The primary near-UV photochemistry of aqueous pyruvic acid
Jan
Thøgersen
 ,
Tobias
Weidner
,
Tobias
Weidner
 and
Frank
Jensen
and
Frank
Jensen
 *
*
Department of Chemistry, Aarhus University, Langelandsgade 140, Aarhus C DK-8000, Denmark. E-mail: frj@chem.au.dk
First published on 5th March 2026
Abstract
Near-UV photolysis of pyruvic acid results in decarboxylation within 0.8 ps. In contrast, pyruvate anions excited by near-UV light relax back to the ground state within 50 ps with no detectable photolysis. The sharply different behavior of the acid and its conjugate base provides a mechanistic explanation for the puzzling pH dependence of the pyruvic acid photolysis.
The photochemistry of pyruvic acid and its conjugated base, pyruvate, is potentially important to the climate as it is estimated that plants and burning of biomass emit 0.85 Tg of pyruvic acid to the atmosphere every year, primarily in the aqueous phase.1 Owing to proliferation, pyruvic acid is omnipresent in aqueous environments, from atmospheric microdroplets to ponds, lakes and oceans.2 The acid constant of pyruvic acid is pKa = 2.5 in bulk water and has been measured to be as low as pKa = 0.7 at water–air interfaces,3,4 where the deprotonated COO− and C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups tilt forward towards bulk.5 Hence, in most aqueous environments pyruvic acid is partly deprotonated with some of the molecules taking the form of pyruvate. Pyruvic acid and pyruvate are readily photolyzed by deep UV light via a strong π* ← π transition, but the ozone layer prevents such light from reaching the biosphere. However, due to a weak π* ← n transition, the two species can be excited by near-UV sunlight. Considering the abundance of pyruvic acid, its near-UV photochemistry is potentially an important source of photoproducts with relevance to the biosphere. Decades of research have led to a general understanding of the photochemistry of aqueous pyruvic acid.6–18 However, important parts of its initial photochemistry are still uncertain. Among the open questions, two stand out: what are the primary photoreactions of aqueous pyruvic acid and why does the yield of the photoproducts decrease with increasing pH?3,8 With these questions unanswered, the well-developed understanding of aqueous pyruvic acid's photochemistry rests on an unsure foundation and reliable insight is needed to improve the predictive accuracy of climate models.
O groups tilt forward towards bulk.5 Hence, in most aqueous environments pyruvic acid is partly deprotonated with some of the molecules taking the form of pyruvate. Pyruvic acid and pyruvate are readily photolyzed by deep UV light via a strong π* ← π transition, but the ozone layer prevents such light from reaching the biosphere. However, due to a weak π* ← n transition, the two species can be excited by near-UV sunlight. Considering the abundance of pyruvic acid, its near-UV photochemistry is potentially an important source of photoproducts with relevance to the biosphere. Decades of research have led to a general understanding of the photochemistry of aqueous pyruvic acid.6–18 However, important parts of its initial photochemistry are still uncertain. Among the open questions, two stand out: what are the primary photoreactions of aqueous pyruvic acid and why does the yield of the photoproducts decrease with increasing pH?3,8 With these questions unanswered, the well-developed understanding of aqueous pyruvic acid's photochemistry rests on an unsure foundation and reliable insight is needed to improve the predictive accuracy of climate models.
In the present work, we use UV pump-IR probe transient absorption spectroscopy with sub-picosecond time resolution assisted by density functional theory to determine the primary reaction dynamics when pyruvic acid is excited by near-UV light (see SI for methods, molecular structures and natural transition orbitals). The results explain the pH dependence and identify the primary photolysis products and thus form a solid foundation for understanding subsequent reactions.
