 Open Access Article
Open Access ArticleExpanding aromaticity tests to include lowest-lying triplet excited states and charged and heterocyclic rings
Maria
Cabrero-Martí
 and
Miquel
Solà
and
Miquel
Solà
 *
*
Institut de Química Computacional i Catàlisi and Departament de Química, Universitat de Girona, 17003 Girona, Catalonia, Spain. E-mail: miquel.sola@udg.edu
First published on 20th January 2026
Abstract
Aromaticity is a pivotal concept in chemistry arising from electron delocalization in closed-loop systems that confers extra energetic stabilization. Its direct observation is not possible, making its quantification challenging. To address this issue, several methods have been developed to quantify this non-observable property based on various physicochemical properties characteristic of these compounds. However, given the indirect nature of these measures, they do not always yield consistent or reliable aromaticity trends. Therefore, it is important to design tools that help identify which descriptors perform most effectively and put forward their weaknesses and strenghts. In a previous study (F. Feixas, E. Matito, J. Poater, M. Solà, On the performance of some aromaticity indices: a critical assessment using a test set, J. Comput. Chem., 2008, 29(10), 1543–1554.), we introduced a series of fifteen aromaticity tests that were used to analyze the advantages and drawbacks of a group of ten aromaticity descriptors. In this work, we propose to extent these initial tests of aromaticity with a series of thirteen tests related to excited state aromaticity, redox processes, and heterocyclic rings to evaluate the ability of twelve aromaticity descriptors. A comprehensive evaluation of the strengths and weaknesses of the analysed indicators of aromaticity reveals that electronic and magnetic indices perform most consistently, although the former show limitations when applied to oxidized species.
1. Introduction
Aromaticity is a crucial concept in the field of chemistry, not exempt from criticism.1–10 Although no consensus exists on a precise definition, chemists continue to rely on this concept because of its fundamental role in the elucidation of some phenomena, such as the particular low reactivity, the interpretation of the molecular structure, and the spectroscopic and magnetic properties of an endless number of compounds.11–13The never-ending ambiguity is caused by the fact that this property, although widely explored, it has never been directly observed.14 The main problem in assessing the aromaticity of a molecule or its parts is that no property serves as a straightforward measure. However, the lack of a precise definition and the absence of a proper operator have not deterred chemists from developing various methodological approaches for its quantification.12
There is a general consensus on certain characteristic properties typically associated with aromatic moieties. The manifestation of these features is apparent in distinct chemical and physical behaviours, which are indicative of their enhanced kinetic and thermodynamic stability in comparison to their open-chain analogues. A molecule is classified as aromatic if it possesses a particular molecular and electronic structure. This characteristic structure is primarily manifested through the presence of a cyclic 2D or 3D moiety with delocalized electrons. Moreover, aromatic species have a tendency towards bond length equalization, and they exhibit distinctive spectroscopic and magnetic signatures. In particular, these compounds have been associated with an unusually high diamagnetic susceptibility, which Pauling,15 London,16 and Lonsdale17 attributed to the induced ring current generated by the delocalized electrons, which results in abnormal chemical shifts.18
Given this situation, the assessment of the aromatic character is usually carried out indirectly through the measurement of the aforementioned common physicochemical properties that are indicative of aromatic character. Thus, even though aromaticity itself cannot be directly observed, chemists have managed to develop numerous indices for the quantification based on energetic, electronic, structural, magnetic, and reactivity measures.12,19 These quantifiers provide an approximate measure of the central property of interest.
Even though this long-standing challenge has been addressed quite effectively, identifying a precise and universally accepted quantitative measure of aromaticity remains elusive. This difficulty arises from the fact that aromaticity is linked to multiple molecular characteristics that are not necessarily interrelated.20–24 The use of various aromaticity criteria derived from different properties has significant consequences. As none of these approaches are free from ambiguity, there is often an absence of correlation among the values. Furthermore, the relative accuracy of each method remains uncertain, as no reliable references exist to assess and compare their effectiveness.
In light of the present circumstances, it is recommended to employ a range of indices from different origins. In front of all the results, when multiple indices based on different measures converge towards the same conclusion, one can confidently establish a solid outcome. Conversely, if the results are not in agreement, any final conclusion will be considerably weaker. Summarizing, while it is undoubtedly a significant challenge, and no unequivocal scale exists, initial indications can be obtained using different descriptors.25–28
Meanwhile, new aromaticity descriptors are frequently introduced in the literature.29–31 The validation of a newly implemented quantifier is usually performed by means of correlation with a series of previously defined descriptors. As said before, the problem is that sometimes poor correlations are found among different indicators of aromaticity. Therefore, validation by comparison does not ensure the quality of a new descriptor. Given the limited reliability of current aromaticity quantifiers, there is a pressing need to develop a comprehensive toolkit that establishes the most suitable descriptors for different families of molecules.
A previous study had been conducted by some of us,19 wherein the performance of a set of aromaticity descriptors was evaluated. In that work, fifteen tests of aromaticity were proposed to identify the most accurate aromaticity descriptors. The tests chosen describe simple situations in which the trends followed by the aromatic character of the different rings are widely accepted by the chemical community. In addition, they were chosen to facilitate a fast application to ensure that the testing set can be quickly and easily extended to incorporate any new quantifier. This approach allows one to readily assess the accuracy of these new indices, and additionally, one can also track whether any improvement has been achieved. By maintaining a straightforward system, we can easily keep our methodology up to date as the field evolves, allowing us to compare all available tools at any given time.
Back in 2008, aromaticity analyses of excited states were not commonly performed, which is why tests involving such states were not included. The present study extends that earlier work to systems not previously examined, including the aromaticity of lowest-lying triplet states, rings undergoing redox processes, and heterocyclic rings. In particular, we set up thirteen new aromaticity tests. We apply the tests to twelve aromaticity descriptors based on different physicochemical properties to identify their strengths and limitations depending on their accuracy in reflecting the expected aromaticity trends across the diverse types of organic molecules.
The tests proposed in this work are depicted in Scheme 1. The initial seven tests are devoted to species in their the lowest-lying triplet state (T1). The following four involve oxidation and/or reduction processes of molecules. In the final two tests, we compare the aromaticity in two heterocyclic isomeric molecules. The results obtained will demonstrate that common descriptors of aromaticity are not optimal to describe certain situations.
