Open Access Article
Open Access ArticlePicosecond time-resolved investigation of NO3˙ in PUREX solvent
Amel
Zorai
 *a,
Daniel
Adjei
*a,
Daniel
Adjei
 a,
Mireille
Benoît
a,
Sergey
Denisov
a,
Mireille
Benoît
a,
Sergey
Denisov
 a,
Philippe
Moisy
a,
Philippe
Moisy
 b,
Jacqueline
Belloni
b,
Jacqueline
Belloni
 a and
Mehran
Mostafavi
a and
Mehran
Mostafavi
 *a
*a
aInstitut de Chimie Physique, UMR 8000 CNRS, Université Paris-Saclay, Bâtiment 349, Rue Magat, 91405 Orsay, France. E-mail: amel.zorai@universite-paris-saclay.fr
bCEA, DES, ISEC, DMRC, Université de Montpellier, Bagnols-sur-Ceze 30207, France
First published on 18th March 2026
Abstract
The early phenomena induced by the irradiation of solvents used in the PUREX process are investigated using picosecond electron pulse radiolysis. Various concentrations of solvent components – namely nitric acid (HNO3), tributyl phosphate (TBP), dodecane, and, to a lesser extent, water are studied, in absence of the spent nuclear fuel. The transient optical absorption spectra at 5 ps constitute of the spectra of solvated electron in TBP and NO3˙ radical, characterized by absorption band with maxima at 640 nm and 670 nm, as well as a band at 440 nm. The yield of each species is determined. While the concentrations of HNO3 and TBP of the environments exhibit opposing trends, the highest NO3˙ yield is recorded in a solution of 1.9 M HNO3/3.3 M TBP/0.8 M H2O (GTotal(NO3˙) = 3.2 × 10−7 mol J−1). This yield arises from both the direct radiolytic effect on HNO3 and the ultrafast indirect effect of energy transfer—via excitation and ionization—from TBP and H2O to HNO3, which act as the primary precursors of NO3˙. Replacement of part of TBP by dodecane has little influence because they behave similarly. Despite the complexity of the system, the partial dose absorbed by each component is used to unravel the mechanism of NO3˙ formation and yields corresponding to direct and indirect effects across the range of concentrations studied. Interestingly, at highest HNO3 concentration the absorbance and yield of NO3˙ is lower. At longer time within a few hundred nanoseconds, the decay of the NO3˙ radical follows pseudo-first-order kinetics and it is attributed to a hydrogen-atom transfer from TBP and dodecane to NO3˙. But the rate constant of this reaction is very low (0.9 × 106 L mol−1 s−1) and, if the spent fuel were present, the oxidation by NO3˙ of uranium and plutonium ions would be expected to be predominant.
Introduction
The PUREX process is the cornerstone of modern nuclear fuel reprocessing, designed to recover valuable fissile materials, primarily uranium and plutonium, from spent nuclear fuel. This process operates in a highly complex chemical environment, further complicated by the intense internal irradiation from radionuclides, which introduces unique physical chemistry challenges.1,2A critical component of the PUREX process is nitric acid (HNO3), which serves two essential functions: it dissolves the spent fuel and regulates the redox potential of the solution; and also plays a pivotal role in separation chemistry by facilitating the formation of extractable neutral complexes, such as U(VI) (UO2(NO3)2) and Pu(IV) (Pu(NO3)4). Tri-n-butyl phosphate (TBP), used as an extractant diluted in alkane at 30% (vol), is particularly effective because it can extract these neutral complexes, as well as molecular nitric acid and water, into the organic phase. The PUREX process efficiently separates uranium and plutonium from the fission products generated during nuclear reactions.3 However, its defining characteristic is that all reactions occur simultaneously with intense auto-radiolysis of the medium, driven by the highly radioactive elements present.
The spent nuclear fuel being radioactive, the solvent used for the extraction of uranium and plutonium during the PUREX process is subjected to some radiolytic degradation. Consequently, the irradiation impact on nitric acid and TBP solutions not only challenges the stability and effective performance of the PUREX process, but also underscores the need to control radiolytic effects to ensure efficient nuclear fuel reprocessing.
Understanding how radiation affects these chemical systems is crucial for optimizing the PUREX process and ensuring the long-term sustainability of nuclear fuel management. The amplitude and mechanism of such degradation have been investigated separately on each component of the medium. Previous pulse radiolysis studies have examined the influence of radiation on either neat nitric acid or TBP separately. That was for the first time, the formation of the nitrate radical NO3˙ was observed using picosecond pulse radiolysis of concentrated aqueous nitric acid solutions.4–9 The radical yield was found to vary with acid concentration, suggesting that both direct ionization and indirect formation by positive hole H2O+˙ trapping by NO3− contribute to the radical formation during the electron pulse. Subsequent studies confirmed that NO3˙ formation in aqueous nitric acid under ionizing radiation is primarily driven by direct effects and rapid electron transfer, with additional contributions from OH˙ radical reactions over longer timescales and at elevated temperature.10,11 Investigations into the ultrafast radiolytic behavior of neat TBP identified key transient species, such as the TBP-solvated electron (e−TBP) and the triplet excited state (3TBP*).12 Another study emphasized that water molecules preferentially solvate and stabilize excess electrons, potentially influencing electron-induced dealkylation during actinide separation.13
However, the behavior of the nitrate radical in the TBP or TBP/dodecane organic phases remains unexplored. This study aims to investigate the formation and decay of NO3˙, whose properties are expected to significantly influence the extraction reactions of uranium and plutonium in the PUREX process. The aim of this study is to determine, despite the complexity of the environment containing undissociated nitric acid, TBP, dodecane and water, the relative contributions of direct and indirect radiation effects leading to the formation of NO3˙. Notably, the solutions used here are close models of the PUREX environment, but they are free of radioactive elements and therefore auto-radiolysis is not expected. Instead, the solutions are irradiated using an external picosecond electron beam source.
