 Open Access Article
Open Access ArticlePhotoinduced electron transfer (PET) in ethaline-like solvents
Saqib Rabbani a,
Mohammedulameen Fakhri
a,
Mohammedulameen Fakhri a,
Andy Lin
a,
Andy Lin a,
Sasha Noble
a,
Sasha Noble ab and
John R. Swierk
ab and
John R. Swierk *a
*a
aDepartment of Chemistry, Binghamton University, Binghamton, New York, 13902, USA. E-mail: jswierk@binghamton.edu
bDepartment of Mechanical Engineering, Binghamton University, Binghamton, New York 13902, USA
First published on 27th April 2026
Abstract
Deep eutectic solvents (DESs) are 21st century solvents composed entirely of hydrogen bond donors and acceptors, which form a eutectic solvent having a significantly lower melting point than its individual components. Due to their tunable nature, DESs are widely employed for applications in electrochemistry, nanotechnology, catalysis and synthesis but their intrinsic electron transfer behaviour is largely unexplored. In order to characterize electron transfer in DESs, steady state reductive emission quenching measurements were performed using tris(2,2′-bipyridine)ruthenium(II), [Ru(bpy)3]2+, with a series of quenchers in several different volumetric compositions of ethaline (choline chloride:ethylene glycol) and water. Using Stern–Volmer analysis, rate constant (kq) values for excited state quenching were calculated and compared with diffusion limited rate constant (kD) data. Finally, the electron transfer data were further analyzed by fitting to Rehm–Weller and Marcus theory models in order to provide a theoretical framework for understanding and predicting electron transfer kinetics.
1. Introduction
Solvents are vitally important for chemical processing and chemical reactions.1 They actively govern reaction rates, pathways, and equilibrium by adjusting solvation, stabilization, and diffusion of different species. Without understanding solvent effects, it is hard to predict the kinetics and thermodynamics of reactions.2 However, the extensive use of conventional organic solvents, especially volatile solvents (e.g., dichloromethane) and petroleum derived hydrocarbons (e.g., toluene), poses a significant problem.3 Their toxicity and environmental persistence, combined with high cost, have driven a need to develop greener alternative solvents. Besides this, many organic solvents are extracted from non-renewable resources, which creates a further concern.4,5Deep eutectic solvents (DESs) were introduced by Abbott and co-workers in 2004.6 They are characterized by relatively low melting points and can be easily synthesized by combining a hydrogen bond donor (HBD) and a hydrogen bond acceptor (HBA) under constant heating and stirring. Unlike many conventional solvents, their preparation does not require additional purification steps such as distillation or recrystallization.7 The bypassing of these energy and resource intensive steps limits chemical waste and energy usage and diminishes the use of auxiliary solvents, thereby contributing to the green and sustainable nature of DESs and allowing them to function as alternatives to conventional solvents in chemical and industrial use. Since DESs show similar physicochemical properties to ionic liquids (ILs), DESs are often substituted for ILs in different applications.8 Finally, DESs are non-volatile in nature thus making their storage very convenient.9
Despite similarities, DESs and ILs are markedly different in terms of their preparation and chemical constituents. ILs are formed by the combination of cations and anions, and their syntheses often require high temperatures and extended reaction times.10 In addition, synthesis of ILs typically involves multiple steps, including cation formation followed by anion exchange. Finally, ILs are often volatile, flammable, and/or toxic, which raises significant safety and environmental concerns.11
In contrast, DESs offer several advantages over ILs. First, the non-toxic HBA and HBD can be used; many HBD and HBA are bio-based or inspired by naturally occurring metabolites.12 This makes DESs environmentally friendly and well-suited for green chemistry applications. Moreover, their synthesis is generally straightforward, involving only the mixing of components followed by heating and stirring for a few hours.12 Importantly, purification can be completed on the individual, solid HBD and HBA before DES formation.
The unique physicochemical properties of DESs, such as their polarity, low volatility, and extensive hydrogen bonding networks, make them attractive media for chemical reactions. These features are important in photochemical processes, such as photoinduced electron transfer (PET), where an electron is transferred from an excited-state molecule to an acceptor molecule, leading to emission quenching and reduced emission.13 The tunable solvation environment provided by DESs allows the optimization of PET dynamics by stabilizing charge transfer states and dictating reaction kinetics.14 For example, the polarity and viscosity of DESs can affect the reorganization energy and charge separation efficiency in PET processes.
Electron transfer in ionic liquids (ILs) has been thoroughly investigated over the last two decades,15–26 whereas there is significantly less work on electron transfer in DESs. Pandey and coworkers explored charge transfer through fluorescence quenching with a variety of fluorophores in DESs27–30 but only reported quenching rate constants and did not investigate electron transfer within a theoretical framework. A variety of studies have focused on electron transfer in DESs using electrochemical methods but only reported rates of electron transfer.31–37 Chang, Li and coworkers used computational methods to explore charge transfer from DESs to SO2 and examined how the DESs stabilized the reduced SO2.38 Burda and Sangoro used femtosecond TAS and ionic conductivity to probe the eutectic composition of ethaline and characterize charge transport through the DES.39 Finally, Smith and Goeltz demonstrated that the choice of HBA could be used to shift the potential for oxidation of a catechol-based DES that exhibited proton-coupled electron transfer.40
Understanding electron transfer in DESs is essential for both understanding fundamental electron transfer dynamics and rationally designing DESs for advanced applications in photocatalysis, solar energy conversion, and sensors. However, significant knowledge gaps persist, specifically in the photophysics of chromophores in DESs, the measurement of electron transfer rates, and the application of foundational theories like those of Rehm–Weller and Marcus to DES systems. To address these knowledge gaps, we investigated aqueous DES mixtures on three key aspects: the photophysics of [Ru(bpy)3]2+, the correlation between experimental and theoretical electron transfer rates, and the applicability of Rehm–Weller and Marcus theories in these neoteric solvents.
2. Experimental
2.1 Materials
For this study, choline chloride (>99%), ethylene glycol (>99%), tris(2,2′-bipyridine)ruthenium(II) hexafluorophosphate [Ru(bpy)3]2+ (>97%), methyl viologen dichloride hydrate (MV, >98%), benzyl viologen dichloride (BV, >97%), and ethyl viologen dibromide (EV, >99%) were purchased from Sigma-Aldrich. 1,1′-Diphenyl-[4,4′-bipyridine]-1,1′-diium chloride (DPV, >97%) and 1,1′-diheptyl-4,4′-bipyridinium dibromide (DHB, >98%) were purchased from TCI America. All these reagents were used as received without any further purification.2,11-Dimethyl-6,7-dihydrodipyrido[1,2-a:2′,1′-c]pyrazinediium dibromide (DMPP-DB), 2,12-dimethyl-7,8-dihydro-6Ff-dipyrido[l,2-a:2′,l′-c]-[l,4]diazepinium dibromide (DMDHP-DB), 1,1′,4,4′-tetramethyl-2,2′-bipyridinium diiodide (TMBP-I2), and 6,7,8,9-tetrahydro-2,13-dimethyldipyrido 〈1,2-a:2′,1′-c〉 〈1,4〉 diazocinediium dibromide (THDDP-DB) were prepared according to literature procedures (SI).41,42
2.2 Preparation and characterization of ethaline/water mixtures
For the preparation of ethaline, a DES composed of choline chloride and ethylene glycol, a published procedure was followed.43 Choline chloride and ethylene glycol were mixed in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 ratio in a round bottom flask, heated, and stirred for 1 hour at 80 °C. A transparent, homogeneous and shelf stable DES was obtained. The pure ethaline DES was then subdivided into seven DES/water mixtures: 0%, 10%, 30%, 50%, 70%, 90%, and 100% water. The DES and aqueous DES mixtures were then stored in a glass desiccator to minimize moisture exposure.
