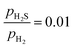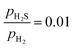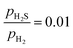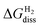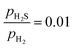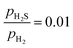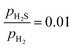Identifying the active nickel sites and hydrogen species in the phosphosulfide overlayer on the Ni2P hydrotreating catalyst: a DFT-D3 study
Alba B.
Vidal
 *a,
José Luis
Peña-Mena
a,
Rafael
Añez
a,
David S.
Coll
b,
Oscar
Hurtado-Aular
c,
Aníbal
Sierraalta
*a,
José Luis
Peña-Mena
a,
Rafael
Añez
a,
David S.
Coll
b,
Oscar
Hurtado-Aular
c,
Aníbal
Sierraalta
 a and
Joaquin L.
Brito
a and
Joaquin L.
Brito
 de
de
aLaboratorio de Química Física y Catálisis Computacional, Centro de Química “Dr Gabriel Chuchani”, Instituto Venezolano de Investigaciones Científicas (IVIC), Apartado 21827, Caracas, Venezuela. E-mail: abvidals@gmail.com
bLaboratorio de Físico Química Teórica de Materiales, Centro de Química “Dr Gabriel Chuchani”, Instituto Venezolano de Investigaciones Científicas (IVIC), Apartado 21827, Caracas, Venezuela
cDepartamento de Química, Universidad Nacional del Sur (UNS), Av. Alem 1253, Bahía Blanca, 8000, Argentina
dLaboratorio de Físico Química de Superficies, Centro de Química “Dr Gabriel Chuchani”, Instituto Venezolano de Investigaciones Científicas (IVIC), Apartado 21827, Caracas, Venezuela
eBiomass to Resources Group, Universidad Regional Amazónica IKIAM, Tena, Ecuador
First published on 5th March 2026
Abstract
Density functional theory (DFT) calculations and an atomistic thermodynamic approach were employed to identify the active nickel sites and determine the chemical nature of the hydrogen species in the nickel phosphosulfide overlayer formed on the (0001) and (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0) surfaces of the Ni2P hydrotreating catalyst. Our results showed that Ni(1) sites are less resistant to sulfur than Ni(2) sites, in agreement with experimental observations. Under HDS conditions, the fully sulfided (0001) surface of Ni2P was found to be structurally similar to the (111) surface of the low-activity sulfide Ni3S2, where closely coordinated Ni atoms are prone to sulfidation. Non-hydrogenated surfaces were always the most stable, suggesting that NiH and SH groups are unlikely to form on these surfaces. In contrast, the most stable (10
0) surfaces of the Ni2P hydrotreating catalyst. Our results showed that Ni(1) sites are less resistant to sulfur than Ni(2) sites, in agreement with experimental observations. Under HDS conditions, the fully sulfided (0001) surface of Ni2P was found to be structurally similar to the (111) surface of the low-activity sulfide Ni3S2, where closely coordinated Ni atoms are prone to sulfidation. Non-hydrogenated surfaces were always the most stable, suggesting that NiH and SH groups are unlikely to form on these surfaces. In contrast, the most stable (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0) surface was identified as a hydrogenated surface with coordinatively unsaturated Ni(2) atoms surrounded by SH groups. Depending on the electronegativity of their ligands (S or Ni), the hydrogen species exhibit either protonic (Hδ+) or hydridic (Hδ−) character. Our results suggest that protons within SH groups are the most likely reactive hydrogen species on the nickel phosphosulfide overlayer under reaction conditions, providing atomic-level insight into the origins of HYD activity in Ni2P catalysts.
0) surface was identified as a hydrogenated surface with coordinatively unsaturated Ni(2) atoms surrounded by SH groups. Depending on the electronegativity of their ligands (S or Ni), the hydrogen species exhibit either protonic (Hδ+) or hydridic (Hδ−) character. Our results suggest that protons within SH groups are the most likely reactive hydrogen species on the nickel phosphosulfide overlayer under reaction conditions, providing atomic-level insight into the origins of HYD activity in Ni2P catalysts.
1. Introduction
Nickel phosphide (Ni2P) has emerged as a promising candidate for a new generation of industrial hydrodesulfurization (HDS) catalysts.1–7 According to the literature, the active phase of the Ni2P catalyst is a nickel phosphosulfide overlayer that forms during the HDS reaction.1,8–12 This overlayer can also be generated by pretreatment with sulfidation agents, such as H2S or CS2 in the presence of H2, or by reducing sulfur-containing precursors, such as nickel hexathiodiphosphate (Ni2P2S6).13–16The active sites in HDS catalysts are primarily linked to coordinatively unsaturated metal sites (CUSs), sulfhydryl (SH) groups, and metal hydrides (MH).17,18 CUSs are S2− anion vacancies located at the edges or corners of catalyst nanoparticles. SH groups, on the other hand, result from the dissociative adsorption of H2 onto surface sulfur atoms. These groups behave as Brønsted acid sites.19,20 MH species are formed when H2 dissociatively adsorbs onto CUSs. Consequently, it is essential to understand the structural changes of Ni2P under reaction conditions and the specific catalytic functions of the active sites.
In an early study, Oyama et al. investigated the catalytic activity of Ni2P supported on low- and high-surface-area siliceous supports (SiO2-L, SiO2-H, and MCM-41) for the HDS of 4,6-dimethyldibenzothiophene (4,6-DMDBT).21 Extended X-ray absorption fine structure (EXAFS) analysis confirmed the presence of two types of nickel sites: Ni(1), with tetrahedral coordination to P, and Ni(2), with square pyramidal coordination to P. The analysis further revealed both Ni(1) and Ni(2) sites are present in large crystallites, whereas Ni(2) sites are more numerous on the small crystallites. Catalytic selectivity measurements showed that small crystallites exhibit higher selectivity toward HYD products, while large crystallites have higher selectivity toward DDS products. Based on product selectivity, it was concluded that the Ni(1) sites are primarily responsible for the DDS pathway, while the Ni(2) sites are highly active for the HYD pathway.
In a later study, Oyama et al. investigated the sulfur-resistant properties of the Ni2P catalyst in deep hydrodesulfurization.3 EXAFS analysis of spent catalysts revealed that Ni(2) sites are bound to sulfur with a lower Ni–S coordination as the particle size decreased. Based on EXAFS results, the authors concluded that the active Ni(2) sites on highly dispersed Ni2P catalysts are much more tolerant of sulfur than the tetrahedral Ni(1) sites. Furthermore, the higher selectivity for HYD products correlates with an increased number of surface Ni(2) sites, which predominate in smaller Ni2P particles.
To elucidate the catalytic mechanism of the HDS reaction on the Ni2P catalyst, it is essential to understand the state of adsorbed hydrogen on the nickel phosphosulfide overlayer. To the best of our knowledge, there are no experimental studies on the chemical nature of hydrogen species adsorbed on Ni2P as an HDS catalyst. Only Nelson et al., using density functional theory (DFT) calculations, have studied the presence of SH groups on the (0001) surface of the Ni2P catalyst.22 The authors calculated the free energy changes associated with H2S adsorption and its dissociation into –SH and sulfur on the (0001) Ni3P2-terminated surface as a function of temperature (100–1000 K) under H2 and H2S partial pressures of 5.0 and 0.1 MPa, respectively. Their results indicated that the relative stability of the adsorbed sulfur species follows the order H2S < SH < S. These results suggest that hydrogen bound as an SH group might not be thermodynamically stable under HDS reaction conditions.
On the other hand, electrocatalysis experiments have shown that incorporating sulfur improves the performance of Ni2P in the hydrogen evolution reaction (HER).23–25 Although the HER and HDS occur under different conditions, both processes involve hydrogen atoms adsorbed on the surface.26 It was found that sulfur incorporation accelerates the adsorption rate of reaction intermediates on the catalyst surface, thereby improving HER kinetics. Therefore, sulfur incorporated into the catalyst may act as an activity promoter or selectivity modifier.27
In this work, we combined DFT calculations with an atomistic thermodynamic analysis to investigate the stability of hydrogen species on the phosphosulfide overlayer formed on the (0001) and (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0) surfaces of the Ni2P catalyst under HDS conditions. Our goal is to elucidate the nature of the active nickel sites and the chemical characteristics of the hydrogen species, which are key to understanding the hydrogenation functionality of the phosphosulfide overlayer.
0) surfaces of the Ni2P catalyst under HDS conditions. Our goal is to elucidate the nature of the active nickel sites and the chemical characteristics of the hydrogen species, which are key to understanding the hydrogenation functionality of the phosphosulfide overlayer.