Result and discussion
Fig. 1a shows a model of pyruvic acid and its UV steady-state absorption spectrum. To a good approximation the spectrum consists of two contributions, a localized carbonyl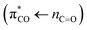 chromophore with acetone as a model responsible for the near-UV absorption, and a localized carboxyl group
chromophore with acetone as a model responsible for the near-UV absorption, and a localized carboxyl group 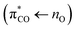 chromophore with propanoic acid as a model, responsible for the deep-UV absorption. In our experiments the carbonyl chromophore of pyruvic acid is excited at 340 nm. We use an aqueous pyruvic acid sample with a concentration of 0.2 M and pH = 1.5. At these conditions ∼15% of the pyruvic acid molecules are deprotonated to form pyruvate. The remaining pyruvic acid molecules are reversibly hydrated to their geminal-diol.18 The extent of hydration depends on pH. Since only the keto form is responsible for the near-UV absorption, this partly explains why the photolysis of pyruvic acid is pH dependent as has been proposed by Rapf et al.3
chromophore with propanoic acid as a model, responsible for the deep-UV absorption. In our experiments the carbonyl chromophore of pyruvic acid is excited at 340 nm. We use an aqueous pyruvic acid sample with a concentration of 0.2 M and pH = 1.5. At these conditions ∼15% of the pyruvic acid molecules are deprotonated to form pyruvate. The remaining pyruvic acid molecules are reversibly hydrated to their geminal-diol.18 The extent of hydration depends on pH. Since only the keto form is responsible for the near-UV absorption, this partly explains why the photolysis of pyruvic acid is pH dependent as has been proposed by Rapf et al.3
Pyruvic acid and pyruvate have comparable near-UV absorption spectra,20 and both species are therefore excited by 340 nm light. However, the infrared absorption spectra of pyruvic acid and pyruvate, presented in Fig. 1b, are markedly different and enable unambiguous identification of the two species. Moreover, we recently reported the primary photochemistry of aqueous pyruvate upon excitation at 340 nm.20 The study showed that all the excited pyruvate anions return to the ground state within 50 ps implying that the photolysis quantum yield is insignificant. Consequently, any photoproducts lasting more than 50 ps originate exclusively from the photolysis of pyruvic acid.
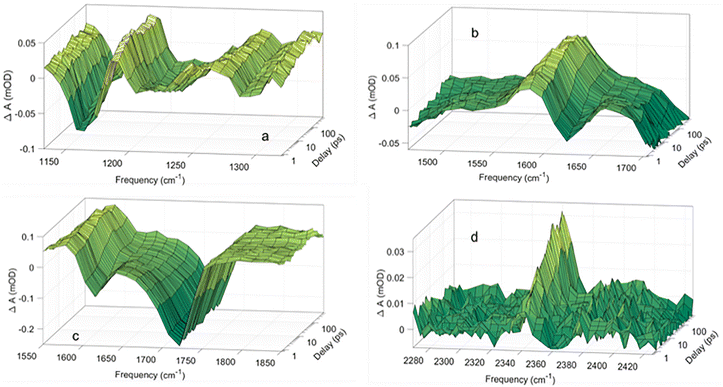
The transient infrared absorption induced by the excitation of pyruvic acid at 340 nm is recorded in the range between 950 cm−1 and 2500 cm−1. The photochemistry of pyruvic acid and pyruvate can be deduced from the segments of the transient absorption spectrum presented in Fig. 2. The full transient absorption spectrum is shown in Fig. SI2. Negative ΔA values imply the photodepletion of ground state pyruvic acid or pyruvate induced by the 340 nm excitation. Positive ΔA values indicate electronically or vibrationally excited states of pyruvic acid or pyruvate molecules or the formation of intermediate or permanent photoproducts.
The negative absorption at 1722 cm−1 is associated with the carboxylic carbonyl stretch of ground state pyruvic acid. Fig. 2(c) shows that the absorption at 1722 cm−1 drops to a minimum immediately after the excitation at t = 0 and stays at this level during the subsequent 560 ps with no sign of recovery.
