 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Validity of kinetic predictions for polymer crystallization from molten and glassy states: the case of poly(p-dioxanone)
Roman
Svoboda
Department of Physical Chemistry, Faculty of Chemical Technology, University of Pardubice, Studentská 573, 532 10 Pardubice, Czech Republic. E-mail: roman.svoboda@upce.cz
First published on 17th February 2026
Abstract
The crystallization of poly(p-dioxanone) (PDX) was examined under different regimes – cooling of a melt, heating from the glassy state, and isothermal annealing reached either from the melt or from the glass. In addition to differential scanning calorimetry (DSC) measurements, supplemental characterization was performed using Raman spectroscopy, thermogravimetry, and optical microscopy. The comparison of the hot and cold crystallization data revealed distinct differences in the PDX crystallization behavior, governed by the presence/amount of nuclei generated on the applied thermal path. The kinetic analysis of the crystallization data employed the extended autocatalytic model combined with Hoffman–Lauritzen or Arrhenius rate expressions. Both frameworks fitted the data well, but their predictive accuracy depended strongly on how the temperature-dependent nucleation Kg(T) and growth E(T) activation energies were implemented (an improvement of the existing models was suggested, based on compensation via the addition of a continuous temperature dependence of the pre-exponential factor A(T)). Predictions of isothermal crystallization from non-isothermal kinetics were only reliable when the nucleation state of the system was appropriately reproduced, underscoring the limitations of using thermal proximity alone as a predictor.
1. Introduction
The process of crystallization in polymers plays a pivotal role in practically all applications of semi-crystalline thermoplastics, i.e., polymers with regular, linear chain structure, small, symmetrical side groups, and strong intermolecular interactions (e.g., hydrogen bonding). In practice, the crystallization of these polymers occurs either from the glassy state upon heating or from the molten state upon cooling. These two distinct pathways are central to how polymers are processed and used in manufacturing, packaging, medical devices, and structural components.Crystallization from the molten state during cooling is fundamental to the primary shaping processes of polymers, such as injection molding, extrusion, and blow molding. For example, in the production of polyethylene terephthalate (PET) bottles, the rate and extent of crystallization during cooling directly affect the bottle's mechanical strength, clarity, and barrier properties. If PET crystallizes too quickly, it may become opaque and brittle; if it crystallizes too slowly or insufficiently, the product may be too soft or deform under load. Control over melt crystallization is therefore essential for ensuring dimensional stability and performance.1,2 Several other examples involve the following: polypropylene (PP) injection molding, where the cooling rate affects spherulite size, transparency, and toughness: fast cooling leads to smaller spherulites (improved toughness), while slow cooling enhances stiffness but reduces impact strength;3 high-density polyethylene (HDPE) pipe manufacturing, where melt crystallization determines pipe rigidity, long-term creep resistance, and thermal performance: improper cooling can lead to uneven crystallinity and dimensional instability;4 polyamide 6 (Nylon 6) in automotive parts, where the crystallinity developed during cooling enhances strength, heat resistance, and chemical durability;5 polyethylene naphthalate (PEN) for high-performance films, which requires controlled crystallization to achieve superior dimensional stability and gas barrier properties in packaging and electronics;6 thermoplastic polyurethane (TPU) moldings, where the crystallized hard segments influence mechanical properties and shape memory behavior in applications like medical catheters and wearable electronics;7 polyketone (PK) fibers require control of the crystalline morphology, critical for high-performance products used in filtration and reinforcement;8 polyoxymethylene (POM/Acetal) crystallization determines low friction and wear-resistant surfaces in precision gear applications;9 melt-spun polyethylene terephthalate (PET) fibers are crystallization-affected with respect to tenacity and shrinkage—crucial for textiles and industrial fibers;10 crystalline polylactic acid (PLA) packaging (specifically compostable hot food containers) utilizes development of the semi-crystalline phase during slow cooling to improve thermal resistance;11 quality of the polybutylene terephthalate (PBT) crystalline phase ensures dimensional precision, flame resistance, and insulation properties for electrical connectors and housings.12
On the other hand, crystallization from the glassy state during heating (also called “cold crystallization”) becomes relevant for polymers that are initially processed into the amorphous state through, e.g., rapid quenching or solvent casting. For example, during 3D printing, the polymers are often printed amorphous or only partially crystallized to maintain flexibility and ease of shaping, as is the case with polylactic acid (PLA) used to manufacture medical implants.13,14 The consequent controlled crystallization realized via thermal annealing increases the stiffness, mechanical stability, and resistance to hydrolytic degradation, so that the implant remains functional for the period of time required for the patient to heal. The cold crystallization is paramount also in the powder-based processing, such as the technique of selective laser sintering (typically polyamides PA12, PA11, and PA6, and thermoplastic polyurethanes (TPU)), where the laser-sintered amorphous particles undergo post-processing heat treatment, enhancing the mechanical strength and dimensional accuracy of the printed part.15–17 Furthermore, in pharmaceutical applications, the semi-crystalline polymer excipients (e.g., polyethyleneglycol (PEG) or poly(ε-caprolactone) (PCL)) are tailor-crystallized to fine-tune the drug release profiles.18,19 For medicinal polymers, such as poly(p-dioxanone) (PPDX) used for the manufacturing of biodegradable sutures and implants, the properties of the crystalline phase embedded in the amorphous matrix define not only the mechanical properties but also the rate of degradation.20 In recycling, post-processing and re-processing, the thermal annealing is used to restore the crystallinity lost during the previous re-extrusion or aging; common examples include poly(ethylene terephthalate) (PET) used for various packaging needs, or poly(trimethylene terephthalate) (PTT) used for textile manufacturing.21,22
From the physico-chemical point of view, polymer crystallization proceeds during the cooling of the melt as a kinetically regulated, multistage ordering process, reflecting a competition between thermodynamic driving forces and molecular constraints imposed by chain connectivity. With the initiation of undercooling, local density, conformational, and orientational fluctuations emerge in the melt. At the microscopic level, nucleation is initiated by the cooperative alignment of chain segments into transient, partially ordered clusters.23–27 In many polymers, this early ordering manifests as a mesomorphic state, characterized by enhanced segmental orientation and conformational regularity without full three-dimensional translational order, which arises due to the segmental alignment and conformational selection being kinetically accessible on shorter time scales than long-range registry and lamellar organization. The mesomorphic domains reduce the nucleation barrier by lowering interfacial free energy with the surrounding melt and often act as precursors or self-nuclei for subsequent crystallization.24–29 The consequent crystal growth then proceeds through the incorporation of chain segments from the surrounding amorphous matrix into lamellar crystals, requiring chain folding, partial disentanglement, and transport toward the growth front.23,24,28–32 These processes are inherently frustrated by limited chain mobility, leading to non-ideal folding, loop and tie-chain formation, and lamellae with finite thickness and defects. As crystallization progresses, the development of crystalline domains imposes spatial and dynamic constraints on the remaining amorphous material, giving rise to a rigid amorphous fraction (RAF) in which chains are immobilized by interfacial confinement rather than structural order. Simultaneously, volumetric contraction and differential mobility generate a mechanically strained amorphous matrix, with residual stresses and frozen-in orientation reflecting the non-equilibrium nature of the transformation.26–32
On the other hand, the crystallization of the amorphous polymer (formed via melt-quench) proceeds during heating from the already present frozen-in structural heterogeneities that were formed during the preceding cooling phase. The potentially formed mesomorphic domains may persist below the glass transition (Tg) as kinetically trapped, partially ordered regions – upon reheating above Tg, the domains regain mobility and can act as very efficient self-nuclei, promoting rapid nucleation without the need for large-scale molecular rearrangements.27,30–32 Crystal growth then proceeds from these pre-aligned segments, often leading to high nucleation densities and comparatively imperfect lamellar structures. Contrary to the crystallization proceeding during cooling, the structural/spatial frustration associated with the RAF formation gets immediately released with rising temperature within the cold-crystallization regime.27,30–32
The final semicrystalline morphology therefore encodes the entire kinetic pathway of cooling (and consequent heating) – the nucleation (either homo- or heterogeneous) can be further mesophase-mediated, which can enhance the crystal density, the associated imperfect growth yields folded and defective lamellae, and the coexistence of mobile amorphous, rigid amorphous, and crystalline regions produces a heterogeneous material whose mechanical and thermal properties are governed as much by frustrated molecular packing as by crystalline order itself.24,30,31
As shown above, the two distinct crystallization pathways play a crucial role in polymer technology and science. Owing to the intricacies associated with the multi-degree ordering of the polymer matrix (as opposed to, e.g., inorganic or low-molecular organic glasses), the prediction of the crystallite morphology, density, and distribution is not straightforward with regard to these two pathways. With the detailed knowledge of the crystallization process being crucial for a large number of applications utilizing thermoplastic polymers, this topic deserves to be explored. Owing to the processing route for the majority of thermoplastics, the theoretical framework for the description of crystallization from the melt23–28 has been developed to a significantly greater extent compared to crystal growth from the amorphous phase, where (largely) the ordinary Arrhenian dependence of the rate constant29–32 is utilized to describe the crystallization kinetics. Note that both these approaches to kinetic analysis will be introduced in detail in Section 2. With the very recent development and thorough testing of a universal kinetic model27,28 for polymer crystallization, the possibility of critically comparing the two approaches to crystal growth in polymers is now open. Even more importantly, the accuracy of the kinetic predictions provided by these two approaches can now be directly confronted with experimental results to determine the best way of tailoring the crystallization process in practice.
