 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
High-resolution spectroscopy of [H,C,N]+: III. Infrared Ã2Σ+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/h2_char_0058_0303.gif) 2Π electronic transition of HCN+
2Π electronic transition of HCN+
Samuel J. P.
Marlton
 *,
Philipp C.
Schmid
*,
Philipp C.
Schmid
 ,
Weslley G. D. P.
Silva
,
Weslley G. D. P.
Silva
 ,
Oskar
Asvany
,
Oskar
Asvany
 and
Stephan
Schlemmer
and
Stephan
Schlemmer
 *
*
I. Physikalisches Institut, Universität zu Köln, Zülpicher Str. 77, 50937 Cologne, Germany. E-mail: marlton@ph1.uni-koeln.de; schlemmer@ph1.uni-koeln.de
First published on 22nd December 2025
Abstract
The Ã2Σ+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π electronic transition of the fundamental HCN+ ion is reported. The spectrum exhibits a rotational, fine, and hyperfine structure that provides insights into the geometries and wavefunctions of the Ã2Σ+ and
2Π electronic transition of the fundamental HCN+ ion is reported. The spectrum exhibits a rotational, fine, and hyperfine structure that provides insights into the geometries and wavefunctions of the Ã2Σ+ and ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π states. The extracted spectroscopic constants for the à state include (in cm−1) Te = 3238.8224(2), B = 1.39568(3), D = 2.27(9) × 10−5, γ = −0.07332(7), γD = 1.87(3) × 10−4, and the Fermi contact hyperfine constant for the HCN+ nitrogen atom bF = 0.0059(1). Based on this value, the Ã2Σ1/2+ state electronic wavefunction is estimated to have 11.5 ± 0.2% s orbital character. The spin–orbit coupling constant for the
2Π states. The extracted spectroscopic constants for the à state include (in cm−1) Te = 3238.8224(2), B = 1.39568(3), D = 2.27(9) × 10−5, γ = −0.07332(7), γD = 1.87(3) × 10−4, and the Fermi contact hyperfine constant for the HCN+ nitrogen atom bF = 0.0059(1). Based on this value, the Ã2Σ1/2+ state electronic wavefunction is estimated to have 11.5 ± 0.2% s orbital character. The spin–orbit coupling constant for the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) state was also determined with high precision ASO = −49.3122(4) cm−1. This work illustrates that leak-out spectroscopy can be applied to measure high-resolution spectra of low energy electronic transitions.
state was also determined with high precision ASO = −49.3122(4) cm−1. This work illustrates that leak-out spectroscopy can be applied to measure high-resolution spectra of low energy electronic transitions.
Introduction
The HCN+ cation is a fundamental molecule with an interesting electronic structure; however, limited details have been reported about its electronic excited states. HCN+ is highly reactive,1,2 making it difficult to isolate for spectroscopic interrogation, so the only reported infrared spectra of HCN+ are vibrational spectra in inert Ne matrices3 and rovibrational and rotational spectra that have recently been measured in cryogenic ion traps as part of this series of publications.4,5No electronic transitions of HCN+ have been directly measured; however, individual states of HCN+ have been observed by photoionisation and photoelectron spectroscopy of the less reactive neutral HCN.6–10 In these investigations, several bands from the HCN+ ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π ground state and a low lying Ã2Σ+ state were observed. These states are separated by only ≈3250 cm−1 because of the similar energies of the 1π and 5σ orbitals, which are illustrated in Fig. 1.6–10
2Π ground state and a low lying Ã2Σ+ state were observed. These states are separated by only ≈3250 cm−1 because of the similar energies of the 1π and 5σ orbitals, which are illustrated in Fig. 1.6–10
In most of these photoelectron and photoionisation spectra, only vibronic transitions are resolved.7–10 The highest resolution was achieved using zero electron kinetic energy (ZEKE) photoionisation spectroscopy (Δ![[v with combining macron]](https://www.rsc.org/images/entities/i_char_0076_0304.gif) = 1 cm−1), which exhibited a partially resolved rotational structure.6 From the ZEKE spectra, it was possible to extract first experimental values of spin–orbit coupling of the ground state (ASO = −49.8(1) cm−1), the rotational constant of the
= 1 cm−1), which exhibited a partially resolved rotational structure.6 From the ZEKE spectra, it was possible to extract first experimental values of spin–orbit coupling of the ground state (ASO = −49.8(1) cm−1), the rotational constant of the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π ground state (B″ = 1.36(1) cm−1), and the rotational constant of the Ã2Σ+ excited state (B′ = 1.37(1) cm−1). Despite being high-quality spectra, it is not clear from these results whether the rotational constant significantly changes in the Ã2Σ+ excited state. This is in contrast to high-level multireference configuration interaction calculations that include vibronic coupling, which predict a significant excited state CN bond contraction, which significantly changes the rotational constant in the excited state.11 Furthermore, higher resolution experimental studies are required to characterise the more subtle spectroscopic properties of HCN+. For example, the couplings between electronic orbital angular momentum (Λ), electron spin (S), nuclear rotation, and nuclear spin of the nitrogen (IN) and hydrogen (IH) are of fundamental interest.
2Π ground state (B″ = 1.36(1) cm−1), and the rotational constant of the Ã2Σ+ excited state (B′ = 1.37(1) cm−1). Despite being high-quality spectra, it is not clear from these results whether the rotational constant significantly changes in the Ã2Σ+ excited state. This is in contrast to high-level multireference configuration interaction calculations that include vibronic coupling, which predict a significant excited state CN bond contraction, which significantly changes the rotational constant in the excited state.11 Furthermore, higher resolution experimental studies are required to characterise the more subtle spectroscopic properties of HCN+. For example, the couplings between electronic orbital angular momentum (Λ), electron spin (S), nuclear rotation, and nuclear spin of the nitrogen (IN) and hydrogen (IH) are of fundamental interest.
In particular, the hyperfine coupling between electronic angular momenta and nuclear spin is valuable because it facilitates quantitative experimental characterisation of the electronic wavefunction.12 Some of these couplings have now been described for the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π state vibrational levels,4,5 but not for excited electronic states. The ability to characterise these subtle couplings in the excited state is also a necessary step towards finding promising candidate polyatomic molecular ions for which laser-cooling might be possible.13
2Π state vibrational levels,4,5 but not for excited electronic states. The ability to characterise these subtle couplings in the excited state is also a necessary step towards finding promising candidate polyatomic molecular ions for which laser-cooling might be possible.13
It is often challenging to characterise the more subtle couplings between angular momenta for gas-phase ions in excited electronic states. For example, hyperfine splitting in excited electronic states is typically on the order of less than 0.1 cm−1 and has been observed for only a few gas-phase molecular ions, almost all of which are diatomics.14–24 To our knowledge, the only polyatomic ions for which excited state hyperfine splitting has been observed in the gas-phase are isotopologues of HCP+ and N2O+.22,23 One reason for this is that excited electronic states often have picosecond or femtosecond lifetimes, and so lifetime broadening makes small splittings between rovibronic lines unresolvable.25 Lifetime broadening becomes an increasingly common feature of larger molecules because they have more degrees of freedom and higher densities of states, which can facilitate fast excited state deactivation by dissociation, internal conversion, or intersystem crossing.
Well established methods for measuring electronic spectra of gas-phase ions are generally not suitable for resolving splittings smaller than 0.01 cm−1. For example, single-photon resonance enhanced photodissociation spectroscopy is often lifetime broadened,25 one-colour multiphoton dissociation can produce broad peaks that are slightly shifted,26,27 messenger tagging does not provide a spectrum of the bare ion,28–30 and one-colour photoionisation spectroscopy of neutral precursors with high energy photons is more Doppler broadened because this broadening increases proportionally to the photon energy. High-resolution infrared spectra of the HCN+ Ã2Σ+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π transition are probably achievable using techniques such as laser induced reactions (LIR),31 laser induced inhibition of cluster growth (LIICG),32 and multiple-colour spectroscopy.29,30 However, these methods typically require sophisticated understanding of some reaction schemes, spectroscopic levels, or subtle experimental conditions to implement. Finally, ions like HCN+ are too reactive to be straightforwardly investigated in jets, discharges, and absorption cells.