In Fig. 2, the same absorption dynamics is observed for every vibrational transition exclusively related to pyruvic acid. More precisely, 96 ± 5% of the excited pyruvic acid molecules have not returned to the vibrationally relaxed ground state of pyruvic acid after 560 ps—either because they dissociate or because they populate long-lived excited states of pyruvic acid. The negative absorption at 1615 cm−1 pertains to the asymmetric carboxylate stretch of pyruvate. It too drops to a minimum right after the excitation, but in contrast to pyruvic acid, the absorption pertaining to pyruvate fully recovers in 50 ps. Fig. 2 shows that the absorption dynamics of all vibrations associated with pyruvate display the same behaviour. Accordingly, all excited pyruvate anions return to the ground state within this time frame, thus confirming that the photolysis quantum yield of aqueous pyruvate is insignificant, as reported earlier.20 The very different dissociation quantum yields of pyruvic acid and pyruvate suggests that the pH dependence of pyruvic acid's photochemistry reflects the extent of deprotonation: at low pH pyruvic acid is fully protonated, and the photolysis yield is significant, while the photolysis yield decreases to zero with increasing pH as pyruvic acid deprotonates to pyruvate.
The dissociation of pyruvic acid is confirmed by the transient absorption associated with the asymmetric stretch of carbon dioxide observed at 2343 cm−1 in Fig. 2d.21–24 The absorption from carbon dioxide rises from a value close to zero after 0.8 ps to a constant level on a ∼15 ps timescale. As discussed in several previous papers,20,25–28 the C–COOH bond itself breaks within 0.8 ps after the excitation and the rise of the absorption at 2343 cm−1 reflects the increasing intensity of the asymmetric CO2 stretch as the dissociating CO2 moiety transforms from its bend geometry in pyruvic acid to the linear geometry of fully hydrated CO2(aq). The formation of carbon dioxide suggests the formation of methyl hydroxy carbene, CH3COH.15,29,30 Our calculations predict that singlet methyl hydroxy carbene has a strong transition at 1274 cm−1 in addition to several weaker transitions below our detection limit, while triplet methyl hydroxy carbene has a single strong transition with a calculated frequency of 1187 cm−1 (Table SI1). Measurements of the IR spectrum of methyl hydroxy carbene in Ar matrices find the strongest transition as a double peak at 1257.4 and 1261.9 cm−1 in addition to weaker transitions at 1337 cm−1, 1301 cm−1 and 1024 cm−1.30Fig. 2 shows positive induced absorption peaks at 1192 cm−1, 1272 cm−1 and 1598 cm−1, in addition to the absorption pertaining to CO2(aq) at 2343 cm−1. The predicted spectra agree with the positive absorption measured at 1192 cm−1 and 1272 cm−1 suggesting the formation of methyl hydroxy carbene in the triplet and singlet excited states, respectively. Our calculations predict that the triplet carbene is 120 kJ mol−1 less stable than the singlet carbene, and the formation of triplet carbene and CO2 from the relaxed geometry of triplet pyruvic acid is endoenergetic by 50 kJ mol−1. Formation of the triplet carbene is thus disfavoured energetically, but could be formed immediately after intersystem crossing to the triplet surface, since the relaxed singlet excited state is essentially isoenergetic with the triplet carbene and CO2. Later, we show that the absorption at 1192 cm−1 may also be due to singlet and triplet excited states of energetically accessible configurations of pyruvic acid. The absorption transients at 1272 cm−1 appear within the instrument limited time of 0.8 ps consistent with the sub-picosecond C–COOH fission indicated by the carbon dioxide formation. The absorption persists for the full 560 ps of the measurements indicating that no secondary reactions involving singlet methyl hydroxy carbene occur within this period. With time methyl hydroxy carbene is likely transformed to acetaldehyde.15,29,30 Acetaldehyde has two strong transitions at 1108 cm−1 and 1716 cm−1 associated with the C–C stretch and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretch respectively (Fig. SI3). However, neither of these transitions are observed in the transient absorption data, thus indicating that the formation of acetaldehyde during the first 560 ps is insignificant, in agreement with the constant absorption assigned to methyl hydroxy carbene.
O stretch respectively (Fig. SI3). However, neither of these transitions are observed in the transient absorption data, thus indicating that the formation of acetaldehyde during the first 560 ps is insignificant, in agreement with the constant absorption assigned to methyl hydroxy carbene.
