Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Steady-state and time-resolved near-infrared photoluminescence of [M1Ag24(SR)18]n− (M = Ag, Au, Pt, Pd, Ni) nanoclusters
Usama Tash,
Weijie Ji,
Avirup Sardar ,
Yitong Wang
,
Yitong Wang and
Rongchao Jin
and
Rongchao Jin *
*
Department of Chemistry, Carnegie Mellon University, Pittsburgh, Pennsylvania 15213, USA. E-mail: rongchao@andrew.cmu.edu
First published on 20th February 2026
Abstract
Silver nanoclusters (NCs) of atomic precision are attractive in terms of optical properties, and among the reported NCs, the thiolate (SR)-protected [Ag25(SR)18]− serves as a model system for uncovering how the electronic structure dictates light emission. Here, we examine the effect of substituting the central Ag atom in [Ag25(SR)18]− with Au, Pt, Pd, or Ni (note: 2− charge for Pt, Pd and Ni doped NCs). Combining steady-state, time-resolved, and oxygen-dependent photoluminescence (PL) spectroscopic studies, we show that dopants introduce distinct electronic effects that affect optical absorption and emission, and that all cases exhibit predominant phosphorescence in the near-infrared region. The incorporation of isoelectronic gold greatly enhances radiative triplet emission, yielding an unusually high quantum yield, whereas Pt or Pd substitution favors nonradiative decay despite retaining the 8-electron closed–shell superatomic configuration by charge compensation. Finally, Ni doping produces a redshift in luminescence while the other dopants lead to a blueshift compared to that of Ag25. Taken together, these results reveal how single-atom substitution modulates both radiative and nonradiative pathways, establishing the doped MAg24 nanoclusters as a versatile platform for tuning near-infrared photophysics at the atomic level.
Introduction
Atomically precise metal nanoclusters (NCs) contain a specific number of atoms in the range of tens to hundreds of metal atoms per core and have received wide interest.1–11 A prototypical example is the 25-atom gold NC with a formula of [Au25(SR)18]− (where SR = thiolate), which possesses a well-defined icosahedral Au13 core and six dimeric staple-like motifs (–SR–Au–SR–Au–SR–), exhibiting a closed–shell superatom electronic configuration (1S21P6) with 8 delocalized valence electrons in the core.12 For its silver analogue, Joshi et al.13 later reported [Ag25(SR)18]− (abbreviated as Ag25), which was found to be isostructural with [Au25(SR)18]−, not only in the geometric structure, but also the superatomic electronic structure. This equivalence enables a direct comparison of the photophysical properties of Ag- and Au-based superatoms within an identical coordination environment.Building on the Ag25 NC, researchers have explored single-atom doping with various heterometals (M) to tune the properties, including both experimental14–21 and theoretical work.22–27 Experimentally, Bakr and co-workers14 achieved doping of Ag25 with one Au atom via galvanic exchange, yielding a [AuAg24(SR)18]− product. The crystal structure of this AuAg24 NC revealed an icosahedral M@Ag12 core analogous to that in the parent Ag25, with the Au atom occupying the central site of the icosahedron. Zheng and co-workers reported the Pd- and Pt-doped ones, i.e., [PdAg24(SR)18]2− and [PtAg24(SR)18]2−, and single-crystal X-ray diffraction (XRD) showed that both PdAg24 and PtAg24 preserve the same framework as [Au25(SR)18]−, i.e., consisting of an M-centered Ag12 icosahedral core capped by six Ag2(SR)3 staples.15 In these heteroatom-doped systems, the dopant's different valence necessitates a change in the NC charge: for example, doping [Ag25]− with a Pt converts the nanocluster to a 2− charge in order to maintain an 8-electron superatom configuration.15 Recently, Ni-doping of Ag25 has also been achieved via a metal-exchange strategy, producing a [NiAg24(SPhMe2)18]0 NC and its two-electron reduced derivative [NiAg24(SPhMe2)18]2−.16 In the neutral NiAg24, the Ni substitution gives a 6-electron open–shell superatom that undergoes Jahn–Teller distortion, whereas upon reduction to the 2− state it becomes a stable 8-electron closed–shell superatom with an undistorted, isotropic core (confirmed by single-crystal XRD).16 In the cases of Au, Pt and Pd doping, the groups of Wu, Zhu and Patra have reported the PL18,19 and ultrafast dynamics21 studies. Theoretically, accurate electronic structure analysis,22–25 absorption spectra,22 HOMO–LUMO gap,25 PL mechanism,26 and vibrational modes27 of MAg24(SR)18 with M = Ag, Au, Pt, and Pd have been tackled by several groups. All these studies have provided a fundamental understanding of how single-atom dopants alter the electronic structure of superatomic silver NCs.
