Open Access Article
Open Access ArticleDopant distributions and band-edge positions in Sr-doped NaTaO3: a first-principles study
Ryusei
Morimoto
 a,
Hiroki
Uratani
a,
Hiroki
Uratani
 *ab,
Hiroshi
Onishi
*ab,
Hiroshi
Onishi
 cde and
Hirofumi
Sato
cde and
Hirofumi
Sato
 af
af
aDepartment of Molecular Engineering, Graduate School of Engineering, Kyoto University, Nishikyo-ku, Kyoto, 615-8510, Japan. E-mail: uratani@moleng.kyoto-u.ac.jp
bPRESTO, Japan Science and Technology Agency (JST), Kawaguchi, Saitama, 332-0012, Japan
cDepartment of Chemistry, Graduate School of Science, Kobe University, Nada-ku, Kobe, 657-8501, Japan
dResearch Center for Membrane and Film Technology, Kobe University, Nada-ku, Kobe, 657-8501, Japan
eDivision of Advanced Molecular Science, Institute for Molecular Science, Okazaki, 444-8585, Japan
fFukui Institute for Fundamental Chemistry, Kyoto University, Sakyo-ku, Kyoto, 606-8103, Japan
First published on 19th January 2026
Abstract
Sr doping is known to enhance the water-splitting activity of NaTaO3 photocatalysts and is often accompanied by surface segregation of Sr, yet, the electronic-structural origin remains unclear. Here, we employ first-principles calculations with explicit enumeration of all Sr configurations in bulk NaTaO3 (NTO) and TaO2-terminated (001) surfaces. In the bulk, the enumeration shows that shorter Sr–Sr separations are energetically preferred. At surfaces, Sr segregates and accumulates strongly. Layer-resolved local density of states indicates that in-gap states are confined to the outermost TaO2 layer, while interior layers retain a near-bulk gap. In Sr-rich surface layers, both band edges shift upward relative to the interior. Population analyses link the valence-band rise to O–O contraction and the conduction-band rise to Ta–Ta elongation. Sr-doping and segregation yields near-surface band bending and a built-in electric field that can promote electron–hole separation and suppress recombination, rationalising the observed activity gains.
Introduction
The breakthrough TiO2 photoelectrolysis reported by Honda and Fujishima initiated systematic studies of photocatalysis.1–4 Under above-gap excitation, nonequilibrium electrons and holes are generated, and their behaviour is governed by the electronic structure. In particulate systems, these excited carriers are guided to catalytic sites by several factors such as internal electric fields and diffusion.5,6 Thus, achieving high efficiency requires an appropriate balance of these mechanisms rather than optical absorption alone.For overall water splitting, metal-oxide photocatalysts, including SrTiO3,7,8 KTaO3,9,10 and NaTaO3,9,10 are often selected because they satisfy both thermodynamic alignment and chemical stability. In these oxides, aliovalent doping with Sr2+,11,12 La3+,13,14 and Al3+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15,16 has also been employed to further enhance the reaction rate. Although such dopants can modify carrier densities, band-edge positions, and related properties, it is challenging to isolate the dominant mechanism in a given composition because several effects change simultaneously. Moreover, substitutional doping is configurational. Even at fixed compositions, many symmetry-inequivalent dopant arrangements and associated relaxations exist; therefore, assessing dopant effects requires explicit configuration enumeration.17,18
15,16 has also been employed to further enhance the reaction rate. Although such dopants can modify carrier densities, band-edge positions, and related properties, it is challenging to isolate the dominant mechanism in a given composition because several effects change simultaneously. Moreover, substitutional doping is configurational. Even at fixed compositions, many symmetry-inequivalent dopant arrangements and associated relaxations exist; therefore, assessing dopant effects requires explicit configuration enumeration.17,18
In the present study, we focus on the role of dopants in NaTaO3 (NTO), a long-studied ultraviolet (UV)-active perovskite. Among reported dopants for NTO, Sr offers a useful probe of mechanism: substantial rate enhancements have been reported despite only modest shifts in the optical onset without invoking oxygen vacancies.19,20 Notably, at low Sr concentrations, Sr has been observed to segregate toward the surface and to form Sr-concentration gradients.21 However, it remains unclear whether the enhanced activity derives primarily from band-edge realignment, mid-gap states, or modified interfacial fields.
