Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Top-down and bottom-up evaluation of thermochemistry of α,ω-alkanediols
Riko Siewert ab,
Olga V. Dorofeeva
ab,
Olga V. Dorofeeva c,
Artemiy A. Samarov
c,
Artemiy A. Samarov d,
Arina V. Elbakari
d,
Arina V. Elbakari e,
Karsten Müller
e,
Karsten Müller ab,
Peter Wasserscheid
ab,
Peter Wasserscheid f and
Sergey P. Verevkin
f and
Sergey P. Verevkin *ab
*ab
aInstitute of Technical Thermodynamics, University of Rostock, Albert-Einstein-Str.2, 18059 Rostock, Germany. E-mail: sergey.verevkin@uni-rostock.de
bCompetence Center CALOR, Department Life, Light & Matter, University of Rostock, Germany
cDepartment of Chemistry, Lomonosov Moscow State University, 119991 Moscow, Russia
dDepartment of Chemical Thermodynamics and Kinetics, Saint Petersburg State University, Peterhof, 198504 Saint Petersburg, Russia
eEngineering and Technology Department, Samara State Technical University, 443100 Samara, Russia
fInstitute of Chemical Reaction Engineering, Friedrich-Alexander-Universität Erlangen-Nürnberg, Egerlandstr. 3, 91058 Erlangen, Germany
First published on 21st April 2026
Abstract
At vapor-liquid equilibrium, the thermodynamic properties of the liquid and vapor phases are intrinsically linked through the thermodynamics of vaporization. However, the standard enthalpies of formation and vaporization of α,ω-alkanediols reported in the literature to date are inconsistent with this fundamental thermodynamic relationship. The aim of this work is to evaluate the existing data and provide recommendations for standard enthalpies of formation and standard enthalpies of vaporization for technical applications. For this purpose, available data are critically analyzed using a “top-down/bottom-up” approach based on gas-phase properties and a bottom-up approach starting from the liquid phase. For those α,ω-alkanediols where discrepancies remain, the relevant thermodynamic properties were re-determined with a combination of combustion calorimetry, vapor pressure measurements and quantum chemical calculations. Based on the comprehensive evaluation, it was shown that the thermodynamic properties of α,ω-alkanediols deviate from the regular trends typically expected for homologous series. Due to the significant contribution of intramolecular hydrogen bond conformers in the gas-phase population, the properties of 1,2-ethanediol, 1,3-propanediol and 1,4-butanediol exhibit distinct particularities compared to long-chained α,ω-alkanediols. Starting from 1,5-pentanediol, the enthalpies of vaporization and the standard enthalpies of formation in the gas phase follow a regular linear trend, attributed to the predominance of conformers without intramolecular hydrogen bonding. The recommended accurate thermodynamic data provide the basis for the development of technical processes utilizing diols from renewable sources in future work.
1. Introduction
α,ω-Alkanediols (such as 1,2-ethanediol, 1,3-propanediol, 1,4-butanediol, etc.) are a fascinating class of organic compounds because their OH groups exhibit interesting physicochemical and thermochemical properties in the liquid and gas phases due to the interplay of inter- and intramolecular hydrogen bonds. These properties are of interest for the development of biofuels, polymer precursors and environmentally friendly solvents.Renewable diols production can reduce dependence on fossil fuels, lower carbon emissions, and promote greener manufacturing processes. Methods like microbial fermentation and catalytic conversion of plant-derived sugars enable the sustainable production of diols like ethylene glycol, propylene glycol, and 1,4-butanediol.1 These bio-based diols help drive the transition toward more environmentally friendly materials and circular economy principles. Long-chain diols (C6–C10) are valuable intermediates in the production of high-performance polyesters, polyurethanes, lubricants, and surfactants. Their longer carbon chains provide enhanced hydrophobicity, flexibility, and durability in materials, making them essential for applications in coatings, adhesives, and biomedical materials. Producing these diols from renewable raw materials, such as plant oils, lignocellulosic biomass, and microbial fermentation, offers a sustainable alternative to petroleum-based sources. Biotechnological processes, including engineered microbial fermentation and enzymatic conversion, enable the efficient synthesis of long-chain diols from fatty acids or sugars. This renewable approach reduces environmental impact, supports the bioeconomy, and promotes greener manufacturing in various industries.
A newly emerging and promising area for the utilisation of renewable diols is their use as liquid organic hydrogen carriers (LOHCs). LOHC systems are an attractive option for the long-term storage of hydrogen in unsaturated organic molecules, such as, e.g., benzyltoluenes,2 alkyl carbazoles.3 These molecules have a similar physical state to conventional diesel fractions, can be transported over long distances and, if necessary, catalytically dehydrogenated to produce molecular hydrogen.
The major drawbacks of these conventional LOHC systems are:
(a) the use of petroleum-based LOHC compounds,
(b) the need to operate at high temperatures for hydrogen release,
(c) the use of noble metal-based catalysts.
A recently published novel strategy appears to overcome all three drawbacks.4,5 It involves diols such as 1,2-propanediol1,6 or 1,4-butanediol,7 two bio-based molecules, as new hydrogen-rich LOHC compounds. For example, the reversible catalytic cyclisation of 1,4-butanediol to γ-butyrolactone releases two moles of hydrogen and can be seen as a very attractive biobased LOHC system.7 Knowledge of the thermodynamic properties of diols and lactones is essential for the technical implementation of such reactions in the context of chemical hydrogen storage technologies.
The focus of this work is the evaluation and revision of the following thermochemical properties of α,ω-alkanediols: standard molar enthalpy of vaporization, ΔglHom, standard molar enthalpy of sublimation, ΔgcrHom, standard molar enthalpy of fusion, ΔlcrHom, and standard molar enthalpy of formation, ΔfHom (298.15 K), in the condensed and gaseous state. These properties are related to each other as follows (referenced to T = 298.15 K):
| ΔfHom (liq, 298.15 K) = ΔfHom (g, 298.15 K) − ΔglHom (298.15 K) | (1) |
| ΔfHom (cr, 298.15 K) = ΔfHom (g, 298.15 K) − ΔgcrHom (298.15 K) | (2) |
| ΔgcrHom (298.15 K) = ΔglHom (298.15 K) + ΔlcrHom (298.15 K) | (3) |
The experimental thermochemistry of the short-chained α,ω-alkanediols: 1,2-ethanediol,8 1,3-propanediol,9 and 1,4-butanediol10 was partially investigated in our earlier studies. In the course of these studies, we found that the experimental thermochemistry of α,ω-alkanediols with a chain length of C6 to C10 is rather confusing. In fact, most of the thermochemical data on the long-chain alkanediols have been reported only in a few publications by the Sabbah group11–14 and the Della Gatta group.15,16 A brief summary of the experimental enthalpies of vaporization, ΔglHom, and enthalpies of formation, ΔfHom (liq or cr), of α,ω-alkanediols available in the literature is shown in Table 1.
| Diol | ΔglHom![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b b |
ΔfHom (liq or cr) |
|---|---|---|
| a The experimental uncertainties are expressed as two times the standard deviation of the mean value.b Original values from ref. 15 and 16 were uniformly adjusted to the reference temperature T = 298.15 K in our previous work.21 | ||
| 1,3-Propanediol | — | −458.7 ± 3.0 (liq)17 to −482.0 ± 2.5 (liq)9 |
| 1,6-Hexanediol | 91.6 ± 4.1 | −362.2 ± 4.8 (cr)18 to −583.9 ± 0.7 (cr)19 |
| 1,7-Heptanediol | 97.5 ± 3.2 | −574 ± 19 (liq)12 |
| 1,8-Octanenediol | 108.7 ± 3.7 | −627 ± 10 (cr)12 |
| 1,9-Nonanediol | 113.3 ± 7.0 | −658 ± 14 (cr)12 |
| 1,10-Decanenediol | 111.0 ± 2.0 | −678.9 ± 6.5 (cr)12 to −699.9 ± 2.1 (cr)20 |
| 1,11-Undecanediol | 128.6 ± 4.0 | |
| 1,12-Dodecanediol | 126.1 ± 5.6 | |
| 1,13-Tridecanediol | 128.2 ± 7.7 | |
| 1,14-Tetradecanediol | 135.4 ± 6.0 | |
| 1,15-Pentadecanediol | 132.0 ± 4.1 | |
| 1,16-Hexadecanediol | 138.8 ± 4.0 | |
The most dramatic situation regarding the consistency of experimental enthalpies of formation is evident for 1,6-hexanediol, where the available values range from −362. 2 to −583.9 kJ mol−1 (Table 1, column 3), and 1,10-decanediol, where the scatter of the available results, at 21 kJ mol−1, is also too large to consider the quality of the data acceptable.
It should be mentioned that most of the combustion results of diols, which lead to the enthalpies of formation, were measured by Knauth and Sabbah,11,12 using a microbomb rocking calorimeter and samples of 5 to 10 milligrams. The uncertainties of 10 to 19 kJ mol−1 (see Table 1, column 3) show that achieving reproducibility of measurements with micro-bombs is a challenging task. That said, the experimental formation enthalpies of diols compiled in Table 1 must be checked for internal consistency within their homologous family.
The Della Gatta group measured the enthalpies of vaporization of α,ω-alkanediols using a combination of the Knudsen method and the torsion effusion method.15,16 The experimental enthalpies of vaporization of α,ω-alkanediols measured at Tav were adjusted by the authors to the reference temperature T = 298.15 K (see Table 1, column 2). It is apparent that the results of the Della Gatta group are strongly aggravated by large uncertainties of up to 7.7 kJ mol−1. These unusually large uncertainties make the interpretation of the trends in the measured enthalpies of vaporization problematic.
For example, the comparison of the vaporization enthalpies in Table 1 for the sequence of the following diols:
| 1,8-Octanediol (ΔglHom (298.15 K) = 108.7 ± 3.7 kJ mol−1), |
| 1,9-Nonanediol (ΔglHom (298.15 K) = 113.3 ± 7.0 kJ mol−1), |
| 1,10-Decanediol (ΔglHom (298.15 K) = 111.0 ± 2.0 kJ mol−1) |
Furthermore, there is no reason or specific interaction that makes the enthalpy of vaporization of 1,9-nonanediol higher than that of 1,10-decanediol, and it is highly unusual that the enthalpies of vaporization of 1,11-undecanediol to 1,15-pentadecanediol are barely distinguishable within the uncertainties attributed to these values.
This unacceptable scatter in the available thermochemical data for each alkanediol has prompted this study, the main aim of which is to evaluate and revise the energetics of the diols using additional experiments, empirical correlations and quantum chemical (QC) calculations, which have now reached a level of accuracy where they can predict enthalpies of formation with the so-called “chemical accuracy” of ∼±4 kJ mol−1. This level of accuracy is achieved by two high-level computational methods, G422 and DLPNO-CCSD(T1)/CBS,23 used in this work. The G4 method was tested on primary alcohols24 and two diols26 and showed good results when using atomization reactions. However, difficulties with using this approach may arise in accounting for conformational diversity of diol molecules. In order to evaluate the energetics of the conformers more reliably, the high-level DLPNO-CCSD(T1)/CBS method was additionally used in combination with isodesmic reactions. In these reactions, the error associated with the accurate consideration of the conformational equilibrium can be eliminated if the products are to some extent similar in conformation to the reagent structures. These advances make it possible to closely match theoretical predictions with experimental data, making quantum chemistry a powerful tool in thermochemistry and molecular design.
This modern QC calculation development enables us to validate thermochemical data for diols with a “top-down” strategy, where the quantum chemistry should provide a plausible expectation for the gas phase enthalpy of formation, ΔfHom (g), to evaluate the experiment in the condensed phase (according to eqn (1)–(3)), e.g. in the liquid state, as shown in Fig. 1.
 | ||
| Fig. 1 Visualization of the “top-down” and “bottom-up” strategies for evaluation of thermochemistry of α,ω-alkanediols via vaporization enthalpy according to eqn (1). | ||
The ΔglHom (298.15 K)-values required by eqn (1) for the “drop” from “top” (quantum chemically calculated enthalpies of formation) to the liquid phase formation enthalpies of the short-chain diols have just been ascertained in our work21 and can be used in this study to evaluate the thermochemistry of α,ω-alkanediols.
α,ω-Alkanediols are flexible molecules with three and more internal rotational degrees of freedom that lead to the existence in the gas-phase an equilibrium mixture with of a large number of conformers with significantly different energetics. Determining the most stable conformer required to calculate the ΔfHom (g) values and is a challenging task for these compounds. At present, the most complete information on conformational equilibrium has been obtained for the two simplest α,ω-alkanediols – 1,2-ethanediol and 1,3-propanediol. Both experimental and theoretical studies including statistical thermodynamics population analysis25–27 show that the most stable conformers of these molecules are stabilized by an intramolecular O–H⋯O–H hydrogen bond between the hydroxyl groups. However, a rather ambiguous interpretation of conformational equilibrium is given for 1,4-butanediol. The results of gas-phase electron diffraction28 and microwave spectroscopy29 studies were interpreted as strong evidence for the mixture of two hydrogen-bonded conformers (denoted as HB here), whereas the analysis of vibrational spectra in the vapor phase25,26,30–34 showed the presence of both HB conformers and those with free hydroxyl groups (denoted as F conformers here). Some studies have found that experimental spectra show good agreement with theoretical spectra with a predominant contribution from HB conformers25,32,33 but others have noted the need to include higher energy F conformers to reproduce the experimental data.26,30,31,34 Previously reported theoretical calculations,26,32–35 also do not provide complete clarity on the stability of the various 1,4-butanediol conformers. And this is due not only to insufficiently high level of the calculation methods used and the problem of accounting for the large anharmonicity induced by low-frequency torsional motions in 1,4-butanediol, which is neglected in the calculation of the zero-point vibrational energy (ZPE), enthalpic correction (H298.15 − H0) and entropy, but also to the approach used to determine the most stable conformers.