Single-atom substitution provides a tunable platform to dissect the photoluminescence (PL) mechanisms. In addition to Au, Pt, Pd, and Ni, recent work by Lee and coworkers also succeeded in doping Ag25 with open d-shell Ru, Os, or Ir in the form of metal hydride (i.e., RuH2, OsH2, IrH, all being d10 with the incorporation of hydrides), and PL mechanistic studies revealed that dopants modulate both the radiative and nonradiative decay channels.28
Studying the PL of both undoped and doped metal NCs provides crucial insight into how atomic and electronic structures dictate light emission. The structure–emission correlations will establish a general design framework for tailoring optical properties across nanocluster systems. Although Ag25 and the doped MAg24 (M = Au, Pt, Pd, Ni) nanoclusters have been previously studied for their PL characteristics, those works primarily focused on qualitative emission behavior and steady-state measurements, with the lifetime information missing (note: recent work by Maity et al.21 probed the lifetimes of M = Ag, Au, Pt and Pd cases, but Ni was still missing). The lifetime information is critical for understanding the PL mechanism.29,30 Several key questions also remain unresolved—particularly regarding the contributions of fluorescence, phosphorescence, and thermally activated delayed fluorescence (TADF).28 Moreover, the quantitative relationship between radiative and nonradiative decay channels has not been firmly established, and the excited-state lifetimes of the MAg24 series have not been systematically measured or compared under identical conditions. The present study therefore revisits the Ag25 series with unified steady-state, time-resolved, and oxygen-dependent analyses, providing insights into the doping effects and PL mechanism.
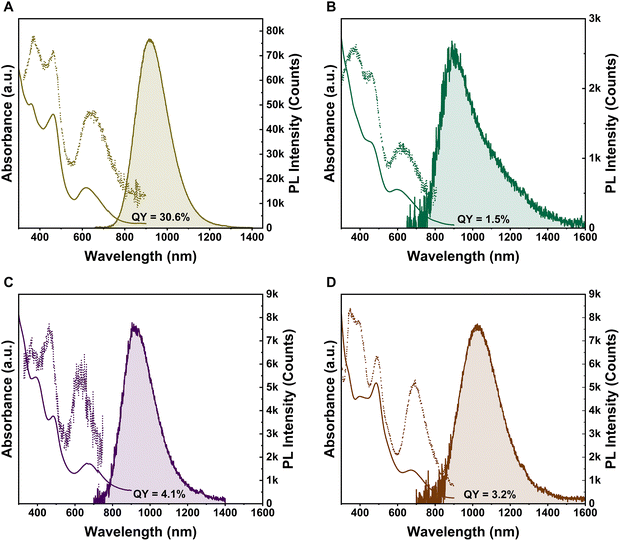
Here, we report a detailed investigation on [Ag25(SR)18]− and its doped analogues [MAg24(SR)18]n− (M = Au, Pt, Pd, Ni) by integrating steady-state with time-resolved and oxygen-dependent PL measurements. By correlating the valence electron count, HOMO–LUMO gap (Eg) modulation, and spin–orbit coupling with changes in PL quantum yield (QY), emission energy, and excited-state lifetimes, this work aims to establish a framework for dopant-controlled emission tuning in superatomic nanoclusters. The single-atom dopants drastically alter the QY from the highest 30.6% for AuAg24 to strong quenching in PtAg24 and confirm that the emission arises predominantly from long-lived triplet state, as evidenced by microsecond (µs) lifetimes and O2 quenching of the PL.
Results and discussion
The syntheses of [Ag25(SR)18]− and [MAg24(SR)18]n− (M = Au, Pt, Pd, Ni) followed the literature reports.13,16 To ensure the purity of samples, we first crystallized all NCs, and the re-dissolved crystals were used in the spectroscopic measurements. In addition, the PL of metal NCs is often quite broad (spanning the entire near-infrared (NIR) range, ∼700 to 1700 nm, depending on the size and composition), thus, we introduced a broadband InGaAs photomultiplier detector for both the stead-state and time-resolved measurements.29,30Doping effect on HOMO–LUMO gap and superatomic electron configuration
The homometal and doped NCs’ optical absorption spectra are shown in Fig. 1; the photon energy scale spectra are shown in Fig. S1. Same as the Au25 NC,12 homometallic Ag25 is a closed–shell superatom22–27 with 8 delocalized electrons (1S21P6) and possesses an Eg of ∼1.48 eV (the onset of absorbance, Fig. S1), which serves as the basis for comparison with the doped NCs. Replacing one Ag with Au, Pt, Pd, or Ni may alter the electron count and modifies the electronic distribution within the icosahedral core. Below we discuss the four doping cases and their effects. | ||
| Fig. 1 UV-vis absorption spectra of [Ag25(2,4-DMBT)18]− and [MAg24(2,4-DMBT)18]n− (M = Au, Pt, Pd, Ni) in CDCl3 at room temperature. The cluster charge (n−) is 1- for Au, 2- for Pt, Pd, and Ni. | ||
| Sample | Eg (eV) | Atom valence electron configuration | Central atom within the NC | Dopant's contribution to the NC's free valence electrons |
|---|---|---|---|---|
