Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Quantitative electronic structure determination of N-heterotriangulene derivatives adsorbed on Au(111)
Jakob Steidela,
Ina Michalskyb,
Milan Kivala b and
Petra Tegeder
b and
Petra Tegeder *a
*a
aPhysikalisch-Chemisches Institut, Ruprecht-Karls Universität Heidelberg, Im Neuenheimer Feld 253, 69120, Heidelberg, Germany. E-mail: tegeder@uni-heidelberg.de; Tel: +49 6221 548475
bOrganisch-Chemisches Institut, Ruprecht-Karls Universität Heidelberg, Im Neuenheimer Feld 270, 69120, Heidelberg, Germany
First published on 10th February 2026
Abstract
N-heterotriangulenes (N-HTAs) are promising organic semiconductors for applications in field effect transistors and solar cells. Thereby the electronic structure of organic/metal interfaces and thin films is essential for the performance of organic-molecule-based devices. Here, we studied the electronic properties of two different N-HTAs, the N-HTA-550 and N-HTA-557, the latter containing an additional 7-membered ring, adsorbed on Au(111) using two-photon photoemission spectroscopy. We quantitatively determined the energetic positions of several occupied and unoccupied molecular (transport levels) and excitonic states (optical gap) in detail. The additional –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C– bridge forming the 7-membered ring in N-HTA-557 resulted in a pronounced increase of the electron affinity by 0.92 eV from 2.19 eV in N-HTA-550/Au(111) to 3.11 eV in N-HTA-557/Au(111) due to the increase of the π-conjugated electron system, while the first ionization potential is nearly unaffected. Structural variation or substitution pattern in N-HTAs foster the opportunity for tailoring their electronic properties.
C– bridge forming the 7-membered ring in N-HTA-557 resulted in a pronounced increase of the electron affinity by 0.92 eV from 2.19 eV in N-HTA-550/Au(111) to 3.11 eV in N-HTA-557/Au(111) due to the increase of the π-conjugated electron system, while the first ionization potential is nearly unaffected. Structural variation or substitution pattern in N-HTAs foster the opportunity for tailoring their electronic properties.
1. Introduction
Organic semiconductors are potential candidates for many applications in (opto)electronic devices such as solar cells, light emitting diodes, or field effect transistors.1–4 The electronic and adsorption properties of the organic compound at the interface to an inorganic electrode and within the molecular film are crucial for device performance. For instance, the orientation of molecules plays a central role for light absorption/emission and charge transport properties. Adsorbate/adsorbate and adsorbate/substrate interactions influence the adsorption geometry and the growth of molecular films, and accordingly, the electronic structure at the organic/inorganic interface (energy level alignment)5–10 and within the molecular film.11–18N-heteropolycyclic aromatic compounds are promising candidates for organic semiconductors as electron-transporting materials for use in field-effect transistors and solar cells.19 The introduction of nitrogen atoms (pyridinic nitrogens) into the π-electron backbone of polycyclic aromatic hydrocarbons increases the electron affinity, i.e. the electron binding capability, while leaving the size of the optical gap nearly unaffected.20–23 Pyrrolic nitrogens make the system more electron rich, that is lower the ionization potential.24
In the present contibution, we studied N-heterotriangulenes (N-HTAs, see Fig. 1), ortho-bridged triphenylamines,24,25 which represent a class of functional molecules with high potential for application in optoelectronic materials,26–29 for example as electron donating compounds in donor/acceptor systems.30 Using structural variation (N-HTA-550 vs. -557) or substitution patterns opens the opportunity for fine-tuning the electronic properties.24,27,31 Therefore, we recently analyzed the electronic properties and absorption spectra of two N-HTA derivatives, N-HTA-550 and N-HTA-557 (Fig. 1), at the interface to Au(111) and within thin molecular films using vibrational and electronic high resolution electron energy loss spectroscopy (HREELS) in combination with quantum chemical calculations. We found that the additional –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C– bridge forming the 7-membered ring in N-HTA-557 resulted in a pronounced reduction of the optical gap size by 0.9 eV from 3.4 eV in N-HTA-550 to 2.5 eV in N-HTA-557 due to an increase of the π-conjugated electron system.32 Herein, we employed two-photon photoemission (2PPE) spectroscopy to determine quantitatively the energetic positions of unoccupied and occupied molecular electronic as well as excitonic states.