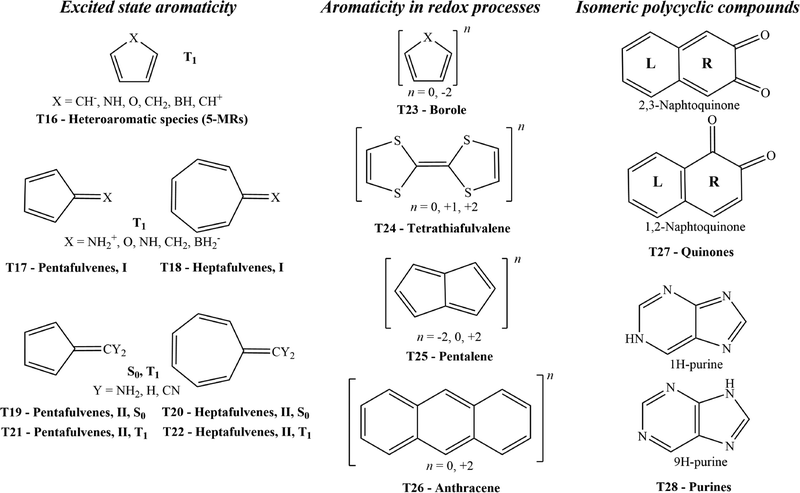 | ||
| Scheme 1 Schematic representation of the thirteen new proposed tests. Numbering starts at test T16 because in our previous work, we proposed fifteen tests (T1–T15). | ||
2. Measures of aromaticity
The twelve descriptors selected for this study are based on structural, magnetic, and electronic measures. Since we are expanding the previous work,19 we selected the same indices that were chosen in that work, and we added the recently introduced electron density of delocalized bonds (EDDB) index.32,33 It is worth noting that in a previous work,34 EDDB was already applied to our initial fifteen tests.19As a geometric-based measure, we have employed the harmonic oscillator model of aromaticity (HOMA) index, defined by Kruszewski and Krygowski.35,36 It is the most widely used structural indicator of aromaticity, due to its straightforward computation and good performance considering its low computational cost. It can be obtained from the following expression:
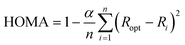 | (1) |
A new parametrization of this index for the excited states has been recently introduced, the HOMER, which stands for Harmonic Oscillator Model of Excited-state aRomaticity.37 There is a key difference in the parameterization process. The Ropt is adjusted to bring both HOMA and HOMER values close to 1 for aromatic rings, however, α in this case, it is adjusted to yield HOMER values close to −1 for a model antiaromatic system. As a result, for non-aromatic systems, values are expected to fall near zero.
As magnetic indices of aromaticity, we have employed three variations of the nucleus-independent chemical shift (NICS) index; NICS(0), NICS(1), and NICS(1)zz. This index was introduced by Schleyer and co-workers in 1996,38 and it has been recognized as the most popular one, owing to the fact that it can also be obtained through experimental measures in some cases.39 For example, NICS and NMR shifts of chemically inert 3He at fullerene centers agree very well.40 NICS is defined as the negative value of the absolute shielding computed at the ring center or at some other interesting point of the system. The aromatic character of the rings is reflected by negative NICS values, whereas positive NICS values indicate antiaromaticity.
Finally, seven aromaticity indicators based on the quantification of cyclic electron delocalization around the rings have been used.27,41 These indices measure the cyclic electron delocalization of mobile electrons in aromatic rings. They use the delocalization index (DI)42–44 between atoms A and B, δ(A, B), which is obtained by the double integration of the exchange–correlation density. For single-determinant wavefunctions, the DI can be expressed in terms of atomic overlaps as:
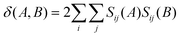 | (2) |
As electronic-based indices, first, we have employed the para-delocalization index (PDI),46,47 which is computed by averaging the DI's between atoms located in para-positions within the six-membered rings (6-MRs). It was inspired by the fact that benzene exhibits greater electron sharing between carbons in para- than in meta-positions.48,49 Therefore, the higher the value, the more aromatic the molecule is expected to be.
Second, we have the aromatic fluctuation index (FLU) by Matito et al. defined in 2005.50,51 It measures aromaticity by assessing the uniformity of cyclic electron delocalization along the ring and comparing it to that of well-known aromatic molecules.
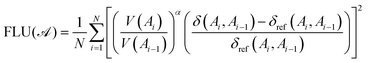 | (3) |
Here A0 ≡ AN and V(A) is the atomic valence of atom A as defined by eqn (4):
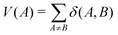 | (4) |
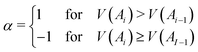 | (5) |
Finally, the δref(Ai, Ai−1) values are δref(C, C) = 1.389e, δref(C, N) = 1.318e, δref(N, N) = 1.518e, and δref(C, O) = 0.970e taken from benzene, pyridine, pyridazine, and furan, respectively, at the B3LYP/6-311++G(d,p) level. Aromatic molecules present FLU values close to zero, and the value increases as the aromaticity of the ring decreases. The original FLU index, designed for closed-shell systems, was adapted for Baird aromaticity analysis by splitting it into α and β components.52 We employ FLU1/2 instead of FLU, since although both follow analogous trends, a key advantage of FLU1/2 is that it displays a wider range, thereby offering clearer trends.53
Third, we have employed a set of four multicenter indices, namely, the Iring, ING, MCI, and INB. The multicenter index (Iring) of Giambiagi et al. studies the electron delocalization around the ring by applying the following eqn (6) for a closed-shell monodeterminantal wavefunction:54
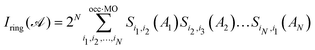 | (6) |
Considering that the electron sharing is not constrained to neighbouring atoms, Bultinck et al. proposed an extension of the Iring named the multicenter electron delocalization index (MCI), whose formula reads:55
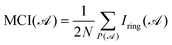 | (7) |
![[scr A, script letter A]](https://www.rsc.org/images/entities/char_e520.gif) ) stands for a permutation operator to generate the N! permutations of the elements in string
) stands for a permutation operator to generate the N! permutations of the elements in string ![[scr A, script letter A]](https://www.rsc.org/images/entities/char_e520.gif) , to ensure that all contributions from every possible rearrangement of the atoms of the ring are taken into account. Generally, when working with organic molecules, these two indices demonstrate a significant correlation since the major contribution to MCI comes from the Kekulé structure. The more aromatic the rings are, the higher the Iring and MCI are.
, to ensure that all contributions from every possible rearrangement of the atoms of the ring are taken into account. Generally, when working with organic molecules, these two indices demonstrate a significant correlation since the major contribution to MCI comes from the Kekulé structure. The more aromatic the rings are, the higher the Iring and MCI are.
These indices are ring-size dependent; therefore, to enable the comparison between rings of different sizes, normalized versions have been introduced. A proposed normalized version of the Iring index is the so-called ING, which is expressed as:56
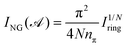 | (8) |
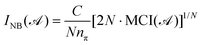 | (9) |
To apply these indices to the lowest-lying triplet excited states (T1), the same strategy used for the FLU index is followed, performing the calculations separately for the α and β components.57
The last electronic index is the electron density of delocalized bonds (EDDB),32–34 which quantifies the electron delocalization throughout the system. It is an electron density (ED) partitioning method, where the ED is divided into several “layers” corresponding to different levels of electron delocalization,58 which are the electron density localized on atoms (EDLA), the electron density of localized bonds (EDLB), and the EDDB, which accounts for the remaining density that cannot be assigned to specific atoms or bonds due to its multicenter delocalized nature. EDDB offers predictions closely aligned with MCI but at much lower computational cost, enhancing efficiency, particularly for large molecules and correlated wavefunctions.
