Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
A comparative study of the lattice dynamics of alkali titanium hexahalides
Junbiao
Guo
 a,
Keith
Refson
a,
Keith
Refson
 b and
Martin T.
Dove
b and
Martin T.
Dove
 *acd
*acd
aInstitute of Atomic and Molecular Physics, Sichuan University, Chengdu, Sichuan 610065, China. E-mail: martin.dove@icloud.com
bISIS Facility, Harwell Campus, Chilton, Didcot, OX11 0QX, UK
cSchool of Mechanical Engineering, Guizhou University of Engineering Science, Xueyan Road, Bijie, Guizhou 55170, China
dSchool of Physical and Chemical Sciences, Queen Mary University of London, Mile End Road, London, E1 4NS, UK
First published on 20th February 2026
Abstract
Lattice dynamics calculations have been performed on a family of vacancy double perovskites of general formula A2TiX6 (A = Rb, Cs, X = F, Cl, Br, I) using density functional theory. This family has attracted interest as potential lead-free optoelectronic materials with tuneable band gaps. We find that most of these materials show stable lattice dynamics, but two, Rb2TiF6 and Rb2TiI6, show instabilities which would lead to a phase transition. In fact Rb2TiF6 normally crystallises in a phase with a different structure, which can be understood on the basis of calculations of the free energy.
1 Introduction
Among the materials used in solar cells, lead-based perovskites show relatively high efficiencies. However, these are not suitable for large-scale applications due to their toxicity and instability.2,3 One approach is to replace lead with tin or germanium, elements from the same group in the periodic table. But they too are chemically unstable, because the Sn2+ + and Ge2+ ions are easily oxidised to Sn4+ and Ge4+. Furthermore, tin is also harmful to the human body.4,5Currently, there is a concentrated search for lead-free perovskites or perovskite derivatives as alternatives. One potential family of materials is the so-called vacancy double perovskites, with chemical formula of A2BX6 (A = K, Rb, Cs, etc., B = Se, Ti, Pd, Mn, etc., X = F, Cl, Br or I). The introduction of halide anions can adjust the band gap by altering the electronic band structure of the material, thereby influencing its optoelectronic properties.6–11 This structure is similar to the normal double perovskite, of general fomula A2B′B″X6, where the B″ cations are replaced by vacancies. These structures have the cubic space group Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m.12 The group IV element titanium exists in a stable tetravalent state in these materials, it has a good biocompatibility, and it is abundantly available. The A2TiX6 vacancy double perovskites hold promise as substitutes for traditional APbX3 perovskites, while maintaining a high conversion efficiency.13,14 The crystal structure of the vacancy double perovskites A2TiX6 with the space group of Fm
m.12 The group IV element titanium exists in a stable tetravalent state in these materials, it has a good biocompatibility, and it is abundantly available. The A2TiX6 vacancy double perovskites hold promise as substitutes for traditional APbX3 perovskites, while maintaining a high conversion efficiency.13,14 The crystal structure of the vacancy double perovskites A2TiX6 with the space group of Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m is shown in Fig. 1. The structure is composed of unconnected TiX6 octahedra on the lattice sites, with A atoms filling the spaces between the octahedra. The point half way between lattice sites along the unit cell edges, a vacant site with halide neighbours, is the site that would contain a cation in the standard perovskite or double perovskite structures.
m is shown in Fig. 1. The structure is composed of unconnected TiX6 octahedra on the lattice sites, with A atoms filling the spaces between the octahedra. The point half way between lattice sites along the unit cell edges, a vacant site with halide neighbours, is the site that would contain a cation in the standard perovskite or double perovskite structures.
 | ||
| Fig. 1 Crystal structure of A2TiX6 (A = Rb, Cs, X = F, Cl, Br, I). The big pink spheres and small green ones denote A and X atoms, respectively. The blue octahedra combined with green spheres represents TiX6. The figure was drawn using CrystalMaker.1 | ||
A number of recent papers have reported on the experimental synthesis of various A2TiX6 crystals, and others have reported density functional theory (DFT) calculations.20–24 The experimental studies have shown that not all elemental combinations can be synthesised, and in some cases the compounds crystallise in a different space group, such as the trigonal P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1.25,26 Most DFT calculations tend to focus on electrical and optical properties, and calculations of lattice vibrations have broadly been neglected. Careful lattice dynamics calculations, comparing known synthesized compounds with those that have not yet been synthesized, help to improve our understanding of stability, and can improve the efficiency of the search for new materials by evaluating the likely stability of any candidate material. Moreover, there are some compounds that can be successfully crystallised, but unfortunately their stability in the environment is questionable. For example, Cs2TiBr6 has been studied, with some research using DFT to calculate its electronic, optical, and thermal properties, indicating that this halide perovskite could be a promising optoelectronic material.27,28 But there is some debate as to whether it decomposes in standard environments.29–31 Such experiments have also been conducted for Cs2TiCl6, and this material seems much more stable than the bromide counterpart.21
m1.25,26 Most DFT calculations tend to focus on electrical and optical properties, and calculations of lattice vibrations have broadly been neglected. Careful lattice dynamics calculations, comparing known synthesized compounds with those that have not yet been synthesized, help to improve our understanding of stability, and can improve the efficiency of the search for new materials by evaluating the likely stability of any candidate material. Moreover, there are some compounds that can be successfully crystallised, but unfortunately their stability in the environment is questionable. For example, Cs2TiBr6 has been studied, with some research using DFT to calculate its electronic, optical, and thermal properties, indicating that this halide perovskite could be a promising optoelectronic material.27,28 But there is some debate as to whether it decomposes in standard environments.29–31 Such experiments have also been conducted for Cs2TiCl6, and this material seems much more stable than the bromide counterpart.21
From this discussion it would seem that there is an important role for lattice dynamics calculations in these materials. In this paper, we have selected A2TiX6 (A = Cs or Rb, X = F, Cl, Br or I) compounds for DFT calculations, focusing on the impact of lattice dynamics on the stability of these perovskite materials. We have found from our lattice dynamics calculations that while some certain compounds may potentially have exciting electrical, optical or thermoelectric properties, they are, in fact, unstable, or may experience a phase transition. Additional calculations, including the Goldschmidt tolerance factor and an analysis of possible space groups induced by soft modes, are also performed to supplement the main results.
