 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Photothermal reshaping and cavity formation in silica-coated gold nanorods using nanosecond pulsed lasers
Tahani
Albogami
 ab,
Lucien
Roach
ab,
Lucien
Roach
 c,
Sarah
Alshehri
c,
Sarah
Alshehri
 abd,
James
McLaughlan
abd,
James
McLaughlan
 e,
Zabeada
Aslam
e,
Zabeada
Aslam
 bf,
Zhan Yuin
Ong
bf,
Zhan Yuin
Ong
 ab,
Stephen D.
Evans
ab,
Stephen D.
Evans
 ab and
Kevin
Critchley
ab and
Kevin
Critchley
 *ab
*ab
aSchool of Physics & Astronomy, University of Leeds, LS2 9JT, UK. E-mail: k.critchley@leeds.ac.uk
bBragg Centre, University of Leeds, LS2 9JT, UK
cCNRS, ENS de Lyon, Laboratoire de Chimie (LCH, UMR 5182), 69342, Lyon, France
dDepartment of Physics, College of Science, University of Bisha, P.O. Box 551, Bisha 61922, Saudi Arabia
eSchool of Electronic & Electrical Engineering, University of Leeds, LS2 9JT, UK
fSchool of Chemical & Process Engineering, University of Leeds, LS2 9JT, UK
First published on 2nd March 2026
Abstract
The aspect ratio of Au nanorods can be precisely engineered using near-infrared nanosecond-pulsed laser irradiation, which enables ultrafast, confined energy deposition inaccessible under continuous-wave irradiation. In this study, we demonstrate a novel method for reshaping Au nanorods encapsulated within thin (<10 nm) silica shells. By leveraging the silica shell as a rigid nanocrucible, nanosecond laser irradiation induces rapid, end-selective shortening of the Au nanorod core, creating terminal cavities of a controllable size. Transmission electron microscopy confirms that while hexadecyltrimethylammonium bromide-coated Au nanorods convert into spherical and ϕ-shaped nanoparticles, the silica shell constrains the laser induced reshaping process, preserving the rod-like morphology while systematically reducing the aspect ratio. Consequently, the distinct longitudinal and transverse plasmon resonances are retained post-irradiation. The reshaping can be precisely controlled by adjusting laser fluence, resulting in a fine-tuned aspect ratio and a significantly narrowed longitudinal resonance, an outcome typically associated with femtosecond laser systems. 4D Scanning transmission electron microscopy reveals that the Au nanorods transform from single crystal to polycrystalline structures upon irradiation, providing direct evidence that nanosecond pulsed irradiation induces complete Au core melting and rapid recrystallization within the shell – with multiple nucleation sites. Despite the polycrystalline structure the resonance peak was narrower than that of the starting nanorod. This method for fabricating Au nanorods with integrated cavities within the offers significant potential for applications in triggered drug delivery, biosensing, and photoacoustic imaging.
1. Introduction
Au nanorods (AuNRs) are intensively investigated for applications in medical imaging and therapeutic applications, principally because of their strong, tunable, longitudinal surface plasmon resonance (LSPR) in the near-infrared (NIR) range.1–4 Having an LSPR band in the NIR region enables AuNRs to be an effective material for analytical,5–7 photo-functional,8–11 diagnostic,12 and biomedical photoacoustic applications.1,13 Irradiating AuNRs at their LSPR dramatically elevates the electron temperature, which then dissipates via non-radiative pathways—chiefly electron–phonon and phonon–phonon coupling—producing heat and causing a rapid rise in the AuNR internal temperature.12 It has been reported that the light-to-heat energy conversion can be up to 100% under particular conditions, such as when the absorption cross-section dominates over the scattering cross-section (σabs ≫ σscat).14 The scattering cross-section of Au nanoparticles scales with D6, whereas the absorption cross-section scales with D3, where D is the particle diameter. Below D ∼ 50 nm Au nanoparticles behave as pure absorbers, because their scattering cross-section is negligible compared to the absorption, while around D ∼ 100 nm the cross-sections are comparable and above this scattering is the dominant effect.12,14Rapid heating of the AuNRs induced by a pulsed laser can cause AuNRs to undergo reshaping, which leads to a reduction in aspect ratio (AR) and resulting in a blueshift or even loss of the LSPR band.15–17 This reshaping behavior is influenced by several factors such as the starting AR, the surfactant types, coating, the surrounding medium (water or organic), the laser types (femtosecond, nanosecond, or continuous wave), and the laser intensity.9,15,16 When the heating rate is greater than cooling rate of AuNR lattice, there can be sufficient energy for surface atoms to break free and transition to a liquid state or, in more extreme cases, melting of whole AuNR can occur.15,18 While reduction of a material's size is known to reduce the melting point, Tm, this is most pronounced when the nanoparticle approaches a few nanometers, therefore, for the majority of reported AuNRs studied, the melting point of Au is close to the bulk value.18,19 However, reshaping does not require the melting temperature of the AuNR to be reached, it only requires that the surface atoms are sufficiently mobile. This phenomenon has been simulated for silver nanoparticles and AuNRs where there is a critical temperature, TS, above which the 'surface' of a nanoparticle can effectively melt and become liquid-like below the melting point, Tm, of the material.16,20–22 Recent in situ studies have provided significant insight into the thermal reshaping of acicular Au nanoparticles, demonstrating that at moderate temperatures, the process is governed by a slow, two-step surface diffusion of Au atoms from the tips to the body.23 When TS < T < Tm the surface atoms are mobile and reshaping will occur to minimize the Gibbs free energy of the particle. This also implies that higher AR AuNRs will be less thermodynamically stable and may undergo faster reshaping.
