 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Generation and benchmarking of a diverse reaction database of quantum mechanical liquid-phase activation Gibbs free energies
Lingfeng
Gui†
 a,
Alan
Armstrong
a,
Alan
Armstrong
 b,
Claire S.
Adjiman
b,
Claire S.
Adjiman
 a,
Fareed Bhasha
Sayyed
c and
Amparo
Galindo
a,
Fareed Bhasha
Sayyed
c and
Amparo
Galindo
 *a
*a
aDepartment of Chemical Engineering, The Sargent Centre for Process Systems Engineering, Imperial College London, London, SW7 2AZ, UK. E-mail: a.galindo@imperial.ac.uk
bDepartment of Chemistry, Imperial College London, Molecular Sciences Research Hub, White City Campus, London, W12 0BZ, UK
cSynthetic Molecule Design and Development, Eli Lilly Services India Pvt Ltd, Devarabeesanahalli, Bengaluru 560103, India
First published on 1st May 2026
Abstract
Many chemical reactions occur in the liquid phase, making the accurate prediction of the liquid-phase activation Gibbs free energy, Δ≠G°,L, crucial for numerous applications. Quantum mechanical (QM) methods with implicit solvation models offer a valuable route to Δ≠G°,L prediction, although they are computationally demanding at high levels of theory and for larger systems. Data-driven surrogate models can address this issue but require extensive training and test datasets. We present here the liquid phase reaction energy database (LiPRED-2026), a QM reaction database containing 4513 Δ≠G°,L values for 28 diverse chemical reactions computed in various solvents at 298.15 K. The reactions have been chosen for their sensitivity to solvent effects and the availability of experimental data. The SMD model is employed to calculate solvation contributions to Δ≠G°,L because it can be used to account for the effect of solvent on the geometries of the reactants and transition states and it is suitable for charged species. The database contains Δ≠G°,L obtained from seven calculation methods, including the thermodynamic cycle method, the direct method, and their variants. Using a subset of the database, a benchmarking study shows that the best methods achieve a mean absolute error of 2.89 kcal mol−1 in absolute Δ≠G°,L and 1.00 kcal mol−1 in relative Δ≠G°,L, respectively, with the lower error for the relative Δ≠G°,L being mainly attributable to error cancellation. The use of a higher level of theory to calculate Δ≠G°,L improves relative Δ≠G°,L values only, but not absolute ones. These results provide valuable insights into the choice of methods and levels of theory appropriate for calculating Δ≠G°,L, while the database can serve for training and testing surrogate models.
1 Introduction
Chemical reactions in the liquid phase hold particular significance in science and engineering. Most biological processes, from DNA replication, protein folding, to cellular respiration, occur in an aqueous environment.1 The production of many chemical products, such as pharmaceuticals, pesticides, dyes and polymers, also involves liquid-phase chemical reactions, typically in organic solvents. In this context, furthering our understanding of solvent effects on chemical reactions is of critical interest.2In modelling liquid-phase reactions, explicit solvation models and implicit solvation models can be used. In the former case, solvent molecules are introduced explicitly. From this perspective, solvent molecules and solute molecules are essentially treated in the same way, though different approximation/parameterisation schemes may be applied to the solute and the solvent molecules. Explicit solvation models are commonly used in classical molecular dynamics and Monte Carlo simulations where the intermolecular interactions are relatively cheap to evaluate. For example, given the importance of water in biological systems, various explicit water models, such as SPC, TIP3P, TIP4P and their other variations, have been developed.3,4 When it comes to more computationally expensive quantum mechanical (QM) modelling, which is particularly relevant for reaction kinetics, implicit solvation models are often adopted, where the solvent environment is represented as a continuum medium characterised by several parameters. The preference for implicit solvation models is often due to the fact that they avoid the introduction of extra degrees of freedom and provide an accurate description of the strong long-range electrostatic interactions.5 Popular implicit solvation models include the polarisable continuum model (PCM),6 the SMx series,7 the COSMO8,9 models, and their variations. There also exist hybrid methods of explicit and implicit solvation models,10,11 wherein one or more layers of solvent molecules are treated explicitly and the remaining solvent molecules are treated implicitly as a continuum.
A significant challenge with using QM methods to calculate the liquid-phase activation Gibbs free energy Δ≠G°,L is their substantial computational expense, especially at higher levels of theory. As a result, the availability of accurate and chemically diverse reaction databases is highly valuable. Such databases, whether derived from QM calculations or experimental measurements, provide essential reference data for benchmarking and validating kinetic models. In particular, ensuring the accuracy and relevance of Δ≠G°,L data is crucial for reliable reaction modeling. Thus, the generation, curation, and benchmarking of diverse reaction databases is beneficial in advancing data-driven approach to reaction prediction.
Many existing reaction databases that catalogue activation energies and reaction kinetics focus on gas-phase reactions predominantly.12–19 However, liquid-phase reactions are often of greater practical interest, which underscores the need for liquid-phase databases. Jorner et al.20 curated a dataset of 449 experimental liquid-phase rate constants for SNAr reactions from the literature, including a broad range of solvents, nucleophiles, and leaving groups. The corresponding Δ≠G°,L values, derived from the rate constants using the Eyring equation, range from 12.5 to 42.4 kcal mol−1. This dataset was used to train and validate a Gaussian process regression model, which achieved a MAE of 0.77 kcal mol−1 on an independent test set. Chung and Green21 presented an extensive dataset comprising over 8.3 million solvation free energies of activation, Δ≠ΔGsolv, and solvation enthalpies of activation, Δ≠ΔHsolv, computed at 298 K using COSMO-RS at the BP-TZVPD-FINE level for 28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 318 neutral reactions and 295 solvents. Δ≠ΔGsolv and Δ≠ΔHsolv are defined as the differences in solvation free energy and solvation enthalpy, respectively, between the transition state and the reactant(s). Δ≠ΔGsolv is particularly useful for calculating the liquid-phase rate constant relative to that in the gas phase or in a different solvent. Among the reactions evaluated, 26
318 neutral reactions and 295 solvents. Δ≠ΔGsolv and Δ≠ΔHsolv are defined as the differences in solvation free energy and solvation enthalpy, respectively, between the transition state and the reactant(s). Δ≠ΔGsolv is particularly useful for calculating the liquid-phase rate constant relative to that in the gas phase or in a different solvent. Among the reactions evaluated, 26![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 448 are unimolecular in both the forward and reverse directions, involving up to two products and generally characterised by high gas-phase energy barriers. Because most of these reactions are uncommon, they were only used to pre-train a machine learning model, which was subsequently fine-tuned using an additional set of 1870 more common reactions. This additional set was generated via the AutoTST framework22 for H-abstraction, H-migration and R-addition reactions. The resulting machine learning surrogate model for Δ≠ΔGsolv and Δ≠ΔHsolv takes only the molecular structures of the reactants and solvents as input and achieves a MAE of 0.68 kcal mol−1 in predicting relative Δ≠G°,L values between two phases on an experimental test set. These relative Δ≠G°,L values were obtained from 165 experimental relative rate constants curated in the earlier work of Chung and Green23 for 15 neutral closed-shell or free radical reactions and 49 solvents over a temperature range from 273 to 392 K.