Experimental methods
Preparation of mixtures
The properties of HNO3 are: MW = 63 g mol−1 (neat HNO3 concentration = 15.68 M), ρHNO3 = 1.4 g mL−1, pKa = −1.37, of TBP (C12H27PO4) are: MW = 266 g mol−1, ρTBP = 0.972 g mL−1, with a dielectric constant around 9 at 25 °C, and of dodecane (C12H26) are: MW = 170.3 g mol−1, ρ(C12H26) = 0.745 g mL−1, dielectric constant ≈ 3.14 TBP and dodecane are slightly polar solvents. High-purity TBP (≥99%) and dodecane (≥99%) were purchased from Sigma Aldrich. Nitric acid (>69%) was purchased from Fischer Scientific. Two protocols were applied to prepare series of nitric acid solutions similar to the organic phase immediately after extraction as used in the PUREX process, one with TBP, the other one with TBP and dodecane.Series 1. Using the liquid–liquid extraction method, a volume of 100 mL of TBP was brought into contact with 100 mL of deionized water at room temperature. An identical procedure was followed for contacting 100 mL of TBP with 100 mL of nitric acid (>69%), TBP immediately turned to yellow (Fig. SI 1) due to the formation of nitrogen dioxide (NO2) and mostly dibutyl phosphate (HDBP) and monobutylphosphate (H2MBP).15–17
These procedures were repeated three times to ensure complete contact between TBP and both water and nitric acid. The organic TBP solutions obtained after this separation (corresponding to the PUREX extraction) were used for subsequent dilutions. Six organic solutions with various nitric acid concentrations [HNO3] were prepared, in particular at 4.5 M that is intermediate in the range of 3–6 M used in the PUREX process. The final water concentration [H2O] was at 0.8 or 1.5 M. These concentrations were based on findings from the referenced paper.18 The final concentrations of the different reagents are summarized in Table SI 1. Only a very small part of nitric acid is dissociated into NO3− nitrate and H3O+, because of the low TBP and dodecane dielectric constants and of the low water concentration.
Series 2 involves diluting TBP in an organic solvent, with dodecane selected for this purpose. A 100 mL sample of TBP was contacted with 100 mL of nitric acid. The extracted solution was then combined with varying amounts of pure TBP and dodecane. Dodecane was chosen as the organic diluent due to its inert nature and compatibility with TBP in the PUREX process. As a hydrocarbon, dodecane is chemically stable, non-reactive with nitric acid or TBP, and crucially, it remains unreactive under irradiation.19 Additionally, its low polarity ensures that it efficiently separates the aqueous and organic phases without interfering with the extraction process. This allows for better control over the distribution of nitric acid and TBP, enabling accurate adjustments in concentration and facilitating the study of extraction dynamics.20 Water and nitric acid concentrations were maintained constant.
The concentration of nitric acid was determined according to the previously described method, and that of water by using the Karl Fischer coulometric titration.18 That of TBP without dodecane was obtained from the complementary volume or with dodecane from initial amounts before extraction.
Pulse radiolysis measurements
Ultra-short electron pulses with a typical width of 5–10 ps are produced by the ELYSE platform based on excitation of a CsTe photocathode using a femtosecond laser pulses at the frequency of 5 Hz.21,22The sample optical cells, made of synthetic fused silica, have optical path length l = 0.5 cm. They have 200 µm thick optical windows to minimize contributions from transient species generated in the quartz cell by the electron beam.
For picosecond pump–probe experiments using optical absorption, the broad band from 355 to 750 nm is generated by focusing part of the fundamental laser beam into a CaF2 crystal, and is split 60/40 into probe and reference paths. Both beams are coupled into optical fibers and sent to a spectrometer for analysis using a cooled CCD camera.
For the detection of the optical absorption signals of transient species in the picosecond to microsecond range, the probe beam is provided by a home-made repetitive lamp and analyzed by a streak camera. The signal absorbances measured may be as low as a few %.