The differences in electronic energy of conformers (ΔEe), ground-state energy (ΔE0, where E0 = Ee + ZPE), enthalpy (ΔH, where H = Ee + ZPE + (H298.15 − H0)) and Gibbs free energy (ΔG, where G = H − TS) are used in QC calculations to estimate the conformer population and determine the most stable conformer. In many cases, all these quantities enable correct identification of the most stable conformer. The conformer population is quite often defined based on the ΔH values. This can be justified if the entropies of conformers are approximately the same and then ΔH ≈ ΔG. To determine the values of ΔG, it is necessary to calculate the entropy of all conformers, but sometimes this is replaced by accounting for the structural degeneracy of each conformer, which depends on its symmetry.26,33,34 This assumption can be applied when the conformers are spatially similar, however, this is not the case for 1,4-butanediol: the HB conformers have a boat-like arrangement with the internal hydrogen bond closing the ring, whereas the F conformers have the extended open-chain structures. These structural features lead to significantly larger values of the product of moments of inertia of F conformers compared to the HB conformers and, as a rule, smaller values of low-lying vibrational frequencies. Therefore, for an accurate calculation of the Gibbs free energies, it is necessary to take into account all molecular parameters of conformers when calculating entropy using the statistical thermodynamics method. However, such calculations have not been performed for 1,4-butanediol in the past,26,32–35 which may be the reason for the discrepancies in the interpretation of the experimental results.
Benchmark relative energies, ΔEe, for 65 conformers of 1,4-butanediol calculated at the CCSD(T)-F12b/cc-pVTZ-F12 level35 have shown that the group of HB conformers is more stable than the F conformers. This result is consistent with a 89% population of HB conformers determined according to the Boltzmann distribution based on the ΔE0 energies of 11 low-energy conformers of 1,4-butanediol calculated at the B3LYP/6-311++G(d,p) level and taking into account the structural degeneracy of each conformer.32 However, a similar calculation for 65 conformers using the ΔE0 values calculated by the MP2/6-311++G(d,p) method33 resulted in a significantly lower value of 46% for the HB conformer population. A similar result, with relative population of 47% for HB conformers, was obtained from MP2/6-311G(d,p) calculations for the 10 lowest-energy conformers.27 Among other long chain α,ω-alkanediols, the relative population of conformers was determined for 1,5-pentanediol and 1,6-hexanediol using MP2 and DFT electronic energies and corrections necessary to estimate the Gibbs free energies.36,37 For these two diols, the extended F conformers were found to be the most abundant.
In the present work, before proceeding to the conformational analysis of long chain diols, it was decided to perform a new calculation of the relative population of 1,4-butanediol conformers based on a more accurate estimation of the Gibbs free energy. This enables us to develop an approach to find the most stable conformers of the remaining diols for which a complete conformational analysis is not possible with our computing capacities. The entropy, ZPE and enthalpic corrections were estimated within the rigid rotor–harmonic oscillator approximation (RRHO), since accounting for the internal rotation in such large flexible molecules still presents great difficulty. Another approximation used in our work is the calculation of the enthalpy of formation only for the most stable conformer. As mentioned above, this is common practice when using isodesmic reactions, but the ΔfHom (g) values calculated from the atomization reaction are often corrected for the conformer population. We abandoned this correction because it was previously shown that it can significantly overestimate the calculated values.27,38 In this context, the highly flexible α,ω-alkanediols selected for experimental and computational studies in this work could either support the validity of the approximations used here, or our study could reveal and quantify the necessary corrections to achieve agreement between experiment and calculations. This means that in the case of α,ω-alkanediols, the alternative “bottom-up” strategy (see Fig. 1) should be applied, where the available experimental enthalpies of formation of the condensed phase need to be critically evaluated using new combustion experiments and structure–property correlations, and the consistent experimental ΔfHom (cr or liq) values in conjunction with the new experimental vaporization (or sublimation) enthalpies of alkanediols can help to address the deficiencies of the QC calculations with the flexible molecules, which exhibit considerable conformational diversity. The combination of “top-down” and “bottom-up” strategies therefore makes it possible to integrate theoretical expectations (“top-down”) with measurements and empirical observations (“bottom-up”) in order to evaluate the available experimental data and recommend reliable thermochemical properties for α,ω-alkanediols.
2. Experimental and theoretical methods
2.1 Thermochemical methods
Commercially available samples of α,ω-alkanediols were used. Their origin and purity are listed in Table S1 in the SI. A self-built high-precision calorimeter with a static bomb39 was used to measure the standard molar energy of combustion of 1,8-octanediol. The absolute vapor pressures of 1,6-hexanediol, 1,7-heptanediol, 1,8-octanediol, 1,9-nonanediol and 1,10-decanediol were measured using the transpiration method.40 The standard molar enthalpies of vaporization and the standard molar enthalpies of sublimation as well as the standard molar entropies of vaporization and the standard molar entropies of sublimation were derived from the temperature dependence of the absolute vapor pressures.2.2 Quantum chemical calculations
Conformational analysis was performed with MMFF94 force field using GMMX conformational searching methodology implemented in the GaussView 6 computational molecular chemistry application.41 The geometry of 10 (1,2-ethanediol) to 1000 (1,10-decanediol) generated structures was further optimized at the B3LYP/6-31G(d,p) level. From these conformers, the group of low-energy conformers was selected for each diol to calculate the Gibbs free energy using the G422 and DLPNO-CCSD(T1)23 methods. The Gaussian 16 software42 and ORCA 5.0.3 program release43 were used in these QC calculations. The results of the calculations are shown in Tables S2–S5.The single-point DLPNO-CCSD(T1)/CBS energies (Ee) were determined using the extrapolation to the basis set limit procedure implemented in the ORCA and the geometries optimized at the B3LYP-D3(BJ)/def2-TZVPP level. The entropy, ZPE and enthalpic corrections were calculated within the RRHO approximation using the rotational constants, vibrational frequencies and symmetry determined from the B3LYP-D3(BJ)/def2-TZVPP calculations. The vibrational frequencies were scaled by a factor of 0.9657 when calculating entropy and by a factor of 0.9883 when calculating the ZPE and enthalpic corrections.44 The Gibbs free energies of the conformers were calculated using the equation G = H − TS, where H = Ee + ZPE + (HT − H0), T = 298.15 K and S is the entropy calculated applying the formulas of statistical thermodynamics. The conformer with the lowest G value was chosen as the most stable, and the enthalpy of formation was calculated for it. The DLPNO-CCSD(T1)/CBS and G4 calculations predicted the same most stable conformers for all diols except 1,4-butanediol, therefore only the results obtained using the DLPNO-CCSD(T1)/CBS method are discussed in more detail below.
The G4 enthalpies of formation were calculated from atomization reaction, whereas 8 isodesmic-type reactions (see Table S5) were used to determine the ΔfHom (g) values by DLPNO-CCSD(T1)/CBS method. The accurate ΔfHom (g) values for small reference species used in these reactions were taken from the active thermochemical tables (ATcT).45 For three reference molecules (1,2-ethanediol, 1,2- and 1,3-propanediol), the experimental data from this work and ref. 46 were used and their accuracy was confirmed by preliminary QC calculations.
3. Top-down strategy: gas-phase enthalpies of formation from quantum chemical calculations
3.1 Conformational analysis and identification of most stable conformers
Relative enthalpies (ΔH), Gibbs free energies (ΔG) and conformer populations (pi) of C2–C10 diols determined in this work are presented in Table S2, as well as quantities used to calculate them. The Cartesian coordinates of most stable HB and F conformers are given in Table S3.The population of 1,2-ethanediol conformers was determined taking into account all 10 possible conformations of this molecule. The HB conformer was found to be the most stable and its overall prevalence compared to F conformers was estimated at 95%. This result agrees with predominance of HB conformers predicted earlier by G4 method (93% of HB conformers)27 using the same approach for Gibbs free energy calculation as in the present work.
A previous population analysis of 1,3-propanediol was performed using the G4 method for 20 conformers whose energy calculated at the B3LYP/6-31G(d,p) level did not exceed 15 kJ mol−1.27 Like for 1,2-ethanediol, the most stable conformer of 1,3-propanediol is HB, but these conformers contribute almost two times less to the total population (51%) compared to 1,2-ethanediol. In this work, we reduced the number of conformers to 11 to see how this would affect the result. In the simplified model, all four HB conformers were detected, but their contribution to the total population, as expected, increased to 70% due to the exclusion of more than half of the higher-energy F conformers. Thus, most likely, the simplified conformer selection of diols based on their relative B3LYP/6-31G(d,p) energies will not miss the most stable conformer.
Conformer population analysis of 1,4-butanediol (Table S2) was performed for all 65 possible conformers detemined using GMMX conformational searching. Cartesian coordinates of these conformers are given in Table S4. The identified conformers agree with those obtained by manual conformational scanning35 with the exception of three conformers (pairs of conformers (30,31), (45,46) and (49,50) in ref. 35 have the same structure). As can be seen in Table S2, the relative population of HB conformers (29%) is significantly smaller than that obtained previously using a more approximate estimate of the entropy contribution to the Gibbs free energy (about 47%26,33).
It should be noted that in the group of F conformers, a subgroup of 26 conformers is distinguished that have a linear carbon backbone (denoted as F-lin conformers here). These straight chain diols are characterized by a continuous linear arrangement of carbon atoms with C–C–C–C angles from 172.5 to 180.0° and an average value of 178.3°. F-lin conformers contribute 50% to the total population of 1,4-butanediol conformers. As can be seen in Fig. 2, the difference between the calculated ΔG values for the lowest-lying HB and F-lin conformers is within the limits of their uncertainties, so it can be assumed that taking into account the anharmonicity of the vibrations may result in the F-lin conformer becoming the most stable (see Section 3.2).
The most stable conformers from all three groups were also revealed when considering only the 20 lowest-energy B3LYP/6-31G(d,p) conformers of 1,4-butanediol, but the population of HB conformers became predominant (50% HB and 43% F-lin). Similar simplified models were used in the present work for the remaining C5–C10 diols. Since F-lin was found to be the most stable conformer in all of these diols, F-lin and F conformers were added to the initially selected 20 conformers. Analysis of the results for C5–C7 diols showed that F-lin conformers dominate among the low-lying conformers of these molecules (Table S2) and therefore, when determining the most stable conformers of C8–C10 diols, the F conformers were not taken into account.
3.2 Approximate treatment of vibrational anharmonicity
Based on the calculated ΔG values (Table 2), it can be concluded that the most stable conformers of C2–C4 diols should be the HB conformers, whereas, starting from 1,5-pentanediol, the F-lin conformations become the most stable. Since for most molecules the calculated ΔG values are within the accuracy of their determination, it is interesting to evaluate how a more accurate calculation accounting for the anharmonicity of vibrations can affect their values. The simplest approaches were used here for such approximate evaluation.| Molecule | Method | ΔfHom | ΔfHom | ΔHc | ΔGc |
|---|---|---|---|---|---|
| HB conformer | F-lin (F) conformerb | ||||
| a Recommended values are given in bold.b F conformers for 1,2-ethanediol and 1,3-propanediol and F-lin for all others.c The ΔH and ΔG values are the difference in these functions between F-lin and HB conformers; values given in parentheses are those approximately corrected for anharmonicity of vibrational frequencies (see text for details). | |||||
| 1,2-Ethanediol | DLPNO | −390.0 ± 2.3 | −379.3 ± 2.3 | 10.7 (10.8) | 8.8 (8.9) |
| G4 | −389.1 | −378.4 | 10.7 | 8.6 | |
| 1,3-Propanediol | DLPNO | −412.4 ± 2.3 | −407.5 ± 2.3 | 4.9 (2.0) | 3.4 (1.0) |
| G4 | −411.5 | −407.7 | 3.8 | 2.5 | |
| 1,4-Butanediol | DLPNO | −433.4 ± 2.6 | −426.4 ± 2.6 | 7.0 (4.2) | 2.2 (0.1) |
| G4 | −433.1 | −428.2 | 4.9 | 1.7 | |
| 1,5-Pentanediol | DLPNO | −449.4 ± 2.4 | −446.6 ± 2.4 | 2.8 (1.6) | −2.9 (−3.4) |
| G4 | −449.9 | −447.0 | 2.9 | −2.6 | |
| 1,6-Hexanediol | DLPNO | −469.7 ± 2.1 | −466.7 ± 2.1 | 3.0 (1.9) | −6.2 (−5.4) |
| G4 | −471.4 | −467.9 | 3.5 | −5.4 | |
| 1,7-Heptanediol | DLPNO | −494.1 ± 2.4 | −487.9 ± 2.4 | 6.2 (3.7) | −2.5 (−2.9) |
| G4 | −495.8 | −489.4 | 6.4 | −2.6 | |
| 1,8-Octanediol | DLPNO | −512.9 ± 2.4 | −508.5 ± 2.4 | 4.5 (2.0) | −4.2 (−5.3) |
| G4 | −516.3 | −510.7 | 5.6 | −3.4 | |
| 1,9-Nonanediol | DLPNO | −536.7 ± 2.3 | −528.7 ± 2.3 | 8.0 (5.8) | −2.1 (−3.5) |
| G4 | −541.1 | −532.0 | 9.1 | −2.6 | |
| 1,10-Decanediol | DLPNO | −555.2 ± 2.4 | −549.5 ± 2.4 | 5.7 (3.2) | −5.2 (−4.3) |
| G4 | −559.0 | −553.4 | 5.7 | −7.5 | |
A free rotor approximation proposed by Radom and co-workers47,48 was used to calculate the ZPEs and enthalpic corrections. This model suggests that for molecules with low-frequency torsional modes, a better result than the RRHO model is achieved by treating frequencies up to 260 cm−1 as tending to zero and replacing their contribution to the enthalpy correction by 1/2RT. Errors in entropies, associated with treating low frequencies as harmonic motions, were reduced using Truhlar's approximation49,50 in which all harmonic frequency values below 100 cm−1 were replaced by 100 cm−1.