448 are unimolecular in both the forward and reverse directions, involving up to two products and generally characterised by high gas-phase energy barriers. Because most of these reactions are uncommon, they were only used to pre-train a machine learning model, which was subsequently fine-tuned using an additional set of 1870 more common reactions. This additional set was generated via the AutoTST framework22 for H-abstraction, H-migration and R-addition reactions. The resulting machine learning surrogate model for Δ≠ΔGsolv and Δ≠ΔHsolv takes only the molecular structures of the reactants and solvents as input and achieves a MAE of 0.68 kcal mol−1 in predicting relative Δ≠G°,L values between two phases on an experimental test set. These relative Δ≠G°,L values were obtained from 165 experimental relative rate constants curated in the earlier work of Chung and Green23 for 15 neutral closed-shell or free radical reactions and 49 solvents over a temperature range from 273 to 392 K.
The Δ≠G°,L can be calculated by subtracting the liquid-phase Gibbs free energy of each reactant from that of the transition state. The calculation of Δ≠G°,Lvia a thermodynamic cycle (TC) may, however, be preferred if the ideal gas-phase activation Gibbs free energy, Δ≠G°,IG, and solvation free energy, ΔG°,solv, can be evaluated more accurately in a separate manner,24 although it has been argued that the TC method may not necessarily always be more accurate than the direct calculation of Δ≠G°,L.24,25 Ho and Ertem24 performed a benchmarking study over an extensive dataset comprised of 175 reaction Gibbs free energy changes and 83 activation free energies within liquid phase, which showed that the accuracies of the direct and TC methods are generally very similar to each other. For the activation Gibbs free energy, Δ≠G°,L, the mean absolute deviation (MAD) between the direct and the TC methods is about 0.24 kcal mol−1 for reactions involving neutral reactants and about 0.96 kcal mol−1 for reactions involving charged reactants, with the direct method generally agreeing better with experiments when charged reactants are involved. However, with the number of experimental Δ≠G°,L values (18 Diels Alder reactions and 13 SN2 reactions) used for comparison in their work, it is difficult to conclude which method is better in general.
Regardless of whether the direct method or the TC method is used, the choice of the level(s) of theory used along with these two methods is key in terms of the accuracy of the Δ≠G°,L calculation. When the solvation free energy is calculated explicitly as in the TC method, it is most appropriately obtained using levels of theory that are consistent with the parameterisation scheme of the solvation model in use. For example, the SMD solvation model used in the current work was parameterised using an average of several relatively low levels of theory in the original work,26 and it is unclear whether one of these levels of theory or the average would produce the best results for Δ≠G°,L. In addition, both the TC method and the direct method involve single-point calculations at a high level of theory, for which QM composite methods, such as G3MP227 and CBS-QB3,28,29 are suitable,24 but for which different options also exist. There is therefore a need to investigate further which method/level of theory combinations (MLoTC) are the most accurate in general and for a specific reaction type.
In the current work, we present the liquid phase reaction free energies database (LiPRED-2026). LiPRED-2026 contains both absolute and relative liquid-phase activation Gibbs free energies, Δ≠G°,L, for 28 diverse organic reactions computed in various solvents at 298.15 K using different MLoTCs. Instead of using reactions generated in silico, LiPRED-2026 encompasses reactions with well-documented solvent effects and mostly with experimental data available. The SMD model26 is chosen over COSMO-type approaches because it is parameterised extensively with experimental data to reproduce solvation Gibbs free energies of both neutral and charged species, and charged reactants are common in organic synthesis. Furthermore, SMD makes it possible to account for the effect of the solvent on the geometries of the reactants and transition state and therefore, on the reaction pathway. The Menschutkin reaction between pyridine and phenacyl bromide serves as the primary case for investigating solvent effects, and Δ≠G°,L is calculated in 531 solvents. A further 2812 calculations are carried our for different reaction–solvent combinations. A subset of reactions from the database is used to benchmark various MLoTCs against experimental Δ≠G°,L data from the literature, providing insights into the balance between computational cost and accuracy.
The remainder of this paper is organised as follows: in Section 2, the computational methods employed in this work are introduced, including the thermodynamic cycle method and its variations (Section 2.1), the direct method (Section 2.2) and the workflow for implementing these methods to calculate Δ≠G°,L (Section 2.3). For Section 3.1, an overview of the LiPRED-2026 database is provided, detailing the reactions (Section 3.1.1) and calculation schemes (Section 3.1.2) involved. In Section 3.2, the results of the benchmarking study of different MLoTCs are presented. Conclusions are presented in Section 4.