Here, we use first-principles calculations on bulk NTO and TaO2-terminated surfaces to isolate the effects of Sr substitution. Configuration enumeration and energetic screening are performed for all possible Sr configurations in given compositions. Short Sr–Sr separations are energetically favoured, implying a thermodynamic drive toward dopant accumulation. When Sr enriches near the surface or forms Sr-rich layers in the bulk, two coupled responses emerge: (i) acceptor-like in-gap states localised on O atoms adjacent to Sr-perturbed TaO6 octahedra and (ii) upward shifts of both band edges. The resulting near-surface band bending establishes a built-in electric field that is expected to promote electron–hole separation and suppress recombination, rationalising activity gains that cannot be explained by band-gap changes alone. These insights suggest a design principle for doped perovskite photocatalysts: controlling dopant distributions and the attendant lattice distortions to engineer band-edge positions and interfacial fields.
Methods
The structural models were constructed as follows. An orthorhombic (Pnma) unit cell of NTO was optimised. Based on this cell, we constructed four bulk supercells, 1 × 1 × 3, 1 × 1 × 5, 2 × 2 × 2, and 2 × 2 × 5, as shown in Fig. 1, hereafter denoted as B1, B2, B3, and B4, respectively. Sr substitution was introduced using a charge-neutral 3Na1Ta scheme, in which three Na sites and one Ta site in a given supercell were replaced by Sr, consistent with NaTaO3–Sr(Sr1/3Ta2/3)O3 stoichiometry. In each size of supercells except B4, all possible Sr (3Na1Ta) configurations are considered (details for construction methods are shown in the SI). For B3, we also examined four additional patterns: B3-1Ta (a Ta site substituted by Sr), B3-1Na (a Na site substituted by Sr), B3-1Ta-c, and B3-1Na-c. In the “-c” models, the total number of valence electrons was adjusted to compensate the nominal charge imbalance caused by Sr substitution at only Na or Ta sites. To model the surfaces, we constructed both symmetric and asymmetric slabs. The symmetric slab S1 consists of a 1 × 1 × 10 supercell with TaO2-terminated (001) surfaces on both sides and a 15 Å vacuum region. Within this geometry, we considered S1-2Ta (two Ta sites substituted by Sr) and S1-4Na2Ta (four Na sites and two Ta sites substituted by Sr to maintain charge neutrality). The asymmetric slab S2 consists of a 1 × 1 × 5 supercell with a TaO2-terminated surface on one side and a NaO-terminated surface on the other, separated by a 15 Å vacuum layer. | ||
| Fig. 1 Representative 3Na1Ta configurations selected from the enumerated set for Sr-doped NaTaO3 bulk supercells. Sr, green; Na, yellow; Ta, blue; O, red. | ||
All first-principles calculations used the projector augmented-wave (PAW) method as implemented in the Vienna ab initio simulation package.22 The plane-wave cutoff was set to 400 eV. Unless otherwise noted, exchange–correlation effects were described by Perdew–Burke–Ernzerhof (PBE) generalised-gradient approximation.23 To assess the impact of nonlocal exchange on the electronic structure, for some cases, we also performed calculations using the hybrid functional HSE06.24 Spin-polarized calculations were tested but the systems converged to a negligible magnetic moment and showed no meaningful changes in the relevant electronic properties (see Table S1). Therefore, in what follows, the non-spin-polarized calculation results are reported. Brillouin-zone sampling was conducted employing Monkhorst–Pack meshes of 5 × 5 × 2, 3 × 3 × 1, 2 × 2 × 2, and 2 × 2 × 1 for B1, B2, B3, and B4, respectively; for slab models, a 3 × 3 × 1 mesh was employed. Dispersion interactions were included for the slab models, using Grimme's D3 correction to PBE (PBE-D3).25 The convergence criterion for electronic self-consistent field calculations was set to 10−7 eV. All structures were relaxed until residual forces were under 0.01 eV Å−1, unless otherwise noted. For NTO unit cell and bulk supercell models, no geometric parameters were fixed. For symmetric