2 Methods
The calculations were performed using the CASTEP software, with plane waves representing the electronic states.32 Norm-conserving pseudopotentials, provided by the NCP19 library in CASTEP, were used to handle the core electrons. For the simulation of A2TiX6 (A = Cs or Rb, X = F, Cl, Br or I), the PBE generalised gradient approximation (GGA) was employed.33,34 A 5 × 5 × 5 Monkhorst–Pack grid35 of wave vectors was selected, and cut-off energies ranging from 800 to 1300 eV were used for different A2TiX6 compounds based on convergence tests.For geometry optimisation, the convergence tolerance for accepting free energy, force, stress, and ionic displacement were set as 10−6 eV per atom, 5 × 10−3 eV Å−1, 10−2 GPa, and 10−4 Å, respectively.
Phonon calculations were performed using density functional perturbation theory (DFPT), and the interpolation method was applied to compute the frequency for different wave vectors within the Brillouin zone.36,37 A 5 × 5 × 5 Monkhorst–Pack grid was employed for calculating the dynamical matrix for interpolation to other wave vectors, which internally within the program sets the range of neighbouring unit cells used for the contribution of distance interactions to the dynamical matrix. The acoustic-sum-rule correction was used to ensure that the frequencies of the acoustic modes tend towards zero value for wave vectors around the Γ point in the Brillouin zone (zero wave vector).
Elastic constants were calculated by fitting the stresses calculated when a range of different strains were imposed on the relaxed crystal structure.
3 Results
3.1 Crystal structure
The calculated values of the lattice parameter a are compared with experimental data and the results of previous calculations in Table 1. Most of the calculated values match well with those of prior calculations or experimental data, with the maximum difference between calculations and experiments being 3.6%. As we will discuss later, from the lattice dynamics calculations Rb2TiF6 and Rb2TiI6 appear to be unstable in the cubic structure.| Formula | a (Å) | a (Å) | a (Å) | Ti–X (Å) | C 11 (GPa) | C 12 (GPa) | C 44 | K |
|---|---|---|---|---|---|---|---|---|
| (This study) | (Other calc.) | (Exp) | (This study) | (GPa) | (GPa) | (GPa) | (GPa) | |
| Rb2TiF6 (unstable) | 8.84 | — | — | 1.886 | 27.59 | 12.56 | 7.21 | 17.57 |
| Rb2TiCl6 | 10.26 | 10.2715 | 10.0116 | 2.362 | 22.42 | 7.38 | 5.82 | 12.39 |
| Rb2TiBr6 | 10.82 | 10.9515 | — | 2.535 | 23.35 | 5.63 | 6.15 | 11.54 |
| Rb2TiI6 (unstable) | 11.63 | 11.9517 | — | 2.774 | 18.71 | 5.84 | 4.06 | 10.13 |
| Cs2TiF6 | 9.27 | 10.109 | — | 1.889 | 24.48 | 11.98 | 6.16 | 16.15 |
| Cs2TiCl6 | 10.60 | 10.6218 | 10.23,16 10.2419 | 2.365 | 18.51 | 6.50 | 4.85 | 10.50 |
| Cs2TiBr6 | 11.10 | 11.0818 | 10.9213 | 2.538 | 17.13 | 5.50 | 4.33 | 9.38 |
| Cs2TiI6 | 11.83 | 11.8818 | 11.6713 | 2.779 | 16.30 | 4.73 | 3.98 | 8.59 |
3.2 Elasticity
For a cubic system, there are only three independent elastic constants C11, C12 and C44. Their calculated values, together with the bulk modulus K = (C11 + 2C12)/3, are given in Table 1. There is an apparent trend that the values of all coefficients systematically decrease with the increase of the masses and ionic radii of both the alkaline cation and the halide anion.3.3 Phonon dispersion curves
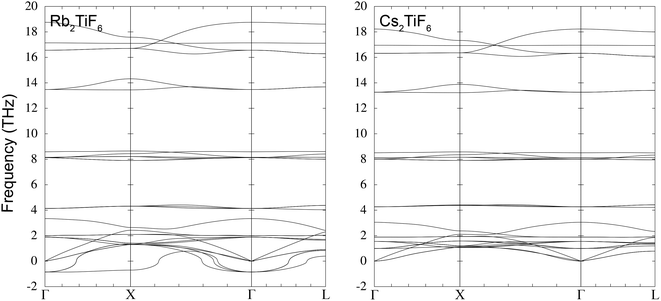
Dispersion curves of the eight compounds A2TiX6 (A = Cs, Rb, X = F, Cl, Br, I) in space group Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m are presented in pairs in Fig. 2 and Fig. S1–S3 (shown in the SI). The irreducible representations of the three acoustic modes are the triple-degenerate T1u, and the decomposition of the 24 optic modes at Γ point (zero wave vector) are the irreducible representations A1g + Eg + T2u + 2T2g + 3T1u + T1g. There is a LO/TO splitting around Γ in all of the dispersion curves, which affects the three optic T1u modes by splitting into singlet and doublet modes, 3(Eu + A2u). The dispersion curves show a number of general trends. In each case the highest six frequencies are the Ti–X stretching modes, which, considering the linear X–Ti–X linkage, are either symmetric (Eg + A1g) or asymmetric (T1u) with regard to the octahedral centre of symmetry. In each case the longitudinal asymmetric stretch mode has highest frequency. Immediately below the stretch modes are the nine modes involving bending of the bonds within the TiX6 octahedra (T2u + T2g + T1u). At yet lower frequencies are the six modes involving the displacements of the A cations and the whole-body TiX6 octahedra (T2g+ T1u). Finally, at the lowest frequencies, are the three modes involving rotations of the TiX6 octahedra (T1g), which have some interesting features we will discuss below. Representations of these mode eigenvectors are given through the example of Cs2TiI6 in Fig. S4 and the list of Γ point frequencies is in Table S1 (given in the SI).