2 Methods
2.1 The thermodynamic cycle method
In the TC method, the liquid-phase activation Gibbs free energy Δ≠G°,L is decomposed according to the thermodynamic cycle in Fig. 1 and expressed as:24 | (1) |
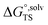 is the solvation free energy of the transition state,
is the solvation free energy of the transition state, 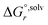 is the solvation free energy of reactant r, P0 is the reference pressure, D is the set of reactant(s) and νr is the stoichiometric coefficient of reactant r. The last term is the standard-state correction from the gas-phase standard state defined by T = 298.15 K and P0 = 1 atm to the solution-phase standard state of 1 mol L−1. Since all calculations in this work are performed using the Gaussian 16 software,30 the remainder of the paper will express the terms in eqn (1) in terms of quantities that can be computed and output by Gaussian 16. Definitions of the thermodynamic quantities reported by Gaussian can be found in the documentation by Ochterski.31 Δ≠G°,IG can be calculated as follows:
is the solvation free energy of reactant r, P0 is the reference pressure, D is the set of reactant(s) and νr is the stoichiometric coefficient of reactant r. The last term is the standard-state correction from the gas-phase standard state defined by T = 298.15 K and P0 = 1 atm to the solution-phase standard state of 1 mol L−1. Since all calculations in this work are performed using the Gaussian 16 software,30 the remainder of the paper will express the terms in eqn (1) in terms of quantities that can be computed and output by Gaussian 16. Definitions of the thermodynamic quantities reported by Gaussian can be found in the documentation by Ochterski.31 Δ≠G°,IG can be calculated as follows: | (2) |
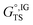 and
and  are the ideal gas Gibbs free energies of the transition state and reactant r, respectively, Eel,IGTS and Eel,IGr are the electronic energies of the transition state and reactants r, respectively, and Gtherm,IGTS and Gtherm,IGr are the thermal corrections to the free energies of the transition state and species r, respectively.
are the ideal gas Gibbs free energies of the transition state and reactant r, respectively, Eel,IGTS and Eel,IGr are the electronic energies of the transition state and reactants r, respectively, and Gtherm,IGTS and Gtherm,IGr are the thermal corrections to the free energies of the transition state and species r, respectively.
 | ||
| Fig. 1 Schematic representation of the thermodynamic cycle method for a multi-molecular reaction; Rr represents reactant r and TS is the transition state. | ||
The solvation free energies 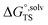 and
and  are calculated by applying the SMD solvation model26 as:
are calculated by applying the SMD solvation model26 as:
 | (3) |
The benefit of using the TC method is that one can calculate Δ≠G°,IG with a high level of theory and  with a level of theory that is consistent with the parameterisation scheme of the solvation model separately.25 Therefore, Δ≠G°,L can be expressed using the combination of different levels of theory as:
with a level of theory that is consistent with the parameterisation scheme of the solvation model separately.25 Therefore, Δ≠G°,L can be expressed using the combination of different levels of theory as:
 | (4) |
In eqn (4), the use of gas-phase thermal corrections is considered appropriate because any solvation-induced changes may already be implicitly incorporated into the “electronic energy” term through the SMD model parameterisation. Using explicit solution-phase thermal corrections may risk double counting, and the use of an ideal-gas partition function is not formally appropriate for solution-phase geometries and frequencies.34 However, Ribeiro et al. argued that, in the SMD parameterisation, the training set, which consists largely of small rigid molecules, was chosen such that solvation-induced structural changes are typically small compared with the intrinsic error of continuum models, rather than with the intention of folding these effects into the parameters.35 Therefore, when solvation-induced changes in geometry and/or vibrational frequencies are expected to be significant, accounting for them explicitly through solution-phase thermodynamic corrections can be justified and may improve accuracy. In later work, Ho and Ertem also noted that solvation-induced changes in thermal motion can be significant, and that explicitly including these contributions can in principle be more accurate, although the benefit is system-dependent and does not always guarantee improved agreement.24
Therefore, when this vibrational change is significant, the vibrational correction term Gtherm,Li,geom–Gtherm,IGi,geom should be added to the solvation free energy components in eqn (4) to account explicitly for the solvation-induced vibrational change. As a result, the Gtherm,IGi,geom term in eqn (4) cancels, leaving only Gtherm,Li,geom. Equivalently, Gtherm,IGi,geom is replaced by Gtherm,Li,geom, such that:
 | (5) |
The TC method with the vibrational correction (eqn (5)) is denoted as “TC-vib” in the remainder of the paper.
Another variation that can be made to the TC method (eqn (4)) is to use a “high” level of theory for the solvation free energy calculations of the TS and reactants. This method is referred to as the “TC-high” method, which can be expressed as:
 | (6) |
Given that the SMD model is parameterised using the following error function,26
 | (7) |
 is the experimental solvation free energy for data point J, ΔGEPj,J is the electrostatic contribution to the solvation free energy calculated at level of theory j for the data point J, GCDSJ is the non-electrostatic contribution to the solvation free energy for the data point J and ND is the number of data points (ND = 2489 in the work of Marenich et al.26). For maximum consistency with the model, the solvation free energy is calculated as the average of the results from the six levels of theory used for the SMD model parameterisation, i.e., M05-2X/MIDI!6D, M052X/6-31G(d), M05-2X/6-31+G(d,p), M05-2X/cc-pVTZ, B3LYP/6-31G(d), and HF/6-31G(d), as this strictly follows the parameterisation scheme of the SMD model. The TC method corrected with this approach is referred to as “TC-SMD” and is expressed as:
is the experimental solvation free energy for data point J, ΔGEPj,J is the electrostatic contribution to the solvation free energy calculated at level of theory j for the data point J, GCDSJ is the non-electrostatic contribution to the solvation free energy for the data point J and ND is the number of data points (ND = 2489 in the work of Marenich et al.26). For maximum consistency with the model, the solvation free energy is calculated as the average of the results from the six levels of theory used for the SMD model parameterisation, i.e., M05-2X/MIDI!6D, M052X/6-31G(d), M05-2X/6-31+G(d,p), M05-2X/cc-pVTZ, B3LYP/6-31G(d), and HF/6-31G(d), as this strictly follows the parameterisation scheme of the SMD model. The TC method corrected with this approach is referred to as “TC-SMD” and is expressed as: | (8) |
A final modification considered is to use the quasi-rigid rotor harmonic oscillator (quasi-RRHO) model to calculate the thermal corrections such that errors caused by low-lying vibrational frequencies in the harmonic approximation can be corrected.36 The method that uses quasi-RRHO is suffixed by “quasi” and expressed as:
 | (9) |
All variants of the TC method used in this work are summarised in Table 1.