For the purpose of this work, we have evaluated the number of cyclically delocalized electrons in a given path (usually the path that follows the perimeter of the ring studied, EDDBP). This function does not account for cross-ring delocalization effects; it is restricted to the delocalization arising from the resonance of the Kekulé forms. Moreover, the EDDBP values have been normalized by dividing them by the number of atoms in the path to express the number of delocalized electrons per atom. Aromatic molecules with a high degree of delocalization yield higher values.
3. Computational details
Geometry optimizations have been performed using Gaussian 1659 at the B3LYP60,61 level of theory with the 6-311++G(d,p) basis set.62 For closed-shell ground state species, geometry optimizations and calculations of aromaticity indices have been carried out using the restricted formalism at the B3LYP/6-311++G(d,p) level of theory. In contrast, open-shell triplet states have been treated with the unrestricted formalism for both geometry optimization and the evaluation of aromaticity descriptors (UB3LYP/6-311++G(d,p)). For comparison purposes, we have kept the same level of theory as in our previous studies,19,63 although we are aware of the superior performance of long-range corrected functionals in aromatic species.64–67 However, we do not expect significant changes in aromaticity trends by changing the functional or the basis set.66,67 The numerical accuracy of the QTAIM calculations done with the AIMPAC program68 has been assessed using two criteria: (i) the integration of the Laplacian of the electron density (∇2ρ(r)) within an atomic basin should be close to zero; (ii) the total number of electrons in a molecule must equal the sum of all the electron populations within the molecule and also match the sum of all localization indices plus half of the delocalization indices. For all atomic calculations, integrated absolute values of ∇2ρ(r) were consistently below 0.001 a.u. For all molecules, the error in the calculated number of electrons was always less than 0.01 a.u.Calculations of electronic and structural descriptors of aromaticity were performed with the ESI-3D program,69 except for EDDBP and HOMER. The EDDBP32–34 indices were obtained using the NBO 7.0 software70 to first derive the natural atomic orbitals (NAO)71 and the one-electron density matrix required by the RunEDDB program.72 The excited-state analogue of HOMA, known as HOMER,37 was computed using the ESIpy program.73 The GIAO method74 has been used for the calculations of NICS38 at the ring center (NICS(0)), determined by the non-weighted mean of the heavy atom coordinates, and at 1 Å above and below the ring plane, NICS(1) and NICS(−1). We have also analysed its out-of-plane component, NICS(1)zz.75 All these indicators of aromaticity have been computed for both closed-shell ground state species and open-shell triplet excited states.
4. Results and discussion
4.1. Triplet state aromaticity – Baird's 4n rule
With the exception of NICS(1) and NICS(1)zz, which correctly order the six species and correctly categorize them, the rest of the indices fail to some extent (see Table S19). The descriptors show inferior performance in the T1 state than in the S0 ground state, as several electronic indices had previously provided correct trends in the S0 state (see T11 in Table 1 and Table S12) but not in the T1 state. However, if we look closely at the results, we can see that MCI and INB only fail to assign a certain aromatic character to the most antiaromatic species (R = CH−). The other multicenter indices, Iring and ING, also fail by attributing similar values to the species with R = −O and –CH2, whereas EDDBP assigns a similar value to the species with R = –NH as well. FLU1/2, HOMA, and HOMER do not work well either. We attribute the failure of the FLU1/2 and HOMA to the fact that they were parameterized for the ground state.
| PDIa | FLU1/2 | MCI | I NB | I ring | I NG | EDDBP | HOMA | HOMERb | NICS(0) | NICS(1) | NICS(1)zz | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a Only applicable to 6-MR's. b Only applied to molecules in their T1 state. c Loss of aromaticity is overemphasized (see ref. 19). d The aromaticity remains almost unchanged with the distortion (see ref. 19). e The trend in aromaticity remains almost unchanged with some oscillations (see ref. 19). f The aromaticity is higher than that of benzene only for a small number of molecules (see ref. 19). g Fails only in ordering one molecule (see ref. 19). h Fails only in ordering one molecule (see text). At the CCSD level, this index passes the test. i The aromaticity of the TS is higher than that of benzene (see ref. 19). j Fails only by assigning nearly identical character to two molecules (see text). k Trend is correct but the classification of some of the species as (anti)aromatic is wrong (see text). l The proportions are not right (see text). | ||||||||||||
| T1 | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Unclearc | N/A | Yes | Yes | Yes |
| T2 | Uncleard | Yes | Yes | Yes | Yes | Yes | Yes | Unclearc | N/A | Uncleare | Yes | Yes |
| T3 | Uncleard | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Yes | Yes | Yes |
| T4 | Uncleard | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Yes | Yes | Yes |
| T5 | Uncleard | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Yes | Yes | Yes |
| T6 | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Unclearf | N/A | No | No | Yes |
| T7 | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | No | No | Yes |
| T8 | N/A | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | No | Yes | Yes |
| T9 | No | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Yes | No | No |
| T10 | No | Unclearg | Unclearh | Unclearh | Unclearh | Unclearh | No | No | N/A | No | No | No |
| T11 | N/A | Unclearg | Yes | Yes | Yes | Unclearg | Unclearg | Unclearg | N/A | Unclearg | Unclearg | Yes |
| T12 | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Yes | Yes | Yes |
| T13 | N/A | No | Yes | Yes | Unclearg | Unclearg | Unclearg | Unclearg | N/A | Yes | Yes | Yes |
| T14 | Yes | No | Yes | Yes | Yes | Yes | Yes | No | N/A | Uncleari | Uncleari | Yes |
| T15 | Yes | No | Yes | Yes | Yes | Yes | Yes | No | N/A | Uncleari | Uncleari | Uncleari |
| T16 | N/A | No | Unclearg | Unclearg | Uncleargj | Uncleargj | Uncleargj | No | No | Unclearg | Yes | Yes |
| T17 | N/A | No | Yes | Yes | Unclearg | Unclearg | Unclearg | Unclearg | Yes | Yes | Yes | Yes |
| T18 | N/A | Unclearg | Yes | Yes | Yes | Yes | Yes | Unclearg | Yes | Yes | Yes | Yes |
| T19 | N/A | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Yes | Yes | Yes |
| T20 | N/A | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Yes | Yes | Yes |
| T21 | N/A | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| T22 | N/A | Yes | Yes | Yes | Unclearg | Unclearg | Yes | Yes | Yes | Yes | Yes | Yes |
| T23 | N/A | N/A | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Yes | Yes | Yes |
| T24 | N/A | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | Yesk | Yes | Yes |
| T25 | N/A | Yes | No | No | No | No | Yes | No | N/A | Yesk | Yesk | Yesk |
| T26 | Unclearg | Yes | Unclearg | Unclearg | Unclearg | Unclearg | Yes | Yes | N/A | No | No | No |
| T27 | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Unclearkl | N/A | Uncleark | Uncleark | Uncleark |
| T28 | Yes | No | No | Unclearj | No | No | Yes | Yes | N/A | Yes | No | No |
The impact of the electron-donating and electron-withdrawing character of the substituents can be comprehended by the varying contributions of the resonance structures in Fig. 1.78–80 In the presence of electron-withdrawing groups (EWGs), which pull electron density away from the ring, the structures 1C and 2C are stabilized with 4 and 6π-electrons within the ring, respectively. Hence, the aromatic character of the 5-MR in pentafulvenes, taking into account Baird's 4n aromaticity rule, is predicted to increase following the trend: NH2+ > O > NH > CH2 > BH2−. Conversely, electron-donating groups (EDGs) enhance the aromaticity in the heptafulvenes in their T1 by stabilizing structure 2A, leaving the 7-MR with 8π-electrons, thus resulting in the opposite trend. To assess the influence of substituents on aromaticity, molecular geometries were optimized imposing a planar conformation. This ensures that the pz orbitals of the substituents are oriented perpendicular to the molecular plane, allowing optimal interaction between these pz orbitals and those of the ring.