C– bridge forming the 7-membered ring in N-HTA-557 resulted in a pronounced reduction of the optical gap size by 0.9 eV from 3.4 eV in N-HTA-550 to 2.5 eV in N-HTA-557 due to an increase of the π-conjugated electron system.32 Herein, we employed two-photon photoemission (2PPE) spectroscopy to determine quantitatively the energetic positions of unoccupied and occupied molecular electronic as well as excitonic states.
2PPE has been proven to be a powerful tool to elucidate the electronic structure of metal/organic interfaces and thin films. This includes electron affinity levels and ionization potentials (transport levels) as well as optical gaps and exciton binding energies.33–54 Recently, we investigated the energy level alignment of a π-expanded N-heterotriangulene derivative adorbed on Au(111) by means of 2PPE and determined transport levels, the optical gap and accordingly the exciton binding energy.54
In the present contribution we thoroughly elucidated the electronic structure of N-HTA-550/Au(111) and N-HTA-557/Au(111) in detail, including the identification of transport levels, the optical gap and hence the exciton binding energy. In particular for applications of these materials in organic field effect transistors or solar cells knowledge about the energetic positions of transport levels is of utmost relevance.
2. Experimental details
The 2PPE experiments were performed under ultra-high vacuum (UHV, < 2 × 10−10 mbar) conditions at a sample temperature of around 90 K. The Au(111) single crystal was prepared by standard procedure of Ar+ sputtering and annealing. The N-HTAs were synthesized according to the procedures reported in ref. 24. The compounds were deposited from an effusion cell held at 373 K for N-HTA-550 and at 393 K for N-HTA-557 onto the Au(111) sample held at 200 K for N-HTA-550 deposition and at 250 K in the case of N-HTA-557. The coverage was determined by temperature-programmed desorption (TPD) measurements (see below). 2PPE allows investigation of occupied, unoccupied electronic and excitonic states in a pump–probe scheme. To discriminate between occupied and unoccupied molecular electronic states photon-energy dependent measurements were performed as described in ref. 37,38,42–44. The tunable femtosecond laser system, which delivers laser pulses over a wide range of photon energies, and the 2PPE setup are described elsewhere.553. Results and discussion
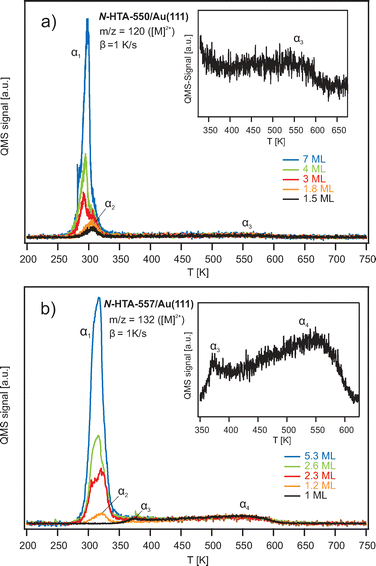
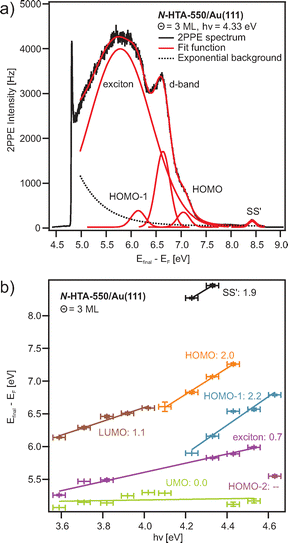
To determine the adsorbate coverage and to analyze the adsorption behavior of the N-HTAs adsorbed on Au(111) we performed coverage-dependent TPD measurements. Fig. 2 shows TPD data for various initial N-HTA-550 (Fig. 2a) and N-HTA-557 (Fig. 2b) coverages. In the case of N-HTA-550/Au(111) three desorption features labeled as α1, α2, and α3 are detected. α1 shows a clear zero-order desorption characteristics (the desorption rate is independent of the coverage) and does not saturate even for higher coverages. Thus, α1 can be assigned to the desorption from the multilayer. We attribute α3 to desorption from the monolayer and α2 to the desorption from the second layer. We defined a monolayer (ML) to the desorption spectrum, in which α3 is saturated. The integral of this spectrum is used as a reference to determine the coverage of all other TPD spectra shown in Fig. 2a. For N-HTA-557/Au(111) (Fig. 2b) α1 is also assigned to the desorption from the multilayer, however it shows a first-order desorption characteristics rather than zero-order. As in the case of N-HTA-550 a desorption peak from the second layer (α2) is observed. Apart from the monolayer desorption (α4), a desorption feature labeled as α3 is detected. We attribute α3 to desorption from a more densely packed compressed phase, as reported for benzene56,57 or other aromatic organic molecules on coinage metal surfaces.41,58–63 Note that the peak area of α4 has been used to define a coverage of 1 ML N-HTA-557/Au(111). Comparing both N-HTA/Au(111) systems, the