| Method | Thermal correction | Solvation free energy |
|---|---|---|
| TC | G therm,IG i,geom | E el,L i,low–Eel,IGi,low |
| TC-vib | G therm,L i,geom | E el,L i,low–Eel,IGi,low |
| TC-SMD | G therm,IG i,geom | E el,L i,SMD–Eel,IGi,SMD |
| TC-high | G therm,IG i,geom | E el,L i,high–Eel,IGi,high |
| TC-quasi | G therm,IG,quasi i,geom | E el,L i,low–Eel,IGi,low |
| Direct | G therm,L i,geom | E el,L i,high–Eel,IGi,high |
| Direct-quasi | G therm,L,quasi i,geom | E el,L i,high–Eel,IGi,high |
2.2 The direct method
The direct method is equivalent to calculating the Gibbs free energy of each species i directly within the continuum solvation model. It can be simply expressed as: | (10) |
The direct method is equivalent to augmenting the TC method result with both the vibrational correction and the high LoT correction for solvation free energy. The standard state correction is also needed if the thermal corrections are obtained using the gas-phase standard state. The direct method using the quasi-RRHO model is then expressed as:
 | (11) |
It should be noted that although the direct method involves fewer constituent terms, it can be more computationally expensive than the TC method as the electronic energies need to be evaluated with the same (potentially high) level of theory in both the gas and liquid phases. The variants of the direct method considered in the current work are summarised in Table 1.
In this study, the term “method” refers exclusively to the variants of the TC method and the driect method, whereas “level of theory” denotes the quantum-mechanical levels used for the high, low, and geom subscripts. The combination of a method with specific levels of theory is abbreviated as “MLoTC”.
The Δ≠G°,L values calculated using eqn (1)–(11) are referred to as absolute Δ≠G°,L values, as they represent the kinetics of a reaction in a specific solvent. In practice, however, relative values are often preferred, because systematic errors associated with the calculations or measurements tend to cancel. The relative Δ≠G°,L values, are determined by subtracting the absolute Δ≠G°,L value in a reference solvent, Δ≠G°,ref, from that in the solvent of interest, Δ≠G°,L, or by subtracting the absolute Δ≠G°,L value for a reference substrate from that for the substrate of interest when comparing substrate effects. In the current work, the reference solvent or reaction is either chosen to be consistent with the reported experimental values or corresponds to the reaction with the lowest free energy in the relevant reaction type series.
2.3 Workflow for calculating Δ≠G°,L
Although Boltzmann averaging would offer a more rigorous treatment of conformational effects, its impact is expected to be limited for most reactions included in the present work, since the majority of species in the current version of the database are either rigid or only mildly flexible.38,39 Furthermore, Wittmann et al.40 reported in a benchmark study of solvation free energies and partition ratios for flexible solutes that Boltzmann-averaged results were very similar to those obtained using only the lowest-energy conformer, supporting the simplified approach adopted here. Further details on the conformer selection for each reaction are provided in the SI.
3 Results and discussion
3.1 Overview of the LiPRED database
The LiPRED database contains 4513 Δ≠G°,L values calculated for 28 reactions using the combination of 7 methods, 3 low levels of theory and 2 high levels of theory. We characterise each reaction with a unique reactant or a unique pair of reactants. All of the reactions included in the database have previously been investigated experimentally and have well-documented solvent effects (see the references cited in Section 3.1.1). The solvents included in the database range from those commonly used in chemical laboratories with available experimental descriptors, such as those in the Minnesota solvent descriptor database,42 to solvents constructed from predefined atom groups,43,44 as well as hypothetical solvents45 defined solely by descriptor values without corresponding molecular structures. The full list of reaction–solvent pairs considered is provided in the Excel sheet available in the Zenodo online repository.3.2 Benchmarking Δ≠G°,L calculations
Experimental Δ≠G°,L of the 46 reaction–solvent pairs in Fig. 3 are collected from literature. Both absolute and relative Δ≠G°,L values are compared in the current work. Experimental absolute Δ≠G°,L values have been reported only for the 3 SN2 reactions involving cyanide as the nucleophile, the 8 DA reactions and the 12 ANS reactions. For the remaining reactions in the set, only relative Δ≠G°,L values have been reported. To assess relative Δ≠G°,L values, the experimental dataset used in the current work is constructed by including the relative Δ≠G°,L values reported in the literature (9 SN2 reactions with chloride as the nucleophile and 14 Menschutkin reactions) and values that are calculated from absolute Δ≠G°,L values by subtracting the smallest experimental absolute Δ≠G°,L values as a reference value for each corresponding reaction type (3 SN2 reactions with cyanide as the nucleophile, 8 DA reactions and 12 ANS reactions).The mean absolute error (MAE), is used as the performance indicator in the current work and is calculated for each MLoTC considered by comparing each computed liquid-phase activation free energy, Δ≠G°,L,cal, to its corresponding experimental liquid-phase activation free energies, Δ≠G°,L,exp:
 | (12) |
To ensure that the MAEs obtained in the benchmarking study are meaningful, we inspected (1) the uncertainties in the experimental data for the reactions in the benchmarking set, where available, and (2) the extent of solvent-induced variation in the experimental Δ≠G°,L values, so that any conclusions drawn from the benchmarking study are not limited by uncertainty or insufficient variation in the experimental data. For example, for the ANS reactions, the reported uncertainty in the rate constants is less than 2 to 3% across all solvents, while the uncertainty in Δ≠G°,L is reported to be around 0.7 kcal mol−1.47 For the DA reactions in water, the reported uncertainties in the rate constants are within 6%,55,56 which corresponds, by the Eyring equation, to an estimated uncertainty of about 0.04 kcal mol−1. Thus, where experimental uncertainties are reported, they are clearly smaller than the MAEs obtained in the benchmarking study (see Fig. 7). Regarding the solvent-induced variation in the experimental Δ≠G°,L values, the relative activation free energies span approximately 0 to 9 kcal mol−1 for the SN2 reactions, 0 to 11 kcal mol−1 for the DA reactions, and about −2 to 4 kcal mol−1 for the Menschutkin reactions. These ranges are substantially larger than the MAEs obtained for both the relative and absolute activation free energies, indicating that the solvent effect is significant compared with the prediction error. Only for the ANS reactions is the solvent-dependent range relatively small, at approximately 0 to 2 kcal mol−1. These solvent-dependent variations are investigated further in Fig. 9.