slabs, the central TaO2 layer was fixed to emulate a bulk-like interior. For asymmetric slabs, the bottom NaO layer was fixed, and a dipole correction along the surface normal was applied. The projected crystal orbital Hamilton population (pCOHP) was employed as implemented in the LOBSTER package.26,27 Density of states and band structures were processed with PyProcar.28 For doped supercells, band structures were analysed using band-structure unfolding to obtain the effective band dispersion in the primitive cell Brillouin zone, following established procedures.29,30 The unfolding was performed with PyProcar. Crystal structures and charge densities were visualised using VESTA.31
Results and discussion
Bulk
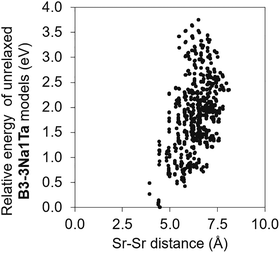
To identify low-energy Sr configurations in bulk NTO, we generated all possible Sr configurations and evaluated single-point energies on unrelaxed structures. We considered 110 configurations for B1-3Na1Ta, 570 for B2-3Na1Ta, and 792 for B3-3Na1Ta. Fig. 2 shows the relative energies of unrelaxed B3-3Na1Ta configurations as a function of Sr–Sr distances (results for B1-3Na1Ta and B2-3Na1Ta are shown in Fig. S1 and S2). In B3-3Na1Ta, the energy correlates with the Sr–Sr distance, although the shortest separation was not the global minimum and lies 0.3 eV above it; the lowest energy configuration is 2.3 eV below the most separated case. For B1-3Na1Ta and B2-3Na1Ta, the configurations with the shortest Sr–Sr separation are the most stable, lying 3.8 eV and 4.2 eV below the most separated cases, respectively. Overall, shorter Sr–Sr separations are energetically favoured.From the enumeration, we selected the lowest energy 3Na1Ta configuration for each supercell. We also built B4-3Na1Ta by adopting the minimum-energy arrangement identified in B3-3Na1Ta. These structures were then fully relaxed and their atomic coordinates are provided in the SI. Fig. 3 summarises the Sr content and the optimised lattice volume for the 3Na1Ta models (corresponding lattice parameters are shown in Table S2). All models showed an approximately isotopic expansion with increasing Sr content. For pristine NTO, the calculated lattice volume agrees with experiment within ∼0.7%. At 36.4 mol% of Sr (B1-3Na1Ta), the cell volume increased by 4.8% relative to pristine NTO.
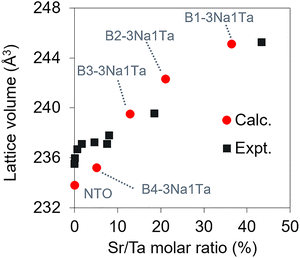 | ||
| Fig. 3 Lattice volume of Sr-doped NaTaO3 as a function of the nominal Sr/Ta molar ratio. Red circles: DFT-optimised structures (this work). Black squares: experimental values in ref. 20. | ||
Linear dependence between the Sr content and the lattice volume in NaTaO3–Sr(Sr1/3Ta2/3)O3 solid solutions has been reported previously.20 In that work, the lattice volume increased by ∼20% from NTO to 200 mol% of Sr. This trend suggests a ∼2.0% increase at 21.1 mol% Sr (B2-3Na1Ta), which is qualitatively consistent with our computations.
We also examined how the Sr substitution site affects the lattice parameters of B3-1Ta and B3-1Na (the details are summarised in Table S3). Replacing Ta by Sr expands the lattice more than replacing Na by Sr. Charge compensation had only a minor effect on the lattice parameters (<0.9% in volume).
The lattice expansion may be driven by the difference in ionic radii between the substituting Sr2+ and the host cation at the substituted site. Na+ (∼1.39 Å) and Sr2+ (∼1.44 Å) have similar sizes at the A site, whereas Sr2+ (∼1.18 Å) is substantially larger than Ta5+ (∼0.64 Å) at the B site;32 this mismatch explains stronger expansion for Sr at Ta sites.
The electronic structures of relaxed NTO and Sr-doped NTO were investigated. Fig. 4a shows the total and projected density of states (PDOS) for NTO, B2-3Na1Ta, and B4-3Na1Ta (other results are provided in Fig. S3a). In all cases, states near the valence-band maximum (VBM) are dominated by O 2p orbitals, while states near the conduction-band minimum (CBM) are dominated by Ta 5d orbitals. Sr-derived states do not appear near the Fermi level even at the highest Sr content; they do not contribute to the band edges.
