Rev., 2017, 117, 6633–6635 CrossRef PubMed.
- M. Freemantle, Chem. Eng. News Arch., 1998, 76, 32–37 CrossRef.
- M. Vafaeezadeh and H. Alinezhad, J. Mol. Liq., 2016, 218, 95–105 CrossRef CAS.
- Z. Dai, R. D. Noble, D. L. Gin, X. Zhang and L. Deng, J. Membr. Sci., 2016, 497, 1–20 CrossRef CAS.
- L. C. Tomé and I. M. Marrucho, Chem. Soc. Rev., 2016, 45, 2785–2824 RSC.
- I. Harmanli, N. V. Tarakina, M. Antonietti and M. Oschatz, J. Am. Chem. Soc., 2021, 143, 9377–9384 CrossRef CAS PubMed.
- D. R. MacFarlane, N. Tachikawa, M. Forsyth, J. M. Pringle, P. C. Howlett, G. D. Elliott, J. H. Davis, M. Watanabe, P. Simon and C. A. Angell, Energy Environ. Sci., 2014, 7, 232–250 RSC.
- M. Watanabe, Electrochemistry, 2016, 84, 642–653 CrossRef CAS.
- M. Watanabe, M. L. Thomas, S. Zhang, K. Ueno, T. Yasuda and K. Dokko, Chem. Rev., 2017, 117, 7190–7239 CrossRef CAS PubMed.
- M. Kerner and P. Johansson, Batteries, 2018, 4, 10–22 CrossRef.
- L. M. McGrath and J. F. Rohan, Molecules, 2020, 25, 6002–6035 CrossRef CAS PubMed.
- F. Lundin, A. Idström, P. Falus, L. Evenäs, S. Xiong and A. Matic, J. Phys. Chem. C, 2022, 126, 16262–16271 CrossRef CAS.
- P. M. Bayley, A. S. Best, D. R. MacFarlane and M. Forsyth, Phys. Chem. Chem. Phys., 2011, 13, 4632–4640 RSC.
- M. Middendorf and M. Schonhoff, J. Phys. Chem. B, 2024, 128, 2939–2947 CrossRef CAS PubMed.
- F. Castiglione, G. Raos, G. B. Appetecchi, M. Montanino, S. Passerini, M. Moreno, A. Famulari and A. Mele, Phys. Chem. Chem. Phys., 2010, 12, 1784–1792 RSC.
- G. Annat, M. Forsyth and D. R. MacFarlane, J. Phys. Chem. B, 2012, 116, 8251–8258 CrossRef CAS PubMed.
- M. T. Clough, C. R. Crick, J. Grasvik, P. A. Hunt, H. Niedermeyer, T. Welton and O. P. Whitaker, Chem. Sci., 2015, 6, 1101–1114 RSC.
- M. Kunze, S. Jeong, G. B. Appetecchi, M. Schönhoff, M. Winter and S. Passerini, Electrochim. Acta, 2012, 82, 69–74 CrossRef CAS.
- N. Abdou, A. Pipertzis, J. Swenson and A. Martinelli, J. Mol. Liq., 2025, 437, 128418–128427 CrossRef CAS.
- G. B. Appetecchi, M. Montanino, A. Balducci, S. F. Lux, M. Winterb and S. Passerini, J. Power Sources, 2009, 192, 599–605 CrossRef CAS.
- Q. Zhou, W. A. Henderson, G. B. Appetecchi and S. Passerini, J. Phys. Chem. C, 2010, 114, 6201–6204 CrossRef CAS.
- Q. Zhou, P. D. Boyle, L. Malpezzi, A. Mele, J.-H. Shin, S. Passerini and W. A. Henderson, Chem. Mater., 2011, 23, 4331–4337 CrossRef CAS.
- P. M. Bayley, A. S. Best, D. R. MacFarlane and M. Forsyth, ChemPhysChem, 2011, 12, 823–827 CrossRef CAS PubMed.
- M. Nádherná, J. Reiter, J. Moškon and R. Dominko, J. Power Sources, 2011, 196, 7700–7706 CrossRef.
- A. Lahiri, T. J. Schubert, B. Iliev and F. Endres, Phys. Chem. Chem. Phys., 2015, 17, 11161–11164 RSC.
- M. Holz and H. Weingartner, J. Magn. Reson., 1991, 92, 115–125 CAS.
- M. Herstedt, M. Smirnov, P. Johansson, M. Chami, J. Grondin, L. Servant and J. C. Lassègues, J. Raman Spectrosc., 2005, 36, 762–770 CrossRef CAS.
- L. Li, S. Zhou, H. Han, H. Li, J. Nie, M. Armand, Z. Zhou and X. Huang, J. Electrochem. Soc., 2011, 158, A74–A82 CrossRef CAS.
- M. Kunze, S. Jeong, E. Paillard, M. Winter and S. Passerini, J. Phys. Chem. C, 2010, 114, 12364–12369 CrossRef CAS.