2 ratio in a round bottom flask, heated, and stirred for 1 hour at 80 °C. A transparent, homogeneous and shelf stable DES was obtained. The pure ethaline DES was then subdivided into seven DES/water mixtures: 0%, 10%, 30%, 50%, 70%, 90%, and 100% water. The DES and aqueous DES mixtures were then stored in a glass desiccator to minimize moisture exposure.
Viscosity measurements were conducted using an in-house Cannon–Fenske viscometer and a TA Discovery HR30 rheometer. The redox potentials (E1/2) of [Ru(bpy)3]2+ and various quenchers in the DES/water mixtures were measured using cyclic voltammetry. A BioLogic SP-50 potentiostat was used along with glassy carbon as the working electrode, aqueous silver–silver chloride as the reference electrode, and a platinum wire as the counter electrode.
2.3 Quenching studies
Emission and absorption measurements were carried out using a Shimadzu RF-6000 spectrofluorophotometer and a Shimadzu UV-2600 spectrophotometer, respectively. For Stern–Volmer emission (phosphorescence) quenching studies, a 0.026 mM [Ru(bpy)3]2+ solution was prepared. Similarly, for all nine quenchers, a series of stock solutions were prepared and used for electron transfer analysis. A four-sided transparent cuvette was used for analysis and each sample was purged with nitrogen gas for at least 30 minutes to remove any dissolved oxygen. For emission lifetime measurements, a 0.026 mM solution of [Ru(bpy)3]2+ in the DES/water mixtures was prepared and the emission lifetime (τ) was measured using transient emission spectroscopy, as previously described in ref. 44.3. Results and discussion
3.1 Viscosity of DES/water mixtures
The viscosities of the DES/water mixtures were measured (Table 1) and modelled using the Grunberg–Nissan model.45 The model used the ideal logarithmic mixing rule, mole fraction of ethaline and water, and an interaction term, G12, which indicates deviation from ideality (SI).45 The mole fractions, calculated from the volumetric percentages and densities of the ethaline/water mixtures, are listed in Table S1. The experimental viscosities were used in eqn (S1) to compute G12, which was optimized using a non-linear least square regression to minimize the error between the experimental and predicted viscosities.| DES | η (Pa s) | kD × 109 (M−1 s−1) | τ (ns) | Φ | kr × 104 (s−1) | knr × 106 (s−1) |
|---|---|---|---|---|---|---|
| a Literature value of Φ of Ru(Bpy)32+ in water.50 | ||||||
| 0 | 0.000991 | 6.67 | 222 | 0.063a | 28.4 | 4.22 |
| 10 | 0.001092 | 6.05 | 561 | 0.030 | 5.37 | 1.73 |
| 30 | 0.00188 | 3.52 | 647 | 0.027 | 4.18 | 1.50 |
| 50 | 0.00368 | 1.80 | 762 | 0.027 | 3.61 | 1.28 |
| 70 | 0.00853 | 0.775 | 785 | 0.032 | 4.03 | 1.23 |
| 90 | 0.0237 | 0.279 | 861 | 0.031 | 3.57 | 1.13 |
| 100 | 0.0326 | 0.203 | 928 | 0.031 | 3.30 | 1.04 |
The resulting G12 value of 4.052 is positive, indicating significant non-ideal behavior in the ethaline–water mixtures. This non-ideality originates from the strength and nature of intermolecular interactions (e.g., hydrogen bonding, van der Waals forces, and ionic interactions) between the components.46
These interactions result in increased structural rigidity and higher viscosity. This effect is most pronounced at the 50% DES mixture, where G12 reaches a maximum (4.447), reflecting a denser hydrogen bond network and more persistent coordination environments, which increases resistance to flow and therefore causes the strong, nonlinear rise in viscosity, characterized by a sharp rise at high DES concentrations. The Grunberg–Nissan model confirms that the viscosity increase is not a linear sum of the pure component viscosities but is supplemented by strong intermolecular interactions, as revealed by the positive G12 value.47
3.2 Photophysical characterization of [Ru(bpy)3]2+
The emission lifetime (τ) of [Ru(bpy)3]2+ was measured in the ethaline/water mixtures (Fig. S5 and Table 1). The emission lifetime of [Ru(bpy)3]2+ increased from 222 ns in 0% DES to 928 ns in 100% DES. This increased τ reflects slower relaxation processes and stabilization of the excited state as the solvent becomes more viscous.48 The quantum yield of emission (Φ) for [Ru(bpy)3]2+ was also calculated49 using eqn (1) in the DES mixtures (Table 1). [Ru(bpy)3]2+ in 0% DES was used as a standard.50
 | (1) |
The Φ values are 2–3% in all ethaline mixtures suggesting reduced emission efficiency per absorbed photon. These low Φ values agree with previous studies,51,52 and are attributed to the involvement of the non-emissive metal-centered, d–d state (3MC).53 The 3MC state introduces an efficient nonradiative decay pathway that competes directly with emission from the 3MLCT state. Even though the 3MC level lies at higher energy and its steady state population remains negligible, thermally assisted access to this surface allows a fraction of the 3MLCT population to cross into 3MC.54 Because the 3MC state relaxes to the ground state on an ultrafast timescale, any population that reaches it is effectively removed from the emissive manifold.55 This nonradiative leakage pathway lowers the fraction of molecules that decay radiatively, leading to a reduced Φ.
Radiative (kr) and non-radiative (knr) decay constants were calculated using eqn (2a) and (2b) and the results are tabulated in Table 1.56
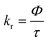 | (2a) |
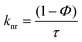 | (2b) |
In eqn (2a) and (2b), Φ and τ represent the quantum yield of emission and emission lifetime. Both kr and knr values dropped by a factor of nine and four, respectively, as the ethaline content was increased. These comparatively larger values of knr as compared to kr suggest that knr is the primary decay mechanism in this case. The decrease in knr is because of increasing viscosity and the structural rigidity of ethaline mixtures, which ultimately suppress the knr. On the other hand, the decrease in the kr is attributed to the unique solvodynamic properties of the ethaline mixtures. The combined effects of a strong hydrogen-bonding network, high ionic strength, and an enhanced refractive index create a restrictive microenvironment that perturbs the emissive 3MLCT state. This environment restricts the electron density redistribution necessary for the radiative transition, thereby attenuating the transition dipole moment and greatly reducing kr. The Φ, which is the fraction of excited molecules that decay via light emission, remains stable or shows a slight decline.
Initially, it is somewhat surprising that the emission lifetime increases while Φ and kr decreases. It is important to note, however, that τ = (kr + knr)−1. Because knr is the primary excited state decay pathway and is ∼2 orders of magnitude larger than kr, the decrease in knr has a more substantial impact on τ and a longer lifetime is observed. In contrast, Φ has more direct dependence on kr so a decrease in this rate constant leads to a smaller value of Φ.
A deeper analysis of [Ru(bpy)3]2+ photophysics in the ethaline/water mixtures was performed by fitting the emission spectra (Fig. S8) using a one mode Franck–Condon analysis.57 This offers a way to probe how the DES/water mixtures influence the metal-to-ligand charge transfer (MLCT) excited state dynamics as the solvent environment transitions from pure water to pure DESs.