The ΔG values approximately accounting for vibrational anharmonicity are given in Table 2 in parentheses. It is worth paying attention to 1,4-butanediol, for which the value of ΔG = G(F-lin) − G(HB) decreased to 0.1 kJ mol−1, which does not exclude that the F-lin conformer may be more stable when taking into account anharmonicity using more accurate methods. A comparison of the calculated enthalpies of formation with the experimental values can also help to clarify which configuration, HB or F-lin, is the most stable conformer of 1,4-butanediol.
3.3 Calculation of gas phase enthalpies of formation
The ΔfHom (g) values for most stable HB and F-lin conformers of α,ω-alkanediols calculated using DLPNO-CCSD(T1) and G4 methods are summarized in Table 2. The details of the DLPNO-CCSD(T1) calculation using isodesmic-type reactions are given in Table S5. As can be seen, the results obtained using the two methods are in good agreement: the deviations are not more than 2 kJ mol−1 for C2–C7 diols and 4.5 kJ mol−1 for the three largest molecules.The values of the enthalpies of formation recommended in Table 2 (marked in bold) were obtained for conformers with lower values of Gibbs free energy: for C2 and C3 diols these are HB conformers, and for C5–C10 these are F-lin conformers. The only exception is 1,4-butanediol, for which the enthalpy of formation is recommended for the F-lin conformer, despite its G value is higher than that of the HB conformer (see Section 7.6).
The QC gas-phase enthalpies of formation of α,ω-alkanediols shown in Table 2 can be considered as the starting point for the “top-down” evaluation strategy. However, as can be seen in Fig. 1, the enthalpies of vaporization are equally required to descend to the liquid phase thermochemistry or to ascend from the liquid phase to the gas phase. The next section deals with this “intermediate” thermochemical property.
4. Enthalpy of vaporization as an intermediate between top-down and bottom-up strategies
4.1 Absolute vapor pressures of α,ω-alkanediols measured using the transpiration method
The vapor enthalpies, which are used in the general eqn (1) to derive the enthalpies of formation of the liquid phase, should be of impeccable quality in order to obtain a reliable validation of the thermochemistry of the diols using the “top-down” strategy. Surprisingly, in contrast to the intensive investigations of the short-chain C2 to C4 diols, only very few vapor pressure measurements of the long-chain C5 to C10 alkanediols were found in the literature. The unusual trends in the enthalpies of vaporization shown in Table 1 also prompted the investigation of 1,5-pentanediol, 1,6-hexanediol, 1,7-heptanediol, 1,8-octanediol, 1,9-nonanediol and 1,10-decanediol using the transpiration method.40 The essential advantage of this method is that the vapor pressure measurements are carried out at temperatures that are as close as possible to the reference temperature T = 298.15 K, so that the enthalpies of vaporization derived from such measurements are relatively less affected by the adjustment to this temperature compared to many other conventional methods.The experimental absolute vapor pressure–temperature dependencies measured in this work using the transpiration method for 1,5-pentanediol, 1,6-hexanediol, 1,7-heptanediol, 1,8-octanediol, 1,9-nonanediol and 1,10-decanediol are summarised in Table S6. They were approximated using a three-parametric equation, as presented in detail in SI, and used to calculate the enthalpies of vaporization ΔglHom (298.15 K), or enthalpies of sublimation ΔgcrHom (298.15 K), according to Kirchhoff's rule, using the heat capacity differences developed in our previous work21 (see Table S7) The final results at the reference temperature T = 298.15 K, ΔglHom (298.15 K), are summarised in Table 3 and compared with the enthalpies of vaporization measured by other methods.
| Diol | Methoda | T-Range/K | ΔglHom/Tav | ΔglHom![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b/298.15 K b/298.15 K |
Ref. |
|---|---|---|---|---|---|
| a Methods: E = ebulliometry; C = calorimetry; n/a = method is not available; T = transpiration method; BP = estimated from boiling points at different pressures (see Table S8); TE = torsion-effusion method; CGC = correlation gas-chromatography.b Vapor pressures treated using eqn (S2) and (S3) with help of heat capacity differences from Table S7 to calculate the enthalpies of vaporization at 298.15 K. Uncertainties of the vaporization enthalpies U(ΔglHom) are the expanded uncertainties (0.95 level of confidence). They include uncertainties from the fitting equation and uncertainties from temperature adjustment to T = 298.15 K as described elsewhere.55 Uncertainties in the temperature adjustment of the literature data on vaporization enthalpies to the reference temperature T = 298.15 K are estimated to account with 20% to the total adjustment.c Weighted mean value (uncertainties were taken as the weighting factor). Values given in bold are recommended for further thermochemical calculations. Data in parentheses were not involved in the evaluation. | |||||
| 1,2-Ethanediol | 65.7 ± 0.2 | 21 | |||
| 1,3-Propanediol | 71.5 ± 0.3 | 21 | |||
| 1,4-Butanediol | 79.0 ± 0.7 | 21 | |||
| 1,5-Pentanediol | E | 391.9–480.1 | 73.2 ± 0.9 | 85.9 ± 2.7 | 51 |
| E | 446.0–514.5 | 64.9 ± 0.4 | 82.1 ± 3.5 | 52 | |
| n/a | 391–479 | 73.4 ± 1.0 | 85.9 ± 2.8 | 53 | |
| C | 298.15 | 86.8 ± 1.0 | 14 | ||
| BP | 352–516 | 73.5 ± 1.5 | 85.5 ± 2.8 | Table S8 | |
| T | 303.4–351.2 | 84.4 ± 0.4 | 87.1 ± 0.5 | Table S6 | |
| 86.6 ± 0.6c | Average | ||||
| 1,6-Hexanediol | 87.0 ± 4.0 | 90.7 ± 0.4 | 21 | ||
| 1,7-Heptanediol | C | 323 | 93.8 ± 1.0 | 96.8 ± 1.2 | 13 |
| TE | 321–361 | 92.4 ± 3.0 | 97.5 ± 3.2 | 16 | |
| CGC | 298.15 | 95.9 ± 0.6 | 21 and 54 | ||
| BP | 375–535 | 77.2 ± 1.2 | 95.5 ± 3.9 | 21 | |
| T | 336.2–373.2 | 87.7 ± 0.3 | 94.4 ± 0.4 | Table S6 | |
| 94.8 ± 0.4c | Average | ||||
| 1,8-Octanediol | TE | 340–372 | 101.1 ± 3.4 | (108.7 ± 3.7) | 15 |
| CGC | 298.15 | 98.7 ± 0.2 | 21 and 54 | ||
| BP | 374–549 | 80.7 ± 1.8 | 98.9 ± 4.1 | 21 | |
| T | 343.2–379.3 | 91.0 ± 0.8 | 99.2 ± 1.0 | Table S6 | |
| 98.7 ± 0.3c | Average | ||||
| 1,9-Nonanediol | TE | 347–373 | 104.4 ± 6.8 | (113.3 ± 7.0) | 16 |
| C | 323 | 110.0 ± 2.0 | (113.6 ± 2.1) | 13 | |
| CGC | 298.15 | 102.7 ± 0.2 | 21 and 54 | ||
| BP | 392–562 | 78.5 ± 3.8 | 102.5 ± 5.2 | 21 | |
| T | 343.2–379.2 | 93.9 ± 0.8 | 103.0 ± 1.0 | Table S6 | |
| 102.7 ± 0.3 | Averagec | ||||
| 1,10-Decanediol | TE | 351–377 | 112.4 ± 4.6 | (122.6 ± 5.0) | 15 |
| E | 457.0–566.3 | 73.6 ± 1.1 | 106.6 ± 6.7 | 16 | |
| CGC | 298.15 | 106.7 ± 0.2 | 21 and 54 | ||
| BP | 405–578 | 76.8 ± 2.1 | 106.2 ± 6.2 | 21 | |
| T | 346.3–370.5 | 97.3 ± 1.2 | 106.6 ± 1.3 | 21 | |
| 106.7 ± 0.3c | Average | ||||
It is important to emphasise that the vapor pressures and enthalpies of vaporization of alkanediols available in the literature were treated in the same way as our new transpiration results (using the heat capacity differences evaluated in Table S7) and adjusted to T = 298.15 K. Only such uniform data treatment allowed a correct comparison of ΔglHom (298.15 K)-values (see Table 3). As a result, a very consistent set of vaporization values for 1,5-pentanediol, 1,7-heptanediol and 1,10-decanediol was compiled. For each of these diols, the weighted average values were estimated and recommended for the thermochemical calculations (bold values in Table 3). The results for 1,8-octanediol and 1,9-nonanediol summarised in Table 3 are fewer in number, but for each of the two diols at least three entries were averaged and considered reliable. It should be recognised that our new transpiration results were extremely helpful in making the selection of reliable results for the long-chain C8 to C10 alkanediols, where the scatter of available vaporization enthalpies of 10–17 kJ mol−1 made the final selection decision much more difficult. Although our selection of the reliable level for the enthalpies of vaporization of α,ω-alkanediols seems reasonable, the large scatter in the data makes additional validation of this selection necessary. The various methods used to validate the results are described in the following section.
4.2 Validation of the vaporization enthalpies by structure–property correlations
The structure–property correlations form the core of physical-organic chemistry, because new experimental results are integrated into the network of already known and reliable data. If the new results are consistent with the trends already known from the available results, they can be recommended for thermochemical calculations. Otherwise, the unusual structural effects must be explained or the experiments repeated to avoid confusion.The simplest types of such correlations are relationships between a property of interest and a chain length (NC), or between a property of interest and the normal boiling temperature (Tb), or between a property of interest and the chromatographic retention index (Jx).
The latter two types are more specific to the enthalpies of vaporization. However, the use of all three types is most successful for the homologous series where a specific contribution for the CH2 fragment is expected. Therefore, the series of α,ω-alkanediols, which is the focus of this work, is predestined for the use of these correlations to validate the experimental thermochemical properties of interest in this study.
 | ||
| Fig. 3 Structure–property correlations with enthalpies of vaporization, ΔglHom (298.15 K) for the data evaluated in Table 3 for α,ω-alkanediols. Left: Chain length dependence with the NC. The numerical data are given in Table S9. Right: With the normal boiling temperatures, Tb, from the literature.56 The numerical data are given in Table S10. Below: With the Kovats indices (Jx) from the literature.57 The numerical data are given in Table S11. | ||
It is obvious that the expected homologous linear behaviour starts first from the C5 diol and the following equation approximates the dependence of the vaporization enthalpies on the chain length:
| ΔglHom (298.15)/kJ mol−1 = 4.01 × NC + 66.6 with R2 = 0.9999 (for C ≥ 5), | (4) |
The reason why the 1,2-, 1,3- and 1,4-diols do not belong to this line is the intra-molecular hydrogen bonding known from the spectroscopic studies,58 which was also observed in the conformers of these diols (see Table S2). Nevertheless, the definite linear increase of the vaporization enthalpies starting from the C5 diol should be considered as evidence for the internal consistency of the values selected in Table 3.
| ΔglHom (298.15 K)/(kJ mol−1) = 0.3117 × Tb − 72.6 with R2 = 0.9985 (for C ≥ 5), | (5) |
| ΔglHom (298.15 K)/(kJ mol−1) = 46.6 + 0.0397 × Jx with R2 = 0.9985 (for C ≥ 5) | (6) |
The high correlation coefficients of eqn (4)–(6) confirm the internal consistency of the ΔglHom (298.15 K)-data used in the correlations. Tables S9–S11 show that the differences between the experimental values and the calculated values calculated are well below than 0.5 kJ mol−1. Therefore, these correlations were used to extrapolate the ΔglHom (298.15 K)-values for C11 to C16 alkanediols (see Tables S9–S11).
| Diol | ΔglHom (exp)a (Table 3) | ΔglHom vs. NC (Table S9) | ΔglHom vs. Tb (Table S10) | ΔglHom vs. Jx (Table S11) |
|---|---|---|---|---|
| a The experimental uncertainties are expressed as two times the standard deviation of the mean value. | ||||
| 1,5-Pentanediol | 86.6 ± 0.6 | 86.7 | 86.9 | 86.9 |
| 1,6-Hexanediol | 90.7 ± 0.4 | 90.7 | 90.7 | 90.7 |
| 1,7-Heptanediol | 94.8 ± 0.4 | 94.7 | 94.2 | 94.3 |
| 1,8-Octanenediol | 98.7 ± 0.3 | 98.7 | 98.7 | 98.7 |
| 1,9-Nonanediol | 102.7 ± 0.3 | 102.7 | 102.7 | 102.7 |
| 1,10-Decanenediol | 106.7 ± 0.3 | 106.7 | 106.8 | 106.9 |
It can be seen from this table that the experimental and empirical results for α,ω-alkanediols are hardly distinguishable, and this fact provides full confidence in the reliability of these values, which is why the experimental vaporization enthalpies, ΔglHom (298.15 K), evaluated in Table 3 (values in bold) can be safely recommended for thermochemical calculations.