 | ||
| Fig. 1 The most relevant resonance structures of fulvenes I (X = NH2+, O, NH, CH2, BH2−) and fulvenes II (X = CY2, Y = NH2, H, CN). | ||
In the preceding ground-state analysis,19 only MCI, INB, and the NICS variants reproduced the exact aromaticity order across both sets of species (see T13 in Table 1 and Table S14). In the T1 state, HOMER, together with the aforementioned indices, accurately captures the trends for the pentafulvenes (see Table S20). HOMA, Iring, and ING fail to properly rank cyclopentadienone and assign similar aromatic characters to the mid-range species. EDDBP fails in the ordering of just R = BH2− and FLU1/2 struggles with the overall ordering. For the heptafulvenes, most indices generally perform well. The exceptions are HOMA and FLU1/2, which both fail switching R = NH2+ and R = O. Moreover, these two species have the same ING value. The quantifiers that rank the molecules correctly are HOMER, NICS, and the rest of the electronic indices, with all indices consistently indicating low aromaticity overall.
Considering the increased contribution of resonance structure 1A with formally 6π-electrons in the presence of an EDG, an increase in the aromaticity is expected in the S0 state of pentafulvenes following the order NH2 > H > CN. In contrast, the aromatic character of the 7-MRs in heptafulvenes decreases in the same order because of the large contribution of the 8π-electron structure 2A. In the T1 state, in which Baird's rule applies,76 EDGs enhance the aromatic character in heptafulvenes in the same order NH2 > H > CN, as the system fulfils the 4n rule, and decrease it in pentafulvenes. On the other hand, stabilizing structures 1C and 2C, EWGs are expected to lead to the converse situation, increasing the aromatic character for heptafulvenes in their ground state in the order CN > H > NH2, due to an increase in the resonance structure with 6π-electrons within the ring, and dropping in the 5-MRs with 4π-electrons in pentafulvenes in the same order. In the T1 state, the EWGs strengthen the aromatic character in pentafulvenes and reduce it in heptafulvenes.78,81–83 Thus, a good aromaticity descriptor should give the following order of aromaticity for the 5-MRs in pentafulvenes in their S0 state NH2 > H > CN, and the same is expected for the heptafulvenes in their triplet excited state. For the 7-MR in heptafulvenes in their S0 state and the pentafulvenes in their T1 state, the opposite order is anticipated.
The complete set of indicators provides the expected trends (see Table S21) for both the S0 and T1 states of penta- and heptafulvenes, with the exception of Iring and ING that provide similar aromaticities to Y = H and CN in T22.
4.2. Redox species
All indices indicate an increase in aromaticity following reduction (see Table S22). Nevertheless, the aromaticity descriptors suggest that the aromatic character of the dianionic species remains very weak and the actual values of the indices are so low that the molecule could reasonably be classified as non-aromatic or at least weakly aromatic, as found in previous studies.85
Finally, after removing another electron and reaching the + 2 charge, the two rings end up with 6π-electrons each, which further enhances the aromatic character, becoming fully aromatic. Furthermore, the bond has completely lost its double-bond character, allowing free rotation of the rings and resulting in a loss of planarity in the overall conformation.94
The entire set of indices effectively describes the aromatic trend (see Table S23). Moreover, almost all the indices correctly describe the non-aromatic character of the neutral species by giving values close to zero, with the exception of NICS(0), that classifies the rings of the neutral species as aromatic.
All indices in the set were suitable for describing the increase in aromaticity following the reduction. However, the increase in aromaticity upon formation of the dicationic species was not consistently captured by the entire collection of quantifiers (see Table S24). The four multicenter indices, Iring, ING, MCI, and INB fail to show the increase in aromaticity after oxidation. This can be attributed to the fact that these quantifiers assess the extent of electron delocalization within the ring. Upon oxidation, the removal of electrons reduces the number of electrons available for delocalization, thereby decreasing the values and diminishing the effectiveness of these electronic indices. Interestingly, although the EDDBP analysis indicates an increase in aromaticity in both directions, the change observed after oxidation is comparatively smaller, which could be argued in a similar manner. Furthermore, HOMA also fails to reflect this increase. Moreover, NICS gives positive values for the aromatic dianion. Given all that, the descriptors that described the expected trend in T25 are FLU1/2, EDDBP, and NICS.
Analysing each species separately, in general, the indices successfully capture the similar aromatic character of the external rings expected in both, the neutral and the dicationic, as well as the resemblance between the external and internal rings in the neutral molecule. The decrease in the aromaticity of the central ring after oxidation was consistently reflected by all indices. Conversely, an increase in the outer rings is indicated only by Iring, FLU1/2, HOMA, and EDDBP indices. Yet most of these indices underestimate the expected variations, as the reported changes are often smaller than the differences observed between rings within the neutral species, where no significant variance is expected. Taking all of the above into account, the only index that aligns with all expected trends in test T26 is FLU1/2 (see Table S25).
4.3. Isomeric molecules
The complete set of indices captures the decrease between left-side rings, from the highly aromatic QB − L to the slightly aromatic QA − L. Still, some indices exhibit limitations (see Table S26). NICS correctly classifies QB − L as aromatic but misclassifies the rest, assigning values shifted toward the antiaromatic region, despite these being expected to be slightly aromatic or non-aromatic. A similar issue is observed with the HOMA index, which indicates non-aromaticity for QA − L and antiaromaticity for QA/B − R. Interestingly, this geometric index is the only one that displays a larger difference between QA − L ≳ QA − R than between QB − L ≫ QA − L, which is inconsistent with the expected aromaticity trend. In contrast, FLU1/2 values increase in line with expectations, making it a reliable quantifier alongside the multicenter indices, which also offer an accurate description.
The increase in aromaticity from 1H-purine to the 9H-purine between the two 6-MRs is correctly captured by all quantifiers (see Table S27). On the contrary, only NICS(0), EDDBP, and HOMA indicate an increase when going from the 5-MR of 1H-purine to 9H-purine. Moreover, this geometric, computationally inexpensive index also provides a good qualitative description of the aromatic character, yielding values close to 1 for the aromatic rings in 9H-purine, and lower values that still fall within the aromatic range (between 0 and 1) for the two rings in 1H-purine.