Fig. 4b shows the band structures of these models (results for other models in Fig. S3b). NTO showed a calculated band gap of 2.80 eV. The lower Sr-content models, B3-3Na1Ta and B4-3Na1Ta, have slightly smaller gaps of ∼2.79 eV and ∼2.75 eV, respectively. These models exhibit no in-gap impurity states. Conversely, the higher Sr-content models, B1-3Na1Ta and B2-3Na1Ta, exhibit several in-gap impurity states associated with Sr substitution. As indicated by the PDOS in Fig. 4a, acceptor-like levels appear just above the VBM. When in-gap impurity states are ignored, the gaps are 2.74 eV for B1-3Na1Ta and 2.44 eV for B2-3Na1Ta.
Impurity bands appear owing to Sr accumulation within a TaO2 layer. In B1-3Na1Ta and B2-3Na1Ta, an Sr(Ta)O6 octahedron (Sr at the Ta site, hereafter Sr(Ta)) is fully surrounded by Sr atoms. This locally Sr-rich environment promotes electron localisation and yields in-gap states. The charge distribution for these impurity bands is localised on O atoms adjacent to the Sr(Ta)O6 octahedra (see Fig. S4 and S5). Although Sr tends to accumulate also in B3-3Na1Ta, this cell does not contain the Sr(Ta)O6 octahedron fully surrounded by Sr at neighbouring A sites. Accordingly, charge localisation is weaker in B3-3Na1Ta than in B1-3Na1Ta and B2-3Na1Ta.
The DOS of NTO, B1-3Na1Ta, B2-3Na1Ta, and B3-3Na1Ta were also computed using HSE06 (Fig. S6). The resulting gap of NTO was about 4.35 eV, which is in better agreement with the experimental value (4.0 eV) than the PBE result.9,33 A similar change in the band gap was observed for B1-3Na1Ta, B2-3Na1Ta, and B3-3Na1Ta. The overall DOS features, including the in-gap states, remained qualitatively unchanged compared with PBE. These results suggest that while the use of hybrid functionals improves the quantitative band-gap value, the key electronic-structure trends discussed here are robust with respect to the choice of the functional.
Experimentally, An et al. reported that in NaTaO3–Sr(Sr1/3Ta2/3)O3 solid solutions the optical band gap increases with Sr content (UV-vis).20 By contrast, our supercell calculations predict a decreasing gap at higher Sr contents and, when Sr accumulates, the in-gap states emerge. This discrepancy likely reflects the limitations of finite periodic supercells. As indicated by Caldes et al.,34 Sr atoms and Ta atoms are mixed in Sr(Sr1/3Ta2/3)O3 (200 mol% Sr vs. Ta). Random solid solutions require configurational averaging over fractional compositions, which small periodic supercells cannot represent adequately. Consequently, artificial Sr ordering/clustered environments, the absence of configurational averaging, and finite-size effects in small supercells likely bias the computed gap relative to the experiment.
Surface
We also examined the Sr configurations in surface slab models. As reported by Zhao and Selloni,35 various surface termination exist. Here, we focus on bare (clean) surfaces without H2O or H adsorption. We used TaO2 terminated symmetric slabs, reported to be the most stable termination without steps and H2O.35 First, to probe favourable Sr sites, we performed single-point calculations for S1-2Ta. We compared five configurations (a-e, Fig. 5a) that systematically vary the Sr depth from the surface top. As shown in Fig. 5b, the total energy decreases as Sr approaches the surface, by up to 5.8 eV relative to the deepest configuration.Starting from the most stable S1-2Ta configuration (a), we constructed S1-4Na2Ta slabs by additionally replacing four Na sites with Sr (p-t, Fig. 5c), and varied the Sr(Na) depth across five configurations. The S1-2Ta slabs have excess electrons due to the termination and substitution, but the S1-4Na2Ta slabs do not. As in the bulk, shorter Sr–Sr separations stabilise the surface: across the five S1-4Na2Ta configurations, and the most concentrated arrangement is lower by 5.2 eV than the most dispersed one (Fig. 5d). This result indicates a tendency for Sr to segregate on the surface, being consistent with earlier EDX mapping reports of Sr surface segregation.21