The analysis of the emission spectra yielded six key photophysical parameters (Table 2), which were used to calculate four additional parameters: the activation barrier (ΔEact) for 3MLCT to 3MC transition, the solvent reorganization energy (λs), and the inner reorganization energy (λi). The meaning and significance of these parameters are as follows:
| DES % | E0–0 (cm−1) | ℏwm (cm−1) | Δv1/2 (cm−1) | SM | γ0 | ln(Fcalc) | λs (eV) | λi (eV) | λ (eV) | ΔEact | E (Ru3+/Ru2+) V vs. Ag/AgCl |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 405 405 |
1447 | 1851 | 0.7598 | 1.7027 | −28.40 | 0.376 | 0.136 | 0.512 | 0.436 | −0.920 |
| 10 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 377 377 |
1472 | 1860 | 0.7482 | 1.6994 | −28.02 | 0.380 | 0.137 | 0.517 | 0.459 | −0.916 |
| 30 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 357 357 |
1479 | 1845 | 0.7306 | 1.7171 | −28.10 | 0.374 | 0.134 | 0.508 | 0.462 | −0.914 |
| 50 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 342 342 |
1474 | 1839 | 0.7151 | 1.7410 | −28.37 | 0.371 | 0.131 | 0.502 | 0.466 | −0.912 |
| 70 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 321 321 |
1484 | 1824 | 0.7106 | 1.7391 | −28.23 | 0.365 | 0.131 | 0.496 | 0.467 | −0.909 |
| 90 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 284 284 |
1489 | 1814 | 0.7100 | 1.7345 | −28.09 | 0.361 | 0.131 | 0.492 | 0.470 | −0.905 |
| 100 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 261 261 |
1488 | 1806 | 0.6971 | 1.7522 | −28.26 | 0.358 | 0.129 | 0.487 | 0.472 | −0.902 |
• E0–0 (0–0 transition energy): this represents the energy of the MLCT transition between the lowest vibrational levels of the ground and excited states. It reflects how the solvent stabilizes the charge-separated excited state.
• ℏwm (average vibrational mode energy): this indicates the energy of the dominant molecular vibration coupled to the electronic transition, providing a measure of vibronic coupling strength.
• Δv1/2 (spectral width) and λs (solvent reorganization energy): the full width at half maximum (Δv1/2) of the spectrum reports on the broadening due to solvent interactions. It is directly used to calculate λs, which quantifies the energy required for the solvent to reorganize around the newly created excited-state dipole.
• SM (Huang–Rhys factor) and λi (inner reorganization energy): the Huang–Rhys factor (SM) describes the strength of coupling between the electronic transition and specific molecular vibrations. The inner reorganization energy (λi) is derived from these factors and represents the energy required to distort the molecular structure upon excitation.
• ln(Fcalc) (the Franck–Condon factor or Frank–Condon vibrational overlap term) and γ0 (Linewidth parameter): the Franck–Condon factor ln (Fcalc) governs the probability of the non-radiative transition back to the ground state, linking directly to the energy gap law. The γ0 parameter accounts for the Gaussian broadening of individual vibronic lines.
As the percentage of DESs increases, the E0–0 values decrease steadily indicating a reduction in energy gap (Table 2). As the ground state potential remains constant (see below), this indicates a stabilization of the 3MLCT state relative to the ground state with increasing ethaline content. Fig. 2 shows the relationship between knr and E0–0, which exhibits a positive slope (0.0079).58
The positive slope between knr and E0–0 indicates that the non-radiative decay of [Ru(bpy)3]2+ in ethaline/water mixtures is not governed by direct decay to the ground state but by a thermally activated decay (TAD) mechanism.53 In this mechanism, the molecule gains thermal energy from the surroundings to reach a higher energy, non-emissive metal-centered, d–d state (3MC) from the emissive 3MLCT. The metal-centered state then rapidly decays to the ground state.59,60 This transition requires crossing an activation barrier (ΔEact) that must be thermally overcome for the population transfer from 3MLCT to the 3MC state.61 TAD can be modelled by:62
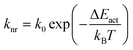 | (3a) |
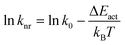 | (3b) |
ΔEact = (ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) k0 − ln k0 − ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) knr) × kBT knr) × kBT
| (3c) |
For Ru(II) complexes, k0 is typically ∼1013–1014 s−1.63 This model allows the calculation of ΔEact at room temperature by using eqn (3c). The calculated ΔEact values are given in Table 2. As the content of ethaline increases, the value of ΔEact also increases. This can be attributed to ethaline's moderate polarity9 and hydrogen bonding,64 which can stabilize the 3MLCT state (demonstrated by the decreasing value E0–0). In turn, this increases the barrier (ΔEact) to the 3MC state and ultimately reduces knr. The increase in ℏwm values with increasing DES concentration also indicates that the ethaline's hydrogen bond donors are restricting the bipyridine vibrational modes. This may be via a specific interaction such as bonding between the OH groups of ethylene glycol and the nitrogen atoms on bipyridine.65
We can also calculate the solvent reorganization energy for the excited state, λs, from the Δv1/2 of the charge-transfer emission band, using a modified Gaussian Marcus–Hush equation (eqn (4)).66
 | (4) |
In this equation, kB is the Boltzmann constant, h is Planck's constant, and c is the speed of light. Vath et al. used this approach to extract λs values from CT absorption/emission bandwidths across different solvents and temperatures.67 Their methodology justifies linking the observed thermal broadening directly to the solvent outer-sphere reorganization energy. Both Δv1/2 and λs values decrease as the solvent goes from a water-rich to an Ethaline-rich environment, which can be linked to the ethaline mixture's increasing viscosity. The increased viscosity can restrict the solvent's reorientation and reduce thermal broadening.
We can also calculate the inner reorganization energy (λi) using SM, which quantifies the ratio of structural distortion by identifying the shift between ground and excited MLCT state potential energy surfaces along vibrational modes. λi is the energy required for structural rearrangement within a molecule as it moves from the excited state to the ground state during emission and can be calculated by using eqn (5).68
| λi = SM × ℏwm | (5) |
In eqn (5), SM stands for the Huang–Rhys Factor and ℏwm stands for the average vibrational mode energy.
For [Ru(bpy)3]2+, λi basically corresponds to vibrations of the bipyridine (bpy) ligand, such as ring stretches and Ru–N bond stretches, that are associated with electron transfer processes.69 The decrease in SM leads to a decrease in λi values and indicates less of a shift between ground and excited state potential energy surfaces with increasing DES concentration. This implies that the bipyridine ligands encounter less geometric distortion in the MLCT state as the ethaline's content is being increased. We suggest that this stems from ethaline's hydrogen bonding stabilizing the bpy− moiety and thus helps delocalize the charge and limits structural changes. The calculation of solvent reorganization (λs) and inner reorganization energy (λi) allows the quantification of total reorganization energy (λ) for emission by the following relationship:70,71 λ = λs + λi. As expected, λ also decreases linearly with increasing DES concentration (Table 2).
The plot between SM and E0–0 shows a linear trend (Fig. 3), which indicates a systematic relationship between the energy gap of the MLCT excited state and the degree of vibronic coupling. This is influenced by the solvent environment. The combined influence exerted by the solvent on E0–0 (electronic stabilization) and SM (vibronic coupling) is evident as the increase in ethaline content enhances both structural solvation and stabilizing dipole–dipole and hydrogen bonding interactions. This linearity also highlights the gradual transition from water's polar and dynamic solvation to ethaline's ionic and viscous environment, which stabilizes the 3MLCT state (lower E0–0) and reduces bpy distortion (lower SM). The observed correlation between ln(knr) and E0–0 should therefore not be interpreted as evidence of rigid, parallel shifts of the potential energy surfaces. Rather, E0–0 reflects stabilization of the emissive 3MLCT minimum, while knr is governed by the barrier-controlled access to the 3MC surface, which is strongly influenced by local solvation, viscosity, and hydrogen-bond interactions characteristic of deep eutectic solvents.