The type of excitation source employed (pulse and width) is important for the photothermal reshaping.4,9,15 Link et al. reported that nanosecond-pulsed laser sources the caused AuNR fragmentation at high fluences (>4 J cm−2), while at lower fluences the AuNRs where quickly melted into nanospheres.15 At a lower fluence of 0.64 J cm−2, nanosecond-pulsed laser sources reportedly produced partial melting resulting in AuNRs with bent and twisted shapes. Low fluence nanosecond-pulsed lasers do not seem to cause surface-type melting, while femtosecond-pulsed lasers can lead to shorter AR AuNRs.15 Link et al. reported that the threshold fluence for complete melting is nearly two orders of magnitude lower for femtosecond laser pulses compared with nanosecond laser pulses.15 Laser reshaping blueshifts the AuNR LSPR peak and significantly impacts the efficacy of AuNRs for photothermal conversion (or photoacoustic contrast) as the peak AuNR absorbance rapidly moves away from the incident laser wavelength.7,24 Therefore, to understand and control the degree of laser-induced reshaping is an important step in realizing AuNRs for many laser-based applications.
Experimental studies show that organic coatings, such as CTAB, can have an impact on the reducing or modifying the nature of the reshaping.25 Furthermore, inorganic coatings such as silica are widely used to enhance the photothermal stability of AuNRs by a post-synthesis modification, and this remains an active area of research. For instance, slow thermal annealing has been used to tune the LSPR of both CTAB-stabilized and silica-shelled AuNRs by physically reshaping the core.26 However, this process relies on equilibrium heating, which differs fundamentally from the rapid, non-equilibrium conditions induced by high-fluence laser pulses. In another study, Au nanoworms where synthesized and coated in silica and exposed to a nanosecond pulsed laser.27 This exposure resulted in a blue-shift in the LSPR that was dependent on the fluence. While the blueshift suggests a reduction in aspect ratio (e.g., similar to the AuNRs), the mechanism and morphology of the Au nanoworms was not fully investigated.27 Khanadeev et al. showed increased photostability of Au nanorods that are coated in silica to nanosecond pulsed laser irradiation.28 Shells of increasing thickness (e.g. from 24 to 57 nm) were found to enhance photostability.
The potential to harness nanosecond-pulsed laser-induced morphological changes of the AuNRs within thin protective shells remains largely unexplored. The aim of our work is to address this gap by providing a detailed investigation into how nanosecond-pulsed laser excitation can be used to controllably reshape CTAB-stabilized AuNRs encapsulated in a thin silica shell. By pinpointing the laser conditions that transform the rod-like core into a more spherical shape, we demonstrate a novel, physical method for engineering hybrid nanostructures with well-defined internal cavities. This laser-based approach presents a compelling alternative to established fabrication routes for such 'void-space' structures, which often rely on complex chemical steps like the selective oxidation of sacrificial layers.29–31 The ability to create these voids on-demand could unlock unique applications for these materials as nanoreactors, advanced drug delivery vehicles, or highly sensitive plasmonic sensors.
2. Materials and methods
2.1. Materials
Gold(III) chloride trihydrate (520918), hexadecyltrimethylammonium bromide (CTAB, H6269), sodium oleate (233978), sodium borohydride (452882) and tetraethyl orthosilicate (TEOS, 78-10-4) were purchased from Sigma-Aldrich. Silver nitrate (11414), hydrochloric acid (12.1 M, UN1789) and ethanol (E/0650DF/17) were purchased from Fisher Scientific. Ascorbic acid (A15613) was purchased from Alfa Aesar. Methanol (20847) was purchased from VWR. All chemicals were used as received without any additional purification. Deionized water was supplied using a Milli-Q system (resistivity of 18.2 MΩ cm).2.2. AuNR synthesis
Aqua regia was used to clean glassware. The AuNRs were synthesized using seedless synthesis method that was developed by Roach et al.32,33 to obtain AuNRs with AR of ∼4.0, the following conditions were applied. First, an aqueous binary surfactant solution consisting of 4 mL of CTAB solution (40 mM), 1.25 mL of sodium oleate solution (12.5 mM) and 4.75 mL of water was prepared. This was followed by the addition of 10 mL of gold(III) chloride trihydrate solution (1 mM), 480 µL of silver nitrate solution (4 mM), 100 µL hydrochloric acid (11.8 M), 150 µL ascorbic acid solution (85.8 mM). The orange color disappeared after the addition of the ascorbic acid, resulting from the reduction of Au3+ to Au+. Finally, 15 µL of freshly prepared ice-cold sodium borohydride solution (10 mM) was injected rapidly into the mixture. The reaction was maintained at 30 °C for 4 h to allow the reaction to complete. The color of the solution turned to a reddish-brown color. The resulting AuNR suspension was centrifuged at 17000 × g for 30 min. The precipitate was isolated and then resuspended in 10 mL water and stored at room temperature and in the dark. These samples are referred to as AuNRs@CTAB for short, despite the surface composition being a mixture of CTAB and sodium oleate.2.3. Coating the AuNRs with Silica
A modified version of the method described by Ashrafi et al. was used to encapsulate the AuNRs in silica.34 To remove any aggregated AuNRs, 10 mL of the cleaned AuNR suspension (at 50 µg mL−1) were centrifuged at 500 × g for 3 min and the supernatant is collected. Then 20 µL of 0.1 M NaOH solution was added to AuNR suspension to adjust the pH to 10. The solution was left for 30 min while being gently stirred. A methanol solution of TEOS 20% (v/v) was prepared. Three injections of the TEOS solution, separated by 30 min intervals were added to the AuNR suspension (30 µL TEOS solution per 1 mL of AuNR suspension). The reaction was maintained at a temperature of (27 ± 1) °C, left continuously stirred for 3 days. The silica-coated AuNRs (AuNRs@SiO2) were centrifuged multiple times (8000 × g, 10 min) and resuspended in 10 mL of deionized water and kept in the dark at room temperature. These samples are referred to as AuNRs@SiO2 from hereon. The porosity of the SiO2 shells was estimated spectroscopically (see Section S1 in the SI).352.4. Finite element method (FEM) simulations