The reference dose Dw was determined from the absorbance at 640 nm of the hydrated electron in neat water.22 The doses absorbed by each component of HNO3/TBP/H2O/C12H26 solutions were calculated by considering their specific electronic density (Tables SI 1 and SI 3). The calculated doses (in J L−1) absorbed by HNO3, TBP, H2O, C12H26, and the corresponding dose fractions f of the total dose are given for the various dose fractions f of the total dose are given for the various concentrations in Fig. 1(a), (b), and Tables SI 1 and SI 3 according to the following equations:
| f(HNO3) = Dose(HNO3)/Total dose | (1) |
| f(HNO3) + f(TBP) + f(H2O) + f(C12H26) = 1 | (2) |
Results and discussions
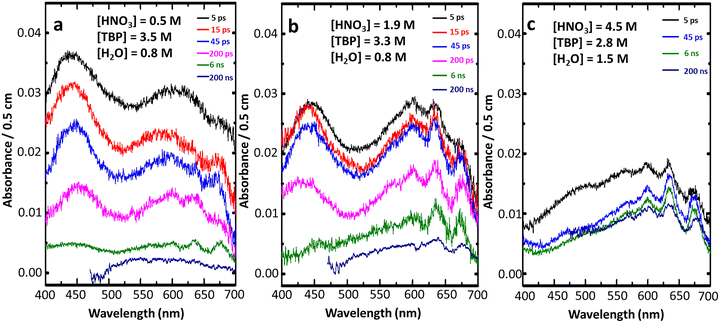
The spectra-kinetics data constitute a large matrix of data for the absorbance at different times and at different wavelengths. The experiments have been twice repeated. Remarkably, despite the low values of the measured absorbances, the uncertainty on their average values is less than ±5%.The time-evolution of the transient optical absorption spectra is presented in Fig. 2 for the HNO3 concentrations of 0.5, 1.9, and 4.5 M.
In solution containing 4.5 M HNO3, the band with peaks at 640 and 670 nm (Fig. 2(c)) correspond fairly well to the radical NO3˙. However, a very weak absorbance of another species is also observed around 440 nm.
The initial spectra at 5 ps for 0.5 and 1.9 M HNO3 (Fig. 2(a) and (b)) are more complex because around 640 nm the shape differs from the radical NO3˙ spectrum. Additionally, an absorption band is also observed around 440 nm and another one in the range 700–780 nm, increases to the near infrared where the NO3˙ radical does not absorb (Fig. 2). It is assigned to e−TBP.12
Fig. 3 presents the kinetics at the wavelengths 440, 640 and 780 nm for the medium with 1.9 M HNO3/3.3 M TBP/0.8 M H2O. Clearly, the rate differences in the decays at these wavelengths (Fig. 3) indicate that three distinct species are early induced by the ultra-short pulse. Note that in neat TBP (HNO3 free)12 the observed transient spectra are initially constituted of the very intense e−TBP spectrum (ε(e−TBP)780nm = 9800 L mol−1 cm−1).12 It overlaps the much weaker band of the longer lived excited state TBP* which is observed alone only in the nanosecond range.
 | ||
| Fig. 3 Kinetics at 440, 640, 780 nm for 1.9 M HNO3/3.3 M TBP/0.8 M H2O. Dose per pulse is Dw = 100 Gy. Optical path l = 0.5 cm. | ||
In 0.5 and 1.9 M HNO3 solutions, the band present in near infrared, where NO3˙ does not absorb, is thus assigned to the electron solvate in TBP as the major component, e−TBP. The absorbance around 640 nm, typically observed in 4.5 M HNO3 environment is assigned to NO3˙.
The 440 nm band, though absorbing in the same range as the less intense one observed in neat TBP,12 belongs to a different species which will be discussed further.
Note that the e−solv spectrum extends also in the visible range down to 400 nm and that it overlaps both other bands.
Primary radiation effects on the components
The radiation energy absorbed within the pulse by the electrons of the molecules constituting the mixture is responsible for the species formation. The major part of it is absorbed by the TBP. However, some fraction is also directly absorbed by the other components HNO3 and H2O, proportionally to their electronic density that increases with their concentration (Fig. 1 and Table SI 1). Therefore, the four components of the medium are directly excited and ionized by their irradiation partial dose, as in neat nitric acid,11 TBP,12 water,23 and dodecane:14| HNO3 ⇝ HNO3˙, HNO3+˙ + e− | (3) |
| TBP ⇝ TBP*, TBP+˙ + e− | (4) |
| H2O ⇝ H2O*, H2O+˙ + e− | (5) |
| C12H26 ⇝ C12H26*, C12H26+˙ + e− | (6) |
| e−→ e−solv | (7) |
Deconvolution of the optical absorption spectrum at 5 ps
The spectral shape/intensity of the early radiation-induced short-lived species in the solutions with 0.5 and 1.9 M HNO3, must be first quantitatively separated in order to allow their shape and yield determination.In solutions either with 0.5 and 1.9 M HNO3, the absorbances due to e−TBP, A(e−TBP)5ps, are obtained at 780 nm (where it alone absorbs), from initial values in kinetics (Fig. 3 and Table SI 2). Then the corresponding partial absorbances of e−TBP at 640 and 440 nm are derived from its known absorption spectrum,12 normalized at the 780 nm values (Table SI 2). The absorbances of the other band at 440 and 640 nm are obtained separately by subtraction of A(e−TBP)5ps from the measured total absorbances (Table SI 2).