Such analysis is the main goal of the present paper. Since the experimental crystallization data need to be collected in all standardly utilized regimes (i.e., non-isothermally during the cooling of a melt, non-isothermally during the heating of a glass, isothermally after cooling from a melt, isothermally after heating from a glass), the choice of suitable polymeric materials is rather limited. In the present work, the crystallization behavior of poly(p-dioxanone) (PDX) will be exploited in this regard. This material was shown32 to exhibit a relatively broad temperature interval, in which the crystallization can be initiated both during cooling from the molten state and during heating from the glassy state (which is the base requirement of the present research). In addition, PDX exhibits32 well resolved polymorphic transitions, formation of the RAF phase, and was even reported to be able to form the mesomorphic phase.33 As such, PDX is a suitable model candidate for exploration of the variations (both kinetic and physico-chemical ones) associated with crystal formation via the two distinct pathways. Apart from PDX being used as a model material, the crystallization data and the corresponding predictions will also be of practical interest, since PDX is used for the manufacturing of chirurgical sutures,34,35 and is currently considered for the production of various medicinal implants.35,36
2. Theoretical background
The kinetics of the simple thermally activated solid-state processes (including crystal growth) can be described by the following set of equations,29 where eqn (1a) is the universal expression used for the solid-state kinetics (multiplication of the rate constant K and kinetic model f), and eqn (1b) is one of the most common and flexible kinetic models, the Šesták–Berggren equation:29,37 | (1a) |
| f(α) = (1 − α)N·αM | (1b) |
As mentioned in the introductory section, the fundamental difference between the two approaches to modeling the hot and cold crystallization lies in the expression of the rate constant K(T). The crystal growth from the molten phase is usually described in terms of the Hoffman–Lauritzen theory:23,24
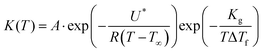 | (2) |
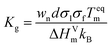 | (3) |
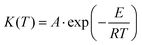 | (4) |
3. Experimental
Poly(p-dioxanone) (PDX) sutures (MediCross, Ostrava, Czech Republic) were used as a source of the PDX polymer and were used as-purchased. The main portion of experimental measurements was performed by means of differential scanning calorimetry (DSC), using the Q2000 calorimeter (TA Instruments, USA) equipped with an autosampler and RCS90 cooling accessory. The measurements were performed in the T-zero heat flow mode; the DSC was calibrated utilizing the melting temperatures and enthalpies of the In, Zn, and H2O standards; the calibrations were performed under standard conditions (hermetically sealed low-mass Al pan with a pin-hole lid, purge gas (N2) flow of 20 cm3 min−1, heating rate q+ = 10 °C min−1). The PDX pieces cut from the original suture were hermetically sealed in a low-mass T-zero Al pan, which is a commercial type of 40 µl Al pan with a mass of ∼20.4 mg and a corresponding lid (∼10.3 mg) for hermetical sealing (the experiments were thus effectively performed in a static air atmosphere). The sample masses varied in the 10.0–10.2 mg range (determined with ±0.05 mg accuracy).Four series of cyclic DSC measurements were performed, each with a new sample to avoid the degradation (thermal depolymerization32) that slowly occurs above the melting temperature Tm. In particular, each of the four temperature programs consisted of a series of cycles (starting with cooling from the molten state, and finishing with heating into the molten state), where a selected variable(s) was changed for each cycle. In the following paragraphs, the four types of temperature programs (always corresponding to one cycle) will be described, with the visual representations given in Fig. 1.
(1) Cyclic non-isothermal experiments with hot crystallization occurring during the cooling of the molten PDX, where the sample was first annealed for 30 s at 130 °C, then cooled at a selected cooling rate q− (which was the only variable in this type of cycle; the cooling rate followed the order 50, 30, 20, 15, 10, 7, 5, 3, 2, 1, 0.5, 0.2, and 0.1 °C min−1) to −35 °C, and then heated back to 130 °C at 20 °C min−1. Note that these cycles were also performed as a test of the critical cooling rate for PDX.
(2) Cyclic non-isothermal experiments with cold crystallization occurring during the heating of the glassy PDX, where the sample was first annealed for 30 s at 130 °C, then cooled at 50 °C min−1 to −35 °C, and then heated at a selected heating rate q+ (varying in the order 50, 30, 20, 15, 10, 7, 5, 3, 2, 1, 0.5, 0.2, and 0.1 °C min−1) to 130 °C.
(3) Cyclic isothermal experiments being initiated by cooling from the molten state, where the sample was first annealed for 30 s at 130 °C, then cooled at 50 °C min−1 to a selected annealing temperature Ta with an isotherm following for the time ta; then the sample was cooled to −30 °C at 10 °C min−1, and then heated to 130 °C at 10 °C min−1. The following Ta in °C (ta in min) combinations were used for the performed cycles: 50(60), 45(60), 40(60), 35(60), 30(60), 25(60), 20(60), 15(60), 10(60), 5(120), 0(240).
(4) Cyclic isothermal experiments being initiated by heating from the glassy state, where the sample was first annealed for 30 s at 130 °C, then cooled at 50 °C min−1 to −30 °C, then heated at 50 °C min−1 to a selected annealing temperature Ta with an isotherm following for the time ta; then the sample was cooled to −30 °C at 10 °C min−1, and then heated to 130 °C at 10 °C min−1. Identical Ta (ta) combinations as for the cycle of the third type were used.
Thermal stability of PDX was (apart from the complex evaluation of the DSC data) directly assessed by thermogravimetry (TGA), using the STA (TGA) 449 F5 Jupiter instrument (Netzsch) equipped with a DSC/TG holder. In this regard, two simple heating scans were performed under air and N2 atmospheres (flow rate 50 cm3 min−1) in the 30–550 °C temperature range (at a heating rate of 10 °C min−1). The TGA measurements were realized in open Al pans (40 µl, ∼21.5–22 mg); the sample masses were ∼10 mg (determined with ±0.005 mg accuracy). The structural characterization of selected PDX samples was done by means of Raman spectroscopy, using the DXR2 Raman microscope (Thermo Fisher Scientific, USA) equipped with a 785 nm excitation diode laser (laser spot size 1.6 µm) and a CCD detector. The measurements were performed with 20 mW laser power on the sample, 3 s duration of a single scan, and 35 scans accumulated in one spectrum, in the spectral range of 50–3375 cm−1. In addition, the selected PDX samples were also investigated with respect to crystalline morphology by means of optical microscopy, using the iScope PLMi (Euromex) optical microscope in the transmission mode, equipped with a series of ×4–×40 objectives and a Moticam visual camera (with a ×1 magnification adapter).