The optical responses of the AuNRs were simulated using COMSOL's radio frequency module. Calculations were performed in the frequency domain in the scattered field formulation using the PARDISO direct solver. Refractive index values for Au were taken from Johnson and Christy.36 Particles were simulated embedded in a medium of water (n = 1.33) surrounded by a perfectly matched layer. The AuNRs were modeled as a cylinder capped by ellipsoidal hemispheres of independently tunable geometry. The silica shell (n = 1.45) was modelled as a larger cylinder capped by ellipsoidal hemispheres. The geometry was chosen to create a shell of constant thickness. A cavity was included in the interior of some of the structures. σabs was calculated through a volume integral of the resistive heat losses inside the particle, Qrh. σscat was calculated through a surface integral of the Poynting vector at the particle surface. For 'angular-averaged' spectra and field maps the polarization of the light was oriented at an angle of cos−1(1/√3) ∼ 54.7° from the long axis of the AuNR. This angle corresponds to the well-known 'magic angle', defined by the condition cos2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ = 1/3. At this angle, second-rank tensor (dipolar) interactions are averaged in the same way as for a full orientational average, which ensures that the relative weighting of longitudinal and transverse dipole contributions matches the true angular average. This follows directly from the standard treatment of dipolar anisotropy.37
θ = 1/3. At this angle, second-rank tensor (dipolar) interactions are averaged in the same way as for a full orientational average, which ensures that the relative weighting of longitudinal and transverse dipole contributions matches the true angular average. This follows directly from the standard treatment of dipolar anisotropy.37
For the simulation of heat generation COMSOL's Heat Transfer module in the time domain was used as second-simulation step following the optical simulation. An optical simulation was run at the wavelength and intensity of the laser at peak pulse power and the calculated Qrh for these conditions was used a heat source. Hence, this simulation assumes that the laser has an infinitely narrow spectral bandwidth. This is not a major inaccuracy due to the spectral linewidth of a laser being significantly narrower than the typical AuNR LSPR peak. To give the corresponding time profile for Qrh, it then was multiplied through by a normalized Gaussian pulse of form,
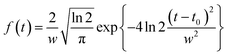 | (1) |
2.5. Characterization
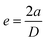 | (2) |
2.6. Nanosecond-pulsed laser irradiation of AuNR samples
The nanosecond-pulsed laser irradiation of AuNRs@CTAB and AuNRs@SiO2 aqueous dispersions, at concentrations of 50 µg mL−1, was investigated using a tunable optical parametric oscillator laser system (Vibrant, OPOTEK, Inc.) at a wavelength of 850 nm (unless stated otherwise). The beam had a pulse width of 7 ns, a repetition frequency of 10 Hz, and a beam diameter of ∼6 mm. A quartz cuvette was filled with the aqueous dispersion of the AuNRs@CTAB and AuNRs@SiO2 particles, which was then continuously stirred with a magnetic bar during pulsed laser irradiation for various exposure times (typically 5 min). The starting temperatures of the aqueous solutions were room temperature (∼21 °C).3. Results
3.1. Characterization of the AuNRs@SiO2 nanoparticles
The optical and morphological properties of the as-synthesized, CTAB-stabilized AuNRs (AuNR@CTAB) were characterized prior to silica coating. UV-vis-NIR extinction spectroscopy revealed two distinct absorption bands (Fig. 1a): a transverse surface plasmon resonance (TSPR) at 511 nm and a LSPR at 830 nm, confirming the anisotropic shape. TEM analysis (Fig. 1b) corroborated this, showing rod-shaped particles with an average length of (47 ± 9) nm, a diameter of (12 ± 2) nm, and a corresponding AR of (3.9 ± 0.8) (N = 100, see Fig. S2 in the SI). Successful encapsulation of these AuNRs within a uniform silica shell was confirmed by TEM (Fig. 1c), which showed a mean shell thickness of (6.8 ± 0.2) nm. The optical signature of this coating process was a predictable redshift of the LSPR peak to (836 ± 1) nm. This shift is the expected consequence of increasing the local refractive index at the Au-dielectric interface upon silica deposition, as reported elsewhere.3 The LSPR peak was also slightly broadened, with the full-width half-maximum (FWHM) increasing from 174 to 204 nm. The coating caused the TSPR peak to be shifted by only 2 nm. These final hybrid particles are hereafter referred to as AuNRs@SiO2. | ||
| Fig. 1 (a) UV-vis-NIR extinction spectra of AuNRs@CTAB (black) and AuNRs@SiO2 (grey). TEM micrographs of (b) AuNRs@CTAB and (c) AuNRs@SiO2 samples. All scale bars are 25 nm. | ||
3.2. Nanosecond-pulsed laser irradiation of AuNRs and AuNRs@SiO2
To investigate the effect of the silica shell on photothermal reshaping, AuNRs@CTAB and AuNRs@SiO2 solutions were irradiated using a nanosecond-pulsed laser (λ = 850 nm) at fluences of 20 and 10 mJ cm−2. The resulting morphological changes were monitored in real-time by observing the evolution of the LSPR and TSPR bands, which are highly sensitive to the AR.At the lower fluence of 10 mJ cm−2 (Fig. 2d), the AuNRs@SiO2 followed a similar reshaping pathway but with less slightly less strength. The new LSPR band appeared at a slightly longer wavelength (665 nm) with its final position was also less blue shifted (621 nm), suggesting that the degree of reshaping is fluence-dependent.
TEM (Fig. 3a–c) was used to examine the form and morphological changes of the AuNRs@CTAB after 0 min, 1 min, and 5 min of nanosecond-pulsed laser exposure (850 nm, 20 mJ cm−2). After 1 min of pulsed laser irradiation, the AuNRs@CTAB noticeably transition from a nanorod morphology to spherical, ellipsoidal, or “ϕ” shapes (Fig. 3b), consistent with previous reports.15 Spherical and ϕ-shaped AuNRs were the most frequently found particles, with yields of 35% and 32%, respectively (N = 100). Approximately, 18% of the AuNRs retained their original shapes after pulsed laser irradiation. The remaining 14% of the particles were lower AR AuNRs. The remaining AuNRs had AR1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) min = 2 ± 1 and e1
min = 2 ± 1 and e1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) min = 1.4 ± 0.4 after 1 min of exposure; and AR5
min = 1.4 ± 0.4 after 1 min of exposure; and AR5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) min = 1.3 ± 0.6 and e5
min = 1.3 ± 0.6 and e5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) min = 1.3 ± 0.4 after 5 min of exposure.
min = 1.3 ± 0.4 after 5 min of exposure.