The Frank–Condon factor which is also called the Frank–Condon vibrational overlap term, ln(Fcalc), counts the vibrational overlap between the ground and excited states during the MLCT emission processes. The negative values observed for ln(Fcalc) mean a small Franck–Condon factor and indicate poor vibrational overlap between the ground and excited states. This poor overlap greatly suppresses knr according to the energy gap law, which is also the primary factor for prolonging τ. This effect is reinforced in ethaline-rich mixtures, where strong ionic and hydrogen bonding interactions rigidify the solvation shell and distort the excited-state geometry,14 further diminishing the vibrational overlap and potentially perturbing the electronic transition dipole strength.
The plot of ln(Fcalc) as a function of ln(knr) is shown in Fig. 4.
This plot illustrates the energy gap law, showing how the rate of non-radiative decay rises in proportion to the Franck–Condon factor, a measure of vibronic overlap that governs this process. In the energy gap law, knr is modelled as follows:
| knr ≈ AFcalc | (6a) |
ln(knr) ≈ ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) A + ln A + ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fcalc Fcalc
| (6b) |
In this case, the slope is set to 1, as knr is directly proportional to Fcalc and the intercept is ln(A), where A is the pre-exponential factor associated with vibrationally induced electronic coupling.56 From the slope of Fig. 4, ln(A) ≈ 42.45 and thus A ≈ 2.73 × 1018 s−1, which suggests that ln(Fcalc) accounts for both the vibrational overlap and the energy barrier to the 3MC state. This extremely large coupling factor (∼1018 s−1) suggests strong electronic coupling between 3MLCT and 3MC surfaces. This magnitude is above the typical vibrational frequency range of ∼1012–1014 for MLCT complexes like [Ru(bpy)3]2+ and further confirms the idea that the nonradiative decay does not follow a simple energy gap law pathway to the ground state.58,72 Instead, the excited state deactivation proceeds through a thermally activated transition involving the 3MLCT to 3MC crossing. Finally, we note that in the present analysis A should not be interpreted as a bare vibrational attempt frequency. In our formulation, Fcalc is not a pure Franck–Condon overlap term, but an effective factor that also incorporates the energetic barrier and multidimensional nuclear reorganization associated with accessing the 3MLCT to 3MC crossing region. Consequently, the pre-factor A represents an effective coupling parameter that absorbs electronic coupling, density of accepting states, and deviations from a single-mode harmonic approximation. Similar large pre-factors are also known in the photophysical literature as well.56,72
The Gaussian linewidth parameter, γ0, reports on the emission band broadening for [Ru(bpy)3]2+ in ethaline mixtures coming from low frequency vibrational modes and solvation inhomogeneity. This can be calculated using eqn (7):58
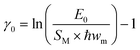 | (7) |
The values of γ0 are gradually increasing with increased DES content (Table 2), indicating enhanced ionic and hydrogen bonding interactions with the solvent. This also indicates increasing static disorder and low frequency mode coupling, which results in broadening of vibronic bands.
Finally, the excited state potentials of [Ru(bpy)3]2+ in all ethaline/water mixtures were calculated using the following Rehm–Weller relationship.73
| E(A+/A*) = E(A+/A−) − E0–0 | (8) |
In this equation, E(A+/A−) corresponds to the ground state reduction potential and E0–0 is the difference in energy between ground and excited state energies. E0–0 can be calculated from emission spectra fitting and the values are listed in Table 2. The ground state reduction potential can be determined using cyclic voltammetry. The voltammogram of [Ru(bpy)3]2+ in ethaline mixtures and reduction potentials are given in Fig. S9 and Table S2, respectively. Using all these data, the excited state potentials are calculated using eqn (8) and the corresponding values are given in Table 2. We observe a slight decrease of 18 mV in the excited state potential increasing from 0% to 100% ethaline.
3.3 Photoinduced electron transfer
In order to understand electron transfer in the DES mixtures, we carried out emission quenching studies using [Ru(bpy)3]2+ and a series of viologens quenchers (Fig. 1). The absorbance spectra of [Ru(bpy)3]2+ was similar in all the DES mixtures with an absorbance maxima at ∼454 nm. The emission spectra of [Ru(bpy)3]2+ in all the DES mixtures, with and without quenchers, are presented in Fig. S19–S27. | ||
| Fig. 1 Photosensitizers and quenchers used in the study. [Ru(bpy)3]2+, MV, EV, THDDP-DB, BV, DHB, DMPP-DB, DPV, DMDHP-DB, and TMBP-I2. | ||
 | ||
| Fig. 2 The plot of nonradiative decay ln (knr) vs. transition energy (E0–0) with a slope of 7.86 × 10−3 ± 2.30 × 10−3, a y-intercept of −114.123 ± 37.596 and a R2 of 0.6998, based on the data given in Table 2. | ||
 | ||
| Fig. 3 The plot of Huang–Rhys factor (SM) vs. transition energy (E0–0) with a slope of 4.20 × 10−4 ± 6.81 × 10−5, a y-intercept of −6.135 ± 1.113 and a R2 of 0.8836, based on the data given in Table 2. | ||
 | ||
| Fig. 4 The plot of Franck–Condon overlap factor ln (Fcalc) vs. nonradiative decay ln (knr) with a slope fixed at 1, a y-intercept of −42.45 ± 9.24 × 10−14 and a R2 of 1, based on the data given in Table 2. | ||
The kinetics of emission quenching can be quantified using the Stern–Volmer relationship:74
 | (9) |
 | (10) |
Here, I0 is the emission intensity in the absence of a quencher, I is the emission intensity in the presence of a quencher, Q is the concentration of a quencher, τ0 is the lifetime of the chromophore, Ksv is the Stern–Volmer constant (L mol−1), and kq is the quenching rate constant (M−1 s−1). Ksv can be derived by plotting I0/I vs. [Q] (Fig. S28–S36 and Table 3). The quenching rate constants (kq) were also calculated and are given in Table 3. In the DES mixtures, the kq values are between 3.8 × 106 and 2.9 × 108 M−1 s−1, which are smaller compared to water and other conventional solvents. When compared to quenching in ionic liquids, the values of kq in ethaline/water mixtures are similar or larger.75–80
| MV | BV | EV | DPV | DHB | DMPP-DB | DMDHP-DB | THDDP-DB | TMBP-I2 | |
|---|---|---|---|---|---|---|---|---|---|
| DES% | Ksv (L mol−1) | ||||||||
| 0 | 322 | 619 | 404 | 1061 | 648 | 415.3 | 208.2 | 52.87 | 18.61 |
| 10 | 1223 | 1222 | 1059 | 1881 | 1356 | 1051.2 | 393.03 | 177.05 | 45.98 |
| 30 | 950 | 1058 | 1008 | 1712 | 1229 | 982.7 | 387.5 | 146.5 | 40.15 |
| 50 | 730 | 797 | 711 | 1359 | 817 | 840.4 | 281.4 | 103.2 | 35.08 |
| 70 | 416 | 415 | 443 | 1194 | 492 | 460.7 | 171.15 | 62.3 | 25.33 |
| 90 | 233 | 189 | 209 | 755 | 214 | 185.7 | 74.51 | 36.7 | 14.04 |
| 100 | 115.76 | 110.22 | 138.68 | 270.1 | 124.61 | 110.1 | 38.2 | 35.5 | 5.96 |
| kq × 109 (L mol−1 s−1) | |||||||||
| 0 | 1.45 | 2.79 | 1.82 | 4.78 | 2.92 | 1.87 | 0.94 | 0.24 | 0.084 |
| 10 | 2.18 | 2.18 | 1.89 | 3.35 | 2.42 | 1.87 | 0.7 | 0.32 | 0.082 |
| 30 | 1.47 | 1.64 | 1.56 | 2.65 | 1.9 | 1.52 | 0.59 | 0.25 | 0.062 |
| 50 | 0.96 | 1.05 | 0.93 | 1.78 | 1.07 | 1.10 | 0.37 | 0.135 | 0.046 |
| 70 | 0.53 | 0.53 | 0.56 | 1.52 | 0.63 | 0.59 | 0.22 | 0.0079 | 0.032 |
| 90 | 0.27 | 0.22 | 0.24 | 0.88 | 0.25 | 0.22 | 0.087 | 0.0043 | 0.007 |
| 100 | 0.13 | 0.12 | 0.15 | 0.29 | 0.14 | 0.12 | 0.042 | 0.0038 | 0.006 |
To better understand the kq values, the diffusion limited rate constants (kD) were calculated using the Smoluchowski equation:22
 | (11) |
Here, R is the ideal gas constant (J K−1 mol−1), T is the temperature (K), and η is the viscosity (Pa s). Experimental viscosities and diffusion limited rate constant (kD) of aqueous DES mixtures are given in Table 1. As expected, with increasing viscosity, the value of kD decreases (Fig. 5).