m are presented in pairs in Fig. 2 and Fig. S1–S3 (shown in the SI). The irreducible representations of the three acoustic modes are the triple-degenerate T1u, and the decomposition of the 24 optic modes at Γ point (zero wave vector) are the irreducible representations A1g + Eg + T2u + 2T2g + 3T1u + T1g. There is a LO/TO splitting around Γ in all of the dispersion curves, which affects the three optic T1u modes by splitting into singlet and doublet modes, 3(Eu + A2u). The dispersion curves show a number of general trends. In each case the highest six frequencies are the Ti–X stretching modes, which, considering the linear X–Ti–X linkage, are either symmetric (Eg + A1g) or asymmetric (T1u) with regard to the octahedral centre of symmetry. In each case the longitudinal asymmetric stretch mode has highest frequency. Immediately below the stretch modes are the nine modes involving bending of the bonds within the TiX6 octahedra (T2u + T2g + T1u). At yet lower frequencies are the six modes involving the displacements of the A cations and the whole-body TiX6 octahedra (T2g+ T1u). Finally, at the lowest frequencies, are the three modes involving rotations of the TiX6 octahedra (T1g), which have some interesting features we will discuss below. Representations of these mode eigenvectors are given through the example of Cs2TiI6 in Fig. S4 and the list of Γ point frequencies is in Table S1 (given in the SI).
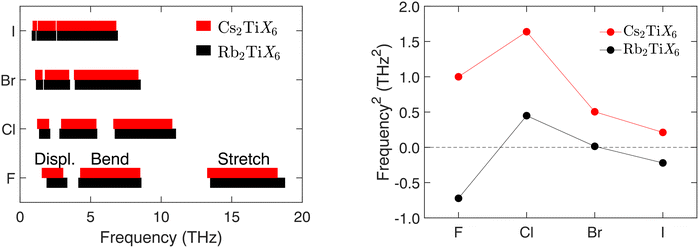
The trends in the frequencies are shown in cartoon form in Fig. 3. In every case, with increasing frequency, the groups of frequencies are the displacement, bond-bending and bond-stretching modes, with no significant overlap. The ranges of frequencies are nearly the same for A = Rb and A = Cs, which is not at all surprising for the bond-bending and bond-stretching modes because these involve only minimal or no displacements of the A cations. For the displacement modes, we might expect the mass difference (ratio of 85.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 132.9 for Rb
132.9 for Rb![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Cs) to give higher frequencies for A = Rb than for A = Cs. This is sometimes seen to be the case, as in the case X = F, but not always. The most striking visual impression from Fig. 3 is the effect from the mass of the X anion. With increasing mass the frequencies of the bond-stretching and bond-bending modes fall considerably, with an effect also on the displacement modes.
Cs) to give higher frequencies for A = Rb than for A = Cs. This is sometimes seen to be the case, as in the case X = F, but not always. The most striking visual impression from Fig. 3 is the effect from the mass of the X anion. With increasing mass the frequencies of the bond-stretching and bond-bending modes fall considerably, with an effect also on the displacement modes.
It is the low-frequency TiX6 rotational modes that do not obviously follow an exact trend with mass. From Fig. 2 and Fig. S3, Table S1, it can be seen that the rotational modes for Rb2TiF6 and Rb2TiI6 in space group Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m show instabilities, in that the frequencies have imaginary values (negative values of the squared frequencies obtained by diagonalisation of the dynamical matrix). The squared frequency values for the range of materials are plotted in Fig. 3. It can be seen that the cases A = Rb and A = Cs follow the same basic trends, with X = Cl having the highest squared frequencies in both cases, with the squared frequencies decreasing on increasing mass from Br to I. Moreover, from Table 1 we see that there is a corresponding increase in the the Ti–X bond lengths, with both effects leading to an increase in the moment of inertia of the TiX6 octahedra. The big surprise is that the case X = F breaks the trend in both cases, having much lower squared frequencies rather than an increase in value. This is not immediately easy to understand. Similarly it is not easy to understand why the frequency-squared values for Cs2TiF6 are systematically higher that for Rb2TiF6, given that the A cation does not contribute to the mode eigenvectors. With a larger unit cell, and larger A–X distances, one might have expected that the potential energy associated with rotations of the TiX6 octahedra would be seen as softer, yet the opposite appears to be the case.
m show instabilities, in that the frequencies have imaginary values (negative values of the squared frequencies obtained by diagonalisation of the dynamical matrix). The squared frequency values for the range of materials are plotted in Fig. 3. It can be seen that the cases A = Rb and A = Cs follow the same basic trends, with X = Cl having the highest squared frequencies in both cases, with the squared frequencies decreasing on increasing mass from Br to I. Moreover, from Table 1 we see that there is a corresponding increase in the the Ti–X bond lengths, with both effects leading to an increase in the moment of inertia of the TiX6 octahedra. The big surprise is that the case X = F breaks the trend in both cases, having much lower squared frequencies rather than an increase in value. This is not immediately easy to understand. Similarly it is not easy to understand why the frequency-squared values for Cs2TiF6 are systematically higher that for Rb2TiF6, given that the A cation does not contribute to the mode eigenvectors. With a larger unit cell, and larger A–X distances, one might have expected that the potential energy associated with rotations of the TiX6 octahedra would be seen as softer, yet the opposite appears to be the case.