The morphological evolution of the AuNRs@SiO2 particles under laser irradiation (850 nm, 20 mJ cm−2) was also characterized by TEM (Fig. 3d–f). In contrast to the uncoated particles, the silica shell maintained the Au core with an anisotropic shape. The shell acted as a rigid nanoscale crucible, confining the AuNR reshaping process. A key consequence of this confined reshaping is the creation of a void within the shell. As the AuNR core melted and contracted to a lower AR, it pulled away from the silica wall, resulting in a distinct cavity, typically at one end (∼92%), but occasionally at both ends of the metallic core. After 5 min of irradiation most of the intact core–shell particles exhibited a well-defined single internal cavity. The reshaping process produced a heterogeneous population of Au core morphologies (Fig. 3f), e.g., 57% of the cores retained a rod-like shape (though with a lower AR), 30% adopted a distinct dumbbell-like morphology, and the remaining 13% were irregular. This reshaping was also accompanied by a change in the end-cap geometry, with the tips of the cores transitioning from a nearly hemispherical shape to a more pointed, prolate form (e > 1). A small fraction of particles appeared unaffected by the laser, and in some instances, the silica shell was observed to have fractured, leaving an empty shell.
Statistical analysis of the TEM images provides quantitative insight into the different reshaping mechanisms for the AuNRs@SiO2 and AuNRs@CTAB (Fig. 4). For the AuNRs@CTAB, the size distributions confirm a rapid and complete melting into near-spherical particles. Before irradiation, the length, diameter, and AR distributions were relatively narrow (Fig. 4a–c). After just 1 min of laser exposure, the length distribution shifts dramatically to shorter values while the diameter distribution broadens and shifts to larger values. This collapse results in a population with a mean AR of ∼2. Continued exposure to 5 min appears to reduce the AR further to slightly narrow these final distributions, suggesting the system settles into a more uniform, thermodynamically stable state.
In contrast, the AuNRs@SiO2 samples show a distinctly different reshaping pathway that is physically constrained by the shell. After 1 min of irradiation at 20 mJ cm−2, the AR distribution is clearly bimodal: it contains a population of un-transformed rods retaining their original AR (∼3.9), alongside a new population of reshaped AuNRs with a mean AR of 2.2 ± 0.6. With continued exposure for 5 min, the initial population was fully depleted, resulting in a single, narrower distribution centered at a final mean AR of 2.2 ± 0.3 (Fig. 4f). The end-cap geometry of the rod before irradiation the caps are slightly oblate-hemispherical (e < 1), but after 5 min irradiation it shifts to a slightly prolate hemispherical (e > 1). Crucially, the reduction in AR was found to be fluence-dependent. A separate sample irradiated at a lower fluence of 10 mJ cm−2 followed the same pathway, but the extent of reshaping was less pronounced. After 5 min, the final mean AR was 2.8, significantly higher than the 2.2 achieved at the higher fluence (Fig. S3 in the SI). This demonstrates that within the silica shell, the final morphology of the Au core is not only controlled but is also tunable by adjusting the incident laser power. Interestingly, the analysis also revealed subtle changes in the silica shell's morphology after five min of irradiation at 20 mJ cm−2. The hybrid nanoparticles total mean length and mean width slightly increased from 57 nm and 22 nm to 60 nm and 26 nm, respectively. However, the shell thickness was observed to decrease slightly by 1.2 nm (Table 1), this could be possibly explained by condensation of the silica shell because of the photothermal heat generation, although this requires further characterization to be stated definitively. The main parameters are summarized in Table 1.
| Sample | Exposure time (min) | L (nm) | D (nm) | AR | End-cap factor, e | Silica thickness (nm) | LSPR (nm) | TSPR (nm) |
|---|---|---|---|---|---|---|---|---|
| AuNRs@CTAB | 0 | 47 ± 9 | 12 ± 3 | 3.9 ± 0.6 | 0.8 ± 0.2 | — | 832 ± 2 | 511 |
| AuNRs@CTAB | 1 | 26 ± 12 | 16 ± 6 | 2 ± 1 | 1.1 ± 0.3 | — | — | 522 |
| AuNRs@CTAB | 5 | 22 ± 6 | 18 ± 6 | 1.4 ± 0.7 | 1.1 ± 0.3 | — | — | 522 |
| AuNRs@SiO2 | 0 | 50 ± 11 | 14 ± 3 | 3.9 ± 0.8 | 0.8 ± 0.1 | 6.8 ± 1.4 | 836 ± 2 | 513 |
| AuNRs@SiO2 | 1 | 36 ± 10 | 14 ± 4 | 3 ± 1 | 1.0 ± 0.2 | 5.5 ± 0.9 | 624 ± 2 | 516 |
| AuNRs@SiO2 | 5 | 35 ± 8 | 16 ± 4 | 2.2 ± 0.3 | 1.3 ± 0.2 | 5.6 ± 1.1 | 611 ± 2 | 517 |
Nanosecond-pulsed laser irradiation of AuNRs@SiO2 nanoparticles induces a controlled reshaping of the Au core within the silica shell, rather than fragmentation or disintegration. This conclusion is supported by basic volumetric calculations, detailed elemental mapping and scanning transmission electron microscopy (STEM) image analysis (see Sections S4 & S5 in the SI, respectively). Based on the analyses provided, laser irradiation of AuNRs@SiO2 nanoparticles induces a controlled reshaping of the Au core within the silica shell, rather than fragmentation or disintegration. This conclusion is supported by detailed elemental mapping, TEM image analysis, and volumetric calculations. Evidence against fragmentation comes from a volumetric analysis of the Au cores before and after laser exposure. By applying thresholding techniques and contour fitting algorithms to TEM images, the 2D shapes of hundreds of AuNR cores were converted into binary images and extrapolated into 3D volumes. The mean volume of the Au cores was found to be approximately the same before and after irradiation at 20 mJ cm−2, a result consistent with a process of melting and recrystallization contained within the silica shell (Fig. S4 in the SI).