| [Ag25(SR)18]− | 1.48 | Ag, Z = 47 [Kr]4d105s1 | No change | 1 |
| [AuAg24(SR)18]− | 1.47 | Au, Z = 79 [Xe] 5d106s1 | No change | 1 |
| [PtAg24(SR)18]2− | 1.45 | Pt, Z = 78 [Xe] 5d96s1 | 5d106s0 | 0 |
| [PdAg24(SR)18]2− | 1.49 | Pd, Z = 46 [Kr] 4d105s0 | No change | 0 |
| [NiAg24(SR)18]2− | 1.41 | Ni, Z = 28 [Ar] 3d84s2 | 3d104s0 | 0 |
Taken together, the doping effects on the NC charge, Eg, and superatomic electron configuration are summarized in Table 1. When Au replaces the central Ag atom, it preserves the 8-electron closed shell, leaving Eg essentially unchanged. To maintain an 8-electron shell closure, Pt or Pd doped NCs adjust their charge accordingly and do not alter the Eg due to the preserved 8e− configuration, whereas Ni can cause the cluster to have either 6 or 8 electrons, depending on its oxidation state. This change in electron filling alters the frontier orbital occupancy and the Eg value, and excited state relaxation behavior for the Ni doped case.16,25 Of note, the Ni doping effect in Ag25 is similar to the case of Ni-doping in Ag29.31
Photoluminescence and quantum yields
Steady-state PL measurements (Fig. 2A) in He-degassed solutions reveal clear dopant-dependent effects. The NIR emission (peaks in 900–1020 nm) differs from earlier reports (peaks around 800 nm).18,19,21 Among the dopants, replacing the central Ag atom with Ni produces a similar peak wavelength (∼1024 nm) compared to Ag25 (peak ∼1017 nm), whereas Pt substitution yields the most blueshift (peak ∼894 nm); Au- and Pd-doped ones emit at intermediate wavelengths in the ∼918–920 nm range, Fig. 2B.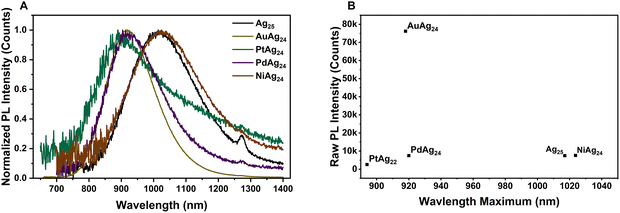 | ||
| Fig. 2 (A) Normalized PL spectra of [Ag25(2,4-DMBT)18]− and [MAg24(2,4-DMBT)18]n− (M = Au, Pt, Pd, Ni) NCs under He. (B) Emission intensities versus peak wavelengths for the dopant cases. | ||
Since the MAg24 series exhibits comparable optical gaps (Eg ≈ 1.4–1.5 eV), the PL peak shifts are best explained by dopant-dependent excited-state relaxation (Stokes shift) rather than differences in the absorption gap alone. Importantly, the Ni-doped cluster studied here is the reduced species [NiAg24(SPhMe2)18]2−, which has an 8e− closed–shell superatomic configuration (1S21P6). We therefore attribute the Ni red-shift to greater structural and vibrational relaxation after excitation: the 3d Ni dopant strengthens the coupling between the excited electrons and the cluster's vibrations, allowing more energy to be lost as vibrational motions before emission. As a result, emission occurs from a more relaxed (lower-energy) excited state, giving rise to a larger Stokes shift and thus lower-energy (more red-shifted) PL.
The most striking effect of doping pertains to the photoluminescence quantum yield (PLQY), Fig. 3. The PL spectral measurements were carried out in deaerated solutions (He purged) to remove the dissolved O2, which is a well-known quencher of triplet excited states. All samples were measured in deuterated chloroform (CDCl3) as the solvent to eliminate solvent absorption of PL. The undoped Ag25 exhibits a PLQY of ∼3.5%. Introducing a single Au atom into Ag25 significantly boosts the PLQY by an order of magnitude, whereas adding a Pt atom drastically quenches PL. Specifically, the PLQY of AuAg24 is ∼30.6%, similar to previous reports,30,32 an exceptionally high value for NCs at room temperature. Pd doping yields a modestly enhanced PLQY (4.1%), and Ni doping with ∼3.2%. The PtAg24 NC shows the lowest PLQY (only ∼1.5%). These results indicate that Au doping is uniquely effective at enhancing radiative efficiency (vide infra), whereas a Pt dopant is detrimental to the PL. The ∼30.6% QY of AuAg24 is one of the highest ever reported for NCs in solution at room temperature, cf., the prototypical [Au25(PET)18]− has a PLQY of ∼1%.30 The 30.6% yield also rivals or exceeds other reported values achieved by surface or aggregation engineering, e.g. ∼8% in aggregated Au22 clusters33 or ∼15% in certain ligand-exchanged Au25-rod derivatives,34 underscoring the remarkable impact of the single Au dopant. The AuAg24 appears to provide an ideal combination of electronic and structural factors that maximize radiative recombination (see more discussions below).