5. Enthalpy of sublimation as an intermediate between top-down and bottom-up strategies
In fact, long-chain α,ω-alkanediols, starting from 1,6-hexanediol, are solid at room temperature (with the exception of 1,7-heptanediol). In this case, reliable data on the sublimation enthalpies, ΔgcrHom (298.15 K), are necessary to reconcile the QC gas-phase enthalpies of formation and the crystal-phase enthalpies of formation according to the top-down strategy (see Fig. 4). | ||
| Fig. 4 Top-down and bottom-up strategies for evaluation of thermochemistry of α,ω-alkanediols via sublimation enthalpies. | ||
The data available in the literature is of very questionable quality, therefore, a critical evaluation using all available methods is necessary, as described in the following sections.
5.1 Experimental sublimation enthalpies from vapor pressure measurements
Surprisingly, no systematic vapor pressure studies on the solid sample for diols with Nc > 6 can be found in the literature. Only the sublimation enthalpies of 1,6-hexanediol, 1,8-octanediol and 1,10-decanediol were measured directly by calorimetry by Knaut and Sabbah.13In this work, for the first time, the vapor pressure–temperature dependences over the solid samples of 1,6-hexanediol, 1,8-octanediol, 1,9-nonanediol and 1,10-decanediol were measured using the transpiration method (see Table S6) and the corresponding sublimation enthalpies, ΔgcrHom (Tav), were derived (see details in SI). Our new results and the available results from ref. 13 were treated consistently in this work to calculate and compare the ΔgcrHom (298.15 K) values. The compilation and comparison of the standard molar sublimation enthalpies of α,ω-alkanediols are listed in Table 5.
| Diol | Methoda | T-Range | ΔgcrHom/Tav | ΔgcrHom/298.15 Kb | Ref. |
|---|---|---|---|---|---|
| a Methods: C = calorimetry; T = transpiration method; PhT = from consistency of phase transition enthalpies (see Table S15).b Vapor pressures from Table S6 were treated using eqn (S2) and (S3) with help of heat capacity differences from Table S7 to calculate the enthalpies of sublimation at 298.15 K. Uncertainties of the sublimation enthalpies U(ΔgcrHom) are the expanded uncertainties (0.95 level of confidence) calculated as described elsewhere.55 They include uncertainties from the fitting equation and uncertainties from temperature adjustment to T = 298.15 K. The uncertainties in the temperature adjustment of the literature data on sublimation enthalpies to the reference temperature T = 298.15 K are estimated to account with 20% to the total adjustment.c Weighted average (uncertainties were taken into account as weighting factors). The values in bold are recommended for further thermochemical calculations.d Sum of ΔglHom (298.15) = (113.6 ± 2.1) kJ mol−1 [Table 3] and ΔlcrHom (298.15 K) = (34.5 ± 0.7) from Table 6. | |||||
| 1,6-Hexanediol (cr) | C | 298.15 | 112.0 ± 0.8 | 13 | |
| T | 290.4–314.2 | 111.8 ± 0.6 | 111.9 ± 0.7 | Table S6 | |
| PhT | 112.2 ± 0.7 | Table S15 | |||
| 112.1 ± 0.4c | Average | ||||
| 1,8-Octanediol (cr) | C | 323.0 | 138.3 ± 1.6 | (140.6 ± 1.7) | 13 |
| T | 304.4–331.2 | 132.1 ± 1.4 | 132.9 ± 1.6 | Table S6 | |
| PhT | 131.8 ± 1.1 | Table S15 | |||
| 132.1 ± 0.9c | Average | ||||
| 1,9-Nonanediol (cr) | C | (148.1 ± 2.2)d | 13 | ||
| T | 298.2–318.7 | 137.2 ± 2.0 | 137.6 ± 2.2 | Table S6 | |
| PhT | 137.2 ± 0.8 | Table S15 | |||
| 137.3 ± 0.8c | Average | ||||
| 1,10-Decanediol (cr) | C | 342.0 | 149.8 ± 1.2 | (151.7 ± 1.3) | 13 |
| T | 315.5–343.7 | 145.8 ± 1.0 | 147.2 ± 1.2 | Table S6 | |
| PhT | 146.8 ± 1.5 | Table S15 | |||
| 147.0 ± 0.9c | Average | ||||
The analysis of the very few available data on the sublimation enthalpies for α,ω-alkanediols revealed a strong ambiguity in these results. While the new and old sublimation enthalpies for 1,6-hexanediol are largely in agreement, the discrepancies of 5 to 7 kJ mol−1 for 1,8-octanediol and 1,10-decanediol are considerable and require explanation or validation.
5.2 Step-by-step validation of sublimation enthalpies
It is also known that the sublimation enthalpy as a thermochemical property hardly obeys the additivity rules, in contrast to the vaporization enthalpy. The reason for this can be seen from eqn (3). The sublimation enthalpy, ΔgcrHom, contains not only the additive contribution of the vaporization enthalpy, ΔglHom, but also the high proportion of intermolecular interactions, which are captured by the fusion enthalpy, ΔlcrHom. Nevertheless, a validation of the experimental sublimation enthalpies is required, even if no direct empirical relationships can be applied. Fortunately, the same eqn (3) suggests a straightforward way to accomplish the required validation as shown in Fig. 5. | ||
| Fig. 5 Relationship between enthalpies of phase transition: crystal–gas, liquid–gas and crystal–liquid determined according to eqn (3). | ||
The reliable vaporization enthalpies, ΔglHom (298.15 K), that contribute to eqn (3), have just been validated in Tables 3 and 4. This is the first step in validating the experimental sublimation enthalpies.
What about the fusion enthalpy, ΔlcrHom (298.15 K), which makes the second contribution to eqn (3)? There has been no lack of experimental studies on the melting of α,ω-alkanediols in the literature (see Table S12). Provided that the literature values for fusion enthalpies measured at Tfus are correctly adjusted from Tfus to the reference temperature T = 298.15 K, the second step in the required validation of sublimation enthalpies is complete, and the result from the summation of ΔlcrHom (298.15 K) and ΔglHom (298.15 K) gives the empirically expected level to support our new sublimation enthalpy measurements. The evaluation of the available data for ΔlcrHom (Tfus) and their adjustment to T = 298.15 K is the subject of the following section.
The experimental data summarised in this table show significant but acceptable variations in the melting parameters for each diol. These variations are natural, as the melting temperatures and phase transition enthalpies are sensitive to the purity of the sample analysed as well as the heating/cooling rates applied during the DSC studies. However, apart from some obvious outliers, at least two matching experimental results were found in the literature for most alkanediols and averaged (see Table S12) for the evaluation and discussion in this section.
The DSC measurements revealed an interesting systematic change in the melting points for α,ω-alkanediols.64 It was observed that the odd-numbered members (hereinafter referred to as “odd”) in this homologous series are less well packed than the even-numbered members (hereinafter referred to as “even”).64 The plot of the averaged ΔlcrHom (Tfus) against Tfus also showed that the energetics of the solid–liquid phase transitions for α,ω-alkanediols are specific to the odd and even members, as shown in Fig. 6.
The fusion enthalpies for C3 to C16 diols are divided into two groups for the even and odd members of the homologous family. Both groups showed their own linear dependence, which was approximated by the following equations:
| odd diols: ΔlcrHom (298.15 K)/(kJ mol−1) = 0.4416 × Tfus − 102.2 with R2 = 0.9967 | (7) |
| even diols: ΔlcrHom (298.15 K)/(kJ mol−1) = 0.9365 × Tfus − 275.0 with R2 = 0.9835 | (8) |
The X-ray data collected by Thalladi et al.64 showed that the even-numbered diols form a layer-like network and the odd-numbered members form a three-dimensional structure. As a result, the odd-numbered members in both series are less tightly packed than the even-numbered members. This quantitative finding is now supported by the collection of fusion enthalpies of α,ω-alkanediols evaluated in Tables S12–S14.
It is worth noting that two compounds, 1,2-ethanediol and 1,4-butanediol, completely fall out of the correlation according to eqn (7) and (8). The obvious explanation for these outliers is that the competition between the strong intramolecular hydrogen bonding and the strong intermolecular hydrogen bonding in both species influences the formation of the crystal lattice. However, it cannot be completely ruled out that the deviation is partly due to the considerable variation (from 4–7 kJ mol−1) in the fusion enthalpies for 1,2-ethanediol and 1,4-butanediol reported in the literature (see Table S12). The very high correlation coefficients R2 obtained for both linear dependencies can be regarded as evidence of good consistency of the fusion parameters within each group and indirectly support the selection of the experimental values for 1,5-pentanediol and 1,7-heptanediol, where the discrepancy between the available data precluded the use of a mean value.
The temperature adjustment of the fusion enthalpies from Tfus to T = 298.15 K was carried out according to the following equation:
| ΔlcrHom (298.15 K)/(J mol−1) = ΔlcrHom (Tfus/K) − (ΔlcrCop,m) × [(Tfus/K) − 298.15 K] | (9) |
The differences in heat capacity between the crystalline and liquid phases, ΔlcrCop,m, were determined in our recent work21 and are listed in Table S7. The summary of the ΔlcrHom (298.15 K)-values adjusted to the reference temperature is listed in Table 6, column 6.
| Diols | Tfusb (K) | ΔlcrHom (Tfus)b (kJ mol−1) | ΔlcrCop,m![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c (J K−1 mol−1) c (J K−1 mol−1) |
(Tfus – 298.15) (K) | ΔlcrHom (298.15 K)d (kJ mol−1) |
|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 |
| a Uncertainties are presented as expanded uncertainties (0.95 level of confidence with k = 2).b Averaged experimental values from Table S12.c The heat capacity differences from Table S7.d The experimental enthalpies of fusion ΔlcrHom measured at Tfus (see Table S12) were adjusted to T = 298.15 K according to eqn (9). | |||||
| Even | |||||
| 1,6- | 316.3 | 22.9 ± 0.2 | −77.5 | 18.2 | 21.5 ± 0.5 |
| 1,8- | 332.2 | 36.2 ± 0.2 | −91.0 | 34.1 | 33.1 ± 1.0 |
| 1,10- | 345.7 | 45.1 ± 0.2 | −104.5 | 47.6 | 40.1 ± 1.5 |
| 1,12- | 352.5 | 53.5 ± 0.6 | −118.0 | 54.4 | 47.1 ± 2.0 |
| 1,14- | 359.8 | 63.4 ± 0.4 | −131.5 | 61.7 | 55.3 ± 2.5 |
| 1,16- | 366.0 | 69.8 ± 0.4 | −145.0 | 67.9 | 60.0 ± 3.0 |
| Odd | |||||
| 1,7- | 290.5 | 26.3 ± 0.6 | −84.3 | −7.6 | 26.9 ± 0.6 |
| 1,9- | 319.1 | 36.5 ± 0.4 | −97.8 | 21.0 | 34.5 ± 0.7 |
| 1,11- | 334.1 | 45.9 ± 0.5 | −111.3 | 36.0 | 41.9 ± 1.3 |
| 1,13- | 350.7 | 53.7 ± 0.7 | −124.8 | 52.6 | 47.1 ± 2.1 |
| 1,15- | 362.7 | 58.0 ± 0.5 | −138.3 | 64.6 | 49.1 ± 2.7 |
These values can now be combined with the vaporization enthalpies ΔglHom (298.15 K) evaluated in Table 3 to verify the experimental sublimation enthalpies of the solid α,ω-alkanediols measured in this work using the transpiration method (see Table 6), as shown in the next section.