2Π transition are probably achievable using techniques such as laser induced reactions (LIR),31 laser induced inhibition of cluster growth (LIICG),32 and multiple-colour spectroscopy.29,30 However, these methods typically require sophisticated understanding of some reaction schemes, spectroscopic levels, or subtle experimental conditions to implement. Finally, ions like HCN+ are too reactive to be straightforwardly investigated in jets, discharges, and absorption cells.
In this study, we show that the recently developed leak-out spectroscopy (LOS) method33—which was recently applied to electronic spectroscopy for the first time34—overcomes some of these disadvantages and yields a high resolution single photon electronic spectrum of bare HCN+. This is the first spectrum of the Ã2Σ+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π transition for HCN+, and the spectrum has a sufficiently high resolution to resolve a rotational, fine, and hyperfine structure. This is encouraging for the potential to measure single-photon electronic spectra of bare gas-phase ions far below the dissociation threshold using leak-out spectroscopy.
2Π transition for HCN+, and the spectrum has a sufficiently high resolution to resolve a rotational, fine, and hyperfine structure. This is encouraging for the potential to measure single-photon electronic spectra of bare gas-phase ions far below the dissociation threshold using leak-out spectroscopy.
This article is a part of a series of publications reporting high-resolution infrared rovibrational4 and pure rotational5 spectra of HCN+. The previous parts of this series described the couplings between electronic and nuclear angular momenta of HCN+ in the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) electronic state. The electronic spectrum of this work was shown in Publication I of this series without analysis.4 In this part, we describe the couplings between electronic and nuclear angular momenta of HCN+ in the
electronic state. The electronic spectrum of this work was shown in Publication I of this series without analysis.4 In this part, we describe the couplings between electronic and nuclear angular momenta of HCN+ in the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) and à states. We then discuss the rovibronic structure of the experimental Ã2Σ+ ←
and à states. We then discuss the rovibronic structure of the experimental Ã2Σ+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π LOS spectrum. The experimental spectrum is fitted with a rovibronic Hamiltonian to extract several spectroscopic constants. These results are contextualised by comparison with a calculated potential energy surface. We then discuss the hyperfine splitting observed in the Ã2Σ+ ←
2Π LOS spectrum. The experimental spectrum is fitted with a rovibronic Hamiltonian to extract several spectroscopic constants. These results are contextualised by comparison with a calculated potential energy surface. We then discuss the hyperfine splitting observed in the Ã2Σ+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π spectrum, which provides further insights into the electronic wavefunction of the HCN+ Ã state. Finally, we report a further rovibrational transition of HCN+ that overlaps with the rovibronic Ã2Σ+ ←
2Π spectrum, which provides further insights into the electronic wavefunction of the HCN+ Ã state. Finally, we report a further rovibrational transition of HCN+ that overlaps with the rovibronic Ã2Σ+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π spectrum. We argue that this rovibrational transition excites an upper (κ) Renner–Teller component of an
2Π spectrum. We argue that this rovibrational transition excites an upper (κ) Renner–Teller component of an ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) state combination band composed of two quanta of ν2 and one quantum of ν1. Together with the previous studies and the earlier parts of this series,4–6,11 these results provide a detailed experimental foundation for understanding the Renner–Teller vibronic coupling in HCN+, as well as the vibronic, rovibronic, fine, and hyperfine structure of the HCN+ Ã electronic state.
state combination band composed of two quanta of ν2 and one quantum of ν1. Together with the previous studies and the earlier parts of this series,4–6,11 these results provide a detailed experimental foundation for understanding the Renner–Teller vibronic coupling in HCN+, as well as the vibronic, rovibronic, fine, and hyperfine structure of the HCN+ Ã electronic state.
Experimental details
Spectra were measured using leak-out spectroscopy (LOS) in the cryogenically cooled 22-pole ion trap instrument referred to as LIRtrap.31,35 This method has been described in detail previously.33 Briefly, HCN+ ions were generated from acetonitrile vapour, which were bombarded with 70 eV electrons in a storage ion source. The ions in the source were pulsed out every 500 ms into a quadrupole mass filter (QMF1), which was set to select m/z 27. The mass selected ions exiting the QMF1 were then trapped and stored in a 22-pole ion trap mounted on a 10 K cold-head.36 After 10 ms of cooling with the neutral Ne (with a continuous number density of approximately 1014 cm−3) in the trap region, the ions were irradiated by light from a continuous optical parametric oscillator system (Toptica TOPO, ≈100 W cm−2, accuracy ≈10−4 cm−1) for 300 ms, the timing of which was controlled with a mechanical shutter. The light frequency was measured using a wavemeter (Bristol 621 A-IR, resolution ≈10−3 cm−1). Photoexcited ions could collide with neutral Ne buffer gas to convert some of their internal energy into kinetic energy. The ions with enhanced kinetic energy could overcome the low potential barrier at the trap exit and leak out of the trap. After exiting the trap, ions pass through a second quadrupole mass filter (QMF2) set to select m/z 27, before being detected using a Daly type detector.37,38 This process was repeated while scanning the OPO frequency to record a rovibrational LOS spectrum.Several experimental configurations that differed slightly from one another were employed in this study. The spectrum was measured at a trap temperature of 27 K with continuous Ne as the buffer gas (n ≈ 2 × 1014 cm−3). This configuration was optimised to observe transitions involving the Ω = 1/2 component of the ground state, which requires elevated temperatures because the Ω = 3/2 component is lower in energy by ≈50 cm−1. This configuration was also optimised to suppress background peaks from isobaric C2H3+ by lowering the trap potential, which increased the trap strength and reduced the leak-out signal of C2H3+ more than that of HCN+. This difference between the leak-out signal of C2H3+ and HCN+ probably reflects a difference in their vibrational to kinetic energy transfer characteristics. A small range of the spectrum was measured at 9 K, with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 mixture of Ne
3 mixture of Ne![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) He that was pulsed into the ion trap. This configuration was optimised to provide a contour fit of the hyperfine splitting, which required low temperature to reduce Doppler broadening and care to avoid saturating the transitions as a result of near-total leak-out of the trapped ion population. Finally, the spectrum was remeasured at 4 K, with a 1
He that was pulsed into the ion trap. This configuration was optimised to provide a contour fit of the hyperfine splitting, which required low temperature to reduce Doppler broadening and care to avoid saturating the transitions as a result of near-total leak-out of the trapped ion population. Finally, the spectrum was remeasured at 4 K, with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 mixture of Ne
3 mixture of Ne![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) He that was pulsed into the ion trap, using the COLtrap machine.38 This configuration was optimised to provide the line positions of hyperfine components.
He that was pulsed into the ion trap, using the COLtrap machine.38 This configuration was optimised to provide the line positions of hyperfine components.
Computational details
The geometries of the![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π and Ã2Σ+ states were optimised using the CASPT2(9,8)/cc-pVQZ method39,40 in the OpenMolcas program.41 The PES in Fig. 4 is constructed by linear interpolation of the xyz coordinates from the optimised
2Π and Ã2Σ+ states were optimised using the CASPT2(9,8)/cc-pVQZ method39,40 in the OpenMolcas program.41 The PES in Fig. 4 is constructed by linear interpolation of the xyz coordinates from the optimised ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π geometry (interpolated coordinate = 0) to the optimised Ã2Σ+ geometry (interpolated coordinate = 1). The electronic energies in Fig. 4 are calculated using the SOC+MRCI+Q(9,8)/cc-pVQZ method40,42 in the ORCA/5.0.2 program.43
2Π geometry (interpolated coordinate = 0) to the optimised Ã2Σ+ geometry (interpolated coordinate = 1). The electronic energies in Fig. 4 are calculated using the SOC+MRCI+Q(9,8)/cc-pVQZ method40,42 in the ORCA/5.0.2 program.43
Results and discussion
Angular momenta and splitting
HCN+ has a relatively complex electronic structure because it has several angular momenta that couple differently in the![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) and à states. These include the rotation of the nuclear framework R, electronic orbital angular momentum Λ, electron spin S, nuclear spin of the nitrogen IN and nuclear spin of the hydrogen IH. We also refer to the total angular momentum exclusive of electron and nuclear spin N, the total angular momentum exclusive of nuclear spin J, the total angular momentum exclusive of the hydrogen nuclear spin F1 and the total angular momentum F. The splittings in the ground
and à states. These include the rotation of the nuclear framework R, electronic orbital angular momentum Λ, electron spin S, nuclear spin of the nitrogen IN and nuclear spin of the hydrogen IH. We also refer to the total angular momentum exclusive of electron and nuclear spin N, the total angular momentum exclusive of nuclear spin J, the total angular momentum exclusive of the hydrogen nuclear spin F1 and the total angular momentum F. The splittings in the ground ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) and excited à states are illustrated in Fig. 2 for a level with one quantum of nuclear rotation (R = 1) and will be discussed with reference to the experimental spectrum.
and excited à states are illustrated in Fig. 2 for a level with one quantum of nuclear rotation (R = 1) and will be discussed with reference to the experimental spectrum.