2. Computational details
2.1. Computational methods
DFT calculations were performed using the Vienna ab initio simulation package (VASP).28,29 The Perdew–Burke–Ernzerhof exchange–correlation functional with a generalized gradient approximation was used.30 The projector augmented wave (PAW) method was used to describe the electron–ion interactions.31 Standard PAW potentials with a valence electron distribution of Ni 3d84s2, P 3s23p3, S 3s23p4, and H 1s1 were used. A plane-wave kinetic energy cut-off of 400 eV was employed. The energy and atomic force convergence criteria were set to 10−5 eV and 0.01 eV Å−1, respectively. The Gaussian smearing method with a width of 0.05 eV was used to describe the partial occupancies of the electronic states. The DFT-D3 correction developed by Grimme et al. was used to capture dispersion interactions, which are important to the energetics of catalytic transformations.32–35 Gamma-centered k-point meshes of 5 × 5 × 9 and 7 × 7 × 7 were used to sample the Brillouin zone for the optimization of bulk Ni2P and Ni3S2, respectively. The calculated lattice parameters for bulk Ni2P are a = b = 5.836 Å and c = 3.307 Å, while for bulk Ni3S2 (rhombohedral representation), a = b = c = 4.034 Å. These values are in good agreement with the reported experimental values for Ni2P (a = b = 5.859 Å, c = 3.382 Å)36 and Ni3S2 (a = b = c = 4.082 Å).37 All surface calculations were performed using symmetric slabs. The (0001) Ni3P2 surface termination was modeled using a nine-atomic-layer (2 × 2) slab with a 3 × 3 × 1 gamma-centered k-point mesh (Fig. S1a). The (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0) AB Ni2P surface termination was modeled with ten-atomic-layer (2 × 2) slabs with a 3 × 5 × 1 Monkhorst–Pack k-point mesh (Fig. S1b). The (111) surface of Ni3S2 was modeled with eight-atomic-layer (2 × 2) slab with a 3 × 3 × 1 gamma-centered k-point mesh (Fig. S1c). A vacuum space of 20 Å was set to avoid interactions between periodic slabs, and all atoms were fully relaxed during the calculations. Bader charge38 and electron density difference (EDD) analyses were performed to assess the charge distribution and the chemical nature of the hydrogen species adsorbed on the surface. The EDD was calculated from the following equation:
0) AB Ni2P surface termination was modeled with ten-atomic-layer (2 × 2) slabs with a 3 × 5 × 1 Monkhorst–Pack k-point mesh (Fig. S1b). The (111) surface of Ni3S2 was modeled with eight-atomic-layer (2 × 2) slab with a 3 × 3 × 1 gamma-centered k-point mesh (Fig. S1c). A vacuum space of 20 Å was set to avoid interactions between periodic slabs, and all atoms were fully relaxed during the calculations. Bader charge38 and electron density difference (EDD) analyses were performed to assess the charge distribution and the chemical nature of the hydrogen species adsorbed on the surface. The EDD was calculated from the following equation:| Δρ = ρ(surface + adsorbate) − ρ(surface) − ρ(adsorbate) | (1) |
2.2. Surface models
To construct slab models of the possible structures of the nickel phosphosulfide overlayer on the Ni2P catalyst, we selected the (0001) and (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0) surfaces. Papawassiliou et al.,40 using 31P solid-state nuclear magnetic resonance (ssNMR), high-resolution transmission electron microscopy (HRTEM), X-ray diffraction (XRD), and DFT calculations, determined that Ni2P nanoparticles synthesized under excess phosphorus predominantly expose these surfaces. For the (0001) surface, the slab model was constructed from a (2 × 2) supercell with a Ni3P2 termination. This surface termination has been reported as the most thermodynamically stable.41,42 The surface is described by a hexagonal supercell (a = b = 11.672 Å) and contains eight P atoms and twelve Ni atoms in the outermost atomic layer, Ni12P8 (Fig. S1a). These Ni atoms are referred to as Ni(1). On the surface, the Ni(1) atoms form four three-fold adsorption sites (Ni3 sites), which are labeled as T sites, as illustrated in Fig. S1. For the (10
0) surfaces. Papawassiliou et al.,40 using 31P solid-state nuclear magnetic resonance (ssNMR), high-resolution transmission electron microscopy (HRTEM), X-ray diffraction (XRD), and DFT calculations, determined that Ni2P nanoparticles synthesized under excess phosphorus predominantly expose these surfaces. For the (0001) surface, the slab model was constructed from a (2 × 2) supercell with a Ni3P2 termination. This surface termination has been reported as the most thermodynamically stable.41,42 The surface is described by a hexagonal supercell (a = b = 11.672 Å) and contains eight P atoms and twelve Ni atoms in the outermost atomic layer, Ni12P8 (Fig. S1a). These Ni atoms are referred to as Ni(1). On the surface, the Ni(1) atoms form four three-fold adsorption sites (Ni3 sites), which are labeled as T sites, as illustrated in Fig. S1. For the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0) surface, the slab model surface was constructed from a (2 × 2) supercell with the AB-type Ni2P termination, which has also been reported as the most thermodynamically stable surface termination.41,42 The surface is described by a rectangular supercell (a = 11.672 Å and b = 6.614 Å) and contains four P atoms and eight Ni atoms in the outermost atomic layer, Ni8P4 (Fig. S1b). This surface contains two distinct types of Ni atoms: four Ni(1) and Ni(2). In Fig. S1b the Ni(2) nickel atoms are represented by light blue spheres to distinguish them from the Ni(1) nickel atoms. On the surface, the Ni atoms adopt a zigzag arrangement along the y-axis. (Fig. S1). This zigzag configuration exhibits two types of Ni3 sites: four T1 sites and four T2 sites. Each T1 site consists of two Ni(1) atoms and one Ni(2) atom, while the T2 site consists of two Ni(2) atoms and one Ni(1) atom. It has been shown that the S atoms are preferentially located at the T1 sites.42 At 50%S, the S atoms are preferentially adsorbed at the T1 sites in an alternating configuration. The adsorption energy difference between T1 and T2 sites was around 1 eV. For the (111) surface of the Ni3S2 catalyst (rhombohedral representation), the slab model was constructed from a (2 × 2) supercell with the Ni3S2 termination. The surface is described by a hexagonal supercell (a = b = 11.360 Å), with eight S atoms and twelve Ni atoms in the outermost atomic layer, Ni12S8 (Fig. S1c). The surface Ni atoms also form four Ni3 sites, which are also labeled as T sites.
0) surface, the slab model surface was constructed from a (2 × 2) supercell with the AB-type Ni2P termination, which has also been reported as the most thermodynamically stable surface termination.41,42 The surface is described by a rectangular supercell (a = 11.672 Å and b = 6.614 Å) and contains four P atoms and eight Ni atoms in the outermost atomic layer, Ni8P4 (Fig. S1b). This surface contains two distinct types of Ni atoms: four Ni(1) and Ni(2). In Fig. S1b the Ni(2) nickel atoms are represented by light blue spheres to distinguish them from the Ni(1) nickel atoms. On the surface, the Ni atoms adopt a zigzag arrangement along the y-axis. (Fig. S1). This zigzag configuration exhibits two types of Ni3 sites: four T1 sites and four T2 sites. Each T1 site consists of two Ni(1) atoms and one Ni(2) atom, while the T2 site consists of two Ni(2) atoms and one Ni(1) atom. It has been shown that the S atoms are preferentially located at the T1 sites.42 At 50%S, the S atoms are preferentially adsorbed at the T1 sites in an alternating configuration. The adsorption energy difference between T1 and T2 sites was around 1 eV. For the (111) surface of the Ni3S2 catalyst (rhombohedral representation), the slab model was constructed from a (2 × 2) supercell with the Ni3S2 termination. The surface is described by a hexagonal supercell (a = b = 11.360 Å), with eight S atoms and twelve Ni atoms in the outermost atomic layer, Ni12S8 (Fig. S1c). The surface Ni atoms also form four Ni3 sites, which are also labeled as T sites.
The slab models of the nickel phosphosulfide overlayer on the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0) and (0001) surfaces were constructed following the surface modification procedure previously reported.42 These slab models take into account the processes of sulfur adsorption and the replacement of surface phosphorus by sulfur at the outermost atomic layer. For the (0001)-Ni12P8 surface, the replacement of P by S was evaluated at 0%, 50%, and 100% of S, where 0%S corresponds to no replacement, 50%S corresponds to replacement with four S atoms, and 100%S corresponds to replacement with eight S atoms. Additionally, sulfur adsorption was evaluated at coverages of 75%S and 100%S, which correspond to the adsorption of three and four S atoms, respectively. Surface terminations are labeled according to the elemental composition of the outermost layer and the number of S atoms adsorbed. For the (10
0) and (0001) surfaces were constructed following the surface modification procedure previously reported.42 These slab models take into account the processes of sulfur adsorption and the replacement of surface phosphorus by sulfur at the outermost atomic layer. For the (0001)-Ni12P8 surface, the replacement of P by S was evaluated at 0%, 50%, and 100% of S, where 0%S corresponds to no replacement, 50%S corresponds to replacement with four S atoms, and 100%S corresponds to replacement with eight S atoms. Additionally, sulfur adsorption was evaluated at coverages of 75%S and 100%S, which correspond to the adsorption of three and four S atoms, respectively. Surface terminations are labeled according to the elemental composition of the outermost layer and the number of S atoms adsorbed. For the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8P4 surface, the replacement of P by S was evaluated at 0%, 50%, and 100% of S, where 0%S corresponds to no replacement, 50%S corresponds to replacement with two S atoms, and 100%S corresponds to replacement with four S atoms. This surface has two types of adsorption sites for sulfur: four T1 sites and four T2 sites (Fig. S1b). For the slab models of the hydrogenated surfaces, we evaluated the dissociative adsorption of one or two H2 molecules, corresponding to hydrogen coverages of 50% and 100%, respectively.