The results for absolute Δ≠G°,L values are summarised in Fig. 7. It can be seen from the comparison of the TC method and the TC-vib method that the addition of the vibrational correction reduces the MAE slightly for all the levels of theory considered, while the use of the TC-high method results in an improvement of the performance of the TC methods only when using the M062X functional. Combining the vibrational correction with the TC-high method, i.e., using the direct method, the use of the M062X funtional further improves the MAE, while the direct methods with levels of theory other than M062X once again perform slightly worse than the original TC methods. Using the average of six levels of theory does not improve the performance even though this choice is most consistent with the parameterisation scheme of the SMD solvation model. Given that Δ≠G°,L is a much more complex quantity than the solvation free energies used to parameterise the SMD model, the performance of the TC-SMD model in predicting Δ≠G°,L does not depend only on being consistent with the SMD parameterisation. For example, the reactions considered here involve species, particularly transition states, that are not represented in the training set. Therefore, it is not surprising that the TC-SMD method, despite being the most consistent with the original SMD parameterisation, does not necessarily lead to improved predictions of Δ≠G°,L.
In general, using the quasi-RRHO model does not improve model performance, and slightly degrades the performance of the TC method, in which the use of the quasi-RRHO model affects only Δ≠G°,IG. Given the presence of empirical components, i.e., the solvation free energies, which are known to contribute a significant portion of the overall error, any benefit provided by quasi-RRHO may be diminished in this case. Further investigation of this aspect could include testing alternative entropy treatments such as 1D-HR57 on a small subset, but is beyond the scope of the present benchmark study.
Focusing on the choice of the high level of theory, the use of G3MP2 consistently provides better performance than that of CBS-QB3. Overall, the direct method, in conjunction with the M062X/G3MP2 level of theory, yields the smallest MAE (2.89 kcal mol−1) for absolute Δ≠G°,L predictions. This is consistent with the conclusion of Ho and Ertem24 that the direct method outperforms the TC method when the transition state is ionic, given that most of the reactions evaluated involve transition states that are either ionic or exhibit a strong ionic character (except for the DA reactions). For the DA reactions, the MAEs achieved using the TC method and the direct method are 1.58 and 1.68 kcal mol−1, respectively, when using M062X/G3MP2. This performance is also comparable to that reported by Chung and Green,23 who evaluated only neutral closed-shell or free radical reactions and obtained an MAE of 1.30 kcal mol−1 with their best level of theory combination: ωB97XD/def2-TZVP for the gas-phase activation free energy and BP-TZVP-G16 for the COSMO-RS solvation evaluation.
At this point, it is useful to note that the MAEs from most of the MLoTCs are above 3 kcal mol−1 and they may be subject to significant systematic error. If the systematic error exists, its impact can be alleviated by calculating relative Δ≠G°,L values, as is commonly done.21,23
The performance of each MLoTC for relative Δ≠G°,L prediction is shown in Fig. 8. Overall, it can be seen that the MAEs for the relative Δ≠G°,L values are 2 kcal mol−1 smaller than that for corresponding absolute Δ≠G°,L, indicating a significant portion of the systematic error is eliminated. In contrast to the case of absolute Δ≠G°,L, the inclusion of the vibrational correction leads to an increase in the MAE for all the levels of theory considered. Similarly, the use of the TC-high method either makes no difference or increases the MAE compared to the TC method. The direct method performs worse than the TC method for all the levels of theory. In principle, the addition of the vibrational correction is expected to be most useful for more flexible systems where solvation-induced changes in vibrational contributions are larger, whereas for predominantly rigid solutes it can lead to a deterioration of the predictions due to the potential double counting of thermal solvation effects already embedded in the parameterisation. In such cases, the standard TC approximation can be a more consistent choice given the underlying solvation model. In addition, similar to the case of absolute Δ≠G°,L predictions, using neither the average of six levels of theory nor the quasi-RRHO model lead to a decrease in the MAE. Overall, CBS-QB3 and G3MP2 provide similar performance. For relative Δ≠G°,L, the MLoTCs affording the smallest MAEs are the TC method + M062X/CBS-QB3 (1.00 kcal mol−1) and TC-high method + M062X/G3MP2 (1.01 kcal mol−1), followed by TC method + M062X/G3MP2 (1.02 kcal mol−1). The MAEs for relative Δ≠G°,L reported by Chung and Green23 are similar across all the levels of theory combinations they evaluated, approximately 0.4 kcal mol−1, which is comparable to the best MAE we obtain here for the DA reaction using TC-vib + M062X/G3MP2 (0.44 kcal mol−1).