4. Results
The main part of the experimental results was obtained by DSC – the non-isothermal crystallization data obtained during cooling and heating are depicted in Fig. 2A and B, respectively; several examples (every other measurement) of the isothermal crystallization curves are shown in Fig. 2C and D. Starting with the experiments focused on crystallization during the cooling of the melt (Fig. 2A, variable q−, fixed q+ = 20 °C min−1), it is clear that at the highest cooling rate of 50 °C min−1, practically no crystallization proceeds, as almost no detectable signal occurs during the cooling part of the cycle and a large crystallization peak is recorded during the consequent heating scan. Interestingly, the heating scan following the cooling at q− = 50 °C min−1 does not exhibit the highest crystallization enthalpy; a slightly stronger crystallization peak occurs during heating following the cooling at q− = 30 °C min−1, which provides an ideal balance between nucleation (the lower the q−, the more time provided for nucleation) and crystal growth (the lower the q−, the more time provided for crystal growth, which then does not already contribute to the exothermic signal evolving during the consequent heating). The borderline cooling rate is q− = 10 °C min−1, which is the lowest q− during which full crystallinity is not achieved during cooling. Note that by “full crystallinity” we are referring to the standard value of χc ≈ 45–55% achievable for this semi-crystalline polymer;47 the corresponding melting enthalpy of a fully crystalline PDX was determined to be ΔHm100% = 141.18 J g−1.48 The heating curves following the individual cooling steps then exhibit an endothermic glass transition effect characterized by the glass transition temperature Tg of ∼−10 °C, followed by the exothermic crystallization signal (manifesting at ∼25–55 °C for the preceding q− ≥ 10 °C min−1) and the endothermic melting signal (manifesting between ∼95 and 120 °C). Depending on the prior q−, a small secondary crystallization peak closely preceding the melting process can occur. This crystallization peak is attributed to the polymorphic α′ → α or disorder → order transition (where α′ represents a mesomorphic phase with loose chain packing, which forms at low T). Correspondingly, the α′ → α transition is missing during the cycles with low q−, where the majority of the crystalline phase was formed at high T (just below the melting temperature Tm).The second set of non-isothermal DSC measurements is depicted in Fig. 2B – with variable q+ and fixed q− = 50 °C min−1. Since all cooling steps were performed at maximum q−, a practically fully amorphous PDX was always formed, and the consequent heating scans were typologically identical, with the effects only shifting to higher T with increasing q+. The only small exception was the cycle performed at the highest q+, where the secondary high-T crystallization (α′ → α transformation) is missing. This is either because the primary crystallization already proceeded at high enough T (due to q+ being 50 °C min−1) for only a negligible amount of the α′ phase being formed, or because at such high q+, the melting of the α′ phase was initiated before it could recrystallize into the α polymorph. A very interesting discrepancy (crucial for the topic of the present paper) was found when monitoring the isothermal crystallization at high Tas – see Fig. 2C. Whereas in the case of cold crystallization (during the heating of the glassy phase), the process gradually slowed down as Ta decreased, the hot crystallization (proceeding during the cooling of a melt) exhibited a non-monotonous trend in the crystallization rate, where the process was slower for Ta = 50 °C, sped up for Tas = 40 and 30 °C, and then again significantly slowed down at even lower Tas, roughly evening out the rates of both processes at Tas ≤ 20 °C. The seemingly anomalous behavior observed for the hot crystallization is caused by the absence of pre-existing nuclei when crystallizing on the path from the molten state. In the case of cold crystallization, the prior formation of the PDX glass (during the cooling at q− = 50 °C min−1) produces a fixed amount of nuclei, and the consequent crystal growth then proceeds from this approximately constant number of active sites. On the other hand, during hot crystallization, the nucleation and crystal growth processes proceed simultaneously, where the former is the rate-limiting step at high T. This explains not only the much higher crystallization rates during the high-T cold crystallization, but also the GT=30°C > GT=50°C data (with “G” being the macroscopic crystallization rate), indicating that the contributions of the nucleation and crystal growth rates even out in the 30–40 °C temperature range. This will be further referred to in Section 5.1 during the mathematical modeling of the two phenomena. As mentioned above, at low T (≤ 20 °C), the DSC-recorded crystallization rates are roughly similar for both crystallization paths (cold and hot), meaning that a sufficiently high number of nuclei was formed during the cooling to these temperatures regardless of the selected path, reaching a saturated state.
In this regard, an even more interesting finding is that associated with the isothermal crystallization at 0 °C, where the sample crystallizing directly after cooling from the melt crystallized faster, but to a significantly lower extent. Whereas the latter can be partially explained by possible inaccuracies in the baseline subtraction, the faster crystallization rate clearly means that the microscopic crystal growth rate is already very low at these temperatures, and that it is the nucleation rate which dictates the crystallization behavior. In particular, the very high density of nuclei formed in the case of the glass-quenched sample hinders the overall crystallization process, as the impingement occurs very early and the competition over the available polymer chains slows down the reordering process. In addition, the nucleation-dominated crystallization could also explain the final difference in the reached crystallinity degrees, as a larger portion of the amorphous matrix can be eventually converted into the crystalline lamellae.
Since certain important parts of Fig. 2A and B are not clear due to the scaling/overlap of the raw data cycles, the selected archetypal heating scans are shown in greater detail in Fig. 3A and B (not necessarily corresponding to the parts of Fig. 2A and B). In Fig. 3A, the trend in the shape of the melting peak measured during the heating scan following the cooling at different q− (shown in Fig. 2A) is depicted for the low q− cycles. Evidently, the high-T crystallization manifesting at low q− results in an absence of the α′ → α transformation (as the majority of the crystalline phase is already formed at high T, and thus as an α polymorph).
In addition, the high-T crystallization also results in the manifestation of the lower-T melting peak shoulder, which gets further split depending on the actual applied q−. We believe that these multiple shoulders correspond to the gradual formation of the rigid amorphous fraction (RAF; as opposed to the mobile amorphous fraction (MAF)49–51) that characterizes the interface between the crystal and the true amorphous phase – note that this layer is formed by the semi-fixated parts of polymer chains, which are already not included in the folded crystalline lamellae. The main/dominant melting peak shoulder then indicates the formation of an additional distinct crystalline phase (high-temperature polymorph/stable phase) – deeper insight into this behavior is reported in ref. 32 and 52. As can be seen in Fig. 3A, the first signs of the α′ → α transformation are apparent for the cycle with q− = 2 °C min−1, where the hot crystallization proceeded in the 45–80 °C temperature range, as opposed to the q− = 1 °C min−1 cycle with 55–85 °C range. The high-temperature limit for the formation of the α′ phase thus appears to be ∼55–60 °C. The differences in the low-q− and high-q− cycles are further contrasted in Fig. 3B, with the added comparison to the typical heating scan following the isothermal crystallization measurement at Ta = 40 °C. The typical feature associated with the heating scans following the isothermal crystallization is the presence of a small endotherm (often characterized as a hybrid process between the glass transition and melting) slightly above Ta corresponding to the stabilization of the RAF phase.52,53 It is worth noting that none of the DSC experiments provided direct evidence for the formation of the mesomorphic phase in PDX (apart from the above-mentioned α′ → α or disorder → order transition), which should manifest as a broad and weak exothermic signal. Nonetheless, the nonexistence of such signal does not prove the nonexistence of the mesomorphic phase, the formation of which can be simply associated with too slow heat evolution (below the DSC detection limit) or be hidden as an underlying signal overlapping with stronger-manifesting physico-chemical phenomena.
To quantify the different types of thermokinetic behavior depicted in Fig. 2, evolution of the trends in characteristic temperatures and enthalpies was determined – the terminology associated with the evaluated processes is schematically defined in Fig. 3C, where the dashed lines indicate the evaluation of the crystallization and melting enthalpies. Note that Tg was evaluated as a half-height midpoint, and the upper index “ons” corresponds to the quantity being evaluated as an extrapolated onset temperature. The trends in characteristic quantities linked with the non-isothermal measurements (Fig. 2A and B) are shown in Fig. 4, and the evaluation of the isothermal data is presented in Fig. 5. The data in tabular form are included in the SI.
Starting with the cold crystallization data (Fig. 2B), the corresponding trends depicted in Fig. 4A show that the most pronounced changes are those in Tonsc1, Tc1, Tc2, ΔHc1, and ΔHm. Whereas the characteristic temperatures behave with increasing q+ as is commonly expected (rapid increase of Tc1 due to the low activation energy, slow increase of Tg due to the high activation energy, practically invariable Tm that only slightly changes as a consequence of the ratios of different polymorphic phases), the opposite trends in the crystallization and melting enthalpies are more interesting. The increasing ΔHc1 with increasing q+ can be partially a consequence of Kirchhoff's law, but as will be shown below, the main contribution still has to be associated with the low-T mesomorphic α′ with loose chain packing exhibiting a generally lower ΔHc1. The seeming discrepancy with ΔHm then arises from the overlap between secondary crystallization and melting. If one neglects the possible inaccuracies associated with the loss of the exo/endo signals due to their overlap, then the low q+ generally produces a larger amount of the α′ phase, and this larger portion then recrystallizes at high temperature to form the more densely packed α polymorph, which consequently translates into the higher overall melting enthalpy – note that the measured melting peak reflects the final state after the secondary (c2) recrystallization, rather than the original primary (c1) crystallization process.