 | ||
| Fig. 5 The plot of viscosity (η) and diffusion limited rate constant (kD) as a function of DES concentration. | ||
The data reveal that kq values decrease as the DES concentration is increased. This could be because of an increase in the overall viscosities of DES mixtures. Alternatively, a decrease in dipolarity or polarizability of the solvent with increasing DES concentration may hinder or slow down the charge transfer between the quenchers and [Ru(bpy)3]2+.81 Water molecules disrupt the hydrogen bond network of ethaline, which alters the molecular arrangement and weakens cohesive forces, leading to a reduction in solvent viscosity with increasing water.82 Due to the high viscosity of DES mixtures, slower solvation might create a more constrained environment around the chromophore. Consequently, this may influence the accessibility of the molecules acting as electron acceptors or donors in the electron transfer process, potentially leading to decreased rates of electron transfer.83 Except for DPV, the kq values for all DES mixtures and quenchers were less than kD, demonstrating that electron transfer was not diffusion controlled. These comparatively smaller kq values when compared to kD are typical for viscous liquids.23,84 This trend can be attributed to variance between the recorded “macroviscosity” of each DES mixture and the “macroviscosity” in the immediate vicinity of the donor–acceptor pair. While bulk viscosity values (Table 1) obtained experimentally show the average resistance to flow for the entire ethaline/water mixture, they do not necessarily show the local dynamic environment immediately surrounding the solute (donor–acceptor pair) molecule. Especially, in DESs, hydrogen bonding networks, ionic clustering, and microheterogeneity can create domains in which solute molecules experience a viscosity that differs greatly as compared to the bulk.14 As a result, in such cases, diffusion, solvation, and electron transfer processes are controlled by local (micro)viscosity rather than the bulk viscosity.85 Therefore, the viscosity of the DES mixtures (Table 1) may not report the local environment and diffusion of donor–acceptor pairs within the DES mixtures.23
The observed kq data for the electron transfer between [Ru(bpy)3]2+ and MV in the ethaline DES (∼1.3 × 108 L mol−1 s−1) can be placed into the context by comparison with literature values in other solvent environments. In conventional molecular–solvent media such as acetonitrile and water, significantly higher rates have been documented (for example, water: ∼1.7 × 109 L mol−1 s−1; buffer: ∼4.6 × 108 L mol−1 s−1), indicating nearly diffusion-controlled or only modestly solvent-hindered quenching.86,87 In contrast, ionic-liquids (IL) frequently show much lower kq values (in the 107 L mol−1 s−1 range) despite their relatively high viscosity, which is attributed to suppressed diffusion, strong ion-pairing and structured solvation shells.88–90 The somewhat higher kq in the neat DES system lies between the IL and molecular-solvent extremes, indicating that while the DES network imposes mobility restrictions (via high viscosity, extensive hydrogen-bonding and ionic framework) it still allows more efficient donor–acceptor encounters compared with classic ILs.
3.4 Rehm–Weller analysis and Marcus theory
Using the kq values determined from Stern–Volmer analysis, the electron transfer can be modelled by both Rehm–Weller91 and Marcus theories.92 The Rehm–Weller analysis gives a simple thermodynamic model to predict if an electron transfer reaction involving an excited molecule is energetically feasible. By comparing the energy of the excited state to the redox potentials of the reacting molecules, it calculates the free energy change (ΔG) for the process.91 Marcus theory on the other hand is a kinetic model and predicts that the rate of an electron transfer reaction does not increase indefinitely with increased thermodynamic driving forces (−ΔG0), but instead follows a parabolic curve.92 This means that the rate initially increases in the normal region before reaching a maximum and then decreasing in the inverted region as the driving force becomes increasingly larger.92The electron transfer data in Table 3 was fit using both Rehm–Weller and Marcus analyses, with the corresponding plots shown in Fig. 6 and Fig. S37. Individual plots are shown in Fig. S38 and S39. In the Rehm–Weller equation,22
| ΔGet = 23.06(EDox − EAred) − E0,0 − S | (12) |
 | ||
| Fig. 6 Experimental electron transfer data plotted using the Rehm–Weller model for thermodynamic analysis. | ||
A plot of log kq vs. ΔG yields information about kmaxdiff, which represents the diffusion-controlled electron transfer limit, and λ, which is the total reorganization energy for electron transfer. Together, the two parameters give an idea about how much structural and solvent reorientation is required for electron transfer to take place. Also, the solvation or coulombic stabilization factor, S reports on if ion-pair stabilization in solvent impacts the observed rates of electron transfer. The calculated ΔGET values are listed in Table S3 of the SI file and ΔGET data reveal a systematic influence of the DES composition on photoinduced electron transfer. For highly reducible electron acceptors, such as DPV and DHB, ΔGET remains highly negative across all DES mixtures, indicating that the corresponding electron transfer reactions are strongly exergonic and likely occur near the diffusion-controlled limit. In contrast, for weaker acceptors such as THDDP-DB and TMBP–I2, ΔGET values approach zero at low DES content and become progressively more negative with increasing DES fraction, suggesting that the ionic environment of the DES enhances charge separation by stabilizing the radical ion pair. The parameters derived from log kq vs. ΔG plot are given in Table 4.
| DES% | λ (kcal mol−1) | λ (eV) | kmaxdiff × 1011 (M−1 s−1) | S |
|---|---|---|---|---|
| 0 | 12 | 0.520 | 7.00 | 2.8 |
| 10 | 11.5 | 0.499 | 5.00 | 2.8 |
| 30 | 11 | 0.477 | 3.00 | 2.8 |
| 50 | 10.3 | 0.447 | 2.00 | 2.0 |
| 70 | 11 | 0.477 | 1.00 | 1.4 |
| 90 | 11.7 | 0.507 | 0.90 | 0.8 |
| 100 | 11.8 | 0.512 | 0.50 | 0.8 |
The λ for electron transfer exhibits a non-monotonic trend across the ethaline/water mixtures. The value of λ decreases from 12.0 kcal mol−1 (0% DES) to a minimum of 10.3 kcal mol−1 (50% DES) and then increases again to 11.8 kcal mol−1 (100% DES). This ∼14% variation reflects solvent-dependent effects on solvent reorganization and intramolecular rearrangements, driven by ethaline's increasing viscosity and ionic interactions. The minimum at 50% DES corresponds to enhanced intermolecular interactions, as evidenced by the elevated G12 interaction factor from the Grunberg–Nissan viscosity model, and is consistent with the non-ideal mixing discussed earlier. The subsequent rise in λ at a higher ethaline content suggests stronger hydrogen-bonding and ionic solvation in ethaline mixtures, increasing the energy required for solvent reorganization.93,94 On the other hand, kmaxdiff decreases considerably with increasing ethaline content, which can be attributed to a significant increase in viscosity and reduced molecular diffusion in the ethaline-rich environment. Finally, the S factor also decreases linearly with ethaline content, showing that the ethaline's environment is partially stabilizing the charge-separated state and reducing the contribution of solvent reorganization to the driving force.