0)-Ni8P4 surface, the replacement of P by S was evaluated at 0%, 50%, and 100% of S, where 0%S corresponds to no replacement, 50%S corresponds to replacement with two S atoms, and 100%S corresponds to replacement with four S atoms. This surface has two types of adsorption sites for sulfur: four T1 sites and four T2 sites (Fig. S1b). For the slab models of the hydrogenated surfaces, we evaluated the dissociative adsorption of one or two H2 molecules, corresponding to hydrogen coverages of 50% and 100%, respectively.
2.3. Thermodynamic approach
Since HDS operating conditions are sulforeductive conditions in which H2 is more abundant than H2S in the gas phase, PH3 could be produced when P atoms are replaced by S atoms in the presence of H2. Therefore, the possibility of complete sulfidation of bulk Ni2P to bulk Ni3S2 under relevant conditions was evaluated by the following reaction:| Ni2P(bulk) + 4/3H2S + 3/2H2 → 2/3Ni3S2(bulk) + PH3 + 4/3H2 | (2) |
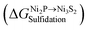 , is then given by
, is then given by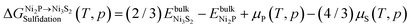 | (3) |
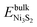 and
and 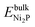 are the DFT total energies of bulk Ni3S2 and Ni2P phases, respectively. The chemical potential of phosphorus, μP(T, p), is determined by the chemical equilibrium with the gas-phase mixture of PH3 and H2,
are the DFT total energies of bulk Ni3S2 and Ni2P phases, respectively. The chemical potential of phosphorus, μP(T, p), is determined by the chemical equilibrium with the gas-phase mixture of PH3 and H2,| μP(T, p) = μPH3(T, p) − (3/2)μH2(T, p) | (4) |
On the other hand, the chemical potential of sulfur, μS(T, p), is determined by the chemical equilibrium with the gas-phase mixture of H2 and H2S,
| μS(T, p) = μH2S(T, p) − μH2(T, p) | (5) |
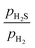 ratio ranges from 0.01 to 0.05, and the temperature varies between 573 and 700 K. In this study, we selected 573 K as the working temperature because several experimental studies using Ni2P catalyst have performed HDS measurements at this temperature.1,2,10,12,13
ratio ranges from 0.01 to 0.05, and the temperature varies between 573 and 700 K. In this study, we selected 573 K as the working temperature because several experimental studies using Ni2P catalyst have performed HDS measurements at this temperature.1,2,10,12,13
The possibility of replacing the surface P atoms with S atoms, as well as the adsorption of S atoms under relevant conditions, can be described as
| NixPy + (z + a)H2S + (3/2z)H2 → NixP(y−z)Sz_aS + zPH3 + (z + a)H2 | (6) |
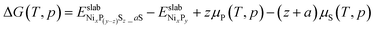 | (7) |
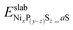 is the DFT total energy of a slab model (either the (0001). or (10
is the DFT total energy of a slab model (either the (0001). or (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0) surface), in which z surface phosphorus atoms have been replaced by sulfur atoms, in addition to the adsorption of a sulfur atoms on the Ni3 sites.
0) surface), in which z surface phosphorus atoms have been replaced by sulfur atoms, in addition to the adsorption of a sulfur atoms on the Ni3 sites. 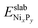 is the DFT total energy of the pristine surface, which is taken as a reference.
is the DFT total energy of the pristine surface, which is taken as a reference.
On the other hand, the Gibbs free energy change associated only with S atom adsorption (ΔGSads), is given by
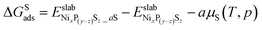 | (8) |
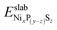 and
and 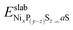 are the DFT total energy of sulfided and clean surfaces, respectively. a is the number of S atoms adsorbed.
are the DFT total energy of sulfided and clean surfaces, respectively. a is the number of S atoms adsorbed.
Finally, the dissociative adsorption of H2 as NiH or SH groups on the nickel phosphosulfide overlayer was evaluated by the following reaction:
| NixP(y−z)Sz_aS + nH2 → NixP(y−z)Sz_aS_2nH | (9) |
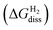 , is given by
, is given by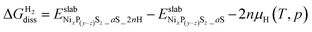 | (10) |
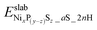 and
and 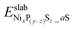 are the DFT total energy of hydrogenated and non-hydrogenated surfaces, respectively. n is the number of dissociatively adsorbed H2 molecules, and μH(T, p) is the hydrogen chemical potential, where μH(T, p) = (½)μH2(T, p).
are the DFT total energy of hydrogenated and non-hydrogenated surfaces, respectively. n is the number of dissociatively adsorbed H2 molecules, and μH(T, p) is the hydrogen chemical potential, where μH(T, p) = (½)μH2(T, p).
3. Results and discussion
3.1. Bulk Ni2P stability under the H2S/H2 gas mixture
Group 8 transition metal catalysts (nickel, palladium, and platinum) are highly susceptible to sulfur poisoning, with nickel being the most susceptible to sulfide formation.43–45 However, nickel phosphide-based catalysts have been shown to be highly resistant to sulfur poisoning.3,7,12,46 Given that Ni3S2 is the most stable stoichiometry of nickel sulfide under HDS reaction conditions,47–49 we investigated the relative stability of bulk Ni2P over bulk Ni3S2 in the presence of the H2S/H2 gas mixture. Bulk Ni2P belongs to the hexagonal crystal system with the P![[6 with combining macron]](https://www.rsc.org/images/entities/char_0036_0304.gif) 2m space group (Fig. 1a). It is characterized by the presence of two distinct types of Ni atoms, denoted as Ni(1) and Ni(2).21,50 Ni(1) atoms have a quasi-tetrahedral coordination geometry with the four nearest-neighbor P atoms, while Ni(2) atoms exhibit a square pyramidal coordination geometry with the five nearest-neighbor P atoms. In Fig. 1a the Ni(2) nickel atoms are represented by light blue spheres to distinguish them from the Ni(1) nickel atoms. Bulk Ni3S2 belongs to the trigonal crystal system with the R32 space group. It can be described in both rhombohedral (Fig. 1b) and hexagonal (Fig. 1c) lattice representations. This bulk only has one type of Ni atom, which has a quasi-tetrahedral coordination geometry with the four nearest-neighbor S atoms.
2m space group (Fig. 1a). It is characterized by the presence of two distinct types of Ni atoms, denoted as Ni(1) and Ni(2).21,50 Ni(1) atoms have a quasi-tetrahedral coordination geometry with the four nearest-neighbor P atoms, while Ni(2) atoms exhibit a square pyramidal coordination geometry with the five nearest-neighbor P atoms. In Fig. 1a the Ni(2) nickel atoms are represented by light blue spheres to distinguish them from the Ni(1) nickel atoms. Bulk Ni3S2 belongs to the trigonal crystal system with the R32 space group. It can be described in both rhombohedral (Fig. 1b) and hexagonal (Fig. 1c) lattice representations. This bulk only has one type of Ni atom, which has a quasi-tetrahedral coordination geometry with the four nearest-neighbor S atoms.