5. Conclusions
As a concluding set of recommendations derived from the analysis of the thirteen tests applied to the twelve aromaticity indices studied, we can say that, first, in general, indicators of aromaticity perform somewhat better in the S0 than in the T1 state; second, NICS and EDDBP are the quantifiers with the highest reliability in these new introduced tests (T16–28), although caution should be exercised when using these magnetic indices in fused rings;21,106,107 third, multicenter indices, which have long been regarded as the most reliable ones, have presented a consistent tendency to encounter difficulties when working on oxidized species; fourth, for the T1 state, HOMER overperforms HOMA, although application of HOMER to fused systems (for instance, biphenylene) remains to be explored; fifth, EDDBP performs as good as MCI but at much lower computational cost. Finally, since no index performs reliably in all cases, we continue to recommend using a set of indices based on different properties to reach more reliable conclusions.Author contributions
M. S.: conceived the project, acquired funding, provided resources, supervised the project, and wrote, reviewed, and edited the manuscript. M. C.: performed DFT calculations, analyzed the data, and wrote, reviewed, and edited the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5cp04577k.Acknowledgements
We are grateful for financial support from the Agencia Española de Investigación (MICIU/AEI/10.13039/501100011033) for project PID2023-147424NB-I00 and the FPI (PRE44_2024) grant to M. C. We also acknowledge the Generalitat de Catalunya for project 2021SGR623 and the ICREA Academia prize 2024 to M. S.Notes and references
- G. Binsch, Aromaticity—An exercise in chemical futility?, Sci. Nat., 1973, 60(8), 369–374, DOI:10.1007/BF00602510.
- A. T. Balaban, Is aromaticity outmoded?, Pure Appl. Chem., 1980, 52(6), 1409–1429, DOI:10.1351/pac198052061409.
- D. Lloyd, What is aromaticity?, J. Chem. Inf. Comput. Sci., 1996, 36(3), 442–447, DOI:10.1021/ci950158g.
- A. Stanger, What is…aromaticity: a critique of the concept of aromaticity—can it really be defined?, Chem. Commun., 2009,(15), 1939–1947, 10.1039/B816811C.
- R. Hoffmann, The many guises of aromaticity, Am. Sci., 2015, 103(1), 18, DOI:10.1511/2015.112.18.
- M. Solà, Why aromaticity is a suspicious concept? Why?, Front. Chem., 2017, 5, 22, DOI:10.3389/fchem.2017.00022.
- G. Merino, M. Solà, I. Fernández, C. Foroutan-Nejad, P. Lazzeretti and G. Frenking, et al., Aromaticity: Quo Vadis, Chem. Sci., 2023, 14(21), 5569–5576, 10.1039/D2SC04998H.
- H. Ottosson, A focus on aromaticity: fuzzier than ever before?, Chem. Sci., 2023, 14(21), 5542–5544, 10.1039/D3SC90075D.
- I. Agranat, Aromaticity-a theoretical notion, Struct. Chem., 2024, 35(3), 715–720, DOI:10.1007/s11224-024-02328-y.
- M. Baranac-Stojanovic, Aromaticity and Antiaromaticity: How to Define Them, Chemistry, 2025, 7(4), 127, DOI:10.3390/chemistry7040127.
- T. Krygowski, M. Cyranski, Z. Czarnocki, G. Häfelinger and A. R. Katritzky, Aromaticity: a theoretical concept of immense practical importance, Tetrahedron, 2000, 56(13), 1783–1796, DOI:10.1016/S0040-4020(99)00979-5.
- M. Solà, A. I. Boldyrev, M. K. Cyrañski, T. M. Krygowski and G. Merino, Aromaticity and antiaromaticity: concepts and applications, John Wiley & Sons, Chichester, 2023 Search PubMed.
- Aromaticity: Modern Computational Methods and Applications, ed. I. Fernández, Elsevier, Dordrecht, 2021 Search PubMed.
- M. Solà, Connecting and combining rules of aromaticity. Towards a unified theory of aromaticity, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2019, 9(4), e1404, DOI:10.1002/wcms.1404.
- L. Pauling, The diamagnetic anisotropy of aromatic molecules, J. Chem. Phys., 1936, 4(10), 673–677, DOI:10.1063/1.1749766.
- F. London, Théorie quantique des courants interatomiques dans les combinaisons aromatiques, J. Phys. Radium, 1937, 8(10), 397–409, DOI:10.1051/jphysrad:01937008010039700.
- K. Y. Lonsdale, Magnetic anisotropy and electronic structure of aromatic molecules, Proc. R. Soc. London, Ser. A, 1937, 159(896), 149–161, DOI:10.1098/rspa.1937.0064.
- R. H. Mitchell, Measuring aromaticity by NMR, Chem. Rev., 2001, 101(5), 1301–1316, DOI:10.1021/cr990359+.
- F. Feixas, E. Matito, J. Poater and M. Solà, On the performance of some aromaticity indices: a critical assessment using a test set, J. Comput. Chem., 2008, 29(10), 1543–1554, DOI:10.1002/jcc.20914.
- H. Szatylowicz, P. A. Wieczorkiewicz and T. M. Krygowski, Aromaticity Concepts Derived from Experiments, Science, 2022, 4(2), 24, DOI:10.3390/sci4020024.
- S. Fias, S. Van Damme and P. Bultinck, Multidimensionality of delocalization indices and nucleus independent chemical shifts in polycyclic aromatic hydrocarbons, J. Comput. Chem., 2008, 29(3), 358–366, DOI:10.1002/jcc.20794.
- A. R. Katritzky, M. Karelson, S. Sild, T. M. Krygowski and K. Jug, Aromaticity as a quantitative concept. 7. Aromaticity reaffirmed as a multidimensional characteristic, J. Org. Chem., 1998, 63(15), 5228–5231, DOI:10.1021/jo970939b.
- M. K. Cyranski, T. M. Krygowski, A. R. Katritzky and P. v. R. Schleyer, To what extent can aromaticity be defined uniquely?, J. Org. Chem., 2002, 67(4), 1333–1338, DOI:10.1021/jo016255s.
- L. Zhao, R. Grande-Aztatzi, C. Foroutan-Nejad, J. M. Ugalde and G. Frenking, Aromaticity, the Hückel 4n + 2 rule and magnetic current, ChemistrySelect, 2017, 2(3), 863–870, DOI:10.1002/slct.201602080.
- J. Poater, I. Garca-Cruz, F. Illas and M. Solà, Discrepancy between common local aromaticity measures in a series of carbazole derivatives, Phys. Chem. Chem. Phys., 2004, 6(2), 314–318, 10.1039/B309965B.
- M. Alonso and B. Herradón, A universal scale of aromaticity for π-organic compounds, J. Comput. Chem., 2010, 31(5), 917–928, DOI:10.1002/jcc.21377.
- F. Feixas, E. Matito, J. Poater and M. Solà, Quantifying aromaticity with electron delocalisation measures, Chem. Soc. Rev., 2015, 44(18), 6434–6451, 10.1039/C5CS00066A.