It is interesting to note that the instabilities seen in the phonon dispersion of Rb2TiF6 and Rb2TiI6 for the rotational modes show very similar characteristics. Both show softening all across the branch from Γ–X along the [100] direction, but in the other two symmetry directions, Γ–X along the [110] direction, and Γ–L (along the [111] direction), the instabilities are confined to wave vectors close to Γ. Given that A2TiX6 is a double perovskite with one octahedral site vacant, the interesting point is that the soft [100] direction with a triple degeneracy at Γ directly reflects the low energy branches of rigid unit modes in cubic single perovskites.38–40 In fact the triple-degenerate mode splits into a singlet state which stays soft along the line, and a doublet mode that increases in frequency, as seen in perovskite. This is seen in all compounds, but as we have discussed, the degree of softening of this mode is different for each compound (Fig. 3).
It is known that the compounds Rb2TiF6 and Cs2TiF6 crystallise in a trigonal structure of space group P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1.25,26,41 This symmetry is not a subgroup of Fm
m1.25,26,41 This symmetry is not a subgroup of Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m, and therefore there will not be a displacive phase transition between the two structures. Thus the existence of the trigonal phase is not directly related to the instability seen in the rotational mode in Rb2TiF6 (and this mode is not unstable in Cs2TiF6, although it does have a low frequency). We present calculations of the phonon dispersion relations of Rb2TiF6 and Cs2TiF6 in space group P
m, and therefore there will not be a displacive phase transition between the two structures. Thus the existence of the trigonal phase is not directly related to the instability seen in the rotational mode in Rb2TiF6 (and this mode is not unstable in Cs2TiF6, although it does have a low frequency). We present calculations of the phonon dispersion relations of Rb2TiF6 and Cs2TiF6 in space group P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1. Broadly we see similar patterns of the branches if we compare the results shown in Fig. 4 with those of the cubic Fm
m1. Broadly we see similar patterns of the branches if we compare the results shown in Fig. 4 with those of the cubic Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m structure, Fig. 2. We see the same distributions of bands for the stretch, bending and displacement modes. However, it is clear that in the P
m structure, Fig. 2. We see the same distributions of bands for the stretch, bending and displacement modes. However, it is clear that in the P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1 structure the rotational mode is quite stable, and in Cs2TiF6 it has a higher frequency than in the cubic phase.
m1 structure the rotational mode is quite stable, and in Cs2TiF6 it has a higher frequency than in the cubic phase.
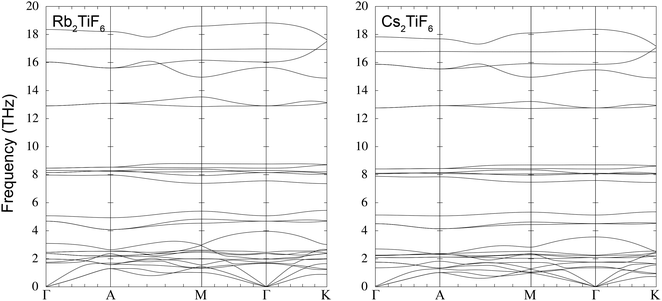 | ||
Fig. 4 Phonon dispersion curves of Rb2TiF6 (left) and Cs2TiF6 (right) in the space group of P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1. The wave vectors lables are Γ = [0, 0, 0], A = [0, 0, 1/2], M = [1/2, 0, 0] and K = [1/3, 1/3, 0]. m1. The wave vectors lables are Γ = [0, 0, 0], A = [0, 0, 1/2], M = [1/2, 0, 0] and K = [1/3, 1/3, 0]. | ||
3.4 Phonon contributions to thermodynamic properties
We now turn our attention to the question of the relative stabilities of the Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m and P
m and P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1 phases of Cs2TiF6, based on the evaluation of the overall free energy G = H − TS, where the symbols have their usual thermodynamic meaning. The phonon contribution to the free energy is obtained from the phonon frequencies using standard equations from statistical mechanics.
m1 phases of Cs2TiF6, based on the evaluation of the overall free energy G = H − TS, where the symbols have their usual thermodynamic meaning. The phonon contribution to the free energy is obtained from the phonon frequencies using standard equations from statistical mechanics.
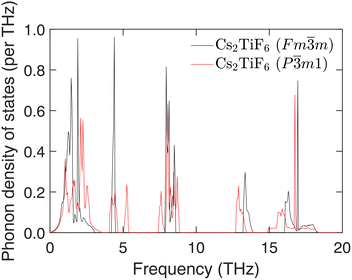
The phonon density of states has been evaluated from calculations for 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 wave vectors spread randomly throughout the whole Brillouin zone, and results are shown in Fig. 5. The consistency with the phonon dispersion curves for symmetry directions, Fig. 2, is clear, particularly with regards to the gaps around 6, 10 and 15 THz. There are some small differences between the two sets of dispersion curves, notably in the external region from 0–3 THz, and in the bending regions at around 5 THz and 7.5 THz. These may have an influence on the differences in phonon free energy.
000 wave vectors spread randomly throughout the whole Brillouin zone, and results are shown in Fig. 5. The consistency with the phonon dispersion curves for symmetry directions, Fig. 2, is clear, particularly with regards to the gaps around 6, 10 and 15 THz. There are some small differences between the two sets of dispersion curves, notably in the external region from 0–3 THz, and in the bending regions at around 5 THz and 7.5 THz. These may have an influence on the differences in phonon free energy.