Fig. 1d shows the phase diagram of the free energy change for complete sulfidation of bulk Ni2P to bulk Ni3S2 as a function of μP(T, p) and μS(T, p). The regions of stability of the bulk phases are represented by the distinct colored areas. The red dotted line in Fig. 1d represents a typical HDS 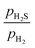 ratio. It is observed that the complete sulfidation of Ni2P to Ni3S2 may occur when μP(T, p) is lower than −6.77 eV. At 573 K, this condition is met when pPH3 is lower than 3 × 10−15 bar with pH2 = 1 bar or when pPH3 is lower than 10−13 bar with pH2 = 10 bar. PH3 is a very strong phosphiding agent, and consequently, competition could exist between the sulfidation and phosphidation processes. Our findings suggest that bulk Ni2P will not undergo complete sulfidation to bulk Ni3S2 as long as there is a trace amount of PH3 in the gas phase. This result correlates with the sulfur resistance observed in Ni2P catalysts under HDS conditions.3,7,12,46
ratio. It is observed that the complete sulfidation of Ni2P to Ni3S2 may occur when μP(T, p) is lower than −6.77 eV. At 573 K, this condition is met when pPH3 is lower than 3 × 10−15 bar with pH2 = 1 bar or when pPH3 is lower than 10−13 bar with pH2 = 10 bar. PH3 is a very strong phosphiding agent, and consequently, competition could exist between the sulfidation and phosphidation processes. Our findings suggest that bulk Ni2P will not undergo complete sulfidation to bulk Ni3S2 as long as there is a trace amount of PH3 in the gas phase. This result correlates with the sulfur resistance observed in Ni2P catalysts under HDS conditions.3,7,12,46
3.2. Phosphosulfide overlayer on the (0001) surface
We revisited the adsorption and replacement processes of sulfur atoms on the (0001) surface by calculating the free energy change as a function of the phosphorus and sulfur chemical potentials, μP(T, p) and μS(T, p). The analysis was carried out considering the thermodynamic limit in which bulk Ni2P transforms into bulk Ni3S2. Fig. 2a presents the corresponding free energy phase diagram. The stability regions of the different surface compositions are indicated by the different shades of green. Fig. 2b–d show the top view of the corresponding surface structures. Surface terminations are labeled according to the elemental composition of the outermost layer and the number of S atoms adsorbed. In Fig. 2a, the red dotted line represents a typical HDS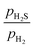 ratio, while the blue dotted line indicates the phase boundary between the bulk Ni2P and Ni3S2. The (0001)-Ni12P8_4S, (0001)-Ni12P4S4_4S, and (0001)-Ni12S8_4S surfaces are the most stable at a
ratio, while the blue dotted line indicates the phase boundary between the bulk Ni2P and Ni3S2. The (0001)-Ni12P8_4S, (0001)-Ni12P4S4_4S, and (0001)-Ni12S8_4S surfaces are the most stable at a 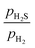 ratio of 0.01. All of these surfaces have all Ni3 sites covered by S atoms. The findings of this study are consistent with those previously reported.42
ratio of 0.01. All of these surfaces have all Ni3 sites covered by S atoms. The findings of this study are consistent with those previously reported.42
Note that the (0001)-Ni12S8_4S surface is the most stable at very low PH3 partial pressures. In the context of HDS experiments, the presence of PH3 in the gas mixture has not been detected, suggesting that only a trace amount of PH3 might be produced by the replacement of surface P atoms by S atoms. In contrast to the bulk phase, a very low pPH3 enables the gradual replacement of all surface P atoms with S atoms, resulting in the formation of a phosphosulfide overlayer on the (0001)-Ni12P8 surface termination. Therefore, our results suggest that the (0001)-Ni12S8_4S surface could be the most stable surface termination under HDS reaction conditions. In this regard, Oyama et al. used EXAFS technique to study the structural properties of supported Ni2P catalysts after the HDS reaction of 4,6-dimethyldibenzothiophene.3 According to EXAFS line shape analysis, catalysts with shorter Ni–Ni bond lengths are more likely to form a phosphosulfide phase. It was found that the tetrahedral Ni(1) sites are less resistant to sulfur than the square-pyramidal Ni(2) sites. On the (0001) surface, the Ni(1) atoms are closely coordinated with another two Ni(1) atoms and, therefore, may be prone to sulfidation. For the Ni2P catalyst, the active sites are believed to be the coordinatively unsaturated surface nickel atoms belonging to the phosphosulfide phase. Our results suggest that the (0001)-Ni12S8_4S surface could not be active under HDS conditions because the Ni3 sites, which consist of Ni(1)-type atoms, would be blocked by sulfur atoms.
It is noteworthy that the surface structure of the (0001)-Ni12S8_4S surface of Ni2P closely resembles that of the (111)-Ni12S8_4S surface of Ni3S2 (Fig. 3a and b). Both surface terminations share the same composition and feature Ni3 sites, where each Ni atom is bonded to two surface S atoms. The Ni–Ni bond distances are 2.646 Å and 2.558 Å, respectively. On the (0001)-Ni12S8_4S surface, the Ni3 sites are capped above by an S atom and below a P atom. On the (111)-Ni12S8_4S surface, the Ni3 sites are capped above and below by an S atom. This results in an almost regular trigonal bipyramid. It is well known that Ni3S2 catalysts exhibit very low activity in various hydrotreating reactions.47,48,51 In this regard, Aray et al. used DFT calculations to investigate the surface stability of the low-index Miller surfaces and the equilibrium morphology of the Ni3S2 catalyst.52 It was found that only the (111) and (11![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ) type surface faces are exposed under HDS conditions. The S-covered surfaces were the most stable in the whole range of the
) type surface faces are exposed under HDS conditions. The S-covered surfaces were the most stable in the whole range of the 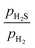 ratio. These findings suggested that the surfaces of the Ni3S2 catalyst may not have coordinatively unsaturated metal sites available for the reaction.
ratio. These findings suggested that the surfaces of the Ni3S2 catalyst may not have coordinatively unsaturated metal sites available for the reaction.
For comparison, we calculated the Gibbs free energy change associated with S atoms adsorption (ΔGSads) on the (0001)-Ni12S8 and the (111)-Ni12S8 surfaces as a function of μS(T, p) (Fig. 3c and d). The clean surface was taken as a reference. Additionally, ΔGSads was calculated for the (0001)-Ni12P8 and (0001)-Ni12P4S4 surfaces (Fig. S2). It is observed that the fully covered surfaces (with four S atoms adsorbed) are the most stable across the entire range of evaluated μS(T, p). For example, at T = 573 K and 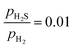 , the energy required to create a sulfur vacancy on the fully covered surface shows the following trend: (111)-Ni12S8_4S (1.06 eV) ≈ (0001)-Ni12P8_4S (1.05 eV) > (0001)-Ni12P4S4_4S (0.63 eV) > (0001)-Ni12S8_4S (0.46 eV). For the (0001) surface, vacancy formation becomes energetically more favorable as surface P atoms are replaced by S atoms; this is due to the strain effect exerted by the S atoms.42 Within our thermodynamic framework, surfaces with coordinatively unsaturated sites consisting of spatially isolated Ni3 ensembles may not be stable under HDS conditions.
, the energy required to create a sulfur vacancy on the fully covered surface shows the following trend: (111)-Ni12S8_4S (1.06 eV) ≈ (0001)-Ni12P8_4S (1.05 eV) > (0001)-Ni12P4S4_4S (0.63 eV) > (0001)-Ni12S8_4S (0.46 eV). For the (0001) surface, vacancy formation becomes energetically more favorable as surface P atoms are replaced by S atoms; this is due to the strain effect exerted by the S atoms.42 Within our thermodynamic framework, surfaces with coordinatively unsaturated sites consisting of spatially isolated Ni3 ensembles may not be stable under HDS conditions.
3.3. Dissociative adsorption of H2 onto the phosphosulfide overlayer on the (0001) surface
Dissociative adsorption of H2 onto the nickel phosphosulfide overlayer can lead to the formation of SH or NiH groups. Fig. 4a–d show the optimized structures of the hydrogenated surfaces resulting from the dissociative adsorption of one and two H2 molecules on the (0001)-Ni12S8_4S and (111)-Ni12S8_4S surfaces. Fig. S3 shows the configurations evaluated for the dissociative adsorption of one H2 molecule on the (0001)-Ni12S8_4S surface. When the SH group forms, the sulfur atom binds to two of the three Ni(1) atoms at the Ni3 site. This leaves the third Ni(1) atom coordinatively unsaturated. Fig. 4e and f display as a function of μH(T, p). The non-hydrogenated surface was taken as a reference. We also calculated
as a function of μH(T, p). The non-hydrogenated surface was taken as a reference. We also calculated 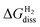 for the (0001)-Ni12P8_4S and (0001)-Ni12P4S4_4S surfaces (Fig. S4). The configurations evaluated for the dissociative adsorption of one H2 molecule on the (0001)-Ni12P8_4S and (0001)-Ni12P4S4_4S surfaces are shown in Fig. S5 and S6, respectively. It is observed that the weakest hydrogen adsorption occurs on the (111)-Ni12S8_4S surface. For example, at T = 573 K and pH2 = 10 bar, the energy required for the dissociative adsorption of one H2 molecule shows the following trend: (111)-Ni12S8_2S_2SH (0.97 eV) > (0001)-Ni12P8_2S_2SH (0.68 eV) > (0001)-Ni12P4S4_2S_2SH (0.62 eV) > (0001)-Ni12S8_2S_2SH (0.39 eV). For the dissociative adsorption of two H2 molecules on fully sulfur-covered surfaces, the corresponding energy trend is: (111)-Ni12S8_4SH (1.94 eV) > (0001)-Ni12P8_4SH (1.44 eV) > (0001)-Ni12P4S4_4SH (1.34 eV) > (0001)-Ni12S8_4SH (1.05 eV). For the (0001) surface, hydrogen dissociative adsorption surface becomes energetically favorable as surface P atoms are replaced by S atoms. Nevertheless, non-hydrogenated surfaces are always the most stable, which suggests that SH groups are unlikely to form on these surfaces.
for the (0001)-Ni12P8_4S and (0001)-Ni12P4S4_4S surfaces (Fig. S4). The configurations evaluated for the dissociative adsorption of one H2 molecule on the (0001)-Ni12P8_4S and (0001)-Ni12P4S4_4S surfaces are shown in Fig. S5 and S6, respectively. It is observed that the weakest hydrogen adsorption occurs on the (111)-Ni12S8_4S surface. For example, at T = 573 K and pH2 = 10 bar, the energy required for the dissociative adsorption of one H2 molecule shows the following trend: (111)-Ni12S8_2S_2SH (0.97 eV) > (0001)-Ni12P8_2S_2SH (0.68 eV) > (0001)-Ni12P4S4_2S_2SH (0.62 eV) > (0001)-Ni12S8_2S_2SH (0.39 eV). For the dissociative adsorption of two H2 molecules on fully sulfur-covered surfaces, the corresponding energy trend is: (111)-Ni12S8_4SH (1.94 eV) > (0001)-Ni12P8_4SH (1.44 eV) > (0001)-Ni12P4S4_4SH (1.34 eV) > (0001)-Ni12S8_4SH (1.05 eV). For the (0001) surface, hydrogen dissociative adsorption surface becomes energetically favorable as surface P atoms are replaced by S atoms. Nevertheless, non-hydrogenated surfaces are always the most stable, which suggests that SH groups are unlikely to form on these surfaces.