The performance of two well-performing MLoTCs, the TC method + M062X/G3MP2 and the direct method + M062X/G3MP2, is further evaluated via the parity plots shown in Fig. 9. In Fig. 9a and c, overall, the direct method yields a higher R2 value of 0.308, compared to 0.250 from the TC method, for the prediction of absolute Δ≠G°,L. Significant systematic deviations are observed in the predicted absolute Δ≠G°,L values for the SN2 reactions and the ANS reactions. The calculated Δ≠G°,L values corresponding to the DA reactions are closest to the experiments. The errors of the TC method can be decomposed into contributions from the gas-phase activation free energies and the solvation free energies. The composite methods employed in this work, such as G3MP2 and CBS-QB3, have been shown in benchmark studies to be accurate for gas-phase reaction barriers.58,59 Therefore, the dominant source of error is more likely to lie in the solvation models. This is supported by the work of Ho25 on the calculation of pKa values and reduction potentials using the TC method with the SMD model, which showed that increasing the accuracy of the electronic structure method does not necessarily improve agreement with experiment. As noted in the description of the SMD parameterisation, the SMD model was parameterised using a dataset that is dominated by neutral solutes, with a smaller fraction of ionic solvation free energies. This imbalance generally leads to better performance for neutral systems, with reported MAEs of 0.6 to 1.0 kcal mol−1 and 4 kcal mol−1 for the solvation free energies of neutral and ionic species, respectively.26 Given the potential propagation of errors from the various solvation free energy terms entering the calculation of Δ≠G°,L, deviations of up to 7 kcal mol−1 (Fig. 10a) for the SN2 reactions, in which both the reactants and the TS involve ionic species, are not abnormal. The slightly better performance of the direct method may arise because species with strong ionic character are subject to larger solvation-induced geometrical and vibrational changes, which are more accurately described by the direct method. This reaction-type dependence indicates that applying SMD to reactions involving charged and highly polar species is more challenging, and leading to the larger errors observed in the absolute free energies of activation of ionic reactions.
Nevertheless, we note that SMD can provide reasonably good performance in the predictions of relative Δ≠G°,L values for ionic reactions, as shown in the parity plots in Fig. 9b and d. In this case, the impact of systematic errors is reduced for both the TC and direct methods (as indicated by the distribution of the data points along the y = x line). The overall trends are predicted well across different types of reactions and solvents, although we note that the R2 value for the direct method (0.830) is slight higher than that for the TC method (0.815), in contrast to the MAE values (1.17 kcal mol−1vs. 1.02 kcal mol−1), suggesting that both methods perform comparably.
We further compare the performance of these two MLoTCs against their counterparts that utilise a low level of theory, M062X/6-31+G(d), in place of the high level of theory calculations, i.e., the gas-phase G3MP2 calculations used in the TC method and the gas- and liquid-phase G3MP2 calculations in the direct method (Fig. 10). Interestingly, employing a high level of theory results in a larger error in the absolute liquid phase activation Gibbs free energies (Fig. 10a) but a smaller error in the relative values (Fig. 10b). This suggests that high levels of theory introduce more systematic error in the resulting activation free energy. This could potentially be corrected through calibration against a large experimental dataset. However, given the marginal improvements observed when using high levels of theory with the SMD model, employing a low level of theory may offer better balance between computational cost and accuracy.
4 Conclusions
In summary, we have introduced the LiPRED-2026 database, comprising 4513 liquid-phase activation Gibbs free energy values calculated for 28 reactions in a range of solvents using the SMD model and seven computational schemes with a range of levels of theory. A subset of the database has been employed to benchmark the accuracy of various MLoTCs in the prediction of both absolute and relative activation free energies. All methods considered perform well for relative activation free energies (or reaction rates). The TC method, in which Δ≠G°,IG and solvation energy are calculated separately, paired with the M062X/CBS-QB3 level of theory provides the best performance in therms of relative Δ≠G°,L, with an MAE of 1.00 kcal mol−1. The combination of the direct method, in which all the calculations are performed in a liquid phase, with the M062X/G3MP2 level of theory has been found to be most effective for predicting absolute activation Gibbs free energies, albeit with an MAE of 2.89 kcal mol−1. The smaller MAE of relative Δ≠G°,L is mainly due to the cancellation of systematic error in absolute Δ≠G°,L. The TC-high method in conjunction with M062X/G3MP2 strikes a good balance between absolute (MAE: 2.99 kcal mol−1) and relative (MAE: 1.01 kcal mol−1) Gibbs free energy predictions. We have also investigated the need to use a high level of theory in both the TC and direct methods. While this improves the accuracy of relative energy predictions, it tends to result in larger errors in absolute energies. Therefore, when comparisons between different solvents are not critical and computational resources are limited, a low level of theory may be sufficient for general applications.The current version of LiPRED-2026 can provide valuable training and test data for developing data-driven models that do not require large amounts of data, such as a linear free energy relationship or a small-scale feedforward neural network model, to predict the kinetics of common, practically significant organic reactions. Such data-lean models have previously been found to be highly effective in solvent selection.45,51 For complex machine learning architectures, particularly those based on deep learning, LiPRED-2026 can support fine-tuning21 and benchmarking, similar to the roles of the fine-tuning and test sets in the work of Chung and Green,21 where the amount of data required is much smaller than that used for pretraining the model.
However, we acknowledge several limitations of the current LiPRED-2026 database that will be addressed in future expansions. First, a single conformer has been considered for each species in a given chemical environment, either in vacuum or in a specific solvent. Although solvation-induced geometric relaxation is captured through the SMD continuum solvation model in methods that include vibrational corrections, such as TC-vib and the direct method, potential solvation-induced changes in conformer populations have not been taken into account in the current work. This may lead to errors for systems with substantial conformational flexibility, for which Boltzmann averaging in both the gas and liquid phases would offer a more rigorous treatment.
Second, although LiPRED-2026 contains 4513 Δ≠G°,L values, the reaction set remains relatively limited. In its current form, LiPRED-2026 is intended mainly as a benchmark and training resource for Δ≠G°,L in the reaction classes currently represented. The 28 reactions currently included do not cover several common reaction classes, such as elimination and radical reactions. Despite its relatively narrow focus, the outcomes of our benchmarking study are consistent with general expectations and provide useful insights on model performance. This motivates future work to extend the benchmarking to other solvation models and to expand substantially both the number and the diversity of reactions included in the database. This latter goal will require the generation of larger sets of reliable experimental data for both absolute and relative Δ≠G°,L values.