- M. Kerner, N. Plylahan, J. Scheers and P. Johansson, Phys. Chem. Chem. Phys., 2015, 17, 19569–19581 RSC.
- P. Gerlach, R. Burges, A. Lex-Balducci, U. S. Schubert and A. Balducci, J. Electrochem. Soc., 2020, 167, 120546 CrossRef CAS.
- C. A. Angell, N. Byrne and J.-P. Belieres, Acc. Chem. Res., 2007, 40, 1228–1236 CrossRef CAS PubMed.
- W. Xu, E. I. Cooper and C. A. Angell, J. Phys. Chem. B, 2003, 107, 6170–6178 CrossRef CAS.
- K. Ueno, H. Tokuda and M. Watanabe, Phys. Chem. Chem. Phys., 2010, 12, 1649–1658 RSC.
- H. Yoon, A. S. Best, M. Forsyth, D. R. MacFarlane and P. C. Howlett, Phys. Chem. Chem. Phys., 2015, 17, 4656–4663 RSC.
- P. Sippel, P. Lunkenheimer, S. Krohns, E. Thoms and A. Loidl, Sci. Rep., 2015, 5, 13922–13929 CrossRef CAS PubMed.
- J. B. Haskins, W. R. Bennett, J. J. Wu, D. M. Hernández, O. Borodin, J. D. Monk, C. W. J. Bauschlicher and J. W. Lawson, J. Phys. Chem. B, 2014, 118, 11295–11309 CrossRef CAS PubMed.
- F. Castiglione, E. Ragg, A. Mele, G. B. Appetecchi, M. Montanino and S. Passerini, J. Phys. Chem. Lett., 2011, 2, 153–157 CrossRef CAS.
- F. Lundin, A. Idström, P. Falus, L. Evenäs, S. Xiong and A. Matic, J. Phys. Chem. C, 2022, 126, 16262–16271 CrossRef CAS.
- F. Castiglione, M. Moreno, G. Raos, A. Famulari, A. Mele, G. B. Appetecchi and S. Passerini, J. Phys. Chem. B, 2009, 113, 10750–10759 CrossRef CAS PubMed.
- M. Gouverneur, J. Kopp, L. van Wullen and M. Schonhoff, Phys. Chem. Chem. Phys., 2015, 17, 30680–30686 RSC.
- O. Holloczki, F. Malberg, T. Welton and B. Kirchner, Phys. Chem. Chem. Phys., 2014, 16, 16880–16890 RSC.
- K. Ueno, H. Tokuda and M. Watanabe, Phys. Chem. Chem. Phys., 2010, 12, 1649–1658 RSC.
- I. Rey, P. Johansson, J. Lindgren, J. C. Lassègues, J. Grondin and L. Servant, J. Phys. Chem. A, 1998, 102, 3249–3258 CrossRef CAS.
- J. C. Lassègues, J. Grondin, R. Holomb and P. Johansson, J. Raman Spectrosc., 2007, 38, 551–558 CrossRef.
- S. Duluard, J. Grondin, J.-L. Bruneel, I. Pianet, A. Grélard, G. Campet, M.-H. Delville and J.-C. Lassègues, J. Raman Spectrosc., 2008, 39, 627–632 CrossRef CAS.
- J.-C. Lassègues, J. Grondin, C. Aupetit and P. Johansson, J. Phys. Chem. A, 2009, 113, 305–314 CrossRef PubMed.
- J. Pitawala, A. Martinelli, P. Johansson, P. Jacobsson and A. Matic, J. Non-Cryst. Solids, 2015, 407, 318–323 CrossRef CAS.
- K. Fujii, S. Seki, S. Fukuda, R. Kanzaki, T. Takamuku, Y. Umebayashi and S.-I. Ishiguro, J. Phys. Chem. B, 2007, 111, 12829–12833 CrossRef CAS PubMed.
- K. Fujii, H. Hamano, H. Doi, X. Song, S. Tsuzuki, K. Hayamizu, S. Seki, Y. Kameda, K. Dokko, M. Watanabe and Y. Umebayashi, J. Phys. Chem. C, 2013, 117, 19314–19324 CrossRef CAS.
Footnote |
| † These authors contributed equally to this work. |
|
| This journal is © the Owner Societies 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  Open Access Article
Open Access Article *a,
Quan Wu†b,
Lars Evenäs
*a,
Quan Wu†b,
Lars Evenäs a,
Anna Martinelli
a,
Anna Martinelli a and
Aleksandar Matic
a and
Aleksandar Matic *b
*b

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.8Pyr14A2 molar ratio, where A1 and A2 represent the FSI or the TFSI anion. All samples were prepared and stored in an argon-filled glovebox with O2 and H2O levels below 1 ppm. Throughout the following text, the samples are named (LiA1)0.2(Pyr14A2)0.8.
0.8Pyr14A2 molar ratio, where A1 and A2 represent the FSI or the TFSI anion. All samples were prepared and stored in an argon-filled glovebox with O2 and H2O levels below 1 ppm. Throughout the following text, the samples are named (LiA1)0.2(Pyr14A2)0.8.