3.4. Phosphosulfide overlayer on the (10![[1 with combining macron]](https://www.rsc.org/images/entities/h3_char_0031_0304.gif) 0) surface
0) surface
Fig. 5a shows the phase diagram of the free energy change for the processes of adsorption and replacement by S atoms on the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0) surface as a function of μP(T, p) and μS(T, p). The stability regions of the different surface compositions are indicated by the different shades of green. Fig. 5b–g show the top views of the corresponding surface structures. In all of these surfaces, S atoms are preferentially adsorbed at the T1 sites. In Fig. 6a, the red dotted line represents a typical HDS
0) surface as a function of μP(T, p) and μS(T, p). The stability regions of the different surface compositions are indicated by the different shades of green. Fig. 5b–g show the top views of the corresponding surface structures. In all of these surfaces, S atoms are preferentially adsorbed at the T1 sites. In Fig. 6a, the red dotted line represents a typical HDS 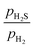 ratio, while the blue dotted line indicates the phase boundary between the bulk Ni2P and Ni3S2. The (10
ratio, while the blue dotted line indicates the phase boundary between the bulk Ni2P and Ni3S2. The (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8P4_2S, (10
0)-Ni8P4_2S, (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8P2S2_2S, and (10
0)-Ni8P2S2_2S, and (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_2S surfaces are the most stable at a
0)-Ni8S4_2S surfaces are the most stable at a 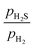 ratio of 0.01. All of these surfaces have the Ni3 sites partially covered by S atoms in an alternating configuration. The findings of this study are consistent with those previously reported.42 It is observed that the (10
ratio of 0.01. All of these surfaces have the Ni3 sites partially covered by S atoms in an alternating configuration. The findings of this study are consistent with those previously reported.42 It is observed that the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_2S surface is the most stable at very low PH3 partial pressures. Therefore, our results suggest that the (10
0)-Ni8S4_2S surface is the most stable at very low PH3 partial pressures. Therefore, our results suggest that the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_2S surface could be the most stable surface termination under HDS reaction conditions. On the (10
0)-Ni8S4_2S surface could be the most stable surface termination under HDS reaction conditions. On the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_2S surface, each T1 site consists of two Ni(1) atoms and one Ni(2) atom, with the Ni(1) atoms shared with the next T1 site. As a result, on the uncovered T1 sites, only the Ni(2) atoms are coordinatively unsaturated (Fig. 5f). Our findings are consistent with the EXAFS results by Oyama et al. that showed the square-pyramidal Ni(2) sites bind to sulfur with a lower Ni-S coordination.3 Therefore, the nickel phosphosulfide phase contains coordinatively unsaturated Ni(2)-type atoms available for the HDS reaction.
0)-Ni8S4_2S surface, each T1 site consists of two Ni(1) atoms and one Ni(2) atom, with the Ni(1) atoms shared with the next T1 site. As a result, on the uncovered T1 sites, only the Ni(2) atoms are coordinatively unsaturated (Fig. 5f). Our findings are consistent with the EXAFS results by Oyama et al. that showed the square-pyramidal Ni(2) sites bind to sulfur with a lower Ni-S coordination.3 Therefore, the nickel phosphosulfide phase contains coordinatively unsaturated Ni(2)-type atoms available for the HDS reaction.
3.5. Dissociative adsorption of H2 onto the phosphosulfide overlayer on the (10![[1 with combining macron]](https://www.rsc.org/images/entities/h3_char_0031_0304.gif) 0) surface
0) surface
Fig. 6a shows the phase diagram of the free energy change for the dissociative adsorption of one and two H2 molecules on the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_2S and (10
0)-Ni8S4_2S and (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_4S surfaces as a function of μH(T, p) and μS(T, p). The (10
0)-Ni8S4_4S surfaces as a function of μH(T, p) and μS(T, p). The (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_2S surface is taken as a reference. For example, the dissociative adsorption of two H2 molecules in the (10
0)-Ni8S4_2S surface is taken as a reference. For example, the dissociative adsorption of two H2 molecules in the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_4S surface was calculated as
0)-Ni8S4_4S surface was calculated as 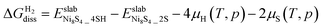 . Fig. 6b–d show the optimized structures for the most stable hydrogenated surfaces. Fig. S7 shows the configurations evaluated for the dissociative adsorption of one H2 molecule on the (10
. Fig. 6b–d show the optimized structures for the most stable hydrogenated surfaces. Fig. S7 shows the configurations evaluated for the dissociative adsorption of one H2 molecule on the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_2S. When H adsorbs to form an SH group, the S atom binds in a bridging position between the Ni(1) and Ni(2) atoms. Similarly, when the H atom adsorbs to form a NiH species, it occupies a bridging position between the Ni(1) and Ni(2) atoms. For the (10
0)-Ni8S4_2S. When H adsorbs to form an SH group, the S atom binds in a bridging position between the Ni(1) and Ni(2) atoms. Similarly, when the H atom adsorbs to form a NiH species, it occupies a bridging position between the Ni(1) and Ni(2) atoms. For the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_2S surface, the dissociative adsorption of one H2 molecule produces two SH groups, leading to the (10
0)-Ni8S4_2S surface, the dissociative adsorption of one H2 molecule produces two SH groups, leading to the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_2SH surface (Fig. 6b). When the SH groups form on this surface, two Ni(1) atoms become coordinatively unsaturated. On the other hand, the dissociative adsorption of two H2 molecules produces two SH groups and two NiH groups, leading to the (10
0)-Ni8S4_2SH surface (Fig. 6b). When the SH groups form on this surface, two Ni(1) atoms become coordinatively unsaturated. On the other hand, the dissociative adsorption of two H2 molecules produces two SH groups and two NiH groups, leading to the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_2SH_2NiH surface (Fig. 6c). For the (10
0)-Ni8S4_2SH_2NiH surface (Fig. 6c). For the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_4S surface, the dissociative adsorption of two H2 molecules produces four SH groups, leading to the (10
0)-Ni8S4_4S surface, the dissociative adsorption of two H2 molecules produces four SH groups, leading to the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_4SH surface (Fig. 6d). We also calculated the phase diagram of the free energy change for the dissociative adsorption of H2 on the (10
0)-Ni8S4_4SH surface (Fig. 6d). We also calculated the phase diagram of the free energy change for the dissociative adsorption of H2 on the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8P4_2S, (10
0)-Ni8P4_2S, (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8P4_4S, (10
0)-Ni8P4_4S, (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8P2S2_2S, and (10
0)-Ni8P2S2_2S, and (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8P2S2_4S surfaces (Fig. S8). The configurations evaluated for the dissociative adsorption of one H2 molecule on the (10
0)-Ni8P2S2_4S surfaces (Fig. S8). The configurations evaluated for the dissociative adsorption of one H2 molecule on the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8P4_2S and (10
0)-Ni8P4_2S and (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8P2S2_2S surfaces are shown in Fig. S9 and S10, respectively.
0)-Ni8P2S2_2S surfaces are shown in Fig. S9 and S10, respectively.
As shown in Fig. 6a, keeping the 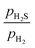 ratio constant at 0.01 while increasing pH2, results in two phase transitions at the most stable surface. First, the (10
ratio constant at 0.01 while increasing pH2, results in two phase transitions at the most stable surface. First, the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_2S surface appears, and then the (10
0)-Ni8S4_2S surface appears, and then the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_4SH surface. The hydrogenated surface (10
0)-Ni8S4_4SH surface. The hydrogenated surface (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_4SH is stable at pH2 above 10 bar. Hydrogenated surfaces resulting from the dissociative adsorption of H2 on the (10
0)-Ni8S4_4SH is stable at pH2 above 10 bar. Hydrogenated surfaces resulting from the dissociative adsorption of H2 on the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_2S surface become stable at lower
0)-Ni8S4_2S surface become stable at lower 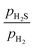 ratios and higher pH2. For instance, the (10
ratios and higher pH2. For instance, the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_2SH surface is stable at pH2 above 10 bar, while the (10
0)-Ni8S4_2SH surface is stable at pH2 above 10 bar, while the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_2SH_2NiH surface is stable at pH2 above 100 bar. These results imply that surfaces with coordinatively unsaturated Ni(1) atoms, or with H atoms adsorbed in a bridging position between the Ni(1) and Ni(2) atoms, may only be stable under strong reducing conditions. This trend was also observed on the surfaces with 0% and 50% of replacement with S (Fig. S8). On the other hand, the energy required for the dissociative adsorption of two H2 molecules on fully sulfur-covered surfaces at T = 573 K and pH2 = 10 bar follows the trend: (10
0)-Ni8S4_2SH_2NiH surface is stable at pH2 above 100 bar. These results imply that surfaces with coordinatively unsaturated Ni(1) atoms, or with H atoms adsorbed in a bridging position between the Ni(1) and Ni(2) atoms, may only be stable under strong reducing conditions. This trend was also observed on the surfaces with 0% and 50% of replacement with S (Fig. S8). On the other hand, the energy required for the dissociative adsorption of two H2 molecules on fully sulfur-covered surfaces at T = 573 K and pH2 = 10 bar follows the trend: (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8P4_4SH (−0.07 eV) > (10
0)-Ni8P4_4SH (−0.07 eV) > (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8P2S2_4SH (−0.14 eV) > (10
0)-Ni8P2S2_4SH (−0.14 eV) > (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni8S4_4SH (−0.42 eV). These results suggest that replacing surface phosphorus atoms with sulfur promotes the H2 dissociation process.