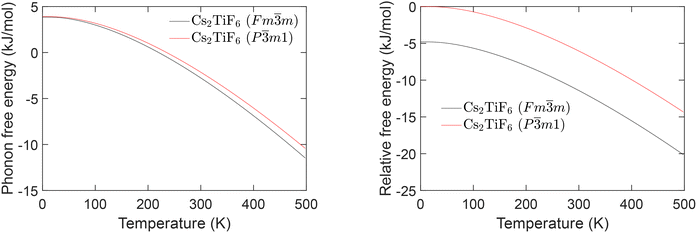
The phonon free energy of the two phases of Cs2TiF6 are compared in Fig. 6. At 0 K the phonon free energy for the Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m phase is 0.05 kJ mol−1 below that of the P
m phase is 0.05 kJ mol−1 below that of the P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1 phase, and on increasing temperature the phonon free energy of the Fm
m1 phase, and on increasing temperature the phonon free energy of the Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m falls faster. This corresponds to having a higher entropy, coming from the balance of the external phonon modes (0–3 THz) having a higher density at lower frequencies in the density of states, Fig. 5. The lattice energy itself certainly favours the Fm
m falls faster. This corresponds to having a higher entropy, coming from the balance of the external phonon modes (0–3 THz) having a higher density at lower frequencies in the density of states, Fig. 5. The lattice energy itself certainly favours the Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m phase over the P
m phase over the P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1 phase, with the former having an energy 4.747 kJ mol−1 below that of the latter. The relative free energy is also shown in Fig. 6. It is clear from this analysis that the Fm
m1 phase, with the former having an energy 4.747 kJ mol−1 below that of the latter. The relative free energy is also shown in Fig. 6. It is clear from this analysis that the Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m phase is the stable phase of Cs2TiF6 over all temperatures, leading to the conclusion that the P
m phase is the stable phase of Cs2TiF6 over all temperatures, leading to the conclusion that the P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1 phase is crystallised as a metastable state.
m1 phase is crystallised as a metastable state.
In the case of Rb2TiF6, however, because some of the phonons are unstable in the Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m phase, we cannot obtain the corresponding comparison of phonon free energy. However, because of the similarities of the general features of the phonon spectra, we might anticipate from comparison with Cs2TiF6 that at temperatures high enough to stabilise the phonons in Rb2TiF6, the entropy of the Fm
m phase, we cannot obtain the corresponding comparison of phonon free energy. However, because of the similarities of the general features of the phonon spectra, we might anticipate from comparison with Cs2TiF6 that at temperatures high enough to stabilise the phonons in Rb2TiF6, the entropy of the Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m phase will also be higher than that of the P
m phase will also be higher than that of the P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1. On the other hand, from the lattice energy calculations we find that the energy of the P
m1. On the other hand, from the lattice energy calculations we find that the energy of the P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1 phase is 4.134 kJ mol−1 lower than that of the Fm
m1 phase is 4.134 kJ mol−1 lower than that of the Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m phase. Thus certainly at lower temperatures the stable structure of Rb2TiF6 is predicted to be that of the P
m phase. Thus certainly at lower temperatures the stable structure of Rb2TiF6 is predicted to be that of the P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1 phase, and from comparison with Cs2TiF6 (Fig. 6) we anticipate that the difference in phonon free energy is unlikely to drive a phase transition at any practical temperatures.
m1 phase, and from comparison with Cs2TiF6 (Fig. 6) we anticipate that the difference in phonon free energy is unlikely to drive a phase transition at any practical temperatures.
3.5 Goldschmidt tolerance factor
The Goldschmidt tolerance factor43t is a geometric criterion used to assess the structural stability of classical perovskite ABX3 compounds,44 and it can be extended to vacancy double perovskite A2BX6 in this study. Together with another geometric factor, the octahedral factor45μ, these two are defined as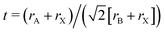 and μ = rB/rX, where rA, rB and rX are the effect ionic radii of the A, B and X ions, respectively.
and μ = rB/rX, where rA, rB and rX are the effect ionic radii of the A, B and X ions, respectively.
Alexander et al.42 analyzed 170 A2BX6 compounds from the ICSD database and identified an optimal range based on these two geometric factors that is applicable to double perovskite structures, within which most cubic perovskites are located. In this work, t and μ are calculated for the eight compounds studied here and compared with the optimal range proposed in that study. The effective ionic radii used in the calculations are taken from Shannon,46 and the results are shown in (Fig. 7). It can be seen that both Rb2TiF6 and Cs2TiF6 lie near the proposed stability boundary, regardless of whether the area is defined by considering all halides or fluorides alone. This is consistent with the fact that these two can crystallize in P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1 phase, even though the cubic phase of Cs2TiF6 is dynamically stable and has lower free energy than its trigonal structure in our calculations. In fact, A2BF6 compounds may exist as multiple polymorphs.47–49 The formation energies among these different phases are very close, as confirmed by calculations in Section 3.6. A vacancy double perovskite phase might be possible for A2BF6, yet it may not correspond to the minimum free energy. Its stability cannot be fully determined by the two structural factors alone. On the other hand, although Rb2TiI6 falls within the proposed range, the region defined by Alexander et al. also contains some of disordered and non perovskite compounds. This range is defined to encompass cubic perovskites, and compared with other compositions, Rb2TiI6 already lies closer to the boundary.
m1 phase, even though the cubic phase of Cs2TiF6 is dynamically stable and has lower free energy than its trigonal structure in our calculations. In fact, A2BF6 compounds may exist as multiple polymorphs.47–49 The formation energies among these different phases are very close, as confirmed by calculations in Section 3.6. A vacancy double perovskite phase might be possible for A2BF6, yet it may not correspond to the minimum free energy. Its stability cannot be fully determined by the two structural factors alone. On the other hand, although Rb2TiI6 falls within the proposed range, the region defined by Alexander et al. also contains some of disordered and non perovskite compounds. This range is defined to encompass cubic perovskites, and compared with other compositions, Rb2TiI6 already lies closer to the boundary.