As can be seen from Table 5, the sublimation enthalpies ΔgcrHom (298.15 K) of 1,6-hexanediol, 1,8-octanediol, 1,9-nonanediol and 1,10-decanediol, calculated according to eqn (3), do not agree in all cases (with the exception of 1,6-hexanediol) with the calorimetric results of Knaut and Sabbah,13 whereas they agree very well with our new experimental values measured using transpiration (see Table S6) for all four diols. This agreement was considered to be the reason for excluding the calorimetric sublimation enthalpies from ref. 13 from the calculation of the average values in Table 5. The weighted average ΔgcrHom (298.15 K) values for 1,6-hexanediol, 1,8-octanediol, 1,9-nonanediol and 1,10-decanediol are given in bold in Table 5 and were recommended for thermochemical calculations according to the “top-down” and “bottom-up” strategies.
An interesting additional option for validating the ΔgcrHom (298.15 K) values for α,ω-alkanediols resulted from a correlation with the fusion enthalpies ΔlcrHom (Tfus) evaluated in Table S12. The reason for this surprisingly good correlation is that both thermochemical properties are closely related to the lattice energy, which increased simultaneously with the growing chain length in the properties involved in the correlation. A good linear correlation (see Table S16):
| ΔgcrHom (298.15 K)/kJ mol−1 = 1.67 × ΔlcrHom (Tfus) + 74.9 with R2 = 0.9899 (for C6–C16) | (10) |
6. Top-down strategy: what ΔfHom (liq or cr)-values can finally be expected for α,ω-alkanediols?
The careful evaluation and revision of the experimental vaporization and sublimation enthalpies of α,ω-alkanediols, which was carried out in Sections 4 and 5, ultimately paves the way for the realization of the “top-down” strategy and the assessment of the expected “theoretical” condensed-phase enthalpies of formation, ΔfHom (liq or cr), from the combination of the QC gas phase formation enthalpies (Table 7, column 2) and the vaporization/sublimation enthalpies evaluated in this work (see Table 7, columns 3 and 4).| Diol | ΔfHom (QC)a | ΔglHom![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b b |
ΔgcrHom![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
ΔfHom (liq or cr)d | ΔfHom (liq or cr)e | Δf |
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 |
| a DLPNO values from Table 2. The uncertainties were defined as sd × t, where sd is the standard deviation from the weighted average value and t is Student's coefficient for the 95% confidence level. The values from this column plotted versus chain-length NC provide the straight line): ΔfHom (g, 298.15 K)/(kJ mol−1) = −343.5 − 20.6 × NC with R2 = 0.9999 (for C ≥ 5).b From Table 3.c From Table 5.d “Theoretical” values calculated using eqn (1) and (2) and data from columns 2 and 3 or 4.e Experimental values from Table 8.f Difference between column 6 and 5.g Measured in this work, see Table 8. | ||||||
| 1,2-Ethanediol (liq) | −390.0 ± 2.3 | 65.7 ± 0.2 | −455.7 ± 2.3 | −455.9 ± 0.6 | −0.2 ± 2.4 | |
| 1,3-Propanediol (liq) | −412.4 ± 2.3 | 71.5 ± 0.3 | −483.9 ± 2.3 | −482.8 ± 1.6 | 1.1 ± 2.8 | |
| 1,4-Butanediol (liq) | −426.4 ± 2.6 | 79.0 ± 0.7 | −505.4 ± 2.7 | −504.7 ± 1.7 | 0.7 ± 3.2 | |
| 1,5-Pentanediol (liq) | −446.6 ± 2.4 | 86.6 ± 0.6 | −533.2 ± 2.5 | −530.2 ± 2.5 | 3.0 ± 3.5 | |
| 1,6-Hexanediol (cr) | −466.7 ± 2.1 | 112.1 ± 0.4 | −578.8 ± 2.1 | −579.8 ± 2.5 | −1.0 ± 3.3 | |
| 1,7-Heptanediol (liq) | −487.9 ± 2.4 | 94.8 ± 0.4 | −582.7 ± 2.4 | (−574 ± 19) | 8.7 ± 19 | |
| 1,8-Octanenediol (cr) | −508.5 ± 2.4 | 132.1 ± 0.9 | −640.6 ± 2.6 | −642.0 ± 1.4g | −1.4 ± 3.0 | |
| 1,9-Nonanediol (cr) | −528.7 ± 2.3 | 137.3 ± 0.8 | −666.0 ± 2.4 | (−658 ± 15) | 8.0 ± 15 | |
| 1,10-Decanenediol (cr) | −549.5 ± 2.4 | 147.0 ± 0.9 | −696.5 ± 2.6 | −698.8 ± 1.0 | −2.3 ± 2.9 | |
The ΔfHom (liq or cr) values given in Table 5, column 5, provide a valuable guideline for evaluating the scattering experimental results found in the literature (see Table 1) as shown in the next section.
7. Bottom-up strategy: condensed-phase enthalpies of formation from combustion calorimetry
7.1 Review and revision of the condensed-phase enthalpies of formation of α,ω-alkanediols
The history of combustion experiments with α,ω-alkanediols began in 193665 with measurements on 1,2-ethanediol, and surprisingly, these early results (albeit with a high degree of uncertainty) still agree with later studies. Table 8 summarizes the available experimental results from combustion calorimetry.| Diol | Δcuo (liq or cr)a/J g−1 | ΔcHom (liq or cr)b/kJ mol−1 | ΔfHom (liq or cr)b/kJ mol−1 | Purityc/mol fraction |
|---|---|---|---|---|
| a Original experimental values from the literature (n/a = not available). Uncertainties correspond to standard uncertainties of the mean.b Results were recalculated in this work according to modern requirements for combustion calorimetry.72 Uncertainties correspond to expanded uncertainties of the mean (0.95 level of confidence).c Purity level given in the original literature.d The weighted mean value (uncertainties were taken as the weighing factor), values in parentheses were not involved in the averaging.e From equilibrium studies (see for details Section 7.3).f From chain-length dependence (Table S22).g From (Table S21). | ||||
| 1,2-Ethanediol (liq) | −19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 176 ± 4366 176 ± 4366 |
−1191.4 ± 7.1 | −453.1 ± 7.1 | 0.99 |
−19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 356 ± 4867 356 ± 4867 |
−1202.6 ± 6.0 | (−441.9 ± 6.1) | — | |
−19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 147 ± 4068 147 ± 4068 |
−1189.6 ± 5.0 | −454.9 ± 5.1 | — | |
−19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 137 ± 4869 137 ± 4869 |
−1189.0 ± 6.0 | −455.9 ± 0.9 | — | |
| n/a52 | −1188.8 ± 0.7 | −455.6 ± 0.8 | 0.999 | |
−19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 066 ± 4611 066 ± 4611 |
−1184.6 ± 5.7 | −459.9 ± 5.7 | 0.9913 | |
| — | −458.2 ± 2.5e | |||
| −455.9 ± 0.6d | ||||
| 1,3-Propanediol (liq) | n/a52 | −1859.0 ± 2.3 | (−464.8 ± 2.5) | 0.999 |
−24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 187 ± 6711 187 ± 6711 |
−1843 ± 10 | −480.8 ± 10.3 | 0.9923 | |
−24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 478 ± 1817 478 ± 1817 |
−1865.1 ± 3.0 | (−458.7 ± 3.0) | — | |
| −483.6 ± 2.2e | — | |||
−24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 171.9 ± 16.09 171.9 ± 16.09 |
−1841.8 ± 2.5 | −482.0 ± 2.5 | ||
| −482.8 ± 1.6d | ||||
| 1,4-Butanediol (liq) | −27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 688 ± 2452 688 ± 2452 |
−2499.1 ± 2.9 | −504.2 ± 3.0 | 0.998 |
−27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 698 ± 1752 698 ± 1752 |
−2499.9 ± 2.0 | −503.3 ± 2.1 | 0.998 | |
−27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 650 ± 6311 650 ± 6311 |
−2496 ± 12 | −508 ± 12 | 0.9939 | |
−27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 679.0 ± 9.070 679.0 ± 9.070 |
−2498.2 ± 1.9 | −505.0 ± 2.0 | — | |
| −504.7 ± 1.7d | ||||
| 1,5-Pentanediol (liq) | n/a65 | −3156 ± 5.0 | −526.5 ± 5.2 | — |
| n/a52 | −3151.1 ± 2.8 | −531.4 ± 2.9 | 0.998 | |
−30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 234 ± 5511 234 ± 5511 |
−3154 ± 12 | −529 ± 12 | 0.9919 | |
| −530.2 ± 2.5d | ||||
| 1,6-Hexanediol (cr) | n/a52 | −3791.9 ± 4.8 | −569.9 ± 5.0 | 0.998 |
| n/a18 | −3999.8 ± 4.6 | (−362.2 ± 4.8) | 0.99 | |
−32![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ± 3712 000 ± 3712 |
−3787.7 ± 9.2 | −574.1 ± 9.2 | 0.9938 | |
−31![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 916.7 ± 1.619 916.7 ± 1.619 |
−3778.0 ± 2.0 | −583.9 ± 3.0 | 0.9999 | |
| −579.8 ± 2.5d | ||||
| 1,7-Heptanediol (liq) | −33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 734 ± 7012 734 ± 7012 |
−4467 ± 19 | (−574 ± 19) | 0.9920 |
| −582.7 ± 1.5f | ||||
| 1,8-Octanenediol (cr) | −34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 778 ± 3312 778 ± 3312 |
−5094 ± 10 | (−627 ± 10) | 0.9991 |
−34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 671.2 ± 1.6 [Table S19] 671.2 ± 1.6 [Table S19] |
−5078.6 ± 0.9 | −642.0 ± 1.4 | 0.9996 | |
| 1,9-Nonanediol (cr) | −35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 771 ± 4412 771 ± 4412 |
−5742 ± 15 | (−658 ± 15) | 0.9981 |
| −668.3 ± 1.7g | ||||
| 1,10-Decanenediol (cr) | 36![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 585.3 ± 7.371 585.3 ± 7.371 |
−6387.3 ± 3.7 | −692.0 ± 3.9 | — |
−36![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 660 ± 1312 660 ± 1312 |
−6400.3 ± 6.4 | −678.9 ± 6.5 | 0.9933 | |
−36![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 540.0 ± 1.520 540.0 ± 1.520 |
−6379.4 ± 0.9 | −699.7 ± 1.0 | 0.9999 | |
| −698.8 ± 1.3d | ||||
The treatment of the primary results of combustion experiments involves numerous important details, references and steps, which were ultimately standardised in the IUPAC compilation published by Sunner and Mansson in 1979.72 For this reason, we have used the standard molar energies of combustion, Δcuo, given in the original literature (see Table 8, column 2) and converted them uniformly to the final condensed standard molar enthalpies of formation, ΔfHom (liq or cr), according to the procedure recommended in the IUPAC book72 (see Table 8, column 4).
Most combustion experiments with α,ω-alkanediols were conducted by Knauth and Sabbah11,12 using a rocking calorimeter with a stainless steel micro bomb. Since the standard molar enthalpies of formation of α,ω-alkanediols derived from combustion experiments by Knauth and Sabbah11,12 are subject to considerable uncertainty, the necessary experimental details of their study must be briefly summarised in order to discuss possible sources of error.
First of all, the purity of the samples used for combustion calorimetry is crucial for the reliability of the formation enthalpies derived using this method. Conventionally, the typical purity of samples is between 0.9995 and 0.9999, expressed in terms of molar fraction. Particularly restricted are quantities of low-molecular impurities such as traces of water or solvents (acetone, methanol, methylene chloride, etc.) as a rule used for recrystallization of samples for combustion experiments. The commercial samples, used by Knauth and Sabbah,11,12 were purified by rectification using a rotary band column with 30 theoretical plates (for liquids) and by sublimation at 313 K under a reduced pressure of approximately 1 Pa (for solids). The sample of 1,6-hexanediol (99+%, “gold label”, Aldrich) was used without further purification. The purity of the samples was determined by by low-temperature differential thermal analysis using the freezing-point method. Despite additional purification, the samples used by Knauth and Sabbah11,12 were significantly below the conventional requirements for combustion calorimetry (see Table 2).
Diols are known to be highly hygroscopic. To prevent contamination of the hygroscopic diols by atmospheric moisture prior to combustion, the liquid and solid diol samples were sealed in small polyethylene containers.11,12 Knauth and Sabbah11,12 used a rocking microcalorimeter calibrated for precise measurements of the combustion energies of small samples (5–10 mg). It is well known,73 that experimental work with a typical microcalorimeter and such small samples is no trivial task.74,75 Apart from purely technical difficulties in recording the very small temperature rise during the combustion experiment, even the seemingly simple problem of reproducible weighing of the tiny samples arose. Nevertheless, accurate measurements with the microcalorimeter are possible, and the reproducibility of the measurements at a level of 0.01% (based on the combustion energy) is generally proven,74,75 at least for the measurements of the primary standard (benzoic acid) and the secondary standards (salicylic acid and succinic acid).
Calibration of the microcalorimeter by Knauth and Sabbah11,12 using benzoic acid yielded a value of 0.025%, which is not perfect but still acceptable. The polyethylene used as a container for the combustion experiments was also measured with the same reproducibility.11,12 In this context, the very poor reproducibility of the combustion experiments with diols (e.g. 0.095% with 1.8-octanediol) does not indicate problems with the equipment, but either problems in the preparation of the samples for the measurements or possibly incomplete combustion of the sample.