The evaluation of the hot crystallization cycles (raw data depicted in Fig. 2A) is shown in Fig. 4B and C. In Fig. 4B, the evaluation of the (second) heating part of the cycles is shown (purposefully before the first, cooling step), where the Tc1 and ΔHc1 data start only at q− = 7 °C min−1 (at lower q−, full crystallinity was achieved already during the first, cooling parts of the cycles). Since all the heating scans were performed at the same q+ = 20 °C min−1, no major variability of the characteristic temperatures can be expected. Hence, instead of plotting Tg, the difference of heat capacity between the undercooled liquid and glassy states (Δcp; evaluated at Tg) is shown. This quantity corresponds to the degree of crystallinity achieved after the first, cooling part of the cycle was finished. Again, the sharp increase of Δcp for cycles with q− > 10 °C min−1 indicates that at these cooling rates, highly/fully amorphous PDX material was obtained. The corresponding evaluation of the first cooling steps is shown in Fig. 4C. If we sum the crystallization enthalpies ΔHcool (the portion achieved during the cooling step) and ΔHc1 (the portion achieved during the consequent heating), the resulting value nicely copies the trend for the melting enthalpy ΔHm. In the case of the hot crystallization cycles, the majority of the crystal formation occurs (at least at low q−) during cooling; hence, the situation is simpler compared to the previously discussed cold crystallization cycles. During slow cooling of the melt, the crystallization occurs at high T, a large portion of the densely packed α polymorphic phase is formed, and the crystallization as well as the corresponding melting enthalpies are similarly high, because no α′ → α recrystallization occurs. At higher q−, a significant portion of the crystalline phase is still formed (or at least pre-nucleated) during the cooling step, which defines the similarity between ΔHcool + ΔHc1 and ΔHm. The higher the q−, the lower the portion of the crystalline phase being formed during the cooling step, and hence, the increasing discrepancy between the two enthalpies – see the magenta and violet lines in Fig. 4C.
The characteristics monitored during the heating scans following the isothermal crystallization steps (where Ta was achieved either by rapid cooling of a melt or by rapid heating of a glass) are shown in Fig. 5. The heating scans were, again, performed at a constant q+ = 10 °C min−1; hence, the characteristic temperatures are practically invariable. Since the Ta range covered only the low-T region (0–50 °C), the determined ΔHm also stays rather invariant, which is in good correspondence with the data shown in Fig. 4B and C. In general, the heating scans following both types of isothermal crystallization experiments (differing in the pathway to Ta) were extremely similar, exhibiting almost identical Tx and ΔHx values.
In addition to the DSC measurements, the selected DSC-crystallized samples were characterized by means of temperature-resolved Raman spectroscopy – see Fig. 6. The full-range Raman spectra of the molten and crystalline PDX samples are shown in Fig. 6A. The main bands can be attributed as follows: the band at 870 cm−1 corresponds to the C–O–C symmetric stretching vibration; the band at 1048 cm−1 corresponds to the stretching vibration of C–C in the aliphatic chain; the band at 1451 cm−1 corresponds to the –CH2– bending vibration; the band at 1732 cm−1 corresponds to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibrations in the ester carbonyl group; and the bands at 483, 1242, 1403, 1610 and 1638 cm−1 correspond to the vibrations in the solvent violet 13 (dye) structure.54–56 It has been hypothesized in ref. 54 that the amount of the amorphous content in PDX can be linked to the proportional manifestation of the high-frequency shoulder of the 1732 cm−1 band.
O stretching vibrations in the ester carbonyl group; and the bands at 483, 1242, 1403, 1610 and 1638 cm−1 correspond to the vibrations in the solvent violet 13 (dye) structure.54–56 It has been hypothesized in ref. 54 that the amount of the amorphous content in PDX can be linked to the proportional manifestation of the high-frequency shoulder of the 1732 cm−1 band.
In Fig. 6B, the Raman spectra are shown that were collected during the following experiment: a small PDX sample was first melted, then quenched at q− = 50 °C min−1 to −30 °C (forming an amorphous matrix), and then left to “fully” crystallize at 20 °C. It is clear that, indeed, the fully amorphous PDX (be it in a liquefied or in a quenched-in form) shows a broad, round band in the 1710–1780 cm−1 range – to the authors’ knowledge, these are the first ever recorded Raman spectra of fully amorphous PDX. As the sample crystallized, a sharp band at 1732 cm−1 started to form. In Fig. 6C, the Raman spectra of the fully amorphous and different DSC-crystallized samples are compared – note that in all cases the crystallized samples were “fully” crystalline (the sample cooled at q− = 10 °C min−1 to −30 °C was left to crystallize to equilibrium at 20 °C; the sample heated at 50 °C min−1 to 70 °C was annealed at that temperature for 30 min and then cooled to laboratory temperature). It is apparent that the proportional manifestation of the two bands directly reflects the presence of the densely packed α polymorphic phase – note that the overall degree of crystallinity changes in a much less pronounced way with the conditions (temperature program) relevant for the samples depicted in Fig. 6C. Nonetheless, further confirmation of this hypothesis is necessary, as the true degree of crystallinity reached during some of these experiments can be only estimated from the present DSC data due to the involvement of the α′ → α recrystallization before the melting enthalpy associated with the low-T crystalline state can be determined.
As the present DSC experiments were performed in hermetically sealed Al pans, i.e., in a static air atmosphere, the high-T PDX stability in air needed to be verified (the kinetic study of the thermal decomposition/depolymerization of PDX reported in ref. 32 was only performed under a N2 atmosphere). The comparison of the TGA measurements performed in N2 and air atmospheres is shown in Fig. 6D. The decomposition/depolymerization evidently proceeds slightly faster and via a distinctly varied mechanism in the air atmosphere, but the difference is still only by 10–20 °C, which makes the presently used upper boundary of the DSC cycles (130 °C) a safe temperature to remain at for several tens of minutes32 – the present total of annealing time at 130 °C was for each sample ∼8 min + the time spent in the near vicinity of the borderline temperature during the slow heating or cooling scans. It is noteworthy that no signs of sample depolymerization were detected during the DSC experiments.
Complementary data to the Raman spectroscopy measurements were obtained by means of optical microscopy. In particular, the four differently DSC-crystallized samples from Fig. 6C were photographed immediately after finishing the defined temperature program – see Fig. 7. Interestingly, the distinct spherulitic morphology with visible lamellae orientation forms only at high T during the very slow cooling of a melt (q− = 0.1 °C min−1). Still recognizable but significantly smaller crystallites form during the faster cooling of the melt (q− = 10 °C min−1). If the melt is quench-amorphized, no spherulites of comparable size (visible under ×10 magnification) can be found in the micrographs, regardless of the crystallization temperature. This is a particularly important finding with regard to the intensity of the 1732 cm−1 Raman band (see Fig. 6C) – the Raman-detected degree of crystallinity is not correlated to the presence of visually identifiable morphology.
5. Discussion
As was mentioned in the introductory part, the discussion will be split into two parts. First, the crystallization kinetics will be calculated for all DSC-measured crystal growth modes and pathways. The emphasis will be put on the accuracy of the model-fitting-based description of the crystallization data, as well as on the identification of the kinetic trends occurring with the key conditions of the given temperature programs (q−, q+, and, ultimately, Ta). In the second part, the kinetic predictions calculated on the basis of each non-isothermal path type will be compared, and their accuracy will be evaluated based on the comparison with the isothermal data.5.1. Crystallization kinetics
| B(T) = (1 − α(T))·(z0,r + z1,r·T) + α(T)·(z0,p + z1,p·(Tf − T)) | (5) |
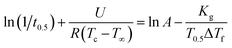 | (6) |
The curve-fitting by the MCHL model (depicted in Fig. 8C and D) gave the sets of kinetic parameters that are introduced in Fig. 8E and F in dependence on q− (and listed in SI). At low q−, fairly standard crystallization behavior is observed, with the MCHL parameters M and N being close46 to the typical nucleation-growth kinetics60,61 (in this case characterized by the Avrami exponent ∼1.5). However, at q− ≥ 7 °C min−1, both kinetic exponents deviate from the approximately constant values – the main feature being the exponent M attaining negative values. Note that since the exponent M is responsible for the autocatalytic behavior, its negative value indicates the mathematical autoretardation (decreasing the crystallization rate with the increasing degree of conversion α), which is the only way how (with fixed Kg value) to account for the overall decreasing crystallization enthalpy (see the identical trend for ΔH in Fig. 8F). The alternative ways of modeling this behavior would be either an implementation of the mutually compensating Kg–T and A–T dependence, or a complex reaction mechanism based on reversible reactions (with the former reflecting the true physico-chemical kinetics more closely). Nonetheless, the development of such a model/description is beyond the scope of the present paper. A partially compensating factor is also expressed by the ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A parameter; however, the negative M values are indeed paramount for allowing the description to be based on such high Kg values. The achieved accuracy of the kinetic description for all fitted data-curves is very high (the correlation coefficients are shown in Fig. 8F), confirming the relevancy of the determined kinetic parameters – note that the MCHL kinetic model has, in general, a pronounced absolute minimum in the hyperspace of the least-squares-based optimization solutions, with no comparable local minima offering acceptable alternatives. We have also tested that the very high Kg values determined for the high q− DSC data are indeed the only possibility how to accurately describe the data with constant/fixed Kg (no other MCHL kinetic parameter can supplement the role of Kg) – this further validates the results depicted in Fig. 8A, E and F.