To exclude artefacts arising from the slab construction, including spurious Sr–Sr interactions between periodically repeated slabs in S1-2Ta and S1-4Na2Ta, we performed two checks. First, the vacuum thickness was increased from 15 Å to 31 Å; the relative energies remained essentially unchanged over this range (Fig. S7). Second, we computed asymmetric, stoichiometric slabs constructed from a 1 × 1 × 5 unit cell with NaO- and TaO2-terminated surfaces separated by a vacuum layer (S2-1Ta and S2-3Na1Ta). Consistent with the symmetric slabs, the energy increases as Sr is placed deeper toward the slab interior (see Fig. S8 for details). Thus, the preference for Sr at the surface (and the corresponding destabilisation when Sr is buried) does not originate from stoichiometry or charge-compensation effects; it persists in asymmetric, stoichiometric slabs as implied in previous study.33
Table 1 shows Bader charges for the NTO slab and S1-4Na2Ta variants p and t (Fig. 5b). Relative to the undoped slab, O atoms adjacent to Sr(Ta) are less negative, whereas O atoms adjacent to Sr(Na) are more negative. The magnitude of both shifts is larger in t than in p. This trend reflects local charge compensation upon the Sr substitution. Sr has two valence electrons, whereas Ta and Na have five and one, respectively. When a Ta site is substituted by Sr (Sr(Ta)), the local region contains fewer electrons than in pristine NTO; when a Na site is substituted by Sr (Sr(Na)), the local region contains more electrons. Consistent with this simple electron-counting picture, O atoms adjacent to Sr(Ta) show less negative Bader charges, and O atoms adjacent to Sr(Na) show more negative Bader charges. When Sr(Ta) and Sr(Na) are close (p), the local electron deficit and excess can partially compensate, whereas at longer separation (t) this compensation is weaker. This local charge compensation accounts for the greater stability of p relative to t. A similar trend in Bader charges is observed for the bulk (Table S4). The correlation between the mean Sr(Na)–Sr(Ta) separation and the single-point energy is stronger than that for Sr(Na)–Sr(Na) or any Sr–Sr separations (Fig. S9–S11), supporting the stabilisation mechanism of p relative to t.
| Structure | Distance between Sr(Ta) and Sr(Na) layers (Å) | Bader charges | |||
|---|---|---|---|---|---|
| O near Sr(Ta) | O near Sr(Na) | Sr(Ta) | Sr(Na) | ||
| NTO slab | −1.29 | −1.29 | |||
| S1-4Na2Ta (p) | 1.94 | −1.22 | −1.28 | +1.52 | +1.53 |
| S1-4Na2Ta (t) | 17.48 | −1.03 | −1.35 | +1.59 | +1.53 |
In subsequent analyses, we focused on the minimum-energy S1-2Ta and S1-4Na2Ta configurations (a and p, respectively); we optimised ionic positions with the central unit cell constrained to maintain the symmetric slab geometry. Electronic DOS for NTO slab, S1-2Ta, and S1-4Na2Ta are shown in Fig. 6a, and the corresponding band structures are presented in Fig. S12. As in the bulk, states near the VBM are dominated by O 2p orbitals, and those near the CBM by Ta 5d orbitals. No Sr derived states are observed near the Fermi energy. For the NTO slab and S1-2Ta, shallow in-gap states appear within ∼+0.6 eV above the VBM. In contrast, the stoichiometric S1-4Na2Ta exhibits deeper impurity states at ∼+2.0 eV above the VBM, localised on O atoms neighbouring Sr(Ta) (see Fig. S13). If we set aside these strongly localised defect states, which are unlikely to contribute to carrier transport, and estimate the underlying band edges from the PDOS, the apparent gaps are 2.70 eV for NTO (within ∼0.1 eV of the bulk), 2.69 eV for S1-2Ta, and ∼2.76 eV for S1-4Na2Ta. Similar results were obtained for S1-4Na2Ta using HSE06 (Fig. S14).
 | ||
| Fig. 6 (a) Total DOS and PDOS for the relaxed NTO slab, S1-2Ta, and S1-4Na2Ta; energies referenced to Fermi energy. (b) Layer-resolved LDOS for slabs. | ||
We further analysed the local density of states (LDOS) layer-by-layer for the TaO2 planes parallel to (001). Fig. 6b shows the TaO2 layer-resolved LDOS for the NTO, S1-2Ta, and S1-4Na2Ta slabs. For all models, L2 to L11 show nearly identical gaps without any in-gap states. In the surface layer (L1), however, the behaviour changes. For the VBM, NTO and S1-2Ta showed shallow in-gap states within ∼0.6 eV above the VBM. These shallow in-gap states are consistent with the non-stoichiometric termination. By contrast, S1-4Na2Ta showed deeper in-gap states (∼2.0 eV above the VBM) despite its stoichiometric composition. For the CBM, the NTO surface layer closely matches the interior. However, in S1-2Ta and S1-4Na2Ta, the conduction band edges are shifted upward by approximately 2.5 eV relative to interior layers. Therefore, Sr substitution at Ta sites raises the surface CBM, and that additional Sr substitution at Na sites (Sr segregation) elevates both the VBM and the CBM relative to interior layers.