This finding was further corroborated by elemental mapping. Energy-dispersive X-ray (EDX) spectroscopic maps of the AuNRs@SiO2 structures show no evidence of Au dispersion or fragmentation outside of the Au core region after 5 min of laser irradiation (Fig. S5 in the SI). The EDX maps provide insight into the behavior of the CTAB surfactant layer. Before irradiation (Fig. S6 in the SI), signals for N and Br overlap with the Au, consistent with the presence of CTAB. Following irradiation, the Br signal becomes weaker and is only present in the region of reshaped Au core, suggesting a possible partial removal or degradation of the CTAB, though this cannot be definitively confirmed from the EDX data alone.
While the Au cores remain intact, they undergo significant morphological changes. To systematically categorize the diverse shapes produced by the reshaping process, the binary images derived from TEM analysis were processed using an unsupervised 16-layer hierarchical clustering model. This computational approach successfully classified the post-irradiation morphologies into four primary types (Fig. S7 in the SI): (a) nanorods: particles that retained their general rod shape, albeit often with a lower AR; (b) irregular rods: elongated particles that lost their uniform cylindrical shape; (c) ovoids: particles that collapsed into more spherical or egg-like shapes; and (d) dumbbell-like: particles exhibiting a distinct, pinched-in center.
3.2. Simulation of AuNRs@SiO2 extinction spectra
In Fig. 5a, FEM simulated spectra are shown for the AuNRs in Fig. 1 (also Fig. S8 in the SI), before and after photothermal reshaping by a 5 mJ cm−2 laser pulse. The respective AuNR@SiO2 dimensions used were (L = 55 nm, D = 14.5 nm, e1 = e2 = 0.6) before and (L = 48 nm, D = 17 nm, e1 = 0.6, e2 = 0.92) after reshaping, with a silica thickness of t = 6 nm. After reshaping, the geometry of the core AuNR was assumed to asymmetric with different end cap geometries (e1 = 0.79 in contact with the SiO2 and e2 = 1.14 inside the cavity, e.g., more prolate). The values of σabs are the angular-averaged values. For the untreated sample, the predicted LSPR peak was at 836 nm (σabs = 4505 nm2), which closely matches the observed experimental LSPR band peak wavelength in Fig. 1a. By comparison, the reshaped geometry with an internal cavity (7 nm in length) had a predicted LSPR peak at 708 nm (σabs = 3848.7 nm2) this compares less favorably with the observed experimental LSPR peak wavelength of 610 nm. The electric field enhancement at the LSPR peak wavelength for each geometry is shown in Fig. 5b and c. The field enhancement of the original AuNR@SiO2 is almost entirely restricted to within the silica shell coating the AuNRs, whereas in the reshaped case there is high field enhancement close to the AuNR tip inside the formed cavity. The calculations here predict a strong local field enhancement, |Ē/Ē0| ∼ 55, within the cavity of the AuNR@SiO2 particle, enabling plasmonic enhancement of Raman scattering. Hence, molecules located within the cavity could be directly measured through Raman spectroscopy.In addition to the optical simulations, computational modelling of the heat generated by a single laser pulse was also performed (Fig. 5d–f and Fig. S9–S17 in the SI). In these models, the laser polarization was aligned perfectly to the longitudinal axis of the AuNRs (i.e. the most efficient illumination configuration). This was assumed to be the most relevant scenario because, for all practical purposes, all AuNRs will experience pulses with close alignment to the light polarization multiple times. The short tumbling time of the particles (τr ∼ 1 µs), compared to the period of the laser pulse (= 100 ms), and the short width of the pulses (τp = 7 ns) particles will essentially be illuminated at a random angle during each pulse. Over 3000 pulses, there is a near certainty that the AuNR will be aligned to the polarization of the light source multiple times.
It can be seen in the electromagnetic simulations that there is precipitous drop in σabs at the laser wavelength (850 nm) after reshaping from 2929 nm2 to 58 nm2, a 98% reduction, which reduces photothermal heat generation to comparatively very low levels. This can be seen in the simulations of the resulting heat generation. The original AuNR@SiO2 particle upon illumination by a pulse with characteristics: τp = 7 ns, λ = 850 nm, and fluence = 5 mJ cm−2 generates a peak temperature increase of ΔT = 352 °C, 1 ns after the peak of the laser pulse. Strong temperature gradients are generated for around 11 ns (the pulse is centered on 10 ns) before rapidly dissipating over the following 40 ns of the simulation (Fig. S10 and S17 in the SI). By comparison, the reshaped AuNR@SiO2 particle only generates a peak temperature increases of ΔT = 7 °C (i.e. ∼98% less, consistent with the drop in σabs). The temperatures achieved by the reshaped AuNR@SiO2 particle are unlikely to result in any further morphological changes. By comparison, the temperatures achieved by the original AuNR@SiO2 are hot enough to decompose organics. The highest predicted temperature increase at the silica-water interface is ΔT = 286 °C, which is close to the known threshold for bubble cavitation (0.8Tc,H2O ∼ 300 °C),40 given the ambient temperature of 20 °C. Hence, for higher pulse fluences we might expect consistent generation of cavitation events.
In addition, simulations were performed of the geometries seen in Fig. 3 which were exposed to a high pulse fluence of 20 mJ cm−2 (Fig. S13–S17 in the SI). Similar trends can be seen with a ∼99% drop in σabs at 850 nm after reshaping. The LSPR of the original AuNR@SiO2 (810 nm) is worse matched to the laser wavelength that in the previous case σabs (850 nm) = 747 nm2 (vs. 2929 nm2 previously), hence despite a fourfold increase in laser pulse fluence, the peak temperature is roughly the same. After reshaping the heat generation at 850 nm would not be expected to induce any further changes in morphology.