A parameter; however, the negative M values are indeed paramount for allowing the description to be based on such high Kg values. The achieved accuracy of the kinetic description for all fitted data-curves is very high (the correlation coefficients are shown in Fig. 8F), confirming the relevancy of the determined kinetic parameters – note that the MCHL kinetic model has, in general, a pronounced absolute minimum in the hyperspace of the least-squares-based optimization solutions, with no comparable local minima offering acceptable alternatives. We have also tested that the very high Kg values determined for the high q− DSC data are indeed the only possibility how to accurately describe the data with constant/fixed Kg (no other MCHL kinetic parameter can supplement the role of Kg) – this further validates the results depicted in Fig. 8A, E and F.
 | (7) |
The results of the two sets of non-linear optimizations based on the combination of eqn (1) and (4) (with either “E = fix” or “E = var”) are shown in Fig. 9E and F. Interestingly, practically the same ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A values were obtained in both optimization series (ln
A values were obtained in both optimization series (ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A was in both series considered a constant for the given data curve, imitating the approach used for hot crystallization in Section 5.1.1), indicating that it is the initial value of the rate constant K(T) that is crucial for the course of the crystallization process. However, there was a striking difference between the two sets of the Šesták–Berggren kinetic exponents M and N, where the “E = fix” option provided nearly constant values of both exponents across the whole range of applied q+ (again characteristically corresponding46 to the nucleation-growth kinetics60,61), while the “E = var” option leads to the apparently uncommon M and N values, requiring very high N values and negative M values to compensate for the E–A dissonance. Although the accuracy of the kinetic description was slightly higher for the “E = var” option (see the correlation coefficients shown in Fig. 9F), the evident necessity for the multifold mutual compensation of the kinetic parameters does not testify to the correctness of this mathematical solution. Considering the similar findings being obtained for the MCHL model in Section 5.1.1, a conclusion can be drawn that the implementation of the temperature-dependent Kg(T) or E(T) will require compensation by a corresponding A(T) master curve to provide a suitable, physically meaningful description of the crystallization data. This is, however, left for a future endeavor; in the meantime, the currently used approach, also known as the single-curve multivariate kinetic analysis (sc-MKA),63 based on the present “E = fix” option, will be a viable method for the description of the temperature-dependent kinetic data. Apart from the above-described analysis, it is worth noting that the accurately determined (through utilizing eqn (5)) crystallization enthalpies (see Fig. 9F) are indeed significantly lower during the cold crystallization compared to the hot crystallization (data depicted in Fig. 8F and listed in SI).
A was in both series considered a constant for the given data curve, imitating the approach used for hot crystallization in Section 5.1.1), indicating that it is the initial value of the rate constant K(T) that is crucial for the course of the crystallization process. However, there was a striking difference between the two sets of the Šesták–Berggren kinetic exponents M and N, where the “E = fix” option provided nearly constant values of both exponents across the whole range of applied q+ (again characteristically corresponding46 to the nucleation-growth kinetics60,61), while the “E = var” option leads to the apparently uncommon M and N values, requiring very high N values and negative M values to compensate for the E–A dissonance. Although the accuracy of the kinetic description was slightly higher for the “E = var” option (see the correlation coefficients shown in Fig. 9F), the evident necessity for the multifold mutual compensation of the kinetic parameters does not testify to the correctness of this mathematical solution. Considering the similar findings being obtained for the MCHL model in Section 5.1.1, a conclusion can be drawn that the implementation of the temperature-dependent Kg(T) or E(T) will require compensation by a corresponding A(T) master curve to provide a suitable, physically meaningful description of the crystallization data. This is, however, left for a future endeavor; in the meantime, the currently used approach, also known as the single-curve multivariate kinetic analysis (sc-MKA),63 based on the present “E = fix” option, will be a viable method for the description of the temperature-dependent kinetic data. Apart from the above-described analysis, it is worth noting that the accurately determined (through utilizing eqn (5)) crystallization enthalpies (see Fig. 9F) are indeed significantly lower during the cold crystallization compared to the hot crystallization (data depicted in Fig. 8F and listed in SI).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 ± 7100 K2 for heating from the glass, and ∼360
500 ± 7100 K2 for heating from the glass, and ∼360![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 ± 8500 K2 for cooling from the melt). Since the Kg values are, in the case of isothermal data, fully compensated by ln
100 ± 8500 K2 for cooling from the melt). Since the Kg values are, in the case of isothermal data, fully compensated by ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A, the following kinetic calculations were, for the sake of simplicity, performed with these two Kg values also in the low-T region, bearing in mind the possible distortion of ln
A, the following kinetic calculations were, for the sake of simplicity, performed with these two Kg values also in the low-T region, bearing in mind the possible distortion of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A.
A.
 | ||
| Fig. 10 (A) Determination of Kg from the isothermal DSC crystallization experiments; “iso(cooling)” and “iso(heating)” indicate the cycles, where Ta was reached during the cooling of a melt and during the heating of a glass, respectively. The isothermal data are compared with akin non-isothermal data from Fig. 8A. The inset shows only the zoomed-in isothermal data with their dominant high-T linear parts fit. (B) Temperature dependence of the rate constant K determined in terms of the Hoffman–Lauritzen theory for the two sets of isothermal crystallization data. (C) Selected isothermal crystallization data (points) obtained from the melt-cooling cycles fit by the MCHL model (red lines). (D) Selected isothermal crystallization data (points) obtained from the glass-heating cycles fit by the MCHL model (red lines). (E) and (F) Evolution of different kinetic parameters obtained from the MCHL fit of the isothermal crystallization data. Solid lines are associated with the quantities depicted on the right axes. | ||
The identical Levenberg–Marquardt-based non-linear optimization (as used in Section 5.1.1) was used to fit the two isothermal data series by the MCHL model – corresponding rate constants are shown in Fig. 10B, and examples of the quality of the curve-fitting are shown in Fig. 10C and D. Note the almost one order of magnitude increase of K(T) caused by the extra nucleation time provided in the case of cold crystallization experiments – see Fig. 10B. It is worth noting that the largely enhanced nucleation rate can be also a consequence of the mesomorphic phase being possibly formed during the cooling step. However, apart from the large difference in the Kg (and correspondingly also the equalizing ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A) values, the MCHL kinetic exponents M and N as well as the overall crystallization enthalpies are extremely similar for both types of the isothermal experiments – as evidenced in Fig. 10E and F (full sets of kinetic parameters are listed in SI). The interpretation of the M and N values46 indicates the 3-dimensional interface-controlled crystal growth for both types of isothermal kinetics. The increasing N values observed for high-T annealing following the heating from the glassy state indicate a possible transition towards the diffusion-controlled mechanism (due to the much higher crystallization rate and competition between a large number of active growth sites), rather than the decrease of the morphological dimensionality of the growing crystallites. However the micrographs from Fig. 7 are inconclusive in this regard. Importantly, the isothermal crystallization enthalpy (ΔH–Ta dependence from Fig. 10F) very well corresponds to the akin data from Fig. 9F (considering the shift in the covered temperature ranges), but the isothermal data show a significantly more temperature-distinguished formation of the loosely packed low-T phase with a correspondingly low ΔH.
A) values, the MCHL kinetic exponents M and N as well as the overall crystallization enthalpies are extremely similar for both types of the isothermal experiments – as evidenced in Fig. 10E and F (full sets of kinetic parameters are listed in SI). The interpretation of the M and N values46 indicates the 3-dimensional interface-controlled crystal growth for both types of isothermal kinetics. The increasing N values observed for high-T annealing following the heating from the glassy state indicate a possible transition towards the diffusion-controlled mechanism (due to the much higher crystallization rate and competition between a large number of active growth sites), rather than the decrease of the morphological dimensionality of the growing crystallites. However the micrographs from Fig. 7 are inconclusive in this regard. Importantly, the isothermal crystallization enthalpy (ΔH–Ta dependence from Fig. 10F) very well corresponds to the akin data from Fig. 9F (considering the shift in the covered temperature ranges), but the isothermal data show a significantly more temperature-distinguished formation of the loosely packed low-T phase with a correspondingly low ΔH.