Author contributions
Lingfeng Gui: conceptualisation, methodology, software, testing, investigation, data curation, writing – original draft, visualisation. Alan Armstrong: conceptualisation, methodology, writing – review & editing, supervision, funding acquisition. Claire S. Adjiman: conceptualisation, methodology, testing, writing – review & editing, supervision, project administration, funding acquisition. Fareed Bhasha Sayyed: conceptualisation, writing – review & editing, supervision. Amparo Galindo: conceptualisation, methodology, testing, writing – review & editing, supervision, project administration, funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
Data underlying this article and not available in the references cited are provided in the Zenodo repository (https://doi.org/10.5281/zenodo.8396100) and in the supplementary information (SI) under a CC BY licence. The Zenodo repository includes the complete quantum mechanical data generated for the liquid-phase activation free energy calculations reported in this work, provided as an Excel file in the “chapter5” folder of the repository. Supplementary information: details of the conformer selection procedure used in this work. See DOI: https://doi.org/10.1039/d6cp00088f.Acknowledgements
Funding from Eli Lilly and Company and the UK EPSRC, through the PharmaSEL-Prosperity Programme (EP/T005556/1), is gratefully acknowledged. AG acknowledges the funding of a Research Chair by the Royal Academy of Engineering and Eli Lilly and Company (RCSRF1819/7/33). The Imperial College Research Computing Service (DOI: https://doi.org/10.14469/hpc/2232) is also gratefully acknowledged for providing support and resources for the quantum mechanical calculations. The authors thank Dr S. P. Kolis (Eli Lilly and Company) for helpful discussions.Notes and references
- J. L. Finney and A. K. Soper, Chem. Soc. Rev., 1994, 23, 1–10 RSC.
- M. Orozco and F. J. Luque, Chem. Rev., 2000, 100, 4187–4226 CrossRef CAS PubMed.
- W. L. Jorgensen, J. Chandrasekhar, J. D. Madura, R. W. Impey and M. L. Klein, J. Chem. Phys., 1983, 79, 926–935 CrossRef CAS.
- P. Mark and L. Nilsson, J. Phys. Chem. A, 2001, 105, 9954–9960 CrossRef CAS.
- C. J. Cramer and D. G. Truhlar, Chem. Rev., 1999, 99, 2161–2200 CrossRef CAS PubMed.
- S. Miertus, E. Scrocco and J. Tomasi, Chem. Phys., 1981, 55, 117–129 CrossRef CAS.
- C. J. Cramer and D. G. Truhlar, Acc. Chem. Res., 2008, 41, 760–768 CrossRef CAS PubMed.
- V. Barone and M. Cossi, J. Phys. Chem. A, 1998, 102, 1995–2001 CrossRef CAS.
- A. Klamt, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2011, 1, 699–709 CAS.
- G. Brancato, N. Rega and V. Barone, J. Chem. Phys., 2008, 128, 144501 CrossRef PubMed.
- M. S. Lee, F. R. Salsbury Jr. and M. A. Olson, J. Comput. Chem., 2004, 25, 1967–1978 Search PubMed.
- J. A. Manion, R. E. Huie, R. D. Levin, D. R. Burgess Jr., V. L. Orkin, W. Tsang, W. S. McGivern, J. W. Hudgens, V. D. Knyazev, D. B. Atkinson, E. Chai, A. M. Tereza, C.-Y. Lin, T. C. Allison, W. G. Mallard, F. Westley, J. T. Herron, R. F. Hampson and D. H. Frizzell, NIST Chemical Kinetics Database, NIST Standard Reference Database 17, Version 7.0 (Web Version), Release 1.6.8, Data version 2015.09, 2015, https://kinetics.nist.gov.
- M. R. McGillen, W. P. L. Carter, A. Mellouki, J. J. Orlando, B. Picquet-Varrault and T. J. Wallington, Earth Syst. Sci. Data, 2020, 12, 1203–1216 CrossRef.
- Q. Zhao, S. M. Vaddadi, M. Woulfe, L. A. Ogunfowora, S. S. Garimella, O. Isayev and B. M. Savoie, Sci. Data, 2023, 10, 145 CrossRef CAS PubMed.
- G. F. von Rudorff, S. N. Heinen, M. Bragato and O. A. von Lilienfeld, Mach. Learn.: Sci. Technol., 2020, 1, 045026 Search PubMed.
- C. A. Grambow, L. Pattanaik and W. H. Green, J. Phys. Chem. Lett., 2020, 11, 2992–2997 CrossRef CAS PubMed.
- C. A. Grambow, L. Pattanaik and W. H. Green, Sci. Data, 2020, 7, 137 CrossRef CAS PubMed.
- K. Spiekermann, L. Pattanaik and W. H. Green, Sci. Data, 2022, 9, 417 CrossRef CAS PubMed.
- V. K. Prasad, Z. Pei, S. Edelmann, A. Otero-de-la Roza and G. A. DiLabio, J. Chem. Theory Comput., 2022, 18, 151–166 CrossRef CAS PubMed.
- K. Jorner, T. Brinck, P.-O. Norrby and D. Buttar, Chem. Sci., 2021, 12, 1163–1175 RSC.
- Y. Chung and W. H. Green, Chem. Sci., 2024, 15, 2410–2424 RSC.
- N. Harms, C. Underkoffler and R. West, ChemRxiv, 2020, preprint DOI:10.26434/chemrxiv.13277870.v2.
- Y. Chung and W. H. Green, J. Phys. Chem. A, 2023, 127, 5637–5651 CrossRef CAS PubMed.
- J. Ho and M. Z. Ertem, J. Phys. Chem. B, 2016, 120, 1319–1329 CrossRef CAS PubMed.
- J. Ho, Phys. Chem. Chem. Phys., 2015, 17, 2859–2868 RSC.
- A. V. Marenich, C. J. Cramer and D. G. Truhlar, J. Phys. Chem. B, 2009, 113, 6378–6396 CrossRef CAS PubMed.
- L. A. Curtiss, P. C. Redfern, K. Raghavachari, V. Rassolov and J. A. Pople, J. Chem. Phys., 1999, 110, 4703–4709 CrossRef CAS.