Similar upward shifts of the VBM and CBM are observed in bulk supercells with Sr accumulation around Sr(Ta) in B1-3Na1Ta and B2-3Na1Ta (see Fig. S15–S17). In B1-3Na1Ta and B2-3Na1Ta, the Sr-rich layer showed upward shifting of the VBM and CBM. B3-3Na1Ta and B4-3Na1Ta also showed upward shifts, with reduced contribution from the O 2p near VBM and Ta 5d near the CBM, although the magnitude is smaller than the other models.
To probe the origin of the upward VBM/CBM shifts, we analysed bond lengths in each TaO2 layers and computed pCOHP. In B2-3Na1Ta, Fig. 7 indicates that Ta–Ta bond lengths are elongated near the Sr accumulated layer, whereas the O–O and Ta–O distances are shortened. By contrast, B4-3Na1Ta showed smaller layer-to-layer variations. In S1-4Na2Ta as well, Ta–Ta lengths are elongated and O–O and Ta–O lengths are shortened at the surface (Fig. S18).
Fig. 8 shows pCOHP for the NTO surface, B2-3Na1Ta, and S1-4Na2Ta. All models show similar features; O–O antibonding contributions appear near the VBM, while Ta–O antibonding and Ta–Ta bonding contributions appear near the CBM. The in-gap states in the S1-4Na2Ta model are contributed from O–O antibonding and Ta–O antibonding characters.
Fig. 9 schematically illustrates a possible mechanism of the band-edge shifts. Shortening of O–O distances enhances O–O antibonding interactions near the VBM, shifting the VBM upward. Elongation of Ta–Ta distances weakens Ta–Ta bonding interactions and, shortening Ta–O length increases Ta–O antibonding interactions, effectively shifting the CBM upward. Sr accumulation at the surface also reduces Ta–Ta overlap, particularly near the conduction-band edge. Consequently, both the VBM and the CBM shift upward toward the surface owing to these local structural changes.
Layer-localised band-edge shifts correspond to band bending near the surface, as suggested previously. Previous studies assumed that only the CBM shifts upward toward the surface, producing internal band bending that suppresses electron–hole recombination.19,21 In our calculations, the CBM shifts upward as the TaO2 layer approaches the surface from the interior. In addition, we found that the VBM shifts upward as well in the Sr segregated model. The concurrent upward shifts of the CBM and VBM establish a built-in electric field across the near-surface region. This field drives electron–hole separation and suppresses recombination. As a result, carrier lifetimes increase, enhancing the availability of electrons and holes for interfacial redox reactions with water.
Conclusions
Using first-principles calculations, we systematically examined Sr substitution in the bulk and TaO2-terminated surfaces of NaTaO3 (NTO). Enumeration and screening of configurations showed that shorter Sr–Sr separations are energetically favoured, and the lattice volume increases monotonically with Sr content. Substitution at Ta (B) sites expands the lattice more than that at Na (A) sites, consistent with ionic radii. At lower Sr contents, the gap is comparable to that of pristine NTO (2.80 eV), whereas Sr accumulation (e.g., 1 × 1 × 3 and 1 × 1 × 5; Sr(Ta)O6 surrounded by Sr at A sites) produces in-gap acceptor-like states near the VBM and reduces the host gap.At the surface, Sr segregates strongly and tends to accumulate, which is a trend that is robust to slab stoichiometry and charge compensation. Layer-resolved LDOS shows that in-gap states are confined to the outermost TaO2 layer, while interior layers retain a nearly constant gap. In both S1-2Ta and S1-4Na2Ta slabs, the VBM and CBM shift upward in the surface layer relative to the interior; similar band-edge upshifts appear in Sr-rich layers of bulk supercells. Analysis of bond metrics and pCOHP links these shifts to O–O contraction (enhanced O–O antibonding raising the VBM) and Ta–Ta elongation together with Ta–O antibonding (raising the CBM).