3.4. Controlled reshaping via off-resonance excitation
To achieve more precise control over the reshaping process, the effects of off-resonance laser excitation was investigated. For this experiment, the AuNRs@SiO2 were synthesized with a slightly lower AR to give a LSPR of ∼800 nm. These particles were then irradiated with a nanosecond-pulsed laser at a fixed wavelength of 850 nm—intentionally above the LSPR peak—for a duration of 5 min (i.e., 3000 pulses), at different fluences. Fig. 6a shows the UV-vis-NIR extinction spectrum, demonstrating a highly controllable, fluence-dependent reshaping of the Au cores. Before irradiation the LSPR maxima was at 800 nm and had a FWHM of 164 nm. Pulsed laser irradiation for 5 min resulted in a blueshift of the maxima which increased with increasing laser fluence. The experiment was repeated with a fresh AuNRs@SiO2 sample, and the combined results are summarized in Fig. 6b. The LSPR band blueshifts and the FWHM values were found to decreased as a function of fluence (Fig. 6b). This confirms that by operating in an off-resonance regime, the final AR of the encapsulated NRs can be precisely tuned in a “dose-dependent” manner. The LSPR band becomes slightly spectrally narrower after irradiation which is caused by spectral hole burning. However, the TSPR peak is weakened, which indicates that the AuNRs are not converted into spheres and that some damping of the TSPR band occurs due to diameter heterogeneity, and enhanced surface scattering from the roughness.STEM images of the AuNRs exposed to 5 mJ cm−2 (λ = 850 nm) results in similar reshaping as the irradiation closer to the plasmon maxima, but with a smaller cavity (Fig. 7). The AuNRs are reshaped and many of the cores can be characterized as dumbbell shaped within the silica shell. There are some of the Au core nanorods that no longer have shells.
 | ||
| Fig. 7 High-resolution (a) and low-resolution (b) brightfield STEM images of the AuNRs@SiO2 particles after 5 min of 5 mJ cm−2 nanosecond-pulsed laser irradiation using a wavelength of 850 nm. | ||
Quantitative analysis of the post-irradiation size distributions provides further insight into the off-resonance reshaping mechanism (Fig. 8). As expected, the TEM data show a clear decrease in the mean length (Fig. 8a) and a corresponding increase in the mean diameter (Fig. 8b) of the Au cores. This geometric shift resulted in a population with a lower average AR. The TEM size analysis does not show evidence of AR distribution narrowing. The end cap geometry does shifts from an oblate hemispherical cap to almost spherical (e = 0.7 to e = 0.9), with a narrower distribution (Fig. S18 in the SI). To directly probe the crystallographic changes induced by pulsed laser irradiation, a STEM capable of spatially mapping electron diffraction patterns was used to characterize AuNR samples (4D-STEM). The resulting diffraction patterns were indexed to the fcc crystal structure of Au and visualized using inverse pole figure (IPF) maps to reveal the orientations of the crystal individual AuNRs (Fig. 9). Before irradiation, the AuNRs@CTAB were confirmed to be single crystals (Fig. 9a–d). The IPF maps show a uniform color across each individual AuNR, indicating that the entire particle is oriented along a single crystallographic zone axis, consistent with a defect-free, monocrystalline structure.
In contrast, after 5 min of irradiation at 5 mJ cm−2, both AuNRs@CTAB and AuNRs@SiO2 samples became polycrystalline. The IPF maps for these samples reveal multiple crystal orientations within a single particle, visualized by multiple color domains (Fig. 9g–m). This confirms that laser-induced heating causes complete melting and recrystallization of the Au core. The AuNRs@CTAB particles reshaped into more spherical forms (consistent with Fig. 3c) and exhibited large grain boundaries characteristic of a polycrystalline nanomaterial (Fig. 9f and h). The AuNRs@SiO2 samples preserved their rod-like morphology, though the cores developed significant internal defects, including twin boundaries (Fig. 9j–l). This confirms that the Au cores of the AuNRs@SiO2 particles have melted and recrystallized with multiple nucleating sites, which occur in different locations of each core.
4. Discussion
The binary surfactant method successfully produced AuNRs with a LSPR peak in the near-infrared and a TSPR peak at 511 nm, consistent with previous work.32,33 The AuNRs exhibited approximately hemispherical end-caps. This cap geometry is known to influence the LSPR peak position but has a negligible effect on its FWHM.38,41,42 Coating the AuNRs with a silica shell induced a slight redshift in both the LSPR and TSPR peaks, which is attributed to the increased refractive index of the medium immediately surrounding the Au core. The silica shells were uniform, following the rod-like shape of the core with a mean thickness of 6.8 nm and an average porosity of ∼43%. The presence of a CTAB layer on the Au surface, confirmed by EDX, was found to be essential for maintaining colloidal stability during the silica growth process.Upon irradiation with a nanosecond-pulsed laser tuned to the LSPR, the shelled (AuNRs@SiO2) and unshelled (AuNRs@CTAB) particles exhibited distinctly different reshaping behaviors. During laser exposure, the LSPR of the unshelled AuNRs@CTAB was rapidly extinguished, accompanied by a corresponding increase in the TSPR band. This spectral change is consistent with the observed morphological transformation into spherical and ϕ-shaped nanoparticles.15 In contrast, the AuNRs@SiO2 samples underwent a more controlled transformation. The LSPR peak blue-shifted from ∼830 nm to ∼620 nm, indicating a decrease in the AR of the AuNR core. Crucially, the TSPR band did not increase in magnitude, confirming that the formation of spheres was suppressed by the shell. The incomplete disappearance of the original 830 nm peak suggests that the silica shell can, in some cases, effectively protect the AuNRs from major reshaping.
The observed reshaping is best explained by the complete melting of the Au core, followed by recrystallization. While simulations on smaller particles show a size-dependent shape transition temperature (Ts) before reaching complete melting, extrapolating these findings suggests a Ts around 1000 K for the large particles used here, i.e. ∼300 K below the bulk melting temperature.21 Under the high-energy conditions of nanosecond laser pulses, complete melting within the shell is the dominant mechanism. When there is no shell present, the AuNRs become spherical, ellipsoidal, or ϕ-shaped. However, the presence of a shell confines the liquid Au and preserves a nanorod shape, albeit a reduced AR with irregular polycrystalline morphology. Importantly, once the LSPR of the core blueshifts past the laser wavelength, absorption ceases, and the reshaping is arrested. This process significantly narrows the FWHM of the LSPR band by ∼60%. This optical finding aligns with reports of laser-induced monodispersity in unshelled AuNRs.16
The silica shell acts as a “nanocrucible” confining the molten Au and preventing it from collapsing into a sphere. This confinement often leads to the formation of a distinct dumbbell-like morphology, as the molten Au minimizes its Gibbs free energy by reducing its contact with the non-wetting silica surface (contact angle, θc > 130°) and beading up at the ends.43 This dumbbell shape is the inverse of the ϕ-shape formed when unshelled AuNRs are irradiated.