desorption behavior in the multilayer regime (zero-order vs. first-order desorption) and in the low-coverage range (compressed phase) differs, indicating that the adsorbate–adsorbate (lateral) as well as the adsorbate–substrate (vertical) interactions are different. Consequently, we assume that the film morphology of the two systems differs.To gain insight into the energy level alignment, i.e., the energetic positions of adsorbate-derived occupied as well as unoccupied electronic states we performed photon-energy-dependent 2PPE measurements.41–44,48,49,51,52,64 Fig. 3(a) shows an exemplarily 2PPE spectrum of 3 ML N-HTA-550 adsorbed on Au(111) recorded with hν = 4.33 eV (for additional 2PPE data see supplementary information (SI)). Several photoemission features could be detected some of them are related to the Au(111) surface such as the gold d-bands (at −2.15 eV and −2.74 eV with respect to Fermi energy (EF) of the Au(111) surface) and the shifted surface state (SS′, at −0.22 eV) others are N-HTA-550-derived 2PPE peaks. On the basis of their photon-energy-dependency they are assigned to occupied and unoccupied molecular states as well as an excitonic state (see Fig. 3b). We detect three photoemission peaks in the photon energy-dependent spectra with a slope of around two, thus they are originating from occupied single-electron states. In the molecular orbital picture, these states can be assigned to the HOMO, HOMO−1 and HOMO−2, possessing binding energies with respect to EF of −1.60 ± 0.16, −2.47 ± 0.15, and −3.71 ± 0.15 eV, respectively. Moreover, we find two peaks which scale linearly with incident photon energy. They stem from intermediate states located at 2.56 ± 0.14 eV and 1.56 ± 0.15 eV with respect to EF. We attribute the states to an unoccupied single-electron state, which is well represented by the LUMO of N-HTA-550, and an higher lying electronic state (see below).
In Fig. 4(a), we summarize the level alignments with respect to the vacuum level. As discussed before,42,43,48,65,66 depending on the excitation mechanism, 2PPE probes transport levels. Unoccupied single-electron states can be populated via an electron transfer from the metal substrate to the molecule creating a negative ion resonance and occupied single-electron states are ionized, creating a positive ion resonance. Thus, we obtain the electron affinities (EA) and the ionization potentials (IP) of the investigated N-HTA-550 molecule. In addition, in 2PPE also intramolecular electronic excitation is possible, which corresponds to exciton generation, i.e. formation of an electron–hole pair, in which the molecule remains overall neutral. The minimal energy required for this process is the optical gap (Eopt), which corresponds to the excitation energy of the lowest excited singlet state. Eopt is smaller than the difference between the lowest IP and the highest EA, which are according to Koopman's theorem given as the negative orbital energies of the HOMO and the LUMO. The so-called transport gap is given as Etransp = IP−EA. The difference between the transport gap and the optical gap corresponds to the exciton binding energy Etransp−Eopt = EB.36,67 Combining our results described above, we obtain a transport gap of 4.19 eV (IP = 6.38 ± 0.16 eV, EA = 2.19 ± 0.14 eV), and an optical gap of 3.19 eV, the excitation energy of the lowest excited singlet state. Hence, EB of the lowest excitonic state amounts to 1.0 eV. Note that with electronic HREELS measurements an optical gap of 3.4 eV has been determined,32 thus 210 meV larger than the value obtained here. With this value the resulting exciton binding energy is 790 meV. Apart from the lowest IP, we determined two higher IPs corresponding to lower lying occupied single-electron states of N-HTA-550, which are described in the molecular orbital picture by the HOMO−1 and HOMO−2. In addition, we identified a further unoccupied molecular state (unoccupied molecular orbital: UMO) located 0.41 ± 0.14 eV above the vacuum level (Evac, unoccupied final state).