To identify the population dynamics of the singlet and triplet excited states of pyruvic acid, we have calculated the IR spectra of these states for energetically accessible conformers of pyruvic acid (Tables SI2 and SI3). The singlet excited state of conformer 2 (shown Fig. SI4) has two intense transitions at 1648 cm−1 (in D2O) and 1219 cm−1 (in H2O). The triplet excited state of conformer 1 and conformer 2 in Fig. SI4 each has two intense transitions at 1595 cm−1 (in D2O) and 1214 cm−1 (in H2O), and 1626 cm−1 (in D2O) and 1146 cm−1 (in H2O), respectively. Within the uncertainty, the calculated values agree with the measured positive transient absorption at 1598 cm−1 (in D2O) and 1192 cm−1 (in H2O) shown in Fig. 2, while the other transitions of these configurations are either hidden by the negative absorption transients associated with the depletion of ground state pyruvic acid or below the instrument detection limit. The absorption transients at 1598 cm−1 and 1192 cm−1 thus suggest the population of the singlet and/or triplet states. Our measurements don’t warrant a spectroscopic distinction between the excited singlet and triplet states. The absorptions at 1598 cm−1 and 1192 cm−1 rise to their final value in less than 0.8 ps and remain constant for the full 560 ps time span of our measurements. These measurements agree with the results reported by Davidson et al.,31 who determined the intersystem crossing yield in aqueous pyruvic acid to 0.22 thereby implying the excitation of both singlet and triplet states. They furthermore measured the triplet excited state lifetime to 74 ns easily consistent with the >560 ps duration of the absorption, we assign to excited state pyruvic acid. Dissociation to carbon dioxide and methyl hydroxy carbene together with the population of the excited pyruvic acid account for all measured absorption transients and are fully consistent with the observed permanent depletion of ground state pyruvic acid. In agreement with our previous conclusions the constant population of excited state pyruvic acid implies that the decarboxylation occurs within the first picosecond. Since our transient absorption spectrometer cannot cover the absorption at 1722 cm−1 and 2343 cm−1 or 1274 cm−1 in one measurement, we are unable to determine the ratio between the two reactions channels. Our measurements do not exclude the existence of decarboxylation channels with time constants of tens of nanoseconds.
The reaction dynamics of the near-UV decarboxylation of aqueous pyruvic acid resemble the photolysis of gas-phase pyruvic acid. Recent studies have shown that near-UV photo-excitation to the lowest singlet excited state of gas-phase pyruvic acid primarily results in decarboxylation forming carbon dioxide and methyl hydroxy carbene with subsequent isomerization to acetaldehyde and to less extent vinyl alcohol.16 The near-UV photochemistry of gas-phase pyruvic acid is largely explained by the mechanistic photodecarboxylation calculations by Chang et al.19 According to these calculations exposure to near-UV light excites pyruvic acid molecules to the lowest excited singlet state. The excited singlet state relaxes by transferring the hydrogen atom from the carboxyl group to the carbonyl. This tautomerization occurs close to a conical intersection connecting the excited state to the ground state. Decarboxylation in the excited singlet state is prohibited by a large (1.0 eV) barrier, but decarboxylation can occur from vibrationally excited states on the electronic ground state. According to Chang et al., decarboxylation can also occur from the lowest triplet state. Due to a small energy gap between the first excited singlet and triplet states calculated to 0.1–0.2 eV19 and measured to 0.29 ± 0.04 eV17 and spin–orbit coupling (17 cm−1), efficient intersystem crossing to the triplet state takes place close to a conical intersection with the lowest singlet states.19 The barrier for decarboxylation in the triplet state is calculated to 0.3 eV19 indicating that decarboxylation can occur with high efficiency. Decarboxylation of gas-phase pyruvic acid may thus take place both from the excited triplet state and from the vibrationally excited ground state. Applying the same mechanism to aqueous pyruvic acid implies that decarboxylation occurs close to the conical intersection in less than ∼0.5 ps after the excitation – either from the excited states or from highly excited vibrational levels on the electronic ground state of pyruvic acid.