A critical question is whether the shell provides sufficient volume for the core to decrease its AR and increase its width. TEM analysis confirms that the Au volume is conserved with no evidence of fragmentation. The necessary extra space is likely created by three possible processes: (1) localized etching of the porous silica shell (possible by superheated water44) (2), thermal decomposition of the ∼3.2 nm thick organic CTAB-oleate layer,45 or (3) the silica shell becomes denser during the heating.
The 4D-STEM analysis provides definitive evidence for the proposed mechanism. The AuNR cores transition from a monocrystalline state before irradiation to a highly polycrystalline state afterward, exhibiting numerous grain boundaries and twin defects. This confirms that the reshaping process involves a full melt-solidification cycle, where rapid heat dissipation during cooling prevents the formation of a perfect single crystal.
Finally, the reduction in the core AR reliably creates a nanocavity between the Au core and the silica shell, typically at one end of the rod. The size of this cavity can be controlled by adjusting laser fluence or irradiating at a wavelength above the LSPR. Compared to chemical etching methods, this laser-driven approach is a simple, rapid, and reproducible technique for fabricating nanocontainers.30,31
5. Conclusion
In conclusion, this study demonstrates a novel and straightforward method to controllably reshape AuNRs within a silica shell using nanosecond pulsed laser irradiation. This process reduces the core AR while preserving the overall rod-like morphology, creating a predictable nanocavity at one end. Unlike unshelled AuNRs which collapse into spheres, the confined cores retain their two characteristic plasmon bands, albeit with a significant blueshift LSPR. The 4D-STEM analysis provided definitive evidence for the underlying mechanism: the AuNRs undergo a monocrystalline-to-polycrystalline transformation, confirming that the core melts and rapidly recrystallizes within its shell. The melting and recrystallisation causes increased roughness and shape heterogeneity. This laser-driven technique offers a rapid route to fabricating AuNRs@SiO2 nanocontainers. Future work should focus on scaling this method and exploring its potential in applications such as targeted drug delivery and nanoreactors.Author contributions
Conceptualization – KC; data curation - KC; formal analysis – TA, SA, LR; funding acquisition – KC; investigation – TA, SA, LR; methodology – JM, ZA; project administration – KC, SDE; supervision – KC, SDE, ZO; validation – TA, SA, LR, ZA; visualization – TA, SA, LR, KC; writing – original draft – TA, SA, LR, KC; writing – review & editing – all authors.Conflicts of interest
There are no conflicts to declare.Data availability
The data associated with this paper is available from University of Leeds at https://doi.org/10.5518/1787.46Supplementary information (SI): additional experimental details and figures. See DOI: https://doi.org/10.1039/d6cp00240d.
Acknowledgements
K. C. thanks the EPSRC (EP/T013753/1) for financial support. We thank BRG Johnson for technical support. L.R. acknowledges financial support from the Région Auvergne-Rhône–Alpes through the “Amorçage Europe” programme (No. 25 003102 01–15704). SA are thankful to the Deanship of Graduate Studies and Scientific Research at University of Bisha for the financial support through the Scholars Research Support Program.References
- X. Huang, S. Neretina and M. A. El-Sayed, Adv. Mater., 2009, 21, 4880–4910 CrossRef CAS PubMed.
- J. Ma, Y. Li, Y. Ying, B. Wu, Y. Liu, J. Zhou and L. Hu, Chem. Med. Chem., 2024, 19, e202300374 CrossRef CAS.
- S. Si, A. G. Majumdar and P. S. Mohanty, BioNanoSci., 2025, 15, 185 CrossRef.
- S. Link and M. A. El-Sayed, Int. Rev. Phys. Chem., 2000, 19, 409–453 Search PubMed.
- B. Nikoobakht, J. Wang and M. A. El-Sayed, Chem. Phys. Lett., 2002, 366, 17–23 CrossRef CAS.
- K. Ueno, S. Juodkazis, M. Mino, V. Mizeikis and H. Misawa, J. Phys. Chem. C, 2007, 111, 4180–4184 CrossRef CAS.
- K. A. Willets and R. P. Van Duyne, Annu. Rev. Phys. Chem., 2007, 58, 267–297 CrossRef CAS PubMed.
- J. W. Cottom, P. Abellan, Q. M. Ramasse, K. Critchley and R. Brydson, J. Phys. Chem. C, 2018, 122, 22041–22053 CrossRef CAS.
- J. Pérez-Juste, B. Rodríguez-González, P. Mulvaney and L. M. Liz-Marzán, Adv. Funct. Mater., 2005, 15, 1065–1071 CrossRef.
- M. Das, N. Sanson, D. Fava and E. Kumacheva, Langmuir, 2007, 23, 196–201 CrossRef CAS PubMed.
- Z. Zhang, Y. Wu, X. Cao, J. Gao, S. Yan, S. Su, Y. Wu, N. Zhou, X. Wang and L. Chen, Nano Lett., 2024, 24, 15127–15135 CrossRef CAS PubMed.
- X. Huang and M. A. El-Sayed, J. Adv. Res., 2010, 1, 13–28 CrossRef.
- H. Takahashi, T. Niidome, A. Nariai, Y. Niidome and S. Yamada, Chem. Lett., 2006, 35, 500–501 CrossRef CAS.
- V. Amendola, R. Pilot, M. Frasconi, O. M. Maragò and M. A. Iatì, J. Phys.: Cond. Matter, 2017, 29, 203002 CrossRef PubMed.
- S. Link, C. Burda, B. Nikoobakht and M. A. El-Sayed, J. Phys. Chem. B, 2000, 104, 6152–6163 CrossRef CAS.