It was not possible to verify the completeness of combustion of such tiny samples using conventional methods (e.g. visual inspection or CO2 recovery76). However, in our opinion, a possible reason for the low reproducibility of the combustion experiments by Knauth and Sabbah11,12 could be the partial loss of polyethylene mass during the sealing of the container using the conventional hot wire method (see details in the IUPAC book72). From our experience, we know that sometimes, if the polyethylene container is too close to the glowing wire, a very small amount of the polyethylene evaporates, leading to a significant loss of mass (especially for the 5–10 mg sample). With this plausible explanation for the poor reproducibility of the combustion experiments by Knauth and Sabbah,11,12 it should be recognised that for most diols, their average enthalpies of formation are in good agreement with the results obtained with other types of calorimeters and typical sample quantities (from 0.5 to 1.0 g).
Even the uniformly treated, available combustion results for each α,ω-alkanediol summarised in Table 8 showed considerable variation in values, so that only with the help of the “theoretical” enthalpies of formation from Table 7, column 5, an apparent outliers for 1,7-heptanediol, 1,8-octanediol and 1,9-nonanediol measured in ref. 12 could be safely discarded (see Table 8, column 4). Given the very uncertain enthalpies of formation for 1,7-heptanediol, 1,8-octanediol and 1,9-nonanediol, we decided to repeat the combustion experiments with 1,8-octanediol using a macro-calorimeter with a static bomb (see SI for necessary details).
7.2 Crystalline-phase enthalpy of formation of 1,8-octanediol from combustion calorimetry
The combustion experiments with 1,8-octanediol refer to the reaction:| C8H18O2(cr) + 11.5 O2 (g) = 8 CO2 (g) + 9 H2O (liq) | (11) |
The standard specific energy of combustion Δcuo (cr) of 1,8-octanediol was determined based on five experiments. The latter value was used to calculate the experimental standard molar enthalpy of combustion, ΔcHom (cr), which refers to the reaction according to eqn (11). The ΔcHom-value was used to obtain the standard molar enthalpy of formation ΔfHom (cr) in the crystalline state, applying Hess's Law to the reaction according eqn (11), and the standard molar enthalpies of formation of CO2(g) and H2O(liq) assigned by CODATA.77 Uncertainties associated with the combustion experiments were estimated according to the procedure recommended by Olofsson.78 The auxiliary information on the combustion experiments is provided in Tables S17–S19. The final results of the combustion experiments with 1,8-octanediols are listed in Table 8. It is difficult to judge whether our new result ΔfHom (cr) = −642.0 ± 1.4 kJ mol−1 is consistent with the previous result ΔfHom (cr) = −627 ± 10 kJ mol−1,12 as the latter value is subject to considerable uncertainty. However, the “theoretical” value ΔfHom (cr) = −640.6 ± 2.6 kJ mol−1 (see Table 7, column 5) from the “top-down” strategy supports our new value.
7.3 Liquid-phase enthalpies of formation of 1,2-ethanediol and 1,3-propanediol from equilibrium studies
A series of equilibrium studies on acetals and ketals synthesis reactions was published by Anteunis et al.79 Two of these, (R1) and (R2) are shown in Fig. 7. | ||
| Fig. 7 The equilibrium reactions of ketal synthesis used to validate the liquid-phase enthalpies of formation of 1,2-ethanediol and 1,3-propanediol (see text). | ||
The reaction enthalpies ΔrHom (liq, 298.15 K) = −(10.0 ± 1.5) kJ mol−1 (ref. 79) for (R1) and ΔrHom (liq, 298.15 K) = −(23.0 ± 1.5) kJ mol−1 (ref. 79) for (R2) measured by NMR-spectroscopy, can be used to independently derive the liquid-phase enthalpies of formation of 1,2-ethanediol and 1,3-propanediol using the Hess's Law and the known enthalpies of formation of acetone, water, and ketals, which are collected in Table S20. The resulting enthalpies of formation are well comparable with the results of combustion calorimetry (see Table 8), and these independently derived values facilitated the selection of a reliable level, particularly in the case of 1,3-propanediol, where the available results differ by about 20 kJ mol−1 (see Table 8, column 4).
7.4 Empirical correlation: liquid phase enthalpies of formation versus Nc
The significant support provided by the “top-down” values for the enthalpies of formation of diols in the condensed phase does not preclude further validation of the experimental combustion results using structure–property correlations, as was already carried out for the vaporization enthalpies in Section 4. From the fusion enthalpies of the solid diols evaluated in Table 6, the set of enthalpies of formation in the liquid phase, ΔfHom (liq, 298.15 K), can be derived for the C6 to C10 diols according to eqn (12):| ΔfHom (liq, 298.15 K) = ΔfHom (cr, 298.15 K) − ΔlcrHom (298.15 K) | (12) |
These values are given in Table S21 and can be used, for example, for chain length correlation. The correlation of the experimental ΔfHom (liq, 298.15 K) values with the number of carbon atoms in the alkyl chain of diols showed a linear dependence (see Table S22):
| ΔfHom (liq, 298.15 K)/(kJ mol−1) = −403.6 − 25.58 × NC with R2 = 0.9996 (for C ≥ 5) | (13) |
| 1 | ΔfHom (liq)expa | ΔfHom (liq)addb | Δc | ΔglHom (exp)d | ΔglHom (add)e | Δf | ΔfHom (g)expg | ΔfHom (g)addh | Δi |
|---|---|---|---|---|---|---|---|---|---|
| 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | |
| a Experimental values from Table 8 and Table S21.b Estimated using group-additivity contributions from Table S23.c Difference between columns 2 and 3 in this table.d Experimental values from Table 3.e Estimated using group-additivity contributions from Table S23.f Difference between columns 5 and 6 in this table.g Experimental values from Table 10.h Estimated using group-additivity contributions from Table S23.i Difference between columns 8 and 9 in this table.j From Table S21.k Calculated from the chain length dependence in Table S22. | |||||||||
| 1,2 | −455.9 | −445.3 | −10.6 | 65.7 | 77.4 | −11.7 | −390.2 | −375.3 | −14.9 |
| 1,3 | −482.8 | −474.2 | −8.6 | 71.5 | 81.9 | −10.7 | −411.3 | −399.1 | −12.2 |
| 1,4 | −504.7 | −502.2 | −2.5 | 79.0 | 81.6 | −2.6 | −425.7 | −422.8 | −2.9 |
| 1,5 | −530.2 | −530.4 | 0.2 | 86.6 | 86.2 | 0.4 | −443.6 | −444.5 | 0.6 |
| 1,6 | −558.3j | −556.2 | −2.1 | 90.7 | 90.5 | 0.2 | −467.7 | −466.2 | −1.7 |
| 1,7 | (−582.7)k | −582.0 | −0.7 | 94.8 | 94.8 | 0.0 | −487.9 | −487.9 | 1.1 |
| 1,8 | −608.9j | −607.8 | 1.1 | 98.7 | 99.0 | −0.3 | −509.9 | −509.6 | 1.7 |
| 1,9 | (−633.8)k | −633.7 | −0.1 | 102.7 | 103.3 | −0.6 | (−531.0) | −531.3 | 0.9 |
| 1,10 | −658.7j | −659.5 | 0.8 | 106.7 | 107.5 | −0.8 | −551.8 | −553.0 | 0.8 |
7.5 Empirical correlation: thermochemical properties from group-additivity
Group additivity (GA) is a well-established empirical method that is commonly used either to predict thermochemical properties or to validate experimental results.80 For molecules that are not too large, have a hydrocarbon skeleton (possibly with a low degree of branching) and only a few functional groups, the GA method provides reliable estimates for unknown properties.81 For the chemical family of alkanols, reliable GA contributions have been developed after careful revision and validation of experimental thermochemical data ΔglHom (298.15 K) and ΔfHom (liq or g, 298.15 K) for alkanes and alkanols.82 A summary of the GA values required for working with diols is listed in Table S23 together with an example of the estimation. A comparison of the experimental and group-additivity-estimated thermochemical properties of α,ω-alkanediols is presented in Table 9.The results shown in Table 9 for the vaporization enthalpies, the enthalpies of formation in the liquid and gaseous phases generally correspond to expectations. The calculated and experimental properties agree within the experimental uncertainties for the long-chain members with C ≥ 5 and are slightly outside their limits for 1,4-butanediol. The properties of 1,2-ethanediol and 1,3-propanediol are obviously affected by intramolecular hydrogen bonding, whereas its influence in 1,4-butanediol is much less. Since the strength of intramolecular hydrogen bonding is individual for all three species, their thermochemical properties do not obey the rules of additivity. So, the GA method used in this work generally supports the selection of reliable values for the thermochemical properties of α,ω-alkanediols.
7.6 Bottom-up strategy: where is the advantage?
Section 6 of this paper has clearly shown that modern advances in quantum chemistry are now an indispensable tool, not to compete, but to help synergistically in the challenging task of evaluating, validating, and critically revising experimental data. In combination with the evaluated phase transition enthalpies, the guiding values ΔfHom (liq or cr, 298.15 K) were derived, and these values were helpful in clarifying the conflicting experimental data.Sections 7.1–7.5 have conclusively demonstrated that the critically evaluated experimental data for ΔfHom (liq or cr, 298.15 K) of α,ω-alkanediols are ultimately consistent and can be recommended for thermochemical calculations. The summary of the evaluated thermochemical properties for α,ω-alkanediols is given in Table 10.
| Diol | ΔfHom (liq or cr)expb | Δgl,crHom![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
ΔfHom (g)expd | ΔfHom (g)QCe | Δf |
|---|---|---|---|---|---|
| a Uncertainties correspond to expanded uncertainties of the mean (0.95 level of confidence).b From Table 8.c From Tables 3 and 5.d Sum of column 2 and 3 in this table.e From Table 7, column 2.f Difference between column 4 and 5 in this table. | |||||
| 1,2-Ethanediol (liq) | −455.9 ± 0.6 | 65.7 ± 0.2 | −390.2 ± 0.6 | −390.0 ± 2.3 | −0.2 |
| 1,3-Propanediol (liq) | −482.8 ± 1.6 | 71.5 ± 0.3 | −411.3 ± 1.6 | −412.4 ± 2.3 | 1.1 |
| 1,4-Butanediol (liq) | −504.7 ± 1.7 | 79.0 ± 0.7 | −425.7 ± 1.8 | −426.4 ± 2.6 | 0.7 |
| 1,5-Pentanediol (liq) | −530.2 ± 2.5 | 86.6 ± 0.6 | −443.6 ± 2.6 | −446.6 ± 2.4 | 3.0 |
| 1,6-Hexanediol (cr) | −579.8 ± 2.5 | 112.1 ± 0.4 | −467.7 ± 2.5 | −466.7 ± 2.1 | −1.0 |
| 1,7-Heptanediol (liq) | −582.7 ± 1.5 | 94.8 ± 0.4 | −487.9 ± 1.6 | −487.9 ± 2.4 | 0.0 |
| 1,8-Octanenediol (cr) | −642.0 ± 1.4 | 132.1 ± 0.9 | −509.9 ± 1.7 | −508.5 ± 2.4 | −1.4 |
| 1,9-Nonanediol (cr) | −668.3 ± 1.7 | 137.3 ± 0.8 | −531.0 ± 1.8 | −528.7 ± 2.3 | −2.3 |
| 1,10-Decanenediol (cr) | −698.8 ± 1.3 | 147.0 ± 0.9 | −551.8 ± 1.6 | −549.5 ± 2.4 | −2.3 |
Our main aim has therefore been achieved. It may appear now that the “bottom-up” strategy is unnecessary – at least for the purposes of this paper. But this is incorrect as will be shown in the following discussion:
Let us compare the experimental (Table 10, column 4) and QC calculated (Table 10, column 5) ΔfHom (g) values. Except for 1,5-pentanediol, the difference between their values does not exceed 3 kJ mol−1, which is significantly better than “chemical accuracy”. This perfect correspondence confirmed that our assumption that the most stable conformer of α,ω-alkanediol adequately represents the gas phase mixture of the conformers is correct.
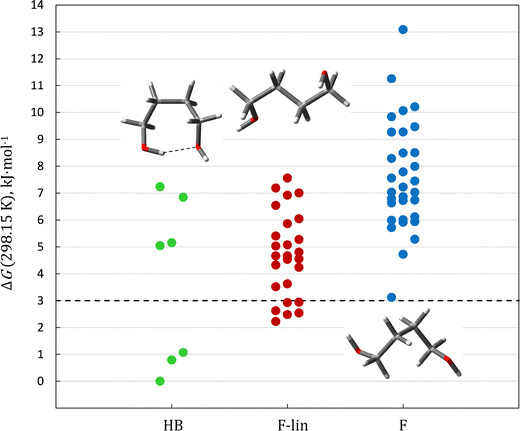
At the same time, as noted above (Section 3), the QC calculations do not provide an unambiguous conclusion about the most stable conformer of 1,4-butanediol (see Table 2). Although an approximate accounting of vibrational anharmonicity (Table 2) suggests that F-lin may be the main conformer of this diol, a comparison of the Gibbs free energies indicates that the most stable conformer should be HB. The experimental data obtained in this work, in our opinion, make it possible to solve this problem, which has long been discussed in the literature. Fig. 8 shows that for the C4–C10 diols, the QC calculations agree within their uncertainties with the experiment when F-lin is considered as the main conformer, whereas for HB conformers the discrepancies go far beyond the calculation errors.