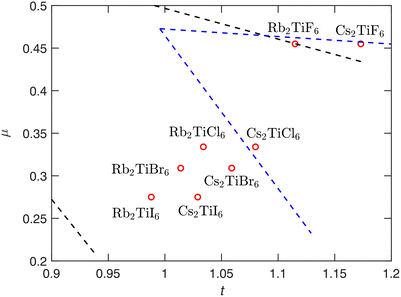 | ||
| Fig. 7 Calculated Goldschmidt tolerance factor t and octahedral factor μ of A2TiX6 (A= Rb, Cs, X = F, Cl, Br, I). The black and blue dashed lines represent the approximate stability boundaries for double perovskite of all halides and only fluorides, respectively, as defined by Alexander et al.42 The region defined by the blue dashed lines extends slightly beyond the black area because, when delineating the region based on fluorides, the starting point selection differs, resulting in a more divergent region. | ||
3.6 Possible space groups
The unstable phonon modes observed in Rb2TiF6 and Rb2TiI6 indicate the structures tend to distort towards lower symmetry. Since P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1 is not a subgroup of Fm
m1 is not a subgroup of Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m, the possible phases can be identified using the tables of Stokes and Hatch.50,51 Based on the unstable modes at the Γ and X points shown in Fig. 2 and Fig. S3, six candidate space groups (C2/m, R
m, the possible phases can be identified using the tables of Stokes and Hatch.50,51 Based on the unstable modes at the Γ and X points shown in Fig. 2 and Fig. S3, six candidate space groups (C2/m, R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) , P
, P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) , P42/nnm, Pn
, P42/nnm, Pn![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) and Pnnn) that preserve the chemical composition for each of Rb2TiF6 and Rb2TiI6 are identified. The parent Fm
and Pnnn) that preserve the chemical composition for each of Rb2TiF6 and Rb2TiI6 are identified. The parent Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m structure is then distorted according to the symmetry constraints of each candidate space group and subsequently fully relaxed. Phonon calculations at the Γ point are performed for all relaxed structures. It was found that, for both Rb2TiF6 and Rb2TiI6, only the C2/m phase has no imaginary phonon frequencies at this point.
m structure is then distorted according to the symmetry constraints of each candidate space group and subsequently fully relaxed. Phonon calculations at the Γ point are performed for all relaxed structures. It was found that, for both Rb2TiF6 and Rb2TiI6, only the C2/m phase has no imaginary phonon frequencies at this point.
To further assess the relative stability of the distorted phases, lattice energies were also calculated. In Rb2TiF6, the lattice energy of the C2/m phase is lower than that of the Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m phase by 3.59 kJ mol−1, while in Rb2TiI6 the corresponding value is 9.83 kJ mol−1. This result demonstrates that, in terms of lattice energies, the monoclinic C2/m phase is also favoured over the cubic structure.
m phase by 3.59 kJ mol−1, while in Rb2TiI6 the corresponding value is 9.83 kJ mol−1. This result demonstrates that, in terms of lattice energies, the monoclinic C2/m phase is also favoured over the cubic structure.
3.7 Heat capacity
For completeness, we show the calculated heat capacities of the members of the family with fully-stable phonons in Fig. 8. For A = Cs we can show results for all four types of anion X, but for A = Rb we can only show results for the two compounds that have stable phonons. The graphs show the expected trends, with faster rises on heating for the heavier X anions reflecting the lower frequencies as seen in the dispersion curves. The heat capacity for the two compounds Rb2TiCl6 and Rb2TiBr6 look very similar to the corresponding case for A= Cs, reflecting the greater importance of the X anion over the A cation in determining the dynamic properties. | ||
| Fig. 8 Calculated heat capacities of Cs2TiX6, X = F, Cl, Br, I (left) and for Rb2TiX6, X = Cl, Br (right). | ||
4 Conclusion
We have compared the lattice dynamics and crystal stabilities of the family of materials of formula A2TiX6, with A = Rb or Cs, and X = F, Cl, Br or I, all with the vacancy double perovskite structure of space group Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m. The lattice dynamics are ordered in the same way in each structure, with the lowest energy modes corresponding to rotations of the TiX6 octahedra, immediately below the external modes involving motions of the A cations and whole-body displacements of the TiX6 octahedra. Above these modes are the bond-bending internal modes of the TiX6 octahedra, and at the highest frequencies are the symmetric and antisymmetric Ti–X stretching modes. The gaps between these different types of modes increases in a consistent manner with decreasing mass of the X anion. The phonon dispersion curves for both A = Rb and or A = Cs are very similar, with largest differences being in the external modes, although not always just through a simple scaling by mass.
m. The lattice dynamics are ordered in the same way in each structure, with the lowest energy modes corresponding to rotations of the TiX6 octahedra, immediately below the external modes involving motions of the A cations and whole-body displacements of the TiX6 octahedra. Above these modes are the bond-bending internal modes of the TiX6 octahedra, and at the highest frequencies are the symmetric and antisymmetric Ti–X stretching modes. The gaps between these different types of modes increases in a consistent manner with decreasing mass of the X anion. The phonon dispersion curves for both A = Rb and or A = Cs are very similar, with largest differences being in the external modes, although not always just through a simple scaling by mass.
We found that the phonons are stable in all but two cases, namely Rb2TiF6 and Rb2TiI6, where the rotational modes show instabilities at the Γ point and along the whole of the lines in reciprocal space in the [100] directions. We also showed that for Rb2TiF6, the Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m phase has a higher free energy than that of the experimentally-observed phase with space group P
m phase has a higher free energy than that of the experimentally-observed phase with space group P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1, thereby explaining why Rb2TiF6 always crystallises in this latter phase. Cs2TiF6 can also be crystallised with the P
m1, thereby explaining why Rb2TiF6 always crystallises in this latter phase. Cs2TiF6 can also be crystallised with the P![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m1 structure, but we found that the free energy of the Fm
m1 structure, but we found that the free energy of the Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m phase is lower at all temperatures. Based on the tables of Stokes and Hatch,50,51 we also explored the possible phases from the soft modes of the cubic Fm
m phase is lower at all temperatures. Based on the tables of Stokes and Hatch,50,51 we also explored the possible phases from the soft modes of the cubic Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m structure in Rb2TiF6 and Rb2TiI6, and found that the monoclinic C2/m phase might be the stable state in both cases.
m structure in Rb2TiF6 and Rb2TiI6, and found that the monoclinic C2/m phase might be the stable state in both cases.