- G. González-Rubio, P. Díaz-Núñez, A. Rivera, A. Prada, G. Tardajos, J. González-Izquierdo, L. Bañares, P. Llombart, L. G. Macdowell, M. Alcolea Palafox, L. M. Liz-Marzán, O. Peña-Rodríguez and A. Guerrero-Martínez, Science, 2017, 358, 640–644 CrossRef.
- S. M. Meyer, J. Pettine, D. J. Nesbitt and C. J. Murphy, J. Phys. Chem. C, 2021, 125, 16268–16278 CrossRef CAS.
- C. Louis and O. Pluchery, Gold Nanoparticles for Physics, Chemistry and Biology, Imperial College Press, London, 2nd edn, 2011, ch. 4 DOI:10.1142/p815.
- K. Dick, T. Dhanasekaran, Z. Zhang and D. Meisel, J. Am. Chem. Soc., 2002, 124, 2312–2317 CrossRef CAS PubMed.
- S. J. Zhao, S. Q. Wang, D. Y. Cheng and H. Q. Ye, J. Phys. Chem. B, 2001, 105, 12857–12860 CrossRef CAS.
- Y. Wang and C. Dellago, J. Phys. Chem. B, 2003, 107, 9214–9219 CrossRef CAS.
- H. B. Liu, J. A. Ascencio, M. Perez-Alvarez and M. J. Yacaman, Surf. Sci., 2001, 491, 88–98 CrossRef CAS.
- J. Lyu, L. Matthews, T. Zinn, D. Alloyeau, C. Hamon and D. Constantin, Nano Lett., 2025, 25, 1544–1549 CrossRef CAS PubMed.
- M. Grzelczak and L. M. Liz-Marzán, Chem. Soc. Rev., 2014, 43, 2089–2097 RSC.
- W. J. Kennedy, S. Izor, B. D. Anderson, G. Frank, V. Varshney and G. J. Ehlert, ACS Appl. Mater. Interfaces, 2018, 10, 43865–43873 CrossRef CAS.
- A. Candreva, F. Parisi, G. Di Maio, F. Scarpelli, I. Aiello, N. Godbert and M. La Deda, Gold Bull., 2022, 55, 195–205 CrossRef CAS.
- M. Yaqub, G. A. Khan, H. Qayyum, M. A. Ghanem, A. Nadeem, Q. Abbas and W. Ahmed, ChemistrySelect, 2025, 10, e00580 CrossRef CAS.
- V. A. Khanadeev, A. V. Simonenko, O. V. Grishin and N. G. Khlebtsov, Nanomaterials, 2023, 13, 1312 CrossRef CAS PubMed.
- E. Choi, M. Kwak, B. Jang and Y. Piao, Nanoscale, 2013, 5, 151–154 RSC.
- Z. Li, J. Zhang, J. Jin, F. Yang, R. Aleisa and Y. Yin, J. Am. Chem. Soc., 2021, 143, 15791–15799 CrossRef CAS PubMed.
- Z. Wu, Y. Liang, Q. Guo, K. Zhang, S. Liang, L. Yang, Q. Xiao and D. Wang, J. Mater. Sci., 2019, 54, 8133–8147 CrossRef CAS.
- L. Roach, P. L. Coletta, K. Critchley and S. D. Evans, J. Phys. Chem. C, 2022, 126, 3235–3243 CrossRef CAS.
- L. Roach, S. Ye, S. C. Moorcroft, K. Critchley, P. L. Coletta and S. D. Evans, Nanotechnology, 2018, 29, 135601 CrossRef PubMed.
- S. J. Ashrafi, F. Yazdian, A. S. H. Zaremi, J. Mohhamadnejad and R. Dinarvand, Biomed. Pharma. J., 2016, 9, 1189–1201 CrossRef.
- P. A. Mercadal, L. A. Perez and E. A. Coronado, J. Phys. Chem. C, 2021, 125, 15516–15526 CrossRef CAS.
- P. B. Johnson and R. W. Christy, Phys. Rev. B, 1972, 6, 4370–4379 CrossRef CAS.
- L. Roach, Synthesis, Functionalisation, and Biomedical Application of Phospholipid-Functionalised Gold Nanorods for Cancer Therapy, PhD thesis, University of Leeds, 2019.
- N. Xu, B. Bai, Q. Tan and G. Jin, Opt. Express, 2013, 21, 21639–21650 CrossRef PubMed.
- B. R. Jany, N. Gauquelin, T. Willhammar, M. Nikiel, K. H. W. van den Bos, A. Janas, K. Szajna, J. Verbeeck, S. Van Aert, G. Van Tendeloo and F. Krok, Sci. Rep., 2017, 7, 42420 CrossRef CAS PubMed.
- S. Wang, L. Fu, Y. Zhang, J. Wang and Z. Zhang, J. Phys. Chem. C, 2018, 122, 24421 CrossRef CAS.
- Z. Hu, S. Hou, Y. Ji, T. Wen, W. Liu, H. Zhang, X. Shi, J. Yan and X. J. A. A. Wu, AIP Adv., 2014, 4, 117137 CrossRef.
- N. Xu, B. Bai, Q. Tan and G. Jin, Opt. Express, 2013, 21, 2987–3000 CrossRef CAS PubMed.
- E. Ricci and R. Novakovic, Gold Bull., 2001, 34, 41–49 CrossRef CAS.
- Z. Zhu, S. Zhang, C. Li, J. Zhang, J. Yu, X. Du, L. He and X. Zhang, Phys. Chem. Chem. Phys., 2018, 20, 1440–1446 RSC.
- S. Gómez-Graña, F. Hubert, F. Testard, A. Guerrero-Martínez, I. Grillo, L. M. Liz-Marzán and O. Spalla, Langmuir, 2012, 28, 1453–1459 CrossRef PubMed.
- T. Albogami, R. Lucien, S. Alshehri, J. McLaughlan, Z. Aslam, Z. Ong, S. D. Evans and K. Critchley, Dataset of Photothermal Reshaping and Cavity Formation in Silica-Coated Gold Nanorods Using Nanosecond Pulsed Lasers, 2025 DOI:10.5518/1787.
| This journal is © the Owner Societies 2026 |