- T. Woller, P. Geerlings, F. De Proft, B. Champagne and M. Alonso, Aromaticity as a guiding concept for spectroscopic features and nonlinear optical properties of porphyrinoids, Molecules, 2018, 23(6), 1333, DOI:10.3390/molecules23061333.
- M. Máximo-Canadas, R. S. S. Oliveira, M. A. S. Oliveira and I. Borges Jr, A New Set of Aromaticity Descriptors Based on the Electron Density Employing the Distributed Multipole Analysis (DMA), ACS Omega, 2025, 10(14), 14157–14175, DOI:10.1021/acsomega.4c11451.
- M. N. C. Zarycz, M. A. Schiel, H. A. Baldoni and R. D. Enriz, SD aromaticity index: a new assessment of aromaticity based on Ramsey spin dipolar contribution to NMR spin–spin coupling constants, Phys. Chem. Chem. Phys., 2025, 27, 22974–22989, 10.1039/D5CP02867A.
- K. Balasubramanian, New Insights into Aromaticity through Novel Delta Polynomials and Delta Aromatic Indices, Symmetry, 2024, 16(4), 391 CrossRef CAS . Available from: https://www.mdpi.com/2073-8994/16/4/391.
- D. W. Szczepanik, E. Zak, K. Dyduch and J. Mrozek, Electron delocalization index based on bond order orbitals, Chem. Phys. Lett., 2014, 593, 154–159, DOI:10.1016/j.cplett.2014.01.006.
- D. W. Szczepanik, M. Andrzejak, K. Dyduch, E. Zak, M. Makowski and G. Mazur, et al., A uniform approach to the description of multicenter bonding, Phys. Chem. Chem. Phys., 2014, 16(38), 20514–20523, 10.1039/C4CP02932A.
- D. W. Szczepanik, M. Andrzejak, J. Dominikowska, B. Pawełek, T. M. Krygowski and H. Szatylowicz, et al., The electron density of delocalized bonds (EDDB) applied for quantifying aromaticity, Phys. Chem. Chem. Phys., 2017, 19(42), 28970–28981, 10.1039/C7CP06114E.
- J. Kruszewski and T. Krygowski, Definition of aromaticity basing on the harmonic oscillator model, Tetrahedron Lett., 1972, 13(36), 3839–3842, DOI:10.1016/S0040-4039(01)94175-9.
- T. M. Krygowski and M. K. Cyranski, Structural aspects of aromaticity, Chem. Rev., 2001, 101(5), 1385–1420, DOI:10.1021/cr990326u.
- E. M. Arpa and B. Durbeej, HOMER: a reparameterization of the harmonic oscillator model of aromaticity (HOMA) for excited states, Phys. Chem. Chem. Phys., 2023, 25(25), 16763–16771, 10.1039/D3CP00842H.
- P. v. R. Schleyer, C. Maerker, A. Dransfeld, H. Jiao and N. J. van Eikema Hommes, Nucleus-independent chemical shifts: a simple and efficient aromaticity probe, J. Am. Chem. Soc., 1996, 118(26), 6317–6318, DOI:10.1021/ja960582d.
- J. Gauss, Calculation of NMR chemical shifts at second-order many-body perturbation theory using gauge-including atomic orbitals, Chem. Phys. Lett., 1992, 191(6), 614–620, DOI:10.1016/0009-2614(92)85598-5.
- M. Bühl and A. Hirsch, Spherical Aromaticity of Fullerenes, Chem. Rev., 2001, 101(5), 1153–1184, DOI:10.1021/cr990332q.
- J. Poater, M. Duran, M. Solà and B. Silvi, Theoretical evaluation of electron delocalization in aromatic molecules by means of atoms in molecules (AIM) and electron localization function (ELF) topological approaches, Chem. Rev., 2005, 105(10), 3911–3947, DOI:10.1021/cr030085x.
- X. Fradera, M. A. Austen and R. F. W. Bader, The Lewis model and beyond, J. Phys. Chem. A, 1999, 103(2), 304–314, DOI:10.1021/jp983362q.
- R. F. W. Bader, A quantum theory of molecular structure and its applications, Chem. Rev., 1991, 91(5), 893–928, DOI:10.1021/cr00005a013.
- J. Poater, M. Solà, M. Duran and X. Fradera, The calculation of electron localization and delocalization indices at the Hartree–Fock, density functional and post-Hartree–Fock levels of theory, Theor. Chem. Acc., 2002, 107(6), 362–371, DOI:10.1007/s00214-002-0356-8.
- R. F. W. Bader, Atoms in molecules: a quantum theory, Oxford University, Oxford, 1990 Search PubMed.
- J. Poater, X. Fradera, M. Duran and M. Solà, The delocalization index as an electronic aromaticity criterion: application to a series of planar polycyclic aromatic hydrocarbons, Chem. – Eur. J., 2003, 9(2), 400–406, DOI:10.1002/chem.200390041.
- J. Poater, X. Fradera, M. Duran and M. Solà, An insight into the local aromaticities of polycyclic aromatic hydrocarbons and fullerenes, Chem. – Eur. J., 2003, 9(5), 1113–1122, DOI:10.1002/chem.200390128.
- R. L. Fulton, Sharing of electrons in molecules, J. Phys. Chem., 1993, 97(29), 7516–7529, DOI:10.1021/j100131a021.
- R. F. W. Bader, A. Streitwieser, A. Neuhaus, K. E. Laidig and P. Speers, Electron delocalization and the Fermi hole, J. Am. Chem. Soc., 1996, 118(21), 4959–4965, DOI:10.1021/ja953563x.
- E. Matito, M. Duran and M. Solà, The aromatic fluctuation index (FLU): a new aromaticity index based on electron delocalization, J. Chem. Phys., 2005, 122(1), 014109, DOI:10.1063/1.1824895.
- E. Matito, M. Duran and M. Solà, Erratum: “The aromatic fluctuation index (FLU): A new aromaticity index based on electron delocalization” [J. Chem Phys. 122, 014109 (2005)], J. Chem. Phys., 2006, 125(5), 059901, DOI:10.1063/1.2222352.
- K. Jorner, F. Feixas, R. Ayub, R. Lindh, M. Solà and H. Ottosson, Analysis of a compound class with triplet states stabilized by potentially Baird aromatic [10]annulenyl dicationic rings, Chem. – Eur. J., 2016, 22(8), 2793–2800, DOI:10.1002/chem.201504924.
- E. Matito, F. Feixas and M. Solà, Electron delocalization and aromaticity measures within the Hückel molecular orbital method, J. Mol. Struct.: THEOCHEM, 2007, 811(1–3), 3–11, DOI:10.1016/j.theochem.2007.01.015.
- M. Giambiagi, M. S. de Giambiagi, C. D. dos Santos Silva and A. P. de Figueiredo, Multicenter bond indices as a measure of aromaticity, Phys. Chem. Chem. Phys., 2000, 2(15), 3381–3392, 10.1039/B002009P.
- P. Bultinck, M. Rafat, R. Ponec, B. Van Gheluwe, R. Carbo-Dorca and P. Popelier, Electron delocalization and aromaticity in linear polyacenes: atoms in molecules multicenter delocalization index, J. Phys. Chem. A, 2006, 110(24), 7642–7648, DOI:10.1021/jp0609176.