0)-Ni8S4_4SH (−0.42 eV). These results suggest that replacing surface phosphorus atoms with sulfur promotes the H2 dissociation process.
As mentioned above, Oyama et al. attribute the high HYD activity observed in small particles to the presence of S-resistant Ni(2) sites.3 Therefore, we used a (2 × 4) supercell to explore the stability of surfaces with SH groups and coordinatively unsaturated Ni(2) atoms. The (2 × 4) supercell size is an expansion along the y-axis. We evaluated the dissociative adsorption of one, two, and four H2 molecules, corresponding to hydrogen coverages of 25%, 50%, and 100%, respectively. The surface compositions evaluated were the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_4S, (10
0)-Ni16S8_4S, (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_6S, and (10
0)-Ni16S8_6S, and (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_8S surfaces, which correspond to sulfur coverages of 50%, 75%, and 100%, respectively. The supercell models are shown in Fig. S11. Fig. 7a shows that the (10
0)-Ni16S8_8S surfaces, which correspond to sulfur coverages of 50%, 75%, and 100%, respectively. The supercell models are shown in Fig. S11. Fig. 7a shows that the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_2S_4SH and (10
0)-Ni16S8_2S_4SH and (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_6SH_2NiH surfaces are the most stable hydrogenated surfaces at a
0)-Ni16S8_6SH_2NiH surfaces are the most stable hydrogenated surfaces at a 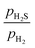 ratio of 0.01. The (10
ratio of 0.01. The (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_6SH_2NiH surface is only stable at pH2 above 104 bar. Additionally, it is found that the (10
0)-Ni16S8_6SH_2NiH surface is only stable at pH2 above 104 bar. Additionally, it is found that the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_4SH surface, which has coordinatively unsaturated Ni(1) atoms, is not stable under any reaction conditions (Fig. S12). Surfaces with H atoms adsorbed in a bridging position between the Ni(1) and Ni(2) atoms, such as the (10
0)-Ni16S8_4SH surface, which has coordinatively unsaturated Ni(1) atoms, is not stable under any reaction conditions (Fig. S12). Surfaces with H atoms adsorbed in a bridging position between the Ni(1) and Ni(2) atoms, such as the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_4SH_4NiH surface, are only stable under strong reducing conditions. At high
0)-Ni16S8_4SH_4NiH surface, are only stable under strong reducing conditions. At high 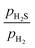 ratios, the (10
ratios, the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_8SH surface is the most stable hydrogenated surface. These results suggest that hydrogenated surfaces, such as (10
0)-Ni16S8_8SH surface is the most stable hydrogenated surface. These results suggest that hydrogenated surfaces, such as (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_2S_4SH, which have coordinatively unsaturated Ni(2) atoms, may be stable under HDS reaction conditions. Note that the SH groups surround the Ni(2) atoms (Fig. 7b). These findings may help explain the high HYD selectivity observed at Ni(2) sites, as reported by Oyama et al.3 Organosulfur compounds adsorbed on Ni(2) sites could react with H atoms from adjacent SH groups, which may result in the formation of hydrogenated intermediates and subsequent C–S bond cleavage. A detailed kinetic analysis of the HDS reaction mechanism, however, is beyond the scope of the present work.
0)-Ni16S8_2S_4SH, which have coordinatively unsaturated Ni(2) atoms, may be stable under HDS reaction conditions. Note that the SH groups surround the Ni(2) atoms (Fig. 7b). These findings may help explain the high HYD selectivity observed at Ni(2) sites, as reported by Oyama et al.3 Organosulfur compounds adsorbed on Ni(2) sites could react with H atoms from adjacent SH groups, which may result in the formation of hydrogenated intermediates and subsequent C–S bond cleavage. A detailed kinetic analysis of the HDS reaction mechanism, however, is beyond the scope of the present work.
To gain insight into the chemical nature of the hydrogen species on the surface, we performed Bader charge and electron density difference analyses. The formal charges of the different adsorbed species on the surface are H−, SH−, and S2− The stabilization of hydrogen as a proton involves an oxidation process, as it is accompanied by the oxidation of S atoms within the SH group. In contrast, the stabilization of hydrogen as a hydride corresponds to a reduction process.53 The Bader charges (q) of the surface atoms for the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_6S, (10
0)-Ni16S8_6S, (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_2S_4SH and (10
0)-Ni16S8_2S_4SH and (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_6SH_2NiH surfaces are summarized in Table 1. The hydrogen atoms are characterized by a different Bader charge, which reflects their chemical identities. For the (10
0)-Ni16S8_6SH_2NiH surfaces are summarized in Table 1. The hydrogen atoms are characterized by a different Bader charge, which reflects their chemical identities. For the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_2S_4SH surface, when H adsorbs as a proton (H+) to form SH groups, each H atom carries a low positive charge of +0.04e. The charge on the S atoms is found to be more negative as S anions (−0.48e) than as SH groups (−0.30e). For the (10
0)-Ni16S8_2S_4SH surface, when H adsorbs as a proton (H+) to form SH groups, each H atom carries a low positive charge of +0.04e. The charge on the S atoms is found to be more negative as S anions (−0.48e) than as SH groups (−0.30e). For the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_6SH_2NiH surface, when H adsorbs as a hydride (H−), each H atom carries a negative charge of −0.17e. Cai et al. reported the Bader charge analysis of hydrogen atoms adsorbed on the RuS2 catalyst. They found that hydrogen atoms bonded to sulfur atoms carry a positive charge ranging from +0.02e to +0.15e, whereas those adsorbed on ruthenium atoms exhibit a negative charge of approximately −0.20e.18 The Bader charges obtained in our study are consistent with their results in terms of both magnitude and sign. We also calculated the EDD to illustrate the redistribution of electron density due to the interaction between these species and the surface. Fig. 8a and b show the EDD plots for the (10
0)-Ni16S8_6SH_2NiH surface, when H adsorbs as a hydride (H−), each H atom carries a negative charge of −0.17e. Cai et al. reported the Bader charge analysis of hydrogen atoms adsorbed on the RuS2 catalyst. They found that hydrogen atoms bonded to sulfur atoms carry a positive charge ranging from +0.02e to +0.15e, whereas those adsorbed on ruthenium atoms exhibit a negative charge of approximately −0.20e.18 The Bader charges obtained in our study are consistent with their results in terms of both magnitude and sign. We also calculated the EDD to illustrate the redistribution of electron density due to the interaction between these species and the surface. Fig. 8a and b show the EDD plots for the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_2S_4SH and (10
0)-Ni16S8_2S_4SH and (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_6SH_2NiH surfaces, respectively. The densities of the S, H, and SH species were used as adsorbate references. In these plots, the blue regions represent electron density accumulation, and the red regions represent electron density depletion. For instance, the electron density accumulates on the adsorbed H atoms on the (10
0)-Ni16S8_6SH_2NiH surfaces, respectively. The densities of the S, H, and SH species were used as adsorbate references. In these plots, the blue regions represent electron density accumulation, and the red regions represent electron density depletion. For instance, the electron density accumulates on the adsorbed H atoms on the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_6SH_2NiH surface, which is consistent with a hydridic character. Additionally, the redistribution of electron density differs between the SH groups due to the local chemical environment that each SH group experiences in its immediate surroundings (Fig. 8b). Overall, these hydrogen species exhibit either proton or hydride character, depending on the electronegativity of their ligands (sulfur or metal).18
0)-Ni16S8_6SH_2NiH surface, which is consistent with a hydridic character. Additionally, the redistribution of electron density differs between the SH groups due to the local chemical environment that each SH group experiences in its immediate surroundings (Fig. 8b). Overall, these hydrogen species exhibit either proton or hydride character, depending on the electronegativity of their ligands (sulfur or metal).18
![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_2S_4SH, and (10
0)-Ni16S8_2S_4SH, and (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_6SH_2NiH surfaces
0)-Ni16S8_6SH_2NiH surfaces
| Surface | q S (e) | q SH (e) | q S(SH) (e) | q H(SH) (e) | q H (e) |
|---|---|---|---|---|---|
| a q S charge of the adsorbed S atoms. b q SH charge of the SH groups. c q S(SH) charge of the S atoms in the SH groups. d q H(SH) charge of the H atoms in the SH groups. e q H charge of the H atoms in the NiH especies. | |||||
(10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_2S_4SH 0)-Ni16S8_2S_4SH |
−0.48 | −0.26 | −0.30 | +0.04 | — |
(10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_6SH_2NiH 0)-Ni16S8_6SH_2NiH |
— | −0.26 | −0.28 | +0.02 | −0.17 |
| −0.26 | −0.31 | +0.05 | |||
4. Conclusions
In this work, we carried out a comprehensive theoretical analysis of the structural and electronic properties of the active nickel and hydrogen species on the phosphosulfide phase formed on the (0001) and (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0) surfaces under HDS reaction conditions. The fully sulfided (0001) surface of Ni2P, composed of Ni(1)-type atoms, closely resembles the (111) surface of the low-activity sulfide Ni3S2, suggesting that surfaces with spatially isolated Ni3 ensembles that are not covered by sulfur are unlikely to be thermodynamically stable. Additionally, hydrogenated (0001) surfaces are not stable because, upon SH group formation, the sulfur atom binds to two of the three Ni(1) atoms within the Ni3 site, leaving the third Ni(1) atom coordinatively unsaturated.