The couplings between these angular momenta were described in detail in parts 1 and 2 of this series for the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) state,4,5 and the couplings of a Σ symmetry vibrationally excited state were also described in part 1 of this series.4 Nevertheless, it will be necessary to describe how these couplings split rovibrational levels in order to understand the experimental spectra reported here.
state,4,5 and the couplings of a Σ symmetry vibrationally excited state were also described in part 1 of this series.4 Nevertheless, it will be necessary to describe how these couplings split rovibrational levels in order to understand the experimental spectra reported here.
Leak-out spectroscopy
The experimental spectrum of the Ã2Σ+ ←![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π vibronic origin transition measured using leak-out spectroscopy (LOS) is shown in Fig. 3 (black trace). This spectrum was measured at 27 K. Spin–orbit coupling (SOC) splits the
2Π vibronic origin transition measured using leak-out spectroscopy (LOS) is shown in Fig. 3 (black trace). This spectrum was measured at 27 K. Spin–orbit coupling (SOC) splits the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π state into two components—
2Π state into two components—![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π3/2 and
2Π3/2 and ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π1/2—which are distinguished by the quantum number of the total electron angular momentum Ω = |Λ + Σ| = |1 ± 1/2|, where Λ and Σ are the quantum numbers for the orbital and spin angular momenta associated with
2Π1/2—which are distinguished by the quantum number of the total electron angular momentum Ω = |Λ + Σ| = |1 ± 1/2|, where Λ and Σ are the quantum numbers for the orbital and spin angular momenta associated with ![[L with combining circumflex]](https://www.rsc.org/images/entities/i_char_004c_0302.gif) z and Ŝz, respectively. The Ω = 3/2 component is lower in energy because the …1π3 configuration is more than half-filled (Fig. 1). The experimental spectrum exhibits both the Ã2Σ1/2+ ←
z and Ŝz, respectively. The Ω = 3/2 component is lower in energy because the …1π3 configuration is more than half-filled (Fig. 1). The experimental spectrum exhibits both the Ã2Σ1/2+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π3/2 and Ã2Σ1/2+ ←
2Π3/2 and Ã2Σ1/2+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π1/2 components, with the Ã2Σ1/2+ ←
2Π1/2 components, with the Ã2Σ1/2+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π3/2 transition being more intense. The Ã2Σ1/2+ ←
2Π3/2 transition being more intense. The Ã2Σ1/2+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π3/2 transition is recognisable as a Ω = 1/2 ← 3/2 type transition because there are no transitions involving J″ = 1/2, but there are transitions involving J′ = 1/2.
2Π3/2 transition is recognisable as a Ω = 1/2 ← 3/2 type transition because there are no transitions involving J″ = 1/2, but there are transitions involving J′ = 1/2.
 | ||
Fig. 3 Electronic spectrum of the HCN+ Ã2Σ+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π origin transition (black trace) measured using LOS at 27 K compared with a simulation (grey sticks) based on a fit using the PGOPHER program.44 2Π origin transition (black trace) measured using LOS at 27 K compared with a simulation (grey sticks) based on a fit using the PGOPHER program.44 | ||
Coupling of the angular momenta of HCN+ split the rovibronic levels of the ground and excited state, as summarised in Fig. 2. For the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π state, the largest splitting arises from spin orbit coupling, giving rise to
2Π state, the largest splitting arises from spin orbit coupling, giving rise to ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π1/2 and
2Π1/2 and ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π3/2 components with a spin–orbit constant of ASO = −49.3122(4) cm−1 (indicated in Fig. 3). This value is reasonably close to previous MRCI calculations (−49.9 cm−1)11 and ZEKE measurements (−49.8 cm−1)6 and −49.3113(3) cm−1 calculated from the rovibrational spectra reported in part I of this series.4 Each J rotational level in the
2Π3/2 components with a spin–orbit constant of ASO = −49.3122(4) cm−1 (indicated in Fig. 3). This value is reasonably close to previous MRCI calculations (−49.9 cm−1)11 and ZEKE measurements (−49.8 cm−1)6 and −49.3113(3) cm−1 calculated from the rovibrational spectra reported in part I of this series.4 Each J rotational level in the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π state is split by Λ-doubling into e and f components. The Ã2Σ+ state does not split by spin orbit coupling because it has Λ = 0. However, the spin (S = 1/2) couples with nuclear rotation to split each N rotational level into two components with J = |N ± S|. Additional splitting from hyperfine coupling will be discussed later.
2Π state is split by Λ-doubling into e and f components. The Ã2Σ+ state does not split by spin orbit coupling because it has Λ = 0. However, the spin (S = 1/2) couples with nuclear rotation to split each N rotational level into two components with J = |N ± S|. Additional splitting from hyperfine coupling will be discussed later.
These couplings between nuclear rotation and electronic angular momenta give rise to a level structure that is shown in the SI, which also illustrates the various possible branches that are observed in the rovibronic spectrum. The selection rules are ΔJ = 0, ±1, with e ↔ f for ΔJ = 0 or e ↔ e and f ↔ f for ΔJ = ±1. This means that there are six branches expected for the Ã2Σ1/2+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π3/2 transition and six branches for the Ã2Σ1/2+ ←
2Π3/2 transition and six branches for the Ã2Σ1/2+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π1/2 transition, which we label by ΔN (in lower case) and ΔJ (in upper case). Eleven of these twelve branches were observed, with the exception of the weak oP branch (ΔN = −2 and ΔJ = −1) of the Ã2Σ1/2+ ←
2Π1/2 transition, which we label by ΔN (in lower case) and ΔJ (in upper case). Eleven of these twelve branches were observed, with the exception of the weak oP branch (ΔN = −2 and ΔJ = −1) of the Ã2Σ1/2+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π1/2 transition. The observed branches were included in a global fit with a nearby HCN+ combination band of Π symmetry that has not been reported before, with the rovibrational ν1 spectrum reported in ref. 4, the rovibrational spectrum of a combination band with Σ symmetry reported in ref. 4, and the rotational spectra reported in ref. 5. We refer to this fit as “fit 1”, which does not include hyperfine coupling. The observed rovibronic and rovibrational lines of HCN+ are captured well by fit 1, with average residuals lower than the wavemeter resolution (0.001 cm−1). Additional lines are observed that arise from isobaric C2H3+.
2Π1/2 transition. The observed branches were included in a global fit with a nearby HCN+ combination band of Π symmetry that has not been reported before, with the rovibrational ν1 spectrum reported in ref. 4, the rovibrational spectrum of a combination band with Σ symmetry reported in ref. 4, and the rotational spectra reported in ref. 5. We refer to this fit as “fit 1”, which does not include hyperfine coupling. The observed rovibronic and rovibrational lines of HCN+ are captured well by fit 1, with average residuals lower than the wavemeter resolution (0.001 cm−1). Additional lines are observed that arise from isobaric C2H3+.