A complete and quantitative deconvolution of the measured spectrum at 5 ps for the 1.9 M HNO3 solution is presented in Fig. 4. The e−TBP spectrum is obtained by a normalization of the published spectrum at A(e−TBP)5ps,780nm = 0.012 (Table SI 2 and Fig. 4). The NO3˙ spectrum is determined via the normalization at the maximum value of A(NO3˙)5ps,640nm = ATotal,5ps,640nm − A(e−TBP)5ps,640nm = 0.0213 (Table SI 2 and Table 1, Fig. 4).
 | ||
| Fig. 4 Deconvolution of the observed spectrum at 5 ps for solution containing 1.9 M HNO3 (Fig. 2) into the transient spectra of e−TBP, NO3˙ and the 440 nm band, in the range of 400 to 650 nm. Optical path l = 0.5 cm. | ||
| [HNO3] (M), (f) | 0.5, (0.029) | 1.9, (0.11) | 4.5, (0.25) |
| [TBP] (M), (f) | 3.5, (0.955) | 3.3, (0.875) | 2.84, (0.726) |
| [H2O] (M), (f) | 0.8, (0.015) | 0.8, (0.015) | 1.5, (0.025) |
| A 780 (e−)TBP (0.5 cm), at 5 ps | 0.017 | 0.012 | 0 |
| G(e−)TBP (10−7 mol J−1) at 5 ps | 0.35 | 0.24 | 0 |
| A 640(NO3˙) (0.5 cm) at 5 ps | 0.016 | 00213 | 0.018 |
| G(NO3˙)Total (10−7 mol J−1) at 5 ps | 2.47 | 3.2 | 2.7 |
| G(NO3˙)Direct (10−7 mol J−1) at 5 ps | 3.4 | 3.4 | 3.4 |
| A 640(NO3˙) Direct effect at 5 ps | 0.00064 | 0.00247 | 0.0058 |
| A 640(NO3˙) Indirect effect at 5 ps | 0.0154 | 0.0189 | 0.0122 |
| G(NO3˙)Indirect (10−7 mol J−1) at 5 ps | 2.4 | 3.17 | 2.46 |
Finally, the 440 nm band is obtained from the difference between the overall measured spectrum and those of e−TBP and of the NO3˙ radical (Fig. 4). The end-of-pulse absorbance values at 440 nm are given in Table SI 2. Note that the maximum wavelength of the total absorbance is slightly shifted in time from 440 to 450 nm when the part due to e−solv vanishes.
Though all three spectra overlap, particularly that of e−TBP in the whole visible domain, the spectra of the 640 and 440 nm bands do not overlap each other at their respective maxima.
The radiolytic initial yield G(e−TBP) = A(e−TBP)5ps,780nm/(l × ε(e−TBP) × Dose) is very low and decreases from 0.35 × 10−7 mol J−1 at 0.5 M HNO3 to 0.24 × 10−7 mol J−1 at 1.9 M HNO3 and 0 at 4.5 M HNO3 (Fig. 3 and Table 1), as compared to the value of 1.6 × 10−7 mol J−1 at 5 ps in neat TBP.12 In fact, the reactions of solvated electrons with HNO3/NO3− are very fast and e−solv is mostly scavenged within the pulse:
| e−solv + HNO3 (NO3−) → HNO3−˙(NO32−˙) | (8) |
Radical NO3˙ yields
The NO3˙ absorbance values at 5 ps and 640 nm A640(NO3˙) = A640(measured) − A640(e−TBP) (Table SI 2 and Fig. 4) and the corresponding concentrations (with ε640 = 1350 L mol−1 cm−1 and ε670 = 1080 L mol−1 cm−1)8 are given in Table 1.The specific formation processes of the radical NO3˙ result in part from the direct-effect of the radiation absorption by HNO3 (reaction (1)) and the fast dissociation of HNO3* and HNO3+˙ (reactions (9)–(11)), that are expected to increase proportionally to the partial dose absorbed by HNO3 or (NO3˙) (Table SI 1):
| HNO3* → NO3˙ + H˙ | (9) |
| HNO3* → NO2 + ˙O˙ | (10) |
| HNO3+˙ → NO3˙ + H+ | (11) |
Each component of the medium also absorbs directly radiation depending on its electron fraction (Fig. 1 and Table SI 1). The primary species concentrations are proportional to these fractions f. The NO3˙ radical may be also induced by the ultrafast excitation energy or charge transfer from the primary radiolytic species of the other components, i.e. TBP and H2O induced by the respective partial dose (Table SI 1) as indirect effects.