0) surfaces under HDS reaction conditions. The fully sulfided (0001) surface of Ni2P, composed of Ni(1)-type atoms, closely resembles the (111) surface of the low-activity sulfide Ni3S2, suggesting that surfaces with spatially isolated Ni3 ensembles that are not covered by sulfur are unlikely to be thermodynamically stable. Additionally, hydrogenated (0001) surfaces are not stable because, upon SH group formation, the sulfur atom binds to two of the three Ni(1) atoms within the Ni3 site, leaving the third Ni(1) atom coordinatively unsaturated.
On the (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0) surface, the SH groups were identified as the stable form of hydrogen adsorption. The (10
0) surface, the SH groups were identified as the stable form of hydrogen adsorption. The (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0)-Ni16S8_2S_4SH surface, which has coordinatively unsaturated Ni(2) atoms surrounded by SH groups, was the most stable under HDS conditions. Hydrogen stabilization as a hydride, with H atoms adsorbed in a bridging position between the Ni(1) and Ni(2) atoms, only occurred under strong reducing conditions. Our results indicated that the formation of the proton-hydride pair may not occur under reaction conditions. Instead, only the proton species within SH groups are likely to play an active role in facilitating hydrogenation reactions on the nickel phosphosulfide overlayer formed on the Ni2P catalyst. It should be emphasized that the surface structures identified in this work correspond to thermodynamically stable surfaces under equilibrium assumptions, rather than a unique description of the dynamic catalytic surface under reaction conditions.
0)-Ni16S8_2S_4SH surface, which has coordinatively unsaturated Ni(2) atoms surrounded by SH groups, was the most stable under HDS conditions. Hydrogen stabilization as a hydride, with H atoms adsorbed in a bridging position between the Ni(1) and Ni(2) atoms, only occurred under strong reducing conditions. Our results indicated that the formation of the proton-hydride pair may not occur under reaction conditions. Instead, only the proton species within SH groups are likely to play an active role in facilitating hydrogenation reactions on the nickel phosphosulfide overlayer formed on the Ni2P catalyst. It should be emphasized that the surface structures identified in this work correspond to thermodynamically stable surfaces under equilibrium assumptions, rather than a unique description of the dynamic catalytic surface under reaction conditions.
Overall, these findings offer insight into processes occurring at the catalyst surface that may influence HYD selectivity at Ni(2) sites. Organosulfur species adsorbed at these sites may interact with protons from nearby SH groups, potentially forming hydrogenated intermediates and facilitating subsequent C–S bond hydrogenolysis.
Conflicts of interest
There are no conflicts to declare.Data availability
The supporting data has been provided as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6cp00205f.Acknowledgements
The DFT calculations were performed using computational resources from Laboratorio de Química Física y Catálisis Computacional (FONACIT Grant 20240PGP66) and Laboratorio de Fisicoquímica Teórica de Materiales (FONACIT Grant 2023PGP320).References
- K. K. Bando, T. Wada, T. Miyamoto, K. Miyazaki, S. Takakusagi, Y. Koike, Y. Inada, M. Nomura, A. Yamaguchi, T. Gott, S. Ted Oyama and K. Asakura, Combined in situ QXAFS and FTIR analysis of a Ni phosphide catalyst under hydrodesulfurization conditions, J. Catal., 2012, 286, 165–171 CrossRef CAS
.
- L. Yang, X. Li, A. Wang, R. Prins, Y. Wang, Y. Chen and X. Duan, Hydrodesulfurization of 4,6-dimethyldibenzothiophene and its hydrogenated intermediates over bulk Ni2P, J. Catal., 2014, 317, 144–152 CrossRef CAS
.
- Y.-K. Lee and S. T. Oyama, Sulfur resistant nature of Ni2P catalyst in deep hydrodesulfurization, Appl. Catal., A, 2017, 548, 103–113 CrossRef CAS
.
- P. J. Topalian, D. R. Liyanage, S. J. Danforth, A. I. d’Aquino, S. L. Brock and M. E. Bussell, Effect of Particle Size on the Deep HDS Properties of Ni2P Catalysts, J. Phys. Chem. C, 2019, 123, 25701–25711 CrossRef CAS
.
- C. E. Miles, T. R. Carlson, B. J. Morgan, P. J. Topalian, J. R. Schare and M. E. Bussell, Hydrodesulfurization Properties of Nickel Phosphide on Boron-treated Alumina Supports, ChemCatChem, 2020, 12, 4939–4950 CrossRef CAS
.
- X. Zhou, X. Li, R. Prins, A. Wang, L. Wang, S. Liu and Q. Sheng, Desulfurization of 2-phenylcyclohexanethiol over transition-metal phosphides, J. Catal., 2020, 383, 331–342 CrossRef CAS
.
- R. H. Bowker, G. H. Layan Savithra, B. A. Carrillo, K. G. Hubach, T. McDonald, S. L. Brock and M. E. Bussell, Effect of particle size on the sulfur resistance of nickel phosphide hydrodesulfurization catalysts, J. Catal., 2023, 425, 70–79 CrossRef CAS
.
- S. Oyama, Effect of Phosphorus Content in Nickel Phosphide Catalysts Studied by XAFS and Other Techniques, J. Catal., 2002, 210, 207–217 Search PubMed
.
- S. T. Oyama, X. Wang, Y.-K. Lee and W.-J. Chun, Active phase of Ni2P/SiO2 in hydroprocessing reactions, J. Catal., 2004, 221, 263–273 CrossRef CAS
.
- Y. Shu, Y. Lee and S. Oyama, Structure-sensitivity of hydrodesulfurization of 4,6-dimethyldibenzothiophene over silica-supported nickel phosphide catalysts, J. Catal., 2005, 236, 112–121 CrossRef CAS
.
- T. Kawai, K. Bando, Y. Lee, S. Oyama, W. Chun and K. Asakura, EXAFS measurements of a working catalyst in the liquid phase: an in situ study of a Ni2P hydrodesulfurization catalyst, J. Catal., 2006, 241, 20–24 CrossRef CAS
.
- S. T. Oyama, T. Gott, K. Asakura, S. Takakusagi, K. Miyazaki, Y. Koike and K. K. Bando, In situ FTIR and XANES studies of thiophene hydrodesulfurization on Ni2P/MCM-41, J. Catal., 2009, 268, 209–222 CrossRef CAS
.
- X. Duan, Y. Teng, A. Wang, V. Kogan, X. Li and Y. Wang, Role of sulfur in hydrotreating catalysis over nickel phosphide, J. Catal., 2009, 261, 232–240 CrossRef CAS
.
- X. Li, Z. Sun, A. Wang, Y. Wang, X. Duan and Y. Chen, XAS study of Ni2P/MCM-41 passivated by O2/He and H2S/H2, Catal. Commun., 2014, 43, 21–24 CrossRef CAS
.
- S. Tian, X. Li, A. Wang, R. Prins, Y. Chen and Y. Hu, Facile Preparation of Ni2P with a Sulfur-Containing Surface Layer by Low-Temperature Reduction of Ni2P2S6, Angew. Chem., Int. Ed., 2016, 55, 4030–4034 CrossRef CAS PubMed
.
- M. He, T. Li, X. Li, A. Wang, Q. Sheng, S. Shang and Z. Yu, Preparation of Ni2P with a Surface Nickel Phosphosulfide Layer by Reduction of Mixtures of Na4P2S6 and NiCl2, ChemCatChem, 2024, e202400823 CrossRef CAS
.
- S. Rangarajan and M. Mavrikakis, DFT Insights into the Competitive Adsorption of Sulfur- and Nitrogen-Containing Compounds and Hydrocarbons on Co-Promoted Molybdenum Sulfide Catalysts, ACS Catal., 2016, 6, 2904–2917 CrossRef CAS
.