5.2. Kinetic predictions of PDX crystallization
A comparison between these simulations and real experimental measurements was made to test the relevance and accuracy of the kinetic predictions based on different sets of kinetic parameters determined for the crystallization of PDX. The isothermal measurements performed at 0, 25 and 50 °C (under both setups, i.e., with achieving Ta either from the liquid or from the glassy state) were used as the experimental verification; the theoretical predictions were then simulated for these Tas using the sets of kinetic parameters obtained from the following curve-fitted data: non-isothermal cooling from the melt at 0.1 and 7 °C min−1, non-isothermal heating from glass at 0.1 and 50 °C min−1, isothermal annealing after cooling from the melt (denoted, e.g., as “q− ≫ Ta = 0 °C”), and isothermal annealing after heating from glass (denoted, e.g., as “q+ ≫ Ta = 0 °C”). Visualization of the corresponding temperature ranges is shown in Fig. 11. In the case of the non-isothermal data, the two borderline data-curves (the highest and the lowest cooling/heating rates with fully manifesting crystallization peak) were selected for each crystallization pathway; the four crystallization peaks well cover the isothermally investigated range (the Tas of interest are indicated by the dashed lines). The three Tas were selected so that the main difference between the two crystallization pathways (the nucleation-determined crystallization rate; see Fig. 10B) was clearly covered.The kinetic predictions were aimed to test the following hypotheses: (1) the closer the non-isothermal crystallization peak to the predicted Ta, the better the prediction; (2) the “q− ≫ Ta” isothermal predictions are more accurate with the kinetic parameters obtained from the melt-cooling measurements, and the “q+ ≫ Ta” predictions are better based on the glass-heating kinetics; (3) the prediction based on the non-isothermal measurement covering a closer T range will be more accurate than the prediction utilizing the isothermal kinetic parameters but from a distanced Ta; (4) the q−/q− ranges (0.1–50 °C min−1 in the absolute values) explored in the present study are sufficient to provide a reliable basis for all relevant isothermal predictions.
The kinetic predictions for the three selected Tas (0, 25 and 50 °C) are shown in Fig. 12. Starting with the lowest Ta = 0 °C, the theoretically closest non-isothermal measurement is that performed at q+ = 0.1 °C min−1 (during cooling, the crystal growth ceases at linear q− sufficient to reach 0 °C before the crystallization initiates or is fully completed). However, practically all non-isothermal measurements provide in this case very poor predictions, differing by at least an order of magnitude on the time scale – the closest prediction (but still unusable for any practical purpose) is that calculated from the cooling at 7 °C min−1 (i.e., the second closest one in Fig. 11). From the isothermal predictions, the one calculated from “q− ≫ Ta = 50 °C” is quite close to both experimental datasets (the exact “fits” have to be, naturally, ruled out from the comparison) – it is actually closer to the experimental “q+ ≫ Ta = 0 °C” data than is the prediction calculated from the “q− ≫ Ta = 0 °C” kinetic parameters. This is due to the crystallization during cooling obeying the Hoffman–Lauritzen kinetics, where the driving force is comparatively low at very high T (lack of nuclei, low undercooling ΔT) and at very low T (slow diffusion). Therefore, the prediction calculated from the “q− ≫ Ta = 25 °C” parameters significantly deviates from the other two akin calculations. Nonetheless, the match between the two experimental datasets with Ta = 0 °C (“q− ≫ …” and “q+ ≫ …”) is also relatively good, indicating that they can be mutually used to predict each other.
For the isothermal annealing at 25 °C, the difference between the two experimental datasets is already large, as the effect of additional nucleation occurring during the “q+ ≫ …” experiment increases. If we again rule out the exact matches (direct fits of the experimental data), then a very good correspondence occurs only between the experimental “q+ ≫ Ta = 25 °C” data and the prediction based on the “q+ = 50 °C min−1” non-isothermal parameters. A relatively acceptable prediction of the same experimental dataset is also that obtained from the “q+ ≫ 50 °C” parameters; otherwise, relatively poor predictions were obtained, which is quite surprising as Ta = 25 °C lies in the middle of the investigated temperature interval, and should be covered by multiple measured datasets (see Fig. 11). This is most probably due to the inherently T-dependent kinetics as expressed by the curvatures in Fig. 8A and 9A.
In the case of the predictions for isothermal annealing at Ta = 50 °C, a good correspondence is found between the experimental “q− ≫ Ta = 50 °C” data and the prediction based on the “q− ≫ Ta = 0 °C” isothermal parameters (similar case as for the first introduced predictions for Ta = 0 °C). In addition, the experimental “q+ ≫ Ta = 50 °C” data are well predicted by the parameters obtained from the “q+ ≫ Ta = 25 °C” fit. Since the former correlation can be again attributed to the specificity of the Hoffman–Lauritzen kinetics (which accounts for the true non-monotonicity of the K–T dependence), the latter case testifies about the reliability of the polymer crystallization kinetic predictions being mainly associated with the state of nucleation alone: for the “q+ ≫ …” series, the material is expected to be already well nucleated and the constant value of Kg (see Fig. 10A) seems to be the deciding factor for the accuracy of the kinetic predictions. Conversely, although the “q− ≫ …” series also exhibits linearity (constant Kg) in the given temperature region, the added influence of the ongoing nucleation clearly affects the overall crystallization rate exceedingly for the corresponding T-closest kinetic prediction (from “q− ≫ 25 °C”) to be accurate.
6. Conclusions
The crystallization behavior of PDX was investigated under various pathways (during cooling, heating, and isothermal annealing) using DSC measurements complemented by Raman spectroscopy, TGA, and optical microscopy experiments. The continuity of the manifestation of the α′ → α transition as well as the gradually increasing crystallinity observed via Raman measurements (probably due to the lamellae thickening) confirms that no sharp polymorphic transition exists for the PDX crystallization data, which justifies the continuity of the temperature-dependent kinetic modeling. The comparative evaluation of kinetic descriptions revealed that while both the temperature-dependent Hoffman–Lauritzen and Arrhenian approaches can provide good fits to experimental data, their predictive power is strongly dependent on the crystallization pathway and temperature regime. In particular, the melt-crystallization kinetics were governed by the interplay between nucleation and growth processes, leading to non-monotonic dependencies, whereas cold crystallization was more strongly determined by pre-existing nuclei formed during quenching. The overall impact of pre-thermal history is thus demonstrated to be crucial for understanding the difference between the two crystallization pathways.The kinetic modeling highlighted that reliable predictions require not only careful selection of the appropriate theoretical framework but also consistent treatment of temperature-dependent parameters. Attempts to introduce continuous temperature dependence of the key quantities (Kg(T), E(T)) into the models often demanded compensatory adjustments of other parameters, stressing the need for more physically grounded master curve approaches, namely those simultaneously implementing the complementary A(T) dependence. On the other hand, the introduction of the T-dependent exponents of the extended MCHL model was proven to be mandatory for the physically meaningful interpretation of the crystallization kinetics. Furthermore, the present data demonstrated that the accuracy of the kinetic predictions for the isothermal polymer crystallization is dominantly driven by the match of the nucleation states between the training and testing datasets, i.e. the data used to determine the kinetic parameters used for the predictions and the actual reality that is being predicted. Accordingly, the thermal proximity (correspondence between the training and testing temperature ranges) is only of secondary importance. These findings emphasize the limitations of using standard non-isothermal measurements for straightforward prediction of isothermal crystallization, especially at temperatures where nucleation becomes rate-determining. The results suggest that future refinement of universal kinetic models must explicitly integrate the contributions of the temperature-dependent nucleation dynamics to enhance predictive reliability.
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
Supplementary information is available. See DOI: https://doi.org/10.1039/d5cp04749h.The data are available in the FigShare database under DOI: https://doi.org/10.6084/m9.figshare.30138169.
Acknowledgements
This work has been supported by the Ministry of Education, Youth and Sports of the Czech Republic (project LM2023037). The author is grateful to Assoc. Prof. Jana Machotová (University of Pardubice, Czech Republic) for her cordial and greatly inspiring discussions.References
- O. Attallah, A. Janssens, M. Azeem and M. Fournet, Fast, high monomer yield from post-consumer polyethylene terephthalate via combined microwave and deep eutectic solvent hydrolytic depolymerization, ACS Sustainable Chem. Eng., 2021, 9, 17174–17185 CrossRef CAS.
- Y. Ko, J. Hinestroza and T. Uyar, Structural investigation on electrospun nanofibers from postconsumer polyester textiles and pet bottles, ACS Appl. Polym. Mater., 2023, 5, 7298–7307 CrossRef CAS.
- X. Zhao, T. Liao, Y. Lu, Z. Jiang and Y. Men, Formation and distribution of the mesophase in ultrasonic micro-injection-molded isotactic polypropylene, Macromolecules, 2021, 54, 5167–5177 Search PubMed.
- N. Tan, L. Lin, T. Deng and Y. Dong, Evaluating the residual stress and its effect on the quasi-static stress in polyethylene pipes, Polymers, 2022, 14, 1458 Search PubMed.