The experimental spectrum in Fig. 3 is compared to a fit produced using the PGOPHER program44 using the Hamiltonian:
 | (1) |
The spectroscopic constants from fit 1 are shown in Table 1. The B rotational constant is 3% larger in the à state (B′ = 1.39581(3) cm−1) than in the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) state (B′ = 1.35276(2) cm−1). This result is in good agreement with the B constants from sophisticated MRCI calculations that take into account vibronic coupling (B′ = 1.3974 cm−1 and B″ = 1.3533 cm−1).11 This agreement also indicates that the à state is mixed with the excited (ν1 + ν2)μ vibrational level of the
state (B′ = 1.35276(2) cm−1). This result is in good agreement with the B constants from sophisticated MRCI calculations that take into account vibronic coupling (B′ = 1.3974 cm−1 and B″ = 1.3533 cm−1).11 This agreement also indicates that the à state is mixed with the excited (ν1 + ν2)μ vibrational level of the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π ground state and that vibronic mixing beyond the Born–Oppenheimer approximation is particularly important for HCN+, as calculated in ref. 11. Because the rotational constant B is determined by the moment of inertia, which is determined by the molecular structure, these values validate the calculated result that the CN bond distance contracts significantly in the à state. Based on previous ZEKE experiments, it was ambiguous whether the CN bond length significantly contracts in the excited state (B′ = 1.37(1) cm−1 and B″ = 1.36(1) cm−1).6 This ambiguity between experiment and theory is now resolved by the higher resolution spectra presented here.
2Π ground state and that vibronic mixing beyond the Born–Oppenheimer approximation is particularly important for HCN+, as calculated in ref. 11. Because the rotational constant B is determined by the moment of inertia, which is determined by the molecular structure, these values validate the calculated result that the CN bond distance contracts significantly in the à state. Based on previous ZEKE experiments, it was ambiguous whether the CN bond length significantly contracts in the excited state (B′ = 1.37(1) cm−1 and B″ = 1.36(1) cm−1).6 This ambiguity between experiment and theory is now resolved by the higher resolution spectra presented here.
![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π electronic spectrum, pure rotational transitions, rovibrational spectrum of the fundamental C–H stretch ν1, and the rovibrational spectra of two combination bands—ν3(σ) + 2ν2(π) and ν1(σ) + ν2(π). Fit 1 refers to this global fit. Fit 2 of the à state refers to a line fit of the 4 K spectrum, which includes hyperfine splitting. The symmetry of single quanta of the ν1(σ) and ν2(π) vibrations is indicated. All values are reported in cm−1. The hyperfine constants from fit 2 are shown in Table 2
2Π electronic spectrum, pure rotational transitions, rovibrational spectrum of the fundamental C–H stretch ν1, and the rovibrational spectra of two combination bands—ν3(σ) + 2ν2(π) and ν1(σ) + ν2(π). Fit 1 refers to this global fit. Fit 2 of the à state refers to a line fit of the 4 K spectrum, which includes hyperfine splitting. The symmetry of single quanta of the ν1(σ) and ν2(π) vibrations is indicated. All values are reported in cm−1. The hyperfine constants from fit 2 are shown in Table 2
![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π 2Π |
||||||
|---|---|---|---|---|---|---|
| Fit 1a | Fit 2a | Rovibb | MRCIc | ZEKEd | Rote | |
| a Values obtained in this work. b Values taken from Ref. 4. c Values taken from Ref. 11. d Values taken from Ref. 6. e Values taken from Ref. 5. | ||||||
| B | 1.35276(1) | 1.35278(2) | 1.35275(1) | 1.3533 | 1.36(1) | 1.3527672(2) |
| A | −49.3120(3) | −49.3122(4) | −49.3113(3) | −49.9 | −49.8(2) | |
| A D | −0.00145(4) | −0.00146(3) | −0.00144(1) | |||
| p | 0.0245(1) | 0.0250(1) | 0.02440(6) | |||
| q | −0.00201(1) | −0.001995(6) | −0.002001(6) | −0.0020026(3) | ||
| D (× 10−6) | 3.3(2) | 3.1(4) | 3.3(1) | 3.28(3) | ||
| Ã2Σ+ | ν 3(σ) + 2ν2(π) | |||||
|---|---|---|---|---|---|---|
| Fit 1a | Fit 2a | MRCIc | ZEKEd | Fit 1a | MRCIc | |
| T e | 3238.8216(2) | 3238.8224(2) | 3233.1 | 3231 | 3272.955(3) | 3269.0 |
| B | 1.39581(3) | 1.39568(3) | 1.3974 | 1.37(1) | 1.3610(1) | 1.3562 |
| A | −4.587(8) | −4.6 | ||||
| γ | −0.07202(2) | −0.07332(7) | ||||
| p | −0.89(2) | |||||
| q | 0.0034(1) | |||||
| D (× 10−6) | 27(1) | 22.7(9) | 1.0(1) | |||
| H (× 10−7) | 70(8) | |||||
| γ D | 0.00012(1) | 0.000187(3) | ||||
| ν 1(σ) | ν 1(σ) + ν2(π) | |||||
|---|---|---|---|---|---|---|
| Fit 1a | Rovibb | MRCIc | Fit 1a | Rovibb | MRCIc | |
| T e | 3056.3413(2) | 3056.3412(1) | 3071.9 | 3340.8480(2) | 3340.8480(2) | 3352.0 |
| B | 1.34340(1) | 1.343387(9) | 1.3445 | 1.36993(2) | 1.36993(1) | 1.3710 |
| A | −48.5992(3) | −48.5987(3) | −49.1 | |||
| A D | −0.00138(1) | −0.001364(6) | ||||
| γ | −0.07294(9) | −0.00463(5) | −0.00457(3) | |||
| p | 0.0287(1) | 0.02862(6) | ||||
| q | −0.00236(1) | −0.002355(7) | ||||
| D (× 10−6) | 3.0(1) | 2.94(7) | 2.8(4) | 2.8(2) | ||
To illustrate the electronic states and their geometry changes investigated in this work, a potential energy surface (PES) for HCN+ is shown in Fig. 4. The PES follows the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π3/2 (purple curve) and
2Π3/2 (purple curve) and ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π1/2 (green curve) spin–orbit components of the ground electronic state and the Ã2Σ+ excited state (blue curve). The x axis in Fig. 4 shows the optimised
2Π1/2 (green curve) spin–orbit components of the ground electronic state and the Ã2Σ+ excited state (blue curve). The x axis in Fig. 4 shows the optimised ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) state geometry at x = 0 and the optimised à state geometry at x = 1. Other x axis values represent interpolation (or extrapolation) between the xyz coordinates of the
state geometry at x = 0 and the optimised à state geometry at x = 1. Other x axis values represent interpolation (or extrapolation) between the xyz coordinates of the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) and à optimised geometries. Both states have linear C∞v optimised geometries, and the CH bond length is similar in the
and à optimised geometries. Both states have linear C∞v optimised geometries, and the CH bond length is similar in the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) ground state (CH
ground state (CH![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) = 1.087 Å) and the à excited state (CHà = 1.086 Å). The calculated CN bond length is much larger in the
= 1.087 Å) and the à excited state (CHà = 1.086 Å). The calculated CN bond length is much larger in the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) ground state (CN
ground state (CN![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) = 1.222 Å) than in the à excited state (CNà = 1.150 Å). Based on our calculations, the Ã2Σ+ ←
= 1.222 Å) than in the à excited state (CNà = 1.150 Å). Based on our calculations, the Ã2Σ+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π origin transition can be expected to be weak because of a low oscillator strength (f = 0.0004 as calculated using MRCI+Q) and unfavorable Franck–Condon factors due to the change in the CN bond distance.
2Π origin transition can be expected to be weak because of a low oscillator strength (f = 0.0004 as calculated using MRCI+Q) and unfavorable Franck–Condon factors due to the change in the CN bond distance.