As pointed out by Burrow et al.17 the photochemistry of gas-phase pyruvic acid is completely different from that of its conjugate base, and the same holds for the aqueous counter part. The near-UV photolysis of aqueous pyruvate is insignificant and all excited pyruvate anions return to the equilibrated ground state in 50 ps, while the photolysis of aqueous pyruvic acid results in significant decarboxylation and population of the lowest excited states with no excited pyruvic acid molecules returning to the equilibrated ground state within the first 560 ps. To the extent the gas-phase mechanisms apply to the aqueous phase, the very different yields from the two species can be explained by energy considerations. Our previous calculations showed that the decarboxylation barrier for pyruvate exceeds the photo-excitation energy at 340 nm and decarboxylation of pyruvate is thus energetically forbidden.20 In the photolysis of pyruvic acid at 340 nm, on the other hand, decarboxylation may occur from the lowest excited states close to conical intersections with the ground state or from highly vibrationally excited states of pyruvic acid's ground state.
Conclusions
The present experimental data show a near-UV photolysis scenario in which ground state pyruvic acid molecules are excited to the lowest excited singlet state. In less than 0.8 ps some of the pyruvic acid molecules dissociate to methyl hydroxy carbene and carbon dioxide, while others intersystem cross to the triplet state. The dissociation products exist for at least 560 ps, and the excited singlet and/or triplet state stays populated for at least as long. Processes occurring on longer timescales including those from long-lived excited states may ultimately affect the overall photochemistry of pyruvic acid. In contrast to pyruvic acid, all pyruvate anions excited by near-UV light return to the ground state within 50 ps. This explains why the photolysis yield of aqueous pyruvic acid excited by near-UV light depends on the acidity: at low pH, pyruvic acid is fully protonated, and the decarboxylation yield is finite, whereas at high pH, pyruvic acid deprotonates to pyruvate which remains intact. These results complement the explanation by Rapf. et al.,3 who assigned the pH dependence of decarboxylation to the pH dependence of hydration.Author contributions
T. W. suggested the project. J. T. recorded and analysed the experimental data. F. J. provided the theoretical calculations. F. J. and J. T. and T. W. wrote the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5cp04995d.Acknowledgements
T. W. and J. T. acknowledge the Novo Nordisk Foundation Facility Grant Nanoscat. No. NNF18OC0032622 The Novo Nordisk Foundation (New Exploratory Research and Discovery – NERD Grant, NNF22OC0074640) for funding. Danish National Research Foundation for financial support through the Center of Excellence for Chemistry of Clouds. (Grant Agreement No. DNRF172). We wish to thank Ditte Thomsen for making the graphical abstract.References
- G. Lin, S. Sillman, J. E. Penner and A. Ito, Atmos. Chem. Phys., 2014, 14, 5451 CrossRef.
- S. A. Epstein, E. Tapavicza, F. Furche and S. A. Nizkorodov, Atmos. Chem. Phys., 2013, 13, 9461 CrossRef.
- R. J. Rapf, M. R. Dooley, K. J. Kappes, R. J. Perkins and V. Vaida, J. Phys. Chem. A, 2017, 121, 8368 CrossRef CAS PubMed.
- A. J. Eugene, E. A. Pillar-Little, A. J. Colussi and M. I. Guzman, Langmuir, 2018, 34, 9307 CrossRef CAS PubMed.
- V. Wank, D. Lesnicki, M. Sulpizi and E. H. G. Backus, J. Chem. Phys., 2025, 163, 224705 CrossRef CAS PubMed.
- M. I. Guzman and A. J. Eugene, Molecules, 2021, 26, 5278 CrossRef CAS PubMed.
- P. A. Leermakers and G. F. Vesley, J. Am. Chem. Soc., 1963, 85, 3776 CrossRef CAS.