- S. Lee, S. Yoon, J. Baek and I. Jeon, Preparation of hexene-functionalized graphitic nanoplatelets for effective interaction with nylon 6, J. Reinf. Plast. Compos., 2022, 42, 819–827 CrossRef.
- T. Lee, H. Yu, M. Forrester, T. Wang, L. Shen and H. Liu, et al., Next-generation high-performance bio-based naphthalate polymers derived from malic acid for sustainable food packaging, ACS Sustainable Chem. Eng., 2022, 10, 2624–2633 CrossRef CAS.
- R. Abedi, B. Maher, L. Amirkhani, M. Rezaei and S. Jamshidi, The relationship between chemical microstructure, crystallinity, mechanical properties, and CO2/N2 gases permselectivity of thermoplastic polyurethane membranes, Colloid Polym. Sci., 2024, 302, 1081–1095 CrossRef CAS.
- R. Xu, L. Yin, J. Chen, J. Xie, Z. Yin and C. Lei, The effect of melt drawing ratio on the crystal structural change and performance of polyketone extrusion cast film, J. Polym. Sci., 2022, 60, 2465–2475 Search PubMed.
- M. Xue, K. Lv, S. Gao, X. Lu, Y. Liu and S. Yan, Synergistic effect of thermoplastic phenolic resin and multiwalled carbon nanotubes on the crystallization of polyoxymethylene, J. Polym. Sci., 2020, 58, 997–1010 CrossRef CAS.
- H. Neve, S. Thorat and S. Radhakrishnan, Melt processing behaviour and structure development in recycled pp blends with recycled pet, Prog. Rubber, Plast. Recycl. Technol., 2022, 39, 141–155 CrossRef.
- J. Amaya, Determination of time and concentration conditions affecting polylactic acid (PLA) production, Polymers, 2025, 17, 2009 CrossRef CAS PubMed.
- F. Aliberti, M. Oliviero, R. Longo, L. Guadagno and A. Sorrentino, Effect of crystallinity on the printability of poly(ethylene terephthalate)/poly(butylene terephthalate) blends, Polymers, 2025, 17, 156 CrossRef CAS PubMed.
- P. Kothavade, P. Yadav, A. Gopal, H. Pol, A. Kafi and S. Bateman, et al., Enhancing the crystallization kinetics and mechanical properties of poly(lactic acid) blends for 3D printing application, ACS Appl. Polym. Mater., 2024, 6, 5754–5762 CrossRef CAS.
- F. Santos, B. Hernandez, R. Santos, M. Machado, M. Souza and E. Sousa, et al., Bioabsorbable polymeric stent for the treatment of coarctation of the aorta (coa) in children: a methodology to evaluate the design and mechanical properties of PLA polymer, Materials, 2023, 16, 4403 CrossRef PubMed.
- T. Czelusniak and F. Amorim, Influence of energy density on polyamide 12 processed by sls: from physical and mechanical properties to microstructural and crystallization evolution, Rapid Prototyping J., 2021, 27, 1189–1205 CrossRef.
- C. Zhang, W. Wu, H. Hu, Z. Rui, J. Ye and H. Shên, Effect of multidimensional filler hybridization on the mechanical properties of thermoplastic polyurethane composites prepared by selective laser sintering, J. Appl. Polym. Sci., 2023, 140, e54041 CrossRef CAS.
- W. Han, L. Kong and M. Xu, Advances in selective laser sintering of polymers, Int. J. Extreme Manuf., 2022, 4, 042002 CrossRef CAS.
- A. Vollrath, C. Kretzer, B. Beringer-Siemers, B. Shkodra, J. Czaplewska and D. Bandelli, et al., Effect of crystallinity on the properties of polycaprolactone nanoparticles containing the dual flap/mpegs-1 inhibitor brp-187, Polymers, 2021, 13, 2557 CrossRef CAS PubMed.
- Z. Azhari, P. Smith, S. McMahon, W. Wang and R. Cameron, Modulating drug release from short poly(ethylene glycol) block initiated poly(l-lactide) di-block copolymers, Pharm. Res., 2022, 40, 1697–1707 CrossRef PubMed.
- J. Loskot, D. Jezbera, Z. Zmrhalová, M. Nalezinková, D. Alferi and K. Lelkes, et al., A complex in vitro degradation study on polydioxanone biliary stents during a clinically relevant period with the focus on Raman spectroscopy validation, Polymers, 2022, 14, 938 CrossRef CAS PubMed.
- F. Ronkay, D. Gere, E. Slezák, E. Szabó, G. Marosi and K. Bocz, Recycled pet packaging materials of improved toughness—importance of devitrification of the rigid amorphous fraction, Macromol. Mater. Eng., 2024, 310, 2400219 Search PubMed.
- J. Sun, W. Zhang, F. Li, Q. Zhuo, C. Qin and J. Wang, et al., Effect of heat treatment with alternating tension on microstructure and elastic recovery of poly(trimethylene terephthalate) fibers, Polym. Adv. Technol., 2022, 33, 2425–2433 Search PubMed.
- S. Kanomi, H. Marubayashi, T. Miyata and H. Jinnai, Reassessing chain tilt in the lamellar crystals of polyethylene, Nat. Commun., 2023, 14, 5531 CrossRef CAS PubMed; J. D. Hoffman, G. T. Davis and J. I. Lauritzen, Jr., in Treatise on Solid State Chemistry, ed. N. B. Hannay, Plenum, New York, 1976, vol. 3, p. 497 Search PubMed.
- J. Xia, L. Ni, C. Zeng, B. Wang, R. Zhang and C. Sun, et al., Temperature-Dependent Triple Crystal Polymorphism of Poly(hexamethylene terephthalate): Long-Range Ordered Mesophase Formation and Crystal Structure–Property Relationships, Macromolecules, 2024, 57, 7369–7380 CrossRef CAS; J. D. Hoffman and J. J. Weeks, Melting process and the equilibriummelting temperature of polychlorotrifluoroethylene, J. Res. Natl. Bur. Stand., Sect. A, 1962, 66, 13–28 CrossRef.
- J. Sheng, W. Chen, K. Cui and L. Li, Polymer crystallization under external flow, Rep. Prog. Phys., 2022, 85, 036601 Search PubMed; M. Burger and V. Capasso, Mathematical modelling and simulation of non-isothermal crystallization of polymers, Math. Models Methods Appl. Sci., 2001, 11, 1029–1053 CrossRef.
- F. Peng, R. Cao, H. Sun, Z. Liu, Y. Zhang and T. Xu, et al., Role of Entanglement in Polymer Crystal Growth and Melting: Molecular Dynamics Simulations, Macromolecules, 2024, 57, 10487–10498 Search PubMed; M. Arshad, A novel kinetic approach to crystallization mechanisms in polymers, Polym. Eng. Sci., 2021, 61, 1502–1517 CrossRef CAS.
- I. Tzourtzouklis, P. Kardasis, G. Papageorgiou and G. Floudas, Phase Diagram, Glassy Dynamics and Crystallization Kinetics of the Biobased Polyester Poly(ethylene 2,5-furanoate) (PEF), Macromolecules, 2024, 57, 11395–11406 CrossRef CAS PubMed; R. Svoboda and J. Machotová, Polymer Crystallization: Universal Macroscopic Description via the Autocatalytic Hoffman-Lauritzen Approach, ACS Omega, 2025, 10, 16602–16619 CrossRef PubMed.
- K. Hall, S. Percec, W. Shinoda and M. Klein, Property Decoupling across the Embryonic Nucleus–Melt Interface during Polymer Crystal Nucleation, J. Phys. Chem. B, 2020, 124, 4793–4804 CrossRef CAS PubMed; R. Svoboda and J. Machotová, Correction to: Polymer Crystallization: Universal Macroscopic Description via the Autocatalytic Hoffman-Lauritzen Approach, ACS Omega, 2025, 10, 56870 CrossRef PubMed.
- L. Ni, C. Sun, S. Xu, W. Xiang, Y. Pan and B. Wang, et al., Thermally Induced Phase Transition of Polybutene-1 from Form I′ to Form II through Melt Recrystallization: Crucial Role of Chain Entanglement, Macromolecules, 2023, 56, 1973–1982 CrossRef CAS; J. Šesták, Science of Heat and Thermophysical Studies: A Generalized Approach to Thermal Analysis, Elsevier, Amsterdam, 2005 Search PubMed.