The materials of general formula A2TiX6 with a structure of vacancy double perovskite are among a group of materials that have been suggested might be potential new electronic ceramics as alternatives to lead-based perovskites. As such there has been much work on synthesis and characterisation, and prior DFT calculations have focused on the electronic properties and band structure, with little attention paid to their lattice dynamics. Lattice dynamics calculations underpin issues such as phase stability and thermodynamics properties. Systematic studies across a range of chemical compositions are useful for gaining understanding and insight into these matters. In the case of A2TiX6 we have understood the trends with changing both ions for the higher-frequency internal modes. For the external modes, and particularly the lowest-frequency rotational mode, we see trends across the range of compositions but these trends do not follow from simple scalings with mass.
Author contributions
Junbiao Guo: investigation; methodology; validation; writing (original draft); writing (review & editing); visualization. Keith Refson: software; writing (review & editing). Martin T. Dove: investigation; methodology; validation; writing (original draft); writing (review & editing); visualization.Conflicts of interest
There are no conflicts to declare.Data availability
The main CASTEP code is available for academic use. Information is available from https://www.castep.org/get_castep. Input and output files for the calculations, and scripts for the analysis, are available upon request.Supplementary information (SI) contains phonon dispersion curves for 6 of the compounds, diagrams of mode eigenvectors, and a table of phonon frequencies at zero wave vector for all materials. See DOI: https://doi.org/10.1039/d5cp04058b.
Acknowledgements
We acknowledge the financial support from National Natural Science Foundation of China, grant numbers 12174274 and 12350710177, from the Joint Fund of Bijie City and Guizhou University of Engineering Science (Bijie Science and Technology Union Contract [2026] number 2), and from Guizhou Provincial Innovation Talent Workstation (KXJC (2025)044 - Qiankehe Platform) (MTD).References
- D. C. Palmer, Z. Kristallogr. Cryst. Mater., 2015, 230, 559–572 CrossRef CAS
.
- A. Babayigit, A. Ethirajan, M. Muller and B. Conings, Nat. Mater., 2016, 15, 247–251 CrossRef CAS PubMed
.
- M. Awais, R. L. Kirsch, V. Yeddu and M. Saidaminov, ACS Mater. Lett., 2021, 3, 299–307 CrossRef CAS
.
- A. D. Jodlowski, D. Rodriguez-Padron, R. Luque and G. de Miguel, Adv. Energy Mater., 2018, 8, 1703120 CrossRef
.
- F. Igbari, Z.-K. Wang and L.-S. Liao, Adv. Energy Mater., 2019, 9, 1803150 CrossRef
.
- R.-I. Biega, Y. Chen, M. R. Filip and L. Leppert, Nano Lett., 2023, 23, 8155–8161 CrossRef CAS PubMed
.
- L. A. Muscarella and E. M. Hutter, ACS Energy Lett., 2022, 7, 2128–2135 CrossRef CAS
.
- M. R. Filip, S. Hillman, A. A. Haghighirad, H. J. Snaith and F. Giustino, J. Phys. Chem. Lett., 2016, 7, 2579–2585 CrossRef CAS PubMed
.
- K. Chakraborty, N. R. Medikondu, P. B. Kanakavalli, V. V. Kamesh, S. Das, M. G. Choudhury and S. Paul, Phys. Scr., 2024, 99, 105554 CrossRef CAS
.
- B. A. Connor, L. Leppert, M. D. Smith, J. B. Neaton and H. I. Karunadasa, J. Am. Chem. Soc., 2018, 140, 5235–5240 CrossRef CAS PubMed
.
- X. Du, D. He, Y. Liu and N. Cheng, J. Appl. Phys., 2020, 128, 235110 CrossRef CAS
.
- B. Cucco, G. Bouder, L. Pedesseau, C. Katan, J. Even, M. Kepenekian and G. Volonakis, Appl. Phys. Lett., 2021, 119, 181903 CrossRef CAS
.
- M.-G. Ju, M. Chen, Y. Zhou, H. F. Garces, J. Dai, L. Ma, N. P. Padture and X. C. Zeng, ACS Energy Lett., 2018, 3, 297–304 CrossRef CAS
.
- S. R. Kavanagh, C. N. Savory, S. M. Liga, G. Konstantatos, A. Walsh and D. O. Scanlon, J. Phys. Chem. Lett., 2022, 10965–10975 CrossRef CAS PubMed
.
- H. Albalawi, B. Ul Haq, G. Nazir, T. Ghrib, N. A. Kattan, Q. Mahmood, A. Aljameel, I. M. Morsi and S. Bouzgarrou, Mater. Today Commun., 2022, 32, 104106 CrossRef CAS
.
- R. L. Lister and S. N. Flengas, Can. J. Chem., 1963, 41, 1548–1551 CrossRef CAS
.
- Q. Mahmood, T. H. Flemban, H. Althib, T. Alshahrani, M. G. B. Ashiq, B. Ul Haq, Y. Tahir, A. Surrati, N. A. Kattan and A. Laref, J. Mater. Res. Technol., 2020, 9, 13043–13053 CrossRef CAS
.
- J. Wei, J. Wu, Y. Wang, Y. Zhang, Z. Ma, C. Qiao and H. Zeng, Mater. Adv., 2023, 4, 3767–3773 RSC
.
- D. Kong, D. Cheng, X. Wang, K. Zhang, H. Wang, K. Liu, H. Li, X. Sheng and L. Yin, J. Mater. Chem. C, 2020, 8, 1591–1597 RSC
.