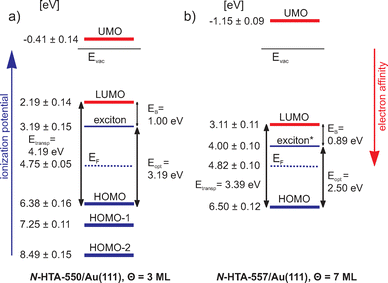 | ||
| Fig. 4 (a) Energy level diagram of 3 ML N-HTA-550/Au(111) with respect to the vacuum level. The blue levels are the ionization potentials as well as an excitonic state and the red ones are the electron affinities, which are in the molecular orbital picture represented by the energies of the HOMOs, LUMO and UMO (unoccupied molecular orbital), respectively. EF is the Au(111) Fermi level and Evac is the vacuum level. (b) Energy level diagram of 7 ML N-HTA-557/Au(111). * denotes the value of the optical gap size determined by electronic HREELS from ref. 32. | ||
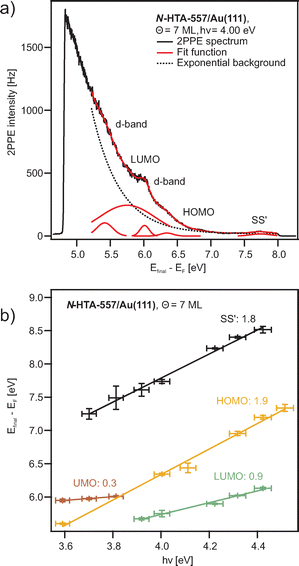
Fig. 5(a) shows an exemplarily 2PPE spectrum of 7 ML N-HTA-557 adsorbed on Au(111) recorded with hν = 4.0 eV (for additional 2PPE data see SI). Similar to the N-HTA-550/Au(111) system we detect photoemission features from the gold substrate (d-bands and SS′) but also adsorbate-induced 2PPE peaks. Based on their photon-energy-dependency they are assigned to the LUMO and HOMO as well as to an unoccupied final state (UMO) (see Fig. 5b). In Fig. 4(b), the level alignment with respect to the vacuum level is summarized. In comparison to N-HTA-550/Au(111) the 2PPE data for N-HTA-557/Au(111) are less structured, i.e. the photoemission signals are very broad and featureless. This is most likely related to the film morphology and surface roughness, which seems to be different in both systems (see above). Nevertheless the HOMO (IP) of N-HTA-557/Au(111) is located at 6.50 ± 0.12 eV and the LUMO (EA) at 3.11 ± 0.11 eV, resulting in a transport gap of 3.39 eV. Thus, the transport gap size decrease by 0.8 eV from 4.19 eV in N-HTA-550/Au(111) to 3.39 eV in N-HTA-557/Au(111), which is due to a pronounced increase of the EA by 0.92 eV as well as a slight rise of the IP by 120 meV in N-HTA-557/Au(111). Hence, the additional –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C– bridge forming the 7-membered ring in N-HTA-557 which leads to an increase of the π-conjugated electron system strongly affects the EA level. Taking into account the optical gap size of 2.5 eV32 measured with electronic HREELS would result in an exciton binding energy of 0.89 eV, which is similar to the value found for N-HTA-550/Au(111).