- J. Cioslowski, E. Matito and M. Solà, Properties of aromaticity indices based on the one-electron density matrix, J. Phys. Chem. A, 2007, 111(28), 6521–6525, DOI:10.1021/jp0716132.
- S. Escayola, C. Tonnelé, E. Matito, A. Poater, H. Ottosson and M. Solà, et al., Guidelines for Tuning the Excited State Hückel-Baird Hybrid Aromatic Character of Pro-Aromatic Quinoidal Compounds, Angew. Chem., Int. Ed., 2021, 60(18), 10255–10265, DOI:10.1002/anie.202100261.
- D. W. Szczepanik, A new perspective on quantifying electron localization and delocalization in molecular systems, Comput. Theor. Chem., 2016, 1080, 33–37, DOI:10.1016/j.comptc.2016.02.003.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb and J. R. Cheeseman, et al., Gaussian 16, Revision A.03, Wallingford, CT, 2016. Software citation Search PubMed.
- A. D. Becke, Density-functional thermochemistry. I. The effect of the exchange-only gradient correction, J. Chem. Phys., 1992, 96(3), 2155–2160, DOI:10.1063/1.462066.
- C. Lee, W. Yang and R. G. Parr, Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density, Phys. Rev. B: Condens. Matter Mater. Phys., 1988, 37(2), 785–789, DOI:10.1103/PhysRevB.37.785.
- R. Krishnan, J. S. Binkley, R. Seeger and J. A. Pople, Self-consistent molecular orbital methods. XX. A basis set for correlated wave functions, J. Chem. Phys., 1980, 72(1), 650–654, DOI:10.1063/1.438955.
- F. Feixas, E. Matito, J. Poater and M. Solà, Aromaticity of distorted benzene rings: exploring the validity of different indicators of aromaticity, J. Phys. Chem. A, 2007, 111(20), 4513–4521, DOI:10.1021/jp0703206.
- D. W. Szczepanik, M. Solà, M. Andrzejak, B. Pawełk, J. Dominikowska and M. Kukułka, et al., The role of the long-range exchange corrections in the description of electron delocalization in aromatic species, J. Comput. Chem., 2017, 38(18), 1640–1654 CrossRef CAS PubMed . Available from: https://onlinelibrary.wiley.com/doi/abs/10.1002/jcc.24805.
- M. Torrent-Sucarrat, S. Navarro, F. P. Cosso, J. M. Anglada and J. M. Luis, Relevance of the DFT method to study expanded porphyrins with different topologies, J. Comput. Chem., 2017, 38(32), 2819–2828, DOI:10.1002/jcc.25074.
- J. Pedersen and K. V. Mikkelsen, A benchmark study of aromaticity indexes for benzene, pyridine and the diazines – I. Ground state aromaticity, RSC Adv., 2022, 12, 2830–2842, 10.1039/D2RA00093H.
- J. Pedersen and K. V. Mikkelsen, A benchmark study of aromaticity indexes for benzene, pyridine and the diazines – II. Excited state aromaticity, J. Phys. Chem. A, 2023, 127, 122–130, DOI:10.1021/acs.jpca.2c07059.
- F. W. Biegler-König, R. F. W. Bader and T. H. Tang, Calculation of the average properties of atoms in molecules. II, J. Comput. Chem., 1982, 3(3), 317–328, DOI:10.1002/jcc.540030306.
- E. Matito, ESI-3D: Electron Sharing Indices Program for 3D Molecular Space Partitioning, University of Girona (UdG), Catalonia, Spain, 2006, Available Online: https://quantchemdev.github.io/resources.html (accessed on 26 May 2023) Search PubMed.
- E. D. Glendening, C. R. Landis and F. Weinhold, NBO 7.0: New vistas in localized and delocalized chemical bonding theory, J. Comput. Chem., 2019, 40(25), 2234–2241, DOI:10.1002/jcc.25873.
- A. E. Reed, R. B. Weinstock and F. Weinhold, Natural population analysis, J. Chem. Phys., 1985, 83(2), 735–746, DOI:10.1063/1.449486.
- D. W. Szczepanik, RunEDDB, Available from: EDDB (https://www.aromaticity.eu).
- J. Grèbol-Tomàs, E. Matito and P. Salvador, Can Aromaticity Be Evaluated Using Atomic Partitions Based on the Hilbert-Space?, Chem. – Eur. J., 2024, 30(44), e202401282, DOI:10.1002/chem.202401282.
- K. Wolinski, J. F. Hinton and P. Pulay, Efficient implementation of the gauge-independent atomic orbital method for NMR chemical shift calculations, J. Am. Chem. Soc., 1990, 112(23), 8251–8260, DOI:10.1021/ja00179a005.
- C. Corminboeuf, T. Heine, G. Seifert, P. V. R. Schleyer and J. Weber, Induced magnetic fields in aromatic [n]-annulenes—interpretation of NICS tensor components, Phys. Chem. Chem. Phys., 2004, 6, 273–276, 10.1039/B313383B.
- N. C. Baird, Quantum organic photochemistry. II. Resonance and aromaticity in the lowest 3ππ* state of cyclic hydrocarbons, J. Am. Chem. Soc., 1972, 94(14), 4941–4948, DOI:10.1021/ja00769a025.
- M. Rosenberg, C. Dahlstrand, K. Kilsa and H. Ottosson, Excited state aromaticity and antiaromaticity: opportunities for photophysical and photochemical rationalizations, Chem. Rev., 2014, 114(10), 5379–5425, DOI:10.1021/cr300471v.
- R. W. Havenith, P. W. Fowler and E. Steiner, Substituent effects on induced current densities in penta-and heptafulvenes, J. Chem. Soc., Perkin Trans. 2, 2002,(3), 502–507, 10.1039/B109812H.
- H. Möllerstedt, M. C. Piqueras, R. Crespo and H. Ottosson, Fulvenes, fulvalenes, and azulene: are they aromatic chameleons?, J. Am. Chem. Soc., 2004, 126(43), 13938–13939, DOI:10.1021/ja045729c.
- S. Yadav, O. El Bakouri, K. Jorner, H. Tong, C. Dahlstrand and M. Solà, et al., Exploiting the Aromatic Chameleon Character of Fulvenes for Computational Design of Baird-Aromatic Triplet Ground State Compounds, Chem. – Asian J., 2019, 14(10), 1870–1878, DOI:10.1002/asia.201801821.
- W. Collier, S. Saebø and C. U. Pittman Jr, Nucleus independent chemical shift evaluation of the aromaticity of pentafulvene and its exocyclic Si, Ge, and Sn derivatives, J. Mol. Struct.: THEOCHEM, 2001, 549(1–2), 1–8, DOI:10.1016/S0166-1280(01)00482-1.
- S. Saebø, S. Stroble, W. Collier, R. Ethridge, Z. Wilson and M. Tahai, et al., Aromatic character of tria-and pentafulvene and their exocyclic Si, Ge, and Sn derivatives. An ab initio study, J. Org. Chem., 1999, 64(4), 1311–1318, DOI:10.1021/jo982279w.