- S. Aslam, A. S. Farooqi, M. Y. Abd Rahman and S. A. M. Samsuri, Phys. Status Solidi A, 2022, 219, 2100671 CrossRef CAS
.
- G. K. Grandhi, A. Matuhina, M. Liu, S. Annurakshita, H. Ali-Loytty, G. Bautista and P. Vivo, Nanomaterials, 2021, 11, 1458 CrossRef CAS PubMed
.
- E. Reyes-Francis, C. Echeverria-Arrondo, D. Esparza, T. Lopez-Luke, T. Soto-Montero, M. Morales-Masis, S.-H. Turren-Cruz, I. Mora-Sero and B. Julian-Lopez, Chem. Mater., 2024, 36, 1728–1736 CrossRef CAS PubMed
.
- D. Liu, G. Yao, S. Jin, J. Chen, B. Lou, Q. Li and R. Sa, J. Phys. Chem. Solids, 2021, 150, 109852 CrossRef CAS
.
- S. M. Liga and G. Konstantatos, J. Mater. Chem. C, 2021, 9, 11098–11103 RSC
.
- Q. Zhou, Y. Zhou, Y. Liu, Z. Wang, G. Chen, J. Peng, J. Yan and M. Wu, J. Mater. Chem. C, 2015, 3, 9615–9619 RSC
.
- I. M. Shlyapnikov, E. A. Goreshnik and Z. Mazej, Inorg. Chem., 2018, 57, 1976–1987 CrossRef CAS PubMed
.
- J. E. Ingall, E. Linscott, N. Colonna, A. J. Page and V. J. Keast, J. Phys. Chem. C, 2024, 128, 9217–9228 CrossRef CAS
.
- Q. Mahmood, M. Hassan, N. Yousaf, A. A. AlObaid, T. I. Al-Muhimeed, M. Morsi, H. Albalawi and O. A. Alamri, Mater. Sci. Semicond. Process., 2022, 137, 106180 CrossRef CAS
.
- M. Chen, M.-G. Ju, A. D. Carl, Y. Zong, R. L. Grimm, J. Gu, X. C. Zeng, Y. Zhou and N. P. Padture, Joule, 2018, 2, 558–570 CrossRef CAS
.
- J. Euvrard, X. Wang, T. Li, Y. Yan and D. B. Mitzi, J. Mater. Chem. A, 2020, 8, 4049–4054 RSC
.
- J. L. Mendes, W. Gao, J. L. Martin, A. D. Carl, N. A. Deskins, S. Granados-Focil and R. L. Grimm, J. Phys. Chem. C, 2020, 124, 24289–24297 Search PubMed
.
- S. J. Clark, M. D. Segall, C. J. Pickard, P. J. Hasnip, M. I. J. Probert, K. Refson and M. C. Payne, Z. Kristallogr. Cryst. Mater., 2005, 220, 191–194 Search PubMed
.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS PubMed
.
- J. P. Perdew, K. Burke, M. Ernzerhof and R. Ernstorfer, Phys. Rev. Lett., 1997, 78, 1396 CrossRef CAS
.
- H. J. Monkhorst and J. D. Pack, Phys. Rev. B, 1976, 13, 5188–5192 CrossRef
.
- S. Baroni, S. de Gironcoli and A. D. Corso, Rev. Mod. Phys., 2001, 73, 515–562 CrossRef CAS
.
- K. Refson, P. R. Tulip and S. J. Clark, Phys. Rev. B: Condens. Matter Mater. Phys., 2006, 73, R4954–12 CrossRef
.
- W. Stirling, J. Phys. C: Solid State Phys., 1972, 5, 2711–2730 CrossRef CAS
.
- A. Giddy, M. Dove, G. Pawley and V. Heine, Acta Crystallogr., Sect. A, 1993, 49, 697–703 CrossRef
.
- L. Tan, V. Heine, G. Li and M. T. Dove, Rep. Prog. Phys., 2024, 87, 126501 CrossRef PubMed
.
- J. Zhou, Y. Wang, Y. Chen, Y. Zhou, B. Milicevic, L. Zhou, J. Yan, J. Shi, R.-S. Liu and M. Wu, Angew. Chem., Int. Ed., 2021, 60, 3940–3945 Search PubMed
.
- A. E. Fedorovskiy, N. A. Drigo and M. K. Nazeeruddin, Small Methods, 2020, 4, 1900426 CrossRef CAS
.
- V. Goldschmidt, Naturwissenschaften, 1926, 14, 477–485 CrossRef CAS
.
- M. Lufaso and P. Woodward, Acta Crystallogr., Sect. B: Struct. Sci., Cryst. Eng. Mater., 2004, 60, 10–20 CrossRef PubMed
.
- C. Li, K. Soh and P. Wu, J. Alloys Compd., 2004, 372, 40–48 CrossRef CAS
.
- R. Shannon, Acta Crystallogr., Sect. A, 1976, 32, 751–767 CrossRef
.
- L. Smrcok, A. Le Bail, M. Boca and A. Rakhmatullin, Cryst. Growth Des., 2020, 20, 3867–3881 CrossRef CAS
.
- G. Benner and R. Hoppe, J. Fluorine Chem., 1990, 48, 219–227 CrossRef CAS
.
- C. Hebecker, H. G. Vonschne and R. Hoppe, Naturwissenschaften, 1966, 53, 154 CrossRef CAS
.
-
H. T. Stokes, D. M. Hatch and B. J. Campbell, ISOSUBGROUP, ISOTROPY Software Suite, https://iso.byu.edu Search PubMed
.
- H. T. Stokes, S. van Orden and B. J. Campbell, J. Appl. Crystallogr., 2016, 49, 1849–1853 CrossRef CAS
.
| This journal is © the Owner Societies 2026 |