There is an avoided crossing between the Ã2Σ1/2+ state and the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π1/2 component close to the Ã2Σ1/2+ state minimum (see Fig. 4b), which involves the two states repelling each other and exchanging electronic character due to SOC (note that the selection rule for spin–orbit interactions between electronic states is ΔΩ = 0). Although the states do not cross, the lower energy Ω = 1/2 state has Ã2Σ1/2+ character at CN = 1.132 Å, the higher energy Ω = 1/2 state has Ã2Σ1/2+ character at CN = 1.138 Å, and the electronic character of both states is mixed at CN = 1.1355 Å as visualised by the changing colour in Fig. 4b. Although this avoided crossing is close to the minimum of the Ã2Σ1/2+ state, there are no apparent effects from this avoided crossing causing distortions of the experimental spectrum. It is possible that this is because the Ã2Σ+ ←
2Π1/2 component close to the Ã2Σ1/2+ state minimum (see Fig. 4b), which involves the two states repelling each other and exchanging electronic character due to SOC (note that the selection rule for spin–orbit interactions between electronic states is ΔΩ = 0). Although the states do not cross, the lower energy Ω = 1/2 state has Ã2Σ1/2+ character at CN = 1.132 Å, the higher energy Ω = 1/2 state has Ã2Σ1/2+ character at CN = 1.138 Å, and the electronic character of both states is mixed at CN = 1.1355 Å as visualised by the changing colour in Fig. 4b. Although this avoided crossing is close to the minimum of the Ã2Σ1/2+ state, there are no apparent effects from this avoided crossing causing distortions of the experimental spectrum. It is possible that this is because the Ã2Σ+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π origin transition we report here is actually below the energy of the avoided crossing. We also note that this crossing is avoided while the molecule is linear (as assumed in Fig. 4), but these states may cross if the bending motion is considered. Future studies could target transitions to vibrationally excited levels of the à state, which are probably more dramatically perturbed by this avoided crossing. Indeed, recent work by Jusko and coworkers investigates a transition to an excited vibrational level of the à electronic state.45
2Π origin transition we report here is actually below the energy of the avoided crossing. We also note that this crossing is avoided while the molecule is linear (as assumed in Fig. 4), but these states may cross if the bending motion is considered. Future studies could target transitions to vibrationally excited levels of the à state, which are probably more dramatically perturbed by this avoided crossing. Indeed, recent work by Jusko and coworkers investigates a transition to an excited vibrational level of the à electronic state.45
Hyperfine splitting
At low temperature (4 K and 9 K), the Doppler width of the lines is small enough that hyperfine splitting is observed (see Fig. 5). This splitting is on the order of 0.004 cm−1 and splits most lines into three observable components. To simulate the spectrum including hyperfine splitting, Ĥhfs was added to the Hamiltonian of eqn (1) as implemented in PGOPHER:44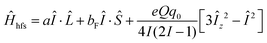 | (2) |
The experimental electronic spectrum was then refit including hyperfine coupling in PGOPHER.44 This fit included lines from the pure rotational spectrum in ref. 5. This was done because fewer hyperfine constants were required to understand the hyperfine splitting for the electronic spectrum in this work than for the pure rotational spectrum in ref. 5, and this provided a good constraint of the ground state constants. This resulted in a fit with an average residual of less than 0.001 cm−1 and is referred to here as “fit 2” in Table 2. The magnitude of the hyperfine splitting arises mostly because of the isotropic interaction between the nuclear spin of the N atom and the electron spin in the à state, which is quantified by the Fermi-contact parameter bF. This large change in bF—five times smaller in the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) state than the à state—reflects the dramatic change in the electronic structure from
state than the à state—reflects the dramatic change in the electronic structure from ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) to Ã. Therefore, the surprising fact that hyperfine splitting is resolved in these infrared spectra is definitive evidence that this is an electronic transition.
to Ã. Therefore, the surprising fact that hyperfine splitting is resolved in these infrared spectra is definitive evidence that this is an electronic transition.
![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π3/2 spectrum (cm−1). When sufficiently resolved, lines from a 35 K spectrum of the Ã2Σ+ ←
2Π3/2 spectrum (cm−1). When sufficiently resolved, lines from a 35 K spectrum of the Ã2Σ+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π1/2 transition and high J lines of the Ã2Σ+ ←
2Π1/2 transition and high J lines of the Ã2Σ+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π3/2 transition were also included. Non-hyperfine constants from fit 2 are given in Table 1
2Π3/2 transition were also included. Non-hyperfine constants from fit 2 are given in Table 1
![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π 2Π |
![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π 2Π |
Ã2Σ+ | |
|---|---|---|---|
| Fit 2 | Ref. 5 | Fit 2 | |
| a(N) | 0.0013(2) | 0.0013(1) | |
| b F(N) | 0.0012(3) | 0.0012(2) | 0.0059(1) |
| d(N) | 0.0019(1) | ||
| a(H) | 0.0013(4) | 0.00013(3) | |
| b F(H) | −0.00192(7) | −0.0017(1) | 0.0002(3) |
| eQq 0 | −0.00018(8) | −0.00019(4) | −0.0005(40) |
| eQq 2 | −0.0005(2) |
The Fermi contact parameter (bF = 0.0059(1) cm−1) of the N atom in the à state provides a direct way of quantifying the electronic wavefunctions in the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) and à states. Comparing the bF parameter in the à state of HCN+ with the borb of an isolated nitrogen (0.0514 cm−1)12 atom gives
and à states. Comparing the bF parameter in the à state of HCN+ with the borb of an isolated nitrogen (0.0514 cm−1)12 atom gives 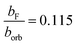 , which means that the à state has only 11.5 ± 0.1% s orbital character on the nitrogen. This is small compared to other similar nitrogen containing neutral molecules in 2Σ+ states.47,48 The relatively low s orbital character is because the singly occupied molecular orbital resembles mostly a pz atomic orbital on the nitrogen atom (see Fig. 1).
, which means that the à state has only 11.5 ± 0.1% s orbital character on the nitrogen. This is small compared to other similar nitrogen containing neutral molecules in 2Σ+ states.47,48 The relatively low s orbital character is because the singly occupied molecular orbital resembles mostly a pz atomic orbital on the nitrogen atom (see Fig. 1).
Another possible effect that could be lowering the percentage of s orbital character in the ÃΣ1/2+ state could be mixing with the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) Π1/2 state as a result of the avoided crossing in Fig. 4. This would result in the upper state having a linear combination of the Σ1/2+ and Π1/2 state configurations. Because the bF is five times lower in the Π state (see Table 2), a greater mixing between the Π and Σ configurations will reduce the bF value in the à state. However, this is quite speculative, and the subtle properties of avoided crossings are challenging to calculate.
Π1/2 state as a result of the avoided crossing in Fig. 4. This would result in the upper state having a linear combination of the Σ1/2+ and Π1/2 state configurations. Because the bF is five times lower in the Π state (see Table 2), a greater mixing between the Π and Σ configurations will reduce the bF value in the à state. However, this is quite speculative, and the subtle properties of avoided crossings are challenging to calculate.
We have also calculated the HCN+ nitrogen bF parameter of the à state using the equation of motion coupled cluster method EOM-CCSD/cc-pVTZ in the Gaussian/16 program.40,49–51 This yielded an à state value of bF = 0.014 cm−1, which is significantly larger than the experimental value (bF = 0.0059(1) cm−1). This could be a further indication of the importance of vibronic coupling for HCN+ and suggests that the à state is not well described within the Born–Oppenheimer approximation.
There are very few polyatomic molecular ions for which the hyperfine structure is observed in excited electronic states. Serendipitously, one of these states is the Ã2Σ+ state of HCP+. Because phosphorus is directly below nitrogen in the periodic table, the Ã2Σ+ state of HCP+ are analogous to the Ã2Σ+ state of HCN+ investigated here. The Ã2Σ+ state of HCP+ have a Fermi contact parameter of bF = 0.106(5) cm−1, which is over an order of magnitude larger than the value we measure for HCN+ (bF = 0.0059(1) cm−1). Additionally, the HCP+ Ã2Σ+ state wavefunction has an s orbital character of 24%, which is significantly larger than that of HCN+ (11.5 ± 0.1%). This might reflect a larger mixing of the phosphorous pz and s orbitals in the highest occupied molecular σ orbital of HCP+ or be the result of the HCN+ avoided crossing discussed above.
A contour fit of the experimental lines in Fig. 5 was attempted to capture some of the unresolved hyperfine structure from the hydrogen nucleus; however, no significant information about the hydrogen hyperfine splitting could be observed. Nevertheless, the contour fit was used to estimate the rotational and translational ion temperature. The rotational temperature was estimated to be 18 ± 5 K based on the relative intensities of the bands in Fig. 5, which arise from the differences in the population of the ground state J = 1.5, J = 3.5, and J = 4.5 levels. The fitted Gaussian width was 0.0020(5) cm−1, which corresponds to a translational temperature of 20 ± 5 K. As expected, these values are higher than the temperature of the trap itself (≈9 K) because of RF heating.52 Additionally, no Lorentzian lineshape was convoluted to account for lifetime broadening, which would further decrease the fitted Gaussian linewidth. The reason we did not include a Lorentzian in the fit is because there is considerable uncertainty in the splitting due to IH, which can lead to dubious results if one attempts to interpret subtle changes in the widths of these lines. It is clear from the narrow linewidths and the fact that the individual hyperfine lines have a similar width to the rovibrational lines that the excited state has a fairly long lifetime and, therefore, the low lying avoided crossing (Fig. 4) does not lead to fast internal conversion to the ground state. Assuming a lifetime broadening of 0.0025 cm−1 yields an extremely conservative lower limit for the excited state lifetime of 2 ns.