| HNO3 + TBP* → HNO3* + TBP → NO3˙ + H˙ + TBP | (12) |
| HNO3 + TBP+˙ → HNO3+˙ + TBP → NO3˙ + H+ + TBP | (13) |
| HNO3 + C12H26* → HNO3* + C12H26 → NO3˙ + H˙ + C12H26 | (14) |
| HNO3 + C12H26+˙→ HNO3+˙ + C12H26 → NO3˙ + H+ + C12H26 | (15) |
| HNO3 + H2O* → HNO3* + H2O → NO3˙ + H˙ + H2O | (16) |
| H2O+˙ + H2O → OH˙ + H3O+ | (17) |
| HNO3 + OH˙ → NO3˙ + H2O | (18) |
| TBP + OH˙ → TBP(–H)˙ + H2O | (19) |
G(NO3˙)Total = f(NO3˙) × G(NO3˙)Direct + f(TBP) × G(NO3˙)Indirect![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) TBP + f(C12H26) × G(NO3˙)Indirect TBP + f(C12H26) × G(NO3˙)Indirect![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) C12H26 + f(H2O) × G(NO3˙)Indirect C12H26 + f(H2O) × G(NO3˙)Indirect![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O H2O | (20) |
We may first remark that the value of GTotal(NO3˙) = 3.2 × 10−7 mol J−1 for the intermediate concentration 1.9 M HNO3 is the largest (Table 1). An excitation yield is certainly involved to account for the measured yield. Considering the opposite variations of the doses absorbed by HNO3 and TBP (Fig. 1(a)), the sum of the direct and indirect effects is the most favored for the medium 1.9 M HNO3/3.3 M TBP. The solvated electron yield G(e−solv) = 0.35 and 0.24 × 10−7 mol J−1 at 5 ps, for 0.5 and 1.9 M HNO3 respectively, is obtained (Table 1) with ε(e−TBP)780nm = 9800 L mol−1 cm−1.12 The values are much lower than 1.6 × 10−7 mol J−1 at 7 ps in neat TBP,12 or than the ionization yields in polar liquid,25 because the solvated electrons are scavenged by the nitric acid.
Direct effects
The parts of direct and indirect effects have been already evaluated separately in highly concentrated aqueous HNO3 solutions and in neat HNO3.11 The NO3˙ yield of direct effects of excitation and ionization on neat HNO3 is G(NO3˙) = 3.4 × 10−7 mol J−1,11 and the same value is taken for HNO3 in TBP solutions (Table 1). The corresponding absorbances are proportional to the partial dose absorbed by HNO3 (Table 1 and Fig. 5). | ||
| Fig. 5 Dependence on the HNO3 concentration of the total NO3˙ absorbance measured at 640 nm, and of the calculated partial absorbances by direct and indirect effects. Optical path l = 0.5 cm (Table 1). | ||
Indirect effects
Note that the indirect effects yields G(NO3˙)Indirect in Table 1 are relative to the effects together from TBP and H2O radiolytic species. The absorbances A640(NO3˙)Indirect are obtained from the difference A640(NO3˙)Total − A640(NO3˙)Direct (Table 1 and Fig. 5).Contrarily to G(NO3˙)Direct, the yields G(NO3˙)Indirect are not constant and do depend on the medium composition. The yields of indirect effects on HNO3 arise first from irradiation reactions (4) and (5) of TBP or H2O with a constant yield. But these reactions are followed by partial transfer viareactions (12) and (13) with a ratio depending on competitive processes in the medium. Namely, the yield G(NO3˙)Indirect increases between [HNO3] = 0.5 and 1.9 M because of the increasing competition of the transfer reaction (13) much favored than the reaction (19):
| TBP+˙ + TBP → TBP + TBP(–H)˙ + H+ | (21) |
However, in 4.5 M HNO3/2.8 M TBP/1.5 M H2O, the indirect yield decrease drastically down to G(NO3˙)Indirect = 2.46 × 10−7 mol J−1 (Table 1). At this high HNO3 concentration, not only e−solv is totally scavenged by HNO3 (reaction (8)), but also a part of its presolvated precursor e− is scavenged:
| e− + HNO3(NO3−) → HNO3−˙(NO3−)− | (22) |
| e− + TBP+˙ → 1TBP* | (23) |
| H2O + TBP˙+ → H2O+˙ + TBP | (24) |
Species absorbing at 440 nm
No absorbance such as the absorption band at 440 nm, arising at the end-of-pulse in the HNO3/TBP/H2O environment (Fig. 2(a) and (b)), was observed in aqueous HNO3 solutions.11 In neat TBP (HNO3 free), a much weaker and longer-lived band was observed at almost the same wavelength and was assigned to an excited state of TBP.12 The same assignment cannot be excluded, provided assuming the huge differences in intensity and stability behavior were just the consequence of the strong acidity of the present environment.The value of this initial absorbance is decreasing when [HNO3] is increased from 0.5 to 1.9 M, and becoming negligible at 4.5 M (or when in parallel [TBP] decreases from 3.5 to 2.7 M). Moreover, the 440 nm absorbance decreases when, at constant HNO3 concentration, that of TBP is reduced by its partial replacement by dodecane (Table SI 2 and Fig. SI 2). Seemingly, the formation of the species absorbing at 440 nm requires an early crossed reaction during both HNO3 and TBP radiolysis.
Time-evolution of the transient species absorbance (t > 5 ps)
As shown in Fig. 3, the absorbance intensity decay kinetics depend on the wavelength, therefore on the short-lived species.At 780 nm, the e−TBP absorbance is alone and decays to 0 within 200 ps viareaction (5) (Fig. 6(b)).
At 680 nm, in 0.5 and 1.9 M solutions, the fast absorbance decay of e−TBP during 200 ps is superimposed to the decay of the NO3˙ absorbance which is very slow (Fig. 6(a)).