Similarly, the electron transfer data were also analysed using the Marcus equation (eqn (S2), SI).95 ΔG for electron transfer (Table S4) shows some dependence on the ethaline content and tends to become less exergonic with increased ethaline content. A comparison between ΔG and λ data (Table S5) splits quenchers into different Marcus kinetic regions. The quenchers with less exergonic or endergonic ΔG values such as TMBP-I2 (∼0.01 to 0.06 eV), THDDP-DB (−0.02 to 0.01 eV) and DMDHP-DB (−0.20 to −0.04 eV) can be classified in the normal region across all ethaline compositions. These quenchers show reduced quenching efficiency because of positive or near-zero ΔG, demanding a greater driving force to approach the barrierless region. On the other hand, for quenchers such as BV, MV, EV, and DMPP-DB, the ΔG values are approaching −λ and thus are near a barrierless reaction. In this region, experimental kq values are approaching optimal electron transfer efficiency with nominal activation barriers. Finally, the most exergonic quenchers such as DPV (−0.46 to −0.65 eV) and DHB (−0.35 to −0.53 eV) fall in the inverted region especially at higher ethaline concentrations (90 and 100% DESs). In these solvent concentrations, ΔG < −λ and kq values are decreasing, probably because of vibronic overlap, even after with a large driving force for electron transfer. This is most obvious in the 100% ethaline concentration where ΔG becomes more negative and the λ value is reduced.
Between the two models, the values of λ are in good agreement with a difference of 1–2 kcal mol−1 between the two models. Comparing the data for λ obtained in neat ethaline, 10.14–11.8 kcal mol−1, the values in DESs are substantially smaller than the 17.7–18.5 kcal mol−1 range reported for Ru(bpy)-related complexes in ethanol–water mixtures, a reduction of roughly 35–43%.86,96 The reduced λ values are mechanistically important and reflect a reduced outer-sphere (solvent) reorganization contribution.
4. Conclusions
The photophysical characterization of [Ru(bpy)3]2+ in ethaline/water mixtures demonstrates that the chromophore has a significantly stabilized 3MLCT state with pronounced luminescence, extended excited state lifetimes, and suppressed non-radiative decay. These effects can be credited to the strong ionic and hydrogen bonding network of ethaline, which effectively rigidifies the chromophores and stabilize its charge transfer state.The unique ethaline environment also influences electron transfer dynamics. The experimental data are well described by both the Rehm–Weller and Marcus models. Rehm–Weller analysis shows that the thermodynamics and intrinsic reorganization aspects of electron transfer are largely unaffected by the specific ethaline/water mixture but the rates of electron transfer are strongly controlled by the viscosity and solvation properties of ethaline, revealing a dual role of ethaline in controlling both energetics and dynamics of electron transfer. The Marcus model demonstrates a shift from the normal to the inverted region for high driving force electron transfer. The data also suggest that ethaline/solvent mixtures could be used to modulate the rate of electron transfer and introduce selectivity in the electron transfer dynamics.
The bimolecular electron transfer rate (kq) of [Ru(bpy)3]2+ with MV in ethaline DESs (1.3 × 108 L mol−1 s−1) is significantly higher than that in typical ILs, comparable to moderate acetonitrile values, but lower than that in water or phosphate buffer. This reflects the intermediate viscosity and structured hydrogen bonding network of ethaline, which slows diffusion relative to water but promotes efficient charge transfer through stabilization of the encounter complex. In addition, the reorganization energies obtained for the [Ru(bpy)3]2+ to MV electron transfer in neat ethaline, 10.14 and 11.8 kcal mol−1 from Marcus and Rehm–Weller models, are notably smaller than the values commonly reported in ethanol–water mixtures. This reduction indicates that the ethaline environment imposes a lower solvent reorganization penalty, consistent with its structured hydrogen bonded and ionic network. A smaller λ supports more efficient electron transfer once the encounter complex is formed.
The conclusions drawn in this work are strictly valid for ethaline and are expected to be most transferable to structurally similar hydrogen-bond-donor-based DESs, rather than universally applicable to all DES classes. Broadly speaking, the data demonstrate that increasing viscosity from ethaline is not solely impacting electron transfer dynamics. Instead, the solvation environment also plays a role in stabilizing the charge transfer state. In addition, it is clear that electron transfer behaviour, generally in DESs and specifically in ethaline, distinctly differs from electron transfer in ILs and that the two solvent systems should be treated independently with respect to charge transfer. The ability to increase excited state stability, while providing tuneable solvent properties to control electron transfer reactions make ethaline a highly promising solvent for electron transfer applications.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary data provides additional details on the following: synthesis of quenchers, viscosity modeling, photophysical characteriation data for [Ru(bpy)3]2+, electrochemical characterization [Ru(bpy)3]2+ and quenchers, and Stern-Volmer quenching data. See DOI: https://doi.org/10.1039/d5cp04727g. 2+, electrochemical characterization [Ru(bpy)3]2+ and quenchers, and Stern-Volmer quenching data.Notes and references
- V. Hessel, N. N. Tran, M. R. Asrami, Q. D. Tran, N. Van Duc Long, M. Escribà-Gelonch, J. O. Tejada, S. Linke and K. Sundmacher, Green Chem., 2022, 24, 410–437 RSC.
- J. J. Varghese and S. H. Mushrif, React. Chem. Eng., 2019, 4, 165–206 RSC.
- D. R. Joshi and N. Adhikari, J. Pharm. Res. Int., 2019, 28, 1–18 CrossRef.
- F. Pena-Pereira, A. Kloskowski and J. Namieśnik, Green Chem., 2015, 17, 3687–3705 RSC.
- A. A. Quintana, A. M. Sztapka, V. d. C. Santos Ebinuma and C. Agatemor, Angew. Chem. Int. Ed, 2022, 61, e202205609 CrossRef CAS PubMed.
- A. P. Abbott, D. Boothby, G. Capper, D. L. Davies and R. K. Rasheed, J. Am. Chem. Soc., 2004, 126, 9142–9147 CrossRef CAS PubMed.
- B. B. Hansen, S. Spittle, B. Chen, D. Poe, Y. Zhang, J. M. Klein, A. Horton, L. Adhikari, T. Zelovich, B. W. Doherty, B. Gurkan, E. J. Maginn, A. Ragauskas, M. Dadmun, T. A. Zawodzinski, G. A. Baker, M. E. Tuckerman, R. F. Savinell and J. R. Sangoro, Chem. Rev., 2021, 121, 1232–1285 CrossRef CAS PubMed.
- G. García, S. Aparicio, R. Ullah and M. Atilhan, Energy Fuels, 2015, 29, 2616–2644 CrossRef.