The opposite result is evident for 1,2-ethanediol and 1,3-propanediol, where only the HB conformers match the experimental enthalpies of formation. This example shows that carefully evaluated experimental values are still indispensable and that the “bottom-up” strategy should not be neglected, even as QC methods continue to advance.
8. Hybrid strategy: bridging the two approaches
The previous section made clear that only a judicious combination of top-down and bottom-up strategies paves the way for a successful experimental and thermochemical work. This combination, known as a hybrid strategy, offers the following advantages:• Improvement of the reliability of predicted enthalpies.
• Cross-validation of computational and experimental methods.
• Time savings by providing guidance for experiments (e.g., avoiding redundant runs).
• Provision of databases with highly reliable thermodynamic values for new organic compounds.
This hybrid strategy is particularly important for the workflow in modern experimental thermodynamics. In the last century, experiments took precedence over theoretical and empirical methods. Experimental thermochemical results were usually published without any validation. The mandatory discussion of the data was generally limited to a comparison with results already available in the literature (if available). Interestingly, the data measured later were considered more reliable. As a consequence, compilations of experimental data available in the literature (e.g. the most popular NIST Webbook83) only contain a list of a given thermochemical property as published in the original work. This means that users of the data are free to choose any value from this list or to calculate the average of the values, regardless of how large the spread of values within the list is.
In this century, the “hybrid strategy” should dominate, and the “top-down” strategy must be applied before the experiments are planned in order to understand the quality of the existing thermochemical properties. This strategy provides important information on whether new experiments are really necessary or whether the available data correspond to the “theoretical” expectations. In addition, every new thermochemical result should be validated using the “top-down” strategy before publication.
In our experience, the new experimental results were in many cases far from the theoretical “expectations”. Such a discrepancy was seen as a good opportunity to further purify the sample and repeat the experiment or to use a different thermochemical method to verify the questionable result. Only if these efforts were unsuccessful and the result remained unchanged using different techniques, the “bottom-up” strategy was applied to uncover possible shortcomings, inconsistencies or simplifications in the theoretical, quantum chemical or empirical methods used for the “top-down” assessments. Consequently, any new result can only be recommended after applying the “hybrid strategy” for thermochemical calculations.
9. Conclusions
Literature data describing the vaporization thermodynamics of different α,ω-alkanediols have been evaluated and significant inconsistencies between standard enthalpies of formation and vaporization have been shown. In this work, a hybrid strategy was applied that combines experimental results with quantum chemical calculations. This way the actual properties are accessed in a two-fold way: starting from the gas phase data with modelling data (top-down) and starting with experimental data for the condensed phase (bottom-up). Based on these results, it could be demonstrated that the available thermodynamic property data for α,ω-alkanediols deviate from the regular trend that could be expected for homologous series. Due to the significant contribution of intramolecularly hydrogen-bonded conformers in smaller molecules with the OH-groups close to each other, the properties of 1,2-ethanediol, 1,3-propanediol, and 1,4-butanediol differ considerably from those of their longer-chain counterparts. Starting from 1,5-pentanediol, the enthalpies of vaporization and the standard enthalpies of formation in the gas phase follow a regular linear trend. It can be assumed that this is due to the predominance of conformers without intramolecular hydrogen bonding.Conflicts of interest
There are no conflicts to declare.Data availability
All data are available in the text and in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6cp00136j.Acknowledgements
This project has been funded by the Free State of Bavaria through the project “Oxo-LOHC-Autotherme und ultratiefe Wasserstoff-Freisetzung aus LOHC-Systeme-Oxo-LOHC” (grant number: 84-6665a2/201/11). AAS was financially supported by the Committee on Science and Higher Education of the Government of St. Petersburg. AVEl was financially supported by the Ministry of Science and Higher Education of the Russian Federation (theme no. FSSE-2025-0006) as part of the state task of the Samara State Technical University (creation of new youth laboratories). OVD acknowledges the support within the framework of the state budget subject “Chemical Thermodynamics and Theoretical Materials Science” (121031300039-1).References
- Y. Amada, H. Watanabe, Y. Hirai, Y. Kajikawa, Y. Nakagawa and K. Tomishige, Production of Biobutanediols by the Hydrogenolysis of Erythritol, ChemSusChem, 2012, 5, 1991–1999 CrossRef CAS PubMed.
- K. Müller, K. Stark, V. N. Emel’yanenko, M. A. Varfolomeev, D. H. Zaitsau, E. Shoifet, C. Schick, S. P. Verevkin and W. Arlt, Liquid Organic Hydrogen Carriers: Thermophysical and Thermochemical Studies of Benzyl- and Dibenzyl-toluene Derivatives, Ind. Eng. Chem. Res., 2015, 54, 7967–7976 CrossRef.
- S. P. Verevkin, V. N. Emel’yanenko, A. Heintz, K. Stark and W. Arlt, Liquid Organic Hydrogen Carriers: An Upcoming Alternative to Conventional Technologies. Thermochemical Studies, Ind. Eng. Chem. Res., 2012, 51, 12150–12153 CrossRef CAS.
- Y. Chen, X. Kong, C. Yang, Y. Liao, G. Gao, R. Ma, M. Peng, W. Shao, H. Zheng, H. Zhang, X. Pan, F. Yang, Y. Zhu, Z. Liu, Y. Cao, D. Ma, X. Bao and Y. Zhu, A catalytic cycle that enables crude hydrogen separation, storage and transportation, Nat. Energy, 2025, 10, 971–980 CrossRef CAS.
- P. Wasserscheid, Impurity-tolerant catalysis, Nat. Energy, 2025, 10, 924–925 CrossRef CAS.
- T. Miyazawa, S. Koso, K. Kunimori and K. Tomishige, Glycerol hydrogenolysis to 1,2-propanediol catalyzed by a heat-resistant ion-exchange resin combined with Ru/C, Appl. Catal., A, 2007, 329, 30–35 CrossRef CAS.
- C. Mevawala, T. Autrey, K. Brooks, M. Bowden, B. L. Tran and K. Müller, 1,4-Butanediol as a Hydrogen Carrier: Liquid- versus Gas-Phase Dehydrogenation, Energy Fuels, 2023, 37, 560–566 CrossRef CAS.
- S. P. Verevkin, Determination of vapor pressures and enthalpies of vaporization of 1,2-alkanediols, Fluid Phase Equilib., 2004, 224, 23–29 CrossRef CAS.
- V. N. Emel’yanenko and S. P. Verevkin, Benchmark thermodynamic properties of 1,3-propanediol: Comprehensive experimental and theoretical study, J. Chem. Thermodyn., 2015, 85, 111–119 CrossRef.
- T. V. Vasiltsova, S. P. Verevkin, E. Bich, A. Heintz, R. Bogel-Lukasik and U. Domanska, Thermodynamic properties of mixtures containing ionic liquids. activity coefficients of ethers and alcohols in 1-methyl-3-ethyl-imidazolium bis(trifluoromethyl-sulfonyl) imide using the transpiration method, J. Chem. Eng. Data, 2005, 50, 142–148 CrossRef CAS.
- P. Knauth and R. Sabbah, Combustion calorimetry on milligram samples of liquid substances with a CRMT rocking bomb calorimeter. Application to the study of α,ω-alkanediols at 298.15 K, J. Chem. Thermodyn., 1989, 21, 203–210 CrossRef CAS.
- P. Knauth and R. Sabbah, Combustion calorimetry on milligram samples of hygroscopic solid substances with a CRMT rocking bomb calorimeter thermochemical study of α,ω-alkanediols at 298.15 K. Part II, J. Chem. Thermodyn., 1989, 21, 779–784 CrossRef CAS.
- P. Knauth and R. Sabbah, Energetics of intra- and intermolecular bonds in α,ω-alkanediols. III. Thermochemical study of 1,6-hexanediol, 1,7-heptanediol, 1,8-octanediol, 1,9-nonanediol, and 1,10-decanediol at 298.15 K, Can. J. Chem., 1990, 68, 731–734 CrossRef CAS.
- P. Knauth and R. Sabbah, Energetics of intra- and intermolecular bonds in α,ω-alkanediols, Struct. Chem., 1990, 1, 43–46 CrossRef CAS.
- V. Piacente, D. Ferro and G. D. Gatta, Vaporization enthalpies of a series of α,ω-alkanediols from vapour pressure measurements, Thermochim. Acta, 1993, 223, 65–73 CrossRef CAS.
- V. Piacente, D. Ferro and G. Della Gatta, Vaporization enthalpies of five odd-numbered (C7 to C15) α,ω-alkanediols, Thermochim. Acta, 1994, 232, 317–321 CrossRef CAS.
- L. Y. Tsvetkova, T. G. Kulagina and B. V. Lebedev, Thermodynamics of the Poly(trimethyl terephthalate) Synthesis Reaction in the Range 0-300 K, Vysokomol. Soedin., Ser. A, 2002, 44, 474–482 CAS.
- I. Contineanu, E. Corlateanu, J. Hersocovici and D. I. Marchidan, Combustion and formation enthalpies of 1,6-hexanediol and adipic acid, Rev. Chim., 1980, 31, 763–764 CAS.
- W. V. Steele, R. D. Chirico, A. Nguyen, I. A. Hossenlopp and N. K. Smith, DIPPR project 871: Determination of ideal-gas enthalpies of formation for key compounds: The 1989 project results, DIPPR Data Ser., 1991, 1, 101–134 CAS.
- W. V. Steele, R. D. Chirico, I. A. Hossenlopp, S. E. Knipmeyer, A. Nguyen and N. K. Smith, DIPPR project 871. Determination of ideal-gas enthalpies of formation for key compounds. The 1990 project results., DIPPR Data Ser., 1994, 2, 188–215 CAS.
- R. Siewert, V. V. Emelyanov, A. A. Samarov, M. Richter, K. Müller and S. P. Verevkin, Empirical Rules in Thermochemistry: Overlooked Overestimations of the Liquid- and Crystal-Phase Heat Capacities of α,ω-Alkanediols and Their Consequences, Liquids, 2025, 5, 20 CrossRef CAS.
- L. A. Curtiss, P. C. Redfern and K. Raghavachari, Gaussian-4 theory, J. Chem. Phys., 2007, 126, 084108 CrossRef PubMed.
- Y. Guo, C. Riplinger, U. Becker, D. G. Liakos, Y. Minenkov, L. Cavallo and F. Neese, Communication: An improved linear scaling perturbative triples correction for the domain based local pair-natural orbital based singles and doubles coupled cluster method [DLPNO-CCSD(T)], J. Chem. Phys., 2018, 148, 011101 CrossRef PubMed.
- D. W. Rogers and A. A. Zavitsas, Enthalpies of reaction, formation, and hydrogenation among long chain alkanes, monoalkenes, acids, alcohols, and amines: G4 computed and experimental results, J. Chem. Thermodyn., 2014, 79, 258–265 CrossRef CAS.
- K. Takahashi, Theoretical study on the effect of intramolecular hydrogen bonding on OH stretching overtone decay lifetime of ethylene glycol, 1,3-propanediol, and 1,4-butanediol, Phys. Chem. Chem. Phys., 2010, 12, 13950 RSC.
- P. Das, P. K. Das and E. Arunan, Conformational Stability and Intramolecular Hydrogen Bonding in 1,2-Ethanediol and 1,4-Butanediol, J. Phys. Chem. A, 2015, 119, 3710–3720 CrossRef CAS PubMed.
- O. V. Dorofeeva and T. A. Suchkova, Theoretical calculation of enthalpy of formation of multiconformational molecules: 1,2-ethanediol, propanediols, and glycerol, Chem. Phys. Lett., 2018, 698, 218–222 CrossRef CAS.
- M. Traetteberg and K. Hedberg, Structure and Conformations of 1,4-Butanediol: Electron-Diffraction Evidence for Internal Hydrogen Bonding, J. Am. Chem. Soc., 1994, 116, 1382–1387 CrossRef CAS.
- L. Evangelisti, Q. Gou, L. Spada, G. Feng and W. Caminati, Conformational analysis of 1,4-butanediol: A microwave spectroscopy study, Chem. Phys. Lett., 2013, 556, 55–58 CrossRef CAS.
- L. P. Kuhn, P. V. R. Schleyer, W. F. Baitinger and L. Eberson, Conformational Effects and Hydrogen Bonding in 1,4-Diols, J. Am. Chem. Soc., 1964, 86, 650–658 CrossRef CAS.
- R. R. Shagidullin, A. V. Chernova and R. R. Shagidullin, Intramolecular hydrogen bonds and conformations of the 1,4-butanediol molecule, Russ. Chem. Bull., 1993, 42, 1505–1510 CrossRef.