- A. P. Scott, I. Agranat, P. U. Biedermann, N. V. Riggs and L. Radom, Fulvalenes, fulvenes, and related molecules: an ab initio study, J. Org. Chem., 1997, 62(7), 2026–2038, DOI:10.1021/jo962407l.
- H. Braunschweig, I. Fernández, G. Frenking and T. Kupfer, Structural evidence for antiaromaticity in free boroles, Angew. Chem., Int. Ed., 2008, 47(10), 1951–1954, DOI:10.1002/anie.200704771.
- J. O. C. Jimenez-Halla, E. Matito, M. Solà, H. Braunschweig, C. Hörl and I. Krummenacher, et al., A theoretical study of the aromaticity in neutral and anionic borole compounds, Dalton Trans., 2015, 44(15), 6740–6747, 10.1039/C4DT03445G.
- C. Hong, J. Baltazar and J. D. Tovar, Manifestations of antiaromaticity in organic materials: Case studies of cyclobutadiene, borole, and pentalene, Eur. J. Org. Chem., 2022,(13), e202101343, DOI:10.1002/ejoc.202101343.
- H. Braunschweig, C. W. Chiu, K. Radacki and T. Kupfer, Synthesis and structure of a carbene-stabilized π-boryl anion, Angew. Chem., Int. Ed., 2010, 49, 2041–2044, DOI:10.1002/anie.200906884.
- S. Dong, W. Wang and J. Zhu, Design of borole-and silylene-based frustrated Lewis pairs for dinitrogen activation promoted by aromaticity: A combined DFT and Machine Learning study, Inorg. Chem., 2025, 64(36), 18195–18205, DOI:10.1021/acs.inorgchem.5c02343.
- E. Hückel, Grundzüge der Theorie ungesättigter und aromatischer Verbindungen, Z. Elektrochem., 1937, 43(9), 752–788 CrossRef . Available from: https://onlinelibrary.wiley.com/doi/abs/10.1002/bbpc.19370430907.
- D. Gibney and J. N. Boyn, Tunable Aromaticity and Biradical Character in Tetrathiafulvalene and Tetraselenafulvalene Derivatives, J. Phys. Chem. A, 2025, 129(39), 9001–9010, DOI:10.1021/acs.jpca.5c05283.
- J. Camacho Gonzalez and A. Muñoz-Castro, Survey of the magnetic behavior of [TTF] and [TTF2]2+ from shielding tensor maps, Chem. Phys. Lett., 2012, 543, 184–187, DOI:10.1016/j.cplett.2012.06.028.
- I. Hargittai, J. Brunvoll, M. Kolonits and V. Khodorkovsky, Tetrathiafulvalene: gas-phase molecular structure from electron diffraction, J. Mol. Struct., 1994, 317(3), 273–277, DOI:10.1016/0022-2860(93)07873-U.
- C. Katan, First-principles study of the structures and vibrational frequencies for Tetrathiafulvalene TTF and TTF-d4 in different oxidation states, J. Phys. Chem. A, 1999, 103(10), 1407–1413, DOI:10.1021/jp983941v.
- S. B. Nielsen, M. B. Nielsen and H. J. A. Jensen, The tetrathiafulvalene dication in the gas phase: its formation and stability, Phys. Chem. Chem. Phys., 2003, 5(7), 1376–1380, 10.1039/B300537B.
- P. J. Mayer, O. El Bakouri, T. Holczbauer, G. F. Samu, C. Janáky and H. Ottosson, et al., Structure-Property Relationships in Unsymmetric Bis (antiaromatics): Who Wins the Battle between Pentalene and Benzocyclobutadiene?, J. Org. Chem., 2020, 85(8), 5158–5172, DOI:10.1021/acs.joc.9b03119.
- I. Willner and M. Rabinovitz, 1,9-Dimethyldibenzo[b,f]pentalene dication and dianion. New 14π and 18π aromatic systems, J. Am. Chem. Soc., 1978, 100(1), 337–338, DOI:10.1021/ja00469a085.
- E. Clar, The Aromatic Sextet, John Wiley & Sons, New York, 1972 Search PubMed.
- M. Solà, Forty years of Clar’s aromatic π-sextet rule, Front. Chem., 2013, 1, 22, DOI:10.3389/fchem.2013.00022.
- G. Portella, J. Poater, J. M. Bofill, P. Alemany and M. Solà, Local Aromaticity of [n]Acenes, [n]Phenacenes, and [n]Helicenes (n = 1–9), J. Org. Chem., 2005, 70(7), 2509–2521, DOI:10.1021/jo0480388.
- J. Poater, M. Duran and M. Solà, Aromaticity determines the relative stability of kinked vs. straight topologies in polycyclic aromatic hydrocarbons, Front. Chem., 2018, 6, 561, DOI:10.3389/fchem.2018.00561.
- J. Dominikowska and M. Palusiak, Does the concept of Clar’s aromatic sextet work for dicationic forms of polycyclic aromatic hydrocarbons?—testing the model against charged systems in singlet and triplet states, Phys. Chem. Chem. Phys., 2011, 13(25), 11976–11984, 10.1039/C1CP20530G.
- H. Szatyłowicz, T. M. Krygowski, M. Palusiak, J. Poater and M. Solà, Routes of π-electron delocalization in 4-substituted-1,2-benzoquinones, J. Org. Chem., 2011, 76(2), 550–556, DOI:10.1021/jo102065e.
- H. Szatylowicz, T. M. Krygowski, M. Solà, M. Palusiak, J. Dominikowska and O. A. Stasyuk, et al., Why 1,2-quinone derivatives are more stable than their 2,3-analogues?, Theor. Chem. Acta, 2015, 134(5), 35, DOI:10.1007/s00214-015-1635-5.
- A. Jezuita, H. Szatylowicz, P. H. Marek and T. M. Krygowski, Aromaticity of the most stable adenine and purine tautomers in terms of Hückel’s 4N + 2 principle, Tetrahedron, 2019, 75(35), 130474, DOI:10.1016/j.tet.2019.130474.
- O. A. Stasyuk, H. Szatyłowicz and T. M. Krygowski, Effect of the H-bonding on aromaticity of purine tautomers, J. Org. Chem., 2012, 77(8), 4035–4045, DOI:10.1021/jo300406r.
- C. Corminboeuf, T. Heine, G. Seifert, P. von Ragué Schleyer and J. Weber, Induced magnetic fields in aromatic [n]-annulenes—interpretation of NICS tensor components, Phys. Chem. Chem. Phys., 2004, 6(2), 273–276, 10.1039/B313383B.
- H. Fallah-Bagher-Shaidaei, C. S. Wannere, C. Corminboeuf, R. Puchta and P. V. R. Schleyer, Which NICS aromaticity index for planar π rings is best?, Org. Lett., 2006, 8(5), 863–866, DOI:10.1021/ol0529546.
| This journal is © the Owner Societies 2026 |