- Q. Zhang, K. De Oliveira Vigier, S. Royer and F. Jérôme, Chem. Soc. Rev., 2012, 41, 7108–7146 RSC.
- R. Ratti, Adv. Chem., 2014, 2014, 1–16 CrossRef.
- J. Płotka-Wasylka, M. de la Guardia, V. Andruch and M. Vilková, Microchem. J., 2020, 159, 1–12 CrossRef.
- R. Germani, M. Orlandini, M. Tiecco and T. Del Giacco, J. Mol. Liq., 2017, 240, 233–239 CrossRef CAS.
- M. Natali, S. Campagna and F. Scandola, Chem. Soc. Rev., 2014, 43, 4005–4018 RSC.
- S. Chatterjee, T. Chowdhury and S. Bagchi, J. Phys. Chem. B, 2024, 128, 12669–12684 CrossRef CAS PubMed.
- N. Siraj, G. Grampp, S. Landgraf and K. Punyain, Z. Phys. Chem., 2013, 227, 105–119 CrossRef CAS.
- H. V. R. Annapureddy and C. J. Margulis, J. Phys. Chem. B, 2009, 113, 12005–12012 CrossRef CAS PubMed.
- H. Y. Fu, Z. G. Xing, X. Y. Cao and G. Z. Wu, Chin. Sci. Bull., 2013, 58, 1882–1886 CrossRef CAS.
- A. Paul and A. Samanta, J. Phys. Chem. B, 2007, 111, 1957–1962 CrossRef CAS PubMed.
- C. L. Bentley, A. M. Bond, A. F. Hollenkamp, P. J. Mahon and J. Zhang, J. Phys. Chem. C, 2014, 118, 22439–22449 CrossRef CAS.
- C. L. Bentley, J. Li, A. M. Bond and J. Zhang, J. Phys. Chem. C, 2016, 120, 16516–16525 CrossRef CAS.
- D. Behar, P. Neta and C. Schultheisz, J. Phys. Chem. A, 2002, 106, 3139–3147 CrossRef CAS.
- R. C. Vieira and D. E. Falvey, J. Phys. Chem. B, 2007, 111, 5023–5029 CrossRef CAS PubMed.
- A. Skrzypczak and P. Neta, J. Phys. Chem. A, 2003, 7800–7803 CrossRef CAS.
- G. Kodis, M. Z. Ertem, M. D. Newton and D. V. Matyushov, J. Phys. Chem. Lett., 2022, 13, 3297–3303 CrossRef CAS PubMed.
- H. Y. Lee, J. B. Issa, S. S. Isied, E. W. Castner, Y. Pan, C. L. Hussey, K. S. Lee and J. F. Wishart, J. Phys. Chem. C, 2012, 116, 5197–5208 CrossRef CAS.
- M. Liang, A. Kaintz, G. A. Baker and M. Maroncelli, J. Phys. Chem. B, 2012, 116, 1370–1384 CrossRef CAS PubMed.
- A. Pandey, A. Yadav and S. Pandey, J. Lumin., 2017, 183, 494–506 CrossRef CAS.
- D. Dhingra, A. Pandey and S. Pandey, J. Phys. Chem. B, 2019, 123, 3103–3111, DOI:10.1021/acs.jpcb.9b01193.
- D. Dhingra, Bhawna, A. Pandey and S. Pandey, J. Phys. Chem. B, 2020, 124, 4164–4173 CrossRef CAS PubMed.
- D. Dhingra, Bhawna, A. Pandey and S. Pandey, J. Phys. Chem. B, 2020, 124, 4164–4173 CrossRef CAS PubMed.
- F. Zhen and P. Hapiot, ChemElectroChem, 2022, 9, 1–6 Search PubMed.
- D. Shen, M. B. Vukmirovic and R. Akolkar, J. Electrochem. Soc., 2019, 166, E526–E532 CrossRef CAS.
- S. Fryars, E. Limanton, F. Gauffre, L. Paquin, C. Lagrost and P. Hapiot, J. Electroanal. Chem., 2018, 819, 214–219 CrossRef CAS.
- P. Qian, G. Cao, M. Muñoz, J. Vura-Weis, B. E. Gurkan, Z. Peng and J. Rodríguez-López, Anal. Chem., 2025, 97, 16239–16249 CrossRef CAS PubMed.
- C. A. Nkuku and R. J. LeSuer, J. Phys. Chem. B, 2007, 111, 13271–13277 CrossRef CAS PubMed.
- A. Renjith and V. Lakshminarayanan, J. Phys. Chem. C, 2018, 122, 25411–25421 CrossRef CAS.
- F. Zhen, L. Percevault, L. Paquin, E. Limanton, C. Lagrost and P. Hapiot, J. Phys. Chem. B, 2020, 124, 1025–1032 CrossRef CAS PubMed.
- S. Spittle, I. Alfurayj, B. B. Hansen, K. Glynn, W. Brackett, R. Pandian, C. Burda and J. Sangoro, JACS Au, 2023, 3, 3024–3030 CrossRef CAS PubMed.
- H. Li, Y. Chang, W. Zhu, C. Wang, C. Wang, S. Yin, M. Zhang and H. Li, Phys. Chem. Chem. Phys., 2015, 17, 28729–28742 RSC.
- P. J. Smith and J. C. Goeltz, J. Phys. Chem. B, 2017, 121, 10974–10978 CrossRef CAS PubMed.
- A. V. Dolganov, L. A. Klimaeva, E. E. Muryumin, S. G. Kostryukov, A. S. Kozlov, O. V. Tarasova and A. V. Knyazev, Russ. J. Phys. Chem. A, 2023, 97, 2307–2313 CrossRef CAS.
- C. Campa, J. Camps, J. Font and P. De March, J. Org. Chem., 1987, 52, 521–525 CrossRef CAS.
- A. R. Harifi-Mood and R. Buchner, J. Mol. Liq., 2017, 225, 689–695 CrossRef CAS.
- B. G. Stevenson, E. H. Spielvogel, E. A. Loiaconi, V. M. Wambua, R. V. Nakhamiyayev and J. R. Swierk, J. Am. Chem. Soc., 2021, 143, 8878–8885 CrossRef CAS PubMed.
- R. Haghbakhsh, A. R. C. Duarte and S. Raeissi, Molecules, 2021, 26, 1–19 CrossRef PubMed.
- D. Lapeña, L. Lomba, M. Artal, C. Lafuente and B. Giner, Fluid Phase Equilib., 2019, 492, 1–9 CrossRef.
- L. He, S. Su, Y. Zhao and J. Long, J. Mol. Model., 2023, 29, 101 CrossRef CAS PubMed.
- A. Vyšniauskas, I. López-Duarte, N. Duchemin, T. T. Vu, Y. Wu, E. M. Budynina, Y. A. Volkova, E. Peña Cabrera, D. E. Ramírez-Ornelas and M. K. Kuimova, Phys. Chem. Chem. Phys., 2017, 19, 25252–25259 RSC.
- J. Hu and C. Y. Zhang, Anal. Chem., 2013, 85, 2000–2004 CrossRef CAS PubMed.
- A. M. Brouwer, Pure Appl. Chem., 2011, 83, 2213–2228 CrossRef CAS.
- J. Van Houten and R. J. Watts, J. Am. Chem. Soc., 1976, 98, 4853–4858 CrossRef CAS.
- K. Suzuki, A. Kobayashi, S. Kaneko, K. Takehira, T. Yoshihara, H. Ishida, Y. Shiina, S. Oishi and S. Tobita, Phys. Chem. Chem. Phys., 2009, 11, 9850–9860 RSC.