However, in the 4.5 M solution (Fig. 6(a)), where no solvated electron is detected at 780 nm, the very small initial decay at 640 nm must be assigned to some intra-spur reaction of NO3˙.
After 200 ps, when the interference of the e−TBP absorbance is over, the NO3˙ absorbance at 640 nm decreases slowly to zero by a pseudo first-order process (Fig. 7). It is worthy to note that the decay rate decreases markedly in 4.5 M HNO3 compared to the concentrations 0.5 or 1.9 M. On the contrary, it increases proportionally to the corresponding TBP concentration as shown in Fig. 7, inset. This suggests a reaction of the radical NO3˙ with TBP:
| NO3˙ + TBP → HNO3 + TBP(–H)˙ | (25) |
 | ||
| Fig. 7 Pseudo first-order decay of the 640 nm absorbance at various HNO3 concentrations. Inset: dependence of the pseudo-first order rate constant on the TBP concentration. | ||
From the slope of the linear dependence of the pseudo first-order rate constant on the TBP concentration (Fig. 7, Inset), the bimolecular rate constant is k21 = 0.9 × 106 L mol−1 s−1. The value of this rate constant is much lower than the diffusion controlled one, as expected for H transfer reactions.
During the PUREX process, the above environment is irradiated by the spent nuclear fuel, which is dissolved by the nitric acid, and the ions of uranium and plutonium are selectively extracted via complexation by nitrate anion and solvation by TBP/C12H26. As the reaction rate constant between NO3˙ and UIV was measured in water as k(UIV + NO3˙) = 1.6 × 106 L mol−1 s−1,2 it is expected that the complexed ions of uranium and plutonium are then oxidized to UVI and PuVI, respectively, either directly by NO3˙ radicals, in competition with reaction (22), or by TBP(–H)˙ and C12H25˙ radicals arising from this reaction. The oxidation yield is that of NO3˙ and is the highest for the environment 1.9 M HNO3/3.3 M TBP/0.8 M H2O (GTotal(NO3˙) = 3.2 × 10−7 mol J−1). Note that in these oxidation reactions the solvents HNO3 and TBP are restored. Eventually, the reduction balance by electrons is supported by HNO3 only.
The absorbance at 440 nm beyond 200 ps decreases according to a first order reaction (Fig. 8). The rates are very close each other and independent of the HNO3 concentration at 0.5 and 1.9 M.
Influence of dodecane
In order to determine the influence of dodecane compared with TBP, data were obtained in replacing part of TBP (C12H27PO4) volumes by dodecane (C12H26). The total dose slightly decreases at increasing dodecane concentration (from 0 to 1.1 M) because of its lower electronic density (Table SI 3, Fig. 1(b)). The NO3˙ absorbances and yields at 50 ns (e−TBP has vanished) are given in Table 2. At constant HNO3 and H2O concentrations, the NO3˙ yields with dodecane are lower than without. Seemingly, the energy transfer to HNO3 is less efficient from irradiated dodecane than from TBP (reactions (12)–(15)).| [C12H26] (M), (f) | 0, (0) | 0.61, (0.11) | 1.11, (0.215) |
| [TBP] (M), (f) | 3.1, (0.84) | 2.6, (0.72) | 2.2, (0.624) |
| [HNO3] (M), (f) | 2.5, (0.15) | 2.5, (0.15) | 2.5, (0.16) |
| [H2O] (M), (f) | 1.5, (0.025) | 1.5, (0.025) | 1.5, (0.025) |
| A(NO3˙)Total,640nm,50ns 0.5 cm | 0.007 | 0.005 | 0.004 |
| Dose (Total) (J L−1) | 100.3 | 96.5 | 94 |
| G(NO3˙)Total,50ns (10−7 mol J−1) | 1.0 | 0.78 | 0.63 |
The influence of dodecane on the decay of the NO3˙ radical absorbance at 640 nm is shown in Fig. 9.
 | ||
| Fig. 9 Dependence on the TBP and dodecane concentrations in HNO3 2.5 M/H2O 1.5 M environments (Table 2) of the kinetics of the NO3˙ radical absorbance at 640 nm in logarithmic scale. Optical path 0.5 cm. | ||
The decays are compared for the same HNO3 2.5 M and H2O 1.5 M concentrations, but variable TBP/C12H26 environments ([C12H26] from 0 to 1.1 M). At any time, the absorbances with dodecane are lower than without. However, clearly, the decays in presence of TBP/dodecane are pseudo-first order as without dodecane. Moreover, the slope is very slightly increasing as much as does the sum: [TBP] + [C12H26]. That indicates that dodecane and TBP behave similarly and with the same bimolecular rate constant in the oxidation reactions (25) and (26) by NO3˙:
| NO3˙ + C12H26 → HNO3 + C12H25˙ | (26) |
Conclusions
Using picosecond pulse radiolysis, the transient optical absorption spectra were thoroughly PUREX model environments with various HNO3/TBP/H2O/C12H26 fractions. The optical absorption spectra at the end of the pulse (5 ps) are composed of the overlapping absorption band around 440 nm, hypothetically assigned to some acidic form of a TBP excited state, a band with specific peaks around 640 nm, assigned to the NO3˙ radical, and a broad spectrum from 400 to the near infrared that is assigned to e−TBP, the electron solvated in TBP. The observed overall spectrum has been deconvoluted into the separate spectra of the short-lived species. The e−TBP decay is extremely rapid via the reaction with HNO3 within the pulse and the yield at 5 ps is very low.The HNO3 and TBP/dodecane concentrations and doses absorbed change with opposite trends, but the total NO3˙ radical yield at 5 ps has the highest value for the 1.9 M HNO3/3.3 M TBP/0.8 M H2O environment (GTotal(NO3˙) = 3.2 × 10−7 mol J−1). The radical NO3˙ is early induced both via direct excitation and ionization of HNO3, and indirectly via the transfer of energies absorbed by TBP/C12H26/H2O to HNO3. Despite the complexity of the environment, the mechanism and separate absorbances and yields of these direct and indirect radiation effects are determined and discussed.