Π[ν3(σ) + 2ν2(π)] combination band
Although the focus of this article is the à ←![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) transition of HCN+, we observed an additional vibrational transition that partially overlaps with the à ←
transition of HCN+, we observed an additional vibrational transition that partially overlaps with the à ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) band, which warrants further discussion. This vibrational transition is centered around 3295 cm−1 and was fit with the same Hamiltonian form as described in ref. 4 (red sticks in Fig. 6). The P, Q, and R branches are clearly apparent and feature Λ type doublets characteristic of a Π ← Π transition. We were unable to assign any lines associated with the Π1/2 ← ΠΩ components. Nevertheless, the P, Q, and R branches can only be reasonably fit to a Π ← Π transition—rather than a Δ ← Π or Σ ← Π transition—and so the upper state must have Π vibronic symmetry.
band, which warrants further discussion. This vibrational transition is centered around 3295 cm−1 and was fit with the same Hamiltonian form as described in ref. 4 (red sticks in Fig. 6). The P, Q, and R branches are clearly apparent and feature Λ type doublets characteristic of a Π ← Π transition. We were unable to assign any lines associated with the Π1/2 ← ΠΩ components. Nevertheless, the P, Q, and R branches can only be reasonably fit to a Π ← Π transition—rather than a Δ ← Π or Σ ← Π transition—and so the upper state must have Π vibronic symmetry.
 | ||
Fig. 6 (a) Portion of the HCN+ Ã2Σ+ ← ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π electronic spectrum and rovibrational spectrum exciting the ν3(σ) + 2ν3(π) combination band in the 2Π electronic spectrum and rovibrational spectrum exciting the ν3(σ) + 2ν3(π) combination band in the ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) ground state (black trace) measured using LOS at ≈35 K. The experimental spectrum is compared with a simulation of the electronic transition (grey lines) and the vibronic transition (red lines) using the PGOPHER program. The vibrational levels are labelled by (v1,v2,v3), which are the vibrational quantum numbers of the CH stretch (v1), the HCN bend (v2), and the CN stretch (v3). The quantum number κ indicates that this band is the upper Renner–Teller component for this vibration (see the term diagram in Fig. 7). (b) Example of a Λ doublet in the ν3(σ) + 2ν3(π) combination band spectrum. ground state (black trace) measured using LOS at ≈35 K. The experimental spectrum is compared with a simulation of the electronic transition (grey lines) and the vibronic transition (red lines) using the PGOPHER program. The vibrational levels are labelled by (v1,v2,v3), which are the vibrational quantum numbers of the CH stretch (v1), the HCN bend (v2), and the CN stretch (v3). The quantum number κ indicates that this band is the upper Renner–Teller component for this vibration (see the term diagram in Fig. 7). (b) Example of a Λ doublet in the ν3(σ) + 2ν3(π) combination band spectrum. | ||
The vibronic symmetry of HCN+ energy levels requires consideration of the Renner–Teller effect. Fig. 7 illustrates the states generated by Renner–Teller coupling in HCN+. The vibrational angular momentum from the ν2(π) bending mode (with quantum number l = v2, v2 − 2, …0 or 1) couples with the total electronic angular momentum (with quantum number Ω) to form the resulting vibronic angular momentum P = |±Ω ± l|. The quantum number K = |±Λ ± l| results from coupling between vibrational and orbital angular momenta. The vibronic term symbols are then 2S+1KP. One quantum of the ν2(π) mode (l = 1) couples with the orbital angular momentum (Λ = 1) to generate two Σ and two Δ vibronic states. Two quanta of the ν2(π) mode (l = 2 and 0) couples with the orbital angular momentum Λ = 1 to generate four Π and two Φ vibronic states. The upper components are labeled κ and the lower components are labeled μ.
The extracted spectroscopic constants from our fit (Table 1) agree well with the calculated values from ref. 11 for the upper (κ, Π3/2) Renner–Teller component of the ν3(σ) + 2ν2(π) combination band, including the surprisingly low spin–orbit coupling constant (ASO = −4.91(1) cm−1). However, the ASO value here only provides an effective fit, because the splitting of the Πp=1/2 and Πp=3/2 vibronic components will need to be treated different from the splitting of ΠΩ=1/2 and ΠΩ=3/2 spin–orbit components. This is consistent with the fact that transitions to Πp=1/2 levels were not observed. Considering the good agreement with calculations, we assign this band as excitation of the upper κ, Π Renner–Teller component of the ν3(σ) + 2ν2(π) combination band (Π(0, 2, 1)κ) as listed—but not analysed—in part I of this series of publications.4
The fact that this combination band is ν3(σ) + 2ν2(π) with Π symmetry validates the previous assignment in ref. 4 that a band with Σ vibronic symmetry corresponds to the lower (μ, Σ) Renner–Teller component of a ν1(σ) + ν2(π) combination band. If the Σ symmetry ν3(σ) + ν2(π) combination band from ref. 4 arose from the upper (κ) Renner–Teller component, then one would expect to observe Δ ← Π transitions slightly lower in energy. Because no such transitions are observed, the Σ symmetry ν3(σ) + ν2(π) combination band observed in ref. 4 can be assigned to the lower (μ) Renner–Teller component.
A thorough analysis of the Renner–Teller coupling of HCN+ is not within the scope of this work. Nevertheless, our results provide a promising indication that the calculations in ref. 11 are accurately capturing the strong Renner–Teller coupling of HCN+.
The Π(0, 2, 1)κ band reported here is the first experimental spectrum of a κ Renner–Teller component of HCN+. The Π(0, 2, 1)μ component has been measured at 2523 cm−1 using ZEKE spectroscopy in ref. 6. These experimental values enable a preliminary estimate of Renner–Teller parameters of HCN+ based on only experimental values. We employed the equations describing a Renner–Teller and spin–orbit active system given in ref. 53, the fundamental vibrational frequency of the CN stretching mode (0, 0, 1) measured at 1785 cm−1 by Wiedmann et al., and the spin–orbit constant for HCN+ determined with our high-resolution spectra (ASO = −49.3113 cm−1). From these results, we estimate an effective Renner–Teller vibrational constant of ω = 580 cm−1 and a splitting parameter of ε = 0.45. However, because of anharmonicity, these values differ from those required to fit the fundamental bending mode μ (292 cm−1) and κ components (821 cm−1).6,11
Future high-resolution experimental studies could provide a detailed characterisation of the Renner–Teller coupling of HCN+ by combining our results with measurements of the Δ(1, 1, 0) components and the Σ(1, 1, 0)κ component (calculated to occur at 3881 cm−1) as well as the Π(0, 2, 1)μ combination band component (measured at 2523 cm−1 using ZEKE).6 The Π(0, 2, 1)κ component in Fig. 6 is the first measured κ Renner–Teller component for HCN+ and serves as a valuable experimental reference point for analysing the vibronic coupling of HCN+.
Conclusions and outlook
In summary, we have measured high resolution electronic spectra of the infrared Ã2Σ+ ←![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π electronic transition origin band of HCN+, which resolved the rotational, fine, and hyperfine structure. Although both states have been observed before, the transition between these states has never been reported, and the spectra presented here have a significantly higher resolution. Furthermore, hyperfine splitting has not been observed before and provides a lens with which the electronic wavefunction can be interrogated. In this case, we find that the à state electronic wavefunction has 11.5(2)% s orbital character on the N atom. The fact that we can observe hyperfine splitting in the infrared is unusual and makes it clear that the observed spectrum corresponds to an electronic transition.
2Π electronic transition origin band of HCN+, which resolved the rotational, fine, and hyperfine structure. Although both states have been observed before, the transition between these states has never been reported, and the spectra presented here have a significantly higher resolution. Furthermore, hyperfine splitting has not been observed before and provides a lens with which the electronic wavefunction can be interrogated. In this case, we find that the à state electronic wavefunction has 11.5(2)% s orbital character on the N atom. The fact that we can observe hyperfine splitting in the infrared is unusual and makes it clear that the observed spectrum corresponds to an electronic transition.