- M. Borgwardt, M. Wilke, I. Y. Kiyan and E. F. Aziz, Phys. Chem. Chem. Phys., 2016, 18, 28893–28900 RSC.
- A. Islam, N. Ikeda, A. Yoshimura and T. Ohno, Inorg. Chem., 1998, 37, 3093–3098 CrossRef CAS.
- J. P. Sauvage, J. P. Collin, J. C. Chambron, S. Guillerez, C. Coudret, V. Balzani, F. Barigelletti, L. De Cola and L. Flamigni, Chem. Rev., 1994, 94, 993–1019 CrossRef CAS.
- K. R. Barqawi, Z. Murtaza, T. J. Meyer, C. Hill and N. Carolina, J. Phys. Chem., 1991, 47–50 CrossRef CAS.
- K. Hanson, M. K. Brennaman, A. Ito, H. Luo, W. Song, K. A. Parker, R. Ghosh, M. R. Norris, C. R. K. Glasson, J. J. Concepcion, R. Lopez and T. J. Meyer, J. Phys. Chem. C, 2012, 116, 14837–14847 CrossRef CAS.
- J. V. Caspar and T. J. Meyer, J. Phys. Chem., 1983, 87, 952–957 CrossRef CAS.
- Q. Sun, S. Mosquera-vazquez, H. A. Goodwin, E. Vauthey and A. Hauser, J. Am. Chem. Soc., 2013, 135, 13660–13663 CrossRef CAS PubMed.
- Q. Sun, B. Dereka, E. Vauthey, M. L. Daku and A. Hauser, Chem. Sci., 2017, 223–230 RSC.
- I. M. Dixon, J.-L. Heully, F. Alary and P. I. P. Elliott, Phys. Chem. Chem. Phys., 2017, 19, 27765–27778 RSC.
- D. Hernández-Castillo, R. E. P. Nau, M. A. Schmid, S. Tschierlei, S. Rau and L. González, Angew. Chem., Int. Ed., 2023, 62, 1–7 Search PubMed.
- J. Van Houten and R. J. Watts, J. Am. Chem. Soc., 1976, 98, 4853–4858 CrossRef CAS.
- A. Pandey, Bhawna, D. Dhingra and S. Pandey, J. Phys. Chem. B, 2017, 121, 4202–4212 CrossRef CAS PubMed.
- A. M. Wright, A. A. Howard, J. C. Howard, G. S. Tschumper and N. I. Hammer, J. Phys. Chem. A, 2013, 117, 5435–5446 CrossRef CAS PubMed.
- N. S. Hush, Intervalence-Transfer Absorption, Part 2. Theoretical Considerations and Spectroscopic Data, in Progress in Inorganic Chemistry, ed. F. A. Cotton DOI:10.1002/9780470166093.ch7.
- P. Vath and M. B. Zimmt, J. Phys. Chem. A, 2000, 104, 2626–2633 CrossRef CAS.
- O. A. Stasyuk, A. J. Stasyuk, M. Solà and A. A. Voityuk, J. Nanostructure Chem., 2024, 14, 293–306 CrossRef CAS.
- J. Bossert and C. Daniel, Coord. Chem. Rev., 2008, 252, 2493–2503 CrossRef CAS.
- S. Chattopadhyay, M. Mukherjee, B. Kandemir, S. E. J. Bowman, K. L. Bren and A. Dey, Chem. Sci., 2021, 12, 11894–11913 RSC.
- S. Jakobsen, K. V. Mikkelsen and S. U. Pedersen, J. Phys. Chem., 1996, 100, 7411–7417 CrossRef CAS.
- E. M. Kober, J. V. Caspar, R. S. Lumpkin and T. J. Meyer, J. Phys. Chem., 1986, 3722–3734 CrossRef CAS.
- R. J. Forster and T. E. Keyes, J. Phys. Chem. B, 1998, 102, 10004–10012 CrossRef CAS.
- H. S. Geethanjali, D. Nagaraja and R. M. Melavanki, J. Mol. Liq., 2015, 209, 669–675 CrossRef CAS.
- M. Álvaro, E. Carbonell, B. Ferrer, H. Garcia and J. R. Herance, Photochem. Photobiol., 2006, 82, 185 CrossRef PubMed.
- R. Lomoth, T. Häupl, O. Johansson and L. Hammarström, Chem. – Eur. J., 2002, 8, 102–110 CrossRef CAS PubMed.
- B. Wu, M. Liang, M. Maroncelli and E. W. Castner, J. Phys. Chem. B, 2015, 119, 14790–14799 CrossRef CAS PubMed.
- B. Wu, M. Liang, N. Zmich, J. Hatcher, S. I. Lall-Ramnarine, J. F. Wishart, M. Maroncelli and E. W. Castner, J. Phys. Chem. B, 2018, 122, 2379–2388 CrossRef CAS PubMed.
- C. M. Gordon and A. J. McLean, Chem. Commun., 2000, 1395–1396 RSC.
- M. Z. Hoffman, F. Bolletta, L. Moggi and G. L. Hug, J. Phys. Chem. Ref. Data, 1989, 18, 219–543 CrossRef CAS.
- A. Pandey, R. Rai, M. Pal and S. Pandey, Phys. Chem. Chem. Phys., 2014, 16, 1559–1568 RSC.
- S. Barani Pour, J. Jahanbin Sardroodi, A. Rastkar Ebrahimzadeh, G. Pazuki and V. Hadigheh Rezvan, Sci. Rep., 2024, 14, 1–18 Search PubMed.
- M. Chakraborty, T. Ahmed, R. S. Dhale, D. Majhi and M. Sarkar, J. Phys. Chem. B, 2018, 122, 12114–12130 CrossRef CAS PubMed.
- D. Y. Chu and J. K. Thomas, Macromolecules, 1990, 23, 2217–2222 CrossRef CAS.
- Y. Bai, P. Sun, M. Zhang, Z. Gao, Z. Yang and Y. Shao, Electrochim. Acta, 2003, 48, 3447–3453 CrossRef CAS.
- T. Hamada, S. Tanaka, H. Koga, Y. Sakai and S. Sakaki, Dalton Trans., 2003, 692–698 RSC.
- J. R. Darwent and K. Kalyanasundaram, J. Chem. Soc., Faraday Trans. 2, 1981, 77, 373–382 RSC.
- C. M. Gordon and A. J. McLean, Chem. Commun., 2000, 1395–1396 RSC.
- A. J. McLean, M. J. Muldoon, C. M. Gordon and I. R. Dunkin, Chem. Commun., 2002, 1880–1881 RSC.
- M. Álvaro, E. Carbonell, B. Ferrer, H. Garcia and J. R. Herance, Photochem. Photobiol., 2006, 82, 185–190 CrossRef PubMed.
- S. Farid, J. P. Dinnocenzo, P. B. Merkel, R. H. Young, D. Shukla and G. Guirado, J. Am. Chem. Soc., 2011, 133, 11580–11587 CrossRef CAS PubMed.
- T. P. Silverstein, J. Chem. Educ., 2012, 89, 1159–1167 CrossRef CAS.
- W. W. Parson, J. Phys. Chem. B, 2021, 125, 7940–7945 CrossRef CAS PubMed.
- P. K. Ghorai and D. V. Matyushov, J. Chem. Phys., 2006, 124, 1–18 CrossRef PubMed.
- R. A. Marcus, Pure Appl. Chem., 1997, 69, 13–29 CrossRef CAS.
- C. D. Clark and M. Z. Hoffman, J. Phys. Chem., 1996, 3654, 14688–14693 CrossRef.
| This journal is © the Owner Societies 2026 |