PL lifetimes and mechanistic insights
Time-resolved photoluminescence decay measurements in the nanosecond (ns) to microsecond (µs) timescales provide insight into the nature of the emissive states in the NCs.21,28–30 Fig. 4 displays the PL decay profiles of the doped NCs (recorded under He inert atmosphere). The PL decays can be well fitted (χ2 ≈ 1.08–1.16) using a bi-exponential function: I(t) = I0 + A1e(−t/τ1) +A2e(−t/τ2), where I0 is the time-zero intensity (or baseline), τ1 and τ2 represent the short- and long-lived lifetimes, and A1 and A2 are the amplitudes (%). The extracted lifetimes and their amplitudes are listed in Table 2. The bi-exponential decay behavior observed in the series indicates the presence of two distinct emissive components.| NC | PL λmax (nm) | PLQY (%) | τ1 (A1) ns | τ2 (A2) ns | Average τ* ns | kr (s−1) | knr (s−1) |
|---|---|---|---|---|---|---|---|
| Ag25 (ref. 30) | 1017 | 3.5 | — | 1100 (100%) | 1100 | 3.18 × 104 | 8.77 × 105 |
| AuAg24 | 918 | 30.6 | — | 1769 (100%) | 1769 | 1.73 × 105 | 3.92 × 105 |
| PtAg24 | 894 | 1.5 | 131 (42.3%) | 967 (57.7%) | 613 | 2.45 × 104 | 1.61 × 106 |
| PdAg24 | 920 | 4.1 | 155 (5.5%) | 1663 (94.5%) | 1580 | 2.59 × 104 | 6.07 × 105 |
| NiAg24 | 1024 | 3.2 | 215 (6.3%) | 1843 (93.7%) | 1740 | 1.84 × 104 | 5.56 × 105 |
Under helium at ambient temperature for all doped NCs, AuAg24 shows a single-exponential decay with a lifetime of 1.769 µs (Fig. 4A), indicative of triplet-state T1 emission (i.e. phosphorescence). The measured lifetime matches the value reported by Maity et al. previously.21,35 PtAg24 (Fig. 4B) displays mixed character, with a TADF channel (τ1 = 130.7 ns, 42.32%) alongside triplet emission (τ2 = 967.6 ns, 57.68%). Its triplet state involvement is further substantiated by O2-induced PL quenching and concomitant 1O2 generation (vide infra). In addition to the biexponential lifetime behavior, spectral deconvolution of the PtAg24 emission (Fig. 5) reveals two distinct emissive components, consistent with the overlapped singlet (S1) and triplet (T1) contributions. Therefore, both TADF (shorter wavelength due to higher energy, centered at 894 nm) and phosphorescence (longer wavelength, centered at 1075 nm), with a S1–T1 peak splitting of 0.23 eV (estimated by the peak energy difference). PdAg24 (Fig. 4C) is triplet-dominated (1.663 µs, 94.51%) with a minor TADF emission (155.3 ns, 5.49%). NiAg24 (Fig. 4D) also exhibits mainly triplet-derived emission (1.843 µs, 93.70%) with a small TADF (215.5 ns, 6.30%). Overall, Au, Pd, and Ni doped MAg24 NCs all feature triplet-derived emission, whereas Pt retains a large (roughly half) contribution from ∼102 ns delayed fluorescence. For the Ag25 and Au/Pd/Ni doped four NCs, their PL peaks are reasonably symmetric, indicating a much smaller S1–T1 splitting (estimated ∼0.1 eV or so).
 | ||
| Fig. 5 Spectral deconvolution of PtAg24 emission into singlet (blue profile) and triplet (red) contributions. | ||
For PtAg24, spectral deconvolution suggests an S1–T1 peak splitting ΔEST ≈ 0.23 eV. This value is larger than that of highly efficient TADF emitters, therefore, the delayed-fluorescence contribution in PtAg24 is expected to be modest; but other factors may promote revserse intersystem crossing (RISC), such as spin–orbit coupling and reorganization energy, which are unknown yet and require future theoretical evaluation. In addition, because TADF originates from thermally assisted reverse intersystem crossing (T1 → S1), the most direct validation of TADF is its temperature dependence, where the delayed fluorescence increases with temperature but is suppressed at low temperatures. In the present work, the room-temperature biexponential decay and spectral deconvolution support a delayed-fluorescence component overlapping with triplet emission, but definitive evidence for TADF will require temperature-dependent PL measurements, which remain to be tested.