The absorbance of the NO3˙ radical decreases with time by a bimolecular reaction with TBP and dodecane. However, the rate constant is very low (k = 0.9 × 106 L mol−1 s−1). In the PUREX process where the environment is irradiated by the spent nuclear fuel, the ions of uranium and plutonium complexed by TBP are oxidized to UVI and PuVI either directly by the early induced NO3˙ radical or by the further TBP(–H)˙/C12H25˙ radicals induced by NO3˙, the most efficiency being observed for the environment 1.9 M HNO3/3.3 M TBP/0.8 M H2O. Its degradation is mostly supported by HNO3 which is reduced via electrons scavenging.
Author contributions
AZ, MB, and SD performed the experiments with the support of DA. Calculations were done by MM and DA. The manuscript was written by JB, MM, AZ, PM, DA and finalized through the contributions of all authors. All authors have given approval to the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Note added after first publication
This article replaces the original version published on 18th March 2026 which contained an error in eqn (10). The remainder of the article remains unchanged.Data availability
A data availability statement (DAS) is required to be submitted alongside all articles.Supplementary information (SI) includes the optical absorption spectra of neat TBP and of various solutions of nitric acid; a table showing the dependence of the dose absorbed by the mixture components on their concentration; and a table showing the how the absorbances and yields of e−TBP and radical were obtained. See DOI: https://doi.org/10.1039/d5cp04724b.
Acknowledgements
This work was supported by CNRS through the NEEDS program and CNRS grant on RadConNOP. The authors deeply thank Jean-Philippe Larbre and Denis Dobrovolskii for their help during the picosecond pulse radiolysis experiments at ELYSE.Notes and references
- R. P. Wayne, I. Barnes, P. Biggs, J. P. Burrows, C. E. Canosa-Mas, J. Hjorth, G. Le Bras, G. K. Moortgat, D. Perner, G. Poulet, G. Restelli and H. Sidebottom, The nitrate radical: Physics, chemistry, and the atmosphere, Atmos. Environ., 1991, 25, 1–203 CrossRef.
- M. A. H. Khan, M. C. Cooke, S. R. Utembe, A. T. Archibald, R. G. Derwent, P. Xiao, C. J. Percival, M. E. Jenkin, W. C. Morris and D. E. Shallcross, Global modeling of the nitrate radical (NO3) for present and pre-industrial scenarios, Atmos. Res., 2015, 164–165, 347–357 CrossRef CAS.
- A. R. Kazanjian, F. J. Miner, A. K. Brown, P. G. Hagan and J. W. Berry, Radiolysis of nitric acid solution: L.E.T. effects, Trans. Faraday Soc., 1970, 66, 2192–2198 RSC.
- M. Daniels, Radiation chemistry of the aqueous nitrate system. III. Pulse electron radiolysis of concentrated sodium nitrate solutions, J. Phys. Chem., 1969, 73, 3710–3717 CrossRef CAS.
- R. K. Broszkiewicz, The radiation-induced formation of NO3 in aqueous solutions, Int. J. Appl. Radiat. Isot., 1967, 18, 25–32 CrossRef CAS.
- P. K. Bhattacharyya and R. D. Saini, Radiolytic yields G(HNO2) and G(H2O2) in the aqueous nitric acid system, Int. J. Radiat. Phys. Chem., 1973, 5, 91–99 CrossRef CAS.
- T. Loegager and K. Sehested, Formation and decay of peroxynitrous acid: a pulse radiolysis study, J. Phys. Chem., 1993, 97, 6664–6669 CrossRef CAS.
- Y. Katsumura, P. Y. Jiang, R. Nagaishi, T. Oishi, K. Ishigure and Y. Yoshida, Pulse radiolysis study of aqueous nitric acid solutions: formation mechanism, yield, and reactivity of NO3 radical, J. Phys. Chem., 1991, 95, 4435–4439 CrossRef CAS.
- G. Garaix, G. P. Horne, L. Venault, P. Moisy, S. M. Pimblott, J. Marignier and M. Mostafavi, Decay Mechanism of NO3˙ Radical in Highly Concentrated Nitrate and Nitric Acidic Solutions in the Absence and Presence of Hydrazine, J. Phys. Chem. B, 2016, 120, 5008–5014 CrossRef CAS PubMed.