C– bridge forming the 7-membered ring in N-HTA-557 which leads to an increase of the π-conjugated electron system strongly affects the EA level. Taking into account the optical gap size of 2.5 eV32 measured with electronic HREELS would result in an exciton binding energy of 0.89 eV, which is similar to the value found for N-HTA-550/Au(111).
Concerning the organic/metal interface, we found for both systems a work function (Φ) decrease from 5.50 eV for the bare Au(111) surface to 4.8 eV at a coverage of 1 ML N-HTA/Au(111), thus a reduction of 700 meV (see Fig. S5 and S11). The reduction in work function can be explained by an induced interface dipole. For higher coverages Φ remains unchanged. For 1 ML N-HTA-550/Au(111) we observed 3 adsorbate-induced photoemission peaks (see Fig. S5 and S6), which can be assigned to the HOMO, HOMO−1 and an unoccupied final state (see Fig. S7). An excitonic state or photoemission related to the LUMO as detected for multilayer coverages are not found, most likely due to the strong electronic coupling between molecular electronic states and metallic band resulting in ultrashort lifetimes. For ≈1 ML N-HTA-557/Au(111) the resulting 2PPE data only resolve electronic states related to the Au(111) surface (see Fig. S11). Note that, no dispersing molecular states at the metal/organic interface of both systems have been observed in our angle-resolved 2PPE measurements, which for instance have been identified for other N-heteropolycycles adsorbed on Au(111).51,52 Thus we exclude hybridization between the molecular states and metal bands.
4. Conclusion
Two N-heterotriangulene (N-HTA) derivatives, the N-HTA-550 and N-HTA-557 adsorbed on Au(111) have been investigated using energy-resolved two-photon photoemission (2PPE) spectroscopy, focusing on the quantitative determination of occupied and unoccupied molecular (transport states) and excitonic states (optical gap) as well as the influence of the –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C– bridge and hence the formation of the 7-membered ring in N-HTA 557 on the electronic structure. For both compounds we were able to assign several 2PPE features to transport levels (electron affinities (EAs) and ionization potentials (IPs)) and excitonic states. Moreover exciton binding energies have been determined. The additional –C
C– bridge and hence the formation of the 7-membered ring in N-HTA 557 on the electronic structure. For both compounds we were able to assign several 2PPE features to transport levels (electron affinities (EAs) and ionization potentials (IPs)) and excitonic states. Moreover exciton binding energies have been determined. The additional –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C– bridge forming the 7-membered ring in N-HTA-557 which leads to an increase of the π-conjugated electron system strongly affected the first EA level and resulted in a pronounced rise of the EA by 0.92 eV from 2.19 eV in N-HTA-550/Au(111) to 3.11 eV in N-HTA-557/Au(111). In contrast, the first IP is barely affected. Using structural variation or substitution pattern may pave the way for fine-tuning the electronic properties of N-HTAs at will.
C– bridge forming the 7-membered ring in N-HTA-557 which leads to an increase of the π-conjugated electron system strongly affected the first EA level and resulted in a pronounced rise of the EA by 0.92 eV from 2.19 eV in N-HTA-550/Au(111) to 3.11 eV in N-HTA-557/Au(111). In contrast, the first IP is barely affected. Using structural variation or substitution pattern may pave the way for fine-tuning the electronic properties of N-HTAs at will.
Author contributions
J. S. performed the 2PPE measurements and analysed the results. I. M. synthesized the N-HTA derivatives. P. T. wrote the manuscript. M. K. supervised I. M. and P. T. supervised J. S. All authors reviewed the manuscript.Conflicts of interest
The authors have no conflicts to declare.Data availability
The data that support the findings of this study are available from the corresponding author upon request. Supplementary information (SI): two-photon photoemission data. See DOI: https://doi.org/10.1039/d5cp04526f.Acknowledgements
Funding by the German Research Foundation (DFG) through the collaborative research center CRC 1249 “N-Heteropolycycles as Functional Materials” (Project Number 281029004-SFB 1249, Projects A05 and B06) is gratefully acknowledged.References
- S. Reineke, F. Lindner, G. Schwartz, N. Seidler, K. Walzer, B. Lüssem and K. Leo, Nature, 2009, 459, 234 CrossRef PubMed.