The quantum yield enhancement and quenching observed above are further analyzed by calculating the radiative (kr) and nonradiative (knr) decay rate constants using eqn (1)–(3) below.
| Φ = τ*kr | (1) |
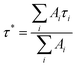 | (2) |
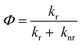 | (3) |
In previous work by Lee and coworker, the radiative rate was correlated with the dopant's electron affinity (and associated superatomic orbital energy shifts) or spin–orbit coupling, while nonradiative losses were found to be governed by how the core structure and vibrations are altered upon doping.28 Herein we performed a detailed analysis of kr and knr, and found that AuAg24 uniquely combines a higher kr and lower knr (kr = 1.73 × 105 s−1, knr = 3.92 × 105 s−1) than the homometallic Ag25 (Table 2) yielding the highest QY (30.6%). The lower knr can be attributed to the rigidifying effect of Au substitution into the icosahedral core, which suppresses electron–phonon coupling.30 As shown in Fig. 6, the average Ag–Ag bond lengths in AuAg24 and Ag25 are nearly identical; however, Ag25 exhibits more pronounced fluctuations in its radial bond-length distribution, indicating greater structural flexibility. In contrast, Au incorporation slightly compresses the core and markedly reduces bond-length dispersion, yielding a more rigid framework. This enhanced rigidity in AuAg24 minimizes vibrational relaxation pathways, thereby lowering nonradiative decay and increasing overall photoluminescence efficiency. Compared to Ag25, PtAg24 exhibits a similar kr but a markedly elevated knr (1.61 × 106 s−1), which explains its lower Φ (1.5%), thus, Pt doping quenches PL, with the nonradiative channels arising from enhanced vibronic coupling. PdAg24 shows a modest improvement in Φ (4.1%) driven primarily by a reduction in knr (6.07 × 105 s−1) with only a minor change in kr. NiAg24 shows both reduced rates (kr = 1.84 × 104 s−1, knr = 5.56 × 105 s−1); such a counteracting effect yields a Φ similar to that of Ag25.
With respect to the dopants (Pt, Pd, Ni), both Pt and Pd cases show a slight contraction in the average M-Ag12 radial bond length (2.752 Å (Pt) and 2.747 Å (Pd) vs. 2.764 Å for homosilver), and the Ni case has the most compact kernel (average radial bond length: 2.706 Å) due to its smallest atomic radius. Compared to the slight increase in the PL for Pd doping and slight decrease for Ni doping, the observed marked PL quenching in Pt dopant may be explained by counteracting from the stronger d-s electronic interactions in the Pt case, resulting in stronger nonradiative decay. In comparison, both Au and Ag have d10 configuration and their d-shell is quite inert, thus, the AuAg24 and Ag25 cases have higher PL than the Pt doping case.
Effect of O2 on the PL
In the MAg24 series, molecular oxygen (3O2 for its ground state) efficiently quenches the PL of NCs due to photosensitization-induced generation of excited-state oxygen, e.g., singlet oxygen (1O2), thus, it can be used to confirm the triplet-state nature of the emission (phosphorescence).36 For AuAg24, the QY drops from 30.6% (under He) to 22.9% (under O2)—a ∼25% decrease (Fig. 7A)—and the amplitude-weighted lifetime shortens from 1.769 to 1.598 µs (Fig. 8A), consistent with the efficient intersystem crossing (ISC) and triplet emission.36,37 PtAg24 exhibits a PL quenching from 1.5% to 1.1% (∼27% variation; Fig. 7B), accompanied by a pronounced lifetime (τ2) shortening from 0.968 to 0.509 µs (Fig. 8B), evidencing triplet states of sufficient lifetime for oxygen sensitization. PdAg24 shows an even stronger response to O2: QY decreases from 4.1% to 1.5% (∼63% variation; Fig. 7C) and τ2 drops from 1.66 to 1.14 µs (Fig. 8C), indicating extensive triplet state energy transfer to O2. NiAg24 also shows strong QY quenching, 3.2% to 1.1% (∼66% variation, Fig. 7D), while its τ2 changes only modestly, Fig. 8D. The coordinated intensity drop and lifetime shortening under O2 substantiate the triplet-state character of the emission for the doped NC series. In addition, all cases show a small emission peak at 1275 nm, which is characteristic of the emission of 1O2 produced (Fig. 7). Overall, the O2 test indicates the involvement of triplet state in the PL process of all the NCs. | ||
| Fig. 7 Oxygen sensitization by AuAg24 (A), PtAg24 (B), PdAg24 (C), and NiAg24 (D) showing PL quenching upon O2 purging compared to He-purged conditions. | ||
 | ||
| Fig. 8 Time-resolved PL decay curves of AuAg24 (A), PtAg24 (B), PdAg24 (C), and NiAg24 (D). All solutions were bubbled with O2. | ||
In purely dynamic (collisional) quenching, O2 acts after excitation by introducing an additional nonradiative deactivation pathway, so the PL intensity and excited-state lifetime typically drop. In the Ni doping case, the intensity decreases while the lifetime remains unchanged, indicating a different mechanism. We rationalize that the highly reactive [NiAg24(SR)18]2− NCs were partially oxidized to [NiAg24(SR)18]0 (Eg ∼ 0.76 eV or 1630 nm wavelength, see ref. 16), and if one considers a Stokes shift, its emission (if any) would be far beyond 1700 nm (our detector's limit). For such a small gap, we predict the oxidized NCs would not be emissive; rather, it would show predominantly nonradiative decay. Since the amount of O2 in the solution is limited, not all [NiAg24(SR)18]2− were oxidized, thus, the survived portion (2− charge state) still showed the same lifetime. Overall, this abnormal case should be related to the high reactivity of [NiAg24(SR)18]2− compared to the other cases that are much less reactive with O2 due to noble metal doping.