- A. J. Lopes Jesus, M. T. S. Rosado, M. L. P. Leitão and J. S. Redinha, Molecular Structure of Butanediol Isomers in Gas and Liquid States: Combination of DFT Calculations and Infrared Spectroscopy Studies, J. Phys. Chem. A, 2003, 107, 3891–3897 CrossRef CAS.
- A. J. L. Jesus, M. T. S. Rosado, I. Reva, R. Fausto, M. E. S. Eusébio and J. S. Redinha, Structure of Isolated 1,4-Butanediol: Combination of MP2 Calculations, NBO Analysis, and Matrix-Isolation Infrared Spectroscopy, J. Phys. Chem. A, 2008, 112, 4669–4678 CrossRef CAS PubMed.
- Y.-L. Cheng, H.-Y. Chen and K. Takahashi, Theoretical Calculation of the OH Vibrational Overtone Spectra of 1- n Alkane Diols (n = 2–4): Origin of Disappearing Hydrogen-Bonded OH Peak, J. Phys. Chem. A, 2011, 115, 5641–5653 CrossRef CAS PubMed.
- S. Kozuch, S. M. Bachrach and J. M. L. Martin, Conformational Equilibria in Butane-1,4-diol: A Benchmark of a Prototypical System with Strong Intramolecular H-bonds, J. Phys. Chem. A, 2014, 118, 293–303 CrossRef CAS PubMed.
- H.-Y. Chen, Y.-L. Cheng and K. Takahashi, Theoretical Calculation of the OH Vibrational Overtone Spectra of 1,5-Pentanediol and 1,6-Hexanediol, J. Phys. Chem. A, 2011, 115, 14315–14324 CrossRef CAS PubMed.
- I. K. Yoo, J. Il Kim and Y. K. Kang, Conformational preferences and antimicrobial activities of alkanediols, Comput. Theor. Chem., 2015, 1064, 15–24 CrossRef CAS.
- O. V. Dorofeeva and O. N. Ryzhova, Gas-Phase Enthalpies of Formation and Enthalpies of Sublimation of Amino Acids Based on Isodesmic Reaction Calculations, J. Phys. Chem. A, 2014, 118, 3490–3502 CrossRef CAS PubMed.
- V. N. Emel’yanenko, S. P. Verevkin and A. Heintz, The Gaseous Enthalpy of Formation of the Ionic Liquid 1-Butyl-3-methylimidazolium Dicyanamide from Combustion Calorimetry, Vapor Pressure Measurements, and Ab Initio Calculations, J. Am. Chem. Soc., 2007, 129, 3930–3937 CrossRef PubMed.
- S. P. Verevkin and V. N. Emel’yanenko, Transpiration method: Vapor pressures and enthalpies of vaporization of some low-boiling esters, Fluid Phase Equilib., 2008, 266, 64–75 CrossRef CAS.
- R. Dennington, T. A. Keith and J. M. Millam, GaussView, version 6, Semichem Inc., Shawnee Mission, KS, 2016 Search PubMed.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, M. C. X. Li, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, D. J. F. B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang and M. Hada, Gaussian 16, Revision C.01, Gaussian, Inc., Wallingford, CT Search PubMed.
- F. Neese, F. Wennmohs, U. Becker and C. Riplinger, The ORCA quantum chemistry program package, J. Chem. Phys., 2020, 152, 224108 CrossRef CAS PubMed.
- M. K. Kesharwani, B. Brauer and J. M. L. Martin, Frequency and Zero-Point Vibrational Energy Scale Factors for Double-Hybrid Density Functionals (and Other Selected Methods): Can Anharmonic Force Fields Be Avoided?, J. Phys. Chem. A, 2015, 119, 1701–1714 CrossRef CAS PubMed.
- B. Ruscic and D. H. Bross, Active Thermochemical Tables (ATcT), ver. 1.130, Argonne National Laboratory, 2023, https://atct.anl.gov/ Search PubMed.
- S. P. Verevkin, V. N. Emel’yanenko and G. Nell, 1,2-Propanediol. Comprehensive experimental and theoretical study, J. Chem. Thermodyn., 2009, 41, 1125–1131 CrossRef CAS.
- A. Nicolaides, A. Rauk, M. N. Glukhovtsev and L. Radom, Heats of Formation from G2, G2(MP2), and G2(MP2,SVP) Total Energies, J. Phys. Chem., 1996, 100, 17460–17464 CrossRef CAS.
- A. P. Scott and L. Radom, Harmonic Vibrational Frequencies: An Evaluation of Hartree−Fock, Møller−Plesset, Quadratic Configuration Interaction, Density Functional Theory, and Semiempirical Scale Factors, J. Phys. Chem., 1996, 100, 16502–16513 CrossRef CAS.
- Y. Zhao and D. G. Truhlar, Computational characterization and modeling of buckyball tweezers: density functional study of concave–convex π⋯π interactions, Phys. Chem. Chem. Phys., 2008, 10, 2813 RSC.
- R. F. Ribeiro, A. V. Marenich, C. J. Cramer and D. G. Truhlar, Use of Solution-Phase Vibrational Frequencies in Continuum Models for the Free Energy of Solvation, J. Phys. Chem. B, 2011, 115, 14556–14562 CrossRef CAS PubMed.
- L. H. Thomas and R. Meatyard, Viscosity and molecular association. Part VI. Association of dihydric alcohols and phenols, J. Chem. Soc. A, Inorg. Phys. Theor., 1966, 92 RSC.
- P. J. Gardner and K. S. Hussain, The standard enthalpies of formation of some aliphatic diols, J. Chem. Thermodyn., 1972, 4, 819–827 CrossRef CAS.
- R. M. Stephenson and S. Malanowski, Handbook of the Thermodynamics of Organic Compounds, Springer, Netherlands, Dordrecht, 1987 Search PubMed.
- P. Umnahanant, S. Kweskin, G. Nichols, M. J. Dunn, H. Smart-Ebinne and J. S. Chickos, Vaporization Enthalpies of the α,ω-Alkanediols by Correlation Gas Chromatography, J. Chem. Eng. Data, 2006, 51, 2246–2254 CrossRef CAS.
- S. P. Verevkin, A. Y. Sazonova, V. N. Emel’yanenko, D. H. Zaitsau, M. A. Varfolomeev, B. N. Solomonov and K. V. Zherikova, Thermochemistry of Halogen-Substituted Methylbenzenes, J. Chem. Eng. Data, 2015, 60, 89–103 CrossRef CAS.
- R. L. Rowley, W. V. Wilding, A. Congote and N. F. Giles, A Systems Approach for Improved Accuracy of Thermophysical Properties in the DIPPR 801 Database: 1, n -Alkanediols as a Case Study, J. Chem. Eng. Data, 2014, 59, 1031–1037 CrossRef CAS.
- C. T. Peng, Prediction of retention indices, J. Chromatogr. A, 2000, 903, 117–143 CrossRef CAS PubMed.
- G. C. Pimentel and A. L. McClellan, Hydrogen Bonding, Annu. Rev. Phys. Chem., 1971, 22, 347–385 CrossRef CAS.
- A. Vetere, New correlations for predicting vaporization enthalpies of pure compounds, Chem. Eng. J., 1979, 17, 157–162 CrossRef CAS.
- E. Kováts, Gas-chromatographische Charakterisierung organischer Verbindungen. Teil 1: Retentionsindices aliphatischer Halogenide, Alkohole, Aldehyde und Ketone, Helv. Chim. Acta, 1958, 41, 1915–1932 CrossRef.
- S. P. Verevkin, Vapour pressures and enthalpies of vaporization of a series of the linear n-alkyl-benzenes, J. Chem. Thermodyn., 2006, 38, 1111–1123 CrossRef CAS.
- V. P. Panchamiya, A. Kuchimanchi, K. G. Kulkarni and S. N. Havaldar, A review on phase change materials: Development, Types, and Applications, J. Phys.: Conf. Ser., 2023, 2426, 012033 CrossRef.
- Ö. F. Ensari and C. Alkan, 1,12-dodecanediol among similar fatty alcohols as a phase change material for thermal energy storage, Sol. Energy Adv., 2025, 5, 100079 CrossRef.
- V. R. Thalladi, R. Boese and H.-C. Weiss, The Melting Point Alternation inα,ω-Alkanediols andα,ω-Alkanediamines: Interplay between Hydrogen Bonding and Hydrophobic Interactions, Angew. Chem., Int. Ed., 2000, 39, 918–922 CrossRef CAS PubMed.
- P. Miller, The free energy of furfural and some of its derivatives, Iowa State Coll. J. Sci., 1936, 10, 91–93 CAS.
- H. Moureu and M. Dode, Heats of Formation of Ethylene Oxide, of Ethandiol and oof Several Homologs, Bull. Soc. Chim. Fr., 1937, 4, 637–647 CAS.
- G. Jung and J. Dahmlos, Thermochemische untersuchungen zur frage der drehbarkeit um C-C-bindunge, Z. Phys. Chem., 1942, 190, 230–240 Search PubMed.
- G. S. Parks, T. J. West, B. F. Naylor, P. S. Fujii and L. A. McClaine, Thermal Data on Organic Compounds. XXIII. Modern Combustion Data for Fourteen Hydrocarbons and Five Polyhydroxy Alcohols, J. Am. Chem. Soc., 1946, 68, 2524–2527 CrossRef CAS.
- L. A. McClaine, Thermodynamic data for some compounds containing carbon, hydrogen and oxygen, PhD thesis, Stanford Univ., 1947, pp. 1–57, (cited in https://webbook.nist.gov/chemistry/).
- N. V. Karyakin, Thermodynamics of Aromatic Heterochain and Heterocyclochain Polymers, NNovg. Gos. Univ., Nizh. Novgorod, 1998 Search PubMed.
- G. S. Parks and H. P. Mosher, Heats of Combustion and Formation of Seven Organic Compounds Containing Oxygen, J. Chem. Phys., 1962, 37, 919–920 CrossRef CAS.
- S. Sunner and M. Månsson, Combustion Calorimetry: Experimental Chemical Thermodynamics, Pergamon, New York, 1979 Search PubMed.
- H.-D. Beckhaus, C. Rüchardt and M. Smisek, Anwendung von kraftfeldrechnungen. VI. Verbrennungsenthalpie und bildungsenthalpie von 4-carbomethoxy-homocuban und homocuban-4-carbonsäure—ein testfall zur berechnung von bildungsenthalpien nach dem kraftfeldverfahren, Thermochim. Acta, 1984, 79, 149–159 CrossRef CAS.
- X. Li, Q.-G. Li, C.-H. Li, L.-F. Duan, J.-H. Jiang, F.-H. Zeng, X. Li and H.-W. Gu, Construction and performance evaluation of a CSC-1 type horizontal rotating micro-bomb combustion-solution isoperibol multifunctional calorimeter, J. Chem. Thermodyn., 2021, 160, 106505 CrossRef CAS.
- M. Sakiyama and T. Kiyobayashi, Micro-bomb combustion calorimeter equipped with an electric heater for aiding complete combustion, J. Chem. Thermodyn., 2000, 32, 269–279 CrossRef CAS.
- G. Hubbard, W. N. Scott and D. W. Waddington, Standard States and Corrections for Combustions in a Bomb at Constant Volume, in Experimental Thermochemistry, ed. F. D. Rossini, Interscience Publishers, New York, 1956, pp. 75–128 Search PubMed.
- J. D. Cox, D. D. Wagman and V. A. Medvedev, CODATA Key Values for Thermodynamics: Final Report of the CODATA Task Group on Key Values for Thermodynamics, CODATA Series on Thermodynamic, Hemisphere Publ. Corp., New York, 1989 Search PubMed.
- G. Olofsson, Assignment of Uncertainties, in Combustion Calorimetry: Experimental Chemical Thermodynamics, ed. S. Sunner and M. Månsson, Pergamon, New York, 1979, pp. 137–161 Search PubMed.
- M. Anteunis and Y. Rommelaere, NMR Experiments on Acetals: XXIX. The Ease of Acetonide Formation of Some Glycols, Bull. Soc. Chim. Belg., 1970, 79, 523–530 CrossRef.
- N. Cohen, Revised group additivity values for enthalpies of formation (at 298 K) of carbon-hydrogen and carbon-hydrogen-oxygen compounds, J. Phys. Chem. Ref. Data, 1996, 25, 1411–1481 CrossRef CAS.
- S. P. Verevkin, V. N. Emel’yanenko, V. Diky, C. D. Muzny, R. D. Chirico and M. Frenkel, New Group-Contribution Approach to Thermochemical Properties of Organic Compounds: Hydrocarbons and Oxygen-Containing Compounds, J. Phys. Chem. Ref. Data, 2013, 42, 033102 CrossRef.
- G. N. Roganov, P. N. Pisarev, V. N. Emel’yanenko and S. P. Verevkin, Measurement and prediction of thermochemical properties. Improved benson-type increments for the estimation of enthalpies of vaporization and Standard Enthalpies of Formation of Aliphatic Alcohols, J. Chem. Eng. Data, 2005, 50, 1114–1124 CrossRef CAS.
- NIST Chemistry WebBook. Available online: https://webbook.nist.gov/chemistry/ (accessed on 01.12.2025).
| This journal is © the Owner Societies 2026 |