- H. Cai, R. Schimmenti, H. Nie, M. Mavrikakis and Y.-H. C. Chin, Mechanistic Role of the Proton–Hydride Pair in Heteroarene Catalytic Hydrogenation, ACS Catal., 2019, 9, 9418–9437 CrossRef CAS
.
- N. Y. Topsoe and H. Topsoe, FTIR Studies of Mo/Al2O3-Based Catalysts, J. Catal., 1993, 139, 641–651 Search PubMed
.
- G. Berhault, M. Lacroix, M. Breysse, F. Maugé, J.-C. Lavalley, H. Nie and L. Qu, Characterization of Acidic Sites of Silica-Supported Transition Metal Sulfides by Pyridine and 2,6 Dimethylpyridine Adsorption: Relation to Activity in CH3SH Condensation, J. Catal., 1998, 178, 555–565 CrossRef CAS
.
- S. Oyama and Y. Lee, The active site of nickel phosphide catalysts for the hydrodesulfurization of 4,6-DMDBT, J. Catal., 2008, 258, 393–400 CrossRef CAS
.
- A. Nelson, M. Sun and A. Junaid, On the structure and composition of the phosphosulfide overlayer on Ni2P at hydrotreating conditions, J. Catal., 2006, 241, 180–188 CrossRef CAS
.
- L. L. D. Thi, P. N. K. Tuyen and T. Y. Vu, Flower-like S-doped-Ni2P mesoporous nanosheet-derived self-standing electrocatalytic electrode for boosting hydrogen evolution, Nanotechnology, 2020, 31, 465401 CrossRef CAS PubMed
.
- X. Wang, Q. Hu, G. Li, S. Wei, H. Yang and C. He, Regulation of the adsorption sites of Ni2P by Ru and S co-doping for ultra-efficient alkaline hydrogen evolution, J. Mater. Chem. A, 2021, 9, 15648–15653 RSC
.
- Y. Wu, X. Chen, L. Su, Q. Wang and S. Ren, A sulfur-doped Ni2P electrocatalyst for the hydrogen evolution reaction, New J. Chem., 2022, 46, 7675–7681 RSC
.
- M. Šarić, P. G. Moses and J. Rossmeisl, Relation between Hydrogen Evolution and Hydrodesulfurization Catalysis, ChemCatChem, 2016, 8, 3334–3337 CrossRef
.
- A. J. McCue and J. A. Anderson, Sulfur as a catalyst promoter or selectivity modifier in heterogeneous catalysis, Catal. Sci. Technol., 2014, 4, 272–294 RSC
.
- G. Kresse and J. Hafner,
Ab initio molecular dynamics for liquid metals, Phys. Rev. B: Condens. Matter Mater. Phys., 1993, 47, 558–561 CrossRef CAS PubMed
.
- G. Kresse and J. Furthmüller, Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set, Phys. Rev. B: Condens. Matter Mater. Phys., 1996, 54, 11169–11186 CrossRef CAS PubMed
.
- J. P. Perdew, K. Burke and M. Ernzerhof, Generalized Gradient Approximation Made Simple, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS PubMed
.
- G. Kresse and D. Joubert, From ultrasoft pseudopotentials to the projector augmented-wave method, Phys. Rev. B: Condens. Matter Mater. Phys., 1999, 59, 1758–1775 CrossRef CAS
.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu, J. Chem. Phys., 2010, 132, 154104 CrossRef PubMed
.
- W. Hujo and S. Grimme, Comparison of the performance of dispersion-corrected density functional theory for weak hydrogen bonds, Phys. Chem. Chem. Phys., 2011, 13, 13942 Search PubMed
.
- J. P. P. Ramalho, J. R. B. Gomes and F. Illas, Accounting for van der Waals interactions between adsorbates and surfaces in density functional theory based calculations: selected examples, RSC Adv., 2013, 3, 13085 RSC
.
- S. Banerjee, A. Kakekhani, R. B. Wexler and A. M. Rappe, Relationship between the Surface Reconstruction of Nickel Phosphides and Their Activity toward the Hydrogen Evolution Reaction, ACS Catal., 2023, 13, 4611–4621 CrossRef CAS
.
- S. Rundqvist, M. Yhland, R. Dahlbom, J. Sjövall, O. Theander and H. Flood, X-Ray Investigations of Mn3P, Mn2P, and Ni2P, Acta Chem. Scand., 1962, 16, 992–998 CrossRef CAS
.
- M. E. Fleet, The crystal structure of heazlewoodite, and metallic bonds in sulfide minerals, Am. Mineral., 1977, 62, 341–345 CAS
.
- R. F. W. Bader, A quantum theory of molecular structure and its applications, Chem. Rev., 1991, 91, 893–928 CrossRef CAS
.
- K. Momma and F. Izumi,
VESTA3 for three-dimensional visualization of crystal, volumetric and morphology data, J. Appl. Crystallogr., 2011, 44, 1272–1276 CrossRef CAS
.
- W. Papawassiliou, J. P. Carvalho, N. Panopoulos, Y. Al Wahedi, V. K. S. Wadi, X. Lu, K. Polychronopoulou, J. B. Lee, S. Lee, C. Y. Kim, H. J. Kim, M. Katsiotis, V. Tzitzios, M. Karagianni, M. Fardis, G. Papavassiliou and A. J. Pell, Crystal and electronic facet analysis of ultrafine Ni2P particles by solid-state NMR nanocrystallography, Nat. Commun., 2021, 12, 4334 CrossRef CAS PubMed
.
- J. He, Á. Morales-García, O. Bludský and P. Nachtigall, The surface stability and equilibrium crystal morphology of Ni2P nanoparticles and nanowires from an ab initio atomistic thermodynamic approach, CrystEngComm, 2016, 18, 3808–3818 RSC
.
- A. B. Vidal, J. L. Peña-Mena, O. Hurtado-Aular, R. Añez and A. Sierraalta, Unraveling the Structure and Surface Chemistry of the Phosphosulfide Phase Formed on Ni2P under Hydrodesulfurization Reaction Conditions: A DFT Study, J. Phys. Chem. C, 2022, 126, 14187–14200 CrossRef CAS
.
- A. Aguinaga, M. Montes, P. Malet, M. J. Capitán, I. Carrizosa and J. A. Odriozola, Extended X-ray absorption fine structure study of sulphur poisoned Ni/SiO2 catalysts, Appl. Catal., A, 1994, 110, 197–205 CrossRef CAS
.
-
J. R. Rostrup-Nielsen, J. Sehested and J. K. Nørskov, Advances in Catalysis, Elsevier, 2002, vol. 47, pp. 65–139 Search PubMed
.
- X. Chen, J. Jiang, F. Yan, K. Li, S. Tian, Y. Gao and H. Zhou, Dry Reforming of Model Biogas on a Ni/SiO2 Catalyst: Overall Performance and Mechanisms of Sulfur Poisoning and Regeneration, ACS Sustainable Chem. Eng., 2017, 5, 10248–10257 CrossRef CAS
.
- R. Prins and M. E. Bussell, Metal Phosphides: Preparation, Characterization and Catalytic Reactivity, Catal. Lett., 2012, 142, 1413–1436 CrossRef CAS
.
- T. Pecoraro and R. Chianelli, Hydrodesulfurization catalysis by transition metal sulfides, J. Catal., 1981, 67, 430–445 CrossRef CAS
.
- J. Quartararo, S. Mignard and S. Kasztelan, Hydrodesulfurization and hydrogenation activities of alumina-supported transition metal sulfides, J. Catal., 2000, 192, 307–315 CrossRef CAS
.
- G. Kishan, L. Coulier, V. H. J. de Beer, J. A. R. van Veen and J. W. Niemantsverdriet, Sulfidation and Thiophene Hydrodesulfurization Activity of Nickel Tungsten Sulfide Model Catalysts, Prepared without and with Chelating Agents, J. Catal., 2000, 196, 180–189 Search PubMed
.
- Y. Lee and S. Oyama, Bifunctional nature of a SiO2-supported Ni2P catalyst for hydrotreating: EXAFS and FTIR studies, J. Catal., 2006, 239, 376–389 CrossRef CAS
.
- R. R. Chianelli, T. A. Pecoraro, T. R. Halbert, W. H. Pan and E. I. Stiefel, Transition metal sulfide catalysis: relation of the synergic systems to the periodic trends in hydrodesulfurization, J. Catal., 1984, 86, 226–230 CrossRef CAS
.
- Y. Aray, D. Vega, J. Rodriguez, A. B. Vidal, M. Elena Grillo and S. Coll, First-Principles Study of Low Miller Index Ni3S2 Surfaces in Hydrotreating Conditions, J. Phys. Chem. B, 2009, 113, 3058–3070 CrossRef CAS PubMed
.
- R. Khare, R. Weindl, A. Jentys, K. Reuter, H. Shi and J. A. Lercher, Di- and Tetrameric Molybdenum Sulfide Clusters Activate and Stabilize Dihydrogen as Hydrides, JACS Au, 2022, 2, 613–622 CrossRef CAS PubMed
.
| This journal is © the Owner Societies 2026 |