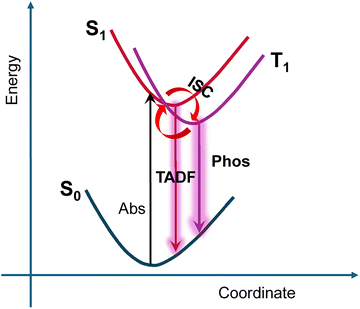
PL mechanism
Based on the above results and analysis, we summarize the PL mechanism in Scheme 1. Following photoexcitation, the exciton first relaxes to the lowest excited state (S1), from which ISC to the T1 state occurs, and accordingly the T1 state gets populated. We estimate the direct S1 emission prior to ISC is minor since no PL lifetime within the range of a few nanoseconds was detected, unlike the case of Au42(SR)32 quantum rods in which strong fluorescence (lifetime < 1 ns) was observed.38 The range of tens of ns to one to two hundreds of ns is attributed to the TADF component,30 whereas the microsecond component typically arises from phosphorescence. With this mechanism, all the five NCs emit predominant phosphorescence, with less from TADF. Except the case of PtAg24, the S1 and T1 energies for other cases are very close since no split peaks are found in their PL spectra, unlike the case of Au42(SR)32 quantum rod in which the fluorescence and the phosphorescence are distinctly split into two peaks.38–40Compared to previous reports,14–21 our current work has contributed to the following aspects: (i) the entire Ag25/MAg24 (Au, Pt, Pd, Ni) series is characterized by unified steady-state and time-resolved PL, as well as oxygen triplet sensitization under the same experimental conditions; (ii) systematic extraction and comparison of kr and knr across all dopants by consistent analysis of lifetime/QY; and (iii) inclusion of NiAg24 in the same mechanistic framework, enabling direct comparison with Au/Pt/Pd in terms of electron counts, emission shifts, and quenching behavior. Together, these efforts provide a quantitative dopant-dependent mapping of radiative vs. nonradiative pathways that was not previously established as a complete, consistent dataset in earlier reports.
Conclusions
In summary, doping a single heteroatom into the [Ag25(SR)18]− nanocluster provides a powerful means of tuning its electronic and photophysical properties. Gold substitution preserves the closed–shell electron count (8e−) and rigidifies the core, thus, giving rise to a high PLQY of 30.6% through significantly enhanced triplet emission. Doping by Pt or Pd maintains the 8-electron configuration through charge compensation but yields contrasting outcomes: Pd gives a small PL enhancement owing to reduced nonradiative loss, while Pt strongly quenches emission due to dominant nonradiative pathways despite strong spin–orbit coupling. Ni uniquely toggles between a distorted 6e− open–shell and a symmetric 8e− closed–shell state, with NIR emission peak position and intensity of the 8e− state being similar to those of Ag25. Overall, dopant identity dictates the balance between electron-count, orbital hybridization, and radiative vs. nonradiative decay, enabling a systematic control of emission energy and quantum yield in silver superatoms. The obtained mechanistic insights will benefit the applications of NCs in catalysis, optics, electronics, and sensing.These dopant-dependent mechanisms enable rational tuning of NIR emission brightness and lifetime via control of kr/knr and triplet-state involvement. Since NIR emission is deep tissue-penetrating and minimally photodamaging, the Ag25/MAg24 family is promising for biosensing. In addition, such doped NCs are also attractive in energy catalysis,41,42 photon upconversion,43,44 solar cells,45 nonlinear optics46 and other applications. Future work will lead to exquisite tailoring for functional NCs with high performance for broader applications.
Conflicts of interest
The authors declare no conflicts of interest.Data availability
The authors confirm that all the data supporting the findings of this study are available within the article and its supplementary information (SI). Supplementary information: experimental section (details of the synthesis and characterization including UV-vis-NIR absorption, steady-state and time-resolved photoluminescence, O2 tests, and quantum-yield determination), and SI Fig. S1. See DOI: https://doi.org/10.1039/d5cp04426j.Acknowledgements
R. J. acknowledges NSF funding (DMR 2419539), and U. T. thanks an Egyptian government fellowship.References
- Y. Li, M. Zhou, Y. Song, T. Higaki, H. Wang and R. Jin, Nature, 2021, 594, 380–384 Search PubMed.