- B. W. D'Andrade and S. R. Forrest, Adv. Mater., 2004, 16, 1585–1595 CrossRef.
- J. Lee, H. Chen, T. Batagoda, C. Coburn, P. I. Djurovich, M. E. Thompson and S. R. Forrest, Nat. Mater., 2016, 15, 92 CrossRef PubMed.
- L. Torsi, M. Magliulo, K. Manoli and G. Palazzo, Chem. Soc. Rev., 2013, 42, 8612–8628 RSC.
- H. Ishii, K. Sugiyama, E. Ito and K. Seki, Adv. Mater., 1999, 11, 605–625 Search PubMed.
- The Molecule-Metal Interface, ed. N. Koch, N. Ueno and A. Wee, Wiley-VCH, Weinheim, 2013 Search PubMed.
- M. Gruenewald, K. Wachter, M. Meissner, M. Kozlik, R. Forker and T. Fritz, Org. Electron., 2013, 14, 2177–2183 CrossRef.
- S. Braun, W. R. Salaneck and M. Fahlman, Adv. Mater., 2009, 21, 1450–1472 Search PubMed.
- M. Oehzelt, N. Koch and G. Heimel, Nat. Commun., 2014, 5, 4174 Search PubMed.
- G. Mercurio, R. J. Maurer, W. Liu, S. Hagen, F. Leyssner, P. Tegeder, J. M. A. Tkatchenko, S. Soubatch, K. Reuter and F. S. Tautz, Phys. Rev. B: Condens. Matter Mater. Phys., 2013, 88, 035421 Search PubMed.
- J. L. Bredas, J. E. Norton, J. Cornil and V. Coropceanu, Acc. Chem. Res., 2009, 42, 1691–1699 CrossRef PubMed.
- A. Köhler and H. Bässler, Electronic Processes in Organic Semiconductors, Wiley-VCH, Weinheim, Germany, 2015 Search PubMed.
- V. May and O. Kühn, Charge and Energy Transfer Dynamics in Molecular Systems, Wiley-VCH, Weinheim, Germany, 2011 Search PubMed.
- D. Beljonne, J. Cornil, L. Muccioli, C. Zannoni, J. L. Brédas and F. Castet, Chem. Mater., 2011, 23, 591–609 CrossRef.
- V. Ruhle, A. Lukyanov, F. May, M. Schrader, T. Vehoff, J. Kirkpatrick, B. Baumeier and D. Andrienko, J. Chem. Theory Comput., 2011, 7, 3335–3345 Search PubMed.
- G. D'Avino, L. Muccioli, F. Castet, C. Poelking, D. Andrienko, Z. G. Soos, J. Cornil and D. Beljonne, J. Phys.: Condens. Matter, 2016, 28, 433002 CrossRef PubMed.
- M. B. Casu, J. Electron Spectrosc. Relat. Phenom., 2015, 204, 39–48 CrossRef.
- R. Forker, M. Gruenewald and T. Fritz, Annu. Rep. Prog. Chem., Sect. C: Phys. Chem., 2012, 108, 34–68 RSC.
- H. Klauk, U. Zschieschang, J. Pflaum and M. Halik, Nature, 2007, 445, 745–748 Search PubMed.
- F. Würthner and M. Stolte, Chem. Commun., 2011, 47, 5109–5115 Search PubMed.
- U. H. F. Bunz, J. U. Engelhart, B. D. Lindner and M. Schaffroth, Angew. Chem., Int. Ed., 2013, 52, 3810–3821 CrossRef PubMed.
- Q. Miao, Adv. Mater., 2014, 26, 5541–5549 CrossRef PubMed.
- U. H. F. Bunz, Acc. Chem. Res., 2015, 48, 1676–1686 CrossRef.