- A. Balcerzyk, A. K. El Omar, U. Schmidhammer, P. Pernot and M. Mostafavi, Picosecond Pulse Radiolysis Study of Highly Concentrated Nitric Acid Solutions: Formation Mechanism of NO3˙ Radical, J. Phys. Chem. A, 2012, 116, 7302–7307 CrossRef CAS PubMed.
- R. Musat, S. A. Denisov, J.-L. Marignier and M. Mostafavi, Decoding the Three-Pronged Mechanism of NO3˙ Radical Formation in HNO3 Solutions at 22 and 80 °C Using Picosecond Pulse Radiolysis, J. Phys. Chem. B, 2018, 122, 2121–2129 CrossRef CAS PubMed.
- F. Wang, G. P. Horne, P. Pernot, P. Archirel and M. Mostafavi, Picosecond Pulse Radiolysis Study on the Radiation-Induced Reactions in Neat Tributyl Phosphate, J. Phys. Chem. B, 2018, 122, 7134–7142 CrossRef CAS PubMed.
- T. Bahry, S. A. Denisov, P. Moisy, J. Ma and M. Mostafavi, Real-Time Observation of Solvation Dynamics of Electron in Actinide Extraction Binary Solutions of Water and n-Tributyl Phosphate, J. Phys. Chem. B, 2021, 125, 3843–3849 CrossRef CAS PubMed.
- Y. Yoshida, T. Ueda, T. Kobayashi, H. Shibata and S. Tagawa, Studies of geminate ion recombination and formation of excited states in liquid n-dodecane by means of a new picosecond pulse radiolysis system, Nucl. Instrum. Methods Phys. Res., Sect. A, 1993, 327, 41–43 CrossRef.
- L. K. Patil, V. G. Gaikar, S. Kumar, U. K. Mudali and R. Natarajan, Thermal Decomposition of Nitrated Tri-n-Butyl Phosphate in a Flow Reactor, Int. Sch. Res. Notices, 2012, 2012, 193862 Search PubMed.
- A. Dodi and G. Verda, Improved determination of tributyl phosphate degradation products (mono- and dibutyl phosphates) by ion chromatography, J. Chromatogr. A, 2001, 920, 275–281 CrossRef CAS PubMed.
- V. S. Smitha, J. S. V. Kumar, M. Surianarayanan, H. Seshadri and N. V. Lakshman, Reactive chemical pathway of tributyl phosphate with nitric acid, Process Saf. Envion. Prot., 2018, 116, 677–684 CrossRef CAS.
- B. Mokili and C. Poitrenaud, Modelling of Nitric Acid and Water Extraction From aqueous Solutions Containing a Salting-Out Agent by Tri-N-Butylphosphate, Solvent Extr. Ion Exch., 1995, 13, 731–754 CrossRef CAS.
- J. P. Holland, J. F. Merklin and J. Razvi, The radiolysis of dodecane-tributylphosphate solutions, Nucl. Instrum. Methods Phys., 1978, 153, 589–593 CrossRef CAS.
- I. Billard, Are molecular solvents, aqueous biphasic systems and deep eutectic solvents meaningful categories for liquid–liquid extraction?, C. R. Chim., 2022, 25, 67–81 CrossRef CAS.
- J. Belloni, H. Monard, F. Gobert, J.-P. Larbre, A. Demarque, V. De Waele, I. Lampre, J.-L. Marignier, M. Mostafavi, J. C. Bourdon, M. Bernard, H. Borie, T. Garvey, B. Jacquemard, B. Leblond, P. Lepercq, M. Omeich, M. Roch, J. Rodier and R. Roux, ELYSE—A picosecond electron accelerator for pulse radiolysis research, Nucl. Instrum. Methods Phys. Res., Sect. A, 2005, 539, 527–539 CrossRef CAS.
- J.-L. Marignier, V. de Waele, H. Monard, F. Gobert, J.-P. Larbre, A. Demarque, M. Mostafavi and J. Belloni, Time-resolved spectroscopy at the picosecond laser-triggered electron accelerator ELYSE, Radiat. Phys. Chem., 2006, 75, 1024–1033 CrossRef CAS.
- G. V. Buxton, C. L. Greenstock, W. P. Helman and A. B. Ross, Critical Review of rate constants for reactions of hydrated electrons, hydrogen atoms and hydroxyl radicals (˙OH/˙O−) in Aqueous Solution, J. Phys. Chem. Ref. Data, 1988, 17, 513–886 CrossRef CAS.
- S. J. Kruse, S. K. Scherrer, G. P. Horne, J. A. LaVerne and T. Z. Forbes, The inorganic chemist's guide to actinide radiation chemistry: a review, Inorg. Chem. Front., 2025, 12, 6398–6434 RSC.
- J. Belloni and J. L. Marignier, Electron-solvent interaction: attachment solvation competition, Int. J. Radiat. Appl. Instrum., Part C, 1989, 34, 157–171 Search PubMed.
| This journal is © the Owner Societies 2026 |