The quality of these spectroscopic constants easily affords accurate simulations of the rovibronic à → ![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) emission of HCN+ for comparison with astronomical measurements such as those possible with the James Webb Space Telescope.54,55 However, the fact that the electronic transition appears to have comparable intensity to vibrational excitation of a nearby ν3(σ) + 2ν2(π) combination band indicates that the electronic transition is fairly weak and will probably be challenging to detect astronomically.
emission of HCN+ for comparison with astronomical measurements such as those possible with the James Webb Space Telescope.54,55 However, the fact that the electronic transition appears to have comparable intensity to vibrational excitation of a nearby ν3(σ) + 2ν2(π) combination band indicates that the electronic transition is fairly weak and will probably be challenging to detect astronomically.
We have recently reported electronic spectra measured using LOS with a low resolution white light fibre laser.34 The electronic spectrum of HCN+ presented here illustrate that electronic transitions can be measured using LOS with a very high resolution for a mass-selected, highly reactive bare ion that is excited well below the dissociation threshold.
Conflicts of interest
There are no conflicts to declare.Data availability
The datasets generated and/or analysed during the current study are available from the corresponding authors upon reasonable request.All data supporting the findings of this study are available within the manuscript and its supplementary information (SI). Supplementary information: rovibrational scheme showing allowed rovibronic transitions and branches, the list of assigned rovibronic lines including hyperfine splitting, the list of assigned rovibronic lines from global fit without hyperfine splitting, and raw leak-out spectroscopy data for the electronic transition. See DOI: https://doi.org/10.1039/d5cp04255k.
Acknowledgements
This work was supported by an ERC advanced grant (MissIons: 101020583), and the Deutsche Forschungsgemeinschaft (DFG) via Collaborative Research Center 1601 (project ID 500700252, sub-project B8 & C4) and DFG project 514067452, INST 216/1069-1 (450096019). The optical cw-OPOs were financed by DFG (INST216/1184-1, project number 504504934 and INST216/1069-1, project number 450096019). WGDPS acknowledges the Alexander von Humboldt Foundation for support through a postdoctoral fellowship.References
- S. Petrie, C. G. Freeman, M. Meot-Ner, M. J. McEwan and E. E. Ferguson, Experimental study of HCN+ and HNC+ ion chemistry, J. Am. Chem. Soc., 1990, 112, 7121–7126 CrossRef CAS.
- P. Dohnal, P. Jusko, M. Jiménez-Redondo and P. Caselli, Measurements of rate coefficients of CN+, HCN+, and HNC+ collisions with H2 at cryogenic temperatures, J. Chem. Phys., 2023, 158(24), 244303 CrossRef CAS PubMed.
- D. Forney, W. E. Thompson and M. E. Jacox, The vibrational spectra of molecular ions isolated in solid neon. IX. HCN+, HNC+, and CN−, J. Chem. Phys., 1992, 97, 1664–1674 CrossRef CAS.
- P. C. Schmid, S. J. P. Marlton, W. G. D. P. Silva, T. Salomon, O. Asvany and S. Schlemmer, High-resolution spectroscopy of [H,C,N]+: I. Rotationally resolved vibrational bands of HCN+ and HNC+, Phys. Chem. Chem. Phys., 2026 10.1039/D5CP04201A.
- W. G. D. P. Silva, P. C. Schmid, D. Gupta, S. Thorwirth, O. Asvany and S. Schlemmer, High-resolution spectroscopy of [H,C,N]+: II. Ground state rotational spectrum of HCN+ (
![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π), Phys. Chem. Chem. Phys., 2026 10.1039/D5CP04204F.
2Π), Phys. Chem. Chem. Phys., 2026 10.1039/D5CP04204F. - R. T. Wiedmann and M. G. White, Vibronic coupling in the
![[X with combining tilde]](https://www.rsc.org/images/entities/char_0058_0303.gif) 2Π and Ã2Σ+ states of HCN+, J. Chem. Phys., 1995, 102, 5141–5151 CrossRef CAS.
2Π and Ã2Σ+ states of HCN+, J. Chem. Phys., 1995, 102, 5141–5151 CrossRef CAS. - A. Lorquet, J.-C. Lorquet, J. Delwiche and M.-J. Hubin-Franskin, Intramolecular dynamics by photoelectron spectroscopy. I. Application to N+2, HBr+, and HCN+, J. Chem. Phys., 1982, 76, 4692–4699 CrossRef CAS.
- C. Fridh and L. Åsbrink, Photoelectron and electron impact spectrum of HCN, J. Electron Spectrosc. Relat. Phenom., 1975, 7, 119–138 CrossRef CAS.
- J. Hollas and T. Sutherley, Geometry of C2N2+ and HCN+ from low-energy photoelectron spectroscopy, Mol. Phys., 1972, 24, 1123–1131 CrossRef CAS.
- J. Eland, T. Field, P. Baltzer and D. Hirst, Photoelectron spectra, electronic structure, coincidence spectra and dissociation mechanisms of the hydrogen cyanide cation, Chem. Phys., 1998, 229, 149–163 CrossRef CAS.
- R. Tarroni, A. Mitrushenkov, P. Palmieri and S. Carter, Energy levels of HCN+ and DCN+ in the vibronically coupled X2Π and A2Σ+ states, J. Chem. Phys., 2001, 115, 11200–11212 CrossRef CAS.
- J. A. Fitzpatrick, F. R. Manby and C. M. Western, The interpretation of molecular magnetic hyperfine interactions, J. Chem. Phys., 2005, 122(8), 084312 CrossRef PubMed.
- M. Nötzold, R. Wild, C. Lochmann and R. Wester, Spectroscopy and ion thermometry of C 2- using laser-cooling transitions, Phys. Rev. A, 2022, 106, 023111 CrossRef.
- I. Dabrowski and G. Herzberg, The spectrum of HeNe+, J. Mol. Spectrosc., 1978, 73, 183–214 CrossRef CAS.
- A. Carrington, D. R. Milverton and P. J. Sarre, Electronic absorption spectrum of CO+ in an ion beam: Analysis of the carbon nuclear hyperfine structure in 13CO+, Mol. Phys., 1978, 35, 1505–1521 CrossRef CAS.
- P. J. Sarre, J. M. Walmsley and C. J. Whitham, High-resolution laser photofragment spectroscopy of CH+, J. Chem. Soc., Faraday Trans. 2, 1986, 82, 1243–1255 Search PubMed.
- P. Sarre, J. Walmsley and C. Whitham, Laser photofragment spectroscopy of near-threshold resonances in SiH+, Philos. Trans. R. Soc., A, 1988, 324, 233–246 CrossRef CAS.
- C. Edwards, C. Maclean and P. Sarre, Laser predissociation spectrum of SH+ (A3Π–X3Σ−) Analysis of proton hyperfine structure, Mol. Phys., 1984, 52, 1453–1466 CrossRef CAS.
- C. Edwards, P. Jackson, P. Sarre and D. Milton, Laser photofragment and emission spectroscopy of the A2Δ–X2Π system of PH+, Mol. Phys., 1986, 57, 595–604 CrossRef CAS.
- P. Cosby, H. Helm and M. Larzilliere, Photofragment spectroscopy of HF+, J. Chem. Phys., 1991, 94, 92–105 CrossRef CAS.
- K. Boudjarane, J. Lacoursière and M. Larzillière, Hyperfine structure of first negative system (B2Σu+–X2Σg+) of 14N2+ and 15N2+ from laser-induced fluorescence measurements, J. Chem. Phys., 1994, 101, 10274–10282 CrossRef CAS.
- M. Larzillière, S. Abed, M. Carré, M. Gaillard, J. Lermé and M. Broyer, l-Type doubling hyperfine structure of N2O+, Chem. Phys. Lett., 1985, 119, 55–60 CrossRef.
- F. X. Sunahori, X. Zhang and D. J. Clouthier, Electronic spectroscopy of jet-cooled HCP+: Molecular structure, phosphorus hyperfine structure, and Renner-Teller analysis, J. Chem. Phys., 2007, 127(10), 104312 CrossRef PubMed.