- I. Michalsky, V. Gensch, C. Walla, M. Hoffmann, F. Rominger, T. Oeser, P. Tegeder, A. Dreuw and M. Kivala, Chem. – Eur. J., 2022, 28, e202200326 CrossRef.
- N. Hammer, T. A. Schaub, U. Meinhardt and M. Kivala, Chem. Rec., 2015, 15, 1119–1131 CrossRef PubMed.
- U. Meinhardt, F. Lodermeyer, T. A. Schaub, A. Kunzmann, P. O. Dral, A. C. Sale, F. Hampel, D. M. Guldi, R. D. Costa and M. Kivala, RSC Adv., 2016, 6, 67372–67377 RSC.
- M. Hirai, N. Tanaka, M. Sakai and S. Yamaguchi, Chem. Rev., 2019, 119, 8291–8331 CrossRef PubMed.
- T. A. Schaub, K. Padberg and M. Kivala, J. Phys. Org. Chem., 2020, 33, e4022 CrossRef.
- M. Krug, M. Wagner, T. A. Schaub, W. Zhang, C. M. Schüßlbauer, J. D. R. Ascherl, P. W. Münich, R. R. Schröder, F. Gröhn and P. O. Dral, et al., Angew. Chem., Int. Ed., 2020, 59, 16233–16240 CrossRef PubMed.
- M. Ajdari, R. Pappenberger, C. Walla, I. Michalsky, F. Maaß, M. Kivala, A. Dreuw and P. Tegeder, J. Phys. Chem. C, 2024, 128, 14399–14406 CrossRef.
- N. Deng and G. Zhang, Org. Lett., 2019, 21, 5248–5251 CrossRef.
- M. Ajdari, R. Pappenberger, C. Walla, M. Hoffmann, I. Michalsky, M. Kivala, A. Dreuw and P. Tegeder, J. Phys. Chem. C, 2023, 127, 542–549 CrossRef.
- X.-Y. Zhu, Surf. Sci. Rep., 2004, 56, 1–83 CrossRef.
- X.-Y. Zhu, J. Phys. Chem. B, 2004, 108, 8778–8793 CrossRef.
- C. Lindstrom and X.-Y. Zhu, Chem. Rev., 2006, 106, 4281–4300 CrossRef.
- M. Muntwiler and X.-Y. Zhu, Dynamics at Solid State Surfaces and Interfaces, Wiley-VCH, 2010 Search PubMed.
- C. Bronner, M. Schulze, S. Hagen and P. Tegeder, New J. Phys., 2012, 14, 043023 CrossRef.
- C. Bronner, G. Schulze, K. J. Franke, J. I. Pascual and P. Tegeder, J. Phys.: Condens. Matter, 2011, 23, 484005 CrossRef.
- N. Armbrust, J. Güdde, P. Jakob and U. Höfer, Phys. Rev. Lett., 2012, 108, 056801 CrossRef PubMed.
- C. Bronner, S. Stremlau, M. Gille, F. Brauße, A. Haase, S. Hecht and P. Tegeder, Angew. Chem., Int. Ed., 2013, 52, 4422–4425 CrossRef PubMed.
- C. Bronner, B. Priewisch, K. Rück-Braun and P. Tegeder, J. Phys. Chem. C, 2013, 117, 27031–27038 CrossRef.
- L. Bogner, Z. Yang, S. Baum, R. F. M. Corso, P. Bäuerle, K. J. Franke, J. I. Pascual and P. Tegeder, J. Phys. Chem. C, 2016, 120, 27268–27275 CrossRef.
- L. Bogner, Z. Yang, M. Corso, R. Fitzner, P. Bäuerle, K. J. Franke, J. I. Pascual and P. Tegeder, Phys. Chem. Chem. Phys., 2015, 17, 27118–27126 RSC.
- A. Stein, F. Maass and P. Tegeder, J. Phys. Chem. C, 2017, 121, 18075–18083 CrossRef CAS.
- D. Gerbert, F. Maass and P. Tegeder, J. Phys. Chem. C, 2017, 121, 15696–15701 CrossRef CAS.
- D. Gerbert and P. Tegeder, J. Phys. Chem. Lett., 2017, 8, 4685–4690 CrossRef CAS PubMed.