These results establish a near-surface band bending driven by local Sr-induced structural distortions, creating a built-in electric field that promotes electron–hole separation and suppresses recombination, thereby enhancing photo-driven water splitting on Sr-modified NTO.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting the findings of this study are presented in the main text and supplementary information (SI). Supplementary information: supporting figures and tables (PDF), and atomic coordinates used for the calculations (VASP POSCAR format). See DOI: https://doi.org/10.1039/d5cp04385a.Acknowledgements
This study was financially supported by the JSPS-KAKENHI (Grant No. 23H03976 and 25H00902), JST-PRESTO (Grant No. JPMJPR23Q2), Toyota Physical and Chemical Research Institute, Japan, and Institute for Quantum Chemical Exploration, Japan. Theoretical computations were partly performed in Research Center for Computational Science, Okazaki, Japan (25-IMS-C019) and Research Institute for Information Technology, Kyushu University, Japan (p240303346).References
- A. Fujishima and K. Honda, Electrochemical Photolysis of Water at a Semiconductor Electrode, Nature, 1972, 238, 37–38 CrossRef CAS PubMed.
- Z. Zhang and J. T. Yates, Band Bending in Semiconductors: Chemical and Physical Consequences at Surfaces and Interfaces, Chem. Rev., 2012, 112, 5520–5551 CrossRef CAS PubMed.
- A. Kudo and Y. Miseki, Heterogeneous photocatalyst materials for water splitting, Chem. Soc. Rev., 2008, 38, 253–278 RSC.
- K. Maeda and K. Domen, Photocatalytic Water Splitting: Recent Progress and Future Challenges, J. Phys. Chem. Lett., 2010, 1, 2655–2661 CrossRef CAS.
- T. Hisatomi, J. Kubota and K. Domen, Recent advances in semiconductors for photocatalytic and photoelectrochemical water splitting, Chem. Soc. Rev., 2014, 43, 7520–7535 RSC.
- S. Zhu and D. Wang, Photocatalysis: Basic Principles, Diverse Forms of Implementations and Emerging Scientific Opportunities, Adv. Energy Mater., 2017, 7, 1700841 CrossRef.
- A. Kudo, A. Tanaka, K. Domen and T. Onishi, The effects of the calcination temperature of SrTiO3 powder on photocatalytic activities, J. Catal., 1988, 111, 296–301 CrossRef CAS.
- K. Domen, S. Naito, M. Soma, T. Onishi and K. Tamaru, Photocatalytic decomposition of water vapour on an NiO–SrTiO3 catalyst, J. Chem. Soc., Chem. Commun., 1980, 543–544 RSC.
- H. Kato and A. Kudo, Water Splitting into H2 and O2 on Alkali Tantalate Photocatalysts ATaO3 (A = Li, Na, and K), J. Phys. Chem. B, 2001, 105, 4285–4292 CrossRef CAS.
- H. Kato and A. Kudo, New tantalate photocatalysts for water decomposition into H2 and O2, Chem. Phys. Lett., 1998, 295, 487–492 CrossRef CAS.
- A. Iwase, H. Kato, H. Okutomi and A. Kudo, Formation of Surface Nano-step Structures and Improvement of Photocatalytic Activities of NaTaO3 by Doping of Alkaline Earth Metal Ions, Chem. Lett., 2004, 33, 1260–1261 CrossRef CAS.
- H. Kadowaki, N. Saito, H. Nishiyama and Y. Inoue, RuO2-loaded Sr2+-doped CeO2 with d0 Electronic Configuration as a New Photocatalyst for Overall Water Splitting, Chem. Lett., 2007, 36, 440–441 CrossRef CAS.
- A. Kudo and H. Kato, Effect of lanthanide-doping into NaTaO3 photocatalysts for efficient water splitting, Chem. Phys. Lett., 2000, 331, 373–377 CrossRef CAS.
- H. Kato, K. Asakura and A. Kudo, Highly Efficient Water Splitting into H2 and O2 over Lanthanum-Doped NaTaO3 Photocatalysts with High Crystallinity and Surface Nanostructure, J. Am. Chem. Soc., 2003, 125, 3082–3089 CrossRef CAS PubMed.
- Y. Goto, et al., A Particulate Photocatalyst Water-Splitting Panel for Large-Scale Solar Hydrogen Generation, Joule, 2018, 2, 509–520 CrossRef CAS.
- T. H. Chiang, et al., Efficient Photocatalytic Water Splitting Using Al-Doped SrTiO3 Coloaded with Molybdenum Oxide and Rhodium–Chromium Oxide, ACS Catal., 2018, 8, 2782–2788 CrossRef CAS.