- H.-W. Hübers, K. M. Evenson, C. Hill and J. M. Brown, The rotational spectrum of the NH+ radical in its X2Π and a4Σ− states, J. Chem. Phys., 2009, 131(3), 034311 CrossRef PubMed.
- G. A. Pino, G. Feraud, M. Broquier, G. Grégoire, S. Soorkia, C. Dedonder and C. Jouvet, Non-radiative processes in protonated diazines, pyrimidine bases and an aromatic azine, Phys. Chem. Chem. Phys., 2016, 18, 20126–20134 RSC.
- D. Bomse, R. Woodin and J. Beauchamp, Molecular activation with low-intensity CW infrared laser radiation. Multiphoton dissociation of ions derived from diethyl ether, J. Am. Chem. Soc., 1979, 101, 5503–5512 CrossRef CAS.
- Y. Zhao, E. de Beer, C. Xu, T. Taylor and D. M. Neumark, Spectroscopy and electron detachment dynamics of C4−, C6−, and C8−, J. Chem. Phys., 1996, 105, 4905–4919 CrossRef CAS.
- T. Salomon, S. Brackertz, O. Asvany, I. Savić, D. Gerlich, M. E. Harding, F. Lipparini, J. Gauss, A. van der Avoird and S. Schlemmer, The He-H3+ complex. II. Infrared predissociation spectrum and energy term diagram, J. Chem. Phys., 2022, 156(14), 144308 CrossRef CAS PubMed.
- S. J. Marlton, J. T. Buntine, P. Watkins, C. Liu, U. Jacovella, E. Carrascosa, J. N. Bull and E. J. Bieske, Probing colossal carbon rings, J. Phys. Chem. A, 2023, 127, 1168–1178 CrossRef CAS PubMed.
- T. E. Douglas-Walker, E. K. Campbell, F. C. Daly, S. Douin, B. Gans, U. Jacovella, C. Maurice, R. Odant and J. Palotas, Ion Spectroscopy in the Context of the Diffuse Interstellar Bands: A Case Study with the Phenylacetylene Cation, ACS Earth Space Chem., 2024, 8(12), 2644–2651 CrossRef CAS PubMed.
- S. Schlemmer, T. Kuhn, E. Lescop and D. Gerlich, Laser excited N2+ in a 22-pole ion trap:: Experimental studies of rotational relaxation processes, Int. J. Mass Spectrom., 1999, 185, 589–602 Search PubMed.
- S. Chakrabarty, M. Holz, E. K. Campbell, A. Banerjee, D. Gerlich and J. P. Maier, A novel method to measure electronic spectra of cold molecular ions, J. Phys. Chem. Lett., 2013, 4, 4051–4054 CrossRef CAS.
- P. C. Schmid, O. Asvany, T. Salomon, S. Thorwirth and S. Schlemmer, Leak-out spectroscopy, a universal method of action spectroscopy in cold ion traps, J. Phys. Chem. A, 2022, 126, 8111–8117 CrossRef CAS PubMed.
- S. J. Marlton, P. C. Schmid, T. Salomon, O. Asvany and S. Schlemmer, Measuring Electronic Transitions Using Leak-Out Spectroscopy, J. Phys. Chem. Lett., 2025, 16, 8758–8763 Search PubMed.
- S. Schlemmer, E. Lescop, J. von Richthofen, D. Gerlich and M. A. Smith, Laser induced reactions in a 22-pole ion trap: C2H2+ + hν 3+ H2 → C2H3+ + H, J. Chem. Phys., 2002, 117, 2068–2075 Search PubMed.
- O. Asvany, F. Bielau, D. Moratschke, J. Krause and S. Schlemmer, Note: New design of a cryogenic linear radio frequency multipole trap, Rev. Sci. Instrum., 2010, 81(7), 076102 CrossRef PubMed.
- N. Daly, Scintillation type mass spectrometer ion detector, Rev. Sci. Instrum., 1960, 31, 264–267 CrossRef CAS.
- O. Asvany, S. Brünken, L. Kluge and S. Schlemmer, COLTRAP: a 22-pole ion trapping machine for spectroscopy at 4 K, Appl. Phys. B:Lasers Opt., 2014, 114, 203–211 Search PubMed.
- K. Andersson, P. A. Malmqvist, B. O. Roos, A. J. Sadlej and K. Wolinski, Second-order perturbation theory with a CASSCF reference function, J. Phys. Chem., 1990, 94, 5483–5488 CrossRef CAS.
- T. H. Dunning Jr, Gaussian basis sets for use in correlated molecular calculations. I. The atoms boron through neon and hydrogen, J. Chem. Phys., 1989, 90, 1007–1023 CrossRef.
- I. Fdez Galvan, M. Vacher, A. Alavi, C. Angeli, F. Aquilante, J. Autschbach, J. J. Bao, S. I. Bokarev, N. A. Bogdanov and R. K. Carlson, et al., OpenMolcas: From source code to insight, J. Chem. Theory Comput., 2019, 15, 5925–5964 Search PubMed.
- P. J. Knowles and H.-J. Werner, An efficient method for the evaluation of coupling coefficients in configuration interaction calculations, Chem. Phys. Lett., 1988, 145, 514–522 Search PubMed.
- F. Neese, Software update: The ORCA program system–Version 5.0, Wiley Interdiscip. Rev.:Comput. Mol. Sci., 2022, 12, e1606 Search PubMed.
- C. M. Western, PGOPHER: A program for simulating rotational, vibrational and electronic spectra, J. Quant. Spectrosc. Radiat. Transfer, 2017, 186, 221–242 Search PubMed.
- M. Jiménez-Redondo; C. Schleif; J. Palotás; J. Sarka; H. Bunn; P. Dohnal; C. Paola and P. Jusko, High Resolution Overtone Spectroscopy of HCN+ and HNC+. Submitted, 2025 Search PubMed.
- R. A. Frosch and H. Foley, Magnetic hyperfine structure in diatomic molecules, Phys. Rev., 1952, 88, 1337 Search PubMed.
- T. Suzuki, S. Saito and E. Hirota, Hyperfine coupling constants of NCO in Ã2Σ+ by sub-Doppler spectroscopy, J. Mol. Spectrosc., 1986, 120, 414–420 CrossRef CAS.
- H. Radford, Hyperfine Structure of the B2Σ+ State of CN, Phys. Rev., 1964, 136, A1571 Search PubMed.
- M. J. Frisch; G. W. Trucks; H. B. Schlegel; G. E. Scuseria; M. A. Robb; J. R. Cheeseman; G. Scalmani; V. Barone; G. A. Petersson; H. Nakatsuji; et al.Gaussian 16 Revision C.01., 2016, Gaussian Inc., Wallingford CT, 2009 Search PubMed.
- H. Koch and P. Jørgensen, Coupled cluster response functions, J. Chem. Phys., 1990, 93, 3333–3344 CrossRef CAS.
- J. F. Stanton and R. J. Bartlett, The equation of motion coupled-cluster method. A systematic biorthogonal approach to molecular excitation energies, transition probabilities, and excited state properties, J. Chem. Phys., 1993, 98, 7029–7039 CrossRef CAS.
- O. Asvany and S. Schlemmer, Numerical simulations of kinetic ion temperature in a cryogenic linear multipole trap, Int. J. Mass Spectrom., 2009, 279, 147–155 Search PubMed.
- G. Herzberg, Molecular Spectra and molecular structure-Vol III, Read Books Ltd, 2013, vol. 1 Search PubMed.
- P. B. Changala, N. L. Chen, H. L. Le, B. Gans, K. Steenbakkers, T. Salomon, L. Bonah, I. Schroetter, A. Canin and M.-A. Martin-Drumel, et al., Astronomical CH3+ rovibrational assignments-A combined theoretical and experimental study validating observational findings in the d203-506 UV-irradiated protoplanetary disk, Astron. Astrophys., 2023, 680, A19 Search PubMed.
- O. Berné, M.-A. Martin-Drumel, I. Schroetter, J. R. Goicoechea, U. Jacovella, B. Gans, E. Dartois, L. H. Coudert, E. Bergin and F. Alarcon, et al., Formation of the methyl cation by photochemistry in a protoplanetary disk, Nature, 2023, 621, 56–59 CrossRef PubMed.
| This journal is © the Owner Societies 2026 |