- W. Bronsch, T. Wagner, S. Baum, M. Wansleben, K. Zielke, E. Ghanbari, M. Györök, A. Navarro-Quezada, P. Zeppenfeld and M. Weinelt, et al., J. Phys. Chem. C, 2019, 123, 7931–7939 CrossRef CAS.
- A. Stein, D. Rolf, C. Lotze, C. Czekelius, K. J. Franke and P. Tegeder, J. Phys.: Condens. Matter, 2019, 31, 044002 CrossRef.
- M. Ajdari, A. Stein, M. Hoffmann, M. Müller, U. H. F. Bunz, A. Dreuw and P. Tegeder, J. Phys. Chem. C, 2020, 124, 7196–7204 CrossRef CAS.
- M. Ajdari, T. Schmitt, M. Hoffmann, F. Maass, H. Reiss, U. H. F. Bunz, A. Dreuw and P. Tegeder, J. Phys. Chem. C, 2020, 124, 13196–13205 CrossRef CAS.
- A. Stein, D. Rolf, C. Lotze, B. Günther, L. H. Gade, K. J. Franke and P. Tegeder, J. Phys. Chem. Lett., 2021, 12, 947–951 CrossRef CAS.
- A. Stein, D. Rolf, C. Lotze, S. Feldmann, D. Gerbert, B. Günther, A. Jeindl, J. J. Cartus, O. T. Hofmann and L. H. Gade, et al., J. Phys. Chem. C, 2021, 125, 19969–19979 CrossRef CAS.
- K. Stallberg, A. Namgalies, S. Chatterjee and U. Höfer, J. Phys. Chem. C, 2022, 126, 12728–12734 CrossRef CAS.
- J. Steidel, I. Michalsky, M. Ajdari, M. Kivala and P. Tegeder, Phys. Chem. Chem. Phys., 2024, 26, 16454–16458 RSC.
- P. Tegeder, J. Phys.: Condens. Matter, 2012, 24, 394001 CrossRef PubMed.
- T. J. Rockey, M. Yang and H.-L. Dai, J. Phys. Chem. B, 2006, 110, 19973–19978 CrossRef CAS PubMed.
- F. Maaß, Y. Jiang, W. Liu, A. Tkatchenko and P. Tegeder, J. Chem. Phys., 2018, 148, 214703 CrossRef.
- M. Schulze, C. Bronner and P. Tegeder, J. Phys.: Condens. Matter, 2014, 26, 355004 CrossRef PubMed.
- S. Hagen, F. Leyssner, D. Nandi, M. Wolf and P. Tegeder, Chem. Phys. Lett., 2007, 444, 85–90 CrossRef CAS.
- P. Tegeder, S. Hagen, F. Leyssner, M. Peters, S. Hecht, T. Klamroth, P. Saalfrank and M. Wolf, Appl. Phys. A: Mater. Sci. Process., 2007, 88, 465–472 CrossRef CAS.
- E. R. McNellis, C. Bronner, J. Meyer, M. Weinelt, P. Tegeder and K. Reuter, Phys. Chem. Chem. Phys., 2010, 12, 6404–6412 RSC.
- L. Óvári, Y. Luo, F. Leyssner, R. Haag, M. Wolf and P. Tegeder, J. Chem. Phys., 2010, 133, 044707 CrossRef.
- C. Bronner and P. Tegeder, New J. Phys., 2014, 16, 053004 CrossRef.
- C. Bronner, M. Schulze, S. Hagen and P. Tegeder, New J. Phys., 2012, 14, 043032 CrossRef.
- E. Varene, I. Martin and P. Tegeder, J. Phys. Chem. Lett., 2011, 2, 252–256 CrossRef CAS.
- C. Bronner, D. Gerbert, A. Broska and P. Tegeder, J. Phys. Chem. C, 2016, 120, 26168–26172 CrossRef CAS.
- M. Knupfer, Appl. Phys. A: Mater. Sci. Process., 2003, 77, 623–626 CrossRef CAS.
| This journal is © the Owner Societies 2026 |