- S. Kasamatsu, O. Sugino, T. Ogawa and A. Kuwabara, Dopant arrangements in Y-doped BaZrO3 under processing conditions and their impact on proton conduction: a large-scale first-principles thermodynamics study, J. Mater. Chem. A, 2020, 8, 12674–12686 RSC.
- F. Tian, A Review of Solid-Solution Models of High-Entropy Alloys Based on Ab Initio Calculations, Front. Mater., 2017, 4, 36 CrossRef.
- H. Onishi, Sodium Tantalate Photocatalysts Doped with Metal Cations: Why Are They Active for Water Splitting?, ChemSusChem, 2019, 12, 1825–1834 CrossRef CAS PubMed.
- L. An and H. Onishi, Electron–Hole Recombination Controlled by Metal Doping Sites in NaTaO3 Photocatalysts, ACS Catal., 2015, 5, 3196–3206 CrossRef CAS.
- L. An, et al., Photoexcited Electrons Driven by Doping Concentration Gradient: Flux-Prepared NaTaO3 Photocatalysts Doped with Strontium Cations, ACS Catal., 2018, 8, 9334–9341 CrossRef CAS.
- G. Kresse and J. Furthmüller, Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set, Comput. Mater. Sci., 1996, 6, 15–50 CrossRef CAS.
- J. P. Perdew, K. Burke and M. Ernzerhof, Generalized Gradient Approximation Made Simple, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS PubMed.
- J. Heyd, G. E. Scuseria and M. Ernzerhof, Erratum: “Hybrid functionals based on a screened Coulomb potential” [J. Chem. Phys.118, 8207 (2003)], J. Chem. Phys., 2006, 124, 219906 CrossRef.
- S. Grimme, S. Ehrlich and L. Goerigk, Effect of the damping function in dispersion corrected density functional theory, J. Comput. Chem., 2011, 32, 1456–1465 CrossRef CAS PubMed.
- V. L. Deringer, A. L. Tchougréeff and R. Dronskowski, Crystal Orbital Hamilton Population (COHP) Analysis As Projected from Plane-Wave Basis Sets, J. Phys. Chem. A, 2011, 115, 5461–5466 CrossRef CAS PubMed.
- S. Maintz, V. L. Deringer, A. L. Tchougréeff and R. Dronskowski, LOBSTER: A tool to extract chemical bonding from plane-wave based DFT, J. Comput. Chem., 2016, 37, 1030–1035 CrossRef CAS PubMed.
- U. Herath, et al., PyProcar: A Python library for electronic structure pre/post-processing, Comput. Phys. Commun., 2020, 251, 107080 CrossRef CAS.
- Z. Wang, J. Zhang and H. Du, Achieving the Sensing Property of Hg0 Molecules on Black Phosphorene Nanosheets Using Anisotropy as a Response Signal, ACS Appl. Nano Mater., 2025, 8, 8417–8423 CrossRef CAS.
- V. Popescu and A. Zunger, Extracting E versus
![[P with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0050_20d1.gif) effective band structure from supercell calculations on alloys and impurities, Phys. Rev. B: Condens. Matter Mater. Phys., 2012, 85, 085201 CrossRef.
effective band structure from supercell calculations on alloys and impurities, Phys. Rev. B: Condens. Matter Mater. Phys., 2012, 85, 085201 CrossRef. - K. Momma and F. Izumi, VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data, J. Appl. Crystallogr., 2011, 44, 1272–1276 CrossRef CAS.
- R. D. Shannon, Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides, Acta Crystallogr., Sect. A, 1976, 32, 751–767 CrossRef.
- Z.-K. Tang, C. Di Valentin, X. Zhao, L.-M. Liu and A. Selloni, Understanding the Influence of Cation Doping on the Surface Chemistry of NaTaO3 from First Principles, ACS Catal., 2019, 9, 10528–10535 CrossRef CAS.
- M. T. Caldes, et al., Solving modulated structures by X-ray and electron crystallography, Micron, 2001, 32, 497–507 CrossRef CAS PubMed.
- X. Zhao and A. Selloni, Structure and stability of NaTaO3(001) and KTaO3(001) surfaces, Phys. Rev. Mater., 2019, 3, 015801 CrossRef CAS.
| This journal is © the Owner Societies 2026 |