- J. A. Montgomery, Jr., M. J. Frisch, J. W. Ochterski and G. A. Petersson, J. Chem. Phys., 1999, 110, 2822–2827 CrossRef.
- J. A. Montgomery, Jr., M. J. Frisch, J. W. Ochterski and G. A. Petersson, J. Chem. Phys., 2000, 112, 6532–6542 CrossRef.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman and D. J. Fox, Gaussian∼16 Revision C.01, Gaussian Inc., Wallingford CT, 2016 Search PubMed.
- J. W. Ochterski, Thermochemistry in Gaussian, Gaussian, inc. technical report, 2000 Search PubMed.
- K. A. Peterson, D. Feller and D. A. Dixon, Theor. Chem. Acc., 2012, 131, 1079 Search PubMed.
- T. Froitzheim, M. Müller, A. Hansen and S. Grimme, ChemRxiv, 2025, preprint DOI:10.26434/chemrxiv-2025-bjxvt.
- J. Ho, A. Klamt and M. L. Coote, J. Phys. Chem. A, 2010, 114, 13442–13444 CrossRef CAS PubMed.
- R. F. Ribeiro, A. V. Marenich, C. J. Cramer and D. G. Truhlar, J. Phys. Chem. B, 2011, 115, 14556–14562 CrossRef CAS PubMed.
- S. Grimme, Chem. – Eur. J., 2012, 18, 9955–9964 CrossRef CAS PubMed.
- T. A. Halgren, J. Comput. Chem., 1996, 17, 490–519 CrossRef CAS.
- S. Grimme, F. Bohle, A. Hansen, P. Pracht, S. Spicher and M. Stahn, J. Phys. Chem. A, 2021, 125, 4039–4054 CrossRef CAS PubMed.
- J. Gorges, S. Grimme, A. Hansen and P. Pracht, Phys. Chem. Chem. Phys., 2022, 24, 12249–12259 RSC.
- L. Wittmann, C. E. Selzer and S. Grimme, Chem. Sci., 2025, 16, 22976–22995 RSC.
- G. Luchini, J. Alegre-Requena, I. Funes-Ardoiz and R. Paton, F1000Research, 2020, 9, 1–14 Search PubMed.
- P. Winget, D. M. Dolney, D. J. Giesen, C. J. Cramer and D. G. Truhlar, Minnesota Solvent Descriptor Database, 2021, https://comp.chem.umn.edu/solvation/mnsddb.pdf Search PubMed.
- E. Grant, Y. Pan, J. Richardson, J. R. Martinelli, A. Armstrong, A. Galindo and C. S. Adjiman, 13th International Symposium on Process Systems Engineering (PSE 2018), Elsevier, 2018, vol. 44, pp. 2437–2442.
- L. Gui, Y. Yu, T. O. Oliyide, E. Siougkrou, A. Armstrong, A. Galindo, F. B. Sayyed, S. P. Kolis and C. S. Adjiman, Comput. Chem. Eng., 2023, 177, 108345 CrossRef CAS.
- L. Gui, A. Armstrong, A. Galindo, F. B. Sayyed, S. P. Kolis and C. S. Adjiman, Mol. Syst. Des. Eng., 2024, 9, 1254–1274 RSC.
- S.-Y. Tang, J. Shi and Q.-X. Guo, Org. Biomol. Chem., 2012, 10, 2673–2682 RSC.
- P. M. E. Mancini, R. D. Martinez, L. R. Vottero and N. S. Nudelman, J. Chem. Soc., Perkin Trans. 2, 1984, 1133–1138 RSC.
- A. D. McFarland, J. Y. Buser, M. C. Embry, C. B. Held and S. P. Kolis, Org. Process Res. Dev., 2019, 23, 2099–2105 CrossRef CAS.
- L. Gui, C. S. Adjiman, A. Galindo, F. B. Sayyed, S. P. Kolis and A. Armstrong, Ind. Eng. Chem. Res., 2023, 62, 874–880 CrossRef CAS.
- M. H. Abraham, J. Chem. Soc. D, 1969, 1307–1308 RSC.
- H. Struebing, Z. Ganase, P. G. Karamertzanis, E. Siougkrou, P. Haycock, P. M. Piccione, A. Armstrong, A. Galindo and C. S. Adjiman, Nat. Chem., 2013, 5, 952–957 CrossRef CAS PubMed.
- A. Diamanti, Z. Ganase, E. Grant, A. Armstrong, P. M. Piccione, A. M. Rea, J. Richardson, A. Galindo and C. S. Adjiman, React. Chem. Eng., 2021, 6, 1195–1211 RSC.
- T. J. Sheldon, C. S. Adjiman and J. Cordiner, Fluid Phase Equilib., 2005, 231, 27–37 CrossRef CAS.
- P. R. Rablen, B. D. McLarney, B. J. Karlow and J. E. Schneider, J. Org. Chem., 2014, 79, 867–879 CrossRef CAS PubMed.
- A. Meijer, S. Otto and J. B. F. N. Engberts, J. Org. Chem., 1998, 63, 8989–8994 CrossRef CAS.
- T. Rispens and J. B. F. N. Engberts, J. Org. Chem., 2002, 67, 7369–7377 CrossRef CAS PubMed.
- J. Pfaendtner, X. Yu and L. J. Broadbelt, Theor. Chem. Acc., 2007, 118, 881–898 Search PubMed.
- Y. Lan, L. Zou, Y. Cao and K. N. Houk, J. Phys. Chem. A, 2011, 115, 13906–13920 Search PubMed.
- L. A. Curtiss, P. C. Redfern and K. Raghavachari, Chem. Phys. Lett., 2010, 499, 168–172 CrossRef CAS.
Footnote |
| † Present address: School of Engineering and Physical Sciences, Heriot-Watt University, Edinburgh, EH14 4AS, Scotland, UK. |
| This journal is © the Owner Societies 2026 |