- S.-F. Yuan, C.-Q. Xu, W.-D. Liu, J.-X. Zhang, J. Li and Q.-M. Wang, J. Am. Chem. Soc., 2021, 143, 12261–12267 CrossRef CAS PubMed.
- Z. R. Yuan, Z. Wang, B. L. Han, C. K. Zhang, S. S. Zhang, Z. Y. Zhu, J. H. Yu, T. D. Li, Y. Z. Li, C. H. Tung and D. Sun, Angew. Chem., Int. Ed., 2022, 61, e202211628 CrossRef CAS PubMed.
- Y. Wang, C. Zhang, T. Xu, Y. Li, H. Xie, D. Sun, N. Feng, S. Li and X. Xin, ACS Appl. Mater. Interfaces, 2022, 17, 17307–17315 CrossRef.
- Y. Zhang, J. Zhang, Z. Li, Z. Qin, S. Sharma and G. Li, Commun. Chem., 2023, 6, 24 CrossRef CAS PubMed.
- Y. Liu, Z. Li, X.-H. Liu, N. Pinna and Y. Wang, Nanoscale Horiz., 2023, 8, 1435–1439 Search PubMed.
- L. Zhan, J. Zhang, B. Ning, Y. He, G. Xiao, Z. Chen and F.-X. Xiao, Inorg. Chem. Front., 2024, 11, 7991–8002 Search PubMed.
- L. Huang, R. Lu, W. Zhang, Y. Fan, Y. Du, K. Ni, Y. Zhu and M. Zhu, Angew. Chem., Int. Ed., 2024, 63, e202412964 CrossRef CAS PubMed.
- H. Seong, M. Choi, S. Park, H.-W. Kim, J. Kim, W. Kim, J. S. Yoo and D. Lee, ACS Energy Lett., 2022, 7, 4177–4184 CrossRef CAS.
- Z. Zhang, R. Yin, Z. Song, M. Zhang, B. Zhang, S. Lu, Q. Yao, D.-E. Jiang, J. Xie and W. Hu, Angew. Chem., Int. Ed., 2025, 64, e202500389 CrossRef CAS PubMed.
- X. Wei, C. Xu, H. Li, X. Kang and M. Zhu, Chem. Sci., 2022, 13, 5531–5538 RSC.
- M. Zhu, C. M. Aikens, M. P. Hendrich, R. Gupta, H. Qian, G. C. Schatz and R. Jin, J. Am. Chem. Soc., 2009, 131, 2490–2492 CrossRef CAS PubMed.
- C. P. Joshi, M. S. Bootharaju, M. J. Alhilaly and O. M. Bakr, J. Am. Chem. Soc., 2015, 137, 11578–11581 Search PubMed.
- M. S. Bootharaju, C. P. Joshi, M. R. Parida, O. F. Mohammed and O. M. Bakr, Angew. Chem., 2016, 128, 934–938 CrossRef.
- J. Yan, H. Su, H. Yang, S. Malola, S. Lin, H. Häkkinen and N. Zheng, J. Am. Chem. Soc., 2015, 137, 11880–11883 CrossRef CAS PubMed.
- M. Kim, K. L. D. M. Weerawardene, W. Choi, S. M. Han, J. Paik, Y. Kim, M.-G. Choi, C. M. Aikens and D. Lee, Chem. Mater., 2020, 32, 10216–10226 CrossRef CAS.
- M. Neumaier, A. Baksi, P. Weis, E. K. Schneider, P. Chakraborty, H. Hahn, T. Pradeep and M. M. Kappes, J. Am. Chem. Soc., 2021, 143, 6969–6980 CrossRef CAS PubMed.
- Q. Yuan, X. Kang, D. Hu, C. Qin, S. Wang and M. Zhu, Dalton Trans., 2019, 48, 13190–13196 RSC.
- X. Liu, J. Yuan, C. Yao, J. Chen, L. Li, X. Bao, J. Yang and Z. Wu, J. Phys. Chem. C, 2017, 121, 13848–13853 CrossRef CAS.
- H. Ji, M. Chen, Y. Sun, W. Liu, M. Jiang, Y. Liu, R. Qin, J. Liu, Z. Hao, F. Zhang, X. Li, L. Zhang and R. Zhang, Sens. Actuators, B, 2025, 439, 137840 CrossRef CAS.
- S. Maity, S. Kolay, S. Ghosh, S. Chakraborty, D. Bain and A. Patra, J. Phys. Chem. Lett., 2022, 13, 5581–5588 CrossRef CAS PubMed.
- P. D’Antoni, L. Sementa, S. Bonacchi, M. Reato, F. Maran, A. Fortunelli and M. Stener, Phys. Chem. Chem. Phys., 2024, 26, 17569–17576 RSC.