- E. C. Griffith, B. K. Carpenter, R. K. Shoemaker and V. Vaida, Proc. Natl. Acad. Sci. U. S. A., 2013, 110, 11714 CrossRef CAS PubMed.
- A. E. Reed Harris, B. Ervens, R. K. Shoemaker, J. A. Kroll., R. J. Rapf, E. C. Griffith, A. Monod and V. Vaida, J. Chem. Phys. A, 2014, 118, 8505 CrossRef CAS PubMed.
- K. J. Kappes, A. M. Deal, M. F. Jespersen, S. L. Blair, J.-F. Doussin, J. Cazaunau, E. Pangui, B. N. Hopper, M. S. Johnson and V. Vaida, J. Phys. Chem. A, 2021, 125, 1036 CrossRef CAS PubMed.
- M. Luo, D. Shemesh, M. N. Sullivan, M. R. Alves, M. Song, R. B. Gerber and V. H. Grassian, J. Phys. Chem. A, 2020, 124, 5071 CrossRef CAS PubMed.
- M. C. Larsen and V. Vaida, J. Phys. Chem. A, 2012, 116, 5840 CrossRef CAS PubMed.
- S. Yamamoto and R. A. Back, Can. J. Chem., 1985, 63, 549 CrossRef CAS.
- R. N. Rosenfeldt and B. Weiner, J. Am. Chem. Soc., 1983, 105, 3485 CrossRef.
- B. R. Samanta, R. Fernando, D. Rösch, H. Reisler and D. L. Osborn, Phys. Chem. Chem. Phys., 2021, 23, 4107 RSC.
- L. J. Sauer and H. F. Davis, J. Phys. Chem. Lett., 2025, 16, 3721 CrossRef CAS PubMed.
- E. M. Burrow, J. Carmona-García, C. J. Clarke, B. F. E. Curchod and J. R. R. Verlet, J. Am. Chem. Soc., 2025, 147, 36987 CrossRef CAS PubMed.
- Y. Pocker, J. E. Meany, B. Nist and C. Zadorojny, J. Phys. Chem., 1969, 73, 2879 CrossRef CAS.
- X.-P. Chang, Q. Fang and G. Cui, J. Chem. Phys., 2014, 141, 154311 CrossRef PubMed.
- J. Thøgersen, F. Madzharova, T. Weidner and F. Jensen, Nat. Commun., 2024, 15, 1 Search PubMed.
- M. Falk and A. G. Miller, Vib. Spectrosc., 1992, 4, 105 CrossRef CAS.
- L. H. Jones and E. McLaren, J. Chem. Phys., 1958, 28, 995 CrossRef CAS.
- T. Schädle, B. Pejcic and B. Mizaikoff, Anal. Methods, 2016, 8, 756 RSC.
- J. Gleim, J. Lindner and P. Vöhringer, J. Chem. Phys., 2022, 156, 094594 CrossRef PubMed.
- M. M. Madsen, F. Jensen and J. Thøgersen, Phys. Chem. Chem. Phys., 2020, 22, 2307 RSC.
- J. Thøgersen, V. Vaida, M. Bregnhøj, T. Weidner and F. Jensen, Phys. Chem. Chem. Phys., 2021, 23, 4555 RSC.
- J. Thøgersen, T. Weidner and F. Jensen, Phys. Chem. Chem. Phys., 2021, 23, 10040 RSC.
- J. Thøgersen, A. S. Chatterley, T. Weidner and F. Jensen, J. Am. Chem. Soc., 2023, 145, 9777 CrossRef.
- S. Mondal, S. Sadhukhan, A. Sinha and M. K. Hazra, J. Am. Chem. Soc., 2025, 147, 211 CrossRef CAS PubMed.
- P. R. Schreiner, H. P. Reisenauer, D. Ley, D. Gerbig, C.-H. Wu and W. D. Allen, Science, 2011, 332, 1300 CrossRef CAS PubMed.
- R. S. Davidson, D. Goodwin and P. F. De Violet, Chem. Phys. Lett., 1981, 78, 471 CrossRef CAS.
| This journal is © the Owner Societies 2026 |