- Y. Liao and Q. Lan, Understanding the Impact of Chain Mobility on Conformational Evolution and Kinetics of Mesophase Formation in Poly(L-lactide) under Low-Pressure CO2″, Polymers, 2024, 16, 1378 CrossRef CAS PubMed; J. Farjas and P. Roura, Modification of the Kolmogorov–Johnson–Mehl–Avrami rate equation for non-isothermal experiments and its analytical solution, Acta Mater., 2006, 54, 5573–5579 CrossRef.
- R. Cao, F. Peng, C. Nie, Y. Zhang, H. Sun and Z. Liu, et al., Primary Nucleation of Polymer Crystal via Particles Fluctuation with Non-Markovian Effect, Macromolecules, 2024, 57, 5979–5990 CrossRef CAS; K. Nakamura, T. Watanabe, K. Katayama and T. Amano, Some aspects of non-isothermal crystallization of polymers—Part I: Relationship between crystallization temperature, crystallinity and cooling conditions, J. Appl. Polym. Sci., 1972, 16, 1077–1091 CrossRef.
- L. Zou and W. Zhang, Molecular Dynamics Simulations of the Effects of Entanglement on Polymer Crystal Nucleation, Macromolecules, 2022, 55, 4899–4906 CrossRef CAS; R. Svoboda, J. Machotová, M. Krbal, D. Jezbera, M. Nalezinková, J. Loskot and A. Bezrouk, Complex thermokinetic characterization of polydioxanone for medical applications: Conditions for material processing, Polymer, 2023, 277, 125978 CrossRef.
- S. Nagarajan, K. Huang, W. Chuang, J. Lin and E. Woo, Thermo-Sensitive Poly(p-dioxanone) Banded Spherulites with Controllable Patterns for Iridescence, J. Phys. Chem. C, 2023, 127, 2628–2638 CrossRef CAS.
- M. Hakim, N. Nahar, M. Saha, S. Islam, H. Reza and S. Sharker, Local drug delivery from surgical thread for area-specific anesthesia, Biomed. Phys. Eng. Express, 2020, 6, 015028 CrossRef PubMed.
- H. Zheng, H. Peng, P. Wang, H. Li, L. Li, Y. Du and G. Lv, In vitro and in vivo evaluation of degradability and biocompatibility of poly(p-dioxanone) hemostatic clips for laparoscopic surgery, J. Appl. Polym. Sci., 2021, 138, 50772 CrossRef CAS.
- F. Zhao, H. Xu, W. Xue, Y. Li, J. Sun and F. Wang, et al., Iodinated poly(p-dioxanone) as a facile platform for x-ray imaging of resorbable implantable medical devices, J. Biomater. Appl., 2020, 35, 39–48 CrossRef CAS PubMed.
- J. Šesták, Šesták–Berggren equation: now questioned but formerly celebrated—what is right, J. Therm. Anal. Calorim., 2017, 127, 1117–1123 CrossRef.
- F. Liu and G. Yang, Effects of anisotropic growth on the deviations from Johnson–Mehl–Avrami kinetics, Acta Mater., 2007, 55, 1629–1639 CrossRef CAS.
- T. Huang and C. Zhou, Surface Diffusion-Controlled Jonhson–Mehl–Avrami–Kolmogorov Model for Hydrogenation of Mg-based Alloys, J. Phys. Chem. C, 2023, 127, 13900–13910 CrossRef CAS.
- M. Castro, F. Domínguez-Adame, A. Sánchez and T. Rodríguez, Model for crystallization kinetics: Deviations from Kolmogorov–Johnson–Mehl–Avrami kinetics, Appl. Phys. Lett., 1999, 2205–2207 CrossRef CAS.
- A. Kolb-Telieps and T. Shu-Song, Investigations to explain the restricted validity of the Avrami-Johnson-Mehl theory for the crystallization of Zr70Ni30, J. Non-Cryst. Solids, 1988, 107, 122–127 CrossRef CAS.
- K. Shirzad and C. Viney, Revisiting time-dependent growth and nucleation rates in the Johnson-Mehl-Avrami-Kolmogorov equation, R. Soc. Open Sci., 2025, 12, 241696 CrossRef PubMed.
- M. Fanfoni and M. Tomellini, Beyond the Kolmogorov Johnson Mehl Avrami kinetics: inclusion of the spatial correlation, Eur. Phys. J. B, 2003, 34, 331–341 CrossRef CAS.
- J. Šesták, Thermophysical Properties of Solids, Their Measurements and Theoretical Analysis, Elsevier, Amsterdam, 1984 Search PubMed.
- R. Svoboda, Johnson-Mehl-Avrami kinetics as a universal description of crystallization in glasses?, J. Eur. Ceram. Soc., 2024, 44, 4064–4082 CrossRef CAS.
- R. Svoboda, On the path to a universal model for crystallization kinetics: Interpretation of the Šesták-Berggren equation in terms of the nucleation-growth concept, J. Eur. Ceram. Soc., 2026, 46, 117716 CrossRef CAS.
- X. Liu, S. Feng, X. Wang, J. Qi, D. Lei, Y. Li and W. Bai, Tuning the mechanical properties and degradation properties of polydioxanone isothermal annealing, Turk. J. Chem., 2020, 44, 1430–1444 Search PubMed.
- K. Ishikiriyama, M. Pyda, G. Zhang, T. Forschner, J. Grebowicz and B. Wunderlich, Heat capacity of poly-p-dioxanone, J. Macromol. Sci., Part B:Phys., 1998, 37, 27–44 Search PubMed.
- M. Lorenzo and M. Righetti, Crystallization-induced formation of rigid amorphous fraction, Polym. Cryst., 2018, 1, e10023 Search PubMed.
- X. Monnier, D. Cavallo, M. Righetti, M. Lorenzo, S. Marina and J. Martín, et al., Physical aging and glass transition of the rigid amorphous fraction in poly(l-lactic acid), Macromolecules, 2020, 53, 8741–8750 CrossRef CAS.
- J. Lee, J. Mangalara and D. Simmons, Correspondence between the rigid amorphous fraction and nanoconfinement effects on glass formation, J. Polym. Sci., Part B: Polym. Phys., 2017, 55, 907–918 CrossRef CAS.
- R. Svoboda and J. Machotová, How Depolymerization-Based Plasticization Affects the Process of Cold Crystallization in Poly(P-Dioxanone), Macromol. Rapid Commun., 2024, 45, 2400369 CrossRef CAS PubMed.
- J. Martín, N. Stingelin and D. Cangialosi, Direct Calorimetric Observation of the Rigid Amorphous Fraction in a Semiconducting Polymer, J. Phys. Chem. Lett., 2018, 9, 990–995 CrossRef PubMed.
- J. Loskot, D. Jezbera, A. Bezrouk, R. Doležal, R. Andrýs, V. Francová, D. Miškář and A. Myslivcová Fučíková, Raman Spectroscopy as a Novel Method for the Characterization of Polydioxanone Medical Stents Biodegradation, Materials, 2021, 14, 5462 CrossRef CAS PubMed.
- M. Jaidann and J. Brisson, Conformation Study of poly(p-dioxanone) fibers by polarized Raman spectroscopy, X-ray diffraction, and conformation analysis, J. Polym. Sci., Part B:Polym. Phys., 2008, 46, 406–417 CrossRef CAS.
- B. Schrader, Infrared and Raman Spectroscopy—Methods and Applications, VCH, Weinheim, Germany, 1995 Search PubMed.
- J. D. Hoffman and J. I. Lauritzen, Jr., Crystallization of bulk polymers with chain folding: theory of growth of lamellar spherulites, J. Res. Natl. Bur. Stand., Sect. A, 1961, 65A, 297–336 Search PubMed.
- K. Levenberg, A method for the solution of certain non-linear problems in least squares, Q. Appl. Math., 1944, 2, 164–168 Search PubMed.
- D. W. Marquardt, An algorithm for least-squares estimation of nonlinear parameters, J. Soc. Ind. Appl. Math., 1963, 11, 431–441 Search PubMed.
- W. A. Johnson and K. F. Mehl, Reaction kinetics in processes of nucleation and growth, Trans. Am. Inst. Min., Metall. Eng., 1939, 135, 416–442 Search PubMed.
- M. Avrami, Granulation, phase change, and microstructure – kinetics of phase change III, J. Chem. Phys., 1941, 7, 177–184 Search PubMed.
- H. E. Kissinger, Reaction kinetics in differential thermal analysis, Anal. Chem., 1957, 29, 1702–1706 CrossRef CAS.
- R. Svoboda, J. Chovanec, S. Slang, L. Beneš and P. Konrád, Single-curve multivariate kinetic analysis: Application to the crystallization of commercial Fe-Si-Cr-B amorphous alloys, J. Alloys Compd., 2022, 889, 161672 CrossRef CAS.
| This journal is © the Owner Societies 2026 |