- F. Alkan, P. Pandeya and C. M. Aikens, J. Phys. Chem. C, 2019, 123, 9516–9527 Search PubMed.
- H. Liu, B. Huang, Y. Shao and Y. Pei, Small, 2024, 20, e2403520 Search PubMed.
- E. Wang and Y. Gao, J. Cluster Sci., 2025, 36, 115 CrossRef CAS.
- K. L. Dimuthu, M. Weerawardene and C. M. Aikens, J. Phys. Chem. C, 2018, 122, 2440–2447 CrossRef.
- A. Tlahuice-Flores, Mol. Simul., 2013, 39, 428–431 CrossRef CAS.
- H. Yi, S. Song, S. M. Han, J. Lee, W. Kim, E. Sim and D. Lee, Angew. Chem., Int. Ed., 2023, 62, e202302591 CrossRef CAS PubMed.
- Y. Wang, Z. Liu, A. Mazumder, C. G. Gianopoulos, K. Kirschbaum, L. A. Peteanu and R. Jin, J. Am. Chem. Soc., 2023, 145, 26328–26338 CrossRef CAS.
- Z. Liu, M. Zhou, L. Luo, Y. Wang, E. Kahng and R. Jin, J. Am. Chem. Soc., 2023, 145, 19969–19981 CrossRef CAS PubMed.
- A. A. A. Ahmed, S. Havenridge, K. Sahoo, L. Thapa, A. Baksi, G. H. Clever, H. Noei, M. Kohantorabi, A. Stierle, C. R. Raj, W. J. Parak, C. M. Aikens and I. Chakraborty, Small, 2024, 21, 2408096 CrossRef PubMed.
- A. Pniakowska, K. K. Ramankutty, P. Obstarczyk, M. Perić Bakulić, Ž. Sanader Maršić, V. Bonačić-Koutecký, T. Bürgi and J. Olesiak-Bańska, Angew. Chem., Int. Ed., 2022, 61, e202209645 CrossRef CAS PubMed.
- Y. Yu, Z. Luo, D. M. Chevrier, D. T. Leong, P. Zhang, D.-E. Jiang and J. Xie, J. Am. Chem. Soc., 2014, 136, 1246–1249 CrossRef CAS.
- P. A. Lummis, K. M. Osten, T. I. Levchenko, M. Sabooni, A. Hazer, S. Malola, B. Owens-Baird, A. J. Veinot, E. L. Albright, G. Schatte, S. Takano, K. Kovnir, K. G. Stamplecoskie, T. Tsukuda, H. Häkkinen, M. Nambo and C. M. Crudden, JACS Au, 2022, 2, 875–885 CrossRef CAS PubMed.
- S. Maity, D. Bain and A. Patra, Nanoscale, 2019, 11, 22685–22723 RSC.
- M. Mitsui, D. Arima, A. Uchida, K. Yoshida, Y. Arai, K. Kawasaki and Y. Niihori, J. Phys. Chem. Lett., 2022, 13, 9272–9278 CrossRef CAS PubMed.
- S. Takano, H. Hirai, T. Nakashima, T. Iwasa, T. Taketsugu and T. Tsukuda, J. Am. Chem. Soc., 2021, 143, 10560–10564 CrossRef CAS PubMed.
- L. Luo, Z. Liu, X. Du and R. Jin, J. Am. Chem. Soc., 2022, 144, 19243–19247 CrossRef CAS PubMed.
- L. Luo, Z. Liu, A. Mazumder and R. Jin, J. Am. Chem. Soc., 2024, 146, 27993–27997 CAS.
- W. Fan, Y. Yang, Q. You, J. Li, H. Deng, N. Yan and Z. Wu, J. Phys. Chem. C, 2023, 127, 816–823 CrossRef CAS.
- K. Kwak and D. Lee, Acc. Chem. Res., 2019, 52, 12–22 CrossRef CAS PubMed.
- S. Li, A. V. Nagarajan, S. Zhao, G. Mpourmpakis and R. Jin, J. Phys. Chem. C, 2021, 125, 24831–24836 CrossRef CAS.
- Y. Niihori, Y. Wada and M. Mitsui, Angew. Chem., Int. Ed., 2021, 60, 2822–2827 Search PubMed.
- Z. Liu, X. Hu, L. Luo, G. He, A. Mazumder, E. Gunay, Y. Wang, E. C. Dickey, L. A. Peteanu, K. Matyjaszewski and R. Jin, J. Am. Chem. Soc., 2025, 147, 28241–28250 CrossRef CAS PubMed.
- M. S. Kim, M. A. Abbas and J. H. Bang, Bull. Korean Chem. Soc., 2016, 37, 791–792 CrossRef CAS.
- R. Antoine and V. Bonacic-Koutecky, Liganded Silver and Gold Quantum Clusters. Towards a New Class of Nonlinear Optical Nanomaterials. Springer, 2018 Search PubMed.
| This journal is © the Owner Societies 2026 |